Abstract
Consumer, industrial, and commercial product usage is a source of exposure to potentially hazardous chemicals. In addition, cleaning agents, personal care products, coatings, and other volatile chemical products (VCPs), evaporate and react in the atmosphere producing secondary pollutants. Here, we show high air emissions from VCP usage (≥ 14 kg person−1 yr−1, at least 1.7× higher than current operational estimates) are supported by multiple estimation methods and constraints imposed by ambient levels of ozone, hydroxyl radical (OH) reactivity, and the organic component of fine particulate matter (PM2.5) in Pasadena, California. A near-field model, which estimates human chemical exposure during or in the vicinity of product use, indicates these high air emissions are consistent with organic product usage up to ~75 kg person−1 yr−1, and inhalation of consumer products could be a non-negligible exposure pathway. After constraining the PM2.5 yield to 5% by mass, VCPs produce ~41% of the photochemical organic PM2.5 (1.1 ± 0.3 μg m−3) and ~17% of maximum daily 8-hr average ozone (9 ± 2 ppb) in summer Los Angeles. Therefore, both toxicity and ambient criteria pollutant formation should be considered when organic substituents are developed for VCPs in pursuit of safer and sustainable products and cleaner air.
Volatile chemical products (VCPs), including cleaning agents, personal care products, coatings, pesticides, printing inks, and adhesives, contain complex mixtures of organic species that span a wide range of volatility and oxidation state. Evaporation of components, such as organic solvents, results in emissions of volatile organic compounds (VOCs), primarily in indoor environments 1. Direct exposure to these VOCs can result in adverse health effects including mucosal irritation, endocrine disruption, asthma, and non-specific symptoms such as headache 2–6. Furthermore, these VOCs escape into ambient air where they contribute to formation of criteria pollutants such as fine particulate matter (PM2.5) and ozone (O3) 1, imposing further risks to public health 7–9. Because of the dramatic reduction in tailpipe emissions in the United States and Europe, recent estimates suggest VCPs may be the largest source of urban VOC emissions 1. However, significant gaps remain in our understanding of the impacts of VCPs on air quality and human health.
Organic aerosol is a substantial component of ambient PM2.5 10, with a large fraction (typically >60%) formed in the atmosphere from secondary chemistry 11,12. Regional chemical transport models struggle to simulate PM2.5, specifically the organic component formed by photochemical activity (i.e., secondary organic aerosol (SOA)), in urban areas 13–18. Li, et al. 19 report high PM2.5 yields for some individual intermediate volatility compounds found in VCPs and consumer products mixtures. Siloxanes, widely used in consumer products and commonly observed in urban air 20, can have PM2.5 yields of 20–50% 21,22. Underestimated VCP emissions, including species that are semi-volatile to intermediate volatility 1,23 or highly oxygenated 24, are likely responsible for some of the missing organic PM2.5 in regional air quality models.
Given the potential contribution of VCPs to both near-field (nominally indoor, during or in the vicinity of product use 70) and far-field (ambient air) exposure pathways, an approach that integrates their resulting impacts is needed. While most direct exposure to VCPs for people 28 will be in the indoor environment, ecosystem and other environmental exposure (e.g. secondary pollutants) resulting from VCPs will be dominated by the far-field environment 27,29,30. Far-field exposure due to residential and commercial releases of VCPs and their resulting criteria pollutant formation are not considered in current chemical exposure estimates 28.
In this work, we show that a model developed for quantifying near-field chemical exposure due to consumer product use can be used to estimate ambient air emissions for far-field air quality modeling. Implementing higher ambient chemical emission estimates in a regional-scale chemical transport model results in organic PM2.5 and ozone predictions that are more consistent with ambient measurements. These simulations demonstrate that VCPs are significant precursors to criteria pollutants and that ambient air measurements can be used to constrain near-field chemical exposure models.
Results
Estimating the implications of VCP usage on air quality and public health requires information on (1) organic product usage (the amount of product used that is carbon-containing compounds), (2) emission to air (described by a population-normalized emission factor , see Methods), and (3) transformation pathways leading to secondary products. Existing near-field chemical exposure models (e.g., the High-throughput Stochastic Human Exposure and Dose Simulation, SHEDS-HT, hereafter called SHEDS 26) help constrain the first two items while far-field ambient chemical transport models (e.g., the Community Multiscale Air Quality Modeling System, or CMAQ) require the latter two as input. Thus, ambient air quality modeling, when evaluated against observed air concentrations, can improve the data, assumptions, and methods upon which near-field chemical exposure models rely.
Organic product usage and emissions to air
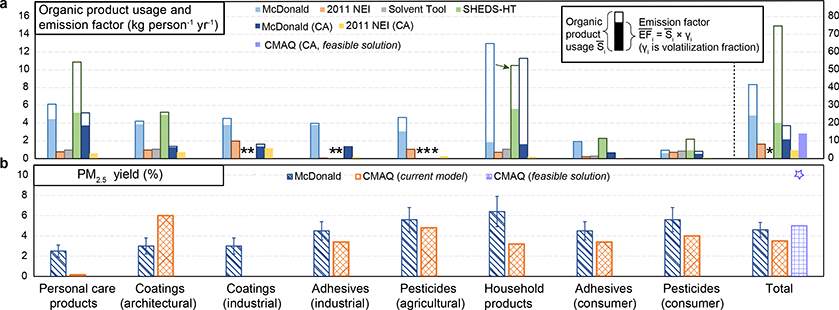
A first step in constraining the contribution of VCPs to air pollution is better quantification of their air emissions to the environment. Fig. 1a compares different ambient air emission estimates (filled bars, see also Supplementary Table 1, Supplementary Table 2 and Methods). In the United States, evaporation of chemicals from such organic products are estimated in the Solvent Sector as part of the EPA National Emission Inventory (NEI) and EPA Solvent Tool. Fig. 1a indicates that these operational NEI methods result in a factor of ~3–4 lower emissions of VCP organic gases per person than a posteriori estimates (see Methods) derived from the near-field SHEDS model 26 and values presented by McDonald et al. 1.
Fig. 1 |. Comparison of organic product usage, emission factors, and PM2.5 yields across different methodologies.
a-b, organic product usage per person (total height of the bars including unfilled portion) and values attenuated by washing down the drain (filled portion of the bars) (a), PM2.5 yields by mass organic gases reacted estimated by McDonald et al. 1 and upper bound estimates for prompt formation in the Community Multiscale Air Quality (CMAQ) Modeling System (b). In a, and values labeled CA indicate they are CA-specific, and otherwise are nation-wide estimates. for household products in SHEDS (one unfilled bar indicated with an arrow) and the total and values (the rightmost bar cluster) use the right y-axis; the asterisks indicate data are not available. Also note estimates are not shown for the CA-component of the 2011 NEI (only values are available for CA-component). In b, the PM2.5 yield of 5% by mass of organic gases was adopted in the CMAQ feasible solution; however, an PM2.5 yield of 10% (represented by star on far right) also resulted in PM2.5 SOA concentrations consistent with measurements.
Even though SHEDS and McDonald et al. use very different input data and approaches, the a posteriori SHEDS model and McDonald et al. arrive at approximately the same nation-wide average air emission factors of ~20 and ~24 kg person−1 yr−1, respectively. The SHEDS estimated for personal care are consistent with McDonald et al., i.e., 5.2 vs 4.5 kg person−1 yr−1 and both methods estimate consumer adhesive emissions at ~2 kg person−1 yr-1. However, SHEDS has the highest for household products, resulting in roughly similar amounts of organic gas emissions as personal care, while McDonald et al. indicates personal care emissions are the highest among VCP subsectors (2× higher than household cleaning). values for California, are generally lower than the nation-wide ones in both 2011 NEI (4.7 vs 8.2 kg person−1 yr−1) and McDonald et al. (10.8 vs 24.3 kg person−1 yr−1) estimates, likely reflecting differences in down-the-drain adjustments applicable to certain categories 31 and more stringent regulations for coatings and adhesives in California 32,33. Summing the per capita emission factors for all sectors available in SHEDS and multiplying by population results in estimates of total residential (excluding industrial and commercial) consumer product emissions of organic gases to air of 1.4 Tg yr−1 in the 2011 NEI, compared to 6.1 Tg yr−1 (~4×) in SHEDS and 3.9 Tg yr−1 (~3×) in McDonald et al. for the United States. In addition to these numbers suggesting the NEI is low on a nation-wide basis, later, we will show an increase in the California component of the NEI to effectively 14 kg person−1 yr−1 (CMAQ feasible solution; the purple filled bar on right in Fig. 1a), also results in improved estimates of criteria pollutants and other air quality endpoints in California, as predicted by a regional-scale chemical transport model.
Differences in organic product usage drive the differences in high vs low emission estimates. The Solvent Tool and EPA NEI methodology assume complete volatilization and air release of organic components in products () yet have lower values than other methods that assume only a fraction of the organic product evaporates (Fig. 1a). This means lower organic product usage (total height of bars in Fig. 1a) in the EPA national-level methodology results in lower values than SHEDS and McDonald et al. These discrepancies of organic product usage are particularly large for sectors such as personal care, household products, and consumer adhesives with SHEDS and McDonald et al. indicating ~10–20× higher organic product usage than NEI methods. Total organic product usage for the VCP sector is estimated at ~75, ~42, and ~8 kg person−1 yr−1 in SHEDS, McDonald et al., and 2011 NEI methodology respectively. These differences indicate constraining organic product usage is of high priority for creating robust emission estimates.
The SHEDS and McDonald et al. approaches result in high air emissions, and volatilization assumptions significantly modulate the magnitude of emissions. In this work, organic species in SHEDS were volatilized into air based on species vapor pressure and nature of application, resulting in an effective volatilization fraction (γ, see Methods) of 27% in a posteriori estimates (Supplementary Table 3). For household products, a large fraction of the organic species (~86% in McDonald et al. and ~89% in SHEDS) were predicted to go down the drain, consistent with the types of product applications in that category, such as laundry detergent and cleaning products. SHEDS (a posteriori) indicates half of the organic compounds in personal care products are ultimately emitted to air. This volatilization fraction is lower for SHEDS (γ = 0.48), than McDonald et al. (γ = 0.73), reflecting different assumptions about the types of applications and/or composition of personal care products. In most other categories, the down-the-drain adjustment is less than a factor of 2 (i.e., 0.5 < γ < 1). Looking across all VCP categories available, ~58% and ~27% of the mass evaporated according to the McDonald et al. and SHEDS methodologies, respectively.
A posteriori SHEDS estimates show an increased importance of inhalation exposure vs dermal uptake and/or ingestion from consumer product usage compared to a priori estimates before the modifications described in Methods. The effective volatilization fraction (γ) was increased from 2% in the a priori SHEDS chemical exposure model to 27% a posteriori, by making more reasonable assumptions for chemical mass retained on surfaces and skin (see Methods). These results illustrate that volatilization assumptions, particularly for dermally applied personal care products, can significantly affect air release estimates. The factor of 10× increase in volatilization factor between a priori and a posteriori implies a factor of 10 increase in post-use inhalation exposure. Using Isaacs et al.26 as a baseline and factoring in the a posteriori volatilization, the inhalation pathway is now estimated to be responsible for 2% of the mean daily intake dose for volatile chemicals in the near-field environment, making it competitive with previous estimates of dermal (7% of dose) and object-to-mouth (3% of dose) but not hand-to-mouth (near 90% of dose) exposure 26. This estimate of the role of inhalation for near-field chemical exposure applies to consumers within residential buildings, and the relative importance of inhalation exposure could be higher for populations in other environments (e.g. commercial or industrial settings). Volatilization should be further explored to more consistently represent the importance of chemicals in both chemical exposure and ambient air quality applications.
Speciation and PM2.5 yields in a regional air quality model
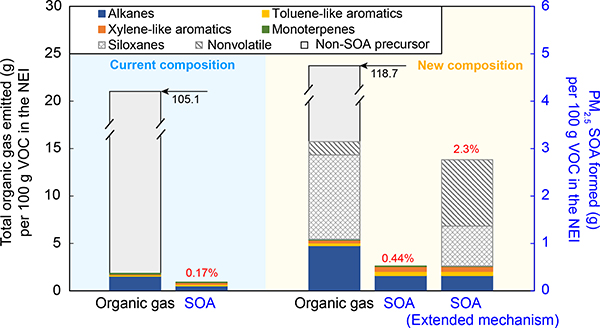
Since VOCs can be precursors for secondary fine particulate matter, another step in determining the role of VCPs in air pollution is to identify transformation pathways leading to secondary products. The predicted ambient air PM2.5 yield depends on the composition of the emissions and the yields of individual species. Individual species in regional chemical transport models like EPA’s CMAQ Modeling System are represented using simplified chemical mechanisms (see Methods) which typically use a reduced number of surrogates to represent VOCs. Existing simplified chemical mechanisms were not developed to provide a robust representation of VCP chemistry (to be illustrated in Fig. 2).
Fig. 2 |. Organic gas emissions and PM2.5 SOA formation potentials for personal care products based on current and new emission composition, respectively, using parameters in CMAQ.
The blue shaded portion indicates the current operational method was used to specify emission composition while updated information was used for the yellow panel. Emission values (in g) that do not correspond to the left y-axis are displayed in black text. PM2.5 yields by mass per organic gases are indicated with red figures.
The challenges associated with representing PM2.5 are illustrated in Fig. 1b, which shows the prompt (rapidly formed, within several hours) sector-specific PM2.5 yields based on a current model system that employs these surrogate methods (see also Supplementary Table 4–6). The PM2.5 yields for VCP sub-sectors in CMAQ vary by an order of magnitude (Supplementary Table 7), ranging from 0.17% for personal care products to as much as 16% for some industrial coatings, with an overall effective value of about 3.5% by mass in California (see Methods). The CMAQ overall PM2.5 yield for VCPs is slightly lower than the estimate from McDonald et al. (i.e., 4.6%). However, if the VCP sector is required to fully close the CMAQ model-measurement gap in PM2.5 SOA, then an even higher yield of 10% is required (discussed in the next section).
If the differences between the CMAQ and McDonald et al. (Fig. 1b) PM2.5 yield estimates are considered, in combination with the previously discussed emission magnitude discrepancies between SHEDS/ McDonald et al. and the NEI (Fig. 1a), then adhesive, pesticide, and personal care product usage account for the biggest potential underestimations in nationwide predictions of PM2.5 mass formation from VCPs at 75 Gg yr−1, 40 Gg yr−1, and 35 Gg yr−1 respectively (Supplementary Table 7). This total missing PM2.5 from these three sub-sectors (140 Gg yr−1) is comparable to SOA formation from mobile sources (~170 Gg yr−1) and accounts for 50% of VCP SOA (~300 Gg yr−1) 1. The potential underestimate in organic PM2.5 from pesticides and adhesives is driven almost entirely by the emissions magnitude, while the aerosol yield (15 × higher) causes the difference for personal care products. As a result, personal care is likely the category where PM2.5 underestimates are driven largely by an incorrect characterization of VOC emission composition across the entire ambient air quality modeling system.
Fig. 2 illustrates how the PM2.5 yield from personal care emissions is underestimated in CMAQ and how it can be improved. A small fraction of the emitted organic gases (~1.7%) are mapped to traditional PM2.5 SOA precursors including alkanes, aromatics, and monoterpenes. Default emission processing and mapping results in an effective PM2.5 yield of 0.17% by mass per organic gases. SOA from personal care products in CMAQ is mainly formed from alkane oxidation which contribute ~57% of the PM2.5, followed by aromatic oxidation (~38%).
Resolving issues associated with the chemical composition of consumer product emissions, especially compounds with lower volatility that are poorly represented in the current CMAQ mechanism model surrogates 37, can increase the PM2.5 yield for personal care products by a factor of 13 (from 0.17% to 2.3% by mass per organic gas, 5th bar in Fig. 2). Updating VOC speciation profiles based on more recent surveys of consumer products from 1997 to 2010 in California 51, results in a ~3× increase in amount of traditional SOA precursors.
The biggest cause of the underestimated PM2.5 SOA formation from personal care VCPs is the lack of surrogate species representing low-vapor-pressure species with high PM2.5 formation potential. For instance, n-heptadecane, a compound present in personal care products with PM2.5 yield of >30% 19, is mapped to an alkane surrogate which has low SOA formation (~3% by mass in CMAQ). Additionally, significant amounts of emitted organic gases are mapped to lumped species labeled as “non-reactive organic gases” and “nonvolatile” species (likely semi- or intermediate-volatility organic compounds considered ineffective ozone precursors) that are not considered in CMAQ regional model calculations of ozone or PM2.5. Accounting for contributions of “non-reactive” organic gases, e.g, siloxanes22,38, and treating the “nonvolatile” species as a precursor like n-pentadecane with a ~50% 39,40 (lower bound) or 100% PM2.5 yield (upper bound, Fig. 2) increases the PM2.5 yield estimated for personal care products by a factor of 4–5 (from 0.44% to as much as 2.3%), suggesting that chemical mechanisms need to be extended to include these non-traditional species.
Constraining emissions and PM2.5 yields with ambient observations
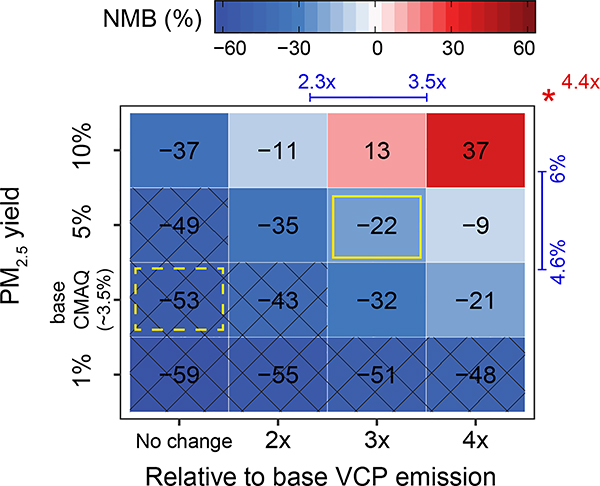
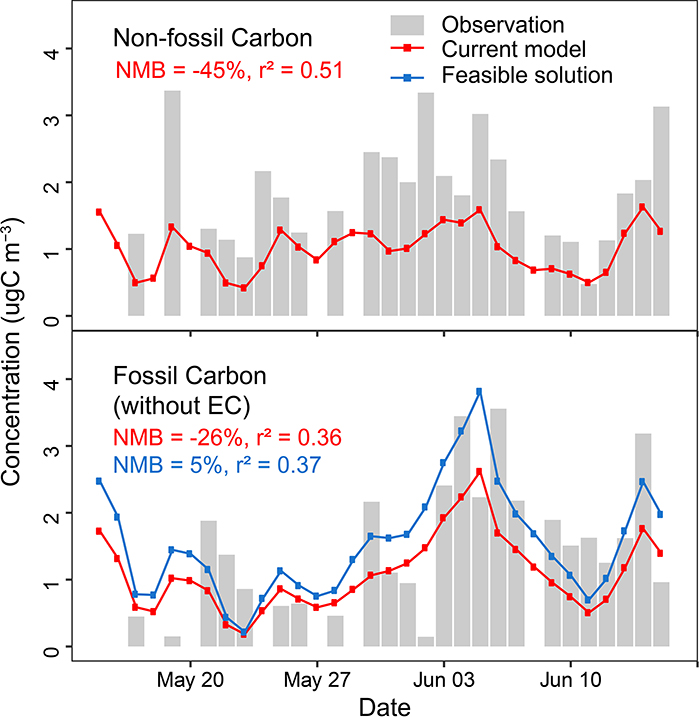
Because the SHEDS and McDonald et al. inventories suggest the 2011 NEI underrepresents VCPs, emission adjustments and PM2.5 yields were implemented in CMAQ to investigate what combinations of parameters improved predictions of ozone, PM2.5, and other metrics for the Los Angeles basin. CMAQ-simulated ambient PM2.5 SOA concentrations in Pasadena, California are driven by both the VCP emission magnitude and PM2.5 yield as reflected by the normalized mean bias (NMB) between measured and predicted SOA (Fig. 3). The current estimate of VCP emissions in the 2011 NEI (CA) and CMAQ model (i.e., VCP emissions of 4.7 kg person−1 yr−1 + effective PM2.5 yield of ~3.5%) results in an underestimation of photochemically-driven prompt SOA of 53% (SOAprompt = SOA with background contributions removed, see Methods). Using the existing SOA parameterization and simply increasing VCP emissions across all subsectors by factors of 2–4 increases SOA production and improves model performance. We consider the 14 kg person−1 yr−1 of VCP emissions (3× NEI (CA) emissions; 1.7× higher than the 2011 NEI nation-wide total estimate) and a 5% PM2.5 yield as a plausible far-field modeling representation of the VCP sector (feasible solution). Yields could be higher or lower in combination with lower or higher emissions, and the feasible solution still underestimates SOA by 22%. With 2011 NEI emissions, some bias remains even with a PM2.5 yield of 10% by mass from organic gases. Additional SOAprompt sources as well as background sources, likely of biogenic or non-fossil origin, may still be missing in CMAQ (Extended Data Fig. 1).
Fig. 3 |. Normalized mean biases (NMB) between CMAQ-predicted and observed prompt SOA concentration (SOAprompt) in Pasadena, California for different combinations of VCP emission magnitude (horizontal axis) and VCP PM2.5 yield (vertical axis) adjustments.
Cross hatching indicates parameters are inconsistent with observations (specifically, NMB ≥ uncertainty in measured OA, i.e., 38% 41). Recommended emissions (top) and PM2.5 yields (right) in McDonald el al. are marked by the range indicated in the margin. Recommended emission magnitude based on SHEDS (consumer emissions only) are indicated with the red asterisk (top). Two cases, the current model (dashed) and feasible solution (solid) are also indicated with yellow-outlined boxes. Base VCP emissions in the CA component of the 2011 NEI are 4.7 kg person−1 yr-1.
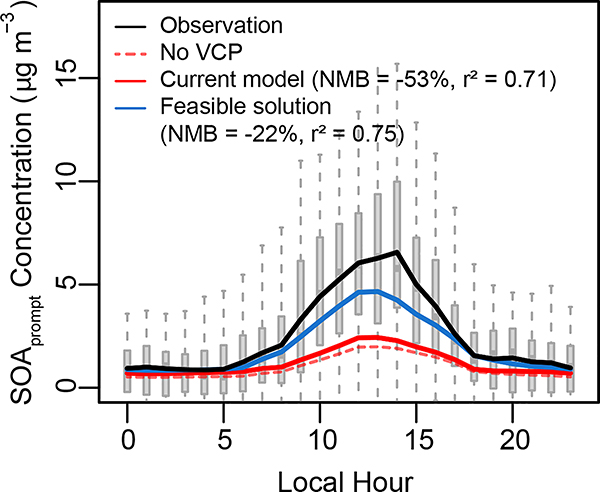
The feasible solution case, supported by the near-field SHEDS model emission estimates and updated VOC speciation profiles, demonstrates that VCPs likely play an important role in PM2.5 SOA formation. The 14 kg person−1 yr−1 VCP emissions with a PM2.5 yield of 5% per mass organic gas also substantially improve the predicted diurnal variation of prompt SOA with a peak concentration of 4.7 μg m−3 (vs 2.4 μg m−3 in the operational CMAQ model system) at Pasadena (Fig. 4). However, simulated SOA was still lower than the observed prompt SOA which reached a diurnally-averaged maximum of 6.6 μg m-3. The base CMAQ system estimated that VCPs contributed 0.2 μg m−3 PM2.5 SOA, compared to 1.0 μg m−3 from non-VCPs (Supplementary Table 8), which means VCPs are not a major source of SOAprompt (only ~20%) in current models. In the feasible solution case, VCPs are predicted to contribute 1.1 ± 0.3 μg m−3 PM2.5, making up 53% of simulated total SOAprompt (2.1 μg m−3) and ~41% of observed SOAprompt at Pasadena (2.6 μg m−3). The strong daytime increase in SOA requires higher PM2.5 yields regardless of emission magnitude (Extended Data Fig. 2).
Fig. 4 |. Diurnal variation of simulated and observed SOAprompt at Pasadena, California.
The simulation with VCP emissions zeroed out is indicated with dashed line. The grey boxes underneath indicate the 25th and 75th percentiles of the observations, with median values marked with horizontal lines in the boxes, and whiskers extending to the 10th and 90th percentiles. The solid lines indicate means of the observations and simulations.
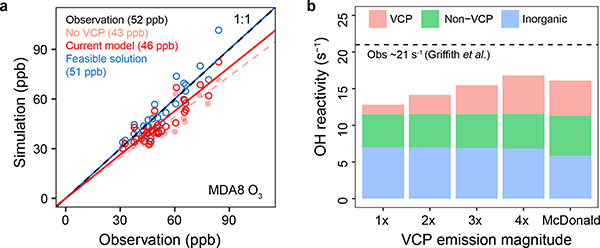
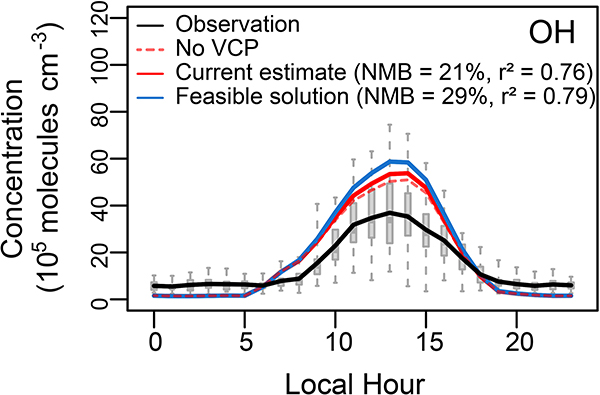
VCP usage results in significant amounts of organic gases being emitted into ambient air and subsequently impacts oxidant levels in addition to PM2.5. In the feasible solution case, VCPs account for ~3.7 ppb of organic gases in ambient air during the daytime (10:00 a.m. - 18:00 p.m.). Increased organic gas emissions from VCPs triggers modest increases in OH and O3 concentrations in the model (Extended Data Fig. 3 and Fig. 5a), which results in faster hydrocarbon reactions, and more SOA. Increased VCP emissions increase the high bias in simulated OH concentration (NMB of 29% vs 21% in the current model), but still within the uncertainty of the measurement (i.e., 36%) 42. Organic gas emissions from VCPs also contribute 9 ± 2 ppb to maximum daily 8-hr average (MDA8) O3 with the feasible solution (vs 3ppb in the current model; Fig. 5a). The higher VCP emissions improve model simulations of O3 compared to observations resulting in the feasible solution regression line overlapping the 1:1 line at Pasadena.
Fig. 5 |. Simulated MDA8 O3 and OH reactivity vs observations at Pasadena.
a-b, MDA8 O3 (a) and OH reactivity with different VCP emission magnitudes (b). In (a), the simulation with VCP emissions zeroed out is indicated with pink color, and averaged MDA8 O3 concentrations are given in the parentheses.
OH reactivity is a measure of how quickly OH is lost via reaction with organic gases and is thus useful for assessing uncertainties in VOC emissions and examining their importance to O3 and PM2.5 formation 63–66. OH reactivity was enhanced in the feasible solution case (15.5 s−1 vs 12.8 s−1 in current model), and was brought to within 30% of the observation (21 s−1, Griffith, et al. 42) with 3× NEI (CA) VCP emissions (Fig. 5b). OH reactivity contributed by VCPs (3.9 s−1 in the feasible solution) is higher than predicted in the default CMAQ modeling system but lower than 4.8 s−1 in McDonald, et al. OH reactivity of 4.5–4.7 s−1 is explained by organic gases from non-VCP sources (e.g., mobile sources, biogenics, etc). Additionally, inorganic species account for ~6.9 s−1 of OH reactivity, higher than the observation-based value (5.8 s−1) due to overestimated NO2 concentrations in the CMAQ simulations. Overall OH reactivity has better agreement with measurements with higher VCP emissions, but some missing OH reactivity in CMAQ remains to be addressed.
Integrating near-field and far-field exposure models
Near-field chemical exposure (e.g. SHEDS) and far-field air quality (e.g. CMAQ) models share common goals in terms of providing estimates of human (and ecosystem) exposure to pollutants. When connected through the CMAQ model, ambient air concentrations of organic aerosol, ozone, and OH reactivity provide top-down constraints on air emissions associated with VCP usage and suggest an emission factor of ~14 kg person−1 yr−1 in California (feasible solution). This value is consistent with a posteriori SHEDS calculations of air emissions of ~20 kg person−1 yr-1. SHEDS indicates this ambient air emission rate is associated with ~75 kg person−1 yr−1 of organic product usage, significantly higher than the current EPA national-level methodology indicating only 8.2 kg person−1 yr-1. In addition to using ambient air concentrations to constrain emissions and organic product usage in near-field modeling, our work suggests additional avenues to better estimate exposure across scales. For example, EPA has undertaken a major effort to curate hundreds of thousands of chemical structures via the CompTox Chemistry Dashboard 43; however, over 250 species in the ambient air emission speciation database used here (Methods) were not found in the Dashboard (as of September 9th, 2019), although chemical identifier curation is ongoing. Curation of species emitted in air in the Dashboard will allow for Quantitative Structure Activity Relationship (QSAR) models developed for chemical properties 44, toxicity 45, and exposure 28 to be applied and facilitate risk evaluations of these compounds. In addition, models like SHEDS can be used to create new speciation profiles for ambient air quality modeling or provide alternative emission factors for use in creating VOC inventories. Ultimately, connecting near-field chemical exposure and far-field air quality models that consider direct exposure to emitted chemicals as well as their oxidation products (e.g. PM2.5 and ozone) will result in improved tools for addressing public health questions.
Discussion
VCPs are ubiquitous in daily life for aesthetic, protective, health care, and other purposes. Because VCPs have the potential to directly expose people to harmful chemicals as well as escape to ambient air, their formulations have continuously evolved over decades in response to air quality and chemical safety drivers. Since the 1990s, VCPs have been targeted for control to reduce the formation of ozone as well as hazardous air pollutants (HAPs) in the United States via the Clean Air Act. This has led to an increase in the prevalence of water and other green solvents in VCPs. Product formulations will likely continue to evolve to improve performance, cost, and regulatory compliance via use of functional substitutes as chemicals with lower health risk and less potential for criteria pollutant formation are identified. However, designing product reformulations aimed at improving public health and the environment will require a wholistic perspective that accounts for multiple pathways of exposure to both the compounds themselves as well as their degradation products, which is not fully realized in current alternative assessment tools nor air quality management. For example, a narrow focus on ozone has led to compounds such as siloxanes being exempt from VOC regulation despite their high potential for SOA formation. Multiple options from solvent substitution to product capture and recycling should be employed to reduce the environmental and health impacts of VCPs.
Methods
In this work, air emissions of VCPs were estimated with multiple inventory methods and a near-field chemical exposure model. Impacts of VCP emissions on ambient criteria pollutants were evaluated via air quality modeling, taking uncertainties in emissions, coupled with model representation of pollutant formation into account (see Extended Data Fig. 4).
VCP emission estimates for ambient air
Usage of volatile chemical products results in evaporative emissions of organic gases to the atmosphere. Air emissions from VCP usage (E) can be estimated based on sector-specific emission factors (EFi) and activity data (Ai) for sector i:
| (1) |
Emissions can be estimated using different activity surrogates such as employment, population, or product use in a sector with appropriate emission factors (Supplementary Table 2). If population-based emission factors (EFs) are reported by the method (e.g. Solvent Tool, SHEDS), they are directly assigned to (i.e., population-normalized emission factors shown in Fig. 1a). If population-based EFs are not provided, values of (in units of kg person−1 yr−1) are computed using emission totals (E) divided by activity data (Ai) in the form of population (using 312 million as 2011 US population and 3.8 million for California). These values quantify how many kilograms of organic gas are released to ambient air per person each year. EFs may consider how organic product usage (Si) (mass per person per year for a population-based activity surrogate, i.e., ) is attenuated by washing down the drain (assumed here to be permanent sink of VOCs) or otherwise sequestered from air. This down-the-drain adjustment is represented by a volatilization factor (γi) which represents the fraction of the organic product (generally a solvent) released into ambient air (0 ≤ γi ≤ 1):
| (2) |
The work here is focused exclusively on the organic content of consumer products that can be released into the atmosphere. The water, electrolytes, and other inorganic components of consumer products are not explicitly discussed or included in Si, γi, or EFi. In this work, five different methods (see also Supplementary Table 2) for calculating VCP emission factors are compared in terms of total sector emissions and category-specific emissions. These methods include:
(1). The 2011 EPA National Emission Inventory (NEI)
The EPA NEI is compiled based on air emission estimates developed by state and local agencies and submitted to EPA, as required by the Air Emissions Reporting Requirements Rule 40 C.F.R. § 51.1008 (2019). In the cases where state or local agencies do not provide emission estimates, EPA provides them as a fallback 46. California VCP emission estimates in the NEI are contributed by the state agency using its own methodology (see section (3) below). 2011 EPA NEI estimates are national-level VOC emission totals aggregated up from state-level reports (including not only nonpoint- but also point- source emissions which are only available for industrial sectors such as industrial surface coating, graphic arts and degreasing, and may use methods from individual jurisdictions or EPA methodology). The EPA NEI Solvent sector emissions are used synonymously with VCP emissions in this work.
The fallback 2011 EPA NEI methodology is based on national-level estimates of solvent use from the Freedonia 2007 report 47 divided by national-level activity data such as population, employment etc., as well as some other sources, e.g. EPA Emissions Inventory Improvement Program (EIIP) technical report 48. Default EPA NEI methodology assumes complete volatilization of the organic components (γ = 1). The NEI inventories VOCs according to a strict definition based on what is likely to lead to ozone formation (40 CFR 51.100). Conversion between the inventoried VOC and total organic gases (which may include non-reactive or other species) is based on other information (e.g. SPECIATE, see below). The difference in regulatory VOC versus total organic gases is small (<30% effect) compared to the differences between the 2011 NEI/ Solvent Tool and other methods which are generally on the order of a factor of 3 (Supplementary Table 9, Fig. 1a).
(2). the Solvent Tool
The Solvent Tool version 1.7 uses the same methods as the 2011 NEI and has been distributed by the EPA since development of the 2014 NEI to provide a way for states to estimate emissions of VOCs from VCPs. EFs in the Solvent Tool v1.7 for some categories were updated from 2011 using the updated Freedonia 2013 report 49. The differences of EFs between the 2011 NEI and the Solvent Tool are negligible (Fig. 1a). EFs in the Solvent Tool apply to nonpoint emissions only 49.
(3). The California emission inventory
The California Air Resources Board (CARB) estimates both total organic gas and VOC emissions from VCPs using sales of products from consumer product surveys from product manufacturers and formulators, multiplied by weight fraction of each organic species (VOC) compound (i.e., VOC content of the products) according to the speciation data from the survey 31. The aggregated category (e.g. hair sprays, hand and body lotion) total organic gases (VOCs) is computed as a sum over each individual product organic gases (VOCs) total, followed by adjustment with volatilization fractions in certain categories (0 < γ < 1).
(4). The SHEDS-HT model
The SHEDS-HT model version 0.1.6 26 and its default inputs were used to estimate the residential release of VOCs from formulated consumer products. Ambient air-quality (e.g., CMAQ) models generally target the actual atmospheric mean state, while exposure models (e.g., SHEDS) are designed to inform risk-based prioritization of chemicals via conservative (upper-bound) exposure estimates for individual species 26,28. SHEDS uses consumer habits and practices (product prevalence in the marketplace, frequency of use, and mass per use) for 324 individual product categories, and predicts uses of these products for a population of simulated individuals representative of the U.S. in terms of age and gender (a standard population size of 10,000 was used here). Releases of individual chemicals were estimated by multiplying the mass of product used by median weight fractions from Material Safety Data Sheets (MSDS) obtained from EPA’s Chemical and Products Database (CPDat) 36 and by volatilization fractions (see below). Mean releases were estimated for all volatile chemicals for which weight fraction data were available. Organic gas emissions for general categories (Supplementary Table 10) were calculated by summing over all chemicals in the category. All volatilized product was assumed released into ambient air without considering air exchange rates or loss in the indoor compartment.
We use the terminology, a priori, to refer to SHEDS predictions using the standard configuration described by Isaacs et al.26. Two critical adjustments were made to standard SHEDS operations (i.e., a priori estimates) resulting in the a posteriori estimates shown in Fig. 1a: (1) product prevalence in the marketplace was adjusted to be more mass conservative across all VOCs and (2) volatilization (γ) was estimated as a function of species vapor pressure and type of product application. By default, SHEDS estimates exposure to individual compounds, and if a species is present in one product, the model will always choose a non-zero value for that species’ occurrence in that product type (100% chemical prevalence). For example, if only one shampoo contains siloxanes, that product is assumed to have 100% of the market share when siloxane exposure is being estimated. Similarly, if a different shampoo is the only one that contains polyethylene glycol, that shampoo will have 100% of the market share when polyethylene glycol exposure is being estimated. This assumption is made due to lack of true knowledge of (1) the fraction of products on the market containing the chemical and (2) the market share of those products. However, it can result in overcounting of mass when individual species are aggregated into a VOC or organic gas total. To remedy this, we opted to randomly sample product composition, even across products in CPDat in which a species was not present (zero). Species were then aggregated up to a total. Furthermore, while SHEDS applies down-the-drain assumptions for products applied to residential and body surfaces, chemicals applied dermally do not volatilize by default. Volatilization was parameterized based on type of usage (e.g., applied to surfaces vs used in an aqueous environment) and volatility of organic species (following McDonald et al. methodology, Supplementary Table 11) to obtain emissions to ambient air.
(5). Recent estimates in literature
McDonald, et al. 1 estimated total organic gas emissions from VCPs using annual sales of products from economic and commodity flow surveys, the organic content usage in each product based on energy and chemical production statistics, and the volatilization fraction of the organic content based on indoor air quality literature and emission control efficiency.
VCP emission processing in air quality models
The composition of VCP emissions (referred to as speciation) in air quality modeling is based on sector-specific speciation profiles from EPA’s SPECIATE database 50. This database provides information to convert inventoried VOCs to specific individual chemical species. SPECIATE 4.5 is the public version of the database in operational use at the time of this work, and consumer product composition is mainly based on the CARB 1997 consumer products survey. For SPECIATE 5.0 51 (released in summer 2019), EPA updated profiles for consumer products and architectural coating using CARB’s 2010 consumer products inventory products update, which is mainly based on the 2003 and 2006 CARB consumer product surveys. Current and new emission composition in Fig. 2 is based on SPECIATE4.5 and SPECIATE 5.0, respectively. Speciation profiles for emission sources such as consumer products and architectural coatings, derived from CARB profiles, include a number of species (e.g. bins) that are not defined in SPECIATE. Those were mapped to compounds or mixtures in SPECIATE 5.0 using Carter’s assignments 52 to avoid loss of mass. In addition, we updated the exempt status of VOCs relevant to VCPs in SPECIATE 5.0 so that the ratio of total organic gases to inventoried non-exempt VOCs is better characterized. Mappings of chemical composites to model-ready surrogates, which are chemical-mechanism-dependent, follow Carter 37,52.
CMAQ model configurations and inputs
The CMAQ model is an ambient air chemical transport model used for research and regulatory purposes. For example, it is used by state and local agencies to design State Implementation Plans (SIPs) and other air quality management strategies. In this work, the CMAQ model uses VCP emissions as input to predict their impact on ambient concentrations of ozone, SOA, and OH reactivity. CMAQ model version 5.3Beta (www.epa.gov/cmaq, https://github.com/USEPA/CMAQ) was used to perform simulations in California (Supplementary Figure 1) with horizontal resolution of 4 km and 35 vertical layers extending up to 50 mb. The simulation spanned the California Nexus of Air Quality and Climate Change (CalNex) campaign 53 (May 15th to June 15th 2010) with the prior 14 days as a spin-up. The Pasadena ground site was located 18 km northeast of downtown Los Angeles (LA) and impacted by emissions in the LA metropolitan area with prevailing southwesterly winds during the daytime (advection of 3–5 h) 15. SAPRC07tic chemical mechanism (see details in Pye et al. 54 and Xie et al. 55) was used to represent gas-phase chemistry, with AERO7 for aerosol chemistry 56–58. The meteorological inputs were provided by Weather Research and Forecasting (WRF) version 3.8.1, with model configurations identical to those in Lu et al. 60
Anthropogenic emissions used in CMAQ were based on the 2011 National Emissions Inventory (2011 NEI), with a 25% reduction of mobile NOx to achieve more consistency with observed NO2 at the AQS sites within the domain (Supplementary Figure 2). Biogenic emissions were calculated online using the Biogenic Emission Inventory (BEIS) model version 3.61, with the Biogenic Emission Landuse Database 4 (BELD4) and multiple data sources for vegetation representation including the US Forest Service Forest Inventory and Analysis (FIA) version 5.1 for 2002–2013 and US Department of Agriculture 2007 census of agriculture data 59.
Treatment of POA and SOA from non-VCPs in CMAQ
Direct emissions of organic PM2.5 (POA, primary organic aerosol) for area-wide anthropogenic emission sectors (gasoline vehicles, diesel vehicles, aircraft, cooking, biomass burning, dust, VCPs, and other) were characterized as semi-volatile with sector-specific volatility profiles for the mobile 60 and non-mobile 14 sources. Atmospheric processing of POA is the same as described in Murphy, et al. 56. A state-of-the-science PM2.5 SOA parameterization for mobile organic gases with intermediate volatility, in an effort to minimize uncertainty in model predictions of the mobile sector SOA, was implemented in CMAQv5.3 as in Lu, et al. 60. Other sources of SOA in the base model include oxidation of traditional VOCs including isoprene, monoterpenes, sesquiterpenes, aromatics, and alkanes. For computational efficiency, SOA from VOCs of primarily anthropogenic origin (benzene, toluene, xylene, polycyclic aromatic hydrocarbons, and alkanes) were parameterized in CMAQv5.3-AERO7 as described in Supplementary Table 4–5.
Treatment of SOA from VCPs in CMAQ
In addition to a base CMAQ v5.3 simulation, a series of simulations were performed (Supplementary Table 12), with VCP organic gas emission perturbations (1×, 2×, 3×, 4× relative to base VCP emissions in the 2011 NEI). Three levels of fixed PM2.5 yields (α in Reaction 1) including 1%, 5% and 10% by mass were explored in concert with the emission perturbations. Due to the diversity of VOCs emitted and created in the atmosphere (Goldstein and Galbally 61 estimated that 104-105 different species have been identified), lumped mechanisms (e.g. SAPRC07) are created to represent the major routes to ozone and PM2.5 formation with a reduced number of organic species (usually ~100 surrogates). SOA yields for VCP subsectors in Fig. 1b and Fig. 2 were calculated offline (i.e., not conducting CMAQ modeling) by mapping emission composition to SAPR07 surrogates with the surrogate-specific yields in CMAQ (Supplementary Table 6). Simulated prompt SOA with base CMAQ v5.3 was equivalent to a simulation with fixed PM2.5 yield of ~3.5%. Organic gases from VCPs participated in gas-phase chemistry leading to ozone formation and oxidant modifications through traditional SAPRC mechanism species (i.e., benzene, toluene, xylene, monoterpenes, etc.) as indicated by SPECIATE 4.5, while the SOA formation from VCPs was imposed by Reaction 1. In Reaction 1 all VCP organic gases were treated using a single surrogate TOGVCP so that a specific yield (α = 1%, 5% and 10%) could be implemented. The reaction rate constant (kOH) for VCP organic gases reacting with OH in Reaction 1 is identical to the averaged kOH for mobile organic gases with intermediate volatility in CMAQ 60, i.e., kOH = 2.18×10−11 cm3 molecule−1 s−1, which is slightly higher than the McDonald et al. estimate of 1.25×10−11 cm3 molecule−1 s−1 1.
| (Reaction 1) |
CMAQ ambient air quality model evaluation
The CMAQ and WRF models were extensively evaluated for the CalNex domain for multiple pollutants (e.g., ozone, total PM2.5 mass and constituents, PM2.5 precursor vapors, etc.) in previous work14, 16, 56, 60, 67–69. The evaluation in this study was designed to focus on rapidly formed photochemically-driven SOA (i.e., prompt SOA or SOAprompt), to isolate fresh SOA from VCPs in an urban atmosphere. Oxygenated organic aerosol (OOA), derived from factor analysis of organic aerosol data from the Aerodyne High-Resolution Time-of-Flight Aerosol Mass Spectrometer (AMS), is considered a measured proxy for SOA and can be further mathematically divided into two components: semi-volatile OOA (SV-OOA) and low-volatility OOA (LV-OOA) 12. Generally, SV-OOA shows strong diurnal variation (peaking at 2–3 p.m.) in Pasadena and is consistent with fresh SOA from urban areas, while LV-OOA, the more oxidized OOA, appears flat throughout the day and is expected to be aged SOA 62. Observed background OA was derived from the minimum in the LV-OOA diurnal circle 15 and is likely associated with biogenic VOC oxidation and transport. Here, the average of the lower 50% of CMAQ simulated SOA concentrations during 00:00 – 04:00 a.m. local time was used to estimate background OA in simulations. Prompt SOA, in both model predictions and observations, was derived by subtracting background OA from the overall SOA concentration. The background adjustment could lead to negative values of prompt OA on an hourly basis, which has minor effects on diurnal cycles or period averaged concentrations (Supplementary Figure 3). A metric Normalized Mean Bias (NMB) was used for the evaluation of simulated SOAprompt with CMAQ:
| (3) |
where Mi and Oi are modelled and observed value at time i, respectively, and N is the number of hourly samples.
OH reactivity from VCP emissions in the simulations was estimated using the concentration of the surrogate TOGVCP with single kOH (Reaction 1). Values calculated with this method compared well with values calculated using SAPRC07 speciation (Supplementary Figure 4). Measured total OH reactivity was based on observed decay of OH due to reactivity with ambient air in a turbulent flow tube reactor. 42
Data availability
The VCP emission estimates in the form of population-based emission factors are available within the supplementary information PDF file. The air quality modeling data for Pasadena during the 2010 CalNex campaign are available at https://doi.org/10.23719/1506136. The CalNex observations are publically accessible at https://www.esrl.noaa.gov/csl/groups/csl7/measurements/2010calnex/. The full CMAQ outputs that support the findings of this study are archived on EPA’s high -performance computing system and can be obtained from the corresponding author upon request.
Code availability
The source code of the operational CMAQ model is available at https://github.com/USEPA/CMAQ, with specific modifications applied in this work accessible at https://doi.org/10.23719/1506136. The code for simulation data processing is available upon request from the corresponding author. The source code of the SHEDS-HT model is available at https://github.com/HumanExposure/SHEDSHTRPackage. The modified SHEDS-HT including inputs and outputs from this study are available at https://github.com/HumanExposure/SHEDS_Applications/. The Solvent Tool is available at ftp://newftp.epa.gov/air/nei/2014/doc/2014v2_supportingdata/nonpoint/Solvent_Tool_v1.7.zip.
Extended Data
Extended Data Fig. 1.
Simulated non-fossil and fossil carbon in the current model and the feasible solution case at Pasadena compared to observations in Woody et al. 14
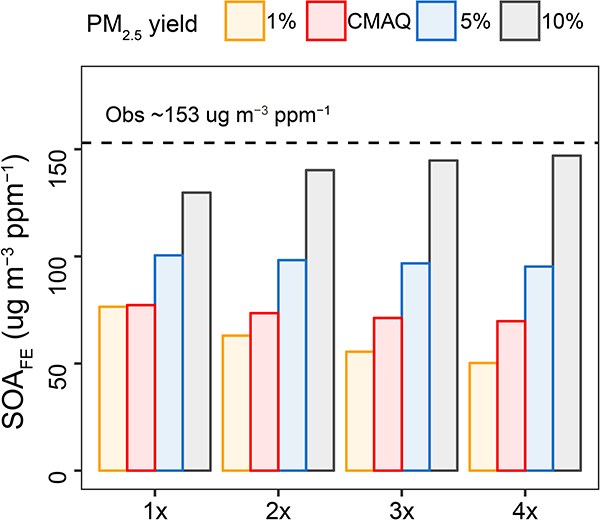
Extended Data Fig. 2. Simulated SOA formation efficiency (SOAFE) over 8:30 am – 12:30 pm at Pasadena.
SOAFE quantifies SOA mass formation per volume of organic gases reacted over a time window, which brings together the organic PM2.5 yield, precursor abundance, oxidant level, and reaction rate constant. See more details in Supplementary Notes.
Extended Data Fig. 3. Simulated OH in the current model estimate and the feasible solution case compared to observations at Pasadena.
The simulation without VCP emissions is indicated with the dashed line.
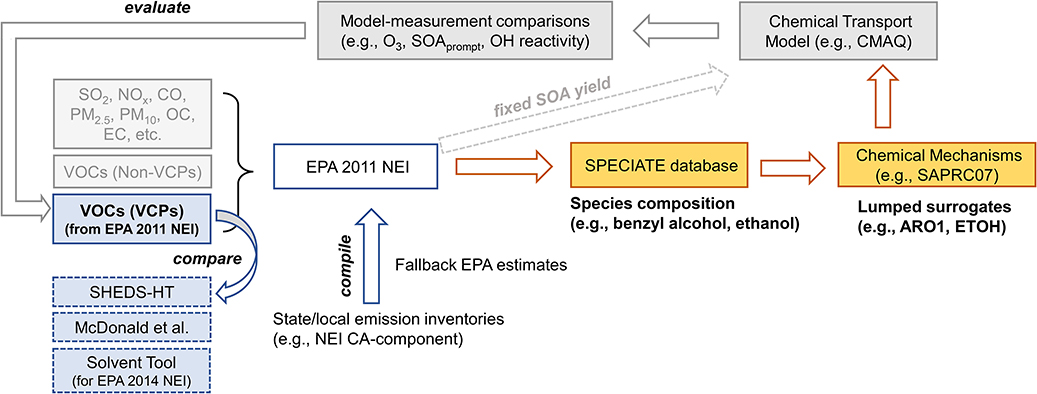
Extended Data Fig. 4. Schematic of the methodology.
This work integrated near-field (i.e., SHEDS-HT) with far-field (i.e., CMAQ modeling with 2011 NEI) modeling and top-down constraints based on ambient measurements. The blue boxes indicate emission estimates that were inter-compared. The emissions in EPA 2011 NEI, containing the estimate for VCP-emitted VOCs (solid outlined), went into air quality modeling. The yellow boxes indicate processing of the NEI including chemical speciation of emissions with the SPECIATE database, and mapping to CMAQ regional model surrogates using a certain chemical mechanism (e.g., SAPRC07). For VCPs, SOA formation was parametrized with a fixed SOA yield (large dashed arrow), and thus emission processing was not required for VCP-emitted VOCs. See more details in Methods.
Supplementary Material
Acknowledgement
We thank EPA’s SPECIATE workgroup, Karl Seltzer, Madeleine Strum, Jennifer Snyder, and Tesh Rao for useful discussion. This project was supported in part by an appointment to the Research Participation Program at the Office of Research and Development, U.S. Environmental Protection Agency, administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and EPA. BCM and SAM were supported by NOAA award NA17OAR4320101 to the University of Colorado Boulder, Cooperative Institute for Research in Environmental Sciences. ALR was supported by the Environmental Protection Agency STAR assistance agreement number RD83587301. The U.S. Environmental Protection Agency through its Office of Research and Development collaborated in the research described here. The research has been subjected to Agency administrative review and approved for publication but may not necessarily reflect official Agency policy. The views expressed in this article are those of the authors and do not necessarily represent the views or policies of the U.S. Environmental Protection Agency nor of the National Oceanic and Atmospheric Administration.
References
- 1.McDonald BC et al. Volatile chemical products emerging as largest petrochemical source of urban organic emissions. Science 359, 760–764, (2018). [DOI] [PubMed] [Google Scholar]
- 2.Trantallidi M et al. Ephect III: Health risk assessment of exposure to household consumer products. Sci. Total Environ 536, 903–913, (2015). [DOI] [PubMed] [Google Scholar]
- 3.Elberling J et al. Mucosal symptoms elicited by fragrance products in a population-based sample in relation to atopy and bronchial hyper-reactivity. Clin. Exp. Allergy 35, 75–81, (2005). [DOI] [PubMed] [Google Scholar]
- 4.Gabb HA & Blake C An informatics approach to evaluating combined chemical exposures from consumer products: A case study of asthma-associated chemicals and potential endocrine disruptors. Environ. Health Perspect 124, 1155–1165, (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dodson RE et al. Endocrine disruptors and asthma-associated chemicals in consumer products. Environ. Health Perspect 120, 935–943, (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brinke JT et al. Development of new volatile organic compound (VOC) exposure metrics and their relationship to “sick building syndrome” symptoms. Indoor air 8, 140–152, (1998). [Google Scholar]
- 7.Pope CA & Dockery DW Health effects of fine particulate air pollution: Lines that connect. J. Air Waste Manag. Assoc 56, 709–742, (2006). [DOI] [PubMed] [Google Scholar]
- 8.Schlesinger RB et al. The health relevance of ambient particulate matter characteristics: Coherence of toxicological and epidemiological inferences. Inhal. Toxicol 18, 95–125, (2006). [DOI] [PubMed] [Google Scholar]
- 9.Ebi Kristie L & McGregor G Climate change, tropospheric ozone and particulate matter, and health impacts. Environ. Health Perspect 116, 1449–1455, (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang Q et al. Ubiquity and dominance of oxygenated species in organic aerosols in anthropogenically‐influenced northern hemisphere midlatitudes. Geophys. Res. Lett 34, L13801, (2007). [Google Scholar]
- 11.Huang RJ et al. High secondary aerosol contribution to particulate pollution during haze events in china. Nature 514, 218, (2014). [DOI] [PubMed] [Google Scholar]
- 12.Jimenez JL et al. Evolution of organic aerosols in the atmosphere. Science 326, 1525–1529, (2009). [DOI] [PubMed] [Google Scholar]
- 13.Ensberg JJ et al. Emission factor ratios, SOA mass yields, and the impact of vehicular emissions on SOA formation. Atmos. Chem. Phys 14, 2383–2397, (2014). [Google Scholar]
- 14.Woody MC et al. Understanding sources of organic aerosol during CalNex-2010 using the CMAQ-VBS. Atmos. Chem. Phys 16, 4081–4100, (2016). [Google Scholar]
- 15.Hayes P et al. Modeling the formation and aging of secondary organic aerosols in Los Angeles during CalNex 2010. Atmos. Chem. Phys 15, 5773–5801, (2015). [Google Scholar]
- 16.Baker K et al. Gas and aerosol carbon in California: Comparison of measurements and model predictions in Pasadena and Bakersfield. Atmos. Chem. Phys 15, 5243–5258, (2015). [Google Scholar]
- 17.Hodzic A et al. Modeling organic aerosols in a megacity: potential contribution of semi-volatile and intermediate volatility primary organic compounds to secondary organic aerosol formation. Atmos. Chem. Phys 10, 5491–5514, (2010). [Google Scholar]
- 18.Shrivastava MK et al. Effects of gas particle partitioning and aging of primary emissions on urban and regional organic aerosol concentrations. J. Geophys. Res.-Atmos 113, D18301, (2008). [Google Scholar]
- 19.Li W et al. Potential of select intermediate-volatility organic compounds and consumer products for secondary organic aerosol and ozone formation under relevant urban conditions. Atmos. Environ 178, 109–117, (2018). [Google Scholar]
- 20.Coggon MM et al. Diurnal variability and emission pattern of decamethylcyclopentasiloxane (D5) from the application of personal care products in two north american cities. Environ. Sci. Technol 52, 5610–5618, (2018). [DOI] [PubMed] [Google Scholar]
- 21.Janechek NJ et al. Physical properties of secondary photochemical aerosol from OH oxidation of a cyclic siloxane. Atmos. Chem. Phys 19, 1649–1664, (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu Y & Johnston MV Aerosol formation from OH oxidation of the volatile cyclic methyl siloxane (CVMS) decamethylcyclopentasiloxane. Environ. Sci. Technol 51, 4445–4451, (2017). [DOI] [PubMed] [Google Scholar]
- 23.Zhao Y et al. Intermediate-volatility organic compounds: A large source of secondary organic aerosol. Environ. Sci. Technol 48, 13743–13750, (2014). [DOI] [PubMed] [Google Scholar]
- 24.Karl T et al. Urban flux measurements reveal a large pool of oxygenated volatile organic compound emissions. Proc. Natl. Acad. Sci, 1186–1191, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cohen Hubal EA et al. Advancing exposure characterization for chemical evaluation and risk assessment. J. Toxicol. Environ. Health, Part B 13, 299–313, (2010). [DOI] [PubMed] [Google Scholar]
- 26.Isaacs KK et al. SHEDS-HT: An integrated probabilistic exposure model for prioritizing exposures to chemicals with near-field and dietary sources. Environ. Sci. Technol. 48, 12750–12759, (2014). [DOI] [PubMed] [Google Scholar]
- 27.Wambaugh JF et al. High-throughput models for exposure-based chemical prioritization in the expocast project. Environ. Sci. Technol 47, 8479–8488, (2013). [DOI] [PubMed] [Google Scholar]
- 28.Ring CL et al. Consensus modeling of median chemical intake for the U.S. Population based on predictions of exposure pathways. Environ. Sci. Technol 53, 719–732, (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Barber MC, Isaacs KK & Tebes-Stevens C Developing and applying metamodels of high resolution process-based simulations for high throughput exposure assessment of organic chemicals in riverine ecosystems. Sci. Total Environ 605, 471–481, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nazaroff W, Weschler CJ, Little JC & Hubal EAC Intake to production ratio: A measure of exposure intimacy for manufactured chemicals. Environ. Health Perspect 120, 1678–1683, (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.California Air Resources Board (CARB). ARB solvent evaporation methodologies-aerosol coatings & consumer products. Preprint at https://ww3.arb.ca.gov/ei/areasrc/arbsolevapaercoatcp.htm (2000).
- 32.California Air Resources Board (CARB). Summary of architectural coating rules in California. Preprint at https://ww3.arb.ca.gov/coatings/arch/rules/voclimits.pdf (2016).
- 33.South Coast Air Quality Management District (SCAQMD). Rule 1168: Adhesive and sealant applications. Preprint at http://www.aqmd.gov/docs/default-source/rule-book/reg-xi/rule-1168.pdf (2005).
- 34.Isaacs K et al. Establishing a system of consumer product use categories to support rapid modeling of human exposure. J. Expo. Sci. Environ. Epidemiol (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.U.S. Environmental Protection Agency. Beta version of the Combined Human Exposure Model (CHEM) for Consumer Products. Available from: https://github.com/HumanExposure (2018).
- 36.Dionisio KL et al. The chemical and products database, a resource for exposure-relevant data on chemicals in consumer products. Sci. Data 5, 180125, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Carter WP Development of a database for chemical mechanism assignments for volatile organic emissions. J. Air Waste Manag. Assoc 65, 1171–1184, (2015). [DOI] [PubMed] [Google Scholar]
- 38.Wu Y & Johnston MV Molecular characterization of secondary aerosol from oxidation of cyclic methylsiloxanes. J. Am. Soc. Mass Spectrom 27, 402–409, (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jathar SH et al. Unspeciated organic emissions from combustion sources and their influence on the secondary organic aerosol budget in the United States. Proc. Natl. Acad. Sci, 10473–10478, (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Presto AA, Miracolo MA, Donahue NM & Robinson AL Secondary organic aerosol formation from high-NOx photo-oxidation of low volatility precursors: n-alkanes. Environ. Sci. Technol 44, 2029–2034, (2010). [DOI] [PubMed] [Google Scholar]
- 41.Bahreini R et al. Organic aerosol formation in urban and industrial plumes near houston and Dallas, Texas. J. Geophys. Res.-Atmos 114, D00F16, (2009). [Google Scholar]
- 42.Griffith SM et al. Measurements of hydroxyl and hydroperoxy radicals during CalNex-LA: model comparisons and radical budgets. J. Geophys. Res.-Atmos 121, 4211–4232, (2016). [Google Scholar]
- 43.Williams AJ et al. The Comptox Chemistry Dashboard: a community data resource for environmental chemistry. J. Cheminformatics 9, 61, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mansouri K, Grulke CM, Judson RS & Williams AJ Opera models for predicting physicochemical properties and environmental fate endpoints. J. Cheminformatics 10, 10, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.U.S. Environmental Protection Agency. Toxicity Estimation Software Tool (TEST). Available from: https://www.epa.gov/chemical-research/toxicity-estimation-software-tool-test (2016).
- 46.U.S. Environmental Protection Agency. 2011 National Emissions Inventory version 2 technical support document. Preprint at https://www.epa.gov/sites/production/files/2015-10/documents/nei2011v2_tsd_14aug2015.pdf (2015).
- 47.Freedonia. US industry forecasts for 2012 & 2017: Solvents. (2008).
- 48.U.S. Environmental Protection Agency. Air Emissions Inventory Improvement Program (EIIP) technical report series. Preprint at https://www.epa.gov/air-emissions-inventories/air-emissions-inventory-improvement-program-eiip (1997).
- 49.Abt Associates. Solvent utilization: documentation for EPA’s nonpoint emissions estimation tool. (2017).
- 50.Simon H et al. The development and uses of EPA’s SPECIATE database. Atmos. Pollut. Res 1, 196–206, (2010). [Google Scholar]
- 51.Abt Associates. Final report SPECIATE version 5.0 database development documentation. Preprint at https://www.epa.gov/sites/production/files/2019-07/documents/speciate_5.0.pdf (2019).
- 52.Carter WPL Development of an improved chemical speciation database for processing emissions of volatile organic compounds for air quality models. Preprint at https://intra.engr.ucr.edu/~carter/emitdb/ (2016).
- 53.Ryerson T et al. The 2010 California research at the nexus of air quality and climate change (CalNex) field study. J. Geophys. Res.-Atmos 118, 5830–5866, (2013). [Google Scholar]
- 54.Pye HOT et al. Modeling the current and future roles of particulate organic nitrates in the southeastern United States. Environ. Sci. Technol 49, 14195–14203, (2015). [DOI] [PubMed] [Google Scholar]
- 55.Xie Y et al. Understanding the impact of recent advances in isoprene photooxidation on simulations of regional air quality. Atmos. Chem. Phys 13, 8439–8455, (2013). [Google Scholar]
- 56.Murphy BN et al. Semivolatile POA and parameterized total combustion SOA in CMAQv5.2: Impacts on source strength and partitioning. Atmos. Chem. Phys 17, 11107–11133, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Pye HOT et al. On the implications of aerosol liquid water and phase separation for organic aerosol mass. Atmos. Chem. Phys 17, 343–369, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Xu L et al. Experimental and model estimates of the contributions from biogenic monoterpenes and sesquiterpenes to secondary organic aerosol in the southeastern United States. Atmos. Chem. Phys 18, 12613–12637, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bash JO, Baker KR & Beaver MR Evaluation of improved land use and canopy representation in beis v3.61 with biogenic VOC measurements in California. Geosci. Model Dev 9, 2191–2207, (2016). [Google Scholar]
- 60.Lu Q et al. Simulation of organic aerosol formation during the CalNex study: updated mobile emissions and secondary organic aerosol parameterization for intermediate-volatility organic compounds. Atmos. Chem. Phys 20, 4313–4332, (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Goldstein AH & Galbally IE Known and unexplored organic constituents in the Earth’s atmosphere. Environ. Sci. Technol 41, 1514–1521 (2007). [DOI] [PubMed] [Google Scholar]
- 62.Hayes P et al. Organic aerosol composition and sources in Pasadena, California, during the 2010 CalNex campaign. J. Geophys. Res.-Atmos 118, 9233–9257, (2013). [Google Scholar]
- 63.Di Carlo P et al. Missing OH reactivity in a forest: Evidence for unknown reactive biogenic VOCs, Science 304, 722–725, (2004). [DOI] [PubMed] [Google Scholar]
- 64.Sheehy PM, Volkamer R, Molina LT & Molina MJ Oxidative capacity of the Mexico City atmosphere - Part 2: A ROx radical cycling perspective. Atmos. Chem. Phys 10, 6993–7008, (2010). [Google Scholar]
- 65.Ren XR et al. OH and HO2 chemistry in the urban atmosphere of New York City, Atmos. Environ 37, 3639–3651, (2003). [Google Scholar]
- 66.Shirley TR, et al. Atmospheric oxidation in the Mexico City Metropolitan Area (MCMA) during April 2003, Atmos. Chem. Phys 6, 2753–2765, (2006). [Google Scholar]
- 67.Baker KR et al. Evaluation of surface and upper air fine scale WRF meteorological modeling of the May and June 2010 CalNex period in California. Atmos. Environ 80, 299–309, (2013). [Google Scholar]
- 68.Kelly JT et al. Fine-scale simulation of ammonium and nitrate over the South Coast Air Basin and San Joaquin Valley of California during CalNex-2010. J. Geophys. Res.-Atmos 119, 3600–3614, (2014). [Google Scholar]
- 69.Markovic M et al. Measurements and modeling of the inorganic chemical composition of fine particulate matter and associated precursor gases in California’s San Joaquin Valley during CalNex 2010. J. Geophys. Res.-Atmos 119, 6853–6866, (2014). [Google Scholar]
- 70.Fantke P et al. Coupled near-field and far-field exposure assessment framework for chemicals in consumer products. Environ. Int 94, 508–518, (2016). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The VCP emission estimates in the form of population-based emission factors are available within the supplementary information PDF file. The air quality modeling data for Pasadena during the 2010 CalNex campaign are available at https://doi.org/10.23719/1506136. The CalNex observations are publically accessible at https://www.esrl.noaa.gov/csl/groups/csl7/measurements/2010calnex/. The full CMAQ outputs that support the findings of this study are archived on EPA’s high -performance computing system and can be obtained from the corresponding author upon request.