Abstract
Cardiac excitation-contraction coupling (ECC) depends on Ca2+ release from intracellular stores via ryanodine receptors (RyRs) triggered by L-type Ca2+ channels (LCCs). Uncertain numbers of RyRs and LCCs form ‘couplons’ whose activation produces Ca2+ sparks, which summate to form a cell-wide Ca2+ transient that switches on contraction. Voltage (Vm) changes during the action potential (AP) and stochasticity in channel gating should create variability in Ca2+ spark timing, but Ca2+ transient wavefronts have remarkable uniformity. To examine how this is achieved, we measured the Vm-dependence of evoked Ca2+ spark probability (Pspark) and latency over a wide voltage range in rat ventricular cells. With depolarising steps, Ca2+ spark latency showed a U-shaped Vm-dependence, while repolarising steps from 50 mV produced Ca2+ spark latencies that increased monotonically with Vm. A computer model based on reported channel gating and geometry reproduced our experimental data and revealed a likely RyR:LCC stoichiometry of ~ 5:1 for the Ca2+ spark initiating complex (IC). Using the experimental AP waveform, the model revealed a high coupling fidelity (Pcpl ~ 0.5) between each LCC opening and IC activation. The presence of ~ 4 ICs per couplon reduced Ca2+ spark latency and increased Pspark to match experimental data. Variability in AP release timing is less than that seen with voltage steps because the AP overshoot and later repolarization decrease Pspark due to effects on LCC flux and LCC deactivation respectively. This work provides a framework for explaining the Vm- and time-dependence of Pspark, and indicates how ion channel dispersion in disease can contribute to dyssynchrony in Ca2+ release.
Keywords: Heart, Ca2+ sparks, Cardiac myocytes, Excitation-contraction coupling, Action potential, Intracellular Ca2+
1. Introduction
Contraction of the heart depends critically on the near-synchronous Ca2+ release from internal stores (the sarcoplasmic reticulum, SR) via a process called ‘Ca2+-induced Ca2+ release’ (CICR) [1]. During normal excitation-contraction coupling (ECC), stochastic activation of surface membrane L-type Ca2+ channels (LCCs) supplies the Ca2+ trigger for CICR. Amplification of the Ca2+ influx via LCCs (iCa) [2,3] is provided by the Ca2+-dependent gating of multiple ryanodine receptors (RyRs, [4,5]) in the SR membrane. RyRs are clustered in dyads where the SR membrane is closely opposed to the surface membrane [6,7]). The local Ca2+ changes arising from LCC and RyR gating in this dyadic space are limited to “couplons” [8] where spatially restricted Ca2+ release events (“Ca2+ sparks” [9]) eventuate. This “local control” [2,8] of CICR normally prevents uncontrolled cell-wide regenerative Ca2+ release and is the result of high local Ca2+ concentrations in the dyad being transduced by relatively insensitive RyRs (that are unlikely to open at cytoplasmic levels of Ca2+) (for review see [10]).
Current models for ECC have focussed on the general features of relevant ion channel gating (e.g. [11,12]), nanoscopic structure of the dyad (e.g. [13,14]) and clustering of RyRs (e.g. [15,16]) in ventricular cells to contribute to a coherent explanation for the probabilistic nature of Ca2+ spark production [8,11,17,18]. However, it is unclear whether our existing knowledge of these processes is sufficient to explain both the membrane voltage- (Vm) and time-dependence of Ca2+ spark initiation (especially in response to an AP). The paucity of kinetic data on the timing of Ca2+ spark initiation is problematic for ECC models that should provide the biophysical underpinning for understanding both normal and abnormal Ca2+ release. Nevertheless, experiments have shown that the normal ventricular Ca2+ transient has remarkable uniformity [2,19] despite the intrinsic stochasticity in LCC and RyR gating that should cause temporally non-uniform Ca2+ release across dyads [2].
In the present study, we have analyzed the Vm-dependence of evoked Ca2+ spark timing and probability and compared the experimental data to predictions of an integrative computer model for Ca2+ spark initiation. We show that simple step command voltage clamp protocols, as are usually employed in biophysical experiments, do not reproduce the physiological latency for AP evoked Ca2+ sparks. An explanation for this unexpected observation is provided by the computer model, which was able to reproduce the experimental data. The model also allows exploration of the likely RyR:LCC stoichiometry that is needed to produce Ca2+ release with a short latency, as well as how many LCC openings are needed to initiate Ca2+ sparks (called the “coupling fidelity” [20]).
2. Methods
2.1. Myocyte isolation and Ca2+ imaging
All procedures were carried out in accordance with the UK Home Office Animals (Scientific Procedures) Act 1986 and conformed to the guidelines from Directive 2010/63/EU of the European Parliament on the protection of animals used for scientific purposes with institutional approval from the University of Bristol ethics committee. Male Wistar rats were euthanised by lethal injection of 140 mg/kg sodium pentobarbital (i.p.) followed by heart removal for enzymatic cell dissociation as described previously [21] and detailed in the Supplement. For Ca2+ imaging, ventricular cells were incubated with 5 μM Fluo-5F/AM (Thermofisher, Massachusetts, USA) then imaged on a confocal microscope (Zeiss, Oberkochen, Germany) in line-scan mode. A light emitting diode placed in the transmitted light detector pathway was used to provide precise timing information in the confocal images. Experiments were carried out at room temperature and additional experimental details are provided in the Supplement.
2.2. Electrophysiology
Ventricular cells were voltage-clamped with 10 mM tetraethylammonium chloride added to the 1 mM Ca2+ Tyrode’s solution, with a Cs+-based, Na+-free pipette solution. Cells were held at a Vm of −80 mV, pre-conditioned to control SR Ca2+ load (4 step depolarizations to 0 mV for 200 ms) and stimulated with either an AP waveform or step protocol at 0.2 Hz. For step protocols, an initial 400 ms ramp to −50 mV was used to inactivate the fast Na+ current. In the repolarizing step protocol (RP), Vm was stepped to 50 mV for 5 ms to activate LCCs, then to test potentials for 100 ms, while in the depolarizing step protocol (DP), Vm was stepped to test potentials for 200 ms. When indicated, 15 μM nifedipine (Sigma-Aldrich, Missouri, U.S.A.) was used to reduce LCC availability [22], so that individual Ca2+ sparks could be resolved [23,24].
2.3. Data analysis and Monte Carlo simulations
Data analysis was performed using custom computer programs written in MATLAB (v2021a, Mathworks, Massachusetts, U.S.A.). The latency of Ca2+ transients and Ca2+ sparks was measured from the start of the test pulse for DP, the end of the conditioning pulse for RP or the time at which the AP passed through −40 mV during phase 0 to the time taken for the rate of rise of fluorescence to exceed 5% of the maximum. Unless otherwise stated, error bars indicate one standard error of the mean (for experimental data), while shaded regions indicate the 95% confidence interval of the mean (for simulated data). Boxes show medians and interquartile ranges. Sample sizes are notated by n and N, which refer to the number of cells and animals, respectively. Statistical tests were performed in Graphpad Prism (v6.01) or MATLAB and a p-value (denoted as p) < 0.05 was considered to be the limit of statistical confidence.
Monte Carlo simulations of CICR latency incorporated stochastic LCC and RyR gating, electrodiffusion and Ca2+ buffering in the junctional space, and detection of Ca2+ by Fluo-5F and confocal microscopy. The Vm- and Ca2+-dependence of LCC gating kinetics was calculated using the LCC open probability (PO,LCC) from existing LCC models [25,26] and mean open time (τO,LCC) from single channel recordings made in near physiological conditions [27]. The Vm-dependence of iCa was estimated using the Goldman-Hodgkin-Katz flux equation (Eq. 11, Supplementary Material) which was scaled to be −0.15 pA at −10 mV with 1 mM extracellular [Ca2+] [28,29]. Dyad Ca2+ kinetics were calculated using a model that used a quadrilateral mesh geometry and included Ca2+ buffering by calmodulin, ATP, Fluo-5F, troponin, mitochondria, phospholipids and RyRs. Surface charge effects due to the surface membrane (see [30]) and junctional SR membrane were included (Fig. 3F). Ca2+-dependent RyR gating was based on published single channel data from rat [18,31]. The simulated detection of the calculated RyR opening was delayed by ~0.3 ms due to the kinetics of Ca2+ binding to Fluo-5F (from the dyad Ca2+ model) plus the effect of microscope blurring (~0.6 ms, [32]). For Ca2+ release latencies where the number of initiating complexes (nIC) was >1, the latency probability distribution was adjusted for missed events by P – (1 – P)nIC.
Fig. 3. Simulation of ECC using reported LCC, RyR and dyad properties.
(A) Time course of average LCC Ca2+ flux by DP to the Vm indicated (5000 simulations each). (B) Vm-dependence of peak LCC Ca2+ flux from A. Note that the averaged single LCC currents in A,B are very similar to reported whole cell ICa data (e.g. [24,34]). (C) and (D) show experimental (black symbols, from Josephson et al. [27]) and model (red lines) for PO,LCC and koff,LCC respectively (E) A distance map for RyRs with RyR positions as shown by Asghari et al. [41]. (F) Calculated [Ca2+]dyad 4 nm from the surface sarcolemma at the indicated lateral distances from an open LCC (iCa = 0.2 pA for 1 ms). The lateral distances shown were chosen so that these data can be directly compared Soeller & Cannell [30], as well as to the RyR distance map shown in panel E. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
3. Results
3.1. Ca2+ release latency during a normal action potential
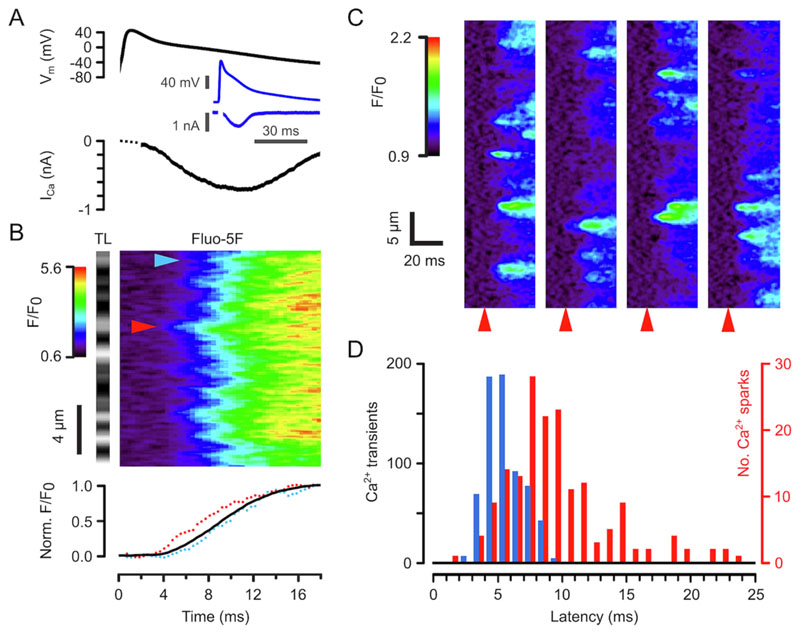
To eliminate cell-to-cell variability in action potential (AP) time course, we voltage-clamped cells using a previously recorded AP as the voltage command [33], with a low affinity fluorescent Ca2+ indicator (Fluo-5F) to report Ca2+ release. Fig. 1A (top panel) shows the early part of the AP (inset showing entire AP) with the nifedipine-sensitive Ca2+ current (ICa) below. It is apparent from the corresponding fluorescent Ca2+ record, imaged at high temporal and spatial resolution (Fig. 1B), that detectable Ca2+ release started after a delay of ~4 ms with peak Ca2+ being reached in ~12 ms. The non-uniformity in the pattern of Ca2+ release in this line-scan image occurred with a periodicity of 1.8 μm, as expected from the resting sarcomere spacing and the diffusion of Ca2+ from SR release sites at z-lines (z-lines can be seen in the corresponding transmitted light image on the left of the Fluo-5F line-scan image) toward the centre of the sarcomere [2]). Variability in the time of initiation of Ca2+ release along the scan line is highlighted by the arrows pointing to two z-lines. The time course of the local Ca2+ transients at these sites are shown in the lower panel in Fig. 1B together with the average time course of the Ca2+ transient. These data show that the time course of Ca2+ release is very similar at different z-lines but differs in the latency for activation [2].
Fig. 1. Ca2+ release latency during an AP.
(A) Top panel shows the AP voltage-clamp waveform and the bottom panel the evoked Ca2+ current (nifedipine-sensitive). The insets show the same data at a slower time scale. Amplifier saturation effects during the upstroke of the AP were not completely removed by subtraction of the current recorded in nifedipine and led to an artifact at early times as indicated in grey. (B) Confocal line scan image of the normalized Fluo-5F fluorescence (F/F0) changes evoked by the AP shown in A. The transmitted light intensity (TL) of the line scan region is shown at the left and indicates z-line positions. The time base is the same for panels A & B. Note the high time resolution and relative uniformity in Ca2+ release at z-lines. The bottom panel shows the mean time course of Fluo-5F fluorescence (black line) together with local time course at the positions marked by the arrow heads. (C) Ca2+ sparks evoked by sequential AP clamp pulses in the presence of nifedipine. Note the much lower probability of Ca2+ release along the scan line and the beat-to-beat variation in position and timing. The red arrows indicate when Vm exceeds −40 mV during the AP upstroke. (D) Ca2+ spark (red bars, 170 events, n/N = 6/3) and Ca2+ transient latencies (blue bars, 721 z-lines, n/N = 7/3). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The eventual uniformity in the Ca2+ transient at each z-line in Fig. 1B suggests that the probability of Ca2+ release in the form of Ca2+ sparks (Pspark) at each sarcomere must approach 1.0 [19]. This prevents individual Ca2+ spark latencies from being resolved since multiple Ca2+ release sites are likely to be within the confocal recording volume (see below and [19]). We therefore used nifedipine to block most LCCs and thereby reduce Pspark to the point where individual electrically stimulated events and their timing could be resolved [23,24]. Fig. 1C shows the effect of adding 15 μM nifedipine to the superfusate, where individual Ca2+ sparks and their latencies are clearly resolvable (time of depolarization marked by red arrows).
Fig. 1D summarises the observed Ca2+ release latencies in response to APs. Ca2+ transient (blue bars) and Ca2+ spark (red bars) latencies were 5.6 ± 0.1 ms (721 z-lines, n/N = 7/3) and 9.6 ± 0.3 ms (170 Ca2+ sparks, n/N = 6/3), respectively. At these times, the AP Vm was passing through 0 mV and − 12 mV (respectively). These voltages are near the peak of the Vm-Ca2+ transient amplitude relationship (see below and [34]).
3.2. Vm-dependence of Ca2+ release latency
To understand the observed Ca2+ spark latency during an AP, we examined its Vm-dependence using a conventional depolarizing protocol (DP), where Vm was stepped from −50 mV to various test potentials to evoke Ca2+ sparks. It is apparent from the line-scan images shown in Fig. 2A that during DP, Ca2+ spark rate had a bell-shaped Vm-dependence (summarised in Fig. 2B) but Ca2+ spark amplitude had no dependence on Vm (Fig. S1 and [23]). Since the maximum rate of rise of a Ca2+ transient (in the absence of nifedipine) should be a reasonable proxy for Ca2+ spark rate (Fig. 2B), the similarity of the Vm-dependence of both (p > 0.999, χ2 test) suggests that nifedipine does not markedly alter the Vm-dependence of LCC gating, adding confidence to the idea that latencies measured in the presence of nifedipine should reflect normal ECC. This is also consistent with the suggestion that nifedipine stabilizes the LCC in a closed state [35] (see [22] for a review).
Fig. 2. Ca2+ spark latencies in response to DP and RP.
(A) Exemplar confocal line scan images showing Ca2+ sparks in the presence of nifedipine at the indicated DP Vm. (B) Ca2+ spark rate during the first 20 ms (838 Ca2+ sparks, n/N = 14/6) (filled circles). The maximum rate of rise for Ca2+ transients for the same DP Vm is shown by blue triangles (218 Ca2+ transients, n/N = 14/8). There was no difference between these Vm dependencies when normalized (p > 0.999, Chi-squared test). (C) Corresponding Vm-dependence of mean (solid circles), median and interquartile range (grey bars) Ca2+ spark latency (1215 Ca2+ sparks, n/N = 14/8). The solid blue line shows an empirical bi-exponential fit to the mean data: (r2 = 0.99). (D) Ca2+ sparks in response to RP (same cell as A). The inset shows the distribution of Ca2+ spark latency during DP to −30 mV. (E) Vm-dependence of Ca2+ spark latency during RP in the presence of nifedipine (1201 Ca2+ sparks, n/N = 15/7). The solid blue line shows an empirical exponential fit: (r2 = 0.96). The inset shows the distribution of Ca2+ spark latency during RP to −30 mV. (F) The coefficient of variation for DP-, RP- and AP-evoked Ca2+ spark latencies. For DP and RP, the Vm were − 30 to 10 mV (p-values from Mann-Whitney tests). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Fig. 2C shows that the mean Ca2+ spark latency was ~58 ms at −40 mV and decreased with Vm to a minimum between −10 and 20 mV (14.5 ± 0.7 ms, 575 Ca2+ sparks, n/N = 14/6) before increasing again. This minimum DP latency was 4.9 ms longer than observed during the AP (c. f. Fig. 1D, p = 0.007, Mann-Whitney test). However, as noted above, the AP was repolarizing around the time where Ca2+ sparks appeared, so measuring Ca2+ spark latency with a conventional DP waveform may not be analogous to the physiological AP. During a pre-pulse to very positive potentials, iCa is strongly reduced and LCCs can open without activating CICR and Ca2+ sparks [36–38]. When Vm is then stepped to more negative potentials iCa increases and triggers CICR with a delay due to the kinetics of Ca2+ accumulation around RyRs and RyR activation kinetics. To analyse this case, we used a 5 ms 50 mV pre-pulse because, at this potential, the reduced iCa did not activate CICR (as evidenced by the lack of Ca2+ sparks during the pre-pulse) and Ca2+ influx via reverse mode Na+-Ca2+ exchange was minimal due to the absence of Na+ in the patch pipette filling solution.
For this repolarizing protocol (RP), Ca2+ sparks were evoked at all test potentials, although the number decreased at Vm > +10 mV (Fig. 2D). Fig. 2E shows that RP Ca2+ spark latency increased approximately exponentially with Vm (solid lines show a 40 mV e-fold increase). Below −90 mV, Ca2+ spark activation was very rapid with a delay that asymptotically approached 3.3 ms. This minimal latency should reflect the delay associated with RyR activation as well as diffusion and detection of Ca2+. At −10 mV, the RP Ca2+ spark latency was 8.7 ± 0.5 ms (189 Ca2+ sparks, n/N = 15/7) which is slightly (0.9 ms) shorter than observed with APs (p < 0.0001, Mann-Whitney test). It follows that the overshoot of the physiological AP and phase 1 repolarization serves an important role to minimise both the Ca2+ transient latency and non-uniformity in local Ca2+ release [21,39,40].
If the latency to first LCC opening were the dominant factor in the delay for Ca2+ spark activation, the observed Ca2+ spark latency distribution should be exponential. However, this was not the case (see insets Figs. 2C & E) at any potential and this observation is underscored by the median latency (bars) not being 69% of the mean latency (filled circles).
Despite the general similarity in the Vm-dependent behaviour of Ca2+ sparks and Ca2+ transients (Fig. 2B), closer inspection of the Ca2+ spark initiation data showed that variability in the AP-evoked latency (as measured by the coefficient of variation, CV = σ/x−) was smaller than seen with RP or DP (Fig. 2F). This might seem counter-intuitive, especially if one considers that each Ca2+ spark occurs at a different Vm during the AP which should add variance due to the Vm-dependence of Ca2+ spark latency. This raises the questions: How does the stochastic gating of LCCs and RyRs contribute to Ca2+ spark initiation and how is the variance in Ca2+ spark timing reduced?
3.3. Reconstructing the Ca2+ spark initiating complex
To examine the sources of variance underlying the timing of Ca2+ sparks, we constructed a Monte-Carlo model that calculates Vm- and Ca2+-dependent LCC gating, Ca2+ diffusion and binding, and RyR Ca2+‒dependent activation rate. The model formulation (for key details, see equations in Supplement) reproduced whole-cell Ca2+ currents (Figs. 3A, S3) and the peak ICa I-V relationship (Fig. 3B) in accord with experimental findings. The underlying single-channel PO,LCC (Fig. 3C, Eq. 9 in Supplement) and on rate (Fig. 3D, Eq. 10 in Supplement) are shown superimposed on reference single channel data [27]. The recent finding that RyR clusters may be more sparsely arranged than previously thought [41] was also incorporated. A prototypic RyR cluster, derived from published data [41], is shown in Fig. 3E and the distance from the individual RyRs is shown in colormap. Fig. 3F shows how [Ca2+]dyad, at distances shown within these colormap levels, changes in response to a 1 ms LCC opening. Shortly after the discovery of Ca2+ sparks, analysis of the evoked Ca2+ spark rate [2,3] suggested that local [Ca2+]dyad levels would need to increase ~100-fold to explain the transition from rest to full activation. Fig. 3F shows that such levels (~10 μM) only develop (after an LCC opens) within a distance that is similar to the half-width of the RyR. This implies that the Ca2+ spark initiating complex (IC) cannot involve all RyRs in the cluster but only those in very close proximity to the open LCC. It follows that the low numbers of RyRs in close proximity to the open LCC should contribute the latency for Ca2+ spark activation. The removal of most of the latency for LCC activation during RP (compare Fig. 2C and E), along with the consequent reduction in uncertainty in LCC activation rate, would allow the Monte-Carlo model kinetic response to RP to be used to investigate the likely RyR:LCC stoichiometry within the IC.
3.4. Stoichiometry of Ca2+ spark initiating complex
As illustrated in Fig. 4A, we used the Vm-dependence of LCC gating and corresponding iCa (Eq. 11 in Supplement) to calculate local [Ca2+] changes that were subject to buffering by the membranes, ATP, calmodulin and Fluo-5F. Experimental RP Ca2+ release latencies were then compared to the results of many simulations (1000’s) that gave the average time to activation of a model IC that had different numbers of RyRs (red arrow in lower RyR Po trace in Fig. 4A). Fig. 4B shows the calculated Ca2+ release latency for RP at 3 potentials (chosen to reflect the Vm at which the majority of Ca2+ sparks occur during an AP, see Fig. 1D) for a range of RyR:LCC stoichiometries. Measured Ca2+ spark latencies at the indicated potentials are shown on the left (and extended by coloured regions across the figure) and should be compared to the behaviour of a single IC at the indicated RyR:LCC stoichiometry. At all three potentials, an RyR:LCC ratio between 3:1 and 8:1 appeared sufficient to explain the experimentally observed latency.
Fig. 4. Ca2+ release latency during voltage-clamp steps.
(A) Diagram showing modelled processes that contribute to the calculated Ca2+ release latency (from top to bottom): (i) DP to 0 mV with finite clamp speed (τ ~ 0.3 ms), (ii) LCC transitions from closed (‘C’) to open (‘O’) states, (iii) the corresponding Ca2+ influx (iCa), (iv) [Ca2+]dyad at an RyR location (indicated by colour, see inset). RyR locations were derived from electron microscopy data [41]. (v) Net RyR on rate and (vi) cumulative RyR open probability (∑po, RyR), where the first opening that triggers Ca2+ release is indicated (red arrow). (B) Simulated mean (solid lines) and 95% confidence intervals for Ca2+ release latencies for various RyR:LCC ratios and RP = ‒15, 0 and 15 mV (4000 simulations for each data point). The measured latencies during RP are shown at left (from Fig. 2E). (C) Simulated Vm-dependence of Ca2+ release latency for a 4:1 (purple) or 8:1 (green) RyR:LCC stoichiometry. (D) Corresponding Ca2+ spark rate. Inset shows RyR arrangements for the simulations. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The Vm-dependence of RP Ca2+ spark latency was reproduced with an RyR:LCC stoichiometry of 4:1 (r2 = 0.88) (purple, Fig. 4C) and the fit only slightly improved (r2 = 0.91) at 8:1 (green). Importantly, the model also fit the Vm-dependence of Ca2+ spark rate reasonably well (Fig. 4D). At first sight, the slight decrease in Ca2+ spark rate during RP to Vm below ‒10 mV might appear paradoxical when iCa increases as Vm becomes more negative (see Eq. 11 in Supplement). However, the model showed that this can be explained by a the Vm-dependence of LCC mean open time (τO,LCC). At very negative potentials, the small τO,LCC (due to increasing deactivation) led to a decrease the average [Ca2+]dyad associated with each LCC opening and a consequent decrease in RyR opening rate (see LCC openings marked by asterisks in Fig. 4A). When Vm was >10 mV Ca2+ spark rate decreased rapidly, reflecting the decreasing iCa and the Ca2+ dependence of RyR opening rate.
3.5. Coupling fidelity in the IC
The computer model also allows us estimate the ability of a single LCC opening to initiate a Ca2+ spark (“coupling fidelity”, Pcpl [20]). Fig. 5A shows the number of LCC openings (no) associated with the initiation of each Ca2+ spark as a function of RP Vm. At very negative potentials, the iCa associated with a single LCC opening (n0 = 1) could trigger a Ca2+ spark ~60% of the time. The remaining ~40% required a second LCC opening (n0 = 2), which is possible because not all LCCs are activated during the RP pre-pulse (note a maximum PO,LCC of ~0.3 in Fig. 3C) and LCCs do not enter an absorbing closed state. Over the Vm range where Ca2+ sparks appear during the AP (Fig. 5A grey region) and in response to an AP (highlighted in red at the right, see below), Ca2+ sparks are most likely to occur on the second LCC opening. In summary, this analysis of no suggests that Pcpl decreases with Vm (Fig. 5B) and is 0.4–0.6 over the physiological range.
Fig. 5. Pspark and Pcpl during RP- and AP-evoked Ca2+ release.
(A) The fraction of Ca2+ sparks (Fspark) that were evoked by no LCC openings at each Vm (40 00 simulations each). The right-hand side shows the corresponding Fspark using the AP shown in Fig. 1A. The mean nO was 2.01 ± 0.01 (10,000 simulations). (B) The Vm-dependence of coupling fidelity (Pcpl) during RP. Pcpl for an AP is shown by a cross on the right. C) The exemplar AP waveform used in the computer model. D) Simulated Pspark using a 4:1 RyR:LCC stoichiometry for 1 (red bars) or 4 (blue bars) ICs. The mean latencies were 9.55 ± 0.06 ms (Pspark = 0.796, 10,000 simulations) and 5.61 ± 0.02 ms (Pspark = 0.997, 10,000 simulations) respectively. E) Average iCa during the AP and proportion involved in triggering Ca2+ transients (blue region). F) The PDFs for Ca2+ spark initiation during an AP (red) or RP to 0 mV (black). The results in E and F are the average of 50,000 simulations and the origin of the reduction Ca2+ release dyssynchrony associated with the AP compared to RP is highlighted by the green shaded regions. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The model predictions were also tested using the AP (Fig. 5C) to give a physiological Vm- and time-dependence to the calculated iCa and resulting Ca2+ spark initiation latencies. As shown in Fig. 5D, the mean simulated AP-evoked Ca2+ release latency in the presence of one IC was 9.55 ± 0.06 ms (red bars), which was not different to that observed experimentally (p = 0.55, Mann-Whitney test, cf. Fig. 1D). This is also consistent with the idea that nifedipine can reduce LCC availability to one per IC or couplon [24]. When the number of ICs in the dyad was increased to 4, the latency for Ca2+ release was approximately halved and was not different to that observed for Ca2+ transients (p = 0.24, Mann-Whitney test, cf. Fig. 1D). Fig. 5E shows the simulated average iCa during an AP (black line), with the fraction that was responsible for Ca2+ transient activation in the blue shaded region and comprised ~20% of the integrated iCa. Fig. 5F shows the probability density functions for Ca2+ spark latency during the AP or RP to 0 mV and explains how the variability in Ca2+ release latency is reduced by the AP waveform. At a latencies <4.5 ms, Pspark is higher for RP because Vm during the AP is > +10 mV, which reduces Pspark and increases latency (see Fig. 2E). At latencies >15 ms, Pspark for the AP is also reduced compared to RP because LCCs are deactivating. The net effect is that the Pspark probability density function over time is narrower than for RP, resulting in less variability in Ca2+ spark timing (CV = 0.56 and 1.21 for AP and RP to 0 mV, respectively) and a hence a more spatially uniform Ca2+ transient.
4. Discussion
The experiments and modelling shown here provide quantitative insight into the control of CICR during APs. While previous studies have shown that AP phase 1 repolarization can increase Ca2+ release synchrony [21,39,40], this effect has been ascribed solely to increasing iCa during phase 1. The present study shows that explanation is incomplete because the kinetics of LCC gating also plays a role during early and later repolarization (Fig. 5F). So, while RP, which can mimic a very fast and deep AP phase 1, can reduce latency compared to DP it does not, by itself, produce the same degree of synchrony in Ca2+ release as an AP. The overshoot (i.e. Vm > 0 mV) of the AP also reduces variance in the timing of CICR and this is central to the production of a more uniform Ca2+ transient. The new latency data (and their empirical equation fits) presented here also form a “ground truth” for the development of future models of cardiac ECC which may find application within in-silico drug screening, and the present computer model illustrates how these data can be used to gain new insight into the operation of CICR at the single channel level.
4.1. Definition of the Ca2+ spark initiation complex
Previous work has shown that a single LCC can initiate a Ca2+ spark at negative potentials [23,24,42]. However, such experiments did not give insight into the number of LCCs, Pcpl or the number of local RyRs involved in Ca2+ spark initiation at physiological potentials. By examining the Vm-dependence of Ca2+ spark latency and fitting a detailed model to observed behaviour we have found that the latency for Ca2+ sparks during the AP can be explained by 4 ICs (at a minimum) each made up of an LCC and at least 3 RyRs. This stoichiometry provides the first functional definition of the physiological couplon, with the local group of RyRs in the IC being most likely to be activated on the second opening of the nearby LCC. The simulations also showed that the apparent RyR:LCC stoichiometry increases as Vm becomes more negative (Fig. 4B), an effect that is due to the increase in iCa activating a larger group of RyRs, whose summed opening rate decreases latency.
Only ~20% of the total LCC current seems to be needed to trigger CICR, and within the parlance of CICR theory forms the “triggering” component of ICa [43]. The remaining excess ensures activation of every couplon during the AP and also provides additional Ca2+ influx to control SR Ca2+ content and releasable Ca2+. The fraction of ICa responsible for triggering Ca2+ sparks is sensitive to the AP waveform due to the interactions between Vm, iCa and LCC gating. A prolongation of AP phase 1 may increase or decrease net Ca2+ influx via ICa (depending on the exact AP time course). However, an increase in the time taken for iCa to increase to the point where Ca2+ spark initiation is likely will increase Ca2+ spark latency (see Fig. S4) and reduce Pcpl.
Typically, two LCC openings (Pcpl ~ 0.5) were needed to trigger CICR to reproduce the observed mean Ca2+ spark latency (which was greater than the mean closed of the LCC) (Fig. 5A). While the number of LCCs needed to produce the observed delay is predicated on the closed time of the LCC, we were constrained by available LCC gating data [27]. The possibility of modal [44] or cooperative gating [45] in LCCs is problematic since these effects could increase the effective closed time and would have prevented the simple LCC gating model from fitting the observed Vm-dependence of Ca2+ spark latency. Since this was not the case, such gating effects may be limited to a subset of clusters at the limits of the Vm range studied (see below) and/or under different conditions. Nevertheless, the finding that Pcpl ~ 0.5 should not be changed by such effects. It is important to note that, while it is the activation of the first IC that leads to a Ca2+ spark, the other ICs (and their RyRs) serve to increase the Ca2+ release flux when they are activated (either by their nearest LCC or by Ca2+ release from the triggering IC).
4.2. Comparison to binding, imaging and biophysical studies
The requirement for an RyR:LCC stoichiometry of 3–8:1 (Fig. 4B) per IC with up to 4 ICs per couplon is compatible with binding studies [46,47] and flux comparisons [48] that indicate a RyR:LCC stoichiometry of ~7:1. Our data is also compatible with super-resolution imaging that suggests a mean of ~8–13 RyRs per “cluster” [49,50] and 2 clusters per couplon [50]. The convergence of these values obtained by very different techniques argues against very large numbers of RyRs in a couplon and we note that previous confocal [51] and electron microscopy quantification [6] would have over-estimated the number of RyRs due to the assumption of tight RyR packing. Correcting the previous optical estimate of 78 RyRs/couplon [51] for a packing density of 40% [41] yields ~30 RyRs/couplon. Similar correction of electron microscopy data [52] suggests 23 RyRs per couplon. Another estimate of the number of RyRs in a couplon was provided by noise analysis [19] and suggested >18 RyRs per couplon, while a peak flux during a Ca2+ spark of 7.9 pA [32] with a single RyR channel current of 0.5 pA [53,54] and PO of 0.5–0.8 [18,31] suggest ~24 RyRs per couplon. Together, these estimates seem to converge toward a mean of ~23 RyRs per couplon, which is in good agreement with the data presented here.
4.3. Coupling fidelity
Some previous studies have suggested that Pcpl is much lower (<0.05, [20,38,55]) than estimated here, although the reported value is dependent on the experimental protocol. Uncertainty in the reported Pcpl may arise from extrapolation to physiological conditions (e.g. the assumed Vm-dependence of iCa and τO,LCC, and the τO,LCC-dependence of Pspark). With a very low Pcpl, a high Pspark (~1.0) during an AP becomes problematic and requires large numbers of LCCs (25–600, [20,38,55]) to be available in the couplon, and such numbers may not be compatible with electrophysiological and binding data. Our data and analysis remove this problem: with Pcpl = 0.5 and ~ 4 ICs in each couplon, Pspark is 0.94 for each LCC opening and with the assumption of 2 openings during the AP (see Fig. 5A) Pspark becomes 0.99.
It is important to note that Pcpl is not the same as the ability of the LCC Ca2+ flux to (eventually) trigger Ca2+ release. Thus, experiments that modulate iCa [42,56] and which have shown that LCCs can trigger a Ca2+ sparks with high fidelity, are consistent with Pcpl as defined here.
4.4. Limitations of the model
Despite the ability of the model to accurately reproduce the Vm-dependence of RP Ca2+ spark latency (Fig. 3C), the fit to DP Ca2+ spark latency at positive potentials developed a systematic error (see Fig. S2). We suggest this is due to a deficiency in the LCC gating model at higher potentials since DP Ca2+ spark latency is highly dependent on the LCC closed and open times. Unfortunately, we could not find single channel gating data in this Vm range that could be used to further refine model LCC behaviour. Nevertheless, the model reproduced a clear inflexion in the RP Ca2+ spark latency curve (Fig. 3C) around ‒10 mV which adds to confidence in the overall behaviour of the model. This inflexion was due to interactions between the Vm- and Ca2+-dependence of LCC inactivation, as well as the Vm-dependence of Pcpl.
4.5. Physiological implications
There should be an optimal RyR:LCC stoichiometry for the IC and the number of ICs in a couplon. Our simulations showed that the steep gradients of Ca2+ within the dyad (Fig. 3F), transduced via the nonlinear RyR opening rate resulted in strong constraints on the number of RyRs in an IC (c.f. Fig. 4B) and ~5 RyRs:1 LCC per IC and 4 ICs per couplon was sufficient to explain both Pspark as well as Ca2+ spark latency. This stoichiometry not only minimises latency, but also latency variance during the AP to produce a more uniform Ca2+ transient. Adding more LCCs to an IC produces redundant Ca2+ influx at later times and this extra Ca2+ has energetic consequences because it has to be pumped out of the cell (eventually). In addition, a decreased delay to first LCC opening does not translate to a similar reduction in Ca2+ release latency because iCa is reduced at earlier times due to the reduced driving force for Ca2+ entry. Adding more RyRs does not markedly increase Pcpl because each additional RyR has to be placed further from the initiating LCC and will be exposed to a lower local [Ca2+] when the LCC opens (see Fig. 3F). In addition, the release flux from the couplon is likely limited by local SR Ca2+ depletion [18] so additional RyRs may not increase the overall CICR amplification but will increase the risk of arrhythmogenic spontaneous Ca2+ sparks during diastole. While dispersion of RyRs in heart failure [49,57] could reduce the number of RyRs in an IC, Pcpl should not change markedly until their mean distance from the LCC is increased significantly. On the other hand, dispersion of functional ICs should lead to a more inhomogeneous Ca2+ transient and may explain the recent observation of “wandering” Ca2+ sparks [58] and compound the problem of IC uncoupling by t-tubule loss [59].
Supplementary Material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.yjmcc.2023.07.005.
Acknowledgements
We would like to thank Dr. M. Stern (NIA NIH, Baltimore) for critical reading of the manuscript and Drs. A.F. James and L. Pannell for supplying some of the cells used in this study (the latter supported by British Heart Foundation FS/CRTF/21/24122).
Sources of funding
The work was supported by the Auckland Medical Research Foundation (81,216 to C.H.T.K.), Health Research Council of New Zealand (08/049 to M.B.C.), British Heart Foundation (PG/20/5/34801 to C.H. T.K. and M.B.C.; IG/13/3/30212 to M.B.C.), Medical Research Council (MR/N002903/1 to M.B.C.), Royal Society (U.K.) Wolfson Merit Award (to M.B.C.) and facilities provided by the Advanced Computing Research Centre at the University of Bristol (http://www.bristol.ac.uk/acrc/).
Abbreviations
- AP
action potential
- CICR
Ca2+-induced Ca2+ release
- DP
depolarizing step voltage protocol
- ECC
excitation-contraction coupling
- IC
initiating complex
- iCa
unitary L-type Ca2+ current
- ICa
L-type Ca2+ current
- LCC
L-type Ca2+ channel
- Pcpl
coupling fidelity
- PO,LCC
LCC open probability
- Pspark
evoked Ca2+ spark probability
- RP
repolarizing step voltage protocol
- RyR
ryanodine receptor (SR Ca2+ release channel)
- SR
sarcoplasmic reticulum
- τO,LCC
LCC mean open time
- Vm
membrane potential
Footnotes
Author contributions
C.H.T.K. and M.B.C. Conceptualization, Methodology, Writing, Funding acquisition; C.H.T.K Formal analysis.
Declaration of Competing Interest
The authors declare no competing interest.
Disclosures
None.
Data availability
The data underlying this article will be shared on reasonable request to the authors.
References
- [1].Fabiato A, Fabiato F. Contractions induced by a calcium-triggered release of calcium from the sarcoplasmic reticulum of single skinned cardiac cells. J Physiol. 1975;249:469–495. doi: 10.1113/jphysiol.1975.sp011026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Cannell MB, Cheng H, Lederer WJ. Spatial non-uniformities in [Ca2+]i during excitation-contraction coupling in cardiac myocytes. Biophys J. 1994;67:15. doi: 10.1016/S0006-3495(94)80677-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Wier WG, Egan TM, López-López JR, Balke CW. Local control of excitation-contraction coupling in rat heart cells. J Physiol. 1994;474:463–471. doi: 10.1113/jphysiol.1994.sp020037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Sutko JL, Kenyon JL. Ryanodine modification of cardiac muscle responses to potassium-free solutions. Evidence for inhibition of sarcoplasmic reticulum calcium release. J Gen Physiol. 1983;82:385–404. doi: 10.1085/jgp.82.3.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Lai FA, Anderson K, Rousseau E, Liu Q-Y, Meissner G. Evidence for a Ca2+ channel within the ryanodine receptor complex from cardiac sarcoplasmic reticulum. Biochem Biophys Res Commun. 1988;151:441–449. doi: 10.1016/0006-291x(88)90613-4. [DOI] [PubMed] [Google Scholar]
- [6].Franzini-Armstrong C, Protasi F, Ramesh V. Shape, size, and distribution of Ca2+ release units and Couplons in skeletal and cardiac muscles. Biophys J. 1999;77:1528–1539. doi: 10.1016/S0006-3495(99)77000-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Jorgensen AO, Shen AC, Arnold W, McPherson PS, Campbell KP. The Ca2+-release channel/ryanodine receptor is localized in junctional and corbular sarcoplasmic reticulum in cardiac muscle. J Cell Biol. 1993;120:969–980. doi: 10.1083/jcb.120.4.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Stern MD. Theory of excitation-contraction coupling in cardiac muscle. Biophys J. 1992;63:497–517. doi: 10.1016/S0006-3495(92)81615-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Cheng H, Lederer WJ, Cannell MB. Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle. Science. 1993;262:740–744. doi: 10.1126/science.8235594. [DOI] [PubMed] [Google Scholar]
- [10].Cannell MB, Kong CHT. Local control in cardiac E–C coupling. J Mol Cell Cardiol. 2012;52:298–303. doi: 10.1016/j.yjmcc.2011.04.014. [DOI] [PubMed] [Google Scholar]
- [11].Sobie EA, Ramay HR. Excitation-contraction coupling gain in ventricular myocytes: insights from a parsimonious model: EC coupling gain model. J Physiol. 2009;587:1293–1299. doi: 10.1113/jphysiol.2008.163915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Agrawal A, Wang K, Polonchuk L, Cooper J, Hendrix M, Gavaghan DJ, Mirams GR, Clerx M. Models of the cardiac L-type calcium current: a quantitative review. WIREs Mech Dis. 2023;15:e1581. doi: 10.1002/wsbm.1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Tanskanen AJ, Greenstein JL, Chen A, Sun SX, Winslow RL. Protein geometry and placement in the cardiac dyad influence macroscopic properties of calcium-induced calcium release. Biophys J. 2007;92:3379–3396. doi: 10.1529/biophysj.106.089425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Koh X, Srinivasan B, Ching HS, Levchenko A. A 3D Monte Carlo analysis of the role of dyadic space geometry in spark generation. Biophys J. 2006;90:1999–2014. doi: 10.1529/biophysj.105.065466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Iaparov BI, Zahradnik I, Moskvin AS, Zahradníková A. In silico simulations reveal that RYR distribution affects the dynamics of calcium release in cardiac myocytes. J Gen Physiol. 2021;153:e202012685. doi: 10.1085/jgp.202012685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Xie Y, Yang Y, Galice S, Bers DM, Sato D. Size matters: ryanodine receptor cluster size heterogeneity potentiates calcium waves. Biophys J. 2019;116:530–539. doi: 10.1016/j.bpj.2018.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Williams GSB, Smith GD, Sobie EA, Jafri MS. Models of cardiac excitation–contraction coupling in ventricular myocytes. Math Biosci. 2010;226:1–15. doi: 10.1016/j.mbs.2010.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Laver DR, Kong CHT, Imtiaz MS, Cannell MB. Termination of calcium-induced calcium release by induction decay: an emergent property of stochastic channel gating and molecular scale architecture. J Mol Cell Cardiol. 2013;54:98–100. doi: 10.1016/j.yjmcc.2012.10.009. [DOI] [PubMed] [Google Scholar]
- [19].Bridge JHB, Ershler PR, Cannell MB. Properties of Ca2+ sparks evoked by action potentials in mouse ventricular myocytes. J Physiol. 1999;518:469–478. doi: 10.1111/j.1469-7793.1999.0469p.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Zhou Y-Y, Song L-S, Lakatta EG, Xiao R-P, Cheng H. Constitutive β2-adrenergic signalling enhances sarcoplasmic reticulum Ca2+ cycling to augment contraction in mouse heart. J Physiol. 1999;521:351–361. doi: 10.1111/j.1469-7793.1999.00351.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Cooper PJ, Soeller C, Cannell MB. Excitation–contraction coupling in human heart failure examined by action potential clamp in rat cardiac myocytes. J Mol Cell Cardiol. 2010;49:911–917. doi: 10.1016/j.yjmcc.2010.04.012. [DOI] [PubMed] [Google Scholar]
- [22].McDonald TF, Pelzer S, Trautwein W, Pelzer DJ. Regulation and modulation of calcium channels in cardiac, skeletal, and smooth muscle cells. Physiol Rev. 1994;74:365–507. doi: 10.1152/physrev.1994.74.2.365. [DOI] [PubMed] [Google Scholar]
- [23].Collier ML, Thomas AP, Berlin JR. Relationship between L-type Ca2+ current and unitary sarcoplasmic reticulum Ca2+ release events in rat ventricular myocytes. J Physiol. 1999;516(Pt 1):117–128. doi: 10.1111/j.1469-7793.1999.117aa.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Santana LF, Cheng H, Gómez AM, Cannell MB, Lederer WJ. Relation between the sarcolemmal Ca 2+ current and Ca 2+ sparks and local control theories for cardiac excitation-contraction coupling. Circ Res. 1996;78:166–171. doi: 10.1161/01.res.78.1.166. [DOI] [PubMed] [Google Scholar]
- [25].O’Hara T, Virág L, Varró A, Rudy Y. Simulation of the undiseased human cardiac ventricular action potential: model formulation and experimental validation. PLoS Comput Biol. 2011;7:e1002061. doi: 10.1371/journal.pcbi.1002061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Grandy SA, Howlett SE. Cardiac excitation-contraction coupling is altered in myocytes from aged male mice but not in cells from aged female mice. Am J Physiol-Heart Circ Physiol. 2006;291:H2362–H2370. doi: 10.1152/ajpheart.00070.2006. [DOI] [PubMed] [Google Scholar]
- [27].Josephson IR, Guia A, Sobie EA, Lederer WJ, Lakatta EG, Stern MD. Physiologic gating properties of unitary cardiac L-type Ca2+ channels. Biochem Biophys Res Commun. 2010;396:763–766. doi: 10.1016/j.bbrc.2010.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Guia A, Stern MD, Lakatta EG, Josephson IR. Ion concentration-dependence of rat cardiac unitary L-type Calcium Channel conductance. Biophys J. 2001;80:2742–2750. doi: 10.1016/S0006-3495(01)76242-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Rose WC, Balke CW, Wier WG, Marban E. Macroscopic and unitary properties of physiological ion flux through L-type Ca2+ channels in guinea-pig heart cells. J Physiol. 1992;456:267–284. doi: 10.1113/jphysiol.1992.sp019336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Soeller C, Cannell MB. Numerical simulation of local calcium movements during L-type calcium channel gating in the cardiac diad. Biophys J. 1997;73:97–111. doi: 10.1016/S0006-3495(97)78051-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Fill M, Gillespie D. Ryanodine receptor open times are determined in the closed state. Biophys J. 2018;115:1160–1165. doi: 10.1016/j.bpj.2018.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Kong CHT, Laver DR, Cannell MB. Extraction of sub-microscopic Ca fluxes from blurred and noisy fluorescent Indicator images with a detailed model fitting approach. PLoS Comput Biol. 2013;9:e1002931. doi: 10.1371/journal.pcbi.1002931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Doerr T, Denger R, Trautwein W. Calcium currents in single SA nodal cells of the rabbit heart studied with action potential clamp. Pflugers Arch. 1989;413:599–603. doi: 10.1007/BF00581808. [DOI] [PubMed] [Google Scholar]
- [34].Cannell MB, Berlin JR, Lederer WJ. Effect of membrane potential changes on the calcium transient in single rat cardiac muscle cells. Science. 1987;238:1419–1423. doi: 10.1126/science.2446391. [DOI] [PubMed] [Google Scholar]
- [35].Hadley RW, Lederer WJ. Nifedipine inhibits movement of cardiac calcium channels through late, but not early, gating transitions. Am J Physiol-Heart Circ Physiol. 1995;269:H1784–H1790. doi: 10.1152/ajpheart.1995.269.5.H1784. [DOI] [PubMed] [Google Scholar]
- [36].Isenberg G, Han S. Gradation of Ca2+-induced Ca2+ release by voltage-clamp pulse duration in potentiated guinea-pig ventricular myocytes. J Physiol. 1994;480:423–438. doi: 10.1113/jphysiol.1994.sp020372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Cannell MB, Cheng H, Lederer WJ. The control of calcium release in heart muscle. Science. 1995;268:1045–1049. doi: 10.1126/science.7754384. [DOI] [PubMed] [Google Scholar]
- [38].Poláková E, Zahradníková A, Jr, Pavelková J, Zahradník I, Zahradníková A. Local calcium release activation by DHPR calcium channel openings in rat cardiac myocytes: local calcium release activation in cardiac myocytes. J Physiol. 2008;586:3839–3854. doi: 10.1113/jphysiol.2007.149989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Sah R, Ramirez RJ, Kaprielian R, Backx PH. Alterations in action potential profile enhance excitation-contraction coupling in rat cardiac myocytes. J Physiol. 2001;533:201–214. doi: 10.1111/j.1469-7793.2001.0201b.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Fowler ED, Wang N, Hezzell MJ, Chanoit G, Hancox JC, Cannell MB. Improved Ca2+ release synchrony following selective modification of Itof and phase 1 repolarization in normal and failing ventricular myocytes. J Mol Cell Cardiol. 2022;172:52–62. doi: 10.1016/j.yjmcc.2022.07.009. [DOI] [PubMed] [Google Scholar]
- [41].Asghari P, Scriven DR, Ng M, Panwar P, Chou KC, van Petegem F, Moore ED. Cardiac ryanodine receptor distribution is dynamic and changed by auxiliary proteins and post-translational modification. ELife. 2020;9:e51602. doi: 10.7554/eLife.51602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Altamirano J, Bers DM. Voltage Dependence of Cardiac Excitation–Contraction Coupling: Unitary Ca2+ Current Amplitude and Open Channel Probability. Circ Res. 2007;101:590–597. doi: 10.1161/CIRCRESAHA.107.152322. [DOI] [PubMed] [Google Scholar]
- [43].Fabiato A. Simulated calcium current can both cause calcium loading in and trigger calcium release from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J Gen Physiol. 1985;85:291–320. doi: 10.1085/jgp.85.2.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Hess P, Lansman JB, Tsien RW. Different modes of ca channel gating behaviour favoured by dihydropyridine Ca agonists and antagonists. Nature. 1984;311:538–544. doi: 10.1038/311538a0. [DOI] [PubMed] [Google Scholar]
- [45].Sato D, Dixon RE, Santana LF, Navedo MF. A model for cooperative gating of L-type Ca2+ channels and its effects on cardiac alternans dynamics. PLoS Comput Biol. 2018;14:e1005906. doi: 10.1371/journal.pcbi.1005906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Wibo M, Bravo G, Godfraind T. Postnatal maturation of excitation-contraction coupling in rat ventricle in relation to the subcellular localization and surface density of 1,4-dihydropyridine and ryanodine receptors. Circ Res. 1991;68:662–673. doi: 10.1161/01.res.68.3.662. [DOI] [PubMed] [Google Scholar]
- [47].Bers DM, Stiffel VM. Ratio of ryanodine to dihydropyridine receptors in cardiac and skeletal muscle and implications for E-C coupling. Am J Physiol-Cell Physiol. 1993;264:C1587–C1593. doi: 10.1152/ajpcell.1993.264.6.C1587. [DOI] [PubMed] [Google Scholar]
- [48].Wang S-Q, Song L-S, Lakatta EG, Cheng H. Ca2+ signalling between single L-type Ca2+ channels and ryanodine receptors in heart cells. Nature. 2001;410:592–596. doi: 10.1038/35069083. [DOI] [PubMed] [Google Scholar]
- [49].Sheard TMD, Hurley ME, Colyer J, White E, Norman R, Pervolaraki E, Narayanasamy KK, Hou Y, Kirton HM, Yang Z, Hunter L, et al. Three-dimensional and chemical mapping of intracellular signaling nanodomains in health and disease with enhanced expansion microscopy. ACS Nano. 2019;13:2143–2157. doi: 10.1021/acsnano.8b08742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Shen X, van den Brink J, Hou Y, Colli D, Le C, Kolstad TR, MacQuaide N, Carlson CR, Kekenes-Huskey PM, Edwards AG, Soeller C, et al. 3D dSTORM imaging reveals novel detail of ryanodine receptor localization in rat cardiac myocytes. J Physiol. 2019;597:399–418. doi: 10.1113/JP277360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Soeller C, Crossman D, Gilbert R, Cannell MB. Analysis of ryanodine receptor clusters in rat and human cardiac myocytes. Proc Natl Acad Sci. 2007;104:14958–14963. doi: 10.1073/pnas.0703016104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Hayashi T, Martone ME, Yu Z, Thor A, Doi M, Holst MJ, Ellisman MH, Hoshijima M. Three-dimensional electron microscopy reveals new details of membrane systems for Ca2+ signaling in the heart. J Cell Sci. 2009;122:1005–1013. doi: 10.1242/jcs.028175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Kettlun C, González A, Ríos E, Fill M. Unitary Ca2+ current through mammalian cardiac and amphibian skeletal muscle ryanodine receptor channels under near-physiological ionic conditions. J Gen Physiol. 2003;122:407–417. doi: 10.1085/jgp.200308843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Gillespie D, Fill M. Intracellular calcium release channels mediate their own countercurrent: the ryanodine receptor case study. Biophys J. 2008;95:3706–3714. doi: 10.1529/biophysj.108.131987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Cheng H, Lederer WJ. Calcium Sparks. Physiol Rev. 2008;88:1491–1545. doi: 10.1152/physrev.00030.2007. [DOI] [PubMed] [Google Scholar]
- [56].Inoue M, Bridge JHB. Variability in couplon size in rabbit ventricular myocytes. Biophys J. 2005;89:3102–3110. doi: 10.1529/biophysj.105.065862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Kolstad TR, van den Brink J, MacQuaide N, Lunde PK, Frisk M, Aronsen JM, Norden ES, Cataliotti A, Sjaastad I, Sejersted OM, Edwards AG, et al. Ryanodine receptor dispersion disrupts Ca2+ release in failing cardiac myocytes. ELife. 2018;7:e39427. doi: 10.7554/eLife.39427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Hou Y, Laasmaa M, Li J, Shen X, Manfra O, Nordén ES, Le C, Zhang L, Sjaastad I, Jones PP, Soeller C, et al. Live-cell photoactivated localization microscopy correlates nanoscale ryanodine receptor configuration to calcium sparks in cardiomyocytes. Nat Cardiovasc Res. 2023;2:251–267. doi: 10.1038/s44161-022-00199-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Dibb KM, Louch WE, Trafford AW. Cardiac transverse tubules in physiology and heart failure. Annu Rev Physiol. 2022;84:229–255. doi: 10.1146/annurev-physiol-061121-040148. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this article will be shared on reasonable request to the authors.