Abstract
The aim of the present study was to explore the risk factors of diabetes peripheral neuropathy (DPN) in type 2 diabetes mellitus, and to propose corresponding nursing intervention measures to provide the basis for early diagnosis, treatment and prevention of DPN. A retrospective analysis was performed on 130 patients with type 2 diabetes mellitus (T2DM) in the communities surrounding our hospital from January 2017 to June 2018. They were divided into a DPN group (62 cases) and non-DPN group (control group, 68 cases). First, a univariate analysis was performed. DPN or non-DPN were used as dependent variables, and statistically significant parameters in univariate analysis were used as independent variables. Multivariate logistic regression analysis was included to analyze the peripheral nerves in type 2 diabetes mellitus. At the same time, the patients with DPN were given individualized comprehensive nursing intervention. Univariate analysis showed that body mass index (BMI), diabetes duration, smoking history, drinking history, family history of diabetes, fasting blood glucose (FBG), postprandial blood glucose (PBG), glycosylated hemoglobin (HbA1c), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), systolic blood pressure, 24-h urine microalbumin excretion (24hmALB), glomerular filtration rate (GFR), serum creatinine in DPN group were significantly different from those in the control group (P<0.05). Logistic regression analysis showed that the duration of diabetes, PBG, glycosylated hemoglobin (HbA1c), HDL-C, 24hmALB, and GFR were independent risk factors for DPN. The quality of life [Physiological function (PF), Role-physical (RP), Role-emotional (RE), General health (GH), Social function (SF), Mental health (MH), Bodily pain (BP), Vitality (VT)] in the DPN group was significantly improved after six months of individualized nursing intervention compared with that before intervention (P<0.05). In conclusion, the high risk factors of DPN should be identified as early as possible, to prevent and delay the occurrence and development of diabetes peripheral neuropathy, so as to improve the quality of life of these patients.
Keywords: diabetes mellitus, type 2, peripheral neuropathy, risk factors, nursing countermeasures
Introduction
Diabetes mellitus (DM) has now become the third most common non-communicable disease after cardiovascular diseases and malignant tumors, that seriously threatens human life and health (1). WHO research data show that, with the improvement of living standards and changes in living behavior, the number of global diabetes patients is predicted to increase from 415 million in 2015 to 642 million by 2040, bringing a severe problem in prevention and control of the disease (2). Diabetes peripheral neuropathy (DPN) is one of the most common complications of type 2 diabetes, and it is also the most common clinical cause of neuropathy (3). The main clinical manifestations of DPN are burning pain in the distal limbs; however the early symptoms are not obvious. If not properly controlled, the symptoms gradually worsen and reversal in the later stages is difficult, eventually resulting in progressive aggravation of pain and serious impact on the quality of life of patients. According to the International Diabetes Federation Survey (4), the prevalence of DPN in patients with DM is as high as 30-50%, with diabetes foot (DF) being the most common DPN manifestation in severe cases, resulting in foot ulcers, gangrene and even amputations. If early evaluation, diagnosis and treatment of patients with DPN can be performed, the incidence of lower extremity ulcers and amputations would be effectively reduced. The incidence of risk could be reduced by 60 and 85%, respectively (4); therefore, early prevention and control is extremely important. DPN pathological changes often occur when they are asymptomatic, thus early assessment of their risk factors and active personalized nursing interventions are the key to prevent and reduce the incidence of DPN (5). This research statistically analyzed the general data parameters and related clinical data parameters of patients with DM. Multivariate logistic regression was used to analyze the independent risk factors for DPN. Individualized comprehensive nursing intervention was applied for patients with DPN in order to provide theoretical basis for comprehensive prevention and treatment of DPN.
Patients and methods
Patients and clinical materials
One hundred and thirty patients with type 2 diabetes mellitus (T2DM) in the community surrounding Weifang People's Hospital from January 2017 to June 2018 were selected as the research subjects. They were divided into a DPN group (62 cases) and non-DPN group (68 cases) according to the presence or absence of peripheral neuropathy. A unified questionnaire was used to investigate the general clinical data of patients, including sex, age, body mass index (BMI), comorbidities, blood pressure and life history. Patient biochemical indexes were regularly tested according to research requirements. Inclusion criteria were as follows: i) T2DM and DPN diagnostic criteria were in line with the 2013 American Diabetes Association (ADA) Guidelines and the ADA Position Statement on Peripheral Diabetes Neuropathy (2017) (6); ii) Peripheral neuropathy included symmetrical peripheral neuropathy and asymmetric neuropathy. Symmetrical peripheral neuropathy refers to multiple peripheral neuropathy with symmetrical distal limbs, distal muscle weakness and muscular atrophy, reduction or disappearance of tendon reflexes, and may also be accompanied by impairment of autonomic nerve function. Asymmetric neuropathy included early onset of proximal weakness of one leg and muscular atrophy, about half of them gradually involved the proximal pelvic girdle muscles of both legs, manifested as difficulty in standing up, walking and stepping on stairs, often accompanied by sharp pain in the deep thigh and lumbosacral area; iii) Patients were without barrier in language or communication, with clear awareness of information; iv) Patients agreed to participate in the research and cooperated with researchers. Exclusion criteria were as follows: i) Patients without the ability to act independently and unable to communicate normally, and who did not cooperate with the researcher; ii) Patients with type 1 diabetes; iii) Patients with secondary diabetes; iv) patients with peripheral nerve pain caused by infectious or other metabolic diseases; v) Patients with other orthopedic and neurological diseases, or severe heart, liver and kidney dysfunction. There was no significant difference in age, sex and other general data between the two groups (P>0.05), which was comparable. This study was approved by the Ethics Committee of Weifang People's Hospital (Slunlh:20161223).
Methods
Laboratory index inspection
Biochemical indexes (blood glucose, blood lipids, creatinine) and urine microalbumin were detected by Siemens automatic biochemical analyzer ADVIA2400 (Siemens), and reagents for blood glucose, blood lipids, creatinine and urine microalbumin were provided by Siemens. HbAlc was detected by the G8 glycosylated hemoglobin analyzer (Japan Tosoh Co., Ltd.). Body mass index (BMI)=weight (kg)/the square of the height (m). Biochemical indicators were evaluated every three months.
Intervention methods
Due to the convenience of community health services, patients were followed up via various forms (telephone, door-to-door follow-up, visits to the community service center).
Intervention measures
Patients in both groups were given routine care. The DPN group received targeted personalized comprehensive nursing intervention on the basis of routine care for six months. Comprehensive interventions were implemented, such as health education, diet intervention, exercise intervention, medication intervention and prevention of complications for diabetes patients. First, health education for patients included oral education, brochures, multimedia promotion, and patient interaction. Multiple modes are used to spread knowledge of DNP, allowing patients to fully understand the risk factors, hazards, and the importance of prevention of DPN. The patients must receive strengthened psychological care and self-intervention management (7). Second, diet intervention is the basis to control blood glucose (8). A personalized diet plan should be developed according to the specific circumstances of the patient. According to the patient's age, physique and condition, nursing staff should calculate calories and nutrients every day, reduce high-calorie and high-sugar foods, and add foods with rich coarse fiber. The proportion of the three meals should be reasonable, and the calories for breakfast, lunch and dinner are 1/5, 2/5 and 2/5, respectively. Patients should quit alcohol and tobacco, change poor lifestyle and behavior habits, develop good eating habits, so as to control blood sugar, which can significantly reduce the occurrence of chronic complications of T2DM (9). Third, the goals of medication, correct medication methods and medication precautions should be explained to the patients. The patients should be guided to improve the awareness in blood sugar self-testing and self-management ability. Fourth, diabetes foot is a common type of diabetes peripheral neuropathy (10), thus patients should strengthen foot care and exercise, promote blood circulation, improve the peripheral nerve nutrition of the patient's limbs, which could significantly reduce the incidence of diabetes foot. Diabetic foot ulcers should be treated as soon as possible. Fifth, exercise can reduce obesity, insulin resistance, and improve the body's regulation of blood glucose (11). Therefore, patients should be encouraged to exercise according to their own conditions, especially distal muscle strength training and endurance training, so as to reduce the incidence of complications (12). All of the above interventions are helpful to prevent and delay the occurrence and development of diabetic peripheral neuropathy.
Evaluation
The SF-36 quality of life table (13) was used for quality of life evaluation. There are 36 items in the SF-36 quality of life table: 10 items for Physiological function (PF), 4 items for Role physical (RP), 3 items for Emotional function (RE), 6 items for General health (GH), 2 items for Social function (SF), 5 items of Mental health (MH), 2 items of Body pain (BP), 4 items of Vigor (VT), and patients were evaluated in 8 dimensions. The higher the score, the better the patient's physical function and mental and psychological status.
Statistical methods
SPSS 25.00 statistical software (IBM Corp.) was used for data processing. The continuous variables were expressed as mean ± standard deviation. t-test was used for the comparison between groups, and χ2 test was used for the comparison between all categorial variable groups. DPN or non-DPN were regarded as dependent variables, and statistically significant parameters in univariate analysis were used as independent variables. Multivariate logistic regression analysis was used to analyze the peripheral nerves in type 2 diabetes mellitus. P<0.05 was considered to indicate a statistically significant difference.
Results
Single factor analysis of general data and laboratory data of the two groups of patients
Statistical analysis was performed on general clinical data of the two groups of patients, including sex, age, diastolic blood pressure, systolic blood pressure, body mass index (BMI), duration of diabetes, smoking history, drinking history, and laboratory data including glomerular filtration rate (GFR), 24 h urinary microalbumin excretion (24hmALB), blood creatinine, fasting blood glucose (FBG), postprandial blood glucose (PBG), glycated hemoglobin (HbA1c), triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C). As shown in Table I, the results revealed that when comparing the general clinical data between the DPN group and the control group, there was no significant difference in sex, age, and diastolic blood pressure (P>0.05); the body mass index (BMI) of the DPN group was significantly larger than that of the control group, with statistically significant difference (P<0.05); the duration of diabetes in the DPN group was significantly longer than that of the control group, and the difference was statistically significant (P<0.001); the population of smoking and drinking history in the DPN group was significantly increased compared with the control group (P<0.05); the number of people with a family history of diabetes in the DPN group was significantly higher compared with the control group (P<0.05), and the systolic blood pressure in the DPN group was significantly higher than that in the control group (P<0.05). When comparing the DPN group with the control group in terms of laboratory data, the DPN group was unable to effectively control the long-term blood glucose, which caused damage to the kidney glomerulus to varying degrees, resulting in a significant reduction in glomerular filtration rate (GFR) and 24-h urine microvolume. The albumin excretion volume (24hmALB) was significantly increased, and the difference was statistically significant compared with the control group (P<0.001). The serum creatinine was significantly increased compared with the control group (P<0.05). Patients in the DNP group were at high blood glucose levels for an extended period of time, with impaired glucose tolerance. The fasting blood glucose (FBG) was significantly increased compared with the control group (P<0.05). Postprandial blood glucose (PBG) and glycated hemoglobin (HbA1c) were significantly increased compared with the control group (P<0.001). Triglycerides (TG) in blood lipids were significantly increased compared with the control group (P<0.05), HDL cholesterol (HDL-C) was significantly decreased compared with the control group (P<0.001).
Table I.
Univariate analysis of general data of the two groups of patients.
| Variable | DPN group (n=62) | Non-DPN group (n=68) | χ2/t value | P-value |
|---|---|---|---|---|
| Sex (n) | ||||
| Male | 34 | 37 | χ2=0.291 | 0.590 |
| Female | 28 | 31 | ||
| Age (years) | 65.32±5.79 | 54.12±5.37 | t=1.554 | 0.171 |
| BMI (kg/m2) | 27.16±2.25 | 21.78±1.69 | t=2.986 | 0.020 |
| Course of disease (years) | 15.25±1.43 | 8.67±1.54 | t=4.230 | <0.001 |
| Smoking history [n (%)] | 35 (56.45) | 24 (38.71) | χ2=5.857 | 0.016 |
| Drinking history [n (%)] | 36 (58.06) | 26 (38.24) | χ2=5.112 | 0.024 |
| Family history of diabetes [n (%)] | 30 (48.39) | 20 (29.41) | χ2=6.660 | 0.010 |
| Blood pressure (mmHg) | ||||
| Systolic pressure | 135.21±4.34 | 122.41±3.48 | t=2.521 | 0.045 |
| Diastolic pressure | 85.36±1.37 | 82.45±1.24 | t=1.727 | 0.135 |
| 24hmALB (mg/24 h) | 298.67±15.37 | 165.43±10.29 | t=7.860 | <0.001 |
| GFR [ml/(min/1.73 m2)] | 65.38±10.24 | 104.69±12.37 | t=-4.679 | <0.001 |
| Serum creatinine (µmol/l) | 91.97±13.64 | 62.51±11.54 | t=2.423 | 0.049 |
| FBG (mmol/l) | 7.97±0.86 | 5.55±0.54 | t=2.842 | 0.029 |
| PBG (mmol/l) | 12.35±1.06 | 7.23±0.78 | t=5.262 | <0.001 |
| HbAlc (%) | 11.78±1.44 | 7.72±0.85 | t=5.712 | <0.001 |
| TG (mmol/l) | 3.45±0.84 | 1.77±0.51 | t=2.608 | 0.045 |
| TC (mmol/l) | 4.67±0.86 | 3.66±0.67 | t=1.015 | 0.349 |
| HDL-C (mmol/l) | 0.86±0.11 | 1.32±0.13 | t=4.689 | <0.001 |
| LDL-C (mmol/l) | 3.79±0.75 | 3.25±0.78 | t=0.547 | 0.604 |
Data are expressed as mean ± SD (%). DPN, diabetes peripheral neuropathy; BMI, body mass index; 24hmALB, 24-h urine microalbumin excretion; GFR, glomerular filtration rate; FBG, fasting blood glucose; PBG, postprandial blood glucose; HbAlc, glycosylated hemoglobin; TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Logistic regression analysis of risk factors related to peripheral neuropathy in type 2 diabetes mellitus
With DPN as the dependent variable, the statistically significant parameters in the univariate analysis were included in multivariate logistic regression analysis as independent variables, including patient body mass index (BMI), duration of diabetes, smoking history, drinking history, family history of diabetes, systolic blood pressure, 24-h urine microalbumin excretion (24hmALB), glomerular filtration rate (GFR), blood creatinine, fasting blood glucose (FBG), postprandial blood glucose (PBG), glycated hemoglobin (HbA1c), triglyceride (TG) and high-density lipoprotein cholesterol (HDL-C). Multivariate logistic regression analysis was included. As shown in Table II, the results revealed that the duration of diabetes, PBG, HbA1c, HDL-C, 24hmALB, and GFR were independent risk factors for DPN.
Table II.
Logistic regression analysis of DPN-related risk factors.
| Variable | B | SE | Wald | P-value | OR | 95% CI |
|---|---|---|---|---|---|---|
| BMI | 0.459 | 0.533 | 3.756 | 0.061 | 1.236 | 0.925-1.364 |
| Duration of diabetes | 1.035 | 0.532 | 14.325 | <0.001 | 3.567 | 1.472-5.631 |
| Smoking history | 0.674 | 0.369 | 3.012 | 0.097 | 1.110 | 0.567-1.964 |
| Drinking history | 0.236 | 0.297 | 2.531 | 0.101 | 2.036 | 0.712-2.036 |
| Family history of diabetes | 0.179 | 0.269 | 3.826 | 0.055 | 1.278 | 0.539-2.115 |
| FBG | 0.486 | 0.452 | 1.378 | 0.214 | 1.634 | 0.768-3.214 |
| PBG | 0.765 | 0.129 | 12.306 | <0.001 | 2.354 | 1.563-2.896 |
| HbAlc | 1.032 | 0.345 | 13.256 | <0.001 | 2.798 | 1.563-3.015 |
| TG | 0.4355 | 0.502 | 0.821 | 0.436 | 1.587 | 0.586-2.124 |
| HDL-C | 0.869 | 0.236 | 11.967 | <0.001 | 1.896 | 1.335-2.036 |
| Systolic pressure | 0.642 | 0.521 | 0.621 | 0.524 | 1.253 | 0.657-1.962 |
| 24hmALB | 0.596 | 0.306 | 13.675 | <0.001 | 2.015 | 1.764-3.125 |
| GFR | 1.158 | 0.234 | 14.012 | <0.001 | 3.269 | 1.564-4.326 |
DPN, diabetes peripheral neuropathy; OR, odds ratio; CI, confidence interval; BMI, body mass index; FBG, fasting blood glucose; PBG, postprandial blood glucose; HbAlc, glycosylated hemoglobin; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; 24hmALB, 24-h urine microalbumin excretion; GFR, glomerular filtration rate.
Comparison of the quality of life of patients in the DPN group before and after comprehensive nursing intervention
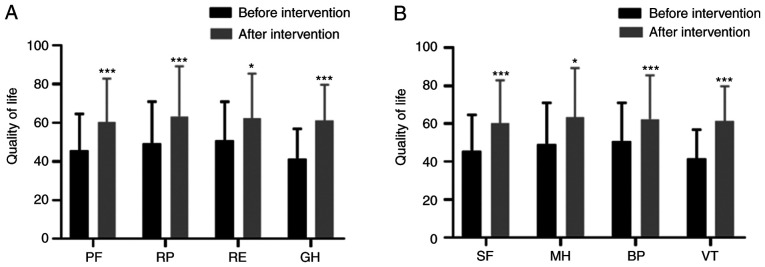
Six months after individualized comprehensive nursing intervention, the quality of life indexes (PF, RP, RE, GH) of patients in the DPN group were significantly higher than these indexes before intervention (t=7.829, t=4.018, t=3.028, t=6.048; P<0.001, P=0.001, P=0.015, P<0.001) (all P<0.05) (Fig. 1A). The quality of life indexes (SF, MH, BP, VT) after intervention were higher than those before the intervention (t=4.258, t=3.856, t=4.233, t=6.278; P=0.001, P=0.003, P=0.001, P<0.001) (Fig. 1B).
Figure 1.
Comparison of life quality in the DPN group before and after comprehensive nursing intervention. (A) The quality of life indexes (PF, RP, RE, GH) of patients in the DPN group were significantly higher than these indexes before intervention (t=7.829, t=4.018, t=3.028, t=6.048; P<0.001, P=0.001, P=0.015, P<0.001). (B) The quality of life indexes (SF, MH, BP, VT) after intervention were higher than those before the intervention (t=4.258, t=2.856, t=4.233, t=6.278; P=0.001, P=0.023, P=0.001, P<0.001). *P<0.05 and ***P<0.001. PF, physiological function; RP, role-physical; RE, role-emotional; GH, general health; SF, social function; MH, mental health; BP, bodily pain; VT, vitality.
Quality of life after comprehensive nursing intervention in the DPN group compared with non-DPN control group
There was no significant difference in the quality of life (PF, RP, RE, GH, SF, MH, BP, VT) between the DPN group and non-DPN group (t=0.103, t=0.208, t=0.234, t=0.318, t=0.391, t=0.233, t=0.086, t=0.278; P=0.901, P=0.835, P=0.823, P=0.761, P=0.709, P=0.824, P=0.934, P=0.792) (all P>0.05) (Table III).
Table III.
Comparison of life quality between the DPN group and the non-DPN group after intervention.
| Project | DPN group after intervention | Non-DPN group | t value | P-value |
|---|---|---|---|---|
| PF | 66.82±14.53 | 64.42±11.98 | 0.103 | 0.901 |
| RP | 61.74±14.31 | 57.16±13.35 | 0.208 | 0.835 |
| RE | 56.37±13.01 | 59.83±13.69 | 0.234 | 0.823 |
| GH | 58.62±13.97 | 55.47±12.85 | 0.318 | 0.761 |
| SF | 60.25±14.29 | 56.80±13.57 | 0.391 | 0.709 |
| MH | 63.42±16.34 | 60.24±14.64 | 0.233 | 0.824 |
| BP | 62.37±14.59 | 64.12±13.30 | 0.086 | 0.934 |
| VT | 61.255±14.66 | 57.82±13.78 | 0.276 | 0.792 |
Data are expressed as mean ± SD (%). PF, physiological function; RP, role-physical; RE, role-emotional; GH, general health; SF, social function; MH, mental health; BP, bodily pain; VT, vitality.
Discussion
At present, the pathogenesis of diabetes peripheral neuropathy (DPN) is unclear, and scholars believe that factors such as metabolic disorders, vascular endothelial damage, chronic microinflammatory response, and oxidative stress are involved in the pathogenesis (14). Toronto Diabetes Neuropathy Committee (15) believes that long-term hyperglycemia, cardiovascular risk and other factors result in metabolic disorders and microvascular changes, leading to chronic, symmetrical sensorimotor polyneuropathy. DPN, as the most common diabetes mellitus (DM) neuropathy today, has certain influence on the peripheral nervous system. Symmetry of numbness, paresthesia and pain are the typical clinical manifestation of DPN. If not controlled, DPN tends to develop toward the center of body, then sock-like sensation loss occurs in the feet, above the ankles, and in both hands (16). As a result, muscle sensory abnormalities and proprioceptive dysfunction in patients may cause joint disease, ulcers, and even gangrene in severe cases (17). Veves et al (18) found that quality of life scores, such as behavior ability, physiological function, vigor, social function and mental health, were significantly reduced because of the impaired ability of action and balance caused by peripheral nerve pain. DPN is also one of the main causes of non-traumatic lower limb amputation. Therefore, early assessment of risk factors and targeted care interventions are important to improve the quality of life of these patients (19).
Metabolic disorders, vascular injury, cytokine abnormalities, oxidative stress, neurotrophic factor deficiency, and immune factors all play roles in the development of DPN (20), of which long-term hyperglycemia caused by metabolic disorders is the main cause of DPN (21). Long-term substandard blood glucose control leads to increased levels of non-enzymatic glycosylation products in peripheral nerve peripheral neurons, which can increase the flux of the polyphenol pathway, activate protein kinase C and stimulate oxidative stress, enhance the formation of late glycosylation end products, and cause corresponding clinical symptoms such as peripheral nerve damage (22). The duration of diabetes is an independent risk factor for DPN. Studies have found that the risk of developing DPN increases by 6.70% each year after the onset of diabetes. The incidence of DPN in patients with DM increases with a gradient of 30.00 (5 years), 60.00 (10 years) and 90.00% (20 years), respectively (23). The glycated hemoglobin (HbA1c) level reflects the blood glucose levels in patients with DM for 8-12 weeks, thus HbA1c can accurately reflect the recent blood glucose control. Research found that for every 1% increase in HbAlc, the risk of DPN increased by 5.30%, indicating that HbA1c is also a risk factor for DPN (24). Type 2 diabetes mellitus (T2DM) is caused by insufficient or relatively insufficient insulin secretion, which causes high blood sugar. Sustained high blood sugar can cause toxic effects on nerve cells and neurological dysfunction, leading to peripheral neuropathy. The results of the present study demonstrated that with the extension of the duration of diabetes and unqualified glycemic control, the prevalence of DPN increases significantly. Thus, the fundamental measures for the prevention of DPN is to control blood sugar.
Abnormal blood lipid metabolism is also a risk factor for DPN. Studies have found that patients with DPN have higher peripheral blood TG, TC content, and lower HDL-C levels (25). Wiggin et al (26) explored the degree of DPN neuropathy and blood lipids through nerve biopsy technology, and found that a decrease in HDL-C levels in patients with DM can accelerate the course of DPN. Padilla et al (27) found that palmitic acid, an important component of blood lipids, is a long-chain saturated fatty acid, which is involved in the occurrence of DPN, further suggesting a close correlation between lipid levels and DPN. In addition, high blood lipids could cause blood viscosity, and indirectly cause peripheral blood vessel blockage. Peripheral blood supply is reduced, and consequently nutrients do not reach peripheral nerve cells through peripheral blood vessels, finally leading to DPN (28). Research has confirmed that diabetes nephropathy and DPN are strongly associated with low HDL-C levels and have been proven to be independent risk predictors of lower limb amputation and wound-related death in diabetes foot ulcers (28). Multivariate logistic regression results showed that HDL-C is an independent risk factor for DPN among lipid metabolism-related parameters. In addition, it has been demonstrated that microvascular complications also play an important role in the development of DPN (29). Diabetes nephropathy is one of the diabetes microvascular complications. A large amount of proteinuria indicates that patients have a high risk of developing DPN, and a low level of GFR [<60 ml/min/1.73 m2)] is an important risk factor for DPN (30). This research also confirmed that high levels of microalbuminuria and low levels of GFR were independent risk factors for DPN. Although smoking history, drinking history and BMI were not independent risk factors for DPN after logistic regression in this study, smoking and drinking are closely related to atherosclerosis. Atherosclerosis may increase the risk of peripheral neuropathy by increasing vascular endothelial dysfunction (31). In addition, obesity is inextricably linked to insulin resistance, which is usually exacerbated by fat accumulation. With the increase in BMI, individuals are prone to insulin resistance (32). The insulin-mediated diastolic function of vascular endothelial cells could be impaired, and the risk of diabetic peripheral neuropathy is increased. The occurrence and development of DPN are associated with multiple risk factors, thus it is necessary to involve multiple risk factors in the early comprehensive evaluation of DPN.
Community health services are integrated with prevention, medical treatment, health care, rehabilitation and health education. Community nursing has the characteristics of convenience, economy and flexibility. In particular, there are obvious advantages in nursing interventions for chronic diseases (33), thus we should make full use of the advantages of community health services. This research showed that the quality of life of the DPN group intervention was significantly improved compared with that before intervention, and the effect was remarkable.
In conclusion, analyzing the risk factors of developing DPN and implementing comprehensive and reasonable interventions in the community can significantly improve the quality of life of patients. There are certain limitations to this study. The intervention time of this study was only 6 months. In the future, we will make full use of the advantages of community services to promote health education, and improve the level of community health services.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
ZL designed the study and drafted the manuscript. XL and BX were responsible for the collection and analysis of the experimental data. SW, TG and HL revised the manuscript critically for important intellectual content. SW and TG were responsible for the inspection of biochemical indexes (blood glucose, blood lipids, creatinine) and urine microalbumin. HL was responsible for the quality of life evaluation.. All authors read and approved the manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Weifang People's Hospital, (Weifang, Shandong, China). Patients who participated in this research, signed an informed consent and had complete clinical data. Signed written informed consents were obtained from all patients and/or guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Ang L, Cowdin N, Mizokami-Stout K, Pop-Busui R. Update on the management of diabetic neuropathy. Diabetes Spectr. 2018;31:224–233. doi: 10.2337/ds18-0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Popkin BM. Nutrition transition and the global diabetes epidemic. Curr Diab Rep. 2015;15(64) doi: 10.1007/s11892-015-0631-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Albers JW, Pop-Busui R. Diabetic neuropathy: Mechanisms, emerging treatments, and subtypes. Curr Neurol Neurosci Rep. 2014;14(473) doi: 10.1007/s11910-014-0473-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hewston P, Deshpande N. Falls and balance impairments in older adults with Type 2 diabetes: Thinking beyond diabetic peripheral neuropathy. Can J Diabetes. 2016;40:6–9. doi: 10.1016/j.jcjd.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 5.Themistocleous AC, Ramirez JD, Shillo PR, Lees JG, Selvarajah D, Orengo C, Tesfaye S, Rice AS, Bennett DL. The pain in neuropathy study (PiNS): A cross-sectional observational study determining the somatosensory phenotype of painful and painless diabetic neuropathy. Pain. 2016;157:1132–1145. doi: 10.1097/j.pain.0000000000000491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pop-Busui R, Boulton AJ, Feldman EL, Bril V, Freeman R, Malik RA, Sosenko JM, Ziegler D. Diabetic neuropathy: A position statement by the American Diabetes Association. Diabetes Care. 2017;40:136–154. doi: 10.2337/dc16-2042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schillinger D, Grumbach K, Piette J, Wang F, Osmond D, Daher C, Palacios J, Sullivan GD, Bindman AB. Association of health literacy with diabetes outcomes. JAMA. 2002;288:475–482. doi: 10.1001/jama.288.4.475. [DOI] [PubMed] [Google Scholar]
- 8.Callaghan BC, Little AA, Feldman EL, Hughes RA. Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database Syst Rev. 2012;13(CD007543) doi: 10.1002/14651858.CD007543.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Miller CK, Edwards L, Kissling G, Sanville L. Nutrition education improves metabolic outcomes among older adults with diabetes mellitus: Results from a randomized controlled trial. Prev Med. 2002;34:252–259. doi: 10.1006/pmed.2001.0985. [DOI] [PubMed] [Google Scholar]
- 10.Im S, Kim SR, Park JH, Kim YS, Park GY. Assessment of the medial dorsal cutaneous, dorsal sural, and medial plantar nerves in impaired glucose tolerance and diabetic patients with normal sural and superficial peroneal nerve responses. Diabetes Care. 2012;35:834–839. doi: 10.2337/dc11-1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu B, Yang Z, Wang M, Yang Z, Gong W, Yang Y, Wen J, Zhang Z, Zhao N, Zhu X, et al. High prevalence of diabetic neuropathy in population-based patients diagnosed with type 2 diabetes in the Shanghai downtown. Diabetes Res Clin Pract. 2010;88:289–294. doi: 10.1016/j.diabres.2010.02.002. [DOI] [PubMed] [Google Scholar]
- 12.Liebreich T, Plotnikoff RC, Courneya KS, Boulé N. Diabetes NetPLAY: A physical activity website and linked email counselling randomized intervention for individuals with type 2 diabetes. Int J Behav Nutr Phys Act. 2009;6(18) doi: 10.1186/1479-5868-6-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Campolina AG, Ciconelli RM. SF-36 and the development of new assessment tools for quality of life. Acta Reumatol Port. 2008;33:127–133. (In Portuguese) [PubMed] [Google Scholar]
- 14.Qu GB, Wang LL, Tang X, Wu W, Sun YH. The association between vitamin D level and diabetic peripheral neuropathy in patients with type 2 diabetes mellitus: An update systematic review and meta-analysis. J Clin Transl Endocrinol. 2017;9:25–31. doi: 10.1016/j.jcte.2017.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Javed S, Alam U, Malik RA. Burning through the pa in: Treatments for diabetic neuropathy. Diabetes Obes Metab. 2015;17:1115–1125. doi: 10.1111/dom.12535. [DOI] [PubMed] [Google Scholar]
- 16.Tesfaye S, Selvarajah D. Advances in the epidemiology, pathogenesis and management of diabetes peripheral neuropathy. Diabetes Metab Res Rev. 2012;28:8–14. doi: 10.1002/dmrr.2239. [DOI] [PubMed] [Google Scholar]
- 17. doi: 10.11604/pamj.2018.30.4.14765. Djibril AM, Mossi EK, Djagadou AK, Balaka A, Tchamdja T and Moukaila R: Epidemiological, diagnostic, therapeutic and evolutionary features of diabetic foot: A study conducted at the medico-surgical clinic, University Hospital Sylvanus Olympio in Lomé. Pan Afr Med J 30: 4, 2018 (In French). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Veves A, Backonja M, Malik RA. Painful diabetic neuropathy: Epidemiology, natural history, early diagnosis, and treatment options. Pain Med. 2008;9:660–674. doi: 10.1111/j.1526-4637.2007.00347.x. [DOI] [PubMed] [Google Scholar]
- 19.Kim SS, Won JC, Kwon HS, Kim CH, Lee JH, Park TS, Ko KS, Cha BY. Prevalence and clinical implications of painful diabetes peripheral neuropathy in type 2 diabetes mellitus: Results from a nationwide hospital-based research of diabetes neuropathy in Korea. Diabetes Res Clin Pract. 2014;103:522–529. doi: 10.1016/j.diabres.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 20.Al-Mahroos F, Al-Roomi K. Diabetic neuropathy, foot ulceration, peripheral vascular disease and potential risk factors among patients with diabetes in Bahrain: A nationwide primary care diabetes clinic-based study. Ann Saudi Med. 2007;27:25–31. doi: 10.5144/0256-4947.2007.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pai YW, Lin CH, Lee IT, Chang MH. Variability of fasting plasma glucose and the risk of painful diabetic peripheral neuropathy in patients with type 2 diabetes. Diabetes Metab. 2018;44:129–134. doi: 10.1016/j.diabet.2018.01.015. [DOI] [PubMed] [Google Scholar]
- 22.Ziegler D, Strom A, Lobmann R, Reiners K, Rett K, Schnell O. High prevalence of diagnosed and undiagnosed polyneuropathy in subjects with and without diabetes participating in a nationwide educational initiative (PROTECT study) J Diabetes Complications. 2015;29:998–1002. doi: 10.1016/j.jdiacomp.2015.09.008. [DOI] [PubMed] [Google Scholar]
- 23.Katulanda P, Ranasinghe P, Jayawardena R, Constantine GR, Sheriff MH, Matthews DR. The prevalence, patterns and predictors of diabetic peripheral neuropathy in a developing country. Diabetol Metab Syndr. 2012;4(21) doi: 10.1186/1758-5996-4-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.McCarter RJ, Hempe JM, Chalew SA. Mean blood glucose and biological variation have greater influence on HbA1c levels than glucose instability: An analysis of data from the diabetes control and complications trial. Diabetes Care. 2006;29:352–355. doi: 10.2337/diacare.29.02.06.dc05-1594. [DOI] [PubMed] [Google Scholar]
- 25.Li G, Sun C, Wang Y, Liu Y, Gang X, Gao Y, Li F, Xiao X, Wang G. A clinical and neuropathological study of Chinese patients with diabetic peripheral neuropathy. PLoS One. 2014;9(e91772) doi: 10.1371/journal.pone.0091772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wiggin TD, Sullivan KA, Pop-Busui R, Amato A, Sima AA, Feldman EL. Elevated triglycerides correlate with progression of diabetes neuropathy. Diabetes. 2009;58:1634–1640. doi: 10.2337/db08-1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Padilla A, Descorbeth M, Almeyda AL, Payne K, De Leon M. Hyperglycemia magnifies Schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res. 2011;1370:64–79. doi: 10.1016/j.brainres.2010.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ikura K, Hanai K, Shinjyo T, Uchigata Y. HDL cholesterol as a predictor for the incidence of lower extremity amputation and wound-related death in patients with diabetic foot ulcers. Atherosclerosis. 2015;239:465–469. doi: 10.1016/j.atherosclerosis.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 29.Isak B, Oflazoglu B, Tanridag T, Yitmen I, Us O. Evaluation of peripheral and autonomic neuropathy among patients with newly diagnosed impaired glucose tolerance. Diabetes Metab Res Rev. 2008;24:563–569. doi: 10.1002/dmrr.859. [DOI] [PubMed] [Google Scholar]
- 30.Hämäläinen H, Rönnemaa T, Halonen JP, Toikka T. Factors predicting lower extremity amputations in patients with type 1 or type 2 diabetes mellitus: A population-based 7-year follow-up research. J Intern Med. 1999;246:97–103. doi: 10.1046/j.1365-2796.1999.00523.x. [DOI] [PubMed] [Google Scholar]
- 31.Wiggin TD, Sullivan KA, Pop-Busui R, Amato A, Sima AAF, Feldman EL. Elevated triglycerides correlate with progression of diabetic neuropathy. Diabetes. 2009;58:1634–1640. doi: 10.2337/db08-1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li L, Chen J, Wang J, Cai D. Prevalence and risk factors of diabetic peripheral neuropathy in type 2 diabetes mellitus patients with overweight/obese in Guangdong province, China. Prim Care Diabetes. 2015;9:191–195. doi: 10.1016/j.pcd.2014.07.006. [DOI] [PubMed] [Google Scholar]
- 33.Davies B, Edwards N, Ploeg J, Virani T. Insights about the process and impact of implementing nursing guidelines on delivery of care in hospitals and community settings. BMC Health Serv Res. 2008;8(29) doi: 10.1186/1472-6963-8-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.