Kv7.2 (KCNQ2) channel dysfunctions cause a rare form of neonatal and infantile epileptic and developmental encephalopathy (MIM 613720).1 Unlike early clinical features and the epilepsy phenotype of KCNQ2 encephalopathy, data about the long-term developmental outcome are lacking. We followed up for over 3 decades a girl with this disease.
The patient, who is aged 30 years and the only child of healthy unrelated parents, presented with pleiomorphic epileptic seizures (clonic, tonic asymmetric, and focal, with cyanosis) on the second day of life. These seizures were unresponsive to IV benzodiazepines and pyridoxine. EEG detected 2 independent foci in both frontotemporal regions. After a stormy onset, the combination of phenobarbital and phenytoin led to seizure improvement within the third month of life.
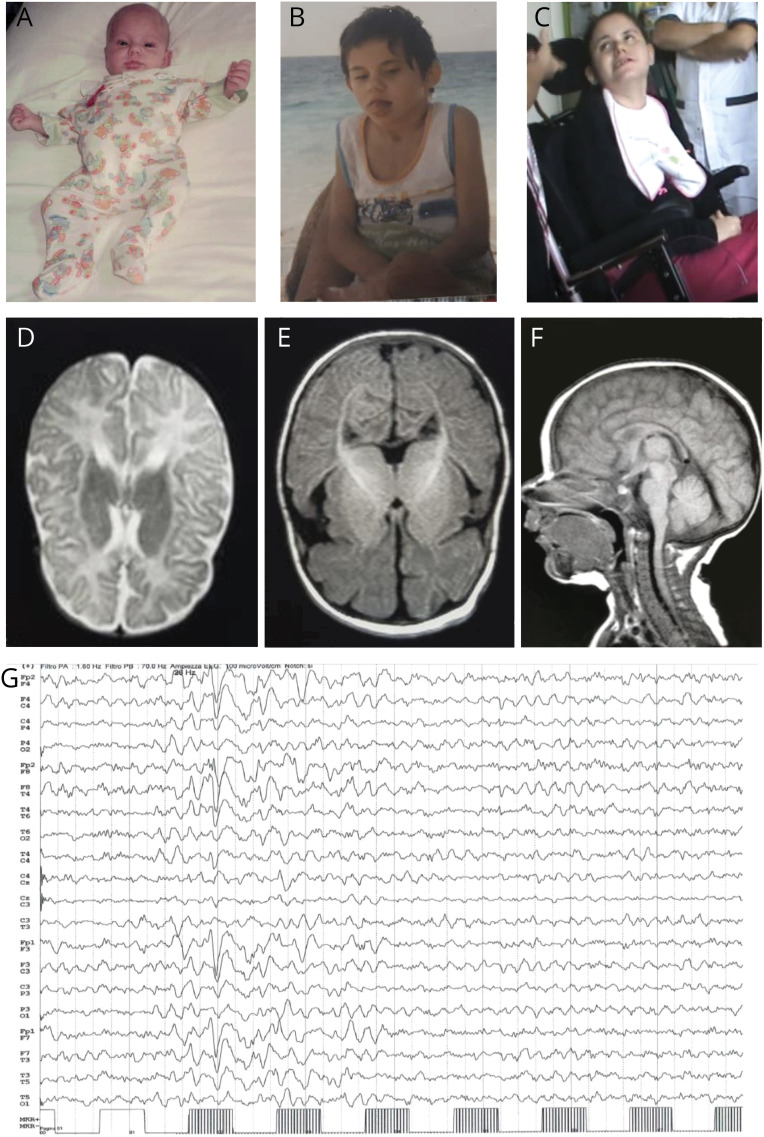
Neurologic examination during the first months of life detected minor facial dysmorphisms (figure), hypokinesia, and diffuse hypotonia. In the following years, progressive developmental delay, subcontinuous Rett-like hand stereotypies (flapping, washing, and mouthing) with reduced functional use of both hands, bradykinesia, hypomimia, severe dystonic quadriparesis, and kyphoscoliosis became obvious. She was never able to walk and had been using a wheelchair since age 7 years. At this stage, the girl showed profound intellectual disability (Griffiths Mental Development Scales: DQ 30) with absent language, hypoactivity, and severely limited social interaction. During late childhood and adolescence, epilepsy showed a relapsing-remitting course with focal clonic or generalized seizures and prolonged episodes of staring and unresponsiveness. Interictal EEG alterations included plurifocal spike and wave abnormalities predominantly in the anterior regions and a photoparoxysmal response at medium and high frequencies (figure, G). Between the ages of 17 and 20 years, seizures were well controlled with valproate and lamotrigine. After age 20 years, the patient remained seizure free with a stable clinical condition (video 1).
Figure. Clinical, neuroradiologic, and EEG features of the reported patient.
(A, B, and C) Minor facial dysmorphisms at age 5 months, 7 years, and 30 years (prominent ears and chin, deep-set eyes, and wide mouth with protruding tongue). (D, E, and F) Brain MRI at age 4 years showing frontotemporal cortical atrophy. (G) EEG at age 28 years showing a photoparoxysmal response at 28 Hz.
Clinical presentation of the reported patient at age 30 years. The patient presented with dystonic quadriparesis, bradykinesia, hypomimia, and hand stereotypies. The video was obtained with written informed parental consent.Download Supplementary Video 1 (15.1MB, mp4) via http://dx.doi.org/10.1212/000510_Video_1
A brain MRI at age 4 years showed prevalent frontoinsular atrophy (figure, D–F). CSF examination at age 7 years detected normal glycorrhachia, lactate, amino acids, biogenic amines, and pterins.
A next-generation sequencing panel that included 140 genes involved in genetic epilepsies revealed the de novo heterozygous KCNQ2 variant c.629G>A (p.Arg3894Gln), which was not present in the Human Gene Mutation Database (portal.biobase-international.com/). Its pathogenetic role is supported by previously published missense variants affecting the same amino acid in the S4 voltage sensor segment of the protein in patients with epileptic and developmental encephalopathies (c.628C>T; p.Arg210Cys and c.629G>A; p.Arg210His) and by multistate structural modeling studies that have demonstrated the involvement of the arginine at position 210 in the S4 segment in the stabilization of the protein through complex electrostatic interactions.1–3 Although there were no therapeutic consequences, the molecular genetic diagnosis had a positive psychological impact on the parents.
The clinical history of our patient highlighted that motor and behavioral abnormalities, rather than epilepsy, characterize the adult phenotype of KCNQ2 encephalopathy. The clinical follow-up of our patient showed the progressive emergence of a behavioral pattern resembling that of Rett syndrome. A Rett-like presentation was previously reported in 8 patients with KCNQ2 encephalopathy.4,5 Two of these patients carried the c.637C>T (p. Arg213Trp) variant, which involves the same region as the c.628C>T variant. One of them presented with neonatal seizures, global developmental delay, a lack of speech, and hand stereotypies.4,5
Some parkinsonian symptoms, such as bradykinesia, hypomimia, hypoactivity, and dystonia (video 1), have previously been reported in classic MeCP2-related Rett syndrome and in other early-onset epileptic and developmental encephalopathies,6 suggesting that several genetically determined neurodevelopmental disorders share a common adult outcome. However, unlike other diseases (including Kleefstra syndrome, MECP2 mutations in males, and CDKL5 encephalopathy in females), our patient did not experience a regression in cognitive or motor performance in adulthood.6
The role of Kv7.2 dysfunction in the expression of abnormal GABAergic parvalbumin-positive interneurons and their dendritic arborization within corticostriatal and nigrostriatal dopaminergic transmission (an impairment also described in Mecp2-null mice) could suggest a possible link between Rett syndrome, KCNQ2 encephalopathy, and other neuropsychiatric disorders in terms of the pathophysiology of hypokinetic movement disorders, dystonia, and hand stereotypies.7 Recent experimental data on KCNQ2+/− mice established a link between loss-of-function Kv7.2 mutations and behavioral disorders that was characterized by increased repetitive and compulsive behaviors, reduced social interest, and impaired social interaction mechanisms.7
From a clinical perspective, the present case highlights the importance of diagnostic reevaluation in adults with early-onset chronic neurologic conditions that were previously labeled according to a syndromic, rather than etiologic, diagnostic classification. In the absence of therapeutic consequences, the prognostic and psychological implications of a genetic diagnosis should not be underestimated.
Appendix. Authors
Study funding
No targeted funding reported.
Disclosure
The authors have no disclosures to declare. Go to Neurology.org/NG for full disclosure.
References
- 1.Weckhuysen S, Ivanovic V, Hendrickx R, et al. Extending the KCNQ2 encephalopathy spectrum: clinical and neuroimaging findings in 17 patients. Neurology 2013;81:1697–1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mercimek-Mahmutoglu S, Patel J, Cordeiro D, et al. Diagnostic yield of genetic testing in epileptic encephalopathy in childhood. Epilepsia 2015;56:707–716. [DOI] [PubMed] [Google Scholar]
- 3.Soldovieri MV, Ambrosino P, Mosca I, et al. Epileptic encephalopathy in a patient with a novel variant in the Kv7.2 S2 transmembrane segment: clinical, genetic, and functional features. Int J Mol Sci 2019;20:E3382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang J, Zhang Q, Chen Y, Yu S, Wu X, Bao X. Rett and Rett-like syndrome: expanding the genetic spectrum to KIF1A and GRIN1 gene. Mol Genet Genomic Med 2019;7:e968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vidal S, Brandi N, Pacheco P, et al. The most recurrent monogenic disorders that overlap with the phenotype of Rett syndrome. Eur J Paediatr Neurol 2019;23:609–620. [DOI] [PubMed] [Google Scholar]
- 6.Willemsen MH, Rensen JH, van Schrojenstein-Lantman de Valk HM, Hamel BC, Kleefstra T. Adult phenotypes in Angelman- and Rett-like syndromes. Mol Syndromol 2012;2:217–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim EC, Patel J, Zhang J, et al. Heterozygous loss of epilepsy gene KCNQ2 alters social, repetitive and exploratory behaviors. Genes Brain Behav 2020;19:e12599. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Clinical presentation of the reported patient at age 30 years. The patient presented with dystonic quadriparesis, bradykinesia, hypomimia, and hand stereotypies. The video was obtained with written informed parental consent.Download Supplementary Video 1 (15.1MB, mp4) via http://dx.doi.org/10.1212/000510_Video_1