Abstract
Objectives
The COVID-19 pandemic presents an urgent need to investigate whether existing drugs can enhance or even worsen prognosis; metformin, a known mammalian target of rapamycin (m-TOR) inhibitor, has been identified as a potential agent. We sought to evaluate mortality benefit among older persons infected with SARS-CoV-2 who were taking metformin as compared to those who were not.
Design
Retrospective cohort study.
Setting and Participants
775 nursing home residents infected with SARS-CoV-2 who resided in one of the 134 Community Living Centers (CLCs) of the Veterans Health Administration (VHA) during March 1, 2020, to May 13, 2020, were included.
Methods
Using a window of 14 days prior to SARS-CoV-2 testing, bar-coded medication administration records were examined for dispensing of medications for diabetes. The COVID-19–infected residents were divided into 4 groups: (1) residents administered metformin alone or in combination with other medications, (2) residents who used long-acting or daily insulin, (3) residents administered other diabetes medications, and (4) residents not administered diabetes medication, including individuals without diabetes and patients with untreated diabetes. Proportional hazard models adjusted for demographics, hemoglobin A1c, body mass index, and renal function.
Results
Relative to those not receiving diabetes medications, residents taking metformin were at significantly reduced hazard of death [adjusted hazard ratio (HR) 0.48, 95% confidence interval (CI) 0.28, 0.84] over the subsequent 30 days from COVID-19 diagnosis. There was no association with insulin (adjusted HR 0.99, 95% CI 0.60, 1.64) or other diabetes medications (adjusted HR 0.71, 95% CI 0.38, 1.32).
Conclusions and Implications
Our data suggest a reduction in 30-day mortality following SARS-CoV-2 infection in residents who were on metformin-containing diabetes regimens. These findings suggest a relative survival benefit in nursing home residents on metformin, potentially through its mTOR inhibition effects. A prospective study should investigate the therapeutic benefits of metformin among persons with COVID-19.
Keywords: Nursing home, diabetes, COVID-19, metformin
On December 31, 2019, an unforeseen health crisis emerged in Wuhan City, China. The World Health Organization reported a series of 44 pneumonia cases,1 related to a new beta-coronavirus, SARS-CoV-2. By January 20, 2020, this newly termed coronavirus disease 2019 (COVID-19) was detected in the United States.2 With COVID-19's rapid worldwide spread came the quick recognition of the major threat posed to the geriatric population, particularly the comorbid and frail.3 Severe COVID-19 can occur in the presence of multiple chronic illnesses and age-related immunosenescence4; grouped living conditions of many older people, particularly those in facilities, further exacerbate the risk of acquiring COVID-19. With urgent worldwide efforts to both prevent and treat COVID-19, there is interest in determining whether existing therapeutic options can be repurposed for use against this disease.
Type 2 diabetes mellitus (T2DM) ranks highly among the multiple morbidities that affect older nursing home residents. Patients with diabetes typically fare worse with infections than persons without diabetes. Metformin, among the most common T2DM medications, could play an important role for patients with diabetes infected with SARS-CoV-2. A recent focus on potential treatment for SARS-CoV-2 infection includes a pathway not usually considered for its “antiviral” property, the mammalian target of rapamycin (mTOR) pathway.5 mTOR complexes are critical in many cell functions including senescence and apoptosis, and they also play a role in viral protein production, processing, and assembly. mTOR and its protein complexes, mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2), are functionally intertwined with the adenosine monophosphate (AMP)–associated protein kinase (AMPK) pathway, and both biological cascades appear to be plausible targets for COVID-19 therapy.5
Metformin's immunomodulatory effects on mTOR inhibition have sparked interest as a potential cancer and antiviral therapeutic. In a study by Sahra and colleagues, the expression of the protein REDD1 (regulated in development and DNA damage responses 1), a known negative regulator of mTOR, rapidly increased following metformin treatment in prostate, breast, and lung cancer cells.6 Metformin may also boast additional anti-inflammatory benefits via alterations of the gut microbiota through carbohydrate metabolism7 and/or antiseptic properties.8 Metformin's potential anti-inflammatory and immunomodulatory effects are not thoroughly understood, but increased interest in the utility of metformin in inflammatory diseases outside of T2DM, such as human immunodeficiency virus,9 multiple sclerosis,10 and systemic lupus,11 is growing. Metformin historically has shown a modest benefit in treating influenza.12 Influenza A virus has been shown to exploit host signaling molecules, including mTORC1 and mTORC2 protein complexes, to increase viral replication.13 Metformin's potential antiviral mechanism of action could be due to increased REDD1, which would lead to subsequent mTOR regulation.6
A recent study demonstrated that patients with diabetes with confirmed COVID-19 infection had elevated risk of severe pneumonia, hyperinflammatory state, and hypercoagulability compared with a similar cohort of individuals without diabetes.14 In both patients with diabetes and individuals without diabetes, the potential role for metformin in treating COVID-19 has been suggested.15 Without a prospective trial, using available cohort data from a population with confirmed SARS-CoV-2 infection that includes patients with diabetes treated with metformin allows for evaluation of theoretical benefits of metformin among persons with COVID-19. In nursing home residents with laboratory confirmation of SARS-CoV-2 infection, we examined the association of metformin on survival.
Methods
This retrospective study was approved by the Institutional Review Board.
Cohort
The cohort included all residents of the 134 Community Living Centers (CLC) operated by the Veterans Health Administration (VHA). CLCs are nursing homes that the VHA operates directly. We evaluated the subset with laboratory-confirmed evidence of SARS-CoV-2 tested during the interval of March 1, 2020, and May 13, 2020, following the VHA directive that all residents be tested.
Diabetes Medication Exposure
Using a window of 14 days prior to SARS-CoV-2 testing, we examined the bar-coded medication administration records for evidence of medications for diabetes. Residents with COVID-19 were divided into 4 groups: (1) residents who were administered metformin alone or in combination with other medications, (2) residents who used long-acting insulin or daily regular insulin (received regular insulin on ≥12 of 14 days), (3) residents who were administered other diabetes medications, and (4) residents who were not administered diabetes medication. Group 4 included individuals without diabetes as well as patients with diabetes who were untreated or diet-controlled.
Outcomes
The primary outcome was for 30-day mortality as measured from the date of SARS-CoV-2 positivity. The secondary outcome was hospital-free survival at 30 days from the date of SARS-CoV-2 positivity.
Covariates
Demographic variables collected from VA administrative records included age, sex, and race. Comorbidities were collected to complete the Elixhauser comorbidity index.16 We collected additional variables that would be related to diabetes and metformin utilization. Estimated glomerular filtration rate (eGFR) was collected from VA laboratory data and when missing was calculated according to the Chronic Kidney Disease Epidemiology equation. Hemoglobin A1c (glycated hemoglobin) was collected from VA laboratory records. Body mass index was calculated from the most recently recorded weight and height. In addition, the length of CLC stay (days), and VA hospitalizations in the prior year (days) were collected from VA administrative records.
Analysis
Characteristics were compared across the populations using an analysis of variance for continuous variables and a chi-square for dichotomous variables. The primary analysis referenced to the group of residents who were not treated with diabetes medications. The cohort was followed for the outcomes until 30 days after positive SARS-CoV-2 reporting date. Both the primary mortality outcome and the secondary 30-day hospital-free survival outcome are time dependent, and a proportional hazard model was used for bivariate analysis and multivariable analysis. The proportional hazard assumption was satisfied for each of the outcomes. Multivariable regression included variables that were significantly disparate among the groups.
Results
Characteristics of the 775 residents with SARS-CoV2 infection are displayed in Table 1 , with groups compared for any differences. The majority were not on diabetes pharmacotherapy (61.4%, n=476). The mean age at time of data collection was 75.5 (±10.8) years, with a significantly lower age in those on metformin (72.3 ±8.3 years) and insulin (73.3 ±9.4 years years). Consistent with a VHA cohort, residents were 97.3% (n=754) male. The percentage of diabetes medication use was higher in black residents who were more often on insulin (40.8% n=42), metformin (30.7%, n=39), and other diabetes medications (33.3%, n=23), relative to those not on medications (20.8%, n=99) (P < .001). Hemoglobin A1c was higher among residents on diabetes medication (insulin 7.7%; metformin 7.5%, other medications 6.5%; no medication 5.8%; P < .001). Body mass index greater than 30 was more common among residents on insulin (32.0%, n=33), metformin (40.9%, n=52), and other diabetes medications (33.3%, n=23), relative to those on no medication for diabetes (21.4%, n=55). Of the comorbidities, there were generally no significant differences within groups. History of a mental illness with psychosis was highest in residents on metformin (51.2%, n=65), whereas those on no diabetes medications (41.6%, n= 198) and those on other diabetes medications (43.5%, n=30) were higher than those on insulin (33.0%, n=34).
Table 1.
Characteristics of Nursing Home Residents with COVID-19
| Overall (n = 775) |
No Diabetes Drugs (n = 476) |
Metformin (n = 127) |
Insulin (n = 103) |
Other Diabetes Drugs (n = 69) |
P Value | |
|---|---|---|---|---|---|---|
| Demographics | ||||||
| Age, mean (SD) | 75.6 (10.8) | 76.9 (11.7) | 72.3 (8.3) | 73.3 (9.4) | 75.6 (9.2) | <.001 |
| Male, n (%) | 754 (97.3) | 461 (96.8) | 125 (98.4) | 100 (97.1) | 68 (98.6) | .70 |
| Female, n (%) | 21 (2.7) | 15 (3.2) | 2 (1.6) | 3 (2.9) | 1 (1.4) | .70 |
| Race (%) | .001 | |||||
| Black | 203 (26.2) | 99 (20.8) | 39 (30.7) | 42 (40.8) | 23 (33.3) | |
| Other | 58 (7.5) | 41 (8.6) | 10 (7.9) | 5 (4.9) | 2 (2.9) | |
| White | 514 (66.3) | 336 (70.6) | 78 (61.4) | 56 (54.4) | 44 (63.8) | |
| Diabetes-related factors | ||||||
| Hemoglobin A1c, %, mean (SD) | 6.54 (1.38) | 5.80 (0.72) | 7.49 (1.44) | 7.68 (1.47) | 6.50 (1.25) | <.001 |
| Diabetes, ICD-10, n (%) | 308 (39.7) | 95 (20.0) | 107 (84.3) | 81 (78.6) | 25 (36.2) | <.001 |
| Diabetes complication, ICD-10, n (%) | 301 (38.8) | 82 (17.2) | 103 (81.1) | 93 (90.3) | 23 (33.3) | <.001 |
| Body mass index, mean (SD) | 27.26 (6.36) | 26.04 (5.74) | 29.65 (6.57) | 29.31 (6.96) | 28.19 (7.01) | <.001 |
| Body mass index ≥30, n (%) | 210 (27.1) | 102 (21.4) | 52 (40.9) | 33 (32.0) | 23 (33.3) | <.001 |
| Obesity, ICD-10, n (%) | 118 (15.2) | 55 (11.6) | 28 (22.0) | 28 (27.2) | 7 (10.1) | <.001 |
| Estimated glomerular filtration rate, mL/min/1.73 m2, mean (SD) | 74.78 (32.04) | 72.72 (29.67) | 73.23 (25.19) | 83.38 (42.90) | 79.00 (38.29) | .016 |
| Renal disease, ICD-10, n (%) | 195 (25.2) | 97 (20.4) | 17 (13.4) | 54 (52.4) | 27 (39.1) | <.001 |
| Other comorbidities | ||||||
| Dementia, n (%) | 537 (69.3) | 333 (70.0) | 86 (67.7) | 69 (67.0) | 49 (71.0) | .90 |
| Pulmonary disease, n (%) | 264 (34.1) | 159 (33.4) | 40 (31.5) | 41 (39.8) | 24 (34.8) | .57 |
| Hypothyroid, n (%) | 91 (11.7) | 55 (11.6) | 15 (11.8) | 17 (16.5) | 4 (5.8) | .20 |
| Tumor, n (%) | 131 (16.9) | 90 (18.9) | 14 (11.0) | 19 (18.4) | 8 (11.6) | .11 |
| Weight loss, n (%) | 138 (17.8) | 97 (20.4) | 14 (11.0) | 15 (14.6) | 12 (17.4) | .08 |
| Alcohol use disorder, n (%) | 96 (12.4) | 65 (13.7) | 11 (8.7) | 12 (11.7) | 8 (11.6) | .49 |
| Drugs use disorder, n (%) | 60 (7.7) | 42 (8.8) | 9 (7.1) | 6 (5.8) | 3 (4.3) | .48 |
| Any substance use disorder, n (%) | 125 (16.1) | 82 (17.2) | 17 (13.4) | 17 (16.5) | 9 (13.0) | .65 |
| Psychiatric diagnosis, n (%) | 541 (69.8) | 338 (71.0) | 88 (69.3) | 73 (70.9) | 42 (60.9) | .39 |
| Psychoses, n (%) | 327 (42.2) | 198 (41.6) | 65 (51.2) | 34 (33.0) | 30 (43.5) | .048 |
| Depression, n (%) | 330 (42.6) | 199 (41.8) | 53 (41.7) | 51 (49.5) | 27 (39.1) | .47 |
| Utilization | ||||||
| Hospital days in prior year, mean (SD) | 9.54 (20.81) | 9.26 (21.32) | 7.52 (18.85) | 12.81 (23.60) | 10.28 (15.39) | .27 |
| Long-stay resident, n (%) | 155 (20.0) | 106 (22.3) | 13 (10.2) | 22 (21.4) | 14 (20.3) | .026 |
| Outcomes | ||||||
| 30-d mortality, n (%) | 160 (20.6) | 108 (22.7) | 16 (12.6) | 24 (23.3) | 12 (17.4) | .07 |
ICD-10, International Classification of Diseases, 10th Edition.
Bolded P values were selected for inclusion in multivariable modeling.
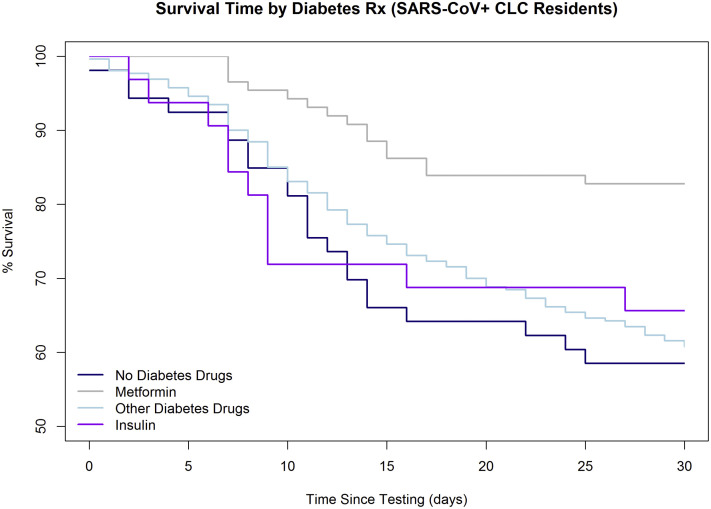
Thirty-day mortality was lowest in SARS-CoV-2 residents taking metformin (12.6%, n=12) compared with those on other diabetes medications (17.4%, n=12), insulin (23.3%, n=24), and no diabetes medications (22.7%, n=108; Table 2 ). Relative to those not receiving diabetes medications, residents taking metformin were at significantly reduced hazard of death (adjusted hazard ratio 0.48, 95% confidence interval 0.28, 0.84) over the subsequent 30 days from COVID-19 diagnosis after adjustment for age, race, hemoglobin A1c, renal function, long stay, and past mental health with psychosis. Figure 1 displays the survival curve based on diabetes medication exposure. There was no difference in hospital-free survival between all treatment groups.
Table 2.
Mortality Among Nursing Home Residents With COVID-19
| 30-d Mortality | No Diabetes Drug | Insulin | Metformin | Other Diabetes Drugs |
|---|---|---|---|---|
| Unadjusted HR (95% CI) | Referent | 1.00 (0.63, 1.57) | 0.42 (0.26, 0.69) | 0.77 (0.42, 1.40) |
| Adjusted HR∗ (95% CI) | Referent | 0.99 (0.60, 1.64) | 0.48 (0.28, 0.84) | 0.71 (0.38, 1.32) |
CI, confidence interval; HR, hazard ratio.
Adjusted for age, body mass index, hemoglobin A1c, estimated glomerular filtration rate, long stay (>90 days), and underlying psychoses.
Fig. 1.
Survival curve by diabetes medication use. Shown is the survival curve in the 30-days post-testing in SARS-CoV2–positive diabetic patients on metformin (gray), insulin (violet), other diabetes drugs (light blue), and no therapy (navy). After adjustment for demographics, diabetes, obesity, renal function, and psychosis, metformin was associated with a 52% reduced hazard of mortality (adjusted hazard ratio 0.48, 95% confidence interval 0.28, 0.84).
Discussion
Diabetes mellitus is a common medical condition estimated by the Centers for Disease Control and Prevention to affect up to 26.8% of American individuals older than 65 years.17 Metformin is a first-line treatment for diabetes and has been demonstrated to have immunomodulatory and antiviral activity via mTOR inhibition. Our data suggest that metformin use may be associated with decreased mortality from SARS-CoV-2 infection among nursing home residents on a population level.
These findings suggest an association between metformin use among nursing home residents with COVID-19 and relative survival benefit. This association could be mediated through the mTOR inhibition effects of metformin. Although it is difficult to demonstrate that mTOR inhibition is causal, the findings are consistent with recent research on everolimus (formerly RAD001), an mTOR inhibitor and rapamycin analogue, which was found to improve age-related immune dysfunction. This study demonstrated improved response to influenza vaccination (gauged by increased antibody titers) in older participants receiving everolimus.18 A newer agent, dactolisib (RTB101, ResTORbio), a combined phosphatidylinositol-3 kinase (PI3k) inhibitor and mTOR inhibitor, is also being investigated for its potential role in ameliorating immune function in older people. Reduced rates of pulmonary infections in infection over a 1-year period were reported in both dactolisib and combination therapy groups despite study participants receiving a limited 6-week course of therapy.19 Dactolisib was additionally administered to older patients at risk for pulmonary infections (ie, with asthma, chronic obstructive pulmonary disease, T2DM) and demonstrated both a 30% reduction in laboratory-confirmed viral respiratory infections and increased pan-antiviral signaling via interferon stimulation.20
A recent review of pathways implicated in both diabetes and SARS-CoV-2 coronavirus infection explores the role of angiotensin-converting enzyme 2 (ACE2) and dipeptidyl peptidase 4 (DPP4), both coronavirus receptors, as potential therapeutic targets.21 Other molecular pathways common to diabetes and SARS-CoV-2 infection may explain the relative benefit seen in the metformin-containing medication groups. Alternatively, its benefit may be related to insulin sensitization effects. Viral-induced interferon-gamma secretion has been demonstrated to increase muscular insulin resistance and circulating insulin levels, which, in turn, increase cluster of differentiation 8 cytotoxic T-cell (CD8+ T-cell) responses.22 Metformin is an inhibitor of the mitochondrial electron transport chain, promoting glycolysis,23 which may increase CD8+ effector function but impair memory T-cell responses.24 Metformin's ability to reduce neutrophil counts and to reduce neutrophil extracellular traps have also been proposed as potential mechanisms for its beneficial use in patients with diabetes and COVID-19.25
Other retrospective studies have also recently suggested that metformin may be associated with decreased mortality from COVID-19. Bramante et al26 reviewed United Health insurance claims from all 50 states in the United States among patients with diabetes hospitalized with COVID-19 and found that metformin was associated with decreased mortality among women. Crouse et al27 found that diabetes was a risk factor for COVID-19 infection among over 25,000 persons tested at the University of Alabama at Birmingham, and they found metformin treatment in patients with diabetes to be associated with decreased mortality from COVID-19. Luo et al28 found in-hospital mortality among patients with diabetes and COVID-19 to be lower among those taking metformin in a study from Wuhan, China. One meta-analysis that included these 3 studies and two small others found metformin use to be associated with decreased COVID-19 mortality.29 A second meta-analysis that included these 3 studies as well as the CORONADO study in France30 describes the observed reduction in mortality from COVID-19 associated with metformin.31 In a more recent retrospective study assessing incidence of lactic acidosis and 28-day mortality in hospitalized individuals with COVID-19, Cheng et al32 noted increased incidence of lactic acidosis in patients on metformin without any increase in mortality between patients with acidosis and those without. Their results differ from our own, though a comparison is challenging as their cohort was aged 18-80 years (median age 63 years) and had already been hospitalized. Of interest are their findings of improving inflammatory marker profiles, notably 28-day trends of CRP, IL-6, IL-2, and TNF-α, in addition to reduced indices of cardiac injury and congestive heart failure in patients with COVID-19 on metformin. Our findings appear to be complementary to those of Cheng et al, and our study is contributory as it represents data from a largely older, nonhospitalized population.
The strengths of our study include the availability of data from more than 7600 nursing home residents within the VA system, all of whom underwent COVID-19 screening. Our control group included individuals without diabetes as well as patients with diabetes who were untreated or diet-controlled. Although there were differences in body mass index and baseline hemoglobin A1c between treatment groups, they had similar rates of hospitalization prior to positive SARS-CoV-2 testing. Furthermore, there was no significant difference in eGFR between all treatment groups, though the insulin group had a larger variance in renal function that could signify different degrees of renal dysfunction and/or the inclusion of hemodialysis patients within that data subset.
The generalizability of the sample is limited by the underlying demographics of the VA population, which is predominantly male, relative to the US nursing home population, which is predominantly female. It is not evident if gender has any effect on our findings, or if any metformin effects would be applicable in female residents.
Another limitation is the analytic strategy of proportional hazards modeling that uses listwise deletion, which reduced our population available for regression analyses size by approximately one-third—most notably due to missing laboratory covariates such as hemoglobin A1c and estimated glomerular filtration rate. Bivariable analysis was unaffected but multivariable analysis was restricted and could not be adjusted for all possible confounding variables. Our data additionally was unable to shed light on the rationale for their specific diabetic regimen as well as reason for hospitalization. As a retrospective study that did not include clinical record review, we are unable to determine the appropriateness of metformin use. Additionally, our study was designed to look at population outcomes rather than the biochemistry of metformin. SARS-CoV-2 activity relies on a multitude of intracellular processes, and the case for mTOR being relevant is made by extrapolation from our data on other viruses, as noted above. Metformin may be beneficial through its effects on other pathways yet unidentified. Additionally, metformin may have non-antiviral effects on inflammation, hypercoagulability, and ACE2 expression that cannot be investigated further with our study design. A final limitation worth noting is the use of other medications that may impact our data through their anti-inflammatory effects. Future analyses in larger data sets should explore the intersection of multiple medications.
Moving forward, there is great potential for additional studies examining the association of metformin use and outcomes in viral infection, both via mTOR inhibition and other effects. There is a growing body of research on the effects of metformin on neoplastic disease. Biochemical studies of metformin in the context of cytotoxic immunity and inflammation may elucidate the mechanisms by which the drug may offer benefits to both diabetic and nondiabetic patients with viral infections.
Metformin is an inexpensive and readily available therapy that could reduce mortality and hospitalization associated with SARS-CoV-2 infection in patients with T2DM for which there are no drug contraindications; further evaluation is warranted. Future clinical studies may include chart review, inflammatory marker collection, coagulation studies, and glycemic trends to better understand the association of SARS-CoV-2 on morbidity and mortality in diabetic residents.
The next step in investigating metformin use in SARS-CoV-2 infection should be a prospective, randomized controlled study. A prospective study could also allow for inclusion of nondiabetic individuals. Other trials of mTOR inhibitors are currently in process33 , 34 and comparison of outcomes in residents treated with these drugs vs metformin, to assess for the magnitude of mTOR-inhibition–dependent benefits, is a worthwhile endeavor.
Conclusions and Implications
The findings of our study suggest a reduction in 30-day mortality following SARS-CoV-2 infection in nursing home residents on metformin. Our work adds to the evidence others have found for metformin's benefit among those with SARS-CoV-2. A prospective randomized controlled trial of the therapeutic benefits of metformin would overcome some of the limitations of our study and provide an opportunity to investigate any benefits in a nondiabetic population.
Acknowledgments
All authors are VA employees. We are indebted to the VHA Offices of Geriatrics and Extended Care, Population Health, and Reporting, Analytics, Performance Improvement, & Deployment. The opinions expressed are those of the authors and do not reflect the official policies of the United States Government or the US Department of Veterans Affairs. We are thankful to Rachel Clements and Elizabeth Archambault for their thorough review and insights.
Ethics: This study was approved by the Providence VAMC Institutional Review Board.
Footnotes
ML and PT contributed equally and have agreed to share first authorship.
This work was funded by VA Health Services Research and Development (CIN 13-419; C19 20-213) and the National Institute of Aging (3P01AG027296-11S2).
The authors declare no conflicts of interest.
References
- 1.World Health Organization Disease outbreak news: Pneumonia of unknown cause—China. https://www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-cause-china/en/ Available at:
- 2.Holshue M.L., DeBolt C., Lindquist S. First case of 2019 novel coronavirus in the United States. N Engl J Med. 2020;382:929–936. doi: 10.1056/NEJMoa2001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boccardi V., Ruggiero C., Mecocci P. COVID-19: A geriatric emergency. Geriatrics. 2020;5:24. doi: 10.3390/geriatrics5020024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nikolich-Zugich J., Knox K.S., Rios C.T. SARS-CoV-2 and COVID-19 in older adults: What we may expect regarding pathogenesis, immune responses, and outcomes. Geroscience. 2020;42:505–514. doi: 10.1007/s11357-020-00186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maiese K. The mechanistic target of rapamycin (mTOR): Novel considerations as an antiviral treatment. Curr Neurovasc Res. 2020;17:1–6. doi: 10.2174/1567202617666200425205122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ben Sahra I., Regazzetti C., Robert G. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer Res. 2011;71:4366–4372. doi: 10.1158/0008-5472.CAN-10-1769. [DOI] [PubMed] [Google Scholar]
- 7.Duca F.A., Côté C.D., Rasmussen B.A. Metformin activates a duodenal AMPK-dependent pathway to lower hepatic glucose production in rats [published correction appears in Nat Med. 2016 Feb;22(2):217] Nat Med. 2015;21:506–511. doi: 10.1038/nm.3787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weinberg E.D. Antimicrobial activities of biguanides. Ann N Y Acad Sci. 1968;148:587–600. doi: 10.1111/j.1749-6632.1968.tb27733.x. [DOI] [PubMed] [Google Scholar]
- 9.Moyo D., Tanthuma G., Cary M.S. Cohort study of diabetes in HIV-infected adult patients: Evaluating the effect of diabetes mellitus on immune reconstitution. Diabetes Res Clin Pract. 2014;103:e34–e36. doi: 10.1016/j.diabres.2013.12.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Negrotto L., Farez M.F., Correale J. Immunologic effects of metformin and pioglitazone treatment on metabolic syndrome and multiple sclerosis. JAMA Neurol. 2016;73:520–528. doi: 10.1001/jamaneurol.2015.4807. [DOI] [PubMed] [Google Scholar]
- 11.Yin Y., Choi S.C., Xu Z. Normalization of CD4+ T cell metabolism reverses lupus. Sci Transl Med. 2015;7:274ra18. doi: 10.1126/scitranslmed.aaa0835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bailey C.J. Metformin: Historical overview. Diabetologia. 2017;60:1566–1576. doi: 10.1007/s00125-017-4318-z. [DOI] [PubMed] [Google Scholar]
- 13.Kuss-Duerkop S.K., Wang J., Mena I. Influenza virus differentially activates mTORC1 and mTORC2 signaling to maximize late stage replication. PLOS Pathogens. 2017;13:e1006635. doi: 10.1371/journal.ppat.1006635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Guo W., Li M., Dong Y. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020:e3319. doi: 10.1002/dmrr.3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sharma S., Ray A., Sadasivam B. Metformin in COVID-19: A possible role beyond diabetes. Diabetes Res Clin Pract. 2020;164:108183. doi: 10.1016/j.diabres.2020.108183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Elixhauser A., Steiner C., Harris D.R., Coffey R.M. Comorbidity measures for use with administrative data. Med Care. 1998;36:8–27. doi: 10.1097/00005650-199801000-00004. [DOI] [PubMed] [Google Scholar]
- 17.Centers for Disease Control and Prevention National Diabetes Statistics Report, 2020. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services, 2020. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf Available at:
- 18.Mannick J., Del Giudice G., Lattanzi M. mTOR inhibition improves immune function in the elderly. Sci Transl Med. 2014;6:268ra179. doi: 10.1126/scitranslmed.3009892. [DOI] [PubMed] [Google Scholar]
- 19.Mannick J.B., Morris M., Hockey H.P. TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci Transl Med. 2018:10. doi: 10.1126/scitranslmed.aaq1564. [DOI] [PubMed] [Google Scholar]
- 20.Mannick J., Tomlinson A., Shergill S. LB2. TORC1 Inhibition with RTB101 as a potential pan-antiviral immunotherapy to decrease the incidence of respiratory tract infections due to multiple respiratory viruses in older adults. Open Forum Infect Dis. 2019;6(Suppl 2):S993–S994. [Google Scholar]
- 21.Drucker D.J. Coronavirus infections and type 2 diabetes—Shared pathways with therapeutic implications. Endocr Rev. 2020;41:1–13. doi: 10.1210/endrev/bnaa011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Šestan M., Marinović S., Kavazović I. Virus-induced interferon-γ causes insulin resistance in skeletal muscle and derails glycemic control in obesity. Immunity. 2018;49:164–177.e166. doi: 10.1016/j.immuni.2018.05.005. [DOI] [PubMed] [Google Scholar]
- 23.Andrzejewski S., Siegel P.M., St-Pierre J. Metabolic profiles associated with metformin efficacy in cancer. Front Endocrinol (Lausanne) 2018;9:372. doi: 10.3389/fendo.2018.00372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sukumar M., Liu J., Ji Y. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J Clin Invest. 2013;123:4479–4488. doi: 10.1172/JCI69589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dalan R. Metformin, neutrophils and COVID-19 infection. Diabetes Res Clin Pract. 2020;164:108230. doi: 10.1016/j.diabres.2020.108230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bramante C., Ingraham N., Murray T. Observational study of metformin and risk of mortality in patients hospitalized with COVID-19. Preprint. medRxiv. 2020 [Google Scholar]
- 27.Crouse A., Grimes T., Li P. Metformin use is associated with reduced mortality in a diverse population with COVID-19 and diabetes. Preprint. medRxiv. 2020 doi: 10.3389/fendo.2020.600439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Luo P., Qiu L., Liu Y. Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis. Am J Trop Med Hyg. 2020;103:69–72. doi: 10.4269/ajtmh.20-0375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hariyanto T.I., Kurniawan A. Metformin use is associated with reduced mortality rate from coronavirus disease 2019 (COVID-19) infection. Obes Med. 2020;19:100290. doi: 10.1016/j.obmed.2020.100290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cariou B., Hadjadj S., Wargny M. Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: The CORONADO study. Diabetologia. 2020;63:1500–1515. doi: 10.1007/s00125-020-05180-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Scheen A.J. Metformin and COVID-19: From cellular mechanisms to reduced mortality. Diabetes Metab. 2020;46:423–426. doi: 10.1016/j.diabet.2020.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cheng X., Liu Y.M., Li H. Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;32:537–547.e3. doi: 10.1016/j.cmet.2020.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harrison C. Drug researchers pursue new lines of attack against COVID-19. Nature Biotechnol. 2020;38:659–662. doi: 10.1038/d41587-020-00013-z. [DOI] [PubMed] [Google Scholar]
- 34.Zheng Y., Li R., Liu S. Immunoregulation with mTOR inhibitors to prevent COVID-19 severity: A novel intervention strategy beyond vaccines and specific antiviral medicines. J Med Virol. 2020;92:1495–1500. doi: 10.1002/jmv.26009. [DOI] [PMC free article] [PubMed] [Google Scholar]