Abstract
While both the spleen and lymph nodes are called secondary lymphoid tissues, how lymphocytes enter these tissues are quite different from each other. This is because the architecture of the two types of organs and the mode of lymphocyte migration into these organs are quite distinct. In the spleen, T cells are passively released in the blood flow from the arterioles in the red pulp and marginal zone area. In contrast, T cells in the blood are actively captured on high endothelial venules in lymph nodes by the coordinated actions of CCR7 and several adhesion molecules. A recent finding indicates that T cells, released in the red pulp and marginal zone areas, actively find their way to the white zone by utilizing the migration track created by periarteriolar stromal cells. This finding adds one more piece to our understanding of lymphocyte migration for effective adaptive immune responses in the spleen.
Subject terms: Chemokines, CD4-positive T cells
Immune cells passively enter the spleen via arterioles. Naive and central memory T cells migrate from the red pulp to the white pulp by following chemotactic gradients. Once they reach the T zone of the white pulp, they search for dendritic cells that can potentially present cognate antigens originating from blood. It has been a mystery as to how exactly T cells in the red pulp reach the T zones in the white pulp. Chauveau et al. reported that T cells that migrate out of the arterioles in the red pulp and marginal zone (MZ) move to the white pulp under the guidance of the chemotactic and adhesion signals created by the periarteriolar stroma cells.1 Thus, T cells undergo directional migration from the red to the white pulp through a route created by specialized stromal cells.
The spleen has multiple functions. It functions as a blood filter by removing particulates and pathogens and recycling old red blood cells in the red pulp.2 It is also a site of extramedullary hematopoiesis, as it harbors hematopoietic progenitor cells and produces myeloid cells in the red pulp.3 It is also a site where adaptive immune responses, which are mediated by T and B cells, occur in the white pulp, and, in this regard, is similar to the T and B cell zones in lymph nodes (LNs) and Peyer’s patches.2 Moreover, it is a site of antibody production by B cells in the MZ and red pulp. A major difference between the spleen and LNs is that the spleen does not have high endothelial cells (HEVs), which are specialized cuboid endothelial cells involved in lymphocyte trafficking from blood into the T cell zone of LNs. Instead, the lymphocytes enter the spleen via arterioles that branch out from the splenic artery. Splenic arterioles end at B cell follicles, MZs, or red pulp and therefore can deliver blood fluid and its content (antigens and/or cells) to these areas but not to T zones. Chauveau et al. visualized the route of released T cells migrating from the blood into the T zones of the white pulp.
The T and B zones in secondary lymphoid tissues are created by distinct stromal cells that produce specific chemotactic and retention signals.4 CCR7-activating chemokines, such as CCL19 and CCL21, are expressed in the T zone, whereas the CXCR5-activating chemokine CXCL13 (also called BLC) is expressed in the B zone. CXCL12 (also called SDF-1), which activates CXCR4 and CXCR7, is expressed in the red pulp, MZ, and dark zone of germinal centers (GCs).5,6 HEVs are present in the T zone of LNs, where they capture circulating naive and central memory T cells in the blood circulation. These T cells express CD62L, CCR7, and basic integrins such as LFA1 to enter lymphoid tissues through HEVs. The chemokine CCL21 (also called SLC) is highly expressed by HEVs and activates CCR7,7 which, as a G-protein-coupled receptor, triggers integrin activation on T cells to firmly adhere to HEVs. HEVs also express peripheral node addressin in peripheral LNs or mucosal addressin (MAdCAM-1) in mesenteric LNs to loosely capture blood lymphocytes, which occurs via a process referred to as the “rolling” of lymphocytes, on HEVs prior to CCR7 activation and firm adhesion.8 Adhered T cells on HEVs undergo extravasation to enter LNs through HEVs and then undergo CCR7-mediated interstitial migration along CCL19 and CCL21 gradients to finally localize to T zones.9
Unlike T cell migration into LNs, lymphocyte entrance into the spleen does not require specific trafficking receptors, such as CCR7 and CD62L. This is because cells and particles in the blood are nonspecifically unloaded into the blood from the arterioles of the spleen. Thus, this passive process is quite different from the active process utilized for lymphocyte entrance into LNs. Once they are released into the red pulp, CCR7-expressing T cells migrate on a migration track created by perivascular stromal cells around red pulp arterioles to reach the T zone1 (Fig. 1). The authors refer to these as “perivascular T-tracks.” Chauveau et al. used intravital two-photon laser-scanning microscopy to image the migration of fluorescent T cells in live mice. They also used photoconvertible green fluorescent protein (GFP)-expressing T cells to track cell migration from the red to the white pulp. This migration is dependent on CCR7-mediated chemotaxis and involves integrin (LFA-1 and VLA-4)-mediated adhesion. However, CCR7 and integrins are not required for T cells to enter the track before migration. However, cell adhesion to the track is inhibited by pertussis toxin; therefore, G-protein coupled signaling is still required for T cells to find the track.1 The whole migration track from the red pulp to the T zone is actually composed of two separate domains, one is which is created by the red pulp perivascular stroma cells traveling through MZ bridging channels, while the other is created by the T zone stroma cells. Both types of stromal cells express CCL19 and CCL21, which chemotactically guide T cell migration to the T zone.1 The expression of these chemokines is rapidly downregulated in an inflammatory condition induced by lipopolysaccharide injection (which models inflammatory or infection conditions).1 Therefore, the red-to-white pulp migration track seems to be active mainly in the steady state but not during immune responses. This is consistent upon considering that the T zone architecture and T cell recruitment are greatly altered during immune and inflammatory responses, which can block the entrance of T cells in the blood into T zones during active immune responses.
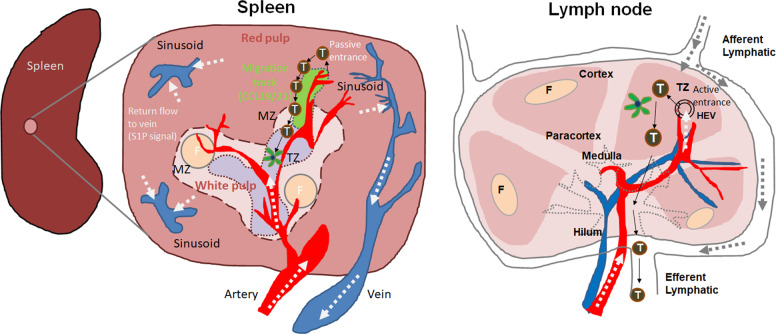
Fig. 1.
T cell trafficking into the spleen versus the lymph nodes. The spleen is largely composed of red and white pulp. T and B cells are concentrated in the white pulp, while blood particulates and red blood cells are filtered in the red pulp. Between the red and white pulp, marginal zones (MZs) are present and contain MZ B cells and macrophages to allow rapid antibody and phagocytic responses, respectively. T cells passively enter the spleen through arterioles in the red pulp and the MZ. Around the arterioles, perivascular stroma cells are present (depicted in green), forming a unidirectional migration track to T zones (TZ) in the white pulp. These cells express CCL19 and CCL21, which chemotactically guide T cells to enter the TZ through MZ bridging channels. The red pulp migration track is connected to white pulp TZ stromal cells. These T cell migration tracks allow the prompt localization of T cells passively released from the blood, promoting the efficient interaction of T cells with dendritic cells in the T cell area. This is in contrast to the HEV-mediated active entrance of T cells into the TZ of lymph nodes. What is not known is where T cells, once they are localized to the T zone, emigrate to go back to the blood circulation. Sphingosine 1-phosphate (S1P) levels are high in blood plasma and marginal sinuses, and lymphocyte exit from the spleen is regulated by S1P-dependent chemotaxis. Whether B cell entrance into follicles (F) utilizes the same routes remains to be determined. Arrows indicate the direction of blood or plasma flows (white), T cell migration (black), or lymphatic drainage (gray)
A remaining question is how can naive and central memory T cells in T zones migrate back into the blood circulation? An early study reported that T cells appear to move into the central area of the periarteriolar lymphoid sheath (PALS) before returning to the blood.10 While this indicates that the PALS may function as an exit route, this has not been demonstrated. Another possible route is through the MZ. This appears to be true for effector T cells, which downregulated CCR7 but upregulated nonlymphoid tissue-homing receptors such as CXCR3.11 This allows effector T cells to be localized to the MZ and to possibly be drained to the vascular sinusoids in the red pulp and eventually to the spleen vein. It is likely that some T cells that do not express CCR7 may use this route to exit the spleen. Activated T cells highly express CD69, which downregulates sphingosine 1-phosphate (S1P) receptor 1 expression.12 As they become resting cells, T cells downregulate CD69 but upregulate S1P1, leading to increased chemotaxis in response to S1P during emigration. The MZ has high levels of S1P, which induces lymphocyte egress.13 This pathway is also utilized in the LNs and thymus by lymphocytes exiting to the blood. However, the route of exit for naive and central memory CD4+ T cells in the white pulp to the blood circulation remains to be identified. T cell activation in the T zone, which is mainly directed by dendritic cells, generates effector T cells. It is unclear whether there is a specialized exit route for these T cells in the white pulp or, more specifically, in the T zone, where the S1P-dependent exit may occur.
In terms of B cells, while it has been proposed that B cells may enter through arterioles in the red pulp and MZ of the spleen,14 it remains unknown how they enter the follicles in the spleen. B cells have the potential to utilize the same tracks as T cells because they express CCR7 before they further localize to B follicles via CXCR5-mediated chemotaxis. T cells and follicular dendritic cells in GCs also express CXCL13 to form B follicles and to expand GCs during B cell activation.15–17 Moreover, CXCL12 is more highly expressed in the dark zone than in the light zone of GCs, which is to segregate B cells at distinct stages of differentiation following B cell receptor activation. CXCL12 is also expressed in the red pulp and leads to the retainment of plasma B cells.6 In LNs, B cells along with T cells enter through HEVs, localize to follicles, and exit via efferent lymphatic vessels. B follicles are divided into low-oxysterol inner and high-oxysterol outer areas.18 Activated B cells express EBI2, the receptor for oxysterol, and follow the oxysterol signal to localize to the outer follicles, which makes it easier for activated B cells to interact with follicular T cells.
The migration track in the red pulp identified by Chauveau et al.1 would allow efficient migration of T cells to T zones in the white pulp. Directional migration via the newly discovered perivascular track is far more efficient than random chemotaxis in the 3D environment, which does not rely on such a track. This migration strategy would increase the efficiency of antigen detection and presentation for robust T cell activation in the spleen. This track may be utilized by other cell types, such as B cells and dendritic cells, which should be validated.
Acknowledgements
C.H.K. is the Judy and Kenneth Betz Endowed Professor in the Mary H Weiser Food Allergy Center. He was supported in part by grants from the NIH (R01AI121302 and R21AI14889801).
Competing interests
The authors declare no competing interests.
References
- 1.Chauveau A, et al. Visualization of T cell migration in the spleen reveals a network of perivascular pathways that guide entry into T zones. Immunity. 2020;52:794–807. doi: 10.1016/j.immuni.2020.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mebius RE, Kraal G. Structure and function of the spleen. Nat. Rev. Immunol. 2005;5:606–616. doi: 10.1038/nri1669. [DOI] [PubMed] [Google Scholar]
- 3.Johns JL, Christopher MM. Extramedullary hematopoiesis: a new look at the underlying stem cell niche, theories of development, and occurrence in animals. Vet. Pathol. 2012;49:508–523. doi: 10.1177/0300985811432344. [DOI] [PubMed] [Google Scholar]
- 4.Kim CH. The greater chemotactic network for lymphocyte trafficking: chemokines and beyond. Curr. Opin. Hematol. 2005;12:298–304. doi: 10.1097/01.moh.0000166496.18773.e3. [DOI] [PubMed] [Google Scholar]
- 5.Allen CD, et al. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nat. Immunol. 2004;5:943–952. doi: 10.1038/ni1100. [DOI] [PubMed] [Google Scholar]
- 6.Hargreaves DC, et al. A coordinated change in chemokine responsiveness guides plasma cell movements. J. Exp. Med. 2001;194:45–56. doi: 10.1084/jem.194.1.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gunn MD, et al. A chemokine expressed in lymphoid high endothelial venules promotes the adhesion and chemotaxis of naive T lymphocytes. Proc. Natl Acad. Sci. USA. 1998;95:258–263. doi: 10.1073/pnas.95.1.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Butcher EC, Williams M, Youngman K, Rott L, Briskin M. Lymphocyte trafficking and regional immunity. Adv. Immunol. 1999;72:209–253. doi: 10.1016/S0065-2776(08)60022-X. [DOI] [PubMed] [Google Scholar]
- 9.Worbs T, Mempel TR, Bolter J, von Andrian UH, Forster R. CCR7 ligands stimulate the intranodal motility of T lymphocytes in vivo. J. Exp. Med. 2007;204:489–495. doi: 10.1084/jem.20061706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nieuwenhuis P, Ford WL. Comparative migration of B- and T-Lymphocytes in the rat spleen and lymph nodes. Cell Immunol. 1976;23:254–267. doi: 10.1016/0008-8749(76)90191-X. [DOI] [PubMed] [Google Scholar]
- 11.Kim CH, et al. Rules of chemokine receptor association with T cell polarization in vivo. J. Clin. Investig. 2001;108:1331–1339. doi: 10.1172/JCI13543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bankovich AJ, Shiow LR, Cyster JG. CD69 suppresses sphingosine 1-phosophate receptor-1 (S1P1) function through interaction with membrane helix 4. J. Biol. Chem. 2010;285:22328–22337. doi: 10.1074/jbc.M110.123299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Matloubian M, et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature. 2004;427:355–360. doi: 10.1038/nature02284. [DOI] [PubMed] [Google Scholar]
- 14.Lortan JE, Roobottom CA, Oldfield S, MacLennan IC. Newly produced virgin B cells migrate to secondary lymphoid organs but their capacity to enter follicles is restricted. Eur. J. Immunol. 1987;17:1311–1316. doi: 10.1002/eji.1830170914. [DOI] [PubMed] [Google Scholar]
- 15.Ansel KM, et al. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature. 2000;406:309–314. doi: 10.1038/35018581. [DOI] [PubMed] [Google Scholar]
- 16.Kim CH, et al. Unique gene expression program of human germinal center T helper cells. Blood. 2004;104:1952–1960. doi: 10.1182/blood-2004-03-1206. [DOI] [PubMed] [Google Scholar]
- 17.Cyster JG, et al. Follicular stromal cells and lymphocyte homing to follicles. Immunol. Rev. 2000;176:181–193. doi: 10.1034/j.1600-065X.2000.00618.x. [DOI] [PubMed] [Google Scholar]
- 18.Yi T, et al. Oxysterol gradient generation by lymphoid stromal cells guides activated B cell movement during humoral responses. Immunity. 2012;37:535–548. doi: 10.1016/j.immuni.2012.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]