Abstract
This review presents recent knowledge on the neuroprotective effects of vitamin D and their usefulness as oral supplementation when combined with other molecules, such as curcumin. A critical look at the effectiveness of vitamin D in this field is also provided. Vitamin D plays a crucial role in neuroprotection and in the cognitive decline associated with aging, where vitamin D’s levels are related to the levels of several neurotrophic factors. An important role of vitamin D has also been observed in the mechanism of neuroinflammation, which is the basis of several aging conditions, including cognitive decline and neurodegeration; furthermore, the neuroprotective effect of vitamin D in the cognitive decline of aging has recently been reported. For this reason, many food supplements created for humans contain vitamin D alone or combined with other molecules with antioxidant properties. However, recent studies also explored negative consequences of the use at a high dosage of vitamin D. Vitamin D in tissues or brain cells can also modulate calbindin-D28K, parvalbumin, and calretinin, and is involved in immune function, thanks also to the combination with curcumin. Curcumin acts as a free radical scavenger and antioxidant, inhibiting lipid peroxidation and oxidative DNA damage. In particular, curcumin is a potent immune-regulatory agent and its administration has been reported to attenuate cognitive impairments. These effects could be exploited in the future to control the mechanisms that lead to the brain decay typical of neurodegenerative diseases.
Keywords: vitamin D, brain aging, neurodegenerative diseases, neuroinflammation, VDR
In recent years it has been discovered that curcumin shows anticarcinogenic, hepatoprotective, thrombo-suppressive, cardioprotective, antiarthritic, and anti-infectious properties. Curcumin has also shown neuroprotective properties in several preclinical studies for neurodegenerative diseases. This suggests that curcumin could be a worthy candidate for nutraceutical intervention in relation to neurodegenerative diseases [1]. Neurodegenerative diseases affect millions of people around the world. In addition, cognitive disorders caused by brain aging are becoming increasingly common, in part because the human life span has increased in recent years [2]. For these reasons, there is an urgent need to develop new and more effective approaches to combat these harmful conditions, both as a treatment and as prevention. While pharmaceutical treatments for neurodegenerative diseases are successful only in some patients, for others, researchers are looking for alternative options due to the lack of efficacy or intolerable side effects. For this reason, this review aimed to expose the main neuroprotective effects of vitD, alone and combined with curcumin, in order to illustrate how the use of natural dietary supplements can be useful in preventing or counteracting neuroinflammation progression. However, it is necessary to take into account that this field of research is still at the beginning of its path and many novelties await us if continue to explore this interesting molecule.
1. Vitamin D General Overview
In the present critical review, alongside the description of the most recent knowledge on the role of the essential micronutrient vitamin D (vitD) in various mechanisms underlying neurodegenerative disorders and other adverse effects, the potential effects of curcumin on the health of the central nervous system (CNS) are discussed, where curcumin is a nutraceutical that is extensively used in herbal medicine. Vitamin D has gained great fame as a nutritionally essential factor since the elucidation of vitamin D’s chemical structure revealed that it is a steroid hormone that is able to exert its effects through a specific receptor, which was discovered in the 1960s [3]. Vitamin D can either be ingested or synthesized in the skin [4,5], and to make it biologically active, the prohormone vitamin D is transported through the bloodstream to the liver, where it is metabolized [6]. 25-hydroxyvitamin D (25(OH)D3) is the major circulating metabolite of vitamin D in plasma and is essential for providing an index of a patient’s vitamin D nutritional status [7,8]. A successive metabolization of 25(OH)D3 generates the hormonally active form of vitamin D, namely, 1,25-dihydroxyvitamin D3 (1,25(OH)2D3), which is responsible for most of the biological actions of vitamin D [6]. It was assumed that the brain’s 1,25(OH)2D3 supply depends on the plasma concentration of 1,25(OH)2D3 (vitD) [9,10,11]. VitD works through two types of receptors: (i) the nuclear vitamin D receptor (VDR) to induce genomic action [12] and (ii) the putative membrane receptor MARRS (membrane-associated, rapid response steroid-binding) to induce non-genomic actions [13].
Maintaining an adequate plasma level of vitamin D can be a problem and vitamin D deficiency is more common than previously thought [14]. Natural food sources of vitamin D are uncommon; therefore, most people rely on skin production following safe exposure to sunlight. Exposure to sunlight is certainly the safest form of vitamin D supply and it could reduce the dependency on supplements [15]; however, many variables influence the amount of UV rays that reach the skin and its effectiveness. These include the time of day, season, latitude, altitude, clothing, use of sunscreen, pigmentation, and age. For this reason, especially in the elderly, exogenous administration through food supplements is necessary [16]. The U.S. National Academy of Science established a recommended daily intake for vitamin D of 15 μg/day (600 units/day) for people under the age of 70 and 20 μg/day (800 units/day) for people over the age of 70 [17]. Due to its broad therapeutic index, vitamin D toxicity is extremely rare. However, close attention should be paid to a prolonged excess of vitamin D intake, which could lead to hypercalcemia, hypercalciuria, and hyperphosphatemia, which are considered to be the initial signs of vitamin D intoxication. In particular, the impairment of the calcium/phosphate balance could lead to cardiovascular damage, such as arrhythmia, cardiac arrest, calcification of the vessels, and hypertension [18]. Recently, the localization of vitamin D3 25-hydroxylase and 25-hydroxyvitamin D3-1α-hydroxylase enzymes in the brain was demonstrated, confirming a local bioactivation of the vitamin D3 prohormone and its presence in the cerebrospinal fluid [19,20,21]. Furthermore, the local catabolism performed by vitamin D3 24-hydroxylase was found [22]. The fact that the CNS can locally perform both its activation and inactivation makes vitD a neurosteroid by definition [23]. Among the many functions that have been attributed to vitamin D, some concern the nervous system. A number of pleiotropic functions were recognized, such as maintaining healthy neuronal development, an adequate trophism of the adult brain, and a slow aging process [24]. VDRs are widely distributed throughout the embryonic and adult brain and appear most prominently in the neuroepithelium and proliferating zones in both rats [25,26,27,28,29] and humans [30]. Their presence has also been noted in neurons and glia of the human prefrontal and the cingulate cortices, thalamus, hypothalamus, cerebellum, substantia nigra, caudate, putamen, amygdala, and hippocampus [30,31]. Since the initial reports of Stumpf et al., on the presence of vitamin-D-specific nuclear binding in the brain and spinal cord [32,33], evidence has accumulated to suggest that both mRNA encoding the VDR and the protein itself are present in the nervous system. Thus, VDR gene expression has been demonstrated in neuronal and glial cells [34,35,36,37,38,39,40]. Expression of the VDR occurs early in the developing rodent brain at embryonic day (E) 11.5 and E12 in the rat dorsal root ganglion, spinal cord, and midbrain. Increasing levels of VDR expression throughout gestation coincides with increasing levels of apoptosis and decreasing levels of mitosis, and appears to be localized to the neuroepithelium and differentiating fields [41,42]. The sites of expression of VDR change during development, leading to the hypothesis that vitD may play a role in brain development [43]. For these reasons, the many functions that vitD exerts on the central nervous system have made it possible to hypothesize its use to counteract the mechanisms that lead to brain aging and neurodegenerative diseases.
2. Calcium Homeostasis in the Brain
Many calcium-binding proteins are present throughout the body, including the CNS [44,45]. It was reported that vitD in brain tissues or cells can modulate calbindin-D28K, parvalbumin, and calretinin [46,47]. These calcium-binding proteins are widely distributed in both the adult and fetal brain. In the adult brain, each of these proteins has a unique distribution and exhibits temporal patterns during development [48,49,50,51] and aging [52]. Calbindin-D28K is required for the normal signaling of synaptically evoked calcium transients [53] in synaptic plasticity [54], long-term potentiation (LTP) [55], memory formation [56,57], and possibly in the regulation of exocytosis (synaptic secretion of neurotransmitters) [58]. Furthermore, in Purkinje cells in the cerebellum, calbindin-D28kK appears to be directly involved in motor control [53,59], which could suggest a possible mechanism explaining motor deficits observed in vitamin-D-deficient rats. Calmodulin, is another important calcium-binding protein in the brain [60], although two studies reported that calcitriol treatment shifted the intracellular distribution of calmodulin [61]. In the brain, calmodulin is involved in neurotransmitter activity [62], N-methyl-D-aspartate (NMDA)-induced synaptic plasticity [63], and short-term plasticity [64]. Calcium/calmodulin-dependent protein kinase II (CAM kinase II) is highly concentrated in neurons and is believed to play a central role in a variety of brain functions, including learning and memory; it has been suggested that CAM kinase II is the molecular basis of long-term synaptic memory [65,66,67,68]. A recent report shows the down-regulation of the L-type voltage-sensitive calcium (Ca2+) channel in hippocampal neurons in the presence of vitD, which has been correlated with a neuroprotective effect against excitotoxic insults [69]. Another way in which vitD might mediate its neuroprotective effect is to induce the synthesis of Ca2+-binding proteins, such as parvalbumin [47].
3. Neurogenesis Is Stimulated by Vitamin D Metabolites and Some of Its Target Gene Products
VitD acts as a prodifferentiation hormone in many tissues. With reference to the brain, it is of interest that the increase in apoptotic cells and decrease in mitosis in the developing rat brain correlates with the appearance of VDRs [41]. Chronic treatment with calcitriol or its analogs increased the neurite outgrowth (and some other markers of differentiation) in human neuroblastoma cells in some significant experiments [70,71]. In rodents, vitD treatments stimulated neurite outgrowth in embryonic hippocampal explant cultures [72] and in a hippocampal progenitor cell line [73]. Adult neurogenesis is a very active area of research [74,75]. Adult stem cells are believed to be located in only two regions of the adult brain. One group of neural stem cells originates in the subventricular region of the forebrain and migrates to the olfactory bulb; the second group originates in the subgranular zone of the hippocampus and differentiates into neural and glial cells in the dentate gyrus [76,77]. In both cases, new neurons are local-circuit interneurons that link motor and sensory neurons [78]. There is a possible involvement of vitD in adult neurogenesis, with specific reference to the brain, where VDRs are widespread in both the olfactory bulb and the dentate gyrus in rodents [79] and were recently observed in the subventricular zone of the neonatal brain [80]. Furthermore, an increased density of hippocampal neurons was observed in some rat strains supplemented for 6–12 months with vitD (20 ng/rat, administered subcutaneously 5 times/week) [81].
3.1. Vitamin D and Neurotrophic Factors
The combination of in vitro, ex vivo, and animal model data provides evidence that vitD has a crucial role in neuronal proliferation, differentiation, neurotransmission, neuroplasticity, and neuroprotection. In addition, vitD levels correlate with the levels of several neurotrophic factors, including nerve growth factors (NGFs) and those of neurotrophins, which play crucial roles in the maintenance and growth of neurons [82,83]. In several cases, the neurotrophin production caused by vitD correlated with the neuroprotective effect, including the survival and migration of developing neurons in the brain [84,85]. Various studies have demonstrated that vitD can act on cells of the nervous system by up-regulating the synthesis of the NGF neurotrophin 3 (NT3), whereas it down-regulates neurotrophin 4 (NT4) [36,38,86] and glial cell line-derived neurotrophic factor (GDNF) [87]. NGF is present mainly in the hippocampus and neocortex, where it affects the growth and survival of developing neurons, neurotransmission, and synaptic plasticity [88]. Additional functions of vitD that are linked to neurotrophins include the enhancement of the synaptic transmission in the hippocampus by NT-3 [89] and its involvement in calcium signaling via NT4/5 [90]. Several studies on hippocampal explants and cultures treated with vitD have displayed increased NGF expression that is concomitant with a neurite outgrowth [36,86,91]. GDNF affects the survival and differentiation of dopaminergic cells and is present at relatively high levels in the developing striatum of the rat [92]. VitD administration leads to an increase in GDNF synthesis in both C6 glioma cells and the brain. In addition, in a stroke model, Wang and colleagues showed that VDH pre-treatment for 8 days can significantly increase the levels of GDNF and attenuate cortical infarction that is induced by a middle cerebral artery (MCA) ligation in rats [84,86].
3.2. Astrocytes as a Target of Neuroprotection
The mechanism behind astrocyte degeneration in age-related neurodegenerative disorders is unknown, although many different genetic abnormalities have been identified that can cause neurodegenerative disorders [93]. Furthermore, many studies in the literature provide indications as to the molecular and cellular factors that determine whether a particular neuron succumbs or resists an age-related disease [94]. The most vulnerable neurons are normally large with myelinated axons extending over long distances, from one region of the nervous system to another or from the central nervous system to peripheral targets [95,96,97,98,99]. This axon projection mechanism could be particularly linked to their vulnerability to aging, including high energy requirements, dependence on axonal transport (antegrade and retrograde) that support function and trophism, and a large cell surface that increases the risk of exposure of cells to toxic environmental conditions [100]. Normally, degeneration is often extended to subpopulations of neurons with a particular neurotransmitter phenotype. For example, amyotrophic lateral sclerosis (ALS) affects cholinergic motor neurons and striatal neurons containing GABA (γ-aminobutyric acid) and dopaminergic neurons, which are the most vulnerable in Huntington’s disease (HD) and Parkinson’s disease (PD), respectively [101]. Among the different neurotransmitters, glutamate could have an active and essential role in neuronal damage and death in all neurodegenerative disorders (see Section 3.3 on excitotoxicity below). Dysfunction and death of neurons negatively affect both the pre- and post-synaptic neurons with which they communicate [102].
3.3. Vitamin D3 and Neurodegenerative Diseases
The neuroprotective effect of vitD has recently been observed in the cognitive decline of aging rats [103], and it has been extensively studied in an animal model of multiple sclerosis (MS) and experimental allergic encephalomyelitis. The hormone prevents the onset and reversibly blocks the progression of clinical signs, but such a protective effect is absent in VDR knockout mice [104]. Some evidence implicates vitD as a candidate in influencing the susceptibility to a number of psychiatric and neurological diseases, such as schizophrenia, autism, PD, amyotrophic lateral sclerosis, epilepsy, and Alzheimer’s disease (AD), where the evidence is especially strong for MS [105,106]. Upon studying the effect of a vitD dietary restriction in the spinal cord and brain of ALS patients, there was a negative effect of vitD deficiency on the antioxidant capacity [107]. The effect of vitD might not be due exclusively to its neuro-immunomodulatory properties [105] since it has recently been reported that the hormone enhances neural stem cell proliferation and differentiation into neurons and oligodendrocytes, which are the myelinating cells of the central nervous system [108,109]. Neural stem cells constitutively express VDRs, which can be up-regulated by vitD. VitD regulates the expression of many AD-related genes. It attenuates amyloid-beta (Aβ)-peptide accumulation by stimulating phagocytosis of Aβ-peptide, probably by modulating the transcription of Toll-like receptors and cytokines, together with enhancing brain-to-blood efflux transport by increasing the P-glycoprotein expression [110]. Alterations in adult neurogenesis appear to be a common hallmark in different neurodegenerative diseases, including PD and AD [111]. Therefore, a hunt for possible molecules that are capable of stimulating neurogenesis is underway in order to develop new treatments for neurodegenerative disorders. Moreover, the combination of anti-neurodegenerative drugs with vitD supplementation might be useful. Indeed, the supplementation of the combination of nemantidine plus vitD has been shown to prevent cognitive decline more efficiently than that of the single compounds [112]. In addition, vitamin D exerts direct neuroprotective effects via the synthesis of Ca2+ ion binding proteins; this is important in neuronal function and in neuronal transmission. Proper levels of neuronal calcium are critical because their excess may result in the formation of reactive oxygen species (ROS) with consequent neuronal damage. Indeed, vitamin D levels are inversely associated with oxidative stress, which leads to neuronal apoptosis or necrosis [113]. VitD also affects neuroplasticity, a process in which neural synapses and pathways are adapted to the needs of environmental and behavioral demands by adjusting the brain to noxious stimuli diseases or environmental cues [114,115,116]. The VDRs in glial cells are involved in the uptake and release of neurotransmitters, including that of GABA neurotransmission within the motor cortex [117], which is the principal “brake” within the brain that affects muscle relaxation via the corticospinal neurons [118].
3.4. Vitamin D and Neuroinflammation
Concurrent administration of vitD with lipopolysaccharide (LPS) significantly inhibits inducible nitric oxide synthase (iNOS) expression in monocytes in the rat brain, suggesting that vitD can also help attenuate immune-induced oxidative damage in the CNS [119]. Lin et al. found a similar effect on zinc-induced toxicity in the CNS, where concurrent administration of vitD reduced apoptosis and oxidative damage [120]. An animal model of chronic relapsing experimental autoimmune encephalomyelitis (EAE), which is a model of MS, was used to study the effects of vitD [121]. VitD has been reported to be able to block the development of disease after its onset in both rats and mice [83,122] by providing an improvement in their condition, which was correlated with the inhibition of iNOS [123,124], CD4 antigen expression [122], and interleukin 12 (IL-12)-dependent TH1 cell development in the CNS [125]. VitD also increases the levels of transforming growth factor β (TGFβ) and IL-4, which were increased in a mouse model and are anti-inflammatory TH2 immune response cytokines [126]. In another EAE system, vitD significantly reduced acute inflammation and the levels of GFAP by inducing inflammatory cell apoptosis [127]. VitD has been observed to reduce the production of inflammatory cytokines tumor necrosis factor alpha (TNFα), IL-6, and nitric oxide (NO) in stimulated microglia [128]. VitD also appears to regulate the expression of N-myc, c-myc, PKC, and TGFβ in neuroblastoma cells [129], suggesting that it may affect neural cell growth in ways other than the well-established induction of NGF and its receptors [72,85,90,130].
3.5. Vitamin D3 and the Oxidative Defense System
An organism possesses defense mechanisms based on its antioxidants’ actions. Both enzymatic (e.g., superoxide dismutase (SOD), catalase, and glutathione reductase (GR)) or non-enzymatic (e.g., glutathione (GSH), melatonin, vitamins (A, C, E), and flavonoids) molecules play an important role in maintaining homeostasis and cell viability. Oxidative stress plays a key role in mitochondrial dysfunction, which will eventually lead to aging and the loss of function [131]. The mitochondrial free radical theory is one of the most studied hypotheses to explain this mechanism; it is based on the endogenous production of ROS and reactive nitrogen species (RNS) and their harmful effects on the mitochondria [132,133]. These species react with lipids, proteins, and nucleic acids, causing oxidative damage that leads to a progressive decline in cellular functions [107,134]. A study on the mechanisms of vitD protection against methamphetamine-induced reductions in brain levels of dopamine and serotonin showed that they include an increase in the levels of glutathione, along with the inhibition of iNOS production, both of which could reduce the toxicity caused to dopamine and serotonin neural terminals [135]. VitD can regulate lipid peroxidation as a form of neuroprotection through inducing the synthesis of parvalbumin, a Ca2+ binding protein, which helps with maintaining the cellular calcium homeostasis and eventually lowers lipid peroxidation [136], as well as through promoting the expression of calcium buffering proteins calbindin-D28K and calbindin-d9K, which reduce the malonaldehyde (MDA) levels [137,138]. During a CNS injury, there is a high release rate of nitric oxide (NO) that reacts with superoxide and eventually produces nitrogenous species, especially peroxynitrite (ONOO−), which is a marker in the spinal cord of ALS patients that inactivates SOD2 through dityrosine formation [139]. The reduction of SOD2 activity is through the post-translational impact of vitD on nuclear factor κB (NF-κB), as activated microglia in ALS use the NF-κB pathway to induce mitochondrial dysfunction inhibition of SOD2 and motor neuron death [140,141]. In addition, a cross-sectional study on type 2 diabetes mellitus patients was performed to investigate the correlation between vitD status and the antioxidant profile in diabetic patients compared to healthy groups, where they were followed up for fasting serum levels of 25-OH-D, calcium, phosphorus, parathyroid hormone, glucose, HbA1C, insulin, total antioxidant capacity (TAC), SOD, GR, and glutathione peroxidase (GSH-Px). These findings have indicated an inverse relationship between the serum levels of 25-OH-D and the activities of both GSH-PX and GR, along with a positive correlation with SOD in diabetic patients. Meanwhile, in healthy subjects, there is an inverse relation between 25-OH-D levels and SOD and GSH-PX levels, and a positive correlation with GR activities. The positive association between the serum level of 25-OH-D and GR is due to the role of GSH in maintaining the intracellular redox balance. VitD acts by increasing the activity of GR and decreasing the GSH-PX activity in healthy subjects to enhance the GSH pool [142]. VitD has also been reported to inhibit the synthesis of iNOS [119,123], which is an enzyme induced in CNS neurons and non-neuronal cells during various insults or diseases, such as ischemia, Alzheimer’s disease, Parkinson’s disease, AIDS, infections, multiple sclerosis, and EAE. iNOS produces nitric oxide, one of the biological effects of which is to damage both neurons and oligodendrocytes when it is produced at high levels [143,144]. VitD has also been reported to up-regulate γ-glutamyl transpeptidase activity and the expression of the corresponding gene in rat brains and reduce the nitrite production in LPS-stimulated primary rat astrocyte culture because γ-glutamyl transpeptidase is largely involved in the glutathione cycle of the brain in the crosstalk between astrocytes and neurons [145,146,147]. In conclusion, regarding the oxidative mechanism involving vitD, treatments with vitamin D3 and other compounds that are capable of improving its effect could be a promising remedy for the treatment of neurodegenerative diseases during aging.
3.6. Curcumin General Overview
Curcumin is the principal curcuminoid of the popular Indian spice turmeric, and its main active ingredient is obtained from the rhizome of Curcuma longa Linn. Curcumin acts as a free radical scavenger and antioxidant, inhibiting lipid peroxidation [148] and oxidative DNA damage. Moreover, it is a particularly potent immuno-regulatory agent that can modulate the activation and function of T-cells, B-cells, neutrophils, natural killer cells, and macrophages [149]. Curcumin treatment effectively inhibits the activation of microglial cells by diminishing the production of nitric oxide [150] and reducing the secretion of pro-inflammatory cytokines, such as IL1β, IL6, and TNF [151]. Recent experiments have also demonstrated that curcumin protects dopaminergic neurons against microglia-mediated neurotoxicity [152], limits brain inflammation [153], and rescues retinal cells from stress-induced cell death [154]. The suppressive effect of curcumin is thought to involve regulating the JAK–STAT inflammatory signaling in activated microglia [155]. A recent report has revealed that curcumin reduces the amyloid-β-stimulated inflammatory responses in primary astrocytes. The deleterious effects of amyloid-β, such as the increased expression of COX-2 and glial fibrillary acidic protein and the decreased peroxisome proliferator-activated receptor gamma (PPARγ), were attenuated using pretreatment with curcumin [156]. Accumulating cell culture and animal model data show that curcumin is a strong candidate for use in the prevention or treatment of major disabling age-related neurodegenerative diseases, such as AD, PD, and strokes [157]. Furthermore, it was shown that curcumin can down-regulate the expression of IL-6, TNF, and various other chemokines [149]. Moreover, it has subsequently been shown that curcumin down-regulates the expression of the NF-κB-regulated gene products, such as COX-2, TNF, 5-LOX, IL-1, IL-6, IL-8, MIP-1α, adhesion molecules, c-reactive protein (CRP), and CXCR-4 [158,159,160,161].
3.7. Neuroprotective Effects of Curcumin
Curcumin has also been reported to bind to COX-2 and 5-LOX and inhibit their activity [162]. In a study on the protective effect of curcumin on dopaminergic neurons from apoptosis in an MPTP mouse model of PD, Curcumin markedly ameliorated the loss of dopaminergic axons in the striatum as well as the death of dopaminergic neurons, further mechanistic studies demonstrated that curcumin inhibits MPTP-induced hyperphosphorylation of c-Jun N-terminal kinase (JNK); it also prevents the degeneration of nigrostriatal neurons by inhibiting the dysfunction of mitochondria through abolishing the hyperphosphorylation of JNKs induced by MPTP [163]. Moreover, curcumin’s inhibitory effect on NO and PGE2 production was associated with decreased iNOS and COX-2 expressions in LPS-stimulated BV2 microglia, where the results of Western blot analysis revealed that iNOS and COX-2 protein levels were undetectable in unstimulated BV2 microglia [151]. A study was done to evaluate the neuroprotective effects of curcuminoids on B35 and SH-SY5Y neuroblastoma cells after subjecting the cells to hydrogen peroxide, followed by curcumin at different concentrations of 5, 10, and 20 μM before and after being damaged. B35 neuroblastoma cells showed an increase in cell viability when treating cells with curcumin before and after the induced damage. Meanwhile, the SH-SY5Y neuroblastoma cells showed an increase in their viability only after the curcumin treatment. Caspase-3 and caspase-9, which are important apoptosis mediators, were examined, where the curcumin inhibited the caspase-3 in a concentration-dependent manner but not caspase-9. These findings suggest that curcumin is a neuroprotectant and an anti-apoptotic agent that acts through the inhibition of caspase-3; hence, it can be introduced as a potential agent to treat or prevent neurodegenerative diseases [164]. The results of a study performed by Yang and his colleagues show that curcumin at a 10 µM concentration had a neuroprotective effect on midbrain dopaminergic neurons; increased the dopamine uptake; decreased the expression of proinflammatory cytokines, such as nitric oxide (NO), prostaglandin E2 (PGE2), IL-1β, and TNF-α; inhibited the transcription of NF-κB and activator protein-1 [153]. However, another group of researchers has identified a possible molecular target for curcumin that may be involved in SH-SY5Y dopaminergic neuron protection during MPP+-induced cytotoxicity (3 mM MPP+). This mechanism involves JNK pathway activation and caspase-3 cleavage, preventing neuronal death [165]. A study of another mechanism for curcumin in apoptosis investigated the effects of curcumin in appoptosin-induced apoptosis in SH-SY5Y cells, where appoptosin (SLC25A38) is a pro-apoptotic protein that is up-regulated in Alzheimer’s disease (AD) brains and promotes the pathological progress. Through pretreating SH-SY5Y cells with curcumin, then transfecting with appoptosin, followed with detecting apoptotic cells with Annexin V staining analysis using flow cytometry, the expression of cleaved caspase-3, appoptosin, and heme oxygenase-1 (HO-1) were examined and the intracellular ROS level was measured to detect the mitochondrial membrane potential. The results showed an overexpression of appoptosin, which was accompanied by reduced HO-1 expression, ROS overproduction, and mitochondrial potential impairment. However, pretreating with curcumin (2.5–20 μmol/L) in a dose-dependent manner attenuated all these pathological changes in appoptosin-transfected SH-SY5Y cells [166].
3.8. Effectiveness of Vitamin D Dietary Supplements in a Clinical Study
Starting from 2011, many systematic reviews and meta-analyses have reported useful data for the development of evidence-based clinical practice guidelines [167] on vitamin D supplementation. Several systematic epidemiologic studies that were based on the association between vitamin D and cognitive decline, dementia, and neurodegenerative conditions have been published [168]. However, the data show discordant results since some studies claim a positive role of vitamin D in the brain, while others even indicate adverse effects. For example, a meta-analysis reported by Suzuki et al. [169] showed that older adults with PD taking supplemental vitamin D in randomized controlled trials showed beneficial muscle effects in terms of strength and balance [169]. Nonetheless, it cannot be distinguished whether vitamin D supplementation specifically delays the progression of PD or whether it just nonspecifically improves muscle strength and balance in older adults. In another study, long-stay patients with baseline 25(OH)D < 40 nmol/L were treated with 9000 IU vitamin D2 for 8–40 weeks or a placebo (lactose tablets). No significant difference in the single cognitive measure (mental assessment score) was found between groups [170]. Barnard and Colon-Emeric suggested that cognitive function, as measured using the Mini-Mental State Examination (MMSE), was not associated with the 25(OH)D concentration, although their conclusions were based on whether the relationship between vitamin D and cognitive test scores in the original studies was statistically significant [171]. Another trial with 4143 participants compared vitamin D3 (400 IU/day) and calcium supplements to a placebo. Rutjes et al. [172] found low- to moderate-certainty evidence of no effect of vitamin D3 and calcium supplements at any time point during 10 years on the overall cognitive function (MD after a mean of 7.8 years = − 0.1 MMSE points, 95% CI − 0.81 to 0.61) or the incidence of dementia (HR = 0.94, 95% CI = 0.72 to 1.24). Based on this result, a pilot study with 60 participants used a higher dose of vitamin D3 (4000 IU on alternate days) and found preliminary evidence that this dose probably had no effect on cognitive function over six months [172]. Therefore, they found no evidence that a vitamin D supplementation strategy for cognitively healthy adults had a meaningful effect on cognitive decline or dementia, although the evidence does not permit definitive conclusions. Interventional trials investigated whether vitamin D supplementation can beneficially influence the brain disease course, but the trials have been methodologically fairly heterogeneous, and study designs have not always been appropriate [173]. Some studies were not longitudinal and could have confounding factors; therefore, the final result could be unclear. Due to this scenario, we investigated even further because vitamin D research has gained increased attention in recent times due to its roles beyond bone health and calcium homeostasis.
A randomized controlled double-blind placebo trial investigated the effect of vitamin D on cognition in healthy elderly subjects above 65 years of age. The vitamin-D-supplemented group (supplement contained 4.0 μg (160 IU) of vitamin D and other trace elements) had a better cognitive performance (p < 0.01) in the different cognitive tests (seven cognitive tests, including the MMSE) compared to the placebo group [174]. Another prospective pre–post interventional study, which included 80-year-old subjects from a memory clinic, found that those who received oral vitamin D3 supplementation (800 IU per day or 100,000 IU per month) experienced improved global cognition and executive functioning abilities over a 16-month follow-up period compared to controls [175]. Bailon et al. [176], in its systematic review of meta-analysis cognition data (using the MMSE) provided suggestive evidence of a possible relationship between vitD and cognitive decline, but the nature of this relationship remained unclear. This discrepancy may be a function of the type of cognitive measure used. Little is known about the function of vitamin D in relation to the different cognitive domains. To date, no treatment study has examined this question where both vitamin D and cognition were measured over a sufficient period in a large at-risk population. In particular, Balion et al. [176] included data from eight cross-sectional and case-control studies with a total of 2749 participants, comparing MMSE scores between participants who were given a 25(OH)D concentration < 50 nmol/L and those who were given a 25(OH)D ≥ 50 nmol/L. This review demonstrated a higher average MMSE score with a higher vitamin D concentration (average difference 1.2; 95% CI 0.5–1.9) with a significant heterogeneity (I2 = 0.65) [168]. Therefore, it is clear that the difference between the studies not only concerns the method applied but also the dosages. It appears from these studies that at low doses, the beneficial effects are observable, while at high doses, these effects become nil. In summary, vitamin D supplementation should be encouraged in the elderly and in AD patients to combat possible cognitive decline; meanwhile, a huge body of evidence emphasizes the importance of vitamin D as a supplement for slowing brain aging. In addition, based on statistical and clinical results, vitamin D treatment may be a safe option for brain supplements due to its potential synergistic benefits if used at a correct dosage and in combination with other molecules. For example, in a pilot study, Annweiler et al. [177] explored the combination of vitD and memantine to improve cognitive parameters in AD patients. Memantine, which is a Japanese drug approved by the FDA (Food and Drug Administration) to treat moderate-to-severe AD, is able to exert its therapeutic effect by acting as a low to moderate affinity, non-competitive NMDA receptor antagonist that blocks the Ca2+ ion flux [178]. Treatment with memantine plus vitamin D was associated with improvement in the MMSE score compared to memantine or vitamin D alone. Patients with AD who took memantine plus vitamin D for 6 months had a statistically and clinically relevant gain in cognition, supporting the hypothesis of the possible synergistic and potential benefits of the combination. This study confirmed a possible beneficial role of vitD when used correctly and its possible beneficial interaction with other molecules.
3.9. Interaction between Vitamin D3 and Curcuminoids
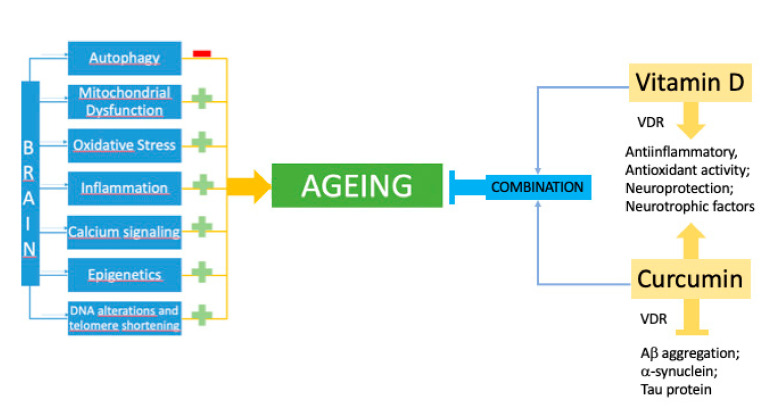
The results of many studies may be relevant to the use of curcuminoids and vitamin D3 in the prevention of neurodegenerative diseases, which are substances that work via antioxidant and anti-inflammatory mechanisms ([179] Figure 1). Indeed, morbidities of aging have been related to defective functions of both T cells and macrophages leading to brain amyloidosis and inflammation [180]. “Inflammaging” may be associated with an increase in incompetent memory T cells and inflammatory cytokines produced by macrophages, whereas the defective clearance of amyloid-beta Aβ1–42 may be related to the defective transcription of immune genes necessary for phagocytosis, β-1,4-mannosyl-glycoprotein, 4-β-N-acetylglucosaminyltransferase, and Toll-like receptors. The approaches used for re-balancing Aβ immunity and inflammation are being pursued in animal models and the peripheral blood mononuclear cells of patients [179,181,182,183]. Increased inflammation may represent the “first hit” and defective transcription of immune genes represent the “second hit” in the pathogenesis of AD.
Figure 1.
Schematic representation of the combined effects of vitD and curcumin to slow down brain aging. VDR: vitamin D receptor.
Gagliardo et al. conducted studies that showed that treatment with curcumin may decrease the predisposition of Aβ to accumulate in cells. Although the role of Aβ1-42 as a potential causative agent in the pathophysiology of neurodegenerative disease has been documented extensively, the exact molecular mechanisms and their biochemical effect on brain tissues are not very clear [179,183]. An in vitro experiment by Alamro et al. on primary neuronal cultures investigated the effect of Aβ1-42 on the primary neuronal cells and the role of vitamin D3 or curcumin used as a natural therapy for AD treatment. It was confirmed that the treatment of primary neuronal cells with Aβ1-42 could cause elevated levels of oxidative stress, which is depicted by an increased amount of lipid peroxidation products and decreased levels of antioxidant enzymes [184]. According to this, curcumin treatment produced a decrease in intracellular Aβ aggregates, an increase of phagocytosis (e.g., the MGAT3 pathway), and a reduction of inflammation. Results of the treatment of PBMCs (peripheral blood mononuclear cells) from AD patients with curcuminoids demonstrated the curcuminoids’ ability to act on cell susceptibility to Aβ accumulation. Data showed that NF-κB and BACE1 were decreased after treatment with curcuminoids. In addition, the decrease in NF-κB led to the suppression of the inflammatory cascade, which is one of the major pathways in aging pathology [185]. In this context, it is necessary to emphasize the role of vitamin D3 as a transcription regulator of genes of the innate immune system [186]. It has been hypothesized that vitamin D3 possesses immunostimulant effects and also induces the phagocytosis and degradation of Aβ via monocyte/macrophage maturation [182,187]. We suggest that increased MGAT3 and VDRs inhibit Aβ accumulation and decrease inflammation. Moreover, in most systems, vitamin D inhibits the activation of NF-κB [188]. VDR stimulation induced by curcuminoids could shift this balance to and increase the inhibitory activity of VDRs on NF-κB [189]. Another study on primary cortical neuronal cells demonstrated that Aβ1-42 caused a significant reduction in mitochondrial health or mitophagy, but treatment with vitamin D3 and curcuminoids could overturn this outcome [183]. The oxidative state of the primary cortical neuronal cells is one of the earliest hallmarks of neurodegenerative diseases [190]. Normally, cells have an antioxidant mechanism, such as GSH and antioxidant enzymes, catalase, and SOD, that can quench free radicals [191,192], but this mechanism is dysregulated during aging. As said before, lipid peroxidation is the main contributor to free-radical-mediated damage to the neuronal membrane; it can also produce some secondary oxidation products that are capable of causing additional cellular damage. These highly reactive electrophilic aldehydes are MDA and 4-hydroxy-2-nonenal (HNE) [193]. Alamro et al. [183] showed that a significant amount of MDA in the Aβ1–42 treated cultures produced high levels of these aldehydes, but the presence of vitamin D3 and curcumin in the culture categorically reduced its formation [190]. The synergistic effect of vitamin D3 and curcumin significantly increased the catalase enzyme expression. These results further reiterated the beneficial therapeutic effect of both vitamin D3 and curcumin in the treatment of AD. The combined effect of vitamin D3 and curcumin has also been reported to accelerate the clearance of β-amyloid by activating the immune cells [183]. These results have been confirmed by SOD enzyme activity analysis, where the activity was significantly reduced with the treatment of Aβ1–42 to the cells, but the synergistic effect of both vitamin D3 and curcumin has a positive effect on up-regulating SOD enzyme activity. Finally, the neuroprotective action of vitamin D3 on neuronal tissue is due to the up-regulation of several genes involved in alleviating oxidative stress, such as reduced GSH and NGF [114]. In recent years, the targeted delivery of NGF to the affected areas of Basal Forebrain-Like Cholinergic Neurons (BFCN) as a potential therapy for the AD patients has gained support due to the regenerative effect of NGF on BFCN [194]. During aging, there is a significant loss of NGF; however, this effect was neutralized in the presence of vitamin D3 or curcumin. Earlier studies have indicated the role of vitamin D3 in the regulation and secretion of NGF and GDNF [195]. Curcumin has also been shown to initiate proliferation and improve the health of neurite growth and synaptic activity [196].
3.10. A New Possible Treatment Approach to Support Brain Health
The data shown so far demonstrate that there are several functions played by vitamin D and curcumin, especially in neuroprotection and maintaining brain health. Furthermore, these substances could play an important role in the early, mild stages of neurodegenerative diseases and in cognitive impairment. Therefore, the question arose about the use of such compounds regarding combined treatment, whether there is a synergistic effect, and whether they are safe to use in such a combination. In a study published in 2015, in which a scopolamine-hydrobromide-induced Alzheimer’s disease rat model was used to reduce the effective action at the synapse by antagonizing muscarinic acetylcholine receptor without changing the concentration of acetylcholine to produce a stage of memory impairment, animals were subjected to a drug treatment schedule for 27 days, where there was a scopolamine group, a scopolamine–curcumin group, a scopolamine–vitamin D group, and a scopolamine–donepezil group. All groups underwent behavioral tests, namely, the rectangular maze and locomotor activity tests, followed by histological analysis for cellular degeneration to determine whether there was amelioration in this stage after treatment, and immunoblotting procedures were used to detect the expression of modified microtubule-associated tau protein. Results show that scopolamine treatment has led to a decrease in transfer latency as a result of significant memory loss, which was obvious in behavioral tests, where a large number of degenerated cells was observed in histological imaging along with a significant presence of abnormal tau protein. In contrast, treatment groups had an ameliorated transfer latency regarding locomotion, indicating improved memory, as seen from the behavioral test results, where the histological examination showed equal cell numbers and similar cell morphology compared to the control groups, which proved the presence of memory regions. Furthermore, there was a strong reduction in tau phosphorylation normalized to β-actin. From these results, they concluded the potential of curcumin and vitD to reverse some cognitive and memory impairment within the same AD-induced model [197].
4. Conclusions
Based on all the studies previously described on vitD and curcumin, which have described the function of these molecules and the possible mechanisms activated in different neurodegeneration models, it could be interesting to consider the administration of vitD and curcumin as a supplement in a continuous way. Although it is good to remember that correct and safe exposure to sunlight could reduce the dependence on exogenous vitamin D supplementation, there are conditions in which the use of supplements is necessary. In the future, it would be useful to explore the existence of a synergistic effect for these compounds when administered in a combined preparation. Further studies will be needed in order to test their neuroprotective and therapeutic abilities to maintain a healthy brain during aging or to prevent neurodegeneration.
Acknowledgments
This review was written as part of the AGING Project, Department of Excellence, Ministero dell’Istruzione, Università e Ricerca (MIUR), Department of Translational Medicine, University of Eastern Piedmont. The authors thank Mariangela Fortunato for her help in revising the English language.
Author Contributions
Investigation, M.F.; data curation, S.R. and V.M.; writing—original draft preparation, M.F., S.R., V.M., and F.U.; supervision, F.U. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Monroy A., Lithgow G.J., Alavez S. Curcumin and neurodegenerative diseases. Biofactors. 2013;39:122–132. doi: 10.1002/biof.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Heemels M.T. Neurodegenerative diseases. Nature. 2016;539:179. doi: 10.1038/539179a. [DOI] [PubMed] [Google Scholar]
- 3.Fernandes de Abreu D.A., Eyles D., Féron F. Vitamin D, a neuro-immunomodulator: Implications for neurodegenerative and autoimmune diseases. Psychoneuroendocrinology. 2009;34(Suppl. S1):265–277. doi: 10.1016/j.psyneuen.2009.05.023. [DOI] [PubMed] [Google Scholar]
- 4.Gartner L.M., Greer F.R. Prevention of rickets and vitamin D deficiency: New guidelines for vitamin D intake. Pediatrics. 2003;111:908–910. doi: 10.1542/peds.111.4.908. [DOI] [PubMed] [Google Scholar]
- 5.Holick M.F., Smith E., Pincus S. Skin as the site of vitamin D synthesis and target tissue for 1,25-dihydroxyvitamin D3. Arch. Dermatol. 1987;123:1677–1683. doi: 10.1001/archderm.1987.01660360108022. [DOI] [PubMed] [Google Scholar]
- 6.How K.L., Hazewinkel H.A., Mol J.A. Dietary vitamin D dependence of cat and dog due to inadequate cutaneous synthesis of vitamin D. Gen. Comp. Endocrinol. 1994;96:12–18. doi: 10.1006/gcen.1994.1154. [DOI] [PubMed] [Google Scholar]
- 7.Kanis J.A. Vitamin D metabolism and its clinical application. J. Bone Joint Surg. Br. 1982;64:542–560. doi: 10.1302/0301-620X.64B5.6754741. [DOI] [PubMed] [Google Scholar]
- 8.Bikle D.D. Vitamin D metabolism, mechanism of action, and clinical applications. Chem. Biol. 2014;21:319–329. doi: 10.1016/j.chembiol.2013.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gascon Barré M., Huet P.M. Apparent (3H) 1,25-dihydroxyvitamin D3 uptake by canine and rodent brain. Am. J. Physiol. 1983;244:E266–E271. doi: 10.1152/ajpendo.1983.244.3.E266. [DOI] [PubMed] [Google Scholar]
- 10.Balabanova S., Richter H.P., Antoniadis G., Homoki J., Kremmer N., Hanle J., Teller W.M. 25-Hydroxyvitamin D, 24,25-dihydroxyvitamin D and 1,25-dihydroxyvitamin D in human cerebrospinal fluid. Klin. Wochenschr. 1984;62:1086–1090. doi: 10.1007/BF01711378. [DOI] [PubMed] [Google Scholar]
- 11.Pardridge W.M., Sakiyama R., Coty W.A. Restricted transport of vitamin D and A derivatives through the rat blood–brain barrier. J. Neurochem. 1985;44:1138–1141. doi: 10.1111/j.1471-4159.1985.tb08735.x. [DOI] [PubMed] [Google Scholar]
- 12.Wang T.T., Tavera-Mendoza L.E., Laperriere D., Libby E., MacLeod N.B., Nagai Y., Bourdeau V., Konstorum A., Lallemant B., Zhang R., et al. Large-scale in silico and microarray-based identification of direct 1,25-dihydroxyvitamin D3 target genes. Mol. Endocrinol. 2005;19:2685–2695. doi: 10.1210/me.2005-0106. [DOI] [PubMed] [Google Scholar]
- 13.Khanal R., Nemere I. Membrane receptors for vitamin D metabolites. Crit. Rev. Eukaryot. Gene Expr. 2007;17:31–47. doi: 10.1615/CritRevEukarGeneExpr.v17.i1.30. [DOI] [PubMed] [Google Scholar]
- 14.Ginde A.A., Liu M.C., Camargo C.A., Jr. Demographic differences and trends of vitamin D insufficiency in the US population, 1988–2004. Arch. Intern. Med. 2009;169:626–632. doi: 10.1001/archinternmed.2008.604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Razzaque M.S. Sunlight exposure: Do health benefits outweigh harm? J. Steroid Biochem. Mol. Biol. 2018;175:44–48. doi: 10.1016/j.jsbmb.2016.09.004. [DOI] [PubMed] [Google Scholar]
- 16.Kennel K.A., Drake M.T., Hurley D.L. Vitamin D deficiency in adults: When to test and how to treat. Mayo Clin. Proc. 2010;85:752–758. doi: 10.4065/mcp.2010.0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ross A.C., Manson J.E., Abrams S.A., Aloia J.F., Brannon P.M., Clinton S.K., Durazo-Arvizu R.A., Gallagher J.C., Gallo R.L., Jones G., et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011;96:53–58. doi: 10.1210/jc.2010-2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Razzaque M.S. Can adverse effects of excessive vitamin D supplementation occur without developing hypervitaminosis D? J. Steroid Biochem. Mol. Biol. 2018;180:81–86. doi: 10.1016/j.jsbmb.2017.07.006. [DOI] [PubMed] [Google Scholar]
- 19.Fu G.K., Lin D., Zhang M.Y., Bikle D.D., Shackleton C.H., Miller W.L., Portale A.A. Cloning of human 25- hydroxyvitamin D-1α-hydroxylase and mutations causing vitamin D-dependent rickets type1. Mol. Endocrinol. 1997;11:1961–1970. doi: 10.1210/mend.11.13.0035. [DOI] [PubMed] [Google Scholar]
- 20.Hosseinpour F., Wikvall K. Porcine microsomal vitamin D3 25-hydroxylase (CYP2D25) J. Biol. Chem. 2000;275:34650–34655. doi: 10.1074/jbc.M004185200. [DOI] [PubMed] [Google Scholar]
- 21.Zhender D., Bland R., Williams M.C., McNinch R.W., Howie A.J., Stewart P.M., Hewison M. Extrarenal expression of 25-hydroxyvitamin D3-1α hydroxylase. J. Clin. Endocrinol. Metab. 2001;86:888–894. doi: 10.1210/jcem.86.2.7220. [DOI] [PubMed] [Google Scholar]
- 22.Naveilhan P., Neveu I., Baude C., Ohyama K.Y., Brachet P., Wion D. Expression of 25(OH) vitamin D3 24-hydroxylase gene in glial cells. NeuroReport. 1993;5:255–257. doi: 10.1097/00001756-199312000-00018. [DOI] [PubMed] [Google Scholar]
- 23.Garcion E., Wion-Barbot N., Montero-Menei C.N., Berger F., Wion D. New clues about vitamin D functions in the nervous system. Trends Endocrinol. Metab. 2002;13:100–105. doi: 10.1016/S1043-2760(01)00547-1. [DOI] [PubMed] [Google Scholar]
- 24.Groves N.J., McGrath J.J., Burne T.H. vitamin D as a neurosteroid affecting the developing and adult brain. Annu. Rev. Nutr. 2014;34:117–141. doi: 10.1146/annurev-nutr-071813-105557. [DOI] [PubMed] [Google Scholar]
- 25.Langub M.C., Herman J.P., Malluche H.H., Koszewski N.J. Evidence of functional vitamin D receptors in rat hippocampus. Neuroscience. 2001;104:49–56. doi: 10.1016/S0306-4522(01)00049-5. [DOI] [PubMed] [Google Scholar]
- 26.Stumpf W.E., Sar M., Clark S.A., DeLuca H.F. Brain target sites for 1,25-dihydroxyvitamin D3. Science. 1982;215:1403–1405. doi: 10.1126/science.6977846. [DOI] [PubMed] [Google Scholar]
- 27.Sutton A.L., MacDonald P.N. Vitamin D: More than a “bone-a-fide” hormone. Mol. Endocrinol. 2003;17:777–791. doi: 10.1210/me.2002-0363. [DOI] [PubMed] [Google Scholar]
- 28.Veenstra T.D., Prufer K., Koenigsberger C., Brimijoin S.W., Grande J.P., Kumar R. 1,25- Dihydroxyvitamin D3 receptors in the central nervous system of the rat embryo. Brain Res. 1998;804:193–205. doi: 10.1016/S0006-8993(98)00565-4. [DOI] [PubMed] [Google Scholar]
- 29.Walbert T., Jirikowski G.F., Prüfer K. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the limbic system of the rat. Horm. Metab. Res. 2001;33:525–531. doi: 10.1055/s-2001-17210. [DOI] [PubMed] [Google Scholar]
- 30.Eyles D.W., Smith S., Kinobe R., Hewison M., McGrath J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005;29:21–30. doi: 10.1016/j.jchemneu.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 31.Eyles D.W., Feron F., Cui X., Kesby J.P., Harms L.H., Ko P., McGrath J.J., Burne T.H.J. Developmental vitamin D deficiency causes abnormal brain development. Psychoneuroendocrinology. 2009;34(Suppl. S1):247–257. doi: 10.1016/j.psyneuen.2009.04.015. [DOI] [PubMed] [Google Scholar]
- 32.Stumpf W.E., Sar M., Rid F.A., Tanaka Y., DeLuca H.F. Target cells for 1,25-dihydroxyvitamin D3 in intestinal tract, stomach, kidney, skin, pituitary, and parathyroid. Science. 1979;206:1188–1190. doi: 10.1126/science.505004. [DOI] [PubMed] [Google Scholar]
- 33.Stumpf W.E., Clark S.A., O’Brien L.P., Reid F.A. 1,25(OH)2 vitamin D3 sites of action in spinal cord and sensory ganglion. Anat Embryol. 1988;177:307–310. doi: 10.1007/BF00315837. [DOI] [PubMed] [Google Scholar]
- 34.Clemens T.L., Garrett K.P., Zhou X.Y., Pike J.W., Haussler M.R., Dempster D.W. Immunocytochemical localization of the 1,25-dihydroxyvitamin D3 receptor in target cells. Endocrinology. 1988;122:1224–1230. doi: 10.1210/endo-122-4-1224. [DOI] [PubMed] [Google Scholar]
- 35.Sutherland M.K., Somerville M.J., Yoong L.K., Bergeron C., Haussler M.R., McLachlan D.R. Reduction of vitamin D hormone receptor mRNA levels in Alzheimer as compared to Huntington hippocampus: Correlation with calbindin-28k mRNA levels. Mol. Brain Res. 1992;13:239–250. doi: 10.1016/0169-328X(92)90032-7. [DOI] [PubMed] [Google Scholar]
- 36.Neveu I., Naveilhan P., Jehan F., Baudet C., Wion D., De Luca H.F., Brachet P. 1,25-Dihydroxyvitamin D3 regulates the synthesis of nerve growth factor in primary cultures of glial cells. Brain Res. Mol. Brain Res. 1994;24:70–76. doi: 10.1016/0169-328X(94)90119-8. [DOI] [PubMed] [Google Scholar]
- 37.Johnson J.A., Grande J.P., Windebank A.J., Kumar R. 1,25-Dihydroxyvitamin D3 receptors in developing dorsal root ganglia of fetal rats. Brain Res. Dev. Brain Res. 1996;92:120–124. doi: 10.1016/0165-3806(95)00204-9. [DOI] [PubMed] [Google Scholar]
- 38.Cornet A., Baudet C., Neveu I., Baron-Van Evercooren A., Brachet P., Naveilhan P. 1,25-Dihydroxyvitamin D3 regulates the expression of VDR and NGF gene in Schwann cells in vitro. J. Neurosci. Res. 1998;53:742–746. doi: 10.1002/(SICI)1097-4547(19980915)53:6<742::AID-JNR11>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 39.Prüfer K., Veenstra T.D., Jirikowski G.F., Kumar R. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanat. 1999;16:135–145. doi: 10.1016/S0891-0618(99)00002-2. [DOI] [PubMed] [Google Scholar]
- 40.Baas D., Prufer K., Ittel M.E., Kuchler-Bopp S., Labourdette G., Sarlieve L.L., Brachet P. Rat oligodendrocytes express the vitamin D(3) receptor and respond to 1,25-dihydroxyvitamin D(3) Glia. 2000;31:59–68. doi: 10.1002/(SICI)1098-1136(200007)31:1<59::AID-GLIA60>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 41.Burkert R., McGrath J., Eyles D. Vitamin D receptor expression in the embryonic rat brain. Neurosci. Res. Commun. 2003;33:63–71. doi: 10.1002/nrc.10081. [DOI] [Google Scholar]
- 42.Erben R.G., Soegiarto D.W., Weber K., Zeitz U., Lieberherr M., Gniadecki R., Moller G., Adamski J., Balling R. Deletion of deoxyribonucleic acid binding domain of the vitamin D receptor abrogates genomic and nongenomic functions of vitamin D. Mol. Endocrinol. 2002;16:1524–1537. doi: 10.1210/mend.16.7.0866. [DOI] [PubMed] [Google Scholar]
- 43.Eyles D., Brown J., Mackay-Sim A., McGrath J., Feron F. Vitamin D3, and brain development. Neuroscience. 2003;118:641–653. doi: 10.1016/S0306-4522(03)00040-X. [DOI] [PubMed] [Google Scholar]
- 44.Schwaller B., Meyer M., Schiffmann S. ‘New’ functions for ‘old’ proteins: The role of the calcium-binding proteins calbindin D-28k, calretinin and parvalbumin, in cerebellar physiology. Studies with knockout mice. Cerebellum. 2002;1:241–258. doi: 10.1080/147342202320883551. [DOI] [PubMed] [Google Scholar]
- 45.Baimbridge K.G., Celio M.R., Rogers J.H. Calcium-binding proteins in the nervous system. Trends Neurosci. 1992;15:303–308. doi: 10.1016/0166-2236(92)90081-I. [DOI] [PubMed] [Google Scholar]
- 46.Alexianu M.E., Robbins E., Carswell S., Appel S.H. 1alpha, 25 dihydroxyvitamin D3-dependent up-regulation of calcium-binding proteins in motoneuron cells. J. Neurosci. Res. 1998;51:58–66. doi: 10.1002/(SICI)1097-4547(19980101)51:1<58::AID-JNR6>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 47.DeViragh P.A., Haglid K.G., Celio M.R. Parvalbumin increases in the caudate putamen of rats with vitamin D hypervitaminosis. Proc. Natl. Acad. Sci. USA. 1989;86:3887–3890. doi: 10.1073/pnas.86.10.3887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Alfahel-Kakunda A., Silverman W.F. Calcium binding proteins in the substantia nigra and ventral tegmental area during development: Correlation with dopaminergic compartmentalization. Brain Res. Dev. Brain Res. 1997;103:9–20. doi: 10.1016/S0165-3806(97)00101-6. [DOI] [PubMed] [Google Scholar]
- 49.Ohshima T., Endo T., Onaya T. Distribution of parvalbumin immunoreactivity in the human brain. J. Neurol. 1991;238:320–322. doi: 10.1007/BF00315329. [DOI] [PubMed] [Google Scholar]
- 50.Schierle G.S., Gander J.C., D’Orlando C., Ceilo M.R., Vogt Weisenhorn D.M. Calretinin-immunoreactivity during postnatal development of the rat isocortex: A qualitative and quantitative study. Cereb. Cortex. 1997;7:130–142. doi: 10.1093/cercor/7.2.130. [DOI] [PubMed] [Google Scholar]
- 51.Ulfig N. Calcium-binding proteins in the human developing brain. Adv. Anat. Embryol. Cell Biol. 2002;165:1–92. [PubMed] [Google Scholar]
- 52.Kishimoto J., Tsuchiya T., Cox H., Emson P.C., Nakayama Y. Age-related changes of calbindin-D28k, calretinin, and parvalbumin mRNAs in the hamster brain. Neurobiol. Aging. 1998;19:77–82. doi: 10.1016/S0197-4580(97)00166-8. [DOI] [PubMed] [Google Scholar]
- 53.Airaksinen M.S., Eilers J., Garaschuk O., Thoenen H., Konnerth A., Meyer M. Ataxia and altered dendritic calcium signaling in mice carrying a targeted null mutation of the calbindin D28k gene. Proc. Natl. Acad. Sci. USA. 1997;94:1488–1493. doi: 10.1073/pnas.94.4.1488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chard P.S., Jordan J., Marcuccilli C.J., Miller R.J., Leiden J.M., Roos R.P., Ghadge G.D. Regulation of excitatory transmission at hippocampal synapses by calbindin D28k. Proc. Natl. Acad. Sci. USA. 1995;92:5144–5148. doi: 10.1073/pnas.92.11.5144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jouvenceau A., Potier B., Poindessous-Jazat F., Dutar P., Slama A., Epelbaum J., Billard J.M. Decrease in calbindin content significantly alters LTP but not NMDA receptor and calcium channel properties. Neuropharmacology. 2002;42:444–458. doi: 10.1016/S0028-3908(01)00202-7. [DOI] [PubMed] [Google Scholar]
- 56.Molinari S., Battini R., Ferrari S., Pozzi L., Killcross A.S., Robbins T.W., Jouvenceau A., Billard J.M., Dutar P., Lamour Y., et al. Deficits in memory and hippocampal long-term potentiation in mice with reduced calbindin D28K expression. Proc. Natl. Acad. Sci. USA. 1996;93:8028–8033. doi: 10.1073/pnas.93.15.8028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Dumas T.C., Powers E.C., Tarapore P.E., Sapolsky R.M. Overexpression of calbindin D(28k) in dentate gyrus granule cells alters mossy fiber presynaptic function and impairs hippocampal-dependent memory. Hippocampus. 2004;14:701–709. doi: 10.1002/hipo.10210. [DOI] [PubMed] [Google Scholar]
- 58.Westerink R.H., Rook M.B., Beekwilder J.P., Wadman W.J. Dual role of calbindin-D28K in vesicular catecholamine release from mouse chromaffin cells. J. Neurochem. 2006;99:628–640. doi: 10.1111/j.1471-4159.2006.04099.x. [DOI] [PubMed] [Google Scholar]
- 59.Barski J.J., Hartmann J., Rose C.R., Hoebeek F., Morl K., Noll-Hussong M., De Zeeuw C.I., Konnerth A., Meyer M. Calbindin in cerebellar Purkinje cells is a critical determinant of the precision of motor coordination. J. Neurosci. 2003;23:3469–3477. doi: 10.1523/JNEUROSCI.23-08-03469.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Toutenhoofd S.L., Strehler E.E. The calmodulin multigene family as a unique case of genetic redundancy: Multiple levels of regulation to provide spatial and temporal control of calmodulin pools? Cell Calcium. 2000;28:83–96. doi: 10.1054/ceca.2000.0136. [DOI] [PubMed] [Google Scholar]
- 61.De Boland A.R., Massheimer V., Fernandez L.M. 1,25-Dihydroxyvitamin D3 affects calmodulin distribution among subcellular fractions of skeletal muscle. Calcif. Tissue Int. 1988;43:370–375. doi: 10.1007/BF02553281. [DOI] [PubMed] [Google Scholar]
- 62.Gnegy M.E. Calmodulin in neurotransmitter and hormone action. Annu. Rev. Pharmacol. Toxicol. 1993;33:45–70. doi: 10.1146/annurev.pa.33.040193.000401. [DOI] [PubMed] [Google Scholar]
- 63.Gnegy M.E. Ca2+/calmodulin signaling in NMDA induced synaptic plasticity. Crit. Rev. Neurobiol. 2000;14:91–129. doi: 10.1615/CritRevNeurobiol.v14.i2.10. [DOI] [PubMed] [Google Scholar]
- 64.Junge H.J., Rhee J.S., Jahn O., Varoqueaux F., Spiess J., Waxham M.N., Rosenmund C., Brose N. Calmodulin and Munc13 form a Ca2+ sensor/effector complex that controls short-term synaptic plasticity. Cell. 2004;118:389–401. doi: 10.1016/j.cell.2004.06.029. [DOI] [PubMed] [Google Scholar]
- 65.Lisman J., Schulman H., Cline H. The molecular basis of CaMKII function in synaptic and behavioural memory. Nat. Rev. Neurosci. 2002;3:175–190. doi: 10.1038/nrn753. [DOI] [PubMed] [Google Scholar]
- 66.Mizuno K., Antunes-Martins A., Ris L., Peters M., Godaux E., Giese K.P. Calcium/calmodulin kinase kinase beta has a male-specific role in memory formation. Neuroscience. 2007;145:393–402. doi: 10.1016/j.neuroscience.2006.11.056. [DOI] [PubMed] [Google Scholar]
- 67.Poulsen D.J., Standing D., Bullshields K., Spencer K., Micevych P.E., Babcock A.M. Overexpression of hippocampal Ca2+/calmodulin-dependent protein kinase II improves spatial memory. J. Neurosci. Res. 2007;85:735–739. doi: 10.1002/jnr.21163. [DOI] [PubMed] [Google Scholar]
- 68.Yamauchi T. Neuronal Ca2+/calmodulin-dependent protein kinase II– discovery, progress in a quarter of a century, and perspective: Implication for learning and memory. Biol. Pharm. Bull. 2005;28:1342–1354. doi: 10.1248/bpb.28.1342. [DOI] [PubMed] [Google Scholar]
- 69.Brewer L.D., Thibault V., Chen K.C., Langub M.C., Landfield P.W., Porter N.M. Vitamin D hormone confers neuroprotection in parallel with downregulation of L-type Calcium channel expression in hippocampal neurons. J. Neurosci. 2001;21:98–108. doi: 10.1523/JNEUROSCI.21-01-00098.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Taniura H., Ito M., Sanada N., Kuramoto N., Ohno Y., Nakamichi N., Yoneda Y. Chronic vitamin D(3) treatment protects against neurotoxicity by glutamate in association with upregulation of vitamin D receptor mRNA expression in cultured rat cortical neurons. J. Neurosci. Res. 2006;83:1179–1189. doi: 10.1002/jnr.20824. [DOI] [PubMed] [Google Scholar]
- 71.Moore T.B., Koeffler H.P., Yamashiro J.M., Wada R.K. Vitamin D3 analogs inhibit growth and induce differentiation in LA-N-5 human neuroblastoma cells. Clin. Exp. Metastasis. 1996;14:239–245. doi: 10.1007/BF00053897. [DOI] [PubMed] [Google Scholar]
- 72.Brown J., Bianco J.I., McGrath J.J., Eyles D.W. 1,25-dihydroxyvitamin D3 induces nerve growth factor, promotes neurite outgrowth and inhibits mitosis in embryonic rat hippocampal neurons. Neurosci. Lett. 2003;343:139–143. doi: 10.1016/S0304-3940(03)00303-3. [DOI] [PubMed] [Google Scholar]
- 73.Obradovic D., Gronemeyer H., Lutz B., Rein T. Cross-talk of vitamin D and glucocorticoids in hippocampal cells. J. Neurochem. 2006;96:500–509. doi: 10.1111/j.1471-4159.2005.03579.x. [DOI] [PubMed] [Google Scholar]
- 74.Lledo P.M., Alonso M., Grubb M.S. Adult neurogenesis and functional plasticity in neuronal circuits. Nat. Rev. Neurosci. 2006;7:179–193. doi: 10.1038/nrn1867. [DOI] [PubMed] [Google Scholar]
- 75.Hagg T. Molecular regulation of adult CNS neurogenesis: An integrated view. Trends Neurosci. 2005;28:589–595. doi: 10.1016/j.tins.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 76.Watts C., McConkey H., Anderson L., Caldwell M. Anatomical perspectives on adult neural stem cells. J. Anat. 2005;207:197–208. doi: 10.1111/j.1469-7580.2005.00448.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Taupin P., Gage F.H. Adult neurogenesis and neural stem cells of the central nervous system in mammals. J. Neurosci. Res. 2002;69:745–749. doi: 10.1002/jnr.10378. [DOI] [PubMed] [Google Scholar]
- 78.Purves D., Fitzpatrick D., Augustine G.J., Katz L.C., Williams S.M., McNamara J.O., Lamantia A.S. Neuroscience. 2nd ed. Sinaur Associates; Sunderland, MA, USA: 2001. [Google Scholar]
- 79.Cianferotti L., Cox M., Skorija K., Demay M.B. Vitamin D receptor is essential for normal keratinocyte stem cell function. Proc. Natl. Acad. Sci. USA. 2007;104:9428–9433. doi: 10.1073/pnas.0702884104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cui X., McGrath J.J., Burne T.H., Mackay-Sim A., Eyles D.W. Maternal vitamin D depletion alters neurogenesis in the developing rat brain. Int. J. Dev. Neurosci. 2007;25:227–232. doi: 10.1016/j.ijdevneu.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 81.Landfield P.W., Cadwallader-Neal L. Long-term treatment with calcitriol (1,25(OH)2 vit D3) retards a biomarker of hippocampal aging in rats. Neurobiol. Aging. 1998;19:469–477. doi: 10.1016/S0197-4580(98)00079-7. [DOI] [PubMed] [Google Scholar]
- 82.Cai Q., Tapper D.N., Gilmour R.F., Jr., deTalamoni N., Aloia R.C., Wasserman R.H. Modulation of the excitability of avian peripheral nerves by vitamin D: Relation to calbindin-D28k, calcium status and lipid composition. Cell Calcium. 1994;15:401–410. doi: 10.1016/0143-4160(94)90015-9. [DOI] [PubMed] [Google Scholar]
- 83.Cantorna M.T., Hayes C.E., DeLuca H.F. 1,25-Dihydroxyvitamin D3 reversibly blocks the progression of relapsing encephalomyelitis, a model of multiple sclerosis. Proc. Natl. Acad. Sci. USA. 1996;93:7861–7864. doi: 10.1073/pnas.93.15.7861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wang Y., Chiang Y.H., Su T.P., Hayashi T., Morales M., Hoffer B.J., Lin S.Z. Vitamin D3 attenuates cortical infarction induced by middle cerebral arterial ligation in rats. Neuropharmacology. 2000;39:873–880. doi: 10.1016/S0028-3908(99)00255-5. [DOI] [PubMed] [Google Scholar]
- 85.Riaz S., Malcangio M., Miller M., Tomlinson D.R. A vitamin D3 derivative (CB1093) induces nerve growth factor and prevents neurotrophic deficits in streptozotocin-diabetic rats. Diabetologia. 1999;42:1308–1313. doi: 10.1007/s001250051443. [DOI] [PubMed] [Google Scholar]
- 86.Saporito M.S., Brown E.R., Hartpence K.C., Wilcox H.M., Vaught J.L., Carswell S. Chronic 1,25-dihydroxyvitamin D3-mediated induction of nerve growth factor mRNA and protein in L929 fibroblasts and in adult rat brain. Brain Res. 1994;633:189–196. doi: 10.1016/0006-8993(94)91539-3. [DOI] [PubMed] [Google Scholar]
- 87.Naveilhan P., Neveu I., Wion D., Brachet P. 1,25-Dihydroxyvitamin D3, an inducer of glial cell line-derived neurotrophic factor. Neuroreport. 1996;7:2171–2175. doi: 10.1097/00001756-199609020-00023. [DOI] [PubMed] [Google Scholar]
- 88.Siegel G.J., Agranoff B.W., Albers R.W., Fisher S.K., Uhler M.D. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th ed. Lippincott Williams & Wilkins; Philadelphia, PA, USA: 1999. [Google Scholar]
- 89.Kang H., Schuman E.M. Long-lasting neurotrophin-induced enhancement of synaptic transmission in the adult hippocampus. Science. 1995;267:1658–1662. doi: 10.1126/science.7886457. [DOI] [PubMed] [Google Scholar]
- 90.Rose C.R., Blum R., Pichler B., Lepier A., Kafitz K.W., Konnerth A. Truncated TrkB-T1 mediates neurotrophin-evoked calcium signaling in glia cells. Nature. 2003;426:74–78. doi: 10.1038/nature01983. [DOI] [PubMed] [Google Scholar]
- 91.Wion D., MacGrogan D., Neveu I., Jehan F., Houlgatte R., Brachet P. 1,25-dihydroxyvitamin D3 is a potent inducer of nerve growth factor synthesis. J. Neurosci. Res. 1991;28:110–114. doi: 10.1002/jnr.490280111. [DOI] [PubMed] [Google Scholar]
- 92.Christophersen N.S., Gronborg M., Petersen T.N., Fjord-Larsen L., Jorgensen J.R., Juliusson B., Blom N., Rosenblad C., Brundin P. Midbrain expression of delta-like 1 homologue is regulated by GDNF and is associated with dopaminergic differentiation. Exp. Neurol. 2007;204:791–801. doi: 10.1016/j.expneurol.2007.01.014. [DOI] [PubMed] [Google Scholar]
- 93.Morrison B.M., Hof P.R., Morrison J.H. Determinants of neuronal vulnerability in neurodegenerative diseases. Ann. Neurol. 1998;44(Suppl. S1):32–44. doi: 10.1002/ana.410440706. [DOI] [PubMed] [Google Scholar]
- 94.Smith D.E., Saji M., Joh T.H., Reis D.J., Pickel V.M. Ibotenic acid-induced lesions of striatal target and projection neurons: Ultrastructural manifestations in dopaminergic and non-dopaminergic neurons and in glia. Histol. Histopathol. 1987;2:251–263. [PubMed] [Google Scholar]
- 95.Mattson M.P. Pathways towards and away from Alzheimer’s disease. Nature. 2004;430:631–639. doi: 10.1038/nature02621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cookson M.R. The biochemistry of Parkinson’s disease. Annu. Rev. Biochem. 2005;74:29–52. doi: 10.1146/annurev.biochem.74.082803.133400. [DOI] [PubMed] [Google Scholar]
- 97.Moore D.J., West A.B., Dawson V.L., Dawson T.M. Molecular pathophysiology of Parkinson’s disease. Annu. Rev. Neurosci. 2005;27:57–87. doi: 10.1146/annurev.neuro.28.061604.135718. [DOI] [PubMed] [Google Scholar]
- 98.Sieradzan K.A., Mann D.M. The selective vulnerability of nerve cells in Huntington’s disease. Neuropathol. Appl. Neurobiol. 2001;27:1–21. doi: 10.1046/j.0305-1846.2001.00299.x. [DOI] [PubMed] [Google Scholar]
- 99.Cleveland D.W., Rothstein J.D. From Charcot to Lou Gehrig: Deciphering selective motor neuron death in ALS. Nat. Rev. Neurosci. 2001;2:806–819. doi: 10.1038/35097565. [DOI] [PubMed] [Google Scholar]
- 100.Mandelkow E.M., Stamer K., Vogel R., Thies E., Mandelkow E. Clogging of axons by tau, inhibition of axonal traffic and starvation of synapses. Neurobiol. Aging. 2003;24:1079–1085. doi: 10.1016/j.neurobiolaging.2003.04.007. [DOI] [PubMed] [Google Scholar]
- 101.Bayatti N., Behl C. The neuroprotective actions of corticotropin releasing hormone. Ageing Res. Rev. 2005;4:258–270. doi: 10.1016/j.arr.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 102.Braak H., Braak E. Evolution of neuronal changes in the course of Alzheimer’s disease. J. Neural. Transm. Suppl. 1998;53:127–140. doi: 10.1007/978-3-7091-6467-9_11. [DOI] [PubMed] [Google Scholar]
- 103.Latimer C.S., Brewer L.D., Searcy J.L., Chen K.C., Popović J., Kraner S.D., Thibault O., Blalock E.M., Landfield P.W., Porter N.M. Vitamin D prevents cognitive decline and enhances hippocampal synaptic function in aging rats. Proc. Natl. Acad. Sci. USA. 2014;111:E4359–E4366. doi: 10.1073/pnas.1404477111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Eyles D.W., Burne T.H., McGrath J.J. Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front. Neuroendocrinol. 2013;34:47–64. doi: 10.1016/j.yfrne.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 105.Trump D.L., Deeb K.K., Johnson C.S. Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010;16:1–9. doi: 10.1097/PPO.0b013e3181c51ee6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Jiang P., Zhu W.Y., He X., Tang M.M., Dang R.L., Li H.D., Xue Y., Zhang L.H., Wu Y.Q., Cao L.J. Association between vitamin D receptor gene polymorphisms with childhood temporal lobe epilepsy. Int. J. Environ. Res. Public Health. 2015;12:13913–13922. doi: 10.3390/ijerph121113913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Palaniappan A.R., Dai A. Mitochondrial aging and the beneficial role of alpha-lipoic acid. Neurochem. Res. 2007;32:1552–1558. doi: 10.1007/s11064-007-9355-4. [DOI] [PubMed] [Google Scholar]
- 108.Shirazi H.A., Rasouli J., Ciric B., Rostami A., Zhang G.X. 1,25-Dihydroxyvitamin D3 enhances neural stem cell proliferation and oligodendrocyte differentiation. Exp. Mol. Pathol. 2015;98:240–245. doi: 10.1016/j.yexmp.2015.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.De la Fuente A.G., Errea O., van Wijngaarden P., Gonzalez G.A., Kerninon C., Jarjour A.A., Lewis H.J., Jones C.A., Nait-Oumesmar B., Zhao C., et al. Vitamin D receptor-retinoid X receptor heterodimer signaling regulates oligodendrocyte progenitor cell differentiation. J. Cell Biol. 2015;211:975–985. doi: 10.1083/jcb.201505119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Landel V., Annweiler C., Millet P., Morello M., Féron F. Vitamin D, Cognition and Alzheimer’s Disease: The Therapeutic Benefit is in the D-Tails. J. Alzheimers Dis. 2016;53:419–444. doi: 10.3233/JAD-150943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Winner B., Winkler J. Adult neurogenesis in neurodegenerative diseases. Cold Spring Harb. Perspect. Biol. 2015;7:a021287. doi: 10.1101/cshperspect.a021287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Annweiler C., Karras S.N., Anagnostis P., Beauchet O. Vitamin D supplements: A novel therapeutic approach for Alzheimer patients. Front. Pharmacol. 2014;5:6. doi: 10.3389/fphar.2014.00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Radak Z., Taylor A.W., Ohno H., Goto S. Adaptation to exercise-induced oxidative stress: From muscle to brain. Exerc. Immunol. Rev. 2001;7:90–107. [PubMed] [Google Scholar]
- 114.DeLuca G.C., Kimball S.M., Kolasinski J., Ramagopalan S.V., Ebers G.C. Review: The role of vitamin D in nervous system health and disease. Neuropathol. Appl. Neurobiol. 2013;39:458–484. doi: 10.1111/nan.12020. [DOI] [PubMed] [Google Scholar]
- 115.Pirota S., Kidgell D.J., Daly R.M. Effects of vitamin D supplementation on neuroplasticity in older adults: A double-blinded, placebo-controlled randomised trial. Osteoporos. Int. 2015;26:131–140. doi: 10.1007/s00198-014-2855-6. [DOI] [PubMed] [Google Scholar]
- 116.Taghizadeh M., Talaei S.A., Djazayeri A., Salami M. Vitamin D supplementation restores suppressed synaptic plasticity in Alzheimer’s disease. Nutr. Neurosci. 2014;17:172–177. doi: 10.1179/1476830513Y.0000000080. [DOI] [PubMed] [Google Scholar]
- 117.Nut D. GABA-A receptors: Subtypes, regional distribution, and function. J. Clin. Sleep Med. 2006;2:S7–S11. [PubMed] [Google Scholar]
- 118.Manini T.M., Clark B.C. Dynapenia and aging: An update. J. Gerontol. A Biol. Sci. Med. Sci. 2012;67:28–40. doi: 10.1093/gerona/glr010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Garcion E., Sindji L., Montero-Menei C., Andre C., Brachet P., Darcy F. Expression of inducible nitric oxide synthase during rat brain inflammation: Regulation by 1,25- dihydroxyvitamin D3. Glia. 1998;22:282–294. doi: 10.1002/(SICI)1098-1136(199803)22:3<282::AID-GLIA7>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 120.Lin A.M., Chen K.B., Chao P.L. Antioxidative effect of vitamin D3 on zinc-induced oxidative stress in CNS. Ann. N. Y. Acad. Sci. 2005;1053:319–329. doi: 10.1196/annals.1344.028. [DOI] [PubMed] [Google Scholar]
- 121.Lemire J.M., Archer D.C. 1,25-dihydroxyvitamin D3 prevents the in vivo induction of murine experimental autoimmune encephalomyelitis. J. Clin. Investig. 1991;87:1103–1107. doi: 10.1172/JCI115072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Nataf S., Garcion E., Darcy F., Chabannes D., Muller J.Y., Brachet P. 1,25 Dihydroxyvitamin D3 exerts regional effects in the central nervous system during experimental allergic encephalomyelitis. J. Neuropathol. Exp. Neurol. 1996;55:904–914. doi: 10.1097/00005072-199608000-00006. [DOI] [PubMed] [Google Scholar]
- 123.Garcion E., Nataf S., Berod A., Darcy F., Brachet P. 1,25-Dihydroxyvitamin D3 inhibits the expression of inducible nitric oxide synthase in rat nervous system during experimental allergic encephalomyelitis. Brain Res. Mol. Brain Res. 1997;45:255–267. doi: 10.1016/S0169-328X(96)00260-4. [DOI] [PubMed] [Google Scholar]
- 124.Garcion E., Sindji L., Nataf S., Brachet P., Darcy F., Montero-Menei C.N. Treatment of experimental autoimmune encephalomyelitis in rat by 1,25-dihydroxyvitamin D3 leads to early effects within the central nervous system. Acta Neuropathol. 2003;105:438–448. doi: 10.1007/s00401-002-0663-0. [DOI] [PubMed] [Google Scholar]
- 125.Mattner F., Smiroldo S., Galbiati F., Muller M., Di Lucia P., Poliani P.L., Martino G., Panina-Bordignon P., Adorini L. Inhibition of Th1 development and treatment of chronic-relapsing experimental allergic encephalomyelitis by a non-hypercalcemic analogue of 1,25-dihydroxyvitamin D(3) Eur. J. Immunol. 2000;30:498–508. doi: 10.1002/1521-4141(200002)30:2<498::AID-IMMU498>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 126.Cantorna M.T., Woodward W.D., Hayes C.E., DeLuca H.F. 1,25-dihydroxyvitamin D3 is a positive regulator for the two anti-encephalitogenic cytokines TGF-beta 1 and IL-4. J. Immunol. 1998;160:5314–5319. [PubMed] [Google Scholar]
- 127.Spach K.M., Pedersen L.B., Nashold F.E., Kayo T., Yandell B.S., Prolla T.A., Hayes C.E. Gene expression analysis suggests that 1,25-dihydroxyvitamin D3 reverses experimental autoimmune encephalomyelitis by stimulating inflammatory cell apoptosis. Physiol. Genomics. 2004;18:141–151. doi: 10.1152/physiolgenomics.00003.2004. [DOI] [PubMed] [Google Scholar]
- 128.Lefebvre d’Hellencourt C., Montero-Menei C.N., Bernard R., Couez D. Vitamin D3 inhibits proinflammatory cytokines and nitric oxide production by the EOC13 microglial cell line. J. Neurosci. Res. 2003;71:575–582. doi: 10.1002/jnr.10491. [DOI] [PubMed] [Google Scholar]
- 129.Veenstra T.D., Windebank A.J., Kumar R. 1,25-dihydroxyvitamin D3 regulates the expression of Nmyc, c-myc, protein kinase C, and transforming growth factor-beta2 in neuroblastoma cells. Biochem. Biophys. Res. Commun. 1997;235:15–18. doi: 10.1006/bbrc.1997.6718. [DOI] [PubMed] [Google Scholar]
- 130.Riaz S.S., Tomlinson D.R. Pharmacological modulation of nerve growth factor synthesis: A mechanistic comparison of vitamin D receptor and beta(2)-adrenoceptor agonists. Brain Res. Mol. Brain Res. 2000;85:179–188. doi: 10.1016/S0169-328X(00)00254-0. [DOI] [PubMed] [Google Scholar]
- 131.Zorica J. Antioxidative defense mechanisms in the aging brain. Arch. Biol. Sci. 2014;66:245–252. [Google Scholar]
- 132.Harman D. The biologic clock: The mitochondria? J. Am. Geriatr. Soc. 1972;20:145–147. doi: 10.1111/j.1532-5415.1972.tb00787.x. [DOI] [PubMed] [Google Scholar]
- 133.Nordberg J., Arnér E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001;31:1287–1312. doi: 10.1016/S0891-5849(01)00724-9. [DOI] [PubMed] [Google Scholar]
- 134.Chakravarti B., Chakravarti D.N. Oxidative modification of proteins: Age-related changes. Gerontology. 2007;53:128–139. doi: 10.1159/000097865. [DOI] [PubMed] [Google Scholar]
- 135.Cass W.A., Smith M.P., Peters L.E. Calcitriol protects against the dopamine- and serotonin-depleting effects of neurotoxic doses of methamphetamine. Ann. N. Y. Acad. Sci. 2006;1074:261–271. doi: 10.1196/annals.1369.023. [DOI] [PubMed] [Google Scholar]
- 136.Wrzosek M., Lukaszkiewicz J., Wrzosek M., Jakubczyk A., Matsumoto H., Piatkiewicz P., Radziwoñ-Zaleska M., Wojnar M., Nowicka G. Vitamin D and the central nervous system. Pharmacol. Rep. 2013;65:271–278. doi: 10.1016/S1734-1140(13)71003-X. [DOI] [PubMed] [Google Scholar]
- 137.Halhali A., Figueras A.G., Diaz L., Avila E., Barrera D., Hernández G., Larrea F. Effects of calcitriol on calbindins gene expression and lipid peroxidation in human placenta. J. Steroid Biochem. Mol. Biol. 2010;121:448–451. doi: 10.1016/j.jsbmb.2010.03.008. [DOI] [PubMed] [Google Scholar]
- 138.Uchino M., Ando Y., Tanaka Y., Nakamura T., Uyama E., Mita S., Murakami T., Ando M. Decrease in cu/zn- and mn-superoxide dismutase activities in brain and spinal cord of patients with amyotrophic lateral sclerosis. J. Neurol. Sci. 1994;127:61–67. doi: 10.1016/0022-510X(94)90136-8. [DOI] [PubMed] [Google Scholar]
- 139.Macmillan-Crow L.A., Cruthirds D.L. Invited review: Manganese superoxide dismutase in disease. Free Radic. Res. 2001;34:325–336. doi: 10.1080/10715760100300281. [DOI] [PubMed] [Google Scholar]
- 140.Frakes A.E., Ferraiuolo L., Haidet-Phillips A.M., Schmelzer L., Braun L., Miranda C.J., Ladner K.J., Bevan A.K., Foust K.D., Godbout J.P., et al. Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron. 2014;81:1009–1023. doi: 10.1016/j.neuron.2014.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Keeney J.T.R., Förster S., Sultana R., Brewer L.D., Latimer C.S., Cai J., Klein J.B., Porter N.M., Butterfield D.A. Dietary vitamin D deficiency in rats from middle to old age leads to elevated tyrosine nitration and proteomics changes in levels of key proteins in brain: Implications for low vitamin D-dependent age-related cognitive decline. Free Radic. Biol. Med. 2013;65:324–334. doi: 10.1016/j.freeradbiomed.2013.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Saedisomeolia A., Taheri E., Djalali M., Djazayeri A., Qorbani M., Rajab A., Larijani B. Vitamin D status and its association with antioxidant profiles in diabetic patients: A cross-sectional study in Iran. Indian J. Med. Sci. 2013;67:29–37. [PubMed] [Google Scholar]
- 143.Dawson V.L., Dawson T.M. Nitric oxide actions in neurochemistry. Neurochem. Int. 1996;29:97–110. doi: 10.1016/0197-0186(95)00149-2. [DOI] [PubMed] [Google Scholar]
- 144.Mitrovic B., St Pierre B.A., Mackenzie-Graham A.J., Merrill J.E. The role of nitric oxide in glial pathology. Ann. N. Y. Acad. Sci. 1994;738:436–446. doi: 10.1111/j.1749-6632.1994.tb21835.x. [DOI] [PubMed] [Google Scholar]
- 145.Garcion E., Thanh X.D., Bled F., Teissier E., Dehouck M.P., Rigault F., Brachet P., Girault A., Torpier G., Darcy F. 1,25-Dihydroxyvitamin D3 regulates gamma 1 transpeptidase activity in rat brain. Neurosci. Lett. 1996;216:183–186. doi: 10.1016/0304-3940(96)87802-5. [DOI] [PubMed] [Google Scholar]
- 146.Garcion E., Sindji L., Leblondel G., Brachet P., Darcy F. 1,25-Dihydroxyvitamin D3 regulates the synthesis of ã-glutamyl transpeptidase and glutathione levels in rat primary astrocytes. J. Neurochem. 1999;73:859–866. doi: 10.1046/j.1471-4159.1999.0730859.x. [DOI] [PubMed] [Google Scholar]
- 147.Dringen R., Guetterer J.M., Hirrlinger J. Glutathione metabolism in brain metabolic interaction between astrocytes and neurons in the defense against reactive oxygen species. Eur. J. Biochem. 2000;267:4912–4916. doi: 10.1046/j.1432-1327.2000.01597.x. [DOI] [PubMed] [Google Scholar]
- 148.Molina-Jijón E., Tapia E., Zazueta C., El Hafidi M., Zatarain-Barron Z.L., Hernandez-Pando R., Medina-Campos O.N., Zarco-Márquez G., Torres I., Pedraza-Chaverri J. Curcumin prevents Cr (VI)—Induced renal oxidant damage by a mitochondrial pathway. Free Radic. Biol. Med. 2011;51:1543–1557. doi: 10.1016/j.freeradbiomed.2011.07.018. [DOI] [PubMed] [Google Scholar]
- 149.Jagetia G.C., Aggarwal B.B. “Spicing up” of the immune system by curcumin. J. Clin. Immunol. 2007;27:19–35. doi: 10.1007/s10875-006-9066-7. [DOI] [PubMed] [Google Scholar]
- 150.Jung K.K., Lee H.S., Cho J.Y., Shin W.C., Rhee M.H., Kim T.G., Kang J.H., Kim S.H., Hong S., Kang S.Y. Inhibitory effect of curcumin on nitric oxide production from lipopolysaccharide-activated primary microglia. Life Sci. 2006;79:2022–2031. doi: 10.1016/j.lfs.2006.06.048. [DOI] [PubMed] [Google Scholar]
- 151.Jin C.Y., Lee J.D., Park C., Choi Y.H., Kim G.Y. Curcumin attenuates the release of pro-inflammatory cytokines in lipopolysaccharide-stimulated BV2 microglia. Acta Pharmacol. Sin. 2007;28:1645–1651. doi: 10.1111/j.1745-7254.2007.00651.x. [DOI] [PubMed] [Google Scholar]
- 152.He L.F., Chen H.J., Qian L.H., Chen G.Y., Buzby J.S. Curcumin protects pre-oligodendrocytes from activated microglia in vitro and in vivo. Brain Res. 2010;1339:60–69. doi: 10.1016/j.brainres.2010.04.014. [DOI] [PubMed] [Google Scholar]
- 153.Yang S., Zhang D., Yang Z., Hu X., Qian S., Liu J., Wilson B., Block M., Hong J.S. Curcumin protects dopaminergic neuron against LPS induced neurotoxicity in primary rat neuron/glia culture. Neurochem. Res. 2008;33:33–2044. doi: 10.1007/s11064-008-9675-z. [DOI] [PubMed] [Google Scholar]
- 154.Mandal M.N., Patlolla J.M., Zheng L., Agbaga M.P., Tran J.T., Wicker L., Kasus-Jacobi A., Elliott M.H., Rao C.V., Anderson R.E. Curcumin protects retinal cells from light-and oxidant stress-induced cell death. Free Radic. Biol. Med. 2009;46:672–679. doi: 10.1016/j.freeradbiomed.2008.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kim H.Y., Park E.J., Joe E.H., Jou I. Curcumin suppresses Janus kinase-STAT inflammatory signaling through activation of Src homology 2 domain-containing tyrosine phosphatase 2 in brain microglia. J. Immunol. 2003;171:6072–6079. doi: 10.4049/jimmunol.171.11.6072. [DOI] [PubMed] [Google Scholar]
- 156.Wang H.M., Zhao Y.X., Zhang S., Liu G.D., Kang W.Y., Tang H.D., Ding J.Q., Chen S.D. PPARgamma agonist curcumin reduces the amyloid-beta-stimulated inflammatory responses in primary astrocytes. J. Alzheimers Dis. 2010;20:1189–1199. doi: 10.3233/JAD-2010-091336. [DOI] [PubMed] [Google Scholar]
- 157.Cole G.M., Teter B., Frautschy S.A. Neuroprotective effects of curcumin. Adv. Exp. Med. Biol. 2007;595:197–212. doi: 10.1007/978-0-387-46401-5_8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Li L., Aggarwal B.B., Shishodia S., Abbruzzese J., Kurzrock R. Nuclear factor-kappaB and IkappaB kinase are constitutively active in human pancreatic cells, and their down-regulation by curcumin (diferuloylmethane) is associated with the suppression of proliferation and the induction of apoptosis. Cancer. 2004;101:2351–2362. doi: 10.1002/cncr.20605. [DOI] [PubMed] [Google Scholar]
- 159.Shakibaei M., Schulze-Tanzil G., John T., Mobasheri A. Curcumin protects human chondrocytes from IL-1 beta-induced inhibition of collagen type II and beta1-integrin expression and activation of caspase-3: An immunomorphological study. Ann. Anat. 2005;187:487–497. doi: 10.1016/j.aanat.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 160.Shishodia S., Amin H.M., Lai R., Aggarwal B.B. Curcumin (diferuloylmethane) inhibits constitutive NF-kappaB activation, induces G1/S arrest, suppresses proliferation, and induces apoptosis in mantle cell lymphoma. Biochem. Pharmacol. 2005;70:700–713. doi: 10.1016/j.bcp.2005.04.043. [DOI] [PubMed] [Google Scholar]
- 161.Skommer J., Wlodkowic D., Pelkonen J. Gene-expression profiling during curcumin-induced apoptosis reveals downregulation of CXCR4. Exp. Hematol. 2007;35:84–95. doi: 10.1016/j.exphem.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 162.Huang X., Moir R.D., Tanzi R.E., Bush A.I., Rogers J.T. Redox-active metals, oxidative stress, and Alzheimer’s disease pathology. Ann. N. Y. Acad. Sci. 2004;1012:153–163. doi: 10.1196/annals.1306.012. [DOI] [PubMed] [Google Scholar]
- 163.Pan J., Li H., Ma J.F., Tan Y.Y., Xiao Q., Ding J.Q., Chen S.D. Curcumin inhibition of JNKs prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease through suppressing mitochondria dysfunction. Transl. Neurodegener. 2012;1:16. doi: 10.1186/2047-9158-1-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Mufti S., Bautista A., Pino-Figueroa A. Evaluation of the neuroprotective effects of curcuminoids on B35 and SH-SY5Y neuroblastoma cells. Med. Aromat. Plants. 2015;4:197. doi: 10.4172/2167-0412.1000197. [DOI] [Google Scholar]
- 165.Yu S., Zheng W., Xin N., Chi Z.H., Wang N.Q., Nie Y.X., Feng W.Y., Wang Z.Y. Curcumin prevents dopaminergic neuronal death through inhibition of the c-Jun N-terminal kinase pathway. Rejuvenation Res. 2010;13:55–64. doi: 10.1089/rej.2009.0908. [DOI] [PubMed] [Google Scholar]
- 166.Zheng K.M., Zhang J., Zhang C.L., Zhang Y.W., Chen X.C. Curcumin inhibits appoptosin-induced apoptosis via upregulating heme oxygenase-1 expression in SH-SY5Y cells. Acta Pharmacol. Sin. 2015;36:544–552. doi: 10.1038/aps.2014.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Hunter J.E., Schmidt F.L. Methods of Meta-Analysis: Correcting Errors and Bias in Research Findings. Sage; Thousand Oaks, CA, USA: 2006. [Google Scholar]
- 168.Aghajafari F., Pond D., Catzikiris N., Cameron I. Quality assessment of systematic reviews of vitamin D, cognition and dementia. BJPsych Open. 2018;4:238–249. doi: 10.1192/bjo.2018.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Suzuki M., Yoshioka M., Hashimoto M., Murakami M., Noya M., Takahashi D., Urashima M. Randomized, double-blind, placebo-controlled trial of vitamin D supplementation in Parkinson disease. Am. J. Clin. Nutr. 2013;97:1004–1013. doi: 10.3945/ajcn.112.051664. [DOI] [PubMed] [Google Scholar]
- 170.Corless D., Ellis M., Dawson E., Fraser F. Using activities of daily living assessments to measure the effectiveness of vitamin D supplements in elderly long-stay patients. Br. J. Occup. Ther. 1987;50:60–62. doi: 10.1177/030802268705000208. [DOI] [Google Scholar]
- 171.Barnard K., Colon-Emeric C. Extraskeletal effects of vitamin D in older adults: Cardiovascular disease, mortality, mood, and cognition. Am. J. Geriatr. Pharmacol. 2010;8:4–33. doi: 10.1016/j.amjopharm.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 172.Rutjes A.W., Denton D.A., Di Nisio M., Chong L.Y., Abraham R.P., Al-Assaf A.S., Anderson J.L., Malik M.A., Vernooij R.W., Martínez G., et al. Vitamin and mineral supplementation for maintaining cognitive function in cognitively healthy people in mid and late life. Cochrane Database Syst. Rev. 2018;12:CD011906. doi: 10.1002/14651858.CD011906.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Koduah P., Paul F., Dörr J.M. Vitamin D in the prevention, prediction and treatment of neurodegenerative and neuroinflammatory diseases. EPMA J. 2017;8:313–325. doi: 10.1007/s13167-017-0120-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Chandra R.K. Effect of vitamin and trace-element supplementation on cognitive function in elderly subjects. Nutrition. 2001;17:709–712. doi: 10.1016/S0899-9007(01)00610-4. [DOI] [PubMed] [Google Scholar]
- 175.Sultan S., Taimuri U., Basnan S.A., Ai-Orabi W.K., Awadallah A., Almowald F., Hazazi A. Low Vitamin D and Its Association with Cognitive Impairment and Dementia. J. Aging Res. 2020;2020:6097820. doi: 10.1155/2020/6097820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Balion C., Griffith L.E., Strifler L., Henderson M., Patterson C., Heckman G., Llewellyn D.J., Raina P. Vitamin D, cognition, and dementia: A systematic review and meta-analysis. Neurology. 2012;79:1397–1405. doi: 10.1212/WNL.0b013e31826c197f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Annweiler C., Herrmann F.R., Fantino B., Brugg B., Beauchet O. Effectiveness of the combination of memantine plus vitamin D on cognition in patients with Alzheimer disease: A pre-post pilot study. Cogn. Behav. Neurol. 2012;25:121–127. doi: 10.1097/WNN.0b013e31826df647. [DOI] [PubMed] [Google Scholar]
- 178.Matsunaga S., Kishi T., Iwata N. Memantine monotherapy for Alzheimer’s disease: A systematic review and meta-analysis. PLoS ONE. 2015;10:e0123289. doi: 10.1371/journal.pone.0123289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Gagliardi S., Franco V., Sorrentino S., Zucca S., Pandini C., Rota P., Bernuzzi S., Costa A., Sinforiani E., Pansarasa O., et al. Curcumin and Novel Synthetic Analogs in Cell-Based Studies of Alzheimer’s Disease. Front. Pharmacol. 2018;9:1404. doi: 10.3389/fphar.2018.01404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Fiala M. Re-balancing of inflammation and abeta immunity as a therapeutic for Alzheimer’s disease-view from the bedside. CNS Neurol. Disord. Drug Targets. 2010;9:192–196. doi: 10.2174/187152710791012044. [DOI] [PubMed] [Google Scholar]
- 181.Mizwicki M.T., Menegaz D., Zhang J. Genomic and nongenomic signaling induced by 1α,25(OH)2-vitamin D3 promotes the recovery of amyloid-β phagocytosis by Alzheimer’s disease macrophages. J. Alzheimers Dis. 2012;29:51–62. doi: 10.3233/JAD-2012-110560. [DOI] [PubMed] [Google Scholar]
- 182.Masoumi A., Goldenson B., Ghirmai S. 1alpha, 25-dihydroxyvitamin D3 interacts with curcuminoids to stimulate amyloid-beta clearance by macrophages of Alzheimer’s disease patients. J. Alzheimers Dis. 2009;17:703–717. doi: 10.3233/JAD-2009-1080. [DOI] [PubMed] [Google Scholar]
- 183.Alamro A.A., Alsulami E.A., Almutlaq M., Alghamedi A., Alokail M., Haq S.H. Therapeutic Potential of Vitamin D and Curcumin in an In Vitro Model of Alzheimer Disease. J. Cent. Nerv. Syst. Dis. 2020;12:1179573520924311. doi: 10.1177/1179573520924311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Kizuka Y., Kitazume S., Fujinawa R., Saito T., Iwata N., Saido T.C. An aberrant sugar modification of BACE1 blocks its lysosomal targetingin Alzheimer’s disease. EMBO Mol. Med. 2015;7:175–189. doi: 10.15252/emmm.201404438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Sebollela A., Freitas-Correa L., Oliveira F.F., Paula-Lima A.C., Saraiva L.M., Martins S.M. Amyloid-beta oligomers induce differential gene expression in adult human brain slices. J. Biol. Chem. 2012;287:7436–7445. doi: 10.1074/jbc.M111.298471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Lin R. Crosstalk between Vitamin D Metabolism, VDR Signalling, and Innate Immunity. BioMed Res. Int. 2016;2016 doi: 10.1155/2016/1375858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Aggarwal B.B., Harikumar K.B. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 2009;41:40–59. doi: 10.1016/j.biocel.2008.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Chen Y., Zhang J., Ge X., Du J., Deb D.K., Li Y.C. Vitamin D receptor inhibits nuclear factor kappaB activation by interacting with IkappaB kinase beta protein. J. Biol. Chem. 2013;288:19450–19458. doi: 10.1074/jbc.M113.467670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Geldmeyer-Hilt K., Heine G., Hartmann B., Baumgrass R., Radbruch A., Worm M. 1,25-dihydroxyvitamin D3 impairs NF-kappaB activation in human naive B cells. Biochem. Biophys. Res. Commun. 2003;407:699–702. doi: 10.1016/j.bbrc.2011.03.078. [DOI] [PubMed] [Google Scholar]
- 190.Bradley-Whitman M.A., Lovell M.A. Biomarkers of lipid peroxidation in Alzheimer disease (AD): An update. Arch. Toxicol. 2015;89:1035–1044. doi: 10.1007/s00204-015-1517-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Brennan L.A., Morris G.M., Wasson G.R., Hannigan B.M., Barnett Y.A. The effect of vitamin C or vitamin E supplementation on basal and H2O2-induced DNA damage in human lymphocytes. Br. J. Nutr. 2000;84:195–202. doi: 10.1017/S0007114500001422. [DOI] [PubMed] [Google Scholar]
- 192.Banakar M.C., Paramasivan S.K., Chattopadhyay M.B. 1alpha, 25-dihydroxyvitamin D3 prevents DNA damage and restores antioxidant enzymes in rat hepatocarcinogenesis induced by diethylnitrosamine and promoted by phenobarbital. World J. Gastroenterol. 2004;10:1268–1275. doi: 10.3748/wjg.v10.i9.1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.AlJohri R., AlOkail M., Haq S.H. Neuroprotective role of vitamin D in primary neuronal cortical culture. Eneurologicalsci. 2019;14:43–48. doi: 10.1016/j.ensci.2018.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Espinet C., Gonzalo H., Fleitas C., Menal M.J., Egea J. Oxidative stress and neurodegenerative diseases: A neurotrophic approach. Curr. Drug Targets. 2015;16:20–30. doi: 10.2174/1389450116666150107153233. [DOI] [PubMed] [Google Scholar]
- 195.Dikmen M. Comparison of the Effects of Curcumin and RG108 on NGF-Induced PC-12 Adh Cell Differentiation and Neurite Outgrowth. J. Med. Food. 2017;20:376–384. doi: 10.1089/jmf.2016.3889. [DOI] [PubMed] [Google Scholar]
- 196.Orme R.P., Bhangal M.S., Fricker R.A. Calcitriol imparts neuroprotection in vitro to midbrain dopaminergic neurons by upregulating GDNF expression. PLoS ONE. 2013;8:e62040. doi: 10.1371/journal.pone.0062040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Khan S., Mas Rina Wait HJ A.H., Kaneez F.S. Effect of curcumin and vitamin D3 on learning and cognition in rat model of alzheimer’s disease. Austin J. Cerebrovasc. Dis. Stroke. 2017;4:1060. doi: 10.26420/austinjcerebrovascdisstroke.2017.1060. [DOI] [Google Scholar]