Abstract
Nearly half of patients with heart failure in the community have heart failure with preserved ejection fraction (HFpEF). Patients with HFpEF are often elderly and their primary chronic symptom is severe exercise intolerance. Left ventricular diastolic dysfunction is associated with the pathophysiology of HFpEF and is an important contributor to exercise intolerance in HFpEF patients. The effects of exercise training on left ventricular diastolic function in HFpEF patients have been examined in several randomised clinical trials. Meta-analysis of the trials indicates that exercise training can provide clinically relevant improvements in exercise capacity without significant change in left ventricular structure or function in HFpEF patients. Further studies are necessary to elucidate the exact mechanisms of exercise intolerance in HFpEF patients and to develop recommendations regarding the most effective type, intensity, frequency, and duration of training in this group.
Keywords: Exercise, inspiratory muscle training, functional electrical stimulation, diastolic function, exercise capacity
Nearly half of patients with heart failure (HF) in the community have HF with preserved ejection fraction (HFpEF) and mortality and morbidity in this group of patients is high.[1–4] However, to date, there is no established pharmacotherapy to improve survival in HFpEF.[5–9] Patients with HFpEF are often elderly and their primary chronic symptom is severe exercise intolerance, which results in a reduced quality of life.[10,11] There is much evidence that left ventricular (LV) diastolic dysfunction is associated with the pathophysiology of HFpEF and that LV diastolic dysfunction contributes importantly to exercise intolerance in HFpEF patients.[12–18] Furthermore, emerging evidence suggests that non-cardiac factors such as skeletal myopathy and vascular dysfunction also contribute to exercise intolerance in this patient group.[4,19–21]
The effect of exercise training on LV diastolic function in HFpEF has been examined in many randomised clinical trials (RCTs). The aim of this brief review is to summarise the RCTs examining the effects of exercise training on LV structure and function, as well as exercise capacity in HFpEF patients.
Pathophysiology of Heart Failure with Preserved Ejection Fraction
HF is defined as the pathological state in which the heart is unable to pump blood at a rate required by the metabolising tissues or can do so only with an elevated filling pressure. Inability of the heart to pump blood sufficiently to meet the needs of the body’s tissues may be due to the inability of the LV to fill (diastolic performance) and/or eject (systolic performance). When the HF is associated with a reduced ejection fraction (EF), the pathological state is called HF with reduced EF (HFrEF). In contrast, when the heart failure occurs in the absence of a reduced EF, the pathological state is called HFpEF.[22] HFrEF and HFpEF have several similarities in LV structural and functional characteristics, including increased LV mass and increased LV end-diastolic pressure. The clearest difference between the two forms of HF is the difference in LV geometry and LV function; HFrEF is characterised by LV dilatation, eccentric LV hypertrophy and abnormal systolic and diastolic function, whereas HFpEF is characterised by concentric LV hypertrophy, a normal or near-normal EF and abnormal diastolic function.[23] Exercise capacity is similarly impaired in HFrEF and HFpEF.[24]
Limited exercise tolerance because of fatigue and dyspnoea is a major symptom and a cause of disability in HFpEF. It results from abnormal central haemodynamics and peripheral non-cardiac factors. Abnormal central haemodynamics includes the inability to maintain (or augment) LV stroke volume adequately or maintaining (or augmenting) LV stroke volume at the expense of exaggerated increase in LV filling pressure during exercise.[25] In addition, limited increase in HR during exercise (chronotropic incompetence) also contributes to limited increase in cardiac output. Peripheral non-cardiac factors contributing to exercise intolerance include impaired vascular function and alterations in the skeletal muscle. In HFpEF patients, arterial stiffness is increased and endothelial function is impaired, both of which contribute to the exercise intolerance.[19] Furthermore, recent studies have shown that alterations in skeletal muscle, such as impaired microvascular function, reduced capillary density and mitochondrial dysfunction, are important contributors to exercise intolerance in HFpEF patients.[4,20,21]
Effect of Exercise Training in Heart Failure with Preserved Ejection Fraction
Many RCTs have reported the effect of exercise training on LV structure and function, as well as exercise capacity in HFpEF. Most of these trials used cycling and/or walking as the primary training modality (Table 1). Other physical training modalities included inspiratory muscle training and functional electrical stimulation of the lower limbs (Tables 2 and 3). Of note, most of the participants in these RCTs were taking standard medications for HF, such as angiotensin converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers and diuretics.
Table 1: Characteristics of Exercise Trials (Cycling or Walking) in HFpEF Patients.
| Study | N (Intervention/Control) | EF/NYHA Class | Session Time/Frequency/Intensity/Duration | Major Exclusion Criteria | Training Modality | Control | Outcomes (Cardiac Structure/Function) | Outcomes (Exercise Capacity) |
|---|---|---|---|---|---|---|---|---|
| Kitzman et al. 2010[27] | 26/27 | ≥50% II–III |
1 h/ Three times a week/~70% of HR reserve/16 weeks |
CAD, pulmonary disease, renal dysfunction (creatinine >2.5 mg/dl) | Walking/cycling | Attention control telephone call | E/A, DT, EF, LVEDV, LV mass | Peak VO2, 6MWD |
| Edelmann et al. 2011[43] | 46/21 | ≥50% II–III |
20–40 min/ Two to three times a week/~60% of peak VO2/24 weeks | Pulmonary disease, CAD, anaemia | Cycling + resistance training | Usual care (maintenance of usual activity level) | E/e’, e’, EF, LVEDV LV mass | Peak VO2, 6MWD |
| Smart et al. 2012[44] | 16/14 | >45% II–III |
30 min/ Three times a week/ ~70% of peak VO2/ 16 weeks | CAD | Cycling | Usual care (maintenance of usual activity level) | E/A, DT, E/e’, e’, EF | Peak VO2 |
| Alves et al. 2012[45] | 20/11 | >55% NR |
30 min/ Three times a week/ ~75% of maximal HR/ 24 weeks | ACS, uncontrolled metabolic disease | Treadmill/ cycling | Usual care | E/A, EF | NR |
| Kitzman et al. 2013[29] | 32/31 | ≥50% II–III |
1 h/ Three times a week/ ~70% of HR reserve/ 16 weeks | CAD, pulmonary disease, anaemia | Walking/ cycling | Attention control telephone call | E/A, DT, EF, LVEDV | Peak VO2, 6MWD |
| Kitzman et al. 2016[46] | 51/49 | ≥50% II–III |
1 h/ Three times a week/ ~70% of HR reserve/ 20 weeks | As above | Walking | Attention control telephone call or caloric restriction | E/A, E/e’, e’, EF, LVEDV, LV mass | Peak VO2, 6MWD |
| Fu et al. 2016[47] | 30/30 | ≥50% II–III |
30 min/ Three times a week/ 80% of peak VO2/ 12 weeks | AF, recent (<4 weeks) ACS or coronary revascularisation, COPD, renal dysfunction (eGFR <30 ml/min) | Cycling | Usual care | E/e’, EF | NR |
6MWD = 6-minute walk distance; ACS = acute coronary syndrome; CAD = coronary artery disease; COPD = chronic obstructive pulmonary disease; DT = E-wave deceleration time; eGFR = estimated glomerular filtration rate; E/A; the ratio of peak early to late diastolic mitral inflow velocities; E/e’ = the ratio of early diastolic mitral inflow to annular velocities; e’ = early diastolic mitral annular velocity; EDV = end-diastolic volume; EF = ejection fraction; HFpEF = heart failure with preserved EF; HR = heart rate; LV = left ventricular; NR = not reported; NYHA = New York Heart Association; peak VO2 = peak exercise oxygen uptake.
Table 2: Characteristics of Exercise Trials (Inspiratory Muscle Training) in HFpEF Patients.
| Study | Intervention/Control (n) | EF/NYHA Class | Major Exclusion Criteria | Session Time/Frequency/Duration | Control | Outcomes (Cardiac Structure/Function) | Outcomes (Exercise Capacity) |
|---|---|---|---|---|---|---|---|
| Palau et al. 2014[35] | 14/13 | >50% ≥II |
Recent (<3 months) ACS or cardiac surgery, pulmonary disease, smokers | 20 min/ twice a day/ 12 weeks | Usual care | E/e’, e’, EF, LV mass | Peak VO2, 6MWD |
| TRAINING-HF Trial. 2019[36] | 15/13 | >50% ≥II |
Recent (<3 months) ACS or cardiac surgery, COPD | 20 min/ twice a day/ 24 weeks | Usual care | E/e’, LA volume | Peak VO2 |
6MWD = 6-minute walk distance; ACS = acute coronary syndrome; COPD = chronic obstructive pulmonary disease; E/e’ = the ratio of early diastolic mitral inflow to annular velocities; e’ = early diastolic mitral annular velocity; EF = ejection fraction; HFpEF = heart failure with preserved EF; LA = left atrial; LV = left ventricular; NYHA = New York Heart Association; peak VO2 = peak exercise oxygen uptake.
Table 3: Characteristics of Exercise Trials (Functional Electrical Stimulation) in HFpEF Patients.
| Study | Intervention/Control (n) | EF/NYHA Class | Major Exclusion Criteria | Session Time/Frequency/Duration | Control | Outcomes (Cardiac Structure/Function) | Outcomes (Exercise Capacity) |
|---|---|---|---|---|---|---|---|
| Karavidas et al. 2013[39] | 15/15 | >50% II or III |
Recent (≤4 weeks) HF decompensation, ACS | 30 min/ 5 days a week/ 6 weeks | Sham stimulation | E/A, E/e’, LA volume | Peak VO2, 6MWD |
| TRAINING-HF 2019[36] | 15/13 | >50% ≥ II |
Recent (<3 months) ACS or cardiac surgery, COPD | 45 min/ 2 days a week/ 12 weeks | Usual care | E/e’, LA volume | Peak VO2 |
6MWD = 6-minute walk distance; ACS = acute coronary syndrome; COPD = chronic obstructive pulmonary disease; E/A; the ratio of peak early to late diastolic mitral inflow velocities; E/e’ = the ratio of early diastolic mitral inflow to annular velocities; e’ = early diastolic mitral annular velocity; EDV = end-diastolic volume; EF = ejection fraction; HFpEF = heart failure with preserved EF; LA = left atrial; LV = left ventricular; NYHA = New York Heart Association; peak VO2 = peak exercise oxygen uptake.
Cycling or Walking
The effect of cycling or walking on LV structure and function, as well as exercise capacity in HFpEF, has been examined in seven RCTs (Table 1). A meta-analysis of these RCTs has recently been reported.[26] In the meta-analysis, the ratio of peak early to late diastolic mitral inflow velocities (E/A), E-wave deceleration time, ratio of early diastolic mitral inflow to annular velocities (E/e), and early diastolic mitral annular velocity (e’) were extracted for the measures of LV diastolic function; peak exercise oxygen uptake (VO2) by expired gas analysis and 6-minute walk distance (6MWD) for the measures of exercise capacity; LV end-diastolic volume and LV mass for the measures of LV structure; and left ventricular ejection fraction (LVEF) for the measure of LV systolic function. HR reserve, which was defined as the difference between peak HR during exercise test and HR before exercise, was also extracted.
In the pooled analyses, cycling and/or walking did not significantly change LV diastolic function in HFpEF patients. There was no significant difference in changes of E/A (weighted mean difference [WMD] 0.030; 95% CI [−0.023–0.082]; I[2]=6.252%; p=0.266), E-wave deceleration time (WMD −2.040; 95% CI [−26.534–22.454] ms; I[2]=50%; p=0.870), or e’ (WMD 0.317; 95% CI [−0.952–1.587] cm/s; I[2]=87.523%; p=0.624) between exercise training and control groups. Similarly, the exercise training did not significantly change LV structure and systolic function. There was no significant difference in changes of LV end-diastolic volume (standardised mean difference [SMD] −0.034; 95% CI [−0.276–0.208]; I[2]=0%; p=0.784), LV mass (SMD 0.072; 95% CI [−0.205–0.350]; I[2]=0%; p=0.609) or LVEF (WMD 0.850; 95% CI [−0.128–1.828]; I[2]=0%; p=0.088) between the exercise training and control groups.
Despite the neutral effect on LV structure and function, cycling and/or walking improved exercise capacity in HFpEF patients. Exercise training significantly increased peak VO2 (WMD 1.660; 95% CI [0.973–2.348] ml/min/kg; I[2]=21%; p<0.001) and 6MWD (WMD 33.883; 95% CI [12.384–55.381] m; I[2]=0%; p<0.01) compared with the control group. Furthermore, exercise training increased HR reserve compared with the control group (WMD 7.521; 95% CI [1.797–13.246] bpm; I[2]=0%; p<0.05).
The meta-analysis clearly showed that exercise training improved exercise capacity without an improvement in LV structure or function in HFpEF patients.[26] To consider the possible mechanisms for these observations, it may be useful to look over the pathophysiological background of exercise intolerance in HFpEF patients. During exercise, oxygen consumption in the metabolising tissues increases dramatically. Normally, this is accomplished by an increase in cardiac output (a product of HR and stroke volume) and an increased use of oxygen by the metabolising tissues. Earlier studies have reported that, in HFpEF patients, stroke volume during exercise increases or is maintained at the expense of increased LV end-diastolic pressure due to diastolic abnormalities, resulting in exertional dyspnoea.[15–18] However, emerging data suggest that chronotropic incompetence, as well as peripheral non-cardiac factors, such as reduced oxygen delivery to exercising skeletal muscle and impaired oxygen use by active muscles during exercise, may play a relatively greater role in limiting exercise performance in HFpEF patients.[4,20,21]
Considering these points, the following mechanisms may underlie the improved exercise capacity with exercise training in HFpEF patients. In pooled analyses, exercise training improved HR reserve but not LV diastolic or systolic function.[26] Thus, improved chronotropic incompetence resulting from exercise training may contribute at least in part to improved exercise capacity in HFpEF patients. Furthermore, in an ancillary study of the included trial, exercise training increased use of oxygen by active muscles but not peak stroke volume during exercise.[27,28] Finally, another included trial reported that exercise training did not improve endothelial function or arterial stiffness, both of which are important determinants of exercise intolerance in HFpEF patients.[29] Taken together, in HFpEF patients, the improved exercise capacity with exercise training may result from improved chronotropic incompetence, as well as increased use of oxygen by active muscles (Figure 1).
Figure 1: Mechanisms of Improved Exercise Capacity With Exercise Training in HFpEF Patients.
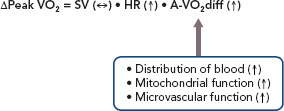
A-VO2 diff = arteriovenous-oxygen difference; HR = heart rate; HFpEF = heart failure with preserved ejection fraction; peak VO2 = peak exercise oxygen uptake; SV = stroke volume.
Although the mechanisms underlying increased use of oxygen by active muscles with exercise training remain elusive, several potential mechanisms have been proposed. First, improvement in skeletal muscle mitochondrial function with exercise training may be a significant contributor to increased use of oxygen in HFpEF patients. Multiple reports support that muscle mitochondrial function is impaired in HFpEF and is a strong factor for reduced use of oxygen.[4,20,21] In an animal model of HFpEF, exercise training prevented the impairment of mitochondrial function.[30] Second, exercise-induced upregulation of endothelial nitric oxide synthetase may increase bioavailability of nitric oxide, thereby improving vascular function and increasing distribution of blood to skeletal muscle.[31] Finally, exercise training may induce anti-inflammatory cytokines, thereby reducing metabolic inflammation and oxidative stress and improving microvascular circulation in skeletal muscle.[31]
The neutral effect of exercise training on LV structure and function should be interpreted with caution. First, exercise intervention period was relatively short (12–24 weeks; Table 1). Further studies are necessary to examine whether longer exercise intervention may favourably affect LV structure and function in HFpEF patients. Second, Doppler measurements of LV diastolic function at rest may be insufficient to detect subtle changes in diastolic function with exercise training. Because more sophisticated measurements of diastolic function, such as left atrial strain, have been developed, the effect of exercise training on the newly developed measurements merits further investigation. Finally, many HFpEF patients experience dyspnoea only during exertion. In these patients, LV filling pressure becomes markedly elevated during exercise. However, no included trials examined the effect of exercise training on LV function or LV filling pressure during exercise. Future trials should examine the effect of exercise training on LV function measures during exercise using exercise echocardiography.
Although minimal clinically important differences in exercise capacity in HFpEF patients have not been established, the reported improvements of 1.660 ml/min/kg in peak VO2 and 33.883 m in 6MWD with exercise training in the meta-analysis[26] appear to be clinically important based on the results of earlier studies. Specifically, a mean change of 15.9–55.2 m in 6MWD has been reported to be associated with a mild to moderate improvement in HF status in HFrEF patients.[32] Additionally, a meta-analysis of 22 RCTs with 3,826 HFrEF patients showed improvements of 1.85 ml/min/kg in peak VO2 and 47.9 m in 6MWD with exercise training.[33] Finally, even small increments in peak VO2 following exercise training have been reported to be associated with improved survival in patients with a wide range of cardiovascular diseases and healthy subjects.[34]
Recent studies have shown that up to one-third of patients fail to demonstrate a meaningful increase in peak VO2 in response to exercise training, despite adequate compliance to training.[34] Factors possibly influencing the response to exercise training are varied and are grouped as cardiac (systolic and diastolic function, chronotropic incompetence), non-cardiac (skeletal myopathy, vascular function, endothelial function, autonomic control), external (adherence, exercise dose and intensity) and comorbidities (obesity, anaemia, kidney diseases and pulmonary diseases). However, which factors predict the response to the training remains to be elucidated and warrants future investigation.
Inspiratory Muscle Training
The effect of inspiratory muscle training on LV structure and function, as well as exercise capacity in HFpEF, has been examined in two RCTs (Table 2).[35,36] Specifically, Palau et al. reported that 12-week inspiratory muscle training did not significantly change LV diastolic function; there was no significant difference in changes of e’ or E/e’ between the training and control groups. Similarly, the training did not significantly change LV systolic function or LV structure; there was no significant difference in changes of LVEF or LV mass between the training and control groups.
Despite the neutral effect on LV structure or function, inspiratory muscle training significantly increased peak VO2 (3.9 ml/min/kg; p<0.001) and 6MWD (67.4 m; p<0.001) compared with control group. Furthermore, the training reduced resting HR (−6 BPM; p<0.05) and increased peak exercise HR (5 BPM; p<0.01) compared with the control group, indicating that the training improved HR reserve.
Similar results have been reported recently by the same investigators.[36] In the Inspiratory Muscle Training and Functional Electrical Stimulation for Treatment of HFpEF (TRAINING-HF) trial, 12-week inspiratory muscle training did not change cardiac function or structure; there was no significant difference in changes of E/e’ or left atrial volume index between exercise training and control groups. Despite the neutral effect on cardiac structure or function, inspiratory muscle training improved peak VO2 (2.98 ml/min/kg; p<0.001) compared with the control group.
Patients with congestive HF reportedly have reduced maximal inspiratory pressure and endurance of inspiratory muscle, both of which contribute to the exercise intolerance.[37] Inspiratory muscle training may delay the development of diaphragmatic fatigue and increase ventilatory efficiency, resulting in an improvement in exercise capacity in HF patients.[38]
The reported improvement in peak VO2 with inspiratory muscle training is greater compared to that with cycling or walking.[35,36] However, there are no RCTs comparing the effect of inspiratory muscle training versus cycling or walking on exercise capacity in HFpEF patients. The comparative effectiveness of inspiratory muscle training and cycling or walking in HFpEF patients merits further investigation.
Functional Electrical Stimulation
The effect of functional electrical stimulation of the lower limbs on cardiac structure and function as well as exercise capacity in HFpEF has been examined in two RCTs (Table 3).[36,39] Specifically, Karavidas et al. reported that 6-week functional electrical stimulation did not significantly change cardiac function or structure; there was no significant difference in changes of E/A, E/e’ or left atrial volume between the stimulation and control groups.[39] Despite the neutral effect on cardiac structure or function, functional electrical stimulation improved 6MWD (52.8 m; p<0.01) compared with control group.
Similar results were reported in the TRAINING-HF trial.[36] Specifically, 12-week functional electrical stimulation did not significantly change cardiac function or structure; there was no significant difference in changes of E/e’ or left atrial volume index between the stimulation and control groups. Despite the neutral effect on cardiac structure or function, functional electrical stimulation improved peak VO2 (2.93 ml/min/kg; p<0.001) compared with the control group.
The possible mechanisms underlying the improved exercise capacity with the functional electrical stimulation in HFpEF patients include the improvement of endothelial function.[39] In HFpEF patients, endothelial function is impaired and is an important contributor to exercise intolerance.[29]
Perspectives
As described above, recent RCTs and meta-analyses of RCTs have shown that physical training such as cycling or walking, inspiratory muscle training and functional electrical stimulation can improve exercise capacity in HFpEF patients. Nevertheless, clinicians should take several points into consideration. First, although not specific to this topic, RCTs usually have strict enrolment criteria, and thus, the findings of the RCTs cannot be generalised to routine clinical practice. Specifically, in most of the trials listed in Tables 1–3, patients with cardiac and non-cardiac comorbidities such as AF, coronary artery disease, chronic obstructive pulmonary diseases, chronic kidney disease and anaemia were excluded. Importantly, registry studies have reported that HFpEF patients commonly have these co-morbidities.[40] Thus, the reported beneficial effect of physical training may not be extended into real-world HFpEF patients. Further studies are warranted to examine the effect of physical training on functional capacity in HFpEF patients with these co-morbidities. Second, the most effective type, intensity, frequency and duration of training are not determined in HFpEF patients. There are several on-going exercise trials in HFpEF patients that are expected to be published (Table 4). Specifically, the Exercise Training In Diastolic Heart Failure (Ex-DHF) trial is designed to investigate whether long-term (12-month) supervised exercise training can improve a clinically meaningful composite outcome score in HFpEF patients.[41] Components of the outcome score are all-cause mortality, hospitalisations, New York Heart Association functional class, global self-rated health, maximal exercise capacity and diastolic function after 6 and 12 months. The Optimizing Exercise Training In Prevention and Treatment of Diastolic Heart Failure (OptimEx-CLIN) trial aims to define the optimal dose of exercise training in patients with HFpEF.[42] Patients with stable symptomatic HFpEF will be randomised (1:1:1) to moderate intensity continuous training, high intensity interval training or a control group. The primary endpoint of the OptimEx-CLIN trial is peak VO2 after 3 months. The results of these trials may provide further insights into exercise prescriptions in HFpEF patients.
Table 4: Characteristics of On-going Exercise Trials in HFpEF Patients.
| Study | Intervention/Control (n) | Entry EF/ NYHA Class | Duration | Training Modality | Control | Primary Endpoint | Secondary Endpoint |
|---|---|---|---|---|---|---|---|
| Ex-DHF[41] | 160/160 | ≥50% II–III |
12 months | Endurance and resistance training | Usual care | Combined outcome score* after 6 and 12 months | Components of the primary endpoint, submaximal exercise capacity, echocardiographic parameters of LV geometry and dimensions, diastolic and systolic function, ventilatory efficacy, HRQoL and NT-proBNP after 6 and 12 months |
| OptimEx-CLIN[42] | 120/60 | >50% II–III |
12 months | Moderate intensity continuous training or high intensity interval training | Usual care | Peak VO2 after 3 months | Peak VO2 after 12 months Echocardiographic parameters of LV diastolic function, HRQoL, endothelial function and NT-proBNP after 3 and 12 months. |
*Components of the outcome score are all-cause mortality, hospitalisations, NYHA functional class, global self-rated health, maximal exercise capacity and diastolic function.
EF = ejection fraction; HFpEF = heart failure with preserved EF; HRQoL = health-related quality of life; LV = left ventricular; NT-proBNP = N-terminal pro b-type natriuretic peptide; NYHA = New York Heart Association; peak VO2 = peak exercise oxygen uptake.
Conclusion
In summary, available evidence suggests that physical training in addition to standard HF medication can provide clinically relevant improvements in exercise capacity without significant changes in LV function or structure in HFpEF patients. Further studies are necessary both to elucidate more exact mechanisms of exercise intolerance and to develop recommendations regarding the most effective training approach, including type, intensity, frequency and duration, in patients with HFpEF.
References
- 1.Vasan RS, Larson MG, Benjamin EJ et al. Congestive heart failure in subjects with normal versus reduced left ventricular ejection fraction: prevalence and mortality in a population-based cohort. J Am Coll Cardiol. 1999;33:1948–55. doi: 10.1016/s0735-1097(99)00118-7. [DOI] [PubMed] [Google Scholar]
- 2.Owan TE, Hodge DO, Herges RM et al. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med. 2006;355:251–9. doi: 10.1056/NEJMoa052256. [DOI] [PubMed] [Google Scholar]
- 3.Tsuchihashi-Makaya M, Hamaguchi S, Kinugawa S et al. Characteristics and outcomes of hospitalized patients with heart failure and reduced vs preserved ejection fraction. Report From the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). Circ J. 2009;73:1893–900. doi: 10.1253/circj.cj-09-0254. [DOI] [PubMed] [Google Scholar]
- 4.Pfeffer MA, Shah AM, Borlaug BA. Heart failure with preserved ejection fraction in perspective. Circ Res. 2019;124:1598–617. doi: 10.1161/CIRCRESAHA.119.313572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yusuf S, Pfeffer MA, Swedberg K et al. Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved Trial. Lancet. 2003;362:777–81. doi: 10.1016/S0140-6736(03)14285-7. [DOI] [PubMed] [Google Scholar]
- 6.Massie BM, Carson PE, McMurray JJ et al. Irbesartan in patients with heart failure and preserved ejection fraction. N Engl J Med. 2008;359:2456–2467. doi: 10.1056/NEJMoa0805450. [DOI] [PubMed] [Google Scholar]
- 7.Cleland JG, Tendera M, Adamus J et al. The perindopril in elderly people with chronic heart failure (PEP-CHF) study. Eur Heart J. 2006;27:2338–45. doi: 10.1093/eurheartj/ehl250. [DOI] [PubMed] [Google Scholar]
- 8.Yamamoto K, Origasa H, Hori M. Effects of carvedilol on heart failure with preserved ejection fraction: the Japanese Diastolic Heart Failure Study (J-DHF). Eur J Heart Fail. 2013;15:110–8. doi: 10.1093/eurjhf/hfs141. [DOI] [PubMed] [Google Scholar]
- 9.Pitt B, Pfeffer MA, Assmann SF et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383–92. doi: 10.1056/NEJMoa1313731. [DOI] [PubMed] [Google Scholar]
- 10.Kitzman DW, Little WC, Brubaker PH et al. Pathophysiological characterization of isolated diastolic heart failure in comparison to systolic heart failure. JAMA. 2002;288:2144–50. doi: 10.1001/jama.288.17.2144. [DOI] [PubMed] [Google Scholar]
- 11.Reddy YNV, Rikhi A, Obokata M et al. Quality of life in heart failure with preserved ejection fraction: importance of obesity, functional capacity, and physical inactivity. Eur J Heart Fail. 2020;22:1009–18. doi: 10.1002/ejhf.1788. [DOI] [PubMed] [Google Scholar]
- 12.Zile MR, Baicu CF, Gaasch WH. Diastolic heart failure--abnormalities in active relaxation and passive stiffness of the left ventricle. N Engl J Med. 2004;350:1953–9. doi: 10.1056/NEJMoa032566. [DOI] [PubMed] [Google Scholar]
- 13.Lam CS, Roger VL, Rodeheffer RJ et al. Cardiac structure and ventricular-vascular function in persons with heart failure and preserved ejection fraction from Olmsted County, Minnesota. Circulation. 2007;115:1982–90. doi: 10.1161/CIRCULATIONAHA.106.659763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zile MR, Gottdiener JS, Hetzel SJ et al. Prevalence and significance of alterations in cardiac structure and function in patients with heart failure and a preserved ejection fraction. Circulation. 2011;124:2491–501. doi: 10.1161/CIRCULATIONAHA.110.011031. [DOI] [PubMed] [Google Scholar]
- 15.Kitzman DW, Higginbotham MB, Cobb FR et al. Exercise intolerance in patients with heart failure and preserved left ventricular systolic function: failure of the Frank-Starling mechanism. J Am Coll Cardiol. 1991;17:1065–72. doi: 10.1016/0735-1097(91)90832-t. [DOI] [PubMed] [Google Scholar]
- 16.Zile MR, Kjellstrom B, Bennett T et al. Effects of exercise on left ventricular systolic and diastolic properties in patients with heart failure and a preserved ejection fraction versus heart failure and a reduced ejection fraction. Circ Heart Fail. 2013;6:508–16. doi: 10.1161/CIRCHEARTFAILURE.112.000216. [DOI] [PubMed] [Google Scholar]
- 17.Maeder MT, Thompson BR, Brunner-La Rocca HP et al. Hemodynamic basis of exercise limitation in patients with heart failure and normal ejection fraction. J Am Coll Cardiol. 2010;56:855–63. doi: 10.1016/j.jacc.2010.04.040. [DOI] [PubMed] [Google Scholar]
- 18.Abudiab MM, Redfield MM, Melenovsky V et al. Cardiac output response to exercise in relation to metabolic demand in heart failure with preserved ejection fraction. Eur J Heart Fail. 2013;15:776–85. doi: 10.1093/eurjhf/hft026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kitzman DW, Haykowsky MJ. Vascular dysfunction in heart failure with preserved ejection fraction. J Card Fail. 2016;22:12–6. doi: 10.1016/j.cardfail.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 20.Haykowsky MJ, Tomczak CR, Scott JM et al. Determinants of exercise intolerance in patients with heart failure and reduced or preserved ejection fraction. J Appl Physiol (1985) 2015;119:739–44. doi: 10.1152/japplphysiol.00049.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kitzman DW, Haykowsky MJ, Tomczak CR. Making the case for skeletal muscle myopathy and its contribution to exercise intolerance in heart failure with preserved ejection fraction. Circ Heart Fail. 2017;10:e004281. doi: 10.1161/CIRCHEARTFAILURE.117.004281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fukuta H, Little WC. Diagnosis of diastolic heart failure. Curr Cardiol Rep. 2007;9:224–8. doi: 10.1007/BF02938354. [DOI] [PubMed] [Google Scholar]
- 23.Fukuta H, Little WC. Contribution of systolic and diastolic abnormalities to heart failure with a normal and a reduced ejection fraction. Prog Cardiovasc Dis. 2007;49:229–40. doi: 10.1016/j.pcad.2006.08.009. [DOI] [PubMed] [Google Scholar]
- 24.Kitzman DW. Exercise intolerance. Prog Cardiovasc Dis. 2005;47:367–79. doi: 10.1016/j.pcad.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 25.Bonow RO, Udelson JE. Left ventricular diastolic dysfunction as a cause of congestive heart failure. Mechanisms and management. Ann Intern Med. 1992;117:502–10. doi: 10.7326/0003-4819-117-6-502. [DOI] [PubMed] [Google Scholar]
- 26.Fukuta H, Goto T, Wakami K et al. Effects of exercise training on cardiac function, exercise capacity, and quality of life in heart failure with preserved ejection fraction: a meta-analysis of randomized controlled trials. Heart Fail Rev. 2019;24:535–47. doi: 10.1007/s10741-019-09774-5. [DOI] [PubMed] [Google Scholar]
- 27.Kitzman DW, Brubaker PH, Morgan TM et al. Exercise training in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. Circ Heart Fail. 2010;3:659–67. doi: 10.1161/CIRCHEARTFAILURE.110.958785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Haykowsky MJ, Brubaker PH, Stewart KP et al. Effect of endurance training on the determinants of peak exercise oxygen consumption in elderly patients with stable compensated heart failure and preserved ejection fraction. J Am Coll Cardiol. 2012;60:120–8. doi: 10.1016/j.jacc.2012.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kitzman DW, Brubaker PH, Herrington DM et al. Effect of endurance exercise training on endothelial function and arterial stiffness in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. J Am Coll Cardiol. 2013;62:58492. doi: 10.1016/j.jacc.2013.04.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bowen TS, Rolim NP, Fischer T et al. Heart failure with preserved ejection fraction induces molecular, mitochondrial, histological, and functional alterations in rat respiratory and limb skeletal muscle. Eur J Heart Fail. 2015;17:263–72. doi: 10.1002/ejhf.239. [DOI] [PubMed] [Google Scholar]
- 31.Schiattarella GG, Rodolico D, Hill JA. Metabolic inflammation in heart failure with preserved ejection fraction. Cardiovasc Res. 2020;cvaa217 doi: 10.1093/cvr/cvaa217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Spertus J, Peterson E, Conard MW et al. Monitoring clinical changes in patients with heart failure: a comparison of methods. Am Heart J. 2005;150:707–15. doi: 10.1016/j.ahj.2004.12.010. [DOI] [PubMed] [Google Scholar]
- 33.van der Meer S, Zwerink M van BM et al. Effect of outpatient exercise training programmes in patients with chronic heart failure: a systematic review. Eur J Prev Cardiol. 2012;19:795–803. doi: 10.1177/1741826711410516. [DOI] [PubMed] [Google Scholar]
- 34.Gevaert AB, Adams V, Bahls M et al. Towards a personalised approach in exercise-based cardiovascular rehabilitation: How can translational research help? A ‘call to action’ from the Section on Secondary Prevention and Cardiac Rehabilitation of the European Association of Preventive Cardiology. Eur J Prev Cardiol. 2020;27:1369–85. doi: 10.1177/2047487319877716. [DOI] [PubMed] [Google Scholar]
- 35.Palau P, Dominguez E, Nunez E et al. Effects of inspiratory muscle training in patients with heart failure with preserved ejection fraction. Eur J Prev Cardiol. 2014;21:1465–73. doi: 10.1177/2047487313498832. [DOI] [PubMed] [Google Scholar]
- 36.Palau P, Dominguez E, Lopez L et al. Inspiratory muscle training and functional electrical stimulation for treatment of heart failure with preserved ejection fraction: The TRAINING-HF Trial. Rev Esp Cardiol (Engl Ed) 2019;72:288–97. doi: 10.1016/j.rec.2018.01.010. [DOI] [PubMed] [Google Scholar]
- 37.Meyer FJ, Borst MM, Zugck C et al. Respiratory muscle dysfunction in congestive heart failure: clinical correlation and prognostic significance. Circulation. 2001;103:2153–8. doi: 10.1161/01.cir.103.17.2153. [DOI] [PubMed] [Google Scholar]
- 38.Stein R, Chiappa GR, Guths H et al. Inspiratory muscle training improves oxygen uptake efficiency slope in patients with chronic heart failure. J Cardiopulm Rehabil Prev. 2009;29:392–5. doi: 10.1097/HCR.0b013e3181b4cc41. [DOI] [PubMed] [Google Scholar]
- 39.Karavidas A, Driva M, Parissis JT et al. Functional electrical stimulation of peripheral muscles improves endothelial function and clinical and emotional status in heart failure patients with preserved left ventricular ejection fraction. Am Heart J. 2013;166:760–7. doi: 10.1016/j.ahj.2013.06.021. [DOI] [PubMed] [Google Scholar]
- 40.Vaduganathan M, Michel A, Hall K et al. Spectrum of epidemiological and clinical findings in patients with heart failure with preserved ejection fraction stratified by study design: a systematic review. Eur J Heart Fail. 2016;18:54–65. doi: 10.1002/ejhf.442. [DOI] [PubMed] [Google Scholar]
- 41.Edelmann F, Bobenko A, Gelbrich G et al. Exercise training in Diastolic Heart Failure (Ex-DHF): rationale and design of a multicentre, prospective, randomized, controlled, parallel group trial. Eur J Heart Fail. 2017;19:1067–74. doi: 10.1002/ejhf.862. [DOI] [PubMed] [Google Scholar]
- 42.Suchy C, Massen L, Rognmo O et al. Optimising exercise training in prevention and treatment of diastolic heart failure (OptimEx-CLIN): rationale and design of a prospective, randomised, controlled trial. Eur J Prev Cardiol. 2014;21:18–25. doi: 10.1177/2047487314552764. [DOI] [PubMed] [Google Scholar]
- 43.Edelmann F, Gelbrich G, Dungen HD et al. Exercise training improves exercise capacity and diastolic function in patients with heart failure with preserved ejection fraction: results of the Ex-DHF (Exercise training in Diastolic Heart Failure. pilot study. J Am Coll Cardiol. 2011;58:1780–91. doi: 10.1016/j.jacc.2011.06.054. [DOI] [PubMed] [Google Scholar]
- 44.Smart NA, Haluska B, Jeffriess L et al. Exercise training in heart failure with preserved systolic function: a randomized controlled trial of the effects on cardiac function and functional capacity. Congest Heart Fail. 2012;18:295–301. doi: 10.1111/j.1751-7133.2012.00295.x. [DOI] [PubMed] [Google Scholar]
- 45.Alves AJ, Ribeiro F, Goldhammer E et al. Exercise training improves diastolic function in heart failure patients. Med Sci Sports Exerc. 2012;44:776–85. doi: 10.1249/MSS.0b013e31823cd16a. [DOI] [PubMed] [Google Scholar]
- 46.Kitzman DW, Brubaker P, Morgan T et al. Effect of caloric restriction or aerobic exercise training on peak oxygen consumption and quality of life in obese older patients with heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2016;315:36–46. doi: 10.1001/jama.2015.17346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fu TC, Yang NI, Wang CH et al. Aerobic interval training elicits different hemodynamic adaptations between heart failure patients with preserved and reduced ejection fraction. Am J Phys Med Rehabil. 2016;95:15–27. doi: 10.1097/PHM.0000000000000312. [DOI] [PubMed] [Google Scholar]


