Abstract
Coronavirus disease-2019 (COVID-19) pandemic continues to threaten patients, societies, and economic and healthcare systems around the world. Like many other diseases, the host immune system determines the progress of COVID-19 and fatality. Modulation of inflammatory response and cytokine production using immunonutrition is a novel concept that has been applied to other diseases as well. Zinc, one of the anti-inflammatory and antioxidant micronutrient found in food with well-established role in immunity, is currently being used in some clinical trials against COVID-19. This review integrates the contemporary studies of role of zinc in antiviral immunity along with discussing its potential role against COVID-19, and ongoing COVID-19 clinical trials using zinc.
Keywords: COVID-19, Clinical trials, Immunity, Zinc
Introduction
As the whole world is grappling with coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), there is a frantic race for finding treatment regimens based on current knowledge until effective vaccine and ad hoc drugs are developed. Indeed, therapeutic approaches against COVID-19, mostly centered on the management of its immunopathology and/or tailored to directly control viral replication, are for now based on off-label prescription. Several human trials are currently in progress to assess the therapeutic indexes of drugs already approved for other diseases (i.e., drug repurposing), and in combination with dietary supplements like vitamin C, vitamin D, vitamin B12, probiotics, and zinc (Zn). Zn is a common theme in both prophylactic and curative COVID-19 clinical studies using nutritional supplements.
Zn, the second most abundant trace metal in the human body after iron, is essential for multiple cellular functions including maintenance of immune health. Notably, “Zn proteome” is estimated to encompass around 3000 proteins [1] including ∼ 750 Zn finger transcription factors, a number of which are deeply implicated in homeostasis and inflammation. Ever since Zn deficiency was demonstrated as a human disease more than 56 years ago, it has been implicated in immune dysfunction, growth retardation, hypogonadism, and cognitive impairment [2, 3]. Importantly, Zn also plays a critical role in antiviral immunity [4, 5]. Readers are referred to specialized reviews on essential role of Zn in human health (including Zn homeostasis and transporters) and other non-viral/viral diseases [5–16].
With so many clinical trials going on against the COVID-19 pandemic, there is scientific curiosity about the nature and basis of these trials [17]. In this review, we will discuss the basis of clinical trials using adjunct Zn therapy for COVID-19 through the prism of its indispensable role in antiviral immunity along with briefly discussing immunopathology of COVID-19.
COVID-19 Immunopathology
As already shown for SARS and Middle East respiratory syndrome (MERS), the host immune response plays a central role also in the pathology associated to SARS-CoV-2 infection, as the fatalities occurring among COVID-19 patients are often preceded by the so-called cytokine storm, a massive systemic release of proinflammatory cytokines such as interleukin (IL)-1b, IL-6, and tumor necrosis factor-α (TNF-α). These events ultimately contribute to acute respiratory distress syndrome (ARDS) and lead to vascular hyperpermeability, diffuse coagulopathy, multi-organ failure, and eventually death [18, 19]. These cytokines are mainly secreted by innate immune cells such as monocytes, macrophages, and dendritic cells (DC), which are supposed to act mainly in the early phases of infections. However, cytokine storm occurs at late stages of SARS-CoV-2 infection, when the adaptive, instead of the innate immune response, is known to become prominent.
A series of timely coordinated actions are required for an effective immune response against viral infections, and the agenda in which these actions move may be outlined by opposing early/innate versus late/adaptive immune response waves. Firstly, innate immune and infected tissue cells initiate primitive and unspecific reactions in an attempt to restrain viral replication by producing type-I interferons (IFN), IFN-α and IFN-β, and to attract blood cells to the site of infection by producing chemokine and TNF-α, which activates endothelial cells enhancing vascular adhesion and diapedesis of leukocytes, and by secreting other proinflammatory cytokines to activate leukocytes. It is peculiar that, while SARS-CoV-2 replication in permissive cells may be effectively inhibited by exogenously added type-I IFN, endogenous production of IFN-α and IFN-β upon infection is inhibited by proteins encoded by SARS-CoV-related viruses, whereas chemokines and inflammatory cytokines are highly produced contributing to an unbalanced response [20, 21].
During this first phase of the immune response, DC migrate to lymph nodes and initiate a complex and slower intercellular cascade, ultimately culminating in the expansion and maturation of adaptive, virus-specific T and B lymphocytes. Seroconversion—i.e., the appearance in the blood of antibodies against immunogenic viral proteins—and probably the clonal expansion of virus-specific T cells, occurs in most COVID-19 patients 10 days after symptom onset [22] and severe disease and cytokine storm are associated in time and magnitude with the immunoglobulin response in SARS-CoV-1 [23] and SARS-CoV-2 infection [24, 25].
Strikingly, a similar cytokine storm is also the most notable side effect of chimeric antigen receptor T (CAR-T) cell therapy, and its severity is forecasted by the extent of tumor burden [26], leading to the hypothesis that a high antigen (i.e., viral) load at the time of the (late) adaptive immune cell activity against SARS-CoV-2, when antibodies and virus-specific T cells arise, may favor a massive second wave of cytokine release by innate cells in a positive feedback loop. Much effort is now trying to decipher, in the immunotherapy setting, the exact mechanisms pushing this positive feedback from adaptive to innate cells, which may deserve our attention to anticipate plausible pathways to interfere with COVID-19 immunopathology. One line of evidence points to cytokines produced by T cells upon antigenic stimulation, namely to TNF-α [27] and granulocyte-macrophage colony-stimulating factor (GM-CSF) [28], as triggers of the secondary systemic toxic cytokine release by innate cells. Importantly, these two cytokines are unnecessary (both) or detrimental (GM-CSF) for on-target effects of CAR-T cells [28], which suggests that they may be dispensable also for the antiviral response to SARS-CoV-2.
There is a clear sex- and age-dependent bias in COVID-19-related mortality. Better outcomes in women, for instance, may be the result of a more pronounced IFN-α production by plasmacytoid DC [29] during initial phases of infection. The more effective viral containment operated by such a response could be assumed to lower the antigen load at the onset of the adaptive wave and prevent the cytokine storm. In studies from patients during the 2003 SARS-CoV-1 outbreak, treatment with synthetic type-I IFN in conjunction with steroids has proven useful to ameliorate pneumonia course [30], and early, in contrast to delayed, type-I IFN treatment rescued mice from death after SARS-CoV-1 infection [31]. On the other hand, aging of the immune system, which is associated to chronic low-level activation of innate cells, also entails a delayed and inflated response upon pathogen encountering [32]. Lastly, with age, adaptive cells become slow in mounting antigen-specific responses to novel pathogens secondary to the loss of the naive B and T cell repertoire due to thymic involution [33].
Immunological Functions of Zn
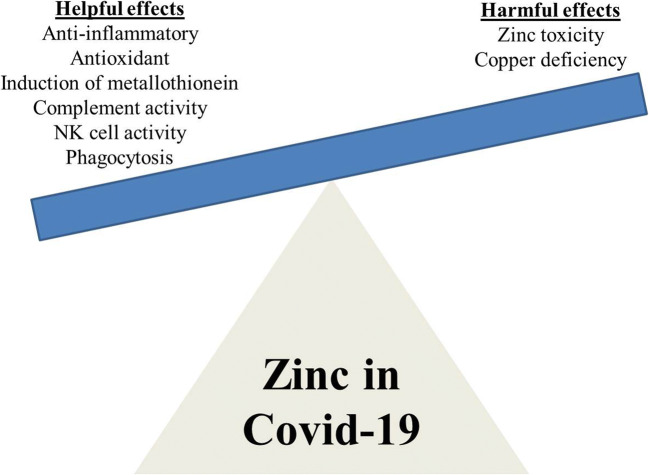
Data gleaned from seminal animal model studies have added considerable knowledge about the principal roles of Zn in the immune system. Zn is an essential micronutrient, which plays an important role in the physiology of the immune system by acting as signaling molecule. Zn not only acts as an anti-inflammatory agent but also functions as an antioxidant stabilizing membrane [34]. Zn deficiency impinges on survival of immune cells and adversely affects important functions such as phagocytosis, target cell killing, and cytokine production (Fig. 1). It is clearly evident from studies on preclinical models that Zn deficiency plays a role in thymus and lymphoid tissue atrophy [35] and declines in the mechanisms of activation of both helper T cell [36] and cytotoxic CD8+ T cell responses [37]. Specifically, Zn deficiency leads to compromised immune system, as evidenced by degeneration of thymus, lymphopenia, and defective lymphocyte responses [3, 5, 34]. Zn deficiency causes immunodeficiency with severe lymphopenia that is characterized in part by a considerable decrease in developing B cell compartments in the bone marrow [3, 38]. Moreover, Zn potentiates IFN-α effect by an order of magnitude [39] which can be used to counteract IFN antagonism by SARS-CoV-2 proteins.
Fig. 1.
Balance between helpful and harmful effects of zinc for COVID-19
A marked neutrophilia is observed in severe COVID-19 patients [40]. Zn gluconate supplementation is able to reduce airway neutrophil infiltration and TNF-α release by inhibiting NF-kB-dependent transcription of inflammatory genes [41]. The Zn finger CCHC-type containing 3 (ZCCHC3) potentiates TLR3 signaling induced by double-strand RNA [42], which is produced during intracellular replication of coronaviruses.
In vitro, Zn deficiency induced enhanced IL-6 and IL-1β production, as well as higher expression of intercellular adhesion molecule 1 (ICAM-1), important for leukocytes’ extravasation, of CD86, a co-stimulatory molecule involved in T cell activation, and of HLA-DR, a MHC-II molecule, in cultured human monocytic cells [43]. Moreover, aged mice have lower plasma Zn concentration, and this was associated to higher IL-6 and ICAM-1 gene expression in splenocytes [43]. Thus, Zn supplementation may be instrumental to reduce inflammatory cytokines, particularly IL-6 and IL-1β, and at the same time to enhance the protective, though inhibited, type-I IFN response in SARS-CoV-2 infection. Zn deficiency indicates towards the decreased interferon-γ (IFN-γ) production, a major component of Th1 cytokine panel [44]. Since Th1 and Th2 responses are mutually inhibited by the effect of their respective signature cytokines IFN-γ and IL-4, such imbalance between Th1 and Th2 might lead to dysfunctions in cell-mediated immune response [34].
There can be a whole lot of factors for the development of Zn deficiency, whether geographic, socioeconomic, nutritional, or as a result of disease pathologies such as chronic viral infections. Notably, during infection, Zn levels decrease significantly and a person’s requirement for Zn may increase with the severity of the infection. Aged individuals, infants, and chronic alcoholics are particularly more susceptible to Zn deficiency, increasing their chances of acquiring life-threatening viral infections [45–48]. As Zn is indispensable for a strong immune response, Zn deficiency (persistent hypozincemia [serum Zn < 70 μg/dL]) can considerably dampen innate as well as adaptive antiviral immunity [4, 5].
The Role of Zn in Antiviral Immunity
There is very scant information available on the role and effect of Zn in SARS and coronavirus disease, even though literature is rapidly increasing [17, 49]. Much of the current knowledge about the use of Zn as an antiviral therapy and immunomodulatory agent has originated from studies done with other viral diseases (Readers are referred to these specialized articles for role of Zn in viral diseases [4, 5]). Zn is an essential constituent of thymulin hormone, which is involved in maturation and differentiation of T cells in thymus gland. Zn not only plays an important role in IL-2 and IFN-γ production but also stimulates macrophages to produce IL-12. IL-12 activates the natural killer cells and T cytotoxic cells. Both IFN-γ and IL-12 play a crucial role in destruction of various pathogens. Among anti-inflammatory cytokines, only IL-10 production is affected by Zn deficiency, whose enhanced levels adversely affect the functioning of macrophages and Th1 response [50].
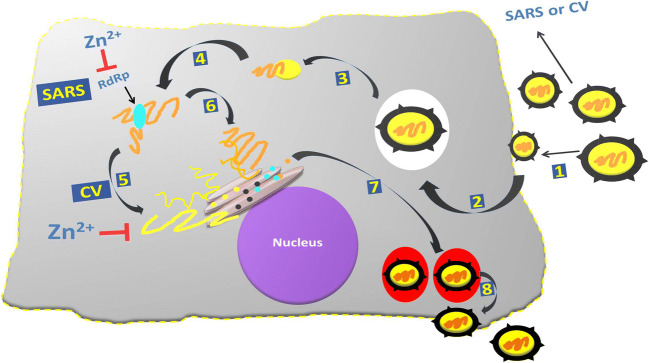
The rationale behind the use of Zn in COVID-19 studies aroused from the observation that Zn ions and Zn ionophores, such as pyrithione (PT), have previously been described as potent inhibitors of various RNA viruses [51]. Also, targeting of RNA-dependent RNA polymerase (RdRps) of RNA viruses like SARS-CoV-2 is more suitable for antiviral drug development as RdRp activity is strictly virus-specific and may be blocked without severely affecting key cellular functions. The basis of Zn therapy in coronavirus infection is shown in Fig. 2. It is known previously that Zn ions are involved in many different cellular processes, proper folding, and activity of various cellular enzymes and transcription factors. On the other hand, Zn2+ is probably an important cofactor for numerous viral proteins as well. Zn2+ can serve as intracellular second messenger and may trigger apoptosis or a decrease in protein synthesis at elevated concentrations [52–54]. Metallothioneins, a family of metal-binding proteins, regulate homeostasis of Zn and copper, alleviate heavy metal poisoning, and lessen the superoxide stress. Various evidence indicates that metallothionein expression is increased in response to bacterial and viral infections, and it is anticipated that upregulated biosynthesis of human metallothionein might play a significant role in nutritional immunity at the time of active infection [55].
Fig. 2.
Schematic representation of different stages of SARS-CoV-1 and coronavirus replication cycle. Viral attachment (1), entry (2), uncoating (3), transcription (4), viral protein translation (5), replication (6), assembly and maturation (7), and finally viral release (8). Data from in vitro studies has demonstrated two mechanisms by which zinc interferes with these viruses’ replication cycle steps that include viral genome transcription for SARS-Cov-1 (5), and viral protein translation and polypeptide processing for CV (6). However, to date, no studies have shown zinc-mediated inhibition of SARS-CoV-1 or CV entry, receptor binding, uncoating, assembly, and/or viral particle release. CV, coronavirus; RdRp, RNA-dependent RNA polymerase; SARS, severe acute respiratory syndrome coronavirus; Zn, zinc
In cell culture studies, inhibition of the replication of various RNA viruses was reported using high Zn2+ concentrations and addition of compounds that stimulate cellular import of Zn2+, such as hinokitiol, pyrrolidine dithiocarbamate, and PT as mentioned above (reviewed in) [5]. Around 10 years ago, an in vitro study demonstrated that the Zn ionophore PT in combination with Zn2+ is a potent inhibitor of the replication of SARS-CoV-1 [51]. In addition, authors also studied the effect of Zn ions on the RdRp of SARS-CoV-1 and demonstrated that Zn2+ directly inhibited the in vitro activity of RdRp (Fig. 2). More specifically, Zn2+ was found to block the SARS-CoV-1 RdRp elongation step along with reduced template binding. Further strengthening their claim, authors also showed that the Zn2+-mediated RdRp inhibition of SARS-CoV-1 could be reversed through the addition of a Zn2+ chelator [51].
Recently, Finzi (2020) has reported that treatment of four COVID-19 cases with high dose of zinc salts initiated the reduction of disease symptoms within 24 h after initiation of high dose zinc salt lozenges [56]. As adjuvant therapy, Zn (in appropriate dose) may provide protection through decreasing lung inflammation, enhancing mucociliary clearance, inhibiting the ventilator-induced lung injury, and immunomodulation in COVID-19 patients [49].
COVID-19: Interventional Clinical Trials of Drugs/Nutritional Supplements with Zn
As of 4 August 2020, 22 studies were found after searching for Zn and COVID-19 at the clinical trial website https://clinicaltrials.gov/ct2/home. However, upon narrowing down the search criteria, 19 studies were clinical trials and the remaining 3 were observational studies (NCT04326725, NCT04407572, NCT04412746). Furthermore, among the remaining 19 studies, one study (NCT04371952 entitled DYNAMIC Study [DoxycYcliNe AMbulatoIre COVID-19]) was based on the fact that doxycycline, a tetracycline antibiotic, is known to chelate Zn from matrix metalloproteinases, which may help in part to inhibit COVID-19 infection by limiting its ability to replicate in the host. There is no intervention of Zn supplementation to the patient, so this study was ruled out for present review. Three more studies (NCT04485130, NCT04491994, NCT04485169) also did not use zinc in the experimental arm, so similarly left out from the present article.
Among the remaining 15 studies (Table 1), 4 studies are exclusively using different combinations of dietary supplement including Zn as an intervention against COVID-19 (NCT04351490, NCT04342728, NCT04323228, NCT04468139 at serial nos. 1, 2, 3, and 4 of Table 1, respectively). None of the four studies is completed yet at the time of writing this review. The NCT04351490 trial is using combination of vitamin D3 with Zn gluconate with the rationale that these two micronutrients are able to modulate the immune response by reducing the inflammatory storm. Vitamin D, a steroid hormone synthesized from cholesterol, has recently been reported to have anti-fibrotic, anti-inflammatory, and immunomodulatory properties on the basis of various studies done with hepatitis C virus infection. Vitamin D impedes T cell proliferation, expression of IFN-γ, expression of IL-2, and CD8 T lymphocyte–mediated cytotoxicity [57].
Table 1.
Interventional clinical trials with zinc ± other drugs/dietary supplements for COVID-19
| S. no. | Clinical trial study title | ClinicalTrial.Org identifier | Intervention (drug/s) | Zinc dose and duration | Types of intervention/allocation | No. of subjects/age/sex |
|---|---|---|---|---|---|---|
| Clinical trials using zinc with dietary supplements | ||||||
| 1 | Impact of zinc and vitamin D3 supplementation on the survival of aged patients infected with COVID-19 | NCT04351490 |
• Dietary supplement: zinc gluconate • Dietary supplement: 25-OH cholecalciferol |
Zinc gluconate 15 mg × 2 per day during 2 months | Treatment/randomized-open label | 3140/60 years and older/all sex |
| 2 | Coronavirus 2019 (COVID-19)—using ascorbic acid and zinc supplementation | NCT04342728 |
• Dietary supplement: ascorbic acid • Dietary supplement: zinc gluconate • Dietary supplement: ascorbic acid and zinc gluconate • Other: standard of care |
Zinc gluconate 50 mg ± ascorbic acid to be taken daily at bedtime for 10 days | Supportive care/randomized-open label | 520/18 years and older/all sex |
| 3 | Anti-inflammatory/antioxidant oral nutrition supplementation in COVID-19 | NCT04323228 |
• Dietary supplement: oral nutrition supplement (ONS) enriched in eicosapentaenoic acid, gammalinolenic acid, antioxidants including zinc • Dietary supplement: isocaloric/isonitrogenous ONS |
ONS containing 5.7 mg zinc for 14 days | Supportive care/randomized-double masking (participant, care provider) | 30/18 years to 65/all sex |
| 4 | The study of quadruple therapy zinc, quercetin, bromelain and vitamin C on the clinical outcomes of patients infected with COVID-19 | NCT04468139 |
Drug: quercetin Dietary supplement: bromelain Drug: zinc Drug: vitamin C |
Zinc 50 mg orally daily dose | Treatment/single group assignment/masking: none (open label) | 60/18 years and older (adult, older adult)/all sex |
| Preventive clinical trials using zinc with hydroxychloroquine | ||||||
| 5 | A study of hydroxychloroquine and zinc in the prevention of COVID-19 infection in military healthcare workers | NCT04377646 |
• Drug: hydroxychloroquine • Drug: hydroxychloroquine (placebo) • Drug: zinc • Drug: zinc (placebo) |
Hydroxychloroquine ± zinc (15 mg) at daily dose up to 2 months | Prevention/randomized/double blind randomized clinical trial with 3 arms-triple masking (participant, care provider, investigator)—random blind allocation | 660/18 years to 65 years/all sex |
| 6 | A study of hydroxychloroquine, vitamin C, vitamin D, and zinc for the prevention of COVID-19 infection | NCT04335084 |
• Drug: hydroxychloroquine • Dietary supplement: vitamin C • Dietary supplement: vitamin D • Dietary supplement: zinc |
No information available on zinc dose | Prevention/randomized-double masking (participant, investigator) | 600/18 years and older/all sex |
| 7 | COVID-19 prophylaxis with hydroxychloroquine associated with zinc for high-risk healthcare workers | NCT04384458 |
• Drug: hydroxychloroquine • Drug: zinc sulfate |
Hydroxychloroquine with zinc sulfate (66 mg) daily for 50 days | Prevention/randomized-open label | 400/18 years to 70 years/all sex |
| 8 | A preventive treatment for migrant workers at high-risk of Covid-19 | NCT04446104 |
• Drug: hydroxychloroquine sulfate • Drug: ivermectin 3 mg tab • Drug: zinc • Drug: povidone-iodine • Dietary supplement: vitamin C |
Zinc tablet 80 mg/vitamin C 500 mg daily for 42 days | Prevention/randomized intervention/parallel assignment/open label | 5000/21 years to 60 years/all sex |
| 9 | Zinc with chloroquine/hydroxychloroquine in treatment of COVID-19 | NCT04447534 |
• Drug: chloroquine • Drug: zinc |
No information available on zinc dose |
Treatment/randomized •Intervention model: parallel assignment •Masking: double (participant, care provider) |
200/18 years and older/all sex |
| Treatment clinical trials using zinc with antiviral drugs/dietary supplements | ||||||
| 10 | Hydroxychloroquine and zinc with either azithromycin or doxycycline for treatment of COVID-19 in outpatient setting | NCT04370782 |
• Drug: hydroxychloroquine • Drug: azithromycin • Drug: zinc sulfate • Drug: doxycycline |
Zinc sulfate 220 mg once daily for 5 days | Treatment/randomized-open label | 750/30 years and older/all sex |
| 11 | A study of quintuple therapy to treat COVID-19 infection | NCT04334512 |
• Drug: hydroxychloroquine • Drug: azithromycin • Dietary supplement: vitamin C • Dietary supplement: vitamin D • Dietary supplement: zinc |
No information available on zinc dose | Treatment/randomized-double masking (participant, investigator) | 600/18 years and older/all sex |
| 12 | International ALLIANCE study of therapies to prevent progression of COVID-19 | NCT04395768 |
• Dietary supplement: vitamin C • Drug: hydroxychloroquine • Drug: azithromycin • Dietary supplement: zinc citrate • Dietary supplement: vitamin D3 • Dietary supplement: vitamin B12 |
Zinc citrate (30 mg) daily | Treatment/randomized-double masking (investigator, outcomes assessor) | 200/18 years and older |
| 13 | New antiviral drugs for treatment of COVID-19 | NCT04392427 | • Drug: treatment group: will receive a combination of nitazoxanide, ribavirin, ivermectin, and zinc | No information available on zinc dose | Treatment/randomized-single masking (outcomes assessor) | 100/12 years and older/all sex |
| 14 | Efficacy of subcutaneous ivermectin with or without zinc and Nigella sativa in COVID-19 patients | NCT04472585 |
• Drug: Nigella sativa/black cumin • Drug: ivermectin injectable solution • Other: placebo • Drug: zinc |
Zinc sulfate 20 mg 3 times a day |
Treatment/randomized, controlled, multi-armed, open-label, interventional study Masking: quadruple (participant, care provider, investigator, outcomes assessor) |
40/18 years and older/all sex |
| 15 | Trial of combination therapy to treat COVID-19 infection | NCT04482686 |
• Drug: ivermectin • Drug: doxycycline Hcl • Dietary supplement: zinc • Dietary supplement: vitamin D3 • Dietary supplement: vitamin C |
No information available on zinc dose | Treatment/randomized intervention model/parallel assignment/masking: triple (participant, care provider, investigator) | 300/18 years to 75 years/all sex |
HCQ, hydroxychloroquine; ONS, oral nutrition supplement
The NCT04342728 trial is primarily studying whether ascorbic acid and Zn gluconate either alone or in combination can decrease the duration of symptoms in newly diagnosed COVID-19 patients. Ascorbic acid, also known as vitamin C which has potent antioxidant properties, has a very long history (> 45 years) in combating or relieving the symptoms of rhinoviruses or common cold [58], and having immune booster properties [59]. Interestingly, various in vitro animal and clinical trials have shown that vitamin C may affect susceptibility to lower respiratory tract infections during the 2003 SARS epidemic [59]. Taking a holistic approach, clinical trial NCT04323228 is using an anti-inflammatory and antioxidant oral nutrition supplement against COVID-19 with rationale that n-3 fatty acids, vitamin A, vitamin C, vitamin E, selenium, and Zn in extraordinary doses may modulate the immune system response and improve the cytokine storm–associated COVID-19 (Readers are referred to specialized articles on antioxidant and/or antiviral properties of various nutrients [4]).
Four clinical studies (NCT04377646, NCT04335084, NCT04384458, NCT04446104 at serial nos. 5, 6, 7, and 8 in Table 1, respectively) are preventive studies, which are studying whether hydroxychloroquine and/or vitamin supplements in combination with Zn are effective as a prophylaxis for asymptomatic healthcare workers/general public. Hydroxychloroquine, derivative of chloroquine having anti-inflammatory, immunosuppressive, and anti-autophagy activities, is an anti-malarial drug and anti-rheumatologic agent. Besides, both hydroxychloroquine and chloroquine constrain pH-dependent steps of SARS-CoV-2 replication by increasing pH in intracellular vesicles [60]. Due to these properties, hydroxychloroquine with Zn and/or azithromycin has been touted as the medicine and as a prophylactic drug for COVID-19 [61–63]. More importantly, chloroquine and hydroxychloroquine work as Zn ionophore, thereby increasing the intracellular Zn concentration [64]. This increase of intracellular Zn concentration will in turn may help in the inhibition of RdRp of SARS-CoV-2 (reviewed in) [51, 61].
The rest of 5 clinical trials (serial nos. 10–15 of Table 1) are aimed at finding better treatment option for COVID-19 by using various combinations of antiviral drugs/dietary supplements with Zn. The results of these studies are still awaited. The common denominator among the above mentioned trials is the use of Zn by virtue of its antioxidant, anti-inflammatory, and antiviral properties [4, 5]. Based on its antioxidant properties, Zn can protect against age-associated macular degeneration, induced by oxidative stress [65]. In general, these properties are the therapeutic basis of the indication of Zn for the treatment of wounds, burns, and acne vulgaris. In addition, Zn is very effective in controlling toxic levels of non-ceruloplasmin copper in Wilson’s disease and other copper-related diseases [7]. Acting synergistically, various antioxidants and anti-inflammatory nutrients/drugs combined with antiviral drugs may be beneficial at least in alleviating the COVID-19 symptoms or halting the progression to more severe form of COVID-19. For a clear picture, results of these randomized control trials will be eagerly awaited.
Word of Caution: Zn Toxicity
The recommended dietary allowance (RDA) for Zn is 11 mg/day for men and 8 mg/day for women [66]. Zn absorption is influenced by some foods; e.g., animal proteins result in greater Zn absorption, while phytates reduce its absorption (reviewed in [67]). Zn elimination half-life is in the range of 0.9–1.2 h.
Zn deficiency is far more widespread in population than Zn toxicity (Zn toxicity is very sporadic and occurs very rarely). In comparison to several other metal ions with similar chemical properties, Zn is relatively harmless. Having said that, caution should be practiced against indiscriminate self-medication and chronic use of Zn supplements without proper medical prescription/monitoring as Zn toxicity has also been reported in the literature [68–70]. Acute Zn intoxication is a rare event though exposure to high doses leads to toxic effects. However, it is well established that copper deficiency is associated with taking up large doses of supplemental Zn over extended periods of time [71–73]. In addition, chronic use of Zn as supplements [68] or as a medication [74] can block intestinal absorption of copper [75]. The finely tuned and synchronized systemic homeostasis and efficient regulatory mechanisms keep a check on the cytotoxic doses of exogenous Zn. It is the endogenous Zn that plays a pivotal role in Zn-induced cytotoxic events in single cells. Brain, in particular, is very sensitive to Zn toxicity.
Zinc as a Two-Edged Sword
Using Zn as a supplement or treatment adjuvant is a two-edged sword. Large doses of Zn (300 mg/day) over 6 weeks can affect the immune response, impairing lymphocyte, and neutrophil function [76]. On this basis, a concern had arisen that long-term Zn treatment can cause suppression of the immune system. On the similar lines, using excess Zn supplementation as an antiviral therapy can also do harm to the immune system. Maywald et al. have shown in an in vitro study that excess Zn supplementation can in fact reduce the expression of IFN-γ (a key cytokine for T-helper-1 immune reactions) by reducing interferon regulatory factor 1 expression in regulatory T cells [77]. However, it has been shown for elderly persons that zinc supplementation increased interferon-alpha (IFN-α) production by leukocytes in vitro as compared to younger subjects [78]. This observation necessitates the need to continuously monitor the levels of Zn to obtain maximum therapeutic efficacy. Cellular Zn intake can be improved by ionophores including chloroquine and some of its derivatives such as hydroxychloroquine [64]. Alternatively, natural ionophores of potential use with a good tolerability profile are quercetin and epigallocatechin gallate [79].
Summary and Future Directions
With no slowing down of COVID-19 pandemic as indicated by 18 million confirmed cases including 0.69 million deaths worldwide as of August 4, 2020 (https://covid19.who.int/), researchers are seeking ways in which to potentially protect people from the SARS-CoV-2 or to alleviate its effects once caught. One such means is Zn therapy in addition to other antiviral drugs. Zn is well tolerated and it is best known for its antioxidant, anti-inflammatory, immunomodulatory, and antiviral activities, the latter possibly mediated by its ability to inhibit RNA virus RdRp [51], thus protecting the body’s cells and tissues from viral infection, oxidative damage, and dysfunction. Based on the current knowledge of beneficial and harmful effects of Zn (Fig. 1), it can be safely concluded that risk to reward ratio is in favor of Zn supplementation in COVID-19. However, the clinical and preclinical data on this aspect is very scanty now and results of current clinical trials employing Zn in COVID-19 can somehow shed more light on the efficacy of Zn against viral infections in vivo. The investigators of the different studies hope to complete the trials in the near future.
Basic and experimental research is still at infancy stage with regard to antiviral mechanisms, clinical benefits, and optimal dose of Zn supplementation as a therapeutic treatment as well as a preventative measure for viral infections including SARS-CoV-2. Generous funding is the need of hour to rigorously pursue this aspect of basic research and provide conclusive evidence to the clinical trials and assumptions based on current knowledge. Although the findings of these trials will be too late for the many thousands of people currently infected with COVID-19, these studies will offer valued data as to the potential alleviation of symptoms by adjunct Zn during future viral/COVID-19 outbreaks and provide platform for future research avenues.
Acknowledgments
The authors duly acknowledge the WHO, NCBI, and NIH for providing free online scientific research articles/information/clinical trials pertaining to COVID-19.
Authors’ Contributions
All authors contributed to the preparation of this review.
Compliance with Ethical Standards
Competing Interests
Dr. Rosanna Squitti is the Chief Scientific Officer of IGEA Pharma N.V.; she has some shares in IGEA Pharma N.V. The other authors declare no competing financial interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Maret W. Zinc and the zinc proteome. Met Ions Life Sci. 2013;12:479–501. doi: 10.1007/978-94-007-5561-1_14. [DOI] [PubMed] [Google Scholar]
- 2.Prasad AS, Miale A, Jr, Farid Z, Sandstead HH, Schulert AR. Clinical and experimental. Zinc metabolism in patients with the syndrome of iron deficiency anemia, hepatosplenomegaly, dwarfism, and hypogonadism. 1963. J Lab Clin Med. 1990;116(5):737–749. [PubMed] [Google Scholar]
- 3.Fukada T, Hojyo S, Hara T, Takagishi T. Revisiting the old and learning the new of zinc in immunity. Nat Immunol. 2019;20(3):248–250. doi: 10.1038/s41590-019-0319-z. [DOI] [PubMed] [Google Scholar]
- 4.Gupta S, Read SA, Shackel NA, Hebbard L, George J, Ahlenstiel G. The role of micronutrients in the infection and subsequent response to hepatitis C virus. Cells. 2019;8(6):603. doi: 10.3390/cells8060603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Read SA, Obeid S, Ahlenstiel C, Ahlenstiel G. The role of zinc in antiviral immunity. Adv Nutr. 2019;10(4):696–710. doi: 10.1093/advances/nmz013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.King JC. Zinc: an essential but elusive nutrient. Am J Clin Nutr. 2011;94(2):679S–684S. doi: 10.3945/ajcn.110.005744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sensi SL, Granzotto A, Siotto M, Squitti R. Copper and zinc dysregulation in Alzheimer’s disease. Trends Pharmacol Sci. 2018;39(12):1049–1063. doi: 10.1016/j.tips.2018.10.001. [DOI] [PubMed] [Google Scholar]
- 8.Sensi SL, Paoletti P, Bush AI, Sekler I. Zinc in the physiology and pathology of the CNS. Nat Rev Neurosci. 2009;10(11):780–791. doi: 10.1038/nrn2734. [DOI] [PubMed] [Google Scholar]
- 9.Fukada T, Yamasaki S, Nishida K, Murakami M, Hirano T. Zinc homeostasis and signaling in health and diseases: zinc signaling. J Biol Inorg Chem. 2011;16(7):1123–1134. doi: 10.1007/s00775-011-0797-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hara T, Takeda TA, Takagishi T, Fukue K, Kambe T, Fukada T. Physiological roles of zinc transporters: molecular and genetic importance in zinc homeostasis. J Physiol Sci. 2017;67(2):283–301. doi: 10.1007/s12576-017-0521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hojyo S, Fukada T. Zinc transporters and signaling in physiology and pathogenesis. Arch Biochem Biophys. 2016;611:43–50. doi: 10.1016/j.abb.2016.06.020. [DOI] [PubMed] [Google Scholar]
- 12.Mezzaroba L, Alfieri DF, Colado Simão AN, Vissoci Reiche EM. The role of zinc, copper, manganese and iron in neurodegenerative diseases. Neurotoxicology. 2019;74:230–241. doi: 10.1016/j.neuro.2019.07.007. [DOI] [PubMed] [Google Scholar]
- 13.Sanna A, Firinu D, Zavattari P, Valera P. Zinc status and autoimmunity: a systematic review and meta-analysis. Nutrients. 2018;10(1):68. doi: 10.3390/nu10010068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Subramanian Vignesh K, Deepe GS., Jr Immunological orchestration of zinc homeostasis: the battle between host mechanisms and pathogen defenses. Arch Biochem Biophys. 2016;611:66–78. doi: 10.1016/j.abb.2016.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vasto S, Mocchegiani E, Candore G, Listì F, Colonna-Romano G, Lio D, Malavolta M, Giacconi R, Cipriano C, Caruso C. Inflammation, genes and zinc in ageing and agerelated diseases. Biogerontology. 2006;7(5-6):315–327. doi: 10.1007/s10522-006-9046-6. [DOI] [PubMed] [Google Scholar]
- 16.Gumulec J, Masarik M, Krizkova S, Adam V, Hubalek J, Hrabeta J, Eckschlager T, Stiborova M, Kizek R. Insight to physiology and pathology of zinc(II) ions and their actions in breast and prostate carcinoma. Curr Med Chem. 2011;18(33):5041–5051. doi: 10.2174/092986711797636126. [DOI] [PubMed] [Google Scholar]
- 17.Rahman MT, Idid SZ (2020) Can Zn be a critical element in COVID-19 treatment? Biol Trace Elem Res. 10.1007/s12011-020-02194-9 [DOI] [PMC free article] [PubMed]
- 18.Nile SH, Nile A, Qiu J, Li L, Jia X, Kai G. COVID-19: pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020;53:66–70. doi: 10.1016/j.cytogfr.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ye Q, Wang B, Mao J. The pathogenesis and treatment of the ‘cytokine storm’ in COVID-19. J Inf Secur. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Møller R, Jordan TX, Oishi K, Panis M, Sachs D, Wang TT, Schwartz RE, Lim JK, Albrecht RA, ten Oever BR. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020;181(5):1036–1045.e9. doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kopecky-Bromberg SA, Martínez-Sobrido L, Frieman M, Baric RA, Palese P. Severe acute respiratory syndrome coronavirus open reading frame (ORF) 3b, ORF 6, and nucleocapsid proteins function as interferon antagonists. J Virol. 2007;81(2):548–557. doi: 10.1128/JVI.01782-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Okba NMA, Müller MA, Li W, Wang C, GeurtsvanKessel CH, Corman VM, Lamers MM, Sikkema RS, de Bruin E, Chandler FD, Yazdanpanah Y, le Hingrat Q, Descamps D, Houhou-Fidouh N, Reusken CBEM, Bosch BJ, Drosten C, Koopmans MPG, Haagmans BL. Severe acute respiratory syndrome coronavirus 2-specific antibody responses in coronavirus disease patients. Emerg Infect Dis. 2020;26(7):1478–1488. doi: 10.3201/eid2607.200841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee N, Chan PK, Ip M, Wong E, Ho J, Ho C, Cockram CS, Hui DS. Anti-SARS-CoV IgG response in relation to disease severity of severe acute respiratory syndrome. J Clin Virol. 2006;35(2):179–184. doi: 10.1016/j.jcv.2005.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lou B, Li T-D, Zheng S-F, Su YY, Li ZY, Liu W, Yu F, Ge SX, Zou QD, Yuan Q, Lin S, Hong CM, Yao XY, Zhang XJ, Wu DH, Zhou GL, Hou WH, Li TT, Zhang YL, Zhang SY, Fan J, Zhang J, Xia NS, Chen Y. Serology characteristics of SARS-CoV-2 infection since exposure and post symptom onset. Eur Respir J. 2020;56:2000763. doi: 10.1183/13993003.00763-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zheng S, Fan J, Yu F, Feng B, Lou B, Zou Q, Xie G, Lin S, Wang R, Yang X, Chen W, Wang Q, Zhang D, Liu Y, Gong R, Ma Z, Lu S, Xiao Y, Gu Y, Zhang J, Yao H, Xu K, Lu X, Wei G, Zhou J, Fang Q, Cai H, Qiu Y, Sheng J, Chen Y, Liang T. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January-March 2020: retrospective cohort study. BMJ. 2020;369:m1443. doi: 10.1136/bmj.m1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brudno JN, Kochenderfer JN. Recent advances in CAR T-cell toxicity: mechanisms, manifestations and management. Blood Rev. 2019;34:45–55. doi: 10.1016/j.blre.2018.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li J, Piskol R, Ybarra R, Chen YJ, Li J, Slaga D, Hristopoulos M, Clark R, Modrusan Z, Totpal K, Junttila MR, Junttila TT. CD3 bispecific antibody-induced cytokine release is dispensable for cytotoxic T cell activity. Sci Transl Med. 2019;11(508):eaax8861. doi: 10.1126/scitranslmed.aax8861. [DOI] [PubMed] [Google Scholar]
- 28.Sterner RM, Sakemura R, Cox MJ, Yang N, Khadka RH, Forsman CL, Hansen MJ, Jin F, Ayasoufi K, Hefazi M, Schick KJ, Walters DK, Ahmed O, Chappell D, Sahmoud T, Durrant C, Nevala WK, Patnaik MM, Pease LR, Hedin KE, Kay NE, Johnson AJ, Kenderian SS. GM-CSF inhibition reduces cytokine release syndrome and neuroinflammation but enhances CAR-T cell function in xenografts. Blood. 2019;133(7):697–709. doi: 10.1182/blood-2018-10-881722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Seillet C, Laffont S, Trémollières F, Rouquié N, Ribot C, Arnal JF, Douin-Echinard V, Gourdy P, Guéry JC. The TLR-mediated response of plasmacytoid dendritic cells is positively regulated by estradiol in vivo through cell-intrinsic estrogen receptor α signaling. Blood. 2012;119(2):454–464. doi: 10.1182/blood-2011-08-371831. [DOI] [PubMed] [Google Scholar]
- 30.Loutfy MR, Blatt LM, Siminovitch KA, Ward S, Wolff B, Lho H, Pham DH, Deif H, LaMere EA, Chang M, Kain KC, Farcas GA, Ferguson P, Latchford M, Levy G, Dennis JW, Lai EK, Fish EN. Interferon alfacon-1 plus corticosteroids in severe acute respiratory syndrome: a preliminary study. JAMA. 2003;290(24):3222–3228. doi: 10.1001/jama.290.24.3222. [DOI] [PubMed] [Google Scholar]
- 31.Channappanavar R, Fehr AR, Vijay R, Mack M, Zhao J, Meyerholz DK, Perlman S. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice. Version 2. Cell Host Microbe. 2016;19(2):181–193. doi: 10.1016/j.chom.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nomellini V, Gomez CR, Kovacs EJ. Aging and impairment of innate immunity. Contrib Microbiol. 2008;15:188–205. doi: 10.1159/000136358. [DOI] [PubMed] [Google Scholar]
- 33.Thomas R, Wang W, Su DM. Contributions of age-related thymic involution to immunosenescence and inflammaging. Immun Ageing. 2020;17:2. doi: 10.1186/s12979-020-0173-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Prasad AS. Zinc: role in immunity, oxidative stress and chronic inflammation. Curr Opin Clin Nutr Metab Care. 2009;12(6):646–652. doi: 10.1097/MCO.0b013e3283312956. [DOI] [PubMed] [Google Scholar]
- 35.Shankar AH, Prasad AS. Zinc and immune function: the biological basis of altered resistance to infection. Am J Clin Nutr. 1998;68(2 Suppl):447S–463S. doi: 10.1093/ajcn/68.2.447S. [DOI] [PubMed] [Google Scholar]
- 36.Fraker PJ, DePasquale-Jardieu P, Zwickl CM, Luecke RW. Regeneration of T-cell helper function in zinc-deficient adult mice. Proc Natl Acad Sci U S A. 1978;75(11):5660–5664. doi: 10.1073/pnas.75.11.5660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Frost P, Rabbani P, Smith J, Prasad A. Cell-mediated cytotoxicity and tumor growth in zincdeficient mice. Proc Soc Exp Biol Med. 1981;167(3):333–337. doi: 10.3181/00379727-167-41174. [DOI] [PubMed] [Google Scholar]
- 38.Bonaventura P, Benedetti G, Albarède F, Miossec P. Zinc and its role in immunity and inflammation. Autoimmun Rev. 2015;14(4):277–285. doi: 10.1016/j.autrev.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 39.Berg K, Bolt G, Andersen H, Owen TC. Zinc potentiates the antiviral action of human IFNalpha tenfold. J Interf Cytokine Res. 2001;21(7):471–474. doi: 10.1089/10799900152434330. [DOI] [PubMed] [Google Scholar]
- 40.Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS (2020) Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis:ciaa248. 10.1093/cid/ciaa248 [DOI] [PMC free article] [PubMed]
- 41.Morgan CI, Ledford JR, Zhou P, Page K. Zinc supplementation alters airway inflammation and airway hyperresponsiveness to a common allergen. J Inflamm (Lond) 2011;8:36. doi: 10.1186/1476-9255-8-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zang R, Lian H, Zhong X, Yang Q, Shu HB. ZCCHC3 modulates TLR3-mediated signaling by promoting recruitment of TRIF to TLR3. J Mol Cell Biol. 2020;12(4):251–262. doi: 10.1093/jmcb/mjaa004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wong CP, Rinaldi NA, Ho E. Zinc deficiency enhanced inflammatory response by increasing immune cell activation and inducing IL6 promoter demethylation. Mol Nutr Food Res. 2015;59(5):991–999. doi: 10.1002/mnfr.201400761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Beck FW, Prasad AS, Kaplan J, Fitzgerald JT, Brewer GJ. Changes in cytokine production and T cell subpopulations in experimentally induced zinc-deficient humans. Am J Phys. 1997;272(6 Pt 1):E1002–E1007. doi: 10.1152/ajpendo.1997.272.6.E1002. [DOI] [PubMed] [Google Scholar]
- 45.Yasuda H, Tsutsui T. Infants and elderlies are susceptible to zinc deficiency. Sci Rep. 2016;6:21850. doi: 10.1038/srep21850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dinsmore W, Callender ME, McMaster D, Todd SJ, Love AH. Zinc absorption in alcoholics using zinc-65. Digestion. 1985;32(4):238–242. doi: 10.1159/000199243. [DOI] [PubMed] [Google Scholar]
- 47.Manari AP, Preedy VR, Peters TJ. Nutritional intake of hazardous drinkers and dependent alcoholics in the UK. Addict Biol. 2003;8(2):201–210. doi: 10.1080/1355621031000117437. [DOI] [PubMed] [Google Scholar]
- 48.Mills PR, Fell GS, Bessent RG, Nelson LM, Russell RI. A study of zinc metabolism in alcoholic cirrhosis. Clin Sci (Lond) 1983;64(5):527–535. doi: 10.1042/cs0640527. [DOI] [PubMed] [Google Scholar]
- 49.Skalny AV, Rink L, Ajsuvakova OP, Aschner M, Gritsenko VA, Alekseenko SI, Svistunov AA, Petrakis D, Spandidos DA, Aaseth J, Tsatsakis A, Tinkov AA. Zinc and respiratory tract infections: perspectives for COVID19 (Review) Int J Mol Med. 2020;46(1):17–26. doi: 10.3892/ijmm.2020.4575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Prasad AS. Clinical, immunological, anti-inflammatory and antioxidant roles of zinc. Exp Gerontol. 2008;43(5):370–377. doi: 10.1016/j.exger.2007.10.013. [DOI] [PubMed] [Google Scholar]
- 51.te Velthuis AJ, van den Worm SH, Sims AC, Baric RS, Snijder EJ, van Hemert MJ. Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6(11):e1001176. doi: 10.1371/journal.ppat.1001176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Alirezaei M, Nairn AC, Glowinski J, Prémont J, Marin P. Zinc inhibits protein synthesis in neurons. Potential role of phosphorylation of translation initiation factor-2alpha. J Biol Chem. 1999;274(45):32433–32438. doi: 10.1074/jbc.274.45.32433. [DOI] [PubMed] [Google Scholar]
- 53.Frederickson CJ, Koh JY, Bush AI. The neurobiology of zinc in health and disease. Nat Rev Neurosci. 2005;6(6):449–462. doi: 10.1038/nrn1671. [DOI] [PubMed] [Google Scholar]
- 54.Lazarczyk M, Favre M. Role of Zn2+ ions in host-virus interactions. J Virol. 2008;82(23):11486–11494. doi: 10.1128/JVI.01314-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rahman MT, Karim MM. Metallothionein: a potential link in the regulation of zinc in nutritional immunity. Biol Trace Elem Res. 2018;182(1):1–13. doi: 10.1007/s12011-017-1061-8. [DOI] [PubMed] [Google Scholar]
- 56.Finzi E. Treatment of SARS-CoV-2 with high dose oral zinc salts: a report on four patients. Int J Infect Dis. 2020;99:307–309. doi: 10.1016/j.ijid.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jin CN, Chen JD, Sheng JF. Vitamin D deficiency in hepatitis C virus infection: what is old? what is new? Eur J Gastroenterol Hepatol. 2018;30(7):741–746. doi: 10.1097/MEG.0000000000001134. [DOI] [PubMed] [Google Scholar]
- 58.Bucher A, White N. Vitamin C in the prevention and treatment of the common cold. Am J Lifestyle Med. 2016;10(3):181–183. doi: 10.1177/1559827616629092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hemilà H. Vitamin C and SARS coronavirus. J Antimicrob Chemother. 2003;52(6):1049–1050. doi: 10.1093/jac/dkh002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Schrezenmeier E, DÃrner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol. 2020;16(3):155–166. doi: 10.1038/s41584-020-0372-x. [DOI] [PubMed] [Google Scholar]
- 61.Derwand R, Scholz M. Does zinc supplementation enhance the clinical efficacy of chloroquine/hydroxychloroquine to win today’s battle against COVID-19? Med Hypotheses. 2020;142:109815. doi: 10.1016/j.mehy.2020.109815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pastick KA, Okafor EC, Wang F, Lofgren SM, Skipper CP, Nicol MR, Pullen MF, Rajasingham R, McDonald EG, Lee TC, Schwartz IS, Kelly LE, Lother SA, Mitjà O, Letang E, Abassi M, Boulware DR. Review: hydroxychloroquine and chloroquine for treatment of SARS-CoV-2 (COVID-19). Version 2. Open Forum Infect Dis. 2020;7(4):ofaa130. doi: 10.1093/ofid/ofaa130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Touret F, de Lamballerie X. Of chloroquine and COVID-19. Antivir Res. 2020;177:104762. doi: 10.1016/j.antiviral.2020.104762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Xue J, Moyer A, Peng B, Wu J, Hannafon BN, Ding WQ. Chloroquine is a zinc ionophore. PLoS One. 2014;9(10):e109180. doi: 10.1371/journal.pone.0109180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Micera A, Bruno L, Cacciamani A, Rongioletti M, Squitti R. Alzheimer’s disease and retinal degeneration: a glimpse at essential trace metals in ocular fluids and tissues. Curr Alzheimer Res. 2019;16(12):1073–1083. doi: 10.2174/1567205016666191023114015. [DOI] [PubMed] [Google Scholar]
- 66.Trumbo P, Yates AA, Schlicker S, Poos M. Dietary reference intakes: vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J Am Diet Assoc. 2001;101(3):294–301. doi: 10.1016/S0002-8223(01)00078-5. [DOI] [PubMed] [Google Scholar]
- 67.Tuerk MJ, Fazel N. Zinc deficiency. Curr Opin Gastroenterol. 2009;25(2):136–143. doi: 10.1097/MOG.0b013e328321b395. [DOI] [PubMed] [Google Scholar]
- 68.Maret W, Sandstead HH. Zinc requirements and the risks and benefits of zinc supplementation. J Trace Elem Med Biol. 2006;20(1):3–18. doi: 10.1016/j.jtemb.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 69.Fosmire GJ. Zinc toxicity. Am J Clin Nutr. 1990;51(2):225–227. doi: 10.1093/ajcn/51.2.225. [DOI] [PubMed] [Google Scholar]
- 70.Plum LM, Rink L, Haase H. The essential toxin: impact of zinc on human health. Int J Environ Res Public Health. 2010;7(4):1342–1365. doi: 10.3390/ijerph7041342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Magee AC, Matrone G. Studies on growth, copper metabolism of rats fed high levels of zinc. J Nutr. 1960;72(2):233–242. doi: 10.1093/jn/72.2.233. [DOI] [PubMed] [Google Scholar]
- 72.Ogiso T, Moriyama K, Sasaki S, Ishimura Y, Minato A. Inhibitory effect of high dietary zinc on copper absorption in rats. Chem Pharm Bull (Tokyo) 1974;22(1):55–60. doi: 10.1248/cpb.22.55. [DOI] [PubMed] [Google Scholar]
- 73.Van Campen DR. Copper interference with the intestinal absorption of zinc-65 by rats. J Nutr. 1969;97(1):104–108. doi: 10.1093/jn/97.1.104. [DOI] [PubMed] [Google Scholar]
- 74.Gupta P, Choksi M, Goel A, Zachariah U, Sajith KG, Ramachandran J, Chandy G, Kurian G, Rebekah G, Eapen CE. Maintenance zinc therapy after initial penicillamine chelation to treat symptomatic hepatic Wilson’s disease in resource constrained setting. Indian J Gastroenterol. 2018;37(1):31–38. doi: 10.1007/s12664-018-0829-x. [DOI] [PubMed] [Google Scholar]
- 75.Członkowska A, Litwin T, Dusek P, Ferenci P, Lutsenko S, Medici V, Rybakowski JK, Weiss KH, Schilsky ML. Wilson disease. Nat Rev Dis Primers. 2018;4(1):21. doi: 10.1038/s41572-018-0018-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chandra RK. Excessive intake of zinc impairs immune responses. JAMA. 1984;252(11):1443–1446. doi: 10.1001/jama.1984.03350110043027. [DOI] [PubMed] [Google Scholar]
- 77.Maywald M, Rink L. Zinc supplementation induces CD4+CD25+Foxp3+ antigenspecific regulatory T cells and suppresses IFN-γ production by upregulation of Foxp3 and KLF-10 and downregulation of IRF-1. Eur J Nutr. 2017;6(5):1859–1869. doi: 10.1007/s00394-016-1228-7. [DOI] [PubMed] [Google Scholar]
- 78.Cakman I, Kirchner H, Rink L. Zinc supplementation reconstitutes the production of interferon-alpha by leukocytes from elderly persons. J Interf Cytokine Res. 1997;17(8):469–472. doi: 10.1089/jir.1997.17.469. [DOI] [PubMed] [Google Scholar]
- 79.Dabbagh-Bazarbachi H, Clergeaud G, Quesada IM, Ortiz M, O’Sullivan CK, Fernández-Larrea JB. Zinc ionophore activity of quercetin and epigallocatechin-gallate: from Hepa 1-6 cells to a liposome model. J Agric Food Chem. 2014;62(32):8085–8093. doi: 10.1021/jf5014633. [DOI] [PubMed] [Google Scholar]