Abstract
Liver involvement in sickle cell disease (SCD) is often referred as sickle cell hepatopathy (SCH) and is a complication of SCD which may be associated with significant mortality. This manuscript is based on a round table workshop between paediatric and adult hepatologists and haematologists and review of the literature. The discussion was prompted by the lack of substantial data and guidance in managing these sometimes very challenging cases. This review provides a structured approach for the diagnosis and management of SCH in children and young adults. The term SCH describes any hepatobiliary dysfunction in the context of SCD. Diagnosis and management of biliary complications, acute hepatic crisis, acute hepatic sequestration and other manifestations of SCH are discussed as well as the role of liver transplantation and haemopoietic stem cell transplantation in the management of SCH.
Introduction
Mortality from sickle cell disease (SCD) has been decreasing in childhood, but has increased for young adults of 20-24 years of age.1 Mortality from liver-related complications in SCD is about 7% of all SCD deaths.1 In this review the term sickle cell hepatopathy (SCH) describes any hepatobiliary dysfunction in SCD. SCH has an estimated incidence of 10-40% in patients with SCD.2,3 To address the clinical need for a structured approach of diagnosing and managing SCH in children and young adults, a multidisciplinary expert workshop was held at King’s College Hospital, London in 2018. The group comprised of specialists from the three UK paediatric liver centres and one centre from France, adult hepatologists, hepatobiliary surgeons and haematologists who are actively involved in the care and research of this condition.
Clinical presentation and diagnosis of SCH
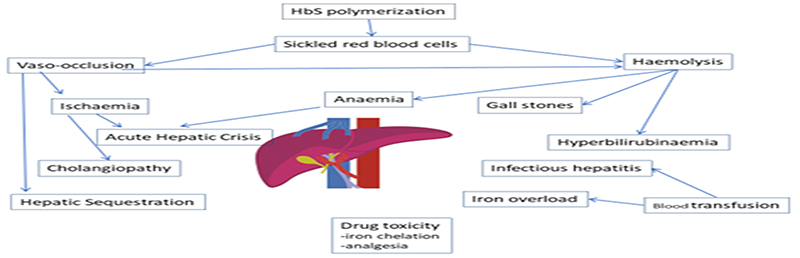
The polymerization of deoxygenated HbS is the origin of all the complications of SCD. This increases the rigidity of the red cell with consequent damage to the vascular endothelium and vaso-occlusion, resulting in a cascade of pathological processes including infarction, haemolysis, inflammation, hypercoagulability and oxidative stress. All these contribute to liver complications seen in SCD (Figure 1 and Table 1).
Figure 1. Pathophysiology of sickle cell hepatopathy.
The polymerization of deoxygenated HbS is the origin of all the complications of SCD. It increases the rigidity of the red cell with consequent damage to the vascular endothelium and vaso-occlusion, resulting in a cascade of pathological processes, including infarction, haemolysis, inflammation, hypercoagulability and oxidative stress. HbS: haemoglobin S
Permission has been obtained from the illustrator Dr Vassiliki Tzomaka to include the above illustration in the figure of this article.
Table 1. Clinical presentation, diagnosis and management of sickle cell hepatopathy.
| Sickle Cell Hepatopathy | |||||
|---|---|---|---|---|---|
| Asymptomatic or Symptoms and Signs of biliary obstruction | Abdominal pain and acute hepatomegaly with jaundice | Jaundice +/-Symptoms and signs of Chronic Liver Disease | |||
| Raised Total/Conjugated Bilirubin, ALT and γ-GT | |||||
| High Ferritin | Biliary Abnormalities on Ultrasound | No Biliary Abnormalities on Ultrasound | Chronic Liver Disease workup | ||
| Liver MRI +/- Liver Biopsy | MRCP/ERCP for intervention | ↓Hb > 20 g/L | Steady Hb +/- ↑INR | Liver biopsy | |
| Iron overload | Cholangiopathy +/- dominant stricture | Biliary sludge, gall-stones +/- biliary obstruction | Acute Hepatic Sequestration | Acute Hepatic Crisis +/- Liver Failure | AILD Wilson disease HBV HCV Drug toxicity |
| Management | |||||
| Iron chelators | Ursodeoxycholic acid +/- dilatation or stent of stricture | ERCP Laparoscopic cholecystectomy Laparotomy | Fluid resuscitation Blood transfusion Exchange transfusion (if ↑LFTs, ↑INR or conjugated bilirubin >50 μmol/L) | IV hydration Blood transfusion HDU/PICU Exchange Transfusion Renal replacement therapy Antibiotics | As per individual diagnosis |
ALT: alanine aminotransferase, γ-GT: gamma-glutamyl transferase, MRI: magnetic resonance imaging, MRCP: magnetic resonance cholangiopancreatography, ERCP: Endoscopic Retrograde Cholangiopancreaticography, Hb: haemoglobin, AILD: autoimmune liver disease, HBV: hepatitis B virus, HCV: hepatitis C virus
SCH varies from isolated conjugated hyperbilirubinaemia to liver failure. The main clinical presentations of SCH are discussed below.
Biliary complications
The most common liver pathologies associated with SCD are biliary complications ranging from biliary sludge and gallstones to cholangiopathy and biliary cirrhosis. Increased haemolysis is mainly responsible for hyperbilirubinemia and gallstones. Gallstones are diagnosed on screening or during episodes of acute biliary obstruction. Their prevalence, depending on the population studied, varies from very rare to up to 15%.4–6 Typical signs of acute cholecystitis are uncommon. More common is intermittent, poorly localized pain of unclear aetiology. Stones and debris leaving the gallbladder may be asymptomatic or cause obstruction leading to abdominal pain, exacerbation of jaundice, pale stools and possibly cholangitis or pancreatitis.
Sickling and occlusion of the end arteries supplying the biliary tree and lack of collateral circulation leads to progressive ischaemic injury of the biliary tree7 resulting in cholangiopathy (prevalence 0.8%)3 (Figure 2 and Figure 3).
Figure 2. Sickling in end arteries of bile ducts.
The diagram on the left illustrates the sickling in the end arteries perfusing the bile ducts. The picture on the right is an MRCP of a child with sickle cholangiopathy showing intrahepatic cholangiopathy with dilatations and strictures but no dominant stricture. MRCP: magnetic resonance cholangiopancreatography
Figure 3. Biopsy changes in sickle cell hepatopathy.
Liver biopsy from an 11-year-old showing sinusoidal dilatation (long arrow) and porto-portal bridging fibrosis (short arrows). Sickle cells are visible within the sinusoids in this biopsy from a 12-year-old (B, main image). In acute hepatic sequestration, large numbers of sickled cells can be seen within congested sinusoids (B, lower inset). Iron overload can be seen within cells of the reticuloendothelial system (upper inset) and hepatocytes (long arrow). A liver biopsy from a 17-year-old demonstrating features of autoimmune liver disease in the form of a porto-lobular hepatitis with plasma cell rich interface activity (C, main image). The same child demonstrated cholangiopathic features in the form of concentric periductal fibrosis (C, lower inset) and periductal lymphoplasmacytic inflammation around portal areas (C, upper inset). Cholangiopathic features in the form of duct loss attributed to ischaemic damage demonstrated in panel D. Duct destruction was a prominent feature of this biopsy from a 7-year-old with sickle cell disease and autoimmune liver disease. The upper inset demonstrates a destructive cholangitis affecting interlobular calibre bile ducts (D, upper inset, H&E x200 magnification) and the lower inset demonstrates plasma cell rich inflammation surrounding septal calibre bile ducts (D, lower inset).
Unconjugated hyperbilirubinaemia in SCD is common, but conjugated hyperbilirubinaemia should be investigated for acute obstruction secondary to gallstones, inspissated bile or a dominant biliary stricture. The initial investigation for all biliary pathology is US examination of the liver. Biliary pathology on US should prompt a Magnetic Resonance Cholangiopancreatography which may indicate the need for Endoscopic Retrograde Cholangiopancreaticography (ERCP).
Acute hepatic crisis
Acute sickling of the erythrocytes in the hepatic sinusoids, anaemia and sinusoidal obstruction by the engorged Kupffer cells leading to ischaemia is the underlying mechanism8,9 of an acute hepatic crisis (also known as acute intrahepatic sickling). This is diagnosed by the association of acute hepatomegaly and pain, increased conjugated hyperbilirubinaemia, variably elevated liver enzymes, with or without liver synthetic failure (defined as prolonged INR >1·5 unresponsive to intravenous vitamin K), and without an obvious biliary obstruction or infection. We include what is known as “sickle cell intrahepatic cholestasis” in the severe presentation of acute hepatic crisis. Sometimes the liver failure presents as part of multiorgan failure.9
The disease severity is independent of HbS level and can be classified as follows:
-
-Mild presentation:
-
○bilirubin less than 80 μmol/L, ≤50% conjugated; (or a bilirubin 2× the steady state)
-
○no liver failure (normal INR).
-
○
-
-Moderate presentation:
-
○bilirubin 80-150 μmol/L, ≥50% conjugated, elevated transaminases, without liver failure; (or a bilirubin 3-5× the steady state)
-
○no liver failure
-
○
-
-Severe presentation:
-
○bilirubin over 150 μmol/L, ≥50% conjugated, elevated transaminases, without liver failure; (or a bilirubin more than 5× the steady state)
-
○elevated conjugated bilirubin (more than 50 μmol/L), with liver failure
-
○
Acute hepatic sequestration
Sequestration of large numbers of sickled erythrocytes in the hepatic vasculature is the underlying mechanism in acute hepatic sequestration (Figure 3). Sometimes it is precipitated by acute infections or exposure to hepatotoxic drugs8,10 and in these cases the patients are cholestatic. It is diagnosed with clinical features of rapid liver enlargement and tenderness with an acute drop of more than 20 g/L in haemoglobin that may be life-threatening.9,11 Liver function may be normal. The proportion of HbS is not predictive of the severity.
Iron overload
Patients with SCD may develop iron overload as a consequence of frequent requirement of blood transfusion (Figure 3). Hepatic iron overload contributes to about 10% of patient deaths.12
Autoimmune liver disease (AILD)
Abnormal liver function tests (LFTs) should be investigated comprehensively to exclude other liver conditions, particularly AILD. In a series of 77 children, 17% had AILD (Figure 3) and 8% viral hepatitis.13 AILD is diagnosed on positive autoantibodies (ANA, SMA, LKM1) and compatible histopathology. About 50% of these children also have a cholangiopathy (“overlap syndrome”), sometimes with positive ANCA antibodies and/or the presence of colitis (prevalence 0.03-0.05%).3,13
Infections and other causes of liver disease
Children with SCD are at risk of acquiring viral hepatitis. They are also at increased risk of bacterial and other infections, due to splenic hypofunction and other immune impairments. Drug induced hepatitis is another consideration.
Monitoring of children with SCD for hepatopathy
Routine monitoring for liver disease is advised at least yearly in SCD patients (Table 2 and Table 3). If serum ALT >2× upper limit of normal (ULN) and/or γ-GT 1·5-2× ULN they should be investigated from a liver disease perspective. Abnormal serum AST (which also reflects rate of haemolysis) and to a lesser degree ALT is described in more than half of asymptomatic patients, but not higher than >2 ULN. Alkaline phosphatase is generally not a good marker of hepatobiliary disease in children given its variation with growth14, whereas γ-GT is more sensitive and specific.15
Table 2. Overview of investigations of liver dysfunction in SCD.
| Overview of Investigations of Liver Dysfunction in SCD |
|---|
Asymptomatic:
|
Signs of biliary obstruction (jaundice, abdominal pain, pale stool, fever):
|
Signs of acute hepatomegaly, abdominal pain +/- jaundice
|
Jaundice with or without symptoms and signs of chronic liver disease
|
If chronic liver disease work up negative and LFTs remain abnormal
|
SCD: sickle cell disease; ALT: alanine aminotransferase; γ-GT: gamma-glutamyl transferase; MRI: magnetic resonance imaging; MRCP: magnetic resonance cholangiopancreatography; ERCP: Endoscopic Retrograde cholangio-pancreaticography; ANA: anti-nuclear antibodies; SMA: smooth muscle antibody; LKM1: liver kidney microsomal antibody; ANCA: anti-neutrophil cytoplasm antibody; KF: Kayser-Fleischer; EBV: Epstein-Barr virus; LFTs: liver function tests.
Table 3. Indications for referral to hepatologist.
| Indications for referral to hepatologist |
|---|
| (Symptomatic) gallstones |
| ALT >2xULN (on two occasions) |
| γ-GT >2x ULN (on two occasions) |
| Increased conjugated bilirubin > 2x known steady state level |
Co-existing or suspecting additional liver disease like
|
| Liver failure (INR >1·5 unresponsive to intravenous vitamin K with abnormal LFTs) |
ALT: alanine aminotransferase, ULN: upper limit of normal, γ-GT: gamma-glutamyl transferase, NAFLD: non-alcoholic fatty liver disease, INR: international normalized ratio, LFTs: liver function tests
Abdominal US should be performed yearly after 5 years of age5,6. Serum ferritin should be monitored annually in children with a history of transfusion and monthly in case of chronic transfusion. Liver MRI is recommended annually for evaluating iron burden in patients receiving more than 10 transfusions per year16 or with a ferritin >1000μg/L.
Liver biopsy in patients with SCD is indicated for the investigation of persistently abnormal LFTs in order to rule out coexisting pathology.13 It should only be performed when the patient is stable to avoid serious complications.17 Patients need admission the night before for preparation and discussion with haematology about performing an exchange transfusion.
Management of sickle cell hepatopathy
Management of biliary complications
Most biliary complications are managed conservatively at the acute presentation with intravenous fluids, analgesia and appropriate antibiotics. Choledocholithiasis is the presenting feature in <10% of children with SCD. The ideal therapeutic option is an initial ERCP +/- sphincterotomy, followed by elective laparoscopic cholecystectomy +/- on-table cholangiogram.19,20 Laparoscopic cholecystectomy for asymptomatic gallstones is controversial. In some countries, cholecystectomy is performed on detection whilst in others the decision is deferred until the young patient can consent.
Cholangiopathy can be helped by ursodeoxycholic acid. It may require dilatation or stenting of dominant strictures by ERCP. Cholangiopathy may progress to biliary cirrhosis and end stage liver disease (ESLD) requiring liver transplant(LT).3
Management of acute hepatic crisis
Usually with acute hepatic crisis, haemoglobin is stable and there is no sequestration. The levels of HbS are not predictive of the severity. Treatment should not be delayed regardless of HbS levels. In mild cases, adequate hydration, usually intravenously is enough. Moderate cases require a simple blood transfusion and daily monitoring of the LFTs until they normalize. Severe cases require:
-
-
care in a high dependency/intensive care unit.
-
-
an urgent exchange transfusion: this can be repeated if there is relapse of liver failure after initial improvement regardless of HbS level.
-
-
twice daily monitoring of LFTs until normalization.
-
-
renal replacement therapy as required.
-
-
broad spectrum antibiotics.
Management of acute hepatic sequestration
The patient may present with symptoms of hypovolaemia and will require emergency support with intravenous fluids. A top-up transfusion will be required to increase the haemoglobin to its steady-state level. If there is liver dysfunction with prolonged INR, or a conjugated bilirubin higher than 50 μmol/L an exchange transfusion is required regardless of HbS%.3,9
Pharmacotherapy for SCH
No medications are proven to improve cholestasis in SCH. Ursodeoxycholic acid does not dissolve pigment gallstones of SCD, but may help in cholangiopathy. Hydroxyurea (hydroxycarbamide) is the only drug approved for the treatment of SCD in Europe21 and in the USA it is recommended for all children. Hydroxyurea induces HbF synthesis, which directly reduces the rate of HbS polymerization22 and therefore reduces the frequency of complications related to it.23,24 Hydroxyurea is metabolized by the liver, but there is no evidence that it causes hepatic damage. No trials explicitly assess its efficacy or safety in SCH, but several studies have shown incidental protection against liver disease, with patients on hydroxyurea less likely to die from liver failure.24–26
Management of iron overload
Deferasirox (DFX) and desferrioxamine (DFO) are licensed iron chelators in SCD.27 DFO is usually given as a subcutaneous or intravenous infusion in heavily iron-loaded patients. DFX is an orally active iron chelator. LFTs should be checked prior to the initiation of treatment, every two weeks during the first month and monthly thereafter. DFX may contribute to hepatic dysfunction, but the most common cause of liver dysfunction in these patients is the iron itself. For persistent and progressive increase in serum transaminase levels that cannot be attributed to other causes, DFX should be interrupted. Once the levels have normalised, cautious re-initiation of treatment at a lower dose followed by gradual dose escalation may be considered. DFX is not recommended in patients with severe hepatic impairment.
Management of other causes of liver disease
Management of the other causes of liver diseases is as in patients without SCD. For AILD remission is achievable with prednisolone with or without azathioprine or mycophenolate mofetil.13 Ursodeoxycholic acid is added in cholangiopathy.
Haemopoietic stem cell transplantation
HSCT from human leucocyte antigen (HLA) identical siblings can be curative therapy in children with SCD with greater than 95% 5-year overall survival rates.28 Lack of HLA-identical siblings who are clinically unaffected poses a significant barrier in the uptake of this mode of therapy. This led to the development of HSCT programmes using alternative donor sources such as matched unrelated29 or haploidentical donors. Outcomes with these programmes have a higher incidence of serious complications and are reserved only for severely affected children.
In children with AILD or cholangiopathy, HSCT should be discussed early, before the development of advanced liver disease precludes the conditioning necessary for HSCT. HSCT should stop the progression of liver disease. Other potentially curative techniques currently under development are gene therapy30 and gene editing.
Liver transplant
Patients with SCD coming to LT either have co-existent liver disease unrelated to the SCD or have SCH. SCH is a rare and not standard indication for LT. Predicting which patients with SCH will progress to ESLD, understanding whether there are interventions which may stabilise the liver disease and identifying the appropriate time to assess them for LT can be difficult.
The published literature of LT in SCH is limited: 19 cases have been reported in total; 13 adults and 6 children.3,31–34 In the early experiences, significant morbidity and mortality (one year post transplant mortality over 50%)31,35 was related to non-liver, but sickle-related end organ damage. Mortality has been substantial in those transplanted in the context of an acute hepatic crisis. These results have been improved upon with better patient selection i.e. transplanting patients with ESLD who are relatively well and stable and with improved haematologic management in the peri-transplant period.
Of the 4 children who underwent LT3,33 more recently, all survived for more than 4 years. One died 6 years following transplantation from a subdural haematoma. No episodes of graft vascular thrombosis occurred. The fourth child reported3 in spite of aggressive exchange transfusions suffered sickle crises, seizures and rejection. The course of SCD does not appear to be modified by LT, and SCH may recur in the graft.
LT is potential treatment for ESLD secondary to SCH, particularly severe progressive cholangiopathy. However isolated liver transplant still leaves the risk of recurrence of cholangiopathy hence sequential HSCT appears to be an attractive option once the patient has recovered from liver transplant surgery. Sequential liver transplant and HSCT has been successfully performed for primary immune deficiency related liver disease.36
Epilogue
With new and evolving treatments for SCD, the prevalence of SCH is likely to decrease. In the meantime, management of SCH lacks evidence base. Clinical trials looking into the usefulness of early cholecystectomy and into the role of hydroxyurea in the prevention of hepatopathy as an endpoint are required. LT should be considered for very selected patients and in centres expert at managing LT and SCD. HSCT with an available suitable donor would be curative for SCD and avoid the risk of recurrence of SCH in the donor liver. This could be an option while evolving therapies become reality.
Search strategy and selection criteria
References for this review were identified through searches of PubMed with the search terms “sickle cell hepatopathy”, “acute hepatic crisis”, “acute hepatic sequestration”, “sickle cell liver disease” and “children”, without a time constraint. References were also identified from authors’ own files. Only papers written in English and in French were reviewed. The final reference list was generated based on originality and relevance to the specific scope of this Review.
What is known about the topic
Liver involvement in sickle cell disease may be associated with significant mortality
Mortality from sickle cell disease in young adults has been increasing
Management of sickle cell hepatopathy requires multidisciplinary and specialist management
What this study adds
A structured approach for the diagnosis of sickle cell hepatopathy
Guidance in the management of sickle cell hepatopathy
Indications for referral of a child with sickle cell disease to a hepatologist
Acknowledgements
Vassiliki Tzomaka
MowatLabs staff
Role of funding source
The workshop was supported by the British Heart Foundation.
Footnotes
Contributors’ Statement
Eirini Kyrana, David Rees, Florence Lacaille, Emer Fitzpatrick, Nigel Heaton, Sue Height, Marianne Samyn, Fulvio Mavilio, Valentine Brousse, Abid Suddle, Subarna Chakaravorty, Emma Drasar and Anil Dhawan: Participated in the round table discussions and presentations and contributed to the writing and editing of the manuscript. Mark Davenport, Anita Verma, Maesha Deheragoda and Maria Sellars contributed in the writing and editing of the manuscript. Girish Gupte, Mark Velangi, Baba Inusa, Nedim Hadzic, Tassos Grammatikopoulos and Jonathan Hind participated in the round table discussions and presentations and reviewed the manuscript.
Conflict of interest statements
Eirini Kyrana, Florence Lacaille, Emer Fitzpatrick, Mark Davenport, Nigel Heaton, Sue Height, Marianne Samyn, Fulvio Mavilio, Valentine Brousse, Abid Suddle, Anita Verma, Mark Velangi, Baba Inusa, Emma Drasar, Nedim Hadzic, Tassos Grammatikopoulos, Jonathan Hind, Maesha Deheragoda, Maria Sellars and Anil Dhawan have nothing to disclose.
David Rees is on advisory boards of Novartis and DSMB of AstraZeneca outside the submitted work. Girish Gupte reports personal fees from Alexion Pharmaceuticals outside the submitted work. Subarna Chakravorty reports personal fees from Novartis outside the submitted work. Valentine Brousse reports personal fees from Bluebirdbio, personal fees from Addmedica, outside the submitted work.
Ethics Committee approval
Not applicable
Contributor Information
Eirini Kyrana, Children’s Live Unit, Leeds General Infirmary, Leeds, UK.
David Rees, King’s College London, Department of Haematological Medicine, King’s College Hospital, Denmark Hill, London.
Florence Lacaille, Gastroenterology-Hepatology-Nutrition Unit, Hôpital Universitaire Necker-Enfants Malades, 149 rue de Sèvres, 75015 Paris.
Emer Fitzpatrick, Institute of Liver Studies, King’s College London, Denmark Hill, London, UK.
Mark Davenport, Department of Paediatric Surgery, King’s College Hospital, Denmark Hill, London SE5 9RS, UK.
Nigel Heaton, Hepatobiliary and Pancreatic Surgery/Liver Transplantation, King’s College Hospital NHS Trust, Denmark Hill, SE9 5RS, London, UK.
Sue Height, Paediatric Haematology, King’s College Hospital NHS Trust, London.
Marianne Samyn, Paediatric Liver, GI and Nutrition Centre, King’s College Hospital NHS Trust, London, UK.
Fulvio Mavilio, Department of Life Sciences, University of Modena and Reggio Emilia Via Campi, 287; 41125 Modena – Italy.
Valentine Brousse, Service de Pédiatrie Générale et Maladies Infectieuses, Hôpital Necker-Enfants Malades, 149 rue de Sèvres, 75015 Paris.
Abid Suddle, Institute of Liver Studies, King’s College Hospital NHS Trust, Denmark Hill, SE9 5RS, London, UK.
Subarna Chakravorty, Paediatric Haematology, King’s College Hospital NHS Trust, Denmark Hill, London, UK.
Anita Verma, Institute of Liver Studies, Kings College Hospital, Denmark Hill, SE9 5RS, London UK.
Girish Gupte, Liver Unit (including small bowel transplantation), Birmingham Women’s and Children’s Hospital, Steelhouse Lane, Birmingham B4 6NH.
Mark Velangi, Department of Haematology, Birmingham Women’s and Children’s Hospital, Steelhouse Lane, Birmingham B4 6NH.
Baba Inusa, Children's sickle cell and thalassaemia centre at Evelina London Children’s Hospital, St Thomas’ Hospital, Westminster Bridge Road, London SE1 7EH.
Emma Drasar, Department of Clinical Haematology, University College London Hospitals, 250 Euston Roads Bloomsbury, London NW1 2PG.
Nedim Hadzic, Paediatric Centre for Hepatology, Gastroenterology and Nutrition, King's College Hospital, Denmark Hill, London SE5 9RS, UK.
Tassos Grammatikopoulos, Paediatric Liver, GI & Nutrition Centre and MowatLabs King's College Hospital NHS Foundation Trust, London and Institute of Liver Studies, King’s College London, Denmark Hill, London, SE5 9RS.
Jonathan Hind, Paediatric Liver, GI and Nutrition Centre, King’s College Hospital NHS Trust, London, UK.
Maesha Deheragoda, Liver Histopathology Laboratory, Institute of Liver Studies, King’s College Hospital, London, UK.
Maria Sellars, Department of Radiology, Kings College Hospital, Denmark Hill, London, UK.
References
- 1.Hamideh D, Alvarez O. Sickle cell disease related mortality in the United States (1999-2009) Pediatr Blood Cancer. 2013;60(9):1482–6. doi: 10.1002/pbc.24557. [DOI] [PubMed] [Google Scholar]
- 2.Shah R, Taborda C, Chawla S. Acute and chronic hepatobiliary manifestations of sickle cell disease: A review. World J Gastrointest Pathophysiol. 2017;8(3):108–16. doi: 10.4291/wjgp.v8.i3.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Allali S, de Montalembert M, Brousse V, et al. Hepatobiliary Complications in Children with Sickle Cell Disease: A Retrospective Review of Medical Records from 616 Patients. J Clin Med. 2019;8(9) doi: 10.3390/jcm8091481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alkindi SY, Pathare A, Al Zadjali S, et al. Serum Total Bilirubin, not Cholelithiasis, is Influenced by UGT1A1 Polymorphism, Alpha Thalassemia and beta(s) Haplotype: First Report on Comparison between Arab-Indian and African beta(s) Genes. Mediterr J Hematol Infect Dis. 2015;7(1):e2015060. doi: 10.4084/MJHID.2015.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Walker TM, Hambleton IR, Serjeant GR. Gallstones in sickle cell disease: observations from The Jamaican Cohort study. J Pediatr. 2000;136(1):80–5. doi: 10.1016/s0022-3476(00)90054-4. [DOI] [PubMed] [Google Scholar]
- 6.McCarville MB, Luo Z, Huang X, et al. Abdominal ultrasound with scintigraphic and clinical correlates in infants with sickle cell anemia: baseline data from the BABY HUG trial. AJR Am J Roentgenol. 2011;196(6):1399–404. doi: 10.2214/AJR.10.4664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahmed M, Dick M, Mieli-Vergani G, Harrison P, Karani J, Dhawan A. Ischaemic cholangiopathy and sickle cell disease. Eur J Pediatr. 2006;165(2):112–3. doi: 10.1007/s00431-005-0005-z. [DOI] [PubMed] [Google Scholar]
- 8.Berry PA, Cross TJ, Thein SL, et al. Hepatic dysfunction in sickle cell disease: a new system of classification based on global assessment. Clin Gastroenterol Hepatol. 2007;5(12):1469–76. doi: 10.1016/j.cgh.2007.08.009. quiz 369. [DOI] [PubMed] [Google Scholar]
- 9.Lacaille F, Lesage F, de Montalembert M. Acute hepatic crisis in children with sickle cell disease. J Pediatr Gastroenterol Nutr. 2004;39(2):200–2. doi: 10.1097/00005176-200408000-00015. [DOI] [PubMed] [Google Scholar]
- 10.Pecker LH, Patel N, Creary S, et al. Diverse manifestations of acute sickle cell hepatopathy in pediatric patients with sickle cell disease: A case series. Pediatr Blood Cancer. 2018;65(8):e27060. doi: 10.1002/pbc.27060. [DOI] [PubMed] [Google Scholar]
- 11.Hatton CS, Bunch C, Weatherall DJ. Hepatic sequestration in sickle cell anaemia. Br Med J (Clin Res Ed) 1985;290(6470):744–5. doi: 10.1136/bmj.290.6470.744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Feld JJ, Kato GJ, Koh C, et al. Liver injury is associated with mortality in sickle cell disease. Aliment Pharmacol Ther. 2015;42(7):912–21. doi: 10.1111/apt.13347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jitraruch S, Fitzpatrick E, Deheragoda M, et al. Autoimmune Liver Disease in Children with Sickle Cell Disease. J Pediatr. 2017;189:79–85 e2. doi: 10.1016/j.jpeds.2017.06.035. [DOI] [PubMed] [Google Scholar]
- 14.Brody JI, Ryan WN, Haidar MA. Serum alkaline phosphatase isoenzymes in sickle cell anemia. JAMA. 1975;232(7):738–41. [PubMed] [Google Scholar]
- 15.Richard S, Billett HH. Liver function tests in sickle cell disease. Clin Lab Haematol. 2002;24(1):21–7. doi: 10.1046/j.1365-2257.2002.0378a.x. [DOI] [PubMed] [Google Scholar]
- 16.Tsitsikas DA, Nzouakou R, Ameen V, Sirigireddy B, Amos RJ. Comparison of serial serum ferritin measurements and liver iron concentration assessed by MRI in adult transfused patients with sickle cell disease. Eur J Haematol. 2014;92(2):164–7. doi: 10.1111/ejh.12230. [DOI] [PubMed] [Google Scholar]
- 17.Zakaria N, Knisely A, Portmann B, et al. Acute sickle cell hepatopathy represents a potential contraindication for percutaneous liver biopsy. Blood. 2003;101(1):101–3. doi: 10.1182/blood-2002-06-1823. [DOI] [PubMed] [Google Scholar]
- 18.Drasar E, Fitzpatrick E, Gardner K, et al. Interim assessment of liver damage in patients with sickle cell disease using new non-invasive techniques. Br J Haematol. 2017;176(4):643–50. doi: 10.1111/bjh.14462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Davenport M, Howard ER, Green DW. Minimally invasive surgery in sickle cell disease. BMJ. 1991;303(6796):249. doi: 10.1136/bmj.303.6796.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Al-Salem AH, Issa H. Laparoscopic cholecystectomy in children with sickle cell anemia and the role of ERCP. Surg Laparosc Endosc Percutan Tech. 2012;22(2):139–42. doi: 10.1097/SLE.0b013e3182471b1c. [DOI] [PubMed] [Google Scholar]
- 21.Qureshi A, Kaya B, Pancham S, et al. Guidelines for the use of hydroxycarbamide in children and adults with sickle cell disease: A British Society for Haematology Guideline. Br J Haematol. 2018;181(4):460–75. doi: 10.1111/bjh.15235. [DOI] [PubMed] [Google Scholar]
- 22.Rees DC. The rationale for using hydroxycarbamide in the treatment of sickle cell disease. Haematologica. 2011;96(4):488–91. doi: 10.3324/haematol.2011.041988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ware RE, Davis BR, Schultz WH, et al. Hydroxycarbamide versus chronic transfusion for maintenance of transcranial doppler flow velocities in children with sickle cell anaemia-TCD With Transfusions Changing to Hydroxyurea (TWiTCH): a multicentre, open-label, phase 3, non-inferiority trial. Lancet. 2016;387(10019):661–70. doi: 10.1016/S0140-6736(15)01041-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Steinberg MH, McCarthy WF, Castro O, et al. The risks and benefits of long-term use of hydroxyurea in sickle cell anemia: A 17.5 year follow-up. Am J Hematol. 2010;85(6):403–8. doi: 10.1002/ajh.21699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Steinberg MH, Barton F, Castro O, et al. Effect of hydroxyurea on mortality and morbidity in adult sickle cell anemia: risks and benefits up to 9 years of treatment. JAMA. 2003;289(13):1645–51. doi: 10.1001/jama.289.13.1645. [DOI] [PubMed] [Google Scholar]
- 26.Voskaridou E, Christoulas D, Bilalis A, et al. The effect of prolonged administration of hydroxyurea on morbidity and mortality in adult patients with sickle cell syndromes: results of a 17-year, single-center trial (LaSHS) Blood. 2010;115(12):2354–63. doi: 10.1182/blood-2009-05-221333. [DOI] [PubMed] [Google Scholar]
- 27.Vichinsky E, Onyekwere O, Porter J, et al. A randomised comparison of deferasirox versus deferoxamine for the treatment of transfusional iron overload in sickle cell disease. British journal of haematology. 2007;136(3):501–8. doi: 10.1111/j.1365-2141.2006.06455.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bernaudin F, Dalle JH, Bories D, et al. Long-term event-free survival, chimerism and fertility outcomes in 234 patients with sickle-cell anemia younger than 30 years after myeloablative conditioning and matched-sibling transplantation in France. Haematologica. 2020;105(1):91–101. doi: 10.3324/haematol.2018.213207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shenoy S, Eapen M, Panepinto JA, et al. A trial of unrelated donor marrow transplantation for children with severe sickle cell disease. Blood. 2016;128(21):2561–7. doi: 10.1182/blood-2016-05-715870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ribeil JA, Hacein-Bey-Abina S, Payen E, et al. Gene Therapy in a Patient with Sickle Cell Disease. N Engl J Med. 2017;376(9):848–55. doi: 10.1056/NEJMoa1609677. [DOI] [PubMed] [Google Scholar]
- 31.Kindscher JD, Laurin J, Delcore R, Forster J. Liver transplantation in a patient with sickle cell anemia. Transplantation. 1995;60(7):762–4. doi: 10.1097/00007890-199510150-00026. [DOI] [PubMed] [Google Scholar]
- 32.Baichi MM, Arifuddin RM, Mantry PS, Bozorgzadeh A, Ryan C. Liver transplantation in sickle cell anemia: a case of acute sickle cell intrahepatic cholestasis and a case of sclerosing cholangitis. Transplantation. 2005;80(11):1630–2. doi: 10.1097/01.tp.0000184446.52454.69. [DOI] [PubMed] [Google Scholar]
- 33.Mekeel KL, Langham MR, Jr, Gonzalez-Peralta R, Fujita S, Hemming AW. Liver transplantation in children with sickle-cell disease. Liver Transpl. 2007;13(4):505–8. doi: 10.1002/lt.20999. [DOI] [PubMed] [Google Scholar]
- 34.Hurtova M, Bachir D, Lee K, et al. Transplantation for liver failure in patients with sickle cell disease: challenging but feasible. Liver Transpl. 2011;17(4):381–92. doi: 10.1002/lt.22257. [DOI] [PubMed] [Google Scholar]
- 35.Lang T, Berquist WE, So SK, et al. Liver transplantation in a child with sickle cell anemia. Transplantation. 1995;59(10):1490–2. doi: 10.1097/00007890-199505270-00025. [DOI] [PubMed] [Google Scholar]
- 36.Hadzic N, Pagliuca A, Rela M, et al. Correction of the hyper-IgM syndrome after liver and bone marrow transplantation. The New England journal of medicine. 2000;342(5):320–4. doi: 10.1056/NEJM200002033420504. [DOI] [PubMed] [Google Scholar]