Abstract
Patients with plasma cell dyscrasias produce free abnormal monoclonal Ig light chains that circulate in the blood stream. Some of them, termed glomerulopathic light chains, interact with the mesangial cells and trigger, in a manner dependent of their structural and physicochemical properties, a sequence of pathological events that results in either light chain–derived (AL) amyloidosis (AL-Am) or light chain deposition disease (LCDD). The mesangial cells play a key role in the pathogenesis of both diseases. The interaction with the pathogenic light chain elicits specific cellular processes, which include apoptosis, phenotype transformation, and secretion of extracellular matrix components and metalloproteinases. Monoclonal light chains associated with AL-Am but not those producing LCDD are avidly endocytosed by mesangial cells and delivered to the mature lysosomal compartment where amyloid fibrils are formed. Light chains from patients with LCDD exert their pathogenic signaling effect at the cell surface of mesangial cells. These events are generic mesangial responses to a variety of adverse stimuli, and they are similar to those characterizing other more frequent glomerulopathies responsible for many cases of end-stage renal disease. The pathophysiologic events that have been elucidated allow to propose future therapeutic approaches aimed at preventing, stopping, ameliorating, or reversing the adverse effects resulting from the interactions between glomerulopathic light chains and mesangium.
Key words: AL-amyloidosis, kidney, kidney repair, light chain deposition disease, mesangium, monoclonal Ig deposition disease, monoclonal light chains, stem cells
The past 2 decades have seen a continuous progress in the way by which multiple myeloma (MM) and other monoclonal gammopathies (MGs) are managed, resulting in an increase in patient survival and improvement in their quality of life.1 However, complications derived from the pathogenic properties of the abnormal monoclonal Ig still represent a challenge, as they can deeply influence the clinical evolution and outcome of the underlying disease.2 Very often, in addition to the intact monoclonal antibody, an excessive amount of the antibody light chain (LC) is secreted into the bloodstream, where it can be detected. Depending on its structural properties, the circulating free monoclonal LC can interfere with the normal physiology of certain organs and systems, causing disease.3 The kidney is one of the most frequently affected organs in plasma cell dyscrasias.2,4 Renal impairment is detected in up to 50% of patients with MM at diagnosis, and at least 5% of them will require dialysis treatment at one point of their clinical course.2
Signs and symptoms of renal damage can also be found in patients in whom the circulating monoclonal Ig is secreted by a non-overtly malignant plasma cell clone, a disorder termed as MG of renal significance (MGRS).2,4,5 Patients with MGRS do not fulfill criteria for MM or any other hematologic malignancy. In fact, only 8% of patients with AL-Am and approximately 15% to 20% of those with LCDD have underlying MM or other overt hematologic malignancies.4 Furthermore, only a small percentage of patients with MGRS exhibit other than renal manifestations. These patients are excellent candidates for renal-based therapeutic intervention, now possible to be designed because of a detailed understanding of pathophysiologic events that occur in AL-Am and LCDD (Tables 1 and 2).2,4, 5, 6, 7
Table 1.
Key points to understand the renal pathobiology of glomerulopathic light chains
|
|
|
|
|
|
AL-Am, light chain–derived amyloidosis; LCDD, light chain deposition disease.
Table 2.
Clinical characteristics of MGRS, MM, AL-Am, and LCDD
| Disease | Clone | Clinical definition | Renal manifestation |
|---|---|---|---|
| MGRS | Any | Any B-cell or plasma cell clonal lymphoproliferationa with both of the following characteristics:
|
Various depending on lesion4,5a,b |
| MM | Plasma cell | ≥10% plasma cells in bone marrow, M spike ≥ 30g/L and one or more CRAB features or biological markers of malignancyc | Most common: acute or subacute renal failure due to cast nephropathy2,6 |
| AL-Am | Plasma cell | Systemic disorder with multiorgan manifestations and monoclonal light chain fibril deposition | Massive proteinuria7 |
| LCDD | Plasma cell | Systemic disorder with primary renal involvement and nonorganized monoclonal light chain deposits | Proteinuria and hypertension2 |
AL-Am, light chain–derived amyloidosis; CLL, chronic lymphocytic leukemia; CRAB features, hypercalcemia, renal failure, anemia, bone disease; LCDD, light chain deposition disease; MGRS, monoclonal gammopathy of renal significance; MM, multiple myeloma.
The International Kidney and Monoclonal Gammopathy consensus definition of MGRS includes all B-cell or plasma cell proliferative disorders (such as smouldering MM, smouldering Waldenström macroglobulinemia, monoclonal B-cell lymphocytosis, low-grade CLL, and low-grade B-cell non-Hodgkin lymphomas) that produce a nephrotoxic monoclonal Ig.
MGRS-associated disorders can affect 1 or more renal compartments. MGRS-associated disorders that affect only the glomeruli: immunotactoid glomerulonephritis, C3 glomerulopathy, and proliferative glomerulonephritis with monoclonal Ig deposits. MGRS-associated disorders that affect only the proximal tubules: light-chain proximal tubulopathy. MGRS-associated disorders that mainly involve the glomeruli but can occasionally affect blood vessels: cryoglobulinemic glomerulonephritis. MGRS-associated disorders that usually affect all renal compartments, including glomeruli, vessels, and the tubule interstitium: Ig-related amyloidosis and monoclonal Ig deposition disease.
Biological markers of malignancy: (1) clonal bone marrow ≥ 60%, (2) serum-free light chain ratio greater than or equal to 100 provided involved free light chain level is 100 mg/l or higher, and (3) more than 1 focal lesion on magnetic resonance imaging.
The mechanism by which the monoclonal LCs cause renal damage depends on their ability to interact with different cell types and renal compartments. Some monoclonal LCs are classified as tubulopathic LCs (TLCs), as they exert their pathogenic activity in the proximal tubules, causing 2 clinically different disorders, Fanconi's syndrome and myeloma (cast) nephropathy.6,8, 9, 10 In contrast to this group, other LCs can interact with components of the glomerulus, specifically with the mesangial cells (MCs) and cause glomerular dysfunction. This group, termed glomerulopathic LCs (GLCs), primarily produce 2 diametrically opposite diseases in terms of their renal pathology: AL-Am and LCDD.11,12
In AL-Am the monoclonal LC deposits in the mesangial space in the form of an insoluble substance named amyloid (Am), whose distinctive component is nonbranching, randomly disposed fibrils of 7 to 13 nm in diameter, but variable in length.11,13 Amyloid deposition is accompanied by destruction of the mesangial matrix that is ultimately replaced by fibrils. As more fibrils deposit, the mesangial damage is accentuated, with more cellular debris and apoptotic MCs accumulating, a sequence of events that leads to renal insufficiency by cumulative glomerular damage.14,15 In contrast, in LCDD, the pathogenic LC deposits as nonordered, amorphous aggregates. This triggers a number of events resulting in mesangial expansion with increased extracellular matrix of abnormal composition (rich in tenascin), often forming mesangial nodules replacing the predominant collagen IV matrix that is present in the normal mesangium.16, 17, 18, 19
The past few decades have seen the development and implementation of several experimental platforms, including animal models that accurately reproduce key events of the renal diseases caused by GLCs.20 The availability of such platforms has been critical for understanding the pathological processes that begin with the interaction of the GLCs with the MC and eventually lead to irreversible glomerular damage. The advancement in the elucidation of the factors driving the propensity of the GLCs to form pathological aggregates, as well as the unraveling of the structural characteristics of these aggregates, have provided a much better understanding of the pathobiology involved.21, 22, 23, 24, 25, 26, 27 These studies are paving the way to the development of innovative therapeutic strategies for repairing the damage that such aggregation causes in glomeruli.28
This review article provides an integrated, global view of the most recently published information regarding the mechanisms by which the GLCs impair renal function, and the structural factors that turn the LCs glomerulopathic. Based on experimental evidence, the mechanism by which MCs may promote LC amyloid deposition is discussed. Also, this article highlights the key contribution of using different experimental platforms for unraveling the sequence of pathological events triggered by the interaction of GLCs with the MCs (Figures 1 and 2).29, 30, 31, 32, 33, 34, 35 The progress and challenges in designing and developing new therapeutic approaches to prevent mesangial injury and repair/heal the injured mesangium are also addressed.
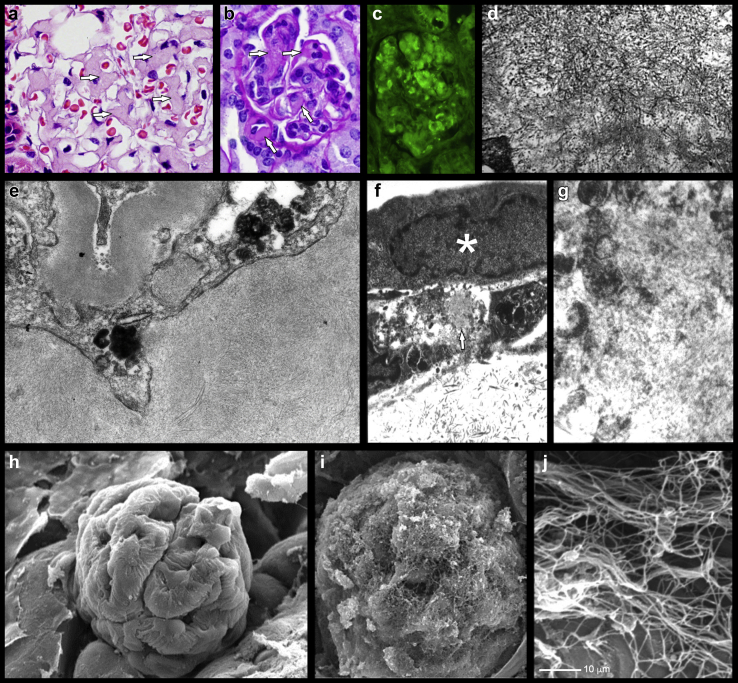
Figure 1.
Light chain–derived amyloidosis (AL-Am). Comparison of findings in experimental platforms and renal biopsy of patient with AL-Am. (a) Hematoxylin and eosin staining in renal biopsy from patient with AL-Am. Note the expanded mesangial areas with eosinophilic, amorphous material replacing normal mesangial matrix (arrows). (b–d) Samples of renal tissue obtained from rats in vivo perfused with an amyloidogenic λ light chains (LCs) through penile vein. (b) Periodic acid–Schiff (PAS) stain showing expanded mesangial areas with similar eosinophilic, amorphous material in some mesangial areas (arrows), as shown in (a). Original magnification ×750. (c) Thioflavin T staining showing fluorescence in areas with amyloid deposition. Original magnification ×750. (d) Transmission electron microscopy (TEM) showing randomly disposed, nonbranching 7- to 13-nm fibrils replacing normal mesangial matrix. Original magnification ×18,500. (e) TEM micrograph showing transformed mesangial cell (MC) with macrophage phenotype and surrounding amyloid fibrils in a renal biopsy of a patient with AL-Am. Sample stained with uranyl and lead citrate. Original magnification ×32,500. (f) TEM micrograph showing MC grown in Matrigel with amyloidogenic LC for 72 hours. Formation of amyloid (arrow) by surrounding transformed MC (with macrophage phenotype, and normal MC [∗]) on top with smooth muscle phenotype not participating in the process of amyloid formation. (g) Magnified area shown with the arrow in (f). Sample stained with uranyl and lead citrate stain. Original magnification is ×7500 and ×18,500 in (f) and (g), respectively. (h–j) Scanning electron microscopy (SEM) image of renal samples taken from rat in vivo perfused with an amyloidogenic λ LCs through penile vein. (h) Normal-appearing rat glomerulus. (i) Fibrillary material in rat glomerulus with advanced amyloid deposition. (h,i) Original magnification ×700. (j) High-magnification (×22,500) SEM micrograph showing details of amyloid fibrils. (d–g) TEM samples stained with uranyl acetate and lead citrate. (e) Reprinted from Teng J, Turbat-Herrera EA, Herrera GA. Extrusion of amyloid fibrils to the extracellular space in experimental mesangial AL-amyloidosis: transmission and scanning electron microscopy studies and correlation with renal biopsy observations. Ultrastruct Pathol. 2014;38:104–115,29 with permission from Taylor & Francis Ltd., http://www.tandfonline.com. (f,g) Reprinted with permission from Tagouri YM, Sanders PW, Picken MM, et al. In vitro AL-amyloid formation by rat and human mesangial cells. Lab Invest. 1996;74:290–302.30 Copyright © 1996, Springer Nature. (h–j) Reprinted with permission from Teng J, Turbat-Herrera EA, Herrera GA. An animal model of glomerular light-chain-associated amyloidogenesis depicts the crucial role of lysosomes. Kidney Int. 2014;86:738–746.31
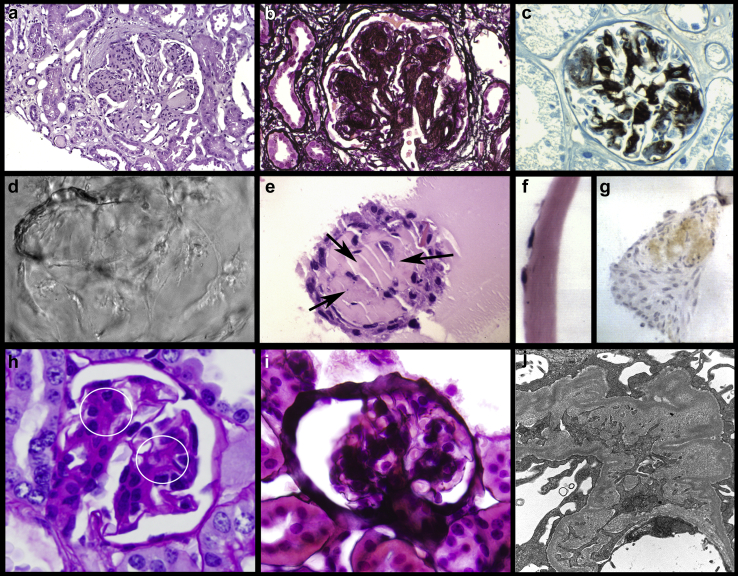
Figure 2.
Comparison of findings in experimental cellular platform and renal biopsy of patient with light chain deposition disease (LCDD). (a–c) Renal biopsy of a patient with LCDD. (a) Hematoxylin and eosin staining showing mesangial nodularity. Similarity with nodules shown in the experimental platforms is striking. Original magnification ×350. (b) Silver methenamine stain highlighting increased matrix in the mesangial areas. Original magnification ×500. (c) Immunohistochemical stain for tenascin showing abundant tenascin deposition in expanded mesangial areas. Analysis performed using Avidin Biotin Complex method method and 3,3′-diaminobenzidine as chromogen. Original magnification ×500. (d–g) Mesangial cells (MCs) grown on Matrigel incubated with monoclonal light chains (LCs) purified from urine of a patient with κ LCDD. (d) Phase-contrast microscopy showing a 3-dimensional view of accumulated material creating a nodule. Original magnification ×300. (e) Light microscopy. Hematoxylin and eosin staining showing nodule with eosinophilic staining proteinaceous material in center, which is similar to the mesangial nodules in renal biopsy (a). Arrows pointing to extracellular matrix in center and mesangial nodules. MCs incubated with LCDD-LC purified from urine of a patient for 72 hours. Original magnification ×850. (f) Control MCs incubated with tubulopathic LC from the urine of a patient with myeloma cast nephropathy. MCs growing on Matrigel as a single layer. Note difference from (e) and (f). (g) MCs incubated in Matrigel and LCDD-LC for 72 hours. Immunohistochemical stain for tenascin. Brown staining in the mesangial nodule indicating abundant tenascin deposition. Avidin-biotin technique, diaminobenzidine as marker. Original magnification ×700. (h–j) In vivo rat model of LCDD. (h) Periodic acid–Schiff (PAS) stain showing expanded mesangial with increased PAS-positive extracellular matrix (circles). (i) Silver methenamine stain showing the silver-positive expanded mesangial matrix. (h,i) Original magnification ×500. (j) TEM micrograph showing increase mesangial matrix and scattered powdery LC deposits. Uranyl acetate and lead citrate stain. Original magnification ×1500. (c) Reprinted with permission from Turbat-Herrera EA, Isaac J, Sanders PW, et al. Integrated expression of glomerular extracellular matrix proteins and beta 1 integrins in monoclonal light chain-related renal diseases. Mod Pathol. 1997;10:485–495.32 Copyright © 1997, Springer Nature. (e) Reprinted with permission from Teng J, Zhang PL, Russell WJ, et al. Insights into mechanisms responsible for mesangial alterations associated with fibrogenic glomerulopathic light chains. Nephron Physiol. 2003;94:28–38.33 Copyright © 2003 Karger Publishers, Basel, Switzerland. (g) From Keeling J, Herrera GA. An in vitro model of light chain deposition disease. Kidney Int. 2009;75:634–645.34 (h,j) Reprinted with permission from Herrera GA, Turbat-Herrera EA, Teng J. Animal models of light chain deposition disease provide a better understanding of nodular glomerulosclerosis. Nephron. 2016;132:119–136.35 Copyright © 2016 Karger Publishers, Basel, Switzerland.
Normal Catabolism of Polyclonal LCs
The LCs are 1 of the 2 types of polypeptide chains composing the human antibodies and are classified into 2 types, kappa (κ) and lambda (λ). They are approximately 214 amino acids in length (∼22 kDa of molecular weight) and fold into 2 separate domains, the variable (VL) and the constant (CL) domain. Both domains adopt the β-sandwich folding motif, typical of the Igs (Figure 3).21, 22, 23, 24,36 The sequence of the VL domain is encoded by 2 different gene segments, named VL and joining (JL) gene segments, whereas the entire CL domain is encoded by CL gene segment. The set of gene segments encoding κ LCs resides in chromosome 2 (2p11.2),37 whereas those encoding the λ type locate at chromosome 22 (22q11.2).38
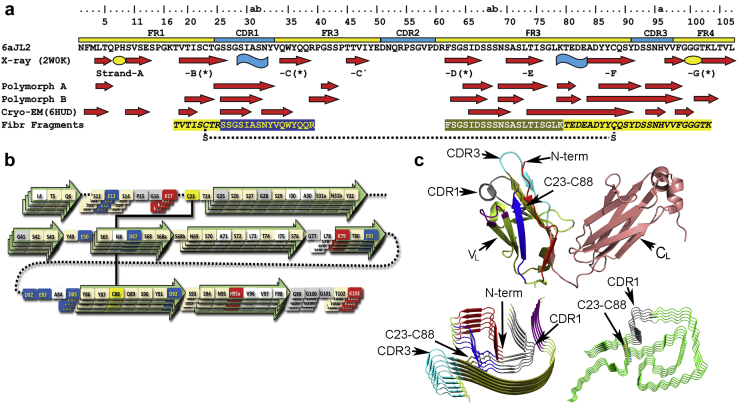
Figure 3.
(a) Comparison of the secondary structure arrangement of the λ6 VL protein 6aJL2 in native state determined by X-ray crystallography (PDB 2W0K) with that of the polymorphs A and B of the amyloid-like fibrils of the λ6 VL protein 6aJL2-R25G (determined by solid-state nuclear magnetic resonance [NMR] analysis24) and the amyloid fibrils of a cardiotoxic λ6 light chain (LC) (determined by cryogenic electron microscopy,21 PDB 6HUD). The sequence of 3 fibrillogenic fragments (Fibr fragments) of protein 6aJL2 identified by limited proteolysis with trypsin is also displayed.22 Note that one of the fibrillogenic fragments is composed of 2 protein segments covalently linked by the conserved intradomain disulfide bond between cysteines 23 and 88 (Cys23-Cys88), represented as a dotted line. β-strands and α-helices are represented as red arrows and blue curved ribbons, respectively. β-strand in native 6aJL2 protein are indicated according to the standard identification (strand-A to -G). ∗The edge strands of the VL β-sandwich. Residue numbering and the complementarity determining (CDRs) and framework (FRs) regions are according to Al-Lazikani et al.36 The green ovals represent the structural motifs “sheet-switch” and “β-bulge” that interrupt β-strands A and G, respectively, playing an anti-aggregation role. Note that most of the regions adopting β-strand conformation the amyloid fibrils overlap with the fibrillogenic fragments. (b) Schematic representation of 6aJL2-G25G fibril organization of polymorph A, according to solid-state NMR analysis. Residues showing in-register parallel orientation are represented by several layers. Protein segments in β-strand are represented as arrows. The disulfide bond Cys23-Cys88 is displayed as a line. Color code: red denotes positively charged (basic) residues, blue denotes negatively charged (acid) residues, beige denotes polar noncharged residues, white denotes hydrophobic residues, yellow denotes cysteine residues, and gray denotes glycine residues. (c) Structure of a native full-length λ6 LC (top) and 2 LC-derived (AL) amyloid fibrils, one λ621 (bottom-left) and the other λ123 (bottom-right). The structure of the λ6 LC was determined by X-ray crystallography as part of a hepcidin-Fab complex (PDB 3H0T). The variable (VL) and constant (CL) domains are indicated. The structure of both AL fibrils was determined by cryogenic electron microscopy (λ6 AL PDB 6HUD, λ1 AL PDB 6IC3). Note that both AL fibrils are assembled by the stacking of the LC monomers, one on top of each other (only 5 monomers are represented). Homologous regions of the VL of the full-length λ6 LC and the monomers of the λ6 AL fibril are colored the same. The N-terminal, CDR1, CDR3, and the disulfide bond between Cys23-Cys88 are indicated in the structures. For comparative purposes, the protein segment corresponding to the CDR1 in the native LC is colored the same in the AL structures.
The functional gene of the LC is assembled by the rearrangement of the VL, JL, and CL gene segments through a somatic recombination linked to B-cell ontogeny.39 Approximately two-thirds of the B lymphocytes of the polyclonal repertoire in healthy individuals express an antibody of κ type.
The serum of healthy individuals contains a small amount of polyclonal free LCs, which originate from the excess production of LCs over the heavy chains that occurs in the normal B cells.40 This small pool of free LCs is efficiently cleared from the blood, with a half-life of 2 to 6 hours, mainly via renal catabolism. In normal conditions, the kidneys catabolize approximately 50 mg per day of polyclonal LCs. Once filtered in the glomerulus and delivered to the proximal tubules, the polyclonal LCs are avidly endocytosed by the cubilin-megalin receptor, a unique receptor for low molecular weight proteins located at the microvillous surface of the proximal tubular cells.41, 42, 43 The LCs are catabolized in the endosomes to short peptides and free amino acids that are returned to the circulation for recycling. The cubilin/megalin complex is also recycled to the plasma membranes directly from the early endosomes via a rapid recycling pathway.41,43 The membrane-bound early endosomes represent the major cellular sorting station from which cargo molecules (in this case LCs) can either be trafficked to late endosomes and/or lysosomes for processing.
Pathogenic Aggregation of Monoclonal LCs
In patients with MGs, a monoclonal LC, either of κ or λ type, is often overproduced and secreted in free state into the bloodstream.44 This exposes the LC to the influence of environmental factors that can promote its misfolding and aggregation.
LC aggregation is a complex phenomenon that results from the interplay between several factors, some of them are protein-dependent, whereas others depend on the biology of the individual where aggregation occurs. Protein-dependent factors are those that determine the intrinsic propensity of the LC to misfold and aggregate, which is ultimately a function of LC sequence. On the other hand, the intrinsic aggregability of the LC is modulated by factors of the biological microenvironment, as the composition of the extracellular matrix,45, 46, 47 biochemical and physiological features of the cells of the targeted tissue,48,49 the efficiency of the physiological mechanisms to regulate pH,50 ionic strength,51,52 temperature,53,54 and redox status.55,56 Also, the rate of synthesis of the monoclonal LC by the abnormal plasma cell clone and the capability of kidneys to catabolize and excrete it influence the onset and progression of its deposition.
Sequence Diversity as a Driving Force of LC Deposition
LC aggregation is linked to the pathogenesis of several renal diseases, such as AL-Am, LCDD, myeloma (cast) nephropathy, and Fanconi syndrome, among others. In AL-Am, the LC deposits as fibrils characterized by an ordered core formed by intermolecular β-sheets.21,23,24 In contrast, in LCDD it forms nonorganized amorphous aggregates that, ultrastructurally, appear as electron-dense granular, punctuate to powdery deposits.11,19 In myeloma (cast) nephropathy and Fanconi syndrome, the LC forms intratubular casts in distal nephrons57 and intracytoplasmic crystals in proximal tubular cells,58 respectively. The ability to form aggregates that differ widely in ultrastructural appearance and pathobiological properties is a hallmark of the LCs59,60 and largely reflects their marked sequence heterogeneity,60 a direct consequence of the mechanisms that generate a diverse repertoire of polyclonal antibodies.39,61
Contribution of the VL Gene Segment to LC Aggregation
There is evidence that the VL gene segment that encodes the LC influences its propensity to form amyloid, as well as the tendency to predominantly deposit in certain organs (organ tropism). Only 5 (IGLV10-44, IGLV2-14, IGLV3-1, IGLV6-57, and IGKV1-33) of the approximately 70 different VL genes that compose the human repertoire encode 55% to 60% of the reported amyloidogenic LCs.62, 63, 64, 65, 66, 67 Such overrepresentation in AL suggests that these genes share some unknown molecular and/or genetic properties that make the LCs derived from them prone to deposit as amyloid in vivo.63,65,66,68 Of them, IGLV6–57, the only member of the λ6 VL subgroup, displays the strongest association with amyloidosis.67,69 It encodes approximately 20% of the amyloidogenic LC of λ type,62 a figure that contrasts with its low frequency (∼2%) in the repertoire of polyclonal bone marrow λ B cells of healthy individuals.64 Moreover, it has been observed that AL-LCs derived from IGLV6-57 are more likely to cause renal deposition, whereas those from gene segments IGLV2-14 and IGLV1-44 are more likely to cause peripheral nerve and cardiac involvement, respectively. It was also reported that the κ1 gene IGKV1-33 confers organ tropism to the LCs, because the LCs derived from it are more likely to cause liver involvement than the others.62,63,65
What makes LC derived from AL-associated VL genes prone to deposition as amyloid has not yet been elucidated. A recent study suggests that a relatively unstable and intrinsically aggregation-prone VL protein encoded in the germline IGLV6-57 gene could be one of the factors that explains its strong association to AL.70 However, not all AL-associated germline VL genes encode intrinsically amyloidogenic proteins.70 The association of some VL gene segments with AL-Am appears to be the result of a combination of several intrinsic and tissue-dependent factors that may differ from one gene to another.70 Understanding how the VL genes, in interplay with other factors, modulate the propensity of LC to form amyloid or to show organ tropism is of great relevance, because the evidence indicates that this has an impact in the clinical evolution of the disease.62,63,65,66
There also appears to be some bias in the use of VL genes in LCDD. Most of the LCs involved in LCDD are encoded by a restricted set of VL genes that belong to κ1 (IGKV1-5), κ3 (IGKV3-11 and IGKV3-15), or κ4 (IGKV4-1) subgroups.71,72 Only 2 λ LCDD LCs have been reported so far, and they were assigned to the λ2 genes IGLV2-23 and DPL12.73 A larger number of patients with LCDD need to be analyzed to establish with certainty the frequency of use of VL genes in this condition.
Contribution of the Somatic Mutations
Changes introduced in the VL domain by somatic hypermutation play a key role in LC amyloid aggregation.45,59,74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84 Because the structural impact of mutations is context-dependent, the exact mechanism by which somatic mutations promote aggregation varies depending on the nature of the change and the targeted residue.45,72,77,79, 80, 81, 82,85, 86, 87, 88 Somatic mutations can promote LC amyloidogenesis by decreasing the thermodynamic stability of the native folding, increasing the probability that the LC adopts non-native states that are aggregation-prone.77,82,83,89 In vitro studies have also shown that some mutations modify the kinetics of unfolding/refolding of the LC, causing the accumulation of non-native aggregation-prone species, an effect that may trigger amyloidogenesis.45,90 As recently reported, increasing the conformational flexibility of the LC, regardless of the impact on its thermodynamic of folding, is another way by which somatic mutations can promote amyloid aggregation.91,92 It has also been found that somatic mutations that disrupt the LC dimer interface and shift the equilibrium toward the less thermodynamically stable monomeric LC, have the potential to favor amyloidogenesis.84,93,94 Because association into a stable dimer protects LC from aggregation,89,94, 95, 96, 97 pharmacologic stabilization the LC dimer has been proposed as an potential therapeutic strategy for AL-Am amyloidosis.94,98
The role of somatic mutations in LCDD is less understood than in AL-Am. Sequence analysis has revealed that LCDD LCs are characterized by mutations that place large hydrophobic residues at solvent exposed positions of the VL.80,99 It is believed that the increase of surface hydrophobicity favors the nonordered aggregate that characterizes LCDD. Although structural analysis suggests that at least some mutations in LCDD LCs may be destabilizing,72,85 it has not been established if the loss of folding stability plays a definitive role in LCDD.
Mechanism of LC Amyloid Aggregation
The general consensus emerged from biophysical and structural studies is that the thermodynamic stability is a major driver of LC amyloid aggregation.50,54,68,74, 75, 76, 77, 78, 79,81, 82, 83,85,100, 101, 102, 103 As a general rule, AL-Am LCs tend to be thermodynamically less stable than their nonpathologic counterparts, a tendency that is more evident when proteins belonging to the same VL subgroup are compared.54,75,78,86,93,104,105 Moreover, it is also possible to turn a stable and poorly amyloidogenic LC into an aggregation-prone entity by incorporating into its sequence destabilizing mutations82,85,86 or incubating it in middle destabilizing conditions, such as low pH,50 sub-denaturing concentration of urea54,102,103 or guanidine hydrochloride,89 high temperature,53 and high hydrostatic pressure.106 Taken together, these findings support the concept that LC amyloidogenesis in essentially a misfolding event and anticipate that non-native folding intermediates are critical components of the aggregation pathway.82 Several laboratories have found evidence that non-native folding intermediates accumulate under conditions that promote amyloid aggregation of LC.89,100,103,107 However, the structural mechanism by which these misfolded species are formed and how they initiate the assembly of AL fibrils is not well understood. Solution nuclear magnetic resonance (NMR) analysis performed with β2-microglobulin108,109 and Fyn SH3110 domain, 2 proteins that, like LCs, adopt native β-sandwich folding, have provided clues to the structural characteristics of proamyloidogenic folding intermediates formed under physiological conditions. It was found that these folding intermediates retain most of the native structure (native-like intermediates), but feature perturbations of segments that normally protect β-sandwich domains from aggregation.109,110 It is thought that such local perturbations result in the unprotected exposition of proamyloidogenic sequences, normally buried in the protein core, to intermolecular contacts that may nucleate aggregation.109,110 As mentioned before, destabilizing mutations,82 oxidative stress,49,111 and interactions with nonphysiological ligands of the LCs, as cupper112 and the glycosaminoglycans,45,46,113 are among several factors that can promote local perturbation of the protective motif of LCs.
Although they are considered central in the mechanism of fibrillar aggregation, the role of proamyloidogenic sequences in LCs has been poorly studied. It was recently reported that the recombinant λ6 VL protein 6aJL2 contains 3 different segments that form amyloid-like fibrils autonomously,22 a distinctive property of the proamyloidogenic sequences114 (Figure 3a). These segments encompass most parts of 6aJL2 protein, with the exception of the β-strand A (N-terminal strand) and the long loop spanning positions Ser40 to Asp60, segments of the VL domain predicted to play protective function.81 The finding that 6aJL2 protein has several proamyloidogenic segments dispersed along its sequence led to suggest that its amyloidogenesis is driven by more than 1 amyloid-prone aggregation hotspot.22 This hypothesis is supported by a recent solution NMR study that showed that several residues located in the segments identified as proamyloidogenic hotspots22 in 6aJL2 protein engage in intermolecular contacts when the protein is incubated in destabilizing conditions that promote fibrillar aggregation.115 Taken together, these studies indicate that misfolding drives the fibrillar aggregation of 6aJL2 protein through a complex process in which more than 1 amyloid-prone hotspot could drive the aggregation.
AL Fibril Structure: What Does It Tell Us About the Mechanism of LC Amyloidogenesis?
The use of solid-state NMR and cryogenic electron microscopy spectroscopy for characterizing the structure of the LC amyloid fibrils has provided key information about the internal order of these aggregates at the atomic level. A recent solid-state NMR analysis of the amyloid-like fibrils of the rVL 6aJL2-R25G, a highly fibrillogenic mutant of 6aJL2 protein,68,78 showed that it adopts a conformation in the fibrillar state that is totally different from its native folding.24 This finding implies that the assembly of 6aJL2-R25G protein into amyloid-like fibrils occurs through a substantial structural conversion that affects the whole molecule.24 Three different polymorphs, termed A, B, and C, were found in the fibril preparations analyzed. Polymorphs A and B, the best characterized and most abundant in the samples, featured a β core formed by residues that were in β-strands in the native protein plus others located in turn regions. The fibril core consisted largely of polar amino acids, because serine, threonine, asparagine, glutamine, cysteine, and tyrosine represented 48.5% of the residues, whereas glycine and hydrophobic residues accounted for 11.8 %, and only 22.0%, respectively.24 A finding that highlights the extensive nature of the conformational rearrangement undergone by 6aJL2-R25G to aggregate is that the complementarity determining region 1 and the loop connecting the β-strands E and F, the only 2 segments in the α-helix in the native 6aJL2, also refolded into β strands to integrate into the fibril core (Figure 3a and b). This indicates that helix-to-strand transitions are part of the mechanism of 6aJL2-R25G fibrillogenesis, something that has been reported in other amyloid precursors, as prion proteins,116 Apo A-I,117 and Aβ peptides.118,119 Comparison between spectra recorded with mixed 13C:15N labeled and a fully 13C,15N-labeled samples established that 6aJL2-R25G fibrils are formed by the stacking of the monomers one on the top of another in parallel orientation and in register24 (Figure 3b). As most of the β-strands composing the fibril core overlap with the fibrillogenic fragments previously identified in 6aJL2,22 it can be inferred that the proamyloidogenic hotspots play a key structural role in 6aJL2 aggregation (Figure 3a).
Solid-state NMR analysis of 2 κ VL proteins, AL-10927 and MAK3, was recently reported. It was found that the composition of the structural core of the fibrils formed by these κ proteins differ from each other and from the fibrils of the λ6 6aJL2-R25G protein.24 This is a not surprising finding given the structural heterogeneity of the LCs. It cannot be excluded that the differences in the composition of the fibril core between these proteins reflect differences in the location and characteristics of the proamyloidogenic sequences.
Two recent studies applied cryogenic electron microscopy to determine the structure of 2 different AL fibrils, one λ6 and the other one λ1, recovered from patients with cardiac amyloidosis.21,23 It was found that only the VL domain composes the β core in both fibrils, with no contribution of the CL. It was also determined that the conformation of both proteins in the fibrillar context is totally different from the β-sandwich folding characteristics of the native VL (Figure 3c). The fibrils were formed by the stacking of the monomers refolded in a relatively flat structure, one on the top of another, and in-register parallel arrangement along the fibril axis21,23 (Figure 3c). These findings agree with the data obtained by solid-state NMR analysis of 6aJL2-R25G amyloid-like fibrils.24 However, there were differences between the 2 fibrils regarding the content of β structure of the fibril core and the topology of the VL domain (Figure 3c).21,23
Taken together, the studies mentioned in this section lead to conclude that the LCs form amyloid fibrils through an extensive structural conversion of the VL domain that refold to adopt a relatively flat structure. The stacking of totally refolded VL domains, one on top of another, assembles the fibrils, which are stabilized by a β core consisting of β-strands arranged in-register to form long intermolecular β-sheets. Finally, it is important to mention that AL fibrils formed by LCs belonging to different VL subgroups feature different topology of the VL polypeptide chain. This finding likely reflects the difference in primarily structure between these LCs and may be part of the structural basis that determines clinical heterogeneity in AL-amyloidosis.
Most of what is known about the structural and biophysical basis of LC amyloid aggregation has been proven in in vitro experiments in which cells and tissue components are generally absent. Therefore, understanding how cells can modulate the propensity of LCs to form amyloid in a tissue-specific manner and at the same time be affected by the ability of these proteins to cause damage, is of great significance to progress in the understanding of AL-amyloidosis pathogenesis. The next sections of this review address the pathological mechanisms triggered by the interaction between GLCs and MCs.
Renal Damage by Monoclonal LCs
Patients with plasma cell dyscrasias produce abnormal LCs that circulate in the bloodstream and reach the kidneys. Approximately 85% of circulating LCs in patients with these conditions result in pathological alterations.11,12,15 Approximately 70% of all nephropathic LCs cause tubular injury (TLCs), whereas the remaining 30% result in glomerular alterations (GLCs). Selected (few) LCs are both TLCs and GLCs.120 Two main diseases are caused by GLCs, AL-Am and LCDD, which differ significantly in the pathogenic mechanisms responsible for glomerular injury.
Organ damage remains the major source of mortality and morbidity in LCDD and AL-Am.121 These diseases target the mesangium122 and repair of the mesangium is essential to avoid further damage that may complicate the regeneration process to an extent that repair may not be possible at all. Glomerulosclerosis is the final pathway of glomerular damage. Using a mathematical model, it has been proposed that when more than 50% of a glomerulus is sclerosed, repair to convert the affected glomerulus into a functional one is no longer possible.123 The interaction of GLCs with glomerular structures is a complex process.33,122,124,125 In very initial steps, GLCs can be seen associated with the peripheral capillary walls, often leading to proteinuria by altering the capillary wall barrier. However, the site where key pathologic glomerular interactions of GLC occur is the mesangium, clearly seen in renal biopsy specimens from patients with early manifestations of these disorders.3,11,12,15,19,126
In AL-AM, as well as in LCDD, current treatments are based in drugs used in MM, as proteasome inhibitors, immunomodulators, and alkylators.127, 128, 129 The goal of therapy is to eliminate the clonal plasma cells producing the toxic LCs to halt and possibly reverse symptomatic organ damage. An important consideration is that several of the drugs used to treat AL-Am/LCDD can result in nephrotoxicity. Most of them produce tubulointerstitial damage, which is beyond the scope of this review, but some may result in glomerular injury.130 Although some of the glomerulopathies that may occur associated with therapy are not centered in the mesangium, others such as thrombotic microangiopathy with mesangiolysis being a crucial pathologic event could benefit in the future from organ-directed therapeutic proposals (i.e., mesenchymal stem cell rescue), as part of the clinical management.
The Normal Mesangium
The mesangial areas provide the main support for the glomerulus and are composed of MCs and matrix. The matrix contains numerous proteins and it is very rich in collagen IV. Other glycoproteins include fibronectin, laminin, proteoglycans such as perlecan and bamacan, and so forth. MCs constitute 30% to 40% of all glomerular cells. They spread from the hilum in an arboreal pattern and are embedded in a matrix that they produce and remodel. There are generally no more than 2 MCs per mesangial area in the adult glomerulus. Two types of MCs have been recognized. The predominant type exhibits distinct morphological and functional characteristics that clearly separate them from endothelial and epithelial cells. These MCs participate in contractile activities maintaining glomerular turgor and integrity. The second type of MCs is uncommon, accounting for fewer than 10% of all MCs and displays phagocytic properties. It has been proposed that these cells are derived from bone marrow and have been shown to express Fc and C3 receptors.131
Ultrastructurally, the great majority of MCs in the normal glomerulus exhibit intracytoplasmic myofilaments and attachment plaques, features of smooth muscle cells. Only rare lysosomes and a small amount of rough endoplasmic reticulum are identifiable in these cells.132, 133, 134
Histochemical stains are used in diagnostic renal pathology to highlight the mesangial matrix and the silver stain is particularly useful in this regard. The mesangial matrix is argyrophilic and when it is replaced by any extraneous material, the normal silver staining disappears.135 The mesangial matrix when viewed by transmission electron microscopy appears as a mesh of moderately electron-dense material. It is composed of a dense network of microfibrils 6 to 8 nm in diameter and of variable length, serving the purpose of anchoring the MCs.
Scanning electron microscopic evaluation of the mesangium provides a view of cell surfaces and interactions with surrounding matrix not readily apparent using any other imaging techniques available. Using scanning electron microscopy, there are well-defined but very few coated vesicles (caveolae) on the surface of normal MCs that increase significantly in numbers when MCs are incubated with GLCs but not with TLCs, providing morphologic evidence of an immediate recognition of certain but not all pathologic LCs.125,136,137 The interaction of GLCs with the surface of MCs initiates pathological events downstream regulated by signals that originate at the cell surface. Caveolins localized to the surface of membrane caveolae provide crucial signaling for downstream events.35
Alterations Produced by GLCs in MCs
Studies carried out in the past 2 decades have allowed us to understand the complex sequence of pathological events that begin once GLC interacts with the mesangium. In large part, this progress was made possible by the availability of a variety of experimental platforms that allow reproducing, in a controlled environment, what happens in patients with AL-Am and LCDD.20,124 Although different in terms of the complexity of their design, they are complementary to each other, permitting crucial evaluation of the step-by-step mechanisms involved (Figures 2 and 3) and delineation of possible key steps amenable to therapeutic intervention.30,33,34,138,139 Three experimental platforms have been used to study interactions between GLCs and MCs. The first uses a cellular approach,30,33,34,139 the second is an ex vivo platform,138 and the third an in vivo animal model.20,31,35
Incubation of GLCs with MCs in culture with or without a matrix results in 2-dimensional (2D), 3D, and 6D cellular models. Amgel or Matrigel are used as matrix to grow MCs. The composition of these 2 artificial matrices mimics that of the normal mesangial matrix and they do not contain unacceptable amounts of growth factors.30,33,34,139 Using the Live Cell imaging system, the events that occur when GLCs are incubated with MCs can be observed for prolonged periods (for several weeks) with sequential photos providing a detailed view of pathologic events. Appropriate fluorescent dyes are used to highlight various cellular compartments or types of cells in the cultures to better characterize the interactions among different cell types, including changes in cell morphology and functional correlates.31,34,35,139
The ex vivo platform uses explanted rat or mice kidneys perfused through the renal artery. This approach has the limitation that the ex vivo perfused kidney can be kept physiologically intact only for up to 96 hours, but it is particularly helpful to ensure that large quantities of monoclonal LCs are directly delivered to the kidney and this “burst” of LCs makes pathologic events express in an overt manner and observing pathogenetic cellular events becomes easier.138 The third platform involves penile injections of LCs in mice. It represents the best approach for extended studies mimicking human diseases and best suited to observe and understand the repair process beyond the early phases.20,31,35 The injection of LCs via a penile route bypasses the systemic circulation preventing LC dilution and results in direct delivery of large amounts of LC to the kidneys.31
There are crucial cellular alterations that occur when MCs are incubated with AL-Am– and LCDD-derived LCs which can be appreciated by light, transmission, and scanning electron microscopy. When incubated with AL-LCs, MCs transform into a macrophage phenotype acquiring numerous lysosomes and losing their smooth muscle features.125 These cells acquire CD68 (a macrophage marker) and lose smoothelin, muscle-specific and smooth muscle actins (smooth muscle cell markers). In contrast, when MCs are exposed to LCDD LCs, they acquire abundant rough endoplasmic reticulum developing a myofibroblastic phenotype (Figure 4).48,140 Transmission electron microscopy clearly depicts these morphological alterations. In both cases, the structural changes in the MCs endow them to perform new functions that will evolve into producing the typical pathological findings of each of the 2 conditions. In the case of AL-Am, the phenotypic change makes MCs able to engage in active phagocytosis of LCs and eventually generate amyloid fibrils. In the case of LCDD, the MCs with additional acquired machinery become ready to produce excess extracellular matrix proteins that will be different from those present in the normal mesangium34,35,133 (Figure 4).
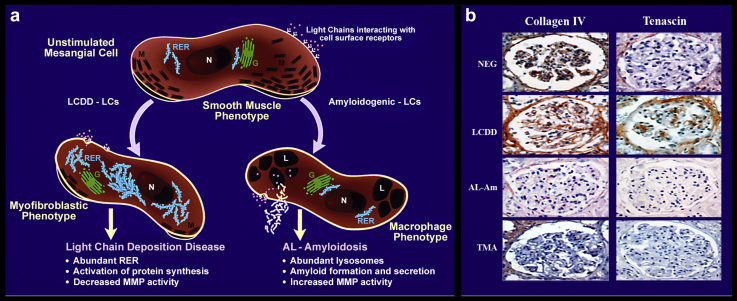
Figure 4.
Impact of glomerulopathic long chains (GLCs) in protein expression in renal glomeruli. (a) Schematic representation of phenotypic transformation that occurs in mesangial cells (MCs) after interaction with GLCs (but not tubulopathic LCs [TLCs]). G, Golgi complex; L, lysosome; M, myofilaments; N, nucleus; RER, rough endoplasmic reticulum. (b) Immunohistochemical stain for collagen IV and tenascin in renal biopsies from patients with light chain deposition disease (LCDD), light chain–derived amyloidosis (AL-Am), thin glomerular basement membrane disease (NEG), and thrombotic microangiopathy (TMA). NEG and TMA biopsies were included as negative and positive controls, respectively. Collagen IV is by far the most prominent extracellular matrix protein in the normal glomeruli with essentially no tenascin. In LCDD, tenascin replaces collagen IV in the expanded (nodular) mesangial areas. No tenascin remains in AL-Am and in TMA, less than normal collagen IV is present. Analysis performed using biotin avidin complex method and 3,3'-diaminobenzidine as chromogen. (a) Reprinted with permission from Keeling J, Teng J, Herrera GA. AL-amyloidosis and light-chain deposition disease light chains induce divergent phenotypic transformations of human mesangial cells. Lab Invest. 2004;84:1322–1338.48 Copyright © 2004, Springer Nature. (b) Reprinted with permission from Keeling J, Herrera GA. Matrix metalloproteinases and mesangial remodeling in light chain-related glomerular damage. Kidney Int. 2005;68:1590–1603.140
Numerous receptors have been described on the surface of MCs, including those for platelet-derived growth factor (PDGF-β),141 transforming growth factor-β (TGF-β),142 mannose,143 CD 71 IgA1 Fc,144 and vascular endothelial growth factor.145 Other pertinent receptors, such as cubilin/megalin and CD89-polymeric Ig (FcαR1), are not present on quiescent or activated MCs. Some of the receptors on MCs are expressed only when exposed to certain stimuli, whereas other receptors are weakly expressed normally and become readily detectable/visible when MCs are activated. Among the surface receptors in MCs are integrins. These mediate cell attachment to the extracellular matrix (especially through β1 integrins) and the effects of the extracellular matrix on a number of cell functions.146 They also mediate interactions with other cells. Therefore, the function of integrins include regulation of MC proliferation and matrix turnover and, therefore, are important in the understanding of the pathogenesis of renal diseases. As such, they play a significant role in the pathogenesis of mesangiopathies in LC-associated glomerular disorders. In glomerular diseases associated with monoclonal LCs, colocalized with tenascin in the center of mesangial nodules in LCDD and at the periphery of mesangial areas partially replaced by amyloid in AL-Am, highlighting the importance of this integrin in the pathogenesis of these disorders.32
When MCs are incubated with GLCs but not TLCs, they exhibit characteristic surface alterations with development of caveolae (coated pits),125,136,137,139 and on transmission electron microscopy (using immunogold labeling techniques) and scanning electron microscopy it is apparent that GLCs interact with MCs through activation of receptors and mediators located in these caveolae124,137 (Figure 5).124,125,139 These stimulate specific downstream events through mediators that ultimately alter extracellular matrices.147, 148, 149 Resulting from these surface interactions is the activation of “dormant” genes that affect MC behavior. The increased release of matrix proteins, such as tenascin, is the essence of the extracellular matrix alterations in nodular glomerulosclerosis associated with LCDD. It occurs in response to signaling events initiated at the cell surface by the binding of LCs to receptors (Figure 4). Furthermore, the trafficking and localization of amyloid precursor proteins within the post-Golgi endocytic/lysosomal system plays a crucial role in the exposure of LCs to secretases that mediate its cleavage to form amyloid fibrils in the acidic lysosomal compartment.150, 151, 152, 153, 154
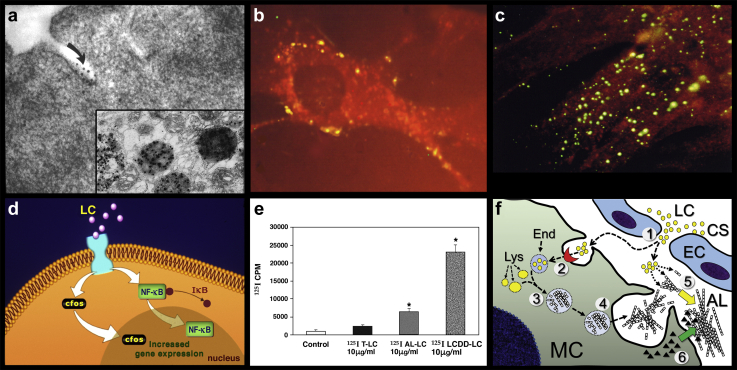
Figure 5.
Mechanism of amyloidogenesis in mesangial cells (MCs) incubated with amyloidogenic light chains (LCs). (a) Initial interaction of the LC with surface caveolae in MCs where receptor resides, after 30 seconds post incubation. The image shows transmission electron microscopy analysis with immunogold labeling (10-nm gold particles) for λ LCs in MCs incubated with an amyloidogenic λ LC. The insert shows the same LC after 5 hours post incubation in mature lysosomes. Original magnification in (a) and in insert is ×35,000 and ×17,500, respectively. (b) Direct fluorescence of MC co-incubated with Texas red-labeled (red) amyloidogenic LC and fluorescein (green)-labeled light chain deposition disease (LCDD) LC. Both LCDD and amyloidogenic LCs compete for the same receptor on the surface of MCs (colocalization showing yellow staining). Note that amyloidogenic LCs are avidly internalized. Original magnification ×500. (c) Internalization of fluorescein isothiocyanate-labeled amyloidogenic LCs into MCs detected by direct fluorescence. Original magnification ×500. (d) Schematic representation of interactions of glomerulopathic LCs (GLCs) with MCs and activation of c-fos and nuclear factor (NF)–κB to activate downward cellular pathways. (e) Comparison of various types of 125I-labeled LCs binding to MCs at 30 minutes of incubation. Note prominent interaction of LCDD LCs with surface of MCs and lesser but significant of amyloidogenic LCs (statistically significant when compared with tubulopathic LCs). The amyloidogenic LCs are avidly internalized. (f) Diagrammatic representation of LC-derived (AL) amyloid formation by MCs: (1) The unstable and misfolding-prone monoclonal LC (yellow circles) is attracted from the glomerular capillary to the MC. (2) The LC internalizes into the MCs by a receptor-mediated mechanism. (3) The early endosome (End) containing the misfolded LC fuses with the lysosomes (Lys), transforming into a late lysosome, where the self-assembly of the LC into amyloid fibrils occur. (4) The fibrils formed inside the lysosomes are extruded from the MC, accumulating in the extracellular space. (5) Soluble monomers of the monoclonal LCs aggregate into the preformed fibrils, which seed the aggregation reaction. Fibrils accumulate in the extracellular space. (6) Matrix metalloproteinases and other proteases secreted by the MCs proteolyze the AL, removing the protease-sensitive LC constant domain (CL), as well as other components of the extracellular matrix. This results in substitution of extracellular matrix by AL amyloid fibrils. Experimental data support steps (1–4). In steps (5) and (6), a possible mechanism of AL amyloid deposition in the glomeruli mediated by MCs is proposed. CS, capillary space; EC, endothelial cells. (a) From Herrera GA, Russell WJ, Isaac J, et al. Glomerulopathic light chain-mesangial cell interactions modulate in vitro extracellular matrix remodeling and reproduce mesangiopathic findings documented in vivo. Ultrastruct Pathol. 1999;23:107–126,124 with permission from Taylor & Francis Ltd., http://www.tandfonline.com. (b,c,e) Reprinted with permission from Teng J, Russell WJ, Gu X, et al. Different types of glomerulopathic light chains interact with mesangial cells using a common receptor but exhibit different intracellular trafficking patterns. Lab Invest. 2004;84:440–451.125 Copyright © 2004, Springer Nature. (d) Reprinted with permission from Herrera GA, Turbat-Herrera EA, Teng J. Understanding mesangial damage and repair: insights from an experimental model of immunoglobulin light chain-associated mesangiopathy. J Cell Biol Cell Metab. 2014;1:003.139 Copyright © 2014 Guillermo A Herrera, et al.
MC apoptosis also occurs as a cellular death pathway when GLCs interact with MCs, and this is likely the main mechanism involved in MC deletion.124,136 In the late phases of both AL-Am and LCDD, there is a marked reduction of MCs. An important part of the process of mesangial regeneration is to provide cellular elements that can carry out the process to fruition, as often there are few or essentially no viable MCs capable of doing so, because even when identified, these MCs may already be in the apoptotic cascade with limited to essentially no possibility of recovery. Once the process of amyloidogenesis begins, the deposited fibrils in the extracellular space, resulting from extrusion from MCs,29 create the nidus for further amyloid formation, eliminating the need for cellular elements. Amyloid fibrils have the potential to nucleate subsequent soluble precursor protein accelerating fibril formation. Amyloid seeding is a recognized potent mechanism of amplification and spread of disease-related fibrils completely independent of cells. Seeding circumvents the nucleation step of the fibrillogenesis, which is the rate-limiting reaction of fibrillogenesis.155, 156, 157
A number of growth factors regulate mesangial homeostasis, including PDGF-β and TGF-β, among others.133 These two are particularly important as the first promotes MC proliferation and TGF-β activation increases mesangial matrix, playing a key role in glomerulosclerosis. TGF-β also enhances the activity of connective tissue growth factor. There is constant mesangial turnover as a physiologic process with continuous engagement of these factors in the process.132,133 MCs are susceptible to injurious stimuli reacting vigorously, first with MC proliferation mediated through PDGF-β. The activation of TGF-β results in a negative feedback loop to inhibit MC proliferation (through PDGF-β inactivation) adversely affecting replacement of injured MCs.17,18,33,122 Once amyloid is deposited extracellularly, it inhibits PDGF-β, thus controlling the mesangial cellular proliferation.140
Other factors that play a significant role in mesangial homeostasis include metalloproteinases (MMPs), tissue inhibitors of MMPs, alpha 2-macroglobulin, and plasminogen activator inhibitor, among others.28,133,140,158 The mesangium can repair itself when the injury is minor or as part of normal homeostasis. However, pathologic processes ensue when injurious agents damage the mesangium to a point where significant matrix modification takes place and return to normal is not possible.
The most important mesangial MMPs are -2, -5, -7, and -9. MMPs-1, -8, and -13 have been shown to play important roles in other renal compartments. Although MMPs have been demonstrated to have some substrate specificity, it has been shown that more than one MMP may degrade the same proteins. The normal mesangium contains predominantly MMPs -2 and -9 (also known as gelatinases or collagenases). The proper balance of these 2 MMPs together with the combined activities of PDGF-β and TGF-β are crucial for maintenance of a healthy mesangium. MMPs play important roles in AL-Am and LCDD. Although in AL-Am glomerular MMPs are activated by the amyloid deposited in the extracellular matrix, in LCDD they are inhibited, thus resulting in mesangial destruction in amyloidosis and increased matrix in LCDD.122,133,158 In renal biopsies, in mesangial amyloidosis there is replacement of the normal matrix resulting in loss of argyrophilia and in LCDD, there is increased matrix, resulting in accentuated silver staining.144 Both show staining with the periodic acid–Schiff, although less in AL-Am for the same reason. MMPs may also play a role in removing the LC CL domain by proteolysis.75,98
Molecular chaperones are a rather diverse group of functionally related proteins that, regardless of their structural and spatial differences, generally (almost invariably) suppress amyloid formation, and are likely to be important in defining the result of aggregation-prone proteins in tissue in vivo. Chaperones may prevent events associated with aggregation by blocking promiscuous intermolecular interactions or by assisting in the refolding of aberrant substrates. Molecular chaperones interact preferentially with soluble oligomeric prefibrillar species.159 In addition, there are other proteins that also interact with amyloid fibrils in an similar fashion and can modulate their pathobiological properties. These proteins are known as accessory amyloid molecules,160 and 2 of them, apolipoprotein E and serum amyloid P, have been speculated to act as pathological chaperones.161 This is an important area for future research.
Understanding the role of complement pathways in the pathogenesis of MGRS-associated C3 glomerulonephritis remains a work in progress. It appears that functional dysregulation of the alternate pathway may be at play. The monoclonal GLCs may act as autoantibodies to complement or related regulatory proteins (i.e., CFH-complement factor H) and by that mechanism in an indirect manner activate a complement-mediated glomerulonephritis.162, 163, 164 These diseases have a direct effect on mesangial pathobiology producing glomerular alterations that are similar to those seen in LCDD (MC proliferation/matrix secretion).
GLCs-MCs: Surface and Downstream Events
Both GLCs interact with the same receptor on MCs. GLCs because of their physicochemical characteristics are attracted, guided to, and interact with MCs using a unique receptor that is present on their surface where caveolae are present: the sortilin-related protein receptor (SORL1).165 SORL1, a transmembrane sorting receptor, is additionally involved in endocytosis, sorting and trafficking of amyloid precursor proteins playing unique essential role in amyloid-beta peptide handling in Alzheimer disease in which the mechanism of amyloid formation in cerebral-vascular smooth muscle cells is very similar to that of the renal mesangium morphologically and functionally.166 Both AL and LCDD LCs compete for this receptor.125 This receptor sorts the GLCs to different cellular compartments and is crucial for signaling downstream cellular events, culminating in pathological events that are diametrically opposite. The activated pathways have been elucidated in vitro and reproduced in in vivo animal platforms. Likewise, the sequence of events that occurs as a result of AL- and LCDD-associated LC interactions with mesangium are also observed in sequential renal biopsies from patients with these diseases, highlighting the translational importance of these investigations as a way to understand MC pathobiology and with the ultimate purpose of designing new therapeutic interventions.11,12,15
Within a few minutes after G (but not T) LCs interact with MCs there is activation of NF-κβ and c-fos with migration of signals from cytoplasm to nuclei.122,136 These initially activated mediators are responsible for eliciting certain activities in the MCs. NF-κβ activation correlates with the production of monocyte chemoattractant protein-1 by human MCs, whereas c-fos has an effect resulting in cytoskeletal rearrangement (MC rounding and development of surface membrane ruffling) through PDGF-β activation and, ultimately, enhancement of MC proliferation. Phenotypic transformation of MCs is also regulated by c-fos activation, which permits transformation and ultimately, downstream events. For example, if c-fos migration from cytoplasm to nucleus is inhibited (i.e., with epillocatechin-3 [EGCG], green tea), amyloid formation is decreased.167, 168, 169 Studies performed with caveolin 1 knockout mice have demonstrated an important signaling role for caveolin 1 in LCDD where downstream glomerular events do not occur.35
MCs in Renal Amyloidosis
Once internalized into MCs, amyloidogenic LCs are transported to the mature lysosomal compartment with the help of Rab proteins.170 SORL1 also plays a pivotal role in internalizing proteins into lysosomes to be catabolized.171 The ability of the MCs to produce fibrils from intact LCs intracellularly and subsequently extrude them into the extracellular space29 is indicative that these cells are crucial participants in the process leading to the deposition of amyloid in the glomerulus, by seeding the aggregation of circulating monomers. It is well known that the fibrillogenesis of most proteins and peptides is a nucleation-dependent reaction.172 Also, it has been extensively proven that the amyloid aggregation can be accelerated by adding preformed fibrils, which act as seeds, catalyzing the aggregation of the soluble monomers.173 Therefore, MCs could trigger and/or enhance AL deposition in glomeruli by their capacity to selectively internalize the circulating full-length AL-LC and provide the required destabilizing environment (lysosome) where the fibrillogenesis is efficiently promoted. The acidic environment characterizing the mature lysosomal compartment could be enough destabilizing as to promote the unfolding and aggregation of the full-length LC, overcoming the protective effect of the CL98,174,175 (Figure 5f). Moreover, proteolysis by lysosomal enzymes is a crucial factor promoting LC fibrillogenesis in MCs.22,154,176 A MC-mediated mechanism of LC amyloidogenesis is not necessarily exclusive, as it can coexist with other aggregation pathways that may or may not depend on previous proteolysis of the monoclonal LC. It has been found that vascular smooth muscle cells can also promote amyloid aggregation of clinically proven amyloidogenic LCs by a mechanism that involves receptor-mediated internalization and trafficking to the mature lysosomal compartment, with the subsequent extrusion of the amyloid fibrils177 (Figure 5). Furthermore, some LCs due to their peculiar physicochemical characteristics may be endocytosed and delivered to the mature lysosomal compartment where in the process of catabolism of these LCs, crystals are formed in a manner similar to what happens in proximal tubular cells in Fanconi syndrome. Therefore, a cell-mediated mechanism for AL amyloid formation may represent a general component of the pathogenesis of AL-Am, not exclusively in the kidney.
Stem Cells in Glomerular Repair
Because kidney repair capability is limited, the development of therapeutic strategies aimed to fix damaged renal structures represents an attractive option that is being investigated by several laboratories. The use of stem cells (SCs) for glomerular regeneration represents a promising approach that may become a realistic goal for the future.
Kidney regeneration is a complex process that requires reprogramming of endogenous renal cells, migration of bone marrow–derived cells, renal cell progenitor differentiation and neoangiogenesis, just to mention a few of the key processes required.178, 179, 180 Intrinsic mechanisms of glomerular repair include the participation of sublethally injured MCs, often already engaged in the apoptotic pathway, but still able to repair themselves and effectively function aided by bone marrow SCs that can migrate to the injured areas, and renal SC pools.181,182 SCs have been shown to play a role in self-renewal and are capable of differentiating into several phenotypes.88,180, 181, 182, 183, 184, 185 For example, bone marrow cells have been shown to differentiate into MCs in murine recipients when the glomerulus was damaged by an antibody-mediated glomerulonephritis.184
Niches of glomerular cells to replace podocytes have been discovered182,186,187 in the glomeruli and their vicinity. The best described niches are located in Bowman’s capsule88,187 and near the juxtaglomerular apparatus where renin-producing cells are present.186,187 In contrast, understanding mesangial repair by glomerular progenitor SCs has lagged behind. There are only a handful of reports focusing specifically on mesangial repair.28,181,183, 184, 185
Renal cell progenitors either located in the kidney or recruited from the circulation, may, in addition, potentiate survival and cellular proliferation of remaining cells, contributing to renal repair. Renal progenitor cells, in general, are identified by cell markers such as CD133, CD24, and CD54.188 Sagrinati et al.189 have demonstrated that a subgroup of parietal epithelial cells coexpress CD24 and 133, as well as other SC-specific transcription factors such as BmI-I, Oct-4 and Nanog, suggesting that they represent SCs. These SCs can migrate to areas of damage and differentiate accordingly.
Glomerular repair/regeneration requires cellular proliferation and differentiation of SC precursors and, only when the appropriate extracellular matrix and proper microenvironment are there, this can occur in an orderly and adequate manner. The degree of glomerular damage will dictate whether intrinsic mechanisms suffice. The reservoir of SCs with plasticity to regenerate the glomerulus is enough to support normal glomerular cellular turnover but insufficient to repair damaged glomeruli in most glomerulonephritis.190
Future Potential Therapeutic Approaches/New Research Directions
Organ damage remains the major source of mortality and morbidity in both AL-Am and LCDD.121 Hematologic responses are not always associated with beneficial organ responses emphasizing the importance of organ-based therapeutic approaches.121 In the case of MGRS, the plasma cell clone may be difficult to identify or a small one, making clone-directed therapies challenging and highlighting the crucial role of organ-based therapies in this setting to prevent further renal damage leading to irreversibility. Emerging therapies to ameliorate the toxicity of GLCs and/or resolve amyloid deposits and excess extracellular matrix in AL-Am and LCDD, respectively, are most welcome. Understanding how monoclonal LCs interact with MCs to lead to pathological events has provided crucial valuable information to design new potential therapeutic interventions at the kidney level. These therapeutic approaches together with measures to provide for or enhance mesangial repair provide hope for organ-based treatments in the future.
Competition at the receptor site for LCs on MCs using structure-based drug design strategies based on crystallography and/or NMR data to generate novel ligands for SORL1 has already been proposed as a treatment strategy.191 This will diminish or abolish the interactions between GLCs and SORL1 and subsequent downstream pathological events. This approach would be of benefit in both LCDD and AL-Am.
Preventing internalization of the LCs into MCs represent another potential strategy to be used specifically in AL-Am. The next area of potential therapeutic intervention is to interfere with the transport of the internalized monoclonal LC from endosomes to the mature lysosomal compartment where amyloid fibrils are formed. This will entail pharmacologically regulating Rab proteins that participate in the intracellular transport of LCs.170
Altering the lysosomal pH is another manipulation to reduce amyloid generation. The formation of fibrils requires a stringent acidic pH (approximately 5) environment in the mature lysosomal compartment. Fibrils are subsequently dumped into the extracellular matrix (Figure 5f). It has been shown experimentally that increasing the lysosomal pH with chloroquine administration decreases the formation of fibrils.150
Finally, interventions at the level of the extracellular compartment aiming at destroying amyloid and/or preventing “seeding,” which propagates amyloid deposition, are alternate potential therapeutic options. Promoting immunotherapeutic clearance of AL deposit with antibodies is an option currently under active research. One approach has been to target serum amyloid P component, a molecule common to all type of amyloid deposits, with a fully humanized monoclonal antibody.192 Another approach under development is to target specifically the LC fibrils. Currently, 2 monoclonal antibodies, 11-1F4 and 2A4, that bind AL-Am fibrils and promote their phagocytic removal, are under clinical evaluation. MAb 11-1F4 was generated by immunizing with aggregates of a κ4 LC variable domain protein193 and MAb 2A4 was generated by immunizing with a short peptide derived from sAA.194 It was shown that both antibodies recognize the AL fibrils formed by both κ and λ LCs.194, 195, 196 These manipulations, although showing some positive effects, have not been entirely satisfactory for the intended purposes. Likewise, the delivery to the mesangium of MMP-7 transfected SCs to foster destruction of the tenascin-rich expanded mesangium represents a strategy that needs to be further investigated to treat LCDD.197
In some instances, more than 1 mechanism is at play. For example, there is experimental evidence that epillocatechin-3 gallate (EGCG, green tea) can interfere with LC amyloidogenesis by a dual mechanism: by stabilizing a nonamyloidogenic conformation in the ECM and inhibiting a mesangial cell signaling pathway (translocation of c-fos signal from cytoplasm to nuclei) linked to MC phenotypic transformation and downstream intracellular amyloid formation in lysosomes.198
The therapeutic approaches aimed at inhibiting the aggregation of the monoclonal LC, or promoting the clearance of the already deposited LC aggregates, or modulate the intracellular signaling pathways and processes that sustain the cytotoxicity of GLCs, either applied independently or as part of an integral therapy, have the potential to change the bad prognosis that characterizes the diseases linked to LC deposition. However, their successful translation into the clinic would require the continued and coordinated efforts of the research community and pharmaceutical industry.
A promising strategy to contend with the organ disfunction caused by monoclonal LC deposition is that aimed to stimulate the regeneration of the injured tissue. As heart and kidney are the most frequently affected organs in disorders with LC deposition, most of the research effort in this area has been focused on understanding how regeneration of these organs can be promoted. The most significative advances in cardiac regeneration in AL-Am was recently addressed in a review article.199 Here, we summarize what has been done regarding regeneration of renal mesangium. Because of the challenges with endogenous glomerular repair, as detailed, the potential use of exogenous mesenchymal SCs has been explored. The models described here of mesangial damage can also be used to assess how exogenous SC therapy may result in kidney repair. Starting with accomplishing mesangial regeneration represents a feasible approach because some diseases begin and progress in the mesangium before affecting other glomerular compartments.35,139,181,200,201 If these diseases can be treated in early stages and mesangial homeostasis can be reestablished, this will represent a significant advance in the management of these patients. In addition, SC therapy has been suggested to delay progression of glomerular disorders.202
Repairing the damaged mesangium presents a number of challenges: (1) absence of functional MCs to satisfactorily engage in the repair process, (2) removal of debris and abnormal extracellular matrix proteins, such as tenascin,11,203 and (3) lack of sufficient growth factors and other effector molecules to participate in and facilitate the glomerular healing process.204, 205, 206, 207
Mesenchymal SCs are undifferentiated cells that inherently possess immunomodulatory208 and site-specific trophic properties and, most importantly, an ability to differentiate into a number of cells types. Exogenous mesenchymal SCs selectively migrate to areas of mesangial damage where they proceed to clean up debris and dispose of apoptotic bodies, damaged matrix and extraneous material such as amyloid fibrils by virtue of transforming to facultative macrophages in the early phase of mesangial repair137,183 (Figure 6). After the clean-up is finished, the SCs transform again developing morphological and functional characteristics of MCs in the late phase of the mesangial repair183 (Figure 6).
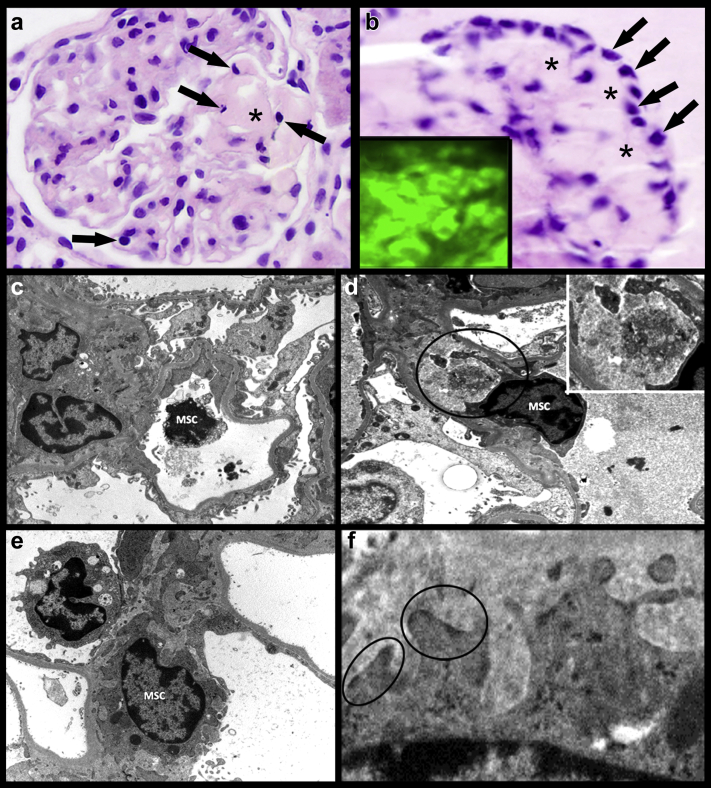
Figure 6.
Interactions of mesenchymal stem cells (MSCs) with mesangial cells (MCs) incubated with amyloidogenic light chains (LCs) in the process of amyloid formation. (a) Hematoxylin and eosin staining of mouse glomerulus with amyloid deposits (asterisk) and MSCs (arrows) surrounding deposits (original magnification ×500). (b) MCs growing on Matrigel incubated with AL-LC and 48 hours later MSCs added. Note the MSCs (arrows) surrounding the amyloid deposits (asterisks). The insert shows green-labeled MSCs, allowing identification of them in the experimental media. (c) MSC in capillary space (original magnification ×7000) and (d) identifying mesangial area with damage and embracing area that needs repair (circles and insert) (original magnification in [d] and insert is ×7000 and ×14,000, respectively). (e) Incorporation of MSCs into the damaged mesangial area (original magnification ×15,500) and (f) eventual morphologic transformation into MCs with attachment plaques and myofilaments (circles) (original magnification ×20,500). Reprinted with permission from Herrera GA, Teng J, Liu X, et al. Mesenchymal stem cells in mesangial repair in a model of immunoglobulin light-chain mediated mesangial injury. J Stem Cell Res Ther. 2014;4:13.137 © 2014 Guillermo A. Herrera, et al.
While in the research laboratory exogenous mesenchymal SCs have been shown to be of great value in repairing damaged mesangial areas by providing paracrine factors, as suggested initially,209 and by transforming into phagocytic cells removing debris/abnormal material, finally becoming functional MCs, confirmed later,181,183, 184, 185 there are a number of obvious challenges as efforts are made trying to move this type of therapy into the clinical arena. From vascular occlusion produced by the infusion of mesenchymal SCs, to delivery of these cells specifically to damaged areas in selected organs, to determine the amounts of these cells needed for the expected results to simply defining ways of how to administer them represent a number of the challenges that need to be considered seriously before going forward with translational applications of this technology to serve patients.205,207 Delivery of exogenous mesenchymal SCs directly to the kidney would avoid some of the difficulties and decrease complications.
Finally, creating the proper “homing” for the SCs to be attracted to the areas of damage and to provide them with the appropriate milieu for development and maturation needs to be a priority to accomplish a successful mesangial repair. In part, this was accomplished in experimental platforms by delivering a “cocktail” of factors that facilitated the repair process together with the SCs, using the ex vivo and in vivo animal models210 (Figure 7,183). This “cocktail” contained pertinent growth factors (PDGF-β and TGF-β), differentiating factor retinoid acid that stimulates cellular phenotypic transformation,211 and curcumin, which has a number of interesting properties that aid in repair. It has been shown that curcumin enhances activity of MMPs, disrupts interactions between integrins and extracellular matrix facilitating matrix removal, and may prime the extracellular matrix to facilitate cleavage.212, 213, 214, 215 The repair process occurred faster and more efficiently when the “cocktail” was administered, solidifying the value of this approach183 (Figure 7). Strategies currently in development for kidney-targeted drug delivery can enhance the efficacy of growth factors and drugs capable of stimulating the migration into the damaged areas and subsequent differentiation of exogenous mesenchymal SCs, reducing the undesirable effects in other tissues.216
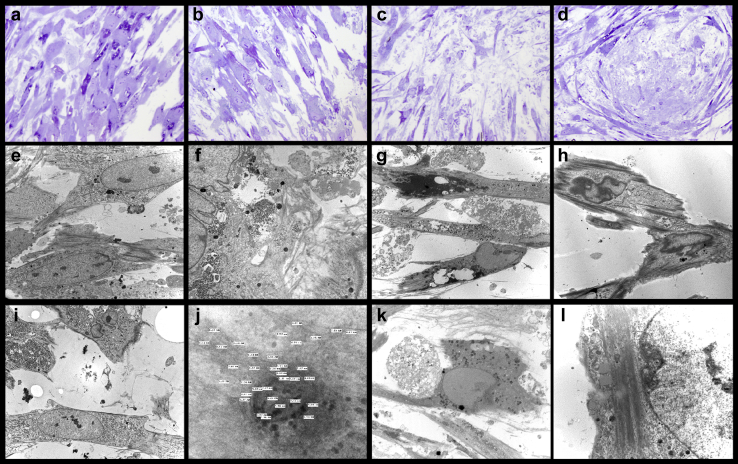
Figure 7.
Process of mesangial repair in vitro illustrating early and late phases of the process and the value of “cocktail” administration. First column (a,e,i): mesangial cells (MCs) in culture with no light chains (LCs). These images portray normal MCs in culture. Note clean background and cells with intracytoplasmic filaments and attachment plaques. Second column (b,f,j): Transformed MCs (macrophage phenotype) incubated with amyloidogenic LCs forming amyloid. Note the presence of amyloid deposits formed by transformed (macrophage-like) MCs. Third column (c,g,k): MCs incubated with amyloidogenic LCs and mesenchymal stem cells (SCs) without “cocktail.” Note the presence of transformed mesenchymal SCs with numerous lysosomes cleaning the damaged mesangial area. Fourth column (d,h,l): MCs incubated with amyloidogenic LCs, mesenchymal SCs, and “cocktail.” The mesenchymal SCs have transformed to MCs with peripheral intracytoplasmic myofilaments and attachment plaques (late repair process). This shows that the repair process is far more efficient (and faster) when the “cocktail” is used. (a–d) Toluidine blue staining. (e–l) Transmission electron microscopy with uranyl and lead citrate staining. Original magnification (a–d) ×500, (e–h) ×1000, (i, k) ×2000, (g) ×2500, and (l) ×12,500. (a–l) From Herrera GA, Teng J, Zeng C, et al. Phenotypic plasticity of mesenchymal stem cells is crucial for mesangial repair in a model of immunoglobulin light chain-associated mesangial damage. Ultrastruct Pathol. 2018;42:262–288,183 with permission from Taylor & Francis Ltd., http://www.tandfonline.com.
Conclusions
Our understanding of how glomerular damage occurs in monoclonal LC-related renal diseases has been significantly advanced in the past decades. This has provided us with delineation of key steps in the pathological processes that are amenable to pharmacologic intervention to prevent, control, modulate, or delay progressive glomerular damage leading to irreversibility. Although abolishing or controlling the injurious agent/s remains critical, especially when systemic in nature, regulating pathological events locally may provide a reasonably sound approach to prevent damage and/or promote enhanced healing. The mesangium has become a clear target for avoiding irreversible changes once the plasma cell neoplastic process is controlled or eradicated.
There are several potential areas where therapeutic intervention at the glomerular level requires further exploration. Although the opportunities that exogenous SC therapy offers are exciting, several limitations including difficulties associated with delivery, proper cellular engraftment, and difficulties in monitoring negative effects that may occur need to be further carefully studied.
Disclosure
All the authors declared no competing interests.
Acknowledgments
This work was supported in part by a Senior Research Grant on Stem Cell from The Amyloidosis Foundation granted to GAH and a Royal Society-backed Newton Mobility Grant (Project ID Number: 006075) granted to LPY. We thank Adrian Hoff for his valuable contribution in the preparation of figures.
References
- 1.Martin T., Huff C.A. Multiple myeloma: current advances and future directions. Clin Lymphoma Myeloma Leuk. 2019;19:255–263. doi: 10.1016/j.clml.2019.03.025. [DOI] [PubMed] [Google Scholar]
- 2.Yadav P., Cook M., Cockwell P. Current trends of renal impairment in multiple myeloma. Kidney Dis (Basel) 2016;1:241–257. doi: 10.1159/000442511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sanders P.W., Herrera G.A. Monoclonal immunoglobulin light chain-related renal diseases. Semin Nephrol. 1993;13:324–341. [PubMed] [Google Scholar]
- 4.Leung N., Bridoux F., Batuman V. The evaluation of monoclonal gammopathy of renal significance: a consensus report of the International Kidney and Monoclonal Gammopathy Research Group. Nat Rev Nephrol. 2019;15:45–59. doi: 10.1038/s41581-018-0077-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Leung N., Bridoux F., Hutchison C.A. Monoclonal gammopathy of renal significance:when MGUS is no longer undetermined or insignificant. Blood. 2012;120:4292–4295. doi: 10.1182/blood-2012-07-445304. [DOI] [PubMed] [Google Scholar]
- 6.Hutchison C.A., Batuman V., Behrens J. The pathogenesis and diagnosis of acute kidney injury in multiple myeloma. Nat Rev Nephrol. 2011;8:43–51. doi: 10.1038/nrneph.2011.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kastritis E., Gavriatopoulou M., Roussou M. Renal outcomes in patients with AL amyloidosis: prognostic factors, renal response and the impact of therapy. Am J Hematol. 2017;92:632–639. doi: 10.1002/ajh.24738. [DOI] [PubMed] [Google Scholar]
- 8.Herrera G.A. Proximal tubulopathies associated with monoclonal light chains:the spectrum of clinicopathologic manifestations and molecular pathogenesis. Arch Pathol Lab Med. 2014;138:1365–1380. doi: 10.5858/arpa.2013-0493-OA. [DOI] [PubMed] [Google Scholar]
- 9.Cheng M., Gu X., Turbat-Herrera E.A. Tubular injury and dendritic cell activation are integral components of light chain-associated acute tubulointerstitial nephritis. Arch Pathol Lab Med. 2019;143:1212–1224. doi: 10.5858/arpa.2018-0032-OA. [DOI] [PubMed] [Google Scholar]
- 10.Ronco P.M., Aucouturier P. The molecular bases of plasma cell dyscrasia-related renal diseases. Nephrol Dial Transplant. 1999;14(Suppl 1):4–8. doi: 10.1093/ndt/14.suppl_1.4. [DOI] [PubMed] [Google Scholar]
- 11.Herrera G.A. Renal manifestations of plasma cell dyscrasias:an appraisal from the patients' bedside to the research laboratory. Ann Diagn Pathol. 2000;4:174–200. doi: 10.1016/s1092-9134(00)90042-x. [DOI] [PubMed] [Google Scholar]
- 12.Sanders P.W., Herrera G.A., Kirk K.A. Spectrum of glomerular and tubulointerstitial renal lesions associated with monotypical immunoglobulin light chain deposition. Lab Invest. 1991;64:527–537. [PubMed] [Google Scholar]
- 13.Herrera G.A., Paul R., Turbat-Herrera E.A. Ultrastructural immunolabeling in the diagnosis of light-chain-related renal disease. Pathol Immunopathol Res. 1986;5:170–187. doi: 10.1159/000157010. [DOI] [PubMed] [Google Scholar]
- 14.Herrera G.A. The contributions of electron microscopy to the understanding and diagnosis of plasma cell dyscrasia-related renal lesions. Med Electron Microsc. 2001;34:1–18. doi: 10.1007/s007950100000. [DOI] [PubMed] [Google Scholar]
- 15.Herrera G.A. Renal lesions associated with plasma cell dyscrasias:practical approach to diagnosis, new concepts, and challenges. Arch Pathol Lab Med. 2009;133:249–267. doi: 10.5858/133.2.249. [DOI] [PubMed] [Google Scholar]
- 16.Floege J., Eitner F., Alpers C.E. A new look at platelet-derived growth factor in renal disease. J Am Soc Nephrol. 2008;19:12–23. doi: 10.1681/ASN.2007050532. [DOI] [PubMed] [Google Scholar]
- 17.Herrera G.A., Shultz J.J., Soong S.J. Growth factors in monoclonal light-chain--related renal diseases. Hum Pathol. 1994;25:883–892. doi: 10.1016/0046-8177(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 18.Zhu L., Herrera G.A., Murphy-Ullrich J.E. Pathogenesis of glomerulosclerosis in light chain deposition disease. Role for transforming growth factor-beta. Am J Pathol. 1995;147:375–385. [PMC free article] [PubMed] [Google Scholar]
- 19.Lin J., Markowitz G.S., Valeri A.M. Renal monoclonal immunoglobulin deposition disease:the disease spectrum. J Am Soc Nephrol. 2001;12:1482–1492. doi: 10.1681/ASN.V1271482. [DOI] [PubMed] [Google Scholar]
- 20.Sirac C., Herrera G.A., Sanders P.W. Animal models of monoclonal immunoglobulin-related renal diseases. Nat Rev Nephrol. 2018;14:246–264. doi: 10.1038/nrneph.2018.8. [DOI] [PubMed] [Google Scholar]
- 21.Swuec P., Lavatelli F., Tasaki M. Cryo-EM structure of cardiac amyloid fibrils from an immunoglobulin light chain AL amyloidosis patient. Nat Commun. 2019;10:1269. doi: 10.1038/s41467-019-09133-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ruiz-Zamora R.A., Guillaume S., Al-Hilaly Y.K. The CDR1 and other regions of immunoglobulin light chains are hot spots for amyloid aggregation. Sci Rep. 2019;9:3123. doi: 10.1038/s41598-019-39781-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Radamaker L., Lin Y.H., Annamalai K. Cryo-EM structure of a light chain-derived amyloid fibril from a patient with systemic AL amyloidosis. Nat Commun. 2019;10:1103. doi: 10.1038/s41467-019-09032-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lecoq L., Wiegand T., Rodriguez-Alvarez F.J. A substantial structural conversion of the native monomer leads to in-register parallel amyloid fibril formation in light chain amyloidosis. Chembiochem. 2019;20:1027–1031. doi: 10.1002/cbic.201800732. [DOI] [PubMed] [Google Scholar]
- 25.Brumshtein B., Esswein S.R., Sawaya M.R. Identification of two principal amyloid-driving segments in variable domains of Ig light chains in systemic light-chain amyloidosis. J Biol Chem. 2018;293:19659–19671. doi: 10.1074/jbc.RA118.004142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Piehl D.W., Blancas-Mejia L.M., Ramirez-Alvarado M. Solid-state NMR chemical shift assignments for AL-09 VL immunoglobulin light chain fibrils. Biomol NMR Assign. 2017;11:45–50. doi: 10.1007/s12104-016-9718-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hora M., Sarkar R., Morris V. MAK33 antibody light chain amyloid fibrils are similar to oligomeric precursors. PLoS One. 2017;12 doi: 10.1371/journal.pone.0181799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Herrera G.A., Turbat-Herrera E.A., Teng J. Glomerular repair: Current status and future expectations. In: Herrera G.A., editor. Experimental Models for Renal Diseases. Pathogenesis and Diagnosis. Karger; Basel: 2011. pp. 351–362. [Google Scholar]
- 29.Teng J., Turbat-Herrera E.A., Herrera G.A. Extrusion of amyloid fibrils to the extracellular space in experimental mesangial AL-amyloidosis:transmission and scanning electron microscopy studies and correlation with renal biopsy observations. Ultrastruct Pathol. 2014;38:104–115. doi: 10.3109/01913123.2013.861568. [DOI] [PubMed] [Google Scholar]
- 30.Tagouri Y.M., Sanders P.W., Picken M.M. In vitro AL-amyloid formation by rat and human mesangial cells. Lab Invest. 1996;74:290–302. [PubMed] [Google Scholar]
- 31.Teng J., Turbat-Herrera E.A., Herrera G.A. An animal model of glomerular light-chain-associated amyloidogenesis depicts the crucial role of lysosomes. Kidney Int. 2014;86:738–746. doi: 10.1038/ki.2014.122. [DOI] [PubMed] [Google Scholar]
- 32.Turbat-Herrera E.A., Isaac J., Sanders P.W. Integrated expression of glomerular extracellular matrix proteins and beta 1 integrins in monoclonal light chain-related renal diseases. Mod Pathol. 1997;10:485–495. [PubMed] [Google Scholar]
- 33.Teng J., Zhang P.L., Russell W.J. Insights into mechanisms responsible for mesangial alterations associated with fibrogenic glomerulopathic light chains. Nephron Physiol. 2003;94:28–38. doi: 10.1159/000071288. [DOI] [PubMed] [Google Scholar]
- 34.Keeling J., Herrera G.A. An in vitro model of light chain deposition disease. Kidney Int. 2009;75:634–645. doi: 10.1038/ki.2008.504. [DOI] [PubMed] [Google Scholar]
- 35.Herrera G.A., Turbat-Herrera E.A., Teng J. Animal models of light chain deposition disease provide a better understanding of nodular glomerulosclerosis. Nephron. 2016;132:119–136. doi: 10.1159/000443282. [DOI] [PubMed] [Google Scholar]
- 36.Al-Lazikani B., Lesk A.M., Chothia C. Standard conformations for the canonical structures of immunoglobulins. J Mol Biol. 1997;273:927–948. doi: 10.1006/jmbi.1997.1354. [DOI] [PubMed] [Google Scholar]
- 37.Malcolm S., Barton P., Murphy C. Localization of human immunoglobulin kappa light chain variable region genes to the short arm of chromosome 2 by in situ hybridization. Proc Natl Acad Sci U S A. 1982;79:4957–4961. doi: 10.1073/pnas.79.16.4957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Emanuel B.S., Cannizzaro L.A., Magrath I. Chromosomal orientation of the lambda light chain locus:V lambda is proximal to C lambda in 22q11. Nucleic Acids Res. 1985;13:381–387. doi: 10.1093/nar/13.2.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brack C., Hirama M., Lenhard-Schuller R. A complete immunoglobulin gene is created by somatic recombination. Cell. 1978;15:1–14. doi: 10.1016/0092-8674(78)90078-8. [DOI] [PubMed] [Google Scholar]
- 40.Nakano T., Matsui M., Inoue I. Free immunoglobulin light chain:its biology and implications in diseases. Clin Chim Acta. 2011;412:843–849. doi: 10.1016/j.cca.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 41.Herrera G.A. Low molecular weight proteins and the kidney: physiologic and pathologic considerations. Ultrastruct Pathol. 1994;18:89–98. doi: 10.3109/01913129409016277. [DOI] [PubMed] [Google Scholar]
- 42.Batuman V., Verroust P.J., Navar G.L. Myeloma light chains are ligands for cubilin (gp280) Am J Physiol. 1998;275:F246–F254. doi: 10.1152/ajprenal.1998.275.2.F246. [DOI] [PubMed] [Google Scholar]
- 43.Christensen E.I., Birn H. Megalin and cubilin:synergistic endocytic receptors in renal proximal tubule. Am J Physiol Renal Physiol. 2001;280:F562–F573. doi: 10.1152/ajprenal.2001.280.4.F562. [DOI] [PubMed] [Google Scholar]
- 44.Sanders P.W. Renal involvement in plasma cell dyscrasias. Curr Opin Nephrol Hypertens. 1993;2:246–252. doi: 10.1097/00041552-199303000-00011. [DOI] [PubMed] [Google Scholar]
- 45.Blancas-Mejia L.M., Hammernik J., Marin-Argany M. Differential effects on light chain amyloid formation depend on mutations and type of glycosaminoglycans. J Biol Chem. 2015;290:4953–4965. doi: 10.1074/jbc.M114.615401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Martin D.J., Ramirez-Alvarado M. Glycosaminoglycans promote fibril formation by amyloidogenic immunoglobulin light chains through a transient interaction. Biophys Chem. 2011;158:81–89. doi: 10.1016/j.bpc.2011.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Stevens F.J., Kisilevsky R. Immunoglobulin light chains, glycosaminoglycans, and amyloid. Cell Mol Life Sci. 2000;57:441–449. doi: 10.1007/PL00000706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Keeling J., Teng J., Herrera G.A. AL-amyloidosis and light-chain deposition disease light chains induce divergent phenotypic transformations of human mesangial cells. Lab Invest. 2004;84:1322–1338. doi: 10.1038/labinvest.3700161. [DOI] [PubMed] [Google Scholar]
- 49.Nieva J., Shafton A., Altobell L.J., 3rd Lipid-derived aldehydes accelerate light chain amyloid and amorphous aggregation. Biochemistry. 2008;47:7695–7705. doi: 10.1021/bi800333s. [DOI] [PubMed] [Google Scholar]
- 50.Velazquez-Lopez I., Valdes-Garcia G., Romero Romero S. Localized conformational changes trigger the pH-induced fibrillogenesis of an amyloidogenic lambda light chain protein. Biochim Biophys Acta Gen Subj. 2018;1862:1656–1666. doi: 10.1016/j.bbagen.2018.04.014. [DOI] [PubMed] [Google Scholar]
- 51.Sikkink L.A., Ramirez-Alvarado M. Salts enhance both protein stability and amyloid formation of an immunoglobulin light chain. Biophys Chem. 2008;135:25–31. doi: 10.1016/j.bpc.2008.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Solomon A., Weiss D.T., Pepys M.B. Induction in mice of human light-chain-associated amyloidosis. Am J Pathol. 1992;140:629–637. [PMC free article] [PubMed] [Google Scholar]
- 53.Sikkink L.A., Ramirez-Alvarado M. Biochemical and aggregation analysis of Bence Jones proteins from different light chain diseases. Amyloid. 2008;15:29–39. doi: 10.1080/13506120701815324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim Y., Wall J.S., Meyer J. Thermodynamic modulation of light chain amyloid fibril formation. J Biol Chem. 2000;275:1570–1574. doi: 10.1074/jbc.275.3.1570. [DOI] [PubMed] [Google Scholar]
- 55.Lu Y., Jiang Y., Prokaeva T. Oxidative post-translational modifications of an amyloidogenic immunoglobulin light chain protein. Int J Mass Spectrom. 2017;416:71–79. doi: 10.1016/j.ijms.2016.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zottig X., Laporte Wolwertz M., Golizeh M. Effects of oxidative post-translational modifications on structural stability and self-assembly of lambda6 immunoglobulin light chain. Biophys Chem. 2016;219:59–68. doi: 10.1016/j.bpc.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 57.Sathick I.J., Drosou M.E., Leung N. Myeloma light chain cast nephropathy, a review. J Nephrol. 2019;32:189–198. doi: 10.1007/s40620-018-0492-4. [DOI] [PubMed] [Google Scholar]
- 58.El Hamel C., Thierry A., Trouillas P. Crystal-storing histiocytosis with renal Fanconi syndrome: pathological and molecular characteristics compared with classical myeloma-associated Fanconi syndrome. Nephrol Dial Transplant. 2010;25:2982–2990. doi: 10.1093/ndt/gfq129. [DOI] [PubMed] [Google Scholar]
- 59.Del Pozo-Yauner L., Becerril B., Ochoa-Leyva L. The structural determinants of the immunoglobulin light chain amyloid aggregation. In: Olivares-Quiroz L., Guzman-Lopez O., Jardón-Valadez H.E., editors. Physical Biology of Proteins and Peptides. Springer; Cham, Switzerland: 2015. pp. 1–28. [Google Scholar]
- 60.Bellotti V., Mangione P., Merlini G. Review:immunoglobulin light chain amyloidosis--the archetype of structural and pathogenic variability. J Struct Biol. 2000;130:280–289. doi: 10.1006/jsbi.2000.4248. [DOI] [PubMed] [Google Scholar]
- 61.French D.L., Laskov R., Scharff M.D. The role of somatic hypermutation in the generation of antibody diversity. Science. 1989;244:1152–1157. doi: 10.1126/science.2658060. [DOI] [PubMed] [Google Scholar]
- 62.Kourelis T.V., Dasari S., Theis J.D. Clarifying immunoglobulin gene usage in systemic and localized immunoglobulin light-chain amyloidosis by mass spectrometry. Blood. 2017;129:299–306. doi: 10.1182/blood-2016-10-743997. [DOI] [PubMed] [Google Scholar]
- 63.Abraham R.S., Geyer S.M., Price-Troska T.L. Immunoglobulin light chain variable (V) region genes influence clinical presentation and outcome in light chain-associated amyloidosis (AL) Blood. 2003;101:3801–3808. doi: 10.1182/blood-2002-09-2707. [DOI] [PubMed] [Google Scholar]
- 64.Perfetti V., Casarini S., Palladini G. Analysis of V(lambda)-J(lambda) expression in plasma cells from primary (AL) amyloidosis and normal bone marrow identifies 3r (lambdaIII) as a new amyloid-associated germline gene segment. Blood. 2002;100:948–953. doi: 10.1182/blood-2002-01-0114. [DOI] [PubMed] [Google Scholar]
- 65.Comenzo R.L., Zhang Y., Martinez C. The tropism of organ involvement in primary systemic amyloidosis: contributions of Ig V(L) germ line gene use and clonal plasma cell burden. Blood. 2001;98:714–720. doi: 10.1182/blood.v98.3.714. [DOI] [PubMed] [Google Scholar]
- 66.Comenzo R.L., Wally J., Kica G. Clonal immunoglobulin light chain variable region germline gene use in AL amyloidosis:association with dominant amyloid-related organ involvement and survival after stem cell transplantation. Br J Haematol. 1999;106:744–751. doi: 10.1046/j.1365-2141.1999.01591.x. [DOI] [PubMed] [Google Scholar]
- 67.Ozaki S., Abe M., Wolfenbarger D. Preferential expression of human lambda-light-chain variable-region subgroups in multiple myeloma, AL amyloidosis, and Waldenstrom's macroglobulinemia. Clin Immunol Immunopathol. 1994;71:183–189. doi: 10.1006/clin.1994.1070. [DOI] [PubMed] [Google Scholar]
- 68.del Pozo Yauner L., Ortiz E., Sanchez R. Influence of the germline sequence on the thermodynamic stability and fibrillogenicity of human lambda 6 light chains. Proteins. 2008;72:684–692. doi: 10.1002/prot.21934. [DOI] [PubMed] [Google Scholar]
- 69.Solomon A., Frangione B., Franklin E.C. Bence Jones proteins and light chains of immunoglobulins. Preferential association of the V lambda VI subgroup of human light chains with amyloidosis AL (lambda) J Clin Invest. 1982;70:453–460. doi: 10.1172/JCI110635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Garay Sanchez S.A., Rodriguez Alvarez F.J., Zavala-Padilla G. Stability and aggregation propensity do not fully account for the association of various germline variable domain gene segments with light chain amyloidosis. Biol Chem. 2017;398:477–489. doi: 10.1515/hsz-2016-0178. [DOI] [PubMed] [Google Scholar]
- 71.Denoroy L., Deret S., Aucouturier P. Overrepresentation of the V kappa IV subgroup in light chain deposition disease. Immunol Lett. 1994;42:63–66. doi: 10.1016/0165-2478(94)90036-1. [DOI] [PubMed] [Google Scholar]
- 72.Vidal R., Goni F., Stevens F. Somatic mutations of the L12a gene in V-kappa(1) light chain deposition disease:potential effects on aberrant protein conformation and deposition. Am J Pathol. 1999;155:2009–2017. doi: 10.1016/s0002-9440(10)65520-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Decourt C., Touchard G., Preud'homme J.L. Complete primary sequences of two lambda immunoglobulin light chains in myelomas with nonamyloid (Randall-type) light chain deposition disease. Am J Pathol. 1998;153:313–318. doi: 10.1016/s0002-9440(10)65573-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Blancas-Mejia L.M., Misra P., Dick C.J. Immunoglobulin light chain amyloid aggregation. Chem Commun (Camb) 2018;54:10664–10674. doi: 10.1039/c8cc04396e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Oberti L., Rognoni P., Barbiroli A. Concurrent structural and biophysical traits link with immunoglobulin light chains amyloid propensity. Sci Rep. 2017;7:16809. doi: 10.1038/s41598-017-16953-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Villalba M.I., Canul-Tec J.C., Luna-Martinez O.D. Site-directed mutagenesis reveals regions implicated in the stability and fiber formation of human lambda3r light chains. J Biol Chem. 2015;290:13039. doi: 10.1074/jbc.A114.629550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.del Pozo-Yauner L., Wall J.S., Gonzalez Andrade M. The N-terminal strand modulates immunoglobulin light chain fibrillogenesis. Biochem Biophys Res Commun. 2014;443:495–499. doi: 10.1016/j.bbrc.2013.11.123. [DOI] [PubMed] [Google Scholar]
- 78.Gonzalez-Andrade M., Becerril-Lujan B., Sanchez-Lopez R. Mutational and genetic determinants of lambda6 light chain amyloidogenesis. FEBS J. 2013;280:6173–6183. doi: 10.1111/febs.12538. [DOI] [PubMed] [Google Scholar]
- 79.Poshusta T.L., Sikkink L.A., Leung N. Mutations in specific structural regions of immunoglobulin light chains are associated with free light chain levels in patients with AL amyloidosis. PLoS One. 2009;4:e5169. doi: 10.1371/journal.pone.0005169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Davis D.P., Gallo G., Vogen S.M. Both the environment and somatic mutations govern the aggregation pathway of pathogenic immunoglobulin light chain. J Mol Biol. 2001;313:1021–1034. doi: 10.1006/jmbi.2001.5092. [DOI] [PubMed] [Google Scholar]
- 81.Hernandez-Santoyo A., del Pozo Yauner L., Fuentes-Silva D. A single mutation at the sheet switch region results in conformational changes favoring lambda6 light-chain fibrillogenesis. J Mol Biol. 2010;396:280–292. doi: 10.1016/j.jmb.2009.11.038. [DOI] [PubMed] [Google Scholar]
- 82.Hurle M.R., Helms L.R., Li L. A role for destabilizing amino acid replacements in light-chain amyloidosis. Proc Natl Acad Sci U S A. 1994;91:5446–5450. doi: 10.1073/pnas.91.12.5446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Marin-Argany M., Guell-Bosch J., Blancas-Mejia L.M. Mutations can cause light chains to be too stable or too unstable to form amyloid fibrils. Protein Sci. 2015;24:1829–1840. doi: 10.1002/pro.2790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bhavaraju M., Hansmann U.H. Effect of single point mutations in a form of systemic amyloidosis. Protein Sci. 2015;24:1451–1462. doi: 10.1002/pro.2730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Helms L.R., Wetzel R. Specificity of abnormal assembly in immunoglobulin light chain deposition disease and amyloidosis. J Mol Biol. 1996;257:77–86. doi: 10.1006/jmbi.1996.0148. [DOI] [PubMed] [Google Scholar]
- 86.Raffen R., Dieckman L.J., Szpunar M. Physicochemical consequences of amino acid variations that contribute to fibril formation by immunoglobulin light chains. Protein Sci. 1999;8:509–517. doi: 10.1110/ps.8.3.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Levinson R.T., Olatoye O.O., Randles E.G. Role of mutations in the cellular internalization of amyloidogenic light chains into cardiomyocytes. Sci Rep. 2013;3:1278. doi: 10.1038/srep01278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Romagnani P. Family portrait:renal progenitor of Bowman's capsule and its tubular brothers. Am J Pathol. 2011;178:490–493. doi: 10.1016/j.ajpath.2010.11.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Qin Z., Hu D., Zhu M. Structural characterization of the partially folded intermediates of an immunoglobulin light chain leading to amyloid fibrillation and amorphous aggregation. Biochemistry. 2007;46:3521–3531. doi: 10.1021/bi061716v. [DOI] [PubMed] [Google Scholar]
- 90.Blancas-Mejia L.M., Tischer A., Thompson J.R. Kinetic control in protein folding for light chain amyloidosis and the differential effects of somatic mutations. J Mol Biol. 2014;426:347–361. doi: 10.1016/j.jmb.2013.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Mukherjee S., Pondaven S.P., Hand K. Effect of amino acid mutations on the conformational dynamics of amyloidogenic immunoglobulin light-chains: a combined NMR and in silico study. Sci Rep. 2017;7:10339. doi: 10.1038/s41598-017-10906-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kazman P., Vielberg M.T., Pulido Cendales M.D. Fatal amyloid formation in a patient's antibody light chain is caused by a single point mutation. Elife. 2020;9 doi: 10.7554/eLife.52300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Baden E.M., Randles E.G., Aboagye A.K. Structural insights into the role of mutations in amyloidogenesis. J Biol Chem. 2008;283:30950–30956. doi: 10.1074/jbc.M804822200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wolwertz M.L., Nguyen P.T., Quittot N. Probing the role of lambda6 immunoglobulin light chain dimerization in amyloid formation. Biochim Biophys Acta. 2016;1864:409–418. doi: 10.1016/j.bbapap.2016.01.009. [DOI] [PubMed] [Google Scholar]
- 95.Zhao J., Zhang B., Zhu J. Structure and energetic basis of overrepresented lambda light chain in systemic light chain amyloidosis patients. Biochim Biophys Acta Mol Basis Dis. 2018;1864:2294–2303. doi: 10.1016/j.bbadis.2017.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Souillac P.O., Uversky V.N., Millett I.S. Elucidation of the molecular mechanism during the early events in immunoglobulin light chain amyloid fibrillation. Evidence for an off-pathway oligomer at acidic pH. J Biol Chem. 2002;277:12666–12679. doi: 10.1074/jbc.M109229200. [DOI] [PubMed] [Google Scholar]
- 97.Buell A.K., Dobson C.M., Knowles T.P. The physical chemistry of the amyloid phenomenon:thermodynamics and kinetics of filamentous protein aggregation. Essays Biochem. 2014;56:11–39. doi: 10.1042/bse0560011. [DOI] [PubMed] [Google Scholar]
- 98.Morgan G.J., Kelly J.W. The kinetic stability of a full-length antibody light chain dimer determines whether endoproteolysis can release amyloidogenic variable domains. J Mol Biol. 2016;428:4280–4297. doi: 10.1016/j.jmb.2016.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Deret S., Chomilier J., Huang D.B. Molecular modeling of immunoglobulin light chains implicates hydrophobic residues in non-amyloid light chain deposition disease. Protein Eng. 1997;10:1191–1197. doi: 10.1093/protein/10.10.1191. [DOI] [PubMed] [Google Scholar]
- 100.Khurana R., Gillespie J.R., Talapatra A. Partially folded intermediates as critical precursors of light chain amyloid fibrils and amorphous aggregates. Biochemistry. 2001;40:3525–3535. doi: 10.1021/bi001782b. [DOI] [PubMed] [Google Scholar]
- 101.Wall J.S., Gupta V., Wilkerson M. Structural basis of light chain amyloidogenicity: comparison of the thermodynamic properties, fibrillogenic potential and tertiary structural features of four Vlambda6 proteins. J Mol Recognit. 2004;17:323–331. doi: 10.1002/jmr.681. [DOI] [PubMed] [Google Scholar]
- 102.Kim Y.S., Cape S.P., Chi E. Counteracting effects of renal solutes on amyloid fibril formation by immunoglobulin light chains. J Biol Chem. 2001;276:1626–1633. doi: 10.1074/jbc.M007766200. [DOI] [PubMed] [Google Scholar]
- 103.Blancas-Mejia L.M., Tellez L.A., del Pozo-Yauner L. Thermodynamic and kinetic characterization of a germ line human lambda6 light-chain protein:the relation between unfolding and fibrillogenesis. J Mol Biol. 2009;386:1153–1166. doi: 10.1016/j.jmb.2008.12.069. [DOI] [PubMed] [Google Scholar]
- 104.Wall J., Schell M., Murphy C. Thermodynamic instability of human lambda 6 light chains:correlation with fibrillogenicity. Biochemistry. 1999;38:14101–14108. doi: 10.1021/bi991131j. [DOI] [PubMed] [Google Scholar]
- 105.Ramirez-Alvarado M. Amyloid formation in light chain amyloidosis. Curr Top Med Chem. 2012;12:2523–2533. doi: 10.2174/1568026611212220007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kim Y.S., Randolph T.W., Stevens F.J. Kinetics and energetics of assembly, nucleation, and growth of aggregates and fibrils for an amyloidogenic protein. Insights into transition states from pressure, temperature, and co-solute studies. J Biol Chem. 2002;277:27240–27246. doi: 10.1074/jbc.M202492200. [DOI] [PubMed] [Google Scholar]
- 107.Simpson E.R., Herold E.M., Buchner J. The folding pathway of the antibody V(L) domain. J Mol Biol. 2009;392:1326–1338. doi: 10.1016/j.jmb.2009.07.075. [DOI] [PubMed] [Google Scholar]
- 108.Rennella E., Cutuil T., Schanda P. Real-time NMR characterization of structure and dynamics in a transiently populated protein folding intermediate. J Am Chem Soc. 2012;134:8066–8069. doi: 10.1021/ja302598j. [DOI] [PubMed] [Google Scholar]
- 109.Jahn T.R., Parker M.J., Homans S.W. Amyloid formation under physiological conditions proceeds via a native-like folding intermediate. Nat Struct Mol Biol. 2006;13:195–201. doi: 10.1038/nsmb1058. [DOI] [PubMed] [Google Scholar]
- 110.Neudecker P., Robustelli P., Cavalli A. Structure of an intermediate state in protein folding and aggregation. Science. 2012;336:362–366. doi: 10.1126/science.1214203. [DOI] [PubMed] [Google Scholar]
- 111.Hu D., Qin Z., Xue B. Effect of methionine oxidation on the structural properties, conformational stability, and aggregation of immunoglobulin light chain LEN. Biochemistry. 2008;47:8665–8677. doi: 10.1021/bi800806d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Pelaez-Aguilar A.E., Valdes-Garcia G., French-Pacheco L. Site-specific interactions with copper promote amyloid fibril formation for lambda6aJL2-R24G. ACS Omega. 2020;5:7085–7095. doi: 10.1021/acsomega.9b03220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.McLaughlin R.W., De Stigter J.K., Sikkink L.A. The effects of sodium sulfate, glycosaminoglycans, and Congo red on the structure, stability, and amyloid formation of an immunoglobulin light-chain protein. Protein Sci. 2006;15:1710–1722. doi: 10.1110/ps.051997606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Esteras-Chopo A., Serrano L., Lopez de la Paz M. The amyloid stretch hypothesis: recruiting proteins toward the dark side. Proc Natl Acad Sci U S A. 2005;102:16672–16677. doi: 10.1073/pnas.0505905102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Rennella E., Morgan G.J., Yan N. The role of protein thermodynamics and primary structure in fibrillogenesis of variable domains from immunoglobulin light chains. J Am Chem Soc. 2019;141:13562–13571. doi: 10.1021/jacs.9b05499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Honda R., Kuwata K. Evidence for a central role of PrP helix 2 in the nucleation of amyloid fibrils. FASEB J. 2018;32:3641–3652. doi: 10.1096/fj.201701183RR. [DOI] [PubMed] [Google Scholar]
- 117.Townsend D., Hughes E., Hussain R. Heparin and methionine oxidation promote the formation of apolipoprotein A-I amyloid comprising alpha-helical and beta-sheet structures. Biochemistry. 2017;56:1632–1644. doi: 10.1021/acs.biochem.6b01120. [DOI] [PubMed] [Google Scholar]
- 118.Chen Y.C. Impact of a discordant helix on beta-amyloid structure, aggregation ability and toxicity. Eur Biophys J. 2017;46:681–687. doi: 10.1007/s00249-017-1235-5. [DOI] [PubMed] [Google Scholar]
- 119.Lo C.J., Wang C.C., Huang H.B. The Arctic mutation accelerates Abeta aggregation in SDS through reducing the helical propensity of residues 15–25. Amyloid. 2015;22:8–18. doi: 10.3109/13506129.2014.980943. [DOI] [PubMed] [Google Scholar]
- 120.Anandh U., Patrick A., Sharma A. Coexistent amyloid fibrils in a patient with combined light chain deposition disease and light chain cast nephropathy. Indian J Nephrol. 2019;29:204–206. doi: 10.4103/ijn.IJN_36_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Weiss B.M., Wong S.W., Comenzo R.L. Beyond the plasma cell:emerging therapies for immunoglobulin light chain amyloidosis. Blood. 2016;127:2275–2280. doi: 10.1182/blood-2015-11-681650. [DOI] [PubMed] [Google Scholar]
- 122.Keeling J., Herrera G.A. The mesangium as target for glomerulopathic light and heavy chains: pathogenic considerations in light and heavy chain-mediated glomerular damage. In: Herrera G.A., editor. Vol. 153. S Karger AG; Basel, Switzerland: 2007. pp. 116–134. (The Kidney in Plasma Cell Dyscrasias). [DOI] [PubMed] [Google Scholar]
- 123.Fogo A.B. The potential for regression of renal scarring. Curr Opin Nephrol Hypertens. 2003;12:223–225. doi: 10.1097/00041552-200305000-00001. [DOI] [PubMed] [Google Scholar]
- 124.Herrera G.A., Russell W.J., Isaac J. Glomerulopathic light chain-mesangial cell interactions modulate in vitro extracellular matrix remodeling and reproduce mesangiopathic findings documented in vivo. Ultrastruct Pathol. 1999;23:107–126. [PubMed] [Google Scholar]
- 125.Teng J., Russell W.J., Gu X. Different types of glomerulopathic light chains interact with mesangial cells using a common receptor but exhibit different intracellular trafficking patterns. Lab Invest. 2004;84:440–451. doi: 10.1038/labinvest.3700069. [DOI] [PubMed] [Google Scholar]
- 126.Markowitz G.S. Dysproteinemia and the kidney. Adv Anat Pathol. 2004;11:49–63. doi: 10.1097/00125480-200401000-00005. [DOI] [PubMed] [Google Scholar]
- 127.Wang Q., Jiang F., Xu G. The pathogenesis of renal injury and treatment in light chain deposition disease. J Transl Med. 2019;17:387. doi: 10.1186/s12967-019-02147-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Jimenez-Zepeda V.H. Light chain deposition disease: novel biological insights and treatment advances. Int J Lab Hematol. 2012;34:347–355. doi: 10.1111/j.1751-553X.2012.01419.x. [DOI] [PubMed] [Google Scholar]
- 129.Varga C., Titus S.E., Toskic D. Use of novel therapies in the treatment of light chain amyloidosis. Blood Rev. 2019;37:100581. doi: 10.1016/j.blre.2019.05.005. [DOI] [PubMed] [Google Scholar]
- 130.Wanchoo R., Abudayyeh A., Doshi M. Renal toxicities of novel agents used for treatment of multiple myeloma. Clin J Am Soc Nephrol. 2017;12:176–189. doi: 10.2215/CJN.06100616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Schreiner G.F. The mesangial phagocyte and its regulation of contractile cell biology. J Am Soc Nephrol. 1992;2:S74–S82. doi: 10.1681/ASN.V210s74. [DOI] [PubMed] [Google Scholar]
- 132.Migliorini A., Ebid R., Scherbaum C.R. The danger control concept in kidney disease: mesangial cells. J Nephrol. 2013;26:437–449. doi: 10.5301/jn.5000247. [DOI] [PubMed] [Google Scholar]
- 133.Herrera G.A. Plasticity of mesangial cells: a basis for understanding pathological alterations. Ultrastruct Pathol. 2006;30:471–479. doi: 10.1080/01913120600932594. [DOI] [PubMed] [Google Scholar]
- 134.Schlondorff D., Banas B. The mesangial cell revisited:no cell is an island. J Am Soc Nephrol. 2009;20:1179–1187. doi: 10.1681/ASN.2008050549. [DOI] [PubMed] [Google Scholar]
- 135.Herrera G.A., Lott R.L. Silver stains in diagnostic renal pathology. J Histotechnol. 1996;19:219–223. [Google Scholar]
- 136.Russell W.J., Cardelli J., Harris E. Monoclonal light chain--mesangial cell interactions:early signaling events and subsequent pathologic effects. Lab Invest. 2001;81:689–703. doi: 10.1038/labinvest.3780278. [DOI] [PubMed] [Google Scholar]
- 137.Herrera G.A., Teng J., Liu X. Mesenchymal stem cells in mesangial repair in a model of immunoglobulin light-chain mediated mesangial injury. J Stem Cell Res Ther. 2014;4:13. [Google Scholar]
- 138.Herrera G.A., Teng J. Mesangial amyloid formation in an isolated rat kidney perfusion model: a new model to study renal amyloidogenesis. In: Skinner JLB M., Connors L.H., Seldin D.C., editors. XIth International Symposium on Amyloidosis. 1st ed. CRC Press, Taylor & Francis Group; Boca Rato, FL: 2007. pp. 307–309. [Google Scholar]
- 139.Herrera G.A., Turbat-Herrera E.A., Teng J. Understanding mesangial damage and repair: insights from an experimental model of immunoglobulin light chain-associated mesangiopathy. J Cell Biol Cell Metab. 2014;1:003. [Google Scholar]
- 140.Keeling J., Herrera G.A. Matrix metalloproteinases and mesangial remodeling in light chain-related glomerular damage. Kidney Int. 2005;68:1590–1603. doi: 10.1111/j.1523-1755.2005.00571.x. [DOI] [PubMed] [Google Scholar]
- 141.Iida H., Seifert R., Alpers C.E. Platelet-derived growth factor (PDGF) and PDGF receptor are induced in mesangial proliferative nephritis in the rat. Proc Natl Acad Sci U S A. 1991;88:6560–6564. doi: 10.1073/pnas.88.15.6560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Hong S.W., Isono M., Chen S. Increased glomerular and tubular expression of transforming growth factor-beta1, its type II receptor, and activation of the Smad signaling pathway in the db/db mouse. Am J Pathol. 2001;158:1653–1663. doi: 10.1016/s0002-9440(10)64121-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Liu Z.H., Striker G.E., Stetler-Stevenson M. TNF-alpha and IL-1 alpha induce mannose receptors and apoptosis in glomerular mesangial but not endothelial cells. Am J Physiol. 1996;270:C1595–C1601. doi: 10.1152/ajpcell.1996.270.6.C1595. [DOI] [PubMed] [Google Scholar]
- 144.Gomez-Guerrero C., Duque N., Egido J. Mesangial cells possess an asialoglycoprotein receptor with affinity for human immunoglobulin A. J Am Soc Nephrol. 1998;9:568–576. doi: 10.1681/ASN.V94568. [DOI] [PubMed] [Google Scholar]
- 145.Merkle M., Ribeiro A., Belling F. Response of VEGF to activation of viral receptors and TNFalpha in human mesangial cells. Mol Cell Biochem. 2012;370:151–161. doi: 10.1007/s11010-012-1406-8. [DOI] [PubMed] [Google Scholar]
- 146.Adler S. Integrin matrix receptors in renal injury. Kidney Int Suppl. 1994;45:S86–S89. [PubMed] [Google Scholar]
- 147.Cohen A.W., Hnasko R., Schubert W. Role of caveolae and caveolins in health and disease. Physiol Rev. 2004;84:1341–1379. doi: 10.1152/physrev.00046.2003. [DOI] [PubMed] [Google Scholar]
- 148.Anderson R.G. The caveolae membrane system. Annu Rev Biochem. 1998;67:199–225. doi: 10.1146/annurev.biochem.67.1.199. [DOI] [PubMed] [Google Scholar]
- 149.Thomsen P., Roepstorff K., Stahlhut M. Caveolae are highly immobile plasma membrane microdomains, which are not involved in constitutive endocytic trafficking. Mol Biol Cell. 2002;13:238–250. doi: 10.1091/mbc.01-06-0317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Isaac J., Kerby J.D., Russell W.J. In vitro modulation of AL-amyloid formation by human mesangial cells exposed to amyloidogenic light chains. Amyloid. 1998;5:238–246. doi: 10.3109/13506129809007296. [DOI] [PubMed] [Google Scholar]
- 151.Mukadam A.S., Breusegem S.Y., Seaman M.N.J. Analysis of novel endosome-to-Golgi retrieval genes reveals a role for PLD3 in regulating endosomal protein sorting and amyloid precursor protein processing. Cell Mol Life Sci. 2018;75:2613–2625. doi: 10.1007/s00018-018-2752-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Aridor M., Balch W.E. Perspectives: drug delivery. Regulating export of ER cargo. Science. 2000;287:816–817. doi: 10.1126/science.287.5454.816. [DOI] [PubMed] [Google Scholar]
- 153.Rivera V.M., Wang X., Wardwell S. Regulation of protein secretion through controlled aggregation in the endoplasmic reticulum. Science. 2000;287:826–830. doi: 10.1126/science.287.5454.826. [DOI] [PubMed] [Google Scholar]
- 154.Epstein W.V., Tan M., Wood I.S. Formation of “amyloid” fibrils in vitro by action of human kidney lysosomal enzymes on Bence Jones proteins. J Lab Clin Med. 1974;84:107–110. [PubMed] [Google Scholar]
- 155.Westermark G.T., Fandrich M., Lundmark K. Noncerebral amyloidoses:aspects on seeding, cross-seeding, and transmission. Cold Spring Harb Perspect Med. 2018;8:a024323. doi: 10.1101/cshperspect.a024323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Walker L.C., Bian F., Callahan M.J. Modeling Alzheimer's disease and other proteopathies in vivo: is seeding the key? Amino Acids. 2002;23:87–93. doi: 10.1007/s00726-001-0113-7. [DOI] [PubMed] [Google Scholar]
- 157.Chaudhuri P., Prajapati K.P., Anand B.G. Amyloid cross-seeding raises new dimensions to understanding of amyloidogenesis mechanism. Ageing Res Rev. 2019;56:100937. doi: 10.1016/j.arr.2019.100937. [DOI] [PubMed] [Google Scholar]
- 158.Keeling J., Herrera G.A. Human matrix metalloproteinases: characteristics and pathologic role in altering mesangial homeostasis. Microsc Res Tech. 2008;71:371–379. doi: 10.1002/jemt.20565. [DOI] [PubMed] [Google Scholar]
- 159.Yerbury J.J., Kumita J.R. Protein chemistry of amyloid fibrils and chaperones:implications for amyloid formation and disease. Current Chemical Biology. 2010;4:10. [Google Scholar]
- 160.Alexandrescu A.T. Amyloid accomplices and enforcers. Protein Sci. 2005;14:1–12. doi: 10.1110/ps.04887005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Dergunov A.D. Role of ApoE in conformation-prone diseases and atherosclerosis. Biochemistry (Mosc) 2006;71:707–712. doi: 10.1134/s0006297906070029. [DOI] [PubMed] [Google Scholar]
- 162.Tan Y., Zhao M.H. Complement in glomerular diseases. Nephrology (Carlton) 2018;23(Suppl 4):11–15. doi: 10.1111/nep.13461. [DOI] [PubMed] [Google Scholar]
- 163.Jokiranta T.S., Solomon A., Pangburn M.K. Nephritogenic lambda light chain dimer:a unique human miniautoantibody against complement factor H. J Immunol. 1999;163:4590–4596. [PubMed] [Google Scholar]
- 164.Schurder J., Rafat C., Vigneron C. Complement-dependent, monoclonal gammapathy-associated thrombotic microangiopathy. Kidney Int. 2017;92:516. doi: 10.1016/j.kint.2017.04.039. [DOI] [PubMed] [Google Scholar]
- 165.Herrera G.A., Teng J., Shen X., Turbat-Herrera E.A. Sortilin-related receptor (SORL-1) mediates glomerulopathic light chain interactions with mesangial cells. Proceedings of the American Society Nephrology Meeting, San Diego, CA. J Am Soc Nephrol. 2018;29:16. [Google Scholar]
- 166.Urmoneit B., Prikulis I., Wihl G. Cerebrovascular smooth muscle cells internalize Alzheimer amyloid beta protein via a lipoprotein pathway:implications for cerebral amyloid angiopathy. Lab Invest. 1997;77:157–166. [PubMed] [Google Scholar]
- 167.Meshitsuka S., Shingaki S., Hotta M. Phase 2 trial of daily, oral epigallocatechin gallate in patients with light-chain amyloidosis. Int J Hematol. 2017;105:295–308. doi: 10.1007/s12185-016-2112-1. [DOI] [PubMed] [Google Scholar]
- 168.Pelaez-Aguilar A.E., Rivillas-Acevedo L., French-Pacheco L. Inhibition of Light Chain 6aJL2-R24G amyloid fiber formation associated with light chain amyloidosis. Biochemistry. 2015;54:4978–4986. doi: 10.1021/acs.biochem.5b00288. [DOI] [PubMed] [Google Scholar]
- 169.Herrera G.A., Turbat-Herrera E., Teng J. Vol. 99. Lab Invest; National Harbor, MD: 2019. Epigallocatechin-3-gallate inhibits renal light chain amyloidogenesis by two different mechanisms [abstract] p. 15. (USCAP 2019). [Google Scholar]
- 170.Hutagalung A.H., Novick P.J. Role of Rab GTPases in membrane traffic and cell physiology. Physiol Rev. 2011;91:119–149. doi: 10.1152/physrev.00059.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Naslavsky N., Caplan S. The enigmatic endosome - sorting the ins and outs of endocytic trafficking. J Cell Sci. 2018;131:jcs216499. doi: 10.1242/jcs.216499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Jarrett J.T., Lansbury P.T., Jr. Amyloid fibril formation requires a chemically discriminating nucleation event:studies of an amyloidogenic sequence from the bacterial protein OsmB. Biochemistry. 1992;31:12345–12352. doi: 10.1021/bi00164a008. [DOI] [PubMed] [Google Scholar]
- 173.Morozova-Roche L.A., Zurdo J., Spencer A. Amyloid fibril formation and seeding by wild-type human lysozyme and its disease-related mutational variants. J Struct Biol. 2000;130:339–351. doi: 10.1006/jsbi.2000.4264. [DOI] [PubMed] [Google Scholar]
- 174.Klimtchuk E.S., Gursky O., Patel R.S. The critical role of the constant region in thermal stability and aggregation of amyloidogenic immunoglobulin light chain. Biochemistry. 2010;49:9848–9857. doi: 10.1021/bi101351c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Blancas-Mejia L.M., Horn T.J., Marin-Argany M. Thermodynamic and fibril formation studies of full length immunoglobulin light chain AL-09 and its germline protein using scan rate dependent thermal unfolding. Biophys Chem. 2015;207:13–20. doi: 10.1016/j.bpc.2015.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Tan M., Epstein W. Polymer formation during the degradation of human light chain and Bence-Jones proteins by an extrct of the lysosomal fraction of normal human kidney. Immunochemistry. 1972;9:9–16. doi: 10.1016/0019-2791(72)90278-9. [DOI] [PubMed] [Google Scholar]
- 177.Vora M., Kevil C.G., Herrera G.A. Contribution of human smooth muscle cells to amyloid angiopathy in AL (light-chain) amyloidosis. Ultrastruct Pathol. 2017;41:358–368. doi: 10.1080/01913123.2017.1349852. [DOI] [PubMed] [Google Scholar]
- 178.Little M.H. Regrow or repair: potential regenerative therapies for the kidney. J Am Soc Nephrol. 2006;17:2390–2401. doi: 10.1681/ASN.2006030218. [DOI] [PubMed] [Google Scholar]
- 179.Rookmaaker M.B., Smits A.M., Tolboom H. Bone-marrow-derived cells contribute to glomerular endothelial repair in experimental glomerulonephritis. Am J Pathol. 2003;163:553–562. doi: 10.1016/S0002-9440(10)63683-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Chou Y.H., Pan S.Y., Yang C.H. Stem cells and kidney regeneration. J Formos Med Assoc. 2014;113:201–209. doi: 10.1016/j.jfma.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 181.Herrera G.A., Zeng C., Turbat-Herrera E.A. Healing the damaged mesangium in nodular glomerulosclerosis using mesenchymal stem cells (MSCs):Expectations and challenges. Ultrastruct Pathol. 2016;40:61–70. doi: 10.3109/01913123.2016.1145776. [DOI] [PubMed] [Google Scholar]
- 182.Romagnani P., Humphreys B.D. Report on ISN Forefronts, Florence, Italy, 12–15 September. 2013: Stem cells and kidney regeneration. Kidney Int. 2014;86:23–27. doi: 10.1038/ki.2014.32. [DOI] [PubMed] [Google Scholar]
- 183.Herrera G.A., Teng J., Zeng C. Phenotypic plasticity of mesenchymal stem cells is crucial for mesangial repair in a model of immunoglobulin light chain-associated mesangial damage. Ultrastruct Pathol. 2018;42:262–288. doi: 10.1080/01913123.2018.1449772. [DOI] [PubMed] [Google Scholar]
- 184.Wong C.Y., Cheong S.K., Mok P.L. Differentiation of human mesenchymal stem cells into mesangial cells in post-glomerular injury murine model. Pathology. 2008;40:52–57. doi: 10.1080/00313020701716367. [DOI] [PubMed] [Google Scholar]
- 185.Wong C.Y., Tan E.L., Cheong S.K. In vitro differentiation of mesenchymal stem cells into mesangial cells when co-cultured with injured mesangial cells. Cell Biol Int. 2014;38:497–501. doi: 10.1002/cbin.10231. [DOI] [PubMed] [Google Scholar]
- 186.Pippin J.W., Sparks M.A., Glenn S.T. Cells of renin lineage are progenitors of podocytes and parietal epithelial cells in experimental glomerular disease. Am J Pathol. 2013;183:542–557. doi: 10.1016/j.ajpath.2013.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Shankland S.J., Pippin J.W., Duffield J.S. Progenitor cells and podocyte regeneration. Semin Nephrol. 2014;34:418–428. doi: 10.1016/j.semnephrol.2014.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Lin C.S., Xin Z.C., Dai J. Commonly used mesenchymal stem cell markers and tracking labels:Limitations and challenges. Histol Histopathol. 2013;28:1109–1116. doi: 10.14670/hh-28.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Sagrinati C., Ronconi E., Lazzeri E. Stem-cell approaches for kidney repair:choosing the right cells. Trends Mol Med. 2008;14:277–285. doi: 10.1016/j.molmed.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 190.Thakkar U.G., Vanikar A.V., Trivedi H.L. Stem cell therapy: an emerging modality in glomerular diseases. Cytotherapy. 2017;19:333–348. doi: 10.1016/j.jcyt.2016.11.003. [DOI] [PubMed] [Google Scholar]
- 191.Yuan Y., Pei J., Lai L. LigBuilder 2: a practical de novo drug design approach. J Chem Inf Model. 2011;51:1083–1091. doi: 10.1021/ci100350u. [DOI] [PubMed] [Google Scholar]
- 192.Richards D.B., Cookson L.M., Barton S.V. Repeat doses of antibody to serum amyloid P component clear amyloid deposits in patients with systemic amyloidosis. Sci Transl Med. 2018;10 doi: 10.1126/scitranslmed.aan3128. [DOI] [PubMed] [Google Scholar]
- 193.Solomon A., Weiss D.T., Wall J.S. Therapeutic potential of chimeric amyloid-reactive monoclonal antibody 11–1F4. Clin Cancer Res. 2003;9:3831S–3838S. [PubMed] [Google Scholar]
- 194.Wall J.S., Kennel S.J., Richey T. Generation and characterization of anti-AA amyloid-specific monoclonal antibodies. Front Immunol. 2011;2:32. doi: 10.3389/fimmu.2011.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Renz M., Torres R., Dolan P.J. 2A4 binds soluble and insoluble light chain aggregates from AL amyloidosis patients and promotes clearance of amyloid deposits by phagocytosis (dagger) Amyloid. 2016;23:168–177. doi: 10.1080/13506129.2016.1205974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Wall J.S., Kennel S.J., Williams A. AL amyloid imaging and therapy with a monoclonal antibody to a cryptic epitope on amyloid fibrils. PLoS One. 2012;7 doi: 10.1371/journal.pone.0052686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Feng X., Zeng C., Teng J. Established human mesenchymal stem cell lines stably secreting matrix metallopeptidase 7 to treat glomerulosclerosis in light chain deposition disease. Proceedings of the Kidney Week 2019, Washington DC. J Am Soc Nephrol. 2019;30:912. [Google Scholar]
- 198.Herrera G.A., Turbat-Herrera E.A., Teng J. Epigallocatechin-3-gallate inhibits renal light chain amyloidogenesis by two different mechanisms. Proceedings of the USCAP 108th Annual Meeting, 2019, National Harbor, MD. Modern Pathol. 2019;32(suppl 2):15. [Google Scholar]
- 199.Joshi S., Evangelisti A., Liao R. Harnessing cardiac regeneration as a potential therapeutic strategy for AL cardiac amyloidosis. Curr Cardiol Rep. 2020;22:1. doi: 10.1007/s11886-020-1252-3. [DOI] [PubMed] [Google Scholar]
- 200.Herrera G.A., Teng J., Turbat-Herrera E.A. Renal amyloidosis:current views on pathogenesis and impact on diagnosis. Contrib Nephrol. 2011;169:232–246. doi: 10.1159/000314795. [DOI] [PubMed] [Google Scholar]
- 201.von Gise H., Christ H., Bohle A. Early glomerular lesions in amyloidosis. Electronmicroscopic findings. Virchows Arch A Pathol Anat Histol. 1981;390:259–272. doi: 10.1007/BF00496558. [DOI] [PubMed] [Google Scholar]
- 202.Eirin A., Lerman L.O. Mesenchymal stem cell treatment for chronic renal failure. Stem Cell Res Ther. 2014;5:83. doi: 10.1186/scrt472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Truong L.D., Pindur J., Barrios R. Tenascin is an important component of the glomerular extracellular matrix in normal and pathologic conditions. Kidney Int. 1994;45:201–210. doi: 10.1038/ki.1994.24. [DOI] [PubMed] [Google Scholar]
- 204.Blanpain C., Daley G.Q., Hochedlinger K. Stem cells assessed. Nat Rev Mol Cell Biol. 2012;13:471–476. doi: 10.1038/nrm3371. [DOI] [PubMed] [Google Scholar]
- 205.Lasagni L., Romagnani P. Glomerular epithelial stem cells: the good, the bad, and the ugly. J Am Soc Nephrol. 2010;21:1612–1619. doi: 10.1681/ASN.2010010048. [DOI] [PubMed] [Google Scholar]
- 206.Nagaishi K., Mizue Y., Chikenji T. Mesenchymal stem cell therapy ameliorates diabetic nephropathy via the paracrine effect of renal trophic factors including exosomes. Sci Rep. 2016;6:34842. doi: 10.1038/srep34842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Nagy A., Quaggin S.E. Stem cell therapy for the kidney: a cautionary tale. J Am Soc Nephrol. 2010;21:1070–1072. doi: 10.1681/ASN.2010050559. [DOI] [PubMed] [Google Scholar]
- 208.Shi Y., Wang Y., Li Q. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol. 2018;14:493–507. doi: 10.1038/s41581-018-0023-5. [DOI] [PubMed] [Google Scholar]
- 209.Kunter U., Rong S., Djuric Z. Transplanted mesenchymal stem cells accelerate glomerular healing in experimental glomerulonephritis. J Am Soc Nephrol. 2006;17:2202–2212. doi: 10.1681/ASN.2005080815. [DOI] [PubMed] [Google Scholar]
- 210.Reilly G.C., Engler A.J. Intrinsic extracellular matrix properties regulate stem cell differentiation. J Biomech. 2010;43:55–62. doi: 10.1016/j.jbiomech.2009.09.009. [DOI] [PubMed] [Google Scholar]
- 211.Park S.H., Park S.H., Kook M.C. Ultrastructure of human embryonic stem cells and spontaneous and retinoic acid-induced differentiating cells. Ultrastruct Pathol. 2004;28:229–238. doi: 10.1080/01913120490515595. [DOI] [PubMed] [Google Scholar]
- 212.Akbik D., Ghadiri M., Chrzanowski W. Curcumin as a wound healing agent. Life Sci. 2014;116:1–7. doi: 10.1016/j.lfs.2014.08.016. [DOI] [PubMed] [Google Scholar]
- 213.Hsu Y.C., Chen M.J., Yu Y.M. Suppression of TGF-beta1/SMAD pathway and extracellular matrix production in primary keloid fibroblasts by curcuminoids:its potential therapeutic use in the chemoprevention of keloid. Arch Dermatol Res. 2010;302:717–724. doi: 10.1007/s00403-010-1075-y. [DOI] [PubMed] [Google Scholar]
- 214.Tan T.W., Tsai H.R., Lu H.F. Curcumin-induced cell cycle arrest and apoptosis in human acute promyelocytic leukemia HL-60 cells via MMP changes and caspase-3 activation. Anticancer Res. 2006;26:4361–4371. [PubMed] [Google Scholar]
- 215.Thangapazham R.L., Sharad S., Maheshwari R.K. Skin regenerative potentials of curcumin. Biofactors. 2013;39:141–149. doi: 10.1002/biof.1078. [DOI] [PubMed] [Google Scholar]
- 216.Liu C.P., Hu Y., Lin J.C. Targeting strategies for drug delivery to the kidney: from renal glomeruli to tubules. Med Res Rev. 2019;39:561–578. doi: 10.1002/med.21532. [DOI] [PubMed] [Google Scholar]