Abstract
Aflatoxin B1 contamination adversely affects human health by impairing long-term physical and cognitive development. Several crops have been associated with aflatoxin B1 contamination and corn is one of them. In the Anuradhapura district of the North Central Province of Sri Lanka, corn is one of the main agricultural produce. Due to poor farming practices in this area, it is possible that aflatoxin B1 is somehow transported from soil to the corn ears. This study was carried out to detect and quantify aflatoxin B1 in corn and corn-grown soils in Anuradhapura. Corn (n = 60) and corn-grown soil (n = 60) samples were randomly collected from 20 minor-scale corn-grown fields with three random replicates. Each sample was prepared for the measurement of aflatoxin B1 levels using the Enzyme-Linked Immunosorbent Assay (ELISA). Though 20 ppb is considered as the poisonous or deleterious level for corn consumption, there were toxin contaminations of up to 60–70 ppb in the corn kernel, while majority of soil had 350–400 ppb of aflatoxin B1 levels. Fifteen corn samples had exceeded the acceptable level while 22 samples were free of aflatoxin B1 and 23 samples were within the acceptable level. The results showed that the presence of aflatoxin B1 in corn is not habitually distributed throughout Anuradhapura district and it increased with the soil aflatoxin B1 concentration. It appears that there is a relationship between corn kernel and corn-grown soil aflatoxin B1 levels.
Keywords: Agricultural sciences, Food safety, Food analysis, Absorbance, Aspergillus flavus, Cereals, Corn, Soil
Agricultural sciences; Food safety; Food analysis; Absorbance; Aspergillus flavus; Cereals; Corn; Soil.
1. Introduction
Aflatoxins are a group of highly toxic secondary metabolites, produced by fungi of the genus Aspergillus spp., where Aspergillus flavus and Aspergillus parasiticus are principally involved in this process (Gams et al. 1985). A. flavus is the most abundant aflatoxin-producing species associated with corn while A. parasiticus is more prevalent in peanuts (Abbas et al. 2004). A. flavus typically produces B1 and B2 toxins, while A. parasiticus produces B1, B2, G1 and G2 aflatoxins. The toxicity levels of aflatoxin B1 decrease in the form of B1>G1>B2>G2 (Lakkireddy et al. 2014). Aflatoxin production depends on many factors such as the substrate, temperature, pH, relative humidity and the presence of other fungi (Gourama and Bullerman 1995). Drought, insect damage and heat during fungal growth increase aflatoxin levels (Garcia and Cotty 2004). Populations of A. flavus in soils under corn cultivation can range from 200 to >300,000 colony-forming units (CFU) g−1 and it can constitute from ≤0.2% to ≤8% of the culturable soil fungal population (Abbas et al. 2008). The fungus can survive between 12 °C to 48 °C, however, the optimum growth is generally at 28 °C–37 °C with a high humidity of above 80%.
Aflatoxin contamination in food is a worldwide problem, where globally, it is the dominant mycotoxin studied to date (Rushing and Selim 2019). The toxin has negative impacts on human health by impairing long-term physical and cognitive development as well as indirectly resulting in malnutrition (Zain 2011). High aflatoxin exposure (both in utero and in early life) leads to stunting and underweight (Razzazi-Fazeli et al. 2004). Given these adverse effects, regulatory control limitations for aflatoxin B1 in food and feed are well established. The US Food and Drug Administration (FDA) has set limits of 20 ppb total aflatoxins for interstate commerce of food and feed and 0.5 ppb of aflatoxins for sale of milk (Lakkireddy et al. 2014). Generally, nuts (mainly peanuts), cereals (mainly maize) and animal feed are considered as most susceptible to aflatoxin contamination (Williams et al. 2002).
In Sri Lanka, aflatoxin B1 is the predominant mycotoxin contaminating most agricultural produce (Yogendrajah et al. 2014). According to a survey conducted in Sri Lanka, edible vegetable oil types obtained from the Sri Lankan market, peanuts, peanut-based products and parboiled rice were reported with significantly high aflatoxin B1 and aflatoxin G1 contents (Bandara et al. 1991; Manage and Dissanayake 2009; Karunarathna et al. 2019). The reported aflatoxin B1 values were higher than raw milled rice, where enhanced fungal growth is expected anyway due to high humidity and temperature in their storage conditions (Bandara et al. 1991; Manage and Dissanayake 2009; Karunarathna et al. 2019). There are several cereals which are grown in Sri Lanka for which aflatoxin B1 contamination levels have not been systematically assessed. Corn is one such agricultural produce for which scientific studies carried out in Sri Lanka are presently non-existent on aflatoxin B1 contamination. Corn is rich in carbohydrates, which in turn provides a good growth medium for Aspergillus flavus conidial and sclerotial development. Corncobs take several years to decompose while other crop residues readily decay in agricultural soil. Thus, it is only logical to expect corn to harbour greater A. flavus levels. Anuradhapura, which is a district located in the North Central Province of Sri Lanka, is an agricultural district and corn is one of the major field crops grown in the area (Handawela 1992). Corn farms have the second highest extent of land next to rice where, around 30,000ha of land is devoted to this crop, annually. Only a few publications to date have quantified the presence of soil and kernel aflatoxin in corn-grown soils to assess soil aflatoxin toxicity, together with kernel aflatoxin concentration, especially in Sri Lanka. Therefore, this study aimed at evaluating the aflatoxin B1 contamination of corn kernel and corn-grown soil in the district of Anuradhapura.
2. Materials and methods
2.1. Sample collection
Corn and soil samples were randomly collected from different areas of twenty minor scale corn-grown fields in the Anuradhapura district. All corn samples were healthy-looking and void of visible mold. These fields had been continuously utilized to grow only corn rather than any other crop, hence their being selected for the study. The names and their locations on the global positioning system (GPS) are shown in Figure 1. Soil and corn samples were collected at the harvesting stage with three random replicates per each field, resulting 60 corn and 60 soil samples altogether. Soil samples were collected from surface level of soil (0.0–4.0 cm in depth). Each corn (seed) and soil samples collected were sealed and stored at 4 °C until further analysis.
Figure 1.
Location of the corn fields in the district of Anuradhapura, Sri Lanka.
2.2. Sample preparation
As per the method by Nesci and Etcheverry (2002), soil samples were initially filtered through a 2 mm sieve and dried under ambient conditions until a constant weight was reached. The corn seeds were removed and homogenized into a paste using a kitchen blender. Both seed paste and dried soil samples were separately weighed (20.0 ± 0.05 g) into conical flasks and 100 mL of 70 % methanol was added. The mixtures were shaken for 5 min and kept still for 10 min. The supernatants were filtered with Whatman #1 filter papers and diluted with deionized water in a volume ratio of 1:3. Finally, the pH was adjusted to a range of 5–9 by using HCl and NaOH. These filtered samples were used for the ELISA (Enzyme-Linked Immunosorbent Assay) analysis for aflatoxin B1. Samples were prepared for analysis as per the instructions provided in the ELISA assay kit (Creative Diagnostics, Shirley, USA, https://www.creative-diagnostics.com/Aflatoxin-B1-ELISA-Kit-3592-464.htm). In brief, the mean values of the absorbance values obtained from the standards and the samples were divided by the absorbance value of the first standard (zero standard) and multiplied by 100% where,
| Absorbance (%) = B/B0 X 100% |
B – Absorbance of standards or samples
B0 – Absorbance of zero standard
For the standard curve, the absorbance value of standards was placed as the y-axis in a semi-logarithmic plot of the concentration of the standards (ppb) as x-axis. The aflatoxin concentration of each sample (ppb), was read from the calibration curve and multiplied by the corresponding dilution factor of each sample followed, to obtain the actual concentration of sample.
2.3. Detection and quantification of Aflatoxin
All samples and reagents in the assay kit were brought to room temperature (25 °C) prior to analysis. The absorbance was measured with a dual wavelength of 450/630 nm using a Thermo-Scientific Multiskan™ GO microplate reader (USA). The experiment was conducted in a completely randomized design (CRD) with three replicates. Exploratory analyses were conducted using histograms, box plots and summary statistics. Shapiro – Wilk normality test, non-parametric one-way ANOVA (Kruskal Wallis) and two sample comparison (Wilcoxon rank sum test) were conducted. The statistical analyses were conducted for three main purposes: to test the normality of the data, the significance among the aflatoxin B1 concentration and its distribution, and to determine the significance of the relationship between corn and corn grown aflatoxin B1, respectively.
3. Results and discussion
The aflatoxin concentrations in all replicates of the corn kernels and soil samples are shown in Table 1. The histograms shown in Figure 2 revealed that aflatoxin B1 is extensively distributed throughout Anuradhapura district. Further, the Shapiro – Wilk normality test revealed that the data were not normally distributed in both corn kernel and soil. This was demonstrated in the w value of 0.85699 and p-value of 0.0001915 (<0.05) for corn kernel, and w value of 0.76076 with 4.335 × 10−8 (<0.05) of p-value for corn grown soil. According to the US FDA (2000), 20 ppb is the poisonous or deleterious level for corn consumption. There were toxin contaminations up to 60–70 ppb in corn kernel while majority of soil represented 350–400 ppb of aflatoxin B1 concentration.
Table 1.
Aflatoxin concentrations in the corn and corn grown soil samples. Values are expressed as average of 3 replicates and the margin of error is ±15%, ND = Not Detected.
| Field Location | Concentration (ppb) |
|
|---|---|---|
| Kernel | Soil | |
| Madatugama | ND | 12.20 |
| ND | ND | |
| ND | ND | |
| Kekirawa | 7.27 | 80.95 |
| ND | 3.69 | |
| ND | 3.26 | |
| Eppawala | ND | ND |
| ND | ND | |
| 8.48 | 49.36 | |
| Mahailluppallama | ND | 64.03 |
| 14.14 | 73.49 | |
| ND | 65.75 | |
| Thambuththegama | ND | 11.61 |
| 6.18 | 6.12 | |
| 5.66 | 5.50 | |
| Rajanganaya | ND | ND |
| 4.70 | 59.32 | |
| 4.00 | 86.69 | |
| Thalawa | 6.34 | 84.64 |
| 6.09 | 68.53 | |
| 10.90 | 11.58 | |
| Sravastipura | 18.71 | 4.56 |
| 0.64 | 273.64 | |
| 7.66 | 79.70 | |
| Nachchadoowa | 3.02 | 60.60 |
| 2.00 | 74.19 | |
| 40.93 | 78.40 | |
| Saliyapura | 25.92 | 89.77 |
| ND | 7.05 | |
| 18.16 | 15.97 | |
| Rambewa | ND | 8.72 |
| 21.05 | 409.41 | |
| ND | 358.09 | |
| Punewa | 2.16 | 384.35 |
| ND | 366.40 | |
| ND | 347.97 | |
| Kebithigollewa | 47.54 | 374.07 |
| 28.22 | 385.26 | |
| 1.47 | 382.33 | |
| Mihintale | 36.04 | 406.11 |
| 42.99 | 358.51 | |
| 8.39 | 384.30 | |
| Galenbindunuwewa | ND | 354.18 |
| ND | 361.24 | |
| 8.46 | 378.63 | |
| Horowpathana | 37.03 | 391.22 |
| 23.43 | 392.68 | |
| 41.48 | 402.24 | |
| Kahatagasdigiliya | 41.31 | 401.23 |
| ND | 388.44 | |
| 1.35 | 395.25 | |
| Palugaswewa | 54.31 | 400.86 |
| ND | 387.89 | |
| ND | 391.80 | |
| Thirappane | 44.59 | 400.89 |
| 64.28 | 369.13 | |
| ND | 337.49 | |
| Maha vilachchiya | 3.66 | 349.87 |
| 6.76 | 372.96 | |
| 55.37 | 374.66 | |
Figure 2.
Frequency of distribution of aflatoxin B1 for the a) corn grain, and b) corn soil.
Majority of the population in Anuradhapura are subsistence farmers who have minor-scale corn-grown fields. They have used Pacific 999, Pacific 989, Jambo and Jet as corn breeder seeds for corn cultivation and a mixture of Triple Super Phosphate (TSP), Urea and Muriate of Potash (MoP) as the main fertilizer. Wilson et al. (1989) have found that high amount of nitrogen-based fertilization increases aflatoxin levels in corn, while Payne (1987) has pointed out that low amount of fertilizer similarly increased aflatoxin B1. Furthermore, Handawela (1992) had identified seven types of soil throughout Anuradhapura, all of which are possible to have contributed to the occurrence of aflatoxin B1. The soil type, landform, irrigation, fertilization and cultivation which results in altering edaphic factors may have had a significant effect on spore numbers in soil which are responsible for aflatoxin B1 contamination (Zhang et al. 2017).
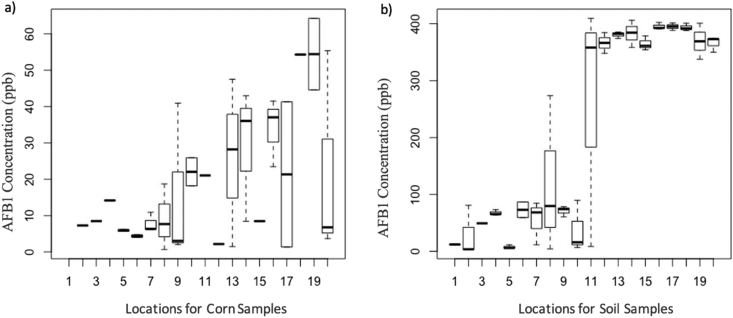
According to the box plot analysis (Figure 3), a p-value of 0.00197 (<0.05) with a w value of 275 was observed for corn kernel, and a w value of 1.026 × 10−9 (<0.05) and a p-value 666 was observed for corn-grown soil. Depending on the Wilcoxon rank sum test, (H0: High levels of AFB1 ≤ 0, Ha: Acceptable levels of aflatoxin B1 > 0), corn samples obtained from Madatugama was free of aflatoxin contamination while Kekirawa, Eppawala, Mahailluppallama, Thambuththegama, Rajanganaya, Thalawa, Sravastipura, Punewa and Galenbindunuwewa were contaminated, but not exceeding the acceptable level. Nachchadoowa, Saliyapura, Rambewa, Kebithigollewa, Mihintale, Horowpathana, Kahatagasdigiliya, Palugaswewa, Thirappane and Maha vilachchiya were contaminated with aflatoxin and it exceeded the acceptable level. Though there are regulatory control limitations for aflatoxin B1 in feed and foodstuff, a suitable threshold level for soil aflatoxin B1 has not been established to date. According to the Wilcoxon rank sum test there were two significant groups of aflatoxin B1 concentrations (H0: High levels of aflatoxin B1 ≤ 0, Ha: low levels of aflatoxin B1 > 0) in corn-grown soil. For instance, Rambewa, Punewa, Kebithigollewa, Mihintale, Galenbindunuwewa, Horowpathana, Kahatagasdigiliya, Palugaswewa, Thirappane and Maha vilachchiy showed high level of aflatoxin B1 concentrations while Madatugama was free of aflatoxin and Kekirawa, Eppawala, Mahailluppallama, Thambuththegama, Rajanganaya, Thalawa, Sravastipura, Nachchadoowa and Saliyapura showed comparatively low level of aflatoxin B1 concentrations.
Figure 3.
Distribution of aflatoxin B1 in the corn kernel, a); and corn-grown soil, b). AFB1 (1-Madatugama, 2-Kekirawa, 3-Eppawala, 4-Mahailluppallama, 5-Thambuththegama, 6-Rajanganaya, 7-Thalawa, 8-Sravastipura, 9-Nachchadoowa, 10-Saliyapura, 11-Rambewa, 12-Punewa, 13-Kebithigollewa, 14-Mihintale, 15-Galenbindunuwewa, 16-Horowpathana, 17-Kahatagasdigiliya, 18-Palugaswewa, 19-Thirappane, 20-Mahavilachchiya).
Non-parametric correlation test (Kendall's rank test and Spearman's rank correlation test) revealed a relationship between corn kernels and corn grown soil with a t-value of 425 and p-value of 0.03309 (<0.05) for corn kernels, and 5742 of s valve with a p-value of 0.01109 (<0.05) for corn-grown soil. Corn kernel samples obtained from Rambewa, Punewa, Kebithigollewa, Mihintale, Galenbindunuwewa, Horowpathana, Kahatagasdigiliya, Palugaswewa, Thirappane and Mahavilachchiya which exceeded the acceptable level of aflatoxin B1, showed higher level of aflatoxin B1 in corn grown soil as well according to the Wilcoxon rank sum test. Thus, it appeared that corn kernel aflatoxin B1 increases in relation to corn grown soil aflatoxin B1 concentration, because soil is considered as the primary inoculum of A. flavus. Horn et al. (1995) also pointed out that soil is considered as the main reservoir of A. flavus which is responsible for secondary metabolite aflatoxin B1 production, albeit spores can be transferred to plants via wind and splashing.
4. Conclusions
Aflatoxin B1 contamination in corn and corn grown soil appears to be an evolving problem in the Anuradhapura district of Sri Lanka. From this study, it was shown that out of the 20 areas studied, Nachchadoowa, Saliyapura, Rambewa, Kebithigollewa, Mihintale, Horowpathana Kahatagasdigiliya, Palugaswewa, Thirappane and Maha vilachchiya had exceeded US FDA regulatory levels for corn consumption while only Madatugama was free of aflatoxin B1 contamination. However, aflatoxin B1 does not appear to be habitually distributed due to possible factors such as unequal weather conditions, farming practices, soil type, corn genotype and fertilization. According to the results, aflatoxin B1 reported in corn-grown soil is relatively larger than in the corn kernel. In addition, high soil aflatoxin B1 concentrations lead to increased aflatoxin B1 in the corn kernel. Therefore, it can be concluded that, there is a possibility of corn kernel contamination related to soil aflatoxin levels. Organic and biological farming practices can be primarily used to overcome these problems. For instance, biocontrol A. flavus strains with a nutrient-supplying carrier can be used to out-compete wild aflatoxigenic A. flavus at the site of application. Nevertheless, there should be proper guidance and regulations to dispose contaminated produce safely. Proper regulatory control limitations for soil aflatoxin B1 should be introduced to identify contaminated soil before cultivation and economically feasible strategies should be introduced to reduce or overcome exposure. Aflatoxin contaminated corn on the other hand can be recommended for ethanol production to recover the expenditure spent on cultivation practices. While phytochemical applications have been researched in studies such as Zhao et al. (2018) and Tiwari and Shankar (2018) to modulate aflatoxin biosynthesis, the feasibility of using such methods within a Sri Lankan agricultural context, where farmers would look for low-cost, time-saving remedies, is uncertain.
Although it may seem obvious from this study that there exists a relationship between aflatoxin B1 contamination in corn kernels and corn-grown soil, the influence of other environmental factors on aflatoxin B1 contamination cannot be excluded. The weather conditions, inclusive of other physical factors such as pH and temperature, also play an important role for the growth and therefore, are an important aspect to be investigated during further studies on aflatoxin B1 contamination in the North Central Province of Sri Lanka. In addition, the effect of soil type, crop rotation, previous usages of soil for the same or different types of crops and other factors related to soil chemistry would also have an effect, and thus, would be investigated in future. Nevertheless, this study serves as a preliminary investigation and is of importance given that recent systematic studies have not been published to date.
Declarations
Author contribution statement
W.M.S.C. Jayaratne, A.H.M.A.K. Abeyratne: Performed the experiments; Analyzed and interpreted the data.
H.K.S. De Zoysa: Conceived and designed the experiments; Analyzed and interpreted the data.
D.M.R.B.N. Dissanayake: Analyzed and interpreted the data.
T.C. Bamunuarachchige, V.Y. Waisundara: Conceived and designed the experiment; Wrote the paper.
S. Chang: Contributed reagents, materials, analysis tools or data.
Funding statement
S. Chang wishes to acknowledge the funding provided by Griffith University, Australia for some components of this study.
Declaration of interests statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
References
- Abbas H.K., Accinelli C., Wilkinson J.R., Zablotowicz R.M. Aspergillus flavus aflatoxin occurrence and expression of aflatoxin biosynthesis genes in soil. Can. J. Microbiol. 2008;54:71–379. doi: 10.1139/w08-018. [DOI] [PubMed] [Google Scholar]
- Abbas H.K., Locke M.A., Zablotowicz R.M. Spatial variability of Aspergillus flavus soil populations under different crops and corn grain colonization and aflatoxins. Can. J. Bot. 2004;82:1768–1775. [Google Scholar]
- Bandara J.M.R.S., Bean G.A., Vithanege A.K. Occurrence of aflatoxins in parboiled rice in Sri Lanka. Mycopathologia. 1991;116:65–70. doi: 10.1007/BF00436366. [DOI] [PubMed] [Google Scholar]
- Food and Drug Administration U.S.A. 2000. Guidance for Industry: Action Levels for Poisonous or Deleterious Substances in Human Food and Animal Feed.https://www.fda.gov/food/guidanceregulation/ucm077969.htm#afla (last accessed on 26th of March 2019) [Google Scholar]
- Gams W., Christensen M., Onions A.H.S., Pitt J.I., Samson R.A. Intrageneric taxa of Aspergillus. In: Samson R.A., Pitt J.I., editors. Advances in Aspergillus and Penicillium Systematics. 1985. pp. 55–62. New York. [Google Scholar]
- Garcia R.J., Cotty P.J. Aspergillus flavus in soils and corn cobs in south Texas: implications for management of aflatoxins in corn–cotton rotations. Plant Dis. 2004;88:1366–1371. doi: 10.1094/PDIS.2004.88.12.1366. [DOI] [PubMed] [Google Scholar]
- Gourama H., Bullerman L.B. Aspergillus flavus and Aspergillus parasiticus: aflatoxigenic fungi of concern in foods and feeds: a review. J. Food Protect. 1995;58(12):1395–1404. doi: 10.4315/0362-028X-58.12.1395. [DOI] [PubMed] [Google Scholar]
- Handawela J. Department of Agriculture, Peradeniya: The Central Environmental Authority; 1992. Anuradhapura District Environmental Profile - Core, Land and Water Management Research Centre.https://core.ac.uk/download/pdf/52178700.pdf [Google Scholar]
- Horn B.W., Greene R.L., Dorner J.W. Effect of corn and peanut cultivation on soil populations of Aspergillus flavus and A. parasiticus in southwestern Georgia. Appl. Environ. Microbiol. 1995;61:2472–2475. doi: 10.1128/aem.61.7.2472-2475.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karunarathna N.B., Fernando C.J., Munasinghe D.M.S., Fernando R. Occurrence of aflatoxins in edible vegetable oils in Sri Lanka. Food Contr. 2019;101:97–103. [Google Scholar]
- Lakkireddy K., Kondapalli K., Rao K.S. Aflatoxin in food and feed: the science of safe food. Res. Rev.: J. Food Sci. Technol. 2014;3:6–11. [Google Scholar]
- Manage P.M., Dissanayake D.M.M.P. Aflatoxin contamination in peanuts commercially available in Sri Lanka. Vidyodaya J. Sci. 2009:151–159. [Google Scholar]
- Nesci A., Etcheverry M. Aspergillus section Flavi populations from field maize in Argentina. Lett. Appl. Microbiol. 2002;34:343–348. doi: 10.1046/j.1472-765x.2002.01094.x. [DOI] [PubMed] [Google Scholar]
- Payne G.A. Aspergillus flavus infection of maize: silks and kernels. In: Zuber M.S., Lillehoj E.B., Renfro B.L., editors. Aflatoxin in Maize: Proceedings of the Workshop. CIMMYT; Mexico: 1987. pp. 119–129. [Google Scholar]
- Razzazi-Fazeli E., Noviandi C.T., Porasuphatana S., Agus A., Böhm J. A survey of aflatoxin B1 and total aflatoxin contamination in baby food, peanut and corn products sold at retail in Indonesia analyzed by ELISA and HPLC. Mycotoxin Res. 2004;20:51–58. doi: 10.1007/BF02946735. [DOI] [PubMed] [Google Scholar]
- Rushing B.R., Selim M.I. Aflatoxin B1: a review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem. Toxicol. 2019;124:81–100. doi: 10.1016/j.fct.2018.11.047. (2019) [DOI] [PubMed] [Google Scholar]
- Tiwari S., Shankar J. Integrated proteome and HPLC analysis revealed quercetin-mediated inhibition of aflatoxin B1 biosynthesis in Aspergillus flavus. Biotechnology. 2018;8(1):47. doi: 10.1007/s13205-017-1067-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams W.P., Windham G.L., Buckley P.M., Daves C. Aflatoxin accumulation in conventionak and transgenic corn hybrids infested with southwestern corn borer (Lepidoptera. Crambidae) J. Agric. Urban Entomol. 2002;19:227–236. [Google Scholar]
- Wilson D.M., Walker M.E., Gascho G.J. Some effects of mineral nutrition on aflatoxin contamination of corn and peanuts. In: Engelhard A.W., editor. Soil-borne Plant Pathogens: Management of Diseases with Macro and Microelements. APS Press; St. Paul Minnesota: 1989. pp. 137–151. [Google Scholar]
- Yogendrajah P., Jacxsens L., Saeger S.D., Meulenaer B.D. Co-occurrence of multiple mycotoxins in dry chilli (Capsicum annum L.) samples from the markets of Sri Lanka and Belgium. Food Contr. 2014;46:26–34. [Google Scholar]
- Zain M.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011;15:129–144. [Google Scholar]
- Zhang C., Selvaraj J., Yang Q., Liu Y. A Survey of Aflatoxin Producing Aspergillus sp. from peanut field soils in four Agroecological zones of China. Toxins. 2017;9:40. doi: 10.3390/toxins9010040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X.X., Zhi Q.Q., Li J.Y., Keller N.P., He Z.M. The antioxidant gallic acid inhibits aflatoxin formation in Aspergillus flavus by modulating transcription factors FarB and CreA. Toxins. 2018;10(7):270. doi: 10.3390/toxins10070270. [DOI] [PMC free article] [PubMed] [Google Scholar]