Abstract
The endocannabinoid system (ECS) has received extensive attention for its neuroprotective effect on the brain. This system comprises endocannabinoids, endocannabinoid receptors, and the corresponding ligands and proteins. The molecular players involved in their regulation and metabolism are potential therapeutic targets for neuropsychiatric diseases including anxiety, depression and neurodegenerative diseases such as Alzheimer’s disease (AD) and Parkinson’s disease (PD). The inhibitors of two endocannabinoid hydrolases, i.e., fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL), have the capacity to increase the level of endocannabinoids indirectly, causing fewer side effects than those associated with direct supplementation of cannabinoids. Their antidepressant and anxiolytic mechanisms are considered to modulate the hypothalamic-pituitary-adrenal axis and regulate synaptic and neural plasticity. In terms of AD/PD, treatment with FAAH/MAGL inhibitors leads to reduction in amyloid β-protein deposition and inhibition of the death of dopamine neurons, which are commonly accepted to underlie the pathogenesis of AD and PD, respectively. Inflammation as the cause of depression/anxiety and PD/AD is also the target of FAAH/MAGL inhibitors. In this review, we summarize the application and involvement of FAAH/MAGL inhibitors in related neurological diseases. Focus on the latest research progress using FAAH/MAGL inhibitors is expected to facilitate the development of novel approaches with therapeutic potential.
Keywords: Alzheimer's disease, Parkinson's disease, FAAH, MAGL, depression, anxiety
Introduction
Cannabis has been used in medicine for thousands of years, and its main active ingredients are cannabinoids. Owing to the interactions between these active chemicals and the body’s homeostasis system, cannabinoids were initially widely used for anticonvulsive, anti-inflammatory, and analgesic therapies, especially to alter neuronal function in the brain [1]. Advances in medicine led to the discovery of the endocannabinoid system (ECS), and many diseases that involve this system are receiving increasing attention [2]. Initially discovered as a complex network of cannabinoid receptors, the ECS is widely distributed in the central and peripheral nervous systems. It is involved in the regulation of several functions, including emotion and motivational behavior. It has also been shown to play a role in the pathophysiology of several mental disorders [3]. Its potential role in the etiology of mental disorders merits further study. Scientists are exploring this new field via the development of new drugs and treatment methods. Therefore, the three currently recognized types of cannabinoids, plant cannabinoids, synthetic cannabinoids and endocannabinoids, as well three components of the ECS, endocannabinoids, endocannabinoid receptors, and their corresponding ligands and proteins, have been used as the main research subjects, and extensive mechanistic and pharmacodynamics studies have been carried out. The structural and functional analogs that interact with the ECS and play a role in the treatment of central nervous system (CNS) disorders are regarded as potential new CNS drugs. Because the action of hydrolase on endocannabinoids increases endocannabinoid levels indirectly and thus causes fewer side effects than direct exogenous supplementation, two types of hydrolases, fatty acids amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL), have been examined as potential new drug targets for CNS disorders. Most neurodegenerative diseases are characterized by impaired memory and cognitive and motor function [4]. The pathogenesis of emotional disorders such as depression is controversial and has multiple explanations. For example, the neuroendocrine hypothesis involving the dysfunction of the hypothalamic–pituitary–adrenal axis (HPA) caused by stimuli such as stress is widely accepted [5]. Endocannabinoid pathways are widely believed to be involved in neurodegenerative diseases and neuropsychiatric diseases [6–8]. The development of FAAH and MAGL hydrolytic enzyme inhibitors and their extensive applications in neurological diseases have been a focus of research. The review explains the endocannabinoid system and eventually discusses two key types of enzyme inhibitors of the ECS: FAAH inhibitors and MAGL inhibitors. The current use of these inhibitors and relevant research with regard to nervous system diseases are summarized; based on the findings, these inhibitors exhibit potential as therapeutic agents in preclinical models of CNS diseases. The discussion on neurological diseases mainly focuses on depression/anxiety and Alzheimer’s disease (AD)/Parkinson’s disease (PD).
The endocannabinoid system
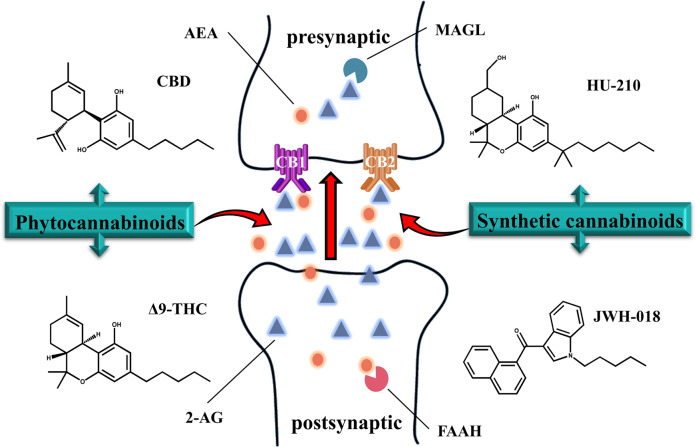
The ECS consists of the following three components: cannabinoid receptor proteins, corresponding ligands (endocannabinoids), and proteins involved in endocannabinoid regulation and metabolism. The ECS has been and is currently being studied extensively. Due to the presence of cannabinoid binding sites throughout the central and peripheral nervous systems and the potent effects of the ECS on neurotransmission, these neuroendocrine interactions are receiving extensive attention and have been elucidated in several studies. Endocannabinoid signaling induces the inhibition of HPA activity. The experimental finding that the exposure of rats to 30 min of restraint stress leads to increased activity of FAAH in rats suggests that endocannabinoid signaling within the amygdala could be an important determinant of neuroendocrine system function. Moreover, stress decreases the endocannabinoid and cannabinoid receptor type 1 receptor (CB1) ligand concentrations and disinhibits excitatory transmission in the basolateral amygdala [9]. It should be emphasized that the excitation, inhibition, or initiation of second messenger cascades by endogenous cannabinoids is mediated by retrograde signaling based on the interaction between the transmitter and receptor. This implies that the local excitation of a postsynaptic neuron, which is prompted by the release of anandamide (AEA) and 2-arachidonoyl-glycerol (2-AG) into the extracellular space and followed by binding with receptors, is localized to the presynaptic membrane [10] (Fig. 1).
Fig. 1.
Schematic diagram of the endocannabinoid system. Endocannabinoid AEA and 2-AG are released after postsynaptic synthesis to retroactively bind to endocannabinoid receptors (CB1 and CB2) at the presynaptic site. Exogenous cannabinoids such as phytocannabinoids (CBD and Δ9-THC) and synthetic cannabinoids (HU-210 and JWH-018) can also be added to the synapse to activate endocannabinoid receptors. FAAH and MAGL exist simultaneously in presynaptic and postsynaptic regions and hydrolyze endogenous cannabinoids. AEA anandamide, 2-AG 2-arachidonoyl-glycerol, FAAH fatty acid amide hydrolase, MAGL monoacylglycerol lipase, CBD cannabidiol, Δ9-THC Δ9-tetrahydrocannabinol
Endocannabinoids: AEA and 2-AG
Endocannabinoids are lipid signaling molecules that potently act as cannabinoid receptors. Two representative endocannabinoids that are widely distributed throughout the central nervous system, N-arachidonoyl-ethanolamine, called anandamide (AEA), and 2-arachidonoyl-glycerol (2-AG), were identified in the pig brain and canine gut [11], respectively. The kidneys are known to be enriched in AEA and enzymes that metabolize AEA, which are associated with many different physiological and pathophysiological functions, including the regulation of sensory and autonomic nerve signals, the regulation of energy expenditure and balance, and the initiation and control of inflammation [12]. The detection of 2-AG in the CNS has garnered extensive attention from scholars. Importantly, the levels of 2-AG are 170 times higher than those of AEA in the brain [13]. The opposing effects of 2-AG and AEA indicate that an increase in the AEA level is related to the improvement of decision-making ability and cognitive flexibility, while an increase in the 2-AG level is related to the destruction of cognitive flexibility and inhibitory response ability [14]. The difference in their effects is attributed to the fact that they are derived from different biosynthetic pathways. The two biosynthetic pathways of 2-AG are the signaling pathway that begins with phosphatidylinositol-4,5-diphosphate and the metabolic pathway that involves triglycerides containing sn2-arachidonic esters [13]. On the other hand, the sequential action of a Ca2+-dependent or Ca2+-independent N-acyltransferase followed by N-acyl-phosphatidylethanolamine-specific phospholipase D appears to be the most relevant biosynthetic pathway of AEA [15]. In addition, AEA and 2-AG exert their biological effects by binding to receptors. AEA and 2-AG molecules that are not involved in the mentioned pathways are degraded by two major hydrolases: FAAH and MAGL. Other metabolic processes, such as oxidation by cyclooxygenase-2 (COX-2), lipoxygenases, and cytochrome P450, have also been indicated [16].
In addition, phytocannabinoids and synthetic cannabinoids are structural or functional analogs of endocannabinoids. Even today, phytocannabinoids, encompassing approximately 120 compounds isolated from Cannabis sativa so far, are poorly defined or have unknown pharmacological profiles [17]. The predominant compound in Cannabis extracts is Δ9-tetrahydrocannabinol (Δ9-THC), which is paralleled by cannabidiol (CBD), the predominant component in natural Cannabis [18]. Both compounds have medical uses and are used as recreational drugs to produce effects on perception, mood, emotion, and cognition [19]. Synthetic cannabinoids (SCs) are a class of heterogeneous compounds developed to explore endogenous cannabinoid systems or for use as potential therapeutic agents [20]. Currently, the classification of SCs into three generations is widely accepted; chronologically, the “JWH group”, “CP group”, and “HU group” are the pioneers, with the second group containing alkyl derivatives, N-methyl piperidine, and benzoylindole. Molecules in which the carbonyl group or indole ring is replaced with other functional groups are considered third generation SCs [21]. Most SCs, as structural analogs of endogenous cannabinoids, can bind to receptors as agonists and elicit cannabimimetic effects similar to those of phytocannabinoids. Due to their greater binding affinity for CB1 and CB2 receptors than THC, SCs show more intense antioxidant, anti-inflammatory, and neuroprotective effects [22].
CB1/CB2 receptor
The discovery and identification of cannabinoid receptor type 1 (CB1) and CB2 were breakthroughs. Their endocannabinoid ligands were discovered in subsequent research, thereby generating new frontiers for correlational research on the ECS. Both CB1 and CB2 receptors are coupled to Gi/o proteins, and they negatively regulate adenylate cyclase and positively regulate mitogen-activated protein kinase [23]. Evidence exists that CB1 can also stimulate adenylyl cyclase via Gs, induce receptor-mediated Ca2+ fluxes and stimulate phospholipases in some experimental models [24]. The distribution of these receptors varies; CB1 receptors have been found to be ubiquitously distributed in the CNS, being highly expressed in the hippocampus, amygdala, prefrontal cortex, hypothalamus, and basal ganglia and expressed at lower level in peripheral neural and other tissues that participate in the regulation of emotions, stress, and responsiveness. Consequently, some psychiatric issues, as well as distress and dysfunction, may be related to the regulation of CB1 activity. For instance, CB1 receptor agonists may cause psychotic episodes and panic reactions, while antagonism may result in symptoms indicative of depression and anxiety-related disorders [25]. In contrast, the CB2 receptor is mainly expressed in peripheral tissues and is mainly expressed in the immune system. Recent studies have also revealed the presence of CB2 receptors in the CNS. The generally accepted and established consensus was that brain CB2 receptors are expressed predominantly in activated microglia, astrocytes, and their subpopulations, with the inducible nature of the CB2 receptor in microglia during neuroinflammation being uncovered later [26]. CB2 receptors also exist on ventral tegmental area dopaminergic neurons, modulating dopaminergic neuronal function and DA-regulated behavior [27]. In addition, there are other endocannabinoid target receptors, such as the transient receptor potential vanilloid type-1 channel (TRPV1), which might be related to the axonal transport and excitability of retinal ganglion cells, release of cytokines from microglia, regulation of retinal blood vessels [28], and TRPV1-like receptors, which can inhibit the release of the excitatory neurotransmitter glutamate in brain areas such as the hippocampus [23]. GRP55, which is expressed in regions within the brain, can also interact with cannabinoids, and it has been reported that CB1 may regulate signaling pathways mediated by GRP55 [29, 30]. In terms of the affinity of endocannabinoids for receptors, 2-AG is a complete agonist of CB1 and CB2 receptors, while AEA has a lower affinity for CB2 receptors than for CB1 receptors [31, 32].
FAAH/MAGL enzymes and their inhibitors
FAAH and MAGL are the principal catabolic enzymes for a class of bioactive lipids called fatty acid amides and are the key enzymes for the hydrolysis of endogenous cannabinoids. Based on the hydrolytic mechanism of FAAH and MAGL, the study of endocannabinoids and their receptor system, as well as their potential therapeutic applications in several nervous system disorders, cancers, and neuroinflammatory diseases, a large number of irreversible/reversible inhibitors have been used to explore the different selectivities of these two enzymes. The reported structures of most reversible FAAH inhibitors contains α-keto heterocycles, oleoyls-like trifluoromethyl ketones, oxime carbamates, and selected β-lactams [33]. Piperidine and piperazine urea compounds are considered the most promising irreversible FAAH inhibitors owing to their high selectivity and potency [34]. Their related roles include reducing inflammation and regulating myocardial lipid metabolism [35]. For example, in an in vitro experiment, the knockdown of FAAH suppressed prostaglandin E2 production and proinflammatory gene expression. Treatment with FAAH inhibitors also had anti-inflammatory effects [36]. However, it has been revealed that 85% of total hydrolyzation of 2-AG in the brain is mediated via another major inhibitor, MAGL [37], and the bulk of the development of MAGL inhibitors occurred in the early 2010s. In comparison, the hydrolase activity of AEA in brain issues can be ascribed to FAAH. MAGL inhibitors constitute three classes of chemical compounds: noncompetitive inhibitors, partially reversible inhibitors, serine-reactive agents, and cysteine-reactive agents [38]. Their functions, such as the regulation of lipid metabolism and anti-inflammation, have been reported in several animal experiments. For instance, they have protective effects in lung ischemia-reperfusion injury [39] and chronic liver injury [40]. In addition, dual FAAH/MAGL inhibition has been the focus of a small proportion of the research, as the individual inhibition of either FAAH or MAGL enzymes is unable to induce a full spectrum of activities [41]. Furthermore, it is thought that some of the pharmacological effects of dual FAAH/MAGL inhibitors are stronger than the complete inhibition of either FAAH or MAGL [42, 43]. For now, the pharmacological effects of FAAH inhibitors are being observed and tolerance tests for their clinical usage are being performed in healthy volunteers [44, 45]. However, the safety limitations of the usage of FAAH/MAGL inhibitors in clinical trials should not be overlooked. For example, the FAAH inhibitor BIA 10-2474 leads to acute and rapidly progressive neurological symptoms, such as headache and altered consciousness. One patient even developed brain death [46]. The underlying mechanism is still unknown. An improper excessive dose was regarded as the chief culprit [47]. In addition, gender differences in the effects of FAAH and MAGL inhibitors in clinical trials need to be further studied, with a focus on females, especially women of reproductive age [48].
Neuropsychiatric disorders regulated by the endocannabinoid system
Depression and anxiety
Depression is a mental illness of great concern, with core symptoms including but not limited to, low mood, anhedonia, and irregular diet and sleep patterns. Clinically, patients with depression often have anxiety-like symptoms, implying that the comorbidity of these two mental diseases is a common phenomenon. Serotonergic and noradrenergic functional maladjustment [49] and HPA axis alterations often result in depression and anxiety. Several clinical trials and animal experiments have demonstrated that the endocannabinoid content in the tissues and serum of patients with depression show marked variation compared with those in healthy individuals. CB1 receptor expression and 2-AG levels have been found to be significantly reduced in the hippocampus following chronic unpredictable stress [50], which is thought to mimic the behavioral and endocrinal changes that promote the development of human clinical depression [51]. An interesting study of alterations in the ECS revealed that the serum level of 2-AG is significantly decreased in patients with major depression, while in patients with mild depression, serum AEA and 2-AG content show an upward trend [52]; this implies that the ECS could be interpreted as a buffer and regulator. There is a strong negative correlation between the serum AEA level and anxiety symptoms in major depressive disorder patients. These pieces of evidence support an interaction between ECS and depression/anxiety.
Several CB1/CB2 receptor agonists and antagonists have been used to explore the possible mechanisms of ECS in depression and anxiety. The antidepressant and anxiolytic effects of these compounds in ameliorating the disorder of the HPA axis and reversing the decline in monoamine neurotransmitter levels have been demonstrated in animal models. For instance, the administration of a CB1 receptor antagonist/inverse agonist increases the concentration of serum corticosterone in a dose-dependent manner, and pretreatment with receptor agonists, transport inhibitors, or FAAH inhibitors significantly reduces or eliminates the inhibition of corticosterone-induced release [53]. CB1 receptor antagonists can block stress-induced corticosterone oversecretion by inhibiting FAAH activity within the basolateral amygdala complex [9]. The amygdala is believed to be a key component of the excitatory drive of the HPA axis [54]. This provides evidence that the roles of the HPA axis and EC system in the regulation of physiological processes in depression highly overlap. Furthermore, CB1/CB2 receptors are stimulated to increase the activity of adrenergic, serotonergic, and dopaminergic neurons, as well as the synthesis and/or release of corresponding neurotransmitters in specific brain regions. Some specific studies are discussed below. Low doses of WIN55,212-2, a selective CB1 receptor agonist, enhance dorsal raphe nucleus 5-HT neuronal activity through a CB1R-dependent mechanism [55]. Numerous studies have focused on the role of CB2Rs in pathological pain of immune origin [56] because CB2 receptors are thought to play a role in the peripheral immune system. The mode of action of CB2 receptor agonists and agonists in reversing monoamine neurotransmitter levels has been poorly characterized. Based on the fact that CB2 receptors are expressed in dopaminergic neurons in the mouse brain, Liu et al. used dopaminergic neuron-specific CB2 receptor conditional knockout mice to confirm that CB2 may play important roles in the modulation of psychomotor behaviors [57]; the mechanism may be related to decreased activity of CB2-mediated dopaminergic neurons in the ventral tegmental area [27]. β-caryophyllene, a naturally available sesquiterpene, has also been reported to have effects mediated by CB2 receptors on depression and anxiety [58]. The mechanism can be ascribed to an increase in the uptake levels or function of glutamate as a subchronic immobilization and acoustic stress-induced inflammatory regulator. The latter hypothesis was also confirmed by experiments by Zoppi et al. in which CB2 receptor agonists inhibited the increase in stress-induced proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), nitric oxide (NO), and COX-2 and regulated depression caused by neuroinflammation. However, CB2 and the glucocorticoid mechanism regulated by the HPA axis were not found to be relevant in this study [26]. Furthermore, one study found that exogenous cannabinoids taken up can interact with CB2 receptors and promote neurogenesis in the hippocampal dentate gyrus in adult rats, which translates to anxiolytic and antidepressant effects [59].
Alzheimer’s disease (AD) and Parkinson’s disease (PD)
AD and PD are neurodegenerative diseases that share similar molecular mechanisms and pathological processes: protein mutation leading to erroneous sorting or misfolding, protein aggregation due to blocked degradation, and inclusion body formation at specific sites. A mutation in the amyloid precursor protein gene that causes the abnormal accumulation of amyloid-β protein (Aβ) in the brain [60] and phosphorylated Tau protein [61] is the focus of attention for AD. It has been observed that CB2 receptors and FAAH are selectively overexpressed in neuritic plaque-associated glia in Alzheimer’s disease brains [62], especially in reactive astrocytes and activated microglial cells [63]. A CB2 agonist demonstrated the ability to stimulate natural Aβ removal in frozen human tissue sections and inhibit the synthesis of pathogenic peptides [64]. The expression patterns of the CB2 receptor and FAAH are closely related to the deposition of Aβ, suggesting that CB2 and FAAH may play a regulatory role in the function of microglial cells in the pathological changes associated with AD. The expression of CB1 receptors in different brain regions of AD patients remains controversial; however, research has indicated that CB1 receptor activity is contingent on the clinical period of AD, with higher CB1 receptor activity found in the early stage of AD and decreased levels found in later stages [65]. Additionally, Tau hyperphosphorylation may be affected by CB1 agonism [66] and cannabidiol [67], with the former downregulating the expression of inducible nitric oxide synthase and the production of NO in Aβ-stimulated C6 cells and the latter exerting its effect through Wnt/β-catenin pathway rescue in Aβ-stimulated PC12 neuronal cells.
PD, also known as tremor paralysis, is a slowly progressing neurodegenerative disease of largely unknown etiology. The main biochemical pathology is the significant degeneration and loss of dopaminergic neurons in the substantia nigra and a significant decrease in the dopamine concentration in the striatum accompanied by dysfunction of the basal ganglia [68], a region that is a crucial regulator of motor activity affected by PD. CB1 receptors are highly distributed in the basal ganglia [69] and exert complex regulatory effects on some important neurotransmitters, playing a role in anti-excitatory neural toxicity [70] and neuroprotection. Clinical observations have reported that the degeneration of dopaminergic neurons is accompanied by increased endocannabinoid system activity, and endocannabinoid system dysfunction is observed in both PD patients and experimental animal models. The upregulation of the CB2 receptor in glial cells in postmortem tissues obtained from PD patients has also been investigated [71]. Mounsey et al utilized the stereotaxic injection of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), a neurotoxin, to establish a PD rat model by stereotactic injection and observed an elevated level of 2-AG [72]. In the same model, CB1 agonists improved the survival of nigrostriatal dopaminergic neurons [73], and CB2 receptor activation may have blocked blood-brain barrier leakage and the neuroinflammation caused by the activation of glial cells, leading to decreased production of proinflammatory chemokines and thus preventing damage to dopaminergic neurons [74].
FAAH/MAGL inhibitor application as a new strategy for future treatment of CNS disorders
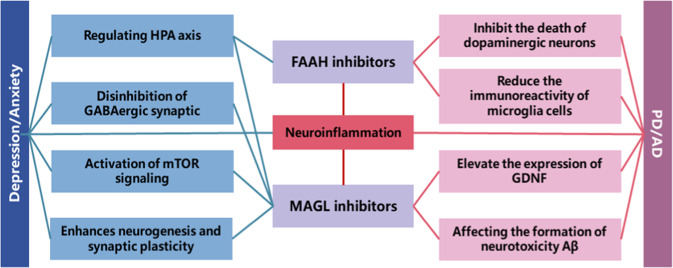
The use of hydrolase inhibitors has been mentioned as a new drug strategy with a strong potential for treating CNS disorders. The direct stimulation of a receptor by an endocannabinoid agonist or antagonist directly increases the content of endocannabinoids; this makes the action of the agonist or antagonist on the receptor less robust than indirect stimulation and causes this strategy to be prone to side effects. Therefore, inhibitors of FAAH and MAGL indirectly increase the excitability of the ECS by reducing the hydrolysis of endocannabinoids. Several FAAH and MAGL inhibitors have been assessed in preclinical studies (Fig. 2 and Table 1).
Fig. 2.
The application of FAAH and MAGL inhibitors in depression/anxiety and Parkinson’s disease/Alzheimer’s disease and the related mechanism. Both endocannabinoid hydrolytic enzyme inhibitors improve the symptoms of depression/anxiety and PD/AD by regulating neuroinflammation. In addition, FAAH inhibitors can coordinate the function of the HPA axis, and MAGL inhibitors can play a role against depression and anxiety by blocking the inhibitory effect of astrocyte-mediated GABAergic synapses or glutamatergic synapses, activating the mTOR signaling pathway, and enhancing neurogenesis and synaptic plasticity. In PD/AD, FAAH and MAGL play a role by inhibiting the death of dopaminergic neurons, reducing the immunoreactivity of microglial cells, and increasing the expression of GDNF. This affects the formation of Aβ. PD Parkinson’s disease, AD Alzheimer’s disease, Aβ amyloid β-protein
Table 1.
Key biochemical indicators in CNS diseases after FAAH and MAGL inhibitors therapy in the animal
| Type of the inhibitors | Name | Biochemical indicators | References |
|---|---|---|---|
| FAAH inhibitors | N-arachidonoyl serotonin | CORT ↓ | [76] |
| URB597 | CORT↓ | [75, 81] | |
| COX-2, PGE2, NO ↓ | [91] | ||
| DOPA ↑ | [104] | ||
| URB694 | CORT ↓ | [79] | |
| ST4070 | CORT ↓ | [80] | |
| SSR411298 | CORT ↓ | [82] | |
| PF-3845 | BDNF ↑ | [106] | |
| MAGL inhibitors | JZL184 | CORT ↓ | [86] |
| IL-1β, TNF-α ↓ | [90] | ||
| Aβ ↓ | [102, 103] | ||
| KML29 | DOPA, GDNF ↑ | [106] |
PGE2 prostaglandin E2, NO nitric oxide, DOPA dopamine, BDNF brain-derived neurotrophic factor, GDNF glial cell line-derived neurotrophic factor, IL-1β interleukin-1β, TNF-α tumor necrosis factor-α, CORT corticosterone, COX-2 cyclooxygenase-2
Depression/anxiety
To produce antidepressant or anxiolytic effects, FAAH inhibitors may alter the response of the HPA axis and modulate its function, while MAGL inhibitors may be beneficial in suppressing CNS inflammation, thus ameliorating the depression or anxiety caused by different physiological mechanisms [75]. As a dual blocker of FAAH and TRPV1, N-arachidonoyl serotonin reverses the prolongation of immobility time in the forced swimming test through a mechanism associated with the normalization of the HPA axis after restraint stress by reversing the increase in plasma corticosterone levels induced by stress [76]. Previous studies suggest that N-arachidonoyl serotonin increases the levels of anandamide and facilitates its action on the CB1 receptor [77], resulting in an anxiolytic-like effect [78]. The above studies suggest that inhibitors of enzymes that hydrolyze endogenous cannabinoids may modulate monoaminergic signaling to play a role.
Similarly, some other FAAH inhibitors, such as URB597, URB694, and ST4070, also have the same influence on blood corticosterone levels in stress paradigms and further ameliorate the conditions of induced stress, such as depression or anxiety [9, 75, 79–81]. SSR411298, a newly synthesized and effective reversible FAAH inhibitor, has robust antidepressant-like activity in the forced swimming test and chronic mild stress model. In experimental anxiety models, anxiolytic-like effects are observed in high-intensity traumatic events [82]. In addition, an increase in time spent in a lit arena in the light/dark test [83] and a decrease in marble burying behavior [84] are observed following treatment with URB597 and PF-3845, respectively. The MAGL inhibitor JZL184 also shows similar results, revealing its anxiolytic effects. A recent study performed comparative profiling of FAAH, MAGL, and dual inhibitors, and the results demonstrated that acute selective FAAH or MAGL inhibitors prevent stress-induced anxiety-like behavior at doses that do not impair cognitive function but are associated with anxiety-like behavior [85].
The MAGL inhibitor JZL184 has been reported to increase corticosterone levels and is thought to participate in the regulation of circulating corticosterone [86]. Nevertheless, the antidepressant effect of MAGL inhibitors is significant, and several special mechanisms have been reported. For example, high or low doses of MAGL may have an antidepressant effect on depressive states associated with acute stress and chronic corticosterone-induced stress. The mechanism has been postulated to involve the disinhibition of the GABAergic synapses or glutamatergic synapses mediated by astrocytes [87], reversing the downregulation of mTOR and ERK signals induced by chronic unpredictable stress [88] and enhancing adult neurogenesis and long-term synaptic plasticity in the hippocampal dentate gyrus [89].
The endogenous cannabinoid 2-AG, which is dominant in vivo, is a complete agonist of the CB2 receptor and appears to be associated with inflammation. As 2-AG is mainly hydrolyzed by MAGL, MAGL inhibitors can contribute to the improvement of neuroinflammation-related depression symptoms. For example, as a MAGL inhibitor, JZL184 inhibits MAGL activity and elevates 2-AG levels in the spleen but not in the frontal cortex. This can be explained by the absence of detectable JZL184 levels in the frontal cortex as opposed to the spleen. However, JZL184 attenuates lipopolysaccharide (LPS)-induced interleukin-1β (IL-1β), interleukin-6 (IL-6), interleukin-10 (IL-10) and TNF alpha levels in the frontal cortex, suggesting the indirect actions of JZL184 on CNS neuroinflammation [90]. This study also provides further evidence for the potential of MAGL inhibitors as antidepressants targeting inflammation. In addition, a study reported that URB597 decreases the expression of COX-2 and inducible nitric oxide in LPS-stimulated microglia and inhibits the release of inflammatory factors such as prostaglandin E2 and NO, suggesting that its antidepressant effect may be related to the mediation of neuroinflammation [91]. In terms of rapid acting antidepressant effects, ketamine and JZL-184 both reverse evolving depressive-like behavior during forced abstinence from alcohol drinking. According to a review by Ogawa et al., FAAH and MAGL inhibitors may require chronic administration for weeks to show clear antidepressant-like effects [75].
Other neuropsychiatric disorders
The FAAH inhibitor AM3506 lowers the percentage of trials with a correct nonmatching response in the delayed nonmatching-to-position procedure [92], signifying a possible role in learning and memory function. However, reports to the contrary are also available, as the most commonly used inhibitor of FAAH, URB597, enhances memory acquisition when used in passive avoidance experiments in rats. This enhancement is impeded by a PPAR-alpha antagonist, supporting the ability of FAAH inhibitors to modulate memory by activating PPAR-alpha [93]. In a model of ethanol-related neuroinflammation and memory decline, the effect of the FAAH inhibitor on hippocampal-dependent memory may involve the regulation of hippocampal microglial cell recruitment and activation [94]. In addition, URB597 has a positive effect on cognitive function in male rats [95] and prevents epileptic-induced changes in short-term plasticity and long-term potentiation (LTP) [96].
Alterations in learning performance induced by the MAGL inhibitor SAR127303 in multiple tests are related to episodic, working and spatial memory, particularly a reduction in LTP of hippocampal CA1 synaptic transmission and acetylcholine release, two features of memory function [97]. In recent years, the potential effects of FAAH inhibitors in several other stress-related disorders, such as post-traumatic stress disorder (PTSD), have also been explored. Chronic treatment with URB597 prevents poststress symptoms (extinction and startle response), while acute delivery ameliorates depressive-like symptoms (anhedonia and reduced locomotion) in addition to PTSD-like symptoms (impaired fear extinction and enhanced fear retrieval) through the upregulation of CB1 induced by the return of shock/situational reminders to the normal level and by improving performance in hippocampal-dependent memory impairment and amygdala-dependent memory enhancement in PTSD [98–100]. Chronic traumatic encephalopathy, a disease that was recently regarded as a target for MAGL, has neuropathological features such as neurodegeneration, TAR DNA-binding protein 43 protein aggregation, and tau phosphorylation, all of which may be significantly inhibited [101].
PD/AD
However, no reports specifically focused on the use of MAGL inhibitors for depression related to neuroinflammation are available, although the capacity of MAGL to reduce the neuroinflammatory response has been implicated in several other related nervous system diseases. For instance, Rea et al. demonstrated that JZL184 treatment significantly reduces the levels of inflammation-induced Iba1-immunoreactive microglia and total Aβ burden in the hippocampus and temporal and parietal cortices in APPSwe/PSEN1ΔE9 mice, a model of Alzheimer’s disease. Thus, neuroinflammatory responses produced by hyperactive glial cells can be alleviated, dramatically affecting the course of AD, including the formation of neurotoxic Aβ [102, 103]. In PD, some scholars found that URB597 can inhibit the death of dopaminergic neurons, reduce the immunoreactivity of microglial cells, and improve the protective effect of MPTP-induced motor changes, thereby improving the symptoms of PD [104]. Johnston et al. reported that when L-DOPA and URB597 were coadministered to determine if URB597 hinders the effects of L-DOPA therapy on PD, URB597 reduced total L-DOPA-induced activity and the magnitude of hyperactivity by 32% and 52%. The lack of effects of URB597 on the antiparkinsonian actions of L-DOPA shows that FAAH inhibitors may play an effective role in treating the side effects of L-DOPA without impeding its therapeutic benefits [105]. Comparative profiling of FAAH and MAGL inhibitors has also been performed. In this study, scientists selected KML29 and PF-3845 as MAGL and FAAH inhibitors with the purpose of investigating the differential influence of dopamine and BDNF in a PD rat model. The final result demonstrated that treatment with KML29 for over five weeks attenuated striatal dopamine depletion in MPTP/probenecid mice, preserved MPTP-induced CB2 expression and elevated the expression of glial-derived neurotrophic factor. While PF-3845 did not show some degree of protection, it reduced CB2 expression. Thus, MAGL inhibition is a better choice for PD treatment [106].
Conclusions and perspectives
We comprehensively summarized cannabinoid compounds, the ECS, and their applications in diseases of the nervous system. We also reviewed reports on the two hydrolysis enzymes (FAAH and MAGL) and their effect on nervous system diseases and depression/anxiety, with a special focus on PD/AD. Although the application of FAAH/MAGL inhibitors in learning and memory, PTSD and other processes has been assessed, the literature survey shows that limited studies on these aspects have been conducted, and further progress should be made. Since MAGL is still widely accepted to be associated with peripheral inflammation, only limited studies have explored the therapeutic potential of MAGL inhibitors for the treatment of neurological diseases. For example, in depression, despite the availability of reports on the mechanism of MAGL inhibitors, the lack of concrete evidence in animal behavioral experiments remains a challenge to be overcome. Currently, the two inhibitors are obtained by chemical synthesis or structural modification, and we suggest that the extraction of active compounds from natural products should be considered as a good source of the inhibitors; additionally a more general in vitro screening model should be established because active substances produced by the metabolism of plants and other organisms can be used for defense or other physiological functions. The chemical structure of many natural products is so complex that it is difficult or impossible to obtain drug candidates by synthetic methods; most natural products have naturally generated chirals, whereas the chiral synthesis of most compounds is more medicinal. The above characteristics indicate that natural products are the original biological “design” and that biological macromolecules/drug targets (enzymes, proteins, etc.) have a natural affinity. Although the efficacy of existing inhibitors has been summarized, the inhibitors to evaluate efficacy at the experiment level in animals, such as URB597 and JZL184, are similar. Hence, the actual effects of other inhibitors need to be further verified in animals. Additionally, comparative studies of the interactions and relationships between these inhibitors and current antidepressants are limited. N-arachidonoyl serotonin has an antidepressive effect as a FAAH inhibitor, and we expect drug combinations involving currently available drugs that increase monoamine release to treat depression and FAAH/MAGL inhibitors to be more effective. From the overall perspective of the endogenous cannabinoid system, the role of receptors besides CB1/CB2 receptors, such as TRPV1, remains poorly understood. Since AEA and 2-AG are two endocannabinoids without any apparent specialty, we believe that compounds other than the enzymes FAAH and MAGL may modulate the metabolism of endocannabinoids in animals, and this should be further explored. For numerous proteins, extracting the substance that conforms to the characteristics of the enzyme can improve the limitations of the current studies conducted on the ECS, and this will serve as an interesting and challenging task for researchers.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81573636, 81773924), Beijing Natural Science Foundation (7182114), CAMS Innovation Fund for Medical Sciences (CIFMS) (2016-I2M-1-004), the Fundamental Research Funds for the Central Universities (2018RC350010), and Project of NDRC and State Administration of Traditional Chinese Medicine (ZYBZH-Y-HUN-24).
Competing interests
The authors declare no competing interests.
References
- 1.Cheer JF, Hurd YL. A new dawn in cannabinoid neurobiology: the road from molecules to therapeutic discoveries. Neuropharmacology. 2017;124:1–2. doi: 10.1016/j.neuropharm.2017.07.004. [DOI] [PubMed] [Google Scholar]
- 2.Katzman MA, Furtado M, Anand L. Targeting the endocannabinoid system in psychiatric Illness. J Clin Psychopharmacol. 2016;36:691–703. doi: 10.1097/JCP.0000000000000581. [DOI] [PubMed] [Google Scholar]
- 3.Marsicano G, Wotjak CT, Azad SC, Bisogno T, Rammes G, Cascio MG, et al. The endogenous cannabinoid system controls extinction of aversive memories. Nature. 2002;418:530–4. doi: 10.1038/nature00839. [DOI] [PubMed] [Google Scholar]
- 4.Basavarajappa BS, Shivakumar M, Joshi V, Subbanna S. Endocannabinoid system in neurodegenerative disorders. J Neurochem. 2017;142:624–48. doi: 10.1111/jnc.14098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Keller J, Gomez R, Williams G, Lembke A, Lazzeroni L, Murphy GM, et al. HPA axis in major depression: cortisol, clinical symptomatology and genetic variation predict cognition. Mol Psychiatry. 2017;22:527–36. doi: 10.1038/mp.2016.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yin AQ, Wang F, Zhang X. Integrating endocannabinoid signaling in the regulation of anxiety and depression. Acta Pharmacol Sin. 2019;40:336–41. doi: 10.1038/s41401-018-0051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Covey DP, Mateo Y, Sulzer D, Cheer JF, Lovinger DM. Endocannabinoid modulation of dopamine neurotransmission. Neuropharmacology. 2017;124:52–61. doi: 10.1016/j.neuropharm.2017.04.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hillard CJ, Beatka M, Sarvaideo J. Endocannabinoid signaling and the hypothalamic-pituitary-adrenal axis. Compr Physiol. 2016;7:1–15. doi: 10.1002/cphy.c160005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hill MN, McLaughlin RJ, Morrish AC, Viau V, Floresco SB, Hillard CJ, et al. Suppression of amygdalar endocannabinoid signaling by stress contributes to activation of the hypothalamic-pituitary-adrenal axis. Neuropsychopharmacology. 2009;34:2733–45. doi: 10.1038/npp.2009.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vinod KY, Hungund BL. Role of the endocannabinoid system in depression and suicide. Trends Pharmacol Sci. 2006;27:539–45. doi: 10.1016/j.tips.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 11.Iannotti FA, Di Marzo V, Petrosino S. Endocannabinoids and endocannabinoid-related mediators: targets, metabolism and role in neurological disorders. Prog Lipid Res. 2016;62:107–28. doi: 10.1016/j.plipres.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 12.Ritter JK, Li G, Xia M, Boini K. Anandamide and its metabolites: what are their roles in the kidney. Front Biosci. 2016;8:264–77. doi: 10.2741/s461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Baggelaar MP, Maccarrone M, van der Stelt M. 2-Arachidonoylglycerol: a signaling lipid with manifold actions in the brain. Prog Lipid Res. 2018;71:1–17. doi: 10.1016/j.plipres.2018.05.002. [DOI] [PubMed] [Google Scholar]
- 14.Fagundo AB, de la Torre R, Jiménez-Murcia S, Agüera Z, Pastor A, Casanueva FF, et al. Modulation of the endocannabinoids N-Achidonoylethanolamine (AEA) and 2-Arachidonoylglycerol (2-AG) on executive functions in humans. PLoS One. 2013;8:e66387. doi: 10.1371/journal.pone.0066387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maccarrone M. Metabolism of the endocannabinoid anandamide: open questions after 25 years. Front Mol Neurosci. 2017;10:166. doi: 10.3389/fnmol.2017.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Biernacki M, Skrzydlewska E. Metabolism of endocannabinoids. Postepy Hig Med Dosw. 2016;70:830–43. doi: 10.5604/17322693.1213898. [DOI] [PubMed] [Google Scholar]
- 17.Turner SE, Williams CM, Iversen L, Whalley BJ. Molecular pharmacology of phytocannabinoids. Prog Chem Org Nat Prod. 2017;103:61–101. doi: 10.1007/978-3-319-45541-9_3. [DOI] [PubMed] [Google Scholar]
- 18.Potter DJ, Hammond K, Tuffnell S, Walker C, Di Forti M. Potency of Δ9-tetrahydrocannabinol and other cannabinoids in cannabis in England in 2016: implications for public health and pharmacology. Drug Test Anal. 2018;10:628–35. doi: 10.1002/dta.2368. [DOI] [PubMed] [Google Scholar]
- 19.Pertwee RG, Cascio MG. Known pharmacological actions of delta-9-tetrahydrocannabinol and of four other chemical constituents of cannabis that activate cannabinoid receptors. Handb Cannabis. Oxford, UK: Oxford University Press; 2014.
- 20.Schafroth MA, Carreira EM. Synthesis of phytocannabinoids. Prog Chem Org Nat Prod. 2017;103:37–59. doi: 10.1007/978-3-319-45541-9_2. [DOI] [PubMed] [Google Scholar]
- 21.Pintori N, Loi B, Mereu M. Synthetic cannabinoids: the hidden side of spice drugs. Behav Pharmacol. 2017;28:409–19. doi: 10.1097/FBP.0000000000000323. [DOI] [PubMed] [Google Scholar]
- 22.Castaneto MS, Gorelick DA, Desrosiers NA, Hartman RL, Pirard S, Huestis MA. Synthetic cannabinoids: epidemiology, pharmacodynamics, and clinical implications. Drug Alcohol Depend. 2014;144:12–41. doi: 10.1016/j.drugalcdep.2014.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pertwee RG. The pharmacology of cannabinoid receptors and their ligands: an overview. Int J Obes. 2006;30:S13. doi: 10.1038/sj.ijo.0803272. [DOI] [PubMed] [Google Scholar]
- 24.Turu G, Hunyady L. Signal transduction of the CB1 cannabinoid receptor. J Mol Endocrinol. 2010;44:75–85. doi: 10.1677/JME-08-0190. [DOI] [PubMed] [Google Scholar]
- 25.Moreira FA, Grieb M, Lutz B. Central side-effects of therapies based on CB1 cannabinoid receptor agonists and antagonists: focus on anxiety and depression. Best Pract Res Clin Endocrinol Metab. 2009;23:133–44. doi: 10.1016/j.beem.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 26.Zoppi S, Madrigal JL, Caso JR, García-Gutiérrez MS, Manzanares J, Leza JC, et al. Regulatory role of the cannabinoid CB2 receptor in stress-induced neuroinflammation in mice. Br J Pharmacol. 2014;171:2814–26. doi: 10.1111/bph.12607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang HY, Gao M, Liu QR, Bi GH, Li X, Yang HJ, et al. Cannabinoid CB2 receptors modulate midbrain dopamine neuronal activity and dopamine-related behavior in mice. Proc Natl Acad Sci U S A. 2014;111:E5007–15. doi: 10.1073/pnas.1413210111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ryskamp DA, Redmon S, Jo AO, Križaj D. TRPV1 and endocannabinoids: emerging molecular signals that modulate mammalian vision. Cells. 2014;3:914–38. doi: 10.3390/cells3030914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kargl J, Balenga N, Parzmair GP, Brown AJ, Heinemann A, Waldhoer M. The cannabinoid receptor CB1 modulates the signaling properties of the lysophosphatidylinositol receptor GPR55. J Biol Chem. 2012;287:44234–48. doi: 10.1074/jbc.M112.364109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Martínez-Pinilla E, Aguinaga D, Navarro G, Rico AJ, Oyarzábal J, Sánchez-Arias JA, et al. Targeting CB1 and GPR55 endocannabinoid receptors as a potential neuroprotective approach for Parkinson’s disease. Mol Neurobiol. 2019;56:5900–10. doi: 10.1007/s12035-019-1495-4. [DOI] [PubMed] [Google Scholar]
- 31.Soni N, Prabhala BK, Mehta V, Mirza O, Kohlmeier KA. Anandamide and 2-AG are endogenously present within the laterodorsal tegmental nucleus: functional implications for a role of eCBs in arousal. Brain Res. 2017;1665:74–9. doi: 10.1016/j.brainres.2017.04.003. [DOI] [PubMed] [Google Scholar]
- 32.Sugiura T, Kondo S, Kishimoto S, et al. Evidence that 2-arachidonoylglycerol but not N-palmitoylethanolamine or anandamide is the physiological ligand for the cannabinoid CB2 receptor. Comparison of the agonistic activities of various cannabinoid receptor ligands in HL-60 cells. J Biol Chem. 2000;275:605–12. doi: 10.1074/jbc.275.1.605. [DOI] [PubMed] [Google Scholar]
- 33.Otrubova K, Ezzili C, Boger DL. The discovery and development of inhibitors of fatty acid amide hydrolase (FAAH) Bioorg Med Chem Lett. 2011;21:4674–85. doi: 10.1016/j.bmcl.2011.06.096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Korhonen J, Kuusisto A, van Bruchem J, Patel JZ, Laitinen T, Navia-Paldanius D, et al. Piperazine and piperidine carboxamides and carbamates as inhibitors of fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL) Bioorg Med Chem. 2014;22:6694–705. doi: 10.1016/j.bmc.2014.09.012. [DOI] [PubMed] [Google Scholar]
- 35.Harasim-Symbor E, Polak A, Pędzińska-Betiuk A, Weresa J, Malinowska B, Lewandowska A, et al. Fatty acid amide hydrolase inhibitor (URB597) as a regulator of myocardial lipid metabolism in spontaneously hypertensive rats. Chem Phys Lipids. 2019;218:141–8. doi: 10.1016/j.chemphyslip.2018.12.007. [DOI] [PubMed] [Google Scholar]
- 36.Tanaka M, Yagyu K, Sackett S, Zhang Y. Anti-inflammatory effects by pharmacological inhibition or knockdown of fatty acid amide hydrolase in BV2 microglial cells. Cells. 2019;8:E491. doi: 10.3390/cells8050491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Blankman JL, Simon GM, Cravatt BF. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chem Biol. 2007;14:1347–56. doi: 10.1016/j.chembiol.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bertrand T, Augé F, Houtmann J, Rak A, Vallée F, Mikol V, et al. Structural basis for human monoglyceride lipase inhibition. J Mol Biol. 2010;396:663–73. doi: 10.1016/j.jmb.2009.11.060. [DOI] [PubMed] [Google Scholar]
- 39.Xiong Y, Yao H, Cheng Y, Gong D, Liao X, Wang R. Effects of monoacylglycerol lipase inhibitor URB602 on lung ischemia-reperfusion injury in mice. Biochem Biophys Res Commun. 2018;506:578–84. doi: 10.1016/j.bbrc.2018.10.098. [DOI] [PubMed] [Google Scholar]
- 40.Habib A, Chokr D, Wan J, Hegde P, Mabire M, Siebert M, et al. Inhibition of monoacylglycerol lipase, an anti-inflammatory and antifibrogenic strategy in the liver. Gut. 2018;68:522–32. [DOI] [PubMed]
- 41.Long JZ, Nomura DK, Vann RE, Walentiny DM, Booker L, Jin X, et al. Dual blockade of FAAH and MAGL identifies behavioral processes regulated by endocannabinoid crosstalk in vivo. Natl Acad Sci U S A. 2009;106:20270–5. doi: 10.1073/pnas.0909411106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sakin YS, Dogrul A, Ilkaya F, Seyrek M, Ulas UH, Gulsen M, et al. The effect of FAAH, MAGL, and dual FAAH/MAGL inhibition on inflammatory and colorectal distension-induced visceral pain models in rodents. Neurogastroenterol Motil. 2015;27:936–44. doi: 10.1111/nmo.12563. [DOI] [PubMed] [Google Scholar]
- 43.Sakin YS, Tanoğlu A, Gülşen M. Dual FAAH and MAGL inhibition might play a key role in visceral pain. Turk J Gastroenterol. 2018;29:625–6. doi: 10.5152/tjg.2018.18401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Deplano A, Cipriano M, Moraca F, Novellino E, Catalanotti B, Fowler CJ, et al. Benzylamides and piperazinoarylamides of ibuprofen as fatty acid amide hydrolase inhibitors. J Enzym Inhib Med Chem. 2019;34:562–76. doi: 10.1080/14756366.2018.1532418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Postnov A, Schmidt ME, Pemberton DJ, de Hoon J, van Hecken A, van den Boer M, et al. Fatty acid amide hydrolase inhibition by JNJ-42165279: a multiple-ascending dose and a positron emission tomography study in healthy volunteers. Clin Transl Sci. 2018;11:397–404. doi: 10.1111/cts.12548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kerbrat A, Ferré JC, Fillatre P, Ronzière T, Vannier S, Carsin-Nicol B, et al. Acute neurologic disorder from an inhibitor of fatty acid amide hydrolase. N Engl J Med. 2016;375:1717–25. doi: 10.1056/NEJMoa1604221. [DOI] [PubMed] [Google Scholar]
- 47.Edan G, Kerbrat A. Inhibitor of fatty acid amide hydrolase—learning from tragic failures. N Engl J Med. 2017;376:393–4. doi: 10.1056/NEJMc1615417. [DOI] [PubMed] [Google Scholar]
- 48.D’Souza DC, Cortes-Briones J, Creatura G, Bluez G, Thurnauer H, Deaso E, et al. Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF-04457845) in the treatment of cannabis withdrawal and dependence in men: a double-blind, placebo-controlled, parallel group, phase 2a single-site randomised controlled trial. Lancet Psychiatry. 2019;6:35–45. doi: 10.1016/S2215-0366(18)30427-9. [DOI] [PubMed] [Google Scholar]
- 49.Ressler KJ, Nemeroff CB. Role of serotonergic and noradrenergic systems in the pathophysiology of depression and anxiety disorders. Depress Anxiety. 2000;12:2–19. doi: 10.1002/1520-6394(2000)12:1+<2::AID-DA2>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 50.Hill MN, Patel S, Carrier EJ, Rademacher DJ, Ormerod BK, Hillard CJ, et al. Downregulation of endocannabinoid signaling in the hippocampus following chronic unpredictable stress. Neuropsychopharmacology. 2005;30:508–15. doi: 10.1038/sj.npp.1300601. [DOI] [PubMed] [Google Scholar]
- 51.Willner P. Validity, reliability and utility of the chronic mild stress model of depression: a 10-year review and evaluation. Psychopharmacology. 1997;134:319–29. doi: 10.1007/s002130050456. [DOI] [PubMed] [Google Scholar]
- 52.Hill MN, Miller GE, Ho WS, Gorzalka BB, Hillard CJ. Serum endocannabinoid content is altered in females with depressive disorders: a preliminary report. Pharmacopsychiatry. 2008;41:48–53. doi: 10.1055/s-2007-993211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Patel S, Roelke CT, Rademacher DJ, Cullinan WE, Hillard CJ. Endocannabinoid signaling negatively modulates stress-induced activation of the hypothalamic-pituitary-adrenal axis. Endocrinology. 2004;145:5431–8. doi: 10.1210/en.2004-0638. [DOI] [PubMed] [Google Scholar]
- 54.Herman JP, Ostrander MM, Mueller NK, Figueiredo H. Limbic system mechanisms of stress regulation: hypothalamo-pituitary-adrenocortical axis. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29:1201–13. doi: 10.1016/j.pnpbp.2005.08.006. [DOI] [PubMed] [Google Scholar]
- 55.Bambico FR, Katz N, Debonnel G, Gobbi G. Cannabinoids elicit antidepressant-like behavior and activate serotonergic neurons through the medial prefrontal cortex. J Neurosci. 2007;27:11700–11. doi: 10.1523/JNEUROSCI.1636-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yang L, Li Y, Ren J, Zhu C, Fu J, Lin D, et al. Celastrol attenuates inflammatory and neuropathic pain mediated by cannabinoid receptor type 2. Int J Mol Sci. 2014;15:13637–48. doi: 10.3390/ijms150813637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu QR, Canseco-Alba A, Zhang HY, Tagliaferro P, Chung M, Dennis E, et al. Cannabinoid type 2 receptors in dopamine neurons inhibits psychomotor behaviors, alters anxiety, depression and alcohol preference. Sci Rep. 2017;7:17410. doi: 10.1038/s41598-017-17796-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bahi A, Al Mansouri S, Al Memari E, Al Ameri M, Nurulain SM, Ojha S. β-Caryophyllene, a CB2 receptor agonist produces multiple behavioral changes relevant to anxiety and depression in mice. Physiol Behav. 2014;135:119–24. doi: 10.1016/j.physbeh.2014.06.003. [DOI] [PubMed] [Google Scholar]
- 59.Jiang W, Zhang Y, Xiao L, Van Cleemput J, Ji SP, Bai G, et al. Cannabinoids promote embryonic and adult hippocampus neurogenesis and produce anxiolytic- and antidepressant-like effects. J Clin Invest. 2005;115:3104–16. doi: 10.1172/JCI25509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Barage SH, Sonawane KD. Amyloid cascade hypothesis: Pathogenesis and therapeutic strategies in Alzheimer’s disease. Neuropeptides. 2015;52:1–18. doi: 10.1016/j.npep.2015.06.008. [DOI] [PubMed] [Google Scholar]
- 61.Iqbal K, Liu F, Gong CX. Tau and neurodegenerative disease: the story so far. Nat Rev Neurol. 2016;12:15–27. doi: 10.1038/nrneurol.2015.225. [DOI] [PubMed] [Google Scholar]
- 62.Benito C, Núñez E, Tolón RM, et al. Cannabinoid CB2 receptors and fatty acid amide hydrolase are selectively overexpressed in neuritic plaque-associated glia in Alzheimer’s disease brains. J Neurosci. 2003;23:11136–41. doi: 10.1523/JNEUROSCI.23-35-11136.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Núñez E, Benito C, Pazos MR, Barbachano A, Fajardo O, González S, et al. Cannabinoid CB2 receptors are expressed by perivascular microglial cells in the human brain: an immunohistochemical study. Synapse. 2004;53:208–13. doi: 10.1002/syn.20050. [DOI] [PubMed] [Google Scholar]
- 64.Tolón RM, Núñez E, Pazos MR, Benito C, Castillo AI, Martínez-Orgado JA, et al. The activation of cannabinoid CB2 receptors stimulates in situ and in vitro beta-amyloid removal by human macrophages. Brain Res. 2009;1283:148–54. doi: 10.1016/j.brainres.2009.05.098. [DOI] [PubMed] [Google Scholar]
- 65.Manuel I, González dSRE, Giralt MT, Ferrer I, Rodríguezpuertas R. Type-1 cannabinoid receptor activity during Alzheimer’s disease progression. J Alzheimers Dis. 2014;42:761–6. doi: 10.3233/JAD-140492. [DOI] [PubMed] [Google Scholar]
- 66.Esposito G, De Filippis D, Steardo L, Scuderi C, Savani C, Cuomo V, et al. CB1 receptor selective activation inhibits beta-amyloid-induced iNOS protein expression in C6 cells and subsequently blunts tau protein hyperphosphorylation in co-cultured neurons. Neurosci Lett. 2006;404:342–6. doi: 10.1016/j.neulet.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 67.Esposito G, De Filippis D, Carnuccio R, Izzo AA, Iuvone T. The marijuana component cannabidiol inhibits beta-amyloid-induced tau protein hyperphosphorylation through Wnt/beta-catenin pathway rescue in PC12 cells. J Mol Med. 2006;84:253–8. doi: 10.1007/s00109-005-0025-1. [DOI] [PubMed] [Google Scholar]
- 68.Kalia LV, Lang AE. Parkinson’s disease. Lancet. 2015;386:896–912. doi: 10.1016/S0140-6736(14)61393-3. [DOI] [PubMed] [Google Scholar]
- 69.Di Filippo M, Picconi B, Tozzi A, Ghiglieri V, Rossi A, Calabresi P. The endocannabinoid system in Parkinson’s disease. Curr Pharm Des. 2008;14:2337–47. doi: 10.2174/138161208785740072. [DOI] [PubMed] [Google Scholar]
- 70.Marsicano G, Goodenough S, Monory K, Hermann H, Eder M, Cannich A, et al. CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science. 2003;302:84–8. doi: 10.1126/science.1088208. [DOI] [PubMed] [Google Scholar]
- 71.Gómez-Gálvez Y, Palomo-Garo C, Fernández-Ruiz J, García C. Potential of the cannabinoid CB(2) receptor as a pharmacological target against inflammation in Parkinson’s disease. Prog Neuropsychopharmacol Biol Psychiatry. 2016;64:200–8. doi: 10.1016/j.pnpbp.2015.03.017. [DOI] [PubMed] [Google Scholar]
- 72.Mounsey RB, Mustafa S, Robinson L, Ross RA, Riedel G, Pertwee RG, et al. Increasing levels of the endocannabinoid 2-AG is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease. Exp Neurol. 2015;273:36–44. doi: 10.1016/j.expneurol.2015.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chung YC, Bok E, Huh SH, Park JY, Yoon SH, Kim SR, et al. Cannabinoid receptor type 1 protects nigrostriatal dopaminergic neurons against MPTP neurotoxicity by inhibiting microglial activation. J Immunol. 2011;187:6508–17. doi: 10.4049/jimmunol.1102435. [DOI] [PubMed] [Google Scholar]
- 74.Chung YC, Shin WH, Baek JY, Cho EJ, Baik HH, Kim SR, et al. CB2 receptor activation prevents glial-derived neurotoxic mediator production, BBB leakage and peripheral immune cell infiltration and rescues dopamine neurons in the MPTP model of Parkinson’s disease. Exp Mol Med. 2016;48:e205. doi: 10.1038/emm.2015.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ogawa S, Kunugi H. Inhibitors of fatty acid amide hydrolase and monoacylglycerol lipase: new targets for future antidepressants. Curr Neuropharmacol. 2015;13:760–75. doi: 10.2174/1570159X13666150612225212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Navarria A, Tamburella A, Iannotti FA, Micale V, Camillieri G, Gozzo L, et al. The dual blocker of FAAH/TRPV1 N-arachidonoylserotonin reverses the behavioral despair induced by stress in rats and modulates the HPA-axis. Pharmacol Res. 2014;87:151–9. doi: 10.1016/j.phrs.2014.04.014. [DOI] [PubMed] [Google Scholar]
- 77.Gobira PH, Lima IV, Batista LA, de Oliveira AC, Resstel LB, Wotjak CT, et al. N-arachidonoyl-serotonin, a dual FAAH and TRPV1 blocker, inhibits the retrieval of contextual fear memory: role of the cannabinoid CB1 receptor in the dorsal hippocampus. J Psychopharmacol. 2017;31:750–6. doi: 10.1177/0269881117691567. [DOI] [PubMed] [Google Scholar]
- 78.Micale V, Cristino L, Tamburella A, Petrosino S, Leggio GM, Drago F, et al. Anxiolytic effects in mice of a dual blocker of fatty acid amide hydrolase and transient receptor potential vanilloid type-1 channels. Neuropsychopharmacology. 2009;34:593–606. doi: 10.1038/npp.2008.98. [DOI] [PubMed] [Google Scholar]
- 79.Carnevali L, Vacondio F, Rossi S, Macchi E, Spadoni G, Bedini A, et al. Cardioprotective effects of fatty acid amide hydrolase inhibitor URB694, in a rodent model of trait anxiety. Sci Rep. 2015;5:18218. doi: 10.1038/srep18218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Danandeh A, Vozella V, Lim J, Oveisi F, Ramirez GL, Mears D, et al. Effects of fatty acid amide hydrolase inhibitor URB597 in a rat model of trauma-induced long-term anxiety. Psychopharmacology. 2018;235:3211–21. doi: 10.1007/s00213-018-5020-7. [DOI] [PubMed] [Google Scholar]
- 81.Marco EM, Rapino C, Caprioli A, Borsini F, Laviola G, Maccarrone M. Potential therapeutic value of a novel FAAH inhibitor for the treatment of anxiety. PLoS One. 2015;10:e0137034. doi: 10.1371/journal.pone.0137034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Griebel G, Stemmelin J, Lopez-Grancha M, Fauchey V, Slowinski F, Pichat P, et al. The selective reversible FAAH inhibitor, SSR411298, restores the development of maladaptive behaviors to acute and chronic stress in rodents. Sci Rep. 2018;8:2416. doi: 10.1038/s41598-018-20895-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Scherma M, Medalie J, Fratta W, Vadivel SK, Makriyannis A, Piomelli D, et al. The endogenous cannabinoid anandamide has effects on motivation and anxiety that are revealed by fatty acid amide hydrolase (FAAH) inhibition. Neuropharmacology. 2008;54:129–40. doi: 10.1016/j.neuropharm.2007.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kinsey SG, O’Neal ST, Long JZ, Cravatt BF, Lichtman AH. Inhibition of endocannabinoid catabolic enzymes elicits anxiolytic-like effects in the marble burying assay. Pharmacol Biochem Behav. 2011;98:21–7. doi: 10.1016/j.pbb.2010.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bedse G, Bluett RJ, Patrick TA, Romness NK, Gaulden AD, Kingsley PJ, et al. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: comparative profiling of FAAH, MAGL and dual inhibitors. Transl Psychiatry. 2018;8:92. doi: 10.1038/s41398-018-0141-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Aliczki M, Zelena D, Mikics E, Varga ZK, Pinter O, Bakos NV, et al. Monoacylglycerol lipase inhibition-induced changes in plasma corticosterone levels, anxiety and locomotor activity in male CD1 mice. Horm Behav. 2013;63:752–8. doi: 10.1016/j.yhbeh.2013.03.017. [DOI] [PubMed] [Google Scholar]
- 87.Wang Y, Gu N, Duan T, Kesner P, Blaskovits F, Liu J, et al. Monoacylglycerol lipase inhibitors produce pro- or antidepressant responses via hippocampal CA1 GABAergic synapses. Mol Psychiatry. 2017;22:215–26. doi: 10.1038/mp.2016.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhong P, Wang W, Pan B, Liu X, Zhang Z, Long JZ, et al. Monoacylglycerol lipase inhibition blocks chronic stress-induced depressive-like behaviors via activation of mTOR signaling. Neuropsychopharmacology. 2014;39:1763–76. doi: 10.1038/npp.2014.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhang Z, Wang W, Zhong P, Liu SJ, Long JZ, Zhao L, et al. Blockade of 2-arachidonoylglycerol hydrolysis produces antidepressant-like effects and enhances adult hippocampal neurogenesis and synaptic plasticity. Hippocampus. 2015;25:16–26. doi: 10.1002/hipo.22344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kerr DM, Harhen B, Okine BN, Egan LJ, Finn DP, Roche M. The monoacylglycerol lipase inhibitor JZL184 attenuates LPS-induced increases in cytokine expression in the rat frontal cortex and plasma: differential mechanisms of action. Br J Pharmacol. 2013;169:808–19. doi: 10.1111/j.1476-5381.2012.02237.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Tham CS, Whitaker J, Luo L, Webb M. Inhibition of microglial fatty acid amide hydrolase modulates LPS stimulated release of inflammatory mediators. FEBS Lett. 2007;581:2899–904. doi: 10.1016/j.febslet.2007.05.037. [DOI] [PubMed] [Google Scholar]
- 92.Panlilio LV, Thorndike EB, Nikas SP, Alapafuja SO, Bandiera T, Cravatt BF, et al. Effects of fatty acid amide hydrolase (FAAH) inhibitors on working memory in rats. Psychopharmacology. 2016;233:1879–88. doi: 10.1007/s00213-015-4140-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mazzola C, Medalie J, Scherma M, Panlilio LV, Solinas M, Tanda G, et al. Fatty acid amide hydrolase (FAAH) inhibition enhances memory acquisition through activation of PPAR-alpha nuclear receptors. Learn Mem. 2009;16:332–7. doi: 10.1101/lm.1145209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Rivera P, Fernández-Arjona M, Silva-Peña D, Blanco E, Vargas A, López-Ávalos MD, et al. Pharmacological blockade of fatty acid amide hydrolase (FAAH) by URB597 improves memory and changes the phenotype of hippocampal microglia despite ethanol exposure. Biochem Pharmacol. 2018;157:244–57. doi: 10.1016/j.bcp.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 95.Hlavacova N, Chmelova M, Danevova V, Csanova A, Jezova D. Inhibition of fatty-acid amide hydrolyse (FAAH) exerts cognitive improvements in male but not female rats. Endocr Regul. 2015;49:131–6. doi: 10.4149/endo_2015_03_131. [DOI] [PubMed] [Google Scholar]
- 96.Colangeli R, Pierucci M, Benigno A, Campiani G, Butini S, Di Giovanni G. The FAAH inhibitor URB597 suppresses hippocampal maximal dentate afterdischarges and restores seizure-induced impairment of short and long-term synaptic plasticity. Sci Rep. 2017;7:11152. doi: 10.1038/s41598-017-11606-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Griebel G, Pichat P, Beeské S, Leroy T, Redon N, Jacquet A, et al. Selective blockade of the hydrolysis of the endocannabinoid 2-arachidonoylglycerol impairs learning and memory performance while producing antinociceptive activity in rodents. Sci Rep. 2015;5:7642. doi: 10.1038/srep07642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Burstein O, Shoshan N, Doron R, Akirav I. Cannabinoids prevent depressive-like symptoms and alterations in BDNF expression in a rat model of PTSD. Prog Neuropsychopharmacol Biol Psychiatry. 2018;84:129–39. doi: 10.1016/j.pnpbp.2018.01.026. [DOI] [PubMed] [Google Scholar]
- 99.Fidelman S, Mizrachi Zer-Aviv T, Lange R, Hillard CJ, Akirav I. Chronic treatment with URB597 ameliorates post-stress symptoms in a rat model of PTSD. Eur Neuropsychopharmacol. 2018;28:630–42. doi: 10.1016/j.euroneuro.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 100.Shoshan N, Segev A, Abush H, Mizrachi Zer-Aviv T, Akirav I. Cannabinoids prevent the differential long-term effects of exposure to severe stress on hippocampal- and amygdala-dependent memory and plasticity. Hippocampus. 2017;27:1093–109. doi: 10.1002/hipo.22755. [DOI] [PubMed] [Google Scholar]
- 101.Zhang J, Teng Z, Song Y, Hu M, Chen C. Inhibition of monoacylglycerol lipase prevents chronic traumatic encephalopathy-like neuropathology in a mouse model of repetitive mild closed head injury. J Cereb Blood Flow Metab. 2015;35:443–53. doi: 10.1038/jcbfm.2014.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chen X, Zhang J, Chen C. Endocannabinoid 2-arachidonoylglycerol protects neurons against β-amyloid insults. Neuroscience. 2011;178:159–68. doi: 10.1016/j.neuroscience.2011.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Pihlaja R, Takkinen J, Eskola O, Vasara J, López-Picón FR, Haaparanta-Solin M, et al. Monoacylglycerol lipase inhibitor JZL184 reduces neuroinflammatory response in APdE9 mice and in adult mouse glial cells. J Neuroinflamm. 2015;12:81. doi: 10.1186/s12974-015-0305-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Viveros-Paredes JM, Gonzalez-Castañeda RE, Escalante-Castañeda A, Tejeda-Martínez AR, Castañeda-Achutiguí F, Flores-Soto ME. Effect of inhibition of fatty acid amide hydrolase on MPTP-induced dopaminergic neuronal damage. Neurologia. 2019;34:143–52. doi: 10.1016/j.nrl.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 105.Johnston TH, Huot P, Fox SH, Wakefield JD, Sykes KA, Bartolini WP, et al. Fatty acid amide hydrolase (FAAH) inhibition reduces L-3,4-dihydroxyphenylalanine-induced hyperactivity in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned non-human primate model of Parkinson’s disease. J Pharmacol Exp Ther. 2011;336:423–30. doi: 10.1124/jpet.110.169532. [DOI] [PubMed] [Google Scholar]
- 106.Pasquarelli N, Porazik C, Bayer H, Buck E, Schildknecht S, Weydt P, et al. Contrasting effects of selective MAGL and FAAH inhibition on dopamine depletion and GDNF expression in a chronic MPTP mouse model of Parkinson’s disease. Neurochem Int. 2017;110:14–24. doi: 10.1016/j.neuint.2017.08.003. [DOI] [PubMed] [Google Scholar]