Abstract
Immune checkpoint inhibitors (ICI), which target immune regulatory pathways to unleash antitumor responses, have revolutionized cancer immunotherapy. Despite the remarkable success of ICI immunotherapy, a significant proportion of patients whose tumors respond to these treatments develop immune-related adverse events (irAEs) resembling autoimmune diseases. Although the clinical spectrum of irAEs is well characterized, their successful management remains empiric. This is in part because the pathogenic mechanisms involved in the break-down of peripheral tolerance and induction of irAEs remain elusive. Herein, we focused on regulatory T cells (Tregs) in individuals with irAEs because these cells are vital for maintenance of peripheral tolerance, appear expanded in the peripheral blood of individuals with cancer and abundantly express checkpoint molecules, hence representing direct targets of ICI immunotherapy. Our data demonstrate an intense transcriptomic reprogramming of CD4+CD25+CD127– Tregs in the blood of individuals with advanced metastatic melanoma who develop irAEs following ICI immunotherapy, with a characteristic inflammatory, apoptotic and metabolic signature. This inflammatory signature was shared by Tregs from individuals with different types of cancer developing irAEs and individuals with autoimmune diseases. Our findings suggest inflammatory Treg reprogramming is a feature of immunotherapy-induced irAEs, and this may facilitate translational approaches aiming to induce robust antitumor immunity without disturbing peripheral tolerance.
Introduction
Immune checkpoint inhibitors (ICI), such as anti-PD1, anti-PDL1 and anti-CTLA4, have shown remarkable efficacy in the treatment of solid tumors. Despite their success, more than half of treated patients do not derive meaningful benefit from such treatments and responses are often accompanied by autoimmune toxicities, termed immune related adverse events (irAEs), which may interfere with therapy and could lead to life-threatening conditions (1–2). These toxicities usually develop within the first few weeks to months after treatment initiation and can manifest as generalized symptoms including fatigue or fever, or can produce organ-specific damage leading to rash, arthritis, colitis, pneumonitis, and adrenal or thyroid insufficiency (3–4). Management of irAEs in general, entails dose interruption/discontinuation of immunotherapy and systemic high-dose corticosteroids (5). To date, it is not possible to predict which patients will develop irAEs, which organs will be involved and how severe an irAE may be, making patient management challenging. Although genetic predisposition, gut microbiome, epitope spreading and cross-presentation of neoantigens have been implicated in the development of irAEs (6–7), the precise molecular mechanisms that drive them remain largely unknown. As autoimmunity and cancer represent two sides of the same coin, in terms of the operation of immune tolerance mechanisms, perhaps it is not surprising that as we manipulate the immune system to treat cancer through the use of ICI, we inevitably disturb the mechanisms that regulate self-tolerance, inducing an array of irAEs.
Immune suppression by Foxp3+regulatory T cells (Tregs) is essential and indispensable for maintenance of tolerance and prevention of autoimmunity, as illustrated by the fact that spontaneous autoimmune disease develops when Tregs are rendered deficient. For example, mutations in the human gene encoding Foxp3 impair Treg development and cause a fatal multi-organ autoimmune disease called immune dysregulation, polyendocrinopathy, enteropathy, and X-linked (IPEX) syndrome (8). In rodents, depletion of Foxp3+ Tregs causes similar autoimmune diseases (9).
In addition to preventing autoimmunity, Tregs suppress the antitumor immune response and promote tumor outgrowth (10). The tumor microenvironment (TME) is unique in that it is nutrient poor, hypoxic, and acidic. This provides a unique proliferative and functional advantage to Tregs, which rely on oxidative phosphorylation, compared with effector T cells, which are primarily glycolytic (11). Consistent with this, increased frequencies of intratumoral Tregs have been reported in almost all solid tumors (12) and the level of Treg infiltration is associated with unfavorable prognosis in various types of cancer (12–13). In the TME, Tregs acquire enhanced suppressor functions and an activated phenotype with expression of chemokine receptors such as CCR4 or CCR8 and up-regulation of immune checkpoint proteins such as CTLA-4, PD1, PDL-1, PDL-2, IL1R2 (14). Thus far, efforts to therapeutically target Tregs in cancer have had limited success and have been accompanied by the development of side effects.
Tregs express high levels of CTLA-4 and PD1, and recent evidence suggests that PD1 expression by Tregs in the TME can predict the efficacy of anti-PD1 immunotherapy (15). Therefore, we hypothesized that ICI could imprint on Tregs and induce their reprogramming, contributing to the development of irAEs. Herein, we demonstrate an intense “inflammatory” phenotype in CD4+CD25+CD127– Tregs isolated from the blood of patients with advanced metastatic melanoma who developed irAEs after ICI therapy. As the use of ICIs in anticancer therapy is rapidly expanding, the frequency of irAEs is expected to significantly increase. Thus, it is urgent that we increase our understanding of the mechanisms underlying the development of irAEs in order to facilitate not only better management of irAEs but also develop prognostic biomarkers.
Materials and Methods
Human subjects
Peripheral blood samples were obtained from individuals diagnosed with cancer or autoimmune diseases and were freshly analyzed directly after collection. Patients with advanced metastatic melanoma, stage III(B-D)-IV, who were receiving treatment with anti-PD1 (n =26), were recruited through the First Department of Internal Medicine, Laikon General Hospital Greece, National and Kapodistrian University of Athens, (Greece). Samples were collected at the beginning of the 3rd cycle of anti-PD1 infusion. Patients with kidney (n=6), non-small cell lung (n=6), liver (n=1) and bladder (n=3) cancer who were receiving treatment with anti-PD1 (between 8th-12th cycle of infusion), were recruited through the oncology clinic of Alexandra University hospital, National and Kapodistrian University of Athens (Greece) and Department of Medical Oncology, University General Hospital of Heraklion (Greece). Individuals with cancer were staged according to the American Joint Committee on Cancer 8th edition (AJCC Manual).
Individuals with rheumatoid arthritis (RA) (n=9) were diagnosed according to the 1987 American College of Rheumatology (ACR) criteria, had moderate disease activity (DAS > 4.2) according to the disease activity score based on the 28 joint counts. RA and healthy individuals (n=14) were recruited through the Rheumatology and Clinical Immunology Department, 4th Clinical Pathology, Attikon University Hospital (Athens, Greece). Individuals with inflammatory bowel disease (IBD) (n=4) who were in remission or had moderate disease activity, based on standard clinical, endoscopical and histological criteria, were recruited through the GI-Unit, 3rd Academic Department of Internal Medicine, Sotiria Hospital, National and Kapodistrian University of Athens. Individuals with Hashimoto’s thyroiditis (n=7) who were diagnosed on the basis of increased TSH, increased anti-TPO and anti-Tg and normal or low T4, T3 levels and receiving thyroxine replacement therapy, were recruited through the Endocrine Unit of 1st Department of Internal Medicine, Laiko Hospital (Athens, Greece). All individuals with autoimmune diseases did not receive steroids for at least 24h prior to blood drawing.
The Clinical Research Ethics Board of all hospitals approved this study. Informed written consent was obtained from all individuals prior to sample collection. All patient studies were conducted in accordance with ethical guidelines of the Declaration of Helsinki. Meta-analysis of healthy and Rhematoid Arthritis (RA) samples was performed from Alissafi et al. Cell metabolism (16). Clinical and demographic characteristics of all the patients included in this study are summarized in Supplementary Table 1. Objective Response Rate (ORR) is defined as the proportion of patients with a complete or a partial response to therapy.
Human cell isolation from peripheral blood
Heparinized blood (20ml) was collected from human subjects. Peripheral blood mononuclear cells (PBMCs) were isolated on Histopaque-1077 (cat.#10771, Sigma) density gradient. Briefly, blood was diluted 1:1 with PBS and carefully layered over Histopaque medium. Ficoll gradients were centrifuged at 1800 rpm for 30 min with no brake at room temperature. The PBMC layer was collected and cells were washed with PBS.
Treg isolation
Tregs were isolated from PBMCs using the CD4+CD25+CD127dim/– Regulatory T Cell Isolation Kit II (cat.#130-094-775, Miltenyi Biotec) according to the manufacturer’s instructions. Briefly, non-CD4+ and CD127high cells were indirectly magnetically labeled with a cocktail of biotin-conjugated antibodies, and anti-biotin monoclonal antibodies conjugated to MicroBeads. The labeled cells were subsequently depleted by separation through MACS® column. In the second step, the CD4+CD25+CD127dim/– Tregs were directly labeled with CD25 MicroBeads II and isolated by positive selection from the pre-enriched CD4+ T-cell fraction, by separation through MACS® Column and eluted as the positively selected cell fraction.
Flow cytometry
For analysis of Tregs, single-cell suspensions from human PBMCs were stained with conjugated antibodies against human CD4 (BioLegend Cat# 317428, RRID:AB_1186122), CD25 (BioLegend Cat# 302604, RRID:AB_314274), and CD127 (BioLegend Cat# 351316, RRID:AB_10900804). For Foxp3 intracellular staining, cells were fixed and stained using the Foxp3 Staining Set (Thermo Fisher Scientific Cat# 12-4774-42, RRID:AB_10670338) according to the manufacturer’s instructions. Cells of interest were acquired on a FACS ARIA III (BD Biosciences) using BD FACSDIVA v8.0.1 software (BD FACSDiva Software, RRID:SCR_001456). Analysis was performed with FlowJo software (FlowJo, RRID:SCR_008520).
RNA sequencing pipeline
Total RNA was extracted using NucleoSpin® RNA XS (Macherey-Nagel #740955.50) as per the manufacturer’s protocol and mRNA libraries were generated using the Illumina TruSeq Sample Preparation kit v2 (Illumina #RS-122-2001). Single-end 75-bp mRNA sequencing was performed on Illumina NextSeq 500 platform. Raw fastq sequencing reads were aligned against the human reference genome sequence (GRCh37/hg19) using the STAR aligned v2.5.0c, discarding all non-uniquely aligned reads (16). For read counting per annotated gene, we utilized the STAR (STAR, RRID:SCR_015899) function “--quantMode GeneCounts”, counting reads matching exons of the Ensembl V75 Genes annotation. Further processing was performed with the R Bioconductor (Bioconductor, RRID:SCR_006442) package edgeR v.3.14.0 (edgeR, RRID:SCR_012802) using unstranded reads. Reads were normalized for intra- and inter-sample variances using the functions “calcNormFactors” and “estimateTagwiseDisp”, and further by the gene-length as per Ensembl V75 Genes annotations, resulting in fragments-per-kilobase-per-million (FPKM) for each gene. Differential gene expression analysis was performed as previously described (16). Genes with p-value<0.05 and fold change |FC|>1.5 were considered statistically significant. Heatmaps and boxplots were created in R with an in-house developed script which is based on ggplot package. Venn diagrams were created using Venny 2.1.0 online tool.
Enrichment analysis
Significant differentially expressed genes (DEGs) were used for gene ontology (GO) analysis using g:Profiler web-server (G:Profiler, RRID:SCR_006809). Gene Set Enrichment Analysis (Gene Set Enrichment Analysis, RRID:SCR_003199) was also performed to reveal enriched signatures in our gene sets based on the Molecular Signatures Database (MSigDB) v7.0. Gene sets were ranked by taking the –log10 transform of the p-value multiplied by the FC. Significantly upregulated genes were at the top and significantly downregulated genes were at the bottom of the ranked list. GSEA pre-ranked analysis was then performed using the default settings. Enrichment was considered significant FDR (q-value) <5%. Pathway analysis was performed using IPA (Ingenuity Pathway Analysis, RRID:SCR_008653). We selected differentially expressed genes for each pairwise comparison using p-value < 0.05 and |FC|≥1.5 thresholds.
Quantification and Statistical Analysis
Statistical analysis was performed using unpaired Student’s t-test in GraphPad Prism v5 software (GraphPad Prism, RRID:SCR_002798). Data are presented as means ± S.E.M. P-value < 0.05 was considered as indicative of statistical significance. All P-values and n are reported in the figure legends. The investigators were not blinded to the identities of the samples. No randomization was performed. Compared samples were collected and analyzed under the same conditions. G*power analysis was performed (with 90% power and 0.001 type 1 error) to calculate the appropriate sample size. No data were excluded. All data showed normal distribution.
Data Sharing Statement
The RNAseq data have been deposited in the European Genome-Phenome Archive (EGA accession number: EGAS00001004694)
Results
Transcriptomic reprogramming of peripheral Tregs in melanoma patients developing irAEs
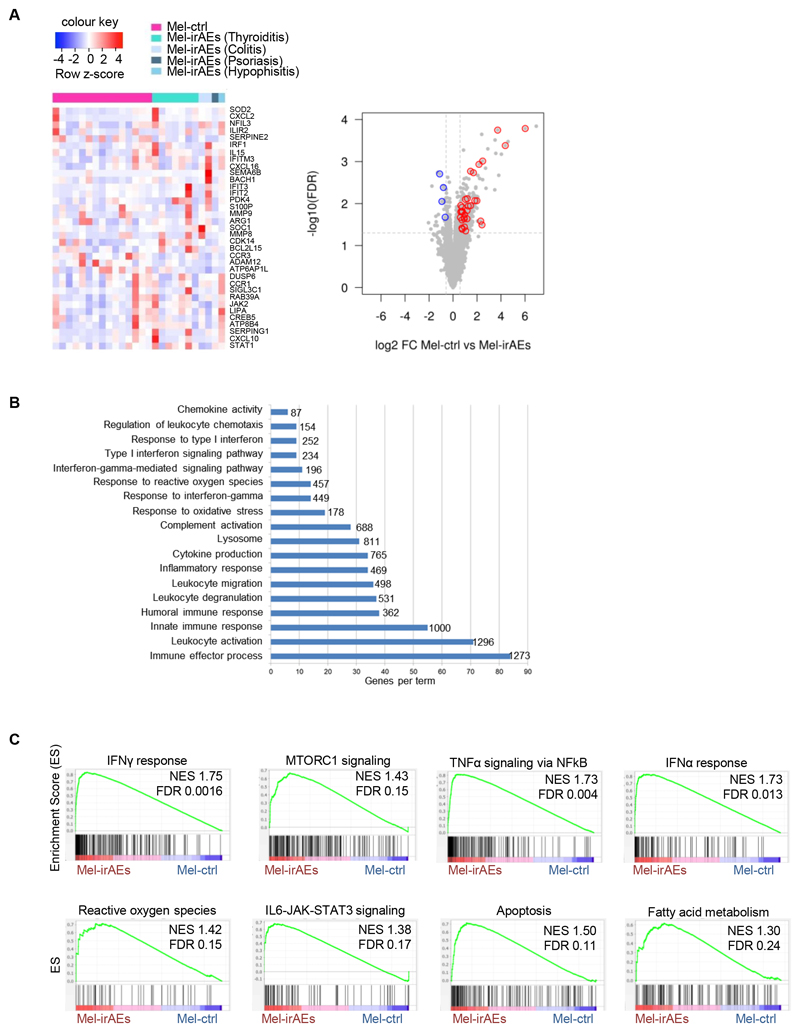
The molecular mechanisms underlying the development of irAEs remain elusive and their delineation will facilitate the design of translational approaches to induce robust antitumor immunity without disturbing peripheral tolerance. Herein, we explored whether ICI immunotherapy reprograms peripheral Tregs during cancer and could contribute to the development of irAEs. To do this, CD4+CD25+CD127– Tregs were isolated from the blood of individuals with advanced melanoma who were treated with anti-PD1 and subjected to transcriptomic analysis. Eleven of the patients experienced irAEs such as hypophisitis, thyroiditis, psoriasis and colitis, and are denoted hereafter as Mel-irAEs, and 15 did not (denoted as Mel-ctrl) (Supplementary Table 1). Although we did not observe significant differences in the frequency of CD4+CD25+Foxp3+CD127– Tregs between Mel-irAEs and Mel-ctrl (Supplementary Fig. 1A), genome-wide RNA-seq analysis revealed 252 differentially expressed genes (DEG) (|FC|≥1.5 and p<0.05) between the two groups (Supplementary Fig. 1B).
Focusing on Treg biology and immune-related genes, we identified a core signature of upregulated transcripts in Mel-irAEs compared to Mel-ctrl Tregs (Fig. 1A). These genes included those encoding chemokines and chemokine receptors involved in inflammation and leukocytic mobility, such as CXCL2, CXCL16, CXCL10, and CCR1; and enzymes such as amino acid catabolizing ARG1 and anti-oxidant SOD2. In contrast, genes implicated in the function of activated Tregs such as CCR3 and SERPINE2 or in metabolic processes such as ADAM12 and ATP6AP1L were significantly downregulated in Mel-irAE Tregs compared with Mel-ctrl Tregs (Fig. 1A and Supplementary Fig. 1B). Pathway analysis of DEGs showed that circulating Mel-irAEs Tregs exhibited an inflammatory-like phenotype, consisted of leukocyte activation, inflammatory response, cytokine production, oxidative stress response, type I IFN signaling and IFNγ signaling, compared to Mel-ctrl Tregs (Fig. 1B). In detail, GSEA demonstrated that Mel-irAEs Tregs were enriched in pro-inflammatory processes such as “IFNγ response” (with transcription factors: IRF1, STAT1, STAT3 and NFKB1A being among the top enriched genes), “IFNα response” (with top enriched genes belonging to the IFN-induced genes i.e. IFITR2-3, IFITM2, IFIH1), “TNFα signaling via NFKb” (with top enriched genes including RELB, IL18, and IL1B) and “IL-6/JAK/STAT3 signaling” (with STAT1, STAT3, TNF, IL1B, IRF1 and IFNGR2 being among the most enriched genes) (Fig. 1C). The enrichment in inflammatory pathways in Mel-irAEs Tregs was retained (370 DEGs) when individuals were categorized according to their objective response rate (ORR) (Supplementary Fig. 1C & Supplementary Table 1).
Figure 1. Treg inflammatory transcriptomic reprogramming in patients with advanced melanoma who develop irAEs.
Transcriptomic analysis of CD4+CD25+CD127– Tregs isolated from blood of advanced melanoma patients undergoing anti-PD1 treatment. (A) Heatmap with scaled cpm expression values (row z-score) (left) and volcano plot (right) of selected DEGs between ‘Mel-ctrl (n=15) vs Mel-irAEs (n=11)’. Categories of irAEs are listed on the heatmap. (B) Bar plot representing pathway analysis of DEGs from ‘Mel-ctrl vs Mel-irAEs’with adjusted p value ≤ 0.05. Each term size is listed on the end of each bar.(C) GSEA plot showing the enrichment of “IFNγ response” (NES 1.75, FDR 0.0016), “MTORC1 signaling” (NES 1.43, FDR 0.15). “Reactive oxygen species” (NES 1.42, FDR 0.15), “IL6-JAK-STAT3 signaling” (NES 1.38, FDR 0.17), “TNFα signaling via NFkB” (NES 1.73, FDR 0.004), “IFNα response” (NES 1.73, FDR 0.013), “Apoptosis” (NES 1.50, FDR 0.11) and “Fatty acid metabolism” (NES 1.30, FDR 0.24) gene set. For DEG analysis, the GLM statistical approach and the thresholds: |FC|≥1.5 and p<0.05 were used.
Treg metabolism drives their proliferation and immunosuppressive nature under steady state (17), whereas their de-stabilized metabolic fitness dictates development of autoimmunity (16). In this context, our GSEA analysis revealed a metabolic rewiring in Mel-irAEs compared with Mel-ctrl Tregs. This rewiring was characterized by “MTORC1 signaling” (with enrichment for TBK1, SERP1, and NAMPT genes), “reactive oxygen species pathway” (with top enriched genes such as MPO, SOD2, and CAT) and “Fatty acid metabolism” (with top enriched genes such as HMGCS1, ACOX1, and ACSL1) (Fig. 1C).
To provide mechanistic insights, IPA was performed, revealing IFNγ, IL-6, IL-1, IL-33, TNF, NFkb complex, STAT1, STAT4, STAT5, IRF1, IRF3, IRF5 and IRF7 as the top activated upstream regulators in Mel-irAEs Tregs (Supplementary Fig. 1D). These data provide insight into the signals that drive intense reprogramming of circulating CD4+CD25+CD127– Tregs in advanced melanoma patients who develop irAEs following ICI immunotherapy toward a pro-inflammatory phenotype accompanied by enrichment in metabolic alterations.
Shared inflammatory and metabolic signatures in autoimmune and irAE Tregs
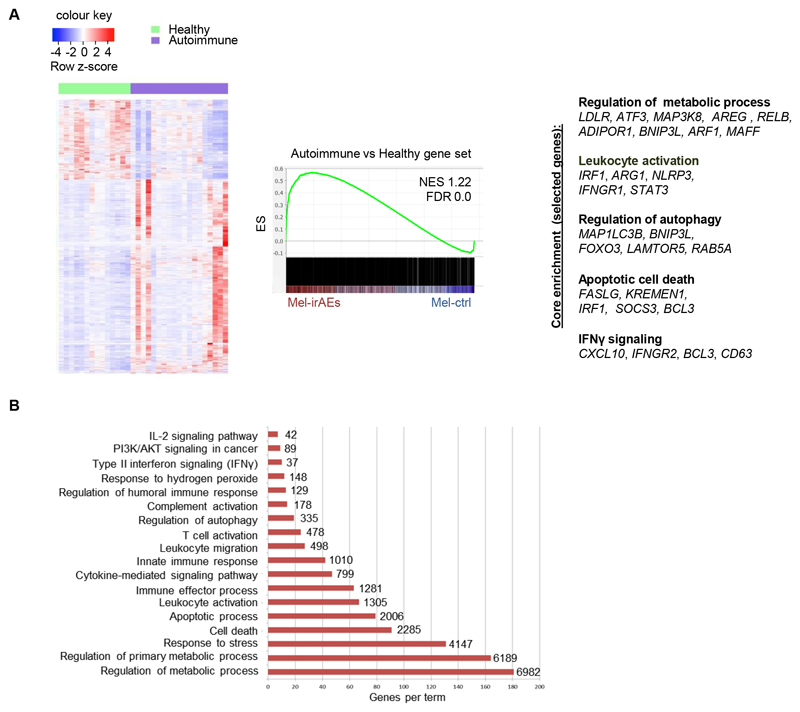
Despite the extensive clinical characterization of irAEs, it remains unclear to what extend they share common pathogenetic features with spontaneously arising autoimmune syndromes. To this end, we performed transcriptomic analysis of CD4+CD25+CD127– Tregs isolated from peripheral blood of healthy individuals and individuals with autoimmune manifestations analogous to the irAEs cohort (Ulcerative Colitis, Chrohn’s Disease, Hashimoto Thyroiditis, and Rheumatoid Arthritis, see Supplementary Table 1, denoted as autoimmune). When compared with Tregs from healthy individuals, autoimmune Tregs demonstrated a de-regulated transcriptomic program that was similar to the Mel-irAEs Treg phenotype (Fig. 2A). GSEA analysis demonstrated an over-representation of Mel-irAEs Treg DEGs in autoimmune versus healthy Tregs, including genes involved in IFNγ signaling (CXCL10, IFNGR2, BCL3, and CD63), leukocyte activation (IRF1, ARG1, NLRP3, IFNGR1, and STAT3), regulation of autophagy (MAP1LC3B, BNIP3L, FOXO3, LAMTOR5, and RAB5A), apoptotic cell death (FASLG, KREMEN1, IRF1, SOCS3, and BCL3) and regulation of metabolic processes (LDLR, ATF3, MAP3K8, ARE, RELB, ADIPOR1, BNIP3L, ARF1, and MAFF) (Fig. 2A and 2B). Our findings demonstrate a shared transcriptomic profile among Tregs from patients with autoimmune diseases and those with irAEs. This profile is enriched for inflammatory, apoptotic and metabolic pathways, and is characteristic of destabilized Tregs (18).
Figure 2. Inflammatory Treg transcriptomic profile: A common denominator of autoimmune diseases and irAEs.
(A) Heatmap of DEGs, with scaled cpm expression values (row z-score), from peripheral blood CD4+CD25+CD127–Tregs isolated from ‘Healthy (n=14) vs Autoimmune (n=20)’ individuals (left) and GSEA plot (right) showing the enrichment of ‘Mel-irAEs vs Mel-ctrl’ with the ‘Autoimmune vs Healthy’ gene set (NES 1.22, FDR 0.0). Selected core enriched genes listed to the bottom of the plot. (B) Bar plot representing pathway analysis of the core enriched genes deriving from comparison in panel A, with adjusted p value ≤ 0.05. Each term size is listed on the end of each bar. For DEG analysis, the GLM statistical approach and the thresholds: |FC|≥1.5 and p<0.05 were used.
Transcriptomic profile of irAE Tregs is common across different types of cancer
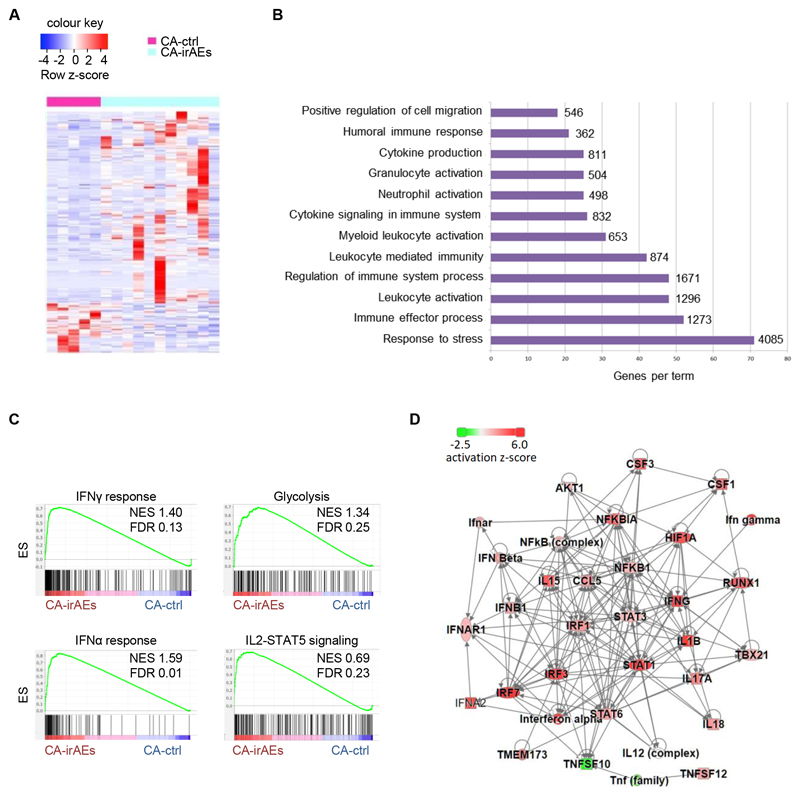
To examine whether inflammatory reprogramming of Tregs is specific to patients with melanoma experiencing irAEs following anti-PD1 immunotherapy, we performed transcriptomic analysis of Tregs from individuals with kidney, liver, bladder and non-small cell lung cancer who did or did not experience irAEs following anti-PD1 immunotherapy (denoted as CA-irAEs or CA-ctrl respectively, Supplementary Table 1). CA-irAEs Tregs had a distinct transcriptomic profile, with 225 DEGs compared with CA-ctrl Tregs (Fig. 3A). Pathway analysis revealed an inflammatory profile in CA-irAEs Tregs, noticeably similar to that of Mel-irAEs Tregs, with leukocyte activation, cytokine production, immune effector process and response to stress being among the top enriched pathways (Fig. 3B). GSEA analysis further confirmed a destabilized inflammatory phenotype in CA-irAEs Tregs that wassimilar tothat seen in Mel-irAEs Tregs. In detail, IFNγ response (through IRF1, IRF2, IRF7, IRF9, STAT1, STAT2, CCL5, CXCL9, CXCL10 and CD274 genes), IFNα response (with top enriched genes such as IRF1, IRF2, IRF7, IRF9, STAT2, CXCL10, IFITs, and IFIs), and IL2–STAT5 signaling (IL1R2, EOMES, BCL2L1, SOCS1, CXCL10, SOCS2 and TNFSF10) among the most enriched pathways (Fig. 3C). Noticeably, among the top activated upstream regulators of circulating Tregs in CA-irAEs were: IFNγ, IFNα, IL-17A, TBX21, NFkB, HIF1A, IRF1, RUNX1, IL-1B, TNFSF10, STAT1, STAT3 and STAT6 (Fig. 3D & Supplementary Fig. 2A). Taken together, these findings identify a distinct pro-inflammatory transcriptomic profile in Tregs isolated from individuals developing irAEs that is common across different types of cancer.
Figure 3. Shared pro-inflammatory transcriptomic profile in Tregs across different types of cancer.
Transcriptomic analysis of CD4+CD25+CD127– Tregs isolated from peripheral blood of CA-ctrl (n=5) and CA-irAEs (n=11) treated with anti-PD1. (A) Heatmap of DEGs with scaled cpm expression values (row z-score). (B) Bar plot representing pathway analysis of DEGs with adjusted p value ≤ 0.05. Each term size is listed on the end of each bar. (C) GSEA plot showing the enrichment of “IFNγ response” (NES 1.40, FDR 0.13), “Glycolysis” (NES 1.34, FDR 0.25), “IFNα response” (NES 1.59, FDR 0.01) and “IL2-STAT5 signaling” (NES 0.69, FDR 0.23) gene set. (D) Upstream regulator network from the DEGs of ‘CA-ctrl vs CA-irAEs’ using IPA analysis; red: represents activation in CA-irAEs; green: represents inactivation in CA-irAEs. For DEG analysis, the GLM statistical approach and the thresholds: |FC|≥1.5 and p<0.05 were used.
A universal Treg signature in individuals with irAEs and autoimmune diseases
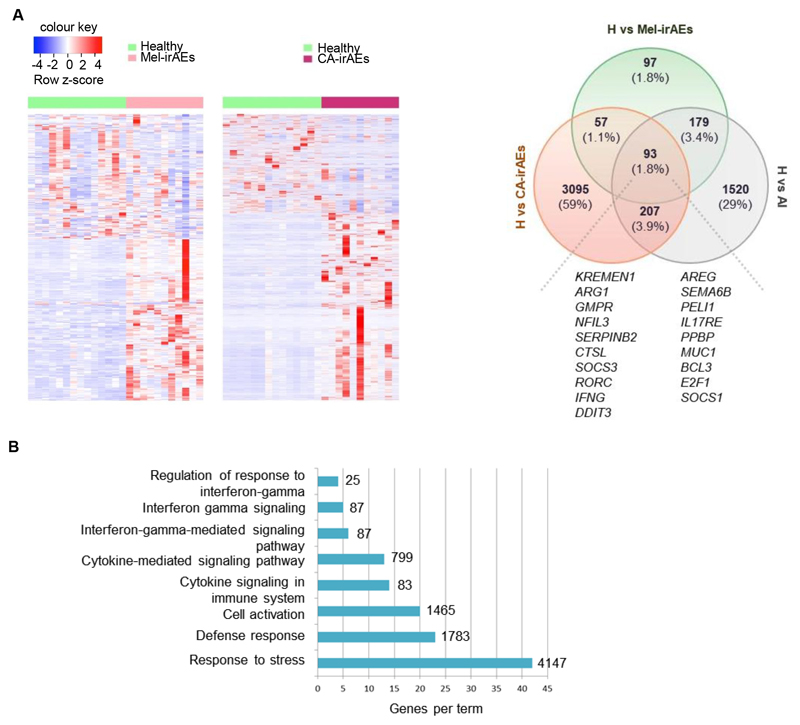
Since peripheral blood remains the most accessible tissue and development of irAEs occurs throughout the body, we reasoned that identification of signatures in circulating Tregs could characterize the development of irAEs irrespective of cancer type. For this reason, we compared gene expression in Mel-irAEs, CA-irAEs, autoimmune and healthy Tregs (Fig. 4A & Supplementary Fig. 2B). Comparison of gene sets among the three disease settings revealed 93 common DEGs (1.8% overlap) comprising a prominent core signature of 19 genes involved in Treg immune-related processes and regulation of inflammatory responses (including SOCS1, SOCS3, IFNG, RORC, IL17RE, ARG1, AREG, and SEMA6B), (Fig. 4A). Pathway analysis illustrated that the 93 shared DEGs were mostly involved in inflammatory processes, with IFNγ-mediated responses, immune activation and response to stress being highly enriched (Fig. 4B). Collectively, our findings reveal a universal Treg inflammatory signature across irAEs in diverse cancer types that is shared with autoimmune Tregs, suggesting that targeting of this signature specifically in Tregs might hold the promise for designing novel therapies to manage irAEs as well as for development of liquid “biopsies”.
Figure 4. Pan-cancer Treg inflammatory signature across irAEs and autoimmunity.
(A) Heatmap of DEGs, with scaled cpm expression values (row z-score), of CD4+CD25+CD127– Tregs isolated from peripheral blood of ‘Healthy (n=14) vs Mel-irAEs (n=11)’ and ‘Healthy (n=14) vs CA-irAEs (n=11)’ (left); and venn diagram showing the overlap between “Healthy vs Mel-irAEs”, “Healthy vs CA-irAEs” and “Healthy vs Autoimmune” DEGs (H vs AI). Selected Treg-related genes are listed to the bottom of the diagram. (B) Bar plot representing pathway analysis of the 93 common DEGs deriving from analysis in panel A, with adjusted p value ≤ 0.05. Each term size is listed on the end of each bar. For IPA and DEG analysis, the GLM statistical approach and the thresholds: |FC|≥1.5 and p<0.05 were used.
Discussion
Herein, we have described an intense inflammatory transcriptional reprogramming of Tregs in the periphery of individuals with cancer developing irAEs following anti-PD1 immunotherapy, with transcripts such as IFNG, STAT1, RORC and STAT3, being highly enriched. This is in agreement with the notion of an increased plasticity or fragility of Tregs in diverse pathogenic settings, including autoimmunity and cancer (13,18). In addition, Tregs from individuals with irAEs demonstrated a metabolic rewiring, which is consistent with the Treg dysfunction observed in individuals with autoimmune diseases (16). The acquired phenotype of Tregs supports the notion that tolerance is disturbed upon ICI immunotherapy in individuals with solid tumors who develop irAEs. The question that remains to be answered is why some individuals develop irAEs while others do not. Development of irAEs maybe occur through similar mechanisms to those that lead to autoimmune diseases, where the balance between effector and regulatory immune responses is driven by diverse parameters such as genetic and environmental factors. Thus, expression of specific HLA alleles, gene polymporphisms and environmental factors such as viral infections, in combination with Treg-mediated breakdown of tolerance could be decisive in determining whether ICI immotherapy leads to the development of irAEs. The notion of the autoimmune nature of irAEs is supported by the observation that TNF blockade, an established therapy in multiple autoimmune diseases, leads to amelioration of irAEs symptoms in experimental mice (19). To this end, a comprehensive analysis of HLA alleles in patients with irAEs may provide solid evidence for genetic susceptibility to developing irAEs upon ICI immunotherapy.
Although Treg accumulation in the TME is a hallmark of tumors, and in several cases correlates with the aggressiveness of tumor cells and impaired responses to immunotherapy, development of irAEs could be mediated by de-regulated immune responses in the peripheral lymphoid compartments. Thus, analysis of blood could shed light on the pathogenic mechanism of irAEs. Given that ICI immunotherapy is administered intraveneously, Tregs, which abundantly express checkpoint molecules (i.e., PD1 and CTLA4) (13), will be targeted systematically, potentially leading to imbalance of peripheral tolerance. Our findings highlight reprogramming of peripheral Tregs upon ICI immunotherapy in individuals with irAEs toward an inflammatory signature. Together, our data suggest peripheral immune responses should be further explored in the effort to map the mechanisms contributing to irAEs development. Identification of mechanisms leading to breakdown of tolerance and irAEs development in ICI-treated cancer patients will deepen our understanding of immune deregulation in cancer and contribute to efficient management of irAEs. It may also provide tools for stratification of patients with increased susceptibility to irAEs.
Supplementary Material
Synopsis.
Regulatory T cells (Tregs) maintain peripheral tolerance. The authors show a common inflammatory signature between Tregs from patients with solid tumors who develop immune-related adverse events and Tregs from patients with autoimmune diseases, suggesting shared underlying mechanisms.
Acknowledgments
We thank Giannis Vatselas for assisting with RNA-seq and for providing technical advice, Anastasia Filia for providing bioinformatics support. Nikos Malissovas for providing technical assistant, Antigoni Pieta for assisting with patient sample collection, Anastasia Apostolidou for assisting with flow cytometry and Dora Togia for financial and administrative management.
Financial support
This research project has received funding from the European Reasearch Council (ERC) under the European Union’s Horizon 2020 research and Innovation programme (grant agreement no 947975) and from the Hellenic Foundation for Research and Innovation (H.F.R.I.) under the “2nd Call for H.F.R.I. Research Projects to support Post-Doctoral Researchers” (Project Number: 166) to T.A. D.T.B., T.A. and P.V. was supported by the ERC under the European Union’s Horizon 2020 research and innovation program (grant agreement no 742390). D.T.B., T.A., and P.V. were supported by the European Union’s Horizon 2020 research and innovation program under grant agreement no 733100.
Footnotes
Authors’ Contributions:
M.G. performed experiments, analyzed data, generated figures and assisted in writing the manuscript. A.B. and A.H. performed experiments and analyzed data. A.K. performed bioinformatic analysis of RNA-seq data. P.K., D.A., R.Z., G.B., E.K., D.M., D.T.B and A.B. performed clinical evaluation of patients and provided human specimens. A.T. assisted with bioinformatics analysis and edited the manuscript. H.G. performed clinical evaluation of individuals, provided human specimens and critically edited the manuscript. T.A. designed the study, performed experiments, analyzed data, generated figures, and wrote the manuscript. P.V. designed and supervised the study, performed data analysis, and wrote the manuscript.
The authors declare no potential conflicts of interest.
References
- 1.Freeman-Keller M, Kim Y, Cronin H, Richards A, Gibney G, Weber JS. Nivolumab in Resected and Unresectable Metastatic Melanoma: Characteristics of Immune-Related Adverse Events and Association with Outcomes. Clin Cancer Res. 2016;22:886–94. doi: 10.1158/1078-0432.CCR-15-1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Michot JM, Bigenwald C, Champiat S, Collins M, Carbonnel F, Postel-Vinay S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer. 2016;54:139–48. doi: 10.1016/j.ejca.2015.11.016. [DOI] [PubMed] [Google Scholar]
- 3.Johnson DB, Reynolds KL, Sullivan RJ, Balko JM, Patrinely JR, Cappelli LC, et al. Immune checkpoint inhibitor toxicities: systems-based approaches to improve patient care and research. Lancet Oncol. 2020;21:e398-e404. doi: 10.1016/S1470-2045(20)30107-8. [DOI] [PubMed] [Google Scholar]
- 4.Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chavez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020;6:38. doi: 10.1038/s41572-020-0160-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, Caterino JM, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714–68. doi: 10.1200/JCO.2017.77.6385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Postow MA, Sidlow R, Hellmann MD. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N Engl J Med. 2018;378:158–68. doi: 10.1056/NEJMra1703481. [DOI] [PubMed] [Google Scholar]
- 7.June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540–7. doi: 10.1038/nm.4321. [DOI] [PubMed] [Google Scholar]
- 8.Bennett CL, Christie J, Ramsdell F, Brunkow ME, Ferguson PJ, Whitesell L, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20–1. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 9.Klocke K, Sakaguchi S, Holmdahl R, Wing K. Induction of autoimmune disease by deletion of CTLA-4 in mice in adulthood. Proc Natl Acad Sci U S A. 2016;113:E2383-92. doi: 10.1073/pnas.1603892113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tanaka A, Sakaguchi S. Regulatory T cells in cancer immunotherapy. Cell Res. 2017;27:109–18. doi: 10.1038/cr.2016.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wahl DR, Byersdorfer CA, Ferrara JL, Opipari AW, Jr, Glick GD. Distinct metabolic programs in activated T cells: opportunities for selective immunomodulation. Immunol Rev. 2012;249:104–15. doi: 10.1111/j.1600-065X.2012.01148.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hatziioannou A, Alissafi T, Verginis P. Myeloid-derived suppressor cells and T regulatory cells in tumors: unraveling the dark side of the force. J Leukoc Biol. 2017;102:407–21. doi: 10.1189/jlb.5VMR1116-493R. [DOI] [PubMed] [Google Scholar]
- 13.Alissafi T, Hatzioannou A, Legaki AI, Varveri A, Verginis P. Balancing cancer immunotherapy and immune-related adverse events: The emerging role of regulatory T cells. J Autoimmun. 2019;104:102310. doi: 10.1016/j.jaut.2019.102310. [DOI] [PubMed] [Google Scholar]
- 14.De Simone M, Arrigoni A, Rossetti G, Gruarin P, Ranzani V, Politano C, et al. Transcriptional Landscape of Human Tissue Lymphocytes Unveils Uniqueness of Tumor-Infiltrating T Regulatory Cells. Immunity. 2016;45:1135–47. doi: 10.1016/j.immuni.2016.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kumagai S, Togashi Y, Kamada T, Sugiyama E, Nishinakamura H, Takeuchi Y, et al. The PD-1 expression balance between effector and regulatory T cells predicts the clinical efficacy of PD-1 blockade therapies. Nat Immunol. 2020;21:1346–58. doi: 10.1038/s41590-020-0769-3. [DOI] [PubMed] [Google Scholar]
- 16.Alissafi T, Kalafati L, Lazari M, Filia A, Kloukina I, Manifava M, et al. Mitochondrial Oxidative Damage Underlies Regulatory T Cell Defects in Autoimmunity. Cell Metab. 2020;32:591–604.:e7. doi: 10.1016/j.cmet.2020.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Newton R, Priyadharshini B, Turka LA. Immunometabolism of regulatory T cells. Nat Immunol. 2016;17:618–25. doi: 10.1038/ni.3466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dominguez-Villar M, Hafler DA. Regulatory T cells in autoimmune disease. Nat Immunol. 2018;19:665–73. doi: 10.1038/s41590-018-0120-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Perez-Ruiz E, Minute L, Otano I, Alvarez M, Ochoa MC, Belsue V, et al. Prophylactic TNF blockade uncouples efficacy and toxicity in dual CTLA-4 and PD-1 immunotherapy. Nature. 2019;569:428–32. doi: 10.1038/s41586-019-1162-y. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.