Abstract
Background
Genetically engineered T cells are powerful anti-cancer treatments but are limited by safety and specificity issues. We herein describe a MHC-unrestricted modular platform combining autologous T cells, transduced with a targetable synthetic agonistic receptor (SAR), with bispecific antibodies (BiAb) that specifically recruit and activate T cells for tumor killing.
Methods
BiAbs of different formats were generated by recombinant expression. T cells were retrovirally transduced with synthetic agonistic receptors. T cell activation, proliferation, differentiation and T cell induced-lysis were characterized in three murine and human tumor models in vitro and in vivo.
Results
Murine T cells transduced with SAR composed of an extracellular domain EGFRvIII fused to CD28 and CD3ζ signaling domains could be specifically recruited towards murine tumor cells expressing EpCAM by anti-EGFRvIII x anti-EpCAM BiAb. BiAb induced selective antigen-dependent activation, proliferation of SAR-T cells and redirected tumor cell lysis. Selectivity was dependent on the monovalency of the antibody for EGFRvIII. We identified FAS ligand as a major mediator of killing utilized by the T cells. Similarly, human SAR-T cells could be specifically redirected towards mesothelin-expressing human pancreatic cancer cells. In vivo, treatment with SAR-T cells and BiAb mediated anti-tumoral activity in three human pancreatic cancer cell xenograft models. Importantly, SAR-activity unlike CAR-activity was reversible in vitro and in vivo.
Conclusions
We describe a novel ACT platform with anti-tumor activity in murine and human tumor models with a distinct mode of action that combines adoptive T cell therapy with bispecific antibodies.
Introduction
Therapeutic use of genetically engineered T cells has been pioneered in hematological indications (1,2). Chimeric antigen receptor (CAR)- based adoptive T cell therapy (ACT) is currently the main clinically used avenue (3). CAR-transduced T cells specific for CD19 have proven effective against treatment-refractory B cell-associated malignancies (4–6). CAR-T cell therapy can be accompanied by cytokine release syndrome and other toxicities originating from excessive T cell activation (7) which frequently require intensive care and limit their applicability (8).
We have recently shown that combining ACT with a bispecific antibody (BiAb) targeting a tumor-associated antigen and a marker antigen unique to the tumor-specific T cells results in enhanced anti-tumor efficacy (9). An advantage of this strategy would be the dependence on a BiAb with a defined half-life. Potential toxicity would vanish with antibody clearance and efficacy could be sustained through repeated dosing of the antibody.
We now hypothesized that a synthetic agonistic receptor (SAR) constituted of an extracellular domain – itself inert and not present on any naturally occurring T cell – fused to intracellular T cell activating domains could be specifically triggered by a BiAb. Triggering would be conditional on binding of the second specificity to a tumor-associated antigen on the tumor cell. This strategy has the advantage that T cell activation can occur only in the presence of the BiAb and only in the vicinity of the antibody-targeted tumor cell. We generated two proof-of-concept synthetic proteins for murine and human studies together with the respective BiAb. Both receptors bear the advantage of comprising as the extracellular domain, an antigen not present on normal cells and restricted to particular tumor entities such as glioblastoma (10).
We herein describe a novel platform for adoptive T-cell therapy combining advantages of CAR T cells and antibody-based therapies with the potential for clinical translation (Supplementary Fig. 1A).
Material and Methods
Statistical analysis
The FACS data was analyzed with FlowJo V10.3 software. Statistical analysis was performed by using GraphPad Prism software 7.0. Differences between experimental conditions were analyzed using the unpaired two-tailed Student’s t test. In all in vivo experiments, two-way ANOVA with correction for multiple testing by the Bonferroni method was used to analyze differences between the groups. p-values < 0.05 were considered to be significant. All statistical tests are two-sided. Data are shown as mean values ± SEM of a minimum of three biological replicates or independent experiments, as indicated.
Cell Lines
The ovalbumin overexpressing murine pancreatic cancer cell line Panc02-OVA and the murine melanoma cell line B16F10 (11) were stably transduced with the pMXs vector (12) containing the full length murine epithelial cell adhesion molecule (EpCAM, UNIPROT entry Q99JW5) cDNA to generate the EpCAM overexpressing cell lines Panc02-OVA-EpCAM and B16- EpCAM. The hepatocellular carcinoma cell line RIL-175 was kindly provided by Prof. Tim Greten, Center for Cancer Research at the National Cancer Institute (USA), and Nicolas Melin, University of Bern (Switzerland). The hepatocellular carcinoma cell lines Hepa1-6 and Hep3B were kindly provided by Dr. Mike Helms, Sanofi (Germany), while Hep-55.1C was provided by CLS Cell Lines Service (Germany). The murine mammary carcinoma cell line 4T1 (11) has a high endogenous EpCAM expression. The Platinum-E packaging cell line was purchased from Cell Biolabs Inc. (Hoelzel Diagnostika, Cologne, Germany). The MIA PaCa human pancreatic cancer and A375 human melanoma cell lines were purchased from DSMZ (Braunschweig, Germany). The SUIT-2 cell line was previously described (13). The Flp-In-HEK293 human embryonic kidney epithelial cells were purchased from Life Technologies (Carlsbad, USA). Human mesothelin overexpressing cell lines MIA PaCa-MSLN, SUIT-2-MSLN and Flp-In-HEK293-MSLN (HEK-MSLN) were generated by transduction with pMXs (14) containing full length human mesothelin (MSLN, UNIPROT entry Q13421) cDNA. Likewise, the human glioblastoma cell-line U251 (purchased from ATCC) was generated to express full-length human EGFRvIII (Uniprot Entry P00533 AA 1-29, 298-646). The cell line MSTO-mesothelin-luciferase (MSTO-MSLN-LUC) was generated by transducing the MSTO-211H cells with lentiviruses (VSV-G pseudotyped) encoding full-length human MSLN and full-length firefly luciferase (ffLuc) sequentially. The cells were transduced with lentivirus encoding hMSLN. The gene encoding full-length human MSLN was cloned into the MCS of the lentiviral vector obtained from System Bioscience, pCDH-EF1a-MCS-T2A-Puro cDNA. The cells were then selected with puromycin at 5ug/mL for 2 - 3 days. Surviving cells were stained and confirmed with surface expression of hMSLN. The generated MSTO-MSLN cells were then transduced with lentivirus encoding ffLuc. The gene encoding full-length ffLuc was cloned into the MCS of the lentiviral vector obtained from System Bioscience, pCDH-CMV-MCS-EF1a-Neo. The cells were then selected with 5 mg/mL of G418 for 2 - 3 days. The packaging cell line Platinum-A has been previously described (9). All cells were cultured in DMEM with 10% fetal bovine serum (FBS, Life Technologies, USA), 1 % penicillin/streptomycin (PS) and 1 % L-glutamine (all from PAA, Germany). 10 μg/ml puromycin and 1 μg/ml blasticidin selection antibiotics (Sigma, Germany) were added to the Plat-A and Plat-E medium. Primary murine T cells were cultured in RPMI 1640 (Lonza, Basel, Switzerland) containing 10 % FBS, 1 % PS, 1 % L-glutamine, 1 % sodium pyruvate and 1 mM HEPES (T cell medium, TCM). 50 μM β-mercaptoethanol were added to the T cell medium before culturing the cells. The cell lines used in experiments were regularly checked every 4 weeks for mycoplasma species with the commercial testing kit MycoAlert (Lonza). All human cell lines were short tandem repeat-profiled in house to verify their origin. Cells were used for a time period no longer than 2 months.
Generation of T cell activating fusion constructs
The constructs were generated by overlap extension PCR and recombinant expression cloning into the retroviral pMP71 vector. The murine EGFRvIII - CD28 - CD3ζ fusion receptor (E3) consists of human EGFRvIII (Uniprot Entry P00533 AA 1-29, 298-646), murine CD28 (Uniprot Entry P31041 AA 151-218) and murine CD3ζ (Uniprot Entry P24161 AA 52164). The Crypto-1 – CD28 – CD3ζ fusion receptor (C3) consists of human cryptic antigen 1 (Uniprot Entry P13385 AA 31-150), murine CD8a (Uniprot Entry P01731 AA 153-196), murine CD28 (AA 178-218) and murine CD3ζ (AA 52-164). The human EGFRvIII – CD28 – CD3ζ fusion receptor (hE3) consists of human EGFRvIII (Uniprot Entry P00533 AA 1-29, 298- 646), human CD28 (Uniprot Entry P10747 AA 153-220) and human CD3ζ (Uniprot Entry P20963 AA 52-164). The control constructs E3del and C3del are lacking the intracellular signaling domains CD28 and CD3ζ.
Construction of 2 + 2 and 2 +1 BiAb
The construction of expression vectors for BiAb was performed by standard recombinant DNA technologies. All antibody chain genes were separately inserted into expression vectors under control of a MPSV or a SV40E hCMV promoter. The plasmids were co-transfected and transiently expressed in HEK293 or CHO cells. The 2 + 2 antibody was based on a muEpCAM-binding murine IgG2a antibody (clone G8.8). Disulfide-stabilized single-chain Fv (ScFv) specific for hEGFRvIII (clone MR1.1) or hCripto-1 (LC020 H3L2D1) were fused with a 3 x G4S connector to the C-terminus of the heavy chain. The 2 + 1 antibody contained two Fabs for muEpCAM or mesothelin (clone SS1) and one Fab for EGFRvIII which was N-terminally fused to one arm of the muEpCAM or mesothelin IgG. In order to obtain high yields of correctly paired molecules the “knobs-into-holes” (KiH) technology was used for heterodimerization. P329G, L234A and L235A (PG LALA) mutations were inserted in CH3 and CH2 domains to prevent binding to FcγRs and C1q (15)(15)(15). To ensure correct pairing of the different chains, the CrossMAbVH-VL technology (anti-EGFRvIII) and charged residues (anti-muEpCAM) were used (16).
Expression, purification and quality control
All antibodies (unless commercially available) were transiently transfected and expressed at Roche Innovation Center Munich or Zurich, where they were purified. The secreted proteins were first purified by protein A affinity chromatography, followed by size exclusion chromatographic steps. The protein concentration of purified protein samples was determined by measuring the optical density (OD) at 280 nm divided by the molar extinction coefficient calculated on the basis of the amino acid sequence. Purity and molecular weight of the molecules after the final purification step were analyzed by CE - SDS gel electrophoresis in the presence or absence of a reducing agent, using the Caliper LabChip GXII system (Caliper Lifescience) according to the manufacturer’s recommendation. The aggregate content of the molecules was analyzed using a TSKgel G3000 SW XL analytical size-exclusion column (Tosoh) in 25 mM K2HPO4, 125 mM NaCl, 200 mM L-arginine monohydrochloride, 0.02 % (w/v) NaN3, pH 6.7 running buffer at 25 °C. All antibodies had a purity of over 95 %.
Animal experiments
Wild type C57BL/6RJ and BALB/cJ mice were purchased from Janvier (St. Bertevin, France) or Charles River (Sulzfeld, Germany). NSG mice (NOD.Cg-Prkdcscid Il2rgtm1WjI/SzJ) were purchased from Charles River (Sulzfeld, Germany). The transgenic mouse strain B6Smn.C3-Fasl<gld>/J was purchased from The Jackson Laboratory (Bar Harbor, USA). Perforin deficient C57BL/6-Prf1tm1Sdz mice were bred at the University of Zurich, Switzerland. The granzyme B knock-out splenocytes were a gift from J. Pardo (Zaragoza, Spain). Suit-2-MSLN and MIA PaCa-MSLN xenograft models were established by subcutaneously injecting 5 x 105 cells into the right flank. MSTO-MSLN-LUC xenograft model was established by subcutaneously injecting 106 cells in a 1 :1 ratio of PBS and Matrigel (Corning life sciences, USA) into the right flank. BiAb and 107 T cells were given intravenously as indicated. For the bispecific antibody depletion, redosing schedule and antibody dosage was carried out as indicated. A total of six BiAb treatments were injected per mouse at 3, 6, 9, 13, 17 and 19 days post tumor injection. Mice were bled 6 h after antibody redosing.
For T cell depletion, C57BL/6RJ mice were injected intravenously with 107 T cells followed by intravenous injection of 1 mg cetuximab each 6 h after T cell transfer. All animal experiments were approved by the local regulatory agency (Regierung von Oberbayern) and adhered to the NIH guide for the care and use of laboratory animals. Endpoints were registered by an observer blinded to the treatment groups as previously defined (9, 12, 17). IVIS® Lumina™ X5 (Perkin Elmer, USA) was used to acquire in vivo images. The Living Image Software 4.7.2 was used for analysis.
Cytotoxicity assays
T cells were incubated with tumor cell lines and BiAb at indicated effector-to-target ratios and concentrations. Transduced T cells were added to the adherent tumor cells and co-cultured as indicated. Lactate dehydrogenase (LDH) levels were measured according to the manufacturer’s protocol (Promega). For impedance-based real-time killing assays the xCELLigence instrument (ACEA Bioscience, USA) was used. The parameter termed Cell Index (CI) is a measure of the relative change in the electrical impedance at a certain frequency (ƒn), to represent the cell status. All the described calculations are based on the RTCA Software version 1.
T cell transduction and expansion
The transduction and expansion of primary human and murine T cells was conducted following previously described protocols (9,12).
Interferon-γ and IL-2 release assays
Human and murine T cell stimulation assays were set up at indicated antibody concentrations and effector-to-target ratios. IFN-γ and IL-2 release was quantified by enzyme-linked immunosorbent assay (ELISA, BD bioscience).
Flow cytometry
Flow cytometry was performed according to previously published protocols (9, 17, 18). For saturation curve analysis, cells were incubated with increasing concentrations (0.01, 0.1, 0.5, 1, 5, 10, 20 or 25 μg/ml) of either the tetra- or trivalent BiAb and stained with the secondary antibody (tetravalent BiAb: Cy2- conjugated affinity pure goat-anti mouse IgG; trivalent BiAb: FITC-conjugated affinity pure goat-anti human IgG, both The Jackson Laboratory).
For phenotype analysis surface staining was performed by anti-mouse CD8a (FITC, clone 53- 6.7, Biolegend), anti-mouse CCR7 (PerCP/Cy5.5, clone 4B12, Biolegend), anti-mouse CD62L (Pacific Blue, clone MEL-14, Biolegend), anti-mouse CD69 (PE, clone H1.2F3, Biolegend), anti-mouse LAG3 (PerCP/Cy5.5, clone C9B7W, Biolegend) anti-mouse PD1 (PE/Cy7, clone 29F.1A12, Biolegend) and anti-human EGFR (APC, clone AY13, Biolegend). Intracellular staining was performed by anti-mouse Ki67 (PE, clone 16A8, Biolegend). For T cell immunophenotyping and tumor infiltrate analysis, surface staining was performed using anti-human CD45 (PacBlue, clone 2D1, Biolegend), anti-human CD3 (AlexaFluor700, clone HIT3a, Biolegend), anti-human CD4 (BV605, clone OKT4, Biolegend), anti-human CD8 (PercP-Cy5.5, clone SK1, Biolegend), and anti-human EGFR (APC, clone AY13, Biolegend). A Fixable Viability Dye (eFluorTM 780, eBioscience) was also included. Cells were washed and resuspended in PBS containing CountBright® absolute counting beads (Life Technologies). Samples were analyzed with a BD FACS Canto II or Fortessa (BD Bioscience, Germany).
Bispecific antibody depletion in vitro assay
T cell and tumor cell co-culture was set up at indicated antibody concentrations and effector-to-target ratios. SAR T cells were co-incubated with BiAb before being added to the tumor cells. For the assays with human cells aMSLN-CD28-CD3ζ CAR T cells were used as a control and co-cultured with tumor cells following the same procedure (no BiAb was added). For the assays with murine cells an aEPCAM-CD28-CD3ζ CAR was used. After 24 h supernatants were collected and T cells transferred onto a new plate with seeded tumor cells. T cells were either re-suspended with medium only or re-dosed with BiAb to control for the depletion. 4 sequential assay depletions were performed. IFN-γ concentration in the supernatants was quantified by ELISA.
Confocal microscopy
E3 T cells were stained with the PKH 26 red, untransduced T cells with PKH 67 green fluorescent cell membrane labeling dye (both Sigma Aldrich) according to the manufacturer s protocol. Labeled cells were mixed in a ratio of 1 to 1, preloaded with 1 μg/ml BiAb and co-incubated with 4T1 tumor cells in a 96-well plate for 8 to 10 h. Upon cluster formation, cells were imaged with a Leica TCS SP5 confocal system (Leica Microsystems, Wetzlar, Germany). A cluster was predefined as a tumor cell surrounded by ≥ 9 T cells.
Results
Impact of BiAb on synthetic agonistic receptor (SAR) T cells
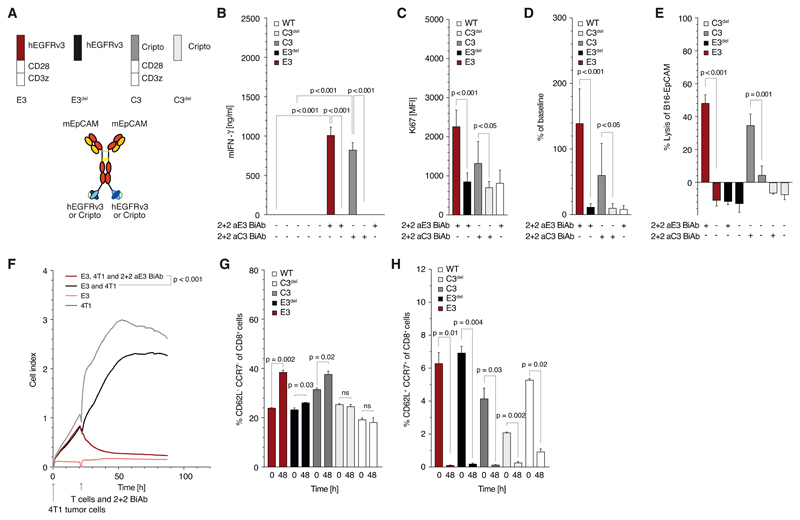
We generated SARs constituted either of the extracellular domains of human EGFRvIII or of human Cripto-1 fused to the murine CD28 and CD3ζ domains (later referred to as E3 and C3, respectively). Proteins devoid of the intracellular domains were created as controls (referred to as E3del and C3del). All constructs could be transduced into murine cytotoxic T cells with comparable efficiencies (Supplementary Fig. 1B, C). Tetravalent 2 + 2 BiAb targeting EGFRvIII or Cripto and murine EpCAM were recombinantly generated (Fig. 1A). We analyzed T cell activation by the BiAb and found a marked and specific activation for both C3 and E3 murine T cells (referred to as C3 or E3 T cells) but not for C3del or E3del T cells measured by IFN-γ release (Fig. 1B).
Figure 1. Synthetic agonistic receptor (SAR) T cells can be triggered by BiAb to induce T cell activation, proliferation and tumor celllysis.
(A) Schematic overview of the constructs used in experiments for panels B to H: tetravalent 2 + 2 anti-EpCAM x anti-EGFRvIII (or anti-Cripto) BiAb and receptor constructs E3, E3del, C3 and C3del (B) Quantification of E3, E3del, C3, C3del and untransduced murine T cell activation when cultured in 96-well plates, coated with the corresponding 2 + 2 BiAb (10 μg/ml). (C, D, G, H) Transduced T cells were co-cultured with Panc02-OVA-EpCAM+ tumor cells and 2 + 2 BiAb (1 μg/ml). The absolute number and the proliferation rate of the T cells was determined by staining cells for intracellular Ki67 before and after co-culture. For phenotypic analysis, T cells were stained for CD8, CCR7 and CD62L surface expression. CD8+CCR7+CD62L+ T cells were defined as central memory T cells and CD8+CCR7-CD62L- T cells as effector memory T cells. (E) Quantification of B16-EpCAM tumor cell lysis when cocultured with E3, E3del, C3 or C3del T cells (E:T 10:1). (F) Real-time lysis of 4T1 tumor cells when cultured with E3 T cells with or without 1 μg/ml of BiAb (E:T 10:1).
All graphs show mean values ± SEM of at least triplicates. Experiments in panels C, D, G and H were performed in duplicates for technical reasons. Each experiment shown is representative of at least three independent experiments. p-values by two-sided unpaired t-test are indicated. For impedance-based cytotoxicity assays the total curve over time was compared. p<0.05 was considered statistically significant.
Activation of C3 or E3 but not of C3del or E3del T cells by BiAb induced mitotic activity in the transduced T cells (Fig. 1C), resulting in enhanced T cell proliferation (Fig. 1D). When co-culturing C3 or E3 T cells with EpCAM+ tumor cells, SAR-transduced but not control-transduced T cells induced lysis of tumor cells (Fig. 1E). In long term assays, E3 T cells mediated complete lysis of 4T1 (Fig. 1F), Panc02-EpCAM+ and B16-EpCAM+ tumor cells (data not shown). Activation of T cells in co-culture with various tumor cells drove T cell differentiation towards an effector memory phenotype (Fig. 1G, H) which was comparable to differentiation seen with a second generation anti-EpCAM CAR (Supplementary Fig. 1D - J). Co-culture with tumor cells resulted in clustering of E3 T cells only in the presence of the BiAb (Supplementary Fig. 2A).
Influence of BiAb valency on antigen-specific T cell activation
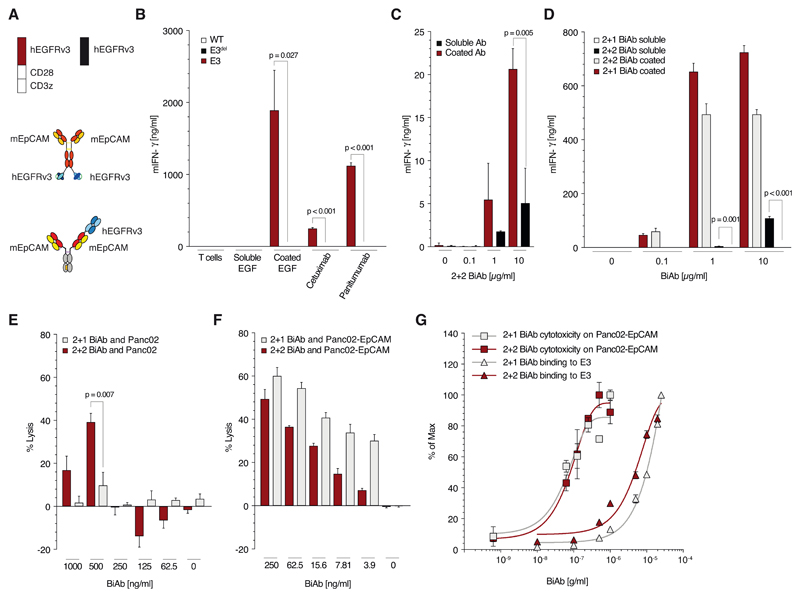
To further analyze the requirements for T cell activation upon SAR T cell engagement, we made use of the natural ligands of EGFR. Soluble EGFR ligands did not trigger activation of the E3 T cells when used under physiological conditions (Fig. 2A, B and Supplementary Fig. 2B, C). Further, following the inhibition of EGFR ligand shedding from the tumor cell surface via the addition of the matrix metalloprotease inhibitor batimastat, SAR T cells were not activated by the upregulated membrane bound ligands compared to the positive control, though basal activation - lower by more than a 100-fold - was observed. (Supplementary Fig. 2D). In contrast, Fc-tagged EGF immobilized to the well induced similar activation to EGFR-binding antibodies immobilized in the same way. This indicates that T cell activation is mediated by crosslinking of the SAR. The BiAb used for the experiments in figure 1 is bivalent for the SAR and may be able to crosslink receptors on its own. Soluble tetravalent 2+2 BiAb was indeed able to trigger T cell activation in the absence of tumor cells (Fig. 2C). We hypothesized that this activation might be due to the bivalency of the BiAb for the SAR. We therefore generated a trivalent heterodimeric 2 + 1 BiAb with only one binding moiety for the agonistic receptor E3 (Fig. 2A). When immobilized, the 2 + 1 BiAb induced similar levels of T cell activation to the 2 + 2 format (Fig. 2D). In contrast, soluble 2 + 1 BiAb failed to induce T cell activation when compared to its tetravalent 2 + 2 format (Fig. 2D). Crosslinking of T cells is therefore abolished when using a trivalent format monovalent for the fusion protein on the T cell. Off-target lysis of EpCAM- tumor cells occurred with the 2 + 2 but not the 2 + 1 BiAb (Fig. 2E). Crucially, on-target lysis of EpCAM+ tumor cells was not altered by the 2 + 1 BiAb format and was comparable for all cell lines tested (Fig. 2F and Supplementary Fig. 2E, F). Similarly, dose response curves were comparable using the 2 + 1 and 2 + 2 BiAb for all cell lines tested (Fig. 2G and Supplementary Fig. 2G, H).
Figure 2. Antibodies monovalent (vs. bivalent) for binding SAR avoid tumor-antigen independent T cell activation.
(A) Schematic overview of the constructs used in panels B to G: trivalent (2+1 format) and a tetravalent (2 + 2 format) anti-EpCAM x anti-EGFRvIII BiAb as well as the constructs E3 and E3del. (B) Quantitative analysis of E3, E3del or untransduced T cell activation when cultured with coated or soluble human EGF or different anti-EGFR antibodies (cetuximab or panitumumab, all at 10μg/ml) for 48 h. (C) Culture of E3 T with either immobilized or soluble 2 + 2 BiAb in increasing concentrations (0, 0.1, 1 or 10 μg/ml). (D) Quantification of E3 T cell activation when cultured with either coated or soluble 2 + 2 BiAb or 2 + 1 BiAb in increasing concentrations (0, 0.1, 1 or 10 μg/ml). (E, F) Co-culture of Panc02-OVA (E) or Panc02-OVA-EpCAM+ (F) tumor cells with E3 T cells (E:T 5:1) preloaded with decreasing amounts (1, 0.5, 0.25, 0.125 or 0.062 μg/ml) of either 2 + 2 BiAb or 2 + 1 BiAb. (G) Saturation curve analysis of E3 T cells for 2 + 2 BiAb and 2 + 1 BiAb BiAb in relation to the lysis curves of Panc02-OVA-EpCAM+ tumor cells.
All graphs show mean values ± SEM of at least triplicates. Cytotoxicity analysis E and F were performed in quadruplicates. Experiments shown are representative of at least three independent experiments. p-values by two-sided unpaired t-test are indicated. p<0.05 was considered statistically significant.
Mechanism underlying tumor cell killing by SAR T cells
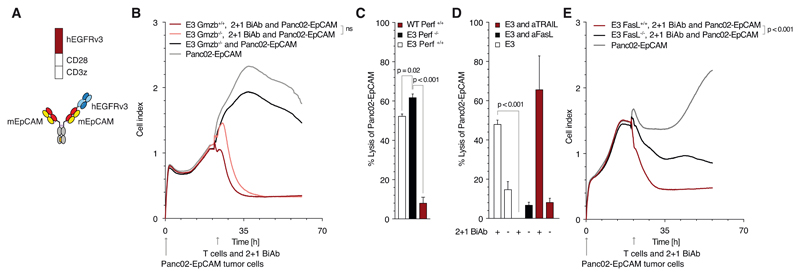
We next investigated the mode of lysis employed by SAR T cells triggered through 2 + 1 BiAb. We found killing by SAR T cells to be independent of granzyme and perforin release (Fig. 3B, C). In contrast, T cells transduced with an EpCAM-specific CAR relied on the release of perforin for efficient killing (Supplementary Fig. 3A). FasL was essential for T cell-mediated lysis in the context of SAR activation for all cell lines tested (Fig. 3D, E and Supplementary Fig. 3B, C). In turn, IFN-γ stimulation of tumor cells resulted in upregulation of FAS expression on all tumor cells (Supplementary Fig. 3D). FasL was, however, dispensable for EpCAM-CAR T cells to efficiently lyse their target (Supplementary Fig. 3E, F, G). As observed for E3 T cells, C3 T cells also killed their target cells in a FasL dependent manner (Supplementary Fig. 3H).
Figure 3. Tumor cell killing by SAR T cells relies on Fas-FasL interactions and is independent of granzyme and perforin release.
(A) Schematic overview of the constructs used in panels B to E: the trivalent antibody and the constructs E3. (B) Real-time Panc02-OVA-EpCAM+ tumor cell killing with either E3 wild-type or granzyme B knock-out murine T cell (E:T 10:1). (C) Quantification of Panc02-OVA-EpCAM+ tumor cell lysis mediated by either E3 wild-type or perforin knock-out murine T cells (E:T 10:1). (D) Impact of T cell-induced tumor cell apoptosis blocking antibodies (anti-CD178 [anti-FasL] or anti CD253 [anti-TRAIL]; each at 10 μg/ml) on Panc02-OVA-EpCAM+ tumor cell lysis by E3 T cells (E:T 10:1). (E) Real-time Panc02-OVA-EpCAM+ tumor cell killing by either E3 wild-type or Fas ligand knock-out murine T cells (E:T 10:1).
All graphs show mean values ± SEM of at least triplicates. Impedance-based cytotoxicity assays were performed in duplicates for technical reasons. Experiments shown are representative of at least three independent experiments. p-values by two-sided unpaired t-test are indicated. For impedance-based cytotoxicity assays the total curve over time was compared. p<0.05 was considered as statistically significant.
The impact and functionality of human SAR T cells combined with BiAb
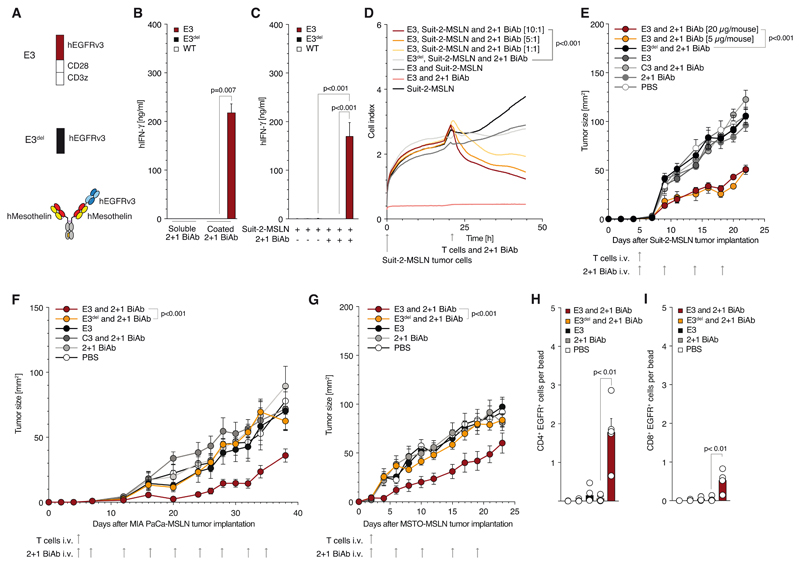
To further assess the impact and functionality of the SAR platform, we generated a fully human version of E3 (Fig. 4A). This construct as well as its deletion construct E3del and the cripto SAR C3 could also be transduced into human T cells with comparable efficiencies (Supplementary Fig. 4A, B). We also generated a 2 + 1 BiAb targeting EGFRvIII with one specificity and the tumor-associated antigen mesothelin (MSLN) with the other specificity (Fig. 4A). Again, plate-bound BiAb but not soluble BiAb activated E3 T cells (Fig. 4B). MSLN-expressing tumor cells were targeted by BiAb and activated E3 but not E3del or untransduced T cells (Fig. 4C). E3 T cells mediated lysis of MSLN-expressing tumor cells (Fig. 4D and Supplementary Fig. 4C, D, E, F). In NSG mice, anti-MSLN CAR T cells were ineffective against a Suit-2-MSLN pancreatic cancer model (Supplementary Fig. 4G). In contrast, E3 T cells plus BiAb mediated anti-tumor responses in three tumor models (Fig. 4E, F, G). In addition, mice where challenged at two tumor sizes (<10 mm2 and <25 mm2), with a therapeutic benefit still being observed when larger tumors were treated (Fig. 4G and Supplementary Fig. 4H). In the MSTO-MSLN-LUC xenograft model, we could observe significant infiltration and persistence of E3 T cells in the treated mice 23 days following adoptive transfer, which was not seen in control groups (Fig. 4 H, I). This tumor T cell infiltrate consisted predominantly of CD4+ SAR T cells, though CD8+ SAR T cells were also present (Supplementary Fig. 4I). In the MSTO-MSLN-LUC cell-line, a Luciferase signal could be used to quantify and show tumor cell persistence at experimental end-point. (Supplementary Fig. 4J). LDH, urea, AST and ALT serum levels remained within the normal range for both E3 T cell plus BiAb and E3del T cell plus BiAb treated mice, showing no systemic parameters of kidney or liver damage in the treated models (Supplementary Fig. 4K, L, M, N).
Figure 4. Anti-human mesothelin x anti-EGFRvIII 2 + 1 BiAb specifically stimulates SAR T cells and mediates tumor killing only in the presence of mesothelin-positive cells.
(A) Schematic overview of the constructs used in experiments for panels B to E: Trivalent BiAb as well as the constructs E3 and E3del. (B) E3, E3del or untransduced T cell activation when cultured with either coated or soluble 2 + 1 BiAb (5 μg/ml). (C) Quantification of E3, E3del and untransduced T cell activation in co-culture with Suit-2-MSLN tumor cell (E:T 10:1). (D) Real-time Suit-2-MSLN tumor cell lysis by E3 or E3del T cells with indicated effector to target ratio. (E) NSG mice inoculated s.c. with Suit-2-MSLN were treated with a single i.v. injection of E3 T cells only (n=5), E3 T cells + 20 μg BiAb (n=10), E3 T cells + 5 μg BiAb (n=5), E3del T cells + 20 μg BiAb (n=5), C3 T cells + 20 μg BiAb (n=5), 20 μg BiAb only (n=5), and PBS (n=5), followed by a total of four BiAb injections as indicated. (F) NSG mice inoculated s.c. with MIA PaCa-MSLN were treated with a single i.v. injection of E3 T cells only (n=6), E3 T cells + 10 μg BiAb (n=10), E3del T cells + 10 μg BiAb (n=6), C3 T cells + 10 μg BiAb (n=6), 10 μg BiAb only (n=6), and PBS (n=6), followed by a total of ten BiAb injections as indicated. (G) NSG mice inoculated s. c. with MSTO-MSLN-LUC were treated with a single i. v. injection of E3 T cells only (n = 5), E3 T cells + 10 μg BiAb (n = 5), 10 μg BiAb only (n = 5) and PBS (n = 5), followed by a total of four BiAb injections as indicated. (H, J) Tumors from the mice in experiment 4G were FACS-analyzed for T cell infiltration. Following a drill down through the CD45 and CD3 gates, the EGFR+ CD4 (H) and CD8 (J) T cell populations were quantified and are depicted as a ratio of cells per bead.
In vitro experiments show mean values ± SEM of at least triplicates and are representative of at least three independent experiments. p-values are based on two-sided unpaired t-test. Impedance-based cytotoxicity assays were performed in duplicates for technical reasons. For impedance-based cytotoxicity assays the total curve over time was compared. In vivo experiments shown are representative of two independent experiments. Analysis of differences between groups for the tumor growth curves were performed using two-way ANOVA with correction for multiple testing by the Bonferroni method. p<0.05 was considered as statistically significant.
Selective activation, applied safety-switches and reversibility of SAR T cells
To ensure no off-target effects or organ damage was being caused by SAR T cells, we cultured pre-activated SAR T cells with hepatocellular carcinoma cell lines, as in vitro models for hepatocytes. Furthermore, with additional dosing of the BiAb, no further SAR T cell activation could be observed (Supplementary Fig. 5A, B). We could also show that glioblastoma cells, expressing EGFRvIII on their surface, were not specifically targeted by SAR T cells (Supplementary Fig. 5C, D) nor could they induce SAR T cell activation (Supplementary Fig. 5E, F).
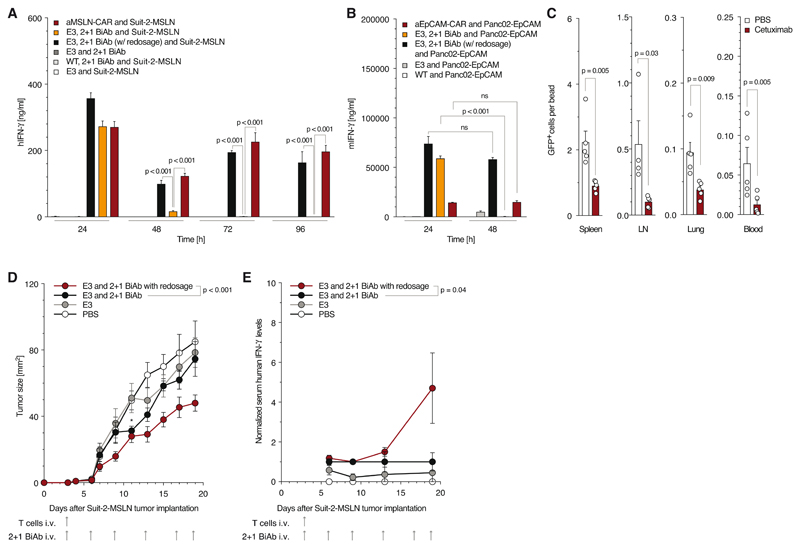
Next, we aimed at demonstrating the intrinsic safety switches of the SAR platform. Activity of SARs unlike CARs is dependent on the presence of the BiAb and should resolve with BiAb elimination. In co-cultures either with human Suit-2-MSLN or murine Panc-EpCAM tumor cells, SAR T cells triggered by the respective BiAb had limited activity over time unlike human anti-MSLN CAR or murine anti-EpCAM CAR T cells. Repeated redosing of BiAb in the assay maintained SAR T cell activity at levels comparable to CAR T cells (Fig. 5A, B). We next compared tumor growth in mice that were redosed with BiAb with mice that received a single dose. Despite transient tumor control (p = 0.026 on day 12), non-redosed mice lost tumor control compared to redosed mice (Fig. 5D). To link these findings to limited T cell activation, mice were bled repeatedly, and serum IFN-γ concentration levels were quantified. We found
Figure 5. E3-MSLN BiAb selectively activates E3 T cells while its depletion results in rapid and complete reversibility of SAR T cell stimulation.
(A, B) For BiAb depletion, Suit-2-MSLN (A) or Panc02-OVA-EpCAM (B) tumor cells were repeatedly co-cultured with E3 T cells with or without readjustment of the BiAb concentration (5 μg/ml). Control condition utilized anti-MSLN-CAR or anti-EpCAM-CAR T cells respectively (E:T 10:1). (C) Analysis of remaining E3-GFP T cells in spleen, lymph node, blood and lung after cetuximab administration. (D, E) NSG mice inoculated s.c. with Suit-2-MSLN (n = 5 / group) were treated with a single i.v. injection of E3 T cells only, E3 T cells + 10 μg BiAb - redosed, E3 T cells + 10 μg BiAb - single dose and PBS, followed by a total of six BiAb injections as indicated. * indicates significance of non-redosed group vs. E3 only at day 12 (p = 0.026) (E). Mice were bled 3, 6, 10 and 16 days after T cell transfer to quantify T cell activation by IFN-γ serum levels (F). Data are reported normalized to the values obtained from the single dose group.
In vitro and in vivo experiments show mean values ± SEM and are representative of at least two independent experiments. p-values are based on two-sided unpaired t-test. Analysis of differences between groups for the tumor growth curves were performed using two-way ANOVA with correction for multiple testing by the Bonferroni method. p<0.05 was considered as statistically significant.
T cell stimulation to be reversible in non-redosed vs. redosed mice as a function of time (Fig. 5E). In summary, we could show that SAR T cell activity is limited to the continued presence of the BiAb, which unlike for CAR T cells could prevent unwanted prolonged T cell activation.
Furthermore, we addressed whether T cells expressing a SAR that can be targeted by the depleting monoclonal antibody cetuximab could be efficiently removed in vivo. Mice transferred with E3 T cells were treated with cetuximab. A single dose of cetuximab efficiently depleted most SAR T cells in the lymph node, blood, lungs and spleen (Fig. 5C). Together, this data demonstrates that SAR T cell action is limited by the half-life of the BiAb and that these cells can be effectively removed from circulation by depleting antibodies.
Discussion
Here, we could demonstrate that BiAb can selectively activate T cells transduced with novel synthetic agonistic receptors (SAR) and mediate MHC-independent tumor cell lysis. The specificity of this process is dependent on the valency of the used BiAb, as only BiAb monovalent for the T cell SAR mediated tumor-associated antigen-specific recognition. We also show, that while T cell degranulation is important for CAR T cell functionality, FasL on the T cell is necessary for SAR T cells to mediate anti-tumoral activity. The activity of SAR T cells is self-limiting due to antibody half-life, while cells can be effectively removed from circulation if needed (19).
Our report demonstrates that an otherwise inert molecule introduced in a T cell can be selectively triggered in vitro and in vivo through binding of a BiAb and subsequent crosslinking of the T cell with a tumor cell. Crosslinking ability of the 2 + 1 BiAb is restored in the presence of an immobilizing tumor-associated binding moiety for the other arm of the BiAb. Earlier work from our group and others could already demonstrate the synergy between ACT and BiAbs (9,20). However, our present study went beyond these approaches by combining a trivalent 2 + 1 BiAb format, as described for clinical stage CD3 BiAbs (also known as T cell bispecific (TCBs)) (21,22) with SAR- T cells. This platform can be adapted to different tumor types by altering either the BiAb tumor antigen target or the SAR.
Choosing the extracellular portion of the SAR will be central to the clinical translation of this therapeutic platform. The antigen must be restricted to the transduced T cells to avoid unwanted binding of the BiAb to non-tumor tissues. There is a lack of truly cancer-specific surface antigens. Introducing one of the few cancer-specific mutations in a surface antigen (EGFR variant 3) or an embryonic antigen (Cripto-1) might be a promising approach (23,24). EGFRvIII is known to mediate a constitutive, ligand-independent signaling due to the formation of transient homodimers which are stabilized by disulfide bonds of free amino - terminal cysteines C16. Although we have not observed ligand-independent signaling in our studies, the dimerization can be abolished by mutating the respective cysteine residue (25). The C16 is not involved in the BiAb engagement and thus could be mutated if any spontaneous dimerization of the SAR occurs.
Currently, most developmental efforts and clinical studies for cell-based immunotherapy focus on CAR T cell therapy (1). CAR T cells kill cancer cells through granzyme- and perforin-mediated lysis (26,27). Given that the signaling moieties used by the SAR in our study are similar to those employed by CARs, an identical mode of action would have been expected. Surprisingly, we found FasL to be an essential molecule for SAR T cell efficacy, and is further supported by reports on FasL activity as the mode of action for T cell-mediated killing (28). The exact underlying mechanism remains unclear, thus further investigation into the precise mechanism of killing is still needed, especially with regards to the influence of the BiAb on immune synapse formation and cell-cell interactions. It needs to be kept in mind that Fas - FasL interactions have been reported to promote tumor growth (29,30), thus under certain conditions FasL involvement might be a detrimental mode of action.
Modular approaches include “universal CAR” (UniCAR) and “switch CAR” which consist of a classical CAR targeting a decoy antigen linked to a tumor targeting nanobody or ScFv against a tumor-associated antigen (31–33), CD16-CAR targeting monoclonal antibodies, (34) or anti-FITC and anti-biotin CAR targeting FITC-tagged antibodies (35) or biotinylated molecules (36,37). These approaches may face immunogenicity issues as a result of the haptens employed and require posttranslational modification of the antibody or molecules, or, in the case of CD16, they can be triggered by circulating or tissue-bound antibodies, limiting their applicability. Moreover, CARs are utilizing scFvs for antigen recognition and are fully functional in their own right upon on- or even potential off-target engagement. Additionally, as CARs by definition do not require additional signals to trigger T cells once engaged, autonomous or tonic signaling that has been reported for several CARs remains a risk with the outlined strategies as well (38). In contrast, SARs are functionally inert molecules which rely on the external trigger of the BiAb for T cell activation. Some of these approaches, such as the anti-biotin CAR, utilize molecule interactions that are up to 106 times stronger than conventional ScFv interactions, thus questioning if target engagement and disengagement would happen in a similar fashion as reported for CARs or BiAbs which is important for efficacy. The platform we describe approximates the CAR approach in terms of efficacy and mode of action and combines, for the first time, modular activation with a target for T cell depletion.
A prerequisite for CAR strategies is tumor cell expression of the targeted antigen. CAR T cells bear the risk of toxicities due to attraction to healthy tissue that expresses the antigen, or resistance to natural T cell brakes. These risks cannot be adequately predicted from preclinical data (39,40). To avoid potentially lethal side effects, “safety switches” have been incorporated into therapeutically administered T cells. A major advantage of the platform we describe is its reversibility with the elimination of the BiAb. The BiAb can also be engineered for modified half-lives according to needs. SAR T cell activity could thus be restored upon redosing of the BiAb including the use of different tumor-targeted BiAbs. In a clinical setting, the adoptive transfer of SAR T cells should be administered first, with repeated dosing of the BiAb following, thus allowing for the careful monitoring of patients for the potential emergence of side effects. Moreover, the BiAb concentration could then be adjusted accordingly, to maintain T cell efficacy and manage possible toxicity. Further, the capacity of this platform to selectively activate SAR T cells without impacting all T cells differentiates the approach from pan-T cell activating bispecific antibodies (41).
This property is in contrast to currently used safety switches for CAR T cell-based treatments which rely on the elimination of the transferred T cells through depleting antibodies or drugs, inducing selective cell death (19,42). The speed and depth of T cell depletion remain to be established for these approaches, particularly whether they are sufficient to counteract severe toxicities. As of yet no successful depletion of CAR T cells upon occurrence of toxicities has been reported in a clinical setting.
In addition, depleting agents may come with side effects of their own (43). Total depletion of the transferred T cells may result in disease relapse. It may make re-dosing of active T cells difficult as an anti-cell product immune response might be generated. These limitations call for safety switches that do not deplete the causing agent but rather tune down their activity. The platform we describe has this potential, as T cell activity will vanish with diminishing levels of BiAb and will resume upon re-dosing. Should the BiAb decay not be sufficient for limiting toxicities, SAR T cells could also be depleted using the clinical grade antibody cetuximab, further enhancing the safety profile of the strategy.
The use of both CD4+ and CD8+ T cell subpopulations has been shown to be required for efficient and durable clinical responses to ACT (44). The SAR used in this study could be successfully transduced in CD4+ and CD8+ human T cells with great efficiency. Higher tumor infiltration and longer persistence in vivo by CD8+, and especially CD4+ SAR T cells emphasize the necessity of both cell types for the observed anti-tumoral responses.
Advanced T cell-based therapies have had limited success against solid tumors. This has mainly resulted from an impaired migration to and activation of the T cells within the tumor tissue (2). SAR T cells achieved long-lasting tumor control in vivo but failed to completely eradicate established tumors. Exchange of the signaling endodomains or equipment with additional structural components were observed to boost the in vivo performance of various chimeric antigen receptors. As such, the impact of further modifications to SAR design must be investigated in search of further improvement in SAR T cell performance, crucially their in vivo functionality.
Moreover, exchanging the intracellular signaling domain CD28 might have an impact on the in vivo persistence and differentiation of SAR T cells (1). The additional engineering of SAR T cells with selected chemokine receptors might overcome their limited infiltration into tumor tissue (17). Further, the use of dominant-negative TGF-β receptors (45) or PD-1-CD28 (12) switch receptors could help shield the modified T cells from the immunosuppressive tumor microenvironment.
In conclusion, we describe a novel modular platform for MHC-unrestricted SAR T cell-based therapy with properties distinct from current approaches and combining advantages from the CAR T cell and antibody worlds. Further preclinical investigations will be needed to demonstrate its translational potential and to further optimize its components.
Supplementary Material
Translational relevance.
Adoptive T cell therapy (ACT) using chimeric antigen receptor-transduced T cells is an effective strategy to treat hematological malignancies. Our modular MHC-unrestricted platform with dependence on monovalent BiAb triggering can overcome antigen loss as an evasion mechanism of solid tumors and off-target cytotoxicity. Synthetic Agonistic Receptor (SAR)-mediated T cell activation occurs only in the vicinity of tumor cells and is restricted by the BiAb half-life. This provides an effective means of control, via BiAb dosing, for the further development of this platform. In summary, we provide evidence for the potential for clinical development and translation of this platform.
Acknowledgments
This study was supported by grants from the international doctoral program “i-Target” funded by the Elite Network of Bavaria, the Melanoma Research Alliance (grant numbers N269626, 409510), the Marie-Sklodowska-Curie “Training Network for the Immunotherapy of Cancer (IMMUTRAIN)” funded by the H2020 program of the European Union, the Else Kröner-Fresenius-Stiftung, the German Cancer Aid, the Ernst-Jung-Stiftung, the Bundesministerium für Bildung und Forschung VIP+ grant ONKATTRACT and the European Research Council Starting Grant (grant number 756017).
Abbreviations
- ACT
Adoptive T cell therapy
- SAR
synthetic agonistic receptor
- BiAb
bispecific antibody
- CAR
chimeric antigen receptor
- EGFRvIII
epidermal growth factor receptor variant III
- EpCAM
epithelial cell adhesion molecule
- MSLN
mesothelin
Footnotes
Conflict of interest statement: Parts of this work have been performed for the doctoral theses of CHK, MRB, MS, MK, MG and RM at the Ludwig-Maximilians-Universität München. CS, GN, MG, SE, CK and SK are inventors of two patent applications related to this work. CS, GN, CK, MG and RM are employees and stock owners of Roche. JD is an employee of TCR2. The authors have declared that no other conflict of interest exists.
Author contributions
CHK, MRB, MS, MK, RK, BC, JD and SL performed experiments and analyzed data. MG and RM generated and purified all BiAbs. CH and FR performed in vivo experiments. JvB, MJ, DL and PD critically reviewed the manuscript and provided reagents. SK, SE, GN, CS and CK supervised the project and did the funding acquisition. SK, CHK and MRB designed the experiments and wrote the manuscript. All authors critically read and approved the final manuscript. The authors thank Prof. Wolfgang Zimmermann for critically reading the manuscript.
References
- 1.Lim WA, June CH. The principles of engineering immune cells to treat cancer. Cell. 2017;168(4):724–40. doi: 10.1016/j.cell.2017.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fesnak AD, June CH, Levine BL. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer. 2016;16(9):566–81. doi: 10.1038/nrc.2016.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rosenberg SA, Restifo NP. Adoptive cell transfer as personalized immunotherapy for human cancer. Science. 2015;348(6230):62–8. doi: 10.1126/science.aaa4967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garfall AL, Maus MV, Hwang WT, Lacey SF, Mahnke YD, Melenhorst JJ, et al. Chimeric antigen receptor T Cells against CD19 for multiple myeloma. N Engl J Med. 2015;373(11):1040–7. doi: 10.1056/NEJMoa1504542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Porter DL, Hwang WT, Frey NV, Lacey SF, Shaw PA, Loren AW, et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med. 2015;7(303):303ra139. doi: 10.1126/scitranslmed.aac5415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507–17. doi: 10.1056/NEJMoa1407222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Teachey DT, Lacey SF, Shaw PA, Melenhorst JJ, Maude SL, Frey N, et al. Identification of predictive biomarkers for cytokine release syndrome after chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Cancer Discov. 2016;6(6):664–79. doi: 10.1158/2159-8290.CD-16-0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fitzgerald JC, Weiss SL, Maude SL, Barrett DM, Lacey SF, Melenhorst JJ, et al. Cytokine release syndrome after chimeric antigen receptor T cell therapy for acute lymphoblastic leukemia. Crit Care Med. 2017;45(2):e124–e31. doi: 10.1097/CCM.0000000000002053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kobold S, Steffen J, Chaloupka M, Grassmann S, Henkel J, Castoldi R, et al. Selective bispecific T cell recruiting antibody and antitumor activity of adoptive T cell transfer. J Natl Cancer Inst. 2015;107(1):364. doi: 10.1093/jnci/dju364. [DOI] [PubMed] [Google Scholar]
- 10.O’Rourke DM, Nasrallah MP, Desai A, Melenhorst JJ, Mansfield K, Morrissette JJD, et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med. 2017;9(399) doi: 10.1126/scitranslmed.aaa0984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kobold S, Merk M, Hofer L, Peters P, Bucala R, Endres S. The macrophage migration inhibitory factor (MIF)-homologue D-dopachrome tautomerase is a therapeutic target in a murine melanoma model. Oncotarget. 2014;5(1):103–7. doi: 10.18632/oncotarget.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kobold S, Grassmann S, Chaloupka M, Lampert C, Wenk S, Kraus F, et al. Impact of a new fusion receptor on PD-1-mediated immunosuppression in adoptive T cell therapy. J Natl Cancer Inst. 2015;107(8):djv146-djv. doi: 10.1093/jnci/djv146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Duewell P, Beller E, Kirchleitner SV, Adunka T, Bourhis H, Siveke J, et al. Targeted activation of melanoma differentiation-associated protein 5 (MDA5) for immunotherapy of pancreatic carcinoma. OncoImmunology. 2015;4(10):e1029698. doi: 10.1080/2162402X.2015.1029698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kitamura T, Koshino Y, Shibata F, Oki T, Nakajima H, Nosaka T, et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp Hematol. 2003;31(11):1007–14. [PubMed] [Google Scholar]
- 15.Hessell AJ, Hangartner L, Hunter M, Havenith CEG, Beurskens FJ, Bakker JM, et al. Fc receptor but not complement binding is important in antibody protection against HIV. Nature. 2007;449:101–4. doi: 10.1038/nature06106. [DOI] [PubMed] [Google Scholar]
- 16.Schaefer W, Regula JT, Bähner M, Schanzer J, Croasdale R, Dürr H, et al. Immunoglobulin domain crossover as a generic approach for the production of bispecific IgG antibodies. Proc Natl Acad Sci USA. 2011;108(27):11187–92. doi: 10.1073/pnas.1019002108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rapp M, Grassmann S, Chaloupka M, Layritz P, Kruger S, Ormanns S, et al. C-C chemokine receptor type-4 transduction of T cells enhances interaction with dendritic cells, tumor infiltration and therapeutic efficacy of adoptive T cell transfer. OncoImmunology. 2016;5(3):e1105428. doi: 10.1080/2162402X.2015.1105428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Voigt C, May P, Gottschlich A, Markota A, Wenk D, Gerlach I, et al. Cancer cells induce interleukin-22 production from memory CD4+ T cells via interleukin-1 to promote tumor growth. Proc Natl Acad Sci USA. 2017;114:12994–9. doi: 10.1073/pnas.1705165114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Paszkiewicz PJ, Frassle SP, Srivastava S, Sommermeyer D, Hudecek M, Drexler I, et al. Targeted antibody-mediated depletion of murine CD19 CAR T cells permanently reverses B cell aplasia. J Clin Invest. 2016;126(11):4262–72. doi: 10.1172/JCI84813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Urbanska K, Lynn RC, Stashwick C, Thakur A, Lum LG, Powell DJ., Jr Targeted cancer immunotherapy via combination of designer bispecific antibody and novel gene-engineered T cells. J Transl Med. 2014;12:347. doi: 10.1186/s12967-014-0347-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bacac M, Colombetti S, Herter S, Sam J, Perro M, Chen S, et al. CD20-TCB with Obinutuzumab pretreatment as next-generation treatment of hematologic malignancies. CliN Cancer Res. 2018;24(19):4785. doi: 10.1158/1078-0432.CCR-18-0455. [DOI] [PubMed] [Google Scholar]
- 22.Bacac M, Fauti T, Sam J, Colombetti S, Weinzierl T, Ouaret D, et al. A novel carcinoembryonic antigen T-cell bispecific antibody (CEA TCB) for the treatment of solid tumors. CliN Cancer Res. 2016;22(13):3286. doi: 10.1158/1078-0432.CCR-15-1696. [DOI] [PubMed] [Google Scholar]
- 23.Johnson LA, Scholler J, Ohkuri T, Kosaka A, Patel PR, McGettigan SE, et al. Rational development and characterization of humanized anti-EGFR variant III chimeric antigen receptor T cells for glioblastoma. Sci Transl Med. 2015;7(275):275ra22. doi: 10.1126/scitranslmed.aaa4963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ligtenberg MA, Witt K, Galvez-Cancino F, Sette A, Lundqvist A, Lladser A, et al. Cripto-1 vaccination elicits protective immunity against metastatic melanoma. Oncoimmunology. 2016;5(5):e1128613. doi: 10.1080/2162402X.2015.1128613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ymer SI, Greenall SA, Cvrljevic A, Cao DX, Donoghue JF, Epa VC, et al. Glioma specific extracellular missense mutations in the first cysteine rich region of rpidermal growth factor receptor (EGFR) initiate ligand independent activation. Cancers. 2011;3(2):2032–49. doi: 10.3390/cancers3022032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Darcy PK, Haynes NM, Snook MB, Trapani JA, Cerruti L, Jane SM, et al. Redirected perforin-dependent lysis of colon carcinoma by ex vivo genetically engineered CTL. J Immunol. 2000;164(7):3705–12. doi: 10.4049/jimmunol.164.7.3705. [DOI] [PubMed] [Google Scholar]
- 27.Isaaz S, Baetz K, Olsen K, Podack E, Griffiths GM. Serial killing by cytotoxic T lymphocytes: T cell receptor triggers degranulation, re-filling of the lytic granules and secretion of lytic proteins via a non-granule pathway. Eur J Immunol. 1995;25(4):1071–9. doi: 10.1002/eji.1830250432. [DOI] [PubMed] [Google Scholar]
- 28.Zeytun A, Hassuneh M, Nagarkatti M, Nagarkatti PS. Fas-Fas ligand-based interactions between tumor cells and tumor-specific cytotoxic T lymphocytes: a lethal two-way street. Blood. 1997;90(5):1952–9. [PubMed] [Google Scholar]
- 29.Chen L, Park S-M, Tumanov AV, Hau A, Sawada K, Feig C, et al. CD95 promotes tumour growth. Nature. 2010;465:492. doi: 10.1038/nature09075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ceppi P, Hadji A, Kohlhapp FJ, Pattanayak A, Hau A, Liu X, et al. CD95 and CD95L promote and protect cancer stem cells. Nat Commun. 2014;5:5238. doi: 10.1038/ncomms6238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Albert S, Arndt C, Feldmann A, Bergmann R, Bachmann D, Koristka S, et al. A novel nanobody-based target module for retargeting of T lymphocytes to EGFR-expressing cancer cells via the modular UniCAR platform. Oncoimmunology. 2017;6(4):e1287246. doi: 10.1080/2162402X.2017.1287246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cartellieri M, Feldmann A, Koristka S, Arndt C, Loff S, Ehninger A, et al. Switching CAR T cells on and off: a novel modular platform for retargeting of T cells to AML blasts. Blood Cancer J. 2016;6(8):e458. doi: 10.1038/bcj.2016.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rodgers DT, Mazagova M, Hampton EN, Cao Y, Ramadoss NS, Hardy IR, et al. Switch-mediated activation and retargeting of CAR-T cells for B-cell malignancies. Proc Natl Acad Sci USA. 2016;113(4):E459. doi: 10.1073/pnas.1524155113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kudo K, Imai C, Lorenzini P, Kamiya T, Kono K, Davidoff AM, et al. T lymphocytes expressing a CD16 signaling receptor exert antibody-dependent cancer cell killing. Cancer Res. 2014;74(1):93–103. doi: 10.1158/0008-5472.CAN-13-1365. [DOI] [PubMed] [Google Scholar]
- 35.Tamada K, Geng D, Sakoda Y, Bansal N, Srivastava R, Li Z, et al. Redirecting gene-modified T cells toward various cancer types using tagged antibodies. Clin Cancer Res. 2012;18(23):6436–45. doi: 10.1158/1078-0432.CCR-12-1449. [DOI] [PubMed] [Google Scholar]
- 36.Urbanska K, Lanitis E, Poussin M, Lynn RC, Gavin BP, Kelderman S, et al. A Universal Strategy for Adoptive Immunotherapy of Cancer through Use of a Novel T-cell Antigen Receptor. Cancer Research. 2012;72(7):1844. doi: 10.1158/0008-5472.CAN-11-3890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Darowski D, Kobold S, Jost C, Klein C. Combining the best of two worlds: highly flexible chimeric antigen receptor adaptor molecules (CAR-adaptors) for the recruitment of chimeric antigen receptor T cells. mAbs. 2019:1–11. doi: 10.1080/19420862.2019.1596511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gomes-Silva D, Mukherjee M, Srinivasan M, Krenciute G, Dakhova O, Zheng Y, et al. Tonic 4-1BB Costimulation in Chimeric Antigen Receptors Impedes T Cell Survival and Is Vector-Dependent. Cell Reports. 2017;21(1):17–26. doi: 10.1016/j.celrep.2017.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ahmed N, Brawley V, Hegde M, Bielamowicz K, Kalra M, Landi D, et al. HER2-Specific Chimeric Antigen Receptor-Modified Virus-Specific T Cells for Progressive Glioblastoma: A Phase 1 Dose-Escalation Trial. JAMA Oncol. 2017 doi: 10.1001/jamaoncol.2017.0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bernhard H, Neudorfer J, Gebhard K, Conrad H, Hermann C, Nahrig J, et al. Adoptive transfer of autologous, HER2-specific, cytotoxic T lymphocytes for the treatment of HER2-overexpressing breast cancer. Cancer Immunol Immunother. 2008;57(2):271–80. doi: 10.1007/s00262-007-0355-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Topp MS, Gökbuget N, Stein AS, Zugmaier G, O’Brien S, Bargou RC, et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 2015;16(1):57–66. doi: 10.1016/S1470-2045(14)71170-2. [DOI] [PubMed] [Google Scholar]
- 42.Casucci M, Perna SK, Falcone L, Camisa B, Magnani Z, Bernardi M, et al. Graft-versus-leukemia effect of HLA-haploidentical central-memory T-cells expanded with leukemic APCs and modified with a suicide gene. Mol Ther. 2013;21(2):466–75. doi: 10.1038/mt.2012.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Collaborative DTSG. Treatment of serious cytomegalovirus infections with 9-(1,3-dihydroxy-2-propoxymethyl)guanine in patients with AIDS and other immunodeficiencies. N Engl J Med. 1986;314(13):801–5. doi: 10.1056/NEJM198603273141301. [DOI] [PubMed] [Google Scholar]
- 44.Turtle CJ, Hanafi L-A, Berger C, Gooley TA, Cherian S, Hudecek M, et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J Clin Invest. 2016;126(6):2123–38. doi: 10.1172/JCI85309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Foster AE, Dotti G, Lu A, Khalil M, Brenner MK, Heslop HE, et al. Antitumor activity of EBV-specific T lymphocytes transduced with a dominant negative TGF-beta receptor. Journal of immunotherapy (Hagerstown, Md : 1997) 2008;31(5):500–5. doi: 10.1097/CJI.0b013e318177092b. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.