Abstract
Checkpoint kinase 1 (CHK1) is a key mediator of the DNA damage response that regulates cell cycle progression, DNA damage repair and DNA replication. Small-molecule CHK1 inhibitors sensitise cancer cells to genotoxic agents and have shown single agent preclinical activity in cancers with high levels of replication stress. However, the underlying genetic determinants of CHK1 inhibitor sensitivity remain unclear. We used the developmental clinical drug SRA737 in an unbiased large-scale siRNA screen to identify novel mediators of CHK1 inhibitor sensitivity and uncover potential combination therapies and biomarkers for patient selection. We identified members of the B-family of DNA polymerases (POLA1, POLE and POLE2) whose silencing sensitised the human A549 non small cell lung cancer (NSCLC) and SW620 colorectal cancer cell lines to SRA737. B-family polymerases were validated using multiple siRNAs in a panel of NSCLC and colorectal cancer cell lines. Replication stress, DNA damage and apoptosis were increased in human cancer cells following depletion of the B-family DNA polymerases combined with SRA737 treatment. Moreover, pharmacological blockade of B-family DNA polymerases using aphidicolin or CD437 combined with CHK1 inhibitors led to synergistic inhibition of cancer cell proliferation. Furthermore, low levels of POLA1, POLE and POLE2 protein expression in NSCLC and colorectal cancer cells correlated with single agent CHK1 inhibitor sensitivity and may constitute biomarkers of this phenotype. These findings provide a potential basis for combining CHK1 and B-family polymerase inhibitors in cancer therapy.
Introduction
DNA is routinely subject to exogenous or endogenous sources of damage (1). Therefore, cells have evolved complex surveillance mechanisms, known as cell cycle checkpoints, to tightly regulate the cell cycle and maintain genomic integrity (2,3). In normal cells, these checkpoints are activated in response to DNA damage, so that cell cycle progression can be co-ordinated with the detection and repair of damaged DNA to prevent genomic instability. In cancer cells, these cell cycle checkpoints are frequently deregulated, leading to high levels of replication stress and genomic instability, an enabling characteristic of cancer (4,5).
Replication stress is often described as the slowing or stalling of replication fork progression during DNA synthesis and is caused by a number of different factors (6,7). As a result of replication fork stalling, DNA polymerases become uncoupled from DNA helicases, which continue to unwind the DNA. This DNA unwinding generates stretches of single-strand DNA (ssDNA) that are protected by binding of replication protein A (RPA; 8). RPA recruits a number of replication stress response proteins and is reliant on the ATR-CHK1 pathway to initially stabilise replication forks and delay the onset of mitosis until replication is resumed and completed (7,9). Checkpoint kinase 1 (CHK1) is a serine/threonine kinase and a critical component of several cell cycle checkpoints acting through multiple mechanisms, including replication fork stabilisation and repair (10,11). Cancer cells with defects in cell cycle control, including those with high levels of replication stress, may therefore be more dependent on cell cycle checkpoints and specifically CHK1 for survival (12). Such cancer-specific dependency could be exploited for therapeutic gain, making CHK1 an attractive anticancer target.
Preclinical validation of CHK1 as a cancer drug target, using siRNA and chemical tools, has led to the development of a number of CHK1 inhibitors (13), including the highly selective, potent and orally bioavailable clinical drug candidate SRA737 (14,15), which is undergoing evaluation in phase I clinical trials (www.clinicaltrials.gov; NCT02797977, NCT02797964). CHK1 inhibitors have been shown to potentiate a number of chemotherapeutic agents (16), in particular gemcitabine (17,18), and demonstrate single agent activity in cancer subtypes with high levels of replication stress (17,19,20). Despite these observations, the underlying genetic determinants of sensitivity to clinically relevant CHK1 inhibitors remain unclear and there are currently no clinically validated biomarkers for patient selection. The identification of genes, which are synthetically lethal in combination with CHK1 inhibition, could lead to novel drug combination studies and potential predictive biomarkers of CHK1 inhibitor sensitivity. This may allow the identification of sensitive patient populations and aid the clinical application and evaluation of CHK1 inhibitors.
Synthetic lethality occurs when the simultaneous loss of function of two gene products results in cell death but the loss of either alone does not (21). Using the clinical drug candidate SRA737, we performed a large-scale synthetic lethal siRNA screen in human cancer cells in order to identify those gene products whose loss is synthetically lethal with CHK1 inhibition. Here, we show that siRNAs targeting POLA1, POLE and POLE2, which encode subunits belonging to the B-family of DNA polymerases, are synthetically lethal in combination with CHK1 inhibition in multiple human non-small cell lung cancer (NSCLC) and colorectal cancer cell lines, suggesting a new potential treatment approach. Moreover, combinatorial depletion of the B-family DNA polymerases and small-molecule CHK1 inhibition resulted in increased replication stress, DNA damage and cell death. Pharmacological blockade of both B-family DNA polymerases and CHK1 led to synergistic inhibition of cancer cell proliferation. Furthermore, we show that low POLA1 and POLE protein expression may represent biomarkers for single agent CHK1 inhibitor sensitivity.
Materials and Methods
Cell lines
Human cell lines were obtained from the ATCC (USA), except SKLU1, which was obtained from the HPA (UK). Lines were validated by DNA profiling and confirmed free of Mycoplasma spp. by PCR using the VenorGeM® Mycoplasma PCR detection kit (Minerva Labs, UK). All cell lines were purchased more than a year prior to the experiments and were propagated for less than three months after thawing. All experiments were performed within 10 passages after thawing. NSCLC cell lines were cultured in RPMI 1640 supplemented with 10% foetal bovine serum (FBS) and colorectal cancer cell lines were cultured in DMEM with 5mM L-glutamine, 1% NEAA and 10% FBS. HBEC3-KT cells were cultured in keratinocyte SFM serum-free medium supplemented with human recombinant epidermal growth factor (5 ng/ml) and bovine pituitary extract (30 μg/ml). Cells were incubated at 37 °C in a humidified atmosphere at 5% CO2.
Drugs
SRA737 was synthesised in-house at the ICR (14,15). Aphidicolin and CD437 were purchased from Sigma-Aldrich (USA), MK-8776 from Selleck Chemicals (USA) and gemcitabine from Eli Lilly (USA). Stock solutions of SRA737, MK-8776, aphidicolin and CD437 were prepared in DMSO and a stock solution of gemcitabine was prepared in 0.9 % NaCl solution. All stock solutions were stored at −80 °C.
Cell lysis and western blotting
Total cell lysates were prepared as described (22) except that NP40 was increased to 0.3%. Protein concentration was determined using a bicinchoninic acid (BCA) assay. Cell lysates (10 to 50 μg) were mixed in Laemmli sample buffer and heated for 3 min at 95 °C. Proteins were separated by SDS-PAGE on pre-cast 3 to 8% tris-acetate or 10% tris-glycine gels (Invitrogen, USA), at 150 V for 60 to 90 min. Proteins were then transferred onto PVDF membranes (Merck Millipore, MA, USA) at 100 V for 90 min. Membranes were incubated in 5% milk or 5% BSA in TNT buffer (50 mmol/L Tris (pH 8.0), 150 mmol/L NaCl and 0.1%Tween 20) for 1 hour at and then overnight at 4 °C with primary antibodies: POLA1 (ab31777), POLE (ab134941), POLE2 (ab57298), CHK1 pS345 (CST-2348), CHK1 (SC-8408), RPA32 (ab125681), cleaved PARP (CST-9541) and GAPDH (ab8245). Membranes were then washed and incubated with horseradish peroxidase-conjugated secondary antibodies (mouse/rabbit) for 1 hour. Proteins were visualised using enhanced chemiluminescence (ECL, Pierce Biotechnology, USA) and hyperfilm (GE Healthcare, UK).
Screening with a small interfering RNA library
The Dharmacon druggable genome siRNA library (Dharmacon, GE Healthcare Life Sciences, UK) consists of siRNAs (four pooled siRNAs per gene) which target the expression of 7593 genes that encode known drug targets or potentially druggable proteins. Of these 7593 genes, 6371 (84 %) were screened which included the kinase, phosphatase and drug targets subsets. Dharmacon druggable human genome siRNA library master plates (384-well) containing lyophilised siRNA were re-suspended in RNAse-free water to a final concentration of 5 μM. Using the automated Echo® 550 Liquid Handler (Labcyte, USA), 0.5 μl of siRNA was then transferred from a master plate to six 96-well daughter plates, to give a final assay concentration of 25 nM. siRNAs were introduced into cells by reverse transfection; 50 μl of HiPerFect in Opti-MEM (Invitrogen, USA) was added to each well at a final assay concentration of 0.3% and incubated for 20 min. Cells (50 μl), at optimum seeding density, were then added to each well, incubated for 48 h at 37 °C and treated with SRA737 at the maximum sub-lethal dose (0.4 μM or 0.8 μM for A549 and SW620, respectively) or DMSO vehicle (0.004 % or 0.008% for A549 and SW620, respectively) in 100 μl medium followed by incubation for 84 h at 37 °C. Plates were fixed with 10% trichloroacetic acid and stained with 0.4% sulphorhodamine-B (SRB) in 1% acetic acid. SRB was solubilised with 10 mM Tris base and plates were read at 490 nm using a Wallac Victor 1420 multilabel counter (Perkin-Elmer, USA). Three independent biological repeats of the screen were carried out for each cancer cell line.
Statistical tests: Robust z-score and Z' factor
A Z’-factor was calculated for the entire screen to ensure a wide assay window for hit identification which was maintained (23). Robust Z-scores were calculated to identify hits from the screen. The difference in robust Z-score, with and without SRA737, was calculated for each siRNA in each biological repeat and averaged. Hits were defined as those genes with a difference in robust Z-score greater than or equal to three, with the most significant differences given higher priority. Additional selection criteria for further hit evaluation were (i) a minimal effect of siRNA alone on cell number and (ii) commonality to both cell lines.
Hit validation: siRNA and protein knockdown
Hits were validated using pooled siRNA (Dharmacon, USA) and individual siRNAs (Qiagen, USA). HiPerFect was used at 0.3% (A549, SW620 cells) or 0.2% (Calu-6, NCI-H1975, RKO and HCT116 cells). HiPerFect in Opti-MEM (40 μl) was added to 10 μl siRNA in RNAse-free water in each well and incubated for 20 min. Cells (50 μl) were then added at the optimal seeding density and incubated for 48 h at 37 °C. Plates were treated with SRA737 (0.4 μM or 0.8 μM for A549 and SW620, respectively) or DMSO vehicle (0.004 % or 0.008% for A549 and SW620, respectively) in 100 μl medium and incubated for 84 h at 37 °C. Plates were fixed and stained with SRB and read and analysed as described above.
Knockdown experiments were carried out in 6-well plates using reverse transfection. siRNA and HiPerFect were added at 3 x the final assay concentrations (500 μl/well) and incubated for 20 min. Next, 500 μl of cells were added to each well and incubated for 6 h at 37 °C. Medium (2 ml) was then added to each well to make 1 x siRNA and HiPerFect concentration and cells were incubated for 48 h at 37 °C. Protein expression levels were determined by western blot.
GI50 determinations for CHK1 inhibitors with POLA1, POLE and POLE2 knockdown
Cells were transfected with non-lethal concentrations of siRNA as described above. Plates were treated with 0.01 to 1 μM SRA737 or 0.1 to 10 μM MK-8776 for 84 h at 37 °C. Plates were fixed and stained with SRB and read and analysed as described above. Data were analysed using GraphPad Prism 6.0.
Combination studies
Cells were plated in 96-well plates at optimum seeding densities and incubated for 36 h at 37 °C. Plates were then treated with single agent SRA737 or aphidicolin, or a combination of the two, at a 1:1 ratio of each GI50 (previously determined) and incubated for four doubling times at 37 °C. Plates were fixed and stained with SRB and read and analysed as described above. Data were analysed using the Chou and Talalay method to generate a combination index (CI, 24).
High content analysis
Cells were plated in 96-well clear-bottom opaque-sided plates, incubated for 36 h at 37 °C and subsequently treated with a GI50 concentration of aphidicolin or SRA737 or a combination of the two, followed by incubation for 24 h at 37 °C. Gemcitabine at a final concentration of 200 nM was added as control. Cells were then fixed in 4% formaldehyde for 20 min and permeabilised with 0.25% Triton™ X-100 for 10 min. Next, the fixed cells were blocked with 3% FBS in TNT for 1 h, incubated overnight at 4 °C with a γH2AX antibody (Upstate, USA #05636) at 1:500 and subsequently washed with PBS. The secondary antibody (Alexa Fluor 488 goat anti-mouse IgG, A11001 Invitrogen, USA) was added at 1:1000 for 1 h, cells were then washed, DAPI was added at 1:1000 for 5 min and cells were washed again before adding 200 μl PBS per well. Images were acquired using an IN Cell Analyser 2200 and were analysed using IN Cell Analyser 1000 workstation (version 3.7.2, GE Healthcare Life Sciences, UK).
Immunofluorescence studies
A549 cells were plated onto 22x22mm coverslips (0.16-0.19mm thickness) in 6-well plates at 5x105 cells per well and incubated for 24 hours. Medium was removed and cells were preincubated with 1.9 ml fresh medium for one hour, after which 100μl of each drug was added at the required concentration. Cells were incubated for a further 24 hours. Medium was then removed, cells washed twice with phosphate-buffered saline (PBS) and fixed with 4% paraformaldehyde (PFA) for 15 minutes at room temperature. PFA was removed and cells washed three times with PBS followed by permeabilisation in Tris-buffered saline (TBS) containing 3% Triton-X100 for 15 minutes at room temperature. Permeabilised cells were washed twice in TBS before blocking in TBS containing 0.1% Tween (0.1%) and 2% BSA (TBST/BSA) for 1 hour at room temperature. Cells were incubated in Anti-phospho-Histone H2A.X (Ser139) Mouse primary antibody (EMD Millipore) at 1:2000 in TBST/BSA overnight at 4°C. Cells were then washed three times in TBST/BSA before incubation in Alexa Fluor 488 Donkey anti-Mouse secondary antibody (Invitrogen) at 1:1000 for one hour in the dark at room temperature. Cells were washed twice with TBST/BSA and then once with TBS. During this final TBS wash, two drops of NucBlue™ Live ReadyProbes™ Reagent (Invitrogen) were added per 1ml of TBS for each coverslip and incubated at room temperature in the dark for 20 minutes. After nuclear staining, slides were mounted using ProLong™ Gold antifade Mountant (ThermoFisher) and left to dry overnight in the dark before imaging.
Fixed sample slides were fitted onto an ASI motorised stage (ASI, Eugene, OR) and visualised using an Olympus IX71 microscope with PlanApo 100x OTIRFM-SP 1.45 NA lens mounted on a PIFOC z-axis focus drive (Physik Instrumente, Karlsruhe, Germany), with illumination using LED light sources (Cairn Research Ltd, Faversham, UK) with appropriate filters (Chroma, Bellows Falls, VT). Samples were visualised using a QuantEM (Photometrics) EMCCD camera, and the entire system was controlled with Metamorph software (Molecular Devices). 3D-maximum projections of volume data were calculated from 31 z-plane images, each 0.2 μm apart. Data were subsequently analysed using Autoquant software (MediaCybernetics, Rockville, MD, USA). All 3D image stacks were subjected to blind 3D deconvolution before analysis. Number and intensity of foci were calculated from all foci present within ≥ 30 cells for each sample examined.
Results
An siRNA screen identifies determinants of CHK1 inhibitor synthetic lethality
To identify gene products whose loss is synthetically lethal with CHK1 inhibition, we performed an unbiased, large-scale siRNA screen with the developmental clinical drug SRA737 and the Dharmacon druggable human genome siRNA library in human A549 NSCLC and SW620 colorectal cancer cells. These cell lines were selected due to their relatively low sensitivity to SRA737 (Supplementary Fig. 1), similar growth characteristics (i.e. doubling time and plating efficiency) and ease of transfection. In total, 6371 genes were screened with or without SRA737 in both cells lines, with three independent biological repeats per cell line.
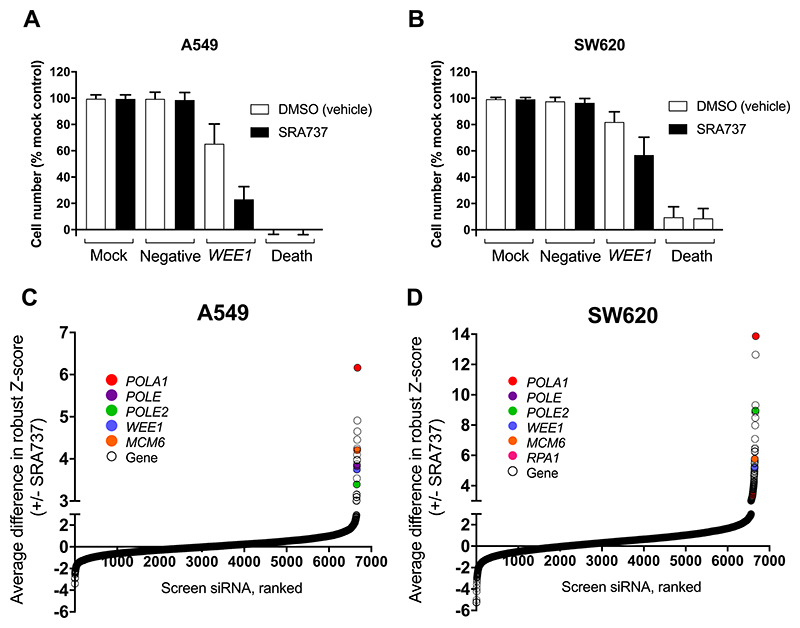
The screen was robust and reproducible, as shown by the fact that the WEE1 siRNA positive control (25) consistently sensitised A549 and SW620 cells to SRA737 and calculated Z’ factors were generally >0.5 (Fig. 1A and B) (23,26). Any plates with Z’-factors <0.5 were rejected from the analysis and re-run. Hits were identified as those genes with a difference in robust Z-score under control (0.004-0.008 % DMSO vehicle) and test (SRA737) conditions of ≥ 3 (Supplementary Tables 1 and 2; Fig. 1C and D).
Figure 1. An siRNA screen identifies knockdown of DNA replication genes as synthetically lethal with CHK1 inhibition in human A549 NSCLC and SW620 colorectal cancer cell lines.
A and B Negative and positive control data for the siRNA library screen in human NSCLC A549 cells (A) and human colorectal cancer SW620 cells (B). Cell number is shown as a percentage of the mock control. Additional controls: Allstars negative siRNA (negative control for off target effects), WEE1 siRNA (positive control for synthetic lethality with SRA737) and death siRNA (positive control for transfection efficiency). C and D The average difference in robust Z-score for A549 (C) and SW620 (D) cells transfected with siRNA and treated with either DMSO (vehicle control) or SRA737. Top hits common to both cell lines are shown as closed coloured circles. A-D Library siRNA (25nm) were introduced into cells by reverse transfection. Mock cells were transfected with lipid only. Cells were then incubated for 48 h followed by treatment with either vehicle (DMSO) control or SRA737 (0.4 or 0.8 μM SRA737, A549 and SW620 respectively) for a further 84 h and then an SRB assay performed. Each cell line was screened with the siRNA library three times (3 biological repeats) to generate the data shown.
A number of the hits with the highest difference in robust Z-score encoded products that were associated with DNA replication (Fig. 1C and D), including MCM6 (mini-chromosome maintenance complex component 6), RPA1 (replication protein A1), POLA1 (DNA polymerase α, catalytic subunit), POLE (DNA polymerase ε, catalytic subunit) and POLE2 (DNA polymerase ε, accessory subunit). POLA1, POLE and POLE2 encode subunits of B-family DNA polymerases, responsible for high-fidelity DNA replication (27), and were identified as hits in both the A549 and SW620 cancer cell lines. POLA1, POLE and POLE2 met all the hit prioritisation criteria (see Materials and Methods section) and were therefore selected for further validation.
Confirmation that knockdown of POLA1, POLE or POLE2 is synthetically lethal with CHK1 inhibition
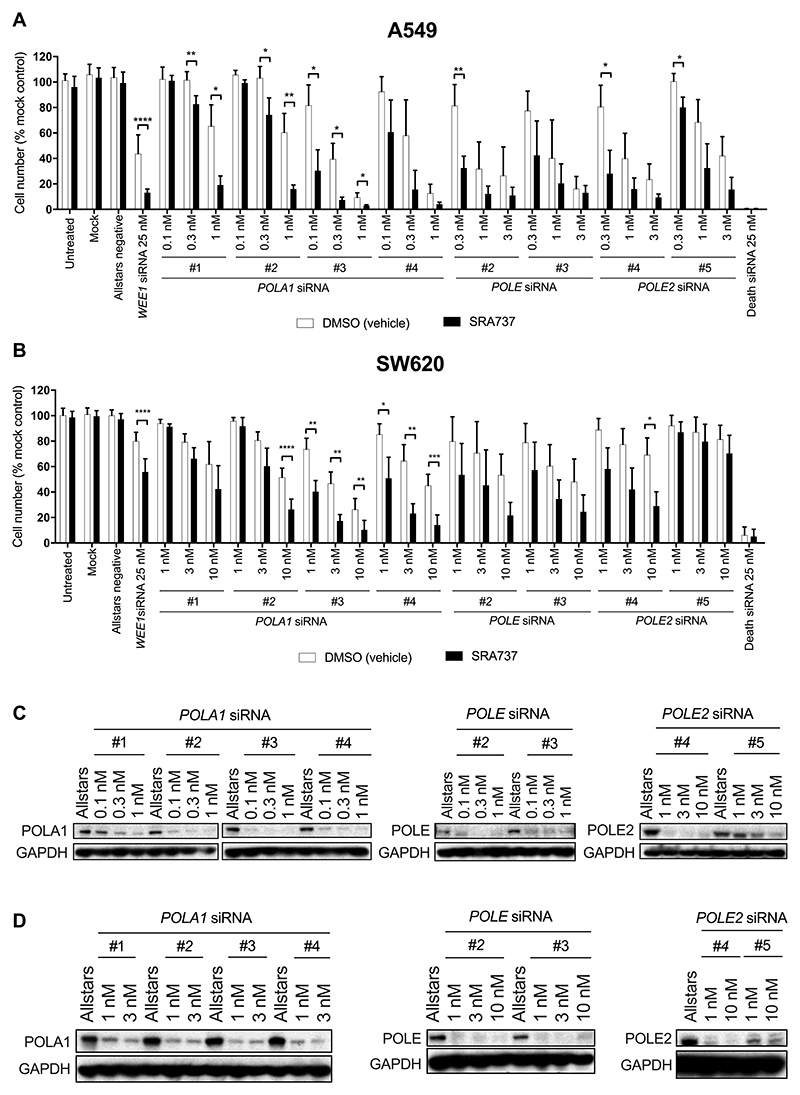
In order to confirm that knockdown of POLA1, POLE and POLE2 is synthetically lethal in the context of pharmacological CHK1 inhibition, cells were transfected with multiple siRNAs, each targeting a different region of the mRNA of each hit gene. Allstars negative (non-targeting) and positive death control siRNAs (Qiagen, USA) were used as controls and to monitor transfection efficiency and toxicity. Both A549 and SW620 human cancer cells were significantly sensitised to SRA737 by siRNA-mediated depletion of POLA1 and POLE2 (Fig. 2A and B), while only A549 cells were also significantly sensitised to SRA737 by transfection with POLE siRNA (Fig. 2A). Depletion of the cognate proteins by the siRNAs was confirmed even at the lowest concentrations tested (Fig. 2C and D).
Figure 2. Knockdown of POLA1, POLE and POLE2 expression is synthetically lethal in combination with CHK1 inhibition.
A and B, The effect of POLA, POLE and POLE2 knockdown on cancer cell line sensitivity to SRA737. Human NSCLC A549 cells (A) and human colorectal cancer SW620 cells (B) were transfected for 48 h with siRNA with a range of siRNA concentrations per cell line as indicated in the figure and then treated with DMSO or SRA737 (0.4 or 0.8 μM SRA737, A549 and SW620 cells respectively) for a further 84 h (n = ≥3 biological repeats) after which an SRB assay performed. Data analysed using the unpaired students t-test *p = <0.05, **p = <0.01. Results are shown as mean ± SD C and D, Western blot analysis showing POLA1, POLE and POLE2 knockdown in A549 (C) and SW620 (D) cells following transfection with 25 nM siRNA. GAPDH was used as a loading control. Blots have been cropped for clarity.
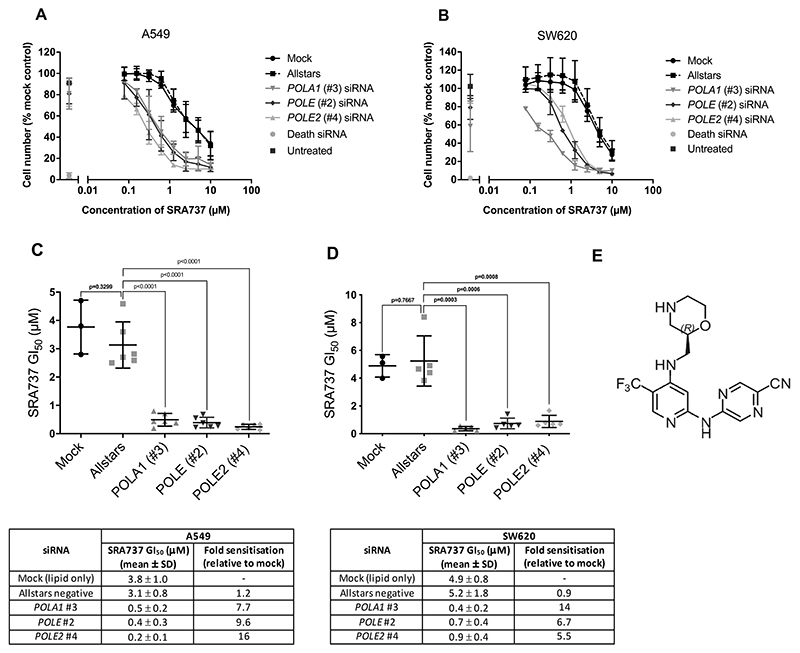
To quantify the extent to which depletion of POLA1, POLE or POLE2 could sensitise cancer cells to SRA737, we determined the GI50 values for SRA737 in both A549 and SW620 cancer cell lines transfected with low concentrations of POLA1, POLE and POLE2 siRNA. Knockdown of POLA1, POLE or POLE2 significantly decreased the GI50 for SRA737 in both cancer cell lines, resulting in between 5- to 16-fold sensitisation to this CHK1 inhibitor drug (Fig. 3A-E). Importantly, we obtained similar results using MK-8776 (SCH 900776, 28), a second CHK1 inhibitor from a different chemotype to SRA737 (15), thus increasing the likelihood that the effects seen represent a genuine and specific synthetic lethal interaction between pharmacological inhibition of CHK1 function and depletion of B-family DNA polymerases (Supplementary Fig. 2A-C).
Figure 3. POLA1, POLE or POLE2 knockdown lowers the GI50 value for SRA737 in human NSCLC A549 and colorectal cancer SW620 cells.
A and B Concentration-response curves for SRA737 following transfection with POLA1, POLE or POLE2 siRNA in NSCLC A549 (A) and colorectal cancer SW620 (B) cells. POLA1 (#3), POLE (#2) and POLE2 (#4) siRNAs were used at 0.1, 0.3 and 0.3 nM respectively in A549 cells and at 1 nM in SW620 cells. Cells were transfected with siRNA for 48 h then treated with DMSO or SRA737 for a further 84 h. Mean cell number (± SD) is shown relative to the untreated mock control (n = ≥3). C and D, Plots of SRA737 GI50 values determined for A549 (C) and SW620 (D) cells, with summary (below the plots) of the mean GI50 values determined for SRA737 and the fold sensitisation to SRA737 as a result of siRNA transfection, relative to GI50 values determined for mock-transfected cells. Bars indicate mean ± SD (n = >3). Data sets were compared using the unpaired students t-test. E, Chemical structure of SRA737.
To confirm that the synthetic lethal interaction that we observed involved an on-target effect of SRA737, we performed CHK1 siRNA knockdown studies in combination with aphidicolin, a tetracyclic diterpene antibiotic which is a potent inhibitor of B-family DNA polymerases α, δ and ε (29–31). We found that A549 cancer cells were indeed sensitised to aphidicolin treatment following siRNA-mediated knockdown of CHK1 (Supplementary Fig. 3A), with reduction in CHK1 protein expression confirmed by western blot (Supplementary Fig. 3B).
Next, we investigated if the synthetic lethal interaction between POLA1, POLE or POLE2 knockdown and CHK1 inhibition could be seen in additional human NSCLC (NCI-H1975 and Calu-6) and colorectal cancer (HCT116 and RKO) cell lines. Encouragingly, all four additional cancer cell lines were significantly sensitised to SRA737 by POLE and POLE2 depletion (Supplementary Figs. 4A-D, 5A and B, 6A and B). Furthermore, NCI-H1975 and Calu-6 cells were significantly sensitised to SRA737 by transfection with POLA1 siRNA (Supplementary Figs. 4A and B). Note, however, that in both HCT116 and RKO colorectal cancer cell lines, transfection with POLA1 siRNA alone markedly reduced cell number (Supplementary Fig. 4C and D), suggesting that these cell lines may have an absolute requirement for POLA1 and/or that knockdown was more efficient in RKO and HCT116 cells versus NCI-H1975 and Calu-6 NSCLC cells (Supplementary Figs. 5 and 6).
Combined pharmacological blockade of CHK1 and B-family DNA polymerases synergistically inhibits cancer cell proliferation
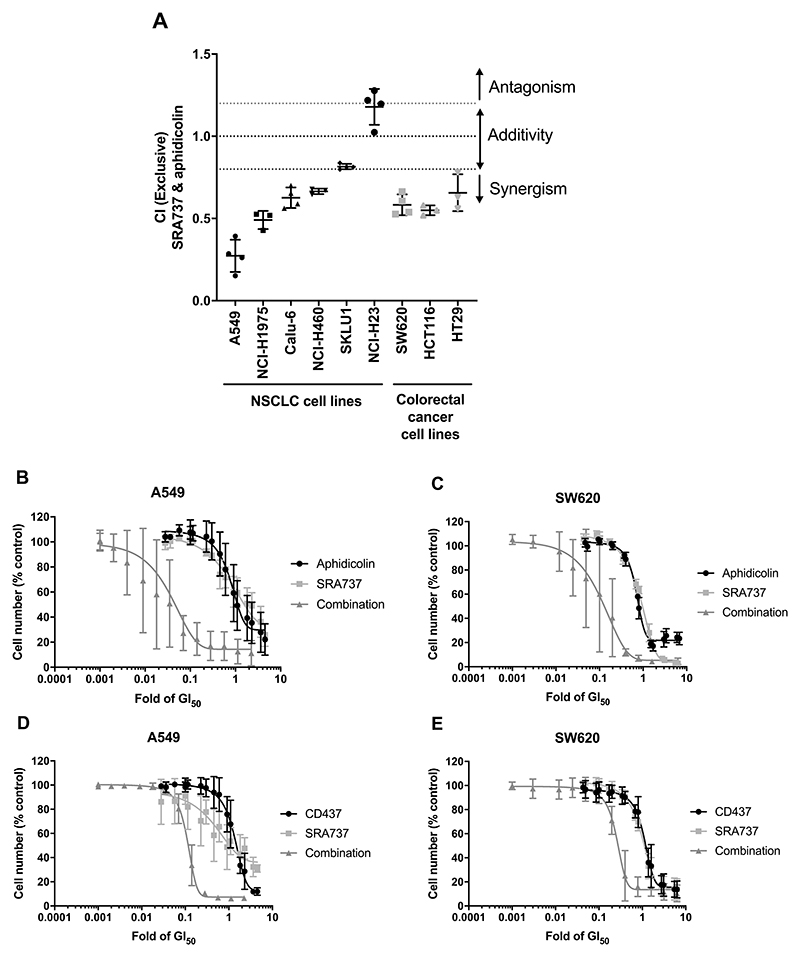
To confirm that the synthetic lethal interaction we observed was due to loss of both B-family polymerase and CHK1 catalytic activity, we adopted an orthogonal validation approach by performing combination studies using small-molecule inhibitors of both CHK1 and B-family DNA polymerases. We tested SRA737 in combination with aphidicolin in a panel of human cancer cell lines. We found that SRA737 and aphidicolin co-treatment was synergistic in 8 out of 9 cancer cell lines tested (Fig. 4A, Supplementary Table 3), including A549 lung and SW620 colorectal cancer cells (Fig. 4B and C).
Figure 4. A synthetic lethal interaction between small-molecule inhibition of both CHK1 and DNA polymerases in human NSCLC and colorectal cancer cell lines.
A Combination index (CI) values for NSCLC and colorectal cancer cell lines treated with 1 x GI50 concentrations of SRA737 and aphidicolin (n = ≥3). CI values calculated using the Chou and Talalay method. Bars indicate mean (± SD) CI values. A one-sided students t-test was used to determine if mean CI values were significantly different from 1 (no interaction), *p = <0.05, **p = <0.01, ***p = <0.001. B and C Concentration-response curves showing the effect on cell numbers of aphidicolin, SRA737 and the combination treatment in NSCLC A549 (B) and colorectal cancer SW620 (C) cell lines (n = 3). D and E Concentration-response curves showing the effect on cell numbers of CD437, SRA737 and a combination treatment in A549 (D) and SW620 (E) cell lines (n = 3). B-E Data are mean (± SD) values of three biological replicates and shown relative to data from cells treated with a vehicle control.
Clinical development of aphidicolin glycinate, a water-soluble analogue of aphidicolin, as an anticancer drug has been hampered by poor pharmacokinetics and toxicity (32). More recently, the structurally distinct retinoid-like agent CD437 has emerged as a DNA polymerase inhibitor with selectivity for α over other B-family polymerases (33). Reassuringly, as with aphidicolin, synergy was also observed between SRA737 and CD437 in both the A549 NSCLC and SW620 colorectal cancer cell lines (Fig. 4D and E). Importantly, the synergy observed between SRA737 plus aphidicolin or SRA737 plus CD437 was comparable to that seen with the developmental clinical combination of SRA737 plus the chemotherapeutic agent gemcitabine (Supplementary Fig. 7A-D and Supplementary Table 3).
In contrast to the results obtained in cancer cell lines, we found no synergy but only an additive effect for SRA737 combined with aphidicolin in the HBEC3-KT normal human bronchial epithelial cell line that is immortalized with CDK4 and hTERT (34, Supplementary Fig. 8). This would suggest that transformed cells may be more susceptible to this drug combination versus their non-transformed counterparts. Overall, our data demonstrate that pharmacological inhibitors of CHK1 and B-family DNA polymerases combine synergistically to inhibit cancer cell proliferation.
Combined depletion of CHK1 and the B-family DNA polymerases increases replication stress, DNA damage and apoptosis
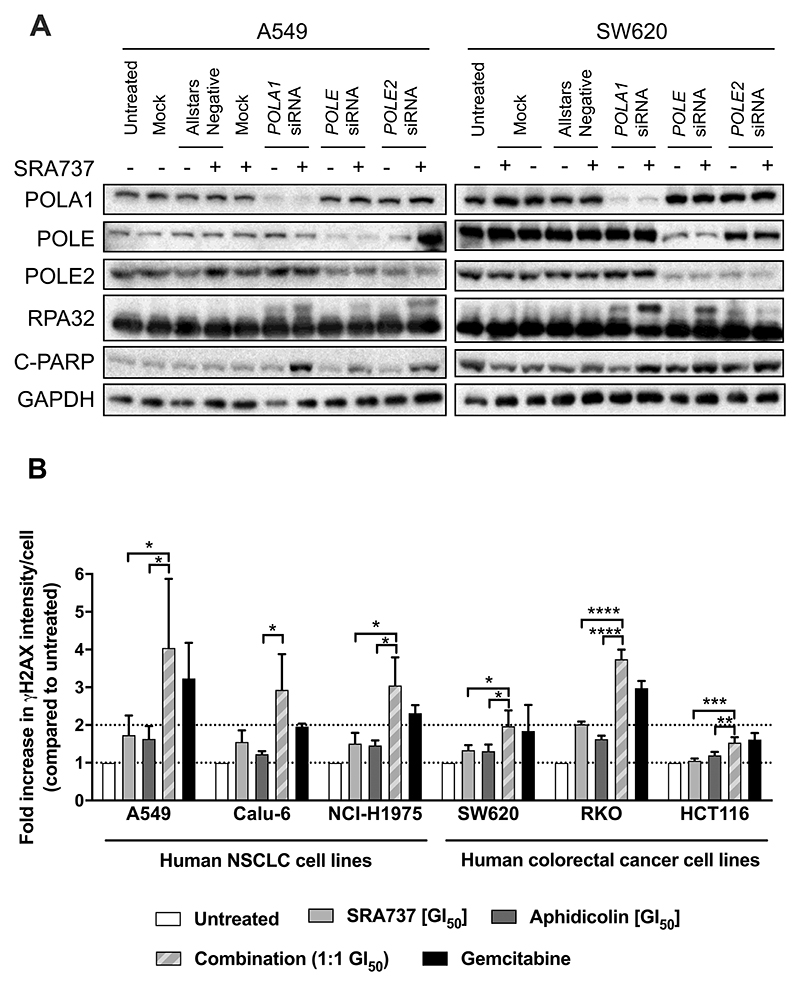
We hypothesised that combined loss of CHK1 and B-family DNA polymerase function would lead to enhanced replication stress, DNA damage and apoptosis. To test this, we examined RPA32 (Replication protein A 32 kDa subunit) which is phosphorylated at multiple sites in response to replication stress, an effect that can be visualised as a band shift on an immunoblot (7, 35–37). We observed an RPA32 band shift after siRNA-mediated depletion of POLA1 alone, indicative of replication stress, but this was not visible with depletion of POLE or POLE2 alone in the A549 or SW620 cell lines (Fig. 5A). However, all combined treatments of SRA737 plus POLA1, POLE or POLE2 siRNA) induced a band shift in RPA32 (Fig. 5A). Furthermore, pharmacological inhibition of B-family polymerases with either aphidicolin or CD437 induced a detectable RPA band shift, which was further enhanced when combined with the CHK1 inhibitor SRA737 (Supplementary Fig. 9).
Figure 5. Combined CHK1 and DNA polymerase inhibition increases replication stress and DNA damage in cancer cells.
A Levels of RPA32 and C-PARP in the lysate of NSCLC A549 and colorectal cancer SW620 cells transfected with 0.1 nM (A549) or 1 nM (SW620) POLA1 #3, POLE #2 or POLE2 #4 siRNA for 48 h prior to 24 h treatment with SRA737. A549 cells were treated with 0.4 μM SRA737 and SW620 cells with 0.8 μM. RPA32 band shift is indicative of replication stress and C-PARP is a marker of apoptosis. GAPDH was used as loading control. Data are representative of two independent experiments. The blot has been cropped for clarity. B Mean (± SD) γH2AX level, relative to that in untreated cells, in cancer cells treated with aphidicolin, SRA737 alone or a 1:1 combination of both agents for 24 h. γH2AX levels were determined by immunofluorescence using an IN Cell Analyser (n = ≥3). Pair-wise comparisons of levels in different treatment groups were carried out using an unpaired students t-test, *p = <0.05, **p = <0.01, ***p = <0.001, ****p = <0.0001.
Persistent and unresolved replication stress can lead to increased DNA damage and cell death (38). We found significantly higher levels of the DNA damage marker γH2AX in 5 out of 6 cancer cell lines co-treated with the CHK1 inhibitor SRA737 and the B-family DNA polymerase inhibitor aphidicolin compared to either treatment alone, as detected by immunofluorescence using an IN Cell Analyser (Fig. 5B). We confirmed that the increase in γH2AX signal was located in foci, which is indicative of direct DNA damage (Supplementary Fig. 10A-F). In addition, all combined treatments of SRA737 plus POLA1, POLE or POLE2 siRNA (apart from SRA737 plus POLE2 depletion in SW620 cells), also caused an increase in the level of cleaved PARP (C-PARP), a marker of apoptosis, when compared to treatment with siRNA or SRA737 alone (Fig. 5A). Taken together, these data indicate that the combined inhibition of the DNA polymerases and CHK1 leads to enhanced replication stress, DNA damage and apoptotic cell death.
POLA1, POLE and POLE2 protein expression correlate with single agent CHK1 inhibitor sensitivity in human NSCLC and colorectal cancer cells lines
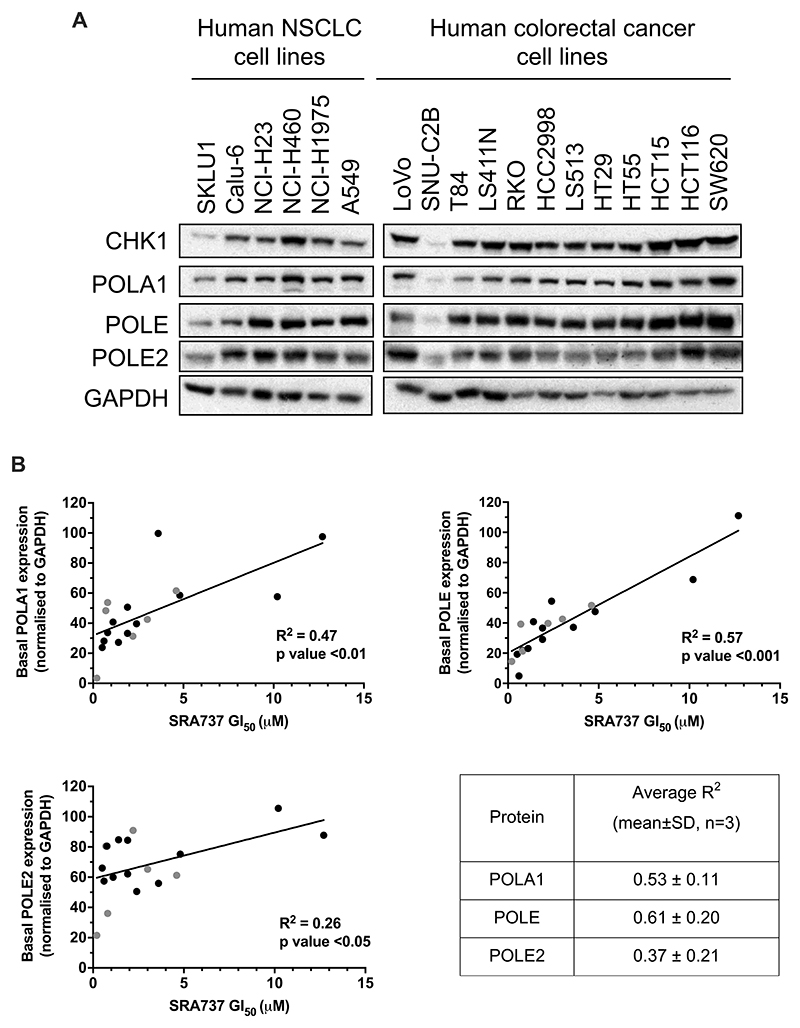
Based on our results with depletion of B-family DNA polymerases, we hypothesised that cancer cells with lower levels of POLA1, POLE and POLE2 protein expression would be more sensitive to CHK1 inhibition compared to those with higher levels of these polymerases. To test this, we measured the level of basal protein expression of POLA1, POLE and POLE2 in the panel of NSCLC and colorectal cancer cell lines (Fig. 6A, genetic status data Supplementary Table 4) and compared these data with GI50 values for single agent SRA737 treatment (Supplementary Table 5). We found a significant correlation between POLA1 and POLE protein expression and SRA737 sensitivity (p ≤ 0.01), and between POLE2 protein expression and SRA737 sensitivity (p < 0.05) (Fig. 6B), suggesting that low basal expression of each of these proteins is indeed indicative of CHK1 inhibitor sensitivity. We also observed that the basal protein expression level of each of these polymerases correlated with the levels of the others (Supplementary Fig. 11A-D), consistent with reports that their expression is co-ordinated (39,40). In contrast, POLA1, POLE and POLE2 basal protein expression did not correlate with sensitivity to the combination of SRA737 and aphidicolin (Supplementary Fig. 12A-C).
Figure 6. Correlation between POLA and POLE basal expression and sensitivity to single agent CHK1 inhibition with SRA737.
A Basal POLA1, POLE and POLE2 expression across a panel of human NSCLC and colorectal cancer cell lines. POLA/E/E2 expression was normalised to the loading control GAPDH. Data are representative of three independent experiments. The blot has been cropped for clarity. B Plots of basal POLA1, POLE and POLE2 protein expression against SRA737 GI50, after western blots were scanned and proteins quantified using Image Studio 2.1 (n =≥3 for GI50). Black and grey dots indicate colorectal cancer and NSCLC cell lines respectively. Estimated regression line shown. Table shows mean (SD) R2 values for POLA1, POLE or POLE2 protein expression versus SRA737 GI50 (n = 3). Data are representative of three independent experiments.
Discussion
CHK1 inhibitors are currently in early clinical trials, both in combination with genotoxic agents and also as single agents (13,16). However, insights into tumour cell vulnerabilities that increase sensitivity to CHK1 inhibitors and biomarkers of this phenotype have been lacking. We used an unbiased large-scale siRNA screen to identify genes which are synthetically lethal when depleted in combination with CHK1 inhibition. We found that the knockdown of members of the B-family of DNA polymerases, POLA1, POLE and POLE2, sensitised human NSCLC and colorectal cancer cells to CHK1 inhibition. This synthetic lethal relationship was identified with the CHK1 inhibitor SRA737 and confirmed with the structurally distinct CHK1 inhibitor MK-8776. Moreover, in addition to validation with multiple siRNAs, the interaction was seen with two chemically distinct B-family DNA polymerase inhibitors, namely aphidicolin and CD437, thus demonstrating the importance of catalytic inhibition. Note that such agreement between two distinct chemotype inhibitors is consistent with best practice for chemical probe use and provides greater confidence, alongside the orthogonal application of genetic depletion as also seen here, that the synthetic lethal interaction involves on-target effects on catalytic function (41). Importantly, we have demonstrated that the synthetic lethal interaction between CHK1 and the B-family DNA polymerases leads to a synergistic pharmacological effect and, moreover, we showed that this occurred in multiple human NSCLC and colorectal cancer cell lines. In addition, the synergy observed is comparable to that seen with the combination of SRA737 plus the chemotherapeutic agent gemcitabine that is currently undergoing clinical evaluation. Observing such robustness in the synthetic lethal interaction is very important and indicates that this effect is real and independent of the range of oncogenic abnormalities in the different cancer cell lines studied. The comparable synergy seen with inhibition of CHK1 plus B-family DNA polymerases and inhibition of CHK1 plus gemcitabine is encouraging, given that gemcitabine is typically considered as the most synergistic genotoxic drug for use in combination with CHK1 inhibition (18).
A recent large-scale analysis has shown that robustness in synthetic lethal interactions is enriched in protein-protein interaction pairs (42). B-family DNA polymerases are responsible for the high-fidelity replication of DNA (27). Several studies have reported a functional relationship between these DNA polymerases and CHK1. Thus CHK1 has been shown to co-immunoprecipitate with DNA polymerase α, indicating a direct interaction between the two proteins (43). Some studies have indicated that DNA polymerase α is required for CHK1 activation (8, 44); conversely, other studies report that depletion of DNA polymerase α by siRNA leads to CHK1 activation (43). Our own data herein show that depletion of the B-family DNA polymerases leads to a robust increased dependence of cancer cells on CHK1 function.
We hypothesised that combined loss of CHK1 and B-family DNA polymerase function leads to enhanced replication stress, DNA damage and apoptosis. This was confirmed by the presence of phosphorylated RPA, γH2AX foci and PARP cleavage, respectively, which were all increased in human cancer cells following partial depletion of B-family DNA polymerases in combination with SRA737 treatment. Moreover, as mentioned, the synergy between SRA737 and aphidicolin or CD437 was comparable to that seen with the combination of SRA737 plus gemcitabine in the NSCLC cell lines tested. Aphidicolin and CD347 both bind to the nucleotide binding site of the DNA polymerase, thus directly inhibiting polymerase activity (31,33). In contrast, gemcitabine inhibits the B-family DNA polymerases indirectly via two mechanisms: (i) depleting the pool of dNTPs available for DNA replication; and (ii) mis-incorporation into replicating DNA which generates lesions capable of stalling the B-family DNA polymerases (45). The shared inhibition of the B-family of DNA polymerases, albeit through a different molecular mechanism, almost certainly underlies the synergy seen with all three agents when combined with CHK1 inhibition.
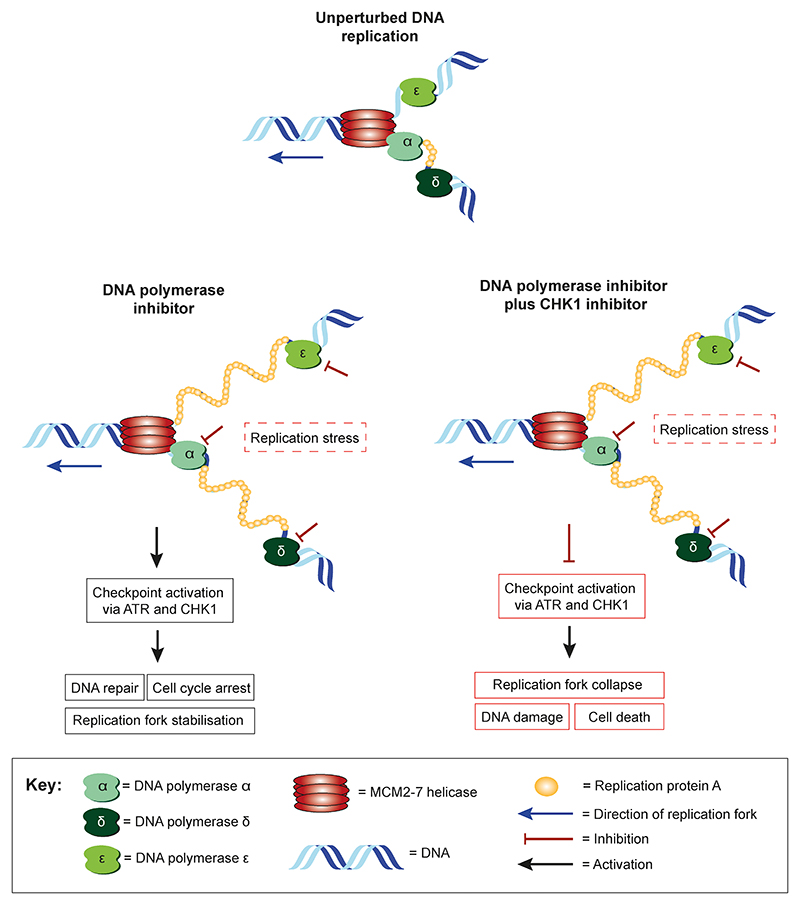
It would be interesting to determine if CHK1 inhibitors are also effective in combination with other agents capable of inducing replication stress. Recently, the neddylation inhibitor MLN4924, which stabilises the re-replication factor CDT1 and potentially increases replication stress, has been shown to enhance the antitumour activity of the CHK1 inhibitor MK-8776, thus further supporting the hypothesis that inducing replication stress sensitises cancer cells to CHK1 inhibitors (46). Based on these findings and our own data, we propose a possible model for how the combined pharmacological inhibition of B-family DNA polymerases and CHK1 may increase replication stress, DNA damage and apoptotic cell death (Fig. 7). This suggests that CHK1 is required to alleviate the replication stress induced by loss of the B-family DNA polymerase function, possibly by stabilising stalled replication forks. Thus, loss of CHK1 when combined with depletion of B-family polymerases may cause intolerable levels of replication stress, DNA damage and eventual cell death. Further work is required to establish if this is the case.
Figure 7. Proposed model showing a potential mechanism for the synthetic lethal interaction between B-family DNA polymerase and CHK1 inhibition.
Schematic illustrating the DNA replication fork under unperturbed conditions, after pharmacological inhibition of B family DNA polymerases, and following combined pharmacological inhibition of B-family DNA polymerase and CHK1.
Germline and somatic mutations in DNA polymerase δ (POLD1) and ε (POLE) have been identified in a number of different cancers, particularly colorectal and endometrial (47). There are over 200 reported mutations in POLE occurring along the entire gene, of which few have been fully characterised and, to date, none have been associated with loss of the protein – perhaps unsurprisingly as DNA polymerase function is essential for viability. Interestingly, the high-mutation rates or hypermutator phenotype associated with colorectal and endometrial cancer have been attributed in part to mutations in the exonuclease domains of the DNA polymerases, leading to loss of their proofreading capabilities (47). We show here low POLA1, POLE and POLE2 protein expression correlate with sensitivity to CHK1 inhibition. This is possibly because cancer cells expressing the B-family DNA polymerases at low levels are more sensitive to increases in replication stress. Thus, low expression of these polymerases at the protein level could potentially be used as biomarkers to predict patient sensitivity to CHK1 inhibitor monotherapy, although this needs to be confirmed with a large panel of cell lines and using clinical samples. In contrast, POLA1 POLE and POLE2 protein expression did not correlate with sensitivity to the combination of SRA737 and aphidicolin, perhaps because the pharmacological inhibition of B-family DNA polymerases α, δ and ε polymerase with aphidicolin overrides low level expression of B-family polymerases as a biomarker for this phenotype.
It is interesting to compare our results to those obtained previously in relevant synthetic lethal screens. Hocke et al. (48) looked for genes that were synthetic lethal with ATR deficiency by screening an siRNA library corresponding to 288 DNA repair genes in a well-characterized genetic ATR knock-in model of DLD1 human colorectal cancer cells as compared to wild type counterparts. They identified as the strongest hit POLD1, which encodes the DNA polymerase δ catalytic subunit. Reasoning that CHK1 is the major downstream effector of ATR they used the staurosporine analog UCN-01 as an inhibitor of CHK1 and showed greater sensitivity in POLD1 -depleted versus control cells. However, UCN-01 is known to be a non-selective inhibitor of CHK1 (49) and follow-up studies in additional colorectal cancer cell lines showed that POLD1 knockdown resulted in a much smaller degree of sensitisation to UCN-01, suggesting that the original finding in DLD1 cells was probably a cell line-specific observation. Moreover, although siRNAs targeting POLD1 were included in our own screen, as well as the screen reported by Davies et al. (25) of siRNAs to cell cycle and DNA repair genes looking for synthetic lethality with the CHK1 inhibitor AR458323 (and which identified WEE1 as synthetic lethal), POLD1 was not identified as a hit in either of these screens. The conflicting results with POLD1 likely reflect differences in the cancer cell lines and/or the CHK1 inhibitors used in the studies. In addition, POLA1 and POLE, both hits in our own screen, were not identified as synthetically lethal hits in the screen by Hocke et. al. (48) since siRNA against these genes proved lethal to DLD1 cells when applied on their own at 10nM. The findings we have reported here indicate that low concentrations of siRNA (less than 0.3 nM) can reduce polymerase levels sufficiently to see synthetic lethality with CHK1, but still leave enough residual activity for cells to survive under control conditions.
In addition to our demonstration that low POLA1, POLE and POLE2 protein expression predispose cells to sensitivity to CHK1 inhibition, previous studies have indicated that amplification and/or elevated expression of MYC family genes may lead to CHK1 inhibitor sensitivity in a number of different cancer types (50, 51). For example, c-MYC overexpression predicts response to single agent treatment with the dual CHK1/CHK2 inhibitor LY2606368 in a panel of small cell lung cancer cell lines. This is thought to be due to the increase in replication stress associated with MYC overexpression (51). In addition, SRA737 has been shown to be effective as a single agent in vivo in mouse models of Eμ-MYC driven B-cell lymphoma and paediatric MYCN-driven neuroblastoma (14,15). Therefore it is conceivable that overexpression of other oncogenes associated with increased replication stress, such as Cyclin E or RAS (52), or replication-associated factors which allow cancer cells to cope with replication stress, such as CDT1 (53), could lead to CHK1 inhibitor sensitivity. Interestingly, a recent study has demonstrated that cyclin F loss predisposes cells to CHK1 inhibition by inducing DNA replication catastrophe (54). Moreover, other direct markers of replication stress, such as pRPA, pCHK1 and ssDNA, may also predict for CHK1 inhibitor sensitivity.
In conclusion, we have shown that both siRNA depletion and pharmacological inhibition of the B-family of DNA polymerases are synthetically lethal in combination with CHK1 inhibition in human lung and colorectal cancer cells. Combination studies revealed that the synergistic interactions between either of two B-family polymerase inhibitors aphidicolin and CD437 with the CHK1 inhibitor SRA737 were comparable to those seen with the promising combination of gemcitabine and SRA737 that is currently undergoing clinical evaluation. Thus, the combination of B-family DNA polymerase inhibition with SRA737 could form the basis, subject to further follow up research, of a potential new future therapeutic approach. The clinical development of the water soluble aphidicolin analog, aphidicolin glycinate, was limited by its rapid clearance, low bioavailability and severe toxicity at the injection site and is no longer being pursued (32). The more recent retinoid-like agent CD437 has not reached the clinic and also inhibits the retinoic acid receptor γ and potentially other off-targets as well as having non-optimized pharmaceutical properties (33). Thus the evaluation of the therapeutic potential of a CHK1 inhibitor in combination with a B-family DNA polymerase inhibitor in animal models and patients must await the emergence of suitable B-family DNA polymerase drug candidates, which the current work encourages. Finally, we found that low POLA, POLE and POLE2 protein expression could potentially be used as biomarkers to predict patient sensitivity to CHK1 inhibitor monotherapy, which could, subject to confirmation and further validation, facilitate the effective clinical use of CHK1 inhibitors as single agents.
Supplementary Material
Statement of Significance.
Findings demonstrate how the therapeutic benefit of CHK1 inhibitors may potentially be enhanced and could have implications for patient selection and future development of new combination therapies.
Acknowledgements
The authors thank Nicky Evans (ICR) for editorial assistance, Dan Mulvihill and Karen Baker (School of Biosciences, University of Kent) for help with confocal microscopy work and both Albert Hallsworth and Vladimir Kirchin (ICR) for their input on this project. PW, MDG, MIW, IC acknowledge program grant support from Cancer Research UK (CRUK grant numbers C309/A11566 and C2739/A22897) and support from The Institute of Cancer Research (ICR), London, UK. PW acknowledges additional grant support from the Wellcome Trust (212969/Z/18/Z) and Cancer Research UK (C35696/A23187) and he is a Cancer Research UK Life Fellow. RFR acknowledges support from the Wellcome Trust (102359/Z/13/Z). MDG and DLC also acknowledge financial support from the University of Kent, Canterbury, UK (cc210/GM02), Kent Cancer Trust (SCP343) and the EB Hutchinson Charitable Trust (A2696). ICR authors acknowledge support from the CRUK Centre at the Institute of Cancer Research.
Footnotes
Conflict of interest statement
IC, MDG, MIW, RFR, PC and PW are current or past employees of The Institute of Cancer Research, which has a commercial interest in the discovery and development of CHK1 inhibitors, including SRA737, and operates a rewards-to-inventors scheme. IC, MDG and MIW have been involved in a commercial collaboration on CHK1 inhibitors with Sareum Ltd and intellectual property arising from the program, including SRA737, was licensed to Sierra Oncology. IC is a consultant for Sierra Oncology, Adorx Ltd, Epidarex LLP and Enterprise Therapeutics Ltd and holds equity in Monte Rosa Therapeutics AG. MDG is on the advisory board of Sierra Oncology, NCL Technology Ventures and Cancer Research UK’s Therapeutic Discovery Laboratories and is a consultant for Azeria Pharmaceuticals, Centauri Therapeutics and Kinsensus Limited. PW was previously a scientific founder, consultant, Scientific Advisory Board member and stock holder of Piramed Pharma (acquired by Roche). He was a scientific founder, consultant, Scientific Advisory Board member, Board member and is a stockholder of Chroma Therapeutics. PW is also a scientific advisory board member for Astex Pharmaceuticals, CV6 Therapeutics and Nextechinvest; has previous or ongoing research funding from Sixth Element Capital, Battle against Cancer Investment Trust, Merck and Astex Pharmaceuticals; is a Non-Executive Board member for Storm Therapeutics, the Royal Marsden NHS Trust and the Chemical Probes Portal; and holds equity in Nextechinvest and Storm Therapeutics.
References
- 1.Ciccia A, Elledge SJ. The DNA damage response: making it safe to play with knives. Mol Cell. 2010;40:179–204. doi: 10.1016/j.molcel.2010.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kastan MB, Bartek J. Cell-cycle checkpoints and cancer. Nature. 2004;432:316–23. doi: 10.1038/nature03097. [DOI] [PubMed] [Google Scholar]
- 3.Bartek J, Lukas J. DNA damage checkpoints: from initiation to recovery or adaptation. Curr Opin in Cell Biol. 2007;19:238–45. doi: 10.1016/j.ceb.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 4.Hartwell LH. Defects in a cell cycle checkpoint may be responsible for the genomic instability of cancer cells. Cell. 1992;71:543–6. doi: 10.1016/0092-8674(92)90586-2. [DOI] [PubMed] [Google Scholar]
- 5.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 6.Lecona E, Fernandez-Capetillo O. Replication stress and cancer: it takes two to tango. Exp Cell Res. 2014;329:26–34. doi: 10.1016/j.yexcr.2014.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zeman MK, Cimprich KA. Causes and consequences of replication stress. Nat Cell Biol. 2014;16:2–9. doi: 10.1038/ncb2897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Byun TS, Pacek M, Yee MC, Walter JC, Cimprich KA. Functional uncoupling of MCM helicase and DNA polymerase activities activates the ATR-dependent checkpoint. Genes Dev. 2005;19:1040–52. doi: 10.1101/gad.1301205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jossen R, Bermejo R. The DNA damage checkpoint response to replication stress: A Game of Forks. Front Genet. 2013;4:26. doi: 10.3389/fgene.2013.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dai Y, Grant S. New insights into checkpoint kinase 1 in the DNA damage response signaling network. Clin Cancer Res. 2010;16:376–83. doi: 10.1158/1078-0432.CCR-09-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Blasius M, Forment JV, Thakkar N, Wagner SA, Choudhary C, Jackson SP. A phospho-proteomic screen identifies substrates of the checkpoint kinase Chk1. Genome Biol. 2011;12:R78. doi: 10.1186/gb-2011-12-8-r78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Forment JV, O’Connor MJ. Targeting the replication stress response in cancer. Pharmacol Ther. 2018;188:155–67. doi: 10.1016/j.pharmthera.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 13.Rundle S, Bradbury A, Drew Y, Curtin NJ. Targeting the ATR-CHK1 axis in cancer therapy. Cancers. 2017;9:41. doi: 10.3390/cancers9050041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Walton MI, Eve PD, Hayes A, Henley AT, Valenti MR, De Haven Brandon A, et al. The clinical development candidate CCT245737 is an orally active CHK1 inhibitor with preclinical activity in RAS mutant SCLC and Eμ-MYC driven B-cell lymphoma. Oncotarget. 2015;7:2329–42. doi: 10.18632/oncotarget.4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Osborne JD, Matthews TP, McHardy T, Proisy N, Cheung KM, Lainchbury M, et al. Multiparameter lead optimization to give an oral checkpoint kinase 1 (CHK1) inhibitor clinical candidate: (R)-5-((4-((Morpholin-2-ylmethyl)amino)-5-(trifluoromethyl)pyridin-2-yl)amino)pyr azine-2-carbonitrile (CCT245737) J Med Chem. 2016;59:5221–37. doi: 10.1021/acs.jmedchem.5b01938. [DOI] [PubMed] [Google Scholar]
- 16.McNeely S, Beckmann R, Bence Lin AK. CHEK again: revisiting the development of CHK1 inhibitors for cancer therapy. Pharmacol Ther. 2014;142:1–10. doi: 10.1016/j.pharmthera.2013.10.005. [DOI] [PubMed] [Google Scholar]
- 17.Walton MI, Eve PD, Hayes A, Valenti MR, De Haven Brandon AK, Box G, et al. CCT244747 is a novel potent and selective CHK1 inhibitor with oral efficacy alone and in combination with genotoxic anticancer drugs. Clin Cancer Res. 2012;18:5650–61. doi: 10.1158/1078-0432.CCR-12-1322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xiao Y, Ramiscal J, Kowanetz K, Del Nagro C, Malek S, Evangelista M, et al. Identification of preferred chemotherapeutics for combining with a CHK1 inhibitor. Mol Cancer Ther. 2013;12:2285–95. doi: 10.1158/1535-7163.MCT-13-0404. [DOI] [PubMed] [Google Scholar]
- 19.Garrett MD, Collins I. Anticancer therapy with checkpoint inhibitors: what, where and when? Trends Pharmacol Sci. 2011;32:308–16. doi: 10.1016/j.tips.2011.02.014. [DOI] [PubMed] [Google Scholar]
- 20.Brooks K, Oakes V, Edwards B, Ranall M, Leo P, Pavey S, et al. A potent Chk1 inhibitor is selectively cytotoxic in melanomas with high levels of replicative stress. Oncogene. 2013;32:788–96. doi: 10.1038/onc.2012.72. [DOI] [PubMed] [Google Scholar]
- 21.Kaelin WG., Jr The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer. 2005;5:689–98. doi: 10.1038/nrc1691. [DOI] [PubMed] [Google Scholar]
- 22.Walton MI, Eve PD, Hayes A, Valenti M, De Haven Brandon A, Box G, et al. The preclinical pharmacology and therapeutic activity of the novel CHK1 inhibitor SAR-020106. Mol Cancer Ther. 2010;9:89–100. doi: 10.1158/1535-7163.MCT-09-0938. [DOI] [PubMed] [Google Scholar]
- 23.Birmingham A, Selfors LM, Forster T, Wrobel D, Kennedy CJ, Shanks E, et al. Statistical methods for analysis of high-throughput RNA interference screens. Nat Methods. 2009;6:569–75. doi: 10.1038/nmeth.1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70:440–6. doi: 10.1158/0008-5472.CAN-09-1947. [DOI] [PubMed] [Google Scholar]
- 25.Davies KD, Cable PL, Garrus JE, Sullivan FX, von Carlowitz I, Huerou YL, et al. Chk1 inhibition and Wee1 inhibition combine synergistically to impede cellular proliferation. Cancer Biol Ther. 2011;12:788–96. doi: 10.4161/cbt.12.9.17673. [DOI] [PubMed] [Google Scholar]
- 26.Zhang J, Chung TDY, Oldenburg KR. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen. 1999;4:67–73. doi: 10.1177/108705719900400206. [DOI] [PubMed] [Google Scholar]
- 27.Hubscher U, Maga G, Spadari S. Eukaryotic DNA polymerases. Ann Rev Biochem. 2002;71:133–63. doi: 10.1146/annurev.biochem.71.090501.150041. [DOI] [PubMed] [Google Scholar]
- 28.Guzi TJ, Paruch K, Dwyer MP, Labroli M, Shanahan F, Davis N, et al. Targeting the replication checkpoint using SCH 900776, a potent and functionally selective CHK1 inhibitor identified via high content screening. Mol Cancer Ther. 2011;10:591–602. doi: 10.1158/1535-7163.MCT-10-0928. [DOI] [PubMed] [Google Scholar]
- 29.Brundret KM, Dalziel W, Hesp B, Jarvis JAJ, Neidle S. X-Ray Crystallographic determination of the structure of the antibiotic aphidicolin : a tetracyclic diterpenoid containing a new ring system. J Chem Soc, Chem Commun. 1972;18:1027–28. [Google Scholar]
- 30.Sheaff R, Ilsley D, Kuchta R. Mechanism of DNA Polymerase a inhibition by aphidicolin. Biochemistry. 1991;30:8590–7. doi: 10.1021/bi00099a014. [DOI] [PubMed] [Google Scholar]
- 31.Baranovskiy AG, Babayeva ND, Suwa Y, Gu J, Pavlov YI, Tahirov TH. Structural basis for inhibition of DNA replication by aphidicolin. Nucleic Acids Res. 2014;42:14013–21. doi: 10.1093/nar/gku1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sessa C, Zucchetti M, Davoli E, Califano R, Cavalli F, Frustaci S, et al. Phase l and clinical pharmacological evalutation of aphidicolin glycinate. J Natl Cancer Inst. 1991;83:1160–4. doi: 10.1093/jnci/83.16.1160. [DOI] [PubMed] [Google Scholar]
- 33.Han T, Goralski M, Capota E, Padrick SB, Kim J, Xie Y, et al. The antitumor toxin CD437 is a direct inhibitor of DNA polymerase alpha. Nat Chem Biol. 2016;12:511–5. doi: 10.1038/nchembio.2082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ramirez RD, Sheridan S, Girard L, Sato M, Kim Y, Pollack J, et al. Immortalization of human bronchial epithelial cells in the absence of viral oncoproteins. Cancer Res. 2004;64:9027–34. doi: 10.1158/0008-5472.CAN-04-3703. [DOI] [PubMed] [Google Scholar]
- 35.Liu S, Opiyo SO, Manthey K, Glanzer JG, Ashley AK, Amerin C, et al. Distinct roles for DNA-PK, ATM and ATR in RPA phosphorylation and checkpoint activation in response to replication stress. Nucleic Acids Res. 2012;40:10780–94. doi: 10.1093/nar/gks849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Treuner K, Findeisen M, Strausfeld U, Knippers R. Phosphorylation of Replication Protein A Middle Subunit (RPA32) Leads to a Disassembly of the RPA Heterotrimer. J Biol Chem. 1999;274:15556–61. doi: 10.1074/jbc.274.22.15556. [DOI] [PubMed] [Google Scholar]
- 37.Ashley AK, Shrivastav M, Nie J, Amerin C, Troksa K, Glanzer JG, et al. DNA-PK phosphorylation of RPA32 Ser4/Ser8 regulates replication stress checkpoint activation, fork restart, homologous recombination and mitotic catastrophe. DNA Repair (Amst) 2014;21:131–9. doi: 10.1016/j.dnarep.2014.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hills SA, Diffley JF. DNA replication and oncogene-induced replicative stress. Curr Biol. 2014;24:R435–44. doi: 10.1016/j.cub.2014.04.012. [DOI] [PubMed] [Google Scholar]
- 39.Bracken AP, Ciro M, Cocito A, Helin K. E2F target genes: unraveling the biology. Trends Biochem Sci. 2004;29:409–17. doi: 10.1016/j.tibs.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 40.Young AP, Nagarajan R, Longmore GD. Mechanisms of transcriptional regulation by Rb-E2F segregate by biological pathway. Oncogene. 2003;22:7209–17. doi: 10.1038/sj.onc.1206804. [DOI] [PubMed] [Google Scholar]
- 41.Blagg J, Workman P. Choose and use your chemical probe wisely to explore cancer biology. Cancer Cell. 2017;32:268–270. doi: 10.1016/j.ccell.2017.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lord CJ, Ryan C. Robust genetic interactions in cancer are enriched in protein-protein interaction pairs. BioRxiv. 2019 doi: 10.1101/646810. [DOI] [Google Scholar]
- 43.Taricani L, Shanahan F, Parry D. Replication stress activates DNA polymerase alpha-associated Chk1. Cell Cycle. 2009;8:482–9. doi: 10.4161/cc.8.3.7661. [DOI] [PubMed] [Google Scholar]
- 44.Michael WM, Ott R, Fanning E, Newport J. Activation of the DNA replication checkpoint through RNA synthesis by primase. Science. 2000;289:2133–7. doi: 10.1126/science.289.5487.2133. [DOI] [PubMed] [Google Scholar]
- 45.Mini E, Nobili S, Caciagli B, Landini I, Mazzei T. Cellular pharmacology of gemcitabine. Ann Oncol. 2006;17(Suppl 5):v7-12.46. doi: 10.1093/annonc/mdj941. [DOI] [PubMed] [Google Scholar]
- 46.Li JA, Song C, Rong Y, Kuang T, Wang D, Xu X, et al. Chk1 inhibitor SCH 900776 enhances the antitumor activity of MLN4924 on pancreatic cancer. Cell Cycle. 2017;17:191–9. doi: 10.1080/15384101.2017.1405194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Barbari SR, Shcherbakova PV. Replicative DNA polymerase defects in human cancers: Consequences, mechanisms, and implications for therapy. DNA Repair (Amst) 2017;56:16–25. doi: 10.1016/j.dnarep.2017.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hocke S, Guo Y, Job A, Orth M, Ziesch A, Lauber K, et al. A synthetic lethal screen identifies ATR-inhibition as a novel therapeutic approach for POLD1-deficient cancers. Oncotarget. 2016;7:7080–95. doi: 10.18632/oncotarget.6857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ruegg UT, Burgess GM. Staurosporine, K-252 and UCN-01: potent but nonspecific inhibitors of protein kinases. Trends Pharmacol Sci. 1989;10:218–20. doi: 10.1016/0165-6147(89)90263-0. [DOI] [PubMed] [Google Scholar]
- 50.Cole K, Huggins J, Laquaglia M, Hulderman CE, Russell MR, Bosse K, et al. RNAi screen of the protein kinome identifies checkpoint kinase 1 (CHK1) as a therapeutic target in neuroblastoma. Proc Natl Acad Sci USA. 2011;108:3336–41. doi: 10.1073/pnas.1012351108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sen T, Tong P, Stewart CA, Cristea S, Valliani A, Shames DS, et al. CHK1 inhibition in small-cell lung cancer produces single-agent activity in biomarker-defined disease subsets and combination activity with cisplatin or olaparib. Cancer Res. 2017;77:3870–84. doi: 10.1158/0008-5472.CAN-16-3409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Macheret M, Halazonetis TD. DNA replication stress as a hallmark of cancer. Ann Rev Pathol. 2015;10:425–48. doi: 10.1146/annurev-pathol-012414-040424. [DOI] [PubMed] [Google Scholar]
- 53.Pozo PN, Cook JG. Regulation and function of Cdt1; A key factor in cell proliferation and genome stability. Genes. 2016;8:2. doi: 10.3390/genes8010002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Burdova K, Yang H, Faedda R, Hume S, Chauhan J, Ebner D, et al. E2F1 proteolysis via SCF-cyclin F underlies synthetic lethality between cyclin F loss and Chk1 inhibition. EMBO J. 2019;38:e101443. doi: 10.15252/embj.2018101443. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.