Abstract
Introduction
Anesthesia during pregnancy can impair fetal neurodevelopment, but effects of surgery remain unknown. The aim is to investigate effects of abdominal surgery on fetal brain development. Hypothesis is that surgery impairs outcome.
Methods
Pregnant rabbits were randomized at 28 days of gestation to 2 h of general anesthesia (sevoflurane group, n = 6) or to anesthesia plus laparoscopic appendectomy (surgery group, n = 13). On postnatal day 1, neurobehavior of pups was assessed and brains harvested. Primary outcome was neuron density in the frontal cortex, and secondary outcomes included neurobehavioral assessment and other histological parameters.
Results
Fetal survival was lower in the surgery group: 54 versus 100% litters alive at birth (p = 0.0442). In alive litters, pup survival until harvesting was 50 versus 69% (p = 0.0352). No differences were observed for primary outcome (p = 0.5114) for surviving pups. Neuron densities were significantly lower in the surgery group in the caudate nucleus (p = 0.0180), but not different in other regions. No differences were observed for secondary outcomes. Conclusions did not change after adjustment for mortality.
Conclusion
Abdominal surgery in pregnant rabbits at a gestational age corresponding to the end of human second trimester results in limited neurohistological changes but not in neurobehavioral impairments. High intrauterine mortality limits translation to clinical scenario, where fetal mortality is close to zero.
Keywords: Fetus, Neurobehavior, Neuron density, Pregnancy, Surgery
Introduction
Commonly used drugs in anesthesia like propofol, sevoflurane, and fentanyl rapidly cross the placenta and can therefore potentially affect fetal brain development (online suppl. Table 1; for all online suppl. material, see www.karger.com/doi/10.1159/000512489) [1–3]. In 2016, the US Food and Drug Administration issued a warning that the exposure of the developing brain to anesthesia might be associated with impaired neurocognitive outcome later in life [4, 5]. This concern was based on preclinical evidence. Both in newborn and pregnant animals, it has been shown that repeated or prolonged exposure to virtually all commonly used anesthetics causes apoptotic neurodegeneration in the developing brain, resulting in persisting neurocognitive impairments [6–13]. Several retrospective clinical studies suggest that in young children, anesthesia exposure could be associated with structural brain deficits that result in developmental disorders later in life [14–19]. Contrariwise, prospective and randomized studies failed to demonstrate such an association [20–22]. The clinical relevance of these observations remains an open debate.
There is much less evidence of the effects of surgery on fetal brain development. Maternal surgery during pregnancy in rodent models has been reported to impair fetal neurobehavioral outcome [23, 24], but rabbit and guinea pig models did not show (long-term) neurological impairments after laparotomy or carbon dioxide pneumoperitoneum [25, 26]. In neonatal rat models, it has been demonstrated that pain increases neuroapoptosis [27], but the opposite effect has also been reported, suggesting a potentially neuroprotective effect [28]. A similar neuro-protective effect has been reported for fetal surgery in sheep [29]. Clinical studies are limited to the observation that maternal surgery during pregnancy is associated with adverse pregnancy outcomes, including a higher incidence of preterm delivery and lower birth weight [30, 31]. The neurodevelopmental effects of surgery ± anesthesia during pregnancy in humans have not been investigated yet.
Exposure to anesthesia without (pronounced) organ manipulation occurs less frequently during pregnancy, but is necessary when pregnant patients require sedation for minimal invasive interventional cardiology (e.g., ablation of atrial flutter), for interventional radiology (e.g., clipping of a cerebral artery aneurysm), or for endoscopy (e.g., endoscopic retrograde cholangiopancreatography and colonoscopy). Moreover, sometimes pregnant women require sedation without surgery on the intensive care unit (e.g., for the treatment of coronavirus disease 19) [31–33]. Much more frequent, however, is the situation in which pregnant woman are exposed to general anesthesia to perform a surgical procedure with organ manipulation. A first reason for surgery is for maternal indications, with the fetus being an innocent bystander. Every year, 0.5–0.7% of pregnant women need to undergo anesthesia for nonobstetric surgery [30, 31]. Most of the procedures are performed during the second trimester. Forty-nine percent of all procedures are intra-abdominal procedures (e.g., laparoscopic appendectomy, detorsion of ovarian mass, cholecystectomy, reduction of internal herniation, and sigmoid resection) [30, 31]. A second, yet less frequent, situation is that the fetus itself needs surgery, for example, for the repair of an open spina bifida (SB) [34, 35]. In 2018 and 2019, a total of 333 fetal procedures were performed in the University Hospitals of Leuven, 34 of which were prenatal SB repairs under general anesthesia.
Recently, we demonstrated that the exposure of pregnant rabbits to sevoflurane anesthesia without organ manipulation transiently impairs neurobehavior and decreases neuron density, when compared to no anesthesia [26]. Because surgery with organ manipulation is much more frequent during pregnancy and because the current evidence of the effects of maternal surgery during pregnancy is conflicting, there is a need to further investigate the effects of clinically relevant maternal surgery on fetal brain development. Therefore, the aim of this study was to investigate the effects of abdominal surgery on fetal brain development in the rabbit model by disentangling the effects for anesthesia from those of surgery. We hypothesized that maternal surgery impairs neurocognitive outcome in comparison with anesthesia without surgery.
Materials and Methods
Nineteen drug-naive time-mated pregnant rabbits (hybrids of New Zealand and the Flemish Giant rabbit) (on average 6 months old) were obtained from a certified breeder. After transport, the animals were acclimatized at least 3 days before the start of the experiments. The rabbits were conventionally housed in individual cages at 21°C and 42% humidity, with a 12-h day-night cycle and free access to water and food. At a gestational age (GA) of 28 days, corresponding to the end of the second trimester in humans [36, 37], does were randomized to either the sevoflurane group (n = 6) or the surgery group (n = 13).
Anesthesia and Surgery
Does underwent 2 h of general anesthesia at GA = 28 days, using a similar anesthesia protocol as previously described (online suppl. 1.1) [26]. After inhalation of 8 vol% sevoflurane, arterial and venous catheters were placed. Propofol was administered intravenously, and the rabbit was intubated. The rabbits were ventilated with one minimum alveolar concentration of sevoflurane in 30% oxygen. Ventilation was adjusted to maintain normocapnia [38]. Fentanyl was administered intravenously, and antibiotics and medroxyprogesterone acetate were given subcutaneously. Does were continuously monitored using pulse oximetry, electrocardiography, invasive arterial blood pressure monitoring, measurement of ventilatory volumes, pressures, and gas analysis (inspiratory and expiratory O2, CO2, and sevoflurane). Esophageal temperature was maintained between 38 and 39.5°C using a heating pad and heating lamp. An arterial blood gas sample was taken 10 min after intubation and 10 min before extubation (sevoflurane group) or immediately before releasing the capnoperitoneum (surgery group). The mean arterial pressure was maintained above 80% of the awake values using phenylephrine [38]. The rabbits were woken up 2 h after intubation.
In the sevoflurane group, there were no additional manipulations. In the surgery group, a laparoscopic appendectomy was performed (online suppl. 1.2). First, a 5-mm camera trocar was placed, the peritoneal cavity was insufflated with CO2 at a pressure of 7 mm Hg, and 2 additional 3-mm trocars were inserted. After dissection and ligation of the appendix, 7 cm of the distal end of the appendix was removed from the abdomen. The abdomen was deflated, trocars removed, and surgical wounds sutured and infiltrated with levobupivacaine.
Ten minutes after intubation and 10 min before the end of anesthesia, a maternal serum sample was obtained and the concentrations of interleukin 6 (IL-6) and interleukin 10 (IL-10) were measured (online suppl. 1.3).
Cesarean Section, Neurobehavioral Testing, and Harvesting
At term (GA = 31 days), an arterial blood sample was taken to obtain a full blood count. A cesarean section was performed under local anesthesia and sedation to standardize fetal stress during birth. After delivery of the pups, the does were euthanized. The pups were kept in a warmed and humidified incubator and fed 3 h after birth (online suppl. 1.4) [26, 39].
Twenty-four hours after birth, the pups underwent neurocognitive testing using a validated scale which assesses different motoric and sensory aspects [26, 39–41]. In addition to the original scale, the number of hops and the number of falling overs were counted as ordinal variables. The videos of the assessment were scored a posterior by 2 blinded observers (online suppl. 2.1).
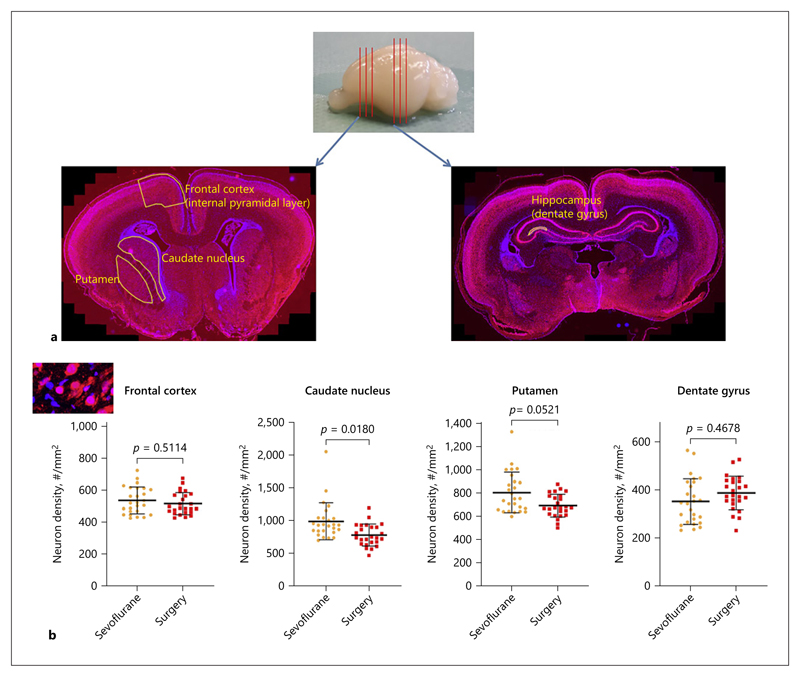
After neurocognitive assessment, the pups were sacrificed to obtain brain histology. Following anesthesia, the pups were transcardially perfused with 0.9% saline followed by formaldehyde. The brains were extracted, embedded in paraffin, and serially cut to obtain slides of the frontal cortex, caudate nucleus, putamen, and hippocampus (Fig. 1a, online suppl. 2.2). These brain regions are involved in the control of memory (hippocampus, caudate nucleus, and putamen), planning/problem solving (frontal cortex), and movement (caudate nucleus and putamen) [42–44].
Fig. 1. Neuron density.
a The brains were cut to obtain 2 sets of 3 consecutive slides (distance: 100 μm between the slides). b Neuron densities counted manually on NeuN-stained slides. Data are shown as individual data points, and bars represent mean and standard deviation. NeuN, neuronal nuclei.
Neuron densities were counted manually on slides stained for the neuronal nuclei (NeuN) marker, using similar methods as before [26, 45–50]. Total cell densities were counted automatically in whole brain regions using Hoechst staining. Synaptophysin, Ki67, and ionized calcium-binding adaptor molecule 1 markers were used to measure synaptogenesis [26, 51], brain proliferation [26, 52, 53], and inflammation [54], respectively (online suppl. 2.3). All slides were digitally scanned, and quantifications were done using Qupath [55] (Centre for Cancer Research & Cell Biology, Northern Ireland, Open source) and ImageJ [56] (Fiji, Los Angeles, CA, USA) software (online suppl. 2.4).
Statistical Analysis
Primary outcome was the neuron density in the frontal cortex. Outcome parameters of the pups were analyzed using a linear mixed-effects model to correct for the fact that the pups of 1 litter are not independent observations (online suppl. 3) [57–61]. Using this model, the sample size was calculated a priori for the primary outcome. Parameters measured on the level of the does were analyzed using the Mann-Whitney U test. The inter- and intrarater reliability of the neurobehavioral evaluation was assessed using Spearman correlation (online suppl. 3). All analyses were performed using SAS software (SAS System for Windows version 9.4; SAS Institute Inc., Cary, NC, USA). The graphs were constructed using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). Data are mentioned as mean ± standard deviation.
Results
Maternal Outcomes
Maternal parameters during anesthesia (and surgery) were comparable (Table 1). Significant differences were limited to the weight of the does and the inspiratory peak airway pressure, which were significantly higher in the surgery group. Blood gas results were comparable (Table 2), with the exception of bicarbonate levels, which were lower in the surgery group. There were no significant differences between both groups in the concentrations of IL-6 and IL-10 measured in maternal serum 10 min before the end of anesthesia (online suppl. Fig. 1). The time of capnoperitoneum was 48 ± 14 min. There were no surgical complications, and all surgeries could be performed without manipulation of the uterus. None of the rabbits delivered prematurely or had a miscarriage. The full blood count and rectal temperature measured 3 days after anesthesia were comparable (online suppl. Table 2).
Table 1. Maternal parameters during anesthesia.
| Sevoflurane | Surgery | p value | |
|---|---|---|---|
| Does, n | 6 | 13 | |
| Weight, kg | 4.3±0.4 | 4.9±0.4 | 0.0106 |
| Inspiratory O2 concentration, % | 28±11 | 25±9 | 0.0793 |
| End-tidal sevoflurane concentration, % | 3.7±0.1 | 3.7±0.1 | 0.1738 |
| End-tidal partial CO2 pressure, mm Hg | 33±4 | 30±3 | 0.2364 |
| Tidal volume, mL/kg | 10.8±1.7 | 11.7±10.7 | 0.2364 |
| Respiratory rate, /min | 36±7 | 38±4 | 0.656 |
| Inspiratory peak pressure, cm H2O | 21±4 | 25±5 | 0.0393 |
| Heart rate, /min | 216±28 | 216±29 | 0.6931 |
| Systolic blood pressure, mm Hg | 69±12 | 69±15 | 0.9301 |
| Diastolic blood pressure, mm Hg | 40±8 | 41±9 | 0.6294 |
| Mean arterial pressure, mm Hg | 50±8 | 50±11 | 1 |
| Pulse oximeter oxygen saturation, % | 98±2 | 98±3 | 0.7589 |
| Esophageal temperature, °C | 38.7±1.1 | 38.2±0.8 | 0.4047 |
| Total dose phenylephrine, μg | 253±191 | 258±137 | 0.7923 |
Maternal parameters during the 2-h general anesthesia (± surgery) period on gestational day 28. Values are displayed as mean±standard deviation. Significant differences (p < 0.05) are highlighted in bold.
Table 2. Maternal arterial blood gas results.
| Sevoflurane | Surgery | p value | |
|---|---|---|---|
| Start of anesthesia | |||
| pH | 7.45±0.09 | 7.41±0.05 | 0.5683 |
| paCO2, mm Hg | 32±2 | 31±2 | 0.3286 |
| paO2, mm Hg | 148±14 | 155±10 | 0.4283 |
| Hemoglobin, g/dL | 9.9±1.2 | 9.7±0.8 | 0.6294 |
| Potassium, mmol/L | 3.4±0.5 | 3.5±0.5 | 1 |
| Glucose, mg/dL | 132±43 | 123±21 | 1 |
| Lactate, mmol/L | 4.1±1.1 | 4.6±1.1 | 0.4552 |
| HCO3-, mmol/L | 22.5±4.2 | 18.6±1.7 | 0.0146 |
| End of anesthesia | |||
| pH | 7.43±0.06 | 7.39±0.07 | 0.2191 |
| paCO2, mm Hg | 32±5 | 29±3 | 0.1748 |
| paO2, mm Hg | 164±53 | 153±17 | 0.5681 |
| Hemoglobin, g/dL | 9.1±1.0 | 9.2±0.9 | 1 |
| Potassium, mmol/L | 3.4±0.5 | 3.5±0.5 | 0.9297 |
| Glucose, mg/dL | 136±27 | 156±33 | 0.2729 |
| Lactate, mmol/L | 4.5±2.0 | 5.2±2.2 | 0.8264 |
| HCO3-, mmol/L | 21.3±5.0 | 15.9±2.8 | 0.0222 |
Maternal arterial blood gas results during anesthesia at gestational day 28. The first sample was taken 10 min after intubation. The second sample was obtained 10 min before the end of the anesthesia time of 2 h (sevoflurane group) or immediately before the end of the capnoperitoneum (surgery group). Values are displayed as mean±standard deviation. Significant differences (p < 0.05) are highlighted in bold.
Fetal Outcomes
Primary Outcome
There were no significant differences in the neuron densities in the frontal cortex (517 ± 70 and 537 ± 85/ mm2, p = 0.5114) (Fig. 1b).
Survival and Biometrics
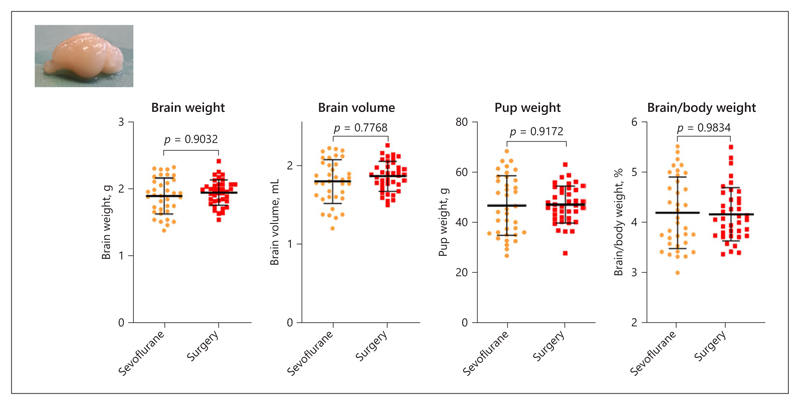
At the moment of cesarean section, all 6 does of the sevoflurane group had litters with alive pups. In contrast, in the surgery group, 6 out of the 13 does (46%) had litters of which all fetuses had died in utero (p = 0.0442) (online suppl. Table 4). For the does with surviving litters, 69% of all pups survived until the moment of harvesting in the sevoflurane group versus 50% in the surgery group (p = 0.0352). There was no difference in postnatal mortality (14% of the total number of pups [death and alive] vs. 12%, p = 0.6203), but there was a difference in the in utero fetal deaths (IUFD) (17% of all pups vs. 38%, p = 0.0079). The number of macerated stillbirths was significantly lower in the sevoflurane group (11 vs. 37%, p = 0.0010), but there was no significant difference in the fresh stillbirths (6 vs. 1%, p = 0.1678). Biometric data were comparable for both groups (Fig. 2).
Fig. 2.
Biometrics. Biometrics of the pups: the weight of the pups 1 day after birth, the weight and volume of the brains of the pups after harvesting, and the brain to body weight ratio. Data are shown as individual data points, and bars represent mean and standard deviation.
Neurobehavioral Assessment
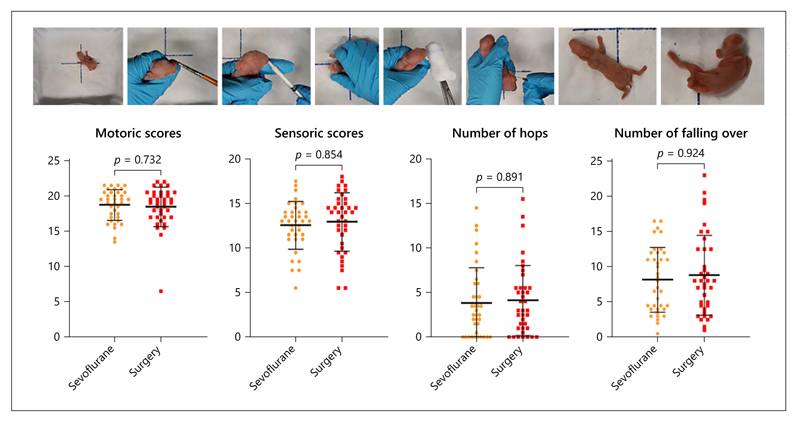
Neurobehavioral assessments of both groups were comparable (Fig. 3). There were no significant differences in motoric and sensory scores and number of hops and falling overs. The interrater reliability was moderate for the motoric scores, good for both the sensory scores and number of hops, and strong for the number of falling overs (online suppl. Fig. 2). The intrarater reliability was for every component good or strong for both observers (online suppl. Fig. 3).
Fig. 3.
Neurobehavioral assessment. The motoric scores, sensoric scores, number of hops, and number of falling overs of the pups 1 day after birth. Data are shown as individual data points, and bars represent mean and standard deviation. Pictures from left to right: open field test, facial/whisker touch response, sucking and swallowing, surface righting reflex, odor aversion, pain sensation, synchronous hop, and falling over.
Brain Histology
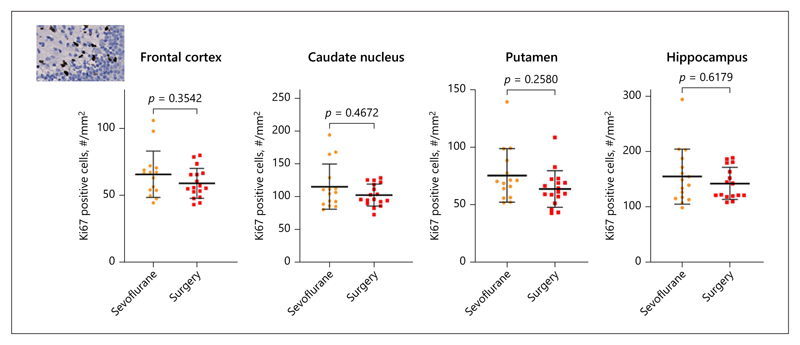
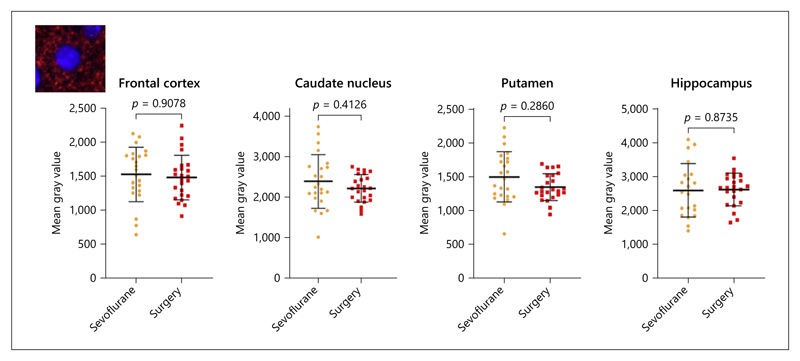
No significant differences were observed in the total cell densities (online suppl. Fig. 4). Neuron densities (Fig. 1b) were significantly lower in the surgery group in the caudate nucleus (781 ± 168 vs. 991 ± 280/mm2, p = 0.0180), tended to be lower in the putamen (694 ± 99 vs. 807 ± 175/mm2, p = 0.0521), and were comparable in the frontal cortex (517 ± 70 vs. 537 ± 85/mm2, p = 0.5114) and dentate gyrus (388 ± 71 vs. 353 ± 95/mm2, p = 0.4678). Proliferation, synaptophysin levels, and inflammation were comparable for both groups (Fig. 4, 5, online suppl. Fig. 5).
Fig. 4.
Proliferation. Proliferation in the brains of the pups measured using the Ki67 marker. Data are shown as individual data points, and bars represent mean and standard deviation.
Fig. 5.
Synaptogenesis. Synaptophysin levels in the brains, measured as the mean gray value. Data are shown as individual data points, and bars represent mean and standard deviation.
Discussion
Principal Findings
We recently reported that anesthesia without organ manipulation in pregnant rabbits resulted in a transiently impaired neurobehavioral outcome and decreased neuron densities, when compared to a sham group without anesthesia [26]. However, general anesthesia in pregnant women is more frequently used to perform surgical procedures, and the combined effects of anesthesia plus surgery on neurodevelopmental outcome remain controversial. The aim of this study was, therefore, to investigate the effects of abdominal surgery on top of general anesthesia on fetal brain development in pregnant rabbits with a GA corresponding to the end of the second trimester in humans, that is, the period during which abdominal surgery has to be performed most frequently [31, 36, 37]. First, there were more macerated IUFD in the surgery group. In survivors, however, there were no significant differences in neuron density in the frontal cortex. Neuron densities were, however, significantly lower in the caudate nucleus and tended to be lower in the putamen in the surgery group, though without any other differences.
Results
Our observations suggest that, in rabbits, abdominal surgery impairs fetal survival. The incidence of macerated IUFD in the surgery group was significantly increased, while there was no difference in fresh stillbirths or postnatal mortality. Several factors may have contributed to the excess IUFD: the application of a 7-mm Hg capnoperitoneum, surgical manipulation, inflammation, a hitherto unknown vital function of the appendix in rabbits, or the susceptibility of rabbit fetuses. However, in guinea pigs, sheep, and humans, it has been demonstrated that laparoscopy during pregnancy is safe for the fetus, that is, it is not associated with an excess of fetal mortality [25, 62–64]. Anesthetic factors seem not to be involved as in the surgery group, anesthesia parameters and blood gas results of pregnant does with surviving pups were comparable with the parameters of does with dead litters. There was also no correlation between CO2 insufflation time and litter mortality (Spearman ρ = -0.249, p = 0.412). The duration of the capnoperitoneum was not correlated with neuron density, when insufflation time was added as a covariate to the mixed-effects model. Also, other maternal parameters were comparable for both groups. The difference in body weight of the does can be explained by pure chance only, as the does were strictly randomized. Higher airway pressures in the surgery group can be attributed to the CO2 pneumoperitoneum required to perform laparoscopy. Both the vital parameters and blood gas results were within the range of healthy awake pregnant rabbits [38]. Exceptions were a higher partial oxygen pressure and higher glucose blood levels in the surgery rabbits, but it is unlikely that these would contribute to IUFD. Though statistically significantly lower, the bicarbonate in the surgery group was above the 5th percentile of healthy awake rabbits, and no acidosis was observed [38]. Hence, we do not think the statistical difference to be of any clinical relevance. Likewise, surgical manipulation and inflammation are unlikely explanations because the uterus was not touched during surgery and inflammatory parameters were not increased. The appendix was only partially removed in this study. Even if an important gastrointestinal or immunological function would have been compromised, it would not explain that most IUFD were probably around or immediately after the operation. Fetal mortality is high in rabbits in all types of experiments in which physiological homeostasis is disturbed (e.g., prematurity [39, 65], surgery to induce fetal growth restriction [66, 67], or SB [68]) when compared to larger animal models (e.g., sheep [69]) and humans. Probably, the susceptibility of fetuses to surgical stimuli in general in small animal models is the explanation for the high IUFD.
When IUFD was added as a covariate to the mixed-effects model, IUFD was not correlated with neuron densities (frontal cortex: p = 0.8727, caudate nucleus: p = 0.7520, putamen: p = 0.5547, and dentate gyrus: p = 0.8082). The conclusions for the group effects also remain the same. This suggests that IUFD and neuron densities are independent outcome parameters.
This study was designed to document potential effects of abdominal surgery on the brain. We did not find obvious impairment by surgery. The differences were limited to a lower neuron density in the caudate nucleus. The loss of neurons in this region involved in movement was not reflected by a decreased motoric score. As many statistical comparisons were performed in this study and only one was significant, it is possible that this difference was observed by random sampling error.
The underlying mechanisms for impairment of neuro-development after exposure to anesthesia during early childhood or pregnancy remain still unknown. One mechanism that has been suggested is (neuro-) inflammation [24, 70–72]. In our study, we could not document signs of increased maternal inflammation. The cytokines IL-6 and IL-10 and leukocyte counts were unchanged, and these have been earlier used in rabbits as a measure of inflammation [73, 74]. IL-6 has been previously shown to be involved in the pathways of neuroinflammation, resulting in impaired fetal brain development [24, 70, 71]. Also, the density of microglial cells (marker of neuroinflammation) did not differ. The absence of inflammation could be an explanation for the limited differences in fetal brain development.
Clinical and Research Implications
This study could not demonstrate an effect of abdominal surgery under anesthesia on brain development. We observed only limited effects for neurohistological parameters, with all but one parameter being unaffected by surgery. We were neither able to detect any neurocognitive impact. Our results are therefore in line with previous findings showing that maternal surgery during pregnancy does not result in major differences compared to anesthesia without surgery [25], or only in slightly impaired neurological outcome [23, 24, 27]. As the high IUFD is probably the consequence of the susceptibility of fetuses in a small animal model, the results should be confirmed in a large animal model.
Strengths and Limitations
We acknowledge that our study has several limitations. First, IUFD rate after abdominal surgery was exorbitant and bears no similarity to the clinical situation, where IUFD rates are close to zero [64]. This questions per se the translational character of our study. Second, abdominal surgery was performed in healthy rabbits without underlying pathology necessitating surgery. Hence, there was no local and/or systemic inflammation. Third, a laparoscopic appendectomy in rabbits is not an exact correlate of this procedure performed in humans due to the different anatomy of the cecum and appendix [75, 76]. Therefore, a laparoscopic appendectomy in rabbits is probably rather the equivalent of more invasive abdominal surgery in humans (e.g., complicated cholecystectomy, reduction of internal herniation, and sigmoid resection). Fourth, rabbits have multiple fetuses, thereby not entirely mimicking the clinical reality in humans. Fifth, it cannot be ruled out that surgery had a more pronounced effect when studying other GAs. However, this is unlikely because the timing of our experiment already reflects the worst case scenario in which surgery is performed at the moment on which the developing brain is most vulnerable to noxious factors, that is, the beginning of brain growth spurt and peak synapto-genesis [36, 37, 77]. Sixth, this study did not include a control group without anesthesia. The primary objective of this study was to elucidate the effects of surgery plus anesthesia versus anesthesia alone on fetal brain development. Of note, the effects of anesthesia versus awake animals on fetal brain development and the validity of our model have already been demonstrated in our previous study [26] and were, therefore, out of the scope of this study. Seventh, even minimal surgery in the rabbit model results in fetal mortality. As this is inherent to this model, the sevoflurane group is probably not the most appropriate group to investigate fetal mortality after abdominal surgery. Sham surgery (e.g., capnoperitoneum without surgery) would have been more appropriate regarding to the mortality.
Our study also has some strengths. The fetal rabbit model has multiple advantages over other animal models. First, the rabbit is a relevant model to investigate factors affecting the brain development [78]. The most vulnerable period of brain development is the brain growth spurt, the period during which the brain is at its peak growth [78–80]. The brain growth spurt occurs in humans as well as in rabbits perinatally, whereas in other often used species, such as rodents, this peak is postnatally, or in nonhuman primates prenatally [78, 80]. The GA of 28 days in rabbits is at the onset of the brain growth spurt and the peak synaptogenesis and corresponds to the end of the human second trimester [36, 37, 77]. Second, both the assessment of neurobehavior and brain histology have been validated, and hence are robust and reliable [26, 39–41]. Third, the rabbit allows the use of well-controlled anesthesia and to use the American Society of Anesthesiologists monitoring [26].
Conclusions
Abdominal surgery in pregnant rabbits at a GA corresponding to the end of the second trimester in humans does not affect fetal brain development. We were unable to observe impairments in neurocognitive function or brain histology in the surviving pups. While we demonstrated a lower neuron density in the caudate nucleus, no differences could be found in other brain regions, neither in biometrics, neurobehavior, total cell densities, proliferation, synaptogenesis, or brain inflammation. The study is limited by a high IUFD rate after abdominal surgery which is in contrast to human abdominal surgery in which fetal mortality is luckily close to zero. This limits the translation of the results of this study to the clinical scenario.
Supplementary Material
Acknowledgements
The authors would like to thank the G-PURE laboratory technicians Katrien Luyten and Sofie Jannes for the assistance in the histological stainings and Rita Van Bree for the enzyme-linked immunosorbent assays readouts of the maternal cytokines. They would like to thank the technicians of the Center for Surgical Technologies: Imke Loret and Rosita Kinnart for their help during the in vivo parts of the animal experiments and Gert Van den Bergh for the logistic assistance and supporting them with laparoscopic material. Finally, the authors would like to thank Steffen Fieuws (Leuven Biostatistics and Statistical Bioinformatics Centre, KU Leuven) for his statistical advice.
Funding Sources
T.B., S.D., M.V.D.V., and S.R. are funded by a Society for Anesthesia and Resuscitation of Belgium (SARB) research grant and by an Obstetric Anaesthetists’ Association (OAA) international grant. L.V.D.V., J.V.D.M., and D.B. are funded by the Erasmus+ Programme of the European Union (Framework Agreement No. 2013-0040). L.J. is funded by the Applied Biomedical Research grant from the Research Foundation - Flanders (FWO) (No. ZL3953000). J.D. is funded by the Great Ormond Street Hospital Charity Fund. Part of the research on fetal surgery is funded by the Welcome Trust (WT101957) and Engineering and Physical Sciences Research Council (ESPRC) (NS/A000027/1). The authors obtained a loan from Karl Storz for the laparoscopic instruments and hardware. None of these funding sources played a role in the study design, data collection, analysis, interpretation, or writing of the article.
Footnotes
Statement of Ethics
The experiment was approved by the Ethics Committee for Animal Experimentation of the Animalium of KU Leuven (P186/2018), and the local guidelines for the care and use of the animals were followed. The guidelines of Animal Research Reporting of In Vivo Experiments (ARRIVE) and SmartTots pre-clinical working group (Chinn G, British Journal of Anaesthesia, 2020) were followed.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Author Contributions
All authors substantially contributed to the manuscript. The study was designed by S.R., J.D., T.B., L.V.D.V., and M.V.D.V. The in vivo parts were done by T.B., L.V.D.V., S.D., S.V., D.E., J.V.D.M., E.G., L.J., and D.B. Histology was performed and analyzed by T.B. The videos of the neurobehavior were evaluated independently by T.B. and E.G. Statistical analysis and writing the first version of the manuscript were done by T.B. All coauthors made substantial contributions to the design of the study; acquisition, analysis, and interpretation of data; and critically revised and approved the final submitted manuscript.
References
- 1.Satoh D, Iwatsuki N, Naito M, Sato M, Hashimoto Y. Comparison of the placental transfer of halothane, enflurane, sevoflurane, and iso-flurane during cesarean section. J Anesth. 1995 Sep;9(3):220–3. doi: 10.1007/BF02479867. [DOI] [PubMed] [Google Scholar]
- 2.Cooper J, Jauniaux E, Gulbis B, Quick D, Bromley L. Placental transfer of fentanyl in early human pregnancy and its detection in fetal brain. Br J Anaesth. 1999 Jun;82(6):929–31. doi: 10.1093/bja/82.6.929. [DOI] [PubMed] [Google Scholar]
- 3.Hu L, Pan J, Zhang S, Yu J, He K, Shu S, et al. Propofol in combination with remifentanil for cesarean section: placental transfer and effect on mothers and newborns at different induction to delivery intervals. Taiwan J Obstet Gynecol. 2017 Aug;56(4):521–6. doi: 10.1016/j.tjog.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 4.FDA. FDA Drug Safety Communication: FDA approves label changes for use of general anesthetic and sedation drugs in young children. 2017 [Google Scholar]
- 5.FDA. FDA Drug Safety Communication: FDA review results in new warnings about using general anesthetics and sedation drugs in young children and pregnant women. 2016 [Google Scholar]
- 6.Jevtovic-Todorovic V, Hartman RE, Izumi Y, Benshoff ND, Dikranian K, Zorumski CF, et al. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J Neurosci. 2003 Feb 1;23(3):876–82. doi: 10.1523/JNEUROSCI.23-03-00876.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kong F, Xu L, He D, Zhang X, Lu H. Effects of gestational isoflurane exposure on postnatal memory and learning in rats. Eur J Pharmacol. 2011 Nov 16;670(1):168–74. doi: 10.1016/j.ejphar.2011.08.050. [DOI] [PubMed] [Google Scholar]
- 8.Brambrink AM, Evers AS, Avidan MS, Farber NB, Smith DJ, Martin LD, et al. Ketamine-induced neuroapoptosis in the fetal and neonatal rhesus macaque brain. Anesthesiology. 2012 Feb;116(2):372–84. doi: 10.1097/ALN.0b013e318242b2cd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fang F, Xue Z, Cang J. Sevoflurane exposure in 7-day-old rats affects neurogenesis, neuro-degeneration and neurocognitive function. Neurosci Bull. 2012 Oct;28(5):499–508. doi: 10.1007/s12264-012-1260-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Creeley C, Dikranian K, Dissen G, Martin L, Olney J, Brambrink A. Propofol-induced apoptosis of neurones and oligodendrocytes in fetal and neonatal rhesus macaque brain. Br J Anaesth. 2013 Jun;110(Suppl 1):i29–38. doi: 10.1093/bja/aet173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Creeley CE, Dikranian KT, Dissen GA, Back SA, Olney JW, Brambrink AM. Isoflurane-induced apoptosis of neurons and oligodendrocytes in the fetal rhesus macaque brain. Anesthesiology. 2014 Mar;120(3):626–38. doi: 10.1097/ALN.0000000000000037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Y, Yin SW, Zhang N, Zhao P. High-concentration sevoflurane exposure in midgestation induces apoptosis of neural stem cells in rat offspring. Neural Regen Res. 2018;13(9):1575–84. doi: 10.4103/1673-5374.237121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu L, Wang S, Feng Y, Zhao W, Zuo W, Zhong L, et al. KIF17 mediates the learning and memory impairment in offspring induced by maternal exposure to propofol during middle pregnancy. Mol Med Rep. 2018;17(4):5428–34. doi: 10.3892/mmr.2018.8479. [DOI] [PubMed] [Google Scholar]
- 14.Wilder RT, Flick RP, Sprung J, Katusic SK, Barbaresi WJ, Mickelson C, et al. Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology. 2009 Apr;110(4):796–804. doi: 10.1097/01.anes.0000344728.34332.5d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.DiMaggio C, Sun LS, Li G. Early childhood exposure to anesthesia and risk of developmental and behavioral disorders in a sibling birth cohort. Anesth Analg. 2011 Nov;113(5):1143–51. doi: 10.1213/ANE.0b013e3182147f42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Flick RP, Katusic SK, Colligan RC, Wilder RT, Voigt RG, Olson MD, et al. Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics. 2011 Nov;128(5):e1053–61. doi: 10.1542/peds.2011-0351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang X, Xu Z, Miao CH. Current clinical evidence on the effect of general anesthesia on neurodevelopment in children: an updated systematic review with meta-regression. PLoS One. 2014;9(1):e85760. doi: 10.1371/journal.pone.0085760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Block RI, Magnotta VA, Bayman EO, Choi JY, Thomas JJ, Kimble KK. Are anesthesia and surgery during infancy associated with decreased white matter integrity and volume during childhood? Anesthesiology. 2017 Nov;127(5):788–99. doi: 10.1097/ALN.0000000000001808. [DOI] [PubMed] [Google Scholar]
- 19.Hu D, Flick RP, Zaccariello MJ, Colligan RC, Katusic SK, Schroeder DR, et al. Association between exposure of young children to procedures requiring general anesthesia and learning and behavioral outcomes in a populationbased birth cohort. Anesthesiology. 2017 Aug;127(2):227–40. doi: 10.1097/ALN.0000000000001735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Davidson AJ, Disma N, de Graaff JC, Withington DE, Dorris L, Bell G, et al. Neurodevel-opmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): an international multicentre, randomised controlled trial. Lancet. 2016 Jan 16;387(10015):239–50. doi: 10.1016/S0140-6736(15)00608-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun LS, Li G, Miller TL, Salorio C, Byrne MW, Bellinger DC, et al. Association between a single general anesthesia exposure before age 36 months and neurocognitive outcomes in later childhood. JAMA. 2016 Jun 7;315(21):2312–20. doi: 10.1001/jama.2016.6967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McCann ME, de Graaff JC, Dorris L, Disma N, Withington D, Bell G, et al. Neurodevelop-mental outcome at 5 years of age after general anaesthesia or awake-regional anaesthesia in infancy (GAS): an international, multicentre, randomised, controlled equivalence trial. Lancet. 2019 Feb 16;393(10172):664–77. doi: 10.1016/S0140-6736(18)32485-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Feng NM, Luo FQ, Wang SQ, Cao MM. Effects of non-obstetric surgery under ketamine anaesthesia in the middle stage of pregnancy on cognition in the offspring and underlying mechanisms. Br J Anaesth. 2019 Mar 1;122(3):e36. [Google Scholar]
- 24.Zou S, Wei ZZ, Yue Y, Zheng H, Jiang MQ, Wu A. Desflurane and surgery exposure during pregnancy decrease synaptic integrity and induce functional deficits in juvenile off-spring mice. Neurochem Res. 2020 Feb;45(2):418–27. doi: 10.1007/s11064-019-02932-z. [DOI] [PubMed] [Google Scholar]
- 25.Garcia-Oria M, Ali A, Reynolds JD, Clary E, Gandsas A, Cummings T, et al. Histologic evaluation of fetal brains following maternal pneumoperitoneum. Surg Endosc. 2001 Nov;15(11):1294–8. doi: 10.1007/s004640080040. [DOI] [PubMed] [Google Scholar]
- 26.Van der Veeken L, Van der Merwe J, Devroe S, Inversetti A, Galgano A, Bleeser T, et al. Maternal surgery during pregnancy has a transient adverse effect on the developing fetal rabbit brain. Am J Obstet Gynecol. 2019 Jul 20;221(4):e1–355. doi: 10.1016/j.ajog.2019.07.029. [DOI] [PubMed] [Google Scholar]
- 27.Shu Y, Zhou Z, Wan Y, Sanders RD, Li M, Pac-Soo CK, et al. Nociceptive stimuli enhance anesthetic-induced neuroapoptosis in the rat developing brain. Neurobiol Dis. 2012 Feb;45(2):743–50. doi: 10.1016/j.nbd.2011.10.021. [DOI] [PubMed] [Google Scholar]
- 28.Liu JR, Liu Q, Li J, Baek C, Han XH, Athira-man U, et al. Noxious stimulation attenuates ketamine-induced neuroapoptosis in the developing rat brain. Anesthesiology. 2012 Jul;117(1):64–71. doi: 10.1097/ALN.0b013e31825ae693. [DOI] [PubMed] [Google Scholar]
- 29.Olutoye OA, Cruz SM, Akinkuotu AC, Sheikh F, Zamora IJ, Yu L, et al. Fetal surgery decreases anesthesia-induced neuroapoptosis in the mid-gestational fetal ovine brain. Fetal Diagn Ther. 2019;46(2):111–8. doi: 10.1159/000491925. [DOI] [PubMed] [Google Scholar]
- 30.Balinskaite V, Bottle A, Sodhi V, Rivers A, Bennett PR, Brett SJ, et al. The risk of adverse pregnancy outcomes following nonobstetric surgery during pregnancy: estimates from a retrospective cohort study of 6.5 million pregnancies. Ann Surg. 2017 Aug;266(2):260–6. doi: 10.1097/SLA.0000000000001976. [DOI] [PubMed] [Google Scholar]
- 31.Devroe S, Bleeser T, Van de Velde M, Verbrugge L, De Buck F, Deprest J, et al. Anesthesia for non-obstetric surgery during pregnancy in a tertiary referral center: a 16-year retrospective, matched case-control, cohort study. Int J Obstet Anesth. 2019 Aug;39:74–81. doi: 10.1016/j.ijoa.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 32.O’Mahony S. Endoscopy in pregnancy. Best Pract Res Clin Gastroenterol. 2007;21(5):893–9. doi: 10.1016/j.bpg.2007.05.007. [DOI] [PubMed] [Google Scholar]
- 33.Blitz MJ, Grünebaum A, Tekbali A, Bornstein E, Rochelson B, Nimaroff M, et al. Intensive care unit admissions for pregnant and nonpregnant women with coronavirus disease 2019. Am J Obstet Gynecol. 2020 Aug;223(2):290–1. doi: 10.1016/j.ajog.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.De Buck F, Deprest J, Van de Velde M. Anesthesia for fetal surgery. Curr Opin Anaesthesiol. 2008 Jun;21(3):293–7. doi: 10.1097/ACO.0b013e3282fe6e70. [DOI] [PubMed] [Google Scholar]
- 35.Adzick NS, Thom EA, Spong CY, Brock JW, 3rd, Burrows PK, Johnson MP, et al. A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med. 2011 Mar 17;364(11):993–1004. doi: 10.1056/NEJMoa1014379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Clancy B, Finlay BL, Darlington RB, Anand KJ. Extrapolating brain development from experimental species to humans. Neurotoxicology. 2007 Sep;28(5):931–7. doi: 10.1016/j.neuro.2007.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Workman AD, Charvet CJ, Clancy B, Darlington RB, Finlay BL. Modeling transformations of neurodevelopmental sequences across mammalian species. J Neurosci. 2013 Apr 24;33(17):7368–83. doi: 10.1523/JNEUROSCI.5746-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bleeser T, Van Der Veeken L, Devroe S, Deprest J, Rex S. Arterial blood pressure and arterial blood gas values in awake pregnant rabbits. Vet Anaesth Analg. 2020 Nov;47(6):849–51. doi: 10.1016/j.vaa.2020.06.006. [DOI] [PubMed] [Google Scholar]
- 39.van der Merwe J, van der Veeken L, Ferraris S, Gsell W, Himmelreich U, Toelen J, et al. Early neuropathological and neurobehavioral consequences of preterm birth in a rabbit model. Sci Rep. 2019 Mar 5;9(1):3506. doi: 10.1038/s41598-019-39922-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Derrick M, Luo NL, Bregman JC, Jilling T, Ji X, Fisher K, et al. Preterm fetal hypoxia-ischemia causes hypertonia and motor deficits in the neonatal rabbit: a model for human cerebral palsy? J Neurosci. 2004 Jan 7;24(1):24–34. doi: 10.1523/JNEUROSCI.2816-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Eixarch E, Batalle D, Illa M, Muñoz-Moreno E, Arbat-Plana A, Amat-Roldan I, et al. Neonatal neurobehavior and diffusion MRI changes in brain reorganization due to intrauterine growth restriction in a rabbit model. PLoS One. 2012;7(2):e31497. doi: 10.1371/journal.pone.0031497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jaeger D, Gilman S, Aldridge JW. Primate basal ganglia activity in a precued reaching task: preparation for movement. Exp Brain Res. 1993;95(1):51–64. doi: 10.1007/BF00229653. [DOI] [PubMed] [Google Scholar]
- 43.Tulving E, Markowitsch HJ. Episodic and declarative memory: role of the hippocampus. Hippocampus. 1998;8(3):198–204. doi: 10.1002/(SICI)1098-1063(1998)8:3<198::AID-HIPO2>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 44.Unterrainer JM, Owen AM. Planning and problem solving: from neuropsychology to functional neuroimaging. J Physiol Paris. 2006 Jun;99(4–6):308–17. doi: 10.1016/j.jphysparis.2006.03.014. [DOI] [PubMed] [Google Scholar]
- 45.Wolf HK, Buslei R, Schmidt-Kastner R, Schmidt-Kastner PK, Pietsch T, Wiestler OD, et al. NeuN: a useful neuronal marker for diagnostic histopathology. J Histochem Cytochem. 1996 Oct;44(10):1167–71. doi: 10.1177/44.10.8813082. [DOI] [PubMed] [Google Scholar]
- 46.Rizzi S, Carter LB, Ori C, Jevtovic-Todorovic V. Clinical anesthesia causes permanent damage to the fetal guinea pig brain. Brain Pathol. 2008 Apr;18(2):198–210. doi: 10.1111/j.1750-3639.2007.00116.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xiong M, Li J, Alhashem HM, Tilak V, Patel A, Pisklakov S, et al. Propofol exposure in pregnant rats induces neurotoxicity and persistent learning deficit in the offspring. Brain Sci. 2014 May 6;4(2):356–75. doi: 10.3390/brainsci4020356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhao T, Li Y, Wei W, Savage S, Zhou L, Ma D. Ketamine administered to pregnant rats in the second trimester causes long-lasting behavioral disorders in offspring. Neurobiol Dis. 2014;68:145–55. doi: 10.1016/j.nbd.2014.02.009. [DOI] [PubMed] [Google Scholar]
- 49.Campos-Pires R, Hirnet T, Valeo F, Ong BE, Radyushkin K, Aldhoun J, et al. Xenon improves long-term cognitive function, reduces neuronal loss and chronic neuroinflammation, and improves survival after traumatic brain injury in mice. Br J Anaesth. 2019 Jul;123(1):60–73. doi: 10.1016/j.bja.2019.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang X, Zhao J, Chang T, Wang Q, Liu W, Gao L. Ketamine exerts neurotoxic effects on the offspring of pregnant rats via the Wnt/β-catenin pathway. Environ Sci Pollut Res. 2020 Jan;27(1):305–14. doi: 10.1007/s11356-019-06753-z. [DOI] [PubMed] [Google Scholar]
- 51.Calhoun ME, Jucker M, Martin LJ, Thinakaran G, Price DL, Mouton PR. Comparative evaluation of synaptophysin-based methods for quantification of synapses. J Neurocytol. 1996 Dec;25(12):821–8. doi: 10.1007/BF02284844. [DOI] [PubMed] [Google Scholar]
- 52.Falini B, Flenghi L, Fagioli M, Stein H, Schwarting R, Riccardi C, et al. Evolutionary conservation in various mammalian species of the human proliferation-associated epitope recognized by the Ki-67 monoclonal antibody. J Histochem Cytochem. 1989 Oct;37(10):1471–8. doi: 10.1177/37.10.2476477. [DOI] [PubMed] [Google Scholar]
- 53.Dong C, Rovnaghi CR, Anand KJ. Ketamine alters the neurogenesis of rat cortical neural stem progenitor cells. Crit Care Med. 2012 Aug;40(8):2407–16. doi: 10.1097/CCM.0b013e318253563c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang Z, Jyoti A, Balakrishnan B, Williams M, Singh S, Chugani DC, et al. Trajectory of inflammatory and microglial activation markers in the postnatal rabbit brain following intrauterine endotoxin exposure. Neurobiol Dis. 2018 Mar;111:153–62. doi: 10.1016/j.nbd.2017.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: open source software for digital pathology image analysis. Sci Rep. 2017 Dec 4;7(1):16878. doi: 10.1038/s41598-017-17204-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012 Jun 28;9(7):676–82. doi: 10.1038/nmeth.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sally G, James AD, Bryce V. A study of clustered data and approaches to its analysis. J Neurosci. 2010 Aug 11;30(32):10601–8. doi: 10.1523/JNEUROSCI.0362-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Aarts E, Verhage M, Veenvliet JV, Dolan CV, van der Sluis S. A solution to dependency: using multilevel analysis to accommodate nested data. Nat Neurosci. 2014 Mar 26;17(4):491–6. doi: 10.1038/nn.3648. [DOI] [PubMed] [Google Scholar]
- 59.Aarts E, Dolan CV, Verhage M, van der Sluis S. Multilevel analysis quantifies variation in the experimental effect while optimizing power and preventing false positives. BMC Neurosci. 2015 Dec 19;16(1):94–15. doi: 10.1186/s12868-015-0228-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Boisgontier MP, Cheval B. The anova to mixed model transition. Neurosci Biobehav Rev. 2016 Sep;68:1004–5. doi: 10.1016/j.neubiorev.2016.05.034. [DOI] [PubMed] [Google Scholar]
- 61.Harrison XA, Donaldson L, Correa-Cano ME, Evans J, Fisher DN, Goodwin CED, et al. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ. 2018;6:e4794. doi: 10.7717/peerj.4794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barnard JM, Chaffin D, Droste S, Tierney A, Phernetton T. Fetal response to carbon dioxide pneumoperitoneum in the pregnant ewe. Obstet Gynecol. 1995 May;85(5 Pt 1):669–74. doi: 10.1016/0029-7844(95)00023-k. [DOI] [PubMed] [Google Scholar]
- 63.Curet MJ, Vogt DA, Schob O, Qualls C, Izquierdo LA, Zucker KA. Effects of CO2 pneumo-peritoneum in pregnant ewes. J Surg Res. 1996 Jun;63(1):339–44. doi: 10.1006/jsre.1996.0272. [DOI] [PubMed] [Google Scholar]
- 64.Chung JC, Cho GS, Shin EJ, Kim HC, Song OP. Clinical outcomes compared between laparoscopic and open appendectomy in pregnant women. Can J Surg. 2013 Oct;56(5):341–6. doi: 10.1503/cjs.022112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Van der Veeken L, Grönlund S, Gerdtsson E, Holmqvist B, Deprest J, Ley D, et al. Long-term neurological effects of neonatal caffeine treatment in a rabbit model of preterm birth. Pediatr Res. 2020 May;87(6):1011–18. doi: 10.1038/s41390-019-0718-8. [DOI] [PubMed] [Google Scholar]
- 66.Eixarch E, Figueras F, Hernández-Andrade E, Crispi F, Nadal A, Torre I, et al. An experimental model of fetal growth restriction based on selective ligature of uteroplacental vessels in the pregnant rabbit. Fetal Diagn Ther. 2009;26(4):203. doi: 10.1159/000264063. [DOI] [PubMed] [Google Scholar]
- 67.Eixarch E, Hernandez-Andrade E, Crispi F, Illa M, Torre I, Figueras F, et al. Impact on fetal mortality and cardiovascular Doppler of selective ligature of uteroplacental vessels compared with undernutrition in a rabbit model of intrauterine growth restriction. Placenta. 2011 Apr;32(4):304. doi: 10.1016/j.placenta.2011.01.014. [DOI] [PubMed] [Google Scholar]
- 68.Grande C, Sancho MA, Conill J, Julià V, Albert A, Martínez A, et al. Creation of a model for myelomeningocele in rabbit embryos. Cir Pediatr. 2002 Jul;15(3):101. [PubMed] [Google Scholar]
- 69.Joyeux L, Engels AC, Van Der Merwe J, Aertsen M, Patel PA, Deprez M, et al. Validation of the fetal lamb model of spina bifida. Sci Rep. 2019 Jun 27;9(1):9327. doi: 10.1038/s41598-019-45819-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zheng H, Dong Y, Xu Z, Crosby G, Culley DJ, Zhang Y, et al. Sevoflurane anesthesia in pregnant mice induces neurotoxicity in fetal and offspring mice. Anesthesiology. 2013 Mar;118(3):516–26. doi: 10.1097/ALN.0b013e3182834d5d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Hirotsu A, Iwata Y, Tatsumi K, Miyai Y, Matsuyama T, Tanaka T. Maternal exposure to volatile anesthetics induces IL-6 in fetal brains and affects neuronal development. Eur J Pharmacol. 2019 Nov 15;863:172682. doi: 10.1016/j.ejphar.2019.172682. [DOI] [PubMed] [Google Scholar]
- 72.Yu X, Ma F, Cao X, Ma X, Hu C. Effects of the application of general anesthesia with propofol during the early stage of pregnancy on brain development and function of SD rat offspring and the intervention of DHA. Neurol Res. 2019 Nov;41(11):1008–14. doi: 10.1080/01616412.2019.1672381. [DOI] [PubMed] [Google Scholar]
- 73.Chen X, Wang Y, Luo H, Luo Z, Liu L, Xu W, et al. Ulinastatin reduces urinary sepsis-related inflammation by upregulating IL-10 and downregulating TNF-α levels. Mol Med Rep. 2013 Jul;8(1):29–34. doi: 10.3892/mmr.2013.1480. [DOI] [PubMed] [Google Scholar]
- 74.Liu SJ, Shi Y, Liu C, Zhang M, Zuo ZC, Zeng CJ, et al. The upregulation of pro-inflammatory cytokines in the rabbit uterus under the lipopolysaccaride-induced reversible immunoresponse state. Anim Reprod Sci. 2017 Jan;176:70–7. doi: 10.1016/j.anireprosci.2016.11.012. [DOI] [PubMed] [Google Scholar]
- 75.Dasso JF, Obiakor H, Bach H, Anderson AO, Mage RG. A morphological and immunohistological study of the human and rabbit appendix for comparison with the avian bursa. Dev Comp Immunol. 2000 Dec;24(8):797–814. doi: 10.1016/s0145-305x(00)00033-1. [DOI] [PubMed] [Google Scholar]
- 76.Malla BK. A study on “vermiform appendix”: a caecal appendage in common laboratory mammals. Kathmandu Univ Med J. 2003 Oct–Dec;1(4):272–5. [PubMed] [Google Scholar]
- 77.Chinn G, Pearn M, Vutskits L, Mintz C, Loepke A, Lee J, et al. Standards for preclinical research and publications in developmental anaesthetic neurotoxicity: expert opinion statement from the SmartTots preclinical working group. Br J Anaesth. 2020 May;124(5):585–93. doi: 10.1016/j.bja.2020.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Harel S, Watanabe K, Linke I, Schain RJ. Growth and development of the rabbit brain. Biol Neonate. 1972;21(5):381–99. doi: 10.1159/000240527. [DOI] [PubMed] [Google Scholar]
- 79.Dobbing J, Sands J. Vulnerability of developing brain. IX. The effect of nutritional growth retardation on the timing of the brain growth-spurt. Biol Neonate. 1971;19(4):363–78. doi: 10.1159/000240430. [DOI] [PubMed] [Google Scholar]
- 80.Dobbing J, Sands J. Comparative aspects of the brain growth spurt. Early Hum Dev. 1979 Mar;3(1):79–83. doi: 10.1016/0378-3782(79)90022-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.