Abstract
The COVID-19 pandemic-related constraints on healthcare access have raised concerns about adverse outcomes from delayed treatment, including the risk of cancer progression and other complications. Further, concerns were raised about a potentially significant backlog of patients in need of cancer care due to the pandemic-related delays in healthcare, further exacerbating any potential adverse outcomes. Delayed access to surgery is particularly relevant to urologic oncology since one-third of new cancers in men (20% overall) arise from the genitourinary (GU) tract and surgery is often the primary treatment. Herein, we summarize the prepandemic literature on deferred surgery for GU cancers and risk of disease progression. The aforementioned data on delayed surgery were gathered in the context of systemic delays present in certain healthcare systems, or occasionally, due to planned deferral in suboptimal surgical candidates. These data provide indirect, but sufficient insight to develop triage schemas for prioritization of uro-oncological cases. Herein, we outline the extent to which the pandemic-related triage guidelines had influenced urologic practice in various regions. To study the adverse outcomes in the pandemic-era, a survey of urologic oncologists was conducted regarding modifications in their initial management of urologic cancers and any delay-related adverse outcomes. While the adverse effects directly from COVID-19 related delays will become apparent in the coming years, the results showing short-term outcomes are quite instructive. Since cancer care was assigned a higher priority at most centers, this strategy may have avoided significant delays in care and limited the anticipated negative impact of pandemic-related constraints.
Keywords: COVID-19, Surgery, Bladder cancer, Prostate cancer, Kidney cancer
1. Introduction
The highly contagious COVID-19 spread at such a rapid rate that within 10 weeks of its recognition, a global pandemic had been declared [1]. It is clear that without any effective therapies or effective vaccines on the horizon, mitigation through social distancing is the only effective strategy to control the spread of COVID-19 [2,3]. The mitigative role of physical distancing naturally applies to health care workers (HCWs) and patients, especially those in need of procedures for various malignancies. Based upon the guidance from public health authorities and medical societies, nonurgent medical care, including elective surgery, had been canceled due to increased risk of exposure of HCWs [4]. Initial recommendations of public health authorities and medical societies for postponement of nonurgent surgery were designed to maintain the health systems’ capacity to accommodate the anticipated surge of cases, to preserve personal protective equipment (PPE), and to avoid unnecessary exposure to COVID-19 in order to maintain a healthy work force [5,6].
While it had been widely anticipated that the rate of COVID-19 infections would be under control by the summer of 2020, quite the opposite has occurred. Nearly 8 months from the onset of the COVID-19 outbreak, daily infection rates have continued to rise throughout most of the United States, Brazil, India and Russia, with new clusters reported in China and the United Kingdom [7,8]. Consequently, of the 155 countries surveyed by the WHO, 42% had partially or completely disrupted services for cancer treatment [1, 9]. It has been reported that patients with COVID-19 infection undergoing surgery had a perioperative respiratory complication rate of over 50% and 30-day mortality rate of 38%, particularly in patients that were older, male, or with comorbidities and malignancy. Over one-third of new cancers in men and 20% of all new cancers are diagnosed in the GU tract [10]. For patients with GU malignancies, much of the survival benefit is derived through surgical extirpation of the primary tumor. Thus, the concerns over potential adverse impact (oncologic and nononcologic) of delayed care of GU cancers, compounded by the climbing number of COVID-19 infections, are likely to remain clinically relevant for the foreseeable future.
Herein, we outline the existing evidence regarding adverse oncologic outcomes from delayed surgical care and the extent of GU cancer care disruption reported from various regions due to various local or regional triage guidelines. These guidelines on the safety of deferred treatment were developed based on reports of delayed care due to causes unrelated to an outbreak. These include factors such as delayed access to care inherent in certain healthcare systems or deferred treatment in unhealthy patients who were deemed unfit for surgery. In order to capture contemporaneous data on the pandemic-related changes in treatment paradigms and potential adverse outcomes, we conducted a survey of urologic oncologists to obtain first-hand account of delays and short-term outcomes which are summarized below.
2. Delayed surgery and adverse outcomes
Regions that may be expecting a surge in the pandemic, and the related constraints on access to healthcare, will need to develop triage and prioritization schemes for elective, but urgent or essential cases. Other than for low-risk prostate cancer and small renal masses, there is no prospective data available on adverse outcomes related to deferred treatment. Most of the prepandemic published literature is based on deferred treatment for patients with significant comorbidities who were deemed poor candidates or from unplanned delays in access to healthcare such as in rural areas or national health services. Below, we provide a targeted review of the historic knowledge on the extent to which delays in surgery may be acceptable and without compromising oncological outcomes.
2.1. Prostate cancer
2.1.1. Low- or intermediate-risk prostate cancer
In men with low-risk prostate cancer (CaP), active surveillance (AS) is the standard management strategy with >98% long-term survival so the issue of surgical case delay is not relevant in this context [11]. While most patients with International Society of Urological Pathology grade group 2 (GG2) CaP undergo treatment, a subgroup with unifocal or low volume GG2 can exhibit indolent behavior and can be managed initially in a fashion similar to GG1 CaP. Although a previous study of AS of GG2 CaP reported increased risk of metastases over a 15-year period, this alone does not preclude AS in this group [12]. A more stringent AS protocol tailored to GG2 patients should be used than the protocol used in that study; one that incorporates MRI of prostate or tissue-based genomic tests to identify grade progression and offer timely treatment [13]. In a pandemic-related triage plan, treatment of contemporary low-volume GG2 CaP (e.g., 1–2 core) can be safely deferred, with monitoring, for a year or longer. Patients with larger volume GG2 (>3 cores) or GG3 CaP who may have been counseled to undergo radical prostatectomy can be reassured of the safety of deferred treatment for 6 to 12 months, without compromising oncological control [14].
2.1.2. High-risk prostate cancer
GG4 or GG5 CaP is associated with high-risk of adverse pathology, increased risk of metastasis, and mortality. Because early treatment is usually recommended for high-risk CaP, data on delayed treatment specifically for this cohort is scant and the rationale entirely undefined. A study of men with a mix of GG3 to GG5 CaP treated with radical prostatectomy within 3 months vs. 3 to 6 months after diagnosis did not find any significant differences in terms of adjuvant therapy, or 5‐year biochemical or metastasis-free survival for any GG CaP [15]. Similarly, a Scandinavian study of long-term follow up (29 years) of clinically localized CaP, >50% of which were classified as high risk, demonstrated a prolonged time interval between diagnosis of CaP and the development of metastasis, suggesting that a short delay in definitive treatment is unlikely to result in adverse oncological control [16]. A study by Boorjian et al., of 3,149 men, including historical cohorts, treated with radical prostatectomy, looked at the risk of biochemical recurrence based on the time interval between biopsy and surgery [17]. They found no association between delayed surgery (up to 12 months) and risk of biochemical recurrence, including in small subgroup with high-risk disease. A more recent retrospective review suggested that delayed prostatectomy for GG4 and GG5 was not associated with worse oncological outcomes [14]. However, this cohort largely consisted of intermediate risk cases, and only 3% of cases were delayed for more than 6 months. In the absence of any Level 1 evidence, the recommendation for surgical delays must rely on scant data from retrospective studies and clinical judgment. It is likely that the screening-based, contemporarily diagnosed high-risk CaP, which is often detected by advanced imaging, is less risky than the traditional cohort, providing some confidence in the safety of short-term deferral for up to 6 months. Men with very high-risk CaP (multiple cores with GG4-5, cT3, PSA > 20), should be prioritized for treatment, however, surgery does not need to be the initial treatment in the setting of a pandemic since effective alternative therapies are available.
2.1.3. Alternatives to surgery
Limited access to surgery in the setting of a pandemic has made other options, such as radiotherapy (RT) and/or androgen deprivation therapy (ADT) a topic of debate [18]. While RT may be equally efficacious for many patients, the feasibility of safely delivering RT in a pandemic depends on the location and logistics of the radiation facility. Switching treatment from surgery to RT in a pandemic due to safety reasons may be difficult to justify considering dozens of required encounters with HCW and increased risk of exposure. Further, switching treatment from surgery to RT for expediency appears oncologically unnecessary, since any treatment of CaP can be safely deferred for a few months.
Due to concerns over disease progression from deferred surgery for high‐risk and/or locally advanced CaP, the use of neoadjuvant ADT while awaiting surgery has been discussed and/or recommended by some experts. This is not an entirely a new concept in high-risk CaP treatment. The efficacy and safety of neoadjuvant ADT for 4 months has been reported previously in a small study. A recent review of the National Cancer Database revealed that the use of neoadjuvant hormonal therapy prior to radical prostatectomy has increased over the last few years [19]. It is important to note that the role of neoadjuvant ADT prior to surgery for high‐risk disease remains undetermined, and is currently a subject of randomized controlled trials. However, if delay of >6-month is anticipated, neoadjuvant ADT for high-risk CaP may be used with informed discussion, in select cases. This approach may provide pathologic control and some reassurance, especially since the clinical use and side effects of short-term ADT are well established from combination with RT.
2.2. Bladder (urothelial) cancer
2.2.1. Nonmuscle invasive bladder cancer
Small, papillary tumors (< 3 cm) when resected are typically low-grade, low-risk, nonmuscle invasive bladder cancer (NMIBC). These are associated with frequent recurrences but very low risk of progression to higher stage [20]. Very small papillary tumors (<1 cm), can be safely fulgurated in the office setting. While cancer staging is important, the risk of anesthesia and hospitalization during a pandemic may outweigh the benefits of a staging resection at that time. Thus, in the setting of pandemic-related constraints, delayed transurethral resection (TURBT) for up to 3 months is oncologically safe, unless gross hematuria requires earlier intervention.
A different set of considerations applies to patients with bladder masses that appear to be in the high-risk NMIBC category, i.e., large, multifocal, with carcinoma-in-situ (CIS). High-grade, T1 tumors ± CIS are at increased risk of progression to muscle-invasive disease. Thus, these patients should be prioritized for TURBT with both a diagnostic (staging) and a therapeutic intent, followed by intravesical therapy within 1 to 2 months. A standard recommendation for patients with high-risk NMIBC is to undergo repeat TURBT within weeks due to the 30% to 40% risk of undetected muscle invasion, i.e., understaging [21]. In the setting of a pandemic, this approach would potentially double the resource utilization, and the risk of COVID-19 exposure to the patient and staff. Thus, it is even more critical to perform a high-quality initial TURBT, with complete and deep resection, such that the risk of understaging can be reduced and repeat TURBT can be safely delayed.
2.2.2. Muscle invasive bladder cancer
Whether considering primary upfront cystectomy or NAC, consideration must be given to the frequency of medical encounters, the risk of readmission or ER visits, and the risk of COVID‐19 infection in the perioperative period vs. during 3‐4 cycles of cisplatin‐based chemotherapy. Radical cystectomy requires hospitalization for 6 to 7 days, with 25%‐30% risk of readmission [22]. Use of NAC requires multiple medical encounters for several weeks to months to undergo infusions, imaging, and laboratory testing. Chemotherapy is also associated with 10% to 20% risk of ER visits or hospital admissions due to complications, with high utilization rate of resources including PPE during each encounter [23]. While deciding on the initial therapy, due consideration should be given to the net survival benefit from NAC, which appears to be relatively modest, and from cystectomy which is curative, providing much of the survival advantage in MIBC. Alternative approaches such as RT with sensitizing chemotherapy can be effective in select patients and may be an attractive option for MIBC with favorable features. However, the frequency of interactions required for this approach defies the only effective mitigative measure against COVID-19, i.e., physical distancing. Further there are concerns regarding chemotherapy administration in the elderly with cancer. In a modeling study, it was estimated that the case fatality rate from COVID-19 infection in a 70-year-old patient may be 2- to 3-folds higher than the 5% survival benefit from chemotherapy [24]. Thus, in the context of pandemic-related limitations on access, the initial treatment should be that which provides the highest cure fraction, e.g., radical cystectomy.
For patients being considered for radical cystectomy, time from diagnosis to surgery is an important predictor of oncologic and survival outcomes. In a single center study of 247 patients, a lag between diagnosis and surgery of >12 weeks was associated with worsening pathologic stage and lower 3-year survival [25]. These adverse outcomes from delayed surgery persisted even after adjustment for nodal status, and clinical and pathological stage. Gore et al. analyzed the Medicare-linked SEER dataset to determine the effect of surgical delay in the subset of 441 patients with stage II bladder cancer [26]. Significantly worse disease-specific and overall survival were noted if the delay between diagnosis and cystectomy was >12 weeks, and those with >24-week delays had a rapid decline in survival.
2.3. Upper tract urothelial cancer
Other than patients with a solitary kidney or small, low-grade tumors, nephroureterectomy (NU) is required in most patients with high-grade upper tract urothelial cancer (UTUC). Waldert et al. have reported delayed NU by >3 months is associated with worse pathologic stage and lymph node metastases [27]. Another study reported that delay of >4 months, compared to 1 month, was associated with significantly worse overall survival [28]. Alternative approaches include repeat endoscopic ablation or partial ureterectomy when anatomically feasible. Repeat endoscopic procedures, which are noncurative, require multiple interactions and resource utilization, and thus, would not be considered a safer option. During the pandemic, the initial intervention for UTUC should be that which provides the most durable disease control should be given priority i.e. partial ureterectomy or nephroureterectomy.
2.4. Kidney cancer
2.4.1. Small renal mass
Small renal masses (SRM; ≤4 cm) are typically associated with slow growth and low risk of progression or metastases [29]. In a prospective study on active surveillance of SRM, the metastasis rate was only 1.1% after 28 months follow-up, with median increase in diameter of 1.3 mm/y. Another prospective study has demonstrated a cancer‐specific survival rate of 100% for patients on active surveillance for SRMs [30]. Considering these favorable data, surgery or ablation for SRMs can be deferred for 12 months or longer, and replaced with periodic imaging to establish growth kinetics.
2.4.2. Large renal mass
Larger renal masses can be classified as cT1b (between 4 and 7 cm) or cT2 (>7 cm). These masses are managed with early surgical excision, so studies of the natural history of large renal masses typically include only those individuals who were deemed suboptimal candidates for surgery. In a small study of 68 patients with cT1b renal masses (median 4.9 cm) and at least 6 months follow-up, delayed intervention in patients with higher linear growth rates (median 0.72 cm/y) did not result in disease progression to metastases [31]. A retrospective study by Touma et al. described the natural history of cT1b tumors that were observed without upfront nephrectomy in older patients (mean age 75.5 years) [32]. Progression to metastases was noted in 24% of patients who had a linear growth rate of 0.98 cm/y, while those remaining free of metastases had a slower growth rate at 0.67 cm/y. The linear growth rate appears to be highly correlated with disease progression in patients with large renal masses. Thus, in situations with limited access to healthcare, an initial period of surveillance with repeat imaging at 3 to 6 months intervals to define growth kinetics appears to be safe.
2.4.3. Inferior vena cava tumor thrombus
Patients with a renal mass and tumor thrombus in the inferior vena cava are at high risk of developing metastases and/or other complications. This is highlighted by the fact that if left untreated, median survival for these patients is only 5 months and 1‐year disease specific survival is <30% [33]. Several reports have demonstrated a durable survival rate following complete excision including radical nephrectomy and IVC thrombectomy. Therefore, surgical care of these patients is considered urgent and should be prioritized to be done in <1 month [34].
2.4.4. Metastatic renal cancer
The role of cytoreductive nephrectomy as a standard of care in the setting of synchronous metastases has recently been challenged since two randomized trials did not show a survival advantage to upfront nephrectomy. The CARMENA trial demonstrated the noninferiority of initial therapy sunitinib alone compared to nephrectomy followed by sunitinib in intermediate and high-risk RCC [35]. Further, the SURTIME trial, evaluating the timing of nephrectomy, demonstrated that initial systemic therapy with delayed nephrectomy was safe and without any difference in short-term disease progression [36]. In the setting of pandemic-related constraints, there is a greater impetus for early initiation of systemic therapy and assessment of objective response before considering cytoreductive nephrectomy.
2.5. Testicular cancer
2.5.1. Clinical stage I
Delays of more than 1 month in seeking primary care or specialist referral are common and significantly associated with worse pathologic stage. This can result in increased intensity of subsequent adjuvant therapy, and possibly affect the survival outcomes [37]. Thus, radical orchiectomy should be performed within 2 weeks of presentation.
Clinical stage I germ cell tumors, both seminoma and nonseminoma, have been managed with various surveillance strategies at an increasing rate over the last decade. Mazzone et al. analyzed (with propensity score matching) the SEER data to determine the rate of postorchiectomy intervention in 5,034 patients with testicular nonseminoma [38]. There was an increasing rate of surveillance and decreasing rate of retroperitoneal lymph node dissection (RPLND), and active treatment compared to surveillance was not associated with lower cancer-specific mortality. In a companion study, of 11,206 patients with testicular seminoma in the SEER database, there was an increase in the utilization of initial surveillance and chemotherapy and decrease in the use of RT [39]. No differences in survival were noted, when surveillance or RT or chemotherapy were compared with each other. In a resource-constrained environment with associated risks of exposure, it is safe to employ surveillance strategy instead of adjuvant chemotherapy, RT, or RPLND.
2.5.2. Metastatic testicular cancer
Since, the mid-1970s, survival of men with metastatic testicular cancer is in large part dependent upon platinum-based chemotherapy, with consolidative surgery playing an important role [40]. In the setting of a pandemic, valid concerns about systemic chemotherapy have been raised such as frequent interactions, repeated exposure and immunosuppression. However, patients with testicular cancer are often young and healthy, and thus, at a lower risk of complications from immunosuppression or COVID-19 exposure. Since chemotherapy for advanced testicular cancer (unlike urothelial cancer) is usually curative, it should be initiated without delay for these patients with otherwise long life expectancy. In case of large tumor burden and constraints on surgical access, radical orchiectomy can be postponed if necessary to expedite platinum-based chemotherapy [41].
2.5.3. Postchemotherapy retroperitoneal mass
The management of postchemotherapy residual retroperitoneal mass depends on the type of primary tumor. For seminomatous tumors, small residual mass (<3 cm) after chemotherapy can be safely observed to allow serial imaging since its very uncommon to find viable tumor in RPLND specimen [42], [43]. Those patients with increasing size of the mass may be selected for RPLND. Larger (>3 cm) seminomatous residual masses typically require RPLND, although the surgery can be quite challenging and 20% to 40% may still only harbor fibrosis [44].
The radiographic size of residual NSGCT mass does not inform the decision to delay RPLND. A report from the German Testicular Cancer Study Group has demonstrated that as the mass size increases from <1 cm to 1.5 and >1.5 cm, the viable cancer detection rate in the RPLND specimen increases from 9.4% to 21% and 35% [45]. Available data on delayed RPLND is scant and suggest that delays >3 months can significantly compromise patient survival [46]. In the absence of any alternative curative therapies for this stage, RPLND for a NSGCT residual mass should be prioritized and performed within a month.
2.6. Penile cancer
Penile cancer is a rare but aggressive tumor which is often associated with delayed presentation. Because surgical excision is the only curative treatment, the primary tumor is often excised promptly and there is lack of clinical data on the effect of delayed surgery on penile cancer outcomes. It is known that if left untreated, most men will die of the disease within 2 years [47]. Lucky et al. reported, delayed presentation of >6 months was associated with locally advanced disease in 43% of men [48]. Due to the rarity of this tumor and scant data, primary lesions should be excised without delay. To avoid risk of exposure to COVID-19, this can be accomplished either spinal or regional anesthesia.
The timing of inguinal lymphadenectomy (ILND) for cN0 patients is debatable. While a short period of observation is often used to differentiate any infection or inflammation related palpable LND, earlier ILND is associated with improved survival [49]. Leitje et al. have demonstrated that prophylactic ILND performed before developing inguinal recurrence is associated with improved disease-specific survival. Patients with initial cN0 disease, ILND within 3 months were associated with significantly higher disease-specific survival than those with late ILND (64.1% vs. 39.5%, respectively [50]). Penile cancer control requires surgical excision at all stages of the disease, thus surgical excision should proceed without delay.
3. Surgical priority guidelines and clinical practice
As COVID-19 pandemic-related constraints were being implemented, several organizations and experts developed documents on prioritization of surgical intervention for various urological conditions [18, [51], [52], [53], [54]]. The prioritization strategies for treatment of various urologic conditions, especially cancers, were based on the criteria and historic knowledge of adverse outcomes from delayed care detailed in Section 2 above. These triage guidelines were reviewed and summarized by Shinder et al., demonstrating general agreement in the recommendations, with some variation [55]. Due to the urgency and need for rapid communication during the early days of the pandemic, none of the initial guidelines and recommendations were based on a formal, systematic review of the literature. Typically, the initial methodology included targeted review of literature and expert opinion from a specialty organization or a group, or solicitation of rapid response from experts, including in open social media format [18]. Subsequently, Katims et al. and others carried out systematic reviews of the literature, generating guidelines for prioritization of care for urologic cancers [56]. Herein, we outline the extent to which the urologic community has adapted these changes in clinical practice over the recent months.
The Société Internationale d'Urologie (SIU) conducted a survey in late March 2020 to assess changes in practice for various conditions in response to COVID-19 [57]. A total of 2,494 urologists (various specialties) from 76 countries completed the survey. The survey questions included whether the urologists would change (prioritize or postpone) any of the 14 common urology procedures, including oncologic procedures, and to assign priority score to these procedures. Overall, oncological conditions, especially conditions with the greatest malignant potential received higher priority score (e.g., median scores for TURP, 1; radical prostatectomy, 5; TURBT, 8). Urologists were less likely to postpone and more likely to continue or prioritize scheduling of oncological procedures. For example, uncomplicated stone management was 2.5 times more likely to be postponed while nephroureterectomy was 2.5 times more likely to be prioritized. In general, respondents’ priority scoring of urological procedures closely matched the priority recommendations by the guidelines published by the European, the British and Australian urologic societies.
One of the surveys of urology providers (physicians, nonphysicians, nurses, advanced practitioners) about the pandemic-related effects was conducted using Twitter accounts of nearly 20 urology-related organizations and individuals [58]. From these accounts with well over 100,000 followers, 678 urology providers from 77 countries completed the full survey. As expected, the regions with severe COVID-19 outbreak reported greater reduction in urologic surgery. A delay in access to urological services was reported across the board, with 50% of respondents concerned about adverse effects of delayed treatment on survival outcomes of their patients. Overall, urologic surgery for benign conditions was reported to have been reduced by nearly 75% to 90%, while urologic cancer surgeries were reduced by 20% to 35%.
In a 12-question survey using social media in late March 2020, Dotzauer et al. received responses from 235 urologists from 44 countries, including 25 from the Unites States. A majority of urologists, 63%, were practicing in WHO designated COVID-19 high-risk countries. As expected, 93% of urologists indicated a change in their clinical practice due to COVID-19. However, the majority of respondents (75%–80%) were able to continue oncologic surgery, with few cancellations [59]. The Papa Giovanni XXIII Hospital in Northern Italy was among those that were overwhelmed earlier by an exponential increase in COVID-19 cases [60]. Most of the medical personnel were reassigned to care for the COVID-19 patients including anesthesiologists. Consequently, a staggering 75% reduction in overall surgical procedures, with a 67% reduction in uro-oncological procedures was noted, compared to the same period in 2019.
With near complete cessation of outpatient procedures for early detection in many regions, combined with substantial reduction in surgery for GU cancers, these authors and others have expressed significant concerns over the potential for adverse oncological outcomes, which could appear in the months or years to follow.
4. Changes in clinical practice and adverse outcomes
While the overall reduction in GU oncology surgical procedures has been reported, data on the use of preferred alternative therapies or the extent of adverse outcomes from deferred treatment have not been available. To understand changes in the treatment of common GU cancers, we surveyed members of the Society of Urologic Oncology (SUO) between June 10 and June 28, using an anonymous e-mail survey sent to 821 members. Complete survey response was received from 140, with response rate of 17.1%. This response rate is within the expected range of online medical surveys which is often <20%, depending upon the frequency and type of follow up reminders (we sent only on follow up reminder, as per policy of the SUO) [61]. Because this survey was conducted during the 6th month of the pandemic, sufficient time had elapsed to potentially demonstrate some of the adverse outcomes (or lack thereof) from modifications in cancer management strategies in response to the pandemic-related delays. The questions included whether the COVID-19 pandemic had necessitated modifications to their standard “initial” management of CaP, (intermediate and high risk), bladder cancer (MIBC), and kidney cancer (T2-4), and which alternative therapies were utilized instead of surgery. We also inquired whether the respondents were personally aware of any patients with GU cancers who had experienced an adverse outcome related to delayed treatment (Table 1 ).
Table 1.
Characteristics of respondents and modifications in treatment due to Covid-19 pandemic
| Question | Response no. | Response percent |
|---|---|---|
| Age (y): | ||
| <40 | 41 | 29.3 |
| 40–50 | 59 | 42.1 |
| 51–60 | 25 | 17.9 |
| 61–70 | 10 | 7.1 |
| >70 | 5 | 3.6 |
| Practice location: | ||
| Large, urban area | 90 | 64.3 |
| Mid-sized city | 42 | 30.0 |
| Small, rural area | 8 | 5.7 |
| Practice type: | ||
| Academic | 113 | 80.7 |
| Private small group | 5 | 3.6 |
| Private, hospital employed | 7 | 5.0 |
| Private, large group | 9 | 6.4 |
| VA, Govt. or public hospital | 6 | 4.3 |
| How was your region affected by COVID-19 infections? | ||
| Minimally | 22 | 15.7 |
| Moderately | 62 | 44.3 |
| Severely | 44 | 31.4 |
| Overwhelmed | 12 | 8.6 |
| Currently, how many "elective" surgical procedures are you able to schedule, compared to your normal schedule? | ||
| <25% | 18 | 12.9 |
| 26–50% | 29 | 20.7 |
| 51–75% | 23 | 16.4 |
| >75% | 70 | 50.0 |
| When do you expect to have no restrictions on elective surgery scheduling? | ||
| within 4–6 weeks | 79 | 56.4 |
| <3 months | 29 | 20.7 |
| >9 months | 4 | 2.9 |
| 3–6 months | 19 | 13.6 |
| 6–9 months | 9 | 6.4 |
| When do you expect to clear the backlog of uro-oncology surgical cases? | ||
| <1 month | 69 | 49.3 |
| 2–3 months | 55 | 39.3 |
| 4–6 months | 12 | 8.6 |
| >6 months | 4 | 2.9 |
| Did the pandemic-related delays in access require you to consider modifications to your standard “initial” treatment for intermediate and high-risk prostate cancer (multiple answers allowed): | ||
| No changes required due to sufficient access to care | 39 | 27.9 |
| Postponed all surgery | 82 | 58.8 |
| Preference for RT | 15 | 10.7 |
| Preference for ADT | 27 | 19.2 |
| Preference for hypo-RT | 5 | 3.6 |
| Did the pandemic-related delays in access require you to consider modifications to your standard “initial” treatment for muscle-invasive bladder cancer (multiple selections allowed): | ||
| No changes required due to sufficient access to care | 99 | 70.7 |
| Preference for cystectomy, without neoadjuvant chemotherapy | 20 | 14.3 |
| Preference neoadjuvant chemotherapy to buy time | 28 | 20.0 |
| Preference of radiation therapy instead of surgery | 5 | 3.6 |
| Did the pandemic-related delays in access require you to consider modifications to your standard “initial” treatment for kidney cancer (T2-T4, M+) (multiple selections allowed): | ||
| No changes required due to sufficient access to care | 61 | 43.6 |
| Postponed surgery for all asymptomatic cases, regardless of size | 18 | 12.9 |
| Preference surgery only for large, complex masses | 65 | 46.4 |
| Preference for systemic therapy for metastatic cases instead of nephrectomy | 14 | 10.0 |
| Are you “personally” aware of any patient who suffered from cancer progression and poor outcome due to delayed access to care? | ||
| Yes | 19 | 13.5 |
| No | 121 | 86.5 |
| How many patients? (Median 2; Range 1–10) | ||
| 1 | 6 | 4.3 |
| 2–3 | 7 | 5.0 |
| >3 | 5 | 3.6 |
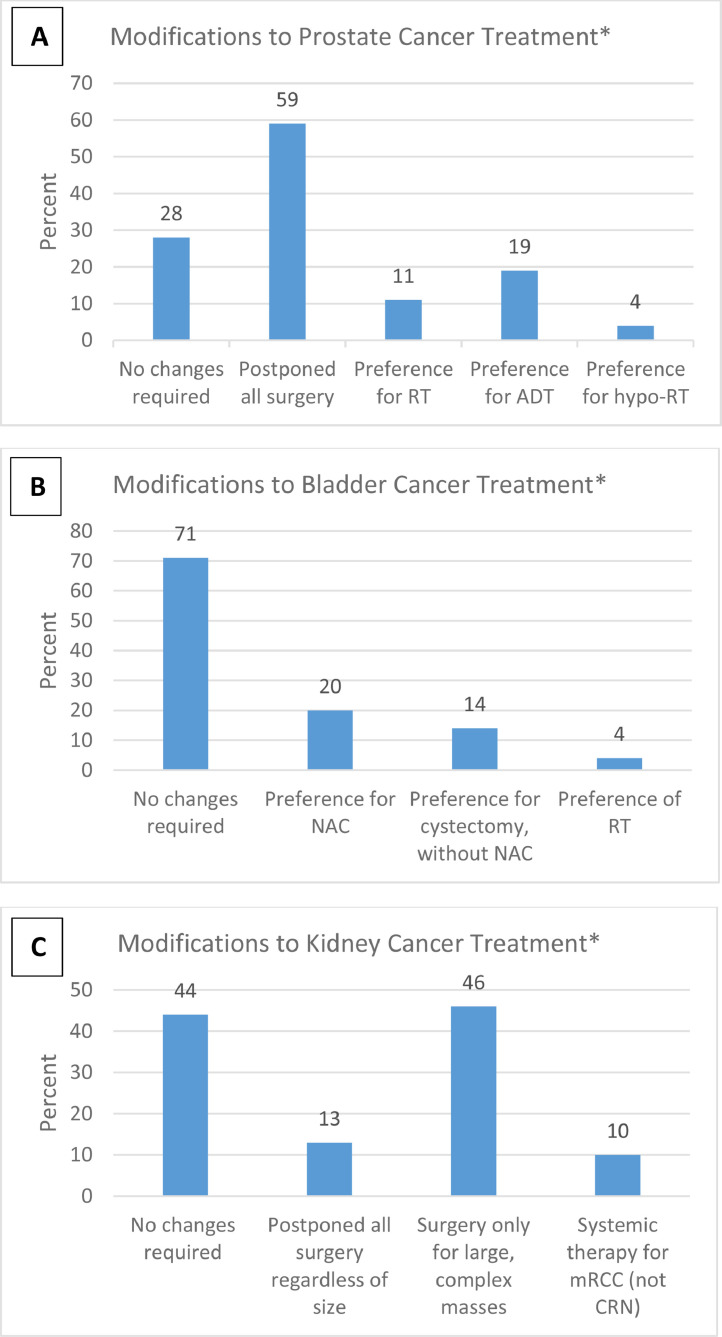
Of the respondents, 72%, 29%, and 56% reported that they had modify their initial management of prostate cancer, bladder cancer and large renal masses, respectively. Of these, cancellation of all surgical cases for prostate cancer and kidney cancer was reported by 59% and 13% of respondents, respectively (Fig. 1 ). As an alternative to surgery for prostate cancer during the pandemic, ADT was preferred by 19% and RT by 15% of the respondents. Of the alternative approaches utilized for MIBC, 20% preferred neoadjuvant chemotherapy during delayed surgery while 14% favored early cystectomy. For Kidney cancer management (excluding small renal mass), surgery was preferentially performed only for the large, complex renal masses. For metastatic renal cancer, only 10% reported a preference for initial systemic therapy instead of upfront cytoreductive nephrectomy. This appears to be in discordance with a number of published guidelines.
Fig. 1.
Modifications to initial treatment in response to COVID-19 related constraints. (A) Prostate cancer; (B) Bladder cancer; (C) Kidney cancer. RT, radiotherapy; ADT, androgen deprivation therapy. *Total may be >100% because multiple choices were allowed.
Nineteen uro-oncologists (13.5%) reported that they were personally aware of patients with GU cancers who experienced an adverse outcome due to COVID-19 related delays in surgery. These were primarily from an academic practice (84%) or a large, urban area (74%) or from regions severely affected by COVID-19 (74%). The median number of patients with a known adverse outcome was 2 (range 1–10). Details of the adverse outcomes were provided for 13 patients by 10 (7%) respondents. These included oncological disease progression in 9 patients (lymph node enlargement, IVC thrombus, lung nodules) and nononcological complications in 4 patients (hematuria, obstruction, ER visits). In response to the extent of backlog of GU cancer surgery, 49% believed that the backlog could be cleared within 1 month while 39% stated 2 to 3 months.
These results suggest that most uro-oncologists have been able to continue surgical treatment along with their standard management pathways for bladder cancer, and to some extent, kidney cancer. Most significant disruption of initial surgical treatment was noted for prostate cancer, with one-third of the respondents using some type of alternative therapy instead of surgery.
5. Conclusion
In regions facing a potential surge of a pandemic and subsequent strain on the healthcare system, the most optimal prioritization strategy would consider patient safety and the adverse outcomes from postponing treatment. Simultaneously, steps should be taken to avoid unnecessary backlog of cases which could further exacerbate the effects of delayed treatment. Significant concerns exist regarding poor oncological outcomes and complications from delayed cancer surgery. While the full impact of disruption in cancer care on survival outcomes will not be fully known for a few years, the survey of uro-oncologists provides an early snapshot, and a first-hand account of the extent of treatment modifications and some of the adverse outcomes. In most regions of the world, treatment for various malignancies, including surgery, had been given priority in most of the triage schemes. Consequently, this strategy may have avoided a significant backlog of surgical cases as evident from the survey results. Since most patients have been able to receive some type of treatment (often within the proposed window of safety), we hypothesize that the COVID-19 pandemic-related disruptions in health care may not have a significant impact on survival outcomes of patients with urologic cancers.
References
- 1.Coronavirus disease (COVID-19) - events as they happen. World Health Organization. Available at:https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen. Accessed 24 July 2020.
- 2.Jarvis CI, Van Zandvoort K, Gimma A, et al. Quantifying the impact of physical distance measures on the transmission of COVID-19 in the UK. BMC Med. 2020;18(1):124. doi: 10.1186/s12916-020-01597-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kobokovich AL, Hosangadi D, Rivers C. Supporting social distancing for COVID-19 mitigation through community-based volunteer networks. Am J public health. 2020;110(8):1167–1168. doi: 10.2105/AJPH.2020.305740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de Leeuw RA, Burger NB, Ceccaroni M, et al. COVID-19 and laparoscopic surgery: scoping review of current literature and local expertise. JMIR Public Health Surveill. 2020;6(2):e18928. doi: 10.2196/18928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Society of American Gastrointestinal and Endoscopic Surgeons. SAGES and EAES recommendations regarding surgical response to COVID‐19 crisis. [cited 2020 Mar 30] Available from: https://www.sages.org/recommendations‐surgical‐response‐covid‐19/
- 6.Association of Surgeons of Great Britain and Ireland. Delivering the emergency general surgery service in the UK during the coronavirus COVID‐19 pandemic. [cited 2020 Apr 18] Available from:https://www.asgbi.org.uk/userfiles/file/news/asgbi‐statement‐the‐ delivery‐of‐emergency‐general‐surgery‐and‐covid‐19.pdf
- 7.Worldometer. “Coronavirus toll update: cases & deaths by country of Wuhan, China Virus - Worldometer.” Worldometers.Info, 2020. www.worldometers.info/coronavirus/.
- 8.CDC. “Coronavirus disease 2019 (COVID-19) in the U.S.” Centers for Disease Control and Prevention, 16 Mar. 2020.www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
- 9.COVIDSurg Collaborative Mortality and pulmonary complications in patients undergoing surgery with perioperative SARS-CoV-2 infection: an international cohort study. Lancet (London, England) 2020;396(10243):27–38. doi: 10.1016/S0140-6736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA A Cancer J Clin. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 11.Tosoian JJ, Mamawala M, Epstein JI, et al. Intermediate and longer-term outcomes from a prospective active-surveillance program for favorable-risk prostate cancer. J Clin Oncol. 2015;33(30):3379–3385. doi: 10.1200/jco.2015.62.5764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Klotz L, Vesprini D, Sethukavalan P, et al. Long-term follow-up of a large active surveillance cohort of patients with prostate cancer. J Clin Oncol. 2015;33(3):272–277. doi: 10.1200/JCO.2014.55.1192. [DOI] [PubMed] [Google Scholar]
- 13.Klotz L. Active surveillance in intermediate-risk prostate cancer. BJU Int. 2020;125(3):346–354. doi: 10.1111/bju.14935. [DOI] [PubMed] [Google Scholar]
- 14.Ginsburg KB, Curtis GL, Timar RE, et al. Delayed radical prostatectomy is not associated with adverse oncologic outcomes: implications for men experiencing surgical delay due to the COVID-19 pandemic. J Urol. 2020;204(4):720–725. doi: 10.1097/JU.0000000000001089. [DOI] [PubMed] [Google Scholar]
- 15.Gupta N, Bivalacqua TJ, Han M, et al. Evaluating the impact of length of time from diagnosis to surgery in patients with unfavourable intermediate-risk to very-high-risk clinically localised prostate cancer. BJU Int. 2019;124(2):268–274. doi: 10.1111/bju.14659. [DOI] [PubMed] [Google Scholar]
- 16.Bill-Axelson A, Holmberg L, Garmo H, et al. Radical prostatectomy or watchful waiting in prostate cancer - 29-year follow-up. N Engl J Med. 2018;379(24):2319–2329. doi: 10.1056/NEJMoa1807801. [DOI] [PubMed] [Google Scholar]
- 17.Boorjian SA, Bianco FJ, Jr, Scardino PT, et al. Does the time from biopsy to surgery affect biochemical recurrence after radical prostatectomy? BJU Int. 2005;96(6):773–776. doi: 10.1111/j.1464-410X.2005.05763.x. [DOI] [PubMed] [Google Scholar]
- 18.Shah P, Kim FJ, Mian BM. Genitourinary cancer management during a severe pandemic: utility of rapid communication tools and evidence-based guidelines. BJUI compass. 2020;1(2):45–59. doi: 10.1002/bco2.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McClintock TR, von Landenberg N, Cole AP, et al. Neoadjuvant androgen deprivation therapy prior to radical prostatectomy: recent trends in utilization and association with postoperative surgical margin status. Ann Surg Oncol. 2019;26(1):297–305. doi: 10.1245/s10434-018-7035-z. [DOI] [PubMed] [Google Scholar]
- 20.Simon M, Bosset PO, Rouanne M, et al. Multiple recurrences and risk of disease progression in patients with primary low-grade (TaG1) non-muscle-invasive bladder cancer and with low and intermediate EORTC-risk score. PLoS One. 2019;14(2) doi: 10.1371/journal.pone.0211721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chang SS, Boorjian SA, Chou R, et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline. J Urol. 2016;196(4):1021–1029. doi: 10.1016/j.juro.2016.06.049. [DOI] [PubMed] [Google Scholar]
- 22.Stimson CJ, Chang SS, Barocas DA, et al. Early and late perioperative outcomes following radical cystectomy: 90-day readmissions, morbidity and mortality in a contemporary series. J Urol. 2010;184(4):1296–1300. doi: 10.1016/j.juro.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 23.Pittman NM, Hopman WM, Mates M. Emergency room visits and hospital admission rates after curative chemotherapy for breast cancer. J Oncol Pract. 2015;11(2):120–125. doi: 10.1200/JOP.2014.000257. [DOI] [PubMed] [Google Scholar]
- 24.Williams M, Calvez K, Mi E, et al. Estimating the risk of death from COVID infection in adult cancer patients. medRxiv. 2020;2018 2020. 2003. [Google Scholar]
- 25.Sánchez-Ortiz RF, Huang WC, Mick R, et al. An interval longer than 12 weeks between the diagnosis of muscle invasion and cystectomy is associated with worse outcome in bladder carcinoma. J Urol. 2003;169(1):110–115. doi: 10.1097/01.ju.0000039620.76907.0d. [DOI] [PubMed] [Google Scholar]
- 26.Gore JL, Lai J, Setodji CM, et al. Urologic diseases in America project. Mortality increases when radical cystectomy is delayed more than 12 weeks: results from a surveillance, epidemiology, and end results-medicare analysis. Cancer. 2009;115(5):988–996. doi: 10.1002/cncr.24052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Waldert M, Karakiewicz P, Raman J, et al. A delay in radical nephroureterectomy can lead to upstaging. BJU Int. 2010;105(6):812–817. doi: 10.1111/j.1464-410X.2009.08821.x. [DOI] [PubMed] [Google Scholar]
- 28.Xia L, Taylor B, Pulido J, et al. Impact of surgical waiting time on survival in patients with upper tract urothelial carcinoma: a national cancer database study. Urol Oncol. 2018;36(1) doi: 10.1016/j.urolonc.2017.09.013. 10.e15-10.e22. [DOI] [PubMed] [Google Scholar]
- 29.Smaldone MC, Kutikov A, Egleston BL, et al. Small renal masses progressing to metastases under active surveillance: a systematic review and pooled analysis. Cancer. 2012;118(4):997–1006. doi: 10.1002/cncr.26369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alam R, Patel HD, Osumah T. Comparative effectiveness of management options for patients with small renal masses: a prospective cohort study. BJU Int. 2019;123(1):42–50. doi: 10.1111/bju.14490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mehrazin R, Smaldone MC, Kutikov A, et al. Growth kinetics and short-term outcomes of cT1b and cT2 renal masses under active surveillance. J Urol. 2014;192(3):659–664. doi: 10.1016/j.juro.2014.03.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Touma NJ, Hosier GW, Di Lena MA, et al. Growth rates and outcomes of observed large renal masses. Can Urolog Assoc J. 2018;13(8):276–281. doi: 10.5489/cuaj.5545. Advance online publication. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reese AC, Whitson JM, Meng MV. Natural history of untreated renal cell carcinoma with venous tumor thrombus. Urol Oncol. 2013;31(7):1305–1309. doi: 10.1016/j.urolonc.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 34.Haddad AQ, Wood CG, Abel EJ, et al. Oncologic outcomes following surgical resection of renal cell carcinoma with inferior vena caval thrombus extending above the hepatic veins: a contemporary multicenter cohort. J Urol. 2014;192(4):1050–1056. doi: 10.1016/j.juro.2014.03.111. [DOI] [PubMed] [Google Scholar]
- 35.Méjean A, Ravaud A, Thezenas S, et al. Sunitinib alone or after nephrectomy in metastatic renal-cell carcinoma. N Engl J Med. 2018;379(5):417–427. doi: 10.1056/NEJMoa1803675. [DOI] [PubMed] [Google Scholar]
- 36.Bex A, Mulders P, Jewett M, et al. Comparison of immediate vs deferred cytoreductive nephrectomy in patients with synchronous metastatic renal cell carcinoma receiving sunitinib: the SURTIME randomized clinical trial. JAMA Oncol. 2019;5(2):164–170. doi: 10.1001/jamaoncol.2018.5543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Öztürk Ç, Fleer J, Hoekstra H, et al. Delay in diagnosis of testicular cancer; a need for awareness programs. PLoS One. 2015;10(11) doi: 10.1371/journal.pone.0141244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mazzone E, Mistretta F, Knipper S, et al. Contemporary assessment of long-term survival rates in patients with stage I nonseminoma germ-cell tumor of the testis: population-based comparison between surveillance and active treatment after initial orchiectomy. Clin Genitourin Cancer. 2019;17(6):e1153–e1162. doi: 10.1016/j.clgc.2019.08.009. https://doi.org/10/1016/j.clgc.2019.08.009. [DOI] [PubMed] [Google Scholar]
- 39.Mistretta FA, Mazzone E, Palumbo C, et al. Contemporary assessment of survival rates in stage I testicular seminoma: a Population-based comparison between surveillance and active treatment after orchiectomy. Clin Genitourin Cancer. 2019;17(4):e793–e801. doi: 10.1016/j.clgc.2019.04.015. [DOI] [PubMed] [Google Scholar]
- 40.Einhorn LH, Donohue J. Cis-diamminedichloroplatinum, vinblastine, and bleomycin combination chemotherapy in disseminated testicular cancer. Ann Intern Med. 1977;87(3):293–298. doi: 10.7326/0003-4819-87-3-293. [DOI] [PubMed] [Google Scholar]
- 41.Reddy BV, Sivakanth A, Naveen Babu G, et al. Role of chemotherapy prior to orchiectomy in metastatic testicular cancer-is testis really a sanctuary site? Ecancermedicalscience. 2014;8:407. doi: 10.3332/ecancer.2014.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Herr HW, Sheinfeld J, Puc HS, et al. Surgery for a post-chemotherapy residual mass in seminoma. J Urol. 1997;157(3):860–862. [PubMed] [Google Scholar]
- 43.Heidenreich A, Pfister D, Witthuhn R, et al. Postchemotherapy retroperitoneal lymph node dissection in advanced testicular cancer: radical or modified template resection. Eur Urol. 2009;55(1):217–224. doi: 10.1016/j.eururo.2008.09.027. [DOI] [PubMed] [Google Scholar]
- 44.Mosharafa AA, Foster RS, Leibovich BC, et al. Is post-chemotherapy resection of seminomatous elements associated with higher acute morbidity? J Urol. 2003;169(6):2126–2128. doi: 10.1097/01.ju.0000060121.33899.4b. [DOI] [PubMed] [Google Scholar]
- 45.Heidenreich A, Paffenholz P, Nestler T, et al. Primary and postchemotherapy retroperitoneal lymphadenectomy for testicular cancer. Oncol Res Treat. 2018;41:370–378. doi: 10.1159/000489508. [DOI] [PubMed] [Google Scholar]
- 46.Hendry WF., Norman AR, Dearnaley DP, et al. Metastatic nonseminomatous germ cell tumors of the testis: results of elective and salvage surgery for patients with residual retroperitoneal masses. Cancer. 2002;94(6):1668–1676. doi: 10.1002/cncr.10440. [DOI] [PubMed] [Google Scholar]
- 47.Ries LAG, Harkins D, Krapcho M, Mariotto A, et al. National Cancer Institute; Bethesda, MD: 2006. SEER cancer statistics review, 1975-2003.https://seer.cancer.gov/csr/1975_2003/ based on November 2005 SEER data submission, posted to the SEER website. [Google Scholar]
- 48.Lucky MA, Rogers B, Parr NJ. Referrals into a dedicated British penile cancer centre and sources of possible delay. Sex Transm Infect. 2009;85(7):527–530. doi: 10.1136/sti.2009.036061. [DOI] [PubMed] [Google Scholar]
- 49.Leijte J, Kirrander P, Antonini N, et al. Recurrence patterns of squamous cell carcinoma of the penis: recommendations for follow-up based on a two-centre analysis of 700 patients. Eur Urol. 2008;54:161–169. doi: 10.1016/j.eururo.2008.04.016. [DOI] [PubMed] [Google Scholar]
- 50.Chipollini J, Tang DH, Gilbert SM, et al. Delay to inguinal lymph node dissection greater than 3 months predicts poorer recurrence-free survival for patients with penile cancer. J Urol. 2017;198(6):1346–1352. doi: 10.1016/j.juro.2017.06.076. [DOI] [PubMed] [Google Scholar]
- 51.Rana RE., Ather MH, Enam SA. Change in surgical practice amidst COVID 19; example from a tertiary care centre in Pakistan. Ann Med Surg. 2020;54:79–81. doi: 10.1016/j.amsu.2020.04.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.COVID-19 and Surgery n.d.https://www.facs.org/COVID-19.Accessed July 19, 2020.
- 53.Ribal MJ, Cornford P, Briganti A, et al. European association of urology guidelines office rapid reaction group: an organisation-wide collaborative effort to adapt the European association of urology guidelines recommendations to the coronavirus disease 2019 era. Eur Urol. 2020;78(1):21–28. doi: 10.1016/j.eururo.2020.04.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Stensland KD, Morgan TM, Moinzadeh A, et al. Considerations in the triage of urologic surgeries during the COVID-19 pandemic. Eur Urol. 2020;77(6):663–666. doi: 10.1016/j.eururo.2020.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shinder BM, Patel HV, Sterling J, et al. Urologic oncology surgery during COVID-19: a rapid review of current triage guidance documents. Urol Oncol. 2020;38(7):609–614. doi: 10.1016/j.urolonc.2020.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Katims AB, Razdan S, Eilender BM, et al. Urologic oncology practice during COVID-19 pandemic: a systematic review on what can be deferrable vs. nondeferrable. Urol Oncol. 2020;38(10):783–792. doi: 10.1016/j.urolonc.2020.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gravas S, Fournier G, Oya M, et al. Prioritising urological surgery in the COVID-19 era: a global reflection on guidelines. Eur Urol Focus. 2020;6(5):1104–1110. doi: 10.1016/j.euf.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Teoh JY, Ong W, Gonzalez-Padilla D, et al. A global survey on the impact of COVID-19 on urological services. Eur Urol. 2020;78(2):265–275. doi: 10.1016/j.eururo.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dotzauer R, Böhm K, Brandt MP, et al. Global change of surgical and oncological clinical practice in urology during early COVID-19 pandemic. World J Urol. 2020:1–7. doi: 10.1007/s00345-020-03333-6. Advance online publication. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Roscigno M, Naspro R, Piccichè A, et al. A snapshot from the department of urology in Bergamo evaluating the timeline of the SARS-CoV-2 outbreak: which patients are we missing? Eur Urol Focus. 2020;6(5):1120–1123. doi: 10.1016/j.euf.2020.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cook DA, Wittich CM, Daniels WL, et al. Incentive and reminder strategies to improve response rate for internet-based physician surveys: a randomized experiment. J Med Internet Res. 2016;18(9):e244. doi: 10.2196/jmir.6318. [DOI] [PMC free article] [PubMed] [Google Scholar]