Abstract
Background
Cardiac amyloidosis (CA) is a life-threatening restrictive cardiomyopathy. Identifying patients with a poor prognosis is essential to ensure appropriate care. The aim of this study was to compare myocardial work (MW) indices with standard echocardiographic parameters in predicting mortality among patients with CA.
Methods
Clinical, biological and transthoracic echocardiographic parameters were retrospectively compared among 118 patients with CA. Global work index (GWI) was calculated as the area of left ventricular pressure–strain loop. Global work efficiency (GWE) was defined as percentage ratio of constructive work to sum of constructive and wasted works. Sixty-one (52%) patients performed a cardiopulmonary exercise.
Results
GWI, GWE, global longitudinal strain (GLS), left ventricular ejection fraction (LVEF) and myocardial contraction fraction (MCF) were correlated with N-terminal prohormone brain natriuretic peptide (R=−0.518, R=−0.383, R=−0.553, R=−0.382 and R=−0.336, respectively; p<0.001). GWI and GLS were correlated with peak oxygen consumption (R=0.359 and R=0.313, respectively; p<0.05). Twenty-eight (24%) patients died during a median follow-up of 11 (4–19) months. The best cut-off values to predict all-cause mortality for GWI, GWE, GLS, LVEF and MCF were 937 mm Hg/%, 89%, 10%, 52% and 15%, respectively. The area under the receiver operator characteristic curve of GWE, GLS, GWI, LVEF and MCF were 0.689, 0.631, 0.626, 0.511 and 0.504, respectively.
Conclusion
In CA population, MW indices are well correlated with known prognosis markers and are better than LVEF and MCF in predicting mortality. However, MW does not perform better than GLS.
Keywords: heart failure with preserved ejection fraction, echocardiography, restrictive cardiomyopathy
Key questions.
What is already known about this subject?
Cardiac amyloidosis has a poor prognosis in the absence of dedicated treatment. Many targeted treatments are in development, but their usefulness depends on the stage of the disease. Therefore, it is important to define prognostic indexes, which will allow a better selection of therapeutic candidates.
What does this study add?
In patients with cardiac amyloidosis, myocardial work indices are well correlated with prognosis markers and are better than left ventricular ejection fraction and myocardial contraction fraction at predicting all-cause mortality.
How might this impact on clinical practice?
In patients with cardiac amyloidosis, myocardial work indices should be used to better assess the severity of the disease and to better select candidates for therapeutic intervention.
Introduction
Cardiac amyloidosis (CA) is an infiltrative and restrictive cardiomyopathy characterised by myocardial deposition of insoluble misfolded proteins, most commonly immunoglobulin light-chain (AL) or transthyretin (ATTR) amyloidosis and its subtype; wild-type and variant-type. The deposit of those proteins disrupts the structure and function of the heart with a thickening of the ventricular wall, an increase of the myocardial mass and a reduction in end-diastolic volume leading to a diastolic dysfunction, an impaired myocardial contraction, and a progressive heart failure and death.1–3 Untreated, CA is life threatening from few months for AL to few years for ATTR.4–6 Consequently, identifying patients with a poor prognosis is fundamental to ensure adequate treatment and a timely referral to specialised centres.
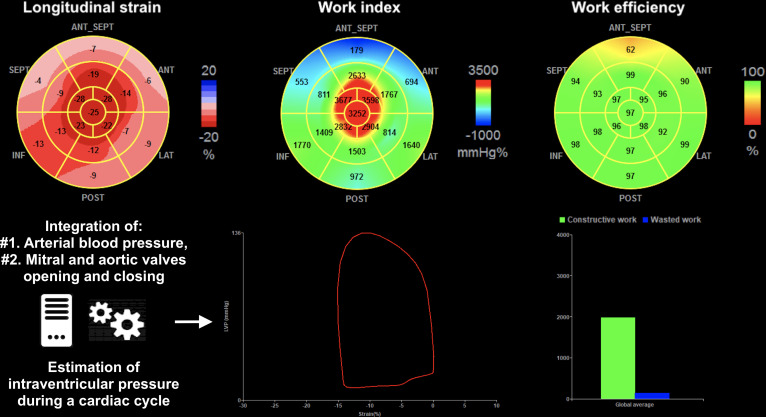
Transthoracic echocardiography (TTE) remains the first imaging examination used for the initial assessment of cardiac involvement but the left ventricular ejection fraction (LVEF) may remain preserved until advanced stages of the disease.7 Myocardial contraction fraction (MCF) has been reported to be superior to LVEF in predicting survival in AL–CA and both variant-type and wild-type ATTR–CA.2 3 More recently, LV global longitudinal strain (GLS) has emerged as a robust and reproducible index to estimate the global and segmental ventricular function, to detect subclinical myocardial dysfunction and to predict outcomes.8–11 However, the GLS depends on the load, which may lead to a misinterpretation of the contractile function of the myocardium.12–14 Thus, myocardial work (MW) is emerging as an alternative tool for studying LV myocardial systolic function incorporating deformation and afterload into its analysis.15 Non-invasive MW is based on LV pressure–strain loop, which explores LV function balanced by afterload with an estimation of intraventricular pressure during a cardiac cycle13 and can be easily calculated at the time of TTE examination using commercial software. The interest of the MW therefore lies in the integration of afterload, which varies in CA according to the degree of restriction.
The aim of this study was to compare MW indices with classic echocardiographic parameters in predicting mortality among patients with CA and to investigate their relationship with known prognosis clinical and biochemical markers.
Methods
Study population
One hundred and eighteen patients with CA followed at the University Hospital of Rangueil (Toulouse, France) were retrospectively included. Patients were included retrospectively from January 2016 to January 2020. Data were collected in February 2020. Analysis began in April 2020. All medical records were reviewed to confirm the diagnosis of CA. Inclusion criteria were as follows: ≥18 years old and presence of variant or wild-type ATTR or AL–CA determined by either an endomyocardial biopsy-proven CA, a positive non-cardiac biopsy for amyloidosis with characteristic TTE parameters and a consistent clinical history of CA or previously established non-biopsy diagnostic criteria of CA.16–18 Exclusion criteria were concomitant moderate to severe valvular heart disease, poor apical acoustic window and inadequate tracking in more than three LV segments. Demographic parameters, cardiovascular factors and comorbidities were collected. Each patient underwent a comprehensive assessment including clinical evaluation, biochemistry, ECG and TTE. Sixty-one patients performed a cardiopulmonary exercise test with the evaluation of peak oxygen consumption. All examinations were performed during the same day.
The investigation conforms to the principles outlined in the Declaration of Helsinki. All patients were informed of the protocol and gave their consent. Our institutional review board approved the study.
Biochemistry
Blood chemistry parameters including creatinine, troponin and N-terminal prohormone brain natriuretic peptide (NT-proBNP) were measured by standard automated commercial techniques. Renal function was expressed as an estimated glomerular filtration rate (eGFR), which was calculated according to the modification of diet in renal disease formula.
Functional analysis
Cardiopulmonary exercise testing was performed on an ergometric bicycle. The initial workload was 20 W and was increased by 10 W every minute. Oxygen consumption was analysed using breath-by-breath analysis at rest, at each step during exercise and during recovery. The test was terminated if patients exhibited signs of exhaustion, or if the maximum physical capacity was reached.
Transthoracic echocardiography TTE
All patients underwent standard two-dimensional TTE using Vivid V.E95 System (GE Vingmed Ultrasound AS, Horten, Norway) equipped with a 2.5 MHz transducer. Images were obtained from the standard parasternal long axis and short axis, apical and subcostal views using high frame rates (>60 frames/s). Bidimensional, colour Doppler, pulsed-wave and continuous-wave Doppler data were stored on a dedicated workstation, EchoPAC V.202 (Advanced Analysis Technologies; GE Medical Systems) for the offline analysis. The following measurements were performed by a single-blinded observer according to the American Society of Echocardiography Guidelines19–21: LVEF using the biplane Simpson’s method from apical two-chamber and four-chamber windows, left atrial volume, LV end-diastolic and end-systolic volume, LV internal dimensions, end-diastolic interventricular septal and posterior wall thickness, LV diastolic function including peak early (E) and late (A) diastolic mitral inflow velocity and its ratio, lateral mitral annular diastolic velocities (e’) and E/e’ ratio. Using these measurements, stroke volume was calculated as end-diastolic volume—end-systolic volume. LV mass was determined using the cube formula described by the American Society of Echocardiography and was indexed to the body surface area. Myocardial volume was defined as LV mass divided by the mean density of myocardium (1.05 g/mL). Then MCF was calculated as stroke volume divided by myocardial volume.22 To calculate LV–GLS, two-dimensional grey scale images were acquired in the standard apical 4-chamber, 3-chamber and 2-chamber views according to the European Association of Cardiovascular Imaging and the American Society of Echocardiography.21 A ‘bull’s-eye’ plot illustrating segmental longitudinal strain was automatically generated. The strain values for the six basal, six mid and five apical segments of the LV were averaged to obtain three regional longitudinal strain values. The apex-to-base gradient in regional LS was examined using a relative apical LS calculated using the formula described by Phelan et al.23 In this report, strain is reported in absolute terms.
MW quantification
According to the European Society of Cardiology and as described by Russell et al,13 MW was calculated using a combination of LV–GLS and a non-invasively estimated LV pressure curve. After calculating GLS, indicating the time of aortic and mitral events by echocardiography and inserting values of brachial blood pressure, the software derived non-invasive pressure–strain loop. Peak systolic LV pressure was assumed to be equal to the peak systolic blood pressure, which was recorded with a brachial cuff. Strain and pressure data were synchronised using the onset R wave on ECG as a common time reference. MW was evaluated from mitral valve closure to mitral valve opening. The area of the loop served as an index of regional and global MW. A bull’s eye with segmental and global work index (GWI) was obtained as well as additional indices: constructive work (work which is performed during shortening in systole adding negative work during lengthening in isovolumetric relaxation), wasted work (negative work performed during lengthening in systole adding work performed during shortening in isovolumetric relaxation) and global work efficiency (GWE, constructive work divided by the sum of constructive and wasted works). By averaging constructive and wasted works for each segment, global constructive and wasted works were estimated for the entire LV. An example of these parameters is presented in figure 1.
Figure 1.
Seventeen-segment bull’s-eye representation of MW indices and GLS, and steps for MW indices analysis. GLS, global longitudinal strain; MW, myocardial work.
Follow-up
Follow-up was assessed in March 2020 by electronic chart review or by phone interview of the patient’s general practitioner/cardiologist, patient or family. All-cause of mortality was the primary endpoint. It was defined using time to death for deceased subjects and time to last known follow-up for those last known to be alive.
Statistical analysis
Continuous variables were expressed as mean±SD. Results of values not normally distributed were presented as medians with IQR. Nominal values were expressed as numbers and percentages. Group comparisons were made using non-parametric Kruskal-Wallis test for continuous variables and Pearson χ2 test for categorical variables, using Mann-Whitney test and Fisher exact test, respectively, for multiple comparisons. Relationships between variables were assessed using Spearman correlation analysis and expressed by R. Comparison between receiver operator characteristic (ROC) curves was made using DeLong test. The best cut-off value was defined as the point with the highest sum of sensitivity and specificity. The intraobserver and interobserver variability of measurements was analysed using the intraclass correlation coefficient from 20 randomly selected patients and from 10 patients with atrial fibrillation reanalysed by two observers blinded to other echocardiographic results. Differences were considered statistically significant for p values of <0.05. All analyses were performed using standard statistical software, SPSS V.20 and MedCalc V.15 (MedCalc, Mariakerke, Belgium).
Results
Study population
The final study population consisted of 118 patients with a confirmed diagnosis of CA. There were 80 (68%) wild-type ATTR–CA, 12 (10%) variant-type and 26 (22%) AL–CA. Baseline characteristics are summarised in table 1. There were 95 (81%) men and the mean age was 78±10 years old. Wild-type patients with ATTR were significantly older. Mean systolic and diastolic blood pressure was 132±21 mm Hg and 77±11 mm Hg and was higher in patients with ATTR. There was no difference in comorbidities between groups except for ischaemic heart disease, which was more frequent in wild-type patients with ATTR. The majority (79%) of patients had symptoms with stage 2 or more New York Heart Association dyspnoea. Seventy-six patients (64%) had diuretics and 28 (24%) received Tafamidis at the time of the evaluation.
Table 1.
Baseline clinical, biological and electrocardiograms characteristics
| Overall population (n=118) |
AL (1) (n=26) |
Wild-type ATTR (2) (n=80) |
Variant-type ATTR (3) (n=12) |
P value | 1 versus 2 | Posthoc analysis 2 versus 3 |
3 versus 1 | |
| Baseline characteristic | ||||||||
| Age—years | 78±10 | 68±12 | 82±7 | 71±6 | <0.001 | <0.001 | <0.001 | 0.722 |
| Male gender, n (%) | 95 (81) | 18 (69) | 68 (85) | 9 (75) | 0.188 | . | ||
| Body mass index—kg/m2 | 25±3 | 24±3 | 26±3 | 26±2 | 0.116 | . | ||
| Systolic blood pressure—mm Hg | 131±20 | 120±19 | 134±20 | 139±17 | 0.010 | 0.010 | 0.260 | 0.007 |
| Diastolic blood pressure—mm Hg | 76±11 | 72±10 | 76±11 | 82±14 | 0.044 | 0.072 | 0.121 | 0.033 |
| Heart rate—bpm | 77±16 | 82±10 | 77±18 | 70±7 | 0.009 | 0.033 | 0.171 | <0.001 |
| Medical history, n (%) | ||||||||
| Current smoker | 1 (1) | 1 (4) | 0 | 0 | 0.170 | |||
| Hypertension | 58 (49) | 8 (31) | 45 (56) | 5 (42) | 0.069 | |||
| Dyslipidaemia | 35 (30) | 6 (23) | 26 (33) | 3 (25) | 0.617 | |||
| Diabetes | 13 (11) | 2 (8) | 11 (14) | 0 | 0.306 | |||
| Ischaemic heart disease | 25 (21) | 3 (12) | 22 (28) | 0 | 0.038 | 0.097 | 0.038 | 0.226 |
| NYHA class, n (%) | ||||||||
| NYHA 1 | 25 (21) | 5 (19) | 17 (21) | 3 (25) | 0.922 | |||
| NYHA 2 | 70 (59) | 15 (58) | 48 (60) | 7 (58) | 0.976 | |||
| NYHA 3 | 20 (17) | 5 (19) | 13 (16) | 2 (17) | 0.940 | |||
| NYHA 4 | 3 (3) | 1 (4) | 2 (3) | 0 | 0.784 | |||
| Medication, n (%) | ||||||||
| Diuretics | 76 (64) | 12 (46) | 60 (75) | 4 (33) | 0.002 | 0.006 | 0.004 | 0.463 |
| Tafamidis | 28 (24) | – | 20 (25) | 8 (67) | <0.001 | 0.004 | ||
| Biochemistry | ||||||||
| NT-proBNP—ng/L | 2423 (1105–4380) | 2108 (691–4645) | 2607 (1408–4675) | 1096 (221–3036) | 0.062 | |||
| Troponin—ng/L | 71±46 | 76±54 | 74±44 | 41±32 | 0.007 | 0.903 | 0.002 | 0.008 |
| eGFR—mL/min/1.73 m2 | 56±21 | 61±26 | 52±19 | 73±16 | 0.002 | 0.084 | 0.001 | 0.209 |
| ECG, n (%) | ||||||||
| Atrial fibrillation | 31 (26) | 2 (8) | 28 (35) | 1 (8) | 0.008 | 0.008 | 0.065 | 0.946 |
| Low voltage | 13 (11) | 8 (31) | 4 (5) | 1 (8) | 0.001 | <0.001 | 0.637 | 0.136 |
| Pseudo infarct pattern | 14 (12) | 4 (15) | 7 (9) | 3 (25) | 0.475 | |||
| Functional analysis | ||||||||
| Peak oxygen consumption—mL/min/kg | 15±5 | 16±5 | 14±5 | 17±6 | 0.087 | |||
Data are presented as number (percent), mean±SD if normally distributed or median (IQR) if not normally distribute.
AL, light chain amyloidosis; ATTR, transthyretin amyloidosis; eGFR, estimated glomerular filtration rate; NT-proBNP, N-terminal pro-hormone brain natriuretic peptide; NYHA, New York Heart Association.
Biochemistry
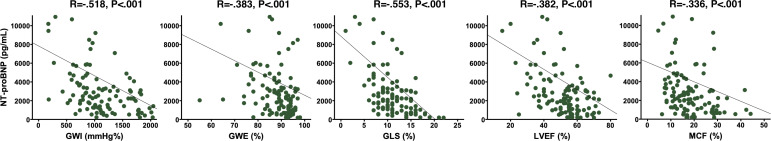
Correlation between MW indices, LVEF, GLS, MCF and biochemical parameters are shown in table 2. NT-proBNP levels were not different between groups. Median NT-proBNP was 2423 (1105–4380) pg/mL and was negatively correlated with GWI and GLS. There was a slight correlation between GWE, LVEF, MCF and NT-proBNP (figure 2).
Table 2.
Correlation between GWI, GWE, LVEF, GLS, MCF and clinical, biochemical and echocardiographic parameters
| GWI | GWE | GLS | LVEF | MCF | |
| Systolic blood pressure | R=0.543* | R=0.158 p=0.088 |
R=0.280† | R=0.294† | R=0.164 p=0.077 |
| Functional status | |||||
| Peak oxygen consumption | R=0.359† | R=0.141 P.278 |
R=0.313† | R=0.110 p=0.400 |
R=0.168 p=0.195 |
| Biochemistry | |||||
| NT-proBNP | R=−0.518* | R=−0.383* | R=−0.553* | R=−0.382* | R=−0.336* |
| eGFR | R=0.309* | R=0.257† | R=0.345* | R=0.205† | R=0.203† |
| Troponin | R=−0.477* | R=−0.287† | R=−0.451* | R=−0.185 p=0.051 |
R=−0.253† |
| Echocardiography | |||||
| LV stroke volume | R=0.488* | R=0.315† | R=0.511* | R=0.420* | R=0.792* |
| Left atrial volume index | R=−0.061 p=0.515 |
R=−0.009 p=0.927 |
R=−0.053 p=0.572 |
R=−0.096 p=0.309 |
R=−0.158 p=0.091 |
| E/A | R=−0.314† | R=0.033 p=0.768 |
R=-.289† | R=−0.242† | R=−0.273† |
| E/e’ | R=−0.167 p=0.071 |
R=−0.123 p=0.186 |
R=−0.237† | R=−0.081 p=0.383 |
R=−0.234† |
| LVEF | R=0.555* | R=0.384* | R=0.595* | – | R=0.530* |
| GLS | R=0.896* | R=0.642* | – | R=0.595* | R=0.567* |
| MCF | R=0.493* | R=0.326* | R=0.567* | R=0.530* | – |
| GWI | – | R=0.625* | R=0.896* | R=0.555* | R=0.336* |
| GWE | R=0.625* | – | R=0.642* | R=0.384* | R=0.493* |
*p<0.001.
†p <0.05.
eGFR, estimated glomerular filtration rate; GLS, global longitudinal strain; GWE, global work efficiency; GWI, global work index; LV, left ventricular; LVEF, left ventricular ejection fraction; MCF, myocardial contraction fraction; NT-proBNP, N-terminal pro-hormone brain natriuretic peptide.
Figure 2.
Correlation between GWI, GWE, LVEF, GLS, MCF and NT-proBNP. GLS, global longitudinal strain; GWE, global work efficiency; GWI, global work index; LVEF, left ventricular ejection fraction; MCF, myocardial contraction fraction; NT-proBNP, N-terminal pro-hormone brain natriuretic peptide.
Among patients with variant-type ATTR, troponin and eGFR were lower and higher, respectively. Mean eGFR in overall population was 57±21 mL/min/1.73 m2 and was moderately correlated with GWI, GWE, GLS, LVEF and MCF. Mean troponin was 71±46 ng/L and was only correlated with GWI, GWE and GLS.
Functional subanalysis
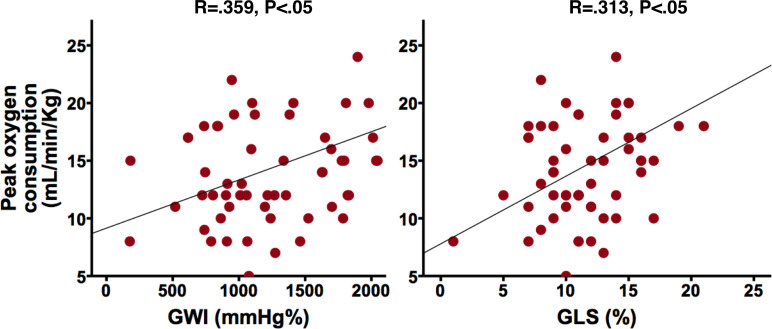
Sixty-one (52%) patients performed a cardiopulmonary exercise test with the evaluation of peak oxygen consumption. Mean peak oxygen consumption was 15±5 mL/min/kg and was more correlated with GWI (R=0.359, p=0.004) than GLS (R=0.313, p=0.014). GWE, LVEF and MCF were not correlated to peak oxygen consumption (table 2 and figure 3).
Figure 3.
Correlation between GWI, GLS and peak oxygen consumption. GLS, global longitudinal strain; GWI, global work index.
Echocardiography
TTE parameters are shown in table 3. LVEF of the whole population was 51%±13% (23%–19%, 21%–18% and 74%–63% patients with reduced, mid-range and preserved LVEF, respectively), whereas all other systolic parameters were impaired with a MCF of 19%±10%, mean GLS of 11%±4%, mean GWI of 1169±538 mm Hg% and mean GWE of 87%±8%. Regional strain analysis showed an apical sparring pattern with mean relative apical longitudinal strain of 1.1 (0.9–1.4). LV filling pressures were increased with a median E/A of 1.5 (0.8–2.7), a deceleration time of 206±84 ms and an E/e’ of 15±7.
Table 3.
Echocardiographic characteristics
| Overall population (n=118) | AL (1)(n=26) | Wild-type ATTR (2)(n=80) | Variant-type ATTR (3)(n=12) | P value | 1 versus 2 | Posthoc analysis 2 versus 3 | 3 versus 1 | |
| Interventricular wall thickness—mm | 17±3 | 15±2 | 18±3 | 17±4 | 0.007 | 0.002 | 0.310 | 0.170 |
| LVEF—% | 51±13 | 52±13 | 50±13 | 56±11 | 0.242 | |||
| LV stroke volume—mL | 50±19 | 50±20 | 50±19 | 52±18 | 0.825 | |||
| MCF—% | 19±9 | 22±12 | 17±9 | 21±9 | 0.094 | |||
| Left atrial volume index—mL/m2 | 43±15 | 38±13 | 45±14 | 43±23 | 0.078 | |||
| E wave—cm/s | 82±23 | 87±27 | 80±21 | 84±25 | 0.478 | |||
| A wave—cm/s | 59±31 | 71±38 | 53±26 | 63±29 | 0.129 | |||
| E/A | 1.5 (0.8–2.7) | 1.3 (0.7–2) | 1,8 (0.9–2.8) | 1 (0.9–1.6) | 0.324 | |||
| e’ wave—cm/s | 6±2 | 6±2 | 6±2 | 7±2 | 0.283 | |||
| E/e’ | 15±7 | 16±7 | 15±7 | 16±12 | 0.728 | |||
| Deceleration time—ms | 206±84 | 188±63 | 212±93 | 208±52 | 0.449 | |||
| GLS—% | 11±4 | 11±2 | 10±4 | 15±5 | 0.006 | 0.214 | 0.003 | 0.010 |
| Relative apical LS | 1.1 (0.9–1.4) | 1 (0.9–1.2) | 1.2 (0.9–1.5) | 0.9 (0.9–1) | 0.043 | 0.048 | 0.068 | 0.456 |
| GWI—mm Hg% | 1169±538 | 1094±422 | 1091±502 | 1755±730 | 0.012 | 0.840 | 0.004 | 0.010 |
| GWE—% | 87±8 | 89±8 | 86±8 | 91±5 | 0.049 | 0.053 | 0.066 | 0.741 |
| Global constructive work—mm Hg% | 1344±562 | 1225±406 | 1290±506 | 1965±822 | 0.013 | 0.654 | 0.005 | 0.008 |
| Global wasted work—mm Hg% | 120±100 | 101±95 | 128±106 | 107±68 | 0.241 | 0.090 | 0.772 | 0.442 |
Data are presented as number (percent), mean±SD if normally distributed or median (IQR) if not normally distributed.
AL, light chain amyloidosis; ATTR, transthyretin amyloidosis; GLS, global longitudinal strain; GWE, global work efficiency; GWI, global work index; LV, left ventricular; LVEF, LV ejection fraction; MCF, myocardial contraction fraction.
There was a strong correlation between GLS and MW indices with correlation coefficients of 0.896 (p<0.001) for GWI and 0.642 (p<0.001) for GWE (table 2).
Predictor of all-cause mortality
The median follow-up duration was 114–19 months. Among the whole population, 28 (24%) died including 8 (31%) with AL–CA, 18 (23%) with wild-type ATTR and 2 (17%) variant-type ATTR. Vital status was unknown for one patient; as a result, 99.2% completed the follow-up.
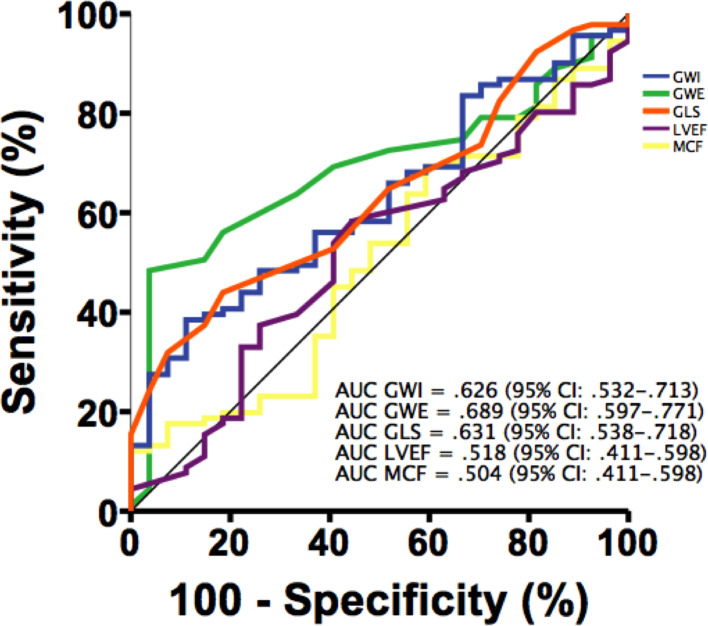
By analysing the ROC curves to predict mortality, the area under the ROC curve of GWE was the greatest with a value of.689 (95 CI 0.597 to 0.771) followed by GLS with a value of 0.631 (95 CI 0.054 to 0.718) then GWI with a value of 0.626 (95 CI 0.532 to 0.713). There was statistical difference of areas under the ROC curves between GWE and MCF (p=0.009), GWE and LVEF (p=0.015) and GWI and MCF (p=0.020). Comparison of areas under the ROC curves between MW indices and GLS was not different (p=0.867 for GWI and p=0.252 for GWE—figure 4).
Figure 4.
Receiver operating characteristic curves comparing GWI, GWE, LVEF, GLS and MCF predicting mortality. AUC, area under the curve; GLS, global longitudinal strain; GWE, global work efficiency; GWI, global work index; LVEF, left ventricular ejection fraction; MCF, myocardial contraction fraction.
The best cut-off values to predict all-cause mortality for GWE, GLS, GWI, MCF and LVEF were 89%, 10%, 937 mm Hg/%, 15% and 52%, respectively. The sensitivity, specificity, positive and negative predictive values of each parameter are presented in table 4.
Table 4.
Performance of echocardiographic parameters to predict all-cause mortality
| Parameter | Cut-off value | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) |
| LVEF | 52% | 58 | 56 | 28 | 82 |
| MCF | 15% | 64 | 44 | 72 | 72 |
| GLS | −10% | 65 | 49 | 29 | 81 |
| GWI | 937 mm Hg/% | 65 | 48 | 29 | 81 |
| GWE | 89% | 64 | 81 | 35 | 87 |
GLS, global longitudinal strain; GWE, global work efficiency; GWI, global work index; LVEF, left ventricular ejection fraction; MCF, myocardial contraction fraction; NPV, negative predictive value; PPV, positive predictive value.
Intraobserver and interobserver variability
The analysis of the interobserver variability of GWI and GWE showed a good reproducibility with an intraclass correlation coefficient of 0.94 (95% CI 0.83 to 0.98) and 0.96 (95% CI 0.89 to 0.99), respectively. The intraobserver variability showed also a good reproducibility with a coefficient of 0.99 (95% CI 0.97 to 1) and 0.97 (95% CI 0.93 to 0.99), respectively. In contrast, MCF showed a moderate reproducibility with an intraclass correlation of 0.85 (95% CI 0.54 to 0.95) and 0.98 (95% CI 0.94 to 0.99) for interobserver and intraobserver variabilities, respectively.
Among patients with atrial fibrillation, the interobserver variability of GWI, GWE, MCF was.90 (95% CI 0.59 to 0.98], 0.68 (95% CI 0.11 to 0.92) and 0.58 (95% CI −0.45 to 0.90), respectively. The intraobserver variability was 0.94 (95% CI 0.74 to 0.99), 0.85 (95% CI 0.38 to 0.96) and 0.79 (95% CI 0.19 to 0.95), respectively.
Discussion
This study evaluates MW indices for predicting mortality in CA population and their relationship with known prognosis parameters such as functional status and biomarkers. GWI and GWE show both a good correlation with NT-proBNP, eGFR and troponin and GWI is associated with peak oxygen consumption. Furthermore, MW indices seem better than MCF and LVEF for predicting all-cause mortality in CA, with a better intraobserver and interobserver reproducibility, even among patients with atrial fibrillation. However, MW indices, as GLS, have better sensitivity than specificity in predicting mortality, giving them good negative predictive value but poor positive predictive value.
A recent study on a population of 100 patients with CA (ATTR, wild-type and variant and AL) shows overlapping results with in particular a prognostic impact of the MW indexes on a composite endpoint including heart failure requiring hospitalisation and all-cause mortality.24 Interestingly, in their study, only GWI has an impact on all-cause mortality, which is not found for GWE and GLS. The authors find a prognostic cut-off value of GWI slightly higher than ours at 1039 mm Hg/%, which probably results from a different population with in particular a higher proportion of AL amyloidosis (34% in their population vs 22% in ours).24 This study does not compare with other biological or echocardiographic prognostic parameters such as MCF, and the lack of impact of GLS on all-cause mortality in their population and the superiority of MW indexes over traditional parameters in our population prompts the use of MW indexes for an integrated approach to prognostic assessment.
The input of non-invasive MW
In a normal heart, there are minimal differences in the timing of contraction on individual myocardial segments, but in CA, the deposit of misfolded proteins in the myocardium is responsible of desynchronised contractions and progressive alteration of LV deformation parameters, which precedes LV dysfunction.8 12 25 MCF and GLS are measures of myocardial deformation or shortening, which can identify decrements in myocardial function that are not detected by LVEF. Previous studies demonstrated the prognostic value of those two parameters as independent predictors of survival.2 3 5 26 27 However, a meta-analysis of 24 studies by Yingchoncharoen et al15 showed that GLS is a load-dependent variable influenced by alterations in loading conditions with a reduction in LV–GLS associated with an increase in afterload. Recently, non-invasive MW was proposed as a new tool to study LV performance, taking into account afterload and myocardial deformation. In CA, the progressive infiltration of the myocardium by amyloid deposits explains the evolution towards restrictive cardiomyopathy, which causes a decrease in cardiac output and a progressive drop in blood pressure. At the restrictive stage, hypotensive treatments are often poorly tolerated and hypotension is accompanied by a worsening of the prognosis.28 It is therefore legitimate to think that the integration of LV afterload, in addition to ventricular function and remodelling parameters, could bring a benefit in the evaluation of the prognosis of patients.
Functional status, biomarkers and outcome
As expected, GWI, GWE, GLS and MCF were impaired, while LVEF was predominantly preserved and were lower than healthy subjects as described by Manganaro et al.29 MW indices showed a strong and significant correlation with NT-proBNP, eGFR, troponin and peak oxygen consumption, which are known prognostic parameters.5 6
Regarding their ability to predict mortality of patients with CA, in our study, GWE, GWI and GLS were better than MCF and LVEF. This may indicate GWI and GWE as being reliable prognostic marker in this population. Also, GWI is highly correlated with blood pressure, which is primordial because low blood pressure is known to be associated with greater mortality in patient with CA.30 Furthermore, we found a good reproducibility between experienced operators indicating GWI and GWE to be robust parameters for clinical assessment.
Regarding MCF and CA, few studies are available2 3 and they did not evaluate the correlation with peak oxygen consumption. In our study, they were not significantly correlated. These results may be due to the small sample size of our study, which limit statistical power. Besides, MCF interobserver echocardiography variability was not evaluated in the previous studies. In our study, it appears that MCF did not show a good reproducibility.
Limitations
Our study has all the limitations associated with retrospective, single-site and limited sample studies. Furthermore, 19% and 18% of patients with reduced and mid-range LVEF, respectively, were included in the study and may introduce bias into the study. Indeed, the predictive role of deformation parameters seems more relevant in patients with preserved LVEF.8 We cannot certify that our results are generalisable only to patients with preserved LVEF.
Almost one half of patients did not perform cardiopulmonary exercise testing with different reasons (poor functional capacity or inability to perform the test) and this may have altered our results especially regarding on correlation between peak oxygen consumption and GWE. Thus, generalisability of our findings to larger cohorts of subjects with CA is uncertain.
Finally, despite integration of LV afterload on deformation parameters, our study shows that MW indices do not perform significantly better that GLS to assess prognosis in patients with CA. Clemmensen et al did not confirm these findings showing a better performance of MW indices over GLS.24 However, one might wonder whether the assessment of MW indices is really necessary, provided that GLS can be calculated in a simple way and avoiding some steps such as the measure of arterial blood pressure and the assessment of valvular timing. Furthermore, despite correct intraobserver and interobserver variability, calculation of MW indices among patients with atrial fibrillation can be challenging and require either tri-plane images acquisition or comparable RR interval loop selection.
Clinical perspectives
As GLS, evaluation of MW by pressure–strain loop is a totally non-invasive, easy and quick approach to assess on myocardial performance on routine TTE, requiring only, in addition, blood pressure measurement and aortic and mitral valve events. Rather than GLS, MW has the advantage to take afterload into its analysis.
MW indices are correlated to known prognosis parameters such as functional status and biomarkers thus they may have clinical value to identify patients with CA with poor prognosis who could profit from a closer monitoring and optimisation of load condition in order to prevent heart failure hospitalisation. However, our study was not designed to compare AL–CA and ATTR–CA, which have different disease trajectory, different life expectancy and specific therapies. Thus, it would be interesting that larger prospective studies evaluate the impact of MW indices in each group.
Conclusion
Non-invasive MW assessed by TTE is an easy and reproducible method of exploration of LV systolic function incorporating myocardial deformation and afterload. MW indices are correlated to known prognosis functional and biochemical parameters and they seem to be better than LVEF and MCF at predicting mortality in CA population. However, MW indices do not perform better than GLS regarding prognosis.
Footnotes
Collaborators: Toulouse Amyloidosis Research Network collaborators: Laurent Alric, MD, PhD (Department of Internal Medicine and Digestive Diseases, Purpan University Hospital, Toulouse, France); Christophe Bureau, MD, PhD (Department of Hepatology-Gastroenterology, Rangueil University Hospital, Toulouse, France); Dominique Chauveau, MD, PhD (Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France); Pascal Cintas, MD, PhD (Department of Neurology, Purpan University Hospital, Toulouse, France); Magali Colombat, MD (Department of Pathology, IUCT Oncopôle, Toulouse, France); Audrey Delas, MD (Department of Pathology, IUCT Oncopôle, Toulouse, France); Delphine Dupin-Deguine, MD (Department of Genetic, Toulouse University Hospital, Toulouse, France); Stanislas Faguer, MD, PhD (Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France); Antoine Huart, MD (Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France); Bénédicte Puissant, MD (Immunology Laboratory, Toulouse University Hospital, Toulouse, France); Grégory Pugnet (Department of Internal Medicine, Toulouse University Hospital, Toulouse, France); Grégoire Prévot, MD (Department of Pneumology, Toulouse University Hospital, Toulouse, France); David Ribes, MD (Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France); Murielle Roussel MD (Department of Hematology, Toulouse University Hospital, Toulouse, France); Laurent Sailler, MD, PhD (Department of Internal Medicine and Digestive Diseases, Purpan University Hospital, Toulouse, France).
Contributors: AR-R analysed the echographic data and wrote the manuscript, EC and KR collected, analysed and interpreted the clinical, biological data. YL-B performed the statistical analysis. MG critically contributed to the manuscript. VB and PF contributed to the discussion and the reviewing. DC critically contributed to the manuscript and revised the last version of the manuscript after the reviewers’ comments. JR and OL designed and led the study, revised the manuscript and gave final approval of the version to be published.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: The study is conformed to the principles outlined in the Declaration of Helsinki. According to French law on ethics, patients were informed that their codified data will be used for the study. According to the French ethic and regulatory law (public health code) retrospective studies based on the exploitation of usual care data should not be submit at an ethic committee but they have to be declared or cover by reference methodology of the French National Commission for Informatics and Liberties. A collection and computer processing of personal and medical data were implemented to analyse the results of the research. Toulouse University Hospital signed a commitment of compliance to the reference methodology MR-004 of the French National Commission for Informatics and Liberties. After evaluation and validation by the data protection officer and according to the General Data Protection Regulation, this study completing all the criteria, it is registered in the register of retrospective study of the Toulouse University Hospital (number’s register: RIPH 2020-22) and cover by the MR-004 (French National Commission for Informatics and Liberties number: 2206723v0). This study was approved by Toulouse University Hospital and confirmed that ethic requirements were totally respected in this report.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request to lairez.o@chu-toulouse.fr.
References
- 1.Rapezzi C, Merlini G, Quarta CC, et al. . Systemic cardiac amyloidoses: disease profiles and clinical courses of the 3 main types. Circulation 2009;120:1203–12. 10.1161/CIRCULATIONAHA.108.843334 [DOI] [PubMed] [Google Scholar]
- 2.Rubin J, Steidley DE, Carlsson M, et al. . Myocardial contraction fraction by M-Mode echocardiography is superior to ejection fraction in predicting mortality in transthyretin amyloidosis. J Card Fail 2018;24:504–11. 10.1016/j.cardfail.2018.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tendler A, Helmke S, Teruya S, et al. . The myocardial contraction fraction is superior to ejection fraction in predicting survival in patients with al cardiac amyloidosis. Amyloid 2015;22:61–6. 10.3109/13506129.2014.994202 [DOI] [PubMed] [Google Scholar]
- 4.Kristen AV, Perz JB, Schonland SO, et al. . Non-Invasive predictors of survival in cardiac amyloidosis. Eur J Heart Fail 2007;9:617–24. 10.1016/j.ejheart.2007.01.012 [DOI] [PubMed] [Google Scholar]
- 5.Gillmore JD, Damy T, Fontana M, et al. . A new staging system for cardiac transthyretin amyloidosis. Eur Heart J 2018;39:2799–806. 10.1093/eurheartj/ehx589 [DOI] [PubMed] [Google Scholar]
- 6.Grogan M, Scott CG, Kyle RA, et al. . Natural History of Wild-Type Transthyretin Cardiac Amyloidosis and Risk Stratification Using a Novel Staging System. J Am Coll Cardiol 2016;68:1014–20. 10.1016/j.jacc.2016.06.033 [DOI] [PubMed] [Google Scholar]
- 7.Siddiqi OK, Ruberg FL. Cardiac amyloidosis: an update on pathophysiology, diagnosis, and treatment. Trends Cardiovasc Med 2018;28:10–21. 10.1016/j.tcm.2017.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park JJ, Park J-B, Park J-H, et al. . Global Longitudinal Strain to Predict Mortality in Patients With Acute Heart Failure. J Am Coll Cardiol 2018;71:1947–57. 10.1016/j.jacc.2018.02.064 [DOI] [PubMed] [Google Scholar]
- 9.Gunasekaran P, Panaich S, Briasoulis A, et al. . Incremental value of two dimensional speckle tracking echocardiography in the functional assessment and characterization of subclinical left ventricular dysfunction. Curr Cardiol Rev 2017;13:32–40. 10.2174/1573403X12666160712095938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Galli E, Vitel E, Schnell F, et al. . Myocardial constructive work is impaired in hypertrophic cardiomyopathy and predicts left ventricular fibrosis. Echocardiography 2019;36:74–82. 10.1111/echo.14210 [DOI] [PubMed] [Google Scholar]
- 11.Chan J, Edwards NFA, Khandheria BK, et al. . A new approach to assess myocardial work by non-invasive left ventricular pressure-strain relations in hypertension and dilated cardiomyopathy. Eur Heart J Cardiovasc Imaging 2019;20:31–9. 10.1093/ehjci/jey131 [DOI] [PubMed] [Google Scholar]
- 12.Edwards NFA, Scalia GM, Shiino K, et al. . Global myocardial work is superior to global longitudinal strain to predict significant coronary artery disease in patients with normal left ventricular function and wall motion. J Am Soc Echocardiogr 2019;32:947–57. 10.1016/j.echo.2019.02.014 [DOI] [PubMed] [Google Scholar]
- 13.Russell K, Eriksen M, Aaberge L, et al. . A novel clinical method for quantification of regional left ventricular pressure-strain loop area: a non-invasive index of myocardial work. Eur Heart J 2012;33:724–33. 10.1093/eurheartj/ehs016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Donal E, Bergerot C, Thibault H, et al. . Influence of afterload on left ventricular radial and longitudinal systolic functions: a two-dimensional strain imaging study. Eur J Echocardiogr 2009;10:914–21. 10.1093/ejechocard/jep095 [DOI] [PubMed] [Google Scholar]
- 15.Yingchoncharoen T, Agarwal S, Popović ZB, et al. . Normal ranges of left ventricular strain: a meta-analysis. J Am Soc Echocardiogr 2013;26:185–91. 10.1016/j.echo.2012.10.008 [DOI] [PubMed] [Google Scholar]
- 16.Austin BA, Tang WHW, Rodriguez ER, et al. . Delayed hyper-enhancement magnetic resonance imaging provides incremental diagnostic and prognostic utility in suspected cardiac amyloidosis. JACC Cardiovasc Imaging 2009;2:1369–77. 10.1016/j.jcmg.2009.08.008 [DOI] [PubMed] [Google Scholar]
- 17.Perugini E, Guidalotti PL, Salvi F, et al. . Noninvasive etiologic diagnosis of cardiac amyloidosis using 99mTc-3,3-diphosphono-1,2-propanodicarboxylic acid scintigraphy. J Am Coll Cardiol 2005;46:1076–84. 10.1016/j.jacc.2005.05.073 [DOI] [PubMed] [Google Scholar]
- 18.Gillmore JD, Maurer MS, Falk RH, et al. . Nonbiopsy diagnosis of cardiac transthyretin amyloidosis. Circulation 2016;133:2404–12. 10.1161/CIRCULATIONAHA.116.021612 [DOI] [PubMed] [Google Scholar]
- 19.Lang RM, Badano LP, Mor-Avi V, et al. . Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of echocardiography and the European association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging 2015;16:233–71. 10.1093/ehjci/jev014 [DOI] [PubMed] [Google Scholar]
- 20.Nagueh SF, Smiseth OA, Appleton CP, et al. . Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of echocardiography and the European association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging 2016;17:1321–60. 10.1093/ehjci/jew082 [DOI] [PubMed] [Google Scholar]
- 21.Voigt J-U, Pedrizzetti G, Lysyansky P, et al. . Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/Industry Task force to standardize deformation imaging. Eur Heart J Cardiovasc Imaging 2015;16:1–11. 10.1093/ehjci/jeu184 [DOI] [PubMed] [Google Scholar]
- 22.King DL, El-Khoury Coffin L, Maurer MS. Myocardial contraction fraction: a volumetric index of myocardial shortening by freehand three-dimensional echocardiography. J Am Coll Cardiol 2002;40:325–9. 10.1016/S0735-1097(02)01944-7 [DOI] [PubMed] [Google Scholar]
- 23.Phelan D, Collier P, Thavendiranathan P, et al. . Relative apical sparing of longitudinal strain using two-dimensional speckle-tracking echocardiography is both sensitive and specific for the diagnosis of cardiac amyloidosis. Heart 2012;98:1442–8. 10.1136/heartjnl-2012-302353 [DOI] [PubMed] [Google Scholar]
- 24.Clemmensen TS, Eiskjær H, Ladefoged B, et al. . Prognostic implications of left ventricular myocardial work indices in cardiac amyloidosis. Eur Heart J Cardiovasc Imaging (Published Online First: June 2020). [DOI] [PubMed]
- 25.Di Bella G, Minutoli F, Pingitore A, et al. . Endocardial and epicardial deformations in cardiac amyloidosis and hypertrophic cardiomyopathy. Circ J 2011;75:1200–8. 10.1253/circj.CJ-10-0844 [DOI] [PubMed] [Google Scholar]
- 26.Buss SJ, Emami M, Mereles D, et al. . Longitudinal left ventricular function for prediction of survival in systemic light-chain amyloidosis: incremental value compared with clinical and biochemical markers. J Am Coll Cardiol 2012;60:1067–76. 10.1016/j.jacc.2012.04.043 [DOI] [PubMed] [Google Scholar]
- 27.Milani P, Dispenzieri A, Scott CG, et al. . Independent prognostic value of stroke volume index in patients with immunoglobulin light chain amyloidosis. Circ Cardiovasc Imaging 2018;11:e006588. 10.1161/CIRCIMAGING.117.006588 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aimo A, Vergaro G, Castiglione V, et al. . Safety and tolerability of neurohormonal antagonism in cardiac amyloidosis. Eur J Intern Med 2020. 10.1016/j.ejim.2020.05.015. [Epub ahead of print: 29 May 2020] (Published Online First: May 2020). [DOI] [PubMed] [Google Scholar]
- 29.Manganaro R, Marchetta S, Dulgheru R, et al. . Echocardiographic reference ranges for normal non-invasive myocardial work indices: results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging 2019;20:582–90. 10.1093/ehjci/jey188 [DOI] [PubMed] [Google Scholar]
- 30.Escher F, Senoner M, Doerler J, et al. . When and how do patients with cardiac amyloidosis die? Clin Res Cardiol 2020;109:78–88. 10.1007/s00392-019-01490-2 [DOI] [PMC free article] [PubMed] [Google Scholar]