Abstract
Many studies have reported that patients with psychosis, even before drug treatment, have mildly raised levels of blood cytokines relative to healthy controls. In contrast, there is a remarkable scarcity of studies investigating the cellular basis of immune function and cytokine changes in psychosis. The few flow-cytometry studies have been limited to counting the proportion of the major classes of monocyte and lymphocytes without distinguishing their pro- and anti-inflammatory subsets. Moreover, most of the investigations are cross-sectional and conducted with patients on long-term medication. These features make it difficult to eliminate confounding of illness-related changes by lifestyle factors, disease duration, and long exposure to antipsychotics. This article focuses on regulatory T cells (Tregs), cornerstone immune cells that regulate innate and adaptive immune forces and neuroimmune interactions between astrocytes and microglia. Tregs are also implicated in cardio-metabolic disorders that are common comorbidities of psychosis. We have recently proposed that Tregs are hypofunctional (‘h-Tregs’) in psychosis driven by our clinical findings and other independent research. Our h-Treg-glial imbalance hypothesis offers a new account for the co-occurrence of systemic immune dysregulation and mechanisms of psychosis development. This article extends our recent review, the h-Treg hypothesis, to cover new discoveries on Treg-based therapies from pre-clinical findings and their clinical implications. We provide a detailed characterisation of Treg studies in psychosis, identifying important methodological limitations and perspectives for scientific innovation. The outcomes presented in this article reaffirms our proposed h-Treg state in psychosis and reveals emerging preclinical research suggesting the potential benefit of Treg-enhancing therapies. There is a clear need for longitudinal studies conducted with drug-naïve or minimally treated patients using more sophisticated techniques of flow-cytometry, CyTOF expression markers, and in vitro co-culture assays to formally test the suppressive capacity of Tregs. Investment in Treg research offers major potential benefits in targeting emerging immunomodulatory treatment modalities on person-specific immune dysregulations.
Keywords: Adaptive immune system, FoxP3, Interleukin-6, Psychosis, Regulatory T cells, Schizophrenia, Tregs
Highlights
-
•
Tregs are cornerstone immune cells controlling the innate and adaptive immune balance.
-
•
Tregs regulate astrocytic-microglial neuroimmune interactions.
-
•
Clinical and experimental research suggest that Tregs are hypofunctional in psychosis, which may explain inflammation and glial dysfunction in psychosis .
-
•
Investment in Treg research will help to uncover novel mechanisms in psychosis.
-
•
Treg-enhancing therapies may guide personalised treatments in psychosis.
1. Introduction
It is widely held that inflammation plays an important role in the pathogenesis of psychosis, based principally on the large number of studies reporting increased circulating concentrations of cytokines and other inflammatory markers (Corsi-Zuelli et al, 2020, 2021; Goldsmith et al., 2016). Meta-analyses show that interleukin-6 (IL-6) stands out as the most consistently raised cytokine in the cerebrospinal fluid (CSF) (Orlovska-Waast et al., 2019) and blood (Goldsmith et al., 2016) of patients relative to healthy controls, including drug-naïve first-episode psychosis (FEP) patients (Pillinger et al., 2018; Upthegrove et al., 2014). In remarkable contrast to the extensive cytokine literature, there have been very few studies of multi-parameter flow cytometry to characterise the cellular components of the immune system. Available studies in psychosis consist of cross-sectional investigations conducted mainly in medicated patients, limited in counting the proportion of major classes of monocyte and lymphocytes without distinguishing their pro- and anti-inflammatory subsets (Miller et al., 2013).
In this article, we focus on regulatory T cells (Tregs) to identify fresh ideas for innovation in mechanisms of neuroimmune dysfunction and treatment in psychosis. Tregs regulate immune homeostasis, suppress inflammation, restrain immune responses (Sakaguchi et al., 2008; Shevach, 2009) and, it is increasingly understood, have regulatory interactions with glial cells in the brain (Ito et al., 2019; Pasciuto et al., 2020; Xie et al., 2015). We briefly recapitulate the role of Tregs in the neuroimmune network and summarise findings that stimulated the development of the hypofunctional Treg (h-Treg) hypothesis of psychosis (Corsi-Zuelli and Deakin 2021). Then, we discuss recent discoveries on Treg-based therapies and consider the dynamics between IL-6 and Tregs and treatment implications. The h-Treg hypothesis requires that Tregs from people with psychosis are hypofunctional. In sections 3, 4, we review the eight available studies in detail, discuss the methodological limitations and inferences that can be drawn. We conclude by summarising the challenges and perspectives for Treg research in psychosis with ideas for the design of future studies. For information on the Junior Key Opinion Leader, see Fig. 1.
Fig. 1.

Fabiana Corsi-Zuelli, BSN, MSc, PhD student in Neuroscience at the University of São Paulo, Ribeirão Preto Medical School, Brazil. Fabiana is interested in the interconnection between the immune and central nervous systems, trying to disentangle immune mechanisms of psychosis. She is a member of the Brazilian STREAM study, supervised by Dr Cristina Del-Ben, and investigates the interplay between immune dysregulation and environmental factors in psychosis, unaffected siblings, and community controls. Fabiana completed her MSc in Neuroscience in 2019 with a joint scholarship at King's College London with Dr Valeria Mondelli, studying the interplay between early-life stress, immune mechanisms, and psychosis. She is now investigating the moderating effect of inflammation on the cannabis-psychosis association. She will next tackle the interaction between the immune and endocannabinoid systems and its relevance to psychosis together with Dr Diego Quattrone (KCL). She is also working on the role of inflammation within the psychosis continuum model and how this may be relevant for the transdiagnostic expression of psychosis. Another topic she is interested in is the interaction between the adaptive and innate immune systems. She is particularly interested in Treg-related mechanisms and the intersection with brain, body, and environment that may be relevant to psychosis development. She has been studying and writing about it with her mentor Prof. Deakin (University of Manchester), with whom she developed the Treg-hypothesis of psychosis. She is looking forward to the next steps of her career development in immunopsychiatry, which will include receiving training in immune cell-based research and conducting investigations with Professors Rachel Upthegrove and Nicholas Barnes at the University of Birmingham. Fabiana receives national and international Fellowships from the São Paulo Research Foundation (FAPESP) and has received prestigious awards in recognition of her work from distinguished societies, including the Society for Biological Psychiatry, British Association for Psychopharmacology, International Brain Research, and the Federation of European Neuroscience Societies.
2. Tregs: role in the neuroimmune network and therapeutic implications
2.1. Tregs overview
Tregs are the ‘guardians’ of immune homeostasis, critical in maintaining a balanced innate and adaptive immune function and limiting inappropriate inflammation (Sakaguchi et al., 2008). In the central nervous system (CNS), Tregs regulate homeostatic neuroimmune interactions, especially between astrocytes and microglia (Ito et al., 2019; Pasciuto et al., 2020; Xie et al., 2015). FoxP3, a Forkhead family transcription factor, is the master regulator of Tregs, critical for their development and suppressive capacity (Buckner, 2010). (Epi)genetic loss of Treg homeostasis has been implicated in the pathogenesis of autoimmune disorders (Ohkura et al., 2020; Shu et al., 2017) and common metabolic diseases, such as type 2 diabetes, insulin resistance (Arroyo Hornero et al., 2020; Eller et al., 2011; Paolino et al., 2021; Qiao et al., 2016; Zeng and Chi, 2013) and cardiovascular diseases (Albany et al., 2019; Meng et al., 2016), which are also recognised co-morbidities of psychosis. In addition, Tregs play key roles in healthy pregnancy by promoting maternal tolerance to foetus antigens (Teles et al., 2013). Hypofunctional Tregs during pregnancy is associated with predisposition to obstetric complications, including placental insufficiency, higher risk of infection, inflammation, premature birth (Martini et al., 2020; Rowe et al., 2013), which are well implicated in abnormal foetal neural development (al-Haddad et al., 2019) and considered non-specific risk factors for neuropsychiatric disorders, including psychosis (Davies et al., 2020). Due to space restrictions, for more details, please refer to Fig. 2 and our recent review (Corsi-Zuelli and Deakin 2021).
Fig. 2.
Loss of Treg-mediated immune homeostasis and increased risk of psychosis
Tregs are the “guardians” of immune homeostasis; they control the balance between the innate and adaptive immune systems, regulate neuron-glial interactions and limit inappropriate inflammation. Middle: There are two main populations of Tregs (light-green cells) based on their developmental origin, thymus-derived (tTregs, pink box) and peripherally-induced (pTregs, blue box). tTregs develop from CD4+CD8− T cells (grey cell), while pTregs are generated extrathymically from naïve CD4+T-cells (marsala cells). FoxP3, a Forkhead family transcription factor, is the master regulator of Tregs, critical for their development and suppressive capacity. Tregs may be more prone to FoxP3 destabilisation in psychosis due to polygenic risk variants influencing the cell suppressive capacity directly (pink box) or indirectly via enhanced IL-6 trans-signalling pathway from innate immune cells (light-purple cells, blue box). IL-6 trans-signalling interferes with the generation of newly immunosuppressive pTregs (faint grey arrow with negative sign), generating instead pathogenic effector T (Teff) cells (bold black arrow with a positive sign) (see Fig. 2 for mechanism). Thus, both IL-6 and Teffs-produced pro-cytokines contribute to or sustain a polygenic hypofunctional Treg state (black inhibitory signs). Top: Symptoms of psychosis may arise from aberrant hypofunctional Treg-glial dysregulation. Tregs in the meninges and brain lymphatic system influence glial function through various immune signals; hypofunctional Tregs fail to maintain a balanced interaction between microglia and astrocytes, which together fail to prevent or adapt to dysfunctional neural development and neurotransmission. Bottom: Genetic and epigenetic predisposition to a hypofunctional Treg signature releases the gain of function of myeloid and effector T cells generating a low-grade inflammatory state that sustains an impaired Treg capacity. Breakdown of the Treg homeostasis is associated with several comorbidities of psychosis. For more information, see the main text and Corsi-Zuelli and Deakin (2021). (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
2.2. H-Treg hypothesis of psychosis
The h-Treg hypothesis (Corsi-Zuelli and Deakin 2021) was prompted by recent lines of evidence from our group and others that are not compatible with the prevalent view that microglial inflammation is central to the pathogenesis of schizophrenia. Deakin et al. (2018) found that minocycline, a potent inhibitor of microglial inflammation, was ineffective in recent-onset patients. The findings were followed by meta-analyses of in vivo Positron Emission Tomography (PET) imaging, providing compelling evidence that the translocator protein TSPO (a putative biomarker for inflamed microglia) is not increased but reduced in psychosis (Plavén-Sigray et al, 2018, 2020), as we had observed in several brain regions of antipsychotic-free patients (Conen et al., 2020). We proposed that microglia are driven into a non-inflamed synaptic pruning state by overactivity of TGF-β mediated astroglial restraint (Conen et al., 2020). We discovered this idea was highly compatible with multiple large-scale transcriptome-wide studies that we reviewed; they consistently reported no change or decreased microglial gene expression but increased astroglial expression in schizophrenia (Gandal et al., 2018a, 2018b; González-Peñas et al., 2019; Ramaker et al., 2017; Toker et al., 2018).
Treg hypofunction provided the missing link between low-grade systemic inflammation in psychosis and central glial dysregulation. This insight was triggered by the observation that low-dose methotrexate had a significant antipsychotic effect (Chaudhry et al., 2020); low-dose methotrexate is standard of care in autoimmune disorders through its ability to restore Tregs and their control of the immune response (Avdeeva et al., 2020; Cribbs et al., 2015; Peres et al, 2015, 2018a). Recent findings had revealed that Tregs have a close regulatory interaction with astrocytes (topic 2.1), and we deduced that methotrexate's efficacy might result from restoring impaired Treg control of astrocytic disinhibition in psychosis. Our discoveries were unexpectedly corroborated by the latest Psychiatric Genetics Consortium report, which identified risk variants of genes associated with the functional capacity of Tregs (FoxP1 and Furin) (Consortium et al., 2020). In the following sections, we briefly discuss novel emergent approaches to enhancing Treg function in neurological and autoimmune disorders and how these are helping to disentangle mechanisms underlying low-grade inflammation and neuroimmune dysfunction that could be relevant to psychosis in the future.
2.3. Tregs control systemic inflammation and neuroimmune dysfunction
There has been increasing interest in recovering the functional capacity of Tregs or transfusing competent Tregs to restore systemic immune control and glial dysfunction. For example, in experimental neuroimmune models, DEpletion of REGulatory T cells (a transgenic murine model that allows highly specific depletion of Tregs using diphtheria toxin) induces astrogliosis (Ito et al., 2019; Krämer et al., 2019) with activation of brain IL-6/STAT3 signalling that can be restored by adoptive cell transfer (ACT) of competent Tregs or intraventricular administration of products derived from competent brain Tregs (amphiregulin) (Ito et al., 2019). In human research, current clinical trials are testing ACT of competent Tregs in several autoimmune and neuroimmune diseases, such as graft-versus-host disease, type-1 diabetes, Crohn's disease, amyotrophic lateral sclerosis, multiple sclerosis (Duggleby et al., 2018; Gliwiński et al., 2017), and most recently COVID-19 (Wang et al., 2021).
In experimental autoimmune encephalomyelitis, Krienke et al. (2021) recently designed a BioNTech mRNA vaccine that codes for disease-related autoantigens optimised for systemic delivery to splenic dendritic cells in the absence of costimulatory signals to treat and prevent the disease. Mice immunised with the newly designed mRNA vaccine had systemic activation of antigen-specific Tregs that suppressed disease-promoting autoreactive T-cells and related cytokines (IFN-γ, TNF-α, IL-6, IL-2, IL-17) without causing systemic immune suppression. This reduced brain and spinal cord demyelination. The vaccine benefit was abolished by antibodies that interfered mostly with Treg functioning (mainly CTLA-4 but also PD-1), as shown by flow cytometry and single-cell RNA sequencing. The study contributes to knowledge on Tregs and neuroimmune dysfunction, but the definition of optimal disease-specific autoantigen is a barrier to clinical translation.
Most recently, Treg research and Treg-based therapies have been expanded to neuropsychiatric disorders such as autism and schizophrenia, in animal models. Xu et al. (2021) induced immune activation in pregnant mice by injection of Toxoplasma gondii antigen and reported abnormally reduced social interaction in the adult male offspring associated with a number of brain changes, including overproduction of IL-6 by astrocytes and abnormal hippocampal neuronal connectivity with no evidence of microglial inflammation. They showed that these changes were mediated by systemic T-cell imbalance (low Tregs; high Th1, Th17). Remarkably, ACT of Tregs from control mice reversed the behavioural and brain changes, with maternal pathogen-activated Tregs especially effective and capable of infiltrating the brain parenchyma. In another study, You et al. (2020) used amphetamine to induce schizophrenia-like behavioural changes in adult mice and considered human umbilical cord-derived mesenchymal stem cells (hUC-MSC) as a Treg-enhancing therapy. The authors showed that a single injection of hUC-MSC reversed aberrant behavioural changes and the systemic pro-inflammatory TNF-α profile. The principal mechanism of hUC-MSC was upregulation of the Treg-related transcriptional factor FoxP3 mRNA in the blood and mesenteric lymph node. Given that hUC-MSC are usually limited in reaching the brain, the authors assume restoration of peripheral Tregs from a single hUC-MSC infusion as sufficient for long-term reversal of behavioural and cytokine changes. In human research, MSC transplants are being tested in children with autism (Chez et al., 2018; Riordan et al., 2019), Crohn's disease (Zhang et al., 2018), diabetes (El-Badawy and El-Badri, 2016), chronic ischemic heart disease (He et al., 2020), and recently in COVID-19 (Maeurer et al., 2021).
There are many challenges to translating these lines of treatment to complex multifactorial disorders, such as psychosis. However, the studies are striking examples of the novel and determining role of Tregs in controlling not only systemic inflammation but also disordered glial functions. Most importantly for this review, Xu et al. (2021) revealed for the first time that restoring impaired Tregs capacity unexpectedly corrects neuroimmune dysfunction that resembles those of clinical schizophrenia discussed in section 2.2. This raises critical translational questions about the phenotype and functional capacity of Tregs in patients with psychosis, their correlations with clinical symptoms and with core inflammatory markers of the disease such as IL-6, and identification of potentially repurposable drugs with Tregs-boosting effects, discussed in the following sections.
2.4. Treg-IL-6 interplay as a novel mechanistic pathway in psychosis?
The functional implications of consistently raised IL-6 in the blood and CSF of patients with psychosis, including drug-naïve patients, are unclear. IL-6 is a pleiotropic cytokine that can signal via both membrane-bound (classic) and soluble (trans) receptors. The classic-signalling is usually associated with its regenerative and anti-inflammatory properties, whereas trans-signalling is frequently associated with its pro-inflammatory actions (Calabrese and Rose-John, 2014). Mendelian randomisation, which uses genetic variants that regulate the levels/activity of a biomarker, suggests a causal role of genetic variants related to IL-6 trans-signalling in psychosis (Hartwig et al., 2017). This indicates that associations are unlikely to be a consequence of confounders, such as lifestyle factors.
It is possible that increased IL-6 trans-signalling contributes to or sustains a hypofunctional Treg state in psychosis (Fig. 3). Extensive preclinical and clinical research in autoimmune disorders suggests that the suppressive capacity of Tregs depends both on the genetic makeup and external cytokine milieu (Attias et al., 2019; Li and Zheng, 2015; Ohkura et al., 2020; Sakaguchi et al., 2008; Shu et al., 2017). Genetic and epigenetic predisposition to hypofunctional Tregs releases increased function of pro-inflammatory myeloid and effector T-cells. During this inflammatory scenario, IL-6 has a well-studied role, particularly via the trans-signalling pathway, in interfering with de novo generation of functional Tregs by destabilising FoxP3 expression from naïve T-cells (Dominitzki, 2007) and skewing the T-cell fate towards pro-inflammatory Th17 RORc+ (Bettelli et al., 2006; Dhuban et al., 2019; Gao et al., 2012; Komatsu et al., 2014).
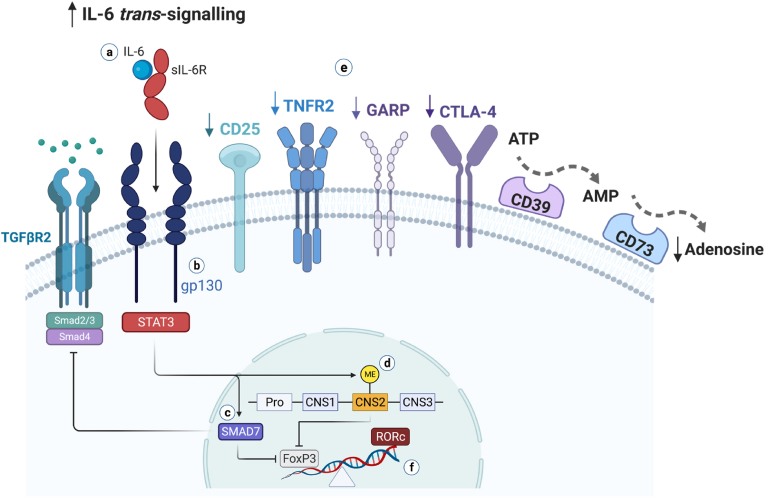
Fig. 3.
Proposed mechanisms for IL-6-sustained hypofunctional Treg state in psychosis.
We summarise mechanisms through which IL-6 might interfere with the immunosuppressive function of Tregs in psychosis (for references, see the main text). a) The IL-6 pro-inflammatory pathway, named trans-signalling, is formed by IL-6 bound to its naturally occurring soluble receptor (sIL-6). The IL-6/sIL-6 complex associates with two molecules of the transmembrane signal transducer glycoprotein (gp130), activating the JAK/STAT3 pathway. b) It has been shown that subpopulations of human Tregs that are hypofunctional to suppress inflammation and related cellular mediators express more gp130; this renders more plasticity to Tregs, turning them more prone to FoxP3 destabilisation upon IL-6/sIL6R biding and conversion to pathogenic T cells under inflammatory conditions. c) IL-6 trans-signalling upregulates the expression of SMAD7, a negative regulator of TGF-β signalling, which inhibits the expression of FoxP3 and the generation of functional Foxp3+ Tregs. d) Complete demethylation of the conserved non-coding sequence 2 (CNS2), a CpG-rich island in the FoxP3 locus (also known as Treg cell-specific demethylated region), is crucial to Treg stability and suppressive function. IL-6-STAT3 induces CNS-2 methylation, resulting in FoxP3 destabilisation. e) Hypofunctional Tregs are deficient in expressing molecules that are implicated in their functional capacity, such as CD25, TNFR2, GARP, CTLA-4, and the adenosine-producing ectoenzymes CD39 and CD73 (see also Table 4 in the main text); adenosine is a potent anti-inflammatory molecule. The hypofunctional Treg state restrains the regulation of pro-inflammatory innate and adaptive forces. f) By inhibiting or destabilising FoxP3 expression, mechanisms c and d skew the T cell fate towards pathogenic T cells, triggering the expression of RORc, the Th17-transcriptional factor that is the polar opposite to FoxP3+ Treg differentiation. Such IL-6-induced FoxP3 destabilisation mechanisms are also likely relevant in naïve T cells, culminating in a self-sustaining state of mild peripheral immune activation with less generation of new competent Tregs. Figure adapted fromCorsi-Zuelli and Deakin (2021).
Interestingly, pharmacological drugs that inhibit IL-6 can restore Tregs in blood. For example, in rheumatoid arthritis (RA) patients, tocilizumab (anti-IL-6R antibody that inhibits IL-6 pro- and anti-inflammatory pathways) significantly increased blood Treg proportions that correlated with a better response to treatment and clinical state (Kikuchi et al., 2015; Li et al., 2016; Samson et al., 2012). Sarantopoulos et al. (2014) showed that tocilizumab increased blood Tregs in patients soon after the first infusion, an effect sustained over at least a year. In another clinical study, tocilizumab restored the balance between transcriptional factors of Tregs (FoxP3) and Th17 (RORc) (Tada et al., 2016). Thiolat et al. (2014) conducted a mechanistic study in RA patients and rodent models to study the effect of anti-IL-6R antibody on Tregs. They found that mice treated with an anti-IL-6R antibody (MR16-1) showed higher frequencies of Tregs that expressed on their cell surface the ATP-converting enzyme CD39 in both their lymph nodes and spleen. In patients, tocilizumab expanded the percentage of functional CD39+ Tregs in the blood of responders versus non-responders as early as a month of treatment. In an experimental in vitro assay, blood CD39+ Tregs from responder patients were functionally capable of suppressing the proliferation of pro-inflammatory T cells. Both in the animal and human studies, the results were not a consequence of changes in Th17 frequencies, suggesting that the mechanistic effect of IL-6 inhibition was primarily on the Treg machinery. Ongoing studies are investigating the inhibition of IL-6 pathways in psychosis.
IL-6 blockade is not the only approach with indirect Treg-boosting effects. Others tested for such purpose include mTOR inhibitors (rapamycin, metformin), low-dose IL-2, TNFR2 agonists, rituximab, vitamin D, and others (Copsel et al., 2020; Eggenhuizen et al., 2020; Fisher et al., 2019; Göschl et al., 2019; Rosenzwajg et al., 2019; Sharabi et al., 2018). Flow-cytometry, CyTOF and co-culture assays of Tregs during clinical trials could advance knowledge on novel mechanisms in psychosis, which we discuss in topic 4.
3. Reviewing clinical studies of cellular immune regulation in schizophrenia
3.1. Overview
We identified eight Treg studies. The great majority were cross-sectional studies conducted with medicated patients with schizophrenia. Only one follow-up study included drug-naïve first-episode schizophrenia patients. Two studies tested Tregs in vitro. Five studies investigated associations between blood Tregs and symptom domains. We discuss the findings in the following sections. The study details are summarised in Table 1, Table 2, Table 3 and Glossary.
Table 1.
Study details.
| Reference | Centre | Design | Sample age (range) | Patients Number |
Comparison group Number and details |
Patients’ details | Medication | Treg markers | Other immune markers | Cytokines/other markers |
|---|---|---|---|---|---|---|---|---|---|---|
| Maxeiner et al. (2009) | Ulm Germany | Cross-sectional | 14-85 | 17 | 28 (not healthy controls) Patients with neurological disorders7 Meningitis 5 Chronic Inflammatory 16 Non-Inflammatory |
Pooled sample of psychiatric patients 12 Schizophrenia spectrum (F20-25) (duration of illness range: 1-14 years) 5 Affective spectrum (F30-33) (duration of illness range: 1-23 years) |
Not mentioned |
Membrane CD4, CD25, CD127, CD69 |
Membrane CD4, CD8, CD25, CD45RO |
Albumin, IgG, IgA, IgM |
| Drexhage et al. (2011) | Rotterdam Netherlands | Cross-sectional Case-control (matched) |
17-39 | 26 | 26 age- and sex-matched Hospital staff, medical students |
Recent-onset (0-3 years of illness) acutely psychotic in-patients with schizophrenia |
All medicated Median 42mg Haloperidol equivalent |
Membrane CD4, CD25 Intracellular FoxP3 |
Membrane CD3, CD4, CD25, CD45RO Intracellular IFN-γ, IL-4, IL-17A |
IFN-γ, IL-17, IL-10, IL-6, IL-4, TNF-α, CCL2, IL5, IL1-β, PTX3 (serum cytometric bead array) TGF-β and sIL-2R (sCD25) (ELISA) |
| Fernandez-Egea et al. (2016) | Cambridge UK | Cross-sectional Case-control (matched) |
18-50 | 18 | 18 healthy controls Age-, sex-, BMI-, smoking-matched, zero illegal drug use |
Chronic, treatment-resistant schizophrenia (mean duration of illness: 18 years) | 17 Clozapine (mean dose: 316mg/day) 1 Risperidone (6mg/day) |
Membrane CD3, CD4, CD25, CD127 HLA-DR, CD45RA |
73 Cell subsets∗ (T, B, myeloid) ∗Human Immune Phenotyping Consortium |
NA |
| Kéri et al. (2017) | Budapest Hungary | Follow-up (8 weeks) Case-control (matched) |
Mean (SD) SCZ 26.7 (7.0) Healthy control 26.4 (5.5) |
35 | 30 healthy controls Age-, sex-, and education-matched |
Drug-naïve, first-episode schizophrenia (mean duration of illness with no treatment: 8 months) | At follow up (8-week treatment) 24 Olanzapine (10-25mg/day) 11 Risperidone (2-6mg/day) |
Membrane CD4, CD25 Intracellular FoxP3 |
Total leucocyte counts CD14+monocytes CD3+CD4+CD25+ activated T cells |
TLR (1, 2, 4, 5, 6) expression in monocytes TLR (2, 5) expression in activated T cells and Tregs |
| Counotte et al. (2018) | Leiden Netherlands | Experimental, random, cross-over Virtual reality controlled experiment |
18-35 | 52 | 80 Pooled sample of ‘Low psychosis liability’ 46 healthy controls 34 unaffected siblings of psychosis patients |
Pooled sample of ‘High psychosis liability’ 38 Recent-onset (up to 5 years) 14 Ultra-high-risk psychosis |
Ultra-high risk 9 antidepressant 2 mood stabilizer 2 stimulants 2 benzodiazepine Recent psychosis 24 antipsychotics 5 antidepressant 1 mood stabiliser 1 stimulants 9 benzodiazepine Sibling 1 lamotrigine (very low dose) |
Membrane CD4, CD25 Intracellular FoxP3 |
CD4+T, CD8+ T NK, B cells, monocytes Membrane CD45, CD3, CD4, CD8, CD19, CD14, CD56, CD15 T (Th1, Th2, Th17) Membrane CD45RO, CD3, CD25, CD4 Intracellular IFN-γ, IL-4, IL-17A |
NA |
| Kelly et al. (2018) | Maryland USA | Cross-sectional | 18-64 | 26 | 17 healthy controls | Schizophrenia/schizoaffective disorder Clinically stable - using the same antipsychotic treatment for 30 days prior inclusion |
9 Clozapine 7 Second generation 10 First generation 9 Antidepressants 8 Anxiolytics |
Membrane CD4, CD25, CD45RA Intracellular FoxP3 |
White blood cell count (total neutrophils, lymphocytes, monocytes eosinophils, basophils) CD3+ and CD4+T Membrane CD3, CD4 |
NA |
| Akkouh et al. (2020) | Oslo Norway | 1.Experimental Human iPSC-derived astrocyte 2. Cross-sectional Whole blood mRNA for Treg markers |
18-65 | 3 iPSC 484 mRNA |
3 iPSC healthy controls 472 mRNA |
iPSC: Chronic Schizophrenia mRNA: Schizophrenia spectrum (353 schizophrenia, 41 schizophreniform, 90 schizoaffective) |
All medicated | CD4+CD25+ CCR6+Tregs (in vitro assay) |
NA | Whole blood mRNA CCR6 and FoxP3 (Treg-related) |
| Sahbaz et al. (2020) | Istanbul Turkey | Cross-sectional Case-control (matched) |
18-65 | 40 | 40 administrative hospital staff age, sex, ethnicity, BMI, education, smoking status-matched | Stable chronic schizophrenia (no change in medication dosage in the past 6 months, no hospitalization in the past 1 year) Mean illness duration: 17.5±10.2 years |
All medicated 7 Clozapine 11 Other atypical 8 Typical 14 Atypical (injection) 21 Typica/Atypical combined Average cumulative chlorpromazine equivalence 713.35±430.45mg |
Membrane CD4, CD25 Intracellular FoxP3 |
Monocyte, NK, B and T cells Membrane CD14, CD3, CD4, CD8, CD19, CD20, CD16, CD56, CD25 |
IL-2, IL-4, IL-6, IL-10, IFN-γ, TNF-α, IL17A Measured in culture supernatant and plasma (cytometric bead array) |
iPSC: Induced Pluripotent Stem Cell; SCZ: Schizophrenia; SD: standard deviation.
Table 2.
Immune cells results.
| Reference | Results |
Confounding | Notes | |||
|---|---|---|---|---|---|---|
| Tregs | Other immune cells | Subgroups | Cytokines/Other markers | |||
| Maxeiner et al. (2009) | Psychiatric patients vs Neurological patients Peripheral blood NS CD4+CD127dim CD4+CD25+CD127dim CD4+CD69+CD127dim CSF High CD4+CD127dim: (Higher vs meningitis) |
Psychiatric patients vs. Neurological patients Peripheral Blood Low CD4+CD25+ activated T cells (Lower vs meningitis) NS CD4+, CD8+, CD4+CD45RO+, CD8+CD45RO+, CD4+CD69+, CD4+CD25+CD69+ CSF NS CD4+, CD8+, CD4+CD45RO+ CD8+CD45RO+, CD4+CD25+, CD4+CD69, CD4+CD25+CD69+ |
Tregs 10/17 Psychiatric patients with Treg percentages (CD4+CD127dim) higher than 10% in the CSF and lower than 10% in the blood vs non-inflamed neurological patients; this subgroup of psychiatric patients overlapped with a cluster of inflamed neurological patients Other 6/17 Psychiatric patients with greater CD4/CD8 ratios in the blood and CSF vs non-inflamed neurological patients |
Albumin, IgG, IgA, IgM All lower vs. meningitis patients |
Not adjusted | Results indicate the percentage, not numbers, of the surface antigens |
| Drexhage et al. (2011) | SCZ vs HC High CD4+ CD25+FoxP3+ |
SCZ vs. HC High monocytes IL-4-containing lymphocytes CD3+CD25+ activated T cells Th17 memory type (CD4+ IL-17A+ CD45RO+) NS Total blood leukocyte counts, lymphocytes IFN-γ and IL-17A-containing lymphocytes Th1 (CD4+ IFN-γ+), Th2 (CD4+IL-4+) |
Tregs One-third of patients (n=7) with lower blood Tregs (<3.25%) Others Subgroups with Inflamed monocyte signature SCZ: 11/19; HC: 3/23 High Th2 and IL-4-containing lymphocytes confined to patients with inflamed monocytes High CD4+CD25+ activated T cells, Tregs and Th17 irrespective of monocyte status |
SCZ vs. HC High sIL2-R NS CCL2, IL-5, IL1-β, PTX3 IL-22, TGF-β: High but NS Lower detection limit IFN-γ, IL-17, IL-10, IL-6, IL-4, TNF-α (SCZ and HC) |
Adjusted for sex and age (matched) | Results represent the percentages, not numbers, of cells within the lymphocyte population PBMCs frozen and stored in liquid nitrogen |
| Fernandez-Egea et al. (2016) | SCZ vs. HC Low Functional, activated (HLA-DR+) and memory Tregs (CD45RA-) |
SCZ vs. HC Low Dendritic cells, central memory T-cells (CD4+CCR7+CD45RA-), CD4+CD161+ naïve T cells, and switched memory B cells (CD27+IgD-) High NK, naïve-B, and CXCR5+ memory T-cells (CD4+CD45RA-) Monocytes (CD16low) |
Immune cluster 1: Lower functional, activated (HLA-DR+) memory Tregs (CD45RA-), dendritic cells, central memory T-cells (CD4+CCR7+CD45RA-), CD4+CD161+ naïve T cells, and switched memory B cells (CD27+IgD-) Immune cluster 2: Higher NK, naïve-B, CXCR5+ memory T-cells (CD4+CD45RA-), monocytes (CD16low) |
NA | Adjusted for age, sex, BMI, and cigarette smoking (matched) Clozapine plasma levels mg/L (patients analyses) |
Results expressed both in percentages and absolute concentration (cells/mm3) |
| Kelly et al. (2018) | SCZ vs HC Significant three-way interaction among group, testing session, and TLR type Reduced TLR2+ Tregs in SCZ vs HC at baseline (but not at follow-up) Increased TLR2+ Tregs in SCZ baseline vs follow-up High TLR5+ Tregs in SCZ vs HC at baseline (but not at follow-up) |
Total leucocyte counts at normal range for patients and controls; NS difference at baseline and follow up Significant three-way interaction among group, testing session, and TLR type Monocytes Higher TLR2+ monocytes in SCZ vs HC at follow up (but not at baseline) Increased TLR2+ monocyte in SCZ at follow-up vs baseline (not in HC) Higher TLR4+ monocytes in SCZ vs HC at baseline (but not at follow-up) Reduced TLR4+ monocyte in SCZ at baseline vs follow-up (not in HC) Higher TLR5+ monocytes in SCZ vs HC at baseline and follow up TLR5+ monocytes NS from baseline to follow-up (patients and controls) T active cells Higher TLR2 in SCZ vs HC at follow-up (but not at baseline) High TLR2 in SCZ baseline vs follow-up Higher TLR5 in SCZ vs. controls at both times |
NA | MFI (TLR density on cell membrane) Significant three-way interaction among group, testing session, and TLR type Monocytes Higher MFI TLR4 and TLR5 in SCZ vs. HC at baseline (but not at follow-up) Higher MFI TLR2 in SCZ vs HC at follow-up (but not at baseline) |
Age, sex, and schooling (matched), medication (patients) Secondary analyses adjusted for: smoking, blood cortisol levels, BMI, waist-to-hip ratio changes during treatment, and DUP. |
Results expressed as the percentages of cells expressing each TLRs, and MFI for TLR density on cell membrane No placebo control available |
| Counotte et al. (2018) | High vs low-psychosis liability NS CD4+CD24+FoxP3+ |
High vs low-psychosis liability NS Th1, Th2, Th7 Trend for decreased lymphocytes in ultra-high risk vs psychosis and controls Trend for increased %CD56+ NK in siblings and psychosis vs. controls |
NA | NA | Adjusted for age, sex, ethnicity, education level, smoking, psychotropic medication | T cell subsets expressed as percentages of total lymphocytes PBMCs were counted and stored in liquid nitrogen |
| Kelly et al. (2018) | SCZ vs. HC High CD25+FoxP3+ NS CD25+FoxP3+CD45RA- (memory Tregs) |
SCZ vs. HC NS White blood cell counts, CD3+ or CD4+ |
NA | NA | Adjusted for age, sex, smoking, BMI medication (patients) | Results represent the percentages, not numbers, of cells within the lymphocyte population |
| Akkouh et al. (2020) | iPSC-astrocyte SCZ vs. controls (after in vitro IL1-β challenge) Lower astrocyte CCL20 gene expression = astrocytes less capable of inducing CCR6+ Tregs chemotaxis |
NA | NA | SCZ vs. HC Low FoxP3 mRNA in whole blood NS CCR6 mRNA in whole blood |
Adjusted for age and sex | - |
| Sahbaz et al. (2020) | SCZ vs. HC (unstimulated condition) High CD4+CD25+FoxP3+ SCZ vs. HC (stimulated condition with anti-CD3 and anti-CD28) Low CD4+CD25+FoxP3+ Within-subject comparison (before x after stimulation with anti-CD3 and anti-CD28) SCZ Low CD4+CD25+FoxP3+ HC NS CD4+CD25+FoxP3+ |
SCZ vs. HC (unstimulated condition) Low CD3+ T cell CD3+CD4+ T cells High CD19+ B cell CD4+CD25+ activated T cell NS CD14+ monocyte, CD8+ T cytotoxic, CD20+ B cell, CD16+CD56+ B Cell SCZ vs. HC (stimulated condition with anti-CD3 and andti-CD28) High activated T cell CD4+CD25+ Within-subject comparison (before vs after stimulation) SCZ or HC NS CD4+CD25+ activated T cell |
NA | Plasma (unstimulated) High IL-6, IL-17A NS IL-2, IL-4, IL-10, TNF-a, IFN-g SCZ vs. HC Supernatant (stimulated condition) High IL-6, IL-17A, IFN-γ, TNF-α Trend IL-2 |
Adjusted for age, sex, ethnicity, BMI, education, smoking status (matched) | Results represent the percentages, not numbers, of cells within the lymphocyte population PBMCs cultured for 72h and stimulated with anti-CD3/anti-CD28 (triggers intracellular signalling pathway and enhances T cell proliferation) NS difference on the evaluated parameters between patients on clozapine vs. other medications |
BMI: body mass index; CSF: cerebrospinal fluid; DUP: duration of untreated psychosis; HC: healthy control; TLR: Toll-like receptor; NA: non-applicable; NS: non-significant; PBMCs: peripheral blood mononuclear cells; SCZ: schizophrenia.
Results reflect differences in the peripheral blood, unless otherwise indicated.
Significant results are highlighted in bold.
Table 3.
Association between Tregs or other immune cells and clinical and stress variables in patients.
| Reference | Clinical instruments | Immune cells and clinical/stress variables correlations in patients |
|---|---|---|
| Maxeiner et al. (2009) | OPCRIT | Not evaluated |
| Drexhage et al. (2011) | DSM-IV criteria (CASH interview) GAF |
GAF at discharge Positive correlation between %Tregs at admission and GAF at discharge in subgroups Treg > 3.25% (n=16) better GAF ≧ 50 at discharge Treg <3.25% (n=7) lower GAF ≦ 49 at discharge (lost after Bonferroni correction for multiple testing) |
| Fernandez-Egea et al. (2016) | Structured interview ICD-10 standards Cognitive performance (BACS) Psychotic symptom severity (CGI-S) - positive, negative, and overall ratings |
Immune cells predictive of diagnosis Cluster 1 including lower Tregs (HLA-DR+ CD45RA-) accounted for 53% of diagnostic variance Cluster 2 including higher CD16low monocytes accounted for 19% of diagnostic variance. Cognitive performance (BACS) and Negative symptom scores (CGI-S) Immune cluster 1 including lower Tregs (CD45RA-) had poor cognitive performance and high negative symptom scores |
| (Kéri et al., 2017) | Structured Clinical Interview for DSM-IV (SCID-CV) PANSS positive, negative and general symptoms RBANS - Cognitive functions (overall score) |
RBARS (Cognition) Monocytes High %TLR4+ and TLR5+ associated with lower cognitive performance at baseline (but not at follow-up), other immune parameters NS Changes in PANSS and RBANS (follow-up minus baseline) not associated with TLR |
| (Counotte et al., 2018) | CAARMS GPTS SIAS CAPE CTQ-SF |
Clinical variables Not evaluated CTQ Significant psychosis liability x CTQ for Th17 only In high-psychosis liability subgroup, childhood trauma associated with increased %Th17 Virtual stress Significant psychosis liability x subjective stress to social virtual experiment for Tregs and NK cells In high-psychosis liability, increased %Tregs and low %NK associated with more subjective stress |
| Kelly et al. (2018) | SCID-V SANS - negative symptoms BPRS - global psychopathology and positive symptoms CDS - depressive symptoms CGI - global illness severity MATRICS - MCCB (cognitive battery) |
SANS - negative symptoms High %Tregs correlated with lower negative symptoms (total SANS), driven primarily by alogia and affective blunting MCCB - Cognition High %Treg trend to better cognitive performance (reasoning and problem solving) High %CD4+ T cells significantly correlated to better visual learning and memory performance |
| Akkouh et al. (2020) | SCID-1 (DSM-IV) | Not evaluated |
| Sahbaz et al. (2020) | SCID - DSM-IV PANSS |
PANSS %Treg NS Supernatant IL-4 and IL-10 negatively correlated with PANSS negative scores %CD19+ and CD20+ B cells positively correlated with PANSS total and global scores |
BACS: The Brief Assessment of Cognition for Schizophrenia; BPRS: The Brief Psychiatric Rating Scale; CASH: The Comprehensive Assessment of Symptoms and History interview; CGI-S: Clinical Global Impression for Schizophrenia; CAARM: Comprehensive Assessment of At-Risk Mental States; CAPE: Community Assessment of Psychic Experiences; CDS: The Calgary Depression Rating Scale; CTQ-SF: Childhood Trauma Questionnaire Short Form; DSM: Diagnostic and Statistical Manual of Mental Disorders; GAF: Global Assessment of Functioning; GPTS: Green Paranoid Thoughts Scale; ICD: International Classification of Diseases; MATRICS-MCBB: The MATRICS Consensus Cognitive Battery; OPCRIT: Operational Criteria Checklist for Psychotic Illness and Affective Illness; PANSS: Positive and Negative Syndrome Scale; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status; SANS: The Scale for the Assessment of Negative Symptoms SIAS: Social Interaction Anxiety Scale.
Significant results are highlighted in bold.
3.2. Increased Tregs
Overall, half of the studies found higher blood Treg percentages in medicated patients with schizophrenia relative to healthy controls (Drexhage et al., 2011; Kelly et al., 2018; Kéri et al., 2017; Sahbaz et al., 2020). From these, three (Drexhage et al., 2011; Kéri et al., 2017; Sahbaz et al., 2020) observed that Treg percentages (CD4+CD25+FoxP3+) in patients were increased concomitantly with greater percentages of activated (CD3+CD25+) or pro-inflammatory (CD4+IL17A+CD45RO+) T-cells. In contrast, Kelly (2018) found increased Treg percentages in clinically stable patients with schizophrenia compared with controls in the absence of elevated proportions of CD3+ or CD4+T-cells, but pro-inflammatory subsets were not evaluated.
Drexhage et al. (2011) performed additional analyses by stratifying their sample based on an inflammatory gene expression signature in monocytes; they found that 11/19 patients and 3/23 controls were in an inflamed monocyte state. They further observed that the dysregulated adaptive immune phenotype of upregulated Tregs alongside higher pro-inflammatory Th17 percentages in patients occurred irrespective of the inflamed innate monocyte state (although slightly more evident in the negative monocyte subgroup), which they interpret as reflecting an altered T-cell set point in schizophrenia.
3.3. Decreased Tregs
Three studies (Drexhage et al., 2011; Fernandez-Egea et al., 2016; Maxeiner et al., 2009) identified lower blood Tregs in subgroups of medicated patients versus controls. In the sample from Drexhage et al. (2011), although blood Treg percentages were overall increased in patients than controls, around one-third of the patients had low Tregs (<3.25% cut-off). Fernandez-Egea et al. (2016) performed detailed flow cytometry of 73 immune cells in treatment-resistant schizophrenia patients medicated with clozapine versus controls. Using partial least squares to predict diagnosis, they identified two distinct immune phenotypes. The first comprised lower numbers of activated and memory Tregs (evaluated by surface markers HLA-DR+ and CD45RA−, respectively) in patients versus controls. The second consisted of higher numbers of classical monocytes (CD16low) in patients than controls. The lower Treg cluster accounted for the highest diagnostic variance of the sample (53%), whereas the monocyte cluster accounted for the lowest variance (19%), interpreted as an immunophenotype of patients with poor response to treatment and more severe outcomes.
Maxeiner et al. (2009) measured T-cells in paired samples of peripheral blood and liquor of a pooled sample of psychiatric patients (the largest proportion of schizophrenia spectrum) compared to two distinct groups of patients with inflammatory or non-inflammatory neurological conditions. They identified a subset (around one-third) of the psychiatric sample with lower blood but higher liquor frequencies of Tregs (CD4+CD25+CD127dim; 10% cut-off) compared to non-inflamed neurological patients, accompanied by greater disinhibition of the systemic T-CD4+/CD8+ ratio. The blood and liquor Treg phenotype overlapped with a cluster of patients with inflammatory neurological conditions, suggesting a potential shared inflammatory process driven by the disturbed Treg populations. However, the pooled psychiatry sample and the lack of a control group hamper further interpretation.
3.4. Unchanged Tregs
One study reported no difference in blood Tregs (CD3+CD4+FoxP3+) or Th17 frequencies in recent-onset treated psychosis patients (Counotte et al., 2018). Nevertheless, the study was a pooled sample of low-versus-high psychosis liability subgroups (recent-onset psychosis and ultra-high risk versus unaffected siblings and controls, respectively).
3.5. A longitudinal study
A single follow-up study included drug-naïve first-episode schizophrenia patients and healthy controls, in which the authors measured the expression of Toll-like receptors (TLR) in subpopulations of blood monocytes and T cells (Kéri et al., 2017). At baseline and compared to controls, they found greater presence of Tregs expressing TLR5 and lower frequency of TLR2-expressing Tregs. Eight-week treatment with either olanzapine or risperidone upregulated the percentages of TLR2-expressing Tregs. The clinical significance of the TLR distinction is unclear. An important limitation is that there was no placebo control group.
3.6. Functional capacity of Tregs in vitro
Akkouh et al. (2020) found evidence for dysregulated astroglia-Treg axis in schizophrenia. Astrocytes cultures from patients' fibroblasts secreted less CCL20 – a chemokine that attracts Tregs to the CSF. In addition, they reported, in a large independent sample (n > 800), that patients had lower expression of the Treg-specific transcriptional factor FoxP3 in their whole blood than controls. However, the connection between the two findings is obscure, and the assessment of Tregs in patients’ blood in this study was only indirect through the measurement of FoxP3 mRNA.
Sahbaz et al. (2020) tested for the first time Tregs competence to regulate pro-immune responses in chronic patients and controls. Although Treg percentages were increased in patients versus controls in unstimulated conditions, after in vitro stimulation with both anti-CD3 and anti-CD28 (a cocktail that triggers intracellular signalling and stimulates T-cell proliferation), the patients regulatory arm had a weaker proliferative response than the controls (also in within-subject analyses) accompanied by increased pro-inflammatory cytokines in culture supernatants (IL-6, IL-17A, IFN-γ, TNF-α). This is the only investigation testing the proliferative capacity of Tregs in psychosis, requiring replication in future, ideally, prospective studies.
3.7. Circulating Tregs and correlations with psychosis symptom domains
The results are summarised in Table 3. There is evidence that low blood Tregs are associated with worst symptomatology. Drexhage et al. (2011) found low blood Treg percentages in one-third of their patient sample at admission that was associated with less favourable global functioning at discharge. Fernandez-Egea et al. (2016) found that more severe negative and cognitive symptoms in treatment-resistant schizophrenia were associated with reduced numbers of experienced Tregs. Finally, Kelly et al., 2018 found that remitted patients with schizophrenia had high blood Treg percentages that were significantly associated with fewer negative symptoms (alogia and affective blunting) and a trend to better cognitive performance (reasoning, problem-solving, processing speed). While in Sahbaz et al. (2020) Treg percentages were not associated with symptomatology, the anti-inflammatory IL-10 (detected in cellular supernatants of Treg assay) was negatively correlated with negative symptoms.
4. Conclusions and future perspectives
4.1. Implications for the h-Treg hypothesis
We discussed the obvious general shortcomings of the literature; studies are few with small samples conducted in medicated patients, and poorly adjusted for the effects of confounding such as body mass index (BMI) and tobacco smoking, and no clinical follow-up to test possible causal mechanisms. Nevertheless, we suggest that the h-Treg hypothesis is compatible with and encouraged by two key aspects of the literature. First, four studies report that greater presence of Tregs is associated with better clinical status in treated patients. This allows that h-Tregs may have a state-dependent causal role in psychosis that could be tested in longitudinal studies in minimally treated patients while adjusting for confounding (e.g., sex, age, BMI, tobacco, and other drugs). A further implication is that other studies reporting increased Tregs in patients without clinical measures do not refute the h-Treg hypothesis. Second, Sahbaz et al. (2020) provide a strong test of the hypothesis by directly measuring the ability of Tregs to limit inflammatory responses to an in vitro challenge and finding it impaired.
We make specific suggestions for further tests of the h-Treg hypothesis below. However, we would reiterate a concluding point from our original proposal that Treg dysfunction will not be the sole primary abnormality in the causation of psychosis. It is clear that a multi-level complex of an interacting multiplicity of factors will be involved, perhaps converging on some final common pathways such as Treg functionality, which may vary across functional subtypes of psychosis and transdiagnostic disorders.
4.2. Better measures of Treg function
The heterogeneity of markers used to detect Tregs in blood across studies may obscure consistent group differences. Tregs are usually identified as CD4+CD25+FoxP3+; however, effector CD4+T-cells can also express CD25. Moreover, FoxP3-expressing Tregs are mixed populations, with some having a minimal suppressive ability. Such limitations have prompted the search for additional markers, such as CD127. Combined CD4+CD25+FoxP3+CD127low has improved Treg purification and should be used for consistency (Liu et al., 2006).
While there is no such ‘best’ functional marker, different molecules combined to CD4+CD25+FoxP3+CD127low can help to delineate Tregs with highly suppressive activity (Hou et al., 2019). For example, Fernandez-Egea et al. (2016) identified Treg subpopulations with more suppressive function (HLA-DR+ CD45RA-) that were reduced in treatment-resistant patients. Other functional markers include CTLA-4, GARP, TNFR2, and others (Table 4). Additionally, CD39 and CD73 are attractive in autoimmune disorders for their key role in adenosine synthesis, a potent anti-inflammatory molecule (Peres et al., 2018b). Techniques such as CyTOF permit concurrent detection of more than 40 different antibodies with high-dimensional single-cell proteomic data (Subrahmanyam and Maecker, 2021) that could be useful in future Treg studies in psychosis.
Table 4.
List of some functional markers, their ligands and main function.
| Functional markers | Ligands | Main function/characteristics |
|---|---|---|
| CTLA4 (CD152) | CD80/CD86 (B7) | Negatively regulates immune responses; restricts CD28 engagement by competing with B7 on antigen-presenting cells |
| HLA-DR+ | – | High FoxP3-expressing Tregs (highly suppressive) |
| GARP (or LRRC32) | TGF-β | A transmembrane protein expressed in activated Tregs that promotes TGF-β release and activation |
| TNFR2 | – | Activates and expands Tregs (highly suppressive Tregs) |
| GITR – glucocorticoid-induced receptor family protein | – | Activates and expands Tregs (highly suppressive Tregs) |
| Integrin αvβ8 | TGF-β | Promotes TGF-β activation and mediates Treg-immune suppression |
| CD39 | ATP and ADP | Converts ATP and ADP to AMP |
| CD73 | AMP | Converts AMP to adenosine |
| TIGIT | CD155/CD112 | Inhibit pro-inflammatory reactions in innate and adaptive cells |
| PD1 | PDL1, PDL2 | Negatively regulates immune responses; inhibits CD28 and TCR signalling |
| ICOS | – | Memory-like Tregs that use IL-10 and TGF-β to supress dendritic cells |
| LAG-3 | MHCII | Outcompetes CD4 to bind MHC II and deliver inhibitory signal to antigen-presenting cells |
| CCR6 | CCL20 | Effector-memory Treg |
| CD28 | CD80/CD86 (B7) | Co-stimulatory molecule; promotes T cell activation, proliferation, and survival |
| CD45RA-/CD45RO+ | – | Memory markers |
| CD45RA + CD25+FoxP3low | – | Naïve-resting Tregs; predominantly thymus-derived (moderate suppressive activity) |
| CD45RA-CD25+FoxP3high | – | Activated-memory Tregs; predominantly thymus-derived (highly suppressive) |
| CD4+CD25+HLA-DR + Tregs | – | Tregs with activated and highly suppressive phenotype |
| CD45RA-FoxP3low | – | Non-Tregs (Th17 signature with RORc upregulation) |
One important consideration is that counting the proportion of blood Tregs is not a direct measure of their functional capacity. The study from Sahbaz et al. (2020) is a great example and, so far, the only testing the proliferative capacity of Tregs in vitro. As in autoimmune disorders (Cribbs et al., 2015; Thiolat et al., 2014), assays of Tregs and T-effector cells (alone and co-culture), determination of cytokines in supernatant, and DNA methylation of the FoxP3 locus are attractive approaches, and perhaps more precise, to test the functional capacity of Tregs. This could be applied to psychosis, ideally in longitudinal studies of untreated patients with placebo control to determine the hypofunctional state of Tregs and ascertain causality.
4.3. Clinical correlates of Treg function in trials of early psychosis
Large clinical trials are needed to move beyond correlational studies and identify the causal roles of immune-regulatory dysfunction in the pathogenesis of psychosis. Any trial of new or repurposed agents, especially those targeting immune mechanisms, should include cellular immunophenotyping together with traditional cytokine measures to develop immune markers of illness subtypes, severity, and stratification. Imaging methods, such as DTI, magnetic resonance imaging (MRI), and functional MRI could be used to assess the relationship between structural and functional brain changes with blood Tregs and other leucocyte populations, including the Treg:Th17 ratio (Poletti et al., 2017), along with careful standardised clinical assessments of symptoms and social and occupational function using transdiagnostic methods. Besides, new PET radioligands (Beaino et al., 2017) are emerging that can be used to assess the functional status of astroglia, microglia, and neuron function in relation to baseline and changes in Tregs function. There is also need for better adjustment for confounding as only half of the studies adjusted for the effects of BMI and smoking in addition to sex and age (Table 2). The identification of genetic variants associated with Tregs activity/function could be useful in future mendelian randomisation studies to understand potential causality and address issues of confounding and reverse causation.
4.4. Hypofunctional Treg hypothesis of psychosis: a role for IL-6?
Associations between low Tregs and worst symptomatology could be reflecting a hyperactive IL-6 state observed in subgroups of patients. We summarise some key mechanisms in Fig. 3. To disentangle that, clinical trials testing the efficacy of IL-6 blockers in psychosis could assess Tregs immune phenotype in vivo and test their suppressive capacity with in vitro co-culture assays before and after IL-6 blockade. Such analyses combined with the Treg:Th17 ratio can help to elucidate mechanisms of IL-6-induced Treg destabilisation. Given the pro-and anti-inflammatory roles of IL-6, in vitro assays are feasible to assess Tregs suppressive capacity in response to drugs that inhibit both- (e.g., tocilizumab) or only the pro-inflammatory pathway of IL-6 (e.g., olamkicept).
Funding
F.C-Z. receives national and international Doctorate Scholarships from the São Paulo Research Foundation (Brazil) (FAPESP; 2019/13229-2 and 2021/07448-3); M.H.F.L acknowledges a fellowship from FAPESP (2020/02642-3), C.M.D-B acknowledges the support from FAPESP (2012/05178-0); P.L-J acknowledges the support from the Center for Research in Inflammatory Diseases (Brazil) (2013/08216-2); B.D acknowledges the support of UK MRC grants MR/K020803/1 and MC_PC_11003, and of Stanley Medical Research Institute clinical trial grants.
Authors contributions
F.C-Z. and B.D developed the hypofunctional Treg hypothesis. F.C.-Z. wrote the manuscript, designed the tables and figures. B.D. supervised the work and together with the other authors critically revised the manuscript. The figures were designed using a premium account from Biorender (https://biorender.com/).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Glossary
- Marker
Immune cell subset
- CD3
T lymphocytes
- CD4
T helper lymphocytes
- CD8
Cytotoxic T lymphocytes
- CD25
Activated T lymphocytes (IL-2 receptor)
- FoxP3
Intracellular Treg transcriptional factor marker
- CD127
Negatively correlated with Treg intracellular FoxP3 marker
- IFN-γ
Th1-producing IFN-γ (intracellular marker)
- IL-4
Th2-producing IL-4 (intracellular marker)
- IL-17A
Th17-producing IL-17A (intracellular marker)
- RORc
Intracellular Th17 transcriptional factor marker
- CD19; CD20
B lymphocytes
- CD56
Natural killer cells
- CD11c; CD123
Dendritic cells
- CD14; CD16
Monocytes
References
- Akkouh I.A., Ueland T., Hansson L., Inderhaug E., Hughes T., Steen N.E., Aukrust P., Andreassen O.A., Szabo A., Djurovic S. Decreased IL-1β-induced CCL20 response in human iPSC-astrocytes in schizophrenia: potential attenuating effects on recruitment of regulatory T cells. Brain Behav. Immun. 2020;87:634–644. doi: 10.1016/j.bbi.2020.02.008. [DOI] [PubMed] [Google Scholar]
- al-Haddad B.J.S., Oler E., Armistead B., Elsayed N.A., Weinberger D.R., Bernier R., Burd I., Kapur R., Jacobsson B., Wang C., Mysorekar I., Rajagopal L., Adams Waldorf K.M. The fetal origins of mental illness. Am. J. Obstet. Gynecol. 2019 doi: 10.1016/j.ajog.2019.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albany C.J., Trevelin S.C., Giganti G., Lombardi G., Scottà C. Getting to the heart of the matter: the role of regulatory T-cells (Tregs) in cardiovascular disease (CVD) and atherosclerosis. Front. Immunol. 2019;10 doi: 10.3389/fimmu.2019.02795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arroyo Hornero R., Hamad I., Côrte-Real B., Kleinewietfeld M. The impact of dietary components on regulatory T cells and disease. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.00253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Attias M., Al-Aubodah T., Piccirillo C.A. Mechanisms of human FoxP3+ Treg cell development and function in health and disease. Clin. Exp. Immunol. 2019 doi: 10.1111/cei.13290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avdeeva A., Rubtsov Y., Dyikanov D., Popkova T., Nasonov E. Regulatory T cells in patients with early untreated rheumatoid arthritis: phenotypic changes in the course of methotrexate treatment. Biochimie. 2020 doi: 10.1016/j.biochi.2020.03.014. [DOI] [PubMed] [Google Scholar]
- Beaino W., Janssen B., Kooij G., van der Pol S.M.A., van Het Hof B., van Horssen J., Windhorst A.D., de Vries H.E. Purinergic receptors P2Y12R and P2X7R: potential targets for PET imaging of microglia phenotypes in multiple sclerosis. J. Neuroinflammation. 2017 doi: 10.1186/s12974-017-1034-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bettelli E., Carrier Y., Gao W., Korn T., Strom T.B., Oukka M., Weiner H.L., Kuchroo V.K. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- Buckner J.H. Mechanisms of impaired regulation by CD4+ CD25+ FOXP3+ regulatory T cells in human autoimmune diseases. Nat. Rev. Immunol. 2010 doi: 10.1038/nri2889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calabrese L.H., Rose-John S. IL-6 biology: implications for clinical targeting in rheumatic disease. Nat. Rev. Rheumatol. 2014 doi: 10.1038/nrrheum.2014.127. [DOI] [PubMed] [Google Scholar]
- Chaudhry I.B., Husain M.O., Khoso A.B., Husain M.I., Buch M.H., Kiran T., Fu B., Bassett P., Qurashi I., ur Rahman R., Baig S., Kazmi A., Corsi-Zuelli F., Haddad P.M., Deakin B., Husain N. A randomised clinical trial of methotrexate points to possible efficacy and adaptive immune dysfunction in psychosis. Transl. Psychiatry. 2020;10:415. doi: 10.1038/s41398-020-01095-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chez M., Lepage C., Parise C., Dang-Chu A., Hankins A., Carroll M. Safety and observations from a placebo-controlled, crossover study to assess use of autologous umbilical cord blood stem cells to improve symptoms in children with autism. Stem Cells Transl. Med. 2018;7:333–341. doi: 10.1002/sctm.17-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conen S., Gregory C.J., Hinz R., Smallman R., Corsi-Zuelli F., Deakin B., Talbot P.S. Neuroinflammation as measured by positron emission tomography in patients with recent onset and established schizophrenia: implications for immune pathogenesis. Mol. Psychiatr. 2020:1–9. doi: 10.1038/s41380-020-0829-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Consortium S.W.G., Ripke S., Walters J.T., O'Donovan M.C. Mapping genomic loci prioritises genes and implicates synaptic biology in schizophrenia. medRxiv. 2020 doi: 10.1101/2020.09.12.20192922. 2020.09.12.20192922. [DOI] [Google Scholar]
- Copsel S.N., Malek T.R., Levy R.B. Medical treatment can unintentionally alter the regulatory T-cell compartment in patients with widespread pathophysiologic conditions. Am. J. Pathol. 2020;190 doi: 10.1016/j.ajpath.2020.07.012. 2000–2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corsi-Zuelli F., Deakin B. Impaired regulatory T cell control of astroglial overdrive and microglial pruning in schizophrenia. Neurosci. Biobehav. Rev. 2021;125:637–653. doi: 10.1016/j.neubiorev.2021.03.004. [DOI] [PubMed] [Google Scholar]
- Corsi-Zuelli F., Loureiro C.M., Shuhama R., Fachim H.A., Menezes P.R., Louzada-Junior P., Mondelli V., Del-Ben C.M. Cytokine profile in first-episode psychosis, unaffected siblings and community-based controls: the effects of familial liability and childhood maltreatment. Psychol. Med. 2020;50:1139–1147. doi: 10.1017/S0033291719001016. [DOI] [PubMed] [Google Scholar]
- Corsi-Zuelli F., Marques L., da Roza D.L., Loureiro C.M., Shuhama R., Di Forti M., Menezes P.R., Louzada-Junior P., Del-Ben C.M. The independent and combined effects of cannabis use and systemic inflammation during the early stages of psychosis: exploring the two-hit hypothesis. Psychol. Med. 2021;19:1–11. doi: 10.1017/S0033291721000726. [DOI] [PubMed] [Google Scholar]
- Counotte J., Drexhage H.A., Wijkhuijs J.M., Pot-Kolder R., Bergink V., Hoek H.W., Veling W. Th17/T regulator cell balance and NK cell numbers in relation to psychosis liability and social stress reactivity. Brain Behav. Immun. 2018;69:408–417. doi: 10.1016/j.bbi.2017.12.015. [DOI] [PubMed] [Google Scholar]
- Cribbs A.P., Kennedy A., Penn H., Amjadi P., Green P., Read J.E., Brennan F., Gregory B., Williams R.O. Methotrexate restores regulatory T cell function through demethylation of the FoxP3 upstream enhancer in patients with rheumatoid arthritis. Arthritis Rheum. 2015;67:1182–1192. doi: 10.1002/art.39031. [DOI] [PubMed] [Google Scholar]
- Davies C., Segre G., Estradé A., Radua J., De Micheli A., Provenzani U., Oliver D., Salazar de Pablo G., Ramella-Cravaro V., Besozzi M., Dazzan P., Miele M., Caputo G., Spallarossa C., Crossland G., Ilyas A., Spada G., Politi P., Murray R.M., McGuire P., Fusar-Poli P. Prenatal and perinatal risk and protective factors for psychosis: a systematic review and meta-analysis. The Lancet Psychiatry. 2020 doi: 10.1016/S2215-0366(20)30057-2. [DOI] [PubMed] [Google Scholar]
- Deakin B., Suckling J., Barnes T.R.E., Byrne K., Chaudhry I.B., Dazzan P., Drake R.J., Giordano A., Husain N., Jones P.B., Joyce E., Knox E., Krynicki C., Lawrie S.M., Lewis S., Lisiecka-Ford D.M., Nikkheslat N., Pariante C.M., Smallman R., Watson A., Williams S.C.R., Upthegrove R., Dunn G. The benefit of minocycline on negative symptoms of schizophrenia in patients with recent-onset psychosis (BeneMin): a randomised, double-blind, placebo-controlled trial. The Lancet Psychiatry. 2018;5:885–894. doi: 10.1016/S2215-0366(18)30345-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhuban K. Bin, Bartolucci S., D'Hennezel E., Piccirillo C.A. Signaling through gp130 compromises suppressive function in human FOXP3+Regulatory T Cells. Front. Immunol. 2019 doi: 10.3389/fimmu.2019.01532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominitzki Sabine, et al. Cutting edge: trans-signaling via the soluble IL-6R abrogates the induction of FoxP3 in naive CD4+CD25 T cells. J. Immunol. 2007;179(4):2041–2045. doi: 10.4049/jimmunol.179.4.2041. [DOI] [PubMed] [Google Scholar]
- Drexhage R.C., Hoogenboezem T. a, Cohen D., Versnel M. a, Nolen W. a, van Beveren N.J.M., Drexhage H. a. An activated set point of T-cell and monocyte inflammatory networks in recent-onset schizophrenia patients involves both pro- and anti-inflammatory forces. Int. J. Neuropsychopharmacol. 2011;14:746–755. doi: 10.1017/S1461145710001653. [DOI] [PubMed] [Google Scholar]
- Duggleby R., Danby R.D., Madrigal J.A., Saudemont A. Clinical grade regulatory CD4+ T cells (Tregs): moving toward cellular-based immunomodulatory therapies. Front. Immunol. 2018;9 doi: 10.3389/fimmu.2018.00252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eggenhuizen P.J., Ng B.H., Ooi J.D. Treg enhancing therapies to treat autoimmune diseases. Int. J. Mol. Sci. 2020 doi: 10.3390/ijms21197015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Badawy A., El-Badri N. Clinical efficacy of stem cell therapy for diabetes mellitus: a meta-analysis. PloS One. 2016;11 doi: 10.1371/journal.pone.0151938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eller K., Kirsch A., Wolf A.M., Sopper S., Tagwerker A., Stanzl U., Wolf D., Patsch W., Rosenkranz A.R., Eller P. Potential role of regulatory T cells in reversing obesity-linked insulin resistance and diabetic nephropathy. Diabetes. 2011 doi: 10.2337/db11-0358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Egea E., Vértes P.E., Flint S.M., Turner L., Mustafa S., Hatton A., Smith K.G.C., Lyons P.A., Bullmore E.T. Peripheral immune cell populations associated with cognitive deficits and negative symptoms of treatment-resistant schizophrenia. PloS One. 2016;11 doi: 10.1371/journal.pone.0155631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher S.A., Rahimzadeh M., Brierley C., Gration B., Doree C., Kimber C.E., Cajide A.P., Lamikanra A.A., Roberts D.J. The role of Vitamin D in increasing circulating T regulatory cell numbers and modulating T regulatory cell phenotypes in patients with inflammatory disease or in healthy volunteers: a systematic review. PloS One. 2019 doi: 10.1371/journal.pone.0222313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandal M.J., Haney J.R., Parikshak N.N., Leppa V., Ramaswami G., Hartl C., Schork A.J., Appadurai V., Buil A., Werge T.M., Liu C., White K.P., Horvath S., Geschwind D.H. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science. 2018;80(359):693–697. doi: 10.1126/science.aad6469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandal M.J., Zhang P., Hadjimichael E., Walker R.L., Chen C., Liu S., Won H., van Bakel H., Varghese M., Wang Y., Shieh A.W., Haney J., Parhami S., Belmont J., Kim M., Moran Losada P., Khan Z., Mleczko J., Xia Y., Dai R., Wang D., Yang Y.T., Xu M., Fish K., Hof P.R., Warrell J., Fitzgerald D., White K., Jaffe A.E., Peters M.A., Gerstein M., Liu C., Iakoucheva L.M., Pinto D., Geschwind D.H. Transcriptome-wide isoform-level dysregulation in ASD, schizophrenia, and bipolar disorder. Science. 2018;80(362) doi: 10.1126/science.aat8127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Z., Gao Y., Li Z., Chen Z., Lu D., Tsun A., Li B. Synergy between IL-6 and TGF-β signaling promotes FOXP3 degradation. Int. J. Clin. Exp. Pathol. 2012;5:626–633. [PMC free article] [PubMed] [Google Scholar]
- Gliwiński M., Iwaszkiewicz-Grześ D., Trzonkowski P. Cell-based therapies with T regulatory cells. BioDrugs. 2017;31:335–347. doi: 10.1007/s40259-017-0228-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldsmith D.R., Rapaport M.H., Miller B.J. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol. Psychiatr. 2016;21:1696–1709. doi: 10.1038/mp.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Peñas J., Costas J., Villamayor M.J.G., Xu B. Enrichment of rare genetic variants in astrocyte gene enriched co-expression modules altered in postmortem brain samples of schizophrenia. Neurobiol. Dis. 2019;121:305–314. doi: 10.1016/j.nbd.2018.10.013. [DOI] [PubMed] [Google Scholar]
- Göschl L., Scheinecker C., Bonelli M. Treg cells in autoimmunity: from identification to Treg-based therapies. Semin. Immunopathol. 2019;41:301–314. doi: 10.1007/s00281-019-00741-8. [DOI] [PubMed] [Google Scholar]
- Hartwig F.P., Borges M.C., Horta B.L., Bowden J., Davey Smith G. Inflammatory biomarkers and risk of schizophrenia: a 2-sample mendelian randomization study. JAMA Psychiatry. 2017;74:1226–1233. doi: 10.1001/jamapsychiatry.2017.3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He X., Wang Q., Zhao Y., Zhang H., Wang B., Pan J., Li J., Yu H., Wang L., Dai J., Wang D. Effect of intramyocardial grafting collagen scaffold with mesenchymal stromal cells in patients with chronic ischemic heart disease. JAMA Netw. Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.16236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M., Komai K., Mise-Omata S., Iizuka-Koga M., Noguchi Y., Kondo taisuke, Sakai ryota, Matsuo K., Nakayama takashi, Yoshie O., Nakatsukasa H., Chikuma S., Shichita T., Yoshimura A. Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nature. 2019 doi: 10.1038/s41586-018-0824-5. [DOI] [PubMed] [Google Scholar]
- Kelly D.L., Li X., Kilday C., Feldman S., Clark S., Liu F., Buchanan R.W., Tonelli L.H. Increased circulating regulatory T cells in medicated people with schizophrenia. Psychiatr. Res. 2018;269:517–523. doi: 10.1016/j.psychres.2018.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kéri S., Szabó C., Kelemen O. Antipsychotics influence Toll-like receptor (TLR) expression and its relationship with cognitive functions in schizophrenia. Brain Behav. Immun. 2017;62:256–264. doi: 10.1016/j.bbi.2016.12.011. [DOI] [PubMed] [Google Scholar]
- Kikuchi J., Hashizume M., Kaneko Y., Yoshimoto K., Nishina N., Takeuchi T. Peripheral blood CD4+CD25+CD127low regulatory T cells are significantly increased by tocilizumab treatment in patients with rheumatoid arthritis: increase in regulatory T cells correlates with clinical response. Arthritis Res. Ther. 2015;17:10. doi: 10.1186/s13075-015-0526-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komatsu N., Okamoto K., Sawa S., Nakashima T., Oh-Hora M., Kodama T., Tanaka S., Bluestone J.A., Takayanagi H. Pathogenic conversion of Foxp3 + T cells into TH17 cells in autoimmune arthritis. Nat. Med. 2014 doi: 10.1038/nm.3432. [DOI] [PubMed] [Google Scholar]
- Krämer T.J., Hack N., Brühl T.J., Menzel L., Hummel R., Griemert E.V., Klein M., Thal S.C., Bopp T., Schäfer M.K.E. Depletion of regulatory T cells increases T cell brain infiltration, reactive astrogliosis, and interferon-γgene expression in acute experimental traumatic brain injury. J. Neuroinflammation. 2019 doi: 10.1186/s12974-019-1550-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krienke C., Kolb L., Diken E., Streuber M., Kirchhoff S., Bukur T., Akilli-Öztürk Ö., Kranz L.M., Berger H., Petschenka J., Diken M., Kreiter S., Yogev N., Waisman A., Karikó K., Türeci Ö., Sahin U. A noninflammatory mRNA vaccine for treatment of experimental autoimmune encephalomyelitis. Science. 2021;80(371):145–153. doi: 10.1126/science.aay3638. [DOI] [PubMed] [Google Scholar]
- Li S., Wu Z., Li L., Liu X. Interleukin-6 (IL-6) receptor antagonist protects against rheumatoid arthritis. Med. Sci. Mon. Int. Med. J. Exp. Clin. Res. 2016;22:2113–2118. doi: 10.12659/MSM.896355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Zheng Y. Regulatory T cell identity: formation and maintenance. Trends Immunol. 2015;36:344–353. doi: 10.1016/j.it.2015.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W., Putnam A.L., Xu-yu Z., Szot G.L., Lee M.R., Zhu S., Gottlieb P.A., Kapranov P., Gingeras T.R., de St, Groth B.F., Clayberger C., Soper D.M., Ziegler S.F., Bluestone J.A. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006;203:1701–1711. doi: 10.1084/jem.20060772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeurer M., Ramalho R., Wang F.-S., Zumla A. Host-directed therapies for COVID-19. Curr. Opin. Pulm. Med. 2021;27:205–209. doi: 10.1097/MCP.0000000000000769. [DOI] [PubMed] [Google Scholar]
- Martini E., Giugliano S., Rescigno M., Kallikourdis M. Regulatory T cells beyond autoimmunity: from pregnancy to cancer and cardiovascular disease. Front. Immunol. 2020;11:509. doi: 10.3389/fimmu.2020.00509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxeiner H.G., Rojewski M.T., Schmitt A., Tumani H., Bechter K., Schmitt M. Flow cytometric analysis of T cell subsets in paired samples of cerebrospinal fluid and peripheral blood from patients with neurological and psychiatric disorders. Brain Behav. Immun. 2009;23:134–142. doi: 10.1016/j.bbi.2008.08.003. [DOI] [PubMed] [Google Scholar]
- Meng X., Yang J., Dong M., Zhang K., Tu E., Gao Q., Chen W., Zhang C., Zhang Y. Regulatory T cells in cardiovascular diseases. Nat. Rev. Cardiol. 2016 doi: 10.1038/nrcardio.2015.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller B.J., Gassama B., Sebastian D., Buckley P., Mellor A. Meta-Analysis of lymphocytes in schizophrenia: clinical status and antipsychotic effects. Biol. Psychiatr. 2013;73:993–999. doi: 10.1016/j.biopsych.2012.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohkura N., Yasumizu Y., Kitagawa Y., Tanaka A., Nakamura Y., Motooka D., Nakamura S., Okada Y., Sakaguchi S. Regulatory T cell-specific epigenomic region variants are a key determinant of susceptibility to common autoimmune diseases. Immunity. 2020;52:1119–1132. doi: 10.1016/j.immuni.2020.04.006. e4. [DOI] [PubMed] [Google Scholar]
- Orlovska-Waast S., Köhler-Forsberg O., Brix S.W., Nordentoft M., Kondziella D., Krogh J., Benros M.E. Cerebrospinal fluid markers of inflammation and infections in schizophrenia and affective disorders: a systematic review and meta-analysis. Mol. Psychiatr. 2019 doi: 10.1038/s41380-018-0220-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paolino M., Koglgruber R., Cronin S.J.F., Uribesalgo I., Rauscher E., Harreiter J., Schuster M., Bancher-Todesca D., Pranjic B., Novatchkova M., Fededa J.P., White A.J., Sigl V., Dekan S., Penz T., Bock C., Kenner L., Holländer G.A., Anderson G., Kautzky-Willer A., Penninger J.M. RANK links thymic regulatory T cells to fetal loss and gestational diabetes in pregnancy. Nature. 2021 doi: 10.1038/s41586-020-03071-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasciuto E., Burton O.T., Roca C.P., Lagou V., Rajan W.D., Theys T., Mancuso R., Tito R.Y., Kouser L., Callaerts-Vegh Z., de la Fuente A.G., Prezzemolo T., Mascali L.G., Brajic A., Whyte C.E., Yshii L., Martinez-Muriana A., Naughton M., Young A., Moudra A., Lemaitre P., Poovathingal S., Raes J., De Strooper B., Fitzgerald D.C., Dooley J., Liston A. Microglia require CD4 T cells to Complete the fetal-to-adult transition. Cell. 2020;182:625–640. doi: 10.1016/j.cell.2020.06.026. e24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peres R.S., Donate P.B., Talbot J., Cecilio N.T., Lobo P.R., Machado C.C., Lima K.W.A., Oliveira R.D., Carregaro V., Nakaya H.I., Cunha T.M., Alves-Filho J.C., Liew F.Y., Louzada-Junior P., Cunha F.Q. TGF-β signalling defect is linked to low CD39 expression on regulatory T cells and methotrexate resistance in rheumatoid arthritis. J. Autoimmun. 2018;90:49–58. doi: 10.1016/j.jaut.2018.01.004. [DOI] [PubMed] [Google Scholar]
- Peres R.S., Donate P.B., Talbot J., Cecilio N.T., Lobo P.R., Machado C.C., Lima K.W.A., Oliveira R.D., Carregaro V., Nakaya H.I., Cunha T.M., Alves-Filho J.C., Liew F.Y., Louzada-Junior P., Cunha F.Q. TGF-β signalling defect is linked to low CD39 expression on regulatory T cells and methotrexate resistance in rheumatoid arthritis. J. Autoimmun. 2018;90:49–58. doi: 10.1016/j.jaut.2018.01.004. [DOI] [PubMed] [Google Scholar]
- Peres R.S., Liew F.Y., Talbot J., Carregaro V., Oliveira R.D., Almeida S.L., França R.F.O., Donate P.B., Pinto L.G., Ferreira F.I.S., Costa D.L., Demarque D.P., Gouvea D.R., Lopes N.P., Queiroz R.H.C., Silva J.S., Figueiredo F., Alves-Filho J.C., Cunhaa T.M., Ferreir S.H., Louzada-Junior P., Cunha F.Q. Low expression of CD39 on regulatory T cells as a biomarker for resistance to methotrexate therapy in rheumatoid arthritis. Proc. Natl. Acad. Sci. U. S. A. 2015 doi: 10.1073/pnas.1424792112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pillinger T., Osimo E.F., Brugger S., Mondelli V., McCutcheon R.A., Howes O.D. A meta-analysis of immune parameters, variability, and assessment of modal distribution in psychosis and test of the immune subgroup hypothesis. Schizophr. Bull. 2018 doi: 10.1093/schbul/sby160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plavén-Sigray P., Matheson G.J., Collste K., Ashok A.H., Coughlin J.M., Howes O.D., Mizrahi R., Pomper M.G., Rusjan P., Veronese M., Wang Y., Cervenka S. Positron emission tomography studies of the glial cell marker translocator protein in patients with psychosis: a meta-analysis using individual participant data. Biol. Psychiatr. 2018;84:433–442. doi: 10.1016/j.biopsych.2018.02.1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plavén-Sigray P., Matheson G.J., Coughlin J.M., Hafizi S., Laurikainen H., Ottoy J., De Picker L., Rusjan P., Hietala J., Howes O.D., Mizrahi R., Morrens M., Pomper M.G., Cervenka S. Meta-analysis of the glial marker TSPO in psychosis revisited: reconciling inconclusive findings of patient–control differences. Biol. Psychiatr. 2020 doi: 10.1016/j.biopsych.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poletti S., de Wit H., Mazza E., Wijkhuijs A.J.M., Locatelli C., Aggio V., Colombo C., Benedetti F., Drexhage H.A. Th17 cells correlate positively to the structural and functional integrity of the brain in bipolar depression and healthy controls. Brain Behav. Immun. 2017;61:317–325. doi: 10.1016/j.bbi.2016.12.020. [DOI] [PubMed] [Google Scholar]
- Qiao Y.C., Shen J., He L., Hong X.Z., Tian F., Pan Y.H., Liang L., Zhang X.X., Zhao H.L. Changes of regulatory T cells and of proinflammatory and immunosuppressive cytokines in patients with type 2 diabetes mellitus: a systematic review and meta-analysis. J. Diabetes Res. 2016 doi: 10.1155/2016/3694957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramaker R.C., Bowling K.M., Lasseigne B.N., Hagenauer M.H., Hardigan A.A., Davis N.S., Gertz J., Cartagena P.M., Walsh D.M., Vawter M.P., Schatzberg A.F., Barchas J.D., Watson S.J., Bunney B.G., Akil H., Bunney W.E., Li J.Z., Cooper S.J., Myers R.M. Post-mortem molecular profiling of three psychiatric disorders. Genome Med. 2017 doi: 10.1186/s13073-017-0458-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riordan N.H., Hincapié M.L., Morales I., Fernández G., Allen N., Leu C., Madrigal M., Paz Rodríguez J., Novarro N. Allogeneic human umbilical cord mesenchymal stem cells for the treatment of autism spectrum disorder in children: safety profile and effect on cytokine levels. Stem Cells Transl. Med. 2019;8:1008–1016. doi: 10.1002/sctm.19-0010. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Rosenzwajg M., Lorenzon R., Cacoub P., Pham H.P., Pitoiset F., El Soufi K., RIbet C., Bernard C., Aractingi S., Banneville B., Beaugerie L., Berenbaum F., Champey J., Chazouilleres O., Corpechot C., Fautrel B., Mekinian A., Regnier E., Saadoun D., Salem J.-E., Sellam J., Seksik P., Daguenel-Nguyen A., Doppler V., Mariau J., Vicaut E., Klatzmann D. Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann. Rheum. Dis. 2019;78:209–217. doi: 10.1136/annrheumdis-2018-214229. [DOI] [PubMed] [Google Scholar]
- Rowe J.H., Ertelt J.M., Xin L., Way S.S. Regulatory T cells and the immune pathogenesis of prenatal infection. Reproduction. 2013 doi: 10.1530/REP-13-0262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahbaz C., Zibandey N., Kurtulmus A., Duran Y., Gokalp M., Kırpınar I., Sahin F., Guloksuz S., Akkoc T. Reduced regulatory T cells with increased proinflammatory response in patients with schizophrenia. Psychopharmacology (Berl) 2020;237:1861–1871. doi: 10.1007/s00213-020-05504-0. [DOI] [PubMed] [Google Scholar]
- Sakaguchi S., Yamaguchi T., Nomura T., Ono M. Regulatory T cells and immune tolerance. Cell. 2008;133:775–787. doi: 10.1016/j.cell.2008.05.009. [DOI] [PubMed] [Google Scholar]
- Samson M., Audia S., Janikashvili N., Ciudad M., Trad M., Fraszczak J., Ornetti P., Maillefert J., Miossec P., Bonnotte B. Inhibition of IL-6 function corrects Th17/Treg imbalance in rheumatoid arthritis patients. Arthritis Rheum. 2012 doi: 10.1002/art.34477. [DOI] [PubMed] [Google Scholar]
- Sarantopoulos A., Tselios K., Gkougkourelas I., Pantoura M., Georgiadou A.-M., Boura P. Tocilizumab Treatment Leads to a Rapid and Sustained Increase in Treg Cell Levels in Rheumatoid Arthritis Patients: comment on the Article by Thiolat et al. Arthritis Rheum. 2014;66:2638. doi: 10.1002/art.38714. 2638. [DOI] [PubMed] [Google Scholar]
- Sharabi A., Tsokos M.G., Ding Y., Malek T.R., Klatzmann D., Tsokos G.C. Regulatory T cells in the treatment of disease. Nat. Rev. Drug Discov. 2018 doi: 10.1038/nrd.2018.148. [DOI] [PubMed] [Google Scholar]
- Shevach E.M. Mechanisms of Foxp3+ T regulatory cell-mediated suppression. Immunity. 2009;30:636–645. doi: 10.1016/j.immuni.2009.04.010. [DOI] [PubMed] [Google Scholar]
- Shu Y., Hu Q., Long H., Chang C., Lu Q., Xiao R. Epigenetic variability of CD4+CD25+ Tregs contributes to the pathogenesis of autoimmune diseases. Clin. Rev. Allergy Immunol. 2017;52:260–272. doi: 10.1007/s12016-016-8590-3. [DOI] [PubMed] [Google Scholar]
- Subrahmanyam P.B., Maecker H.T. Methods in Molecular Biology. 2021. Mass cytometry analysis of T-helper cells; pp. 49–63. [DOI] [PubMed] [Google Scholar]
- Tada Y., Ono N., Suematsu R., Tashiro S., Sadanaga Y., Tokuda Y., Ono Y., Nakao Y., Maruyama A., Ohta A., Koarada S. The balance between Foxp3 and Ror-γt expression in peripheral blood is altered by tocilizumab and abatacept in patients with rheumatoid arthritis. BMC Muscoskel. Disord. 2016;17:290. doi: 10.1186/s12891-016-1137-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teles A., Zenclussen A.C., Schumacher A. Regulatory T cells are baby's best friends. Am. J. Reprod. Immunol. 2013 doi: 10.1111/aji.12067. [DOI] [PubMed] [Google Scholar]
- Thiolat A., Semerano L., Pers Y.M., Biton J., Lemeiter D., Portales P., Quentin J., Jorgensen C., Decker P., Boissier M.C., Louis-Plence P., Bessis N. Interleukin-6 receptor blockade enhances CD39+ regulatory T cell development in rheumatoid arthritis and in experimental arthritis. Arthritis Rheum. 2014 doi: 10.1002/art.38246. [DOI] [PubMed] [Google Scholar]
- Toker L., Mancarci B.O., Tripathy S., Pavlidis P. Transcriptomic evidence for alterations in astrocytes and parvalbumin interneurons in subjects with bipolar disorder and schizophrenia. Biol. Psychiatr. 2018:1–10. doi: 10.1016/j.biopsych.2018.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upthegrove R., Manzanares-Teson N., Barnes N.M. Cytokine function in medication-naive first episode psychosis: a systematic review and meta-analysis. Schizophr. Res. 2014;155:101–108. doi: 10.1016/j.schres.2014.03.005. [DOI] [PubMed] [Google Scholar]
- Wang Y., Zheng J., Islam M.S., Yang Y., Hu Y., Chen X. The role of cd4+foxp3+ regulatory t cells in the immunopathogenesis of covid-19: implications for treatment. Int. J. Biol. Sci. 2021 doi: 10.7150/ijbs.59534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie L., Choudhury G.R., Winters A., Yang S.-H., Jin K. Cerebral regulatory T cells restrain microglia/macrophage-mediated inflammatory responses via IL-10. Eur. J. Immunol. 2015;45:180–191. doi: 10.1002/eji.201444823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z., Zhang Xiaoyun, Chang H., Kong Y., Ni Y., Liu R., Zhang Xiaolin, Hu Y., Yang Z., Hou M., Mao R., Liu W., Du Y., Yu S., Wang Z., Ji M., Zhou Z. Rescue of maternal immune activation-induced behavioral abnormalities in adult mouse offspring by pathogen-activated maternal Treg cells. Nat. Neurosci. 2021 doi: 10.1038/s41593-021-00837-1. [DOI] [PubMed] [Google Scholar]
- You M.-J., Bang M., Park H.-S., Yang B., Jang K.B., Yoo J., Hwang D.-Y., Kim M., Kim B., Lee S.-H., Kwon M.-S. Human umbilical cord-derived mesenchymal stem cells alleviate schizophrenia-relevant behaviors in amphetamine-sensitized mice by inhibiting neuroinflammation. Transl. Psychiatry. 2020;10:123. doi: 10.1038/s41398-020-0802-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng H., Chi H. The interplay between regulatory T cells and metabolism in immune regulation. OncoImmunology. 2013 doi: 10.4161/onci.26586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J., Lv S., Liu X., Song B., Shi L. Umbilical cord mesenchymal stem cell treatment for Crohn's disease: a randomized controlled clinical trial. Gut Liver. 2018;12:73–78. doi: 10.5009/gnl17035. [DOI] [PMC free article] [PubMed] [Google Scholar]