Abstract
Simple Summary
Certain chemotherapy agents (gemcitabine, platinum-based agents) have been suggested to increase the risk of venous thromboembolism in cancer patients. Our aim was to evaluate, whether treatment with these agents can be used to better predict the risk of cancer-associated venous thromboembolism. Within a prospective observational cohort study, including 1409 patients, we found that treatment with gemcitabine and/or platinum-based agents is only of limited value in predicting the risk of venous thromboembolism beyond known risk factors included in an established risk prediction model (tumor type, blood levels of D-dimer). These findings suggest that a large part of the observed rate of venous thromboembolism in patients treated with these agents might be related to the underlying thrombotic risk rather than the agent itself.
Abstract
Gemcitabine and platinum-based agents could increase the risk of venous thromboembolism (VTE) in patients with cancer. We evaluated the additive predictive utility of these agents towards cancer-associated VTE beyond a recently developed and externally validated clinical prediction model, which was based on tumor entity and continuous D-dimer levels. Analysis was performed in the derivation cohort of this model, obtained from the Vienna Cancer and Thrombosis Study (CATS), a prospective observational cohort study (n = 1409). Patients were followed for the occurrence of VTE for a maximum of two years. Competing-risk analysis was performed to obtain cumulative incidences and to conduct between-group comparisons of VTE risk. Cumulative two-year incidences of VTE were not elevated with gemcitabine treatment (10.2% vs. 7.5%, p = 0.148), whereas they were higher for platinum-based therapy (11.6% vs. 5.9%, p < 0.001). In a multivariable analysis, adjusting for tumor site category and D-dimer, gemcitabine was not associated with increased risk of VTE (subdistribution hazard ratio (SHR) 0.82, 95% confidence interval (CI) 0.53–1.28, p = 0.390), whereas platinum-based therapy predicted for a numerically increased VTE risk (SHR 1.44, 95% CI 0.96–2.17, p = 0.080). Similar results were obtained in a sensitivity analysis (updated cohort, n = 1870). Our findings suggest limited additional value of chemotherapy for the prediction of cancer-associated VTE, beyond a validated clinical prediction model.
Keywords: cancer, venous thromboembolism, chemotherapy, gemcitabine, platinum compounds
1. Introduction
Various tumor-, patient- and treatment-related factors contribute to the high risk of venous thromboembolism (VTE) in patients with cancer [1]. Among antineoplastic treatments, especially platinum-based agents and gemcitabine have been particularly implicated in increasing risk of cancer-associated thromboembolism [2,3,4]. Consequently, treatment with either platinum-based agents or gemcitabine has previously been incorporated in the PROTECHT-score, a modified version of the Khorana score for the prediction of VTE in patients with cancer [5]. The performance of this score has been evaluated in two independent cohort studies that reported c-indices of 0.59 and 0.61, indicating only moderate discrimination [6,7].
Recently, we developed and externally validated the Vienna Cancer and Thrombosis model (CATS model), a simple clinical prediction model for VTE in patients with cancer based solely on tumor entity and continuous pretherapeutic levels of D-dimer [8]. The aim of the present analysis was to evaluate platinum-based or gemcitabine therapy for the prediction of cancer-associated VTE and to explore the additive predictive value of these variables beyond those incorporated in the validated CATS score.
2. Results
2.1. Study Cohort
Among the 1409 patients included in the present analysis, 235 (17%) received gemcitabine and 501 (36%) received platinum-based therapy. One-hundred-sixty-seven (12%) patients received both gemcitabine and platinum-based therapy. Importantly, baseline clinical characteristics strongly differed between patients with and without gemcitabine or platinum-based therapy (Table 1). Patients receiving gemcitabine or platinum-based therapy had a significantly higher prevalence of high or very high VTE risk tumor types according to the classification of the CATS score (95% vs. 68% in patients treated with gemcitabine vs. no gemcitabine, respectively, and 99% vs. 58% in patients treated with platinum vs. no platinum, respectively). They also more frequently had higher histological tumor grades (G3/G4 gemcitabine vs. no gemcitabine, 43% vs. 36%, p = 0.05 and platinum vs. no platinum, 41% vs. 36%, p = 0.04), higher cancer stage (stage IV gemcitabine vs. no gemcitabine, 69% vs. 46%, p < 0.001 and platinum vs. no platinum, 67% vs. 40%, p < 0.001), and higher levels of D-dimer (median (interquartile range) gemcitabine vs. no gemcitabine, 1.2 (0.6–2.6) vs. 0.7 (0.3–1.3), p < 0.001 and platinum vs. no platinum, 1.0 (0.5–2.0) vs. 0.6 (0.3–1.2), p < 0.001). Therefore, risk of VTE in patients treated with platinum-based agents or gemcitabine was suspected to be confounded by known strong independent risk factors for cancer-associated VTE [1,9,10].
Table 1.
Baseline characteristics of study cohort.
| Variable | Overall (n = 1409) |
No Gemcitabine (n = 1174) |
Gemcitabine (n = 235) |
p 1 | No Platinum (n = 908) |
Platinum (n = 501) |
p 1 |
|---|---|---|---|---|---|---|---|
| Clinical Variables | |||||||
| Age at entry (years) | 62.9 (54.2–68·9) | 63.0 (53.6–68·9) | 62.6 (56.1–69.6) | 0.256 | 63.7 (53.9–70.4) | 61.2 (54·4–67.2) | 0.003 |
| BMI (kg/m2) | 25.1 (22.1–28.3) | 25.3 (22.5–28.5) | 24.0 (20.9–26.5) | <0.0001 | 25.4 (22.5–28.7) | 24.5 (21.6–27.3) | <0.0001 |
| Male sex | 760 (54%) | 640 (55%) | 120 (51%) | 0.333 | 472 (52%) | 288 (57%) | 0.047 |
| Tumor site | |||||||
| Low/intermediate risk of VTE | 378 (27%) | 366 (31%) | 12 (5%) | <0.0001 | 373 (41%) | 5 (1%) | <0.0001 |
| Breast | 226 (16%) | 215 (18%) | 11 (5%) | <0.0001 | 224 (25%) | 2 (0%) | <0.0001 |
| Prostate | 153 (11%) | 151 (13%) | 1 (<1%) | <0.0001 | 149 (16%) | 3 (1%) | <0.0001 |
| High Risk of VTE | 854 (61%) | 733 (62%) | 121 (51%) | <0.0001 | 467 (51%) | 387 (77%) | <0.0001 |
| Lung | 289 (21%) | 213 (18%) | 76 (32%) | <0.0001 | 88 (10%) | 201 (40%) | <0.0001 |
| Colorectal | 171 (12%) | 170 (14%) | 1 (<1%) | <0.0001 | 68 (7%) | 103 (21%) | <0.0001 |
| Kidney | 42 (3%) | 33 (3%) | 9 (4%) | 0.401 | 39 (4%) | 3 (1%) | <0.0001 |
| Lymphoma | 247 (18%) | 241 (21%) | 6 (3%) | <0.0001 | 227 (25%) | 20 (4%) | <0.0001 |
| Other sites | 105 (7%) | 76 (6%) | 29 (12%) | 0.002 | 45 (5%) | 60 (12%) | <0.0001 |
| Very high risk of VTE | 177 (13%) | 75 (6%) | 102 (43%) | <0.0001 | 68 (7%) | 109 (22%) | <0.0001 |
| Pancreas | 116 (8%) | 14 (1%) | 102 (43%) | <0.0001 | 49 (5%) | 67 (13%) | <0.0001 |
| Stomach | 61 (4%) | 61 (5%) | 0 (0%) | <0.0001 | 19 (2%) | 42 (8%) | <0.0001 |
| Tumor characteristics | |||||||
| Newly diagnosed cancer | 997 (71%) | 819 (70%) | 178 (76%) | 0.066 | 606 (67%) | 391 (78%) | <0.0001 |
| Tumor grade G3/G4 | 518 (38%) | 418 (36%) | 100 (43%) | 0.05 | 315 (36%) | 203 (41%) | 0.04 |
| Tumor stage (UICC/AnnArbor) | / | / | / | <0.0001 | / | / | <0.0001 |
| Stage I | 138 (10%) | 133 (12%) | 5 (2%) | / | 116 (14%) | 22 (4%) | / |
| Stage II | 309 (23%) | 278 (25%) | 31 (13%) | / | 264 (31%) | 45 (9%) | / |
| Stage III | 221 (16%) | 184 (17%) | 37 (16%) | / | 124 (15%) | 97 (19%) | / |
| Stage IV | 672 (50%) | 511 (46%) | 161 (69%) | / | 336 (40%) | 336 (67%) | / |
| Biomarker levels | |||||||
| D-dimer (µg/mL) | 0.7 (0.4–1.5) | 0.7 (0.3–1.3) | 1.2 (0.6–2.6) | <0.0001 | 0.6 (0.3–1.2) | 1.0 (0.5–2.0) | <0.0001 |
| VTE prediction model | |||||||
| CATS score: predicted 6-month VTE risk (%) 2 | 5.0 (3.3–6.3] | 4.8 (2.8–5.70 | 7.4 (5.2–11.0) | <0.0001 | 4.6 (2.6–5.5) | 5.8 (5.0–8.6) | <0.0001 |
| Outcomes | |||||||
| Mortality | 532 (37.8%) | 377 (32.1%) | 155 (66.0%) | / | 253 (27.9%) | 279 (55.7%) | / |
| VTE events | 111 (7.9%) | 87 (7.4%) | 24 (10.2%) | / | 53 (5.9%) | 58 (11.6%) | / |
Continuous variables are summarized by median and corresponding interquartile range and categorical data by absolute frequency and percentages. 1 p-Value from rank-sum test or Chi-square test (as appropriate) for the comparison of baseline variables between gemcitabine vs. no gemcitabine and platinum vs. no platinum therapy. 2 Based on tumor site risk category and continuous levels of D-dimer. BMI, body mass index; UICC, Union for International Cancer Control; VTE, venous thromboembolism.
2.2. Crude VTE Incidence According to Chemotherapeutic Agent
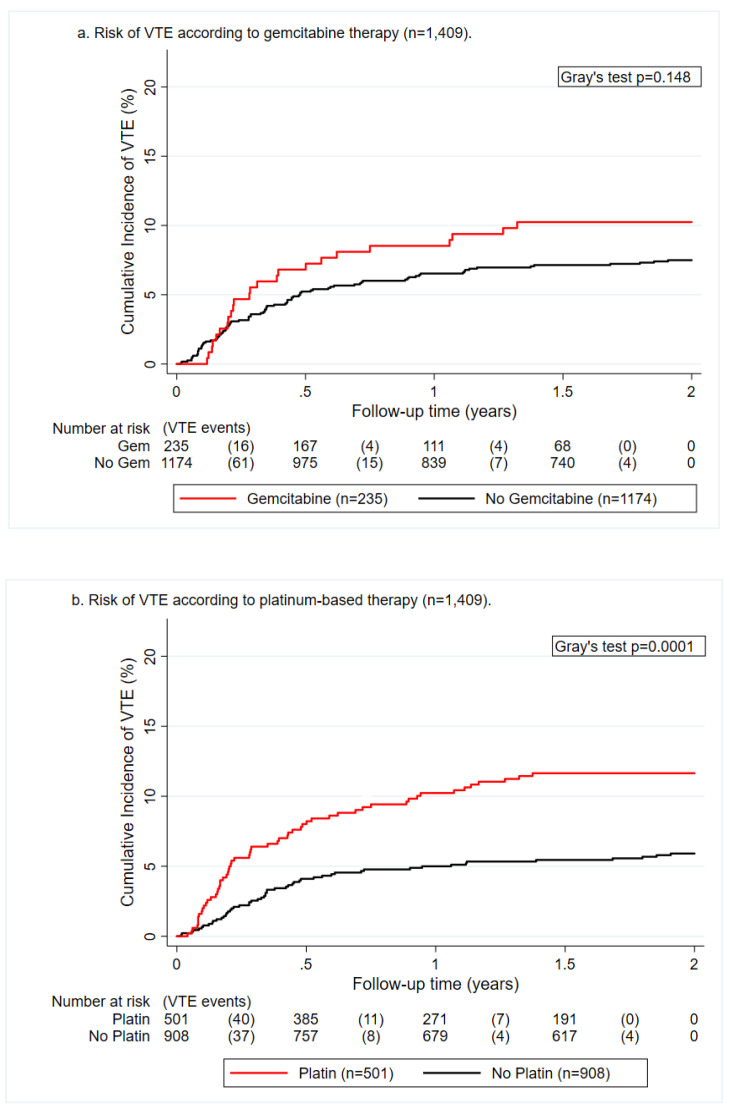
Over a median follow-up of 24 months, we observed 111 patients with VTE. Crude two-year cumulative VTE incidences did not significantly differ between patients treated with gemcitabine as compared with those without gemcitabine (10.2%, 95% confidence interval (CI) 6.8–14.5 vs. 7.5%, 95% CI 6.1–9.1, Gray’s test, p = 0.148, Figure 1a). The cumulative incidence of VTE at 2 years was significantly higher in patients undergoing platinum-based therapy as compared with those without platinum therapy (11.6%, 95% CI 9.0–14.6 vs. 5.9%, 95% CI 4.5–7.6, p < 0.001, Figure 1b). Patients treated with both platinum-based agents and gemcitabine during the study period had a VTE risk that was comparable to the remainder of the patients (9.0%, 95% CI 5.3–14.0 vs. 7.8%, 95% CI 6.4–9.4, p = 0.593).
Figure 1.
Cumulative incidence function of venous thromboembolism (VTE) risk according to gemcitabine (a) and platinum-based chemotherapy (b).
2.3. Prediction of Cancer-Associated Thrombosis Beyond the CATS Score
Gemcitabine was not associated with risk of VTE in the univariable analysis (SHR 1.39, 95% CI 0.88–2.18, p = 0.156). This association remained similar upon multivariable adjustment for tumor type (SHR 0.83, 95% CI 0.53–1.31, p = 0.432), D-dimer (SHR 1.18, 95% CI 0.75–1.85, p = 0.481), and tumor type and D-dimer (SHR 0.82, 95% CI 0.53–1.28, p = 0.390, Model #1–3 in Table 2). These results suggest a lack of independent predictive utility of gemcitabine therapy towards cancer-associated VTE beyond tumor type or levels of D-dimer.
Table 2.
Baseline characteristics of study cohort.
| Model | Variable | SHR | 95%CI | p |
|---|---|---|---|---|
| #1 | Gemcitabine | 0.83 | 0.53–1.31 | 0.432 |
| Tumor type: Low/moderate VTE risk | Ref. | Ref. | Ref. | |
| Tumor type: High VTE risk | 2.48 | 1.37–4.48 | 0.003 | |
| Tumor type: Very high VTE risk | 5.48 | 2.82–10.68 | <0.0001 | |
| #2 | Gemcitabine | 1.18 | 0.75–1.85 | 0.481 |
| D-dimer (per doubling) | 1.44 | 1.25–1.66 | <0.0001 | |
| #3 | Gemcitabine | 0.82 | 0.53–1.28 | 0.390 |
| Tumor type: Low/moderate VTE risk | Ref. | Ref. | Ref. | |
| Tumor type: High VTE risk | 2.26 | 1.24–4.10 | 0.008 | |
| Tumor type: Very high VTE risk | 4.27 | 2.10–8.66 | <0.0001 | |
| D-dimer (per doubling) | 1.31 | 1.11–1.53 | 0.001 | |
| #4 | Platinum-based therapy | 1.46 | 0.97–2.21 | 0.073 |
| Tumor type: Low/moderate VTE risk | Ref. | Ref. | Ref. | |
| Tumor type: High VTE risk | 2.03 | 1.09–3.77 | 0.025 | |
| Tumor type: Very high VTE risk | 3.89 | 1.87–8.11 | <0.001 | |
| #5 | Platinum-based therapy | 1.84 | 1.26–2.69 | 0.002 |
| D-dimer (per doubling) | 1.40 | 1.20–1.63 | <0.0001 | |
| #6 | Platinum-based therapy | 1.44 | 0.96–2.17 | 0.080 |
| Tumor type: Low/moderate VTE risk | Ref. | Ref. | Ref. | |
| Tumor type: High VTE risk | 1.85 | 0.99–3.46 | 0.055 | |
| Tumor type: Very high VTE risk | 3.08 | 1.45–6.57 | 0.004 | |
| D-dimer (per doubling) | 1.31 | 1.1–1.53 | 0.001 |
Multivariable competing risk models of gemcitabine therapy, platinum-based therapy, tumor type, and D-dimer for prediction of VTE in patients with cancer (n = 1409). 95% CI, 95% confidence interval; p, Wald test p-value; Ref., reference category; SHR, subdistribution hazard ratio; VTE, venous thromboembolism.
We observed a strong univariable association between platinum-based therapy and risk of VTE (SHR 2.04, 95% CI 1.41–2.97, p < 0.001). Magnitude and strength of association were weakened upon multivariable adjustment for tumor type (SHR 1.46, 95% CI 0.97–2.21, p = 0.073), D-dimer (SHR 1.84, 95% CI 1.26–2.69, p = 0.002), and both tumor type and d-dimer (SHR 1.44, 95% CI 0.96–2.17, p = 0.080, Model #4–6 in Table 2). These findings suggest a potential weak association between platinum-based therapy and risk of cancer-associated VTE beyond tumor type or levels of D-dimer.
2.4. Sensitivity Analysis
In order to further explore the putative association of therapy with platinum-based agents or gemcitabine with VTE, a sensitivity analysis was performed with an updated cohort of patients recruited within the Vienna Cancer and Thrombosis study (n = 1870). Consistent with the main analysis, therapy with gemcitabine was not associated with risk of VTE in the univariable analysis (SHR 1.17, 95% CI 0.76–1.80, p = 0.478). Therapy with platinum-based agents was associated with higher VTE risk in the univariable analysis (SHR 1.50, 95% CI 1.09–2.07, p = 0.014). This association was weakened upon adjustment for tumor site category and D-dimer levels in the multivariable analysis (SHR 1.21, 0.86–1.70, p = 0.280).
3. Discussion
The present findings suggest that platinum-based therapy and gemcitabine do not significantly improve prediction of VTE in patients with cancer beyond an established risk prediction model. Univariable analyses confirmed a relative increase in risk of VTE with platinum-based therapy and gemcitabine as reported previously [2,4]. This concept is further supported by data on vascular toxicity of these agents from preclinical work and clinical observations [11]. Platinum-based agents have been shown to induce endothelial cell dysfunction, including proinflammatory changes and an increased expression of cell adhesion molecules, whereas preclinical data on vascular and hemostatic effects of gemcitabine remain scarce and inconclusive [12,13,14,15].
Nonetheless, our findings suggest that the high burden of VTE in patients treated with platinum agents or gemcitabine are possibly not be fully causal, but to a large degree be mediated by underlying prothrombotic risk factors that are correlated with treatment with these agents. For example, gemcitabine is one mainstay of treatment for pancreatic cancer, and platinum-based agents are a treatment standard for gastric cancer, and both these tumor entities are associated with the highest known risk of VTE [16,17].
A limitation of our analysis is that treatment with platinum-based agents and gemcitabine was not analyzed as a time-dependent variable and applied cumulative doses were not considered. Although time-dependent and dose-specific analyses could have yielded a more refined appreciation of the association between these treatments and cancer-associated VTE risk, time-dependent variables are not useful for clinical prediction purposes as their information only becomes available over time, and thus cannot be used for clinical decision making.
4. Methods
This analysis was conducted within a prospective, observational cohort study, the Vienna Cancer and Thrombosis Study (CATS). The design, procedures, as well as inclusion and exclusion criteria have been previously reported in detail [18], and the study was approved by the Ethics Committee of the Medical University of Vienna (Project identification code: 126/2003, date of approval: 2 September 2003). Briefly, patients with newly diagnosed cancer or recurrent or progressive disease after remission were eligible for inclusion. Patients were followed prospectively for the occurrence of objectively confirmed, independently adjudicated VTE for a maximum of 2 years.
We used the same dataset as the CATS model derivation study (n = 1423) [8], with risk of VTE stratified according to tumor type, i.e., very high risk (pancreas and gastric), high risk (lung, colorectal, oesophagus, kidney, lymphoma, bladder/urothelial, uterus, cervical, ovarian, and others), and intermediate/low risk (breast and prostate). D-dimer levels were obtained from blood withdrawn at study inclusion and measured with the STA-Liatest assay (Diagnostica-Stago, Asnières, France). Data on chemotherapy during the 2-year observation period were obtained from the in-house pharmacy prescription records, with missing chemotherapy data in 14 patients (analysis cohort, n = 1409).
Statistical Analysis
Baseline differences between groups were analyzed by means of the rank sum test or Chi-square test, as appropriate. Between-group differences in risk of VTE were assessed with competing risk cumulative incidence estimators and Fine and Gray proportional subdistribution hazards regression models [19,20]. A sensitivity analysis was conducted with the most recent dataset available from CATS, consisting of 1870 patients.
5. Conclusions
Therapy with gemcitabine was not associated with an increased risk of VTE. Platinum-based therapy could be a weak independent risk factor for cancer-associated VTE, with only limited predictive value beyond tumor site category and D-dimer levels. These findings do not support the use of these variables in clinical prediction models for VTE in patients with cancer.
Acknowledgments
Open Access Funding by the Austrian Science Fund (FWF).
Author Contributions
Conceptualization, data curation, formal analysis, methodology, writing—original draft, F.M.; Conceptualization, supervision, Writing—review and editing, N.v.E.; Conceptualization, formal analysis, methodology, writing—review and editing, F.P.; Writing—review and editing, M.D.N.; Writing—review and editing, T.F.; Writing—review and editing, M.P.; Project administration, supervision, writing—review and editing, I.P.; Conceptualization, project administration, supervision, writing—original draft, C.A. All authors have read and approved the submission of the manuscript.
Funding
This work was supported by the Anniversary Fund of the Austrian National Bank (grant number 17828) and the Austrian Science Fund (FWF) Special Research Program (SFB) 54.
Conflicts of Interest
F.M. reports no potential conflict of interest. N.v.E. has received advisory board honoraria from LEO Pharma, Daiichi Sankyo, and Bayer, that were transferred to his institute. F.P. reports no potential conflict of interest. M.D.N. reports personal fees from Bayer, Daiichi Sankyo, BMS-Pfizer, Leo Pharma, Sanofi, and Aspen, outside the submitted work. T.F. reports honoraria from MSD; Merck Darmstadt, Roche, BMS, Accord; Sanofi and Boehringer Ingelheim and advisory board participation for MSD; Merck Darmstadt, Amgen, Pfizer, and Sanofi. M.P. has received honoraria for lectures, consultation, or advisory board participation from the following for-profit companies: Bayer, Bristol-Myers Squibb, Novartis, Gerson Lehrman Group (GLG), CMC Contrast, GlaxoSmithKline, Mundipharma, Roche, BMJ Journals, MedMedia, Astra Zeneca, AbbVie, Lilly, Medahead, Daiichi Sankyo, Sanofi, Merck Sharp & Dome, and Tocagen. The following for-profit companies have supported clinical trials and contracted research conducted by M.P. with payments made to his institution: Böhringer-Ingelheim, Bristol-Myers Squibb, Roche, Daiichi Sankyo, Merck Sharp & Dome, Novocure, GlaxoSmithKline, AbbVie. I. Pabinger reports occasional honoraria for lectures and advisory board meetings from Bayer AG and Pfizer outside the submitted work. Cihan Ay reports honoraria for lectures from Bayer, Daiichi-Sankyo, BMS/Pfizer, and Sanofi and participation in advisory boards from Bayer, Boehringer-Ingelheim, Daiichi-Sankyo, and BMS/Pfizer outside the submitted work. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- 1.Ay C., Pabinger I., Cohen A.T. Cancer-associated venous thromboembolism: Burden, mechanisms, and management. Thromb. Haemost. 2017;117:219–230. doi: 10.1160/TH16-08-0615. [DOI] [PubMed] [Google Scholar]
- 2.Seng S., Liu Z., Chiu S.K., Proverbs-Singh T., Sonpavde G., Choueiri T.K., Tsao C.-K., Yu M., Hahn N.M., Oh W.K., et al. Risk of venous thromboembolism in patients with cancer treated with Cisplatin: A systematic review and meta-analysis. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012;30:4416–4426. doi: 10.1200/JCO.2012.42.4358. [DOI] [PubMed] [Google Scholar]
- 3.Moore R.A., Adel N., Riedel E., Bhutani M., Feldman D.R., Tabbara N.E., Soff G.A., Parameswaran R., Hassoun H. High incidence of thromboembolic events in patients treated with cisplatin-based chemotherapy: A large retrospective analysis. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2011;29:3466–3473. doi: 10.1200/JCO.2011.35.5669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Qi W.-X., Lin F., Sun Y.-J., Tang L.-N., Shen Z., Yao Y. Risk of venous and arterial thromboembolic events in cancer patients treated with gemcitabine: A systematic review and meta-analysis. Br. J. Clin. Pharmacol. 2013;76:338–347. doi: 10.1111/bcp.12203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Verso M., Agnelli G., Barni S., Gasparini G., Labianca R. A modified Khorana risk assessment score for venous thromboembolism in cancer patients receiving chemotherapy: The Protecht score. Intern. Emerg. Med. 2012;7:291–292. doi: 10.1007/s11739-012-0784-y. [DOI] [PubMed] [Google Scholar]
- 6.Di Nisio M., Van Es N., Rotunno L., Anzoletti N., Falcone L., De Tursi M., Natoli C., Tinari N., Cavallo I., Valeriani E., et al. Long-term performance of risk scores for venous thromboembolism in ambulatory cancer patients. J. Thromb. Thrombolysis. 2019;48:125–133. doi: 10.1007/s11239-019-01845-6. [DOI] [PubMed] [Google Scholar]
- 7.Van Es N., Di Nisio M., Cesarman G., Kleinjan A., Otten H.-M., Mahé I., Wilts I.T., Twint D.C., Porreca E., Arrieta O., et al. Comparison of risk prediction scores for venous thromboembolism in cancer patients: A prospective cohort study. Haematologica. 2017;102:1494–1501. doi: 10.3324/haematol.2017.169060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pabinger I., Van Es N., Heinze G., Posch F., Riedl J., Reitter E.-M., Di Nisio M., Cesarman-Maus G., Kraaijpoel N., Zielinski C.C., et al. A clinical prediction model for cancer-associated venous thromboembolism: A development and validation study in two independent prospective cohorts. Lancet Haematol. 2018;5:e289–e298. doi: 10.1016/S2352-3026(18)30063-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ahlbrecht J., Dickmann B., Ay C., Dunkler D., Thaler J., Schmidinger M., Quehenberger P., Haitel A., Zielinski C., Pabinger I. Tumor grade is associated with venous thromboembolism in patients with cancer: Results from the Vienna Cancer and Thrombosis Study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012;30:3870–3875. doi: 10.1200/JCO.2011.40.1810. [DOI] [PubMed] [Google Scholar]
- 10.Ay C., Vormittag R., Dunkler D., Simanek R., Chiriac A.-L., Drach J., Quehenberger P., Wagner O., Zielinski C., Pabinger I. D-dimer and prothrombin fragment 1 + 2 predict venous thromboembolism in patients with cancer: Results from the Vienna Cancer and Thrombosis Study. J. Clin. Oncol. 2009;27:4124–4129. doi: 10.1200/JCO.2008.21.7752. [DOI] [PubMed] [Google Scholar]
- 11.Herrmann J., Yang E.H., Iliescu C.A., Cilingiroglu M., Charitakis K., Hakeem A., Toutouzas K., Leesar M.A., Grines C.L., Marmagkiolis K. Vascular toxicities of cancer therapies. Circulation. 2016;133:1272–1289. doi: 10.1161/CIRCULATIONAHA.115.018347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yu M., Han J., Cui P., Dai M., Li H., Zhang J., Xiu R. Cisplatin up-regulates ICAM-1 expression in endothelial cell via a NF-κB dependent pathway. Cancer Sci. 2008;99:391–397. doi: 10.1111/j.1349-7006.2008.00696.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shi Y., Inoue S., Shinozaki R., Fukue K., Kougo T. Release of cytokines from human umbilical vein endothelial cells treated with platinum compounds in vitro. Jpn. J. Cancer Res. 1998;89:757–767. doi: 10.1111/j.1349-7006.1998.tb03281.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mikuła-Pietrasik J., Witucka A., Pakuła M., Uruski P., Begier-Krasińska B., Niklas A., Tykarski A., Książek A. Comprehensive review on how platinum- and taxane-based chemotherapy of ovarian cancer affects biology of normal cells. Cell. Mol. Life Sci. 2018;76:681–697. doi: 10.1007/s00018-018-2954-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dasanu C.A. Gemcitabine: Vascular toxicity and prothrombotic potential. Expert Opin. Drug Saf. 2008;7:703–716. doi: 10.1517/14740330802374262. [DOI] [PubMed] [Google Scholar]
- 16.Bang Y.-J., Van Cutsem E., Feyereislova A., Chung H.C., Shen L., Sawaki A., Lordick F., Ohtsu A., Omuro Y., Satoh T., et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet (Lond. Engl.) 2010;376:687–697. doi: 10.1016/S0140-6736(10)61121-X. [DOI] [PubMed] [Google Scholar]
- 17.Von Hoff D.D., Ervin T., Arena F.P., Chiorean E.G., Infante J., Moore M., Seay T., Tjulandin S.A., Ma W.W., Saleh M.N., et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013;369:1691–1703. doi: 10.1056/NEJMoa1304369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ay C., Simanek R., Vormittag R., Dunkler D., Alguel G., Koder S., Kornek G., Marosi C., Wagner O., Zielinski C., et al. High plasma levels of soluble P-selectin are predictive of venous thromboembolism in cancer patients: Results from the Vienna Cancer and Thrombosis Study (CATS) Blood. 2008;112:2703. doi: 10.1182/blood-2008-02-142422. [DOI] [PubMed] [Google Scholar]
- 19.Ay C., Posch F., Kaider A., Zielinski C., Pabinger I. Estimating risk of venous thromboembolism in patients with cancer in the presence of competing mortality. J. Thromb. Haemost. 2015;13:390–397. doi: 10.1111/jth.12825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fine J.P., Gray R.J. A proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc. 1999;94:496–509. doi: 10.1080/01621459.1999.10474144. [DOI] [Google Scholar]