Abstract
Lime peel contains essential oils are used as anti-oxidants or anti-cancer compounds. As a traditional medicine, lime has been widely used as a substitute for antibiotics. This study aimed to identify active compounds in peel extracts from Citrus aurantifolia that grows in Indonesia. Extraction was carried out by maceration using three different solvents: ethyl acetate, chloroform, and n-hexane. The extracts were analyzed using gas chromatography-mass spectrometry. The results showed that lime peel contained many important compounds and that 28, 27, and 24 different chemical compounds, both minor and major constituents, were extracted by ethyl acetate, chloroform, and n-hexane, respectively. Four compounds were found in all three solvent extracts, namely, D-limonene, phytol, α-tocopherol, and 5, 7-dimethoxycoumarin. Forty-seven compounds were uniquely present in one solvent, including 17 in ethyl acetate, 17 in chloroform, and 13 in n-hexane. Among the active compounds extracted, several are of biological importance, for example, stigmasterol, D-limonene, Vitamin E, and α-tocopherol. It can be concluded that a variety of distinct compounds are extracted from the same lime peel sample when different solvents are used and that some of these are bioactive compounds with anti-oxidant, anti-microbial, or anti-cancer properties.
Key words: Anti-microbial, bioactive compounds, Citrus aurantifolia, gas chromatography-ass spectrometry, lime peel
INTRODUCTION
Citrus aurantifolia, also known as pecel orange, is a type of lime in the Citrus genus that is widespread in Asia and Central America. Lime peel contains essential oils, also known as ethereal oils (aetherolea), which are widely used in the perfume chemical industry. Furthermore, in the food industry, the oils are used to add the aroma of oranges to food and drinks; in the health sector, the oils are used as anti-oxidants or anti-cancer compounds. The technology used to process lime peels has evolved from the original distillation methods (carried out using a simple tool fashioned from ordinary drums) to more advanced processes using stainless steel kettles.
Lime contains numerous beneficial chemical compounds, e.g., citric acid, amino acids (tryptophan, lysine), essential oils (citral, limonene, phellandrene, camphor lemon, cadinene, geranyl acetate, linalyl acetate, acetaldehyde, nonaldehyde), resin, glycoside, and citric acid. It also contains fats, calcium, phosphorus, iron, sulfur, as well as Vitamins B1 and C. In addition, lime juice contains saponins and flavonoids, namely, hesperidin (hesperetin 7-rutinoside), tangeretin, naringin, eriocitrin, and eriocitrocide. Hesperidin is beneficial to health due to its anti-inflammatory and anti-oxidant properties and is an inhibitor of prostaglandin synthesis. Hesperidin also inhibits azoxymethane-induced colon carcinogenesis in rabbits and inhibits N-butyl-N-(4-hydroxybutyl) nitrosamine-induced bladder carcinogenesis in rats.[1] Lime has also been shown to contain citral, limonene, fenchone, terpineol, bisabolene, and other terpenoids.[2] Previous studies have reported that D-Limonene is cytotoxic to cancer cells but not to hepatocytes.[3]
Lime has been used as a cough medicine, antipyretic, or a medication to treat aches and pain. It is also used to treat a wide variety of conditions, including dysentery, constipation, hemorrhoids, irregular menstruation, diphtheria, acne, dizziness and vertigo, barking cough, flu and fever, smoking cessation, tonsillitis, and plaque. In the present study, we aimed to analyze the active compounds extracted from the peel of the Indonesian lime (C. aurantifolia) using ethyl acetate, chloroform, and n-hexane and discuss their potential anti-oxidant, anti-bacterial, and anti-cancer properties.
MATERIALS AND METHODS
Lime peel extraction
Lime peel (C. aurantifolia) samples were obtained from citrus farms in Maribaya Village, West Java, Indonesia. The peels were cut into small pieces and dried at room temperature for 24 h.[4] Dried lime peel was then mashed into powder, and the powder subjected to three rounds of extraction by maceration in 70% ethanol. The extract was filtered and evaporated in a rotary evaporator until a thick extract was obtained. The thick extract was partitioned using three solvents, namely, n-hexane, chloroform, and ethyl acetate. Each final fraction obtained was evaporated in a rotary evaporator and analyzed by gas chromatography-mass spectrometry (GC-MS) (Shimadzum, bnv, Japan).[5,6]
Gas chromatography-mass spectrometry analysis
A total of 1 μL of the extract was injected into the GC-MS using a direct injection system at an injection port temperature of 270°C. The column temperature gradient was initially set at 130°C for 4 min, then increased to 170°C with an increment of 6.5°C/min, after which the temperature was raised again to 215°C with an increment of 2.75°C/min and maintained for 12 min. Finally, the temperature was increased to 230°C at a rate of 4°C/min and maintained at 230°C for 3 min. The detector temperature used was 280°C with MS detector electrons at 70 eV, while the ion source temperature was 250°C. Helium gas was regulated at a pressure of 1 kg/cm2 while hydrogen and nitrogen gas pressures were 0.5 kg/cm2. The gas flow rates were regulated to 30 mL/min, 400 mL/min, 30.1 mL/min, and 46.4 mL/min for hydrogen, oxygen, nitrogen, and helium, respectively.[7]
RESULTS
Gas chromatography analysis
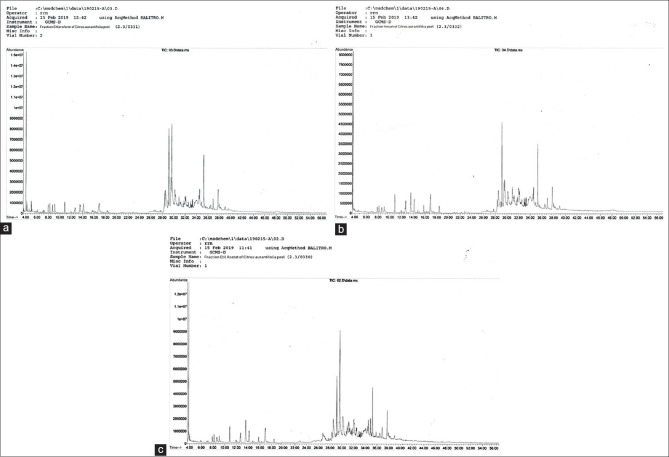
The spectrograms of lime peel extracts using the three solvents [Figure 1] showed that very different compounds were extracted in n-hexane when compared with the other two solvents. In contrast, the chloroform and ethyl acetate spectrograms were more or less similar. At retention times between 10 and 18 min, the chloroform and ethyl acetate spectra are identical, while peak heights were higher in the shown for n-hexane. The three solvents used have different dielectric properties. The dielectric constant indicates the electrical capacity of a material and is influenced by its polarity. Among the three solvents used, n-hexane has the lowest dielectric constant (1.89), while ethyl acetate has the highest dielectric constant (6.02). The dielectric constant of chloroform (4.8) is closer to that of ethyl acetate than to that of n-hexane, and this may explain why the spectrogram of its extract looks more like that of ethyl acetate.
Figure 1.
Gas chromatography-mass spectrometry spectrograms of lime peel extracted using solvents (a) chloroform, (b) n-hexane, and (c) ethyl acetate
Ethyl acetate is semi-polar, and the highest number of compounds were extracted using this solvent. Chloroform is considered a nonpolar solvent with a dielectric constant of 4.8, which makes it a slightly polar solvent resulting in an increased ability to dissolve chemicals. Meanwhile, n-hexane is a nonpolar organic compound commonly used to remove fat from ingredients, and as shown in Table 1, many substances that are soluble in n-hexane during extraction are nonpolar, including octacosane [Table 1: No. 21] and fatty acid derivatives [Table 1: No. 9, 11, 13 and 15].
Table 1.
Chemical compounds in lime peel extracted with three different solvents
| n | Ethyl acetate | n-Hexane | Chloroform |
|---|---|---|---|
| 1 | D-Limonene | D-Limonene | D-Limonene; |
| 2 | (+)-4-Carene | δ-elemene | 3,7-dimethyl-1,3,6-octatriene |
| 3 | Z-citral | β-elemene | 1,1,2,4,4,5-hexachloro-1,5-hexadiene |
| 4 | δ-elemene | caryophyllene | Z-3,7-dimethyl-2,6-octadiene-1-ol |
| 5 | β-elemene | γ-elemene | Z-citral |
| 6 | Methyleugenol | bicyclo [3.1.1] hept-2-en trans- α -bergamotene | Geraniol |
| 7 | Bicyclogermacrene | β-bisabolene | 3,7-dimethyl-2,6-octadienal |
| 8 | β-bisabolene | E, E-α-farnesene | Cyclohexene-4-etheyl-4-methyl-3-(1-methylethenyl)-1-(1-methylethyl)-(3R) |
| 9 | Farnesene | n-hexadecanoic acid | Trans-4,11,11-trimethyl-8-methylenebicyclo[7,2,0]undeca-4-ene |
| 10 | Pentopyranose | Coumarin, 5,7-dimethoxy | Bicyclogermacrene |
| 11 | Hexadecanoic acid | Linoleic acid, methyl ester | 1,3,6,10-dodecatetraene, 3,7,11-trimethyl |
| 12 | Coumarin, 5,7-dimethoxy | Phytol | Hexadecanoic acid |
| 13 | Phytol | Linolenic acid | Coumarin, 5,7-dimethoxy |
| 14 | 1H-inden-1-one, 2,3-dihydo-5,6-dimethoxy-3-methyl-3,4-dimethoxybenzylideneacetone | 1(S)-methyl-2(R)- phenylcyclopropan-ol | 9,12-octadiecadienoic acid (z, z)-methyl ester |
| 15 | Phenol, 2-methyl-6-(2-propenyl)- | Linolenic acid butyl-9,12,15-octadecatrienoate | Phytol |
| 16 | Isopimpinellin | Naphth [2,3-b] azet-2 (1H)-one, 1-phenil; 1-phenylnaphtho [2,3-b] azet-2H (1H)-one | Linolenic acid |
| 17 | Cyclohexene, 6-butyl-1-nitro-;6-butyl-1-nitro-1-cyclohexene | Bicyclo[2,2,1] heptane, 7,7-dimethyl-2-methylene-cyclohexanol, 5-methyl-2-(1-methylethenyl)- | 1-methyl-2,3,6,7-tetrahydro-5H-inden-5-one |
| 18 | Neryl acetate | 7,10,13-hexadecatrienoic acid, methyl ester- 1,3,14,16-nonadecatetraene | Isopimpinellin |
| 19 | Phthalic acid | Bicyclo [3,1,1]hepatene, 6,6-dimethyl-2-methylene | 4-methylene-2,8,8-trimethyl-2-ninyl-9,12,15-octadecatrienoic acid, 2.3-dihydroxypropyl ester, (z, z, z)-1,5-cyclodecadiene, |
| 20 | Naphth [2,4-b] azet-2 (1H)-one, 1-phenyl-; 8H-dibenzo [a, g] quinolizin-8-one | Trans-geranylgeraniol camphene | ethanol, 2-(3,3-dimethylcyclohexylidene)- |
| 21 | (3E,13E)-1,3,12-nonadecatriene | Octacosane | bicyclo [4,2,0] oct-2-ene, 3,7-dimethyl-7-(4-methyl-3-pentenyl)-8-(2,6,10-trimethyl |
| 22 | Cyclooctane, 3-ethenyl- | α-tocopherol | bicyclo [2,2,1] heptane, 2,2-dimethyl-3-methylene |
| 23 | 9,12-octadecadienoic acid (Z, Z)-,2-hydroxy-1-(hydroxymethyl) ethyl ester | Stigmasterol | 16-(acetoxymethyl)-21-oxobenzo [g] indolo [2,3-a] quinolizine-19-yl acetate] |
| 24 | Nonadecane | Stigmast-5-en-3-ol, (3β) | Tetracosane |
| 25 | 2,8-diisopropyl-peri-xanthenoxanthene-4,10-quinone | α-tocopherol | |
| 26 | α-tocopherol | Stigmasterol | |
| 27 | Stigmasta-5,22-dien-3-ol, (3β, 22E) | Stigmast-5-en-3-ol, (3β) | |
| 28 | (23S)-ethylcholest-5-en-3β-ol stigmats-5-en-3-ol, (3β) |
GC-MS analysis showed that a total of 79 chemical compounds were found in the three lime peel extracts [Table 1]. The names of the chemical compounds in the ethyl acetate, n-hexane, and chloroform extracts are listed in Table 1. A few more compounds were extracted by chloroform and ethyl acetate than by n-hexane. Although the spectrograms of chloroform and ethyl acetate extracts were identical, both ethyl acetate and n-hexane extracted many of the same compounds. For instance, the components of atsiri oils such as limonene, farnesene, and bisabolene are extracted by both ethyl acetate and n-hexane, while chloroform extracts only contained D-limonene, the main ingredient of orange peel. There were four compounds that were extracted in all three solvents, namely, limonene, phytol, α-tocopherol, and 5, 7-dimethoxycoumarin. Four compounds were extracted in both ethyl acetate and chloroform, namely, Z-citral, bicyclogemacrene, hexadecanoic acid, and isopimpinellin. Four compounds were extracted in both ethyl acetate and n-hexane, namely, δ-elemene, β-elemene, β-bisabolene, and farnesene. Finally, three compounds were extracted in n-hexane and chloroform, namely, stigmasterol, stigma-5-en-3-ol (3 β), and linolenic acid.
DISCUSSION
Important compounds in lime peel
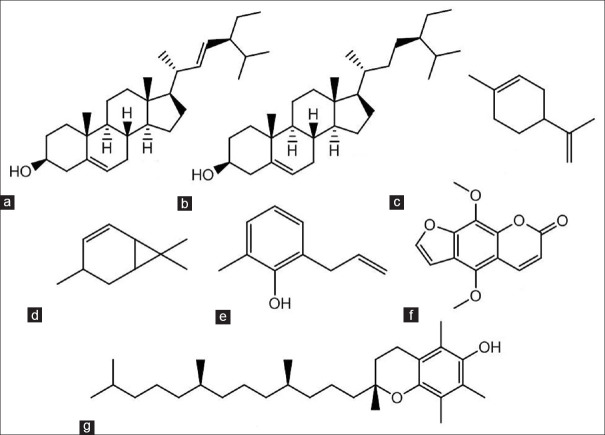
Among the compounds in the three extracts, there are several important active compounds that have anti-oxidant, anti-bacterial, or anti-carcinogenic properties. These important compounds are shown in Figure 2.
Figure 2.
Some important compounds found in the lime peel extracts. (a) Stigmasta-5, 22-dien-3-ol, (3 β, 22E), (b) stigmast-5-en-3-ol, (3 β), (c) D-limonene, (d) (+)-4-Carene, (e) phenol, 2-methyl-6-(2-propenyl), (f) isopimpinellin, and (g) α-tocopherol
Among the important and biologically active compounds identified is α-tocopherol [Figure 2g]. This compound is an anti-oxidant that is commonly used in food fortification. α-Tocopherol is a type of Vitamin E with the label number “E307.” Vitamin E itself comes in eight different structural forms, four of which are tocopherols, and the remaining are tocotrienols. The tocopherols with hydroxyl groups could be able to donate hydrogen atoms resulted in reducing free radicals and hydrophobic side chains. This condition allows for the penetration of cell membranes of the target. Compared with the Vitamin E compounds, α-tocopherol is more easily absorbed and has better accumulation profiles in humans.[8]
Another important compound is D-limonene [Figure 2c]. D-limonene is a compound sourced from the peel of oranges, mandarins, limes, and grapefruit. Its name is derived from “lemon,” and it is often used as a flavoring agent in food. D-limonene is different from the less widely found limonene known as L-limonene, which is found in mint oil. Structurally, these compounds are classified as terpenoids with a closed chain or as cycloalkenes.
D-limonene is commonly used to make hand sanitizers, perfumes, botanical pesticides, and chemical solvents. D-limonene compounds are also believed to be able to prevent or treat various diseases such as bronchitis, diabetes, gallstones, and dyslipidemia. A study revealed that D-limonene is effective in preventing and treating cancer.[9] In addition to D-limonene, compounds shown in Figure 2d and e are also terpenoid compounds that are used in active ingredients, notably for their anti-oxidant properties.
Anti-microbial and anti-cancer compounds
Some of the compounds extracted from the three solvents mentioned above have significant anti-bacterial properties, including caryophyllene, which was in the n-hexane fraction. The structures of caryophyllene compounds are shown in Figure 3. β-caryophyllene compounds are known to have anti-bacterial properties as well as anti-cancer properties (e.g., through selective cytotoxicity against human colorectal cancer cells). β-caryophyllene also suppresses tumor motility, cell invasion, and tumor aggregation.[10] Furthermore, a study showed that caryophyllene compounds not only have anti-cancer properties, but also act as antibiotics.[5]
Figure 3.
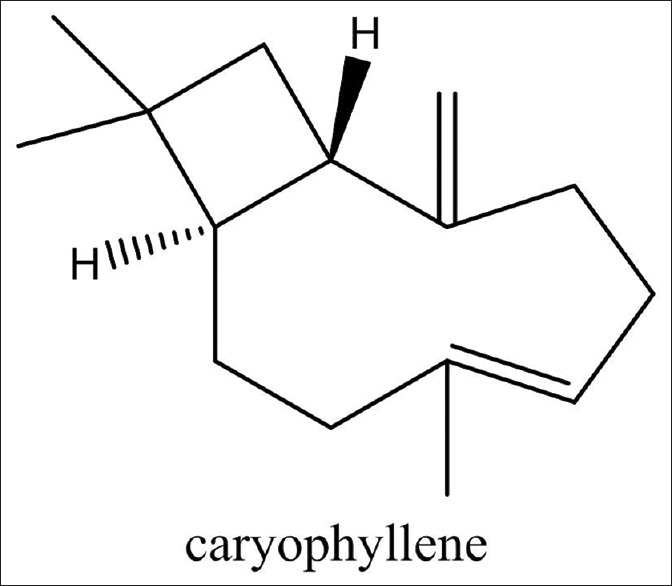
Chemical structure of caryophyllenes
In addition to caryophyllene compounds, other essential oils also contain compounds such as β-elemene, δ-elemene, and bicyclogermacrene (detected in the n-hexane fraction), which have anti-microbial properties. Zhu et al. reported that bicyclogermacrene, β-elemene, δ-elemene, and γ-elemene extracted from rhizoma were not only able to inhibit the growth of the gram-positive bacteria Propionebacterium acnes and Staphylococcus aureus, but could also inhibit the growth of the fungus Malassezia furfur.[11] Thus, Zhu et al. showed that elemene compounds can have both anti-bacterial and anti-fungal functions. The structure of the bicyclogermacrene and groups of elemene compounds are shown in Figure 4.
Figure 4.
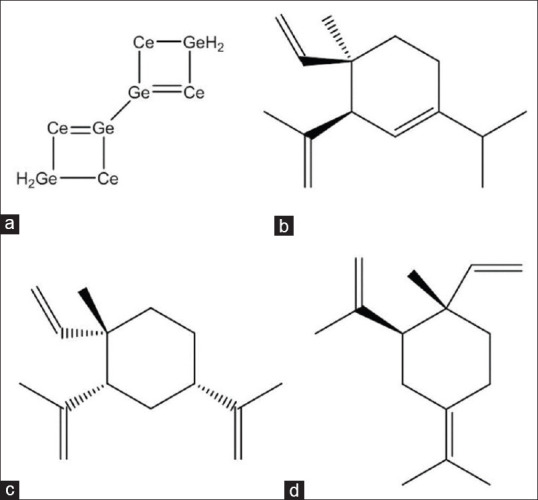
(a) Bicyclogermacrene, (b) δ-elemene, (c) β-elemene, dan (d) γ-elemene
Furthermore, bicyclogermacrene has unique properties that make it an active anti-infectious agent that also accelerates wound healing.[12] Together with these germacrene compounds, several other compounds found in essential oils sourced from various plants are active against a variety of infective pathogens and can accelerate cell growth.[12]
CONCLUSION
GC-MS analysis of active compounds extracted from lime peel using ethyl acetate, n-hexane, and chloroform showed that all three solvents extracted the main components of lime peel, such as limonene and α-tocopherol. The total number of compounds extracted in ethyl acetate, chloroform, and n-hexane were 28, 27, and 24, respectively. Four of the compounds detected were found in all solvents, whereas 47 compounds were uniquely present in one solvent. Of the 79 compounds in total, some are bioactive compounds, namely α-tocopherol, D-limonene, and phytol. Limonene-derived compounds, such as δ-elemene, β-elemene, γ-elemene, and also bicyclogermanecene, have anti-bacterial and anti-cancer properties that can potentially be used in medicines.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
The author thanks the Integrated Laboratory of the Institut Pertanian Bogor, West Java and the Central Jakarta Regional Health Laboratory (Labkesda) for GCMS testing in this study.
REFERENCES
- 1.Aprioku JI, Briggs OE. Citrus aurantifolia (Lime) juice negatively influences estrous cycle of Wistar rats. IOSR J Pharm. 2018;8:38–43. [Google Scholar]
- 2.Manohar S, Khan SI, Kandi SK, Raj K, Sun G, Yang X, et al. Synthesis, antimalarial activity and cytotoxic potential of new monocarbonyl analogues of curcumin. Bioorg Med Chem Lett. 2013;23:112–6. doi: 10.1016/j.bmcl.2012.11.004. [DOI] [PubMed] [Google Scholar]
- 3.Shah B, Shaikh MV, Chaudagar K, Nivsarkar M, Mehta A. Anti-leukemic and anti-angiogenic effects of d-Limonene on K562-implanted C57BL/6 mice and the chick chorioallantoic membrane model. Animal Model Exp Med. 2018;1:328–33. doi: 10.1002/ame2.12039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Krishnan KR, James F, Mohan A. Isolation and characterization of n-hexadecanoic acid from Canthium parviflorum leaves. J Chem Pharm Res. 2016;8:614–7. [Google Scholar]
- 5.García-Pérez JS, Cuéllar-Bermúdez SP, Arévalo-Gallegos A, Rodríguez-Rodríguez J, Iqbal HM, Parra-Saldivar R. Identification of Bioactivity, Volatile and Fatty Acid Profile in Supercritical Fluid Extracts of Mexican arnica. Int J Mol Sci. 2016;17:1528. doi: 10.3390/ijms17091528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Munish P, Verma ML, Mahale K. Handbook on Flavonoids: Dietary Sources, Properties and Health Benefits. Hauppauge, New York: Biochemistry Research Trends: Nutrition and Diet Research Progress. Nova Science Publishers, Inc; 2012. Processing of citrus peel for the extraction of flavonoids for biotechnologycal applications; pp. 443–59. [Google Scholar]
- 7.Fan S, Chang J, Zong Y, Hu G, Jia J. GC-MS analysis of the composition of the essential oil from dendranthema indicum var. Aromaticum using three extraction methods and two columns. Molecules. 2018;23:576. doi: 10.3390/molecules23030576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rigotti A. Absorption, transport, and tissue delivery of vitamin E. Mol Aspects Med. 2007;28:423–36. doi: 10.1016/j.mam.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 9.Gould MN. Cancer chemoprevention and therapy by monoterpenes. Environ Health Perspect. 1997;105(Suppl 4):977–9. doi: 10.1289/ehp.97105s4977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dahham SS, Tabana YM, Iqbal MA, Ahamed MB, Ezzat MO, Majid AS, Majid AM. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil of Aquilaria crassna. Molecules. 2015;20:11808–29. doi: 10.3390/molecules200711808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhu J, Lower-Nedza AD, Hong M, Jie S, Wang Z, Yingmao D, et al. Chemical composition and antimicrobial activity of three essential oils from Curcuma wenyujin. Nat Prod Commun. 2013;8:523–6. [PubMed] [Google Scholar]
- 12.Parsaeimehr A. Bioactive molecules of herbal extracts with anti-infective and wound healing properties. Microbiology for Surgical Infections Diagnosis, Prognosis and Treatment. 2014:205–20. [Google Scholar]