Abstract
Background
Outcome of infants with tracheostomy have not been well described in the literature. Our objective was to describe the respiratory, growth, and survival outcomes of infants with tracheostomy.
Methods
A retrospective study was conducted on 204 infants born between 2005 and 2015 with tracheostomy at <1 year of age and follow-up in the Infant Tracheostomy and Home Ventilator Clinic up to 4 years of age.
Results
The mean age at tracheostomy was 4.5 months with median age of 3 months. Median age of decannulation was 32 months. The time from tracheostomy placement to complete discontinuation of mechanical ventilation was 15.4 months and from tracheostomy to decannulation was 33.8 months. Mortality rate was 21% and median age of death was 18 months. Preterm infants with acquired airway and lung disease (BPD) and born at <28 weeks’ gestation had a significantly higher survival rate compared to term infants. The z-scores for weight and weight for length improved from the time of discharge (mean chronological age 6.5 months) to first year and remained consistent through 3 years.
Conclusions
Premature infants had a higher rate of discontinuation of mechanical ventilation and decannulation compared to term infants. These infants showed consistent growth and comparable survival rate.
Impact
Infants with tracheostomy and ventilator dependence followed in a multidisciplinary clinic model may have improved survival, growth, and earlier time to decannulation.
Preterm infants with acquired airway and lung disease (BPD) with tracheostomy had a higher survival rate compared to term infants with various tracheostomy indications.
The age at tracheostomy in infants was 4.5 months and of decannulation was 37 months. Time from tracheostomy to complete discontinuation of mechanical ventilation was 15.4 months.
Addition of this data to the sparse literature will be crucial in counseling the families and education of medical staff.
Background
Pediatric tracheostomy is on the rise, with 40% of tracheostomies performed in children <1 year of age.1 Indications for tracheostomy in infants include congenital airway anomalies, acquired airway and lung abnormalities (tracheobronchomalacia (TBM), subglottic stenosis, vocal cord paralysis), bronchopulmonary dysplasia [BPD], neurologic and cardiac anomalies, and underlying risk factors (genetic anomalies).1,2 The significant increase in the rate of tracheostomy in infants is partly attributed to the increased survival of extremely preterm infants with severe BPD who require long-term ventilation for several years.3–5 Prior studies indicate that rehospitalization, equipment malfunction, and tracheostomy at <1 year of age are significant risk factors for mortality.1,6
These findings have prompted children’s hospitals to develop dedicated teams to care for these infants, not only during their initial inpatient stay but also on an outpatient basis in collaborative multidisciplinary teams involving several pediatric specialties.7 The varying outcomes reported have led to confusion about the optimal management of these infants who require long-term ventilation, with some neonatal intensive care units (NICUs) preferring long-term intubation over tracheostomy and home discharge.1,8 Significant variation in practice has led to conflicting opinions on indications, feasibility, and long-term prognosis of infants <1 year old undergoing tracheostomy.1,8 Decannulation time, mortality, and neurodevelopmental outcomes vary significantly based on the indication for tracheostomy and underlying diagnosis.8
Although the decannulation time, mortality, and comorbidities have been described in pediatric tracheostomy, these data have not been described in infants receiving a tracheostomy at <1 year of age.8 Further, it has not been clearly established whether outcomes are improved with a multidisciplinary approach to the care of these medically complex infants. Thus the goal of this study was to address the knowledge gap by assessing the respiratory outcomes (complete discontinuation of mechanical ventilation, decannulation from tracheostomy) and survival until 4 years of age. Our second goal was to study common comorbid conditions and growth outcomes until 3 years of age in our established infant home ventilator program. Patients frequently graduate from our clinic between 3 and 4 years of age. Therefore, we chose to study our second goal until 3 years of age. These data will show that a multidisciplinary, standardized approach will result in improved survival and earlier and timely decannulation of infants. These data will also enhance the sparse literature in this area and clarify outcomes for these infants, ultimately allowing medical providers to better counsel families.
Methods
Composition and operations of the multidisciplinary infant tracheostomy and home ventilator team
The infant home ventilator team is composed of four neonatologists, two advanced practice nurse practitioners, a nurse coordinator, a social worker, and a nutritionist. Differing from other centers, neonatologists primarily run the infant home ventilator team and pulmonologists are involved in collaboration with the inpatient team and then in the multidisciplinary clinic after NICU discharge. The home ventilator team rounds weekly on all infants with tracheostomy in the hospital and offers recommendations to the primary inpatient team. The home ventilator team participates in the management of these medically complex infants during their inpatient stay and attends care conferences, providing tracheostomy education and counseling on the expectations after discharge from the hospital. As part of discharge planning, the clinic team meets the patient and family, beginning several weeks before their discharge, to become familiar with their medical and psychosocial needs. The infant tracheostomy and home ventilator clinic serve as a comprehensive, multidisciplinary, family-centered medical home for medically complex infants who require ventilator support in the home. In addition, primary care services, including same-day sick visits, regular growth and neurodevelopmental surveillance, anticipatory guidance, and routine and seasonal immunizations, are also provided to most patients. A 24/7 on-call physician is available to parents, caregivers, and private duty nurses for live telephone consultations. A dedicated psychologist provides counseling and psychosocial support to families and caregivers. Dedicated otolaryngologists, pulmonologists, cardiologists, and a gastroenterologist provide on-site interdisciplinary care and support. Otolaryngologists are present in the clinic twice each month, whereas pulmonologists and gastroenterologists are present in the clinic once per month. Cardiology clinic visits are coordinated to be on the same day as the home ventilator clinic visit. There is a clinic coordinator nurse who helps coordinate the services for each patient and is the single point of contact for the family. Additional subspecialty care with other subspecialists is scheduled as needed. Early intervention occupational and physical rehabilitation services are provided at home through state-sponsored programs, with close follow-up evaluation as needed in the rehabilitation clinic. During rehospitalization, care is provided in the pediatric intensive care unit (PICU), with care oversight provided by the home ventilator team. Most patients are transitioned to the care of local pediatricians/primary care providers by 3–4 years of age with continued pulmonology and other subspecialty support, at which time the majority of these patients have already been liberated from the ventilator and have been decannulated.
Study population
The subjects in this study were recruited from the Children’s Mercy Infant Tracheostomy and Home Ventilator Clinic from 2005 to 2015. This clinic was established in 2005 to provide a smooth transition for ventilator-dependent infants discharged from the intensive care nursery and the PICU to the home setting. Prior to hospital discharge, parents and home caregivers are trained on all aspects of the care of infants with chronic home ventilation based on the clinical practice guidelines set forth by the American Thoracic Society.9 Local home nursing agencies provide private duty nursing support at home, and one of the durable medical equipment company supply all the equipment needed at home.
Study design
Data were abstracted from electronic medical records and entered in the database of the Center for Infant and Pulmonary Disorders (CIPD), located at Children’s Mercy Kansas City in Kansas City, MO. The CIPD houses the Infant Pulmonary Disorders Data Repository, which provided de-identified data for this study with Children’s Mercy institutional review board approval (IRB). Requirement of obtaining informed consent was waived by IRB. Inclusion criteria for this study included infants who received tracheostomy with or without ventilator support at the time of the first hospital discharge, regardless of underlying etiology, and who were followed in the Infant Tracheostomy and Home Ventilator Clinic at Children’s Mercy Kansas City. Two hundred and four (n = 204) infants met the inclusion criteria. Five patients were excluded from the study as these patients had upper airway disorders due to underlying genetic condition. Diagnoses were obtained by the ICD-9 and ICD-10 codes (The International Classification of Diseases, Ninth and Tenth Revisions) in the electronic medical record.
Demographic data
Demographic data included sex, race, gestational age (GA), weight, length, and head circumference at birth. Subjects were divided into six main groups based on the primary indication for tracheostomy, namely: (a) upper airway diseases (laryngo-tracheomalacia, subglottic stenosis, upper airway obstruction, or a combination of these diagnoses); (b) lower airway diseases (TBM, bronchomalacia); (c) combination of upper or lower airway disease and parenchymal lung disease as with BPD; (d) congenital heart disease with associated chronic lung disease (atrioventricular canal, tetralogy of Fallot, hypoplastic left heart syndrome; (e) neurologic disease (epilepsy, Arnold Chiari malformation, hydrocephalus, hypoxic ischemic encephalopathy, bilateral vocal cord paralysis, grade 4 intraventricular hemorrhage cerebral dysgenesis, or a combination of these diagnoses); and (f) others (chromosomal abnormalities, Pierre Robin sequence, severe micrognathia, giant omphalocele, diaphragmatic paralysis, spinal muscular dystrophy, choanal atresia, tracheoesophageal fistula, congenital hypoventilation syndrome, obstructive or central sleep apnea syndrome). If the upper airway anomaly was not related to any genetic abnormality, it was categorized into upper airway disease. Patients with upper airway disease associated with genetic abnormality were excluded. Discharge destination, child protective services involvement, as well as health insurance coverage as surrogates for socioeconomic status were collected.
Outcome variables at the time of discharge and post discharge
Outcome variables at discharge (age at tracheostomy, need for mechanical ventilation, and presence of gastrostomy tube) and outpatient variables (complete discontinuation of mechanical ventilation, need for polysomnogram, age at decannulation) were collected at the time of discharge and during each home vent clinic visit, respectively, until 4 years of age. The presence of comorbidities (pulmonary hypertension, dysphagia, asthma, seizures, obstructive sleep apnea, and adrenal insufficiency) was reported as well. The use of medications (inhaled and enteral) as continuous variables was obtained at 6 months, 1, 2, and 3 year’s chronologic age. Inhaled medications were classified into inhaled steroids and bronchodilators, while enteral medications were further classified based on organ systems, such as gastrointestinal, cardiac, neurologic, endocrine, and renal. Growth outcomes (weight and weight for length) reported as z-scores were abstracted at discharge and at 6, 12, 24, and 36 months chronologic age. All deaths that occurred after hospital discharge were reviewed and reported.
Statistical analysis
Statistical analysis included general descriptive statistics of patients and comparisons between the six defined groups. Continuous variables were assessed using one-way analysis of variance and comparison of categorical variables using chi-square or Fisher’s exact tests. Logistic regression models to predict death were also computed and included variables with cardiac diagnoses, number of oral medications or inhaled medications at discharge, neurologic diagnoses, GA, birth weight, and birth weight to length. For logistic regression, we divided the entire cohort into four groups based on GA. Group 1 consisted of infants born at <28 weeks’ GA, group 2 consisted of infants born between 29 and 32 weeks’ GA, group 3 consisted of infants born between 33 and 36 weeks’ GA, and group 4 consisted of infants born at >37 weeks’ gestation. Furthermore, changes in weight z-scores and weight-to-length ratios across time points (6, 12, 24, and 36 months) were compared through Wilcoxon signed-rank tests. Statistical analysis was done using the SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Description of the cohort
A total of 204 infants met the inclusion criteria from 2005 through 2015, and no patients were excluded from the study. Fifty-three percent were males and 47% were females. The majority were Caucasians (61%), followed by African Americans (18%), Hispanics (6%), Asians (1%), and others (Native Americans, Hawaiians, and multiracial—14%). The primary indications for tracheostomy were abnormalities related to (a) upper airway (13.7%), (b) lower airway (16.7%), (c) combined airway and lungs (BPD) (40.2%), (d) cardiac (7.3%), (e) neurologic (6.9%), and (f) others (15.2%) (Table 1). For all patients, the mean GA at birth was 31.8 weeks, mean birth weight 1733 g, mean birth length 40.1 cm, and mean head circumference at birth was 28.2 cm. However, infants with airway and respiratory conditions (groups a–c) were significantly more immature and smaller at birth compared to those with cardiac, neurologic, and other conditions (30 weeks’ gestation vs 34–36 weeks and 1.3–1.6 kg vs 2.1–2.5 kg) (Table 1). The mean age at tracheostomy was 4.5 (±4.2) months, with median age of 3 months (Q1—2, Q3—5), and was similar for all primary indications for tracheostomy. All infants were ventilator dependent at the time of tracheostomy. At the time of discharge, 173/204 (85.2%) patients continued to require assisted mechanical ventilation, while 39/204(19.1%) were weaned off assisted ventilation.
Table 1.
Demographic data for the entire cohort with comparisons between groups based on reasons for tracheostomy.
| Total cohort | Upper airway | Lower airway | Combined airway and lung with BPD | Cardiac | Neurologic | Other | P value | |
|---|---|---|---|---|---|---|---|---|
| N = 204 (100%) | N = 28 (13.73%) | N = 34 (16.67%) | N = 82 (40.2%) | N = 15 (7.35%) | N = 14 (6.86%) | N = 31 (15.2%) | ||
| Sex | 0.112 | |||||||
| Male (N (%)) | 107 (52.5) | 12 (42.9) | 22 (64.7) | 44 (53.7) | 8 (53.3) | 3 (21.4) | 18 (58.1) | |
| Female (N (%)) | 97 (47.5) | 16 (57.1) | 12 (35.3) | 38 (46.3) | 7 (46.7) | 11 (78.6) | 13 (41.9) | |
| Gestational age (weeks) (mean (SD)) | 31.8 (6.0) | 30.0 (6.0) | 30.8 (6.1) | 30.8 (5.6) | 33.8 (6.2) | 36.8 (3.1) | 34.2 (5.7) | <0.001 |
| Birth weight (grams) (mean (SD)) | 1733 (1205) | 1379 (1031) | 1491 (1203) | 1604 (1230) | 2234 (1306) | 2572 (812) | 2131 (1113) | 0.011 |
| Birth length (cm) (mean (SD)) | 40.1 (8.6) | 38.8 (8.3) | 38.3 (8.6) | 38.2 (8.1) | 42.2 (9.5) | 47.9 (5.9) | 44.5 (7.9) | 0.001 |
| Head circumference (cm) (mean (SD)) | 28.2 (5.4) | 27.0 (5.0) | 27.1 (6) | 27.2 (5.2) | 30.9 (5.4) | 32.7 (1.8) | 30.2 (4.6) | 0.004 |
| Age at tracheostomy (months) (mean (SD)) | 4.5 (4.2) | 4.7 (4.7) | 3.9 (2.6) | 4.9 (4.6) | 3.5 (2.2) | 5.7 (4.9) | 3.6 (4.8) | 0.385 |
| G-tube at discharge | 0.454 | |||||||
| Yes (N (%)) | 189 (93.0) | 24 (85.7) | 33 (97.0) | 75 (92.0) | 15 (100) | 14 (100) | 28 (90.3) | |
| No (N (%)) | 15 (7.0) | 4 (14.3) | 1 (3.0) | 7 (8.0) | 0 (0) | 0 (0) | 3 (9.7) | |
| Sleep study before discharge | 0.307 | |||||||
| Yes (N (%)) | 61 (30.2) | 10 (37.0) | 9 (27.0) | 20 (25.0) | 8 (53.3) | 5 (35.7) | 9 (29.0) | |
| No (N (%)) | 141 (69.5) | 17 (63.0) | 25 (73.0) | 61 (75.0) | 7 (46.7) | 9 (64.3) | 22 (71.0) | |
| No data (N) | 2 | 1 | 0 | 1 | ||||
| Discharge destination | 0.950 | |||||||
| Home (N (%)) | 178 (89.9) | 27 (96.4) | 30 (90.9) | 67 (85.9) | 14 (93.3) | 13 (92.9) | 27 (90.0) | |
| Foster (N (%)) | 19 (9.6) | 1 (3.6) | 3 (9.1) | 10 (12.8) | 1 (6.7) | 1 (7.1) | 3 (10.0) | |
| Chronic care facility (N (%)) | 1 (0.5) | 0 (0) | 0 (0) | 1 (1.3) | 0 (0) | 0 (0) | 0 (0) | |
| No data (N) | 6 | 0 | 1 | 4 | 0 | 0 | 1 | |
| Child protective services involvement | 0.662 | |||||||
| Yes (N (%)) | 18 (9.1) | 2 (7.1) | (9.1) | 10 (12.8) | 0 (0) | 0 (0) | 3 (10.0) | |
| No (N (%)) | 180 (90.9) | 26 (92.9) | 30 (90.9) | 68 (87.2) | 15 (100) | 14 (100) | 27 (90.0) | |
| No data (N) | 6 | 0 | 1 | 4 | ||||
| Health insurance at 12 months | 0.328 | |||||||
| Medicaid (N (%)) | 141 (71.2) | 22 (78.6) | 19 (57.6) | 57 (72.2) | 10 (71.4) | 12 (85.7) | 21 (70) | |
| Private (N (%)) | 20 (10.1) | 2 (7.1) | 6 (18.2) | 7 (8.9) | 3 (21.4) | 0 (0) | 2 (6.7) | |
| Combination (N (%)) | 34 (17.2) | 4 (14.3) | 8 (24.2) | 13 (16.5) | 0 (0) | 2 (14.3) | 7 (23.3) | |
| Self-pay (N (%)) | 3 (1.5) | 0 (0) | 0 (0) | 2 (2.2) | 1(7.1) | 0 | 0 | |
| No data (N) | 6 | 0 | 1 | 3 | 1 | 0 | 1 | |
| Time from tracheostomy to weaning off from home ventilator (months) (mean (SD)) | 15.4 (14.9) | 26.0 (23.0) | 10.1 (7.0) | 16.2 (16.5) | 13.0 (5.1) | a | 14.3 (10.3) | 0.287 |
| Age at weaning off from home ventilator (months) (mean (SD)) | 23.1 (18.1) | 17.08 (8.9) | 24.5 (8.1) | 27.2 (23.0) | 25.7 (15.7) | 11.3 (13.4) | 12.2 (8.5) | 0.04 0 |
| Time from tracheostomy to decannulation (months) (mean (SD)) | 33.8 (19.3) | 36.8 (20.5) | 28 (12.6) | 37.1 (22.6) | 26.8 (4.6) | a | 28.2 (9.3) | 0.413 |
| Age at decannulation (months) (mean (SD)) | 38.1 (20.7) | 39.8 (19.9) | 31.5 (12.6) | 41.9 (23.3) | 30.8 (3.4) | a | 28.2 (12.6) | 0.036 |
| Decannulation | ||||||||
| Yes (N (%)) | 80 (39.2) | 10 (35.7) | 14 (41.2) | 41 (50.0) | 4 (26.7) | 1 (7.1) | 10 (32.3) | 0.031 |
| No (N (%)) | 124 (60.8) | 18 (64.3) | 20 (58.8) | 41 (50.0) | 11 (73.3) | 13 (92.9) | 21 (67.7) | |
| Death (N (%)) | 43 (21.1) | 5 (18.0) | 7 (20.6) | 16 (19.5) | 3 (20.0) | 4 (28.6) | 8 (25.8) | 0.929 |
| Comorbidities | ||||||||
| AI (N (%)) | 31 (15.2) | 7 (25.0) | 4 (11.8) | 16 (19.5) | 3 (20.0) | 0 (0) | 1 (3.2) | 0.060 |
| PHTN (N (%)) | 61 (29.9) | 4 (14.3) | 16 (47.1) | 30 (36.6) | 8 (53.3) | 0 (0) | 3 (9.7) | <0.001 |
| Dysphagia (N (%)) | 113 (55.4) | 19 (67.9) | 15 (44.1) | 43 (52.4) | 8 (53.3) | 10 (71.4) | 18 (58.1) | 0.372 |
| Asthma (N (%)) | 45 (22.1) | 4 (14.3) | 10 (29.4) | 26 (31.7) | 1 (6.7) | 1 (7.1) | 3 (9.7) | 0.025 |
| Seizures (N (%)) | 67 (32.8) | 8 (28.6) | 14 (41.2) | 24 (29.3) | 2 (13.3) | 8 (57.1) | 11 (35.5) | 0.154 |
| OSA (N (%)) | 72 (35.3) | 12 (42.9) | 14 (41.2) | 35 (42.7) | 5 (33.3) | 3 (21.4) | 3 (9.7) | 0.012 |
AI adrenal insufficiency, PHTN pulmonary hypertension, OSA obstructive sleep apnea.
aUnable to analyze due to low numbers.
The majority of infants (93%) were G-tube dependent for their nutritional support at discharge; the proportion of infants who were able to feed orally (7%) had tracheostomy only and did not need assisted ventilation at discharge. No patients were discharged with nasogastric tube (NG). Discharging these patients’ home with a NG tube was felt at higher risk for dislodgement and complications. The mean age at hospital discharge for all infants was 6.5 months and median age was 7 months (Q1—4, Q3—9). One hundred and seventy-eight infants out of the total cohort (89.9%) were discharged to the parents, while 18/204 (9.1%) had child protective services involvement and were placed in foster care. One infant was transferred to an out-of-state chronic care facility. In the Kansas City area, long-term care facilities and day programs are not available, and infants with chronic conditions stay in the NICU until the time of discharge. This could be because of less densely populated areas in Kansas making it financially difficult to pull in enough patients. Seventy-one percent of families had Medicaid coverage, 10.1% had private health insurance, while 17.2% had a combination of both. The population geographic distribution based on zip code of each study subject’s discharge residence is shown in Supplementary Fig. S1.
Outcomes post-hospital discharge
In our study, the mean age of complete discontinuation of mechanical ventilation was 23.1 (SD 18.2) months of age (median of 21 months [Q1—15, Q3—27]) and decannulation at mean age of ~38.1 (SD 20.7) months (median of 32 months (Q1—26.5, Q3—47); however, those infants with upper airway diagnoses (group a) had a shorter duration of ventilator dependence of 17.08 (±8.9) months; P = 0.040 (Table 1).
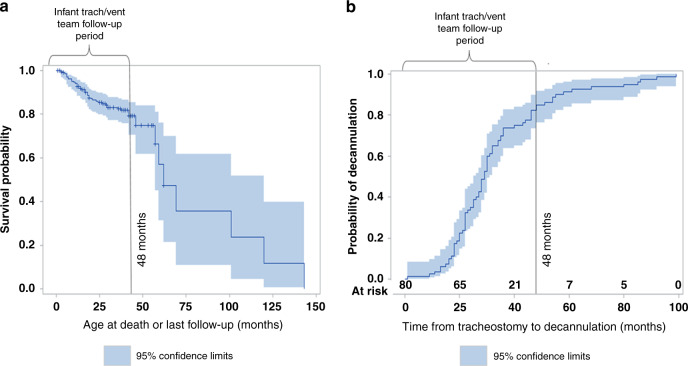
Eighty of the entire cohort of 204 infants were decannulated, with 61 (30%) decannulated by 48 months of age; the highest rate of decannulation was among the infants with BPD (50%) (Table 1). The average time from tracheostomy to decannulation was 33.8 months (19.3) (Fig. 1), and the average age at decannulation was 38.1 (±20.7) months (median of 32 months (Q1—26.2, Q3—46.2) and was similar across all groups. There were 61/204 (30%) of infants who had a sleep study prior to decannulation. Laryngotracheoplasty (airway reconstruction surgery) was done in 41/204 (20.1%) of infants and we believe that it led to earlier decannulation in most cases. These data represent the infants followed up in the home ventilator clinic until 4 years of age. If the infants were not decannulated by 4 years of age, they were transitioned to the pulmonology service for ongoing care, and outcomes have not been established for that population at this time but will be addressed in forthcoming publications.
Fig. 1. Survival and decannulation age trends for the entire cohort.
a Survival trends of the entire cohort on survival probability curve showing 21% mortality. b Time from tracheostomy to decannulation showing average decannulation age of 37 months.
Growth outcomes
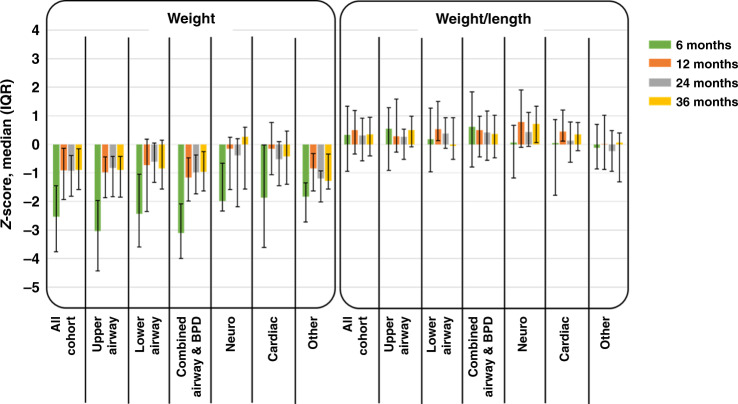
In terms of growth, the z-scores for weight (W) and weight for length (W/L) improved significantly from the time of hospital discharge to the first year of age and stayed consistent through 3 years of age. All weight z-scores were less than average weights (Table 2 and Fig. 2). Essentially, z-score weight comparisons from 6–12, 6–24, and 6–36 months were all significantly different overall and for each of the groups. Comparisons for 12–24, 12–36, and 24–36 months were mostly not statistically different. In weight to length, z-scores skewed more toward the average throughout the time period (Fig. 2).
Table 2.
Results from the Wilcoxon signed-rank test with various comparisons and P values.
| 6–12 months | 6–24 months | 6–36 months | 12–24 months | 12–36 months | 24–36 months | |
|---|---|---|---|---|---|---|
| Weight | ||||||
| All | <0.0001 | <0.0001 | <0.0001 | 0.7675 | 0.1175 | 0.0041 |
| Upper airway | <0.0001 | 0.0002 | 0.0008 | 0.439 | 0.1327 | 0.0663 |
| Lower airway | <0.0001 | 0.0035 | 0.0390 | 0.5787 | 0.6584 | 0.9333 |
| Combined airway | <0.0001 | <0.0001 | <0.0001 | 0.1776 | 0.0783 | 0.1781 |
| Cardiac | 0.0015 | 0.0034 | 0.0063 | 0.0022 | 0.4854 | 0.4143 |
| Neuro | 0.0425 | 0.1934 | 0.1641 | 0.75 | 0.9219 | 0.3203 |
| Other | 0.0003 | 0.0948 | 0.0082 | 0.616 | 0.3788 | 0.0198 |
| Weight/length | ||||||
| All | 0.2365 | 0.6345 | 0.8042 | 0.3026 | 0.4844 | 0.4119 |
| Upper airway | 0.3097 | 0.6022 | 0.5958 | 0.4684 | 0.9636 | 0.3683 |
| Lower airway | 0.2248 | 0.9854 | 0.5678 | 0.0233 | 0.0188 | 0.2971 |
| Combined airway | 0.5055 | 0.7057 | 0.7193 | 0.2727 | 0.2948 | 0.8454 |
| Cardiac | 0.1675 | 0.4238 | 0.3013 | 0.1677 | 0.7869 | 0.5879 |
| Neuro | 0.2661 | 0.8457 | 0.1719 | 0.7646 | 0.8457 | 0.2402 |
| Other | 0.6017 | 0.8831 | 0.5678 | 0.6265 | 0.3682 | 0.953 |
Bold and italic represents statistically significant P values.
z-Score weight comparisons from 6–12, 6–24, and 6–36 months were all significantly different overall and for each of the groups. Comparisons for 12–24, 12–36, and 24–36 months were mostly not statistically different. In weight to length, z-scores were mostly not statistically different.
Fig. 2. Growth trends for the entire cohort until 3 years of age.
Median z-scores with IQR error bars over 3-year period for the entire cohort and by reasons for tracheostomy showing the z-scores for weight (W) and weight for length (W/L) improved significantly from the time of hospital discharge to the first year of age and stayed consistent through 3 years of age.
Comorbidities and medication use
Dysphagia was the most common long-term comorbidity present, in 55% of all infants, followed by obstructive sleep apnea (35.3%), seizures (32.8%), pulmonary hypertension (29.9%), and asthma (22.1%). Most infants received multiple medications depending on the underling primary and comorbid diagnoses. All the chronic medications used until the age of 3 years were divided into indication by organ system, namely: (a) gastrointestinal (ranitidine, omeprazole, lansoprazole, erythromycin, probiotics); (b) neurologic or antiseizure medications (levetiracetam, phenobarbital, clonidine, lorazepam); (c) cardiac (enalapril, propranolol, sotalol, flecainide); (d) renal, mostly diuretics (furosemide, chlorothiazide, spironolactone); (e) endocrine (hydrocortisone, prednisolone, levothyroxine); and (f) respiratory, in the form of inhaled steroids (fluticasone, beclomethasone, budesonide). The use of medications belonging to all the organ categories for the entire cohort was increased at 1 year of age and then gradually decreased over the next 2 years (Fig. 3). In the entire cohort, 7.3% infants (15/204, sildenafil—13, bosentan—2) were on pulmonary hypertension medications at discharge and 7.6% (16/204, sildenafil—14, bosentan—2) infants at 1 year of age. Endocrine medications at discharge were levothyroxine (22/204), hydrocortisone (8/204), prednisone (5/204), growth hormone (1/204): at 1 year of age, levothyroxine (18/204), hydrocortisone (6/204), prednisone (5/204), growth hormone (1/204): at 2 years of age, levothyroxine (19/204), hydrocortisone (1/204), prednisone (1/204), growth hormone (1/204); and at 3 years of age levothyroxine (17/204), hydrocortisone (1/204), prednisone (0/204), growth hormone (1/204).
Fig. 3. Oral and inhaled medications use of the entire cohort over 3-year period by organ system.
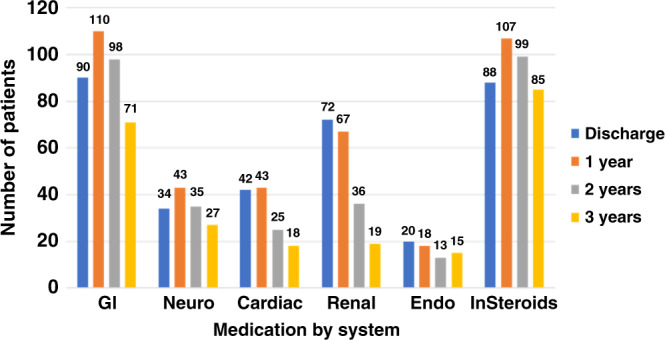
GI gastrointestinal, Neuro neurologic system, Cardiac cardiovascular system, Endo endocrine, InSteroids inhaled steroids.
In the entire cohort, 88/204 (43.1%) infants were on inhaled steroids, 78/204 (38.2%) on inhaled bronchodilators, and 60/204 (29.4%) on both at the time of discharge. At 1 year, 107/204 (52.4%) infants were on inhaled steroids, 66/204 (32.3%) on inhaled bronchodilators, and 54/204 (26.4%) on both. At 2 years, 99/204 (48.5%) infants were on inhaled steroids, 95/204 (46.5%) on inhaled bronchodilators, and 78/204 (38.2%) on both. At 3 years, 85/204 (41.6%) infants were on inhaled steroids, 85/204 (41.6%) on inhaled bronchodilators, and 71/204 (34.8%) on both.
Risk factors associated with death
The overall mortality rate was 43/204 (21.1%), with corresponding mean age at death equal to 28.8 (±30.5) months and median age of 18 months (Q1—10, Q3—31) (Fig. 1). Of those who died, 36/44 (81.8%) died at <48 months of age. Seventeen (17/44) patients died due to presumed cardiac arrest at home, and out of those, 8 patients died due to tracheostomy-related reasons (accidental decannulation, plugged tracheostomy tube) and 9 patients had no clear documentation.
Preterm infants with acquired airway and lung disease (BPD) who were born at <28 weeks’ gestation and received tracheostomy had a significantly higher survival rate as compared to term infants with various tracheostomy indications when solely looked at the GA in an unadjusted model (P value 0.0002, odds ratio 0.119; Table 3). When adjusted to control for birth weight, length, and oral and inhaled medications, this was no longer the case. Logistic regression analysis showed that higher number of inhaled medications at discharge decreased the risk of death by 40%; however, the higher number of oral medications increased risk of death by 24% in an unadjusted model. This finding was still significant when adjusted for GA, birth weight, and length (Table 3).
Table 3.
Backward logistic regression model looking at the outcome variable of death in a cohort of infants with tracheostomy and ventilator dependence and cluster analysis within GA subgroups.
| Effect | B-estimate | P value unadjusted | Odds ratio (95% CI) unadjusted | P value adjusted | Odds ratio (95% CI) adjusted with ROC AUC = 0.8193 |
|---|---|---|---|---|---|
| GA Group 1 (vs 4) | −2.1296 | 0.0002 | 0.119 (0.039–0.365) | 0.2907 | 0.218 (0.013–3.672) |
| GA Group 2 (vs 4) | 0.1476 | 0.7502 | 1.159 (0.467–2.876) | 0.9647 | 1.045 (0.146–7.476) |
| GA Group 3 (vs 4) | 0.3018 | 0.5760 | 1.352 (0.470–3.894) | 0.1014 | 3.354 (0.778–14.269) |
| Birth weight | 0.0947 | 0.1142 | 1.099 (0.977–1.236) | ||
| Birth weight length | 0.0877 | 0.0179 | 1.092 (1.015–1.174) | 0.9640 | 0.996 (0.839–1.182) |
| Cardiac diagnosis | −0.0712 | 0.9153 | 0.931 (0.251–3.460) | ||
| Neuro diagnosis | 0.4379 | 0.4788 | 1.549 (0.461–5.205) | ||
| # oral medications at death | 0.2169 | 0.0013 | 1.242 (1.088–1.418) | 0.0245 | 1.330 (1.037–1.705) |
| # inhaled medications at death | −0.5298 | 0.0205 | 0.589 (0.376–0.922) | 0.0312 | 0.420 (0.191–0.925) |
Group 1: <28 weeks’ gestation; group 2: 29–32 weeks; group 3: 33–36 weeks; group 4: >37 weeks.
Discussion
Published studies have reported tracheostomy outcomes for pediatric patients ranging in age from 1 to 18 years. However, no study has specifically examined tracheostomy outcomes for patients <1 year of age, who make up the majority of the pediatric tracheostomy population.1,8,10 To our knowledge, this is the first and largest tracheostomy and home ventilator follow-up study to examine the respiratory, growth, and survival outcomes of infants who received a tracheostomy at <1 year of age and were followed up after hospital discharge by a dedicated multidisciplinary home ventilator team until 3–4 years of age. This study examined the outcomes of these infants during their initial hospitalization in either the NICU or PICU. Most of these infants were born outside of our regional hospital and were transferred in for ongoing tertiary care. The rate of tracheostomy in infants <1 year of age has been on the rise, primarily due to improved survival of extremely premature infants with better resuscitation strategies and technology in NICUs.11 Most children’s hospitals have moved toward a dedicated multidisciplinary care model with a ventilator care program for this vulnerable population; however, outcome data for these infants are scarce in the literature.7,11
The overall mortality rate in this cohort of infants with tracheostomy for various indications was 21.1% by 4 years of age, with median age of death at 18 months. Our experience is very similar to that of Cristea et al., who reported a mortality rate of 18.6% in their cohort, and a median age of death at 27 months.12 Acute cardiorespiratory arrest related to accidental decannulation and plugged tracheostomy tube were the most common tracheostomy-related causes of death that is consistent with other centers, highlighting a need for close monitoring and ongoing caregiver education on recognition and management of life-threatening events at home.13,14 Ong et al. were able to reduce the tracheostomy-related mortality significantly by a quality improvement initiative of Trach safe practices by surveillance bronchoscopies, training of community-based nurses, and including a clinic survey on near missed events at clinic visits.13 The mortality rate significantly decreased after 2 years of age. Our unexpected findings in unadjusted and adjusted statistical model, that the use of inhaled medications at hospital discharge was associated with decreased risk of death by 40%, and that the number of oral medications at discharge was associated with increased risk of death by 24%, needs further elucidation. Our general observation is that patients with underlying cardiac, neurological, and genetic abnormalities are on several oral medications, and our use of the “number of oral medications” as a surrogate for either “severity of primary disease” or “presence of other comorbidity” may have led to this association. Recently, it was reported that infants with severe BPD are exposed to an alarming number of medications of unclear efficacy and safety, with marked variation between centers. Therefore, controlled trials are urgently needed for examining the use of both inhaled and oral medications in this high-risk populations.15–17
In our study, the mean age of complete discontinuation of mechanical ventilation was 23.1 (SD 18.2) months of age (median of 21.5 [Q1—15.2, Q3—27]) and decannulation at a mean age of 38.1 (SD 20.5) months (median of 32 (Q1—26.2, Q3—46.25). These results were consistent with those Cristea et al. reported on the outcomes for infants with severe BPD with a median age of complete discontinuation of mechanical ventilation at 24 months of age and decannulation at 37.5 months of age.12,18 The decannulation rates for pediatric tracheostomy patients, as published in the literature, range from 31 to 75%.19–26 Infants with severe BPD have increased chance of decannulation and survival due to improved care and ventilation to support constant lung growth.12 Some of the variations in the rate and timing of decannulation may be inherent to the original indication for placing a tracheostomy, variation in the included age groups, or to center-specific practices. In our study, 30% of infants were decannulated by 4 years of age. According to Cristea et al., 59% of their infants were decannulated by 6 years. Salley et al. reported that 30% were decannulated by 5 years, with a median time to decannulation of 30 months of age.8,12 It is important to note that those preterm infants with airway anomalies were liberated from the ventilator earlier, as most of them did not have severe lung disease. Since 2013, our practice has incorporated the use of polysomnogram to determine the patient’s favorability for decannulation as reported by Henningsfield et al.27,28 As use of polysomnogram was started recently, only 30% of the cohort had a sleep study/polysomnogram prior to decannulation. Additionally, the waiting period to schedule a polysomnogram may have resulted in an unintended delay in the timing of decannulation.
Until recently, no data existed describing the association between tracheostomy placement and change in nutritional and developmental status in preterm infants with severe BPD. In 2018, Luo et al. reported that tracheostomy was associated with improved growth and developmental outcomes in severe BPD patients while in the hospital.29 To our knowledge, growth and nutritional outcomes of infants with tracheostomy after hospital discharge have not been studied. We followed the z-scores of infants in our cohort over a 3-year period after birth. We found significant improvement in weight and weight for length z-scores by 6 months to 1 year of age that continued through 3 years of age with no significant reduction in their scores over time. This important finding was consistent in every subcategory of our infants with tracheostomy. We attributed this finding to the role that our dedicated dietitian monitors the growth of each infant closely during each clinic visit, by adjusting fluid and nutritional intake based on the infant’s caloric needs during baseline status and when infants are ill or during the period when the infants are in the process of weaning from the home ventilator. Additionally, almost all these infants have gastrostomy tubes that provided a guaranteed route for maximum nutritional support for several years. Gastrostomy tubes also plays important role during the periods of illness and avoids readmissions to the hospital.
BPD is the most common complication of prematurity.30,31 The survival of extremely premature infants who remained ventilator dependent at 36 weeks’ postmenstrual age has resulted in increased performance of tracheostomy for long-term assisted ventilation for infants who are cared for either in the intensive care units or at home. Acquired airway abnormalities, specifically TBM alone or in association with BPD, are some of the most common indications for tracheostomy in our preterm population. TBM has been recognized only recently as a common complication in infants with BPD and postulated to be the result of prolonged exposure to mechanical forces on the developing preterm airways.32,33 Infants with cardiac, neurologic, and other conditions for which they received tracheostomy were born with higher GA and may not necessarily have BPD by strict definition. These infants could be more appropriately categorized as having chronic lung disease of infancy.34 We believe it is important to distinguish between these tracheostomy indications and clinical phenotypes because their ultimate outcomes may be different.
Although no national standard exists for when to perform tracheostomy for purposes of prolonged mechanical ventilation in infants, the mean age at tracheostomy for our cohort was 4.5 months and was consistent across all tracheostomy indications. This age is similar to what has been reported in the literature.8 Although all of the infants were ventilator dependent at the time of tracheostomy, 19% of them were successfully weaned off prior to hospital discharge. The median age at hospital discharge of 7 months (Q1—4, Q3—9) may not be a true measure of the patient’s need for continued hospitalization, since many factors could affect individual discharge needs, including patient safety concerns, availability of skilled primary caregivers and home care nurses, as well as psychosocial and socioeconomic barriers, as evidenced by our finding that almost 9% of infants in our cohort were discharged to foster care.
Children who require tracheostomy and ventilatory support have multiple comorbidities. Dysphagia (oropharyngeal phase) was the most common comorbidity, identified in as many as 55% of our cohort. The other comorbidities identified were obstructive sleep apneas, seizures, pulmonary hypertension, and asthma or airway hyperactivity. Each of these comorbidities, and the use of medications to treat them, was specific to each of the primary tracheostomy indications. For purposes of comparison, we assigned each medication per intended target organ system. Therefore, the more the medications, the more severe the disease or the greater the number of comorbidities. In our previous study, we reported that oxygen and diuretic use in infants with BPD increased around 1 year of age and then gradually decreased by 2 years of age.6 In this study, we found a similar trend for gastrointestinal and renal medications; however, cardiac medications, endocrine medications, neurologic medications, and inhaled corticosteroids did not decrease significantly over the 3-year period. A probable reason for this difference could be the weaning of diuretics for severe BPD patients. At the same time, inhaled steroids continue to be needed to treat the reactive airway disease or asthma that is frequently triggered by viral infection in these infants. On the other hand, infants with underlying cardiac, neurologic, and endocrine diagnoses did not show a downward trend in medication use throughout the first 3 years of age, probably because their underlying disease conditions were chronic and static.
A significant strength of our study is the relatively large number of infants in the cohort who had regular and consistent outpatient follow-up care by a dedicated team throughout the first 3–4 years of age. This large number allows for more extensive statistical modeling that can better help guide families and care providers on important prognostic factors, outcomes, and follow-up care plans throughout the first few critical years of life. Specific to our cohort of infants with severe BPD with tracheostomy, we found through regression analysis that they have a much higher rate of survival compared to term infants with tracheostomy, as well as a higher rate of decannulation. We postulate that the improved overall survival outcomes in these very high-risk infants is the result of providing a dedicated multidisciplinary approach that ensures timely, regular, and consistent delivery of both primary and subspecialty care, along with optimizing nutritional support to enhance pro-growth state, incorporating developmental surveillance in every clinic visit, as well as addressing the caregivers’ psychosocial issues. Our study also has several limitations, which include its retrospective nature and the short timespan over which the cohort data were collected. Our study was small enough and we were unable to further analyze the patients in “others” category and is also a limitation of this study.
In conclusion, we found that our cohort of premature infants, who received tracheostomy at <1 year of age during their initial hospitalization, had a higher rate of complete discontinuation of mechanical ventilation and of decannulation compared to term infants with various tracheostomy indications. In addition, these infants showed consistent growth and a comparable rate of survival by 3–4 years of age. A dedicated multidisciplinary team providing both primary and subspecialty care appears to be the ideal delivery care model for this vulnerable, high-risk infant population.
Supplementary information
Acknowledgements
The authors thank Linda Gratny MD, founder and former director of the Children’s Mercy Infant Tracheostomy and Home Ventilator Program; Daniel Oh and Jessica Anyaso, medical students at the University of Missouri Kansas City (UMKC), for their contribution in data abstraction; and the Medical Writing Center at Children’s Mercy for their help in editing the manuscript. No funding was received to complete this project.
Author contributions
Concept and design—G.A., J.B.T., W.M., C.L., V.S. Acquisition of data—G.A., S.M., J.N.M. Analysis and interpretation of data—G.A., J.B.T., J.N.-M., V.S., C.L., W.M. Drafting the article or revising it critically for important intellectual content—G.A. wrote the first draft of manuscript; W.M., J.B.T., C.L., and V.S. edited. Final approval of the version to be published—all authors.
Competing interests
The authors declare no competing interests.
Patient consent
Patient consent was not required for this study.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version of this article (10.1038/s41390-020-01183-x) contains supplementary material, which is available to authorized users.
References
- 1.Muller RG, Mamidala MP, Smith SH, Smith A, Sheyn A. Incidence, epidemiology, and outcomes of pediatric tracheostomy in the United States from 2000 to 2012. Otolaryngol. Head Neck Surg. 2019;160:332–338. doi: 10.1177/0194599818803598. [DOI] [PubMed] [Google Scholar]
- 2.Lewis CW, Carron JD, Perkins JA, Sie KCY, Feudtner C. Tracheotomy in pediatric patients: a national perspective. Arch. Otolarngol. Head Neck Surg. 2003;129:523–529. doi: 10.1001/archotol.129.5.523. [DOI] [PubMed] [Google Scholar]
- 3.Overman AE, et al. Tracheostomy for infants requiring prolonged mechanical ventilation: 10 years’ experience. Pediatrics. 2013;131:e1491–e1496. doi: 10.1542/peds.2012-1943. [DOI] [PubMed] [Google Scholar]
- 4.Isaiah A, Moyer K, Pereira KD. Current trends in neonatal tracheostomy. JAMA Otolaryngol. Head Neck Surg. 2016;142:738–742. doi: 10.1001/jamaoto.2016.1107. [DOI] [PubMed] [Google Scholar]
- 5.Ehrenkranz RA, et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116:1353–1360. doi: 10.1542/peds.2005-0249. [DOI] [PubMed] [Google Scholar]
- 6.Akangire G, et al. Factors leading to rehospitalization for tracheostomized and ventilator-dependent infants through 2 years of age. J. Perinatol. 2017;37:857–863. doi: 10.1038/jp.2017.54. [DOI] [PubMed] [Google Scholar]
- 7.Gien J, et al. Retrospective analysis of an interdisciplinary ventilator care program intervention on survival of infants with ventilator-dependent bronchopulmonary dysplasia. Am. J. Perinatol. 2017;34:155–163. doi: 10.1055/s-0036-1584897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Salley, J., Kou, Y.-F., Shah, G. B., Mitchell, R. B. & Johnson, R. F. Survival analysis and decannulation outcomes of infants with tracheotomies. Laryngoscope130, 2319–2324 (2020). [DOI] [PubMed]
- 9.Sterni LM, et al. An Official American Thoracic Society Clinical Practice Guideline: pediatric chronic home invasive ventilation. Am. J. Respir. Crit. Care Med. 2016;193:e16–e35. doi: 10.1164/rccm.201602-0276ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roberts J, et al. Pediatric tracheostomy: a large single-center experience. Laryngoscope. 2020;130:E375–E380. doi: 10.1002/lary.28160. [DOI] [PubMed] [Google Scholar]
- 11.Zhu H, et al. Hospitalizations in children with preexisting tracheostomy: a national perspective. Laryngoscope. 2015;125:462–468. doi: 10.1002/lary.24797. [DOI] [PubMed] [Google Scholar]
- 12.Cristea AI, Carroll AE, Davis SD, Swigonski NL, Ackerman VL. Outcomes of children with severe bronchopulmonary dysplasia who were ventilator dependent at home. Pediatrics. 2013;132:e727–e734. doi: 10.1542/peds.2012-2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ong T, et al. The Trach Safe Initiative: a quality improvement initiative to reduce mortality among pediatric tracheostomy patients. Otolaryngol. Head Neck Surg. 2020;163:221–231. doi: 10.1177/0194599820911728. [DOI] [PubMed] [Google Scholar]
- 14.Ortmann LA, Manimtim WM, Lachica CI. Outcomes of tracheostomy in children requiring surgery for congenital heart disease. Pediatr. Cardiol. 2017;38:296–301. doi: 10.1007/s00246-016-1512-6. [DOI] [PubMed] [Google Scholar]
- 15.Bamat NA, et al. Medication use in infants with severe bronchopulmonary dysplasia admitted to United States children’s hospitals. J. Perinatol. 2020;39:1291–1299. doi: 10.1038/s41372-019-0415-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Puia-Dumitrescu M, et al. Medications and in-hospital outcomes in infants born at 22–24 weeks of gestation. J. Perinatol. 2020;40:781–789. doi: 10.1038/s41372-020-0614-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Greenberg JM, et al. Respiratory medication use in extremely premature (<29 weeks) infants during initial NICU hospitalization: results from the prematurity and respiratory outcomes program. Pediatr. Pulmonol. 2020;55:360–368. doi: 10.1002/ppul.24592. [DOI] [PubMed] [Google Scholar]
- 18.Wang CS, Kou Y-F, Shah GB, Mitchell RB, Johnson RF. Tracheostomy in extremely preterm neonates in the United States: a cross-sectional analysis. Laryngoscope. 2020;130:2056–2062. doi: 10.1002/lary.28304. [DOI] [PubMed] [Google Scholar]
- 19.Carron JD, Derkay CS, Strope GL, Nosonchuk JE, Darrow DH. Pediatric tracheotomies: changing indications and outcomes. Laryngoscope. 2000;110:1099–1104. doi: 10.1097/00005537-200007000-00006. [DOI] [PubMed] [Google Scholar]
- 20.de Trey L, Niedermann E, Ghelfi D, Gerber A, Gysin C. Pediatric tracheotomy: a 30-year experience. J. Pediatr. Surg. 2013;48:1470–1475. doi: 10.1016/j.jpedsurg.2012.09.066. [DOI] [PubMed] [Google Scholar]
- 21.French LC, et al. Tracheotomy in the preschool population: indications and outcomes. Otolaryngol. Head Neck Surg. 2007;137:280–283. doi: 10.1016/j.otohns.2007.02.021. [DOI] [PubMed] [Google Scholar]
- 22.Funamura JL, Durbin-Johnson B, Tollefson TT, Harrison J, Senders CW. Pediatric tracheotomy: indications and decannulation outcomes. Laryngoscope. 2014;124:1952–1958. doi: 10.1002/lary.24596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McPherson ML, et al. A decade of pediatric tracheostomies: indications, outcomes, and long-term prognosis. Pediatr. Pulmonol. 2017;52:946–953. doi: 10.1002/ppul.23657. [DOI] [PubMed] [Google Scholar]
- 24.Özmen S, Özmen ÖA, Ünal ÖF. Pediatric tracheotomies: a 37-year experience in 282 children. Int. J. Pediatr. Otorhinolaryngol. 2009;73:959–961. doi: 10.1016/j.ijporl.2009.03.020. [DOI] [PubMed] [Google Scholar]
- 25.Zenk J, et al. Tracheostomy in young patients: indications and long-term outcome. Eur. Arch. Oto-Rhino-Laryngol. 2009;266:705–711. doi: 10.1007/s00405-008-0796-4. [DOI] [PubMed] [Google Scholar]
- 26.Bandyopadhyay A, et al. Retrospective analysis of factors leading to pediatric tracheostomy decannulation failure: a single-institution experience. Ann. Am. Thorac. Soc. 2017;14:70–75. doi: 10.1513/AnnalsATS.201607-553OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Henningfeld JK, et al. Liberation from home mechanical ventilation and decannulation in children. Pediatr. Pulmonol. 2016;51:838–849. doi: 10.1002/ppul.23396. [DOI] [PubMed] [Google Scholar]
- 28.Bashir A, Henningfeld JK, Thompson NE, D’Andrea LA. polysomnography provides useful clinical information in the liberation from respiratory technology: a retrospective review. Pediatr. Pulmonol. 2018;53:1549–1558. doi: 10.1002/ppul.24164. [DOI] [PubMed] [Google Scholar]
- 29.Luo J, et al. Improved growth and developmental activity post tracheostomy in preterm infants with severe BPD. Pediatr. Pulmonol. 2018;53:1237–1244. doi: 10.1002/ppul.24087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bancalari E, Jain D. Bronchopulmonary dysplasia: 50 years after the original description. Neonatology. 2019;115:384–391. doi: 10.1159/000497422. [DOI] [PubMed] [Google Scholar]
- 31.Akangire G, et al. Clinical outcomes among diagnostic subgroups of infants with severe bronchopulmonary dysplasia through 2 years of age. Am. J. Perinatol. 2018;35:1376–1387. doi: 10.1055/s-0038-1655761. [DOI] [PubMed] [Google Scholar]
- 32.Hysinger E, Friedman N, Jensen E, Zhang H, Piccione J. Bronchoscopy in neonates with severe bronchopulmonary dysplasia in the NICU. J. Perinatol. 2019;39:263–268. doi: 10.1038/s41372-018-0280-y. [DOI] [PubMed] [Google Scholar]
- 33.Hysinger EB, et al. Tracheobronchomalacia is associated with increased morbidity in bronchopulmonary dysplasia. Ann. Am. Thorac. Soc. 2017;14:1428–1435. doi: 10.1513/AnnalsATS.201702-178OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Healy F, Hanna BD, Zinman R. Pulmonary complications of congenital heart disease. Paediatr. Respir. Rev. 2012;13:10–15. doi: 10.1016/j.prrv.2011.01.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.