Abstract
World Health Organisation declared COVID-19 a pandemic on March 11, 2020. It was temporarily named as 2019-nCoV then subsequently named as COVID-19 virus. A coronavirus is a group of viruses, known to be zoonotic, causing illness ranging from acute to mild respiratory infections. These are spherical or pleomorphic enveloped particles containing positive sense RNA. The virus enters host cells, its uncoated genetic material transcribes, and translates. Since it has started spreading rapidly, protective measures have been taken all over the world. However, its transmission has been proved to be unstoppable and the absence of an effective drug makes the situation worse. The scientific community has gone all-out to discover and develop a possible vaccine or a competent antiviral drug. Other domains of biological sciences that promise effective results and target somewhat stable entities that are proteins, could be very useful in this time of crisis. Proteomics and metabolomics are the vast fields that are equipped with sufficient technologies to face this challenge. Various protein separation and identification techniques are available which facilitates the analysis of various types of interactions among proteins and their evolutionary lineages. The presented review aims at confronting the question: ‘how proteomics can help in tackling SARS-CoV-2?’ It deals with the role of upcoming proteome technology in these pandemic situations and discusses the proteomics approach towards the COVID-19 dilemma.
Keyword: Proteomics, Metabolomics, COVID-19, SARS-CoV-2, Severe acute respiratory syndrome, Naso-oropharyngeal swab, Blood, Plasma, Sputum, Feces, Biomarkers, Targeted therapy
Introduction
Revolving from simple pneumonia to a pandemic, COVID-19 has come a long way in contributing towards the mortality rate globally. The disease is caused by a novel coronavirus SARS-CoV-2 (severe acute respiratory syndrome coronavirus-2). Apart from this, the other two viruses of the coronavirus family i.e., MERS-CoV (Middle East respiratory syndrome coronavirus) and SARS-CoV (severe acute respiratory syndrome coronavirus) have also been the cause of mass destruction since the beginning of the 21st century (Walls et al. 2020). Coronaviruses (CoVs) are the largest group of viruses belonging to the order Nidovirales, which includes Coronaviridae, Arteriviridae, and Roniviridae families. Coronaviridae or CoVs includes a clan of positive-sense, single-stranded, non-segmented, enveloped RNA viruses of vertebrates. These viruses contain the largest genomes of 25–31 kb and are infectious when introduced into permissive cells. They can be further classified into four other genera, i.e., alpha-coronavirus, beta-coronavirus, gamma-coronavirus, and delta coronavirus (Yang et al. 2015). Six human coronaviruses (HCoVs) are known to date, i.e., HCoVs-NL63 and HCoVs-229E (alpha CoVs), HCoVs-OC43, HCoVs-HKU1 (beta CoVs), SARS-CoV, and MERS-CoV. Due to their vast distribution, the frequent recombining ability of their genome, large genetic diversity, and zoonotic behaviour, novel coronaviruses have been evident to appear periodically in humans (Wu and McGoogan 2020).
The origin of this highly contagious disease is reported to be epidemiologically linked with a seafood market in Wuhan, Hubei Province, China (Zhou and Yang 2020). The zoonotic transmission of this beta coronavirus from the seafood market of Wuhan has stormed the world by spiking death rates. It was reported in 213 countries and territories affecting more than 36,738,690 people, accounting for 1,066,412 deaths. At the time of writing this manuscript, as of 00:00 GMT + 0, 09 October 2020, India has reported 6,903,812 cases so far, 106,521 deaths and 894,084 currently active cases (https://www.worldometers.info/coronavirus). Similar to a chain reaction, the rapid human-to-human transmissibility has turned a majority of the world’s population into viral carriers and incubators (Chan et al. 2020). The numbers of cases are increasing with more than 316,256 new cases being reported daily (https://www.worldometers.info/coronavirus). The infected individual might remain asymptomatic for days and the initial flu-like symptoms culminate into an elevated inflammatory response (Yuan et al. 2020). The sudden outbreak and accelerated spreading of COVID-19 [SARS-CoV-2] infection have caused substantial public concerns. Within about two months, close to one million individuals worldwide had been infected, leading to about 1,78,371 deaths (Heng et al. 2020).
Most COVID-19 [SARS-CoV-2] studies have focused on its epidemiological and clinical characteristics (Ghinai et al. 2020; Li et al. 2020). About 80% of patients infected with SARS-CoV-2 displayed mild symptoms with good recovering capabilities. They usually recover with or even without conventional medical treatment and, therefore, are classified as mild or moderate COVID-19 (Baden et al. 2020). However, about 20% of patients suffer from respiratory failure and need immediate oxygen therapy or other inpatient interventions, including mechanical ventilation (Murty et al. 2020; Wu et al. 2020). These patients are classified as clinically severe and are mainly diagnosed empirically based on a set of clinical characteristics, such as respiratory rate (≥ 30 times/min), mean oxygen saturation (≤ 93% in the resting state), or arterial blood oxygen partial pressure/oxygen concentration (≤ 300 mmHg) (Baden et al. 2020). However, patients exhibiting these clinical manifestations have already progressed to a clinically severe phase and require immediate access to specialized intensive care; otherwise, they may die rapidly. Therefore, it is critical to developing new approaches to predict the cases which might progress to clinically severe condition. Besides, effective therapy for severe patients remains hypothetical, largely due to limited understanding of COVID 19 SARS-CoV-2 pathogenesis.
The current review aims at analyzing the current status of clinical proteomics and metabolomics with particular emphasis on SARS-CoV2 biology and approaches for its treatment. Therapeutic strategies majorly aim at proteins and not nucleic acids as drug targets. Though the technologies available to date, such as microarray, succeeds in identifying an enormous count of differentially expressed genes but fail to justify their multiple protein products and functionality. On the other hand, proteome analysis aims not only in identifying the differential expression of proteins but also to take account of protein–protein interactions, post-translational modifications, the temporal pattern of expression, and cellular and sub-cellular distribution. Hence, differential and functional proteomics generate information that leads to improved interpretation of the cellular pathways and how they are linked in cells and living organisms. For many decades, proteomics has proved its versatility and efficacy for the development of the novel potential drug targets for constantly appearing diseases posing challenges to humankind. Currently, the world of proteomics has stuck with the dilemma to overcome the proposed oppositions of the SARS-CoV2 virus for drug and vaccine development.
Loopholes in current strategies
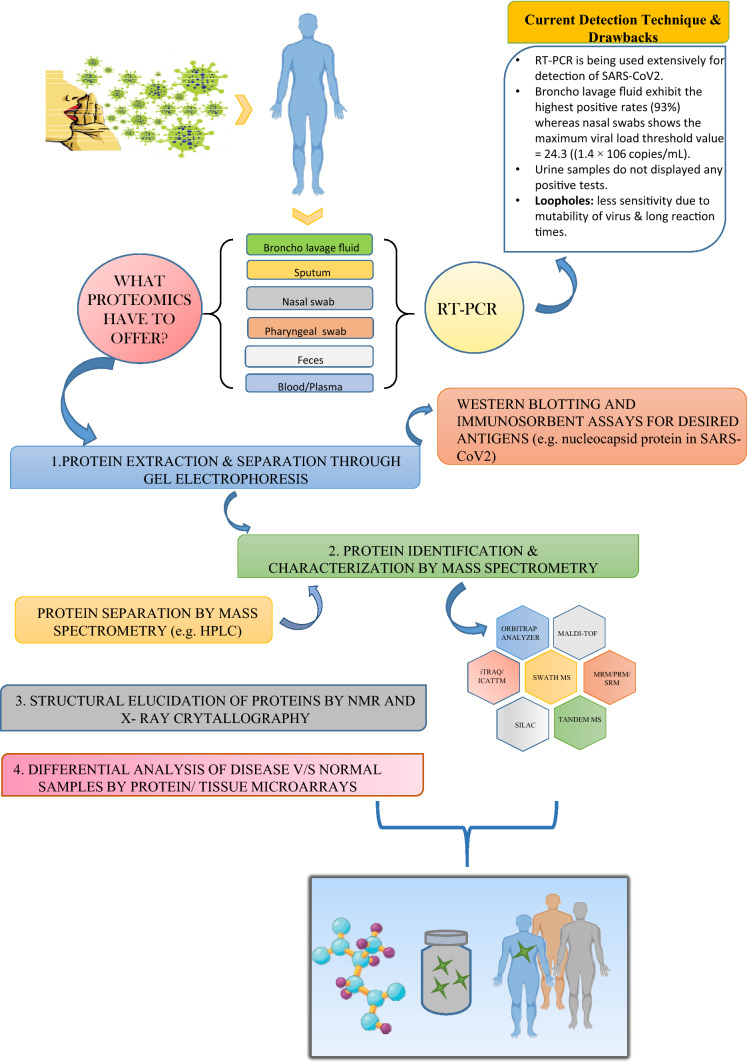
Currently, reverse transcription-polymerase chain reaction is the most reliable method to detect ssthe viral genes in the COVID-19-positive patients. The method is widely available and well established, exhibits many problems in its performance. Due to the potent mutability of the viral genes, the technique might not be sensitive enough to detect the virus if it is mutated. Furthermore, RT-PCR tends to exhibit low throughput due to intermediate and long reaction times (Tahamatan et al. 2020). Beyond these facts, serological tests involving the presence of specific antibodies are also important as they identify false-negative RT-PCR responses. They also track how effectively the patient’s immune system is working against the infection by quantifying the metabolites and antibodies and are essentially helpful for plasma transfusion therapies (Funari et al. 2020). These tests are based on the detection of antigens from nasopharyngeal swabs using antibodies. A positive test confirms the presence of the virus, however, a negative test is inclusive as the sensitivity of the antigen test is between 34 and 80% (Bruning 2018).
How can proteomics pave the way to counter the SARS-CoV-2?
Structural proteome analysis of earlier SARS epidemic in 2003 revealed a large array of proteins that could be targeted for this pandemic too. The SARS-CoV genome encodes 28 proteins of which the structure of 16 proteins or their functional domain has been determined until today. Interestingly, out of these 16 proteins, eight of them have novel functional domains that indicate the distinctiveness of coronavirus proteins (Bartlam et al. 2007). Stukalov et al. identified the interactions of SARS-CoV and SARS-CoV-2 with cellular proteins. They used affinity purification followed by mass spectrometry analysis and statistical modeling of the MS1-level quantitative data which allowed the identification of 1484 interactions between 1086 cellular proteins and 24 SARS-CoV bait proteins. Particularly, they discovered that SARS-CoV-2 targets several cellular regulators involved in innate immunity (ORF7BMAVS,—UNC93B1), stress response components (N-HSPA1A), and DNA-damage response mediators (ORF7a-ATM, -ATR). In a nutshell, SARS-CoV-2 interacts with specific protein complexes contributing to a range of biological processes (Stukalov et al. 2020). Another study elaborated on the use of SELDI-TOF (surface-enhanced laser desorption/ionization) in early detection of the SARS virus. The analysis results in an array of proteins that are specific to the infectious agents (Marzulli et al. 2005).
Wide arrays of techniques are available at hand for the segregation and identification of proteins from complex mixtures which could render a helping hand in this global pandemic. The most common separating methods include: one- and two-dimensional gel electrophoresis and high-performance liquid chromatography (HPLC) whereas mass spectrometry (MS) is the yardstick in the world of protein identification (Verills 2006). The overall workflow of proteomics approaches for COVID-19 patient shown in Fig. 1.
Fig. 1.
A schematic representation of protemics techniques for SARS-CoV2
Conventional techniques
The conventional techniques for purification and segregation of proteins include affinity-based chromatography, ion-exchange chromatography and size-exclusion chromatography. Enzyme-linked immunosorbent assay (ELISA) and western blotting techniques can be used for analysis of selective proteins. Though, still in use but these techniques restrict the analysis to smaller number of proteins and also incapable to define expression level of proteins. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis, two-dimensional and two-dimensional differential gel electrophoresis are used for separation of complex protein samples (Aslam et al. 2017).
A current intervention in the 2D-Gel Electrophoresis has allowed the researchers to analyze the protein complement of the genome targeted. The technique now provides high-throughput and high-resolution separation of proteins. An improvement, such as the introduction of IPG (immobilized pH gradients) strips and their employment with a single pH range, has not only increased the sensitivity but also specify the location of proteins in a cell or tissue extracts (Bjellqvist et al. 1982; Scherl et al. 2002; Cordwell et al. 2000). A step ahead of 2D-GE, the introduction of cyanine fluorescent dyes for the pre-labeling of proteins has made this approach a valuable tool in proteomics. Due to the presence of Cye dye fluorophores which enable the co-migration of multiple proteins and their simultaneous detection, 2D-DIGE allows faster and more reliable gel matching. Labeling before the fractionation prevents the alteration due to gel to gel variation and provides a good dynamic range. Hence, multiple proteins can be analyzed directly in one gel (Tonge et al. 2001; Pasquali et al. 2017; Eggeling et al. 2001). The efficacy of 2D-DIGE has been validated for numerous applications for various cancers and other diseases (Seike et al. 2004; Isaaq et al. 2007).
Advanced technologies
Mass spectrometry
Protein identification and profiling through mass spectrometry techniques have now overpowered the loopholes of various other proteome technologies (including 2D gels) which require a large amount of purified protein for the analyses. MALDI-TOF or Matrix-assisted ionization-time of flight MS has accelerated the identification of proteins isolated by 2D-gel electrophoresis and other methods to the elevated levels because of its sensitivity and speed (Aebersold et al. 2003). Combining the latter with identification of protein sequence, searching through available databases has facilitated the process of drug discovery and development in an enormous amount. Although a very powerful technique, 2D-GE sometimes fails to separate proteins of very high and low molecular weight, thus methods, such as free-flow electrophoresis (FFE), have been developed which enables the resolution of a complex mixture of proteins by combining 2D-GE with the liquid-based IEF method (Hoffmann et al. 2001).
Mass spectrometry can also be seen as the backbone of the various proteomic technologies. It uplifts the studies, enlightens the new findings, and facilitates in taking a step ahead in the analysis. MS-based viral peptide detection has earlier been used for the detection of viral proteins which affect respiratory pathways (Foster et al. 2015; Majchrzykiewicz-Koehorst et al. 2015). It had been a phenomenal technique in the characterization of SARS viruses in the later epidemic. A study employing MALDI-TOF MS used convalescent sera from several SARS patients to detect proteins in the culture supernatants from cells exposed to lavage of another SARS patient. The technique identified a very prominent protein of molecular weight 46 kDa approx. which was found to be a novel nucleocapsid protein, further established to be the major immunogen. Another 139 kDa spike glycoprotein was also examined by MALDI-TOF which was the possible target for the prophylactic intervention in encountering SARS (Krokhin et al. 2003). SARS-CoV-2-specific peptides have also been reported and shortlisted for targeted mass spectrometric studies by Gouveia et al. They used infected cell lysate to identify these peptides and validated these peptides in a nasopharyngeal swab. (Gouveia et al. 2020a, b). Another study using a gargle solution of three patients reported a peptide from nucleoprotein using a 180 min gradient. The study involves LC–MS analysis on a nano-HPLC system coupled to an Orbitrap Fusion Tribid mass spectrometer with nano-ESI source after acetone precipitation and tryptic digestion of the proteins contained within the gargle solution (Ihling et al. 2020). Three peptides have also been reported using 10.5 min run time with 83% sensitivity and 96% specificity (Cardozo et al. 2020). A simple and rapid method that could be used to detect the presence of the virus even in the recovered patients has also been developed. The technique employed multiple reaction monitoring mass spectrometry (MRM-MS) and detected two peptides, QIAPGQTGK and AIVSTIQRKYK, from structural spike glycoprotein and replicase polyprotein 1 ab, respectively, in 2.3 min gradient with a sensitivity of 90% and specificity 100%. They used this method for asymptomatic recovered patients which tested negative for RT-PCR analyses. Hence, suggested the possible application of this detection mechanism in asymptomatic subjects also (Singh et al. 2020). One such study developed an automated antibody capture-based workflow coupled with targeted high-field asymmetric ion mobility spectrometry (FAIMS)-parallel reaction monitoring (PRM) assays on an Orbitrap Exploris 480 mass spectrometer. The study is based on analysis of 363 nasopharyngeal residual swab samples from patients. It involves low-flow LC method followed by a PRM MS that incorporates ion mobility for selected viral peptides. The study confirms the nucleocapsid protein to be the major target antigen, based on the experiments done on nasopharyngeal swabs from COVID-19 patients, recombinant viral proteins and purified virus. They established a collective machine learning-based model for identifying COVID-19-positive samples employing fragment ion intensity in the PRM data. Particularly, their study resulted in 97.8% sensitivity and 100% specificity comparable to RT-PCR-based molecular testing (Renuse et al. 2020). SWATH-MS (Sequential Windowed Acquisition of All Theoretical Fragment Ion Mass Spectra) that allows complete recording of all the fragment ions of the detectable peptide precursor present is a biological sample. To maintain sensitivity, selectivity and accuracy, SWATH-MS incorporates data-independent acquisition and targeted data analysis. It has been used for biomarker discovery in various diseases and, hence, can be used for COVID-19 as well. (Collins et al. 2017; Rosenberger et al. 2017).
Coupling basic protein analytical techniques with mass spectrometry enhances the sensitivity (to the femtomole levels), resolution, and accuracy of the examination. Protein complexes are cleaved into smaller fragments and detected by the mass spectrometer. Peptides can be detected in various ways using different mass spectrometers. Time-of-flight (TOF) MS instruments involve the peptides to fly down a flight tube and the time taken to reach the detector indicates the peptide mass. Quadrupoles, ion traps, and FTICR (Fourier-transform ion cyclotron resonance) are some other types of mass analyzers that make the mass spectrometers more profound and reliable for peptide analysis (Lim et al. 2004). The mass spectrum obtained gives the list of peptide masses that are extensively searched in genome databases, translated and trypsin digested in silico. Another progression in the field of mass spectrometry is fractionating protein complexes multiple times (usually twice) into individual amino acids, it is termed as tandem mass spectrometry or MS/MS. This allows the determination of de novo peptide sequences and, hence, its identification through databases against known proteins (Seidler et al. 2010).
Chemical labelling techniques
Tagging protein fragments with heavy or light isotopes before exposing them to mass spectrometers makes the qualitative and quantitative analyses further easy. Labeling techniques, such as isotope-coded affinity tags (ICATTM) (Smolka et al. 2001), O18-water labeling (Ye et al. 2009), isotope tags for relative and absolute quantification (iTRAQ) (Wiese et al. 2007), and stable isotope labeling with amino acids in cell culture (SILAC) (Mann 2006), allow the subsequent identification and quantification effortlessly.
A recent study involving chemical labelling identifies the host cell pathways that are modulated by SARS-CoV2 and thus showed that inhibition of these pathways prevents the virus from replicating. A human cell culture system was established for infection and further isotope labelled for the LC–MS/MS proteome analysis. Their analysis revealed that SARS-CoV2 reshapes the central cellular pathways, such as carbon metabolism, proteostasis, nucleic acid metabolism, translation, splicing and carbon metabolism. Viral replication was inhibited by small molecules that target these pathways. This study can be a boon in these situations as it reveals the cellular infection profile of SARS-CoV2 for which drugs could be developed. Their finding provides insights for the development of therapies for the treatment of coronavirus (Bojkova et al. 2020).
Protein chips
Protein chips or protein microarrays could be an astonishing approach to detect and characterize the peptides available in a protein complex. Chip technology has been widely used in the field of nucleic acids but its applicability in proteomics is not very well versed. As proteins are extensively heterogeneous, a simple chip for all types of proteins has not been achieved yet. While DNA is stable, proteins tend to lose their three-dimensional structure and, hence, their functionality and specificity as the conditions fall out of a narrow range (Eickhoff et al. 2002). Though, a variety of proteins and peptide arrays have been developed for the analysis of specific or group of proteins, Surface-Enhanced Laser desorption-ionization (SELDI) Protein ChipR, developed by Ciphergen Biosystems, Inc., involves affinity capture of specific subgroups of proteins based on their biochemical or physical properties which is further analyzed with automated MS (Merchant et al. 2000). A low abundant protein that fails to be detected by 2D-GE can be efficiently detected by this technique. Forward-phase arrays and reverse-phase arrays are the two microarray platforms being developed in this series. These two arrays work just opposite to each other. In forward-phase array, an antibody is immobilized on the solid surface and the sample protein mixture is poured on the latter whereas, in reverse-phase array, the sample is immobilized on the surface and then reacted with a specific antibody. Bound molecules are detected by secondary antibody or direct labeling (Liotta et al. 2003; Phizicky et al. 2003). Functional microarray chips can be used to study protein–protein interactions, DNA–protein interactions, and drug-target identification (Phizicky et al. 2003).
Despite all the challenges, protein microchips have also been used in the detection and characterization of SARS viruses. A study involving screening of specific IgG antibodies of 13 recombinant proteins associated with four structural proteins (S, E, N, and M and five putative uncharacterized proteins of SARS-CoV (3a, 3b, 6, 7a and 9b). The results suggested that anti-S and anti-N antibodies are diagnostic markers and S3 is immunogenic thus a potential candidate for vaccine development (Qiu et al. 2005). Zhu et al. has constructed a protein microarray that includes proteins from SARS coronavirus (SARS-CoV) and five other additional coronaviruses. They also developed a computer algorithm that uses multiple classifiers to predict samples from SARS patients and used to predict 206 sera from Chinese fever patients. The test also identified patients with sera reactive against other coronavirus proteins. They further correlated these results with an indirect immunofluorescence test and suggested that viral infection can be scrutinized for months after infection. Thus, this study proved protein microarrays can serve as a sensitive, rapid, and simple tool for large scale identification of viral-specific antibodies in sera (Zhu et al. 2006). A significant number of studies have been published that proves protein microarrays to be a valuable technique for diagnosis and screening of viral peptides (Lu et al. 2005; Reusken et al. 2013) A recently published study involves the development of an Opto-microfluidic sensing platform to rapidly detect antibodies against SARS-CoV2 spike protein in diluted human plasma with high sensitivity. The technique was developed based on localized surface plasmon resonance (LSPR) involving gold nanospikes in a microfluidic device which is further coupled with an optical probe. The platform achieves the limit of detection of 0.5 pM and takes up to 30 min to analyze a sample. This could be a great improvisation in the diagnosis of the COVID-19 (Funari et al. 2020).
Tissue microarrays (TMA)
Taking Immunohistochemistry a level ahead, tissue chenmicroarrays involve the analysis of protein expression in a multitude of tissue samples and IHC to be used in a high-throughput setting (Andersson et al. 2006). TMA plays an important role in target validation of results from cDNA arrays and expression profiling of tissues which have the major significance in proteomic research (Avninder et al. 2008). This technology has been proven significant in various types of cancers including breast cancer, adenocarcinoma, lung cancer, etc. (Jacquemier et al. 2005).
Structural elucidation by nuclear magnetic resonance (NMR)
Techniques, such as nuclear magnetic resonance spectroscopy and X-ray crystallography, have been in use to determine the structural parameters since a long time. NMR studies for SARS have provided countless insights in its structural factors. Innumerable articles have been published elaborating various structural aspects of the virus (Mahajan et al. 2015; Johnson et al. 2010). Structural elucidation of SARS-CoV 2 involving NMR has also provided us with helpful perceptions about this new virus. One such study analyses glycan structures of the receptor-binding domain of SARS-CoV2 spike glycoproteins that are expressed in human HEK293F cells. The study provides strong evidence for the presence of glycan structure which were not found in earlier MS-based analysis. A number of different possible interacting isotopes have been analyzed. The study proposed 3D models of these interacting complexes (Lenza et al. 2020).
Metabolome analysis of SARS
Metabolomics deals with the identification, quantification, and characterization of endogenous and exogenous metabolites. It is based on the idea that endogenous metabolites can accurately indicate even a minute variation in the metabolism. Thus, due to its high sensitivity and specificity, metabolomics can be considered as a valid tool for exploring the interrelationship between the virus and the human body. When a virus enters the body, it alters the cellular metabolism and triggers new pathways; hence, the analysis of the metabolites produced turns out to be crucial for elucidating new features related to the infection and development of better non-invasive, early methods of diagnosis. Although very few numbers of studies have been published regarding the metabolomics approach in viral infections, COVID-19 has already been analyzed by this approach (Yu et al. 2011; Mussap et al. 2013; Noto et al. 2014; Beale et al. 2019). Metabolomic and lipidomic analyses performed on the plasma of COVID-19 patients revealed a correlation between metabolite and lipid alteration and course of disease in these patients which suggested that the development of COVID-19 affected their whole-body metabolism. Particularly, malic acid (TCA cycle) and carbamoyl phosphate (urea cycle) exhibit alterations in energy metabolism and hepatic dysfunction, respectively. Moreover, carbamoyl phosphate is shown to be down-regulated in patients with severe conditions compared with patients having mild symptoms. Interestingly, guanosine monophosphate (GMP) is significantly altered between healthy and COVID-19 patients, alterations in GMP levels also differ among patients with mild and fatal symptoms. The findings of this study align with the severity of the disease and its progression. Their work provides valuable knowledge about plasma biomarkers associated with SARS-CoV-2 against which therapeutic markers can be developed (Wu et al. 2007).
Proteomics for SARS
Proteomics has been widely used in the treatment and analysis of cancer tumors, such as brain cancer ovarian tumors, breast and lung cancer, and various types of adenocarcinomas (Petricoin et al. 2002; Gast et al. 2009; Okano et al. 2006; Khalil et al. 2007). Their potential drug targets and novel biomarkers have been discovered (Srinivas et al. 2002).
It has been established that infectious diseases have always been a major cause of death worldwide. The pandemic in which we are living now has been aroused earlier also. Not just once, but SARS viruses have ascertained that the same clan of virus can be the reason for contagion for thrice. Hence, SARS viruses have always been the topic of great concern and current proteome research. Sera from patients have been analysed and a potential biomarker, truncated α-1 trypsin, has been identified. This protein marker is observed to be elevated in SARS patients as compared to healthy individuals (Yi 2004).
Drug resistance mechanism and identification of the biomarkers that are resistant to the specific strains are some of the milestones which are required to be overcome for the effective treatment of SARS and other infectious diseases. Another major setback of these infections is the similarity of symptoms among SARS and non-SARS patients. A study overpowered this obstacle and suggested that serum proteomic fingerprints could be used to identify SARS cases at their onset; thus, correct treatment could be administered simply by differentiating between SARS and non-SARS patients. By the application of SELDI (surface-enhanced laser ionization) protein chip technology, they compared 39 patients with early-stage SARS infection and 39 non-SARS patients. Their proteomic pattern was analyzed by bioinformatics and biostatistical analyses. The study showed twenty features to be significantly different in the two groups. Fifteen were increased in the SARS group and 5 were decreased (Pang et al. 2006).
An attempt to map the humoral antibody response to SARS-CoV-2 proteins can be made which may aid in the development of antibody-based assays for diagnostic and therapeutic purposes (Srivastava et al. 2020). A recent study demonstrated that some serological antibodies can neutralize the viral entry into the host cells through the angiotensin-converting enzyme 2 (ACE2) receptor. Essentially, the number of commercially available antibodies for SARS-CoV can also target SARS-CoV2 proteins (Wang et al. 2020). Another study reported a comprehensive SARS-CoV2 human protein–protein interaction map using affinity-purification mass spectrometry. In this study, 332 high-confidence, SARS-CoV2 human protein–protein interactions were defined, 66 human proteins of them could be targeted by several existing FDA approved drugs or drugs under clinical trials (Gordon et al. 2020). Integrated multi-omics studies in combination with host–pathogen interactomics analyses can lead to the recognition of therapeutic targets for this novel infection which is required for drug remodeling and development of new drugs and vaccines. Host proteome and metabolome profiles should also be considered using a nasopharyngeal swab, bronchoalveolar lavage fluid, and blood samples from patients from SARS-CoV2 for understanding its complex pathogenesis and host immune response against the virus (Srivastava et al. 2020). Another research observed dysregulation of macrophage, platelet degranulation, and complement system pathways and massive metabolic suppression in patients with severe COVID-19 when compared to non-severe patients (Shen et al. 2020). Comparative studies among patients at different levels of SARS-CoV2 (i.e., asymptomatic stage, early febrile, defervescence, and convalescent stages) must be made which are highly informative to determine how nonsevere or asymptomatic patients progress towards severe or deadly symptoms (Srivastava et al. 2020). Recently a study identified 27 potential biomarkers that are differentially expressed depending on the WHO severity grade of COVID-19 patients. The markers also check the level of inflammatory factors, coagulation factors, and inflammation modulators and indicate the upstream and downstream levels of interleukin 6 (Messner et al. 2020).
Another recent study uses Single Molecule Array assays (Simoa) for the quantitative proteome studies of the SARS-CoV-2 spike, S1 subunit, and nucleocapsid antigens in the plasma of coronavirus patients. The results showed the changes in 31 SARS-CoV-2 biomarkers in 272 longitudinal plasma samples obtained for 39 coronavirus patients. S1 and N antigens were detectable in 41 out of 64 coronavirus-positive patients. It is the first study to detect the SARS-CoV-2 antigens in the blood plasma of COVID-19-positive patients. The data revealed that the viral antigens in the blood are associated with disease progression. (Ogata et al. 2020). A study revealed four pathways that are evidently modulated during SARS-CoV2 infection in vitro. The study involves an integrative proteo-transcriptomic analysis of SARS-CoV2-infected Huh 7 cells. Western blot validation of effector molecules of identified pathways revealed a dose-dependent activation of Akt, MTOR, S6K1 and 4E-BPI at 24 h post infection (hpi). Virus production was significantly reduced by the inhibition of the mTOR signaling pathway using Akt inhibitor MK-2206. Further, the authors suggested profound studies are required for the treatment of COVID-19 patients (Appelberg et al. 2020).
Conclusion
SARS-CoV2 is highly contagious which posed considerable difficulty for systems-level molecular studies and limits its processing in high-end research facilities. However, an urgent effort from molecular biology and proteomics researchers can eliminate this impediment. This would provide us an overview of the novel biomarkers and define point-of-care clinical healthcare deliveries which are aimed at delivering less costly care closer to the patient’s home. In this developing world, there exist a challenge of more effective care for SARS-CoV2, and point-of-care testing (PoCT) may play a much greater role here in the future.
Acknowledgements
Authors are gratefully acknowledged to Sir Ganga Ram Hospital, Delhi, India for providing necessary support.
Funding
None.
Compliance with ethical standards
Conflict of interest
None.
Ethical approval
This review does not contain any studies with human participants or animals performed by the author.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Aebersold R, Mann M. Mass spectrometry-based proteomics. Nature. 2003;422:198–207. doi: 10.1038/nature01511. [DOI] [PubMed] [Google Scholar]
- Andersson A-C, Stromberg S, Backvall H, Kampf C, Uhlen M. Analysis of protein expression in cell microarrays: a tool for antibody-based proteomics. J Histochem Cytochem. 2006;54(12):1413–1423. doi: 10.1369/jhc.6A7001.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Appleberg, et al. Dysregulation in Akt/mTOR/HIF-1 signaling identified by proteo-transcriptomics of SARS-CoV-2 infected cells. Emerg Microb Infect. 2020;9(1):1748–1760. doi: 10.1080/22221751.2020.1799723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aslam B, Basit M, Nisar MA, Khurshid M, Rasool MH. Proteomics: technologies and their applications. J Chromatogr Sci. 2017;55(2):182–196. doi: 10.1093/chromsci/bmw167. [DOI] [PubMed] [Google Scholar]
- Avninder S, Ylaya K, Hewitt SM. Tissue microarray: a simple technology that has revolutionized research in pathology. Technol Rev. 2008;54(2):158–162. doi: 10.4103/0022-3859.40790. [DOI] [PubMed] [Google Scholar]
- Baden LR, Rubin EJ. Covid-19—the search for effective therapy. N Eng J Med. 2020;382:1851–1852. doi: 10.1056/NEJMe2005477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartlam M, Xu Y, Rao Z. Structural proteomics of the SARS coronavirus: a model response to emerging infectious diseases. J Struct Funct Genomics. 2007;8(2):85–97. doi: 10.1007/s10969-007-9024-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beale DJ, Oh DY, Karpe AV, Tai C, Dunn MS, Tilmanis D, Plombo EA, Hurt AC. Untargeted metabolomics analysis of the upper respiratory tract of ferrets following influenza A virus infection and oseltamivir treatment. Metabolomics. 2019;15(3):33. doi: 10.1007/s11306-019-1499-0. [DOI] [PubMed] [Google Scholar]
- Bjellqvist B, Ek K, Righetti PG, Gianazza E, Görg A, Westermeier R, Postel W. Isoelectric focusing in immobilized pH gradients: principle, methodology and some applications. J Biochem Biophys Methods. 1982;6(4):317–339. doi: 10.1016/0165-022X(82)90013-6. [DOI] [PubMed] [Google Scholar]
- Bojkova D, Klann K, Koch B, Widera M, Krause D, Ciesek S, Cinatl J, Münch C. Proteomics of SARS-CoV-2-infected host cells reveals therapy targets. Nature. 2020;583:469–472. doi: 10.1038/s41586-020-2332-7. [DOI] [PubMed] [Google Scholar]
- Bruning, et al. Rapid detection and monitoring of human coronavirus infections. N Microb N Infect. 2018;24:52–55. doi: 10.1016/j.nmni.2018.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan JF-W, Yuan S, Kok KH, To KK-W, Chu H, Yang H, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. The Lancet. 2020;395(10223):514–523. doi: 10.1016/S0140-6736(20)30154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins B, et al. Multi-laboratory assessment of reproducibility, qualitative and quantitative performance of SWATH-mass spectrometry. Nat Commun. 2017;8:291. doi: 10.1038/s41467-017-00249-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cordwell SJ, Nouwens AS, Verrills NM, Basseal DJ, Walsh BJ. Subproteomics based upon protein cellular location and relative solubilities in conjunction with composite two-dimensional electrophoresis gels. Electrophoresis. 2000;21:1094–1103. doi: 10.1002/(SICI)1522-2683(20000401)21:6<1094::AID-ELPS1094>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Eggeling FV, Gawriljuk A, Fiedler W, Ernst G, Claussen U, Klose J. Fluorescent dual colour 2D-protein gel electrophoresis for rapid detection of differences in protein pattern with standard image analysis software. Int J Mol Med. 2001;8(4):373–377. doi: 10.3892/ijmm.8.4.373. [DOI] [PubMed] [Google Scholar]
- Eickhoff H, Konthur Z, Lueking A, Lehrach H, Walter G, Nordhoff E, Nyarsik L, et al. Protein array technology: the tool to bridge genomics and proteomics. In: Hoheisel J, et al., editors. Chip technology. Berlin: Springer; 2002. pp. 103–112. [DOI] [PubMed] [Google Scholar]
- Foster MW, Gerhardt G, Robitaille L, Plante P-L, Boivin G, Corbeil J, Moseley MA. Targeted proteomics of human metapneumovirus in clinical samples and viral cultures. Anal Chem. 2015;87(20):10247–10254. doi: 10.1021/acs.analchem.5b01544. [DOI] [PubMed] [Google Scholar]
- Funari R, Chu K-Y, Shen AQ. Detection of antibodies against SARS-CoV-2 spike protein by gold nanospikes in an opto-microfluidic chip. Biosens Bioelectron. 2020;169:112578. doi: 10.1016/j.bios.2020.112578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gast MC-W, Schellens JHM, Beijnen JH. Clinical proteomics in breast cancer: a review. Breast Cancer Res Treat. 2009;116(1):17–29. doi: 10.1007/s10549-008-0263-3. [DOI] [PubMed] [Google Scholar]
- Ghinai I, et al. First known person-to-person transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in the USA. The Lancet. 2020;395(10230):1137–1144. doi: 10.1016/S0140-6736(20)30607-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon DE, Jang GM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459–468. doi: 10.1038/s41586-020-2286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gouveia D et al. (2020a) Proteotyping SARS-CoV-2 virus from nasopharyngeal swabs: a proof-of-concept focused on a 3 min mass spectrometry window. J Proteome Res [DOI] [PubMed]
- Gouveia D, Grenga L, Gaillard J-C, Gallais F, Bellanger L, Pible O, Armengaud J. Shortlisting SARS-CoV-2 peptides for targeted studies from experimental data-dependent acquisition tandem mass spectrometry data. Proteomics. 2020;20:e2000107. doi: 10.1002/pmic.202000107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heng, et al. Coronavirus disease 2019 (COVID-19): current status and future perspectives. Int J Antimicrob Agents. 2020;55(5):105951. doi: 10.1016/j.ijantimicag.2020.105951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann P, Ji H, Moritz RL, Connolly LM, Frecklington DF. Continuous free flow electrophoresis separation of cytosolic proteins from the human colon carcinoma cell line LIM 1215: a non two dimensional gel electrophoresis based proteome analysis strategy. Proteomics. 2001;1:807–818. doi: 10.1002/1615-9861(200107)1:7<807::AID-PROT807>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- https://www.worldometers.info/coronavirus. Accessed 9 Oct 2020
- Ihling C, Hagemann S, Kehlen A, Hüttelmaier S, Arlt C, Sinz A (2020) Mass spectrometric identification of SARS-CoV-2 proteins from gargle solution samples of COVID-19 patients. J Proteome Res [DOI] [PubMed]
- Issaq HJ, Veenstra TD. The role of electrophoresis in disease biomarker discovery. Electrophoresis. 2007;28:1980–1988. doi: 10.1002/elps.200600834. [DOI] [PubMed] [Google Scholar]
- Jacquemier J, Ginestier C, Rougemont J, et al. Protein expression profiling identifies subclasses of breast cancer and predicts prognosis. Can Res. 2005;65(3):767–769. [PubMed] [Google Scholar]
- Johnson M, Jaudzems K, Wüthrich K. NMR Structure of the SARS-CoV nonstructural protein 7 in solution at pH 6.5. J Mol Biol. 2010;402(4):619–628. doi: 10.1016/j.jmb.2010.07.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khalil AA. Biomarker discovery: a proteomic approach for brain cancer profiling. Cancer Sci. 2007;98(2):201–213. doi: 10.1111/j.1349-7006.2007.00374.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krokhin O, et al. Mass spectrometric characterization of proteins from the SARS virus. Mol Cell Proteomics. 2003;2(5):346–356. doi: 10.1074/mcp.M300048-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenza M et al. (2020) Structural characterization of the N‐linked glycans in the receptor binding domain of the SARS‐CoV‐2 spike protein and their interactions with human lectins using NMR spectroscopy. J German Chem Soc [DOI] [PMC free article] [PubMed]
- Li R, Pei S, Chen B, Song Y, Zhang T, Yang W, Shaman J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2) Science. 2020;368(6490):489–493. doi: 10.1126/science.abb3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim MS, Elenitoba-Johnson KSJ. Proteomics in pathology research. Lab Invest. 2004;84(10):1227–1244. doi: 10.1038/labinvest.3700167. [DOI] [PubMed] [Google Scholar]
- Liotta LA, Espina V, Mehta A, Calvert V, Rosenblatt K, Geho D. Protein microarrays: meeting analytical challenges for clinical applications. Cancer Cell. 2003;3(4):317–325. doi: 10.1016/S1535-6108(03)00086-2. [DOI] [PubMed] [Google Scholar]
- Lu D-D, Chen S-H, Zhang S-H, Zhang M-L, Zhang W, Bo X-C, Wang S-Q. Screening of specific antigens for SARS clinical diagnosis using a protein microarray. Analyst. 2005;130:474–482. doi: 10.1039/b415888a. [DOI] [PubMed] [Google Scholar]
- Mahajan M. Bhattacharya S (2015) NMR structures and localization of the potential fusion peptides and the pre-transmembrane region of SARS-CoV: implications in membrane fusion. Biochim Biophys Acta Biomembr. 1848;2:721–730. doi: 10.1016/j.bbamem.2014.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majchrzykiewicz-Koehorst JA. Rapid and generic identification of influenza A and other respiratory viruses with mass spectrometry. J Virolog Method. 2015;215:75–83. doi: 10.1016/j.jviromet.2014.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann M. Functional and quantitative proteomics using SILAC. Nat Rev Mol Cell Biol. 2006;7(12):952–958. doi: 10.1038/nrm2067. [DOI] [PubMed] [Google Scholar]
- Mazulli T, Low DE, Poutanen SM. Proteomics and severe acute respiratory syndrome (SARS): emerging technology meets emerging pathogen. Clin Chem. 2005;51(1):6–7. doi: 10.1373/clinchem.2004.041574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merchant M, Weinberger SR. Recent advancements in surface-enhanced laser desorption/ionization-time of flight-mass spectrometry. Electrophoresis. 2000;21(6):1164–1167. doi: 10.1002/(SICI)1522-2683(20000401)21:6<1164::AID-ELPS1164>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Messner CB et al (2020) Clinical classifiers of COVID-19 infection from novel ultra-high-throughput proteomics. medRxiv. [DOI] [PMC free article] [PubMed]
- Murty P, Reddy PV (2020) Mental Health in the times of COVID-19 Pandemic. Guidance for General Medical and Specialised Mental Health Care Settings 03–07
- Mussap M, Antonucci R, Noto A, Fanos V. The role of metabolomics in neonatal and pediatric laboratory medicine. Clin Chim Acta. 2013;426:127–138. doi: 10.1016/j.cca.2013.08.020. [DOI] [PubMed] [Google Scholar]
- Noto A, Dessi A, Puddu M, Mussap M, Fanos V. Metabolomics technology and their application to the study of the viral infection. J Matern Fetal Neo-natal Med. 2014;27(Suppl 2):53–57. doi: 10.3109/14767058.2014.955963. [DOI] [PubMed] [Google Scholar]
- Ogata AF, Maley AM, Wu C, Giloba T, Norman M, Lazarovits R, Mao C-P,Newton G, Chang M, Nguyen K (2020) Ultra-sensitive serial profiling of SARS-CoV-2 antigens and antibodies in plasma to understand disease progression in COVID-19 patients with severe disease. Clin Chem [DOI] [PMC free article] [PubMed]
- Okano T, Kondo T, Kakisaka T, Fujii K, Yamada M, Kato H. Plasma proteomics of lung cancer by a linkage of multi-dimensional liquid chromatography and two-dimensional difference gel electrophoresis. Proteomics. 2006;6(13):3938–3948. doi: 10.1002/pmic.200500883. [DOI] [PubMed] [Google Scholar]
- Pang RTK, Poon TCW. Serum proteomic fingerprints of adult patients with severe acute respiratory syndrome. Clin Chem. 2006;52(3):421–429. doi: 10.1373/clinchem.2005.061689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquali M, Serchi T, Planchon S, Renaut J. 2D-DIGE in proteomics. Methods Mol Biol. 2017;1654:245–254. doi: 10.1007/978-1-4939-7231-9_17. [DOI] [PubMed] [Google Scholar]
- Petricoin EF, Ardekani AM. Use of proteomic patterns in serum to identify ovarian cancer. The Lancet. 2002;359(9306):572–577. doi: 10.1016/S0140-6736(02)07746-2. [DOI] [PubMed] [Google Scholar]
- Phizicky E, Bastiaens PIH, Zhu H, Snyder M, Fields S. Protein analysis on a proteomic scale. Nature. 2003;422(6928):208–213. doi: 10.1038/nature01512. [DOI] [PubMed] [Google Scholar]
- Qiu M, et al. Antibody responses to individual proteins of SARS coronavirus and their neutralization activities. Microbes Infect. 2005;7(5–6):882–889. doi: 10.1016/j.micinf.2005.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renuse S et al (2020) Development of mass spectrometry-based targeted assay for direct detection of novel SARS-CoV-2 coronavirus from clinical specimens. medRxiv
- Reusken C, et al. Specific serology for emerging human coronaviruses by protein microarray. Eur Surveill Eur Commun Dis Bull. 2013;18(14):20441. doi: 10.2807/1560-7917.es2013.18.14.20441. [DOI] [PubMed] [Google Scholar]
- Rosenberger G, Liu Y, Röst H, Ludwig C, Buil A, Bensimon A, Soste M, Spector T, Dermitzakis E, Collins B, Malmström L, Aebersold R. Inference and quantification of peptidoforms in large sample cohorts by SWATH-MS. Nat Biotechnol. 2017;35:781–788. doi: 10.1038/nbt.3908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherl A, Couté Y, Déon C, Callé A, Kindbeiter K, Sanchez J-C. Functional proteomic analysis of human nucleolus. Mol Biol Cell. 2002;13(11):4100–4109. doi: 10.1091/mbc.e02-05-0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidler J, Zinn N, Boehm ME, Lehmann WD. De novo sequencing of peptides by MS/MS. Proteomics. 2010;10(4):634–649. doi: 10.1002/pmic.200900459. [DOI] [PubMed] [Google Scholar]
- Seike M, Kondo T, Fujii K, Yamada T, Gemma A, Kudoh S. Proteomic signature of human cancer cells. Proteomics. 2004;4:2776–2778. doi: 10.1002/pmic.200300795. [DOI] [PubMed] [Google Scholar]
- Shen B, et al. Proteomic and metabolomic characterization of COVID-19 patient sera. Cell. 2020;182(1):59–72. doi: 10.1016/j.cell.2020.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh et al. (2020) A rapid and sensitive method to detect SARS-CoV-2 virus using targeted-mass spectrometry. J Proteins Proteomics 1–7 [DOI] [PMC free article] [PubMed]
- Smolka M, Zhou H, Aebersold R. Quantitative protein profiling using two-dimensional gel electrophoresis, isotope-coded affinity tag labeling, and mass spectrometry. Mol Cell Proteomics. 2001;1(1):19–29. doi: 10.1074/mcp.M100013-MCP200. [DOI] [PubMed] [Google Scholar]
- Srinivas PR, Verma M, Zhao Y, Srivastava S. Proteomics for cancer biomarker discovery. Clin Chem. 2002;48(8):1160–1169. [PubMed] [Google Scholar]
- Srivastava S, Ray S. COVID-19 pandemic: hopes from proteomics and multiomics research. OMICS. 2020;24:1–3. doi: 10.1089/omi.2019.0197. [DOI] [PubMed] [Google Scholar]
- Stukalov A, et al. Multi-level proteomics reveals host-perturbation strategies of SARS-CoV-2 and SARS-CoV. BioRxiv. 2020;193:3080. [Google Scholar]
- Tahmtan A, Ardebili A. Real-time RT-PCR in COVID-19 detection: issues affecting the results. Exp Rev Mol Diagnos. 2020;20(5):453–454. doi: 10.1080/14737159.2020.1757437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tonge R, Shaw J, Middleton B, Rowlinson R, Rayner S, Young J. Validation and development of fluorescence two-dimensional differential gel electrophoresis proteomics technology. Proteomics. 2001;1:377–396. doi: 10.1002/1615-9861(200103)1:3<377::AID-PROT377>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Verrills NM. Clinical proteomics: present and future prospects. Clin Biochem Rev. 2006;27(2):99–116. [PMC free article] [PubMed] [Google Scholar]
- Walls AC, Park Y-J, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;180:281–292. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Hou X, Wu X (2020) SARS-CoV-2 proteome microarray for mapping COVID-19 antibody interactions at amino acid resolution. BioRxiv [DOI] [PMC free article] [PubMed]
- Wiese S, Reidegeld KA, Meyer HE, Bettina W. Protein labeling by iTRAQ: a new tool for quantitative mass spectrometry in proteome research. Proteomics. 2007;7(3):340–350. doi: 10.1002/pmic.200600422. [DOI] [PubMed] [Google Scholar]
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323:1239. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- Wu D, et al. plasma metabolomic and lipidomic alterations associated with COVID-19. Nat Sci Rev. 2007;7(7):1157–1168. doi: 10.1093/nsr/nwaa086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu D, Wu T, Liu Q, Yang Z. The SARS-CoV-2 outbreak: what we know. Intern J Infect Dis. 2020;94:44–48. doi: 10.1016/j.ijid.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang D, Leibowitz JL. The structure and functions of coronavirus genomic 3′ and 5′ ends. Elsevier Public Health Emerg Collect. 2015;206:120–133. doi: 10.1016/j.virusres.2015.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye X, Luke B, Andresson T, Blonder J. 18O stable isotope labeling in MS-based proteomics. Brief Functi Genomics. 2009;8(2):136–144. doi: 10.1093/bfgp/eln055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi R, et al. The use of proteomics in the discovery of serum biomarkers from patients with severe acute respiratory syndrome. Proteomics. 2004;4(11):3477–3484. doi: 10.1002/pmic.200400897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Y, Clippinger AJ, Pierciey FJ, Alwine JC. Viruses and metabolism: alterations of glucose and glutamine metabolism mediated by human cytomegalovirus. Adv Virus Res. 2011;80:49–67. doi: 10.1016/B978-0-12-385987-7.00003-8. [DOI] [PubMed] [Google Scholar]
- Yuan M, Wu NC, Zhu X, Lee C-CD, So RTY. A highly conserved cryptic epitope in the receptor binding domains of SARS-CoV-2 and SARS-CoV. Science. 2020;368(6491):630–633. doi: 10.1126/science.abb7269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P, Yang X-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu H, Hu S, Jona G, Zhu X, Kreiswirth N, Willey BM, Mazzulli T, Liu G, Song Q, Chen P, Cameron M, Tyler A, Wang J, Wen J, Chen W, Compton S, Snyder M. Severe acute respiratory syndrome diagnostics using a coronavirus protein microarray. Proc Nat Acad Sci USA. 2006;103(11):4011–4016. doi: 10.1073/pnas.0510921103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu N, Zhang D, Wang W, Li X, Bo Y, Song J. A novel coronavirus from patients with pneumonia in China, 2019. N Eng J Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]