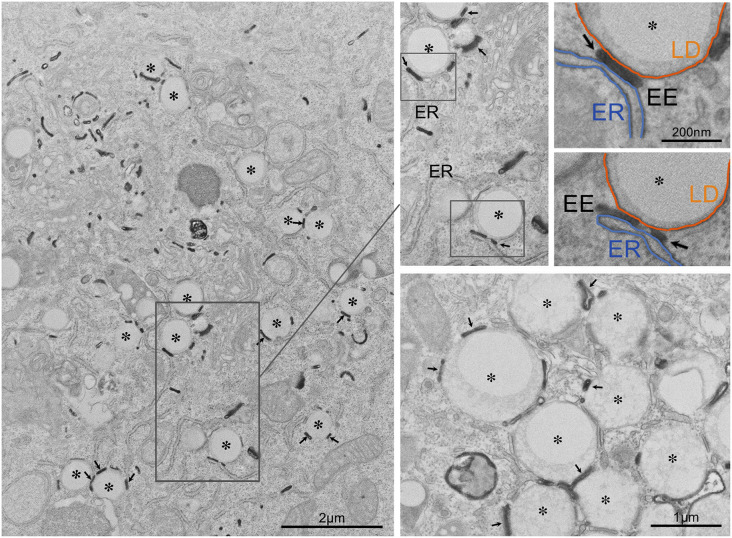
Lipid droplets (LDs) play a crucial role in the storage and distribution of lipids to control cellular metabolism and bioenergetics (1). How LDs organize the lipid supply to different cellular fates is just starting to be elucidated. Formation of membrane contact sites (MCSs; defined as regions where the two membranes of similar or different organelles are <30 nm from each other) between LDs and target organelles, such as the endoplasmic reticulum (ER) or mitochondria, is emerging as an important mechanism regulating lipid transfer (1, 2). MCSs can ensure timely and local lipid supply, avoiding lipotoxicity and missorting of valuable energetic and metabolic substrates. In support of the biological relevance of these mechanisms, up to 26 different Rab GTPases reside on LDs, representing 20% of the LD proteome (3). Interestingly, a subpopulation of these proteins [e.g., Rab5 (a, b, and c), Rab8 (a and b), Rab11 (a), Rab13, Rab14, Rab15, and Rab21] is also associated with the early endosomal system. Early endosomes (EEs) are the major endocytic compartments mediating sorting of proteins, lipids, and lipoproteins. Early biochemical studies proposed the existence of physical interactions between LDs and EEs (4). The existence of these contacts in mammalian cells has not been further explored. The figure illustrates that EE tubules, defined by the presence of internalized transferrin, interact with LDs in mammalian cells. BHK cells were cotransfected with a plasmid encoding human transferrin receptor (TfR plus plasmid encoding GFP) and then incubated with transferrin-HRP for 30 min at 37°C. Cells were then processed for HRP detection and processed for electron microscopy using a mild fixation/low membrane-contrast staining method to optimize transferrin-HRP visualization. Transferrin-HRP-labeled EE tubules (arrows) were specifically associated with LDs (asterisks) as shown in the low magnification overview (left panel) and at higher magnification in the lower right panel. The two pseudocolored panels show higher magnification views of the neighboring panels, with ER in blue and the LD monolayer in orange; note the tripartite interaction with the EE tubule (EE, arrows). The images clearly demonstrate that LDs can form a distinct MCS with tubules of the EE but also that LD-EE contacts can interact in tripartite fashion with the ER. While hitherto elusive in mammalian cells, the existence and function of three-way contacts between lysosomes, LDs, and ER have been well described in yeast (2). With EEs acting as central hubs in lipid trafficking, these observations provide a new perspective on the complexity of LD interactions in mammalian cells.
EQUIPMENT: Transmission electron microscope (JEOL 1011) with Morada CCD camera (Olympus)
REAGENTS: Lipofectamine 2000 (Life Technologies), Human transferrin peroxidase conjugated (#009-0334, Rockland)
REFERENCES
- 1.Bosch M., Parton R.G., and Pol A.. Lipid droplets, bioenergetic fluxes, and metabolic flexibility. Semin. Cell Dev. Biol. Epub ahead of print. March 4, 2020; doi: 10.1016/j.semcdb.2020.02.010. [DOI] [PubMed] [Google Scholar]
- 2.Henne M. 2019. And three’s a party: lysosomes, lipid droplets, and the ER in lipid trafficking and cell homeostasis. Curr. Opin. Cell Biol. 59: 40–49. [DOI] [PubMed] [Google Scholar]
- 3.Bersuker K., Peterson C.W.H., To M., Sahl S.J., Savikhin V., Grossman E.A., Nomura D.K., and Olzmann J.A.. 2018. A proximity labeling strategy provides insights into the composition and dynamics of lipid droplet proteomes. Dev. Cell. 44: 97–112 e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liu P., Bartz R., Zehmer J. K., Ying Y. S., Zhu M., Serrero G., and Anderson R. G.. 2007. Rab-regulated interaction of early endosomes with lipid droplets. Biochim. Biophys. Acta. 1773: 784–793. [DOI] [PMC free article] [PubMed] [Google Scholar]