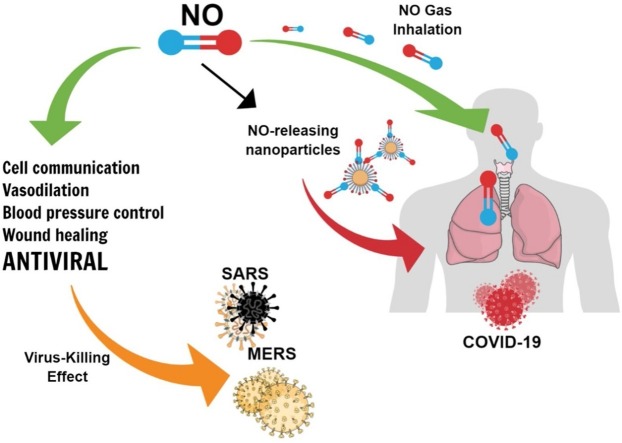
Graphical abstract
Abbreviations: CuNPs, Copper Nanoparticles; IDO, Indoleamine 2,3–dioxygenase; iNOS, Inducible NO Synthase; NO, Nitric Oxide; NOx, Nitrogen Reactive Species; ONOO−, Peroxynitrite; RNA, Ribonucleic acid; SARS-CoV-2, Severe Acute Respiratory Syndrome Corona Virus 2; AgNPs, Silver Nanoparticles; SNAP, S-nitroso-N-acetylpenicillamine; O2•−, Superoxide; UVA, Ultraviolet radiation A; FDA, US Food and Drug Administration; WHO, World Health Organization
Keywords: Inhaled nitric oxide, COVID-19, SARS-CoV, Nanoparticles, Antiviral
Abstract
The endogenous free radical nitric oxide (NO) plays a pivotal role in the immunological system. NO has already been reported as a potential candidate for use in the treatment of human coronavirus infections, including COVID-19. In fact, inhaled NO has been used in clinical settings for its antiviral respiratory action, and in the regulation of blood pressure to avoid clot formation. In this mini-review, we discuss recent progress concerning the antivirus activity of NO in clinical, pre-clinical and research settings, and its beneficial effects in the treatment of clinical complications in patients infected with coronaviruses and other respiratory viral diseases, including COVID-19. We also highlight promising therapeutic effects of NO donors allied to nanomaterials to combat COVID-19 and other human coronavirus infections. Nanomaterials can be designed to deliver sustained, localized NO release directly at the desired application site, enhancing the beneficial effects of NO and minimizing the side effects. Challenges and perspectives are presented to open new fields of research.
1. Introduction
Severe acute respiratory syndrome corona virus 2 (SARS-CoV-2, or 2019-nCoV) was first identified and reported in Wuhan, China, in December 2019, and rapidly spread to over 210 countries and territories (Keni et al., 2020; Lai et al., 2020). At the time of writing, September 2020, the World Health Organization (WHO) has confirmed 32 million cases in all 5 regions of the world, and over 1 million deaths. SARS-CoV-2 is transmitted human-to-human by direct contact or droplets, leading to an infection with 6.4-day incubation and 6.8 % mortality, calculated using recent data (Lai et al., 2020). Therefore, a rush in the study and development of possible treatments and vaccines has been observed from numerous scientists using different approaches (Martinez, 2020). A promising strategy under consideration is the use of inhalation-based therapies, including inhaled nitric oxide (NO) (Gianni et al., 2020a). This short communication aims to highlight important impacts of NO application, reporting the effectiveness of NO against coronaviruses, including SARS-CoV-2, and evidencing promising strategies to leverage this research field.
NO is a free radical gas that plays key roles in biological systems, as first demonstrated by the winners of the Nobel Prize for Medicine and Physiology in 1998, Robert F. Furchgott, Louis J. Ignarro and Ferid Murad (Ignarro, 1999). Despite being involved in processes such as cell communication (O’Toole et al., 2016), vasodilation, control of blood pressure (Seabra et al., 2015), and wound healing (Seabra, 2017), NO presents important antimicrobial and antitumoral properties, depending on its concentration (Hasan et al., 2017; Pieretti et al., 2019). In 1988, NO was related to soft tissue relaxation in the penis, allowing the organ to become engorged. Eight years later, Viagra came to the U.S. market (Gur et al., 2017).
Due to its small size, neutral charge, and relative lipophilicity, NO diffusion through cell membranes is facilitated even in the absence of channels or receptors (Dioguardi, 2011). NO action relies on certain main mechanisms, such as impairment of pathogen replication, interference in the electron transport chain, promotion of S-nitrosation reactions in cysteine residues of important pathogen enzymes, and generation of oxygen and nitrogen reactive species (NOx) (Liu et al., 2017; Seabra et al., 2016). Moreover, NO quickly reacts with superoxide (O2 • −), leading to highly toxic and oxidant peroxynitrite (ONOO−) (Pacher et al., 2008).
NO has been widely studied for combating tumor cells, bacteria, fungi, protozoa, and viruses (Rolim et al., 2019; Santiago-Olivares et al., 2019; Schairer et al., 2012). Inhaled treatments of gaseous NO have been proposed as an efficient route for administering the gas, but systemic administration is also possible using NO donors, which are molecules able to release NO in the biological medium, and/or by allying NO and NO donors with nanomaterials (Quinn et al., 2015). The use of NO donors and nanomaterials increases the stability of NO and enables targeted, controlled NO release at desirable concentrations, which is fundamental for NO bioactivity (Quinn et al., 2015). There are several combinations of NO and/or NO donors with nanoparticles, which can be designed for specific applications (Seabra and Durán, 2017).
There are important recent review articles based on the potential use of NO to combat COVID-19 and other human coronavirus infections (Adusumillia et al., 2020; Hedenstierna et al., 2020; Martel et al., 2020). To the best of our knowledge, these review articles are mainly focused on the administration of gaseous NO (inhaled NO), on discussion of the pathogenesis of viral infections, and on inflammatory responses modulated by NO in vivo. The present review focuses not only on the use of inhaled NO but also on the administration of NO donors in combination with nanomaterials. NO-releasing nanomaterials have been extensively studied in different biomedical applications, including their antimicrobial effects. Due to the versatility of nanomaterials, NO donors allied to a nanoplatform might be important application in the fight against COVID-19 and other human coronavirus infections, and we hope that this review will open the way to new studies in this area.
2. NO potential against human coronaviruses and respiratory viral diseases
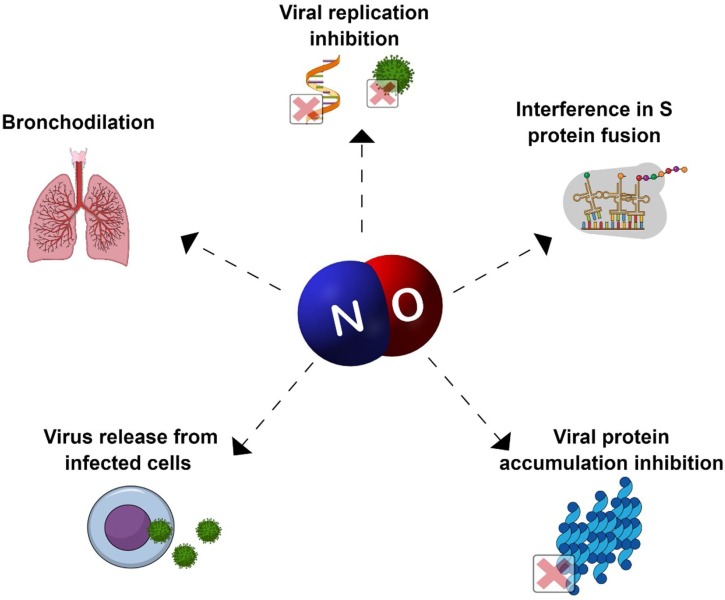
The antimicrobial properties of NO usually rely on antifungal and antibacterial effects, although it is well-known that NO not only presents antiviral activity, but also acts to modulate host response (Akaike and Maeda, 2000). NO has already been reported as a potential candidate for the treatment against coronaviruses and other respiratory viral diseases, such as influenza (Arabi et al., 2015; Keyaerts et al., 2004; Murphy et al., 1998). Fig. 1 shows an overview of NO antiviral activity previously reported against different viral diseases (Akaike and Maeda, 2000; Akerstrom et al., 2005; Arabi et al., 2015; Lin et al., 1997).
Fig. 1.
Diagram of NO actions against viral infections.
To date, seven coronavirus strains have been identified: HCoV-229E, SARS-CoV, HCoV−OC43, HCoV-NL63, HCoV-HKU1, MERS-CoV and SARS-CoV-2 (Kiyuka et al., 2018; Sanchez-Nadales et al., 2020). Of these, NO has been used against MERS-CoV, SARS-CoV and HCoV-NL63. More recently, it has been extensively evaluated against SARS-CoV-2 (Arabi et al., 2015; Keyaerts et al., 2004; Sanchez-Nadales et al., 2020; Tavazzi et al., 2020). Few reports demonstrate the use of NO against MERS-CoV (Al-Dorzi et al., 2016; Arabi et al., 2015) and this topic should be further explored. Inhaled NO treatment was administered in combination with traditional therapies, such as antibiotics, to a hypertense male patient who tested positive for MERS-CoV (Arabi et al., 2015). Treatments allied to inhaled NO demonstrated positive results, and the patient was discharged to his home 21 days from the beginning of NO treatment (Arabi et al., 2015). Moreover, inhaled NO was administered for MERS-CoV in infected healthcare workers and the results also demonstrated that the patients were successfully healed, evidencing that NO might have positive effects in the treatment with low harmful side effects (Al-Dorzi et al., 2016). More recently, inhaled NO treatment was also employed in a 22-year old immunocompromised patient who tested positive for both SARS-CoV-2 and HCoV-NL63 (Sanchez-Nadales et al., 2020). The authors have hypothesized that NO might have played a pivotal role in the treatment, related to the loss of alveolar perfusion and the hypoxic pulmonary vasoconstriction effect (Sanchez-Nadales et al., 2020). Taken together, these reports suggest beneficial effects of administration of NO in patients, and further studies are required.
In addition to the few publications reporting NO trials in the treatment of coronaviruses, this molecule has already been evaluated against other respiratory viral diseases, such as influenza A/H1N1 (Avnon et al., 2015; Kovačević et al., 2015; Murphy et al., 1998). During the 2009 H1N1 pandemic, NO was used in combination with mechanical ventilation in 67 % of the patients under experimental trial, particularly patients with thrombotic complications. In general, authors indicated that critically ill patients were administered deep vein thrombosis prophylaxis in combination with other treatments, such as inhaled NO (Avnon et al., 2015). Interestingly, considering that NO is an endogenous signaling molecule, it has also been reported that serum NO levels are higher in patients that survived acute respiratory distress syndrome caused by H1N1, in comparison with non survivors, as well as an increase of oral NO levels after 21-days of virus incubation; this offers a strong association of NO levels with improved clinical outcomes (Avnon et al., 2015; Murphy et al., 1998). NO can react with important biomolecules controlling physiological and pathophysiological functions. Indeed, NO can nitrosate thiol groups on the surface of red blood cells and the beta chain of hemoglobin, preventing hemolysis and oxidative damage (Zhang et al., 2015). Previous studies have suggested the potential beneficial effects of NO, opening new areas in this research topic.
Although positive biological effects have been reported for the administration of NO donors, further studies are required to better evaluate the levels of inflammatory mediators and the activity of important heme-containing enzymes, such as indoleamine 2,3–dioxygenase (IDO), directly involved in the inflammatory responses in respiratory viral infections (Anderson and Reiter, 2020). During virus infection, a significant increase in the levels of inflammatory mediators can be observed, known as a “cytokine storm”, associated with endothelial and platelet activation; this can lead to vascular permeability, thrombocytopenia, shock (observed in severe cases), pulmonary hypertension and pneumonia (Belladona and Orabona, 2020). IDO and inducible nitric oxide synthase (iNOS) are expressed upon monocyte activation. It has been reported that NO generated by iNOS inhibited the SARS CoV replication cycle (Akerstrom et al., 2005). Interferon-gamma, a molecule with antiviral activity and a regulator of inflammatory responses to pathogens, is a potent inducer of IDO and iNOS (Kang et al., 2018), in addition to other inflammatory factors, such as tumor necrosis factor-α (TNFα) and interleukins 6 and 1β (Turski et al., 2020). For instance, both these enzymes were highly expressed in monocytes of patients infected with dengue virus (DENV-4), compared to healthy controls, during an epidemic in Campo Grande (MS, Brazil) (Fialho et al., 2017). Inflammatory processes and cytokines stimulate iNOS, which in turn enhance NO production against virus infection. IDO, induced by inflammation or immune responses, participates in tryptophan catabolism and can be induced by IFN-γ in monocytes, displaying immunosuppressive and antimicrobial activities (Mellor and Munn, 2004; Hara et al., 2008). IDO enzymatic activity helps to control pulmonary inflammation and hypertension, allowing immunoregulation in COVID-19 patients (Belladona and Orabona, 2020). It should be noted that IDO activity can be either suppressed by NO or induced by NOS inhibition (Hara et al., 2008). IDO is a cytosolic immune-modulatory enzyme containing heme iron for which NO has a high affinity, thus endogenous NO and NO donors can inactivate this enzyme (Thomas et al., 1994). Some papers reported that blockage of IDO activity might enhance antiviral IFN-I/II (interferon type I/II) innate and adaptive T-cell responses in Japanese encephalitis (Kim et al., 2016) and respiratory viral infections (van Wissen et al., 2002). As IDO and NO have been implicated in T cell immune tolerance (Ye et al., 2017), the effects of NO donors on IDO and on cytokine production should be further investigated.
2.1. NO potential against SARS-CoV and SARS-CoV-2
NO inhaled treatment against COVID has already been proposed in 2020 in two different countries, China and United States of America (Berra et al., 2020; Gianni et al., 2020a; Lei et al., 2020). The motivation comes from the historical use of NO-based treatment against SARS-CoV, and also against other respiratory diseases caused by viruses. After the SARS-CoV outbreak in 2002, three different studies demonstrated the potential of NO compounds in the inhibition of SARS-CoV replication (Akerstrom et al., 2005; Åkerström et al., 2009; Keyaerts et al., 2004). In a study that overviewed therapeutic strategies available in 2008 for SARS-CoV treatment, NO was highlighted as a potential drug already available and used on infected humans, while NO donors were reported as promising drugs undergoing pre-clinical trials (Wong and Yuen, 2008). in vitro studies performed with the NO donor S-nitroso-N-acetylpenicillamine (SNAP) indicated that NO was able to inhibit the replication cycle of SARS-CoV in a concentration-dependent mechanism. Moreover, the same experiment showed that viral protein and RNA synthesis were also hindered (Åkerström et al., 2009). Years later, in-depth studies of the same research group into the mechanism of NO against SARS-CoV evidenced that the intermediate peroxynitrite was not responsible for the replication cycle inhibition; the authors demonstrated that the antivirus mechanism relied on two major points: (i) interference in the fusion of S protein and its receptor due to reduced palmitoylation of the S protein, and (ii) a reduction in RNA production, probably due to an effect on cysteine proteases (Akerstrom et al., 2005). A similar pattern was reported by Keyaerts and coworkers, evidencing an IC50 of SNAP against SARS-CoV of 222.3 ± 83.7 μM, and no activity for the non-nitrosated molecule, confirming that the effect relies on NO release (Keyaerts et al., 2004). Moreover, NO produced from inducible NO synthase (iNOS) has also demonstrated the ability to inhibit the replication cycle of SARS-CoV (Åkerström et al., 2009).
2.2. Current use of NO against COVID-19
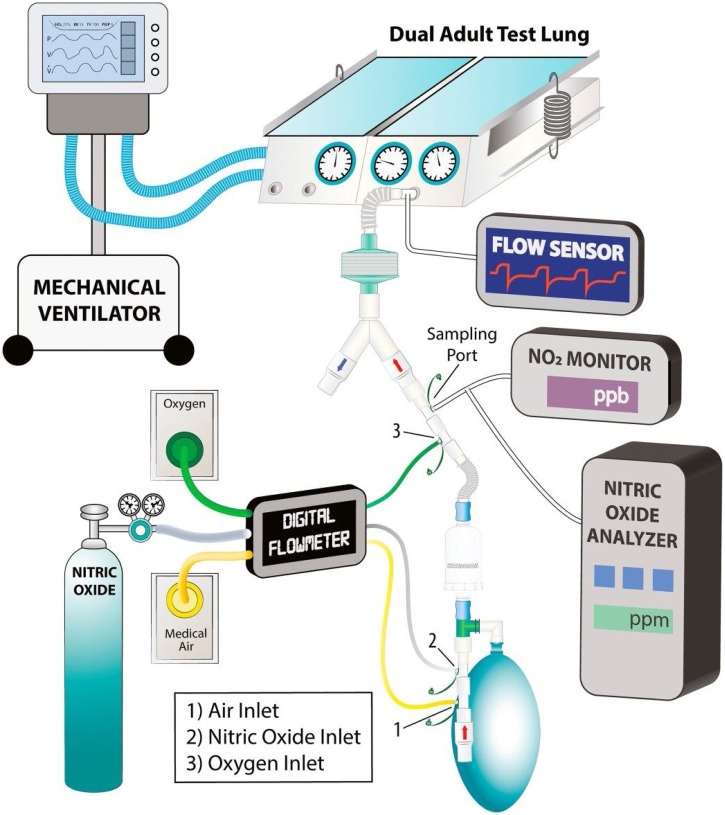
Thus, the protocols published to date against SARS-CoV-2 rely on the efficient results previously demonstrated. Three different strategies were proposed to comprehend the treatment under different conditions, using a randomized experiment evolving 2 centers: Boston and Xi’an (Berra et al., 2020; Gianni et al., 2020a; Lei et al., 2020). Firstly, NO application in severely ill patients is proposed in two different conditions: (i) high doses of gaseous NO, up to 180 ppm, during short periods of time (20−30 min), and (ii) low doses of gaseous NO, up to 80 ppm, during 48 h-treatment (Berra et al., 2020; Lei et al., 2020). Interestingly, a second protocol proposed inhaled NO to prevent COVID-19 in healthcare providers who are in direct contact with patients infected with SARS-CoV-2 (Gianni et al., 2020a). The individuals concerned will receive a maximum of 160 ppm of inhaled NO for a short period of time during 14 days. It is expected that the number of healthcare providers contaminated will decrease from 15 % to 5% (Gianni et al., 2020a). The three strategies proposed have different parameters for the treatment period, concentration and people involved; they promise to increase knowledge about the use of NO against COVID-19 in both treatment and prevention. Although a promising strategy, the inhalation of gaseous NO is not the only possible treatment and/or prevention against COVID-19 or future viral diseases. Interestingly, the same authors that first proposed the use of inhaled NO developed a system for high dose NO inhalation, minimizing NO concentration oscillations and the inhalation of NO2, which might have a promising application in this field (Gianni et al., 2020b) (Fig. 2 ).
Fig. 2.
Diagram of the experimental setup proposed for the use of inhaled NO. The mechanical ventilator connected to the right lung acts as a diaphragm of the left lung. The left lung is connected with the tested delivery system. Reproduced from reference Gianni et al., 2020b with permission from Elsevier.
Inhaled NO trials against COVID-19 have also been reported in other conditions. The trials have varied mostly in the patients’ condition: (i) breathing patients in non-severe condition and (ii) sedated and ventilated patients in severe condition. A trial carried out in Italy evaluated the efficiency of inhaled NO in ten patients in severe conditions (Ferrari et al., 2020). The dose evaluated was 20 ppm for 30 min, in patients already treated with invasive mechanical ventilation (Ferrari et al., 2020). Although NO acts by improving arterial oxygenation, the same pattern was not observed in all patients, especially those with severe hypoxemia. This highlights the fact that even though NO presents promising use in the treatment of patients with COVID-19, further studies must be performed to understand which patients might benefit (Ferrari et al., 2020). Interestingly, a similar trial employing inhaled NO at a concentration of 25 ppm led to complementary results (Tavazzi et al., 2020). The treatment did not improve oxygenation in patients with refractory hypoxemia, although promising results were confirmed for patients with right ventricular dysfunction (Tavazzi et al., 2020).
A different pattern was observed in spontaneously breathing patients. Parikh and coworkers evaluated the potential of inhaled NO treatment in breathing patients diagnosed with COVID-19 (Parikh et al., 2020). Thirty-nine patients with different characteristics, with or without pre-existent diseases, received a dosage of 30 ppm of NO for 2 days. Of all the patients treated with inhaled NO, 53.9 % did not require mechanical ventilation and the SpO2/FiO2 ratio also improved in those patients (Parikh et al., 2020). This suggests that NO might not only prevent hypoxic respiratory failure, but also contribute an antiviral mechanism (Parikh et al., 2020). A higher dose of NO (200 ppm/30 min) was evaluated in pregnant women diagnosed with COVID-19 (Safaee Fakhr et al., 2020). In this trial, six pregnant women received 39 treatments with inhaled NO after hospitalization with COVID-19 with hypoxic respiratory failure. NO treatment in high dosages was shown to be well tolerated, and evidenced an improvement in the cardiopulmonary system (Safaee Fakhr et al., 2020). All six pregnant patients subjected to NO treatment were discharged from hospital, and half of them had already delivered healthy neonates (Safaee Fakhr et al., 2020). A single-patient trial evaluated home treatment with inhaled NO, differing from previous reports (Zamanian et al., 2020). Low doses of 20 ppm were administered 12−14 h per day for 11 days with a nasal canula, remotely monitored (Zamanian et al., 2020). Positive results were observed, opening venues for further trials employing inhaled NO at home as an alternative to hospital treatments (Zamanian et al., 2020).
In the war against COVID-19, NO may not only have a potent antiviral activity but also improve ventilation perfusion in the lungs. In other words, NO demonstrates potential for the treatment of patients infected with COVID-19 both in severe and non-severe conditions, improving oxygenation and antiviral mechanisms, and preventing aggravation of the disease (Ferrari et al., 2020; Parikh et al., 2020). Patients with coronaviruses are susceptible to acute respiratory distress syndrome, which can lead to pulmonary fibrosis (Chen et al., 2004). Inhalation of NO gas enhanced arterial oxygenation in patients with acute respiratory distress syndrome (Chen et al., 2004). Indeed, NO gas is approved by US Food and Drug Administration (FDA) for neonates in the treatment of pulmonary hypertension, and recently it has been authorized for use as rescue treatment in patients with hypoxic COVID-19 symptoms (Gebistorf et al., 2016). Moreover, NO is synthesized endogenously in the blood vessels, regulating blood pressure and thus avoiding clot formation and destroying potential toxins (Ignarro, 1999).
Even though NO has demonstrated important results in the treatment of COVID-19, some authors emphasize that further studies are necessary to better understand which patients will benefit most from inhaled NO treatment. A similar pattern has been previously demonstrated in adults and children with acute respiratory distress syndrome, in which treatment with inhaled NO led to a transient improvement in oxygenation, but did not alter the mortality rate in critically ill patients (Gebistorf et al., 2016). Moreover, it was observed that NO administration might impair the kidney function of the patients treated (Gebistorf et al., 2016).
The data presented in this review are fundamental for revealing promising parameters for further trials employing NO treatment against COVID-19 and/or other respiratory diseases, in order to confirm NO potential, target patients and safe dosages.
3. Promising advances in NO allied to nanomaterials
It should be noted that NO is a free radical, and its stability is improved by the use of NO donors; these fall into different classes depending on the molecule, e.g. organic nitrates, S-nitrosothiols, metal complexes and N-diazeniumdiolates (Wang et al., 2002). Besides inhaled NO directed for pulmonary treatments, FDA has already approved the use of a few NO donors, such as Nitropress (Opasich et al., 2009). More recently, it has been demonstrated that the combination of NO donors with nanoplatforms has been an important approach, which can even improve the antimicrobial properties of NO (Douglass et al., 2019). Recent works have demonstrated the antimicrobial activity of NO donors allied to metal or metal/oxide nanoparticles, such as copper (CuNPs), silver (AgNPs), and polymeric nanoparticles. A composite based on SNAP and CuNPs enabled a tunable release of NO from SNAP, through catalyzation with CuNPs (Pant et al., 2017). NO release was adjusted through the concentration of CuNPs, which promoted a NO flux 6 times higher at the concentration of 5% when compared to free SNAP after 3 and 24 h evaluation. The combination of NO and nanoparticles promoted enhanced antibacterial activity against both Staphylococcus aureus (S. aureus) and Pseudomonas aeruginosa (P. aeruginosa). The growth of these strains treated only with SNAP were 3.5 ± 1.3 × 104 and 1.8 ± 1.3 × 104 respectively, while the values deceased significantly when combined with 3% of CuNPs, reaching 8.9 ± 1.8 × 102 and 2.4 ± 1.8 × 102 respectively (Pant et al., 2017). A similar pattern was observed for the combination of NO and AgNPs, in which the synergistic effect led to a notable inhibition of Escherichia coli (E. coli). Interestingly, a recent work reported that silver nanoparticle inhalation (3−7 nm) could be effective in early stage COVID treatment (Zachar, 2020). In the same work, the authors indicate that the minimal inhibitory concentration (10 μg mL−1) was achievable in the lower airways, but could be an effective initial method for the upper airways to suppress progression of the infection. Therefore, its combination with NO could be a supplementary therapy for infectious respiratory diseases that should be addressed in future studies. On the other hand, the encapsulation of NO donors in polymeric nanoparticles is a promising alternative. The encapsulation of AgNPs and S-nitroso-mercaptosuccinic acid (S-nitroso-MSA) in alginate nanoparticles promoted sustained release of NO and a potent antibacterial effect against S. aureus, E. coli and Staphylococcus mutans with low cytotoxicity to non-tumoral cells (Urzedo et al., 2020). The effectiveness of this strategy is not limited to antibacterial applications: the encapsulation of NO donors in chitosan nanoparticles demonstrated important results against leishmaniasis, a parasitic disease (Cabral et al., 2019). These nanoparticles efficiently inactivated Leishmania (L.) amazonensis promastigotes while no toxic effects were observed in macrophages.
More studies are required to better investigate how endogenous NO (produced by NOS) and exogenous NO, from a donor, might be related. It has been demonstrated that both sources of NO (endogenous and exogenous) can have antiviral activity (Akerstrom et al., 2005). An important advantage of nanotechnology is the ability to design a nanocarrier for sustained, localized drug release (in this case, NO release) for pulmonary application (Cavalcanti and de Britto Lira Nogue, 2020; Lammers et al., 2020). In other words, nanomaterials can be prepared for pulmonary delivery of NO/NO donors, depending on nanoparticle size, chemical structure, chemical surface and charge. Versatile nanocarriers can be designed to deliver therapeutic amounts of NO direct to the desired application site, i.e. the site of the viral infection. Chitosan nanoparticles have been explored for pulmonary delivery of active drugs due to their biocompatibility and biodegradability, and their mucoadhesive and non-immunogenic properties (Islam and Ferro, 2016; Cavalcanti and de Britto Lira Nogue, 2020; Tatlow et al., 2020).
Thus, important effects that have already been observed for NO against SARS-CoV; these could be improved by combination with nanomaterials, promoting sustained, localized release of NO in therapeutic concentrations (Keyaerts et al., 2004). NO allied to nanomaterials might thus find important applications against coronaviruses, including in the treatment of COVID-19.
4. Conclusions and perspectives
Although several drugs have been evaluated against COVID-19, further studies are still required to better elucidate their efficacy, tolerability and side effects (Magro, 2020). In this war against COVID-19, the use of NO/NO donors might find important medical application. There is still little progress on the antiviral effect of NO, especially against coronaviruses. The three protocols proposed against COVID should significantly contribute to this area, and possibly evidence a promising alternative treatment for the current pandemic. The antiviral effects of NO should be further explored. Interestingly, it has been reported that ambient ultraviolet A radiation (UVA) exposure is associated with lower COVID-19 specific mortality, independent of vitamin D (Cherrie et al., 2020). UVA triggers generation of NO from NO stores in the skin; this is released into the blood circulation, increasing the bioavailability of NO and reducing blood pressure (Cherrie et al., 2020).
Finally, we would like to encourage investment in promising advances in NO allied with nanomaterials; this treatment has already shown important results in the area of nanomedicine, but is still undeveloped in the field of viral infections, that are of huge importance not only during the SARS-CoV-2 pandemic, but also for the treatment of future viral diseases that may arise. Definitely, NO is bad news for COVID-19.
Declaration of Competing Interest
The authors report no declarations of interest.
Acknowledgements
This research was funded by FAPESP (2018/08194-2, 2018/02832-7), DI20-1003, CNPq (404815/2018-9, 313117/2019-5). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) - Finance Code 001 and REDES Project 180003.
References
- Adusumillia N.C., Zhang D., Friedman J.M., Friedman A.J. Harnessing nitric oxide for preventing, limiting and treating the severe pulmonary consequences of COVID-19. Nitric Oxide. 2020;103:4–8. doi: 10.1016/j.niox.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akaike T., Maeda H. Nitric oxide and virus infection. Immunology. 2000;101:300–308. doi: 10.1046/j.1365-2567.2000.00142.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akerstrom S., Mousavi-Jazi M., Klingstrom J., Leijon M., Lundkvist A., Mirazimi A. Nitric oxide inhibits the replication cycle of severe acute respiratory syndrome coronavirus. J. Virol. 2005;79:1966–1969. doi: 10.1128/jvi.79.3.1966-1969.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Åkerström S., Gunaan V., Keng C.T., Tan Y.-J., Mirazimi A. Dual effect of nitric oxide on SARS-CoV replication: viral RNA production and palmitoulation of the S protein are affected. Virology. 2009;395:1–9. doi: 10.1016/j.virol.2009.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Dorzi H.M., Aldawood A.S., Khan R., Baharoon S., Alchin J.D., Matroud A.A., Al Johany S.M., Balkhy H.H., Arabi Y.M. The critical care response to a hospital outbreak of Middle East respiratory syndrome coronavirus (MERS-CoV) infection: an observational study. Ann. Intensive Care. 2016;6:101. doi: 10.1186/s13613-016-0203-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson G., Reiter R.J. COVID-19 pathophysiology: interactions of gut microbiome, melatonin, vitamin D, stress, kynurenine and the alpha 7 nicotinic receptor: treatment implications. Melatonin research. 2020;3:322–345. doi: 10.32794/mr11250066. [DOI] [Google Scholar]
- Arabi Y.M., Harthi A., Hussein J., Bouchama A., Johani S., Hajeer A.H., Saeed B.T., Wahbi A., Saedy A., AlDabbagh T., Okaili R., Sadat M., Balkhy H. Severe neurologic syndrome associated with Middle East respiratory syndrome corona virus (MERS-CoV) Infection. 2015;43:495–501. doi: 10.1007/s15010-015-0720-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avnon L.S., Munteanu D., Smoliakov A., Jotkowitz A., Barski L. Thromboembolic events in patients with severe pandemic influenza A/H1N1. Eur. J. Intern. Med. 2015;26:596–598. doi: 10.1016/j.ejim.2015.08.017. [DOI] [PubMed] [Google Scholar]
- Belladona M.L., Orabona C. Potential benefits of tryptophan metabolism to the efficacy of tocilizumab in COVID-19. Front. Pharmacol. 2020;11:959. doi: 10.3389/fphar.2020.00959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berra L., Lei C., Su B., Dong H., Fakhr B.S., Grassi L.G., Di Fenza R., Gianni S., Pinciroli R., Vassena E., Morais C.C.A., Bellavia A., Spina S., Kacmarek R., Carroll R. Protocol for a randomized controlled trial testing inhaled nitric oxide therapy in spontaneously breathing patients with COVID-19. medRxiv. 2020 doi: 10.1101/2020.03.10.20033522. 2020.03.10.20033522. [DOI] [Google Scholar]
- Cabral F.V., Pelegrino M.T., Sauter I.P., Seabra A.B., Cortez M., Ribeiro M.S. Nitric oxide-loaded chitosan nanoparticles as an innovative antileishmanial platform. Nitric Oxide. 2019;93:25–33. doi: 10.1016/j.niox.2019.09.007. [DOI] [PubMed] [Google Scholar]
- Cavalcanti I.D.L., de Britto Lira Nogue MarianeCajubá. Pharmaceutical nanotechnology: which products are been designed against COVID-19? J. Nanopart. Res. 2020;22:276. doi: 10.1007/s11051-020-05010-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L., Liu P., Gao H., Sun B., Chao D., Wang F., Zhu Y., Hedenstierna G., Wang C.G. Inhalation of nitric oxide in the treatment of severe acute respiratory syndrome: a rescue trial in beijing. Clin. Infect. Dis. 2004;39:1531–1535. doi: 10.1086/425357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherrie M., Clemens T., Colandrea C., Feng Z., Webb D., Dibben C., Weller R.B. Ultraviolet a radiation and COVID-19 deaths: a multi country study. medRxiv. 2020 doi: 10.1111/bjd.20093. 2020.07.03.20145912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dioguardi F.S. To give or not to give? Lessons from the arginine paradox. J. Nutrigenet. Nutrigenomics. 2011 doi: 10.1159/000327777. [DOI] [PubMed] [Google Scholar]
- Douglass M.E., Goudie M.J., Pant J., Singha P., Hopkins S., Devine R., Schmiedt C.W., Handa H. Catalyzed nitric oxide release via Cu nanoparticles leads to an increase in antimicrobial effects and hemocompatibility for short-term extracorporeal circulation. ACS Appl. Bio Mater. 2019;2:2539–2548. doi: 10.1021/acsabm.9b00237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrari M., Santini A., Protti A., Andreis D.T., Iapichino G., Castellani G., Rendiniello V., Costantini E., Cecconi M. Inhaled nitric oxide in mechanically ventilated patients with COVID-19. J. Crit. Care. 2020;60:159–160. doi: 10.1016/j.jcrc.2020.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fialho L.G., Torrentes-Carvalho A., Cunha R.V., Faria N., Gandini M., Cipitelli M., de-Oliveira-Pinto L.M., Azeredo E.L., Kubelka C.F. Induced nitric oxide synthase (iNOS) and indoleamine 2,3-dioxygenase (IDO) detection in circulating monocyte subsets from Brazilian patients with Dengue-4 virus. Virol. Rep. 2017;7:9–19. doi: 10.3389/fphar.2020.00959. [DOI] [Google Scholar]
- Gebistorf F., Karam O., Wetterslev J., Afshari A. Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) in children and adults. Cochrane Database Syst. Rev. 2016 doi: 10.1002/14651858.CD002787.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gianni S., Fakhr B.S., Cesar C., Morais A., Di Fenza R., Pinciroli R., Houle T., Mueller A.L., Bellavia A., Carroll R., Berra L. Nitric oxide gas inhalation to prevent COVID-2019 in healthcare providers. medRxiv. 2020 doi: 10.1101/2020.04.05.20054544. 2020.04.05.20054544. [DOI] [Google Scholar]
- Gianni S., Morais C.C.A., Larson G., Pinciroli R., Carroll R., Yu B., Zapol W.M., Berra L. Ideation and assessment of a nitric oxide delivery system for spontaneously breathing subjects. Nitric Oxide. 2020;104–105:29–35. doi: 10.1016/j.niox.2020.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gur S., Chen A.L., Kadowitz P. Nitric oxide donors and penile erectile function. Nitric oxide donors: Novel biomedical applications and perspectives. 2017:121–140. doi: 10.1016/B978-0-12-809275-0.00005-3. [DOI] [Google Scholar]
- Hara T., Ogasawara N., Akimoto H., Takikawa O., Hiramatsu R., Kawabe T., Isobe K., Nagase F. High-affinity uptake of kynurenine and nitric oxide-mediated inhibition of indoleamine 2,3-dioxygenase in bone marrow-derived myeloid dendritic cells. Immunol. Lett. 2008;116:95–102. doi: 10.1016/j.imlet.2007.11.016. [DOI] [PubMed] [Google Scholar]
- Hasan S., Thomas N., Thierry B., Prestidge C.A. Biodegradable nitric oxide precursor-loaded micro- and nanoparticles for the treatment of Staphylococcus aureus biofilms. J. Mater. Chem. B. 2017;5:1005–1014. doi: 10.1039/c6tb03290g. [DOI] [PubMed] [Google Scholar]
- Hedenstierna G., Chen L., Hedenstierna M., Lieberman R., Fine D.H. Nitric oxide dosed in short bursts at high concentrations may protect against Covid 19. Nitric Oxide. 2020;103:1–3. doi: 10.1016/j.niox.2020.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ignarro L.J. Nitric oxide: a unique endogenous signaling molecule in vascular biology. Biosci. Rep. 1999;19:51–71. doi: 10.1023/a:1020150124721. [DOI] [PubMed] [Google Scholar]
- Islam N., Ferro V. Recent advances in chitosan-based nanoparticles pulmonary drug delivery. Nanoscale. 2016;8:14341–14358. doi: 10.1039/C6NR03256G. [DOI] [PubMed] [Google Scholar]
- Kang S., Brown H.M., Hwang S. Direct antiviral mechanisms of interferon-gamma. Immune Netw. 2018;18:e33. doi: 10.4110/in.2018.18.e33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keni R., Alexander A., Nayak P.G., Mudgal J., Nandakumar K. COVID-19: emergence, spread, possible treatments, and global burden. Front. Public Heal. 2020;8:216. doi: 10.3389/fpubh.2020.00216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyaerts E., Vijgen L., Chen L., Maes P., Hedenstierna G., Van Ranst M. Inhibition of SARS-coronavirus infection in vitro by S-nitroso-N- acetylpenicillamine, a nitric oxide donor compound. Int. J. Infect. Dis. 2004;8:223–226. doi: 10.1016/j.ijid.2004.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S.B., Choi J.Y., Uyangaa E., Patil A.M., Hossain F.M.A., Hur J., Park S.-Y., Lee J.-H., Kim K., Eo S.K. Blockage of indoleamine 2,3-dioxygenase regulates Japanese encephalitis via enhancement of type I/II IFN innate and adaptive T-cell responses. J. Neuroinflammation. 2016;13:79. doi: 10.1186/s12974-016-0551-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyuka P.K., Agoti C.N., Munywoki P.K., Njeru R., Bett A., Otieno J.R., Otieno G.P., Kamau E., Clark T.G., van der Hoek L., Kellam P., Nokes D.J., Cotten M. Human coronavirus NL63 molecular epidemiology and evolutionary patterns in rural coastal Kenya. J. Infect. Dis. 2018;217:1728–1739. doi: 10.1093/infdis/jiy098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovačević P., Dragić S., Vidović J., Zeljković S., Momčičević D., Rajkovača Z., Kovačević T. Serum levels of nitric oxide as a predictor of survival in acute respiratory distress syndrome caused by H1N1 pneumonia? Signa Vitae. 2015;10:63–73. doi: 10.22514/SV101.042015.7. [DOI] [Google Scholar]
- Lai C.C., Shih T.P., Ko W.C., Tang H.J., Hsueh P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int. J. Antimicrob. Agents. 2020;55 doi: 10.1016/j.ijantimicag.2020.105924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lammers T., Sofias A.M., van der Meel R., Schiffelers R., Storm G., Tacke F., Koschmieder S., Brümmendorf T.H., Kiessling F., Metselaar J.M. Dexamethasone nanomedicines for COVID-19. Nat. Nanotechnol. 2020;15:622–624. doi: 10.1038/s41565-020-0752-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei C., Su B., Dong H., Bellavia A., Di Fenza R., Fakhr B.S., Gianni S., Grassi L.G., Kacmarek R., Morais C.C.A., Pinciroli R., Vassena E., Berra L. Protocol of a randomized controlled trial testing inhaled nitric oxide in mechanically ventilated patients with severe acute respiratory syndrome in COVID-19 (SARS-CoV-2) medRxiv. 2020 doi: 10.1101/2020.03.09.20033530. 2020.03.09.20033530. [DOI] [Google Scholar]
- Lin Y.-L., Huang Y.-L., Ma S.-H., Yeh C.-T., Chiou S.-Y., Chen L.-K., Liao C.-L. Inhibition of Japanese encephalitis virus infection by nitric oxide: antiviral effect of nitric oxide on RNA virus replication. J. Virol. 1997;71:5227–5235. doi: 10.1128/JVI.71.7.5227-5235.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C., Wen L., Xiao Q., He K. Nitric oxide-generating compound GSNO suppresses porcine circovirus type 2 infection in vitro and in vivo. BMC Vet. Res. 2017:13. doi: 10.1186/s12917-017-0976-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magro G. COVID-19: Review on latest available drugs and therapies against SARS-CoV-2. Coagulation and inflammation cross-talking. Virus Res. 2020;286 doi: 10.1016/j.virusres.2020.198070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martel J., Ko Y.-F., Young J.D., Ojcius D.M. Could nasal nitric oxide help to mitigate the severity of COVID-19? Microb. Infect. 2020;22:4–5. doi: 10.1016/j.micinf.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez M.A. Compounds with therapeutic potential against novel respiratory 2019 coronavirus. Antimicrob. Agents Chemother. 2020;64:e00399–20. doi: 10.1128/aac.00399-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellor A.L., Munn D.H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat. Rev. Immunol. 2004;4:762–774. doi: 10.1038/nri1457. [DOI] [PubMed] [Google Scholar]
- Murphy A.W., Platts-Mills T.A.E., Lobo M., Hayden F. Respiratory nitric oxide levels in experimental human influenza. Chest. 1998;114:452–456. doi: 10.1378/chest.114.2.452. [DOI] [PubMed] [Google Scholar]
- O’Toole E., Doucet M.V., Sherwin E., Harkin A. Elsevier Inc.; 2016. Novel Targets in the Glutamate and Nitric Oxide Neurotransmitter Systems for the Treatment of Depression, in: Systems Neuroscience in Depression; pp. 81–113. [DOI] [Google Scholar]
- Opasich C., Cioffi G., Gualco A. Nitroprusside in decompensated heart failure: what should a clinician really know? Curr. Heart Fail. Rep. 2009;6:182–190. doi: 10.1007/s11897-009-0026-4. [DOI] [PubMed] [Google Scholar]
- Pacher P., Beckman J.S., Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2008;87:315–424. doi: 10.1161/CIRCULATIONAHA.110.956839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pant J., Goudie M.J., Hopkins S.P., Brisbois E.J., Handa H. Tunable nitric oxide release from S-Nitroso-N-acetylpenicillamine via catalytic copper nanoparticles for biomedical applications. ACS Appl. Mater. Interfaces. 2017;9:15254–15264. doi: 10.1021/acsami.7b01408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parikh R., Wilson C., Weinberg J., Gavin D., Murphy J., Reardon C.C. Inhaled nitric oxide treatment in spontaneously breathing COVID-19 patients. Ther. Adv. Respir. Dis. 2020;14 doi: 10.1177/1753466620933510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieretti J.C., Pelegrino M.T., Nascimento M.H.M., Tortella G.R., Rubilar O., Seabra A.B. Small molecules for great solutions: can nitric oxide-releasing nanomaterials overcome drug resistance in chemotherapy? Biochem. Pharmacol. 2019;176 doi: 10.1016/j.bcp.2019.113740. [DOI] [PubMed] [Google Scholar]
- Quinn J.F., Whittaker M.R., Davis T.P. Delivering nitric oxide with nanoparticles. J. Control. Release. 2015;205:190–205. doi: 10.1016/j.jconrel.2015.02.007. [DOI] [PubMed] [Google Scholar]
- Rolim W.R., Pieretti J.C., Renó D.L.S., Lima B.A., Nascimento M.H.M., Ambrosio F.N., Lombello C.B., Brocchi M., De Souza A.C.S., Seabra A.B. Antimicrobial activity and cytotoxicity to tumor cells of nitric oxide donor and silver nanoparticles containing PVA/PEG films for topical applications. ACS Appl. Mater. Interfaces. 2019;11:6589–6604. doi: 10.1021/acsami.8b19021. [DOI] [PubMed] [Google Scholar]
- Safaee Fakhr B., Wiegand S.B., Pinciroli R., Gianni S., Morais C.C.A., Ikeda T., Miyazaki Y., Marutani E., Di Fenza R., Larson G.M., Parcha V., Gibson L.E., Chang M.G., Arora P., Carroll R.W., Kacmarek R.M., Ichinose F., Barth W.H., Kaimal A., Hohmann E.L., Zapol W.M., Berra L. High concentrations of nitric oxide inhalation therapy in pregnant patients with severe coronavirus disease 2019 (COVID-19) Obstet. Gynecol. Publish Ah. 2020:1–5. doi: 10.1097/AOG.0000000000004128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Nadales A., Treminio-Quezada M., Abad H., Navarro-Motta J., Contreras-Chavez P., Kachru A., Chu C. Critical care management for novel 2019 SARS-CoV-2 and HCoV-NL63 coinfection in a young immunocompromised patient: a Chicago experience. Case Reports Crit. Care. 2020;2020:1–8. doi: 10.1155/2020/8877641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santiago-Olivares C., Rivera-Toledo E., Gómez B. Nitric oxide production is downregulated during respiratory syncytial virus persistence by constitutive expression of arginase 1. Arch. Virol. 2019;164:2231–2241. doi: 10.1007/s00705-019-04259-0. [DOI] [PubMed] [Google Scholar]
- Schairer D.O., Chouake J.S., Nosanchuk J.D., Friedman A.J. The potential of nitric oxide releasing therapies as antimicrobial agents. Virulence. 2012;3:271–279. doi: 10.4161/viru.20328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seabra A.B. Elsevier; 2017. Nitric Oxide Donors. [Google Scholar]
- Seabra A.B., Durán N. Nitric Oxide Donors. 2017. Nitric oxide donors for treating neglected diseases; pp. 25–53. Chapter 2. [DOI] [Google Scholar]
- Seabra A.B., Justo G.Z., Haddad P.S. State of the art, challenges and perspectives in the design of nitric oxide-releasing polymeric nanomaterials for biomedical applications. Biotechnol. Adv. 2015;33:1370–1379. doi: 10.1016/j.biotechadv.2015.01.005. [DOI] [PubMed] [Google Scholar]
- Seabra A.B., Pelegrino M.T., Haddad P.S. Elsevier Inc.; 2016. Can Nitric Oxide Overcome Bacterial Resistance to Antibiotics?, in: Antibiotic Resistance: Mechanisms and New Antimicrobial Approaches; pp. 187–204. [DOI] [Google Scholar]
- Tatlow D., Tatlow C., Tatlow S., Tatlow S. A novel concept for treatment and vaccination against Covid‐19 with an inhaled chitosan‐coated DNA vaccine encoding a secreted spike protein portion. Clin. Exp. Pharmacol. Physiol. 2020;00:1–5. doi: 10.1111/1440-1681.13393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavazzi G., Marco P., Mongodi S., Dammassa V., Romito G., Mojoli F. Inhaled nitric oxide in patients admitted to intensive care unit with COVID-19 pneumonia. Crit. Care. 2020;24:508. doi: 10.1186/s13054-020-03222-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas S.R., Mohr D., Stocker R. Nitric oxide inhibits indoleamine 2,3-dioxygenase activity in interferon-gamma primed mononuclear phagocytes. J. Biol. Chem. 1994;269:14457–14464. [PubMed] [Google Scholar]
- Turski W., Wnorowski A., Turski G.N., Turski C.A., Turski L. AhR and IDO1 in pathogenesis of Covid-19 and the “Systemic AhR Activation Syndrome:” a translational review and therapeutic perspectives. Restor. Neurol. Neurosci. 2020;38:343–354. doi: 10.3233/RNN-201042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urzedo A.L., Gonçalves M.C., Nascimento M.H.M., Lombello C.B., Nakazato G., Seabra A.B. Multifunctional alginate nanoparticles containing nitric oxide donor and silver nanoparticles for biomedical applications. Mater. Sci. Eng. C. 2020;112 doi: 10.1016/j.msec.2020.110933. [DOI] [PubMed] [Google Scholar]
- van Wissen M., Snoek M., Smids B., Jansen H.M., Lutter R. IFN-γ amplifies IL-6 and IL-8 responses by airway epithelial-like cells via indoleamine 2,3-dioxygenase. J. Immunol. 2002;169:7039–7044. doi: 10.4049/jimmunol.169.12.7039. [DOI] [PubMed] [Google Scholar]
- Wang P.G., Xian M., Tang X., Wu X., Wen Z., Cai T., Janczuk A.J. Nitric oxide donors: chemical activities and biological applications. Chem. Rev. 2002;102:1091–1134. doi: 10.1021/cr000040l. [DOI] [PubMed] [Google Scholar]
- Wong S.S.Y., Yuen K.Y. The management of coronavirus infections with particular reference to SARS. J. Antimicrob. Chemother. 2008;62:437–441. doi: 10.1093/jac/dkn243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye Q.-X., Xu L.-H., Shi P.-J., Xia T., Fang J.P. Indoleamine 2,3‑dioxygenase and inducible nitric oxide synthase mediate immune tolerance induced by CTLA4Ig and anti-CD154 hematopoietic stem cell transplantation in a sensitized mouse model. Exp. Ther. Med. 2017;14 doi: 10.3892/etm.2017.4722. 1884-1881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zachar O. Formulations for COVID-19 early stage treatment via silver nanoparticles inhalation delivery at home and hospital. Sci. Prepr. 2020:14. doi: 10.14293/S2199-1006.1.SOR-.PPHBJEO.v1. [DOI] [Google Scholar]
- Zamanian R.T., Pollack C.V., Gentile M.A., Rashid M., Fox J.C., Mahaffey K.W., de Jesus, Perez V. Outpatient inhaled nitric oxide in a patient with vasoreactive idiopathic pulmonary arterial hypertension and COVID-19 infection. Am. J. Respir. Crit. Care Med. 2020 doi: 10.1164/rccm.202004-0937LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang R., Hess D.T., Qian Z., Hausladen A., Fonseca F., Chaube R., Reynolds J.D., Stamler J.S. Hemoglobin βCys93 is essential for cardiovascular function and integrated response to hypoxia. Proc. Natl. Acad. Sci. U.S.A. 2015;112:6425–6430. doi: 10.1073/pnas.1502285112. [DOI] [PMC free article] [PubMed] [Google Scholar]