Abstract
Background
Sipjeondaebo-tang (SJDBT, Shi-quan-da-bu-tang in Chinese) is a widely prescribed herbal medicine in traditional Korean medicine. This study aimed to evaluate the effectiveness and safety of SJDBT for treating chronic fatigue syndrome (CFS).
Methods
Ninety-six eligible participants were randomly allocated to either the SJDBT or placebo groups in a 1:1 ratio. Nine grams of SJDBT or placebo granules were administered to the patients for 8 weeks. The primary outcome was the response rate, defined as the proportion of participants with a score of 76 or higher in the Checklist Individual Strength assessment. Other measurements for fatigue severity, quality of life, and qi/blood/yin/yang deficiency were included. Safety was assessed throughout the trial.
Results
At week 8, the response rate did not significantly differ between the groups (SJDBT: 35.4%; placebo: 54.2%; P = 0.101, effect size [95% confidence interval] = 0.021 [-0.177, 0.218]). However, the scores of the visual analogue scale (P = 0.001, -0.327 [-0.506, -0.128]), Fatigue Severity Scale (P = 0.020, 0.480 [0.066, 0.889]), and Chalder fatigue scale (P = 0.004, -0.292 [-0.479, -0.101]) for the SJDBT group showed significant improvements in fatigue severity at the endpoint. Quality of life was not significantly different. Furthermore, SJDBT significantly ameliorated the severity of qi deficiency compared to that in the placebo group. No serious adverse events were observed.
Conclusion
This trial failed to show a significant improvement in fatigue severity, as assessed by the CIS-deprived response rate. It merely showed that SJDBT could alleviate the severity of fatigue and qi deficiency in patients with CFS. However, the further study is needed to confirm the details.
Keywords: Chronic fatigue syndrome, Complementary therapies, Herbal medicine, Randomized controlled trial
1. Introduction
According to the criteria of the US Centers for Disease Control and Prevention (CDC), chronic fatigue syndrome (CFS) or myalgic encephalomyelitis is defined as self-reported persistent or relapsing fatigue lasting 6 or more consecutive months, accompanied by symptoms such as muscle and joint pain, headaches, sore throat, tender lymph nodes, and cognitive difficulties.1, 2 A meta-analysis revealed that the point prevalence of CFS in 14 pooled studies was 0.76%–3.28%, depending on the diagnosis method.1 Another recent systematic review presented a similar meta-analysis with 1,085,976 participants, in which the prevalence of CFS was 0.89%–1.14%.3 Furthermore, a Korean study examining 1648 adult patients concluded the prevalence of CFS as 0.6% in the study population.4 The incidence of the condition varies globally, from 14.8 in the United Kingdom (2001–2013)5 to 25.8 in Norway (2008–2012)6 per 100,000 people.
Cognitive behavioral therapy has been recommended for CFS treatment.7 A randomized controlled trial (RCT) showed that compared to standard care, cognitive behavioral therapy effectively reduced the symptoms of fatigue.8 However, the evidence remains weak and limited.9 The efficacy of therapeutic drugs, such as rintatolimod and rituximab, has also been explored; however, the results have been debatable.9 A recent placebo-controlled RCT of rituximab treatment failed to demonstrate a significant difference in outcomes.10 Therefore, practitioners often seek complementary or alternative ways to manage CFS, including rehabilitation programs11 or exercise therapy.12
Clinical experts in traditional medicine encounter many patients complaining of primary fatigue. Therefore, researchers have attempted to accumulate clinical evidence on the effectiveness of acupuncture, electroacupuncture, or qigong therapies,13 as well as moxibustion therapy.14 A systematic review revealed that the use of herbal medicines had better effects in terms of CFS management than did the control treatments; however, the included studies were highly heterogeneous.13
Sipjeondaebo-tang (SJDBT) is an herbal medicine widely used in Korea. It is clinically used in patients with symptoms such as fatigue, loss of appetite, night sweats, or cold hands and feet and has been authorized by the Ministry of Food and Drug Safety in Korea (K-MFDS). Panax ginseng, a primary herbal ingredient, showed anti-fatigue effects in 90 patients diagnosed with idiopathic chronic fatigue.15 Furthermore, the effectiveness of SJDBT has been investigated for the management of cancer-related fatigue in women with breast cancer.16 However, no clinical trials evaluating the efficacy of SJDBT in patients with CFS have been reported. Therefore, this study aimed to elucidate the effectiveness and safety of SJDBT granule administration for 8 weeks compared with placebo control for the management of fatigue severity in adult patients diagnosed with CFS by using the CDC criteria.
2. Methods
This article followed the Consolidated Standards of Reporting Trials (CONSORT) 2010 Statement: updated guidelines for reporting parallel group randomized trials,17, 18 and the CONSORT Extension for Chinese Herbal Medicine Formulas 2017: Recommendations, Explanation, and Elaboration.19
2.1. Trial registration
The study was initially registered in the Clinical Research Information Service (CRIS, https://cris.nih.go.kr/cris/en/, KCT0002684) on January 11, 2018. No critical methodological changes were introduced after the initial ethics committee approval.
2.2. Study design, setting, and ethics
This was a prospective, parallel-group, double-blind, randomized, placebo-controlled trial conducted in 96 adult patients aged 19–65 years diagnosed with CFS by using the US CDC criteria at two sites: Kyung Hee University Korean Medicine Hospital at Gangdong (Seoul) and Woosuk University Korean Medicine Medical Center (Jeonju), the Republic of Korea. The study protocol and informed consent forms were reviewed and approved by the Institutional Review Board of Kyung Hee University Korean Medicine Hospital at Gangdong on July 17, 2017 (KHNMCOH 2017-06-004-001) and the Institutional Review Board of Woosuk University Korean Medicine Medical Center on March 14, 2018 (WSOH IRB D1802-01-010), respectively.
During the screening visit, information regarding demographic characteristics, medical history, and concomitant medication was collected after the signed informed consent form was obtained from the participants. For screening assessments, a physical examination, vital sign check, laboratory tests (blood and urine tests), pregnancy test (when available), heart rate variability (HRV) examinations, and body composition measurements were included. The eligible participants were randomly allocated to the SJDBT or placebo group in a 1:1 ratio. The participants were administered 9 g of SJDBT or placebo granules for 8 consecutive weeks. The validated self-reported outcomes for fatigue severity, quality of life (QoL), and sleep quality were evaluated at baseline and the endpoint. The safety of the SJDBT granules was assessed by performing the vital sign check, laboratory tests, HRV examination, and adverse event (AE) reporting. The trial schedule is shown in Supplement 1.
2.3. Eligibility criteria
The inclusion criteria were as follows: a) male or female participants aged 19–65 years diagnosed with CFS by using the US CDC criteria, b) Checklist Individual Strength (CIS) score higher than 76 points at baseline, and c) provision of voluntarily signed consent forms. The CFS patients were required to show the following symptoms based on the CDC criteria: a) persistent or recurring fatigue for 6 or more consecutive months not attributable to ongoing exertion or other medical conditions associated with fatigue, b) fatigue that markedly interfered with personal activities and work, and c) four or more concurrent symptoms (impairment in short-term memory or concentration; sore throat; tender cervical or axillary lymph nodes; muscle pain; multijoint pain without joint swelling or redness; headaches of a new type, pattern, or severity; unrefreshing sleep; and post-exertional malaise lasting more than 24 h).2
The participants were excluded if they met the following criteria: a) hypersensitivity to herbal medicines, b) body mass index (BMI) of 45 kg/m2 or higher; c) two-fold or higher levels of the upper normal limit of aspartate aminotransferase (AST), alanine aminotransferase (ALT), serum bilirubin, or serum creatinine at screening assessment; d) positive results for hepatitis B surface antigen, hepatitis C antibodies, or human immunodeficiency virus tests at screening assessment; e) clinically meaningful gastrointestinal disorders that may affect absorption, distribution, metabolism, or excretion of the investigational products; drug-induced or alcoholic hepatitis, liver cirrhosis, fatty liver requiring therapeutic intervention, renal failure or other renal diseases requiring dialysis, anorexia, bulimia, uncontrollable hypertension with systolic blood pressure of 180 mmHg or higher or diastolic blood pressure of 110 mmHg or higher, uncontrollable diabetes mellitus with 9% or higher hemoglobin A1C, uncontrollable edema, tuberculosis, multiple sclerosis, hypothyroidism, uncontrollable asthma, major depressive disorder, bipolar affective disorder, schizophrenia, dementia, delusional disorder, malignant tumor within 5 years before trial enrolment, myocardial infarction, unstable angina pectoris, stroke, cerebral ischemic attack, heart failure, uncontrolled arrhythmia, coronary revascularization procedure within 6 months before the trial, drug/alcohol abuse within 1 year before the trial, or any other severe diseases that could affect the trial; f) pregnant (positive result of the human chorionic gonadotropin test) or lactating women; g) ongoing intake of immunosuppressive medication; h) participation in other clinical trials within 3 months before the trial; or i) other inappropriate reasons related to study participation (clinically significant medical or psychiatric findings).
2.4. Interventions
2.4.1. SJDBT group
SJDBT granules (product name: Deciten Granule) were produced by Hanpoong Pharm. Co Ltd. (http://hpeng.hanpoong.co.kr, Seoul, Republic of Korea). One pouch (3 g) of SJDBT contained 1.0 g each of Ginseng Radix (Panax ginseng C. A. Meyer), Astragali Radix (Astragalus membranaceus Bunge), Poria Sclerotium (Poria cocos Wolf), Atractylodis Rhizoma Alba (Atractylodes japonica Koidzumi or Atractylodes macrocephala Koidzumi), Angelicae Gigantis Radix (Angelica gigas Nakai), Paeoniae Radix (Paeonia lactiflora Pallas), Cnidii Rhizoma (Cnidium officinale Makino or Ligusticum chuanxiong Hort), Cinnamomi Ramulus (Cinnamomum cassia J.Presl), and Rehmanniae Radix Preparata (Rehmannia glutinosa Liboschitz ex Steudel), and 0.5 g of Glycyrrhizae Radix et Rhizoma (Glycyrrhiza uralensis Fischer, Glycyrrhiza glabra Linné, or Glycyrrhiza inflata Batal). The SJDBT granules were manufactured by extraction, filtration, and vacuum evaporation, and validated/quality-controlled following the Korean Pharmacopoeia. This herbal medication has been used for the management of fatigue, loss of appetite, night sweats, and cold hands and feet with the authorization of the K-MFDS.
The participants allocated to the SJDBT group were administered 9 g of SJDBT granules daily (3 g each time, 3 times a day, before or between meals) for 8 weeks. This dosage regimen was ascertained in line with the original use/dosage of Deciten Granule approved by the K-MFDS.
2.4.2. Placebo group
The placebo granules were formulated to be identical to SJDBT in color and taste by using food coloring and flavoring agents. They contained corn starch, lactose hydrate, citric acid hydrate, caramel coloring, and ginseng-flavored powder. All ingredients used in the preparation of the placebo were standardized following the Korean Food Standards Codex and Korean Food Additives Codex. The participants allocated to the placebo group received 9 g of placebo granules daily (3 g each time, 3 times a day, before or between meals) for 8 weeks.
2.5. Concomitant therapies
The participants were not allowed to use any of the following therapies or receive exercise therapy during the treatment period: antipsychotics, antidepressants, dementia medications, systemic steroidal drugs, nonsteroidal anti-inflammatory drugs (NSAIDs), immunomodulators, immunosuppressants, transfusions, and other herbal medicines. However, they were permitted the use of NSAIDs for less than 5 days (except for the 3 days preceding the evaluation visit).
2.6. Outcomes
Data were collected using case report forms. An independent monitor regularly visited each site to verify data, detect protocol deviations, and monitor any safety issues. Every schedule for evaluating each outcome is shown in Supplement 1. No changes in outcome assessments were included after the trial commenced.
2.6.1. Primary outcome: CIS
The severity of fatigue was measured with CIS in Likert scale, with 20 items in several domains: subjective fatigue experience, concentration, motivation, and physical activity levels.20 The participants were required to rate each item on a scale of 1 (“No, that is not true”) to 7 (“Yes, that is true”), recalling the previous 2 weeks (total score range: 20–140). The higher the total score, the greater the fatigue severity. Furthermore, a total score of 76 was determined as the cut-off point for clinically significant fatigue.21
2.6.2. Secondary outcome: visual analogue scale (VAS)
The fatigue intensity was evaluated with the VAS.22 The participants were asked to mark across a 100-mm horizontal line (0 for “Not at all tired” and 10 for “extremely tired”), recalling the previous week. The fatigue VAS is unidimensional, focusing only on the severity of fatigue; therefore, it is widely used to measure fatigue symptoms for a variety of disorders.22
The participants were also required to make marks on the VAS line for each concurrent symptom, defined in the CDC CFS criteria (impairment in short-term memory or concentration; sore throat; tender cervical or axillary lymph nodes; muscle pain; multijoint pain without joint swelling or redness; headaches of a new type, pattern, or severity; unrefreshing sleep; post-exertional malaise lasting more than 24 h). They were given separate 100-mm horizontal lines for each symptom, with 0 and 10 denoting no symptom and very serious symptoms respectively.
2.6.3. Secondary outcome: fatigue severity scale (FSS)
The severity of fatigue was assessed with the FSS, which was originally developed for multiple sclerosis and systemic lupus erythematosus.23 It is a 7-point Likert scale, with nine items (1 for “Strongly disagree” and 7 for “Strongly agree”) to evaluate the physical, social, or cognitive effects of fatigue. The participants were asked to rate their fatigue severity in the previous week by using this scale. The higher the score, the greater the fatigue severity. It is recommended that this scale be used for patients with systemic lupus erythematosus.22
2.6.4. Secondary outcome: Chalder fatigue scale (ChFi)
The physical and mental fatigue was measured with the ChFi. It is a self-reported outcome measure for the evaluation of physical and mental fatigue in patients with CFS.24 The participants were asked to rate their fatigue levels in the previous month by using this scale. The scale consists of 11 items and each item is rated from 0 to 9. A higher score implies a severe level of fatigue. It has been developed to assess disabling fatigue severity in hospital and community populations, especially for systemic lupus erythematosus, primary Sjögren’s syndrome, rheumatoid arthritis, psoriatic arthritis, fibromyalgia syndrome, upper-extremity or carpal tunnel disorder, and CFS.22
2.6.5. QoL, overall happiness level, and sleep quality
The EuroQoL 5-dimension 5-level questionnaire (EQ-5D-5 L) was used to measure the participants’ QoL. It has five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and each dimension has 5 levels (no problems, slight problems, moderate problems, severe problems, and extreme problems).25
Happiness was assessed with 100-mm VAS to measure how happy the participants felt in their own lives, with scores of 0 and 10 indicating “Not at all happy” and “very happy,” respectively.
The Korean version of the Pittsburgh Sleep Quality Index (PSQI-K) was used to evaluate sleep quality by measuring 7 domains: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleep medication, and daytime dysfunction over the last month. The higher the global score of PSQI-K, the worse the subjective sleep quality.26
2.6.6. Qi, blood, yin, yang deficiency
The Qi blood yin yang deficiency questionnaire (QBYY-Q) is a self-reported instrument for the evaluation of qi, blood, yin, and yang deficiencies in terms of pattern/syndrome identification of traditional medicine that has been especially validated for chronic fatigue.27, 28 QBYY-Q separately evaluates qi, blood, yin, and yang deficiencies, and the participants were required to rate each item on a scale of 1 (“Not at all”) to 4 (“Very agree”). A higher score implied more severe qi, blood, yin, and yang deficiencies.
2.6.7. Safety assessment
Participants’ vital signs (systolic blood pressure [SBP], diastolic blood pressure [DBP], and pulse rate) were regularly assessed. Additionally, the participants were screened using laboratory tests (white blood cell, red blood cell, and platelet counts; hemoglobin, hematocrit, albumin, total protein, inorganic phosphorus, AST, ALT, alkaline phosphatase, total cholesterol, glucose, blood urea nitrogen, total bilirubin, uric acid, creatinine, C-reactive protein, sodium, potassium, chloride, calcium, cortisol, lactic acid dehydrogenase, and creatine phosphokinase levels; and pH/specific gravity/protein/glucose of urine) at baseline and the end of treatment. AEs were monitored and reported throughout the study period. To assess any additional AEs, the investigator summoned the participants between 4 and 6 weeks after the assigned treatment was completed.
2.7. Sample size
The null hypothesis assumed that the response rate of the SJDBT group was the same as that of the placebo group, provided that the response rate was defined as the proportion of patients with a CIS score of 76 or higher. No previous randomized clinical study has evaluated the effectiveness of SJDBT in CFS patients. Furthermore, there are no trials comparing the effectiveness of any herbal medications with placebo control in CFS patients, based on a systematic review.13 We searched RCTs of any drugs for CFS with statistically significant results. Therefore, we assumed response rates of 17% and 0% for the SJDBT and placebo groups, respectively, based on a double-blind randomized placebo-controlled study performed in CFS patients,29 despite there being no commonalities in drug mechanism and administration period between the two studies. With an assumed 5% significance level (bilateral) and 80% power of the test, the sample size was calculated as 39 participants per group. Therefore, 96 eligible participants were enrolled in both groups with a 1:1 allocation ratio, considering a 20% drop-out rate. The equation used is as follows:
where pt and pc denote that the response rates of the SJDBT and placebo groups, respectively.
2.8. Randomization, allocation concealment, and blinding
An independent researcher generated random sequence numbers with random blocks and site stratification (R version 3.4, Blockrand package) in a 1:1 allocation ratio. The information of the random numbers and allocated groups was provided by the independent researcher to the pharmaceutical company via an encoded email. The pharmaceutical company put labels by using only those random numbers on SJDBT and placebo packages, which were identically shaped. The allocation was concealed throughout the study as the sequence numbers were documented only by the independent researcher and company. The participants were allocated in the order of screening completion. The patients and clinical physicians were blinded throughout the entire study period as the SJDBT and placebo granules were identical in shape, weight, and packing.
2.9. Statistical methods
The full analysis set (FAS), defined as the participants who were administered SJDBT or placebo and in whom the primary outcome was assessed at least once, was statistically analyzed to evaluate the effectiveness of the test substance. Missing data for the FAS were imputed with the last observation carried forward method. The per protocol set (PPS), defined as the participants who completed all the planned study procedures and did not critically violate the study protocol, was incidentally analyzed to evaluate effectiveness. The safety assessment set (SAS), defined as the participants who were administered SJDBT or placebo and were assessed for any of the safety outcomes at least once, was statistically analyzed for safety evaluation.
The primary outcome was the response rate, defined as the proportion of the patients with a CIS score of 76 or higher. Various assessment tools of fatigue measurement have been used, and there is no single dominant scale for chronic fatigue assessment as yet.22 Since a previous study defined a CIS score of 76 points as the cut-off for clinical significance,21 we adopted the response rate, derived from the CIS score, as the primary measurement. The mean changes in CIS, VAS, FSS, EQ-5D-5 L, PSQI-K, ChFi, and QBYY-Q scores were also analyzed, including those for the subscale scores, if available. The continuous variables are presented as mean ± standard deviation (SD) values, while categorical variables are represented as counts and percentages. Continuous variables were compared between the SJDBT and placebo groups by using an independent t-test or Wilcoxon rank-sum test. The effect sizes calculated by Cohen’s or Rosenthal's formula were addressed with the 95% confidence interval. Categorical variables were compared between groups using the Chi-square or Fisher’s exact test. The effect size, phi, was also calculated with the 95% confidence interval.
AEs were statistically analyzed using the Chi-square or Fisher’s exact test. The measured results of vital signs, HRV, and laboratory tests were statistically analyzed with an independent t-test or Wilcoxon rank-sum test. The continuous variables for safety assessment were compared within each group by using the paired t-test or Wilcoxon signed-rank test. The ratio of normal and abnormal (with clinical significance) results of each laboratory test was also compared within each group by using the McNemar’s test. Statistical analyses were performed using the R software, version 3.6.2 (2019-12-12), at a 5% significance level (bilateral).
3. Results
3.1. Baseline characteristics
In total, 106 participants were screened, of whom 96 were enrolled at Kyung Hee University Korean Medicine Hospital at Gangdong and Woosuk University Korean Medicine Medical Center, the Republic of Korea between May 28, 2018, and September 4, 2019. Patients diagnosed with CFS were randomly allocated to the SJDBT or placebo groups in a 1:1 ratio. Six patients were dropped from the SJDBT group due to withdrawal of consent, deviation from eligibility criteria, or AEs, while five participants were dropped from the placebo group due to withdrawal of consent, deviation of eligibility criteria, loss to follow-up, or AEs. (It was found out, after random allocation, that the CIS scores of four participants at screening assessment were wrongly calculated. All of them were dropped before starting drug administration.)
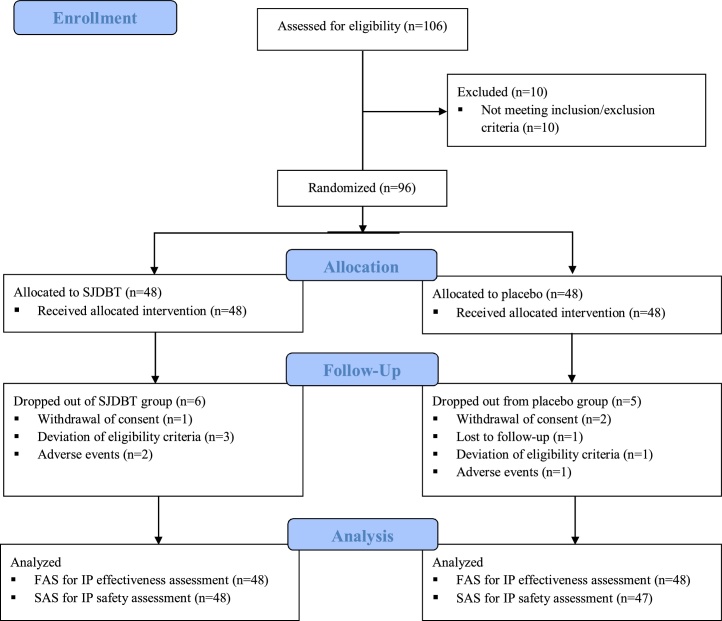
All participants were included in the FAS analysis since they were administered the test substance, and the primary outcome was assessed more than once. One participant in the placebo group was excluded from the SAS analysis because he/she was lost to follow-up after the first safety assessment visit. The CONSORT diagram is shown in Fig. 1. Demographic characteristics are summarized in Table 1. Age, sex, height, weight, and BMI did not significantly differ between the groups. With regard to body composition measurements, skeletal muscle mass, total body water, and the level of basal metabolism significantly differed between the groups. However, the effectiveness variables were not statistically adjusted as no associated clinically critical meaning was observed. Furthermore, drinking, smoking, and exercise tendencies did not differ between the SJDBT and placebo groups. Drug compliance was 89.1 ± 13.6% in the SJDBT group and 89.4 ± 12.0% in the placebo group; the intergroup difference was not significant.
Fig. 1.
CONSORT diagram of the study flow.
Abbreviations: FAS, Full Analysis Set; SAS, Safety Assessment Set; SJDBT, Sipjeondaebo-tang.
Table 1.
Baseline characteristics.
| Variable | SJDBT group (n = 48) | Placebo group (n = 48) | P-value |
|---|---|---|---|
| Agea (y) | 41.5 ± 8.2 | 40.6 ± 9.8 | 0.635 |
| Sexc | |||
| Male, n (%) | 17 (35.4%) | 12 (25.0%) | 0.374 |
| Female, n (%) | 31 (64.6%) | 36 (75.0%) | |
| Heightb (cm) | 166.4 ± 8.0 | 163.8 ± 7.2 | 0.089 |
| Weightb (kg) | 65.4 ± 12.0 | 62.1 ± 11.2 | 0.210 |
| BMIa (kg/m2) | 23.5 ± 3.1 | 23.0 ± 2.9 | 0.446 |
| Body composition | |||
| Skeletal muscle massb (kg) | 26.0 ± 5.7 | 23.8 ± 5.8 | 0.024* |
| Body fat massb (kg) | 18.4 ± 6.1 | 18.6 ± 5.2 | 0.585 |
| Percent body fatb (%) | 27.8 ± 7.0 | 29.8 ± 6.6 | 0.156 |
| Percent abdominal fatb (%) | 1.1 ± 1.2 | 0.9 ± 0.0 | 0.564 |
| Total body waterb (kg) | 34.9 ± 7.5 | 32.0 ± 7.0 | 0.024* |
| Basal metabolismb (kcal) | 1388.4 ± 201.1 | 1312.5 ± 204.1 | 0.028* |
| Drinkingc | |||
| Yes, n (%) | 28 (58.3%) | 27 (56.2%) | 1.000 |
| No, n (%) | 20 (41.7%) | 21 (43.8%) | |
| Smokingc | |||
| Never, n (%) | 36 (75.0%) | 38 (79.2%) | 0.370 |
| Used to smoke, n (%) | 5 (10.4%) | 7 (14.6%) | |
| Yes, n (%) | 7 (14.6%) | 3 (6.2%) | |
| Exercisec | |||
| Yes, n (%) | 18 (37.5%) | 12 (25.0%) | 0.271 |
| No, n (%) | 30 (62.5%) | 36 (75.0%) |
The results are expressed as mean ± SD values or n (%). BMI, body mass index; SJDBT, Sipjeondaebo-tang.
Independent two sample t-test.
Wilcoxon rank sum test.
Chi-squared test.
P < 0.05.
3.2. Fatigue severity
The numbers of patients whose CIS score exceeded 76 points after 8 weeks were 17 (35.4%) and 26 (54.2%) in the SJDBT and placebo groups, respectively, indicating no significant intergroup difference (P = 0.101). Furthermore, the mean change in CIS scores was not significantly different between the two groups (P = 0.210) (Table 2).
Table 2.
Fatigue severity.
| Variable | SJDBT group (n = 48) |
Placebo group (n = 48) |
P-value | Effect size [95% CI]c | ||||
|---|---|---|---|---|---|---|---|---|
| Baseline | Week 8 | Change | Baseline | Week 8 | Change | |||
| CISa | 97.0 ± 14.4 | 70.1 ± 15.0 | −26.1 ± 19.2 | 95.2 ± 10.6 | 74.0 ± 19.7 | −20.8 ± 21.9 | 0.210 | 0.258 [-0.147, 0.660] |
| Fatigue (VAS)b | 83.1 ± 7.6 | 46.1 ± 20.9 | −36.2 ± 23.4 | 79.4 ± 11.9 | 56.3 ± 24.1 | −21.4 ± 23.8 | 0.001** | −0.327 [-0.506, -0.128] |
| Additional symptoms based on CDC criteria (VAS) | ||||||||
| Post-exertional malaiseb | 69.6 ± 24.1 | 36.8 ± 22.2 | −32.6 ± 29.2 | 68.1 ± 22.4 | 48.1 ± 22.3 | −18.9 ± 28.3 | 0.016* | −0.247 [-0.430, -0.067] |
| Unrefreshing sleepb | 80.8 ± 12.7 | 42.2 ± 22.8 | −37.5 ± 23.4 | 80.6 ± 12.6 | 50.5 ± 27.7 | −28.4 ± 27.0 | 0.045* | −0.205 [-0.403, 0.000] |
| Impairment in short-term memory or concentrationa | 66.6 ± 21.7 | 38.4 ± 21.0 | −27.4 ± 24.4 | 62.7 ± 23.7 | 37.8 ± 23.6 | −24.9 ± 24.1 | 0.611 | 0.103 [-0.298, 0.503] |
| Muscle painb | 69.4 ± 20.5 | 33.7 ± 20.7 | −34.6 ± 26.0 | 65.0 ± 23.1 | 44.0 ± 25.5 | −20.1 ± 22.1 | 0.004** | −0.294 [-0.481, -0.086] |
| Multi-joint paina | 43.1 ± 29.6 | 22.5 ± 21.3 | −20.3 ± 33.5 | 42.4 ± 30.2 | 29.3 ± 26.7 | −12.9 ± 25.2 | 0.486 | 0.250 [-0.155, 0.652] |
| Headachesb | 50.8 ± 30.6 | 18.6 ± 19.2 | −30.7 ± 32.9 | 46.5 ± 28.0 | 25.4 ± 24.7 | −20.4 ± 34.1 | 0.106 | −0.165 [-0.350, 0.031] |
| Lymph nodesb | 14.7 ± 20.5 | 10.0 ± 13.9 | −4.2 ± 18.5 | 20.1 ± 27.8 | 12.8 ± 20.5 | −7.1 ± 20.8 | 0.720 | 0.037 [-0.166, 0.223] |
| Sore throatb | 49.3 ± 31.1 | 17.4 ± 21.8 | −31.2 ± 34.6 | 41.4 ± 32.9 | 17.0 ± 24.2 | −24.2 ± 28.2 | 0.209 | −0.129 [-0.329, 0.099] |
| FSSa | 5.3 ± 0.7 | 3.7 ± 1.0 | −1.6 ± 1.2 | 5.0 ± 0.7 | 3.9 ± 1.2 | −1.0 ± 1.3 | 0.020* | 0.480 [0.066, 0.889] |
| ChFib | 63.3 ± 13.4 | 39.7 ± 14.4 | −22.9 ± 19.0 | 56.3 ± 12.6 | 44.7 ± 17.0 | −11.6 ± 16.5 | 0.004** | −0.292 [-0.479, -0.101] |
The results are expressed as mean ± SD values. CDC, Centers for Disease Control and Prevention; ChFI, Chalder fatigue scale; CI, confidence interval; CIS, Checklist Individual Strength;FSS, Fatigue Severity Scale; SJDBT, Sipjeondaebo-tang; VAS, visual analogue scale.
Independent two sample t-test.
Wilcoxon rank sum test.
Cohen's d using pooled variance for independent t-test or Rosenthal's formula for Wilcoxon rank sum test.
P < 0.05, ** P < 0.001.
However, the overall fatigue score in VAS in the SJDBT group differed from the placebo group, with a significant difference observed at the end of treatment (P = 0.001). At week 8, VAS scores for post-exertional malaise (P = 0.016), unrefreshing sleep (P = 0.045), and muscle pain (P = 0.004), among additional symptoms of the US CDC criteria, significantly decreased in the SJDBT group compared to those in the placebo group (Table 3). The mean changes in the FSS score differed significantly between the SJDBT and placebo groups at the treatment endpoint (P = 0.020). Additionally, the Chfi scores decreased by a significantly greater extent in the intervention group than in the control group at week 8 (P = 0.004) (Table 2).
Table 3.
QoLs, overall happiness level, sleep quality, and Qi/blood/yin/yang deficiency.
| Variable | SJDBT group (n = 48) |
Placebo group (n = 48) |
P | Effect size [95% CI]b | ||||
|---|---|---|---|---|---|---|---|---|
| Baseline | Week 8 | Change | Baseline | Week 8 | Change | |||
| EQ-5D-5La | 0.783 ± 0.098 | 0.848 ± 0.052 | 0.063 ± 0.094 | 0.812 ± 0.066 | 0.843 ± 0.059 | 0.032 ± 0.060 | 0.089 | 0.172 [-0.035, 0.368] |
| VAS score for happinessa | 53.6 ± 20.1 | 64.8 ± 17.2 | 11.3 ± 23.0 | 59.3 ± 20.4 | 65.9 ± 19.1 | 6.9 ± 21.4 | 0.346 | 0.097 [-0.119, 0.287] |
| PSQI-Ka | 9.0 ± 2.5 | 6.6 ± 2.4 | −2.4 ± 2.5 | 8.4 ± 3.1 | 6.9 ± 2.9 | −1.5 ± 3.0 | 0.139 | −0.151 [-0.346, 0.038] |
| QBYY-Q | ||||||||
| Qi deficiencya | 13.0 ± 3.2 | 8.9 ± 2.9 | −3.9 ± 3.4 | 11.9 ± 2.9 | 9.6 ± 3.2 | −2.2 ± 3.2 | 0.043* | −0.206 [-0.395, -0.006] |
| Blood deficiencya | 8.5 ± 2.0 | 6.2 ± 1.7 | −2.2 ± 2.2 | 8.0 ± 1.8 | 6.7 ± 2.1 | −1.4 ± 2.1 | 0.079 | −0.180 [-0.377, 0.023] |
| Yin deficiencya | 5.6 ± 2.0 | 5.2 ± 2.0 | −0.4 ± 1.5 | 5.4 ± 1.8 | 4.5 ± 1.5 | −0.9 ± 1.5 | 0.118 | −0.096 [-0.282, 0.120] |
| Yang deficiencya | 8.5 ± 3.2 | 7.2 ± 2.6 | −1.3 ± 2.3 | 8.8 ± 2.6 | 7.8 ± 2.7 | −1.0 ± 1.6 | 0.768 | −0.031 [-0.241, 0.169] |
The results are expressed as mean ± SD values. CI, confidence interval; EQ-5D-5 L, EuroQoL 5-dimensions, 5-level questionnaire; PSQI-K, Korean version of Pittsburgh Sleep Quality Index; QBYY-Q, Qi blood yin yang deficiency questionnaire; QoL, quality of life; SJDBT, Sipjeondaebo-tang; VAS, visual analogue scale.
Wilcoxon rank sum test.
Cohen’s d using pooled variance for independent t-test or Rosenthal's formula for Wilcoxon rank sum test.
P < 0.05.
3.3. QoL, overall happiness level, and sleep quality
The EQ-5D-5 L, VAS for happiness, and PSQI-K scores demonstrated no change at the end of SJDBT or placebo administration at 8 weeks, indicating no statistical significance. The mean ± SD and P values are shown in Table 3.
3.4. Qi, blood, yin, yang deficiency
The scores for qi deficiency were significantly different between the SJDBT and placebo groups at the determined endpoint (P = 0.043). The scores for blood, yin, and yang deficiencies did not change significantly after the 8-week administration. Table 3 presents the QBYY-Q results.
3.5. Safety assessments
Sixteen participants reported AEs (nine and seven participants in the SJDBT and placebo groups, respectively) including hot flushes, headache, heartburn, migraine, dermatitis, nausea, lower abdominal pain, dizziness, heavy stomach, loss of appetite, and muscle pain. All reported AEs were clinically judged as “mild” and “unrelated to SJDBT or placebo granules.” Based on the Chi-square test, the P-value for AE occurrence was 0.820.
At week 8, no laboratory test result significantly differed between the groups, with most results remaining significantly unaltered within each group, except for AST and calcium in the SJDBT group and hematocrit and inorganic phosphorus in the placebo group. However, in each group, results that were significantly altered at week 8 remained within the normal ranges. SBP, DBP, pulse rate, and each component of the HRV examination did not differ significantly between the two groups and within each group.
4. Discussion
The response rate did not significantly differ between the SJDBT and placebo groups at the end of the 8-week administration period. However, the changes in VAS, FSS, and Chfi scores indicated significant improvements in fatigue severity after SJDBT administration for 8 weeks, compared with placebo. The results for QoL, subjective happiness levels, and sleep quality did not significantly differ between the two groups. The qi deficiency level also decreased significantly in the SJDBT group. During the study period, no serious or treatment-related AEs were reported. The trial primarily failed to show significant improvements in fatigue severity, based on the CIS-derived response rate. It secondarily indicated possibilities that SJDBT could alleviate the severity of fatigue in patients with CFS; however, the findings warrant further confirmation.
The pathogenic mechanism of CFS remains to be elucidated. Several studies have explored the relationship between immune dysfunction and CFS. For example, a case-control study analyzed cerebrospinal fluid from 32 CFS patients and 19 healthy subjects and found that the levels of many cytokines were significantly different between the groups, including decreased levels of IL-12 p40 in the CFS group.30 It is meaningful that an in vitro study showed that SJDBT enhanced immunity by augmenting the production of IL-12 p40.31
Investigations have explored the effectiveness of herbal medicines for CFS management. A literature review summarized that several herbal medicines, such as Yukmijihwang-hwan, Bojungikgi-tang, Soshiho-tang, Insamyangyung-tang, Ukgan-san, or Sanjoin-tang, have been used for the management of fatigue or related symptoms. However, none of those had adequate evidence for patients with CFS.32 A recent systematic review analyzed five clinical studies that explored the effectiveness of Chinese herbal medicine for the management of CFS. Although some included studies reported that herbal medicines had better effects, the review concluded that the meta-analysis was not appropriate due to a high level of heterogeneity.13 Above all, all of the included studies were not placebo-controlled, which could induce a bias in efficacy measurement. We tried to design an unbiased RCT comparing SJDBT (one of the most widely used herbal medications in traditional Korean medicine) with adequately produced placebo granules, grounded on the methodology of evidence-based medicine.
The physiology and pathology in traditional medicine suggest that fatigue could be due to deficiencies in qi/blood/yin/yang in terms of pattern/syndrome identification.32 This study subordinately showed that SJDBT significantly improved the severity of qi deficiency following 8 weeks of administration when compared with placebo granules. It is meaningful that Panax ginseng, which is one of the major herbs for the management of qi deficiency pattern/syndrome and among the main herbs in SJDBT, significantly reduced the VAS score for fatigue severity compared to placebo. However, Panax ginseng is not solely used for patients in the practice of traditional Korean medicine.15 Therefore, the result that SJDBT reduced the level of qi deficiency in CFS patients obtained in this study has potential for consideration in future studies.
This was the first placebo-controlled randomized trial evaluating the effectiveness and safety of an herbal medication, SJDBT, for the management of fatigue severity in patients with CFS. There are no adequately designed RCTs comparing herbal medicines with placebo for CFS; thus, this trial can serve as a reference for further studies in terms of its strengths and weaknesses. Although the trial failed to demonstrate a significant primary outcome (response rate), it showed significant changes in VAS, FSS, and Chfi scores, which affords the possibilities that SJDBT might reduce the levels of fatigue in patients with CFS.
There are a few limitations of this study. First, this study set the response rate as the primary outcome, based on the clinical significant result in a previous study.21 Since the response rate was derived from the CIS score in this study, it might not have reflected the severity of fatigue directly. In future research, CIS, VAS, FSS, or Chfi scores should be considered as the primary outcome. The sample size calculation in this study might be debatable as well. Since there were no RCTs with SJDBT and no placebo-controlled trials with any herbal medications, we calculated the sample size only based on the target disease and significant results, which might not have been suitable. A pilot study should also be considered before undertaking this research. Furthermore, the study population could be more specific. This study showed that the qi deficiency significantly improved with SJDBT administration, implying that SJDBT could be evaluated focused on CFS patients diagnosed with qi deficiency pattern/syndrome. Finally, this study did not assess how successful the blinding was. Considering that the drug type was granule and herbal drugs usually have a unique color, odor, or taste, it could be difficult to produce a perfectly identical placebo granule. Therefore, it is necessary to assess the success or failure of blinding in the future.
In conclusion, this trial primarily failed to show a significant improvement in fatigue severity, as assessed by the CIS-deprived response rate. Fatigue on VAS showed that SJDBT could alleviate the severity of fatigue in patients with CFS. However, further studies are needed to confirm the details.
Author contributions
Conceptualization: MH. Methodology: MH and SS. Formal Analysis: SS. Investigation: MH and SJP. Data Curation: SS. Writing – Original Draft: SS. Writing – Review & Editing: MH and SJP. Visualization: SS. Supervision: MH. Project Administration: MH. Funding Acquisition: MH.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HB16C0055). The funding did not influence the study design, data collection, study management, and data analysis and interpretation.
Ethical statement
The study protocol and informed consent form were reviewed and approved by the Institutional Review Board of Kyung Hee University Korean Medicine Hospital at Gangdong, Seoul, Republic of Korea (KHNMCOH 2017-06-004-001) on July 17, 2017 and the Institutional Review Board of Woosuk University Korean Medicine Medical Center on March 14, 2018 (WSOH IRB D1802-01-010), respectively. There have been no critical methodological changes after the initial ethics committee approval.
Data availability
Data are available upon request from the corresponding author.
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.imr.2020.100664.
Supplementary material
The following are Supplementary data to this article:
References
- 1.Johnston S., Brenu E.W., Staines D., Marshall-Gradisnik S. The prevalence of chronic fatigue syndrome/myalgic encephalomyelitis: a meta-analysis. Clin Epidemiol. 2013;5:105–110. doi: 10.2147/CLEP.S39876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fukuda K., Straus S.E., Hickie I., Sharpe M.C., Dobbins J.G., Komaroff A. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med. 1994;121:953–959. doi: 10.7326/0003-4819-121-12-199412150-00009. [DOI] [PubMed] [Google Scholar]
- 3.Lim E.-J., Ahn Y.-C., Jang E.-S., Lee S.-W., Lee S.-W., Son C.-G. Systematic review and meta-analysis of the prevalence of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) J Transl Med. 2020;18:100. doi: 10.1186/s12967-020-02269-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim C.H., Shin H.C., Won C.W. Prevalence of chronic fatigue and chronic fatigue syndrome in Korea: community-based primary care study. J Korean Med Sci. 2005;20:529–534. doi: 10.3346/jkms.2005.20.4.529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Collin S.M., Bakken I.J., Nazareth I., Crawley E., White P.D. Trends in the incidence of chronic fatigue syndrome and fibromyalgia in the UK, 2001-2013: a clinical practice research datalink study. J R Soc Med. 2017;110:231–244. doi: 10.1177/0141076817702530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bakken I.J., Tveito K., Gunnes N. Two age peaks in the incidence of chronic fatigue syndrome/myalgic encephalomyelitis: a population-based registry study from Norway 2008-2012. BMC Med. 2014;12:167. doi: 10.1186/s12916-014-0167-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wyller V.B., Reme S.E., Mollnes T.E. Chronic fatigue syndrome/myalgic encephalo-myelitis--pathophysiology, diagnosis and treatment. Tidsskr Nor Laegeforen. 2015;135:2172–2175. doi: 10.4045/tidsskr.15.1180. [DOI] [PubMed] [Google Scholar]
- 8.Price J.R., Mitchell E., Tidy E., Hunot V. Cognitive behaviour therapy for chronic fatigue syndrome in adults. Cochrane Database Syst Rev. 2008;2008 doi: 10.1002/14651858.CD001027.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Castro-Marrero J., Sáez-Francàs N., Santillo D., Alegre J. Treatment and management of chronic fatigue syndrome/myalgic encephalomyelitis: all roads lead to Rome. Br J Pharmacol. 2017;174:345–369. doi: 10.1111/bph.13702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fluge Ø, Rekeland I.G., Lien K. B-lymphocyte depletion in patients with myalgic Encephalomyelitis/Chronic fatigue syndrome: a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2019;170:585–593. doi: 10.7326/M18-1451. [DOI] [PubMed] [Google Scholar]
- 11.Taylor R.R. Rehabilitation programs for individuals with chronic fatigue syndrome. J Chronic Fatigue Syndr. 2006;13:41–55. doi: 10.1300/J092v13n01_05. [DOI] [Google Scholar]
- 12.Larun L., Brurberg K.G., Odgaard-Jensen J., Price J.R. Exercise therapy for chronic fatigue syndrome. Cochrane Database Syst Rev. 2019;10 doi: 10.1002/14651858.CD003200.pub8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang Y.-Y., Li X.-X. Traditional Chinese medicine for chronic fatigue syndrome: a systematic review of randomized clinical trials. Complement Ther Med. 2014;22:826–833. doi: 10.1016/j.ctim.2014.06.004. [DOI] [PubMed] [Google Scholar]
- 14.Kim H.G., Yoo S.R., Park H.J., Son C.-G. Indirect moxibustion (CV4 and CV8) ameliorates chronic fatigue: a randomized, double-blind, controlled study. J Altern Complement Med. 2013;19:134–140. doi: 10.1089/acm.2011.0503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim H.-G., Cho J.-H., Yoo S.-R. Antifatigue effects of Panax ginseng C.A. Meyer: a randomised, double-blind, placebo-controlled trial. PLoS One. 2013;8:e61271. doi: 10.1371/journal.pone.0061271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheon C., Kang S., Ko Y. Sipjeondaebo-tang in patients with breast cancer with fatigue: a protocol for a pilot, randomised, double-blind, placebo-controlled, cross-over trial. BMJ Open. 2018;8:e021242. doi: 10.1136/bmjopen-2017-021242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Moher D., Hopewell S., Schulz K.F. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ. 2010;340:c869. doi: 10.1136/bmj.c869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schulz K.F., Altman D.G., Moher D. CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials. BMJ. 2010;340:c332. doi: 10.1136/bmj.c332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cheng C., Wu T., Shang H. CONSORT extension for chinese herbal medicine formulas 2017: recommendations, explanation, and elaboration. Ann Intern Med. 2017;167:112–121. doi: 10.7326/M16-2977. [DOI] [PubMed] [Google Scholar]
- 20.Vercoulen J.H., Swanink C.M., Fennis J.F., Galama J.M., van der Meer J.W., Bleijenberg G. Dimensional assessment of chronic fatigue syndrome. J Psychosom Res. 1994;38:383–392. doi: 10.1016/0022-3999(94)90099-x. [DOI] [PubMed] [Google Scholar]
- 21.Bültmann U., de Vries M., Beurskens A.J., Bleijenberg G., Vercoulen J.H., Kant I. Measurement of prolonged fatigue in the working population: determination of a cutoff point for the checklist individual strength. J Occup Health Psychol. 2000;5:411–416. doi: 10.1037//1076-8998.5.4.411. [DOI] [PubMed] [Google Scholar]
- 22.Hewlett S., Dures E., Almeida C. Measures of fatigue: Bristol rheumatoid arthritis fatigue multi-dimensional questionnaire (BRAF MDQ), Bristol rheumatoid arthritis fatigue numerical rating scales (BRAF NRS) for severity, effect, and coping, chalder fatigue questionnaire (CFQ), checklist. Arthritis Care Res. 2011;63(Suppl 1):S263–86. doi: 10.1002/acr.20579. [DOI] [PubMed] [Google Scholar]
- 23.Krupp L.B., LaRocca N.G., Muir-Nash J., Steinberg A.D. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol. 1989;46:1121–1123. doi: 10.1001/archneur.1989.00520460115022. [DOI] [PubMed] [Google Scholar]
- 24.Morriss R., Wearden A., Mullis R. Exploring the validity of the chalder fatigue scale in chronic fatigue syndrome. J Psychosom Res. 1998;45:411–417. doi: 10.1016/S0022-3999(98)00022-1. [DOI] [PubMed] [Google Scholar]
- 25.Herdman M., Gudex C., Lloyd A. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L) Qual Life Res. 2011;20:1727–1736. doi: 10.1007/s11136-011-9903-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Buysse D.J., Reynolds C.F., 3rd, Monk T.H., Berman S.R., Kupfer D.J. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 27.Kim J., Ku B., Kim J., Kim Y., Kim K. Study on reliability and validity of the qi blood yin yang deficiency questionnaire. J Physiol Pathol Korean Med. 2014;28:346–354. internal-pdf://145.144.205.231/DRSRDH_2014_v28n3_346.pdf [Google Scholar]
- 28.Kim J., Ku B., Kim K.H. Validation of the qi blood yin yang deficiency questionnaire on chronic fatigue. Chin Med. 2016;11:24. doi: 10.1186/s13020-016-0092-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Blockmans D., Persoons P., Van Houdenhove B., Bobbaers H. Does methylphenidate reduce the symptoms of chronic fatigue syndrome? Am J Med. 2006;119:167. doi: 10.1016/j.amjmed.2005.07.047. e23-167.e30. [DOI] [PubMed] [Google Scholar]
- 30.Hornig M., Gottschalk G., Peterson D.L. Cytokine network analysis of cerebrospinal fluid in myalgic encephalomyelitis/chronic fatigue syndrome. Mol Psychiatry. 2016;21:261–269. doi: 10.1038/mp.2015.29. [DOI] [PubMed] [Google Scholar]
- 31.Chino A., Sakurai H., Choo M.-K. Juzentaihoto, a Kampo medicine, enhances IL-12 production by modulating Toll-like receptor 4 signaling pathways in murine peritoneal exudate macrophages. Int Immunopharmacol. 2005;5:871–882. doi: 10.1016/j.intimp.2005.01.004. [DOI] [PubMed] [Google Scholar]
- 32.Chen R., Moriya J., Yamakawa J.-I., Takahashi T., Kanda T. Traditional Chinese medicine for chronic fatigue syndrome. Evid Based Complement Alternat Med. 2010;7:3–10. doi: 10.1093/ecam/nen017. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available upon request from the corresponding author.