Turkana pastoralists who move to cities exhibit poor cardiometabolic health, consistent with evolutionary mismatch.
Abstract
The “mismatch” between evolved human physiology and Western lifestyles is thought to explain the current epidemic of cardiovascular disease (CVD) in industrialized societies. However, this hypothesis has been difficult to test because few populations concurrently span ancestral and modern lifestyles. To address this gap, we collected interview and biomarker data from individuals of Turkana ancestry who practice subsistence-level, nomadic pastoralism (the ancestral way of life for this group), as well as individuals who no longer practice pastoralism and live in urban areas. We found that Turkana who move to cities exhibit poor cardiometabolic health, partially because of a shift toward “Western diets” high in refined carbohydrates. We also show that being born in an urban area independently predicts adult health, such that life-long city dwellers will experience the greatest CVD risk. By focusing on a substantial lifestyle gradient, our work thus informs the timing, magnitude, and evolutionary causes of CVD.
INTRODUCTION
It has become increasingly clear that the spread of “Western,” industrialized lifestyles is contributing to a rapid rise in metabolic and cardiovascular diseases (CVDs) worldwide (1–5). Since the Industrial Revolution, modern advancements in agriculture, transportation, and manufacture have had a profound impact on human diets and activity patterns, such that calorie-dense food is often easily accessible and adequate nutrition can be achieved with a sedentary lifestyle. This state of affairs, which is typical in Western, industrialized societies but rapidly spreading across developing countries, stands in stark contrast to the ecological conditions experienced over the vast majority of our evolutionary history. Consequently, the “mismatch” between human physiology—which evolved to cope with a mixed plant- and meat-based diet, activity-intensive foraging, and periods of resource scarcity—and Western, industrialized lifestyles has been hypothesized to explain the current epidemic of cardiometabolic disease (1–4).
Attempts to test the evolutionary mismatch hypothesis thus far have largely focused on comparing cardiometabolic health outcomes between industrialized nations and small-scale, subsistence-level groups (e.g., hunter-gatherers, horticulturalists, and pastoralists). Arguably, the diets and activity patterns of these subsistence-level groups are relatively in line with their recent evolutionary history, and these populations can thus be thought of as “matched” to their evolutionary past (1, 5). In support of the evolutionary mismatch hypothesis, essentially, all subsistence-level populations studied to date show minimal type 2 diabetes, hypertension, obesity, and heart disease relative to the United States and Europe (5–13). Two other classes of studies provide further support: (i) Indigenous populations that have recently transitioned to market-based economies show higher rates of obesity and metabolic syndrome compared to subsistence-level groups [e.g., (14, 15)] and (ii) comparisons between rural and urban areas in developing countries have found higher rates of hypertension, type 2 diabetes, and obesity in the urban, industrialized setting (16–20).
Despite the groundwork that has been laid so far in understanding how Western lifestyles influence health, most prior studies leave two major gaps. First, the participants’ genetic backgrounds are either heterogeneous (in the case of urban versus rural comparisons within a country) or confounded with lifestyle (in the case of subsistence-level versus U.S. or Europe comparisons). This makes it difficult to disentangle genetic versus environmental contributions to health. A more robust study design would be to compare health between individuals living their ancestral, traditional way of life versus individuals from the same genetic background living a modern, industrialized lifestyle. This type of “natural experiment” is difficult to come by [but see (13)], and large-scale work that has assessed acculturation effects on cardiometabolic health within a single group has therefore been limited to more modest lifestyle gradients [e.g., work with the Tsimane (15, 17), Shuar (10), or Yakut (18, 21)]. A second major gap is that research to date has focused on industrialization and acculturation effects at particular life stages, mainly in adulthood, despite strong evidence that early-life conditions influence adult health and that life-course perspectives are likely important (19, 20). Of particular relevance is the hypothesis that individuals use cues during development to predict what the adult environment will be like and develop phenotypes well suited for those conditions. Under such a “predictive adaptive response” (PAR) framework, industrial transitions are thought to be especially detrimental because individuals may be born in resource-poor environments but exposed to resource-rich environments as adults; individuals are thus phenotypically “prepared for” scarcity but encounter plenty instead, leading to a within-lifetime environmental mismatch and subsequent cardiometabolic disease (22–24). Despite the popularity and potential significance of this idea, little work has robustly and empirically tested it against other evolutionary explanations for why early-life resource scarcity is commonly associated with poor adult cardiometabolic health (20, 25–28). In particular, the “developmental constraints” (DC) hypothesis alternatively predicts that early-life nutritional challenges will be unavoidably costly and associated with poor health outcomes no matter the adult environment (20, 25, 28).
To address these gaps, we collected interviews and cardiometabolic health biomarker data from the Turkana, a subsistence-level, pastoralist population from a remote desert in northwest Kenya (Fig. 1) (29, 30). The Turkana and their ancestors have practiced nomadic pastoralism in arid regions of East Africa for thousands of years (30), and present-day, traditional Turkana still rely on livestock for subsistence: 62% of calories are derived from fresh or fermented milk, and another 12% of calories come from animal meat, fat, or blood (29) [specifically, the Turkana herd dromedary camels, zebu cattle, fat-tailed sheep, goats, and donkeys (29)]. The remaining calories are derived from wild foods or products obtained through occasional trade (e.g., cereals, tea, and oil) (29). However, as infrastructure in Kenya has improved in the past few decades, small-scale markets have expanded into northwest Kenya, leading some Turkana to no longer practice nomadic pastoralism and to rely more heavily on the market economy; specifically, these individuals make and sell charcoal or woven baskets or keep animals in a fixed location for trade rather than subsistence. In addition to the emergence of this “nonpastoralist” (but still relatively subsistence-level) subgroup, some individuals have left the Turkana homelands entirely and now live in highly urbanized parts of central Kenya (Fig. 1). The Turkana situation thus presents a unique opportunity, in that individuals of the same genetic background can be found across a substantial lifestyle gradient ranging from relatively matched to extremely mismatched with their recent evolutionary history. Further, because many Turkana are currently migrating between rural and urban areas within their lifetime, we were able to empirically test the PAR hypothesis by asking whether individuals who experienced rural conditions in early life but urban conditions in adulthood exhibited worse cardiometabolic health than individuals whose early and adult environments were similar. We tested this idea against the DC hypothesis, which predicts that early-life challenges incur simple long-term costs that are not contingent on the adult environment (20, 25, 28).
Fig. 1. Sampling and dataset overview.
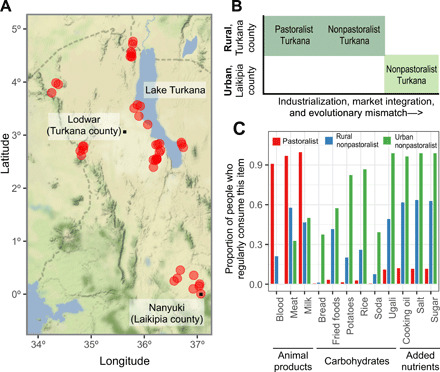
(A) Sampling locations throughout northern and central Kenya are marked with red dots; the county borders are marked with dashed lines. In both Laikipia and Turkana counties, the largest city (which is generally central within each county) is marked with a black dot. (B) Schematic describing the three lifestyle groups that were sampled as part of this study. (C) The proportion of people from each lifestyle group who reported that they consumed a particular item “regularly,” defined as “one to two times per week,” “more than two times per week,” or “every day.” People who reported that they consumed a particular item “rarely” or “never” were categorized as not consuming the item regularly. Animal products are a staple of the traditional pastoralist diet (85), while carbohydrates and added nutrients, which can only be obtained through trade, are indicative of market integration.
Capitalizing on this natural experiment, we sampled 1226 adult Turkana in 44 locations from the following groups: (i) individuals practicing subsistence-level pastoralism in the Turkana homelands, (ii) individuals that do not practice pastoralism but live in the same remote, rural area, and (iii) individuals living in urban centers (Fig. 1). We found that cardiometabolic profiles across 10 biomarkers were favorable in pastoralist Turkana, and rates of obesity and metabolic syndrome were low, similar to other subsistence-level populations (6–12). Comparisons within the Turkana revealed a nonlinear relationship between the extent of industrialization or evolutionary mismatch and cardiometabolic health: No significant biomarker differences were found between pastoralists and nonpastoralists from rural areas. However, we found strong, sometimes sex-specific, differences in health between these two groups and nonpastoralists living in urban areas, although metabolic dysfunction among urban Turkana did not reach the levels observed in the United States. Using formal mediation analyses (31, 32), we show that consumption of processed, calorically dense foods (primarily carbohydrates and cooking fats) and indices of market integration may explain health shifts in urban Turkana. Last, we show that a proxy of urbanization (population density) experienced around the time of birth is associated with worse adult cardiometabolic health, independent of adult lifestyle. In other words, the health costs of living an industrialized lifestyle in early life and adulthood are additive, such that within-lifetime environmental mismatches do not appear to exacerbate health issues as has been previously suggested (22, 24).
RESULTS
Traditional, pastoralist Turkana are at low risk for cardiometabolic disease
To characterize the health of the Turkana people, we collected extensive interview and biomarker data from adult Turkana sampled throughout Kenya (Table 1 and Fig. 1). We measured body mass index (BMI), waist circumference, total cholesterol, triglycerides, high- and low-density lipoproteins (HDLs and LDLs), body fat percentage, systolic and diastolic blood pressure, and blood glucose levels (Table 2). We also created a composite measure of health, defined as the proportion of measured biomarkers that exceed cutoffs set by the U.S. Centers for Disease Control and Prevention (CDC) or the American Heart Association as being indicative of disease (see Supplementary Materials and Methods).
Table 1. Sample sizes by age, sex, and lifestyle, as well as summary statistics for select variables indicating market integration.
NHANES, National Health and Nutrition Examination Survey; MI, market integration. M, Male; F, Female.
| Pastoralist | Nonpastoralist, rural | Nonpastoralist, urban | U.S. (NHANES) | |||||
| Age category | M | F | M | F | M | F | M | F |
| 18–30 | 16 | 23 | 69 | 160 | 69 | 39 | 656 | 882 |
| 31–40 | 36 | 38 | 68 | 115 | 66 | 41 | 391 | 431 |
| 41–50 | 40 | 40 | 67 | 87 | 41 | 19 | 399 | 399 |
| 51–60 | 15 | 9 | 61 | 65 | 29 | 17 | 311 | 313 |
| 61–69 | 1 | 0 | 27 | 26 | 22 | 12 | 168 | 180 |
| >70 | 1 | 1 | 15 | 8 | 11 | 6 | 0 | 0 |
| Select variables indicating MI | ||||||||
| Proportion who frequently use cooking oil |
0.05 | 0.05 | 0.50 | 0.31 | 0.88 | 0.94 | ||
| Proportion who frequently use salt |
0.05 | 0.03 | 0.38 | 0.48 | 0.95 | 0.95 | ||
| Proportion who frequently use sugar |
0.04 | 0.03 | 0.38 | 0.47 | 0.95 | 0.95 | ||
| Mean population density (persons per 5 km2) |
21.28 | 14.46 | 32.03 | 45.83 | 172.87 | 192.96 | ||
| SD of population density (persons per 5 km2) |
10.75 | 9.96 | 53.02 | 68.75 | 306.78 | 217.26 | ||
Table 2. Sample sizes for each biomarker of metabolic and cardiovascular health.
BP, blood pressure.
| Variable | n | Males | Females | Units |
| Waist circumference | 1204 | 515 | 689 | cm |
| BMI | 1198 | 511 | 687 | kg/m2 |
| Body fat | 835 | 367 | 468 | Percentage |
| Total cholesterol | 406 | 201 | 205 | mg/dl |
| HDL cholesterol | 406 | 201 | 205 | mg/dl |
| Triglycerides | 405 | 200 | 205 | mg/dl |
| LDL cholesterol | 353 | 171 | 182 | mg/dl |
| Blood glucose | 436 | 213 | 223 | mg/dl |
| Systolic BP | 1196 | 512 | 684 | mmHg |
| Diastolic BP | 1143 | 476 | 667 | mmHg |
| Composite | 1195 | 511 | 684 | Proportion |
As has been observed in other subsistence-level populations (5), we found extremely low levels of cardiometabolic disease among traditional, pastoralist Turkana: No individuals met the criteria for obesity (BMI > 30) or metabolic syndrome (33), and only 6.4% of individuals had hypertension [blood pressure > 135/85 (33)]. Further, across eight cardiometabolic biomarkers that have been measured consistently in other subsistence-level populations (6–12), the means observed in the Turkana were generally within the range of what others have reported (table S1, A and B). Mean body fat percentage (mean ± SD for females = 20.45 ± 4.57%) and BMI (19.99 ± 2.14 kg/m2) were on the lower extremes but were similar to other pastoralists (mean BMI in the Fulani and the Maasai = 20.2 and 20.7 kg/m2, respectively) and to a small study of the Turkana conducted in the 1980s [mean BMI = 17.7 kg/m2 (34)]. Notably, the only biomarkers that were strongly differentiated in traditional, pastoralist Turkana were HDL (72.69 ± 14.72 mg/dl) and LDL cholesterol levels (60.89 ± 20.22 mg/dl), both of which were even more favorable than what has been observed in other subsistence-level groups, including the Fulani and the Maasai (range of reported means for HDL = 34.45 to 49.11 mg/dl and LDL = 72.70 to 92.81 mg/dl). It remains to be seen why the Turkana HDL/LDL profiles appear as strong and consistent outliers relative to other subsistence-level groups, but one possibility is that there has been selection on Turkana lipid traits as a result of their unique diet, ecology, and lifestyle. This possibility could be explored in future evolutionary genetic and metabolic studies.
Pastoralist Turkana and rural nonpastoralist Turkana have similar biomarker profiles, while urban Turkana exhibit poorer metabolic health
Next, we sought to understand the shape of the relationship between industrialization and cardiometabolic health within the Turkana, by comparing biomarker values across the three lifestyle categories. Using linear models controlling for age and sex, we found that Turkana practicing traditional pastoralism did not differ in any of the 10 measured biomarkers relative to nonpastoralist Turkana living in similarly rural areas (all P values > 0.05; Fig. 2 and table S2A), despite there being major dietary difference between these groups (Fig. 1). Pastoralist and rural nonpastoralist Turkana did significantly differ in our composite measure, with nonpastoralist Turkana exhibiting more biomarker values above clinical cutoffs [average proportion of biomarkers above cutoffs = 4.02 and 6.82% for pastoralists and nonpastoralists, respectively; P value = 1.39 × 10−3; false discovery rate (FDR) < 5%; Fig. 2].
Fig. 2. Pastoralist and rural nonpastoralist Turkana have similar health profiles, while biomarkers of metabolic dysfunction are elevated in urban Turkana but not as extremely as in the U.S.
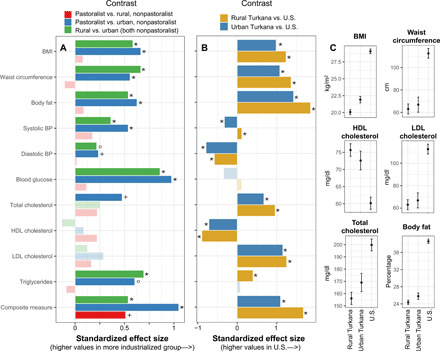
(A) Effect sizes for contrasts between pastoralist, rural nonpastoralist, and urban nonpastoralist Turkana (from linear models controlling for age and sex; table S2A). Effect sizes are standardized, such that the x axis represents the difference in terms of SDs between groups. BP, blood pressure. (B) Standardized effect sizes for contrasts between rural Turkana (pastoralist and rural nonpastoralist grouped together), urban nonpastoralist Turkana, and the U.S. (from linear models controlling for age and sex; table S2B). In (A) and (B), lighter colored bars represent effect sizes that were not significant [false discovery rate (FDR) > 5%], and analyses of body fat and blood glucose focus on females only (see Supplementary Materials and Methods). Symbols correspond to FDR significance thresholds as follows: *FDR < 0.1%, °FDR < 1%, and +FDR < 5%. (C) Predicted values for a typical rural Turkana (pastoralist and rural nonpastoralist grouped together), urban Turkana, and U.S. individual are shown for a subset of significant biomarkers. Estimates and error bars were obtained using coefficients and their SEs from fitted models, for a female of average age (see Supplementary Materials and Methods).
Notably, biomarker values for both pastoralist and nonpastoralist, rural Turkana were consistently more favorable than among Turkana living in urban areas in central Kenya. People living in urban areas exhibited composite measures indicative of worse cumulative cardiometabolic health (average proportion of biomarkers above cutoffs = 13.42%), higher BMIs and body fat percentages, larger waist circumferences, higher blood pressure, and higher levels of total cholesterol, triglycerides, and blood glucose (all FDR < 5%; Fig. 2 and table S2A). The only tested variables that did not exhibit differences between urban and rural Turkana (both pastoralists and nonpastoralists) were the HDL and LDL cholesterol levels, which were favorable in all Turkana regardless of lifestyle (tables S1A and S2A). Using standardized effect sizes, we found that the biomarkers that differed most between the two rural groups and urban residents were blood glucose, triglycerides, and BMI (Fig. 2). For example, the average urban Turkana resident has a 9.69 and 8.43% higher BMI relative to pastoralist and nonpastoralist rural Turkana, respectively.
For all 11 measures, we explored the possibility of age by lifestyle category and sex by lifestyle category interactions. We found no evidence that age modifies the response to lifestyle change (FDR > 5% for all biomarkers; likelihood ratio test comparing models with versus without the interaction term). However, we did find that inclusion of a sex by lifestyle category term improved the model fit for blood glucose levels (P value from a likelihood ratio test = 1.838 × 10−4) and body fat percentage (P value = 4.234 × 10−3; table S2A). In both cases, women experienced worse health with increasing market integration and industrialization, while men did not (fig. S1). The nature of this interaction is consistent with several previous studies (10, 21, 35, 36); however, the specific reasons behind the heightened sensitivity of women to acculturation (in our study and elsewhere) remain unknown. Previous work has speculated that these sex-specific effects are driven by social and behavioral factors that affect diet and activity patterns (e.g., rate of acquisition of wage labor jobs) and that change more markedly for women versus men during industrial transitions (10). It is likely that this general explanation applies to the Turkana as well, although follow-up work is needed to understand the specifics.
Biomarker profiles are more favorable among both rural and urban Turkana relative to the U.S.
We next asked whether the biomarker levels observed among urban Turkana approached those observed in a fully Western, industrialized society (specifically, the United States). We note that a caveat of these analyses is that they must include different genetic backgrounds since Turkana individuals are rarely found in fully industrialized countries.
To compare metabolic health between the U.S., rural Turkana (grouping pastoralists and nonpastoralists since these groups were minimally differentiated in previous analyses), and urban Turkana, we downloaded data from the CDC’s National Health and Nutrition Examination Survey (NHANES) conducted in 2006 (37), focusing on adults (ages 18-65) to recapitulate the age distribution of our Turkana dataset (see Table 1 for sample sizes). Comparisons between NHANES and our Turkana dataset revealed that, while urban Turkana exhibit biomarker values indicative of poorer health than rural Turkana, urban Turkana have more favorable metabolic profiles than the U.S. (Fig. 2, figs. S2 and S3, and table S2B). This pattern held for all measures except (i) blood glucose levels, where no differences were observed (P value for U.S. versus rural Turkana = 0.166, U.S. versus urban Turkana = 0.074); (ii) triglycerides, where urban Turkana could not be distinguished from the U.S. (P = 0.627); (iii) systolic blood pressure, where urban Turkana exhibited higher values than the U.S. (4.55% higher; P = 3.02 × 10−6; FDR < 5%); and (iv) diastolic blood pressure, where mean values for both urban and rural Turkana were unexpectedly higher than the U.S. (rural Turkana, 8.77% higher than U.S., P = 4.43 × 10−65; urban Turkana, 11.69% higher than U.S., P = 3.58 × 10−31, FDR < 5% for both comparisons; fig. S4). These differences in diastolic blood pressure remained after removing all U.S. individuals taking cardiometabolic medications (rural Turkana, 6.91% higher than U.S., P = 3.27 × 10−67; urban Turkana, 10.37% higher than U.S., P = 2.21 × 10−35). However, two pieces of evidence suggest that the higher diastolic blood pressure values observed in the Turkana are not pathological: (i) Values for rural Turkana (77.43 ± 15.22 mmHg) are similar to estimates from other subsistence-level populations without cardiometabolic disease (range of published means = 70.9 to 79.9 mmHg; table S1A) and (ii) few rural Turkana meet the criteria for hypertension relative to the U.S. (fig. S3). Future work is needed to understand the environmental and/or genetic sources of the observed differences in blood pressure between Turkana and U.S. individuals.
For measures that exhibited differences between urban Turkana and the U.S. in the expected directions, these effect sizes were consistently much larger in magnitude than the differences that we observed between rural and urban Turkana (Fig. 2). For example, while the average urban Turkana experiences a 9% higher BMI than their rural counterparts, the average U.S. individual has a BMI that is 44 and 32% higher than rural and urban Turkana, respectively. Similarly, while the average proportion of biomarkers above clinical cutoffs is 6.22% in rural Turkana and 13.42% in urban Turkana, this number rises to 38.84% in the U.S.
Health shifts in urban Turkana are weakly mediated by increased consumption of market-derived carbohydrates and processed foods and more strongly by indices of urbanicity
We next sought to identify the specific dietary, lifestyle, or environmental inputs that drive differential health outcomes between urban and rural Turkana. To do so, we turned to interview data collected for each individual (see Supplementary Materials and Methods), which revealed substantial variation in diet, market access, and “urbanicity” [a term that we use to mean living in an urbanized area and engaging in an urban lifestyle, following (38); Figs. 1 and 3]. We paired these interview data with mediation analyses (31, 32) to formally test whether the effect of a predictor (X) on an outcome (Y) was direct or, instead, indirectly explained by a third variable (M) such that X➔M➔Y (Fig. 3). Using this statistical framework, we tested whether the following factors could explain the decline in metabolic health observed in urban Turkana: increased consumption of market-derived, calorically dense foods (e.g., carbohydrates such as soda, bread, rice, as well as fats such as cooking oil), reduced consumption of traditional animal products, poorer health habits, ownership of more market-derived goods and modern amenities (e.g., cell phone, finished floor, and electricity), occupation that is more market integrated (e.g., formal employment), and residence in a more populated or developed area (measured via population density, distance to a major city, and female education levels) (see Supplementary Materials and Methods). In particular, we predicted that lifestyle effects on health would be mediated by a shift toward a diet that incorporates more carbohydrates and fewer animal products in urban Turkana. These analyses focused on biomarkers for which our sample sizes were the largest since dietary data were not available for all individuals (see table S3 for sample sizes).
Fig. 3. Urban-rural health differences are mediated by both dietary and other factors.
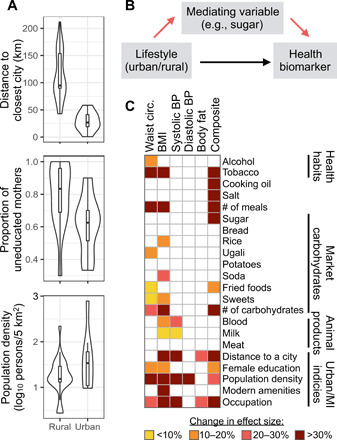
(A) Key measures of urbanicity and market integration used in mediation analyses, with means and distributions shown for urban and rural Turkana. (B) Schematic of mediation analyses. Specifically, mediation analyses test the hypothesis that lifestyle effects on health are explained by an intermediate variable, such as consumption of particular food items (red arrows); alternatively, lifestyle effects on health may be direct (black arrow) or mediated by a variable that we did not measure. (C) Summary of mediation analysis results, where colored squares indicate a variable that was found to significantly explain urban-rural health differences in a given biomarker. Significant mediators are colored on the basis of how much the lifestyle effect (urban/rural) decreased when a given mediator was included in the model. MI, market integration. Full results and sample sizes for mediation analyses are presented in table S3.
In support of our predictions, urban-rural differences in waist circumference, BMI, and our composite measure of health were mediated by greater consumption of processed, calorically dense foods (including carbohydrates and fats) and lower consumption of traditional animal products (milk and blood) in urban Turkana (Fig. 3 and table S3). Notably, the total number of different carbohydrate items that an individual consumed was a strong and consistent predictor across these three biomarkers, suggesting that individual dietary components may matter less than overall exposure to refined carbohydrates. However, it is important to note that refined carbohydrates are commonly processed with oil or other additives, and it is therefore likely a combined effect of exposure to both carbohydrates and fats that drives the negative health effects that we observe.
Contrary to our predictions, we did not find that dietary differences mediated urban-rural differences in systolic blood pressure, diastolic blood pressure, or body fat percentage. Instead, these measures were explained by variables that captured how industrialized and market-integrated a given individual’s lifestyle was, which was also important for waist circumference, BMI, and our composite measure of health in addition to dietary effects. For example, fine-scale measures of population density and degree of market reliance of occupation both significantly mediated five of six tested biomarkers (Fig. 3 and table S3). Further, these indices of urbanicity and market integration tended to be stronger mediators than dietary variables (Fig. 3).
To understand the degree to which the mediators that we identified explain the relationship between lifestyle and a given biomarker, we compared the magnitude of the lifestyle effect in our original models (controlling for age and sex, without any mediators) to the effect estimated in the presence of all significant mediators. If the mediators fully explain the relationship between lifestyle and a given biomarker, then we would expect the estimate of the lifestyle effect to be zero in the second model. These analyses revealed that the mediators that we identified explain most of the relationship between lifestyle and waist circumference (effect size decrease = 90.7%), BMI (79.9%), systolic blood pressure (74.9%), and composite health (64.1%) but explain only a small portion of lifestyle effects on diastolic blood pressure (10.0%) and body fat (23.5%; table S3).
Cumulative exposure to urban environments across the life course compromises metabolic health
Last, we were interested in understanding whether early-life conditions had long-term effects on health, above and beyond the effects of adult lifestyle that we had already identified. We were motivated to ask this question because work in humans and nonhuman animals has demonstrated strong associations between diet and ecology during the first years of life and fitness-related traits measured many years later (20, 25, 39, 40). Two major hypotheses have been proposed to explain why this “embedding” of early-life conditions into long-term health occurs. First, the PAR hypothesis posits that organisms adjust their phenotype during development in anticipation of predicted adult conditions. Individuals that encounter adult environments that “match” their early conditions are predicted to gain a selective advantage, whereas animals that encounter mismatched adult environments should suffer a fitness cost (19, 22, 25, 41, 42). In contrast, the DC or “silver spoon” hypothesis predicts a simple relationship between early environmental quality and adult fitness: Individuals born in high-quality environments experience a fitness advantage regardless of the adult environment (25, 28, 43). Under DC, poor-quality early-life conditions cannot be ameliorated by matching adult and early-life environments; instead, the effects of environmental adversity accumulate across the life course.
We found no evidence that individuals who experienced matched early-life and adult environments had better metabolic health in adulthood than individuals who experienced mismatched early-life and adult conditions (P > 0.05 for all biomarkers). In particular, we tested for interaction effects between the population density of each individual’s birth location (estimated for their year of birth) and a binary factor indicating whether the adult environment was urban or rural (table S4A; see table S4B for parallel analyses using population density to define the adult environment as a continuous measure). This analysis was possible given the within-lifetime migrations of many Turkana between urban and rural areas: Only 19.52 and 33.01% of urban and rural Turkana, respectively, were sampled within 10 km of their birthplace, and the correlation between birth and sampling location population densities was low (R2 = 0.115; P < 10−16; fig. S5).
While we observed no evidence for interaction effects supporting PAR, we did find strong main effects of early-life population density on adult waist circumference (b = 0.272, P = 7.33 × 10−8), BMI (b = 0.266, P = 1.35 × 10−7), body fat (b = 0.306, P = 1.57 × 10−5), diastolic blood pressure (b = 0.124, P = 2.01 × 10−2), and our composite measure of health (b = 0.296, P = 3.40 × 10−4; all FDR < 5%), in support of DC. For all biomarkers, being born in a densely populated location was associated with poorer adult metabolic health (Fig. 4 and table S4A). Furthermore, the early-life environment effect was on the same order of magnitude as the effect of living in an urban versus rural location in adulthood (see table S4A and Supplementary Materials and Methods). For example, BMIs are 5.69% higher in urban versus rural areas, while individuals born in areas from the 25th versus 75th percentile of the early-life population density distribution exhibit BMIs that differ by 3.34%. Similarly, the effect of the adult environment (urban compared to rural) on female body fat percentages is 11.14%, while the effect of early-life population density (25th compared to 75th percentile) is 20.7% (table S4A).
Fig. 4. Early-life population density predicts biomarkers of adult health.
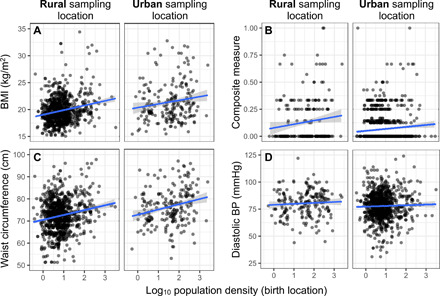
The relationship between the population density of each individual’s birth location and (A) BMI, (B) our composite measure of health, (C) waist circumference, and (D) diastolic blood pressure are shown for individuals sampled in rural and urban locations, respectively. Notably, while the intercept for a linear fit between early-life population density and each biomarker differs between rural and urban sampling locations (indicating mean differences in biomarker values as a function of adult lifestyle), the slope of the line does not. In other words, we find no evidence that the relationship between early-life conditions and adult health is contingent on the adult environmnt (as predicted by PAR). Instead, being born in an urban location predicts poorer metabolic health regardless of the adult environment.
DISCUSSION
By sampling a relatively endogamous population across a substantial lifestyle gradient, we show that (i) traditional, pastoralist Turkana exhibit low levels of cardiometabolic disease and (ii) increasing industrialization, in both early life and adulthood, has detrimental, additive effects on metabolic health (in opposition of popular PAR models that have rarely been tested empirically in humans) (20, 22, 24). Our findings offer strong support for the evolutionary mismatch hypothesis, more so than existing studies that cannot disentangle lifestyle and genetic background effects (6–12, 44, 45, 46) or that assess lifestyle effects across much more modest gradients (10, 17, 21, 47, 48). Our work also provides some of the first multidimensional, large-scale data on acculturation and industrialization effects on cardiometabolic health in pastoralists [see also (34, 49, 50)], which have received less attention than other subsistence modes [e.g., horticulturalists such as the Shuar and Tsimane (10, 15, 51, 52)].
Our observation that pastoralist Turkana do not suffer from cardiometabolic diseases echoes long-standing findings from other subsistence-level groups (6–12). However, it also provides empirical support for a more recent and controversial hypothesis: that many types of mixed plant- and meat-based diets are compatible with cardiometabolic health (1, 53, 54) and that mismatches between the distant human hunter-gatherer past and the subsistence-level practices of horticulturalists or pastoralists do not lead to disease (55). In other words, contemporary hunter-gatherers are most aligned with human subsistence practices that evolved ~300 thousand years ago (56), but they do not exhibit better cardiometabolic health relative to horticulturalists or pastoralists, whose subsistence practices evolved ~12 thousand years ago (tables S1A to S3) (57). Instead, we find evidence consistent with the idea that extreme mismatches between the recent evolutionary history of a population and lifestyle are needed to produce the chronic diseases now prevalent worldwide; in the Turkana, this situation appears to manifest in urban, industrialized areas but not in rural areas with changing livelihoods but limited access to the market economy.
Because our study assessed health in individuals who experience no, limited, or substantial access to the market economy, we were able to determine that industrialization has nonlinear effects on health in the Turkana. In particular, we find no differences between pastoralists and nonpastoralists in rural areas for 10 of 11 variables (Fig. 2), despite nonpastoralists consuming processed carbohydrates that are atypical of traditional practices (Fig. 1 and table S1A). Nevertheless, rural nonpastoralists still live in remote areas, engage in activity-intensive subsistence activities, and rely far less heavily on markets than urban Turkana. Given the mosaic of lifestyle factors that can change with modernization, often in concert, our results suggest that this type of lifestyle has not crossed the threshold necessary to produce cardiometabolic health issues. This “threshold model” may help explain heterogeneity in previous studies, where small degrees of evolutionary mismatch and market integration have produced inconsistent changes in cardiometabolic health biomarkers (10, 58, 59).
While our dataset does not capture every variable that mediates urban-rural health differences in the Turkana, we were able to account for a substantial portion (>60%) of lifestyle effects on waist circumference, BMI, systolic blood pressure, and composite health. In line with our expectations, increases in these biomarkers in urban areas were mediated by greater reliance on processed, calorically dense foods (i.e., refined carbohydrates and cooking fats) and reduced consumption of animal products (Figs. 1 and 3). However, our mediation analyses also show that broader measures of lifestyle modernization (e.g., population density, distance to a major city, and female education levels) have stronger explanatory power than diet alone. It is likely that these indices serve as proxies for unmeasured, more proximate mediators, such as psychosocial stress, nutrient balance, total caloric intake, or total energy expenditure, all of which vary by industrialization and can affect health (1, 12, 60–63). The fact that the number of meals eaten per day (which is typically one in rural areas and two to three in urban areas) was a strong mediator for three of six variables points to total caloric intake, while the importance of occupation suggests activity levels are also probably key. More generally and as expected, our mediation analyses suggest that the link between lifestyle change and health is complex, multifactorial (e.g., driven by a suite of dietary and other factors), and potentially quite different for different biomarkers. Work with the Turkana is under way to address some of the unmeasured sources of variance that we hypothesize to be especially critical, namely, total caloric intake and total energy expenditure.
In addition to the pervasive influence of adult lifestyle on metabolic physiology that we observe in the Turkana, our analyses also revealed appreciable effects of early-life environments. In particular, controlling for the adult environment (urban or rural), birth location population density was a significant predictor of BMI, waist circumference, diastolic blood pressure, our composite measure of health, and body fat. Further, the impact of early-life and adult conditions appears to be on the same order of magnitude, although they do, in some cases, vary up to twofold. In particular, we observed 2 to 6% differences in BMI, waist circumference, and diastolic blood pressure, as a function of each life stage, while body fat and our composite measure show changes in the 11 to 20% range (note, however, that the measures that we used to quantify early-life and adult conditions are not the same, making direct effect size comparisons difficult; table S4A).
We did not find evidence that individuals who grew up in rural versus urban conditions were more prepared for these environments later in life, as predicted by PAR. These findings agree with work in preindustrial human populations and long-lived mammals, which have found weak or no support for PARs (26, 27, 64–66). Together, these findings suggest that because early-life ecological conditions are often a poor predictor of adult environments for long-lived organisms, a strategy “matching” individual physiology to an unpredictable adult environment is unlikely to evolve (67–69). Instead, our work joins others in concluding that challenging early-life environments simply incur long-term health costs (26, 27, 64–66). While previous work in subsistence-level groups has clearly shown an effect of early-life environments (including acculturation) on health outcomes (52, 59), it has not explicitly tested whether PAR versus DC models explain these associations. Our attempt to do so here in the context of urbanicity exposure suggests that rapid industrial transitions are unlikely to create health problems because of within-lifetime environmental mismatches (26, 27, 70). Instead, our findings suggest that greater cumulative exposure to urban, industrialized environments across the life course will create the largest burdens of cardiometabolic disease.
Limitations
Our study has several limitations. First, with the exception of our biomarker measurements, most of our data are self-reported. It is possible that recall may be imperfect, answers may be exaggerated to appear impressive (e.g., in interviews about the ownership of market goods), or participants may not wish to reveal private details (e.g., in interviews about health habits or covariates). On the basis of our conversations with study participants, we expect intentionally provided misinformation to be rare, but there are two areas where self-reporting contributes to specific limitations worthy of discussion. First, because our age data are self-reported, this key covariate is likely noisy and more so for rural than for urban participants (who are more likely to know their exact birth date). We do not have a reason to believe that this issue affects our estimate of the lifestyle-cardiometabolic health relationship, but it does likely complicate our ability to identify age by lifestyle interactions, which is a critical area for future study. Second, because our diet data are self-reported, we are currently unable to tease apart the precise nutritional components that drive health variation in the Turkana. Our mediation analyses reveal that several market-derived foods are key contributors, suggesting that broad exposure to processed, high-energy foods (including both carbohydrates and fats) is important for cardiometabolic health. Future work that estimates the total caloric intake and the intake of fat, protein, carbohydrates, and micronutrients [as in (71)] in the Turkana are planned.
A second limitation is that we do not know how our biomarker values are related to outcomes such as heart disease or mortality in the Turkana. We are relying on work in Western cohorts that has related lipid profiles, blood glucose, blood pressure, and measures of adiposity to these outcomes (72), but it is possible that those relationships are different in the Turkana [e.g., work in the United States has already demonstrated how the shape of the BMI-mortality curve may differ by race/ethnicity (73)]. Further, certain biomarkers may not linearly track disease and mortality risk: Notably, in Western cohorts, the effect of BMI on all-cause mortality risk is J shaped, such that underweight individuals experience some increase in risk, normal BMI individuals experience the lowest risk, and overweight and obese individuals experience the greatest risk (74). It is therefore difficult to draw conclusions about the relationship between any one biomarker, lifestyle change, and long-term outcomes in the present study. However, two pieces of evidence suggest that the changing biomarker profiles that we observe in urban Turkana are meaningful. First, work in Western countries has consistently shown that when individuals simultaneously cross clinical thresholds for several biomarkers, as we observe in urban Turkana, risk of cardiovascular events and all-cause mortality increases (72). Second, Kenya has seen a marked rise in CVD in recent decades, with 13% of hospital deaths in 2014 attributed to CVD. CVD risk is much higher in urban relative to rural areas across the country, with hypertension and type 2 diabetes estimated to be at least fourfold more prevalent in urban settings (75).
Last, a third limitation of our study is that we lack data on several key factors known to modify or mediate the relationship between lifestyle change and cardiometabolic health, such as total energy expenditure (1), total caloric intake and nutritional composition of the diet (71), and parasite load (76). Our ongoing research with the Turkana is in the process of gathering data on these sources of variance.
Conclusions
The hypothesis that mismatches between evolved human physiology and Western lifestyles cause disease has become a central tenet of evolutionary medicine, with potentially profound implications for how we study, manage, and treat a long list of conditions thought to arise from evolutionary mismatch (77). However, this hypothesis has been difficult to robustly test in practice because of inadequate population comparisons and the multiple types of mismatch to be considered. Leveraging the lifestyle change currently occurring in the Turkana population, we show that cardiometabolic health is worse in urban relative to rural areas but that small deviations from traditional, ancestral practices in rural areas do not produce health effects. To build upon our results, we advocate for more within-population comparisons spanning large lifestyle gradients, combined with longitudinal sampling designs [e.g., (72)]. Longitudinal study of other populations undergoing industrial transitions would also be invaluable for assessing the generality of the early-life effects on adult cardiometabolic health that we observe here and for identifying the specific early-life ecological, social, or behavioral factors that drive long-term variation in health.
MATERIALS AND METHODS
Experimental design and sampling overview
Data were collected between April 2018 and March 2019 in Turkana and Laikipia counties in Kenya. During this time, researchers visited locations where individuals of Turkana ancestry were known to reside (Fig. 1). At each sampling location, healthy adults (>18 years old) of self-reported Turkana ancestry were invited to participate in the study, which involved a structured interview and measurement of 10 cardiometabolic biomarkers. Participation rates of eligible adults were high (>75%). GPS coordinates were recorded on a handheld Garmin GPSMAP 64 device at each sampling location. Additional details on the sampling procedures can be found in Supplementary Materials and Methods.
This study was approved by Princeton University’s Institutional Review Board for Human Subjects Research (Institutional Review Board no. 10237) and Maseno University’s Ethics Review Committee (MSU/DRPI/MUERC/00519/18). We also received county-level approval from both Laikipia and Turkana counties for research activities and research permits from Kenya’s National Commission for Science, Technology, and Innovation (NACOSTI/P/18/46195/24671). Written, informed consent was obtained from all participants after the study goals, sampling procedures, and potential risks were discussed with community elders and explained to participants in their native language (by both a local official, usually the village chief, and by researchers or field assistants).
Data filtering and processing
Individuals were excluded from analyses if they met any of the following criteria: (i) pregnancy, (ii) extreme outlier values for a given biomarker, (iii) missing data on primary subsistence activity, (iv) missing interview data, and (v) missing gender or age. For the early-life effects analyses, we also excluded individuals that did not report a birth location or for whom GPS coordinates for the reported birth location could not be identified on a map. Those missing birth locations had similar health profiles as individuals for whom a birth location could be assigned (all FDR > 5% for linear models testing for an effect of birth location missingness on each biomarker, controlling for age, sex, and lifestyle; table S4C). Thus, although the sample sizes for our early-life effects analyses are smaller than for analyses focused on current environmental/lifestyle effects (table S4A), this sample size reduction is not systematically biased in a way that is likely to affect the results.
Before statistical analyses, all biomarkers (except the composite measure of health) were mean centered and scaled by their SD, using the “scale” function in R (78). Consequently, all reported effect sizes are standardized and represent the effect of a given variable on the outcome in terms of increases in SDs.
Statistical analyses
Testing for lifestyle effects on measured biomarkers
For each of the 10 measured biomarkers, we used the following linear model to test for effects of lifestyle controlling for covariates
| (1) |
where yi is the normalized (mean centered and scaled by the SD) biomarker value for individual i, li is lifestyle (pastoralist; nonpastoralist, rural; or nonpastoralist, urban), ai is age (in years), si is sex (male or female), and ei represents residual error. To determine whether a given biomarker exhibited a lifestyle by sex interaction, we used a likelihood ratio test to compare the fit of model 1 with the following model
| (2) |
In model 2, βl × s represents a lifestyle by sex interaction effect. If the P value for the likelihood ratio test comparing models 1 and 2 was less than 0.05, then we concluded that a lifestyle by sex interaction existed, and we tested for the effects of lifestyle within each gender separately (controlling for age). These analyses revealed lifestyle associations with body fat and blood glucose in females but not males (fig. S1). All additional analyses for these biomarkers therefore analyzed data from females alone.
Analyses of blood glucose levels included a covariate noting whether the individual had fasted overnight before the time of blood collection (which was always in the morning). For analyses of the composite measure of health, we used the same approach and the same main and interaction effects described for models 1 and 2 paired with generalized linear models with a binomial link function to accommodate count data. Specifically, the composite measure of health was modeled as the number of biomarkers that exceeded clinical cutoffs/the number of biomarkers measured for a given individual. Only individuals with three or more measured biomarkers were included in this analysis.
For all 11 measures (10 biomarkers and 1 composite measure), we extracted the P values associated with the lifestyle effect (βl) from our models and corrected for multiple hypothesis testing using a Benjamini-Hochberg FDR (79). We considered a given lifestyle contrast to be significant if the FDR-corrected P value was less than 0.05 (equivalent to a 5% FDR threshold). The results of all final models are presented in table S2A.
Identifying factors that mediate lifestyle effects on measured biomarkers
For each biomarker that was significantly associated with lifestyle (table S2A), we were interested in identifying specific variables that mediated urban-rural differences in health. However, we did not perform mediation analyses for lipid traits and for blood glucose, as sample sizes for these biomarkers were smaller to begin with, and, after overlapping with our dietary data, we could only include 50 to 60 urban individuals. Therefore, we focused mediation analyses on waist circumference, BMI, diastolic and systolic blood pressure, body fat, and our composite measure of health (sample sizes for mediation analyses are presented in table S3).
To implement mediation analyses, we used an approach similar to (80, 81) to estimate the indirect effect of lifestyle on a given biomarker through the following potential mediating variables: alcohol and tobacco use (yes/no); consumption of meat, milk, blood, cooking oil, sugar, salt, bread, rice, ugali, potatoes, soda, fried foods, and sweets (frequency of use measured on a 0 to 4 scale); total number of unique carbohydrate items consumed; number of meals eaten per day; distance to the nearest city (in kilometers); log10 population density; main subsistence activity; proportion of mothers in the sampling location with no formal education; and a tally of the number of market-derived amenities an individual had (see Supplementary Materials and Methods). Occupation was coded to reflect integration in the market economy as follows: 0 = animal keeping, farming, fishing, hunting, and gathering; 1 = charcoal burning and mat making; 2 = casual worker, petty trade, and self-employment; and 3 = formal employment. To estimate female education levels in a given area, we calculated the fraction of women sampled in a given area with >0 children who reported having received no education. Population density, distance to a city, the proportion of mothers with no formal education, and the number of owned market goods are all measures that have been used in the literature to describe how urban a given individual/location is (82, 83). Population density estimates were derived from NASA’s Socioeconomic Data and Applications Center (https://doi.org/10.7927/H49C6VHW). Specifically, we used the Gridded Population of the World database (version 4.11) to estimate the number of persons per square kilometer for each sampling location based on our GPS coordinates (see the Supplementary Materials).
For all mediation analyses, we used two categories to describe lifestyle, urban and rural, given minimal health differences between pastoralist and nonpastoralist Turkana living in rural areas. For biomarkers with no sex by lifestyle interaction, we estimated the strength of the indirect effect of each mediator as the difference between the effect of lifestyle (urban versus rural) in two linear models: the “unadjusted” model that did not account for the mediator (equivalent to model 1 in the “Testing for lifestyle effects on measured biomarkers” section) and the effect of lifestyle in an “adjusted” model that also incorporated the mediator. If a given variable is a strong mediator, then the effect of lifestyle will decrease when this variable is included in the model and absorbs variance otherwise attributed to lifestyle. For each biomarker, the adjusted model was implemented as follows
| (3) |
Where βm represents the effect of the potential mediator on the outcome variable (all other variables are as defined in model 1). For body fat percentage, which displayed lifestyle effects in females but not males (fig. S1), we modeled females only and removed the sex term (βs) from models 1 and 3. For the composite measure of health, we used generalized linear models with a binomial link function instead of linear models.
To assess the significance of each mediating variable, we estimated the decrease in βl in model 1 relative to model 3 across 1000 iterations of bootstrap resampling. We deemed a variable to be a significant mediator if the lower bound of the 95% confidence interval (for the decrease in βl) did not overlap with 0. As a measure of effect size, we report the proportion of 1000 bootstrap resampling iterations for which the effect of lifestyle (βl) was reduced when the potential mediating variable was included in the model (table S3); a proportion of >0.975 is equivalent to a 95% confidence interval that does not overlap with 0. As another measure of effect size, we report the percent change in the lifestyle effect estimated from model 1 relative to model 3 for each biomarker-mediator pair (without bootstrapping and using the full dataset to estimate each effect size; Fig. 3). Further, to understand the degree to which the total set of mediators that we identified explain the relationship between lifestyle and a given biomarker, we report the percent change in effect size for the lifestyle effect estimated from model 1 versus a model that included all the same covariates and all significant mediators for a given biomarker.
Testing for early-life effects on biomarkers of adult health
Several research groups (26, 27, 64, 65, 84) have operationalized the PAR and DC hypotheses by asking whether there is evidence for interaction effects between early-life and adult environments (in support of PAR) or whether early-life adversity is instead consistently associated with compromised adult health (in support of DC). We took a similar approach to disentangle these hypotheses. For biomarkers with no sex and lifestyle interaction, we first asked whether there was any evidence for interaction effects between adult lifestyle and lifestyle/urbanicity during early life using the following linear model
| (4) |
where di represents the log10 population density for the birth location of individual i during their birth year, li represents adult lifestyle (urban versus rural), and βl×d captures the interaction effect between these two variables. For the two variables with lifestyle effects on health in females but not males (body fat percentage and blood glucose levels), we modeled females only and removed the sex term (βs). For the composite measure of health, we used generalized linear models with a binomial link function instead of linear models. For each of the 11 models, we extracted the P value associated with βl×d and corrected for multiple hypothesis testing (79). In all cases, the nominal and FDR-corrected P value was >0.05, suggesting that PARs do not explain early-life effects on health in the Turkana.
Next, we reran the appropriate version of model 4 for each measure after removing the interaction effect (βl×d). For biomarkers with no sex and lifestyle interaction, this model was equivalent to
| (5) |
For each of the 11 models, we extracted the P value associated with the early-life effect (βd) and corrected for multiple hypothesis testing (79). We considered a given variable to show support for the DC hypothesis if the FDR-corrected P value was less than 0.05. Results for all models described in this section are presented in table S4A. Results for parallel analyses that use log10 population density for the sampling location to define the adult environment (rather than a binary urban/rural lifestyle variable) are presented in table S4B. All statistical analyses were performed in R (78).
Supplementary Material
Acknowledgments
We thank S. Lowasa, C. Waigwa, and F. Lotukoi for invaluable assistance with data collection, as well as D. M. Mukhongo, S. Ngatia, E. Loowoth, and B. Mbau for contributions to the project. We thank M. Ndegwa for expert database and project management and J. Orina and D. Rubenstein for logistical help in Kenya. We are also grateful to the staff of Mpala Research Centre for essential support, especially F. Hassan, C. Nzomo, B. Wanjohi, G. Chege, T. Maina, and J. Nakolonyo. Last, thanks to the Ayroles and Graham laboratories, as well as J. Altmann, for feedback on this work. Funding: This work was supported by an award to J.F.A. through Princeton University’s Dean for Research Innovations Funds and the Mpala funds. A.J.L. was supported by a postdoctoral fellowship from the Helen Hay Whitney Foundation. Author contributions: A.J.L., D.M., and J.F.A. designed the research. A.J.L., D.M., J.K., and J.F.A. performed the research. A.J.L. analyzed data. A.J.L., M.G., and J.F.A. wrote the paper, with contributions from all coauthors. Competing interests: The authors declare that they have no competing interests. Data and materials availability: Deidentified data underlying the main analyses presented in this paper (contrasting cardiometabolic biomarker values across a lifestyle gradient) have been deposited in Dryad in the following location: https://doi.org/10.5061/dryad.2z34tmpjk. All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
SUPPLEMENTARY MATERIALS
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/6/43/eabb1430/DC1
REFERENCES AND NOTES
- 1.Pontzer H., Wood B. M., Raichlen D. A., Hunter-gatherers as models in public health. Obes. Rev. 19, 24–35 (2018). [DOI] [PubMed] [Google Scholar]
- 2.Manus M. B., Evolutionary mismatch. Evol. Med. Public Heal. 2018, 190–191 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.W. Leonard, in Evolution in Health and Disease, S. Stearns, J. Koella, Eds. (Oxford Univ. Press, 2010), pp. 265–276. [Google Scholar]
- 4.Gluckman P. D., Low F. M., Buklijas T., Hanson M. A., Beedle A. S., How evolutionary principles improve the understanding of human health and disease. Evol. Appl. 4, 249–263 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eaton S. B., Konner M., Shostak M., Stone agers in the fast lane: Chronic degenerative diseases in evolutionary perspective. Am. J. Med. 84, 739–749 (1988). [DOI] [PubMed] [Google Scholar]
- 6.Glew R. H., Williams M., Conn C. A., Cadena S. M., Crossey M., Okolo S. N., VanderJagt D. J., Cardiovascular disease risk factors and diet of fulani pastoralists of northern Nigeria. Am. J. Clin. Nutr. 74, 730–736 (2001). [DOI] [PubMed] [Google Scholar]
- 7.Kaplan H., Thompson R. C., Trumble B. C., Wann L. S., Allam A. H., Beheim B., Frohlich B., Sutherland M. L., Sutherland J. D., Stieglitz J., Rodriguez D. E., Michalik D. E., Rowan C. J., Lombardi G. P., Bedi R., Garcia A. R., Min J. K., Narula J., Finch C. E., Gurven M., Thomas G. S., Coronary atherosclerosis in indigenous South American Tsimane: A cross-sectional cohort study. Lancet 389, 1730–1739 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Leonard W. R., Crawford M. H., Comuzzie A. G., Sukernik R. I., Correlates of low serum lipid levels among the Evenki herders of Siberia. Am. J. Hum. Biol. 6, 329–338 (1994). [DOI] [PubMed] [Google Scholar]
- 9.Mbalilaki J. A., et al. , Daily energy expenditure and cardiovascular risk in Masai, rural and urban Bantu Tanzanians. Br. J. Sports Med. 44, 121–126 (2010). [DOI] [PubMed] [Google Scholar]
- 10.Liebert M. A., Snodgrass J. J., Madimenos F. C., Cepon T. J., Blackwell A. D., Sugiyama L. S., Implications of market integration for cardiovascular and metabolic health among an indigenous Amazonian Ecuadorian population. Ann. Hum. Biol. 40, 228–242 (2013). [DOI] [PubMed] [Google Scholar]
- 11.Glew R. H., Chuang L.-T., Berry T., Okolie H., Crossey M. J., VanderJagt D. J., Lipid profiles and trans fatty acids in serum phospholipids of semi-nomadic fulani in Northern Nigeria. J. Health Popul. Nutr. 28, 159–166 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Raichlen D. A., Pontzer H., Harris J. A., Mabulla A. Z. P., Marlowe F. W., Snodgrass J. J., Eick G., Berbesque J. C., Sancilio A., Wood B. M., Physical activity patterns and biomarkers of cardiovascular disease risk in hunter-gatherers. Am. J. Hum. Biol. 29, e22919 (2017). [DOI] [PubMed] [Google Scholar]
- 13.P. Baker, J. Hanna, T. Baker, The Changing Somoans: Behavior and Health in Transition (Oxford Univ. Press, 1986). [Google Scholar]
- 14.Mbanya J. C. N., Motala A. A., Sobngwi E., Assah F. K., Enoru S. T., Diabetes in sub-saharan Africa. Lancet 375, 2254–2266 (2010). [DOI] [PubMed] [Google Scholar]
- 15.Gurven M., Stieglitz J., Trumble B., Blackwell A. D., Beheim B., Davis H., Hooper P., Kaplan H., The Tsimane Health and Life History Project: Integrating anthropology and biomedicine. Evol. Anthropol. 26, 54–73 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lagranja E. S., Phojanakong P., Navarro A., Valeggia C. R., Indigenous populations in transition: An evaluation of metabolic syndrome and its associated factors among the Toba of northern Argentina. Ann. Hum. Biol. 42, 84–90 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gurven M., Blackwell A. D., Rodríguez D. E., Stieglitz J., Kaplan H., Does blood pressure inevitably rise with age?: Longitudinal evidence among forager-horticulturalists. Hypertension 60, 25–33 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Josh Snodgrass J., Health of indigenous Circumpolar populations. Ann. Rev. Anthropol. 42, 69–87 (2013). [Google Scholar]
- 19.Bateson P., Barker D., Clutton-Brock T., Deb D., D’Udine B., Foley R. A., Gluckman P., Godfrey K., Kirkwood T., Lahr M. M., Namara J. M., Metcalfe N. B., Monaghan P., Spencer H. G., Sultan S. E., Developmental plasticity and human health. Nature , 419–421 (2004). [DOI] [PubMed] [Google Scholar]
- 20.Lea A. J., Archie E. A., Tung J., Alberts S. C., Developmental plasticity: Bridging research in evolution and human health. Evol. Med. Public Health 2017, 162–175 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Snodgrass J. J., Leonard W. R., Sorensen M. V., Tarskaia L. A., Alekseev V. P., Krivoshapkin V., The emergence of obesity among indigenous Siberians. J. Physiol. Anthropol. 25, 75–84 (2006). [DOI] [PubMed] [Google Scholar]
- 22.Gluckman P. D., Hanson M. T., Spencer H. G., Predictive adaptive responses and human evolution. Trends Ecol. Evol. 20, 527–533 (2005). [DOI] [PubMed] [Google Scholar]
- 23.Barker D. J. P., Gluckman P. D., Godfrey K. M., Harding J. E., Owens J. A., Robinson J. S., Fetal nutrition and cardiovascular disease in adult life. Lancet 341, 938–941 (1993). [DOI] [PubMed] [Google Scholar]
- 24.Gluckman P. D., Hanson M. A., Low F. M., Evolutionary and developmental mismatches are consequences of adaptive developmental plasticity in humans and have implications for later disease risk. Philos. Trans. R Soc. B Biol. Sci. 374, 20180109 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Monaghan P., Early growth conditions, phenotypic development and environmental change. Philos. Trans. R. Soc. Lond. B Biol. Sci. 363, 1635–1645 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hayward A. D., Lummaa V., Testing the evolutionary basis of the predictive adaptive response hypothesis in a preindustrial human population. Evol. Med. Public Health 2013, 106–117 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hayward A. D., Rickard I. J., Lummaa V., Influence of early-life nutrition on mortality and reproductive success during a subsequent famine in a preindustrial population. Proc. Natl. Acad. Sci. U.S.A. 110, 13886–13891 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.A. Grafen, in Reproductive success, T. H. Clutton-Brock, Ed. (University of Chicago Press, 1988), pp. 454–471. [Google Scholar]
- 29.Galvin K., Nutritional ecology of pastoralists in dry tropical Africa. Am. J. Hum. Biol. 4, 209–221 (1992). [DOI] [PubMed] [Google Scholar]
- 30.Lamphear J., The people of the grey bull: The origin and expansion of the Turkana. J. Afr. Hist. 29, 27–39 (1988). [Google Scholar]
- 31.Mackinnon D. P., Fairchild A. J., Fritz M. S., Mediation analysis. Annu. Rev. Psychol. 58, 593–614 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Baron R., Kenny D., The moderator-mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. J. Pers. Soc. Psychol. 51, 1173–1182 (1986). [DOI] [PubMed] [Google Scholar]
- 33.Grundy S. M., Cleeman J. I., Daniels S. R., Donato K. A., Eckel R. H., Franklin B. A., Gordon D. J., Krauss R. M., Savage P. J., Smith S. C. Jr., Spertus J. A., Costa F.; American Heart Association; National Heart, Lung, and Blood Institute , Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation 112, 2735–2752 (2005). [DOI] [PubMed] [Google Scholar]
- 34.Corbett S., Gray S., Campbell B., Leslie P. W., Comparison of body composition among settled and nomadic Turkana of Kenya. Ecol. Food Nutr. 42, 193–212 (2003). [Google Scholar]
- 35.Price A. J., Crampin A. C., Amberbir A., Kayuni-Chihana N., Musicha C., Tafatatha T., Branson K., Lawlor D. A., Mwaiyeghele E., Nkhwazi L., Smeeth L., Pearce N., Munthali E., Mwagomba B. M., Mwansambo C., Glynn J. R., Jaffar S., Nyirenda M., Prevalence of obesity, hypertension, and diabetes, and cascade of care in sub-Saharan Africa: A cross-sectional, population-based study in rural and urban Malawi. Lancet Diabetes Endocrinol. 6, 208–222 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.NCD Risk Factor Collaboration (NCD-RisC) , Rising rural body-mass index is the main driver of the global obesity epidemic in adults. Nature 569, 260–264 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.National Health and Nutrition Examination Survey Data from the Centers for Disease Control and Prevention (CDC) and the National Center for Health Statistics (NCHS) (2019) (Hyattsville, MD).
- 38.McDade T. W., Adair L. S., Defining the “urban” in urbanization and health: A factor analysis approach. Soc. Sci. Med. 53, 55–70 (2001). [DOI] [PubMed] [Google Scholar]
- 39.Moore S. E., Cole T. J., Poskitt E. M. E., Sonko B. J., Whitehead R. G., McGregor I. A., Prentice A. M., Season of birth predicts mortality in rural Gambia. Nature 388, 434 (1997). [DOI] [PubMed] [Google Scholar]
- 40.Roseboom T., de Rooij S., Painter R., The Dutch famine and its long-term consequences for adult health. Early Hum. Dev. 82, 485–491 (2006). [DOI] [PubMed] [Google Scholar]
- 41.Gluckman P. D., Hanson M. A., Spencer H. G., Bateson P., Environmental influences during development and their later consequences for health and disease: Implications for the interpretation of empirical studies. Proc. R. Soc. B. 272, 671–677 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gluckman P. D., Hanson M. A. T., Living with the past: Evolution, development, and patterns of disease. Science 305, 1733–1736 (2004). [DOI] [PubMed] [Google Scholar]
- 43.Lindström J., Early development and fitness in birds and mammals. Trends Ecol. Evol. 14, 343–348 (1999). [DOI] [PubMed] [Google Scholar]
- 44.Ebrahim S., Kinra S., Bowen L., Andersen E., Ben-Shlomo Y., Lyngdoh T., Ramakrishnan L., Ahuja R. C., Joshi P., Das S. M., Mohan M., Smith G. D., Prabhakaran D., Reddy K. S.; Indian Migration Study group , The effect of rural-to-urban migration on obesity and diabetes in India: A cross-sectional study. PLOS Med. 7, 1000268 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Teo K., Chow C. K., Vaz M., Rangarajan S., Yusuf S.; PURE Investigators-Writing Group , The Prospective Urban Rural Epidemiology study: Examining the impact of societal influences on chronic noncommunicable diseases in low-, middle-, and high-income countries. Am. Heart J. 158, 1–7.e1 (2009). [DOI] [PubMed] [Google Scholar]
- 46.Silva H. P., Padez C., Moura E. A. F., Filgueiras L. A., Obesity, hypertension, social determinants of health and the epidemiologic transition among traditional Amazonian populations. Ann. Hum. Biol. 43, 371–381 (2016). [DOI] [PubMed] [Google Scholar]
- 47.Dressler W. W., Mata A., Chavez A., Viteri F. E., Arterial blood pressure and individual modernization in a Mexican community. Soc. Sci. Med. 24, 679–687 (1987). [DOI] [PubMed] [Google Scholar]
- 48.Dressler W. W., Modernization, stress, and blood pressure: New directions in research. Hum. Biol. 71, 583–605 (1999). [PubMed] [Google Scholar]
- 49.Little M. A., Lessons learned from the south Turkana ecosystem project. Hum. Ecol. Spec. Issue No. 10, 137–149 (2001). [Google Scholar]
- 50.E. Fratkin, E. A. Roth, As Pastoralists Settle: Social, Health, and Economic Consequences of Pastoral Sedentarization in Marsabit District, Kenya (Kluwer Academic Publishers, 2004). [Google Scholar]
- 51.Blackwell A. D., Pryor G. III, Pozo J., Tiwia W., Sugiyama L. S., Growth and market integration in Amazonia: A comparison of growth indicators between Shaur, Shiwiar, and Nonindigenous school children. Am. J. Hum. Biol. 21, 161–171 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Urlacher S. S., Liebert M. A., Snodgrass J. J., Blackwell A. D., Cepon-Robins T. J., Gildner T. E., Madimenos F. C., Amir D., Bribiescas R. G., Sugiyama L. S., Heterogeneous effects of market integration on sub-adult body size and nutritional status among the Shuar of Amazonian Ecuador. Ann. Hum. Biol. 43, 316–329 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Konner M., Eaton S. B., Paleolithic nutrition. Nutr. Clin. Pract. 25, 594–602 (2010). [DOI] [PubMed] [Google Scholar]
- 54.A.E. Caldwell, S. Eaton, M. Konner, in Oxford Handbook of Evolutionary Medicine, M. Brune, W. Schiefenhoevel, Eds. (Oxford Univ. Press, 2019), pp. 209–267. [Google Scholar]
- 55.D. E. Lieberman, The Story of the Human Body: Evolution, Health, and Disease (Pantheon Books, 2013). [PubMed] [Google Scholar]
- 56.G. Conroy, H. Pontzer, Reconstructing Human Origins: A Modern Synthesis (Norton, 2012). [Google Scholar]
- 57.Zeder M. A., Domestication and early agriculture in the Mediterranean Basin: Origins, diffusion, and impact. Proc. Natl. Acad. Sci. U.S.A. 105, 11597–11604 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Steffen P. R., Smith T. B., Larson M., Butler L., Acculturation to western society as a risk factor for high blood pressure: A meta-analytic review. Psychosom. Med. 68, 386–397 (2006). [DOI] [PubMed] [Google Scholar]
- 59.T. McDade, C. Nyberg, in Human Evolutionary Biology, M. P. Muehlenbein, Ed. (Cambridge Univ. Press, 2010), pp. 581–602. [Google Scholar]
- 60.Sapolsky R. M., The influence of social hierarchy on primate health. Science 308, 648–652 (2005). [DOI] [PubMed] [Google Scholar]
- 61.Taylor S. E., Repetti R. L., Seeman T., Health psychology: What is an unhealthy environment and how does it get under the skin? Annu. Rev. Psychol. 48, 411–447 (1997). [DOI] [PubMed] [Google Scholar]
- 62.Solon-Biet S. M., Mitchell S. J., de Cabo R., Raubenheimer D., Le Couteur D. G., Simpson S. J., Macronutrients and caloric intake in health and longevity. J. Endocrinol. 226, R17–R28 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Huneault L., Mathieu M.-È., Tremblay A., Globalization and modernization: An obesogenic combination. Obes. Rev. 12, 64–72 (2011). [DOI] [PubMed] [Google Scholar]
- 64.Douhard M., Plard F., Gaillard J.-M., Capron G., Delorme D., Klein F., Duncan P., Loe L. E., Bonenfant C., Fitness consequences of environmental conditions at different life stages in a long-lived vertebrate. Proc. Biol. Sci. 281, 20140276 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lea A. J., Altmann J., Alberts S. C., Tung J., Dudley R., Bronstein J. L., Developmental constraints in a wild primate. Am. Nat. 185, 809–821 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pigeon G., Festa-Bianchet M., Pelletier F., Long-term fitness consequences of early environment in a long-lived ungulate. Proc. Biol. Sci. 284, 20170222 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Botero C., Weissing F. J., Wright J., Rubenstein D. R., Evolutionary tipping points in the capacity to adapt to environmental change. Proc. Natl. Acad. Sci. U.S.A. 112, 184–189 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nettle D., Frankenhuis W. E., Rickard I. J., The evolution of predictive adaptive responses in human life history. Proc. R. Soc. B. 280, 20131343 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Nettle D., Bateson M., Adaptive developmental plasticity: What is it, how can we recognize it and when can it evolve? Proc. R. Soc. B 282, 20151005 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wells J. C. K., A critical appraisal of the predictive adaptive response hypothesis. Int. J. Epidemiol. 41, 229–235 (2012). [DOI] [PubMed] [Google Scholar]
- 71.Kraft T. S., Stieglitz J., Trumble B. C., Martin M., Kaplan H., Gurven M., Nutrition transition in 2 lowland Bolivian subsistence populations. Am. J. Clin. Nutr. 108, 1183–1195 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ho J. S., Cannaday J. J., Barlow C. E., Mitchell T. L., Cooper K. H., FitzGerald S. J., Relation of the number of metabolic syndrome risk factors with all-cause and cardiovascular mortality. Am. J. Cardiol. 102, 689–692 (2008). [DOI] [PubMed] [Google Scholar]
- 73.Adams K. F., Schatzkin A., Harris T. B., Kipnis V., Mouw T., Ballard-Barbash R., Hollenbeck A., Leitzmann M. F., Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N. Engl. J. Med. 355, 763–778 (2006). [DOI] [PubMed] [Google Scholar]
- 74.Aune D., Sen A., Prasad M., Norat T., Janszky I., Tonstad S., Romundstad P., Vatten L. J., BMI and all cause mortality: Systematic review and non-linear dose-response meta-analysis of 230 cohort studies with 3.74 million deaths among 30.3 million participants. BMJ 353, i2156 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kenya STEPwise Survey for non communicable diseases risk factors (2015).
- 76.Trumble B. C., Stieglitz J., Blackwell A. D., Allayee H., Beheim B., Finch C. E., Gurven M., Kaplan H., Apolipoprotein E4 is associated with improved cognitive function in Amazonian forager-horticulturalists with a high parasite burden. FASEB J. 31, 1508–1515 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gurven M. D., Lieberman D. E., WEIRD bodies: Mismatch, medicine and missing diversity. Evol. Hum. Behav. , (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.R Core Development Team, R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2015).
- 79.Benjamini Y., Hochberg Y., Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. 57, 289–300 (1995). [Google Scholar]
- 80.Snyder-Mackler N., Sanz J., Kohn J. N., Brinkworth J. F., Morrow S., Shaver A. O., Grenier J.-C., Pique-Regi R., Johnson Z. P., Wilson M. E., Barreiro L. B., Tung J., Social status alters immune regulation and response to infection. Science 354, 1041–1045 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lea A. J., Akinyi M. Y., Nyakundi R., Mareri P., Nyundo F., Kariuki T., Alberts S. C., Archie E. A., Tung J., Dominance rank-associated immune gene expression is widespread, sex-specific, and a precursor to high social status in wild male baboons. bioRxiv , 366021 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Novak N. L., Allender S., Scarborough P., West D., The development and validation of an urbanicity scale in a multi-country study. BMC Public Health 12, 530 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cyril S., Oldroyd J. C., Renzaho A., Urbanisation, urbanicity, and health: A systematic review of the reliability and validity of urbanicity scales. BMC Public Health 13, 513 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Uller T., Developmental plasticity and the evolution of parental effects. Trends Ecol. Evol. 23, 432–438 (2008). [DOI] [PubMed] [Google Scholar]
- 85.K. Galvin, M. Little, in Turkana Herders of the Dry Savanna: Ecology and Biobehavioral Response of Nomads to an uncertain Environment, M. A. Little, P. Leslie, Eds. (Oxford Univ. Press, 1999), pp. 125–146. [Google Scholar]
- 86.M. A. Little, B. Johnson, in South Turkana Nomadism: Coping with an Unpredictably Varying Environment, R. Dyson-Hudson, J. T. McCabe, Eds. (HRAFlex Books, 1985). [Google Scholar]
- 87.Wairimu E., Obare G., Odendo M., Factors affecting weather index-based crop insurance in Laikipia County, Kenya. J. Agric. Ext. Rural Dev. 8, 111–121 (2016). [Google Scholar]
- 88.Lalampaa P. K., Wasonga O. V., Rubenstein D. I., Njoka J. T., Effects of holistic grazing management on milk production, weight gain, and visitation to grazing areas by livestock and wildlife in Laikipia County, Kenya. Ecol. Process. 5, 17 (2016). [Google Scholar]
- 89.R. J. Hijmans, raster: Geographic Data Analysis and Modeling. (2019).
- 90.R. J. Hijmans, geosphere: Spherical Trigonometry. (2017).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/6/43/eabb1430/DC1


