Abstract
Abiotic factors (e.g., temperature, precipitation) vary markedly along elevational gradients, and differentially affect major groups of pollinators. Ectothermic bees, for example, are impeded in visiting flowers by cold and rainy conditions common at high elevations, while endothermic hummingbirds may continue foraging under such conditions. Despite the possibly far-reaching effects of the abiotic environment on plant-pollinator interactions, we know little about how these factors play out at broad ecogeographic scales. We address this knowledge gap by investigating how pollination systems vary across elevations in 26 plant clades from the Americas. Specifically, we explore Cruden’s 1972 hypothesis that the harsh montane environment drives a turnover from insect to vertebrate pollination at higher elevations. We compared the elevational distribution and bioclimatic attributes for a total of 2232 flowering plants and found that Cruden’s hypothesis only holds in the tropics. Above 30° N and below 30°S, plants pollinated by vertebrates (mostly hummingbirds) tend to occur at lower elevations than those pollinated by insects. We hypothesize that this latitudinal transition is due to the distribution of moist, forested habitats favored by vertebrate pollinators, which are common at high elevations in the tropics but not in the temperate Americas.
Keywords: altitudinal gradient, hummingbird habitats, pollinator shift, precipitation
Abstract
Patrones opuestos de reemplazo de polinizadores en montañas tropicales y templadas en América
Factores abióticos como temperatura y precipitación varían marcadamente a lo largo de gradientes altitudinalese impactan diferencialmente a grupos de polinizadores. Por ejemplo, las abejas son ectotermas y por tanto su visita a flores se reduce en ambientes fríos y lluviosos, propio de ecosistemas de alta montaña. Por otro lado, los colibríes, que son endotermos, podrían seguir visitando flores en aquellas condiciones. A pesar del gran impacto que los factores abióticos pueden tener en interacciones planta-animal, sabemos poco acerca de dicho efecto a grandes escalas ecogeográficas. Para llenar este vacío de conocimiento, en este estudio investigamos cómo cambian los sistemas de polinización en gradientes altitudinales en 26 clados de plantas en Norte y Sur América. Específicamente, exploramos la hipótesis de Cruden de 1972, en la quecondiciones en ambientes de alta montaña causan un reemplazo de polinización por insectos a polinización por vertebrados. Comparamos la distribución altitudinal y factores climáticos de un total de 2232 especies de plantas y encontramos que la hipótesis de Cruden solamente aplica en el trópico. Por encima de 30°N y por debajo de 30°S, las plantas polinizadas por vertebrados (en su mayoría colibríes) tienden a ocurrir en zonas de elevación más bajas que especies polinizadas por insectos. Proponemos que esta transición latitudinal se debe a la distribución de hábitats húmedos y boscosos, preferidos por polinizadores vertebrados. Tales hábitats son abundantes en montañas tropicales (p.e., bosques de niebla) mas no en zonas templadas de Norte y Sur América.
Introduction
Biotic interactions are among the most prominent factors limiting, promoting and structuring organismal diversity on earth (Schemske et al. 2009, Sinnott-Armstrong et al. 2021). For plant-pollinator interactions, associations are unevenly distributed across space due to variation in pollinator availability and importance in different regions and ecosystems (Ollerton 2017, Dellinger et al. 2021, Orr et al. 2021). Ecogeographical variation in plant-pollinator relationships emerges across broad latitudinal and altitudinal gradients as major groups of pollinating animals vary in their distributions (e.g. Classen et al. 2015, McCabe & Cobb 2021, Dellinger et al. 2021). For example, bats, rodents and passerine birds are important pollinators particularly in tropical regions (Ratto et al. 2018), while insects and hummingbirds act as pollinators across latitudes in the Americas. Similarly, diverse insect pollinator assemblages (i.e. bees, beetles, wasps, butterflies), common at low to mid elevations, are narrowed to bumblebee- and particularly fly-dominated systems in many high elevation communities (Warren et al. 1988, Arroyo et al. 1982, Primack & Inouye 1993, Lefevbre et al. 2019, Adedoja et al. 2020, McCabe & Cobb 2021). Although these patterns suggest a critical role of climatic factors in structuring plant-pollinator associations, we currently lack broad-scale ecogeographic assessments of the distribution of pollination modes.
Major turnovers in pollinator groups across latitudinal and altitudinal gradients may reflect differences in the pollinators’ tolerance of extrinsic abiotic climatic stressors (Ollerton 2017, Lefevbre et al. 2019). For example, cool and wet weather generally restricts the activity of ectothermic animals like many bees and other small insects (McCallum et al. 2013, Classen et al. 2015, Cozien et al. 2019), while large endothermic animals often continue to forage through moderate to heavy rain (hummingbirds, sunbirds; Sun et al. 2017, Lawson & Rands 2019). In his groundbreaking 1972 paper, Cruden used this observation to hypothesize that vertebrate (particularly bird) pollinated plants will be more abundant in habitats (cold mountains) and seasons (rainy) with reduced bee activity, as vertebrates will be more reliable pollinators in such habitats and seasons. His empirical studies of pollinator communities along four elevational transects in the Mexican Sierra Madre mountains supported this hypothesis (Cruden 1972). The environment-induced differences in activity between ecto- and endothermic pollinators observed by Cruden have critical evolutionary implications (Arroyo et al. 1982, Krömer et al. 2006, Dellinger et al. 2021). Specifically, if cold and rainy conditions prevalent in mountains consistently reduce bee, but not bird pollinator activity, we may expect to find repeated evolutionary pollinator shifts in plant lineages that occur across elevational gradients.
While Cruden’s hypothesis on insect-vertebrate pollinator turnover across elevational (and thus climatic) gradients has been widely accepted, some reports indicate that this pattern does not hold across latitudes. In temperate regions, insects are often the dominant pollinators in high elevation ecosystems (McCallum et al. 2013, Woodard 2017, McCabe & Cobb 2021), a pattern which may relate to the ability of many pollinator species, such as bumblebees and syrphid flies, to regulate their body temperature (Heinrich 1974; Oyen & Dillon 2018). Hummingbirds, by contrast, are more important pollinators in more mesic, lower elevation temperate areas (Grant & Grant 1968, Hamilton and Wessinger 2022), so called ‘hummingbird habitats’ (Stebbins 1989). Whether more mesic habitats represent environments of increased hummingbird pollinator importance more broadly remains to be investigated.
Plant clades featuring evolutionary shifts in pollinators (e.g., between bee and hummingbird pollination, Thomson & Wilson 2008) and occurring across ecogeographic gradients, represent ideal systems to test whether there is a strong link between the abiotic environment and pollination strategy. If certain abiotic environmental conditions do indeed favor pollination by vertebrates over insects (i.e., through a significant reduction of bee pollinator efficiency, Thomson & Wilson 2008), we would expect to see vertebrate pollination associated with these conditions repeatedly across different plant lineages. To our knowledge, however, no attempts have been made to systematically test whether pollination strategies of clades which have colonized montane environments and shifted pollinators follow Cruden’s altitude/climate-driven bee-hummingbird pollinator turnover, nor have the potential bioclimatic thresholds linked to such turnovers been identified.
Here, we address these research gaps by investigating ecogeographic patterns of pollination systems across 2232 plant taxa from 26 clades, serving as evolutionary replicates, across the entire Americas. We chose to focus on the Americas since there, in contrast to other continents, vertebrate pollinators are represented across latitudes by hummingbirds (Cronk & Ojeda 2008), allowing for a broad-scale test of Cruden’s (1972) altitude/climate-driven pollinator turnover hypothesis. In addition to testing for associations between elevation and pollination systems, we consider how environmental factors (temperature, precipitation, cloud cover) and biome type relate to pollination mode and whether moist, shady habitats do indeed represent hummingbird habitats. Together, these analyses provide the hitherto broadest assessment of ecogeographic factors underlying the distribution of pollination strategies in the Americas.
Methods
Selection of study clades and scoring pollination mode
For selecting study clades, we employed the following criteria: 1) clades encompassing both bee and hummingbird pollination in order to span insects and vertebrates, with 2) molecular phylogenetic information available, and 3) occurring across elevational gradients in the Americas. We note that, in targeting clades with evolutionary pollinator transitions that may be associated with elevation, our criteria exclude clades with narrow elevational ranges and clades with invariant pollination strategies, which could be informative about community level questions (see Discussion). We also required that at least some members of the clade have empirical studies of pollination, allowing for system-specific predictions of pollination mode in the remaining taxa. For the latter, we relied on literature from the experts in each system to classify the taxa (see literature cited in Table S1). In order to find clades meeting these requirements, we began with the lists of Tripp & Manos (2008) and Abrahamczyk & Renner (2015), and we supplemented the dataset with other well studied groups (e.g., Bromeliaceae, Loranthaceae, Merianieae) using keyword searches in google scholar (i.e. clade name + pollinat*). This resulted in an initial list of 2563 taxa across 26 groups.
In summarizing the pollination literature for these taxa, we scored the principal pollinator for each species as one of five categories: bee, other insect, hummingbird, mixed hummingbird-insect and other vertebrates (e.g., bats, other birds; Table S1). These categories allow us to capture some of the diversity of systems within insect and vertebrate pollination and align with commonly used functional groupings of pollinators in Neotropical systems (Fenster et al. 2004). In total, we were able to score 27.6% of the taxa for empirical pollinator observations. For the remainder, we follow trait-based (pollination syndrome) scoring from the literature (Table S1).
GBIF occurrence data and environmental variables
All following steps were performed in the programming environment R (R Development Core team 2021). We screened the initial plant taxon list (n = 2563) using Taxonstand (Cayuela et al. 2021) to correct spelling mistakes and synonyms. Next, we submitted the list to GBIF to search for occurrence data for each taxon (rgbif, Chamberlain et al. 2021). We applied strict filtering using the function occ_issues, filtering out records with a continent-country mismatch, country-coordinate mismatch, records where the continent classification was derived from coordinates, invalid continents or coordinates, zero coordinates, coordinates out of range, presumed swapped coordinates, invalid geodetic datum, fuzzy taxon matches and non-metric, non-numeric or unlikely elevation (leaving 732,047 records). Next, we filtered data using CoordinateCleaner (Zizka et al. 2018) removing records located in country centroids, the sea, around gbif headquarters, duplicates or records with equal longitude and latitude (leaving 548,251 records). We then removed records outside of the Americas and calculated median elevation, median latitude and median longitude for each taxon, leaving 1807 taxa. Taking the median per taxon further minimizes bias due to potential single erroneous occurrence points.
Through this data extraction process, 29% of the taxa (756) were removed due to a lack of elevation information. In order to retain more species in the final dataset, we repeated the GBIF query for these 756 species, filtering for coordinate-related issues as above but not removing records lacking elevation information (8913 records). For each of these records, we extracted an elevation value from a global 30 arc sec elevation raster (Amatulli et al. 2018, raster, Hijmans 2021) and, again, calculated median elevation, latitude and longitude per taxon. To verify compatibility of both datasets, we use the same process to extract elevation information for the 1807 which already had elevation data, and found that the reported and extracted elevations produced a high positive correlation (R2 = 0.97).
We ran an additional, manual screening of the taxon list to identify misspelled taxon names or synonyms that had not been corrected by Taxonstand, and downloaded additional data as specified above. Finally, since some rare taxa may have gotten lost by the strict filtering settings, we ran a last GBIF query for the remaining taxa with more relaxed filtering settings (only filtering through CoordinateCleaner, adding five taxa). This left us with a final dataset of 2232 taxa across 26 study groups (Table S1, Fig. S1), with a median number of 22 occurrence points per taxon (range 1 – 8238). Of these, seventy-six taxa were represented by a single occurrence record, distributed evenly across pollination strategies with 35 insect- and 33 vertebrate-pollinated).
In order to evaluate the associations between pollination modes and climate, we downloaded layers for mean annual temperature (bio1) and precipitation (bio12) from the worldclim dataset at 30 arc sec resolution (Hijmans 2017). In addition, we downloaded data on mean annual cloud cover (https://www.earthenv.org/cloud) since high cloud cover strongly impacts flight activity of poikilothermic insects, but not of birds (Cruden 1972). Next, for each pruned occurrence record, we extracted the respective precipitation, temperature and cloud cover value and calculated the median per taxon. The final dataset is available in the online repository Dryad (https://datadryad.org/stash/share/6-aChxVVGL1Mox8UNtwYD8JQsqCUGPK_pLUhvmzZbrY).
Ecogeographic modelling
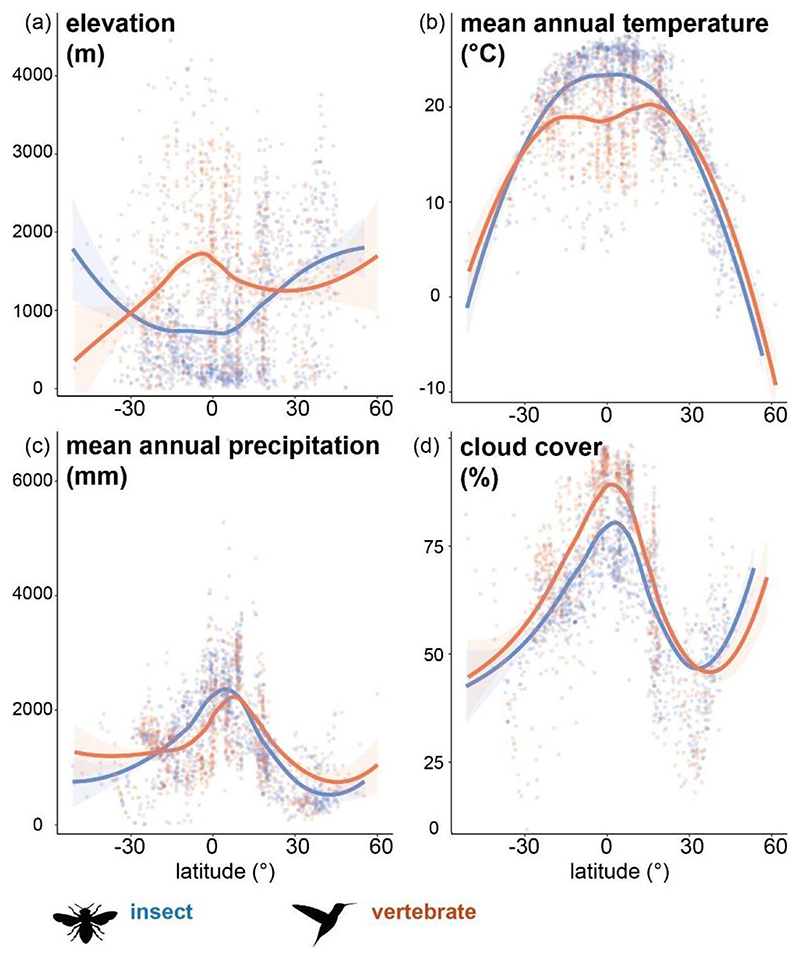
To summarize distribution patterns in our dataset, we plotted occurrence data on a map of the Americas (maptools, Bivand & Lewin-Koh 2021). We used Whittaker biomes (Whittaker 1975, Ricklefs 2008, plotbiomes, Stefan & Levin 2021) to assess whether vertebrate- or insect-pollinated species in our sample consistently associate with different major ecogeographic areas. We used Chi-square statistics to test for differences in biome occupation. To visualize the relative contribution of different biomes to Chi-square statistics, we plotted standardized residuals using correlation plots (corrplot, Wei & Simko 2021). Standardized residuals higher than +/- 2 indicate a strong contribution of the respective biome (Fig. 1).
Figure 1. Geographical and biome distribution of insect- and vertebrate-pollinated species included in our dataset.
(a) Insect-pollinated species (n = 1262) and (b) vertebrate-pollinated species (n = 970) across the Americas; each dot represents the median occurrence of each species. (c) Vertebrate-pollinated species in our dataset were found primarily under intermediate mean annual temperature (10-20°C) and mean annual precipitation (e.g. in temperate seasonal forests), while insect-pollinated species also occurred under cooler and drier (e.g. in temperate grasslands, shrublands) and warmer (e.g. in tropical seasonal forests) conditions. (d) Contribution of biomes (absolute standardized residuals) to total X2, positive values (blue) indicate a positive relation, negative values (red) indicate a negative relation, the size of the circle indicates the strength of the relationship.
We used generalized linear mixed effects models (GLMMs, lme4, Bates et al. 2015) to test whether associations between pollination mode and elevation or climate depend on latitude. Since elevation was strongly positively correlated with precipitation and temperature (Fig. S2), we only included elevation, latitude and cloud cover in our model. Further, we merged the five pollinator groups into a binary response variable (insect versus vertebrate) for two reasons. First, visualizing our data showed strikingly similar patterns among species classified as bee or insect pollinated, and among species classified as hummingbird, mixed-hummingbird or other-vertebrate pollinated (Fig. S3, S4). Second, species initially classified as other-vertebrate pollinated were restricted to the tropics, which would bias model estimation at higher latitudes. We used spline correlograms (Bjornstad 2020) to rule out issues related to spatial autocorrelation both in our raw data and fitted models (see below). We then constructed binomial GLMMs (logit link), testing for an interaction of elevation, cloud cover and latitude. We included the respective study clades as a random effect variable, testing models with random intercepts, and random slopes (e.g. elevation|taxonomic group) and intercepts. Since initial models did not converge, we scaled and centered the numeric predictor variables and optimized models using bobyqa (Bates et al. 2014) across 2e5 iterations. For selecting the best-fit model, we ran stepwise model simplification (drop1) to identify the most parsimonious model and used Chi-squares and ANOVA to check whether the simplified model led to a significant reduction in the residual sum of squares. We also compared model fit using AICs (Table S4). To validate the final model, we visually checked Pearson residuals, predicted versus original values and the ROC (receiver operating characteristic curve, pROC, Robin et al. 2011, Fig. S5). We visualized fixed and random effects using sjPlot (Lüdecke 2021, Fig. S6), effects (Fox & Weisberg 2018) and ggplot2 (Wickham 2016). We back-transformed model coefficients before plotting using custom settings in sjPlot (plot_model function, Lüdecke 2021).
Next, to assess the robustness of our results regarding the relationships between pollination, elevation and latitude, we ran four additional sets of analyses. First, to assess potential bias arising from uneven sampling across study groups and pollination strategies, we randomly subsampled our dataset to 25% 100 times, reran model selection, refit the best-fit models and summarized model coefficients (Table S5, S6). Second, to assess potential bias from using pollination syndromes in the literature to classify taxa, we re-ran the model selection with only the 616 species from 23 clades (258 insect- and 358 vertebrate-pollinated) that had empirically documented pollinators (Table S7, Fig. S7, S8). Third, to check whether mid-domain effects (i.e., artificial peaks in species richness due to geometric constraints when reducing distribution data to median values, Colwell et al. 2016) bias our results, we randomly resampled the pruned occurrence data to one occurrence point per species 100 times, refit the best-fit model and summarized results through boxplots (Fig. S9). Finally, to test the effect of grouping mixed hummingbird-insect pollinated plants with vertebrate-pollinated plants (see above) to produce a binary response variable, we repeated the analyses comparing insect (n = 1263) or vertebrate (n = 845) versus mixed insect-hummingbird (n = 124) pollination in turn (Table S8, Fig. S10).
As our results showed marked differences in elevational patterns between (sub-)tropical and temperate zones, we split the original dataset into widely circumscribed (including subtropics) ‘tropics’ (-30° - +30° latitude; tropics are defined +/- 23.46°), ‘temperate North’ (> 30° latitude) and ‘temperate South’ (< -30° latitude). For each partition, we used the approach outlined above to determine the best-fit model and estimate the effects of mean annual temperature, precipitation and cloud cover on pollination mode.
While we accounted for some phylogenetic structure in our GLMMs by specifying the study groups as random effects, we additionally ran phylogenetic generalized linear mixed models for more accurate assessment of phylogenetic non-independence of data points. To this end, we downloaded the angiosperm-wide phylogeny provided by Zanne et al. (2014) and Smith & Brown (2018) through Jin & Qian (2019, V.PhyloMaker) and pruned it to the species included in our sample. Approximately 41% of our study species (932) were covered by this phylogeny, with even sampling across the distribution range and pollination strategies (472 insect-pollinated species, 460 vertebrate-pollinated; Fig. S4, Table S2). On this subset, we built binary phylogenetic GLMMs including elevation, latitude and cloud cover under a Brownian motion model of trait evolution (binaryPGLMM-function in ape, Paradis & Schliep 2019). We refrained from Bayesian fitting to avoid phylogenetic variance getting trapped around zero (a common problem in binomial GLMMs, Paradis & Schliep 2019). Since cloud cover did not show significant effects, we reduced the model to only include an interaction between elevation and latitude (Table S9).
Results
Contrasting patterns of pollinator turnover across latitudes
Our analysis of pollination modes across the final set of 2232 taxa belonging to 26 clades from 22 families showed marked variation in associations with elevation across latitudes. Considering the five categories (bee (n=1141), insect (n=122), hummingbirds (n=758), mixed hummingbird-insect systems (n=124), other vertebrate pollinators (n=87)), we found that species with hummingbird, mixed or other vertebrate pollination become more abundant at higher elevations in the Tropics (above ca. 1500m) but not in temperate mountains (Fig. S3, S4). Instead, in temperate regions, in our study clades, vertebrate pollination dominates at lower elevations (below 1000m in the southern hemisphere, below 1300m in the northern hemisphere). The bee- and insect-pollinated species show a contrasting pattern, most abundant at lower elevations in the Tropics (below1000m) and at higher elevations in the temperate zone (ca. 1000-2000m). These results suggest that Cruden’s (1972) hypothesis on a turnover of insect and vertebrate pollination at high elevations holds only for tropical lineages.
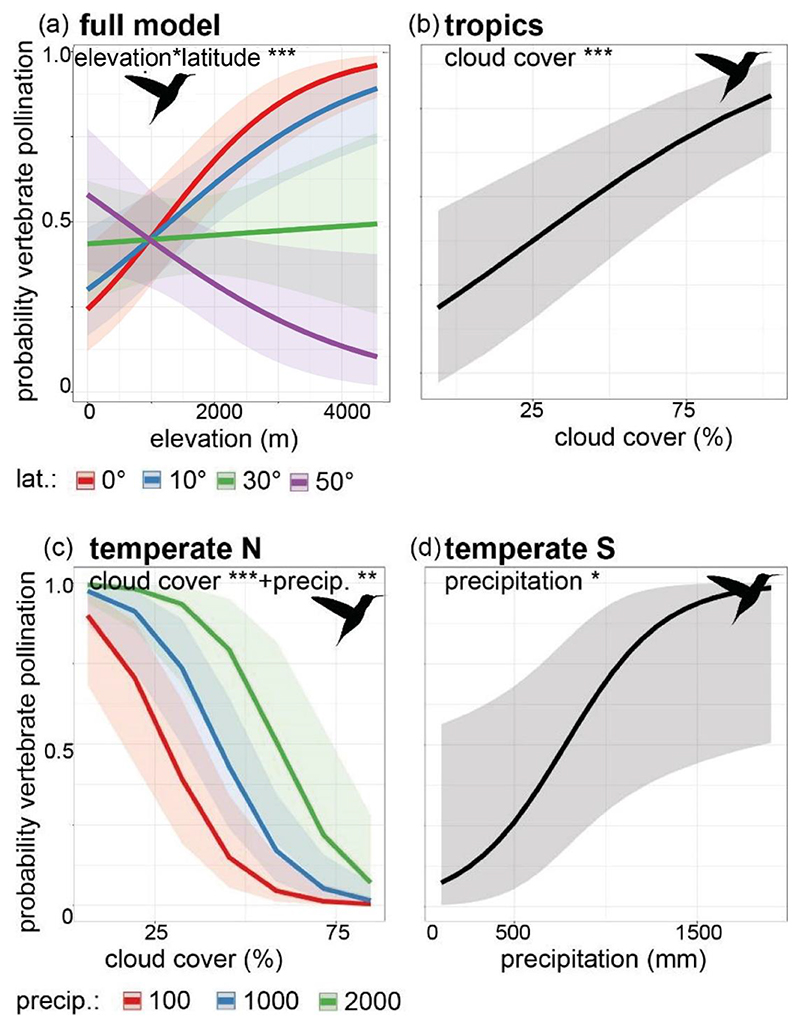
We further explored this contrast using GLMMs, and given the similarity of the curves for all insect and all vertebrate/mixed pollinated plants (Fig. S3), we grouped the bee and generalized insect-pollinated species into one insect category, and the remaining partly or fully vertebrate-pollinated species into one vertebrate category. Using insect pollination as the reference level, we found that both elevation and latitude, but not cloud cover, had strong and significant effects on vertebrate pollination (likelihood-ratio 21.4, estimate -0.03, z-value –4.679, p < 0.001, AUC 0.806, Table 1, Fig. 2, Table S4, Fig. S5). We recovered the same model in our sensitivity analyses, when randomly selecting 25% of the data (96 of 100 replicates showed p < 0.01 effects of elevation*latitude, Table S5, S6), when resampling the dataset to include one randomly picked occurrence point per species (all 100 runs showed p < 0.01 effects of elevation*latitude, Fig. S9), and when accounting for phylogenetic relatedness (Table S9, Fig. S11). Moreover, reducing the dataset to only taxa with empirical observations (n=616) resulted in a best-fit model with the same significant interaction between latitude and elevation as the full model (likelilhood-ratio 14.42, estimate -0.03, z-value -3.703, p < 0.001, AUC 0.82, Table S7). In this subset, the pattern of vertebrate-pollinated species occurring at higher elevations than insect-pollinated species in the tropics is well-supported (Fig. S7), but elevational differences between the two pollination modes are not clear in the temperate zones (note the wide confidence intervals for the slopes, Fig. S7, S8).
Table 1. Best-fit GLMMs on the effect of elevation, latitude and bioclimatic variables on pollination mode.
Models on the full dataset and the tropics included random slopes and intercepts with elevation and temperature, respectively, for the study clades; see Table S4 for details on model selection, insect pollination was used as reference level, significant factors are highlighted in bold.
| (Intercept) | -0.05157 | 0.29089 | -0.177 | 0.859 |
| elevation | 0.86595 | 0.19050 | 4.546 | <0.001 |
| abs(latitude) | -0.00498 | 0.00624 | -0.798 | 0.425 |
| elevation:abs(latitude) | -0.02729 | 0.00583 | -4.679 | <0.001 |
| tropics | ||||
| (Intercept) | 0.94678 | 0.33336 | 2.840 | 0.00451 |
| cloud cover | 0.31511 | 0.06725 | 4.685 | <0.001 |
| temperate North | ||||
| (Intercept) | -0.9452 | 0.4313 | -2.192 | 0.02841 |
| precipitation | 0.5996 | 0.2051 | 2.923 | <0.01 |
| cloud cover | -13.084 | 0.2418 | -5.412 | <0.001 |
| temperate South | ||||
| (Intercept) | 0.1035 | 0.7781 | 0.133 | 0.8942 |
| precipitation | 14.595 | 0.6877 | 2.122 | 0.0338 |
Figure 2. Relation between pollination mode, elevation and different bioclimatic variables across latitudes.
(a) Marked differentiation in the distribution of insect- and vertebrate-pollinated species across latitudes, with vertebrate-pollinated species occurring at higher elevations than insect-pollinated species in the tropics, but not in temperate regions. (b) Vertebrate-pollinated species associate with lower mean annual temperatures in tropical regions than insect-pollinated species, but with slightly warmer conditions in temperate zones. (c) Variable relationships among pollination strategies and mean annual precipitation, with bee-pollinated species in our dataset occurring under slightly wetter conditions than vertebrate-pollinated species around the equator. (d) Vertebrate-pollinated species occur in areas with higher cloud cover than bee-pollinated species in the temperate South and the tropics.
We also examined whether these patterns of pollination mode varying with latitude and elevation were apparent in the 26 individual clades (used as random effects in our GLMM). Indeed, 25 of the 26 clades show the pattern of having a higher probability of vertebrate pollination at high elevations in the tropics, and at low elevations at higher latitudes (note the crossing red and purple lines in Fig. S6). A notable exception is found in the tropical Centropogonids (Campanulaceae), where insect-pollinated species (genus Lysipomia) commonly occur on tropical mountain summits (Páramo) above vertebrate-pollinated relatives.
Finally, we returned to consider the mixed insect-hummingbird category and whether it indeed more closely follows the pattern recovered for vertebrate-pollinated plants, as suggested by visual inspection (Fig. S3). We repeated the GLMM analyses, first contrasting only the insect vs. mixed taxa, and even with vertebrate pollination only represented in the mixed taxa, we recovered the same effects of elevation and latitude (estimate -0.037, likelihood-ratio 21.8, z-value -4.614, p < 0.001, AUC 0.845, removing the elevation*latitude interaction increases the AIC from 701 to 721, Table S8; see also Fig. S10a,b). By contrast, models with elevation are not among the top models when we compare mixed vs. vertebrate-pollinated taxa (Table S8), and we observe no differences across elevation (best fit model including latitude only: estimate -0.987, likelihood-ratio 7.22, z-value -2.337, p < 0.001; Fig. S10c). Together, these analyses support the observation that the mixed insect-vertebrate-pollinated species more closely track the patterns of the vertebrate-pollinated taxa, and thus that lineages that transition to high-elevation habitats in the tropics may benefit from some, if not exclusive, vertebrate pollination.
Cloud cover and moist conditions predict vertebrate pollination across latitudes
Given that Cruden’s (1972) hypothesis was based on associations of pollinator activity with climatic conditions, we next examined possible environmental drivers of pollinator turnover, focusing on mean annual temperature, precipitation and cloud cover (Fig. 2b-d). To avoid confounding effects due to varying patterns across latitudes (Fig. 2a), we split the dataset into tropical (grossly defined as +/- 30°, hence including subtropics) and temperate zones (North and South). Temperature was not significant in any of the models for either data partition and never retained as fixed effect in the best-fit models (Table 1, S4). However, precipitation emerged as a significant predictor in both the tropics and southern temperate regions, with vertebrate pollination associated with higher precipitation. We also recovered an association of high cloud cover with vertebrate pollination in the tropics and the northern temperate zone (Table 1, Figs. 3, S12).
Figure 3. Results of the best-fit GLMM models investigating the effects of different bioclimatic variables across latitudes.
(a) The probability of vertebrate-pollination increases significantly across elevation in the tropics (0°, +/- 10° latitude) while it gradually decreases towards higher latitudes (+/- 30°, +/- 50°). (b) The probability of vertebrate-pollination increases significantly with increased cloud cover in the tropics (i.e. montane cloud forests, n = 1917). (c) In the temperate zones of the Northern hemisphere, the probability of vertebrate pollination decreased significantly with increasing cloud cover, but less so when precipitation was high (n = 265). (d) In the temperate zones of the Southern hemisphere, the probability of vertebrate pollination increased with increasing precipitation (n = 50). Insect pollination was used as reference level in the binomial GLMMs (and is hence not depicted), 95% confidence intervals are given, *** p < 0.001, ** p < 0.01, * p < 0.05.
In addition, since climatic factors are associated with particular biomes (Whittaker 1975), we tested whether insect and vertebrate pollination also show an association with biome. We found that indeed, insect- and vertebrate-pollinated species differ significantly in biome occupation (X2 = 149.81, df = 7, p-value < 0.001, Table S3, Fig. 1c,d). While both pollination strategies were found in each biome (except Tundra), vertebrate-pollinated species were significantly more common in temperate seasonal forests and temperate rainforests, where temperature and precipitation are intermediate (e.g. 10°C-20°C, Fig. 1, Table S3). Insect-pollinated species occurred significantly more often under warmer conditions in tropical seasonal forests/savannas, and under both cooler and drier conditions in wood-/shrublands, boreal forests and temperate grasslands (Fig. 1, Table S3).
Discussion
Altitude-driven pollinator turnover across the Americas
Cruden (1972) hypothesized that bird pollination is advantageous for flowering plants occurring in the often cold and rainy high elevation habitats. Our study found that this hypothesis holds in the (sub)tropics (i.e. between ca. 30° N and 30°S latitude), but not in temperate regions, where insects are more important pollinators also at high elevations. Although our dataset is largely comprised of bee- and hummingbird-pollinated species (51% and 34% respectively), the overall pattern holds for other types of pollinators (Fig. S3, S4). Temperature is tightly correlated with elevation (Fig. 2a, b, S2) and decreases at higher elevations globally, a phenomenon known as the elevational air temperature gradient (Blüthgen 1980). Not surprisingly, temperature was a poor predictor of the importance of vertebrate vs. insect pollination across latitudes. Indeed, insect pollinators as a group are not restricted from high elevations simply based on thermal constraints due to smaller body size and physiology, since bumblebees can forage in near freezing temperatures (Bruggemann 1958, Oyen & Dillon 2018) and thus are effective high elevation pollinators (McCallum et al. 2013, Woodard 2017). Similarly, flies use sun-basking to elevate their body temperature and are important high-elevation pollinators in many ecosystems (González et al. 2009, McCabe & Cobbs 2021, Pelayo et al. 2021).
We found that indicators of mesic vs. arid conditions (mean annual precipitation and cloud cover) were predictors of the importance of vertebrates vs. insects, even across latitudes (Table 1). Consistent with the notion that cloudy and rainy weather deters insect pollinators (Cruden 1972, Kearns 1992, Michener 2000), we found that vertebrate pollination was positively associated with higher precipitation in temperate zones (Fig. 3c,d), and with higher cloud cover in both the tropics and temperate north (Fig. 3b,c). Birds, in particular, are able to forage in wet weather (Lawson & Rands 2019), although bats may have reduced foraging capabilities in rain (Voigt et al. 2011). This strong relationship between pollination mode and precipitation has been recovered in previous smaller-scale studies, where precipitation was negatively correlated with insect pollination, but positively correlated with vertebrate pollination (Cruden 1972, Devoto et al. 2005, González et al. 2009, Dalsgaard et al. 2009, Chalcoff et al. 2012). In accordance with this, our biome analysis suggested that vertebrate pollination is important in cool mesic and wet forests (e.g. cloud forests), while insect pollination is important in dry forests and savannas (Fig.1c,d). Insect pollination in moist biomes appears constrained to warmer conditions (Fig. 1c). Together, these results suggest that – across latitudes – shady, moist, forested ecosystems are key habitats for endothermic vertebrate pollinators (‘hummingbird habitats’, Stebbins, 1989).
Evolutionary history, pollinator niche saturation and seasonality
The associations between pollinator importance and elevation must be considered in light of the evolutionary history and ecological context of the tropics vs. temperate regions. Tropical montane cloud forests feature abundant ‘hummingbird habitats’, as evidenced by notable radiations of several hummingbird lineages in tropical mountains (McGuire et al. 2014). By contrast, hummingbirds have not diversified extensively in tropical lowlands, and only recently (5 mya) have diversified in temperate zones, resulting in ca. 300 hummingbird species in the tropics, and only about 20 in temperate zones (McGuire et al. 2014, Abrahamczyk & Renner 2015). This pattern may suggest that the strong relation between tropical mountains and vertebrate pollination is purely an artifact of the evolutionary radiation of hummingbirds in the cloud forest environment. Even if true, this conclusion would underscore how certain environmental conditions – the abundant hummingbird habitats in tropical cloud forests – favor a particular pollinator group, and result in repeated evolutionary pollinator shifts across plant clades that have colonized this environment (Thomson & Wilson 2008, Lagomarsino et al. 2016, Dellinger et al. 2021, Hamilton & Wessinger 2022).
A similar case can be made for the importance of bumblebee pollination at high elevation in temperate regions. The temperate mountains included in our study (mostly southern Andes, Rocky Mountains) feature habitats that are suitable for species-rich bumblebee pollinator communities, including dry, cool sparse forests or open grassland (Rundel 1994, McCabe & Cobbs 2021). Bumblebees appear to successfully occupy the available pollination niche space in these habitats (Pyke et al. 2011). By contrast, tropical mountains included here are mostly wet, and comparatively few large, cold-adapted bee species (e.g., from the genera Bombus, Centris, Eufriesia) reach the montane cloud forest or Páramo zones (Roubik 1989, Gonzalez & Engel 2004, Perillo et al. 2021). The abundance and diversity of cold-adapted bees in tropical mountains may further be limited by competition for pollination niche space with abundant vertebrate pollinators (Temeles et al. 2016). Such competitive interactions, along with differences in abiotic tolerances, have been suggested as structuring the distribution of moth and hummingbird pollination (Cruden 1976), and should be considered for a full description of the drivers of broad-scale patterns of plant-pollinator interactions.
Finally, latitudinal differences in seasonality likely contribute to the dominance of vertebrate pollinators in tropical, but not temperate mountains. While tropical forests do present some seasonal variation, floral nectar resources are generally available year-round, and hummingbirds may track resources through small-scale (altitudinal) migration (Abrahamczyk et al. 2011, Maglianesi et al. 2015). By contrast, temperate zones are characterized by marked seasonal variation in nectar resources, due to constrained flowering periods that are defined by snow melt at high elevations and summer drought at lower elevations (Inouye & Wielgolaski 2013). During their short summer breeding seasons, the long-distance migratory temperate hummingbirds require nectar resources to be abundant and near to nesting sites in trees (Grant & Grant 1968, Stebbins 1989, McKinney et al. 2012). Truncated flowering periods and limited nesting sites at high elevations (above the treeline) possibly limit hummingbird-pollinated plants to lower elevations in temperate zones.
Parallel trends outside the Americas
While we focused on the ecogeographic patterns in the Americas because of the many temperate and tropical plant clades with bee-bird pollinator shifts, the broader findings may extend in other parts of the world. For example, pollination activities by insects, but not birds, are reduced during rainy and cloudy days in a lowland evergreen forest in South China (Sun et al. 2017), underscoring that continued flower visitation during rain is not a singularity of hummingbirds. Moreover, vertebrate pollination has increasing importance during the rainy season in tropical African mountains (i.e., Janeček et al. 2015, Klomberg et al. 2022). Seasonality seems equally relevant in explaining flowering of (generalist) nectar-feeding bird-adapted plants in temperate Asia and Australia (Ford et al. 1979, Funamoto 2019) and rodent-pollinated plants in South Africa (Wiens 1983). In both cases, vertebrate-adapted plants tend to flower primarily during rainy and cool winter months (Chen et al. 2019), underscoring the increased importance of vertebrate pollination under wet conditions.
Patterns of pollinator turnover across elevations are less clear (Abrahamczyk 2019), but due in large part to a paucity of studies. For example, pollinator interaction networks from Mount Cameroon (5.7° N) suggest continued importance of insect pollinators across elevation (i.e., butterflies, Mertens et al. 2021; bees, Klomberg et al. 2022) while another study from Mt. Kilimanjaro (3° S) shows a decline with elevation (bees, Classen et al. 2015). Importantly, bumblebees are absent from sub-Saharan Africa and Australia, potentially freeing niche space at high elevations (e.g., for other bees, moths, beetles; Adedoja et al. 2020), while they are important pollinators in the Oriental region (i.e., India, tropical China, Southeast Asia, Corlett 2004) and mountains of temperate East Asia (Funamoto 2019, Paudel et al. 2019). Further, studies of specific plant genera suggest a turnover from insect to vertebrate pollination with increasing elevation in African and Asian tropical mountains (i.e., Mucuna, Kobayashi et al. 2021), some in plant clades that otherwise feature abundant insect pollination (i.e., Melastomataceae, Dellinger et al., 2022). Similar to our study, the pattern may potentially be reversed outside of the tropics, for example in temperate Himalayan Rhododendron, where bird-pollinated species reportedly occur in moist montane forests, while bumblebee-pollinated species are found in the higher elevation Alpine zone (Basnett et al. 2019).
Conclusions and future directions
Building on decades of comparative pollination studies across North, Central, and South America, we uncovered consistent patterns showing that both Cruden (1972) and Stebbins (1989) were right. In the tropics, where vertebrate pollinators are most diverse, they serve as the most important high elevation pollinators in our study clades, providing service in the rainy conditions that deter insect pollinators (Cruden 1972). However, in temperate regions, vertebrate pollination is more common at low to intermediate elevations, where those moist conditions are more common (e.g. wooded ravines, Stebbins 1989), leaving insects (especially large-bodied bees and flies) as the more important pollinators in dry montane habitats. These patterns highlight the importance of pollinator physiology as driving the turnover in plant-pollinator interactions along climatic gradients (Arroyo et al. 1982, Warren et al. 1988, Lefevbre et al. 2019, McCabe & Cobb 2021).
This study also points to several fruitful areas for future studies that seek to understand ecogeographic variation in pollination systems. First, we categorized species by the major pollinator in order to capture broad differences in the distribution of vertebrate and insect pollination. However, these relationships are naturally continuous (with each pollinator species varying in importance, and across space and time, Mayfield et al. 2001). Delving into this continuous variation, while not possible at the scale of thousands of species, would give greater insight into the climatic thresholds where turnovers in pollination systems occur. Our analyses of the mixed insect-hummingbird-pollinated species hint at this more complex dynamic, as they actually peak at a higher elevation than primarily bird-pollinated species (Fig. S3). This result is consistent with the idea that a more generalized system provides a successful “back-up” strategy in the harshest environments (Bergamo et al. 2021). Examining the degree of generalization (or specialization) would be a welcome complement to our study, and depending on the thermal tolerance of different pollinator species, may present relationships with temperature that did not emerge from our study at the level of entire functional groups. Further, evolutionary pollinator shifts can clearly occur without species moving to a new/different abiotic environment (Hamilton & Wessinger 2022), exemplified by several clades featuring repeated pollinator shifts also in lowland rainforest environments (e.g. Costus, Vargas et al. 2020, Kay & Grossenbacher 2022). In parallel, plant clades may retain the same functional pollinator group even across ecogeographic gradients, or be restricted to narrow elevational ranges (and were hence excluded from our study since they did not meet our selection criteria). Including such clades, and detailed pollinator and trait data, in future studies would allow tackling intriguing questions such as what variables (i.e., competition for/among pollinators, phenological mismatch, co-flowering, flower developmental constraints) may drive or hinder evolutionary pollinator shifts in addition to or irrespective of the abiotic environment. Using community-level data would further allow for more complete tests of Cruden’s hypotheses and explore whether, even at large biogeographic scales, the relative abundance of insect- and vertebrate-pollinated plants changes in correlation with climtic factors.
Finally, although our sampling is largely based on clades with hummingbird and bee pollination spanning wide elevational ranges, we predict that the broad associations between wet forests and vertebrate pollination will hold beyond our study clades from American mountains. Support for this hypothesis comes from e.g., the intriguing absence of hummingbird-pollinated plants in the Patagonian Steppe (but high abundance in temperate Southern forests, Armesto et al. 1996, Devoto et al. 2005) and singular observations of vertebrate pollination in otherwise insect-pollinated lineages in mountains/wet seasons in the Paleotropics (Janeček et al. 2015, Funamoto 2019). Conducting studies similar to ours at a global scale, or including community-level data, will be necessary, however, to begin to partition the relative importance of abiotic and biotic factors in driving pollinator shifts and structuring broad-scale distribution patterns of plant-pollinator interactions.
Supplementary Material
Acknowledgements
We thank the labs of Stacey Smith and Erin Manzitto-Tripp for inspiring discussions on this topic and helpful feedback on the project. We particularly thank Miranda Sinnott-Armstrong for support with and recommendations for data analyses. We further thank Andreas Berger for sharing data on pollination in Rubiaceae with us. This work was supported by FWF-grant T-1186 to ASD, NSF DEB-2052904 to CAW, and NSF-1553114 to SDS.
Footnotes
Author Contribution
All authors conceived the study, ASD and AH compiled the species datasets, ASD ran the analyses and drafted the manuscript, AH, CW and SDS contributed to improving and revising the manuscript.
Data and Code Availability Statement
All data analyzed in the manuscript and exemplary R code for analyses have been deposited in Dryad (10.5061/dryad.bcc2fqzfg).
References
- Abrahamczyk S. Comparison of the ecology and evolution of plants with a generalist bird pollination system between continents and islands worldwide. Biological Reviews. 2019;94:1658–1671. doi: 10.1111/brv.12520. [DOI] [PubMed] [Google Scholar]
- Abrahamczyk S, Renner SS. The temporal build-up of hummingbird/plant mutualisms in North America and temperate South America. BMC Evol Biol. 2015;15:104. doi: 10.1186/s12862-015-0388-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abrahamczyk S, Kluge J, Gareca Y, Reichle S, Kessler M. The influence of climatic seasonality on the diversity of different tropical pollinator groups. Plos One. 2011;6:e27115. doi: 10.1371/journal.pone.0027115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adedoja O, Kehinde T, Samways MJ. Asynchrony among insect pollinator groups and flowering plants with elevation. Scientific Reports. 2020;10:13268. doi: 10.1038/s41598-020-70055-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amatulli G, Domisch S, Tuanmu M-N, Parmentier B, Ranipeta A, Malczyk M, Jetz W. A Suite of Global, Cross-Scale Topographic Variables for Environmental and Biodiversity Modeling. Scientific Data. 2018;5:180040. doi: 10.1038/sdata.2018.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armesto JJ, Smith-Ramírez C, Sabag C. High-latitude rainforests and associated ecosystems of the west coast of the Americas. Springer; New York, NY: 1996. The importance of plant-bird mutualisms in the temperate rainforest of southern South America; pp. 248–265. [Google Scholar]
- Arroyo MTK, Primack R, Armesto J. Community studies in pollination ecology in the high temperate Andes of central Chile. I. Pollination mechanisms and altitudinal variation. American Journal of Botany. 1982;69:82–97. [Google Scholar]
- Basnett S, Ganesan R, Devy SM. Floral triats determine pollinator visitation in Rhododendron species across an elevation gradient in the Sikkim Himalaya. Alpine Botany. 2019 [Google Scholar]
- Bates D, Mullen KM, Nash JC, Varadhan R. minqa: Derivative-free optimization algorithms by quadratic approximation. R package version 124. 2014
- Bates D, Maechler M, Bolker B, Walker S. Fitting Linear Mixed-Effects Models Using lme4. Journal of Statistical Software. 2015;67(1):1–48. [Google Scholar]
- Bergamo PJ, Streher NS, Zambon V, Wolowski M, Sazima M. Pollination generalization and reproductive assurance by selfing in a tropical montane ecosystem. Naturwissenschaften. 2021;108:50. doi: 10.1007/s00114-021-01764-8. [DOI] [PubMed] [Google Scholar]
- Bivand R, Lewin-Koh N. maptools: Tools for Handling Spatial Objects. R package version 11-1. 2021.
- Bjornstad ON. ncf: Spatial Covariance Functions. R package version 13-2. 2022. https://CRAN.R-project.org/package=ncf .
- Blüthgen J. Allgemeine Klimageographie. Berlin: de Gruyter; 1980. [Google Scholar]
- Bruggemann PF. Insects and environments of the high arctic; Proceedings of the 10th International Congress of Entomology; Montreal, Canada: 1958. [Google Scholar]
- Cayuela L, Macarro I, Stein A, Oksanen J. Taxonstand: Taxonomic Standardization of Plant Species Names. R package version 24. 2021 [Google Scholar]
- Chalcoff VR, Aizen MA, Ezcurra C. Erosion of a pollination mutualism along an environmental gradient in a south Andean treelet, Embothrium coccineum (Proteaceae) Oikos. 2012;121:471–480. [Google Scholar]
- Chamberlain S, Barve V, Mcglinn D, Oldoni D, Desmet P, Geffert L, Ram K. rgbif: Interface to the Global Biodiversity Information Facility API. R package version 352. 2021 [Google Scholar]
- Chen K-H, Lu J-Y, Wang C-N. Effective pollination of Aeschynanthus acuminatus (Gesneriaceae) by generalist passerines, in sunbird-absent East Asia. Scientific Reports. 2019;9:17552. doi: 10.1038/s41598-019-53035-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Classen A, Peters MK, Kindeketa WJ, Appelhans T, Eardley CD, Gikungu MW, Hemp A, Nauss T, Steffan-Dewenter I. Temperature versus resource constraints: which factors determine bee diversity on Mount Kilimanjaro, Tanzania? Global Ecology and Biogeography. 2015;24:642–652. [Google Scholar]
- Colwell RK, Gotelli NJ, Ashton LA, Beck J, Brehm G, Fayle TM, Fiedler K, Forister ML, Kessler M, Kitching RL, Klimes P, et al. Midpoint attractors and species richness: Modelling the interaction between environmental drivers and geometric constraints. Ecology Letters. 2016;19:1009–1022. doi: 10.1111/ele.12640. [DOI] [PubMed] [Google Scholar]
- Corlett RT. Flower visitors and pollination in the Oriental (Indomalayan) Region. Biol Rev. 2004;79:497–532. doi: 10.1017/s1464793103006341. [DOI] [PubMed] [Google Scholar]
- Cozien RJ, van der Niet T, Johnson SD, Steenhuisen SL. Saurian surprise. Ecology. 2019;100:1–4. doi: 10.1002/ecy.2670. [DOI] [PubMed] [Google Scholar]
- Cronk Q, Ojeda I. Bird-pollinated flowers in an evolutionary and molecular context. Journal of Experimental Botany. 2008;59:715–727. doi: 10.1093/jxb/ern009. [DOI] [PubMed] [Google Scholar]
- Cruden RW. Pollinators in high elevation ecosystems: relative effectiveness of birds and bees. Science. 1972;176:1439–1440. doi: 10.1126/science.176.4042.1439. [DOI] [PubMed] [Google Scholar]
- Cruden RW, Kinsman S, Stockhouse RE, II, Linhart YB. Pollination, fecundity, and the distribution of moth-flowered plants. Biotropica. 1976;8:204–210. [Google Scholar]
- Dalsgaard B, Martín González AM, Olesen JM, Ollerton J, Timmermann A, Andersen LH, Tossas AG. Plant-hummingbird interactions in the West Indies: floral specialization gradients associated with environment and hummingbird size. Oecologia. 2009;159:757–766. doi: 10.1007/s00442-008-1255-z. [DOI] [PubMed] [Google Scholar]
- Dellinger AS, Pérez-Barrales R, Michelangeli FA, Penneys DS, Fernández-Fernández DM, Schönenberger J. Low bee visitation rates explain pollinator shifts to vertebrates in tropical mountains. New Phyt. 2021;231:864–877. doi: 10.1111/nph.17390. [DOI] [PubMed] [Google Scholar]
- Dellinger AS, Scheer LM, Artuso S, Fernández-Fernández DM, Sornoza F, Penneys DS, Schönenberger J, et al. Bimodal pollination systems in Andean Melastomataceae involving birds, bats and rodents. Am Nat. 2019;194:104–116. doi: 10.1086/703517. [DOI] [PubMed] [Google Scholar]
- Devoto M, Medan D, Montaldo NH. Patterns of interaction between plants and pollinators along an environmental gradient. Oikos. 2005;109:461–472. [Google Scholar]
- Fleming TH, Muchhala N. Nectar-feeding bird and bat niches in two worlds: pantropical comparisons of vertebrate pollination systems. Journal of Biogeography. 2008;35:764–780. [Google Scholar]
- Ford HA, Paton DC, Forde N. Birds as pollinators of Australian plants. New Zealand Journal of Botany. 1979;17:509–519. [Google Scholar]
- Fox J, Weisberg S. Visualizing Fit and Lack of Fit in Complex Regression Models with Predictor Effect Plots and Partial Residuals. Journal of Statistical Software. 2018;87(9):1–27. [Google Scholar]
- González AMM, Dalsgaard B, Ollerton J, Timmermann A, Olesen JM, Andersen L, Tossas AG. Effects of climate on pollination networks in the West Indies. Journal of Tropical Ecology. 2009;25:493–506. [Google Scholar]
- Gonzalez VH, Engel MS. The tropical Andean bee fauna (Insecta: Hymenoptera. Apoidea), with examples from Colombia. Entomologische Abhandlungen. 2004;62:65–75. [Google Scholar]
- Grant KA, Grant V. Hummingbirds and their flowers. Columbia University Press; New York: 1968. [Google Scholar]
- Heinrich B. Thermoregulation in endothermic insects. Science. 1974;185:747–756. doi: 10.1126/science.185.4153.747. [DOI] [PubMed] [Google Scholar]
- Hijmans RJ. WorldClim 2: new 1km spatial resolution climate surfaces for global land areas. International Journal of Climatology. 2017;37:4302–4315. [Google Scholar]
- Hijmans RJ. raster: Geographic Data Analysis and Modeling. R package version 34-10. 2021.
- Inouye DW, Wielgolaski FE. In: Phenology: An Integrative Environmental Science. Schwartz M, editor. Springer; Dordrecht: 2013. Phenology at high altitudes; pp. 249–272. [Google Scholar]
- Janeček Š, Bartoš M, Njabo KY. Convergent evolution of sunbird pollination systems of Impatiens species in tropical Africa and hummingbird systems of the New World. Biol J Linn Soc. 2015;115:127–133. [Google Scholar]
- Jin Y, Hong Q. V PhyloMaker: an R package that can generate very large phylogenies for vascular plants. Ecography. 2019;42:1353–1359. [Google Scholar]
- Kay KM, Grossenbacher DL. Evolutionary convergence on hummingbird pollination in Neotropical Costus provides insight into the causes of pollinator shifts. New Phytologist. 2022;236:1572–1583. doi: 10.1111/nph.18464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kearns CA. Anthophilous fly distribution across an elevational gradient. The American Midland Naturalist. 1992;127:172–182. [Google Scholar]
- Klomberg Y, Tropek R, Mertens JEJ, Kobe IN, Hodeček J, Raška J, Fominka NT, Souto-Vilarós D, Janecková P, Janeček S. Spatiotemporal variation in the role of floral traits in shaping tropical plant-pollinator interactions. Ecology Letters. 2022 doi: 10.1111/ele.13958. [DOI] [PubMed] [Google Scholar]
- Kobayashi S, Panha S, Seesamut T, Nantarat N, Likhitrakarn N, Denda T, Izawa M. First record of non-flying mammalian contributors to pollination in a tropical montane forest in Asia. Ecology and Evolution. 2021;11:17604–17608. doi: 10.1002/ece3.8361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krömer T, Kessler M, Herzog SK. Distribution and flowering ecology of bromeliads along two climatically contrasting elevational transects in the Bolivian Andes. Biotropica. 2006;38:183–195. [Google Scholar]
- Lagomarsino LP, Condamine FL, Antonelli A, Mulch A, Davis CC. The abiotic and biotic drivers of rapid diversification in Andean bellflowers (Campanulaceae) New Phytol. 2016;210:1430–1442. doi: 10.1111/nph.13920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson DA, Rands SA. The effects of rainfall on plant-pollinator interactions. Arthropod-Plant Interactions. 2019;13:561–569. [Google Scholar]
- Lefevbre V, Villemant C, Fontaine C, Daugeron C. Altitudinal, temporal and trophic partitioning of flower-visitors in Alpine communities. Scientific Reports. 2018;8:4706. doi: 10.1038/s41598-018-23210-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lüdecke D. sjPlot: Data Visualization for Statistics in Social Science. R package version 289. 2021.
- Maglianesi MA, Blüthgen N, Böhning-Gaese K, Schleuning M. Functional structure and specialization in three tropical plant-hummingbird interaction networks across an elevational gradient in Costa Rica. Ecography. 2015;38:1119–1128. [Google Scholar]
- Mayfield MM, Waser NM, Price MV. Exploring the ‘Most Effective Pollinator Principle’ with complex flowers: bumblebees and Ipomopsis aggregata. AoB. 2001;88:591–596. [Google Scholar]
- McCabe LM, Cobb NS. From bees to flies: global shifts in pollinator community along elevational gradients. Front Ecol Evol. 2021;8:626124 [Google Scholar]
- McCallum KP, McDougall FO, Seymour RS. A review of the energetics of pollination biology. J Comp Physiol B. 2013;183:867–876. doi: 10.1007/s00360-013-0760-5. [DOI] [PubMed] [Google Scholar]
- McGuire JA, Witt CC, Remsen JV, Jr, Corl A, Rabosky DL, Altshuler DL, Dudley R. Molecular phylogenetics and the diversification of hummingbirds. Current Biology. 2004;24:910–916. doi: 10.1016/j.cub.2014.03.016. [DOI] [PubMed] [Google Scholar]
- McKinney AM, CaraDonna PJ, Inouye DW, Barr B, Bertelsen CD, Waser NM. Asynchronous changes in phenology of migrating Broad-tailed hummingbirds and their early-season nectar resources. Ecology. 2012;93:1987–1993. doi: 10.1890/12-0255.1. [DOI] [PubMed] [Google Scholar]
- Mertens JEJ, Brisson L, Janeček S, Klomberg Y, Maicher V, Sáfián S, Delabye S, Potocký P, Kobe IN, Pyrcz T, Tropek R. Elevational and seasonal patterns of butterflies and hawkmoths in plant-pollinator networks in tropical rainforests of Mount Cameroon. Scientific Reports. 2021;11:9710. doi: 10.1038/s41598-021-89012-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michener CD. The bees of the World. John Hopkins University Press; Baltimore: 2000. p. 913. [Google Scholar]
- Miller RB. Hawkmoths and the geographic patterns of floral variation in Aqueligia caerulea. Evolution. 1981;35:763–774. doi: 10.1111/j.1558-5646.1981.tb04936.x. [DOI] [PubMed] [Google Scholar]
- Ollerton J. Pollinator Diversity: Distribution, Ecological Function, and Conservation. Ann Rev Ecol, Evol, Syst. 2017;48:353–376. [Google Scholar]
- Orr MC, Hughes AC, Chesters D, Pichering J, Zhu C-D, Ascher JS. Global patterns and drivers of bee distribution. Current Biology. 2021;31:451–458. doi: 10.1016/j.cub.2020.10.053. [DOI] [PubMed] [Google Scholar]
- Oyen KJ, Dillon ME. Critical thermal limits of bumblebees (Bombus impatiens) are marked by stereotypical behaviors and are unchanged by acclimation, age or feeding status. J Exp Biol. 2018;221:jeb165589. doi: 10.1242/jeb.165589. [DOI] [PubMed] [Google Scholar]
- Paradis E, Schliep K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics. 2019;35:526–528. doi: 10.1093/bioinformatics/bty633. [DOI] [PubMed] [Google Scholar]
- Paudel BR, Kessler A, Shrestha M, Zhao JL, Li QJ. Geographic isolation, pollination syndromes, and pollinator generalization in Himalayan Roscoea spp. (Zingiberaceae) Ecosphere. 2019;10:e02943 [Google Scholar]
- Pelayo RC, Llambi LD, Gámez LE, Barrios YC, Ramírez LA, Torres JE, Cuesta F. Plant phenology dynamics and pollination networks in summits of the high tropical Andes: A baseline for monitoring climate change impacts. Frontiers in Ecology and Evolution. 2021;9:679045 [Google Scholar]
- Perillo LN, Castro FSD, Solar R, Neves FDS. Disentangling the effects of latitudinal and elevational gradients on bee, wasp and ant diversity in an ancient neotropical mountain range. Journal of Biogeography. 2021;48:1564–1578. [Google Scholar]
- Primack R, Inouye D. Factors affecting pollinator visitation rates: a biogeographical comparison. Curr Sci. 1993;63:257–261. [Google Scholar]
- Pyke GH, Inouye DW, Thomson JD. Activity and abundance of bumble bees near Crested Butte, Colorado: diel, seasonal, and elevation effects. Ecological Entomology. 2011;36:511–521. [Google Scholar]
- Ratto F, Simmons BI, Spake R, Zamora-Gutierrez V, MacDonald MA, Merriman JC, Dicks LV, et al. Global importance of vertebrate pollinators for plant reproductive success. A meta-analysis. Front Ecol Env. 2018;16:82–90. [Google Scholar]
- Ricklefs RE. The economy of nature. W. H. Freeman and Company; 2008. Chapter 5 Biological Communities, The biome concept. [Google Scholar]
- Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JC, Müller M. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics. 2011;12:77. doi: 10.1186/1471-2105-12-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roubik D. Cambridge Tropical Biology Series. Cambridge University Press; Cambridge: 1989. Ecology and natural history of tropical bees; p. 514. [DOI] [PubMed] [Google Scholar]
- Rundel PW. In: Tropical Alpine Environments: Plant Form and Function. Rundel WP, Smith AP, Meinzer FC, editors. Cambridge University Press; Cambridge: 1994. Tropical alpine climates; pp. 21–44. [Google Scholar]
- Schemske DW, Mittelbach GG, Cornell HV, Sobel JM, Roy K. Is there a latitudinal gradient in the importance of biotic interactions? Ann Rev Ecol Evol Syst. 2009;40:245–269. [Google Scholar]
- Sinnott-Armstrong MA, Donoghue MJ, Jetz W. Dispersers and environment drive global variation in fruit colour syndromes. Ecology Letters. 2021;24:1387–1399. doi: 10.1111/ele.13753. [DOI] [PubMed] [Google Scholar]
- Smith SA, Brown JW. Constructing a broadly inclusive seed plant phylogeny. American Journal of Botany. 2018;105:302–314. doi: 10.1002/ajb2.1019. [DOI] [PubMed] [Google Scholar]
- Stebbins LG. In: The evolutionary ecology of plants. Beck JH, Linhart YB, editors. CRC Press; 1989. Adaptive shifts toward hummingbird pollination; pp. 39–60. [Google Scholar]
- Stefan V, Levin S. plotbiomes: Plot Whittaker biomes with ggplot2. R package version 0009001. 2021.
- Stiles FG. Ecology, flowering phenology and hummingbird pollination of some Costa Rican Heliconia species. Ecology. 1975;56:285–301. [Google Scholar]
- Sun SG, Huang ZH, Chen ZB, Huang SQ. Nectar properties and the role of sunbirds as pollinators of the golden-flowered tea (Camellia petelotii) AJB. 2017;104:468–476. doi: 10.3732/ajb.1600428. [DOI] [PubMed] [Google Scholar]
- Temeles EJ, Newman JT, Newman JH, Cho SY, Mazzotta AR, Kress WJ. Pollinator competition as a driver of floral divergence: an experimental test. Plos One. 2016;11:e0146431. doi: 10.1371/journal.pone.0146431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson JD, Wilson P. Explaining evolutionary shifts between bee and hummingbird pollination: convergence, divergence, and directionality. Int J Plant Sci. 2008;169:23–38. [Google Scholar]
- Tripp EA, Manos PS. Is floral specialization and evolutionary dead-end? Pollination system transitions in Ruellia (Acanthaceae) Evolution. 2008;62:1712–1737. doi: 10.1111/j.1558-5646.2008.00398.x. [DOI] [PubMed] [Google Scholar]
- Vargas OM, Goldston B, Grossenbacher DL, Kay KM. Patterns of speciation are similar across mountainous and lowland regions for a neotropical plant radiation (Costaceae: Costus) Evolution. 2020;74:2644–2661. doi: 10.1111/evo.14108. [DOI] [PubMed] [Google Scholar]
- Voigt CC, Schneeberger K, Voigt-Heucke SL, Lewanzik D. Rain increases the energy cost of bat flight. Physiology. 2011;7 doi: 10.1098/rsbl.2011.0313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren SD, Harper KT, Booth GM. Elevational distribution of insect pollinators. Am Midl Nat. 1988;120:325–330. [Google Scholar]
- Wei T, Simko V. R package ‘corrplot’: Visualization of a Correlation Matrix (Version 090) 2021.
- Wessinger CA, Rausher MD, Hileman LC. Adaptation to hummingbird pollination is associated with reduced diversification in Penstemon. Evol Lett. 2019;3:521–533. doi: 10.1002/evl3.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whittaker RH. Communities and Ecosystems. 2nd. MacMillan Publishing Co; New York: 1975. [Google Scholar]
- Wickham H. ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag; New York: 2016. [Google Scholar]
- Wiens D, Rourke JP, Casper BB, Rickart EA, Lapine TR, Peterson CJ. Non-flying mammal pollination of southern, African Proteas: a non-coevolved system. Ann Missouri Bot Gard. 1983;70:1–31. [Google Scholar]
- Zanne AE, Tank DC, Cornwell WK, Eastman JM, Smith SA, FitzJohn RG, Beaulieu JM, et al. Three keys to the radiation of angiosperms into freezing environments. Nature. 2014;506:89–92. doi: 10.1038/nature12872. [DOI] [PubMed] [Google Scholar]
- Zizka A, Silvestro D, Andermann T, Azevedo J, Duarte Ritter C, Edler D, Antonelli A, et al. CoordinateCleaner: standardized cleaning of occurrence records from biological collection databases. Methods in Ecology and Evolution, R package version 20-18. 2019 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data analyzed in the manuscript and exemplary R code for analyses have been deposited in Dryad (10.5061/dryad.bcc2fqzfg).