Highlights
-
•
Bi-phasic illness observed in COVID 19 leading to ARDS after week two of illness.
-
•
Haemochromatosis compromises host defence mechanisms and predisposes to infection.
-
•
Excess iron may increase SARS-CoV-2 replication leading to a hyperinflammatory state.
-
•
Hepatotoxicity may have been exacerbated by Anakinra with COVID-19 and HLH.
Keywords: Hereditary haemochromatosis, Coronavirus, Haemophagocytic lymphohistiocytosis
Abstract
Background
Syndromes of iron overload have been shown to increase the risk of severe clinical disease in viral infections. Immune dysfunction is similarly described in hereditary haemochromatosis (HH). We present here the case of a 51-year-old man who developed severe coronavirus disease 2019 (COVID-19) complicated by suspected haemophagocytic lymphohistiocytosis (HLH). He was found to have HH post-mortem and we propose a link between his iron overload and the development of severe COVID-19.
Case report
The initial clinical presentation consisted of cough, shortness of breath and fever. Pancytopenia, markedly elevated ferritin and d-dimer were present. Computed tomography (CT) showed bilateral ground glass changes consistent with COVID-19, widespread lymphadenopathy and splenomegaly. A subsequent combined nose and throat swab was positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). HLH was suspected based upon the H-score and Anakinra, an IL-1 receptor antagonist, was commenced. Liver function acutely worsened and magnetic resonance cholangiopancreatography (MRCP) revealed hepatic haemosiderosis. Intense splenic and cervical lymph node uptake were seen on a positron emission tomography (PET) scan and high doses of intravenous steroids were administered due to concerns over haematological malignancy.
Results
Day fourteen of admission heralded the start of progressive clinical deterioration with rapid increase in oxygen demands. Continuous positive airway pressure (CPAP) was trialled without success and the patient unfortunately died seventeen days into admission. Results returned after his death showed homozygous C282Y mutation of the HFE gene consistent with a diagnosis of HH. Post-mortem examination revealed widespread haemosiderin deposition in the liver along with lung pathology in keeping with severe COVID-19 and widespread splenic infarctions.
Conclusion
An association between HH and COVID-19 is not currently described in the literature. What does exist, however, is an evidence base for the detrimental impacts iron overload has on viral infections in general and the negative effects of HH on the immune system. We therefore postulate that the underlying metabolic and immune disturbances seen in HH should be considered a potential risk factor for the development of severe COVID-19. This case also adds to the evidence that hyperinflammation appears to be a unique and interesting characteristic of this novel viral disease.
Introduction
The key role of iron in viral infections is well described, although not fully understood. Iron is an essential factor for successful viral replication within living hosts and replete intracellular iron stores provide favourable conditions for this process 1, 2.
Excess iron is known to increase the risk of severe clinical disease and worsen prognosis in several viral illnesses [2]. Immune function is also understood to be affected by iron overload with a link to several immune defects and autoimmune conditions [3]. We present the case of a 51-year-old male who developed severe, ultimately fatal, coronavirus disease 2019 (COVID-19) complicated by suspected haemophagocytic lymphohistiocytosis (HLH). He was subsequently found to have undiagnosed hereditary haemochromatosis (HH) post-mortem and we hypothesise a link between his iron overload and the development of severe COVID-19.
Case
This 51-year-old man presented with a one-week history of worsening shortness of breath, persistent dry cough and pyrexia. He had a past medical history of an episode of community acquired pneumonia in 2018 requiring a short hospital admission. He was not taking any regular medications, consumed around 80 units of alcohol per week, smoked 5–10 cigarettes per day and had a normal body mass index.
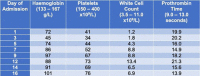
On examination he appeared acutely ill, pale and mildly jaundiced with non-tender hepatomegaly palpable 1 cm below the costal margin. Investigations revealed pancytopenia (table 1 ) with a mildly elevated bilirubin and gamma-glutamyl transferase (GGT). Ferritin was 21,095 μg/l (reference range 30–400 μg/l), D-dimer 13,344 ng/ml (reference range 0–500 ng/ml) and transferrin saturation 100%. HIV, hepatitis B and C screening were negative. EBV serology was IgG positive, but IgM negative. Blood film showed reactive changes with no immature forms and blood cultures were negative.
Table 1.
Haematology laboratory results throughout admission.
 |
Admission chest X-ray showed mild right basal consolidation with hilar lymphadenopathy (Fig. 1 a). Computed tomography (CT) was reported as showing lymphadenopathy in the neck, thorax and upper abdomen as well as bilateral ground glass opacities in the upper lobes of the lungs (Fig. 1b), sclerotic bony lesions in the spine and iliac bones, and likely splenic infarcts. The CT report was concluded as being concerning for haematological malignancy and COVID-19. Transthoracic echocardiogram showed no valvular abnormality or evidence of endocarditis and three sets of repeat blood cultures were negative.
Fig. 1.
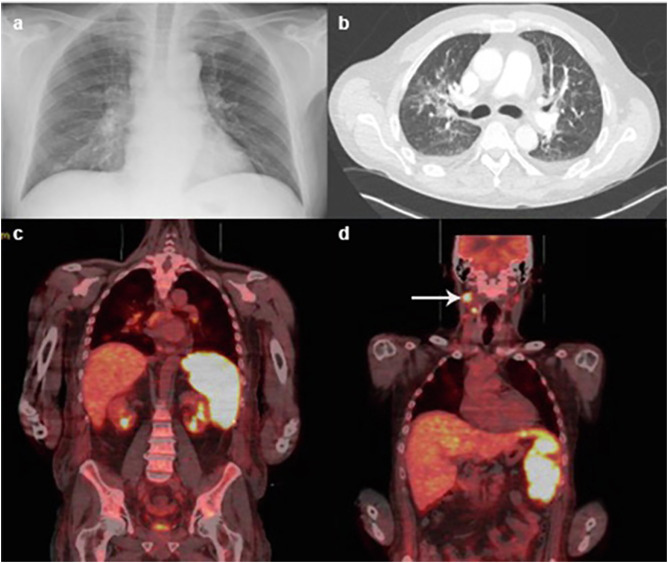
Radiology images throughout admission. (a) chest X-ray from day one of admission reported as showing mild right basal consolidation and hilar lymphadenopathy. (b) Axial view of CT chest from day two of admission showing bilateral ground glass opacities. (c) Coronal view of PET scan showing intense diffuse FDG uptake in the spleen. (d) coronal view of PET scan showing increased FDG uptake in cervical lymph nodes.
Swabs taken on day one and three of admission were negative by RT-PCR for SARS-CoV-2. However, these initial swab results were felt to represent false negatives given the strong suspicion of COVID-19 based on his clinical presentation, admission imaging and community transmission dynamics. A further third combined nose and throat swab was taken on day ten of admission and was positive. The patient was treated with one day of oral amoxicillin-clavulanic acid 625 mg, followed by a five-day course of intravenous piperacillin-tazobactam 4.5 g three times a day. HLH was suspected (H-score [4] on day two of admission suggested a 96–98% probability) and Anakinra, an IL-1 receptor antagonist, was administered at a dose of 100 mg once a day for six days. He also received a total of eight days intravenous methylprednisolone 800–1000 mg in addition to six days of oral prednisolone 50–75 mg. Over the course of the admission two bone marrow trephine biopsies were performed showing evidence of dysplasia but no clear evidence of haemophagocytosis or malignancy.
Liver function tests (LFTs) began to deteriorate on day five of admission. At this point he had completed five days of antibiotics and three days of Anakinra, suggesting potential drug induced hepatotoxicity. Clinically, biochemically and radiologically there were no features of chronic liver disease nor features of portal hypertension. Peak LFTs were: bilirubin 346 μmol/l, alanine transaminase (ALT) 161 u/l, GGT 624 u/l and alkaline phosphatase (ALP) 256 u/l (graph 1 ). Serum albumin was mildly reduced throughout admission from a normal baseline; 24 g/l at its lowest. Ultrasound demonstrated an enlarged fatty liver with normal ducts and splenomegaly of 20 cm. MRCP was advised by the hepatology team to further characterise the liver parenchyma given the uncertainty regarding the aetiology of his liver dysfunction. This was performed on day thirteen revealing hepatic haemosiderosis and follow-up investigations were arranged. A PET scan was performed after discussion with radiology to further investigate the tonsillar mass identified on prior imaging and look for evidence of possible malignancy. It demonstrated intense fluorodeoxyglucose (FDG) uptake in the spleen (Fig. 1c) and cervical lymph nodes (Fig. 1d). The bone lesions seen on CT were not PET-positive and were deemed to be benign after further discussion with a specialist radiologist. Further radiology opinion regarding the splenic findings concluded that they would be in keeping with a hyperinflammatory syndrome given the diffuse intense uptake. Unfortunately, a planned cervical lymph node biopsy was ultimately not possible due to significant interval reduction in lymphadenopathy, likely secondary to parenteral steroid and Anakinra administration.
Graph 1.
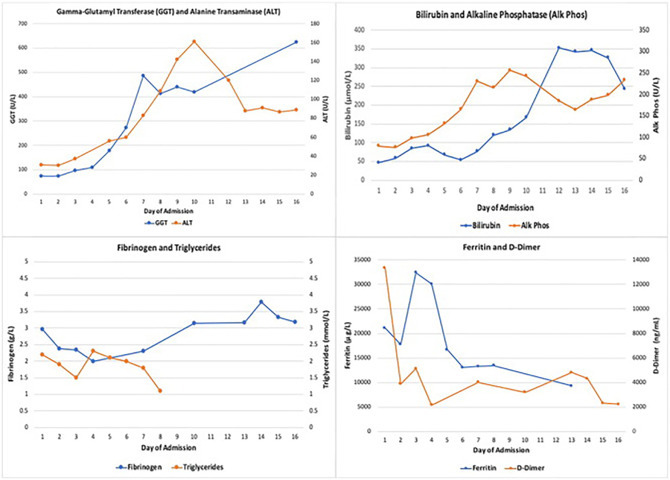
Trend in LFTs and HLH markers throughout admission.
Repeat chest radiography on day eleven showed increasing diffuse bilateral consolidation, typical of COVID-19. Progressive respiratory deterioration began on day fourteen of admission (day twenty-one of symptoms) with the patient becoming increasingly hypoxic, requiring 10–15 litres/minute of oxygen by a non-rebreathe mask. He was trialled with CPAP but showed no improvement, became progressively encephalopathic and unfortunately passed away on day seventeen of admission.
Approximately three weeks following his death, results of genetic testing were returned that revealed a homozygous C282Y mutation of the Human Homeostatic Iron Regulator (HFE) gene. This was deemed confirmatory of a diagnosis of hereditary haemochromatosis in combination with a serum ferritin level of 21,095 ug/l, transferrin saturation of 100% and iron excess within the liver identified on both MRCP and at post-mortem.
A post-mortem was performed given the complexity of his admission and the unclear picture surrounding potential intercurrent malignant disease. The liver showed extensive deposition of haemosiderin in hepatocytes consistent with hereditary haemochromatosis (Fig. 2 a–c) and fatty changes consistent with a history of alcohol excess (Fig. 2b). There was no macroscopic or microscopic evidence of hepatic cirrhosis. Post-mortem histology of the liver also showed fibrous expansion of portal areas and occasional fine fibrous bridging consistent with Ishak stage 3/6. The lungs were congested and oedematous. Lung histology showed acute diffuse alveolar damage with formation of hyaline membranes (Fig. 3 a) and bronchiolar fibrin (Fig. 3c). Occasional small fibrin thrombi were also present (Fig. 3b). The findings are consistent with COVID-19. The spleen showed two wedge shaped infarctions (Fig. 4 a and b) and widespread small foci of necrosis (Fig. 4c). Few blood vessels adjacent to areas of infarctions showed intimal haemorrhage and intimal lymphocytic inflammatory cell infiltrate (Fig. 5 c and d) however, the significance of the vascular changes in the spleen is uncertain due to the extensive necrosis. There was no evidence of neoplasia including lymphoma. Only reactive hilar lymph nodes were identified. There was no lymphadenopathy elsewhere. The kidneys showed occasional glomerular and peritubular capillary thrombi (Fig. 5a and b). There was no evidence of haemosiderin deposition in other organs.
Fig. 2.
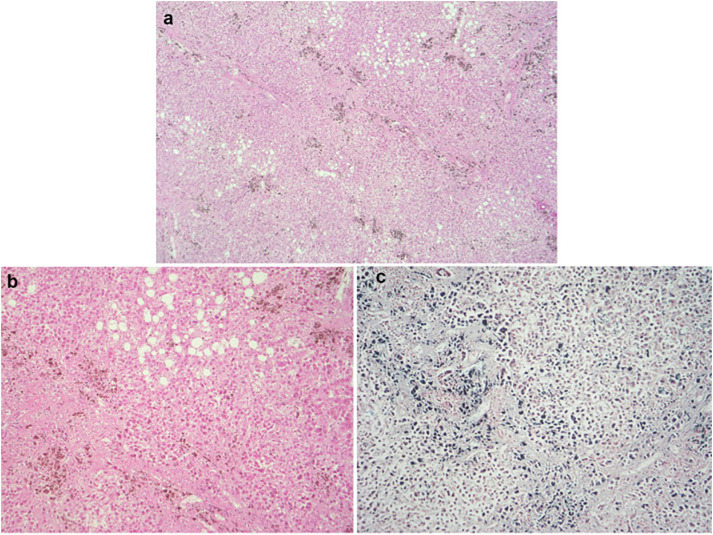
Microscopic appearances of liver tissue from post-mortem. (a and b) Diffuse haemosiderin deposition in hepatocytes and patchy fatty changes (haematoxylin and eosin stain). (c) Pearl's stain highlights haemosiderin deposition.
Fig. 3.
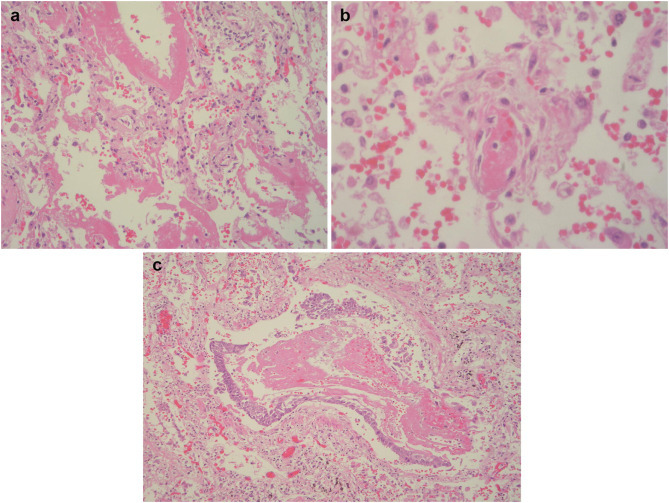
Microscopic appearances of lung tissue from post-mortem. (a) Hyaline membranes in alveoli. (b) Fibrin thrombus in a capillary, congestion and alveolar haemorrhage. (c) Accumulation of fibrin in a bronchiole and epithelial reactive metaplasia (haematoxylin and eosin stain).
Fig. 4.
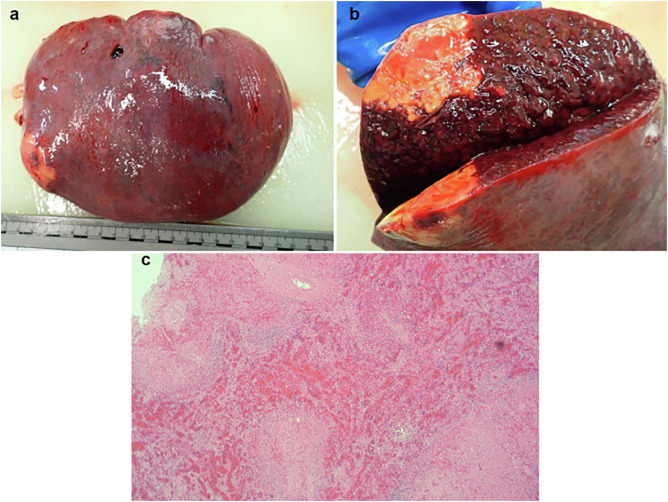
Macroscopic and microscopic appearances of splenic tissue from post-mortem. (a and b) Enlarged spleen and cut section showing wedge shaped infarction and several small foci of necrosis. (c) Several foci of necrosis in the spleen with marked haemorrhage and congestion in the adjacent splenic parenchyma (haematoxylin and eosin stain).
Fig. 5.
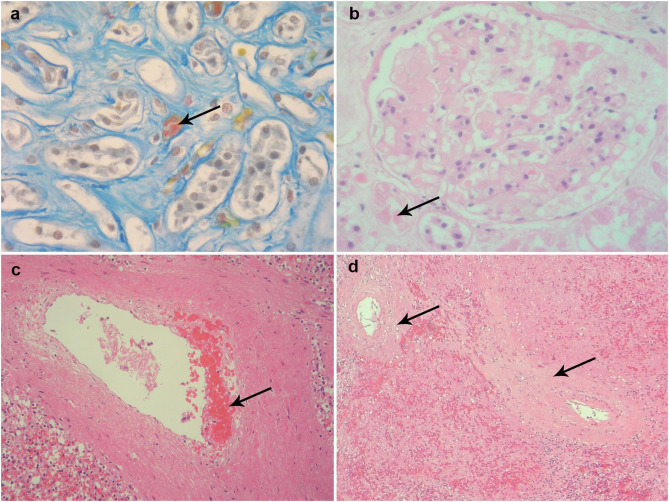
Microscopic appearances of vascular structures from various tissue samples at post-mortem. (a and b) Histological changes in the kidneys with fibrin thrombi in peritubular capillaries (Martius Scarlet Blue) and in glomerular capillaries (haematoxylin and eosin stain) respectively. (c and d) Histological changes in splenic blood vessel showing intimal haemorrhage and lymphocytic infiltration of uncertain significance (haematoxylin and eosin stain) respectively.
Discussion
The case that we present had an atypical COVID-19 clinical course and the patient's underlying diagnosis of hereditary haemochromatosis may have predisposed him to developing severe bi-phasic manifestations of COVID-19 and subsequent fatal outcome.
Hereditary haemochromatosis results in the accumulation of iron in essential organs. This pathological process results from the over-absorption of iron from the intestine and can lead to progressive end-organ damage [5]. HH can be associated with several different mutations but most commonly the C282Y mutation of the HFE gene. This mutation leads to abnormal structure of the HFE protein which, under normal circumstances, is expressed ubiquitously on cell surfaces. Wild-type HFE acts as an iron ‘sensor’ and negatively regulates iron uptake, as well as regulating the production of the hormone hepcidin [6]. Loss of structural integrity of the mutant HFE protein results in reduced expression on the extracellular surface and causes cellular uptake of iron to increase [7]. Secondary effects include the partial or complete loss of hepcidin production resulting in reduced macrophage iron storage and increased iron in the bloodstream [3]. Due to low penetrance, clinical disease does not occur in all individuals with this mutation.
Conditions like haemochromatosis compromise host defence mechanisms, and predispose to infection, autoimmune conditions and some cancers. It has been demonstrated that low levels of hepcidin may be associated with an increased susceptibility to infection [8]. There is also increasing evidence of a direct link between immune function and HH, with the HFE protein existing at the interface between the immune system and iron metabolism [3]. This relates, in part, to the similarity between the structure of the HFE protein and major histocompatibility complex (MHC) class I molecules. Individuals with HH have been shown to have defective cellular antigen presentation secondary to reduced MHC class I molecule cell surface expression [9]. This may have implications for mounting an initial host immune response to invading pathogens.
Abnormal T lymphocyte function is also well described in HH and can arise from a multitude of interferences, most significantly affecting CD8 T lymphocytes. Defects in CD8 T lymphocyte differentiation is thought to be one such example which results in reduced numbers of circulating and hepatic CD8 T lymphocytes in these patients [10]. Additional abnormalities in HH include diminished T cell activation and signalling, as well as reduced cytotoxic capacity of cytotoxic T lymphocytes [3]. Direct effects from excess iron can also negatively impact the immune system, manifesting as inhibited surface expression of adhesion molecules and the CD4 co-receptor on T lymphocytes [3].
Bacteria, fungi and viruses have been shown to have increased growth with excess levels of iron 11, 12. Viruses depend on iron in order to efficiently replicate within living host cells. It has been shown that iron overload in human immunodeficiency virus (HIV), hepatitis B (HBV) and hepatitis C (HCV) is a risk factor for severe disease and increased mortality. Homozygous and heterozygous mutations in HFE C282Y have been shown to cause hepatic iron overload promoting steatosis and liver fibrosis in HCV-infected patients [1]. It has been further suggested that limiting iron availability to infected cells by using iron chelators could be a beneficial therapy in viral infections [2].
In this case the clinical presentation, laboratory results and post-mortem findings are certainly in keeping with a syndrome on the hyperinflammatory spectrum. This encompasses both HLH and macrophage activation syndrome (MAS). Despite the absence of confirmatory bone marrow diagnosis, it is important to note that haemophagocytosis is not always demonstrated in patients with HLH and MAS and its absence does not exclude diagnosis 13, 14. Hyperinflammation appears to be the common link between HLH and MAS, as opposed to haemophagocytosis [15]. As such, utilising diagnostic criteria that consider the clinical picture without relying solely on tissue diagnosis is important. The diagnosis of HLH in adults is based on the HLH-2004 diagnostic criteria in conjunction with clinical judgment. Patients need to meet ≥ 5 of 8 diagnostic criteria [16]. However due to this patient's underlying haemochromatosis the ferritin level would have been elevated making the score less reliable. It is difficult to consider the association of this acute COVID-19 illness with pancytopenia, deranged liver enzymes, elevated LDH, hyperferritinaemia, splenomegaly and widespread lymphadenopathy without inferring a syndrome of significant immune dysregulation. Treatment of MAS-HLH often includes high-dose pulse methylprednisolone, cyclosporine A can be added in patients with an insufficient immediate response, as well as IL-1–blocking therapy with anakinra. Experience with anti–IL-6 blockade with tocilizumab is increasing [16].
The case shows a patient with hyperinflammation and COVID-19 with an underlying diagnosis of HH that was treated with Anakinra, that went on to develop hepatotoxicity, possibly as a result. Interestingly another patient we reported with COVID-19-associated HLH also developed deranged liver enzymes which appeared to respond to a change in antibiotics [17]. In a small study of 80 adults looking at characteristics with secondary HLH deranged liver function tests were identified in 70 (87%) of patients [18]. Anakinra is a recombinant IL-1 receptor antagonist and has previously been evaluated as a method to counteract the cytokine storm observed in severe sepsis and septic shock. As IL-1 serum concentrations have been shown to be significantly increased in patients with COVID-19 compared to healthy controls it is thought that Anakinra may benefit these patients [19]. However, there is limited data about the toxicity of Anakinra in COVID-19 patients. The reason for the deterioration of the patient’s liver function is likely multifactorial due to HH, medication and part of HLH syndrome.
High dose intravenous and then oral steroids were used to treat a potential haematological malignancy in this man, and were administered before respiratory deterioration and oxygen dependence. Recently the RECOVERY trial announced there was significant benefit in COVID-19 patients who required respiratory support being prescribed dexamethasone. Initial data indicates one death being prevented by treatment of around eight ventilated patients or around twenty-five patients requiring oxygen alone [20]. There was no benefit in the use of dexamethasone in patients not requiring oxygen, and a potential signal of harm. A meta-analysis by WHO of clinical trials involving corticosteroids in COVID-19 is underway and eagerly awaited.
Whilst this complex case did initially carry some clinical uncertainties that were not allayed until post-mortem, one aspect that was evidently clear was the bi-phasic nature of his illness. Relatively mild symptoms of COVID-19 were observed at presentation and clinical stability, with little or no oxygen requirements, continued throughout the initial stages of his admission. Week three of his symptoms, however, heralded a dramatic and irreversible deterioration that resulted in acute respiratory failure and was the ultimate cause of his death. This pattern has been repeatedly observed as one subtype of the COVID-19 clinical syndrome [21]. It is felt to signify that effective host control of the virus in the initial stages of infection is crucial to preventing progression towards severe disease [21]. We propose that a degree of immune dysfunction secondary to HH impeded this response. Excess intracellular and circulating iron may have provided ample material to fuel explosive viral replication in the presence of an already dysregulated immune system, subsequently provoking a hyperinflammatory HLH-like state. The potential benefits or harms of high dose steroids in this situation, combined with emerging RECOVERY trial data is unclear.
A definitive link between this man’s underlying HH and the observed hyperinflammatory, bi-phasic COVID-19 illness is not certain. As far as we can tell at the time of writing there is no current publication that outlines such a link. It is the opinion of our group, however, that there is sufficient evidence existing in current literature relating to the effects of hereditary haemochromatosis on other viral infections and immune function to infer that a potential pathological association may exist, and further investigation is required.
Informed consent
The patient’s next of kin has provided informed consent for us to write this report. The patient was deceased at the time of writing.
Funding source
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
None.
CRediT authorship contribution statement
Matthew J. Riley: Conceptualization, Formal analysis, Investigation, Writing - original draft, Visualization. Scott Rory Hicks: Conceptualization, Formal analysis, Investigation, Writing - original draft, Visualization. Sharon Irvine: Conceptualization, Writing - review & editing, Supervision. Tom J. Blanchard: Conceptualization, Writing - review & editing, Supervision. Edward Britton: Formal analysis, Investigation, Writing - review & editing. Howida Shawki: Formal analysis, Investigation, Writing - review & editing, Visualization. Muhammad Sajid Pervaiz: Formal analysis, Investigation, Writing - review & editing. Tom Fletcher: Conceptualization, Writing - review & editing, Supervision.
References
- 1.Schmidt S.M. The role of iron in viral infections. Front. Biosci. 2020;25(4):893–911. doi: 10.2741/4839. [DOI] [PubMed] [Google Scholar]
- 2.Drakesmith H., Prentice A. Viral infection and iron metabolism. Nat Rev Microbiol. 2008;6(7):541–552. doi: 10.1038/nrmicro1930. [DOI] [PubMed] [Google Scholar]
- 3.Reuben A. The hemochromatosis protein HFE 20 years later: an emerging role in antigen presentation and in the immune system. Immun Inflammation Dis. 2017;5(3):218–232. doi: 10.1002/iid3.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fardet L. Development and validation of the HScore, a score for the diagnosis of reactive hemophagocytic syndrome. Arthritis Rheumatol. 2014;66(9):2613–2620. doi: 10.1002/art.38690. [DOI] [PubMed] [Google Scholar]
- 5.Pietrangelo A. Hereditary hemochromatosis: pathogenesis, diagnosis, and treatment. Gastroenterology. 2010;139(2) doi: 10.1053/j.gastro.2010.06.013. [DOI] [PubMed] [Google Scholar]
- 6.Hentze M.W. Two to Tango: regulation of mammalian iron metabolism. Cell. 2010;142(1):24–38. doi: 10.1016/j.cell.2010.06.028. [DOI] [PubMed] [Google Scholar]
- 7.Lebrón J.A. Crystal structure of the hemochromatosis protein HFE and characterization of its interaction with transferrin receptor. Cell. 1998;93(1):111–123. doi: 10.1016/s0092-8674(00)81151-4. [DOI] [PubMed] [Google Scholar]
- 8.Ashrafian H. Hepcidin: the missing link between hemochromatosis and infections. Infect Immun. 2003;71(12):6693–6700. doi: 10.1128/IAI.71.12.6693-6700.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Almeida Sérgio F, et al. HFE cross-talks with the MHC class I antigen presentation pathway. Blood 2005; 106(3):971–7. [DOI] [PubMed]
- 10.Macedo M.F. Low numbers of CD8 T lymphocytes in hereditary haemochromatosis are explained by a decrease of the most mature CD8 effector memory T cells. Clin Exp Immunol. 2010;159(3):363–371. doi: 10.1111/j.1365-2249.2009.04066.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Khan F.A., Fisher M.A., Khakoo R.A. Association of hemochromatosis with infectious diseases: expanding spectrum. Int J Infect Diseases. 2007;11(6):482–487. doi: 10.1016/j.ijid.2007.04.007. [DOI] [PubMed] [Google Scholar]
- 12.Weinberg E.D. Iron loading and disease surveillance. Emerg Infect Dis. 1999;5(3):346–352. doi: 10.3201/eid0503.990305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Anon. Macrophage activation syndrome clinical presentation: history, physical, causes; 2019. Available at: https://emedicine.medscape.com/article/1380671-clinical [accessed 26 June 2020].
- 14.Janka G.E. Familial hemophagocytic lymphohistiocytosis. Eur J Pediatr. 1983;140(3):221–230. doi: 10.1007/BF00443367. [DOI] [PubMed] [Google Scholar]
- 15.Weaver L.K., Behrens E.M. Hyperinflammation, rather than hemophagocytosis, is the common link between macrophage activation syndrome and hemophagocytic lymphohistiocytosis. Curr Opin Rheumatol. 2014;26(5):562–569. doi: 10.1097/BOR.0000000000000093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.La Rosee P. 2019 Recommendations for the management of hemophagocytic lymphohistiocytosis in adults. Blood. 2019;133:2454–2477. doi: 10.1182/blood.2018894618. [DOI] [PubMed] [Google Scholar]
- 17.Haigh K. Hyperinflammation with COVID-19: The key to patient deterioration? Clin Infect Pract. 2020;7–8 doi: 10.1016/j.clinpr.2020.100033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Georgiadou S. Efficient management of secondary haemophagocytic lymphohistiocytosis with intravenous steroids and γ –immunoglobulin infusions. World J Clin Cases. 2019;7(21):3394–3406. doi: 10.12998/wjcc.v7.i21.3394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cavalli G. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. Lancet Rheumatol. 2020;2(6) doi: 10.1016/S2665-9913(20)30127-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Horby P, et al., Effect of dexamethasone in hospitalized patients with COVID-19: preliminary report. medRxiv 2020.06.22.20137273; doi: https://doi.org/10.1101/2020.06.22.20137273.
- 21.Chen J. Clinical progression of patients with COVID-19 in Shanghai, China. J Infect. 2020;80(5) doi: 10.1016/j.jinf.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]


