Abstract
Coronavirus disease 2019 (COVID-19) caused by “Severe Acute Respiratory Syndrome Coronavirus-2” (SARS-CoV-2) infection emerged in Wuhan, a city of China, and spread to the entire planet in early 2020. The virus enters the respiratory tract cells and other tissues via ACE2 receptors. Approximately 20% of infected subjects develop severe or critical disease. A cytokine storm leads to over inflammation and thrombotic events. The most common clinical presentation in COVID-19 is pneumonia, typically characterized by bilateral, peripheral, and patchy infiltrations in the lungs. However multi-systemic involvement including peripheral thromboembolic skin lesions, central nervous, gastrointestinal, circulatory, and urinary systems are reported. The disease has a higher mortality compared to other viral agents causing pneumonia and unfortunately, no approved specific therapy, nor vaccine has yet been discovered. Several clinical trials are ongoing with hydroxychloroquine, remdesivir, favipiravir, and low molecular weight heparins. This comprehensive review aimed to summarize coagulation abnormalities reported in COVID-19, discuss the thrombosis, and inflammation-driven background of the disease, emphasize the impact of thrombotic and inflammatory processes on the progression and prognosis of COVID-19, and to provide evidence-based therapeutic guidance, especially from antithrombotic and anti-inflammatory perspectives.
Keywords: COVID-19, SARS-CoV-2, inflammation, thrombosis, clinical outcomes, d-dimer, disseminated intravascular coagulation, antithrombotic therapy, heparin, low molecular weight heparins, oral anticoagulants
Introduction
The outbreak of coronavirus disease-2019 (COVID-19) caused by “Severe Acute Respiratory Syndrome coronavirus-2” (SARS-CoV-2) emerged in Wuhan, a city in China, in the last days of 2019 (1). The virus quickly spread almost everywhere on the planet and the World Health Organization declared a pandemic on the 11th of March 2020 (2). SARS-CoV-2 enters the human body through the mucosal cells of the nasopharynx. It moves to and enters the alveolar endothelial cells via angiotensin-converting-enzyme 2 (ACE-2) receptors (3, 4). Lungs, myocardium (5), endothelial tissue, testes, gastrointestinal tract, and kidneys (6) express ACE-2 receptors which make them targets of SARS-Co-V-2. It can move to the brain tissue by passing through the blood-brain barrier (7) and cause a multi-systemic disease (8). Clinically, patients can be classified under three major groups according to the severity of the disease: asymptomatic or mildly symptomatic, severe, and critically ill subjects. The last two groups are usually treated in hospitals since respiratory failure can develop and mechanical ventilation may become necessary. Overall fatality rates reported for COVID-19 differ among countries but remain high, with no specific curative treatment to date. Recent data indicate that COVID-19 patients can develop critical disease in the presence of underlying conditions including hypertension, diabetes mellitus, coronary heart disease, chronic obstructive pulmonary disease, immunological and inflammatory diseases, cancer, and the use of immunomodulatory or immune-suppressive drugs (9). An analysis of all COVID-19 cases reported in China established a fatality rate of approximately 0.9%, 10.5%, 7.3%, 6%, 6.3%, and 6.0% in patients with no comorbidities, cardiovascular disease, diabetes mellitus, hypertension, chronic respiratory disease, and cancer, respectively (10-13). Of note, the fatality rate was 14.8% for patients over 80 years old (10, 13).
Autopsy studies report respiratory failure, thrombosis, and septicemia as the main causes of death in COVID-19 patients (14). There are increasing data putting forward the roles of thrombosis and inflammation in disease prognosis. Arterial and venous thrombosis are reported among patients with COVID-19 and there is a strong association between thrombotic events and mortality. A recent study established the presence of thrombi in small and mid-sized pulmonary arteries in various amounts in all 11 patients who underwent autopsy (15). Another post mortem study aimed to compare the lungs of patients who died from COVID-19 and acute respiratory distress syndrome secondary to influenza A (H1N1) (16). The main histopathological finding was diffuse alveolar damage with perivascular T lymphocyte infiltration of the peripheral lung tissue in both groups. However COVID-19 patients’ lungs showed severe endothelial injury, widespread thrombosis with microangiopathy, and increased angiogenesis (16).
This review aims to summarize coagulation abnormalities and inflammatory mechanisms and pathways involved in the pathogenesis of COVID-19, discuss the role of thrombosis and inflammation in COVID-19 progression and clinical outcomes, and provide evidence-based therapeutic guidance, especially from antithrombotic and anti-inflammatory perspectives.
Methodology
To gather currently available evidence-based data on MEDLINE via the PubMed interface, MeSH, terms including “COVID-19,” “thrombosis,” and “inflammation” were used until 01.06.2020. All of the relevant literature associated with COVID-19, thrombosis, and inflammation were evaluated before generating this comprehensive review. The data mainly consisted of retrospective observational studies, case reports, epidemiological statistics, and expert consensus documents.
Pathogenesis of SARS-CoV-2 infection
SARS-CoV-2 attaches and enters into cells via ACE2 receptors, starts a local inflammation, and activates the endothelium (17). These are followed by tissue damage, increased irregular cytokine release, and interruption of angiotensin II metabolism, with increased angiotensin II and decreased angiotensin [1-7], leading to induced inflammation, endothelial activation, and migration of white blood cells and platelets (18). Endothelial activation in the lungs causes the ACE-1 shedding phenomenon, described as a transient release of ACE-1 from cellular membranes in large amounts followed by a quick increase in Angiotensin II levels (19). The later induces inflammation, coagulation, and the capillary leak phenomenon (19). However, when ACE-1 disappears, angiotensin II also decreases to very low levels causing more vasodilation and capillary leak, resulting in endothelial permeability impairment. Decreased angiotensin II levels induce ACE-2 synthesis and may cause susceptibility to SARS-CoV-2 entry to the tissues (20). Under these circumstances, the inappropriate release of cytokines, the cytokine storm, occurs (20).
The attachment and adhesion processes of coronaviruses to human cells are mediated by their large spike (S) proteins protruding from the viral surface, mainly by S2 protein in the case of SARS-CoV-2 (21). SARS-CoV-2 ectodomain S protein attaches to ACE2 protein with a 10 to 20 fold higher affinity than when they attach to SARS-CoV (22). The internalization process recruits the hemagglutinin cleavage pathway in which transmembrane serine protease 2 (TMPRSS2) appears to play a key role (23, 24). Therefore, TMPRSS2 antagonists including camostat mesylate and nafamostat are under investigation since they have inhibited cellular invasion of SARS-CoV-2 in vitro via reducing the protease activity of TMPRSS2 (23, 25).
Clinical spectrum of COVID-19 as a multi-systemic disease
COVID-19 clinically presents with a very wide spectrum of symptoms including almost all systems. Although it affects mainly the respiratory system, particularly the alveolar tissue, different systemic involvements are reported around the world. Furthermore, the severity of respiratory involvement differs between subjects. Some patients experience few and non-specific symptoms including fever and dry cough, others present with combined mild pneumonia and mild dyspnea, or severe pneumonia with dyspnea, tachypnoea, and hypoxia. Approximately 5% of COVID-19 patients develop critical respiratory disease requiring mechanical ventilation (26).
The cardiovascular system is also frequently targeted by SARS-Co-V2. Myocyte necrosis and mononuclear cell infiltration are reported in cardiac muscle autopsy specimens in a study performed in China (27). Additionally, COVID-19 may cause thrombosis in coronary arteries leading to myocardial infarction (28). Peripheral arterial and venous thrombosis are also frequently found in patients with COVID-19 (29, 30). The virus is shown to cross the blood-brain barrier and reach the central nervous system (31). Some researchers revealed that the virus can move to the respiratory center in the brain via the olfactory nerve (31). Gastrointestinal tract mucosa, liver, and hepatic veins are also reported as targets of COVID-19 (32). Urinary system involvements including the kidneys and testicles are reported (33). A Spanish study described five main types of skin lesions seen in COVID-19 patients and the disease can present only with skin lesions, especially among children without respiratory symptoms (34).
Of note, the respiratory distress seen in COVID-19 differs from acute respiratory distress syndrome (ARDS) caused by other infectious agents and other cytokine releasing syndromes. COVID-19 respiratory distress begins with early and profound hypoxemia, without prominent respiratory dysfunction at the beginning. Furthermore, pulmonary compliance is moderately decreased in patients with COVID-19 and the hyperperfusion of poorly or non-ventilated lung segments is reported, explaining profound hypoxemia (19, 35).
Disturbances of hemostasis and coagulation system among patients with COVID-19
Coagulation abnormalities are common among patients with COVID-19. Mild thrombocytopenia (36), elevated fibrinogen, lactate dehydrogenase (LDH), liver enzymes, D-dimer (37, 38), and fibrin degradation products (FDPs) (37) are frequently reported. A slightly prolonged prothrombin time, shortened activated partial thromboplastin time (aPTT) at the beginning and prolonged aPTT in the later phases of the infection, increased Von Willebrand (vWF) activity, vWF antigen, and FVIII levels (39, 40) are also observed. The presence of lupus anticoagulant (41, 42) was also reported in recent studies. Prolongation of aPTT, which can be detected in the case of clotting factor deficiency or the presence of coagulation inhibitors including antibodies to coagulation factors or lupus anticoagulant, is common among COVID-19 patients. Lupus anticoagulant increases the thrombotic risk and has been investigated in patients with COVID-19 in a few studies (41, 42). In one of these studies, lupus anticoagulant was present in 41% of the patients (41) while in the other study it was determined in 91% of the COVID-19 patients (42). Furthermore, low levels of factor XII were reported (42).
The coagulation function of 40 critically ill COVID-19 patients was evaluated by rotational thromboelastometry (ROTEM) and other standard methods on the first, fifth, and tenth days of admission to the intensive care unit. Prothrombin time was prolonged on the tenth day when compared to the first day, and the highest fibrinogen and aPTT levels were observed on the day of admission (when compared to the 5th and 10th days). The authors revealed that ROTEM results were consistent with a persistent hypercoagulable state (43).
The coagulopathy seen in COVID-19 differs from that seen in other infections and has been defined as “sepsis-induced coagulopathy,” characterized by increased D-dimer and fibrinogen levels (44, 45). This situation can progress to disseminated intravascular coagulation (DIC), where prolonged PT, increased D-dimer, thrombocytopenia, and hypofibrinogenemia are commonly reported (43). Of note, DIC appears to be very common, especially among critically ill COVID-19 patients, since current evidence indicate that 71% of the patients who died from COVID-19 have met the criteria set by the International Society on Thrombosis and Haemostasis (ISTH) for the diagnosis of DIC (37).
Changes in coagulation parameters are summarized in Table 1.
Table 1.
Changes in coagulation parameters in patients with COVID-19
| Platelet count | Slightly decreased (36, 57) |
| Prothrombin time | Slightly prolonged (39, 40, 43) |
| International normalized ratio | Slightly increased (39, 40, 43) |
| aPTT | Decreased first, then increased (41, 42) |
| Protein C | Increased (29) |
| Protein S | Decreased (29) |
| Antithrombin | Decreased (29) |
| Factor VIII | Increased (39, 40) |
| vWF activity | Increased (39, 40) |
| vWF antigen | Increased (39, 40) |
| Factor XII | Decreased (42) |
| Fibrinogen | Increased (37, 38, 44, 45) |
| FDPs | Increased (37, 38, 44, 45) |
| D-dimer | Increased (37, 38, 44, 45) |
| Lupus anticoagulant | Positive (up to 91%) (41, 42) |
| ROTEM | Hypercoagulable state (43) |
aPTT - activated partial thromboplastin time, FDPs - fibrin degradation products, ROTEM - rotational thromboelastometry, vWF - von Willebrand Factor
Increased Thrombosis associated clinical events in COVID-19
Thrombosis plays a pivotal role in COVID-19 associated clinical outcomes. There is increasing evidence supporting the impact of thrombosis on disease worsening, tissue damage, and deaths. A recent study aimed to assess and compare thrombotic risks between COVID-19 patients having respiratory distress syndrome and non-COVID-19 ARDS patients (46). A total of 64 thrombotic events in 150 patients (mainly pulmonary embolisms, hemodialysis fistula circuit clotting, etc.) were observed, and significantly more thrombotic complications were present among COVID-19 associated respiratory distress syndrome patients when compared to non-COVID-19 ARDS patients, despite regular anticoagulant therapy (46).
The most plausible mechanism underlying increased thrombosis in COVID-19 appears to be an inflammatory reaction triggered by the infection itself, which results in damages in the microvascular system and causes abnormal activation of the coagulation system. These are followed by increased vascular inflammation and the generation of excessive microthrombi (16).
The most common thrombotic events seen during COVID-19 are venous thromboembolism and pulmonary microvascular thrombosis. However thrombotic events can occur everywhere including heart, venous, or arterial systems. Recent studies also demonstrated that changes in coagulation parameters provide prognostic information in COVID-19. For example, a 3.5 fold increase in D-dimer, 1.9 fold increase in FDPs, 14% prolonged prothrombin time, and 71% DIC (vs. 0.6% DIC in survivors) was reported in patients who died from COVID-19 compared to those who survived (32). Furthermore, thrombotic rather than embolic pulmonary lesions were the main cause of death in COVID-19 in an autopsy study (15). In another study, venous thromboembolism (VTE) was determined in 69% of anticoagulated COVID-19 patients in the intensive care unit (47). Peripheral arterial thrombosis was also reported in critically ill COVID-19 subjects (48). Cases of myocardial infarction associated with increased thrombosis were also reported among patients with COVID-19 (49).
Antithrombotic therapy in COVID-19
Thrombosis is supposed to be one of the key mechanisms increasing the morbidity and mortality in COVID-19. Therefore, several different antithrombotic molecules are under investigation both in the prevention and treatment of thrombotic events in patients with COVID-19. However, there is no evidence-based consensus on antithrombotic regimes and there are still controversial remarks regarding the administration of antithrombotics and their doses. Some experts recommend LMWH, UFH, or fondaparinux for all hospitalized patients at prophylactic doses used for VTE and limb compression in case of prominent contraindications for anticoagulant therapy throughout their hospital stay (50). Additional therapy for 7 to 14 days after discharge can be considered for subjects with increased VTE risk (immobility, obesity, previous history of VTE, active cancer, etc.), perhaps at intermediate dose LMWH twice daily (50). However, none of the researchers and experts support the administration of UFH or LMWHs at therapeutic doses, especially without evidence of DVT or other thrombotic lesions (29, 50, 51).
On the other hand, there are several patients on warfarin or direct oral anticoagulants (DOACs) admitted to the hospitals with COVID-19. During the therapy, severe drug interactions were reported between the different antivirals, anticoagulants, and other therapeutics used in these patients (52). High instability of PT and INR are common among treated COVID-19 patients, probably due to the variability of vitamin K metabolism, liver involvement, diet, co-medications, and heart failure (52, 53). Of note, antiviral drugs interact with DOACs and may cause significantly increased blood DOACs levels, putting patients in danger of bleeding (54). Therefore, some researchers suggest to switch temporarily from oral anticoagulants to LMWHs or heparin and set individualized therapy considering the pharmacological characteristics of oral anticoagulants, the potential of polypharmacy, and the possible necessity of mechanical ventilation in intensive care units (50, 52). Patient-based evaluation of the need for anticoagulation may be the best approach until evidence-based guidelines are published.
The ISTH interim recommends prophylactic doses of LMWH in almost all COVID-19 patients without an increased risk of bleeding (55). This strategy may be appropriate for non-severe COVID-19 patients, however, severe and critically ill COVID-19 patients experience a higher degree hypercoagulable state in which therapeutic doses of heparin or LMWHs may be necessary (56). The Swiss Society of Haematology recommends thromboprophylaxis for all in-hospital COVID-19 patients according to a risk stratification score unless there is a contraindication. Monitoring of coagulation parameters is also recommended including D-dimer, prothrombin time, fibrinogen, platelet count, LDH, creatinine, and alanine aminotransferase routinely, and factor Xa and antithrombin if necessary, as well as a dose-adjusted anticoagulant therapy with regards to renal function (57).
Since post mortem studies established the presence of thrombotic microangiopathy in lung tissues, severe and critically ill COVID-19 patients who recovered from the disease may develop chronic thromboembolic pulmonary hypertension (CTEPH) in the future. Echocardiographic studies may help us to elucidate whether these individuals develop CTEPH. A follow-up program including echocardiographic assessment and lung tissue imaging can help clinicians to monitor both pulmonary artery pressure and fibrotic processes in the lungs.
To reduce thrombosis in patients with COVID-19, several molecules are proposed by investigators. Nafamostat mesylate (NM), a molecule used for pancreatitis and DIC in Japan for several years, is now proposed as a therapeutic in COVID-19 with its suppressive actions against SARS-CoV-2 (58). A combination of heparin with NM may be a more powerful option for the treatment of the thrombotic state in COVID-19 (59).
Systemic anticoagulation at therapeutic doses improved mortality among COVID-19 patients requiring mechanical ventilation (29.1% vs. 62.7%, mortality rates of anticoagulated and non-anticoagulated patients, respectively) (51). Interestingly, the authors revealed no significant difference in mortality rates among patients who received anticoagulation (22.5%) and those who did not (22.8%) when all patients were included in the analysis (51). There are still debates on how to choose the optimal anticoagulant therapy regime in patients with COVID-19. Some experts indicate that therapeutic doses of heparin may be dangerous, and possibly cause harm via contributing to hemorrhagic microangiopathy, which can result in major and fatal bleeding (60).
Another old antithrombotic molecule dipyridamole is being used among a limited number of COVID-19 patients. Dipyridamole has both antiviral and antioxidant properties shown to suppress SARS-CoV-2 replication in vitro (61). In a recent concept trial involving 31 patients with COVID-19, dipyridamole significantly decreased D-dimer (p<0.05), increased lymphocyte and platelet recovery in the circulation, and improved clinical outcomes in severe COVID-19 patients when compared to the controls (61).
The association between coagulation and inflammation following the invasion of SARS-CoV-2 may be explained in part by dysfunctional ACE2-activated renin-angiotensin system, promotion of platelet adhesion and aggregation, and release of inflammatory cytokines that activate the coagulation cascade (62-65).
Early clinical observations indicate that COVID-19 patients may provide immediate and perhaps late benefits from anticoagulant therapy. However, there is still no terminated randomized clinical study and the therapeutic regimes should be determined as concerns clinical evidence.
The role of inflammation revisited in COVID-19
SARS-CoV-2 is found in different tissues including type II alveolar epithelial cells, pulmonary macrophages, hilar lymph nodes, spleen, and testes (66). Inflammation plays an important role in the progression of the disease and its clinical outcomes. Inappropriately increased inflammation causes a cytokine storm leading to diffuse alveolar damage, renal insufficiency, myocarditis, central nervous system involvement, and multi-organ failure, associated with poor clinical prognosis. Coronavirus infection causes a release of proinflammatory cytokines, associated with a systemic inflammatory response syndrome (SIRS), and accelerates cellular death in the lungs, liver, heart, kidneys, and adrenal parenchymal tissue (67).
The cytokine storm is characterized by markedly increased levels of interleukins (ILs), mostly IL-6, IL-2, IL-7, granulocyte colony-stimulating factor, interferon-γ inducible protein 10 (IP-10), monocyte chemoattractant protein 1 (MCP-1), macrophage inflammatory protein 1 alpha (MIP1-a) and tumor necrosis factor (TNF)-alpha, which may promote lymphocyte apoptosis (68-70). These cytokines levels are shown to be increased in patients with severe COVID-19 (71, 72). The cytokine storm results in tissue damage, thrombotic microangiopathy, endothelitis, and endothelial dysfunction.
Monocytes and their derivate tissue macrophages are cells of innate immunity interacting with microbial threats by producing inflammatory cytokines. These cells eliminate pathogens and promote tissue repair. Apart from eliminating infectious agents, monocytes play roles in atherosclerosis, angiogenesis, thrombosis, and tissue repair (73). An inappropriate macrophage over-response can be damaging to the host (for example the macrophage activation syndrome induced by severe infections including COVID-19) (74).
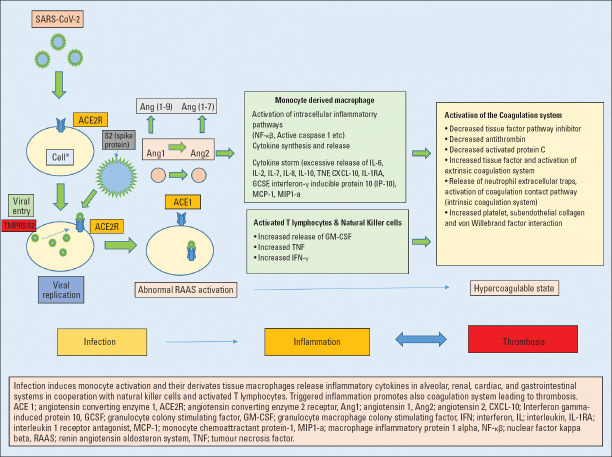
Mechanisms involved in SARS-CoV-2 infection, inflammation, and thrombosis are summarized in Figure 1.
Figure 1.
Infection, inflammation, and thrombosis in COVID-19
Antiviral, immunomodulatory, and anti-inflammatory therapy in COVID-19
There is still no specific curative antiviral therapy nor vaccine for COVID-19 but several clinical and vaccine studies are in progress. From the beginning of the pandemic, several licensed and commercially available medicines were assessed for treatment. Current antiviral treatment strategies mostly use hydroxychloroquine/chloroquine, remdesivir, favipiravir, and/or lopinavir/ritonavir.
In the treatment of COVID-19, 4-aminoquinolines including chloroquine (CQ) and its less toxic hydroxy-analog, hydroxychloroquine (HCQ), are frequently used since the beginning of the pandemic based on recent data obtained from SARS coronavirus disease in some countries. Although there is no strong evidence, HCQ is recommended by local health authorities therapy guidelines and preliminary results indicate some benefit over harm, especially when it is administered in the early phases of the disease. HCQ is supposed to diminish viral entry into mucosal tissues and viral replication at the initial phase of the disease. US health authorities appear to be against HCQ use since there are little evidence and potential harm mainly severe arrhythmias. In the USA, remdesivir was the only molecule that has obtained approval by the Food and Drug Administration for the treatment of COVID-19 at the time this article was written.
HCQ and CQ recruit several mechanisms of action from the inhibition of viral attachment and entry in respiratory tract cells to the inhibition of new viral particle maturation and spread (75-78). Aminoquinolines are frequently used in patients with rheumatic diseases including systemic lupus erythematosus and anti-phospholipid syndrome due to their anti-inflammatory and antithrombotic effects (79).
Favipiravir is an RNA dependent RNA polymerase (RdRp) inhibitor shown to be effective in the treatment of influenza and Ebola virus infections and has been assessed in COVID-19 (80, 81). A Chinese controlled non-randomized trial compared the effect of favipiravir versus lopinavir/ritonavir in COVID-19 (82). Favipiravir was associated with faster viral clearance and a higher improvement rate in chest imaging (82).
Remdesivir is a nucleotide analog prodrug that inhibits viral RNA polymerase and shows in vitro activity against SARS-CoV-2. In a small cohort of patients hospitalized with the diagnosis of severe COVID-19, compassionate-use of remdesivir improved the clinics of 68% of patients (83).
Lopinavir/ritonavir, a drug already in use among HIV/AIDS therapy, was considered in COVID-19. A randomized study performed in hospitalized adult patients with severe COVID-19 showed no benefit with lopinavir-ritonavir treatment beyond standard care (84).
On the other hand, immunomodulatory therapy is being considered in patients with cytokine storm and severe COVID-19. There are several options for immunomodulatory therapy and clinical trials are under investigation. For example, the IL-6 receptor antagonist tocilizumab is one of the most used molecules with this purpose and clinical randomized trials are still going on. Furthermore, IL-1 receptor antagonist - anakinra, anti-IL-1β - canakinumab, anti-TNF - infliximab, adalimumab, and golimumab, anti-GM-CSF - TJ003234 and lenzilumab, M-CSF receptor inhibitor - axatilimab, anti-IFNγ - emapalumab, anti-CCR5 - leronlimab, and anti-complement C5 - eculizumab are some of the promising drugs under investigation (74).
Modulation of the excessive immune response may prevent tissue damage, reduce autoimmune processes, and help clinicians in saving lives. To balance inflammatory response, researchers hope that steroids may also provide some contributions. Corticosteroid therapy can be another option that is limited to severe COVID-19 patients with refractory sepsis, septic shock, and ARDS (85-87). Since corticosteroids bind to cytoplasmic receptors, change the transcription of mRNA, and reduce inflammatory mediator production, they may be useful in limiting COVID-19 associated inflammatory damages (85-87). However, it should not be under-considered that the administration of corticosteroids may be like a double-edged sword and may delay viral clearance and increase the risk of secondary infections and mortality (85-87).
Statins’ anti-inflammatory, antithrombotic, and pleiotropic effects have been shown in coronary artery disease (88) and they may play a role in the modulation of the inflammatory response in COVID-19 (89-91). A downregulation in ACE2 has been established during COVID-19 and statins are known to increase ACE2 expression in cells. Thus statins may contribute to reverse downregulated ACE2 expression and may limit inflammation, thrombosis, and endothelial dysfunction triggered by a dysfunctional ACE2 activated renin-angiotensin system (62-65, 89-91). Of note, renin-angiotensin-aldosterone system inhibitors have no negative effect on disease progression and prognosis in COVID-19, and patients who are using these drugs may adhere to their therapy safely (92, 93).
The interactions between antiviral, anti-inflammatory, and antithrombotic agents frequently used for the treatment of COVID-19 are summarized in Table 2.
Table 2.
The interaction between antiviral, anti-inflammatory, and antithrombotic agents used for the treatment of COVID-19*
| Heparin/LMWHs/Fondaparinux | DOACs | Warfarin | Antiaggregant agents (ASA, P2Y12 antagonists, and dipyridamole) | |
|---|---|---|---|---|
| CQ/HCQ | No clinically significant interaction expected | Coadministration is expected to increase dabigatran and edoxaban concentrations | No clinically significant interaction expected | No clinically significant interaction expected |
| Concentrations of apixaban and rivaroxaban may increase due to P-gp inhibition | ||||
| Remdesivir | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected |
| Favipiravir | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected |
| Lopinavir/Ritonavir | No clinically significant interaction expected | Increased concentrations of DOACs, potential increase in bleeding especially with apixaban and rivaroxaban | Coadministration is expected to decrease warfarin concentrations. | Potentially decrease in dipyridamole’s antiplatelet effect |
| Dabigatran dose might need to be reduced when used together | INR monitoring should be increased | Substantial increase in exposure to ticagrelor (contraindicated) | ||
| Dose reduction for edoxaban should be considered | Diminished clopidogrel’s antiplatelet effect (contraindicated) | |||
| No clinically significant interaction expected with ASA and prasugrel | ||||
| Tocilizumab | No clinically significant interaction expected | No clinically significant interaction expected with dabigatran and edoxaban | Doses may need to be increased to maintain therapeutic effect with warfarin. | No clinically significant interaction expected with ASA and dipyridamole |
| Doses may need to be increased to maintain therapeutic effects with apixaban and rivaroxaban | INR monitoring is recommended | Doses may need to be increased to maintain therapeutic effect with clopidogrel, prasugrel, and ticagrelor | ||
| Sarilumab | No clinically significant interaction expected | No clinically significant interaction expected with dabigatran and edoxaban | Doses may need to be increased to maintain therapeutic effect with warfarin. | No clinically significant interaction expected with ASA and dipyridamole |
| Doses may need to be increased to maintain therapeutic effect with apixaban and rivaroxaban | INR monitorization is recommended | Doses may need to be increased to maintain therapeutic effect with clopidogrel, prasugrel, and ticagrelor | ||
| Anakinra | No clinically significant interaction expected | No clinically significant interaction expected with dabigatran and edoxaban | Doses may need to be increased to maintain therapeutic effect with warfarin. | No clinically significant interaction expected with ASA and dipyridamole |
| Doses may need to be increased to maintain therapeutic effect with apixaban and rivaroxaban | INR monitorization is recommended | Doses may need to be increased to maintain therapeutic effect with clopidogrel, prasugrel, and ticagrelor | ||
| Interferon β | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected | No clinically significant interaction expected |
| Atazanavir | No clinically significant interaction expected | Increased concentrations of apixaban, increased bleeding risk and the combination should be avoided | Monitor INR during coadministration and for the first weeks after stopping atazanavir | Diminished clopidogrel response, prasugrel should be preferred in presence of atazanavir |
| Increased dabigatran exposure and more pronounced anticoagulant response | Coadministration may lead to a substantial increase in exposure to ticagrelor and bleeding risk | |||
| Increase in rivaroxaban’s plasma concentrations and increased bleeding risk | Atazanavir could potentially increase dipyridamole exposure | |||
| Coadministration is expected to increase edoxaban’s concentration (half dose edoxaban is recommended) | No clinically significant interaction expected with ASA and prasugrel | |||
| Ribavirin | No clinically significant interaction expected | No clinically significant interaction expected | The dose of warfarin should be increased by approximately 40% to maintain the desired level of anticoagulation | No clinically significant interaction expected |
| Nitazoxanide | No clinically significant interaction expected | No clinically significant interaction expected | Nitazoxanide may increase the effect of warfarin due to protein binding displacement. | No clinically significant interaction expected |
| Monitorization of INR is recommended |
https://www.covid19-druginteractions.org/checker University of Liverpool, UK.
ASA - acetylsalicylic acid, CQ - chloroquine, DOACs - direct oral anticoagulants, HCQ - hydroxychloroquine, INR - international normalized ratio, LMWHs - low molecular weight heparins (enoxaparin)
Conclusion
In severe or critical COVID-19, inflammatory mechanisms are activated. The release of various cytokines in excessive amounts causes a SIRS called the “cytokine storm,” responsible for increased tissue damages. The inflammation of endothelial tissue, occurrence of endothelial dysfunction, activation of the coagulation system, and generation of a prothrombotic milieu are key mechanisms underlying COVID-19 associated clinical events. Microangiopathy, thrombosis, and angiogenesis highly influence disease progression. Anticoagulant therapy can limit tissue damage associated with microvasculature thrombosis. Debates on timing and dosing of antithrombotic therapy as well as the drug of choice are still up to date and low molecular weight heparins appear to be one step ahead compared to others because of the possible interaction between antiviral agents and DOACs. The unstable status of PT-INR in warfarin therapy also limits its use in COVID-19. There is almost a consensus on the prophylactic use of LMWHs in all hospitalized patients unless there are contraindications. NM alone or in combination with heparin, and dipyridamole can be considered in appropriate patients. Individualized antithrombotic regimes are recommended regarding patients’ clinical status. Favipiravir, remdesivir, and hydroxychloroquine are promising antimicrobials but should be investigated in randomized controlled trials. Immunomodulatory therapies for cytokine storm and SIRS have a high ability to limit the immune system driven host damages.
Footnotes
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
References
- 1.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization. Coronavirus Disease 2019 (COVID-19) Situation report –46. [Accessed on March 12 2020]. Available Online: URL;https://www.who.int/docs/default-source/coronaviruse/situationreports/20200306-sitrep-46-covid-19.pdf?sfvrsn=96b04adf_2 .
- 3.Iba T, Levy JH. Derangement of the endothelial glycocalyx in sepsis. J Thromb Haemost. 2019;17:283–94. doi: 10.1111/jth.14371. [DOI] [PubMed] [Google Scholar]
- 4.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–8. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rizzo P, Vieceli Dalla Sega F, Fortini F, Marracino L, Rapezzi C, Ferrari R. COVID-19 in the heart and the lungs:could we “Notch”the inflammatory storm? Basic Res Cardiol. 2020;115:31. doi: 10.1007/s00395-020-0791-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Su H, Yang M, Wan C, Yi LX, Tang F, Zhu HY, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98:219–27. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ye M, Ren Y, Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav Immun. 2020;88:945–6. doi: 10.1016/j.bbi.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Du Y, Tu L, Zhu P, Mu M, Wang R, Yang P, et al. Clinical Features of 85 Fatal Cases of COVID-19 from Wuhan. A Retrospective Observational Study. Am J Respir Crit Care Med. 2020;201:1372–9. doi: 10.1164/rccm.202003-0543OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Emami A, Javanmardi F, Pirbonyeh N, Akbari A. Prevalence of Underlying Diseases in Hospitalized Patients with COVID-19:a Systematic Review and Meta-Analysis Arch Acad Emerg Med. 2020;8:e35. [PMC free article] [PubMed] [Google Scholar]
- 10.Fu L, Wang B, Yuan T, Chen X, Ao Y, Fitzpatrick T, et al. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China:A systematic review and meta-analysis. J Infect. 2020;80:656–65. doi: 10.1016/j.jinf.2020.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Epidemiology Working Group for NCIP Epidemic Response, Chinese Center for Disease Control and Prevention [The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China] Zhonghua Liu Xing Bing Xue Za Zhi. 2020;41:145–51. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 12.Ruan S. Likelihood of survival of coronavirus disease 2019. Lancet Infect Dis. 2020;20:630–1. doi: 10.1016/S1473-3099(20)30257-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China:a retrospective cohort study. Lancet. 2020;395:1054–62. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S. COVID-19 Autopsies, Oklahoma, USA. Am J Clin Pathol. 2020;153:725–33. doi: 10.1093/ajcp/aqaa062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lax SF, Skok K, Zechner P, Kessler HH, Kaufmann N, Koelblinger C, et al. Pulmonary Arterial Thrombosis in COVID-19 With Fatal Outcome:Results From a Prospective, Single-Center, Clinicopathologic Case Series. Ann Intern Med. 2020;173:350–61. doi: 10.7326/M20-2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N Engl J Med. 2020;383:120–8. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu Z, Xiao X, Wei X, Li J, Yang J, Tan H, et al. Composition and divergence of coronavirus spike proteins and host ACE2 receptors predict potential intermediate hosts of SARS-CoV-2. J Med Virol. 2020;92:595–601. doi: 10.1002/jmv.25726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fehr AR, Perlman S. Coronaviruses:an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Leisman DE, Deutschman CS, Legrand M. Facing COVID-19 in the ICU:vascular dysfunction, thrombosis, and dysregulated inflammation. Intensive Care Med. 2020;46:1105–8. doi: 10.1007/s00134-020-06059-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guzik TJ, Mohiddin SA, Dimarco A, Patel V, Savvatis K, Marelli-Berg FM, et al. COVID-19 and the cardiovascular system:implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res. 2020;116:1666–87. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020;181:281–92. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu X, Chen P, Wang J, Feng J, Zhou H, Li X, et al. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci. 2020;63:457–60. doi: 10.1007/s11427-020-1637-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–80. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stopsack KH, Mucci LA, Antonarakis ES, Nelson PS, Kantoff PW. TMPRSS2 and COVID-19:Serendipity or Opportunity for Intervention? Cancer Discov. 2020;10:779–82. doi: 10.1158/2159-8290.CD-20-0451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Perrotta F, Matera MG, Cazzola M, Bianco A. Severe respiratory SARS-CoV2 infection:Does ACE2 receptor matter? Respir Med. 2020;168:105996. doi: 10.1016/j.rmed.2020.105996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China:Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239–42. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 27.National Health Commission of the People's Republic of China. Chinese Clinical Guidance for COVID-19 Pneumonia Diagnosis and Treatment (7th edition) 2020. Available Online:URL; http://kjfy.meetingchina.org/msite/news/show/cn/3337.html .
- 28.Stefanini GG, Montorfano M, Trabattoni D, Andreini D, Ferrante G, Ancona M, et al. ST-Elevation Myocardial Infarction in Patients With COVID-19:Clinical and Angiographic Outcomes. Circulation. 2020;141:2113–6. doi: 10.1161/CIRCULATIONAHA.120.047525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, et al. Global COVID-19 Thrombosis Collaborative Group, Endorsed by the ISTH, NATF, ESVM, and the IUA, Supported by the ESC Working Group on Pulmonary Circulation and Right Ventricular Function. COVID-19 and Thrombotic or Thromboembolic Disease:Implications for Prevention, Antithrombotic Therapy, and Follow-Up:JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:2950–73. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lodigiani C, Iapichino G, Carenzo L, Cecconi M, Ferrazzi P, Sebastian T, et al. Humanitas COVID-19 Task Force. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Asadi-Pooya AA, Simani L. Central nervous system manifestations of COVID-19:A systematic review. J Neurol Sci. 2020;413:116832. doi: 10.1016/j.jns.2020.116832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wong SH, Lui RN, Sung JJ. Covid-19 and the digestive system. J Gastroenterol Hepatol. 2020;35:744–8. doi: 10.1111/jgh.15047. [DOI] [PubMed] [Google Scholar]
- 33.Peng L, Liu J, Xu W, Luo Q, Chen D, Lei Z, et al. SARS-CoV-2 can be detected in urine, blood, anal swabs, and oropharyngeal swabs specimens. J Med Virol. 2020 doi: 10.1002/jmv.25936. 10.1002/jmv.25936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Galván Casas C, Català A, Carretero Hernández G, Rodríguez-Jiménez P, Fernández-Nieto D, Rodríguez-Villa Lario A, et al. Classification of the cutaneous manifestations of COVID-19:a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71–7. doi: 10.1111/bjd.19163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gattinoni L, Coppola S, Cressoni M, Busana M, Rossi S, Chiumello D. COVID-19 Does Not Lead to a “Typical”Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2020;201:1299–300. doi: 10.1164/rccm.202003-0817LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lippi G, Plebani M, Henry BM. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections:A meta-analysis. Clin Chim Acta. 2020;506:145–8. doi: 10.1016/j.cca.2020.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–7. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lippi G, Favaloro EJ. D-dimer is associated with severity of coronavirus disease 2019 (COVID-19):a pooled analysis. Thromb Haemost. 2020;120:876–8. doi: 10.1055/s-0040-1709650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harenberg J, Favaloro E. COVID-19:progression of disease and intravascular coagulation –present status and future perspectives. Clin Chem Lab Med. 2020;58:1029–36. doi: 10.1515/cclm-2020-0502. [DOI] [PubMed] [Google Scholar]
- 40.Lippi G, Plebani M. Laboratory abnormalities in patients with COVID-2019 infection. Clin Chem Lab Med. 2020;58:1131–4. doi: 10.1515/cclm-2020-0198. [DOI] [PubMed] [Google Scholar]
- 41.Harzallah I, Debliquis A, Drénou B. Lupus anticoagulant is frequent in patients with Covid-19. J Thromb Haemost. 2020;18:2064–5. doi: 10.1111/jth.14867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bowles L, Platton S, Yartey N, Dave M, Lee K, Hart DP, et al. Lupus Anticoagulant and Abnormal Coagulation Tests in Patients with Covid-19. N Engl J Med. 2020;383:288–90. doi: 10.1056/NEJMc2013656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pavoni V, Gianesello L, Pazzi M, Stera C, Meconi T, Frigieri FC. Evaluation of coagulation function by rotation thromboelastometry in critically ill patients with severe COVID-19 pneumonia. J Thromb Thrombolysis. 2020;50:281–6. doi: 10.1007/s11239-020-02130-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18:1094–9. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Iba T, Levy JH, Warkentin TE, Thachil J, van der Poll T, Levi M. Scientific and Standardization Committee on DIC and the Scientific and Standardization Committee on Perioperative and Critical Care of the International Society on Thrombosis and Haemostasis. Diagnosis and management of sepsis-induced coagulopathy and disseminated intravascular coagulation. J Thromb Haemost. 2019;17:1989–94. doi: 10.1111/jth.14578. [DOI] [PubMed] [Google Scholar]
- 46.Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X, et al. CRICS TRIGGERSEP Group (Clinical Research in Intensive Care and Sepsis Trial Group for Global Evaluation and Research in Sepsis). High risk of thrombosis in patients with severe SARS-CoV-2 infection:a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089–98. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Llitjos JF, Leclerc M, Chochois C, Monsallier JM, Ramakers M, Auvray M, et al. High incidence of venous thromboembolic events in anticoagulated severe COVID-19 patients. J Thromb Haemost. 2020;18:1743–6. doi: 10.1111/jth.14869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mestres G, Puigmacià R, Blanco C, Yugueros X, Esturrica M, Riambau V. Risk of peripheral arterial thrombosis in COVID-19. J Vasc Surg. 2020;72:756–7. doi: 10.1016/j.jvs.2020.04.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Januzzi JL., Jr Troponin and BNP use in COVID-19. Cardiology Magazine:American College of Cardiology. 2020 [Google Scholar]
- 50.Marietta M, Ageno W, Artoni A, De Candia E, Gresele P, Marchetti M, et al. COVID-19 and haemostasis:a position paper from Italian Society on Thrombosis and Haemostasis (SISET) Blood Transfus. 2020;18:167–9. doi: 10.2450/2020.0083-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Paranjpe I, Fuster V, Lala A, Russak AJ, Glicksberg BS, Levin MA, et al. Association of Treatment Dose Anticoagulation With In-Hospital Survival Among Hospitalized Patients With COVID-19. J Am Coll Cardiol. 2020;76:122–4. doi: 10.1016/j.jacc.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Testa S, Paoletti O, Giorgi-Pierfranceschi M, Pan A. Switch from oral anticoagulants to parenteral heparin in SARS-CoV-2 hospitalized patients. Intern Emerg Med. 2020;15:751–3. doi: 10.1007/s11739-020-02331-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Foerster KI, Hermann S, Mikus G, Haefeli WE. Drug-Drug Interactions with Direct Oral Anticoagulants. Clin Pharmacokinet. 2020;59:967–80. doi: 10.1007/s40262-020-00879-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Testa S, Prandoni P, Paoletti O, Morandini R, Tala M, Dellanoce C, et al. Direct oral anticoagulant plasma levels'striking increase in severe COVID-19 respiratory syndrome patients treated with antiviral agents:The Cremona experience. J Thromb Haemost. 2020;18:1320–3. doi: 10.1111/jth.14871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Thachil J, Tang N, Gando S, Falanga A, Cattaneo M, Levi M, et al. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020;18:1023–26. doi: 10.1111/jth.14810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Barrett CD, Moore HB, Yaffe MB, Moore EE. ISTH interim guidance on recognition and management of coagulopathy in COVID-19:A comment. J Thromb Haemost. 2020;18:2060–3. doi: 10.1111/jth.14860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Casini A, Alberio L, Angelillo-Scherrer A, Fontana P, Gerber B, Graf L, et al. Thromboprophylaxis and laboratory monitoring for in-hospital patients with COVID-19 - a Swiss consensus statement by the Working Party Hemostasis. Swiss Med Wkly. 2020;150:w20247. doi: 10.4414/smw.2020.20247. [DOI] [PubMed] [Google Scholar]
- 58.Inoue J, Yamamoto M. Identification of an existing Japanese pancreatitis drug, Nafamostat, which is expected to prevent the transmission of new coronavirus infection (COVID-19) Available Online: URL;https://www.u-tokyo.ac.jp/focus /en/artic les/z0508_00083.html .
- 59.Asakura H, Ogawa H. Potential of heparin and nafamostat combination therapy for COVID-19. J Thromb Haemost. 2020;18:1521–2. doi: 10.1111/jth.14858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cattaneo M, Bertinato EM, Birocchi S, Brizio C, Malavolta D, Manzoni M, et al. Pulmonary Embolism or Pulmonary Thrombosis in COVID-19?Is the Recommendation to Use High-Dose Heparin for Thromboprophylaxis Justified? Thromb Haemost. 2020;120:1230–2. doi: 10.1055/s-0040-1712097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liu X, Li Z, Liu S, Sun J, Chen Z, Jiang M, et al. Potential therapeutic effects of dipyridamole in the severely ill patients with COVID-19. Acta Pharm Sin B. 2020;10:1205–15. doi: 10.1016/j.apsb.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Boccia M, Aronne L, Celia B, Mazzeo G, Ceparano M, D'Agnano V, et al. COVID-19 and coagulative axis:review of emerging aspects in a novel disease. Monaldi Arch Chest Dis. 2020;90 doi: 10.4081/monaldi.2020.1300. doi:10.4081/monaldi.2020.1300. [DOI] [PubMed] [Google Scholar]
- 63.Wang J, Saguner AM, An J, Ning Y, Yan Y, Li G. Dysfunctional Coagulation in COVID-19:From Cell to Bedside. Adv Ther. 2020;37:3033–9. doi: 10.1007/s12325-020-01399-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Langer F, Kluge S, Klamroth R, Oldenburg J. Coagulopathy in COVID-19 and Its Implication for Safe and Efficacious Thromboprophylaxis. Hamostaseologie. 2020;40:264–9. doi: 10.1055/a-1178-3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135:2033–40. doi: 10.1182/blood.2020006000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tian S, Hu W, Niu L, Liu H, Xu H, Xiao SY. Pulmonary Pathology of Early-Phase 2019 Novel Coronavirus (COVID-19) Pneumonia in Two Patients With Lung Cancer. J Thorac Oncol. 2020;15:700–4. doi: 10.1016/j.jtho.2020.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chousterman BG, Swirski FK, Weber GF. Cytokine storm and sepsis disease pathogenesis. Semin Immunopathol. 2017;39:517–28. doi: 10.1007/s00281-017-0639-8. [DOI] [PubMed] [Google Scholar]
- 68.Singh S, Sharma A, Arora SK. High producer haplotype (CAG) of -863C/A, -308G/A and -238G/A polymorphisms in the promoter region of TNF-alpha gene associate with enhanced apoptosis of lymphocytes in HIV-1 subtype C infected individuals from North India. PLoS One. 2014;9:e98020. doi: 10.1371/journal.pone.0098020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liao YC, Liang WG, Chen FW, Hsu JH, Yang JJ, Chang MS. IL-19 induces production of IL-6 and TNF-alpha and results in cell apoptosis through TNF-alpha. J Immunol. 2002;169:4288–97. doi: 10.4049/jimmunol.169.8.4288. [DOI] [PubMed] [Google Scholar]
- 70.Aggarwal S, Gollapudi S, Gupta S. Increased TNF-alpha-induced apoptosis in lymphocytes from aged humans:changes in TNF-alpha receptor expression and activation of caspases. J Immunol. 1999;162:2154–61. [PubMed] [Google Scholar]
- 71.Sarzi-Puttini P, Giorgi V, Sirotti S, Marotto D, Ardizzone S, Rizzardini G, et al. COVID-19, cytokines and immunosuppression:what can we learn from severe acute respiratory syndrome? Clin Exp Rheumatol. 2020;38:337–42. [PubMed] [Google Scholar]
- 72.Jackson SP, Darbousset R, Schoenwaelder SM. Thromboinflammation:challenges of therapeutically targeting coagulation and other host defense mechanisms. Blood. 2019;133:906–18. doi: 10.1182/blood-2018-11-882993. [DOI] [PubMed] [Google Scholar]
- 73.Pamukcu B, Lip GY, Devitt A, Griffiths H, Shantsila E. The role of monocytes in atherosclerotic coronary artery disease. Ann Med. 2010;42:394–403. doi: 10.3109/07853890.2010.497767. [DOI] [PubMed] [Google Scholar]
- 74.Merad M, Martin JC. Pathological inflammation in patients with COVID-19:a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20:355–62. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kumar S, Maurya VK, Prasad AK, Bhatt MLB, Saxena SK. Structural, glycosylation and antigenic variation between 2019 novel coronavirus (2019-nCoV) and SARS coronavirus (SARS-CoV) Virusdisease. 2020;31:13–21. doi: 10.1007/s13337-020-00571-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fantini J, Di Scala C, Chahinian H, Yahi N. Structural and molecular modelling studies reveal a new mechanism of action of chloroquine and hydroxychloroquine against SARS-CoV-2 infection. Int J Antimicrob Agents. 2020;55:105960. doi: 10.1016/j.ijantimicag.2020.105960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271–80. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Quiros Roldan E, Biasiotto G, Magro P, Zanella I. The possible mechanisms of action of 4-aminoquinolines (chloroquine/hydroxychloroquine) against Sars-Cov-2 infection (COVID-19):A role for iron homeostasis? Pharmacol Res. 2020;158:104904. doi: 10.1016/j.phrs.2020.104904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Oestereich L, Lüdtke A, Wurr S, Rieger T, Muñoz-Fontela C, Günther S. Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model. Antiviral Res. 2014;105:17–21. doi: 10.1016/j.antiviral.2014.02.014. [DOI] [PubMed] [Google Scholar]
- 81.MDVI LLC. Phase 3 efficacy and safety study of favipiravir for treatment of uncomplicated influenza in adults [Internet] Bethesda (MD): National Library of Medicine; [update 2015 Nov 11;cited 2020 Mar 7] Available from: URL;https://clinicaltrials.gov/ct2/show/NCT02008344 . [Google Scholar]
- 82.Cai Q, Yang M, Liu D, Chen J, Shu D, Xia J, et al. Experimental Treatment with Favipiravir for COVID-19:An Open-Label Control Study. Engineering (Beijing) 2020 doi: 10.1016/j.eng.2020.03.007. doi:10.1016/j.eng.2020.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med. 2020;382:2327–36. doi: 10.1056/NEJMoa2007016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19. N Engl J Med. 2020;382:1787–99. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Alhazzani W, Møller MH, Arabi YM, Loeb M, Gong MN, Fan E, et al. Surviving Sepsis Campaign:Guidelines on the Management of Critically Ill Adults with Coronavirus Disease 2019 (COVID-19) Crit Care Med. 2020;48:e440–69. doi: 10.1097/CCM.0000000000004363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Giwa AL, Desai A, Duca A. Novel 2019 coronavirus SARS-CoV-2 (COVID-19):An updated overview for emergency clinicians. Emerg Med Pract. 2020;22:1–28. [PubMed] [Google Scholar]
- 87.Mehta N, Mazer-Amirshahi M, Alkindi N, Pourmand A. Pharmacotherapy in COVID-19;A narrative review for emergency providers. Am J Emerg Med. 2020;38:1488–93. doi: 10.1016/j.ajem.2020.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Undas A, Brozek J, Musial J. Anti-inflammatory and antithrombotic effects of statins in the management of coronary artery disease. Clin Lab. 2002;48:287–96. [PubMed] [Google Scholar]
- 89.Lee KCH, Sewa DW, Phua GC. Potential role of statins in COVID-19. Int J Infect Dis. 2020;96:615–7. doi: 10.1016/j.ijid.2020.05.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Rodrigues-Diez RR, Tejera-Muñoz A, Marquez-Exposito L, Rayego-Mateos S, Santos Sanchez L, Marchant V, et al. Statins:Could an old friend help in the fight against COVID-19? Br J Pharmacol. 2020 doi: 10.1111/bph.15166. 10.1111/bph.15166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Eroğlu I, Çelik Eroğlu B, Uyaroğlu OA, Sain Güven G. Blocking angiotensin earlier with RAS blockers, statins, and heparin in high-risk COVID-19 patients:Is the remedy here? Anatol J Cardiol. 2020;24:19–20. doi: 10.14744/AnatolJCardiol.2020.73232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.de Abajo FJ, Rodríguez-Martín S, Lerma V, Mejía-Abril G, Aguilar M, García-Luque A, et al. MED-ACE2-COVID19 study group. Use of renin-angiotensin-aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital:a case-population study. Lancet. 2020;395:1705–14. doi: 10.1016/S0140-6736(20)31030-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Gormez S, Ekicibasi E, Degirmencioglu A, Paudel A, Erdim R, Gumusel HK, et al. Association between renin-angiotensin-aldosterone system inhibitor treatment, neutrophil-lymphocyte ratio, D-Dimer and clinical severity of COVID-19 in hospitalized patients:a multicenter, observational study. J Hum Hypertens. 2020:1–10. doi: 10.1038/s41371-020-00405-3. [DOI] [PMC free article] [PubMed] [Google Scholar]