Abstract
Objective
Fukuoka consensus guidelines classify pancreatic cystic lesions (PCLs) presumed to be intraductal papillary mucinous neoplasms (IPMNs) into Fukuoka positive (FP) (subgroups of high-risk (HR) and worrisome features (WFs)) and Fukuoka negative (FN) (non-HR feature/WF cysts). We retrospectively estimated 5-year risk of pancreatic cancer (PC) in FN, WF and HR cysts of patients with PCL-IPMN.
Design
From Mayo Clinic databases, we randomly selected 2000 patients reported to have a PCL; we excluded inflammatory or suspected non-IPMN cysts and those without imaging follow-up. We re-reviewed cross-sectional imaging and abstracted clinical and follow-up data on PCL-IPMNs. The study contained 802 patients with FN cysts and 358 with FP cysts.
Results
Patients with PCL-IPMN had median (IQR) follow-up of 4.2 (1.8–7.1) years. Among FN cysts, 5-year PC risk was low (2–3%) regardless of cyst size (p=0.67). After excluding events in the first 6 months, 5-year PC risk remained low (0–2%) regardless of cyst size (p=0.61). Among FP cysts, HR cysts (n=66) had greater 5-year PC risk than WF cysts (n=292) (49.7% vs 4.1%; p<0.001). In HR cysts, 3-year PC risk was greatest for obstructive jaundice versus enhancing solid component or main pancreatic duct >10 mm (79.8% vs 37.3% vs 39.4%, respectively; p=0.01).
Conclusions
Fukuoka guidelines accurately stratify PCL-IPMNs for PC risk, with FN cysts having lowest and HR cysts having greatest risk. After 6-month follow-up, WF and FN cysts had a low 5-year PC risk. Surveillance strategies should be tailored appropriately.
BACKGROUND
Pancreatic cyst lesions (PCLs) are routinely identified on imaging studies performed for unrelated reasons. Prevalence is estimated to range from 0.2% to 19.6% and increases with age.1-4 When a PCL is identified, a clinical and imaging assessment is made to determine the nature of the cyst. Many cysts are deemed to be most consistent with intraductal papillary mucinous neoplasms (IPMNs),5 which we call PCL-IPMNs. Two international consensus guidelines clarify the management and surveillance of PCL-IPMNs, the Sendai guidelines6 and, more recently, the Fukuoka guidelines,7 named for the Japanese cities where they were developed. The new American Gastroenterological Association guidelines recommend less intensive surveillance than the international consensus guidelines.8 However, all guidelines recommending surveillance or surgical resection are based on low-quality evidence.6,7
The Fukuoka guidelines classify PCL-IPMNs into Fukuoka positive (FP) (cyst subgroups with high-risk (HR) and worrisome features (WFs)) and Fukuoka negative (FN) (non-HR/WF cysts). HR cysts are recommended to undergo surgery because of high cancer prevalence; WFs cysts, endoscopic ultrasonography (EUS) for further risk stratification; and FN cysts, periodic surveillance at various intervals. Our goal was to determine whether this classification system of PCL-IPMNs provides meaningful risk stratification to inform about imaging surveillance of PCL-IPMNs. We retrospectively assembled a cohort of 2000 patients with cysts, of whom 1082 were deemed to have PCL-IPMNs. The cysts were further subclassified as FN or FP at baseline. In patients with FN cysts, we determined the 3-year and 5-year risk of FP features, pancreatic surgery for PCL and pancreatic cancer (PC). In FP cysts, we determined the 3-year and 5-year risk of pancreatic cyst surgery and PC. In addition, we sought to determine whether cyst diagnosis of suspected IPMNs had a greater risk of surgery or PC than the pancreatic cysts that undergo surveillance with the assumption that they are IPMNs or presumed IPMNs.
PATIENTS AND METHODS
Study design
Mayo Clinic Institutional Review Board approved the study. We assembled a retrospective cohort of patients with pancreatic cysts seen at Mayo Clinic in Rochester, Minnesota, between 1 January 2000 and 31 December 2012, who had provided consent for minimal-risk chart review studies. We identified patients from the electronic health records using International Classification of Diseases, ninth edition, codes 577.2 (cyst and pseudocyst of pancreas) and 211.6 (benign neoplasm of pancreas, except islets of Langerhans). A total of 6143 patients were identified. Simple random sampling was used to select a sample of 2000 patients. Study data were collected and managed with Research Electronic Data Capture electronic data capture tools hosted at Mayo Clinic in Rochester.9
Inclusion and exclusion criteria
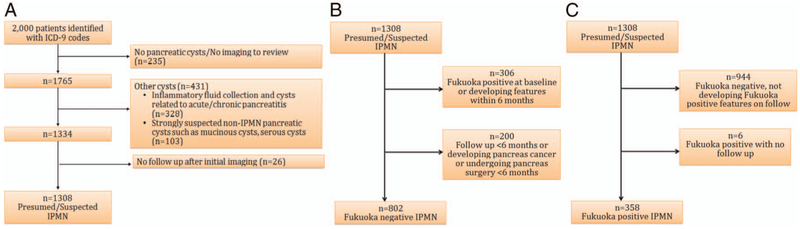
We excluded patients for the following reasons: no imaging to review or no pancreatic cysts (n=235); inflammatory fluid collections secondary to acute pancreatitis and pancreatic cysts associated with parenchymal calcification (n=328); strongly suspected non-IPMN pancreatic cysts based on imaging, such as mucinous cystic neoplasm (n=40); serous cystic neoplasm (SCN) (n=32); cystic pancreatic neuroendocrine tumour (n=4); solid pseudopapillary neoplasm (n=3); pancreatic adenocarcinoma with cystic degeneration (n=5); and pancreatic cysts associated with polycystic kidney disease or Von Hippel-Lindau disease (n=19). We also excluded patients who had no imaging or clinical follow-up after the initial study (n=26) (figure 1A). In addition, for the FN group, we excluded patients with PC or undergoing pancreas surgery (n=32) within 6 months of initial cyst diagnosis and patients with follow-up of <6 months (n=168) (figure 1B). The study contained 802 patients with FN cysts and 358 with FP cysts. Among 358 patients with FP cysts, 59 patients initially presented as FN cysts and developed FP features on follow-up.
Figure 1.
Flow charts of selection from suspected or presumed intraductal papillary mucinous neoplasm (IPMN). (A) Exclusion of non-IPMN cysts. (B) Selection of Fukuoka-negative cysts. (C) Selection of Fukuoka-positive cysts. ICD, International Classification of Diseases.
Study variables
We abstracted multiple demographic and clinical variables: age at cyst diagnosis, sex and race (white and Caucasian). We re-reviewed all imaging studies and EUS reports. Three investigators (SM, AA-K and NdP) abstracted the data. One investigator (SM) re-reviewed each imaging study and recorded the findings. These findings were then compared with the radiology report. When a discrepancy was found between the investigator’s findings and the radiology report or when data of some variables were not recorded in the radiology report, a senior GI radiologist (NT) was consulted and images were re-reviewed.
Data noted on imaging study variables included indication for initial imaging study, type of imaging study (CT, MRI, MR cholangiopancreatography or EUS) and date of imaging. For each scan, the total number of cysts was recorded. If a patient had <5 cysts on the imaging study, individual data for characteristics of each cyst were recorded; if the patient had >5 cysts, data on the largest cyst were recorded. The recorded cyst characteristics included location, maximum diameter, presence of septa, wall thickening, calcification, communication with the main pancreatic duct (MD) and a solid component with or without enhancement. Data on other, non-cyst variables that were abstracted included maximum MD diameter (head, body and tail), presence of MD irregularity, presence and dimension of mural nodule in MD, uncinate duct dilation, parenchymal atrophy, parenchymal calcifications and bile duct dilation. For patients who underwent EUS and fine-needle aspiration of the cyst, we recorded the cytological characteristics and carcinoembryonic antigen (CEA) level.
For patients who underwent pancreas surgery, we recorded whether the procedure was performed for PCL or other indication, date of surgery, type of surgery and final histopathological diagnosis. For patients with PC, we recorded whether the cancer developed at site of pancreatic cyst or away from pancreatic cyst. We noted the date of diagnosis of PC based on the date of histopathological diagnosis, when available. When no histopathological result was available, we selected the date on which imaging study was suggestive of PC. We also recorded the date of last follow-up in the clinic or through contact with the patient.
Cyst definitions
On the basis of initial imaging study, cysts were classified into the following categories: (1) suspected branch duct (BD) IPMN or a PCL >5 mm in diameter communicating with the MD and/or cyst fluid analysis with CEA >192 ng/mL and/or multifocal cysts, at least one of which clearly communicated with MD;7 (2) presumed BD-IPMN or PCL without clear communication with MD and/or CEA >192 ng/mL (when measured) that were clinically managed as BD-IPMNs;5 (3) suspected main duct (MD) IPMN or segmental or diffuse dilation of MD >5 mm without any obvious cause of pancreatic duct obstruction;7 (4) indeterminate BD-IPMN/SCN or a cluster of microcysts or macrocysts lacking communication with MD and no central scar and/or CEA level between 5 and 192 ng/mL; and (5) suspected mixed IPMNs with cysts meeting criteria for both MD-IPMN and BD-IPMN. Definitions of non-IPMN cysts are described in online supplementary material 1. Categories of IPMN (1–5) are PCLs managed as IPMN (PCL-IPMN) and are the subject of the present study.
Classification based on Fukuoka criteria
PCL-IPMNs were further classified as FP (including those with HR features or WFs) or FN (neither HR feature nor WF). HR cysts had one or more of the following: obstructive jaundice with cystic lesion of the pancreas head, enhancing solid component within cyst or MD >10 mm in head, body or tail. Cysts with WFs had one or more of the following: pancreatitis without another identifiable aetiological basis, cyst >3 cm, thickened or enhancing cyst walls, MD measuring 5–9 mm in head, body or tail, non-enhancing mural nodule, lymphadenopathy and an abrupt change in calibre of pancreatic duct with distal pancreatic atrophy FN cysts had neither HR features nor WFs.
Outcomes
Among PCL-IPMNs that were FP, we determined 3-year and 5-year risks of undergoing pancreatic cyst surgery or PC development. In PCL-IPMNs that were FN, we determined the 3-year and 5-year risk of FP features—HR or WF—undergoing pancreatic cyst surgery or PC development.
Outcome ascertainment
Outcomes were identified by chart review. Pancreas cyst surgery was ascertained by review of surgical reports of the included patients. Pancreas cancer was diagnosed if surgical histopathology was consistent with PC or imaging suggested malignancy and EUS-guided biopsy was positive for adenocarcinoma and/or CA-19-9 >300 U/mL.
Statistical analysis
Continuous patient characteristics and baseline cyst features are presented as median and IQR and differences between subgroups were assessed with the Kruskal-Wallis test. Categorical characteristics and features are presented as number and percentage and were compared through χ2 test or Fisher’s exact test when expected counts were small. Kaplan-Meier method was used to calculate the 3-year and 5-year survival rates both overall and by subgroup with the log-rank test being used to test for differences in survival rates between groups. The patients in the study were broken down into groups based on cyst size (<1 cm, 1 to <2 cm, 2 to <3 cm, 3+ cm), Fukuoka risk level (worrisome vs high-risk features) and presumed versus probable IPMN.
RESULTS
Of 2000 patients, 1101 fulfilled the study inclusion criteria, including 194 suspected IPMNs (146 (75.3%)) suspected BD-IPMNs, 18 (9.3%) suspected MD-IPMNs and 30 (15.5%) suspected mixed IPMNs) and 907 presumed IPMNs (70 (7.7%) indeterminate BD-IPMNs or SCNs and 837 (92.3%) presumed BD-IPMNs). Table 1 shows the baseline cyst characteristics between the FN and FP cysts.
Table 1.
Baseline demographic and cyst characteristics on initial imaging in Fukuoka-negative and Fukuoka-positive groups
| Characteristic | Fukuoka negative* (n=802) | Fukuoka positive* (n=299) | Total (N=1101) | p Value |
|---|---|---|---|---|
| Female sex | 477 (59.5) | 147 (49.2) | 624 (56.7) | 0.002 |
| Age, mean (SD), years | 65.8 (12.9) | 65.6 (14.3) | 65.7 (13.3) | 0.74 |
| Race | <0.001 | |||
| White | 737 (91.9) | 248 (83.5) | 985 (89.6) | |
| Caucasian | 28 (3.5) | 12 (4.1) | 40 (3.6) | |
| Unknown | 37 (4.6) | 37 (12.5) | 74 (6.7) | |
| Type of imaging | 0.03 | |||
| CT | 570 (71.1) | 205 (68.6) | 775 (70.4) | |
| MRI/MRCP | 79 (9.9) | 19 (6.4) | 98 (8.9) | |
| EUS | 153 (19.1) | 75 (25.1) | 228 (20.7) | |
| Indication for initial imaging study | <0.001 | |||
| Abdominal pain | 158 (19.7) | 64 (21.4) | 222 (20.2) | |
| Jaundice | 9 (1.1) | 8 (2.7) | 17 (1.5) | |
| Pancreatitis | 23 (2.9) | 32 (10.7) | 55 (5.0) | |
| Other | 592 (73.8) | 186 (62.2) | 778 (70.7) | |
| Unknown | 20 (2.5) | 9 (3.0) | 29 (2.6) | |
| Cyst diagnosis | <0.001 | |||
| Suspected branched duct—IPMN | 97 (12.1) | 49 (16.4) | 146 (13.3) | |
| Suspected main duct—IPMN | 0 (0.0) | 18 (6.0) | 18 (1.6) | |
| Suspected mixed—IPMN | 0 (0.0) | 30 (10.0) | 30 (2.7) | |
| Presumed branched duct—IPMN | 705 (87.9) | 202 (67.6) | 907 (82.4) | |
| No. of cysts | 0.04 | |||
| 1 | 558 (69.6) | 211 (70.6) | 769 (69.8) | |
| 2 | 108 (13.5) | 26 (8.7) | 134 (12.2) | |
| 3 | 35 (4.4) | 8 (2.7) | 43 (3.9) | |
| 4 | 6 (0.7) | 3 (1.0) | 9 (0.8) | |
| ≥5 | 95 (11.8) | 51 (17.1) | 146 (13.3) | |
| Location of cyst in pancreas† | <0.001 | |||
| Head | 298 (37.2) | 152 (53.0) | 450 (41.3) | |
| Neck | 88 (11.0) | 23 (8.0) | 111 (10.2) | |
| Body | 228 (28.4) | 49 (17.1) | 277 (25.4) | |
| Tail | 188 (23.4) | 63 (22.0) | 251 (23.0) | |
| Size of largest cyst (mm)† | <0.001 | |||
| Median (range) | 10 (0.0–29.0) | 28.3 (0.0–130.0) | 11 (0.0–130.0) | |
| Cyst size group† (cm) | <0.001 | |||
| <1 | 395 (49.3) | 9 (13.5) | 434 (39.8) | |
| 1 to <2 | 319 (39.8) | 55 (19.0) | 374 (34.3) | |
| 2 to <3 | 88 (11.0) | 57 (19.7) | 145 (13.3) | |
| ≥3 | 0 (0.0) | 138 (47.8) | 138 (12.6) | |
| Family history of pancreatic cancer | 0.13 | |||
| Yes | 34 (4.2) | 19 (6.4) | 53 (4.8) | |
| No | 753 (93.9) | 278 (93.0) | 1031 (93.6) | |
| Unknown | 15 (1.9) | 2 (0.7) | 17 (1.5) | |
| Follow-up, years, median (range) | 4.9 (0.5–16.5) | 2.2 (0.0–15.6) | 4.2 (0.0–16.5) | <0.001 |
Values are presented as number and percentage of patients unless specified otherwise.
Missing in the Fukuoka-positive group are location of cyst (n=12), size of largest cyst and cyst size group (n=10).
EUS, endoscopic ultrasonography; IPMN, intraductal papillary mucinous neoplasm; MRCP, MR cholangiopancreatography.
Fukuoka-negative cysts
Of 802 cysts that were FN at baseline, 395 cysts were <1 cm, 319 cysts were 1 to <2 cm and 88 cysts were 2 to <3 cm.
Risk of FP features according to cyst size
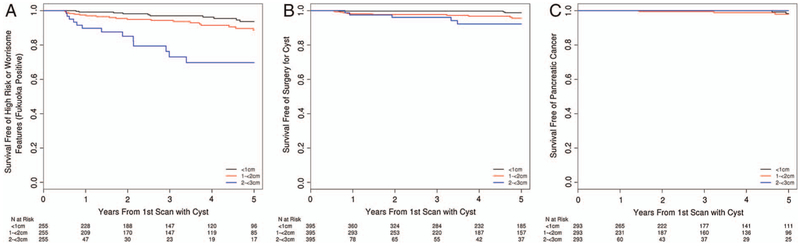
On 5-year follow-up, 42 of 802 FN cysts had FP features (4 (9.5%) HR features and 38 (90.5%) WFs). The median time to develop FP features was 1.9 years (range 0.5–5 years). The risk of FP features for cysts <1 cm, 1 to <2 cm and 2 to <3 cm was 3.00% (95% CI 0.58% to 5.36%), 5.68% (95% CI 2.42% to 8.78%) and 26.93% (95% CI 11.78% to 39.48%) at 3 years and 6.35% (95% CI 2.29% to 10.23%), 11.38% (95% CI 6.13% to 16.34%) and 30.25% (95% CI 14.01% to 43.42%) at 5 years, respectively. At 5 years, cysts between 2 and <3 cm had a higher risk of FP features than the other sizes (p<0.001) (figure 2A).
Figure 2.
Survival-free rates among patients with Fukuoka-negative cysts at baseline by cyst size. (A) Fukuoka-positive findings. (B) Pancreatic cyst surgery. (C) Pancreatic cancer.
Risk of undergoing pancreatic cyst surgery according to cyst size
On 5-year follow-up, 17 patients with FN cysts underwent pancreatic cyst surgery (<1 cm: 3, 1 to <2 cm: 11; and 2 to <3 cm: 5). The risk of undergoing pancreatic cyst surgery for cysts <1 cm, 1 to <2 cm and 2 to <3 cm was 0.26% (95% CI 0.00% to 0.76%), 2.30% (95% CI 0.60% to 3.97%) and 3.92% (95% CI 0.00% to 8.18%) at 3 years and 1.21% (95% CI 0.00% to 2.60%), 4.40% (95% CI 1.73% to 6.99%) and 7.77% (95% CI 0.82% to 14.22%) at 5 years. Cysts of 2 to <3 cm had a higher risk of undergoing pancreatic cyst surgery than smaller cysts (p<0.001) (figure 2B) (see online supplementary material 2).
Risk of PC according to cyst size
On 5-year follow-up, five patients with FN cysts developed PC (<1 cm: 2, 1 to <2 cm: 3; and 2 to <3 cm: 0). The risk of PC for cysts <1 cm, 1 to <2 cm and 2 to <3 cm was 0.00% (95% CI 0.00% to 0.00%), 0.49% (95% CI 0.00% to 1.43%) and 0.00% (95% CI 0.00% to 0.00%) at 3 years and 1.66% (95% CI 0.00% to 3.92%), 2.04% (95% CI 0.00% to 4.38%) and 0.00% (95% CI 0.00% to 0.00%) at 5 years. No difference was found in risk of PC according to cyst size between the three groups (p = 0.61) (figure 2C) (see online supplementary material 3).
Fukuoka-negative cysts (including those with pancreas cyst surgery and pancreas cancer within first 6 months)
Fukuoka-positive cysts
Among the 1101 PCL-IPMNs, 299 had FP features at baseline and 59 FN cysts had FP features on follow-up. Thus, 358 cysts had FP features, of which 66 (18.4%) had HR features and 292 (81.6%) had WFs (see online supplementary material 4). Among the HR cysts, 16 (24.2%) had MD dilation >10 mm; 40 (60.6%), enhancing solid component; and 10 (15.2%), obstructive jaundice with bile duct dilation secondary to cyst in the pancreas head. Among cysts with WFs, 18 (6.2%) had pancreatitis without any identifiable cause; 150 (52.3%), a cyst >3 cm; 49 (16.8%), thickened or enhancing cyst walls or a non-enhancing solid component in the cyst; 53 (18.2%), MD 5 to <10 mm in head, body or tail; 54 (18.5%), lymphadenopathy; 11 (3.8%), a non-enhancing mural nodule; and 7 (2.4%), an abrupt change in calibre of pancreatic duct with distal pancreatic atrophy.
Risk of undergoing pancreas surgery according to HR features or WFs
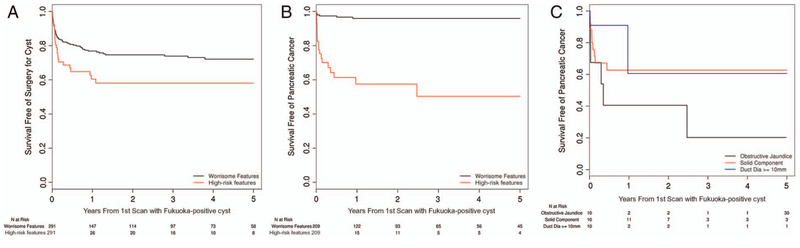
On 5-year follow-up, 89 FP cysts underwent pancreatic cyst surgery (WF 65 and HR 24). The 3-year and 5-year risks of undergoing pancreas surgery for FP cysts with HR features were 41.94% (95% CI 27.22% to 53.68%) and 41.94% (95% CI 27.22% to 53.68%); for those with WFs, the 3-year and 5-year risks were 26.12% (95% CI 20.19% to 31.61%) and 27.89% (95% CI 21.55% to 33.72%) (p=0.020) (figure 3A). Among patients with HR features at 3 years, a significant difference existed in the risk of pancreatic cyst surgery among those with enhancing solid component compared with MD >10 mm and with obstructive jaundice secondary to cyst in pancreas head (30.73% (95% CI 13.80% to 44.34%) vs 82.40% (95% CI 40.06% to 94.83%) vs 20.00% (95% CI 0.00% to 41.32%); p=0.036) (see online supplementary material 2).
Figure 3.
Survival-free rates among patients with Fukuoka-positive cysts by high-risk or worrisome features. (A) Pancreatic cyst surgery. (B) Pancreatic cancer. (C) Pancreatic cancer by high-risk feature subgroups of obstructive jaundice secondary to cystic lesion in head of pancreas, solid component in cyst and main pancreatic duct diameter >10 mm.
Risk of PC according to HR features or WFs
On 5-year follow-up, 25 FP cysts developed PC (WF 7 and HR 18). The 3-year and 5-year risks of PC in FP cysts with HR features were 49.68% (95% CI 26.27% to 65.65%) and 49.68% (95% CI 26.27% to 65.65%); for those with WFs, the 3-year and 5-year risks were 4.09% (95% CI 1.03% to 7.05%) and 4.09% (95% CI 1.03% to 7.05%) (p<0.001) (figure 3B). Among patients with HR features, a significant difference was found in the risk of PC at 3 years in patients with enhancing solid component compared with MD >10 mm and with obstructive jaundice secondary to cyst in the pancreas head (37.27% (95% CI 14.52% to 53.97%) vs 39.39% (0.00% to 73.35%) vs 79.75% (95% CI 0.00% to 96.01%); p=0.014) (figure 3C) (see online supplementary material 3).
Outcomes 6 months after development of FP features
Six months after development of FP features, 3 cysts with WF and 15 with HR underwent surgery. The 5-year risk of pancreatic cyst surgery was 10.48% (95% CI 0.00% to 21.03%) for HR and 10.68% (95% CI 5.22% to 15.38%) for WF (p=0.89) (see online supplementary figure S1A).
Six months after development of FP features, two cysts with WF and two with HR developed PC. The 5-year risk of PC was greater for HR than WF (17.97% (95% CI 0.00% to 38.67%) vs 1.49% (95% CI 0.00% to 3.52%); p=0.02) (see online supplementary figure S1B and supplementary material 5).
Suspected and presumed IPMNs
FN-suspected IPMNs had a higher risk of PC than FN-presumed IPMNs (p=0.04). FP-suspected IPMNs had a higher risk of undergoing surgery (p<0.001) and PC (p<0.001) than FP-presumed IPMNs (see online supplementary figure S2 and supplementary material 6).
Risk for surgery based on year of diagnosis of cyst
In FN cysts, the risk for undergoing pancreas cyst surgery was greater when cyst was diagnosed in earlier time period (2000–2005) compared with recent time period (2006–2012)—5.27% (95% CI 2.30% to 8.15%) vs 1.87% (95% CI 0.38% to 3.33%); p=0.027 (see online supplementary figure S3A). However, this was not observed in FP cysts—30.05% (95% CI 21.17% to 37.94%) vs 32.23% (95% CI 23.99% to 39.57%); p=0.73 (see online supplementary figure S3B).
DISCUSSION
This study provides insight into the natural history of pancreatic cysts that are monitored in clinical practice per IPMN guidelines. Currently, such cysts are risk-stratified by various Fukuoka features. We show that cysts without Fukuoka features, which we refer to as FN cysts, have a low 5-year risk of PC. By comparison, cysts with FP features have a high risk of PC cancer within the first 6 months of feature development, especially cysts with HR Fukuoka features. Subsequent risk decreases sharply in cysts without HR features. In addition, pancreatic cysts with higher likelihood of being IPMN, or suspected IPMN had a greater risk of undergoing cyst surgery and developing PC than the presumed IPMN category of cysts. Metachronous PC rarely developed in PCL-IPMNs in our cohort.
International consensus guidelines for management of IPMNs—the initial Sendai and the revised Fukuoka guidelines—have been widely followed. Few studies have investigated the surveillance recommendations proposed in the guidelines, which are largely based on expert opinion. We categorised cysts on the basis of Fukuoka recommendations and noted that the risk of HR stigmata or WF was low, especially for cysts <2 cm. It was significantly higher for cysts between 2 and <3 cm, largely driven by increase in size to >3 cm. Earlier studies have not specifically looked at Fukuoka positivity as an outcome because most of these studies predated the Fukuoka guidelines. However, similar outcomes have been investigated, such as increase in cyst size, dilation of MD or development of solid nodules. In a large study by Chung et al,10 7.6% of cysts <2 cm grew in size to >2 cm. In another study, 18% cysts ‘progressed’, defined as a cyst size increase of >1 cm, MD dilation of >2 mm, and appearance of mural nodules.11 In contrast, our study shows lower rates of progression in cysts <2 cm in size but shows comparable rates in those 2 to <3 cm.
The risk of undergoing pancreas surgery for cysts <1 cm, 1 to <2 cm and 2 to <3 cm was 1.4%, 4.3% and 7.6% at 5 years, respectively. Of patients who underwent surgery, 20% had high-grade dysplasia or invasive cancer on histopathological examination. A large study by Gaujoux et al11 showed similar findings, with 6.5% of cysts undergoing surgery on follow-up, of which 25% had invasive cancer. In addition, the study showed that cysts >2 cm had significantly greater risk of undergoing surgery than those <2 cm. The risk for surgery for FN cysts was higher in the time period preceding Sendai guidelines (2000–2005) compared with after Sendai guidelines (2006–2012), reflecting a change in practice pattern with a higher threshold to operate patients in the more recent time period.
Risk of PC was low in FN cysts: five cancers developed among a total of 813 cysts. Although there was no difference in risk of PC between different cyst size groups, our study was underpowered to detect any such difference given low rates of cancer in this population. Studies on conservatively monitored cysts estimate a risk of malignancy from 0% to 12% on conservative follow-up.10,13-24 These studies included a heterogeneous population of pancreatic cysts and represented both FN and FP IPMNs. Our results suggest a low risk of malignancy in FN BD-IPMNs. However, drawing conclusions regarding surveillance intervals is difficult because few patients had cancer. Development of PC away from PCL-IPMN was initially described in a Japanese study, where 8% BD-IPMN patients developed metachronous PC on follow-up.23 However, these observations were not replicated in studies from the USA and Europe,25-27 in which metachronous PC was observed in 0.1–1% of PCL-IPMNs. Although the absolute risk is quite low, it is nearly three times greater than age-matched and gender-matched controls.27 Our observations are similar, with only 3 of 1101 patients developing metachronous PC in our entire cohort.
Risk of malignancy is derived from surgical series, based on certain concerning features (HR and WF) identified on preoperative imaging. However, the actual risk of surgery or malignancy when these cysts are conservatively observed is unclear. The present study shows that cysts with HR stigmata had a greater 5-year risk of surgery (41.2% vs 28.8%) and an even greater risk of cancer (49.7% vs 5.1%) than those with WFs. A substantial number of operations in cysts with WFs likely reflect a low threshold in sending patients for surgery. Six months after diagnosis, the overall risk of surgery was much lower and was not different between the two groups (10.1% vs 10.9%). However, cysts with HR stigmata had greater risk of malignancy (18.0% vs 1.6%). Interestingly, no difference in risk of cancer occurred between cysts with WF and FN cysts. These findings are similar to some of the recent studies, which show 13–14% risk of cancer on conservative follow-up28,29 in FP cysts.
We further stratified cysts with HR stigmata and found that obstructive jaundice that developed secondary to cyst in the head of pancreas had a higher risk of cancer than cysts with solid component or MD dilation. However, we also observed that most of the cysts with MD dilation >10 mm underwent surgery compared with those that had obstructive jaundice. Patients with obstructive jaundice possibly had advanced malignancy and did not undergo surgery. Whether cysts with MD >10 mm have lower risk of cancer cannot be concluded because most of these cysts underwent surgery and whether they would have developed cancer in future is unclear.
One limitation of current guidelines is that recommendations for surveillance are based on the presumed histopathological diagnosis of the cysts. Since majority of cysts rarely undergo surgery, their histological subtype is never known and they are managed as presumed IPMNs.5,30 Demonstration of communication of cysts with MD, typical of IPMN, is not often possible on cross-sectional imaging or EUS. In our study, this communication with the MD was visible in about 15% of the BD-IPMNs, which is similar to a study by Lee et al,2 who found such communication in 13% of their MR imaging scans. In a recent meta-analysis conducted by the American Gastroenterological Association, studies discussing conservative follow-up of ‘suspected IPMNs’ had three times greater risk of invasive cancer than studies describing pancreatic cysts or ‘presumed IPMNs’ (0.72% vs 0.24%/year).8 In our study, we also found nearly three times higher risk of cancer for FP cysts in suspected versus presumed IPMN categories (26% vs 8%). Similarly, the risk of PC in FN cysts was greater for suspected versus presumed IPMN categories (6% vs 1%).
The present study has important implications. It validates the stratification proposed in Fukuoka guidelines and suggests that cysts with HR stigmata, especially with obstructive jaundice, have the greatest risk of PC, followed by those with WFs. The study provides valuable prognostic information for patients presenting in clinic with these cysts. Often, these patients are elderly with multiple comorbidities and therefore have a high risk of surgical morbidity and death. Thus, the risk of surgical complications can be weighed against the chance of having PC and an informed decision can be made. Close observation is recommended for cysts within 6 months of presentation. If the cysts are stable for 6 months, then their risk of cancer is less and the cysts can be safely observed at less frequent intervals than currently proposed. The study also highlights a lower risk of cancer in cysts with WFs, and we suggest a higher threshold to recommend surgery for these patients. The smaller cysts have a low risk of cancer, and the cost-effectiveness of surveillance needs to be determined. The study also shows that most of the cysts seen do not have classic features to subcategorise them into a histopathological subtype.
Earlier studies describing the natural history of pancreatic cysts had several limitations such as small number of cysts, lack of morphological description of cysts and cyst classification not per Fukuoka guidelines, and thus the outcomes were not assessed on the basis of Fukuoka categories. In the current study, we analysed a large cohort of patients seen at Mayo Clinic in Rochester, Minnesota, and included various cysts—from small subcentimeter cysts to large cysts with concerning features. The cysts were classified according to Fukuoka categories and the outcomes determined accordingly. Patients were randomly selected from health records and were not limited to those seen exclusively in the pancreas clinic. A study be Lee et al2 suggested that two of three pancreatic cysts are not reported by radiologists. Similarly in our study, we re-reviewed all scans of the patients and identified cysts in many old scans that were not reported. Thus, we could accurately determine the natural history of these pancreatic cysts. Our study was not limited to surgically resected pancreatic cysts and instead considered all patients with pancreatic cysts. It provides an estimated risk of surgery and PC of patients seen in the outpatient clinic because the majority of them do not have concerning features and can be counselled regarding the future risk of cancer.
Our study has several limitations. It was a retrospective study, and cysts were managed before Fukuoka guidelines were published. Hence, a lot of heterogeneity was present in the follow-up of these cysts. In addition, most cysts were presumed IPMNs. At our institution, an EUS-guided fine-needle aspiration of cyst contents, and biochemical and molecular characterisation of the aspirate is not routinely performed. Some of these cysts may have been benign and not IPMNs, but the study does reflect everyday practice. EUS reports were reviewed to abstract information. It is possible that some morphological features were not identified by endosonographer and, therefore, were considered absent in the study.
In conclusion, the results of our study show that Fukuoka guidelines allow risk stratification of cysts for PC. However, overall risk of PC is low in FN and FP cysts with WFs beyond 6-month follow-up. Identifying the small subset of cysts destined to develop PC continues to be a challenge. Surveillance strategies should be tailored appropriately.
Supplementary Material
Significance of this study.
What is already known on this subject?
Pancreatic cysts suspected to be branched duct intraductal papillary mucinous neoplasms can develop pancreatic cancer.
Various guidelines (Sendai, Fukuoka, AGA) have been proposed for management of these cysts supported by low-quality evidence.
Estimated risk for pancreatic cancer for cysts managed with imaging surveillance ranges from 0% to 12%.
What are the new findings?
Among Fukuoka-negative cysts, 5-year risk of pancreatic cancer is low (0–2%) regardless of cyst size.
Beyond 6 months of developing Fukuoka-positive features, risk of pancreatic cancer in cysts with worrisome features is not different from Fukuoka-negative cysts.
Cysts with high-risk stigmata (solid component, main duct dilation >10 mm or obstructive jaundice) have the highest risk of developing pancreatic cancer.
How might it impact on clinical practice in the foreseeable future?
This study validates the risk stratification proposed in Fukuoka guidelines and suggests that cysts with high-risk stigmata, especially with obstructive jaundice, have the greatest risk of pancreatic cancer, followed by those with worrisome features.
Fukuoka-negative cysts have a low risk of cancer, and the cost-effectiveness of surveillance needs to be determined.
Close observation is recommended for cysts within 6 months of presentation. If the cysts are stable for 6 months, then their risk of cancer is less and the cysts can be safely observed at less frequent intervals than currently proposed.
Acknowledgements
The authors acknowledge the revision and edits provided by scientific publications services at Mayo Clinic, Rochester, MN.
Funding This project was supported by CTSA Grant Number UL1TR000135 from the National Center for Advancing Translational Science (NCATS).
Footnotes
Disclaimer The contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health.
Competing interests None declared.
Ethics approval Institutional review board.
Provenance and peer review Not commissioned; externally peer reviewed.
REFERENCES
- 1.Kimura W, Nagai H, Kuroda A, et al. Analysis of small cystic lesions of the pancreas. Int J Pancreatol 1995;18:197–206. [DOI] [PubMed] [Google Scholar]
- 2.Lee KS, Sekhar A, Rofsky NM, et al. Prevalence of incidental pancreatic cysts in the adult population on MR imaging. Am J Gastroenterol 2010;105:2079–84. [DOI] [PubMed] [Google Scholar]
- 3.Zhang XM, Mitchell DG, Dohke M, et al. Pancreatic cysts: depiction on single-shot fast spin-echo MR images. Radiology 2002;223:547–53. [DOI] [PubMed] [Google Scholar]
- 4.Laffan TA, Horton KM, Klein AP, et al. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol 2008;191:802–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Farrell JJ, Fernández-del Castillo C. Pancreatic cystic neoplasms: management and unanswered questions. Gastroenterology 2013;144:1303–15. [DOI] [PubMed] [Google Scholar]
- 6.Tanaka M, Chari S, Adsay V, et al. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006;6:17–32. [DOI] [PubMed] [Google Scholar]
- 7.Tanaka M, Fernández-del Castillo C, Adsay V, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 2012;12:183–97. [DOI] [PubMed] [Google Scholar]
- 8.Scheiman JM, Hwang JH, Moayyedi P. American gastroenterological association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology 2015;148:824–48. e22. [DOI] [PubMed] [Google Scholar]
- 9.Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 2009;42:377–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chung JW, Chung MJ, Park JY, et al. Clinicopathologic features and outcomes of pancreatic cysts during a 12-year period. Pancreas 2013;42:230–8. [DOI] [PubMed] [Google Scholar]
- 11.Maguchi H, Tanno S, Mizuno N, et al. Natural history of branch duct intraductal papillary mucinous neoplasms of the pancreas: a multicenter study in Japan. Pancreas 2011;40:364–70. [DOI] [PubMed] [Google Scholar]
- 12.Gaujoux S, Brennan MF, Gonen M, et al. Cystic lesions of the pancreas: changes in the presentation and management of 1,424 patients at a single institution over a 15-year time period. J Am Coll Surg 2011;212:590–600; discussion 00–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Allen PJ, D’Angelica M, Gonen M, et al. A selective approach to the resection of cystic lesions of the pancreas: results from 539 consecutive patients. Ann Surg 2006;244:572–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arlix A, Bournet B, Otal P, et al. Long-term clinical and imaging follow-up of nonoperated branch duct form of intraductal papillary mucinous neoplasms of the pancreas. Pancreas 2012;41:295–301. [DOI] [PubMed] [Google Scholar]
- 15.Brounts LR, Lehmann RK, Causey MW, et al. Natural course and outcome of cystic lesions in the pancreas. Am J Surg 2009;197:619–22; discussion 22–3. [DOI] [PubMed] [Google Scholar]
- 16.Khannoussi W, Vullierme MP, Rebours V, et al. The long term risk of malignancy in patients with branch duct intraductal papillary mucinous neoplasms of the pancreas. Pancreatology 2012;12:198–202. [DOI] [PubMed] [Google Scholar]
- 17.Lahav M, Maor Y, Avidan B, et al. Nonsurgical management of asymptomatic incidental pancreatic cysts. Clin Gastroenterol Hepatol 2007;5:813–17. [DOI] [PubMed] [Google Scholar]
- 18.Lee SH, Shin CM, Park JK, et al. Outcomes of cystic lesions in the pancreas after extended follow-up. Dig Dis Sci 2007;52:2653–9. [DOI] [PubMed] [Google Scholar]
- 19.Ohno E, Itoh A, Kawashima H, et al. Malignant transformation of branch duct-type intraductal papillary mucinous neoplasms of the pancreas based on contrast-enhanced endoscopic ultrasonography morphological changes: focus on malignant transformation of intraductal papillary mucinous neoplasm itself. Pancreas 2012;41:855–62. [DOI] [PubMed] [Google Scholar]
- 20.Salvia R, Crippa S, Falconi M, et al. Branch-duct intraductal papillary mucinous neoplasms of the pancreas: to operate or not to operate? Gut 2007;56:1086–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tada M, Kawabe T, Arizumi M, et al. Pancreatic cancer in patients with pancreatic cystic lesions: a prospective study in 197 patients. Clin Gastroenterol Hepatol 2006;4:1265–70. [DOI] [PubMed] [Google Scholar]
- 22.Tanno S, Nakano Y, Nishikawa T, et al. Natural history of branch duct intraductal papillary-mucinous neoplasms of the pancreas without mural nodules: long-term follow-up results. Gut 2008;57:339–43. [DOI] [PubMed] [Google Scholar]
- 23.Uehara H, Nakaizumi A, Ishikawa O, et al. Development of ductal carcinoma of the pancreas during follow-up of branch duct intraductal papillary mucinous neoplasm of the pancreas. Gut 2008;57:1561–5. [DOI] [PubMed] [Google Scholar]
- 24.Woo SM, Ryu JK, Lee SH, et al. Branch duct intraductal papillary mucinous neoplasms in a retrospective series of 190 patients. Br J Surg 2009;96:405–11. [DOI] [PubMed] [Google Scholar]
- 25.Wu BU, Sampath K, Berberian CE, et al. Prediction of malignancy in cystic neoplasms of the pancreas: a population-based cohort study. Am J Gastroenterol 2014;109:121–9. quiz 30 [DOI] [PubMed] [Google Scholar]
- 26.Crippa S, Bassi C, Salvia R, et al. Low progression of intraductal papillary mucinous neoplasms with worrisome features and high-risk stigmata undergoing non-operative management: a mid-term follow-up analysis. Gut Published Online First: 7 January 2016. doi: 10.1136/gutjnl-2015-310162 [DOI] [PubMed] [Google Scholar]
- 27.Chernyak V, Flusberg M, Haramati LB, et al. Incidental pancreatic cystic lesions: is there a relationship with the development of pancreatic adenocarcinoma and all-cause mortality? Radiology 2015;274:161–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Piciucchi M, Crippa S, Del Chiaro M, et al. Outcomes of intraductal papillary mucinous neoplasm with “Sendai-positive” criteria for resection undergoing non-operative management. Dig Liver Dis 2013;45:584–8. [DOI] [PubMed] [Google Scholar]
- 29.Roch AM, DeWitt JM, Al-Haddad MA, et al. Nonoperative management of main pancreatic duct-involved intraductal papillary mucinous neoplasm might be indicated in select patients. J Am Coll Surg 2014;219:122–9. [DOI] [PubMed] [Google Scholar]
- 30.Correa-Gallego C, Ferrone CR, Thayer SP, et al. Incidental pancreatic cysts: do we really know what we are watching? Pancreatology 2010;10:144–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.