THIS PAST YEAR MARKED THE 20TH ANNIVERSARY OF THE DISCOVERY OF the Janus kinase (JAK)–signal transducer and activator of transcription (STAT) pathway.1 Arising from efforts to understand the molecular mechanisms of interferon action, the elucidation of this pathway has provided many insights into disease mechanisms and has become the basis for new pharmacologic agents. It is therefore an appropriate time to take stock of our knowledge of this pathway and to consider the ways in which these insights are affecting the practice of medicine.
CYTOKINES AND JAK–STAT SIGNALING
Interferons, erythropoietin, growth hormone, and prolactin, all discovered more than half a century ago, are a few of the dozens of cytokines that have been found to play critical roles in cell growth and differentiation, metabolism, hematopoiesis, host defense, and immunoregulation. It would be hard to overstate the influence of cytokines — as well as anticytokine and anticytokine-receptor antibodies — on medicine.
Understanding the molecular basis of cytokine action has been of great interest, not only for the purpose of basic research but also to further our understanding of disease pathogenesis and the development of new therapies. The term “cytokine” can be confusing because it refers to a broad collection of secreted factors that belong to different structural families and that use different types of receptors and distinct modes of signaling. One large subgroup, the type I and type II cytokine-receptor superfamily, encompasses receptors that bind interferons, many (but not all) interleukins, and colony-stimulating factors. These cytokines all use the same mechanism of signal transduction: the JAK–STAT pathway.2 Because erythropoietin, thrombopoietin, growth hormone, prolactin, and leptin use the same class of receptor, they can also be included in this family of cytokines. Other cytokines, such as tumor necrosis factor, interleukins 1 and 8, transforming growth factor β, and macrophage colony-stimulating factor, bind different classes of receptors and do not use the JAK–STAT pathway as their essential mode of signaling.3
Four JAKs — JAK1, JAK2, JAK3, and tyrosine kinase 2 (TYK2) — selectively associate with the cytoplasmic domains of various cytokine receptors (Fig. 1). JAK3 and TYK2 are primarily important for immune responses. JAK1 and JAK2 have broad functions, with roles that range from host defense and hematopoiesis to growth and neural development; deletion of Jak1 or Jak2 is lethal in mice.2 Cytokine binding activates JAKs, which in turn phosphorylate cytokine receptors. This process allows the selective binding of members of the STAT family: STAT1, STAT2, STAT3, STAT4, STAT5A, STAT5B, and STAT6. These DNA-binding proteins become tyrosine-phosphorylated, which allows them to dimerize, translocate to the nucleus, and regulate gene expression. In mice, complete deficiency of Stat3 or Stat5a and Stat5b is lethal, a finding that is consistent with the broad, critical functions of these proteins. STAT1, STAT2, STAT4, and STAT6 have more restricted functions, playing roles in host defense and immunoregulation.
Figure 1. Disorders Associated with Mutations of JAKs and STATs.
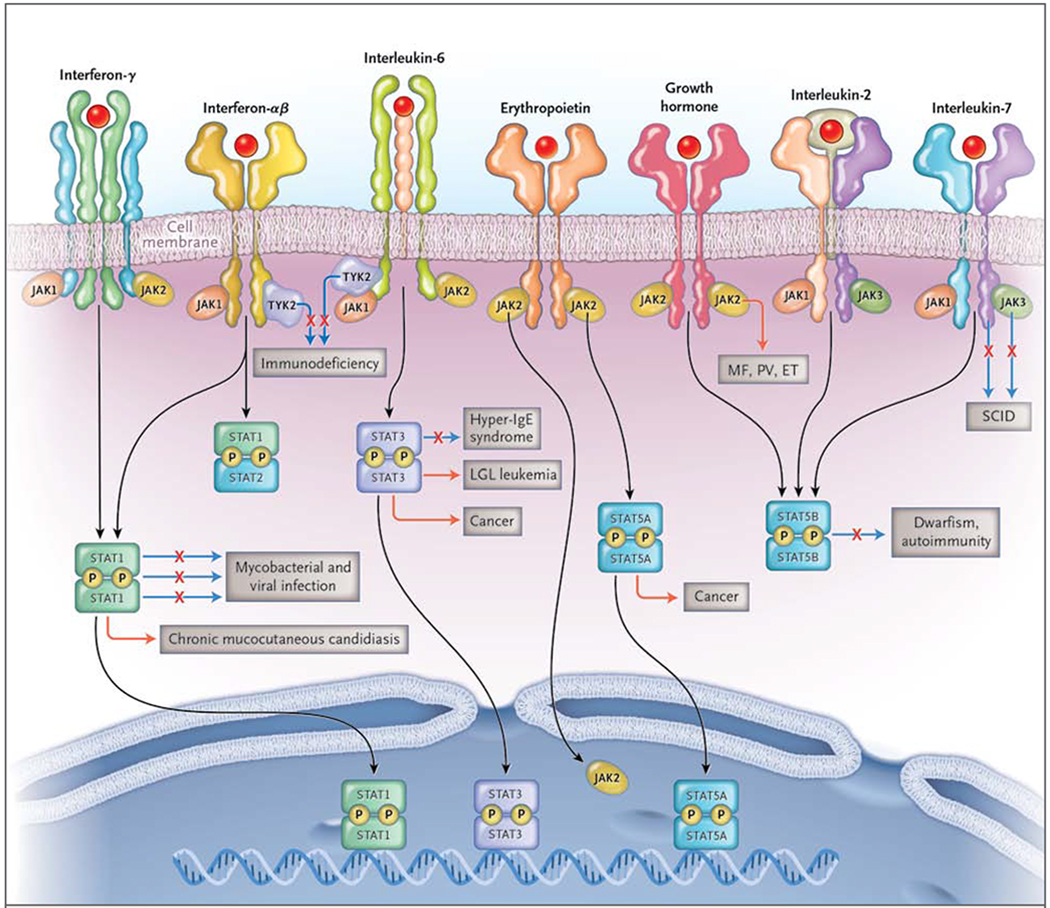
A major subset of cytokines includes those that signal through Janus kinases (JAKs) and signal transducers and activators (STATs). These cytokines include (but are not limited to) interferons, such as interferon-γ, interferon-α, and interferon-β; interleukin-6; erythropoietin; growth hormone; interleukin-2; and interleukin-7. There are four JAKs: JAK1, JAK2, JAK3, and tyrosine kinase 2 (TYK2). Activated JAKs phosphorylate (P) and activate STATs and other pathways. Some JAKs are associated with many different cytokine receptors, but JAK3 associates with only one subunit, the common interleukin-2Rγ chain, or γc. Loss-of-function mutations (denoted by a solid line, with an X) of the genes encoding γc and JAK3 result in severe combined immunodeficiency (SCID). Mutation of TYK2 also results in immunodeficiency. Gain-of-function JAK2 mutations (denoted by an orange line) underlie polycythemia vera (PV), essential thrombocytosis (ET), and myelofibrosis (MF). Constitutive activation and mutation of JAKs is also associated with a variety of cancers. Activated JAKs, in turn, activate STATs and other pathways. Activated STATs translocate to the nucleus, bind DNA, and regulate gene expression. Mutations of STAT1 result in several distinct disorders. Autosomal dominant loss-of-function mutations result in susceptibility to mycobacteria only. Autosomal recessive mutations cause susceptibility to mycobacteria and viruses. In contrast, autosomal dominant gain-of-function mutations cause chronic mucocutaneous candidiasis, other infections, and aneurysms. Loss-of-function mutations of STAT3 result in the hyper-IgE syndrome. Mutations in STAT3 also cause large granular lymphocytic (LGL) leukemia. Constitutive STAT3 and STAT5 activation (an orange line) is associated with many cancers. Mutations of STAT5B result in a syndrome characterized by dwarfism and autoimmunity. In addition to their effect on STATs, JAKs can have direct nuclear effects by phosphorylating histones.
PRIMARY IMMUNODEFICIENCY
Dramatic examples of the importance of JAKs and STATs have come from studying patients with primary immunodeficiencies.4 Among the four JAKs, mutations of JAK3 and TYK2 are known causes of primary immunodeficiency, including severe combined immune deficiency (SCID). Mutations of STAT1, STAT3, and STAT5B are also associated with specific clinical syndromes.
SCID AND JAK3 MUTATIONS
The cause of SCID is impaired development or function of lymphocytes. The receptor chain shared by interleukins 2, 4, 7, 9, 15, and 21 is the common interleukin-2Rγ chain, γc; the gene encoding this protein is located at Xq13. Mutations in the gene encoding interleukin-2Rγ (IL2RG) cause X-linked SCID, which is characterized by markedly diminished T-cell and natural killer (NK) cell numbers and function but preserved B-cell numbers with impaired function (T−B+NK−). The dependence of γc signaling on JAK3 was established when it was determined that most patients who have T−B+NK− SCID without γc mutations have JAK3 mutations.4 In contrast, mutations in the interleukin-7 receptor α chain specifically impair T-cell development, leading to T−B+NK+ SCID, since interleukin-15 signaling, which is required for NK-cell development, is maintained.
TYK2
There are only two instances in which autosomal recessive mutations in TYK2 have been reported in humans, and their phenotypes differed. One child had atopic dermatitis and moderately elevated IgE levels as well as severe bacterial, viral, and fungal infections.5 The other child had severe infection after vaccination with bacille Calmette–Guérin (BCG), neurobrucellosis, and infection with herpes simplex virus (HSV), but only very minor elevations in IgE levels and no atopy.6
STAT1
Human mutations in STAT1 were first identified in patients with disseminated BCG infection or nontuberculous mycobacterial infection in childhood.7 These mutations were dominant-negative for interferon-γ signaling but recessive for interferon-α signaling, which resulted in susceptibility to mycobacterial infection but normal viral control. Whereas dominant-negative mutations lead to relatively mild mycobacterial disease, complete STAT1 deficiency blocks both interferon-γ and interferon-α signaling. Patients with complete deficiency are susceptible to viral and mycobacterial infections, which usually lead to early death.8 Between the complete recessive loss of interferon-γ and interferon-α signaling and the presence of dominant-negative inhibition of interferon-γ signaling alone are the recessive hypomorphic mutations.9 These biallelic mutations result in recurrent infections with intramacrophagic bacteria (e.g., salmonella, BCG, and nontuberculous mycobacteria) and herpes viruses (e.g., HSV, cytomegalovirus, and varicella–zoster virus), but with treatment there is adequate immunity to support survival.10
Heterozygous gain-of-function or hypermorphic mutations that cause chronic mucocutaneous candidiasis have recently been described.11,12 Excessive STAT1 activation causes exaggerated responses to interferon-γ and inhibits the production of interleukin-17. Patients with hypermorphic mutations in STAT1 are susceptible to other complications, including autoimmunity, cerebral aneurysms, and squamous-cell carcinoma. These mutations are also associated with the disseminated dimorphic yeast infections coccidioidomycosis and histoplasmosis and with the IPEX-like (immune dysregulation, polyendocrinopathy, enteropathy, X-linked) syndrome, in which the transcription factor FOXP3 and regulatory T cells are intact.13 It will be of interest to learn precisely how hyperactivating mutations in STAT1 are related to epithelial, invasive, and opportunistic infections as well as cancer and vascular disease.
STAT3
The search for the genetic cause of the autosomal dominant hyper-IgE (or Job’s) syndrome culminated in the surprising identification of dominant-negative mutations in STAT3.14,15 The hyper-IgE syndrome is characterized by eczema, staphylococcal boils, cyst-forming pneumonias, chronic mucocutaneous candidiasis, and extremely high levels of IgE, along with numerous nonimmunologic features, including scoliosis, fractures, characteristic facies, failure of primary tooth deciduation, and coronary-artery aneurysms.16,17 The mutations that cause this complex, multisystem disease are located mainly in the DNA-binding and Src homology 2 (SH2) domains of STAT3, leading to interference with the functioning of the normal allele. STAT3 was known to be crucial for cytokines that drive the differentiation of interleukin-17–producing T cells. Accordingly, interleukin-17–producing cells are diminished in patients with the hyper-IgE syndrome, providing the first evidence of the critical role this cytokine plays in humans.18–20 Because STAT3 is also critical for CD8 T-cell memory, STAT3 mutations are associated with recurrent infection with varicella–zoster virus and increased circulating levels of Epstein–Barr virus.21 However, STAT3 mediates signaling through at least six classes of receptors,4 making it a multifaceted disease with features beyond immune-cell defects, including coronary-artery aneurysms without coronary atherosclerosis.
STAT5
Recessive STAT5B deficiency results in immunodeficiency, autoimmunity, and growth failure.22 Patients with STAT5B deficiency have severe opportunistic infections, variable lymphocyte counts, and normal-to-high levels of immunoglobulins. These effects are explained by the roles STAT5B plays in interleukin-2 and growth-hormone signaling.23,24 STAT5B is important for the expression of FOXP3 and interleukin-2Rα (CD25) and for the differentiation of regulatory T cells.25 STAT5 also down-regulates interleukin-17,26 and both factors underlie the autoimmunity associated with STAT5B deficiency.
IMMUNOREGULATION AND GENETIC LINKS TO IMMUNE-MEDIATED DISEASE
Animal models have provided powerful evidence that the JAK–STAT pathway and the cytokines that use this pathway play critical roles in the pathogenesis of autoimmunity, allergy, asthma, and other immune-mediated diseases. More recently, genomewide association studies have but-tressed the argument that cytokines and cytokine signaling are relevant to human disease. Inherited variation in genes encoding cytokines and cytokine receptors and their respective JAKs and STATs are associated with a significantly increased risk of immune-mediated disease.
Multiple genes in the interleukin-23 signaling pathway contribute to a surprising range of autoimmune diseases, including inflammatory bowel disease, psoriasis, ankylosing spondylitis, and Behçet’s disease.27–29 Polymorphisms of JAK2 and STAT3 are associated with these disorders. Similarly, STAT4 is activated by interleukin-12 and interferon type I, and polymorphisms of STAT4 are associated with systemic lupus erythematosus, rheumatoid arthritis, and Sjögren’s syndrome.30 Interleukins 4 and 13 activate STAT6, and STAT6 polymorphisms are associated with elevated IgE levels and atopic dermatitis.31,32
CANCER
In addition to having roles in host defense and autoimmunity, the JAKs have critical roles in hematologic cancers.33 The initial evidence of these roles derived from the recognition of the constitutive activation of JAKs and STATs in patients with cancer.34,35 JAK2 mutations are associated with myeloproliferative neoplasms, clonal cancers arising from hematopoietic progenitor cells, which include polycythemia vera, essential thrombocythemia, and primary myelofibrosis.36–39 The most frequent JAK2 mutation, V617F, occurs in more than 95% of patients with polycythemia vera and in 32 to 57% of patients with essential thrombocythemia or primary myelofibrosis. The selective association of the V617F mutation with myeloproliferative neoplasms is diagnostically useful, helping to distinguish these disorders from non-malignant syndromes that affect blood counts. Less frequently, myeloproliferative neoplasms harbor mutations in exon 12 of JAK2 in the absence of the V617F mutation; in these instances, patients typically have an isolated erythrocytosis, which may represent a distinct clinical syndrome.40
The presence of a myeloproliferative neoplasm in a first-degree relative increases the risk of disease by a factor of 5 to 7.41 An extended germline haplotype encompassing the JAK2 locus is responsible for much of this familial predisposition, increasing the risk of disease by a factor of 3 to 4.42–44 When a myeloproliferative neoplasm develops, the somatic V617F mutation is present preferentially on the germline-encoded JAK2 allele that confers susceptibility, suggesting that this allele harbors cis-acting elements that increase the likelihood of the V617F mutation.
The JAK2 V617F mutant is located within the JH2 “kinaselike” domain,45,46 which is catalytically active and can phosphorylate and activate the kinase domain.47 Thus, the mutation creates a constitutively active kinase that can render hematopoietic cells independent of exogenous growth factors, thereby causing polycythemia vera and other myeloproliferative processes in mice.48,49 These observations establish the oncogenic importance of the V617F mutation.
Somatically acquired mutations in JAK2 have been detected in high-risk patients with B-cell acute lymphoblastic leukemia (ALL) (9%) and in patients with B-cell ALL associated with Down’s syndrome (34%), most often affecting the R683 residue.50–52 Other JAKs can be activated by mutations in hematologic cancers, including JAK1 in patients with T-cell ALL, patients with B-cell ALL who have a poor prognosis, and patients with acute myeloid leukemia,52–55 and JAK3 in patients with T-cell ALL, patients with adult T-cell leukemia or lymphoma, and patients with NK-cell or Τ-cell lymphoma.56–58
The function of receptors that associate with JAKs can also be altered by chromosomal rearrangements or mutations in cancer, leading to constitutive JAK activity. Activating mutations affecting the thrombopoietin receptor MPL occur in approximately 9% of patients with myelofibrosis, all of whom lack the JAK2 V617F mutation, which leads to the constitutive activation of JAK2 by the MPL receptor.59 In approximately 50% of patients with B-cell ALL, the cytokine receptor CRLF2, which binds JAK2, is overexpressed by chromosomal rearrangements, including 34% of patients with Down’s syndrome and 9% of high-risk children with B-cell ALL.52,60–62 Many leukemias with alterations in CRLF2 also have JAK2 mutations, suggesting that this receptor functions as a platform for constitutive signaling by mutant JAK2. Approximately 10% of patients with T-cell ALL have mutant interleukin-7 receptor α subunits, leading to constitutive JAK1 activation.63,64 Gain-of-function mutations of the granulocyte colony-stimulating factor receptor are associated with acute myeloid leukemia in conjunction with severe congenital neutropenia.65 Outside the hematopoietic lineage, in-frame deletions affecting glycoprotein 130, the signaling component of the interleukin-6 receptor, are present in 60% of patients with inflammatory hepatocellular adenomas, causing JAK2 activation.66
JAKs can also be activated by autocrine cytokine secretion in several subtypes of lymphoma. In primary mediastinal B-cell lymphoma and Hodgkin’s lymphoma, autocrine interleukin-13 signaling activates JAK2, and in 30 to 50% of patients, its activity is further intensified by amplification of the JAK2 locus.67,68 Thus, JAK2 inhibition is lethal to the cell lines for both types of lymphoma.68
Autocrine secretion of interleukins 6 and 10 activates JAKs in the activated B-cell-like (ABC) subtype of diffuse large-B-cell lymphoma, promoting the survival of malignant cells.69–71 In many cases, this autocrine cytokine loop is initiated by activating mutations affecting MYD88, an adaptor protein in toll-like receptor signaling.70 The most common MYD88 mutant, termed L265P, occurs in 29% of patients with ABC diffuse large-B-cell lymphoma, and it is also present in 36% of patients with primary central nervous system lymphoma72 and 69% of patients with leg-type primary cutaneous lymphoma, both of which phenotypically resemble ABC diffuse large-B-cell lymphoma. MYD88 L265P is also common in Waldenstrom’s macroglobulinemia73 (90% of cases) and occurs in a subset of marginal-zone lymphomas70,74 (10 to 11% of cases) and chronic lymphocytic leukemias75,76 (3 to 10% of cases). In ABC diffuse large-B-cell lymphoma, this MYD88 mutant spontaneously coordinates an active signaling complex composed of the interleukin-1—associated kinases IRAK1 and IRAK4, thereby engaging the nuclear factor κB and p38 MAP kinase pathways and leading to the synthesis of interleukin-6 and interleukin-10 and autocrine JAK activation.70
The downstream effectors of JAK signaling in hematopoietic cancer include the PI-3 kinase and Ras pathways and the STAT transcription factors.33 The JAK2 V617F mutation does not cause myeloproliferative disease in mice lacking STAT5,77–79 and STAT3 is required for the survival of ABC diffuse large-B-cell lymphoma cells.69,80 STAT3 is mutated in 40% of large granular leukemias,81 with its SH2 domain altered at some of the same positions in which it is altered in patients with the hyper-IgE syndrome, albeit with different amino acid substitutions. Constitutive STAT activation, which is common in epithelial, liver, and breast cancers, fosters the proliferation and survival of malignant cells and tumor-promoting inflammation while mitigating antitumor immunity.82–85 The complex role of STATs makes them a challenging but important subject for research.
In the past few years, researchers have uncovered a noncanonical, epigenetic role for JAK signaling in the nucleus in leukemias and lymphomas.68,86 JAK2 translocates into the nucleus in leukemias with the V617F mutation and in primary mediastinal B-cell lymphoma and Hodgkin’s lymphoma with amplification of the JAK2 locus. There, JAK2 phosphorylates the histone H3 tail on tyrosine 41, counteracting the formation of heterochromatin and promoting gene expression, including expression of the oncogene MYC in both types of lymphoma.68,86 The JAK2 amplicon in these lymphomas also includes JMJD2C, which encodes a chromatin modifier that counteracts the formation of heterochromatin and cooperates with JAK2 in activating genes epigenetically.68
PHARMACOLOGIC INHIBITION OF CYTOKINE SIGNALING WITH JAK INHIBITORS
HEMATOLOGIC DISEASE
The discovery of JAK2 mutations in myeloproliferative neoplasms prompted the evaluation of JAK inhibitors for the treatment of patients with these neoplasms. The JAK1–JAK2 inhibitor ruxolitinib was initially tested in clinical trials involving 153 patients with myelofibrosis.87–89 Evidence that the drug blocked JAK signaling included loss of STAT3 phosphorylation and expression of interleukin-6 and tumor necrosis factor (TNF). These alterations occurred in patients with and those without the V617F mutation, indicating either that wild-type JAK2 plays a pathogenetic role in myelofibrosis or that the blockade of other kinases contributes to the therapeutic effect. Overall, among patients with splenomegaly, spleen size decreased by more than half in 44%, and the decrease in size was accompanied by a marked reduction of associated symptoms.88,89 Ruxolitinib has now been approved by the Food and Drug Administration for the treatment of myeloproliferative neoplasms (the side effects of ruxolitinib and other JAK inhibitors are discussed, below).
The genetic and functional evidence described above with regard to lymphomas provides an impetus for the evaluation of JAK2 inhibitors in primary mediastinal B-cell lymphoma and Hodgkin’s lymphoma and for the evaluation of JAK1 inhibitors in ABC diffuse large-B-cell lymphoma.68 In ABC diffuse large-B-cell lymphoma cells, the MYD88 L265P mutant not only induces autocrine secretion of interleukins 6 and 10, which enhance survival, but also induces autocrine secretion of interferon-β.70,90 Interferon-β can limit tumor growth, but its production is held in check by the transcription factor interferon regulatory factor 4 (IRF4). Lenalidomide, a drug that has shown activity against ABC diffuse large-B-cell lymphoma in early-phase clinical trials,91 interferes with IRF4 expression, thereby unleashing interferon-β secretion and killing the lymphoma cells. IRF4 expression can also be suppressed by the BTK kinase inhibitor ibrutinib, which blocks the chronic active B-cell-receptor signaling that typifies this lymphoma subtype. The synergistic activity of ibrutinib and lenalidomide in killing ABC diffuse large-B-cell lymphoma cells, both in vitro and in xenografts, sets the stage for clinical trials of this drug combination.
AUTOIMMUNITY
Because of the importance of cytokines in autoimmune diseases, the targeting of JAKs appeared to be a logical strategy to pursue in treating these disorders as well. In fact, a number of JAK inhibitors are in various stages of preclinical development or are being tested in clinical trials (Table 1). One JAK inhibitor, tofacitinib, has been approved for the treatment of arthritis in patients for whom methotrexate is not effective.
Table 1.
JAK-Related Disorders and JAK Inhibitors.*.
| JAK | Activating Cytokines | Disorders Caused by Loss-of-Function Mutations | Disorders Caused by Gain-of-Function (Activating) Mutations | Pharmacologic Inhibitors |
|---|---|---|---|---|
| JAK1 | Common γ-chain cytokines; interleukin-6 family; interleukins 10, 13, and 22; granulocyte colony-stimulating factor; interferons α, β, and γ | T-cell acute lymphocytic leukemia, B-cell acute lymphocytic leukemia, acute myeloid leukemia, ABC diffuse large-B-cell lymphoma, acute myeloid leukemia with severe congenital neutropenia | Ruxolitinib, baricitinib, tofacitinib, GLPG0634, ASP015K, AZD1480 | |
| JAK2 | Erythropoietin and other hormonelike cytokines; interleukin-3 family; interleukin-6 family; interleukins 12 and 23; interleukin-13; granulocyte colony-stimulating factor; interferon-γ | Myeloproliferative neoplasm, polycythemia vera, essential thrombocythemia, primary myelofibrosis, Down’s syndrome–associated B-cell acute lymphoblastic leukemia, B-cell acute lymphocytic leukemia, primary mediastinal B-cell lymphoma, Hodgkin’s lymphoma | Ruxolitinib, baricitinib, tofacitinib, pacritinib, lestaurtinib, AZD1480 | |
| JAK3 | Common γ-chain cytokines | SCID | T-cell acute lymphocytic leukemia, adult T-cell leukemia–lymphoma, natural-killer T-cell lymphoma | Tofacitinib, VX-509, ASP015K |
| TYK2 | Interferons α and β; interleukins 6, 10, and 13; granulocyte colony-stimulating factor; interleukins 12 and 23 | Primary immunodeficiency |
The hormonelike cytokines include growth hormone, prolactin, thrombopoietin, and leptin. The interleukin-3 family comprises cytokines that use the common β subunit, including interleukins 3 and 5 and granulocyte colony-stimulating factor. The interleukin-6 family comprises cytokines that use glycoprotein 130, including interleukins 6, 11, and 27; oncostatin M; ciliary neurotrophic factor; cardiotrophin-1; and leptin. Cytokines that use the common γ chain include interleukins 2, 4, 7, 9, 15, and 21. ABC denotes activated B-cell-like, JAK Janus kinase, and SCID severe combined immune deficiency.
Tofacitinib inhibits JAK3 and JAK1 and, to a lesser extent, JAK2 but has little effect on other kinases.92 Functionally, tofacitinib affects both innate and adaptive immune responses and was found to be effective in preclinical models.93 It has also shown efficacy in the treatment of rheumatoid arthritis in phase 3 trials.94,95 In one study, patients with rheumatoid arthritis who had been receiving methotrexate were given tofacitinib, adalimumab (a TNF-α inhibitor), or placebo. The response rates for patients receiving tofacitinib were similar to the rates among patients receiving adalimumab. In a second study, patients with rheumatoid arthritis in whom conventional therapies had not been effective were found to have a response to tofacitinib. The efficacy of tofacitinib in ulcerative colitis and psoriasis has also been reported recently.96,97
Patients receiving tofacitinib had an increased incidence of infection, including tuberculosis and herpes zoster. Anemia and neutropenia were also noted, presumably in relation to JAK2 inhibition and interference with signaling by erythropoietin and other colony-stimulating factors. Increases in lipid levels were also seen, a response that may be related to the blockade of interleukin-6 signaling. JAKs are important for NK-cell–mediated resistance to tumors, but it remains to be determined whether JAK inhibitors augment the risk of cancer in rheumatoid arthritis.98 The side effects of other first-generation JAK inhibitors (ruxolitinib and baricitinib) appear to be similar, and the described side effects are largely related to the mechanism of action — that is, inhibition of cytokine action. As small molecules, these drugs may also have unrelated, off-target effects.
Cytokines are important in a wide range of diseases, from cancer to immune-mediated disease, and the potential usefulness of JAK inhibitors is equally broad. To what extent JAK inhibitors will be used in conjunction with or in place of other immunomodulatory agents remains to be seen. The inhibitors now in clinical use do not selectively inhibit a single JAK; the question of whether more selective, second-generation agents will be as efficacious but with reduced adverse effects is an important area for future investigation. The comparative safety profiles of first- and second-generation JAK inhibitors will become better understood as they are tested in larger studies in various clinical settings. (Table 1 reviews the cytokines and mutations associated with activation of JAKs, as well as the drugs that inhibit activation.)
On the basis of their critical functions, it would seem logical to target STATs in a variety of disorders, from autoimmune diseases and allergies to cancer and atherosclerosis.99,100 Despite considerable effort, clinically useful STAT inhibitors have yet to become a reality. Unlike JAKs, which are enzymes, STATs have been more challenging targets; because of their enormous potential, work in the area of STAT inhibitors continues. (Table 2 reviews the cytokines and the mutations that influence the activation of STATs.)
Table 2.
STAT-Related Disorders*.
| STAT | Activating Cytokines | Disorders Caused by Loss-of-Function Mutations | Disorders Caused by Gain-of-Function (Activating) Mutations |
|---|---|---|---|
| STAT1 | Interferons, other cytokines | Mycobacterial infections, viral infections | Chronic mucocutaneous candidiasis, fungal infections, aneurysms |
| STAT3† | Interleukin-6 and many other interleukins | Hyper-IgE syndrome | Large granular leukemia, ABC diffuse large-B-cell lymphoma, other cancers |
| STAT4‡ | Interleukins 12, 23, α, β | ||
| STAT5A | Prolactin, other hormonelike cytokines, interleukin-2, other cytokines | Multiple cancers | |
| STAT5B | Growth hormone, other hormonelike cytokines, interleukin-2, other cytokines | Immunodeficiency, growth failure, autoimmunity | |
| STAT6§ | Interleukins 4 and 13 |
STAT denotes signal transducer and activator of transcription.
Polymorphisms of STAT3 are associated with ankylosing spondylitis.
Polymorphisms of STAT4 are associated with rheumatoid arthritis, systemic lupus erythematosus, and other autoimmune diseases.
Polymorphisms of STAT6 are associated with allergic disease.
CONCLUSIONS
What began as an effort to understand basic mechanisms in interferon-mediated gene regulation has resulted in a new framework for the study of cell signaling. Moreover, the discovery of this pathway has clarified the pathophysiology of diseases ranging from primary immunodeficiencies to various cancers. The prospect of targeting the JAK–STAT pathway is now a reality, and we are probably just beginning to see the spectrum of diseases for which such targeting might be useful.
Footnotes
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
REFERENCES
- 1.Stark GR, Darnell JE Jr. The JAK-STAT pathway at twenty. Immunity 2012;36: 503–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Leonard WJ, O’Shea JJ. Jaks and STATs: biological implications. Annu Rev Immunol 1998;16:293–322. [DOI] [PubMed] [Google Scholar]
- 3.O’Shea JJ, Sims J, Siegel RM, Farber JM. Cytokines In: Hochberg MC, Silman AJ, Smolen JS, Weinblatt ME, Weisman MH, eds. Rheumatology. Philadelphia: Elsevier, 2011:91–103. [Google Scholar]
- 4.Casanova JL, Holland SM, Notarangelo LD. Inborn errors of human JAKs and STATs. Immunity 2012;36:515–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Minegishi Y, Saito M, Morio T, et al. Human tyrosine kinase 2 deficiency reveals its requisite roles in multiple cytokine signals involved in innate and acquired immunity. Immunity 2006;25: 745–55. [DOI] [PubMed] [Google Scholar]
- 6.Kilic SS, Hacimustafaoglu M, Boisson-Dupuis S, et al. A patient with tyrosine kinase 2 deficiency without hyper-IgE syndrome. J Pediatr 2012;160:1055–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dupuis S, Dargemont C, Fieschi C, et al. Impairment of mycobacterial but not viral immunity by a germline human STAT1 mutation. Science 2001;293:300–3. [DOI] [PubMed] [Google Scholar]
- 8.Dupuis S, Jouanguy E, Al-Hajjar S, et al. Impaired response to interferon-alpha/ beta and lethal viral disease in human STAT1 deficiency. Nat Genet 2003;33: 388–91. [DOI] [PubMed] [Google Scholar]
- 9.Chapgier A, Kong XF, Boisson-Dupuis S, et al. A partial form of recessive STAT1 deficiency in humans. J Clin Invest 2009; 119:1502–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boisson-Dupuis S, Kong XF, Okada S, et al. Inborn errors of human STAT1: allelic heterogeneity governs the diversity of immunological and infectious phenotypes. Curr Opin Immunol 2012;24: 364–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.van de Veerdonk FL, Plantinga TS, Hoischen A, et al. STAT1 mutations in autosomal dominant chronic mucocutaneous candidiasis. N Engl J Med 2011;365: 54–61. [DOI] [PubMed] [Google Scholar]
- 12.Liu L, Okada S, Kong XF, et al. Gain-of-function human STAT1 mutations impair IL-17 immunity and underlie chronic mucocutaneous candidiasis. J Exp Med 2011;208:1635–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sampaio EP, Bax HI, Hsu AP, et al. A novel STAT1 mutation associated with disseminated mycobacterial disease. J Clin Immunol 2012;32:681–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Minegishi Y, Saito M, Tsuchiya S, et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature 2007;448: 1058–62. [DOI] [PubMed] [Google Scholar]
- 15.Holland SM, DeLeo FR, Elloumi HZ, et al. STAT3 mutations in the hyper-IgE syndrome. N Engl J Med 2007;357:1608–19. [DOI] [PubMed] [Google Scholar]
- 16.Grimbacher B, Holland SM, Gallin JI, et al. Hyper-IgE syndrome with recurrent infections — an autosomal dominant multisystem disorder. N Engl J Med 1999; 340:692–702. [DOI] [PubMed] [Google Scholar]
- 17.Chandesris MO, Melki I, Natividad A, et al. Autosomal dominant STAT3 deficiency and hyper-IgE syndrome: molecular, cellular, and clinical features from a French national survey. Medicine (Baltimore) 2012;91(4):e1–e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Milner JD, Brenchley JM, Laurence A, et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008;452:773–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.de Beaucoudrey L, Puel A, Filipe-Santos O, et al. Mutations in STAT3 and IL12RB1 impair the development of human IL-17-producing T cells. J Exp Med 2008;205:1543–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ma CS, Chew GY, Simpson N, et al. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J Exp Med 2008;205:1551–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Siegel AM, Heimall J, Freeman AF, et al. A critical role for STAT3 transcription factor signaling in the development and maintenance of human T cell memory. Immunity 2011;35:806–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kofoed EM, Hwa V, Little B, et al. Growth hormone insensitivity associated with a STAT5fr mutation. N Engl J Med 2003;349:1139–47. [DOI] [PubMed] [Google Scholar]
- 23.Udy GB, Towers RP, Snell RG, et al. Requirement of STAT5b for sexual dimorphism of body growth rates and liver gene expression. Proc Natl Acad Sci U S A 1997; 94:7239–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Imada K, Bloom ET, Nakajima H, et al. Stat5b is essential for natural killer cell-mediated proliferation and cytolytic activity. J Exp Med 1998;188:2067–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yao Z, Kanno Y, Kerenyi M, et al. Non-redundant roles for Stat5a/b in directly regulating Foxp3. Blood 2007;109:4368–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Laurence A, Tato CM, Davidson TS, et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 2007;26:371–81. [DOI] [PubMed] [Google Scholar]
- 27.Cho JH, Gregersen PK. Genomics and the multifactorial nature of human autoimmune disease. N Engl J Med 2011;365: 1612–23. [DOI] [PubMed] [Google Scholar]
- 28.Duerr RH, Taylor KD, Brant SR, et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 2006;314:1461–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Remmers EF, Cosan F, Kirino Y, et al. Genome-wide association study identifies variants in the MHC class I, IL10, and IL23R-IL12RB2 regions associated with Behcet’s disease. Nat Genet 2010;42: 698–702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Remmers EF, Plenge RM, Lee AT, et al. STAT4 and the risk of rheumatoid arthritis and systemic lupus erythematosus. N Engl J Med 2007;357:977–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Granada M, Wilk JB, Tuzova M, et al. A genome-wide association study of plasma total IgE concentrations in the Framingham Heart Study. J Allergy Clin Immunol 2012;129:840–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paternoster L, Standl M, Chen CM, et al. Meta-analysis of genome-wide association studies identifies three new risk loci for atopic dermatitis. Nat Genet 2011;44: 187–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen E, Staudt LM, Green AR. Janus kinase deregulation in leukemia and lymphoma. Immunity 2012;36:529–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Migone TS, Lin JX, Cereseto A, et al. Constitutively activated Jak-STAT pathway in T cells transformed with HTLV-I. Science 1995;269:79–81. [DOI] [PubMed] [Google Scholar]
- 35.Yu CL, Meyer DJ, Campbell GS, et al. Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the Src oncoprotein. Science 1995;269: 81–3. [DOI] [PubMed] [Google Scholar]
- 36.Baxter EJ, Scott LM, Campbell PJ, et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005;365:1054–61. [Erratum, Lancet 2005;366:122.] [DOI] [PubMed] [Google Scholar]
- 37.James C, Ugo V, Le Couedic JP, et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005;434:1144–8. [DOI] [PubMed] [Google Scholar]
- 38.Kralovics R, Passamonti F, Buser AS, et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 2005;352:1779–90. [DOI] [PubMed] [Google Scholar]
- 39.Levine RL, Wadleigh M, Cools J, et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005; 7:387–97. [DOI] [PubMed] [Google Scholar]
- 40.Scott LM, Tong W, Levine RL, et al. JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med 2007;356:459–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Landgren O, Goldin LR, Kristinsson SY, Helgadottir EA, Samuelsson J, Bjorkholm M. Increased risks of polycythemia vera, essential thrombocythemia, and myelofibrosis among 24,577 first-degree relatives of 11,039 patients with myeloproliferative neoplasms in Sweden. Blood 2008;112:2199–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jones AV, Chase A, Silver RT, et al. JAK2 haplotype is a major risk factor for the development of myeloproliferative neoplasms. Nat Genet 2009;41:446–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kilpivaara O, Mukherjee S, Schram AM, et al. A germline JAK2 SNP is associated with predisposition to the development of JAK2(V617F)-positive myeloproliferative neoplasms. Nat Genet 2009; 41:455–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Olcaydu D, Harutyunyan A, Jager R, et al. A common JAK2 haplotype confers susceptibility to myeloproliferative neoplasms. Nat Genet 2009;41:450–4. [DOI] [PubMed] [Google Scholar]
- 45.Giordanetto F, Kroemer RT. Prediction of the structure of human Janus kinase 2 (JAK2) comprising JAK homology domains 1 through 7. Protein Eng 2002; 15:727–37. [DOI] [PubMed] [Google Scholar]
- 46.Ungureanu D, Wu J, Pekkala T, et al. The pseudokinase domain of JAK2 is a dual-specificity protein kinase that negatively regulates cytokine signaling. Nat Struct Mol Biol 2011;18:971–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bandaranayake RM, Ungureanu D, Shan Y, Shaw DE, Silvennoinen O, Hubbard SR. Crystal structures of the JAK2 pseudokinase domain and the pathogenic mutant V617F. Nat Struct Mol Biol 2012; 19:754–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mullally A, Lane SW, Ball B, et al. Physiological Jak2V617F expression causes a lethal myeloproliferative neoplasm with differential effects on hematopoietic stem and progenitor cells. Cancer Cell 2010;17:584–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li J, Spensberger D, Ahn JS, et al. JAK2 V617F impairs hematopoietic stem cell function in a conditional knock-in mouse model of JAK2 V617F-positive essential thrombocythemia. Blood 2010; 116:1528–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bercovich D, Ganmore I, Scott LM, et al. Mutations of JAK2 in acute lymphoblastic leukaemias associated with Down’s syndrome. Lancet 2008;372:1484–92. [DOI] [PubMed] [Google Scholar]
- 51.Kearney L, Gonzalez De Castro D, Yeung J, et al. Specific JAK2 mutation (JAK2R683) and multiple gene deletions in Down syndrome acute lymphoblastic leukemia. Blood 2009;113:646–8. [DOI] [PubMed] [Google Scholar]
- 52.Mullighan CG, Zhang J, Harvey RC, et al. JAK mutations in high-risk childhood acute lymphoblastic leukemia. Proc Natl Acad Sci U S A 2009;106:9414–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Asnafi V, Le Noir S, Lhermitte L, et al. JAK1 mutations are not frequent events in adult T-ALL: a GRAALL study. Br J Haematol 2010;148:178–9. [DOI] [PubMed] [Google Scholar]
- 54.Flex E, Petrangeli V, Stella L, et al. Somatically acquired JAK1 mutations in adult acute lymphoblastic leukemia. J Exp Med 2008;205:751–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Xiang Z, Zhao Y, Mitaksov V, et al. Identification of somatic JAK1 mutations in patients with acute myeloid leukemia. Blood 2008;111:4809–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Elliott NE, Cleveland SM, Grann V, Janik J, Waldmann TA, Dave UP. FERM domain mutations induce gain of function in JAK3 in adult T-cell leukemia/lymphoma. Blood 2011;118:3911–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Koo GC, Tan SY, Tang T, et al. Janus kinase 3-activating mutations identified in natural killer/T-cell lymphoma. Cancer Discov 2012;2:591–7. [DOI] [PubMed] [Google Scholar]
- 58.Zhang J, Ding L, Holmfeldt L, et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature 2012;481:157–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pikman Y, Lee BH, Mercher T, et al. MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med 2006;3(7):e270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Russell LJ, Capasso M, Vater I, et al. Deregulated expression of cytokine receptor gene, CRLF2, is involved in lymphoid transformation in B-cell precursor acute lymphoblastic leukemia. Blood 2009;114: 2688–98. [DOI] [PubMed] [Google Scholar]
- 61.Harvey RC, Mullighan CG, Chen IM, et al. Rearrangement of CRLF2 is associated with mutation of JAK kinases, alteration of IKZF1, Hispanic/Latino ethnicity, and a poor outcome in pediatric B-progenitor acute lymphoblastic leukemia. Blood 2010;115:5312–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yoda A, Yoda Y, Chiaretti S, et al. Functional screening identifies CRLF2 in precursor B-cell acute lymphoblastic leukemia. Proc Natl Acad Sci U S A 2010;107: 252–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shochat C, Tal N, Bandapalli OR, et al. Gain-of-function mutations in interleukin-7 receptor-alpha (IL7R) in childhood acute lymphoblastic leukemias. J Exp Med 2011;208:901–8. [Errata, J Exp Med 2011;208:preceding 901, 1333.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zenatti PP, Ribeiro D, Li W, et al. Oncogenic IL7R gain-of-function mutations in childhood T-cell acute lymphoblastic leukemia. Nat Genet 2011;43:932–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hunter MG, Avalos BR. Granulocyte colony-stimulating factor receptor mutations in severe congenital neutropenia transforming to acute myelogenous leukemia confer resistance to apoptosis and enhance cell survival. Blood 2000;95:2132–7. [PubMed] [Google Scholar]
- 66.Rebouissou S, Amessou M, Couchy G, et al. Frequent in-frame somatic deletions activate gp130 in inflammatory hepatocellular tumours. Nature 2009;457:200–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kapp U, Yeh WC, Patterson B, et al. Interleukin 13 is secreted by and stimulates the growth of Hodgkin and Reed-Sternberg cells. J Exp Med 1999;189: 1939–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rui L, Emre NC, Kruhlak MJ, et al. Cooperative epigenetic modulation by cancer amplicon genes. Cancer Cell 2010; 18:590–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ding BB, Yu JJ, Yu RY, et al. Constitutively activated STAT3 promotes cell proliferation and survival in the activated B-cell subtype of diffuse large B-cell lymphomas. Blood 2008;111:1515–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ngo VN, Young RM, Schmitz R, et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011;470: 115–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lam LT, Wright G, Davis RE, et al. Cooperative signaling through the signal transducer and activator of transcription 3 and nuclear factor-KB pathways in subtypes of diffuse large B-cell lymphoma. Blood 2008;111:3701–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Montesinos-Rongen M, Godlewska E, Brunn A, Wiestler OD, Siebert R, Deckert M. Activating L265P mutations of the MYD88 gene are common in primary central nervous system lymphoma. Acta Neuropathol 2011;122:791–2. [DOI] [PubMed] [Google Scholar]
- 73.Treon SP, Xu L, Yang G, et al. MYD88 L265P somatic mutation in Waldenstrom’s macroglobulinemia. N Engl J Med 2012; 367:826–33. [DOI] [PubMed] [Google Scholar]
- 74.Yan Q, Huang Y, Watkins AJ, et al. BCR and TLR signalling pathways are recurrently targeted by genetic changes in splenic marginal zone lymphomas. Haematologica 2012;97:595–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Puente XS, Pinyol M, Quesada V, et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 2011;475:101–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wang L, Lawrence MS, Wan Y, et al. SF3B1 and other novel cancer genes in chronic lymphocytic leukemia. N Engl J Med 2011;365:2497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Funakoshi-Tago M, Tago K, Abe M, Sonoda Y, Kasahara T. STAT5 activation is critical for the transformation mediated by myeloproliferative disorder-associated JAK2 V617F mutant. J Biol Chem 2010; 285:5296–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Walz C, Ahmed W, Lazarides K, et al. Essential role for Stat5a/b in myeloproliferative neoplasms induced by BCR-ABL1 and JAK2(V617F) in mice. Blood 2012; 119:3550–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yan D, Hutchison RE, Mohi G. Critical requirement for Stat5 in a mouse model of polycythemia vera. Blood 2012; 119:3539–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Scuto A, Krejci P, Popplewell L, et al. The novel JAK inhibitor AZD1480 blocks STAT3 and FGFR3 signaling, resulting in suppression of human myeloma cell growth and survival. Leukemia 2011;25: 538–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Koskela HL, Eldfors S, Ellonen P, et al. Somatic STAT3 mutations in large granular lymphocytic leukemia. N Engl J Med 2012;366:1905–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 2009; 9:798–809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Song JI, Grandis JR. STAT signaling in head and neck cancer. Oncogene 2000; 19:2489–95. [DOI] [PubMed] [Google Scholar]
- 84.He G, Karin M. NF-kB and STAT3 — key players in liver inflammation and cancer. Cell Res 2011;21:159–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bromberg J Stat proteins and oncogenesis. J Clin Invest 2002;109:1139–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Dawson MA, Bannister AJ, Gottgens B, et al. JAK2 phosphorylates histone H3Y41 and excludes HP1alpha from chromatin. Nature 2009;461:819–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Verstovsek S, Kantarjian H, Mesa RA, et al. Safety and efficacy of INCB018424, a JAK1 and JAK2 inhibitor, in myelofibrosis. N Engl J Med 2010;363:1117–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Verstovsek S, Mesa RA, Gotlib J, et al. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. N Engl J Med 2012;366:799–807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Harrison C, Kiladjian JJ, Al-Ali HK, et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med 2012;366:787–98. [DOI] [PubMed] [Google Scholar]
- 90.Yang Y, Shaffer AL III, Emre NC, et al. Exploiting synthetic lethality for the therapy of ABC diffuse large B cell lymphoma. Cancer Cell 2012;21:723–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hernandez-Ilizaliturri FJ, Deeb G, Zinzani PL, et al. Higher response to lenalidomide in relapsed/refractory diffuse large B-cell lymphoma in nongerminal center B-cell-like than in germinal center B-cell-like phenotype. Cancer 2011;117: 5058–66. [DOI] [PubMed] [Google Scholar]
- 92.Ghoreschi K, Jesson MI, Li X, et al. Modulation of innate and adaptive immune responses by tofacitinib (CP-690,550). J Immunol 2011;186:4234–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Changelian PS, Flanagan ME, Ball DJ, et al. Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 2003;302:875–8. [DOI] [PubMed] [Google Scholar]
- 94.Fleischmann R, Kremer J, Cush J, et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med 2012;367:495–507. [DOI] [PubMed] [Google Scholar]
- 95.van Vollenhoven RF, Fleischmann R, Cohen S, et al. Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N Engl J Med 2012;367:508–19. [DOI] [PubMed] [Google Scholar]
- 96.Sandborn WJ, Ghosh S, Panes J, et al. Tofacitinib, an oral Janus kinase inhibitor, in active ulcerative colitis. N Engl J Med 2012;367:616–24. [DOI] [PubMed] [Google Scholar]
- 97.Papp KA, Menter A, Strober B, et al. Efficacy and safety of tofacitinib, an oral Janus kinase inhibitor, in the treatment of psoriasis: a Phase 2b randomized placebo-controlled dose-ranging study. Br J Dermatol 2012;167:668–77. [DOI] [PubMed] [Google Scholar]
- 98.Bellucci R, Nguyen HN, Martin A, et al. Tyrosine kinase pathways modulate tumor susceptibility to natural killer cells. J Clin Invest 2012;122:2369–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Turkson J, Jove R. STAT proteins: novel molecular targets for cancer drug discovery. Oncogene 2000;19:6613–26. [DOI] [PubMed] [Google Scholar]
- 100.Nelson EA, Sharma SV, Settleman J, Frank DA. A chemical biology approach to developing STAT inhibitors: molecular strategies for accelerating clinical translation. Oncotarget 2011;2:518–24. [DOI] [PMC free article] [PubMed] [Google Scholar]


