Abstract
Background
Treatment for major depressive disorder (MDD) is imprecise and often involves trial-and-error to determine the most effective approach. To facilitate optimal treatment selection and inform timely adjustment, the current study investigated whether neurocognitive variables could predict an antidepressant response in a treatment-specific manner.
Methods
In the two-stage Establishing Moderators and Biosignatures of Antidepressant Response for Clinical Care (EMBARC) trial, outpatients with non-psychotic recurrent MDD were first randomized to an 8-week course of sertraline selective serotonin reuptake inhibitor or placebo. Behavioral measures of reward responsiveness, cognitive control, verbal fluency, psychomotor, and cognitive processing speeds were collected at baseline and week 1. Treatment responders then continued on another 8-week course of the same medication, whereas non-responders to sertraline or placebo were crossed-over under double-blinded conditions to bupropion noradrenaline/dopamine reuptake inhibitor or sertraline, respectively. Hamilton Rating for Depression scores were also assessed at baseline, weeks 8, and 16.
Results
Greater improvements in psychomotor and cognitive processing speeds within the first week, as well as better pretreatment performance in these domains, were specifically associated with higher likelihood of response to placebo. Moreover, better reward responsiveness, poorer cognitive control and greater verbal fluency were associated with greater likelihood of response to bupropion in patients who previously failed to respond to sertraline.
Conclusion
These exploratory results warrant further scrutiny, but demonstrate that quick and non-invasive behavioral tests may have substantial clinical value in predicting antidepressant treatment response.
Keywords: Biomarkers, bupropion, cognitive control, depression, placebo response, reward learning, sertraline, treatment response
Introduction
Major depressive disorder (MDD) is a serious public health problem that affects more than 240 million people worldwide (James et al., 2018). Being prevalent, debilitating and recurrent, it is associated with significant personal, societal and economic costs (Greenberg, Fournier, Sisitsky, Pike, & Kessler, 2015; Kessler et al., 2003). Unfortunately, the treatment of MDD continues to be challenging as clinicians typically rely on trial-and-error to find the most effective approach. In the STAR*D study, which provided every patient with up to four open-label treatment steps each 12 weeks in length, it was found that only ~50% of MDD patients benefited (i.e. responded by showing ⩾50% improvement in symptoms) from the selective serotonin reuptake inhibitor (SSRI) citalopram (Trivedi et al., 2006), and over one-third were resistant to two or more antidepressants (Rush et al., 2006; Souery, Papakostas, & Trivedi, 2006). Within primary care, the response rate to first-line antidepressants is even lower at ~30% (Katon et al., 1996). To worsen these issues, it takes at least 4 weeks to assess whether a particular antidepressant is working. This can result in unnecessarily long trials that can heighten the risk of suicidal behavior, treatment discontinuation and patient morbidity. Identifying variables that can predict response to different antidepressants would help clinicians to decide, as early as possible, whether a particular treatment might be suitable for the patient.
Emerging research suggests that quick and non-invasive behavioral tests, which index specific neurocognitive impairments in MDD, may be predictors or moderators of antidepressant response. Executive function, psychomotor speed and/or memory tests have been found to predict outcome to treatment by fluoxetine for 4 weeks (Gudayol-Ferré et al., 2010), 8 weeks (Dunkin et al., 2000) and 12 weeks (Taylor et al., 2006); escitalopram for 8 weeks (Etkin et al., 2015) and 12 weeks (Alexopoulos et al., 2015); citalopram for 6 weeks (Kalayam & Alexopoulos, 2003) and 8 weeks (Sneed et al., 2007); bupropion for 8 weeks (Herrera-Guzmán et al., 2008) and 8–12 weeks (Bruder et al. 2014); duloxetine for 6 weeks (Mikoteit et al., 2015); agomelatine for 6–8 weeks (Cléry-Melin & Gorwood, 2017); as well as ketamine after 24 h (Murrough et al., 2014, 2015) and 12 days (Shiroma, Albott, et al., 2014). However, some investigators found no evidence of an association between cognitive performance and response/remission to 8 weeks of sertraline (Etkin et al., 2015), venlafaxine (Etkin et al., 2015), and escitalopram (Alexopoulos et al., 2007), as well as 12 weeks of fluoxetine (Gudayol-Ferré et al., 2012). Although the cause of these discrepancies is unclear, they likely stem partly from differences in specific tasks used (Groves, Douglas, & Porter, 2018). All these studies, however, have focused on predicting response to a single antidepressant. Depressed patients who fail to benefit from an adequate trial of SSRI are often switched to a non-SSRI agent (Fredman et al., 2000). Yet, it remains unknown whether pretreatment cognitive performance could differentiate between responders to a second antidepressant, which is administered immediately after nonresponse to a pharmacologically distinct class of medication, and non-responders resistant to both arms of treatment.
More recently, several reports have suggested that early improvements in cognitive performance may be associated with antidepressant treatment response. However, they mostly focused on ‘hot’ cognition, which is related to the processing of emotional information. Specifically, greater improvements in early emotional recognition and processing were found to predict treatment outcome with citalopram (Shiroma, Thuras, Johns, & Lim, 2014; Tranter et al., 2009), escitalopram (Godlewska, Browning, Norbury, Cowen, & Harmer, 2016), and reboxetine (Tranter et al., 2009). Surprisingly, previous studies investigating changes in ‘cold’, non-emotional cognitive variables in MDD have mostly compared performance before and after treatment (Beblo, Baumann, Bogerts, Wallesch, & Herrmann, 1999; Hammar et al., 2009; Herrera-Guzmán et al., 2010; Hinkelmann et al., 2012; Reppermund et al., 2007; Reppermund, Ising, Lucae, & Zihl, 2009). Only one study reported that improvements in cognitive speed, psychomotor function, motivation, and sensory perception from baseline to week 2 were predictive of treatment response to agomelatine after 6 weeks – although these were based on a self-report questionnaire rather than objective behavioral tasks (Gorwood et al., 2015). Thus, the utility of early changes in ‘cold’ cognition as predictors of antidepressant response is still not well understood.
The current study sought to explore the two aforementioned gaps in the literature by using data from the two-staged Establishing Moderators and Biosignatures of Antidepressant Response for Clinical Care (EMBARC) trial (Trivedi et al., 2016). Task-based measures of reward processing, cognitive control, verbal fluency, psychomotor, and cognitive processing speed were collected at baseline and 1 week after the onset of an 8-week clinical trial, where outpatients with recurrent and non-psychotic MDD were randomized to receive the SSRI sertraline or placebo (stage 1). Our first goal was to examine whether changes in any behavioral tests within the first week might differentially predict eventual response to antidepressant treatment. Participants who achieved satisfactory response at the end of stage 1 continued on another 8-week course of the same medication, whereas nonresponders were crossed-over under double-blinded conditions. Accordingly, sertraline non-responders received bupropion, and placebo non-responders took sertraline in stage 2. This allowed us to pursue our second goal: to identify putative pre-treatment cognitive variables that might distinguish patients who benefit from a non-serotonergic antidepressant (bupropion), after failure to respond to an SSRI (sertraline), from non-responders who are resistant to both classes of medication.
Methods
Participants
Outpatients and healthy volunteers were recruited at four sites in the United States (Columbia University, New York; Massachusetts General Hospital, Boston; University of Texas Southwestern Medical Center, Dallas; and University of Michigan, Ann Arbor) after approval by the institutional review board of each site. All enrolled participants provided written informed consent and were aged between 18 and 65 years. Patients also met the criteria for MDD based on the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID), scored ⩾14 on the Quick Inventory of Depressive Symptomatology Self-Report (Rush et al., 2003) at both screening and randomization visits, and were free of antidepressant medication for >3 weeks prior to completing any study measures. Exclusion criteria included: history of bipolar disorder or psychosis, substance dependence (except for nicotine) in the past 6 months or substance abuse in past 2 months, active suicidality, or unstable medical conditions. In total, 634 patients were assessed for eligibility; 338 were excluded, leaving 296 individuals who were randomized in stage 1. Forty healthy controls were also enrolled. Data from participants who passed quality control criteria for at least one of the cognitive tasks at baseline and completed at least 4 weeks of treatment in stage 1 were included here.
Clinical measure of depression
17-item Hamilton Rating Scale for Depression (HAMD17) (Hamilton, 1960)
This is a clinician-administered scale used to assess severity of symptoms of depression experienced over the past week. The HAMD17 was administered at each study visit for baseline, stage 1 (weeks 1, 2, 3, 4, 6, and 8), and stage 2 (weeks 9, 10, 12, and 16). Patients were defined as responders if they completed at least 4 weeks of treatment and showed a decrease in HAMD17 score of ⩾50% at the last observation compared to when the treatment started.
Neurocognitive measures
Probabilistic reward task (PRT)
This is a signal detection test that differentially rewarded correct responses to two difficult-to-discriminate stimuli in a 3:1 ratio, in order to assess the extent to which participants modulated their behavior as a function of reward (Pizzagalli, Jahn, & O’Shea, 2005). Performance was analyzed in terms of response bias, which is an objective measure of reward responsiveness (i.e. the tendency to choose the more rewarded stimulus). Details can be found in online Supplementary methods.
Eriksen flanker task (EFT)
On every trial, participants had to indicate, via a button press, whether an arrow in the center of the screen was pointing to the left or right. Crucially, this central arrow was presented with adjacent arrows that either pointed in the same direction (i.e. congruent condition) or opposite direction (i.e. incongruent condition) (Eriksen, 1995). Inhibitory control was indexed by the interference metric (RTincongruent trials – RTcongruent trials). Details can be found in online Supplementary methods.
Choice reaction time task (CRT)
One of four possible stimuli was presented on each trial and participants had to press the button that corresponded to that stimulus as quickly as they could (Thorne, Genser, Sing, & Hegge, 1985). There were 60 trials in total. Psychomotor processing speed was assessed by the median reaction time of correct trials, which is demographically-adjusted and z-scored to account for known age, gender, and education effects on scores.
A-not-B reasoning test (ABRT)
Participants were required to determine the accuracy of a statement describing the order of a pair of letters (‘AB’ and ‘BA’). The statements could be: (i) _ comes before _, (ii) _ comes after _, (iii) _ does not come before _, and (iv) _ does not come after _, in all permutations of A and B in the blanks (Baddeley, 1968). There were 32 trials in total. Cognitive processing speed was assessed by the median reaction time of correct trials, which is demographically-adjusted and z-scored to account for known age, gender, and education effects on scores.
Verbal fluency test (VFT)
Participants had to produce words beginning with a specific letter within a time limit of 1 min (Benton, Hamsher, & Sivan, 1983). Three different letters (‘F’, ‘A’, and ‘S’) were used and fluency was indexed by the total number of words produced across all three letters, which is demographically-adjusted and z-scored to account for known age, gender, and education effects on scores.
Statistical analysis
Aim 1
For each task, we selected subjects who passed pre-determined quality control criteria at baseline and week 1. Separate logistic regressions were used to evaluate whether early changes from baseline in CRT, ABRT, and VFT – whose outcomes were converted to demographically-adjusted z-scores and are those used in the EMBARC study and prior studies (Gorlyn et al., 2008; Keilp, Sackeim, & Mann, 2005) – were associated with a difference in likelihood of response to sertraline v. placebo. The outcome variable was Responder (yes, no), and covariates were Treatment (sertraline, placebo), baseline score, change in score from baseline to week 1, interaction between Treatment and baseline score, interaction between Treatment and change score, and Site (Columbia, Massachusetts, Texas, Michigan). Because CRT, ABRT, and VFT analyses used demographically-adjusted z-scores, we entered age, gender, and education as additional covariates in logistic regressions for the PRT and EFT to harmonize analyses across tasks. For tasks in which early changes in performance differentially predicted response to placebo v. sertraline, additional sets of analyses were conducted. First, we broke the full logistic regression into two simpler analyses that included Treatment, either baseline score or change score as well as its interaction with Treatment, and Site. Second, analysis of covariances (ANCOVAs) were used to examine how placebo responders and non-responders compared to healthy controls. The outcome variable was change score from baseline to week 1, factor was Group (responders, non-responders, and controls) and covariates were Site and baseline score.
Aim 2
For each task, we selected subjects who passed the quality control criteria at baseline, were non-responders to sertraline or placebo in stage 1 and completed at least 4 weeks of stage 2 treatment with bupropion (after switching from sertraline) or sertraline (after switching from placebo). Separate logistic regressions were utilized to evaluate whether baseline performance in CRT, ABRT, and VFT was associated with a difference in likelihood of response to bupropion v. sertraline. The outcome variable was Responder (yes, no), and covariates were Treatment (bupropion, sertraline), baseline score, interaction between Treatment and baseline score, and Site (Columbia, Massachusetts, Texas, Michigan). To harmonize analyses across tasks, we used similar logistic regressions for PRT and EFT but with additional covariates of age, gender, and education. For tasks in which baseline performance differentially predicted response to bupropion v. sertraline, ANCOVAs were conducted to compare bupropion responders and non-responders with healthy controls. The outcome variable was pretreatment task score, factor was Group (responders, non-responders, controls) and covariates were Site, age, gender, and education. Independent samples’ t test also assessed whether responders and non-responders to bupropion and sertraline differed in baseline HAMD, week 8 HAMD, and change in HAMD from baseline to week 8.
The logistic regression analyses were not corrected for multiple comparisons as the tasks were carefully selected based on prior findings suggesting their potential for predicting response for antidepressants (Gorlyn et al., 2008; Vrieze et al., 2013) and we wanted to examine the value of each test as a predictor.
Results
Early changes in psychomotor and cognitive processing speeds were associated with better response to placebo
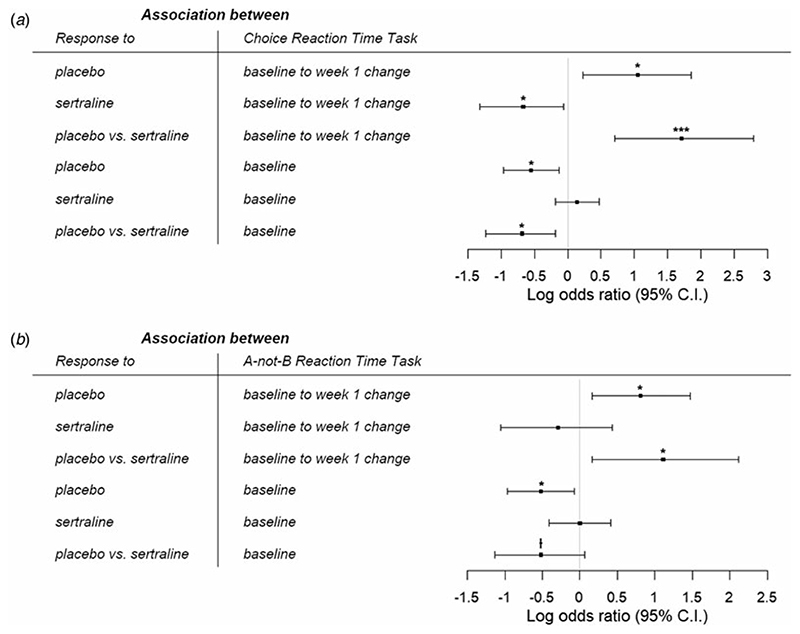
For CRT [sertraline: N = 113, age = 37.1 (13.8) years; placebo: N = 125, age = 38.0 (12.8) years], the full logistic regression revealed that greater improvement in reaction time from baseline to week 1 was associated with increased likelihood of response to placebo [B = 1.05, 95% confidence interval (CI) = 0.23–1.86, p = 0.012], but lower probability of sertraline response (B = −0.67, 95% CI = −1.32 to -0.06, p = 0.037). Importantly, these relationships were significantly different (B = 1.71, 95% CI = 0.71–2.79, p = 0.001), suggesting that early changes in CRT differentially predicted response to placebo and sertraline (Fig. 1a). There was also a significant difference in associations between baseline reaction time and likelihood of response to placebo v. sertraline (B = −0.69, 95% CI = −1.23 to −0.18, p = 0.010). Slower baseline reaction time was related to reduced odds of placebo response (B = −0.55, 95% CI = −0.97 to -0.13, p = 0.011), but not associated with probability of response to sertraline (B = 0.14, 95% CI = −0.18 to 0.47, p = 0.392). See online Supplementary Table S1 for details.
Fig. 1.
Log odds ratio for the associations between likelihood of response to placebo and sertraline with (a) choice reaction time task and (b) A-not-B reaction time task. Greater improvements in psychomotor and cognitive processing speeds within the first week, as well as better pretreatment performance, were specifically associated with higher likelihood of response to placebo. ⍿p <0.10, *p <0.05, **p <0.01, ***p <0.001.
In two simpler logistic regressions that separately examined the effects of baseline and change scores, we found that early changes in CRT within the first week still differentially predicted outcome to placebo and sertraline (B = 1.03, 95% CI = 0.19–1.91, p = 0.018, online Supplementary Table S2). However, there was no longer a difference in relationships between baseline CRT and response to placebo v. sertraline (B = −0.25, 95% CI = −0.69 to 0.19, p = 0.270, online Supplementary Table S3).
An ANCOVA comparing early change in CRT performance among placebo responders, non-responders, and healthy volunteers revealed a significant effect of group (F(2,157) = 4.94, p = 0.008, partial η2 = 0.059). Post-hoc tests found that placebo responders did not differ from controls (t(84) = 0.272, p = 0.79, Cohen’s d = 0.05) whereas non-responders had less improvement from baseline to week 1 than healthy individuals (t(117) = −2.38, p = 0.055, Cohen’s d = 0.46) and responders (t(124) = −2.77, p = 0.019, Cohen’s d =0.51).
For ABRT [sertraline: N = 102, age = 36.8 (13.4) years; placebo: N = 114, age = 37.7 (12.7) years], we similarly found in the full logistic regression that greater improvement in reaction time from baseline to week 1 was related to higher likelihood of response to placebo (B = 0.81, 95% CI = 0.16–1.47, p = 0.015). However, change in ABRT within the first week was not associated with the odds of sertraline response (B = −0.29, 95% CI = −1.05 to 0.43, p = 0.429); and crucially, these relationships were significantly different (B = 1.11, 95% CI = 0.16–2.12, p = 0.027), suggesting that early change in ABRT differentially predicted response to placebo and sertraline. We also found a trending difference in associations between baseline reaction time and probability of response to sertraline v. placebo (B = −0.52, 95% CI = −1.13 to 0.07, p = 0.088). Slower baseline ABRT led to reduced likelihood of response to placebo (B = −0.52, 95% CI = −0.97 to -0.07, p = 0.025), but was not related to sertraline response (B = −0.002, 95% CI = −0.41 to 0.41, p = 0.994) (Fig. 1b). See online Supplementary Table S4 for details.
Simpler logistic regressions investigating the baseline and change scores separately revealed that early changes in ABRT within the first week still differentially predicted outcome to placebo and sertraline, albeit at a trend level (B = 0.74, 95% CI = 0.018–1.52, p = 0.052, online Supplementary Table S5). In contrast, the difference in relationships between baseline ABRT and response to placebo v. sertraline was no longer significant (B = −0.14, 95% CI = −0.60 to 0.32, p = 0.553, online Supplementary Table S6).
A significant effect of group was found when comparing early change in ABRT performance between placebo responders, nonresponders, and healthy individuals (F(2,144) = 3.32, p = 0.039, partial η2 = 0.044). Post-hoc tests revealed that placebo responders had greater improvement from baseline to week 1 than non-responders (t(113) = 2.49, p = 0.043, Cohen’s d = 0.48), but there was no difference between controls v. responders (t(80) = 1.93, p = 0.168, Cohen’s d = 0.42) and controls v. non-responders (t(106) = 0.30, p = 0.76, Cohen’s d = 0.06).
In contrast, neither baseline nor early change in performance for the VFT, PRT, and EFT differentially predicted response to placebo v. sertraline. Details of all these analyses can be found in online Supplementary Tables S7–S9. At the request of an anonymous reviewer, we also repeated all the analyses by adding an additional covariate of smoking status and found that conclusions from all p value significance tests remained the same.
Pretreatment reward responsiveness, cognitive control, and verbal fluency are associated with bupropion response
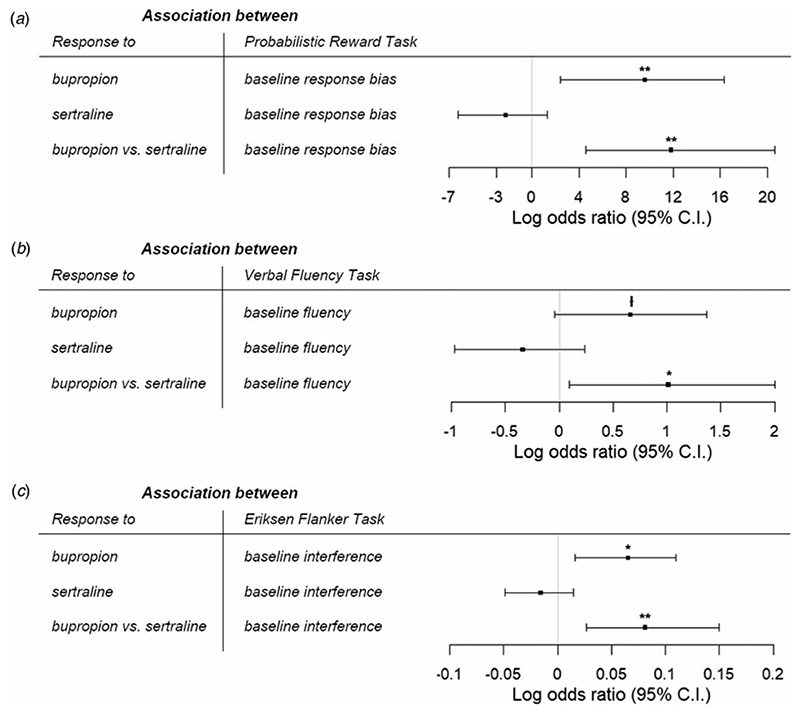
We found that greater pretreatment response bias was associated with higher likelihood of response to bupropion [after switching from sertraline; N = 38, age = 38.4 (14.7) years] (B = 9.59, 95% CI = 2.46–16.3, p = 0.008). However, there was no relationship between response bias and probability of response to sertraline [after previous non-response to placebo; N = 49, age = 41.1 (13.1) years] (B = −2.20, 95% CI = −6.27 to 1.35, p = 0.249). Critically, these associations were significantly different from each other (B = 11.8, 95% CI = 4.60–20.6, p = 0.003), suggesting that baseline response bias differentially predicted response to bupropion and sertraline (Fig. 2a). An ANCOVA comparing baseline response bias among bupropion responders, non-responders, and healthy volunteers revealed a significant effect of group (F(2,67) = 6.99, p = 0.002, partial η2 = 0.173). Post-hoc tests found no difference between responders and controls (t(53) = 0.585, p = 0.56, Cohen’s d = 0.16), whereas non-responders had significantly lower response bias than healthy people (t(59) = 3.27, p = 0.005, Cohen’s d = 0.86) and responders to bupropion (t(37) = 3.22, p = 0.006, Cohen’s d = 1.00).
Fig. 2.
Log odds ratio for the associations between likelihood of response to bupropion and sertraline with (a) probabilistic reward task, (b) verbal fluency task, and (c) Eriksen flanker task. Better response bias, greater verbal fluency, and higher response interference were specifically associated with greater likelihood of response to bupropion in patients who previously failed to respond to sertraline. ⍿p < 0.10, *p < 0.05, **p < 0.01, ***p < 0.001.
Results for the VFT [bupropion: N = 42, age = 38.0 (14.4) years; sertraline: N = 52, age = 40.6 (13.4) years] were similar to the PRT. There was a significant difference in associations between baseline verbal fluency and likelihood of response to bupropion v. sertraline (B = 1.01, 95% CI = 0.097–2.00, p = 0.035). Specifically, greater verbal fluency was related to higher probability of bupropion response at a trend level (B = 0.66, 95% CI = −0.046 to 1.37, p = 0.067), but not associated with odds of response to sertraline (B = −0.34, 95% CI = −0.97 to 0.24, p = 0.259) (Fig. 2b). There was a significant effect of group (F(2,76) = 6.20, p = 0.003, partial η2 = 0.140) when comparing baseline performance among bupropion responders, non-responders, and healthy volunteers. Specifically, bupropion responders and controls did not differ in verbal fluency (t(56) = 0.505, p = 0.62, Cohen’s d = 0.15), but non-responders performed worse than healthy individuals (t(64) = −3.45, p = 0.003, Cohen’s d = 0.88) and responders (t(41) = −2.29, p = 0.074, Cohen’s d = 0.71).
For the EFT [bupropion: N = 36, age = 37.4 (13.5) years; sertraline: N = 50, age = 40.4 (13.5) years], we also found a significant difference in the relationships between baseline interference and odds of response to bupropion v. sertraline (B = 0.081, 95% CI = 0.027–0.15, p = 0.007). Greater baseline interference (i.e. poorer cognitive control) was surprisingly associated with increased likelihood of bupropion response (B = 0.065, 95% CI = 0.016–0.11, p = 0.010), whereas there was no relationship between pretreatment interference and probability of response to sertraline (B = −0.016, 95% CI = −0.049 to 0.015, p = 0.321) (Fig. 2c). There was a trending effect of group (F(2,64) = 2.73, p = 0.073, partial η2 = 0.079) when comparing pretreatment performance between bupropion responders, non-responders, and controls. Healthy individuals did not differ from responders (t(50) = −2.05, p = 0.134, Cohen’s d = 0.65) or non-responders (t(58) = 0.38, p = 0.71, Cohen’s d = 0.10), but responders had greater interference than non-responders at a trend level (t(35) = 2.22, p = 0.089, Cohen’s d = 0.74).
Importantly, for each treatment, responders and non-responders did not differ in their HAMD17 at baseline, at week 8, and their change in HAMD17 from baseline to week 8 (see online Supplementary Tables S15 and S16). This indicates that even though the tasks were administered at baseline, they can be used to distinguish responders from non-responders in stage 2. Together, these findings suggest that reward processing, verbal fluency and cognitive control are capable of distinguishing bupropion responders who did not previously respond to sertraline from non-responders resistant to both classes of medication.
In contrast, pretreatment performance in CRT and ABRT did not differentially predict response to bupropion and sertraline. See online Supplementary Tables S10–S14 for details of these analyses. We also repeated all analyses by adding an additional covariate of smoking status and found that conclusions from all p value significance tests remained the same.
Discussion
Treatment for MDD is challenging and often proceeds via trial-and-error with limited success. To facilitate optimal treatment selection and inform timely adjustments, we sought to identify cognitive variables that can predict response in a treatment-specific manner by analyzing data from the EMBARC clinical trial. Several key findings emerged.
First, greater improvements in psychomotor and cognitive processing speeds within the first week, as well as better pretreatment performance, were specifically associated with higher likelihood of response to placebo. Moreover, the improvement of placebo responders in CRT was comparable to healthy individuals, which suggests they might possess a resilience factor. In contrast, non-responders had less CRT improvement than controls, suggesting the presence of a deficient factor. High placebo responses are commonly reported in clinical trials of novel antidepressants (Enck, Bingel, Schedlowski, & Rief, 2013; Schatzberg, 2015) and treatment with placebo has been found to induce distinct changes in brain functioning of depressed individuals (Enck et al., 2013; Leuchter, Cook, Witte, Morgan, & Abrams, 2002; Mayberg et al., 2002). Together, these findings suggest that, rather than having no effect, the administration of placebo is actually an active form of treatment. Accordingly, identifying MDD patients likely to respond to placebo in advance might have real-world clinical implications. Instead of a long-term antidepressant prescription, MDD patients identified as placebo responders could be treated with briefer, lower-cost interventions that are associated with fewer side effects (Enck et al., 2013). Previous studies in this area have largely focused on demographic variables and depressive symptom severity (Entsuah & Vinall, 2007; Fournier et al., 2010; Holmes, Tiwari, & Kennedy, 2016; Kirsch et al., 2008). More recently, Trivedi et al. analyzed 283 baseline variables from the EMBARC study and found that a higher likelihood of placebo response was predicted by baseline theta current density in the rostral anterior cingulate cortex (rACC) and several pretreatment clinical variables, such as anxious arousal, anhedonia, and neuroticism (Trivedi et al., 2018). However, they did not include early changes in cognition. Data from the sertraline arm were also not examined and thus, some of these predictors might not be specific to placebo. For example, Pizzagalli and coworkers demonstrated that increased baseline rACC theta activity represents a nonspecific marker of treatment outcome to both placebo and sertraline (Pizzagalli et al., 2018). Thus, our results extend the findings from these previous studies, suggesting the baseline and early changes in CRT and ABRT might be more specific predictors of placebo response.
Second, greater improvement in CRT within the first week was specifically associated with lower likelihood of response to sertraline. To the best of our knowledge, this is the first time that early changes in objective measures of ‘cold’ cognition have been reported to predict response to SSRIs. Several prior studies have focused on improvements in ‘hot’ cognition instead, consistently finding that early increases in emotional processing are associated with subsequent improvement in depressive symptoms during treatment with SSRIs (Godlewska et al., 2016; Shiroma, Thuras, et al., 2014; Tranter et al., 2009). Gorwood et al. also examined early changes in ‘cold’ cognition and found that improvements in various domains such as psychomotor function, motivation, cognitive speed, and sensory perception within the first 2 weeks all predicted response to the melatonin agonist, agomelatine, after 6 weeks (Gorwood et al., 2015). However, that study utilized a self-report questionnaire of cognition, which is inherently subjective and might be a less accurate measure of cognitive ability than behavioral tasks.
Third, better reward responsiveness, poorer cognitive control, and greater verbal fluency were associated with greater likelihood of response to bupropion in patients who previously failed to respond to sertraline. Furthermore, bupropion responders had comparable response bias and verbal fluency to healthy volunteers, whereas non-responders performed worse than controls. These findings suggest that responders to bupropion possess a resilience factor whereas a deficient factor might be present in non-responders. Prior studies have investigated cognitive predictors of treatment response to various antidepressants, including bupropion (Alexopoulos et al., 2007, 2015; Bruder et al., 2014; Cléry-Melin & Gorwood, 2017; Dunkin et al., 2000; Etkin et al., 2015; Groves et al., 2018; Gudayol-Ferré et al., 2010, 2012; Herrera-Guzmán et al., 2008; Kalayam & Alexopoulos, 2003; Mikoteit et al., 2015; Murrough et al., 2014, 2015; Shiroma, Albott, et al., 2014; Sneed et al., 2007; Taylor et al., 2006). For example, Herrera-Guzmán et al. (2008) showed that bupropion responders had lower pretreatment cognitive processing speed (as indexed by the Stockings of Cambridge test) compared to non-responders. Another study reported that baseline cognitive control (based on the Stroop interference effect) and verbal fluency were not significantly different in eventual responders and non-responders to bupropion (Bruder et al., 2014). In contrast, we found that lower cognitive control and higher verbal fluency predicted bupropion response, but cognitive processing speed did not. These discrepancies might have occurred due to differences in tasks used and smaller sample sizes in previous studies (N = ~20 v. N = ~40 here). Also, our findings may be specific to patients receiving secondary treatment with bupropion after failure to respond to sertraline. With regard to reward processing, our finding that bupropion responders have greater response bias on the PRT than non-responders has been reported in a recent publication (Ang et al., 2020), in which greater reward responsiveness and resting state frontostriatal functional connectivity were associated with response to bupropion, and is in line with substantial evidence showing that reward processes are modulated by dopaminergic system in the brain (Berridge, Robinson, & Aldridge, 2009). It is also consistent with a recent study showing depressed individuals with enhanced baseline response bias respond more favorably to pramipexole, a selective dopamine agonist (Whitton et al., 2020). In sum, our study is the first to address cognitive predictors of response to the noradrenaline/dopamine reuptake inhibitor (NDRI) bupropion following a failure to respond to the SSRI sertraline. This might have significant clinical value in identifying patients who are likely to respond to secondary treatment with bupropion and those who are unlikely to benefit from both SSRIs and NDRIs, so that they can be recommended alternative forms of treatment.
Limitations of this study should be acknowledged. First, multiple logistic regressions were conducted, but none of the findings would survive multiple comparisons. Although this might increase the chances of committing a type 1 error, our study is exploratory in nature and we were specifically interested in identifying whether each of the cognitive tasks could be a potential predictor of treatment outcome (Huberty & Morris, 1989). This liberal approach may not be stringent enough and thus, our findings are tentative and require replication. Second, this study adopted relatively strict inclusion criteria in order to minimize clinical heterogeneity. Thus, it is unclear whether findings will generalize to other depressed samples, such as those with psychosis or substance dependence. Third, this study did not exclude participants who had tobacco use disorder. Although chronic cigarette smoking has been associated with poorer cognitive performance across multiple domains (Durazzo, Meyerhoff, & Nixon, 2012; Nooyens, van Gelder, & Verschuren, 2008), all conclusions remained after accounting for an additional covariate of smoking status in our analyses.
Conclusion
Cognitive tasks that are quick, non-invasive, and easy to administer may have important clinical value as predictors of response to antidepressant treatment. The current study showed that psychomotor and cognitive processing speed after 1 week were associated with enhanced clinical response to placebo. Reward sensitivity, cognitive control, and verbal fluency at baseline also differentiated bupropion responders, who did not respond to sertraline previously, from non-responders resistant to both classes of medication. These initial results warrant further scrutiny for possible implementation in clinical care.
Supplementary Material
The supplementary material for this article can be found at https://doi.org/10.1017/S0033291720004286.
Acknowledgements
The authors are grateful to Daniel G. Dillon, Ph.D., for his support in the EMBARC study, as well as Boyu Ren, Ph.D., for providing advice on our statistical analyses.
Financial support
The EMBARC study was supported by the National Institute of Mental Health under award numbers U01MH092221 (M.H. Trivedi) and U01MH092250 (P.J. McGrath, R.V. Parsey, and M.M. Weissman). Y.S.A. was supported by a Kaplen Fellowship in Depression from Harvard Medical School, as well as the National Science Scholarship from the Agency for Science, Technology and Research (A*STAR) in Singapore. D.A.P. was partially supported by R37 MH068376. This study was supported by the EMBARC National Coordinating Center at UT Southwestern Medical Center, Madhukar H. Trivedi, M.D., Coordinating PI, and the Data Center at Columbia and Stony Brook Universities. The funder has no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or the decision to submit the manuscript for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Conflict of interest.
In the last 3 years, the authors report the following financial disclosures, for activities unrelated to the current research: Dr Carmody: owns stock in Vertex Pharmaceuticals and CRISPR Therapeutics. Dr Fava: Dr Fava reports the following lifetime disclosures: http://mghcme.org/faculty/faculty-detail/maurizio_fava. Dr Weissman: funding from NIMH, the National Alliance for Research on Schizophrenia and Depression (NARSAD), the Sackler Foundation, and the Templeton Foundation; royalties from the Oxford University Press, Perseus Press, the American Psychiatric Association Press, and MultiHealth Systems. Dr Oquendo: funding from NIMH; royalties for the commercial use of the Columbia-Suicide Severity Rating Scale; equity in Mantra, Inc. Her family owns stock in Bristol Myers Squibb. Dr McInnis: funding from NIMH; consulting fees from Janssen and Otsuka Pharmaceuticals. Dr Trivedi: reports the following lifetime disclosures: research support from the Agency for Healthcare Research and Quality, Cyberonics Inc., National Alliance for Research in Schizophrenia and Depression, National Institute of Mental Health, National Institute on Drug Abuse, National Institute of Diabetes and Digestive and Kidney Diseases, Johnson & Johnson, and consulting and speaker fees from Abbott Laboratories Inc., Akzo (Organon Pharmaceuticals Inc.), Allergan Sales LLC, Alkermes, AstraZeneca, Axon Advisors, Brintellix, Bristol-Myers Squibb Company, Cephalon Inc., Cerecor, Eli Lilly & Company, Evotec, Fabre Kramer Pharmaceuticals Inc., Forest Pharmaceuticals, GlaxoSmithKline, Health Research Associates, Johnson & Johnson, Lundbeck, MedAvante Medscape, Medtronic, Merck, Mitsubishi Tanabe Pharma Development America Inc., MSI Methylation Sciences Inc., Nestle Health Science-PamLab Inc., Naurex, Neuronetics, One Carbon Therapeutics Ltd., Otsuka Pharmaceuticals, Pamlab, Parke-Davis Pharmaceuticals Inc., Pfizer Inc., PgxHealth, Phoenix Marketing Solutions, Rexahn Pharmaceuticals, Ridge Diagnostics, Roche Products Ltd., Sepracor, SHIRE Development, Sierra, SK Life and Science, Sunovion, Takeda, Tal Medical/Puretech Venture, Targacept, Transcept, VantagePoint, Vivus, and Wyeth-Ayerst Laboratories. Dr Pizzagalli: funding from NIMH, Brain and Behavior Research Foundation, the Dana Foundation, and Millennium Pharmaceuticals; consulting fees from BlackThorn Therapeutics, Boehreinger Ingelheim, Compass Pathway, Engrail Pharmaceuticals, Otsuka Pharmaceuticals, and Takeda Pharmaceuticals; one honorarium from Alkermes; stock options from BlackThorn Therapeutics. Dr Pizzagalli has a financial interest in BlackThorn Therapeutics, which has licensed the copyright to the Probabilistic Reward Task through Harvard University. Dr Pizzagalli’s interests were reviewed and are managed by McLean Hospital and Partners HealthCare in accordance with their conflict of interest policies. All other authors report no biomedical financial interests or potential conflicts of interest.
Ethical standards.
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
References
- Alexopoulos GS, Manning K, Kanellopoulos D, McGovern A, Seirup JK, Banerjee S, Gunning F. Cognitive control, reward-related decision making and outcomes of late-life depression treated with an antidepressant. Psychological Medicine. 2015;45(14):3111–3120. doi: 10.1017/S0033291715001075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexopoulos GS, Murphy CF, Gunning-Dixon FM, Kalayam B, Katz R, Kanellopoulos D, et al. Foxe JJ. Event-related potentials in an emotional go/no-go task and remission of geriatric depression. Neuroreport. 2007;18(3):217–221. doi: 10.1097/WNR.0b013e328013ceda. [DOI] [PubMed] [Google Scholar]
- Ang Y-S, Kaiser R, Deckersbach T, Almeida J, Phillips ML, Chase HW, et al. Pizzagalli DA. Pretreatment reward sensitivity and frontostriatal resting-state functional connectivity are associated with response to bupropion after sertraline non-response. Biological Psychiatry. 2020;88(8):657–667. doi: 10.1016/j.biopsych.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baddeley AD. A 3 min reasoning test based on grammatical transformation. Psychonomic Science. 1968;10(10):341–342. doi: 10.3758/BF03331551. [DOI] [Google Scholar]
- Beblo T, Baumann B, Bogerts B, Wallesch C-W, Herrmann M. Neuropsychological correlates of major depression: A short-term follow-up. Cognitive Neuropsychiatry. 1999;4(4):333–341. doi: 10.1080/135468099395864. [DOI] [Google Scholar]
- Benton AL, Hamsher K, Sivan AB. Multilingual aphasia Examination. 3rd ed AJA Associates; Iowa City, IA: 1983. [Google Scholar]
- Berridge KC, Robinson TE, Aldridge JW. Dissecting components of reward: ‘liking’, ‘wanting’, and learning. Current Opinion in Pharmacology. 2009;9(1):65–73. doi: 10.1016/j.coph.2008.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruder GE, Alvarenga JE, Alschuler D, Abraham K, Keilp JG, Hellerstein DJ, et al. McGrath PJ. Neurocognitive predictors of antidepressant clinical response. Journal of Affective Disorders. 2014;166:108–114. doi: 10.1016/j.jad.2014.04.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cléry-Melin M-L, Gorwood P. A simple attention test in the acute phase of a major depressive episode is predictive of later functional remission: Cléry-M Elin et al. Depression and Anxiety. 2017;34(2):159–170. doi: 10.1002/da.22575. [DOI] [PubMed] [Google Scholar]
- Dunkin JJ, Leuchter AF, Cook IA, Kasl-Godley JE, Abrams M, Rosenberg-Thompson S. Executive dysfunction predicts nonresponse to fluoxetine in major depression. Journal of Affective Disorders. 2000;60(1):13–23. doi: 10.1016/s0165-0327(99)00157-3. [DOI] [PubMed] [Google Scholar]
- Durazzo TC, Meyerhoff DJ, Nixon SJ. A comprehensive assessment of neurocognition in middle-aged chronic cigarette smokers. Drug and Alcohol Dependence. 2012;122(1–2):105–111. doi: 10.1016/j.drugalcdep.2011.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enck P, Bingel U, Schedlowski M, Rief W. The placebo response in medicine: Minimize, maximize or personalize? Nature Reviews Drug Discovery. 2013;12(3):191–204. doi: 10.1038/nrd3923. [DOI] [PubMed] [Google Scholar]
- Entsuah R, Vinall P. Potential predictors of placebo response: Lessons from a large database. Drug Information Journal. 2007;41(3):315–330. doi: 10.1177/009286150704100304. [DOI] [Google Scholar]
- Eriksen CW. The flankers task and response competition: A useful tool for investigating a variety of cognitive problems. Visual Cognition. 1995;2(2–3):101–118. doi: 10.1080/13506289508401726. [DOI] [Google Scholar]
- Etkin A, Patenaude B, Song YJC, Usherwood T, Rekshan W, Schatzberg AF, et al. Williams LM. A cognitive-emotional biomarker for predicting remission with antidepressant medications: A report from the iSPOT-D trial. Neuropsychopharmacology. 2015;40(6):1332–1342. doi: 10.1038/npp.2014.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fournier JC, DeRubeis RJ, Hollon SD, Dimidjian S, Amsterdam JD, Shelton RC, Fawcett J. Antidepressant drug effects and depression severity: A patient-level meta-analysis. JAMA. 2010;303(1):47. doi: 10.1001/jama.2009.1943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fredman SJ, Fava M, Kienke AS, White CN, Nierenberg AA, Rosenbaum JF. Partial response, nonresponse, and relapse with selective serotonin reuptake inhibitors in major depression: A survey of current ‘next-step’ practices. The Journal of Clinical Psychiatry. 2000;61(6):403–408. doi: 10.4088/jcp.v61n0602. [DOI] [PubMed] [Google Scholar]
- Godlewska BR, Browning M, Norbury R, Cowen PJ, Harmer CJ. Early changes in emotional processing as a marker of clinical response to SSRI treatment in depression. Translational Psychiatry. 2016;6(11):e957. doi: 10.1038/tp.2016.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorlyn M, Keilp JG, Grunebaum MF, Taylor BP, Oquendo MA, Bruder GE, et al. Mann JJ. Neuropsychological characteristics as predictors of SSRI treatment response in depressed subjects. Journal of Neural Transmission. 2008;115(8):1213–1219. doi: 10.1007/s00702-008-0084-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorwood P, Vaiva G, Corruble E, Llorca P-M, Baylé FJ, Courtet P. The ability of early changes in motivation to predict later antidepressant treatment response. Neuropsychiatric Disease and Treatment. 2015;11:2875–2882. doi: 10.2147/NDT.S92795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenberg PE, Fournier A-A, Sisitsky T, Pike CT, Kessler RC. The economic burden of adults with major depressive disorder in the United States (2005 and 2010) The Journal of Clinical Psychiatry. 2015;76(02):155–162. doi: 10.4088/JCP.14m09298. [DOI] [PubMed] [Google Scholar]
- Groves SJ, Douglas KM, Porter RJ. A systematic review of cognitive predictors of treatment outcome in major depression. Frontiers in Psychiatry. 2018;9:382. doi: 10.3389/fpsyt.2018.00382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gudayol-Ferré E, Herrera-Guzmán I, Camarena B, Cortés-Penagos C, Herrera-Abarca JE, Martínez-Medina P, et al. Guàrdia-Olmos J. The role of clinical variables, neuropsychological performance and SLC6A4 and COMT gene polymorphisms on the prediction of early response to fluoxetine in major depressive disorder. Journal of Affective Disorders. 2010;127(1–3):343–351. doi: 10.1016/j.jad.2010.06.002. [DOI] [PubMed] [Google Scholar]
- Gudayol-Ferré E, Herrera-Guzmán I, Camarena B, Cortés-Penagos C, Herrera-Abarca JE, Martínez-Medina P, et al. Guàrdia-Olmos J. Prediction of remission of depression with clinical variables, neuropsychological performance, and serotonergic/dopaminergic gene polymorphisms: Prediction of remission of depression. Human Psychopharmacology: Clinical and Experimental. 2012;27(6):577–586. doi: 10.1002/hup.2267. [DOI] [PubMed] [Google Scholar]
- Hamilton M. A rating scale for depression. Journal of Neurology, Neurosurgery, and Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammar Ã, Sørensen L, Ardal G, Oedegaard KJ, Kroken R, Roness A, et al. Lund A. Enduring cognitive dysfunction in unipolar major depression: A test–retest study using the Stroop paradigm. Scandinavian Journal of Psychology. 2009;51(4):304–308. doi: 10.1111/j.1467-9450.2009.00765.x. [DOI] [PubMed] [Google Scholar]
- Herrera-Guzmán I, Gudayol-Ferré E, Lira-Mandujano J, Herrera-Abarca J, Herrera-Guzmán D, Montoya-Pérez K, Guardia-Olmos J. Cognitive predictors of treatment response to bupropion and cognitive effects of bupropion in patients with major depressive disorder. Psychiatry Research. 2008;160(1):72–82. doi: 10.1016/j.psychres.2007.04.012. [DOI] [PubMed] [Google Scholar]
- Herrera-Guzmán I, Herrera-Abarca JE, Gudayol-Ferré E, Herrera-Guzmán D, Gómez-Carbajal L, Peña-Olvira M, et al. Joan G-O. Effects of selective serotonin reuptake and dual serotonergic-noradrenergic reuptake treatments on attention and executive functions in patients with major depressive disorder. Psychiatry Research. 2010;177(3):323–329. doi: 10.1016/j.psychres.2010.03.006. [DOI] [PubMed] [Google Scholar]
- Hinkelmann K, Moritz S, Botzenhardt J, Muhtz C, Wiedemann K, Kellner M, Otte C. Changes in cortisol secretion during antide-pressive treatment and cognitive improvement in patients with major depression: A longitudinal study. Psychoneuroendocrinology. 2012;37(5):685–692. doi: 10.1016/j.psyneuen.2011.08.012. [DOI] [PubMed] [Google Scholar]
- Holmes RD, Tiwari AK, Kennedy JL. Mechanisms of the placebo effect in pain and psychiatric disorders. The Pharmacogenomics Journal. 2016;16(6):491–500. doi: 10.1038/tpj.2016.15. [DOI] [PubMed] [Google Scholar]
- Huberty CJ, Morris JD. Multivariate analysis versus multiple univariate analyses. Psychological Bulletin. 1989;105(2):302–308. doi: 10.1037/0033-2909.105.2.302. [DOI] [Google Scholar]
- James SL, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Murray CJL. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: A systematic analysis for the Global Burden of Disease Study 2017. The Lancet. 2018;392(10159):1789–1858. doi: 10.1016/S0140-6736(18)32279-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalayam B, Alexopoulos GS. A preliminary study of left frontal region error negativity and symptom improvement in geriatric depression. The American Journal of Psychiatry. 2003;160(11):2054–2056. doi: 10.1176/appi.ajp.160.11.2054. [DOI] [PubMed] [Google Scholar]
- Katon W, Robinson P, Von Korff M, Lin E, Bush T, Ludman E, Walker E. A multifaceted intervention to improve treatment of depression in primary care. Archives of General Psychiatry. 1996;53(10):924–932. doi: 10.1001/archpsyc.1996.01830100072009. [DOI] [PubMed] [Google Scholar]
- Keilp JG, Sackeim HA, Mann JJ. Correlates of trait impulsiveness in performance measures and neuropsychological tests. Psychiatry Research. 2005;135(3):191–201. doi: 10.1016/j.psychres.2005.03.006. [DOI] [PubMed] [Google Scholar]
- Kessler RC, Berglund P, Demler O, Jin R, Koretz D, Merikangas KR, et al. Wang PS. The epidemiology of major depressive disorder: Results from the national comorbidity survey replication (NCS-R) JAMA. 2003;289(23):3095. doi: 10.1001/jama.289.23.3095. [DOI] [PubMed] [Google Scholar]
- Kirsch I, Deacon BJ, Huedo-Medina TB, Scoboria A, Moore TJ, Johnson BT. Initial severity and antidepressant benefits: A meta-analysis of data submitted to the Food and Drug Administration. PLoS Medicine. 2008;5(2):e45. doi: 10.1371/journal.pmed.0050045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leuchter AF, Cook IA, Witte EA, Morgan M, Abrams M. Changes in brain function of depressed subjects during treatment with placebo. American Journal of Psychiatry. 2002;159(1):122–129. doi: 10.1176/appi.ajp.159.1.122. [DOI] [PubMed] [Google Scholar]
- Mayberg HS, Silva JA, Brannan SK, Tekell JL, Mahurin RK, McGinnis S, Jerabek PA. The functional neuroanatomy of the placebo effect. American Journal of Psychiatry. 2002;159(5):728–737. doi: 10.1176/appi.ajp.159.5.728. [DOI] [PubMed] [Google Scholar]
- Mikoteit T, Hemmeter U, Eckert A, Brand S, Bischof R, Delini-Stula A, et al. Beck J. Improved alertness is associated with early increase in Serum brain-derived neurotrophic factor and antidepressant treatment outcome in major depression. Neuropsychobiology. 2015;72(1):16–28. doi: 10.1159/000437439. [DOI] [PubMed] [Google Scholar]
- Murrough JW, Burdick KE, Levitch CF, Perez AM, Brallier JW, Chang LC, et al. Iosifescu DV. Neurocognitive effects of ketamine and association with antidepressant response in individuals with treatment-resistant depression: A randomized controlled trial. Neuropsychopharmacology. 2015;40(5):1084–1090. doi: 10.1038/npp.2014.298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murrough JW, Wan L-B, Iacoviello B, Collins KA, Solon C, Glicksberg B, Burdick KE. Neurocognitive effects of ketamine in treatment-resistant major depression: Association with antidepressant response. Psychopharmacology. 2014;231:481–488. doi: 10.1007/s00213-013-3255-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nooyens ACJ, van Gelder BM, Verschuren WMM. Smoking and cognitive decline among middle-aged men and women: The Doetinchem Cohort Study. American Journal of Public Health. 2008;98(12):2244–2250. doi: 10.2105/AJPH.2007.130294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizzagalli DA, Jahn AL, O’Shea JP. Toward an objective characterization of an anhedonic phenotype: A signal-detection approach. Biological Psychiatry. 2005;57(4):319–327. doi: 10.1016/j.biopsych.2004.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizzagalli DA, Webb CA, Dillon DG, Tenke CE, Kayser J, Goer F, et al. Trivedi MH. Pretreatment rostral anterior cingulate cortex theta activity in relation to symptom improvement in depression: A randomized clinical trial. JAMA Psychiatry. 2018;75(6):547. doi: 10.1001/jamapsychiatry.2018.0252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reppermund S, Ising M, Lucae S, Zihl J. Cognitive impairment in unipolar depression is persistent and non-specific: Further evidence for the final common pathway disorder hypothesis. Psychological Medicine. 2009;39(4):603–614. doi: 10.1017/S003329170800411X. [DOI] [PubMed] [Google Scholar]
- Reppermund S, Zihl J, Lucae S, Horstmann S, Kloiber S, Holsboer F, Ising M. Persistent cognitive impairment in depression: The role of psychopathology and altered hypothalamic-pituitary-adrenocortical (HPA) system regulation. Biological Psychiatry. 2007;62(5):400–406. doi: 10.1016/j.biopsych.2006.09.027. [DOI] [PubMed] [Google Scholar]
- Rush AJ, Trivedi MH, Ibrahim HM, Carmody TJ, Arnow B, Klein DN, et al. Keller MB. The 16-item Quick Inventory of Depressive Symptomatology (QIDS), clinician rating (QIDS-C), and self-report (QIDS-SR): A psychometric evaluation in patients with chronic major depression. Biological Psychiatry. 2003;54(5):573–583. doi: 10.1016/s0006-3223(02)01866-8. [DOI] [PubMed] [Google Scholar]
- Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Fava M. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: A STAR*D report. American Journal of Psychiatry. 2006;163(11):1905–1917. doi: 10.1176/ajp.2006.163.11.1905. [DOI] [PubMed] [Google Scholar]
- Schatzberg AF. Issues encountered in recent attempts to develop novel antidepressant agents: Developing new antidepressant agents. Annals of the New York Academy of Sciences. 2015;1345(1):67–73. doi: 10.1111/nyas.12716. [DOI] [PubMed] [Google Scholar]
- Shiroma PR, Albott CS, Johns B, Thuras P, Wels J, Lim KO. Neurocognitive performance and serial intravenous subanesthetic ketamine in treatment-resistant depression. The International Journal of Neuropsychopharmacology. 2014;17(11):1805–1813. doi: 10.1017/S1461145714001011. [DOI] [PubMed] [Google Scholar]
- Shiroma PR, Thuras P, Johns B, Lim KO. Emotion recognition processing as early predictor of response to 8-week citalopram treatment in late-life depression: Emotion recognition in late-life depression. International Journal of Geriatric Psychiatry. 2014;29(11):1132–1139. doi: 10.1002/gps.4104. [DOI] [PubMed] [Google Scholar]
- Sneed JR, Roose SP, Keilp JG, Krishnan KRR, Alexopoulos GS, Sackeim HA. Response inhibition predicts poor antidepressant treatment response in very old depressed patients. The American Journal of Geriatric Psychiatry. 2007;15(7):553–563. doi: 10.1097/JGP.0b013e3180302513. [DOI] [PubMed] [Google Scholar]
- Souery D, Papakostas GI, Trivedi MH. Treatment-resistant depression. The Journal of Clinical Psychiatry. 2006;67(Suppl 6):16–22. [PubMed] [Google Scholar]
- Taylor BP, Bruder GE, Stewart JW, McGrath PJ, Halperin J, Ehrlichman H, Quitkin FM. Psychomotor slowing as a predictor of fluoxetine nonresponse in depressed outpatients. The American Journal of Psychiatry. 2006;163(1):73–78. doi: 10.1176/appi.ajp.163.1.73. [DOI] [PubMed] [Google Scholar]
- Thorne DR, Genser SG, Sing HC, Hegge FW. The Walter Reed performance assessment battery. Neurobehavioral Toxicology and Teratology. 1985;7(4):415–418. [PubMed] [Google Scholar]
- Tranter R, Bell D, Gutting P, Harmer C, Healy D, Anderson IM. The effect of serotonergic and noradrenergic antidepressants on face emotion processing in depressed patients. Journal of Affective Disorders. 2009;118(1-3):87–93. doi: 10.1016/j.jad.2009.01.028. [DOI] [PubMed] [Google Scholar]
- Trivedi MH, McGrath PJ, Fava M, Parsey RV, Kurian BT, Phillips ML, et al. Weissman MM. Establishing moderators and biosignatures of antidepressant response in clinical care (EMBARC): Rationale and design. Journal of Psychiatric Research. 2016;78:11–23. doi: 10.1016/j.jpsychires.2016.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L, et al. STAR*D Study Team Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: Implications for clinical practice. American Journal of Psychiatry. 2006;163(1):28–40. doi: 10.1176/appi.ajp.163.1.28. [DOI] [PubMed] [Google Scholar]
- Trivedi MH, South C, Jha MK, Rush AJ, Cao J, Kurian B, et al. Fava M. A novel strategy to identify placebo responders: Prediction index of clinical and biological markers in the EMBARC trial. Psychotherapy and Psychosomatics. 2018;87(5):285–295. doi: 10.1159/000491093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vrieze E, Pizzagalli DA, Demyttenaere K, Hompes T, Sienaert P, de Boer P, et al. Claes S. Reduced reward learning predicts outcome in major depressive disorder. Biological Psychiatry. 2013;73(7):639–645. doi: 10.1016/j.biopsych.2012.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitton AE, Reinen JM, Slifstein M, Ang Y-S, McGrath PJ, Iosifescu DV, et al. Schneier FR. Baseline reward processing and ventrostriatal dopamine function are associated with pramipexole response in depression. Brain. 2020;143(2):701–710. doi: 10.1093/brain/awaa002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.