Abstract
The use of natural compounds for treating chronic bone diseases holds remarkable potential. Among these therapeutics, naringin, a flavanone glycoside, represents one of the most promising candidates due to its multifaceted effect on bone tissues. This review provides an up-to-date overview on naringin applications in the treatment of bone disorders, such as osteoporosis and osteoarthritis, and further highlights its potential for stem cell pro-osteogenic differentiation therapies. A critical perspective on naringin clinical translation is also provided. Such topic is discussed in light of recently developed biomaterial-based approaches that potentiate its bioavailability and bioactivity. Overall, the reported pro-osteogenic, anti-resorptive and anti-adipogenic properties establish this flavanone as an exciting candidate for application in bone tissue engineering and regenerative medicine.
Keywords: Biomaterials, Bone diseases, Naringin, Osteogenesis, Osteoporosis, Tissue engineering
Introduction
Currently bone diseases and injuries represent a significant healthcare burden at a worldwide scale due to the ineffectiveness of currently applied medical treatments for various skeletal disorders [1]. In fact, to date, a fully curative treatment for diseases such as osteoporosis, osteoarthritis, osteomyelitis or Paget’s disease has yet to be developed. Such reality is further accentuated in what concerns critical size bone injuries that are unable to self-regenerate [2]. For these cases, current strategies involve the use of bone grafts with embedded osteogenic growth factors to accelerate bone healing [2]. However currently used osteogenic molecules are frequently associated with high production costs or deleterious side-effects limiting wide applicability and therapeutic efficacy [3]. In an effort to discover new approaches that increase the toolbox of effective treatments for bone pathologies, a great focus has been put on the pursuit of natural-based products due to their availability, cost-effectiveness and biological activity.
Plant-based derivatives and marine-derived compounds represent one of the most cost-effective sources of new bioactive molecules with promising therapeutic effects for different diseases including those of skeletal tissues [4]. In this context, various natural formulations (e.g. Drynaria fortunei or Erythrina variegata) have presented a remarkable potential for improving joint-related (e.g. osteoarthritis) osteoarticular degradation, or for treating bone injuries and bone-related disorders, such as osteoporosis [4,5]. Typically, these formulations can modulate multiple signaling pathways and exert an effect in different cellular targets [6]. Such is extremely valuable in the context of bone diseases considering their multi-factorial pathogenesis, particularly in the case of osteoarthritis and osteoporosis [7]. In addition, their capacity for improving osteoporotic bone healing is highly related to its pro-osteogenic effect in both osteoprogenitor cells as well as stem cells [4]. Hence, research interest in this pro-osteogenic potential has been steadily growing for applications in stem cell-based therapies for tissue engineering.
Currently, the application for these naturally small molecules in bone is particularly focused in osteoporosis and on the pro-osteogenic differentiation of mesenchymal/stromal stem cells. Regarding the latter, such bioinspired approaches have recently shown promising pro-osteogenic potential for tissue engineering and regenerative medicine applications [4,8].
For example, a catechin hydrate-coated substrate markedly enhanced the osteogenic differentiation and mineralization of hASCs [9]. In addition, catechin-coated polycaprolactone nanofiber scaffolds implanted in a critical-sized calvarial defect prompted a significant improvement in bone formation and density, as well as collagen deposition [9]. Another natural compound, icariin, a prenylated flavonol glycoside, has exhibited equally promising properties for bone tissue engineering [4]. In a mouse calvarial defect model, icariin was transplanted in calcium phosphate cement and resulted in enhanced bone and blood vessel formation [10]. Other in vivo studies have reported significant cartilage repair in mice bearing osteochondral defects, as well as bone formation and anti-adipogenic behavior ameliorating osteoporotic status of ovariectomized mice [11,12].
In addition, resveratrol, formononetin and ginsenosides have all markedly improved in vivo bone formation and mineralization, in both defect and ovariectomized animal models [4]. Also, oleanolic acid as well as green tea polyphenols have exhibited beneficial effects by reducing bone erosion and inflammation-induced bone loss in vivo [4,13].Moreover, oral administration of quercetin-loaded solid lipid nanoparticles increased quercetin levels by 3.5-fold of free drug and effectively reversed the osteoporotic status of ovariectomized rats to Sham levels [14].
Therefore, current research into natural small molecules suggests that they are a desirable source of potentially innovative pharmaceutics. In the toolbox of naturally available compounds for treatment of bone disorders, naringin, a flavanone glycoside, is currently gathering special attention in pre-clinical studies and represents one of the most promising candidates for treating various bone diseases and promoting stem cells differentiation towards a pro-osteogenic phenotype.
Naringin – Flavonoid with Multiple Therapeutic Targets in Bone Tissues
Naringin, also known as naringenin 7-O-neohesperidose, is a natural flavonoid present in several fruits of the Citrus genus. Commercial grapefruit (Citrus paradisi) juice is the richest source of naringin (43.5 mg / 100 mL), where this compound is significantly more concentrated than in their hand-squeezed equivalents (23.0 mg / 100 mL) [15]. Similarly, industrial bergamot (Citrus bergamia) juice represents another valuable source of this compound, with naringin contents around 26.1 mg / 100 mL [15]. Furthermore, this compound is present in moderate quantity in Citrus aurantium (1.97 mg / mL) and even in certain bitter commercial orange juices (2.13 mg / 100 mL) [15]. In addition, naringin is also considered the main effective component in the basket fern Drynaria fortunei, a traditional Chinese medicine for osteoporosis [16].
Naringin-enriched natural-sources have been found to hold remarkable potential for various biomedical applications. In fact, Citrus paradisi juice has been described to increase the bioavailability of various drugs by decreasing first-pass metabolism, either via inhibition of cytochrome P-450 3A4 drug-metabolizing intestinal enzyme, or by inhibiting the P-glycoprotein-induced efflux from the enterocytes [17]. Industrial bergamot juice has also shown antiproliferative activity in human hepatocellular carcinoma (HepG2 cells) and plays a protective role in the treatment of rheumatoid arthritis due to its anti-oxidant and anti-inflammatory activity. Interestingly it is also capable of mimicking the mechanism of statins, exerting significant hypolipidemic and hypocholesterolemic effects in humans [18–20].
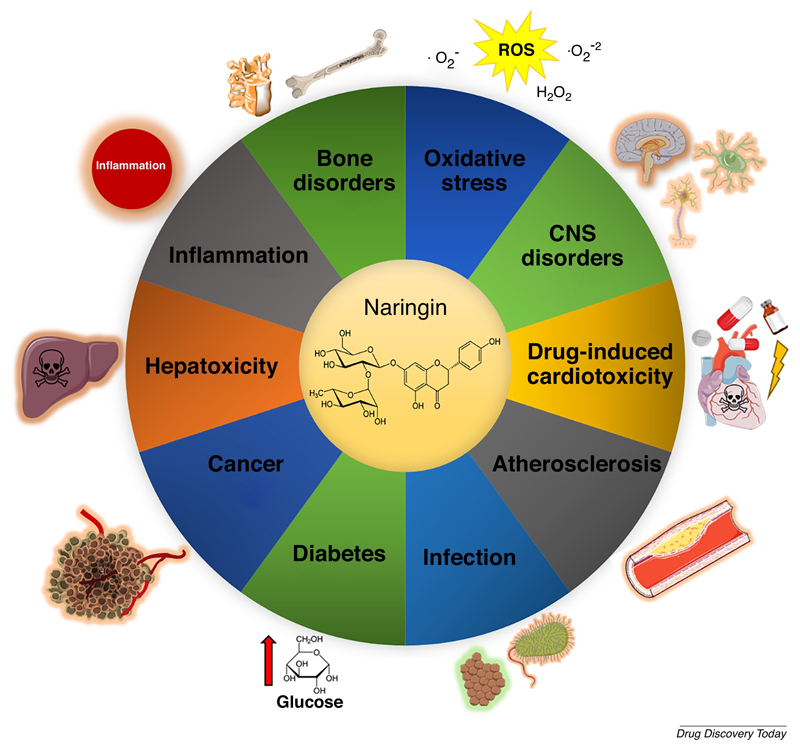
Naturally, these findings raised attention towards understanding the molecular effects of the main bioactives present in these sources, such as the flavanone naringin. Since, numerous pre-clinical studies investigating naringin bioactivity highlighted promising applications in different diseases (Figure 1) [16,21].
Figure 1.
Overview of various conditions in which naringin has been described to display protective / therapeutic effects. For a more general description of the activity of naringin in these conditions, please see reference [21]. ROS – reactive oxygen species. CNS – central nervous system.
Among the different applications of naringin, recent research has been focusing on the pro-osteogenic effects of naringin in osteoporosis, or as a naturally-inspired compound for directing mesenchymal/stromal cells osteogenic differentiation. This unique biological activity in the context of bone disorders will be further discussed in the context of recent in vitro and in vivo pre-clinical reports.
Pre-clinical in vitro studies
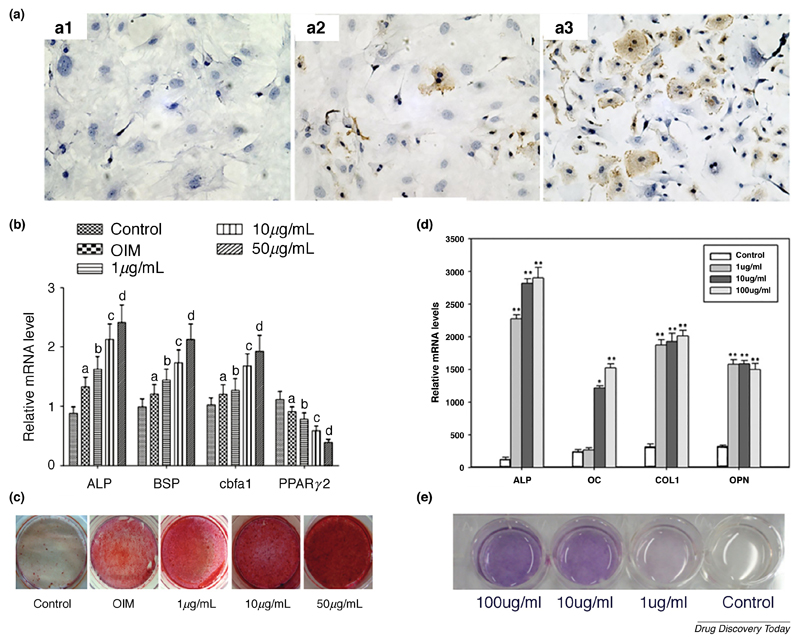
The pro-osteogenic effect of naringin is well described in the literature, suggesting potential applications as bone therapeutic or as mediator of mesenchymal stem cells (MSCs) osteogenic lineage differentiation [16]. Different studies have shown that naringin has a significant effect in the proliferation or differentiation of osteoprogenitor cells such as murine pre-osteoblasts (MC3T3-E1) and cells with osteoblastic phenotype, including human and murine primary fetal osteoblasts (respectively, hOB and pOB) [22,23]. Fan and his team found that naringin dose-dependently improved the proliferation of rabbit bone marrow-derived MSCs (BM-MSCs), and that naringin (1 µM) significantly increased the mRNA expression of osteocalcin (OC), alkaline phosphatase (ALP) and collagen type I (Col I) across studied time points (3, 7, 14, 21 days) [24]. Moreover, the authors observed that naringin improved cells osteogenic commitment by inhibiting peroxisome proliferator activated receptor ɣ (PPARɣ) expression, a key regulator in promoting MSCs adipogenesis. The naringin-induced downregulation of PPARɣ levels was linked to the corresponding upregulation of microRNA-20a expression [24]. In fact, the effect of naringin on the expression of the previous markers (OC and PPARɣ) was effectively reversed by transfecting the BM-MSCs with anti-microRNA-20a antibody. Alternatively, in rat BM-MSCs, naringin increased the proliferation over 9 days in a dose dependent manner (up to 10 µg/mL) [25]. In these cells, the highest dose (100 µg/mL) was shown to decrease proliferation over long culture periods (9 days). After naringin treatment, there was a 5 to 7-day delay before ALP expression peak was observed, where the dose of 10 µg/mL exhibited the best osteogenic performance. These findings were supported by OC cell immunostaining after naringin treatment, in which 10 µg/mL led to the highest increase in OC expression (Figure 2A). Another study performed by Yu and co-workers further expands the knowledge regarding naringin-mediated activation of signaling pathways that are related to proliferation or osteogenesis [26]. These researchers found that naringin promotes rat BM-MSCs osteogenesis via activation of the Notch signaling pathway [26]. Naringin significantly enhanced the mRNA levels of osteogenic markers (ALP, bone sialoprotein (BSP) and core-binding factor a1), while it decreases adipogenic regulator (PPARɣ2) both in a dose-dependent manner (Figure 2B). In addition, increased calcium node deposition in cultured cells also followed a dose-dependent behavior (Figure 2C). Interestingly, treatment with Notch-inhibitor DAPT partly reduced naringin-induced ALP activity stimulation, calcium deposits and osteogenic mRNA transcript levels, whilst suppressing the inhibitory effect on PPARɣ2. Such findings strongly support the use of naringin as a pro-osteogenic natural compound. This is highly important in the context of stem cell-based therapies since up to date the main pharmaceutical approach has been the use of dexamethasone to guide lineage differentiation [27]. However, this glucocorticoid has been described to strongly induce adipogenesis even in osteogenic medium, which can be counterproductive for the optimizing the effectiveness of such therapies [28]. The described simultaneous anti-adipogenic and osteogenic activity of naringin, suggests a tremendous potential of this natural compound for stem cell-based therapies.
Figure 2.
Naringin increases the expression of osteogenic markers, leading to an enhanced mineralization. (A) OC immunostaining of rat BM-MSCs after induction with different naringin doses for 6 days, A1 – control, A2 – 1 µg/mL, A3 – 10 µg/mL. (B) RT-PCR gene expression analysis of osteogenic markers after a 14-day treatment of rat BM-MSCs with different naringin doses. Data is represented in mean ± s.d. (n=5). ap < 0.05 versus control group; bp < 0.05 versus the OIM group; cp < 0.01 versus the 1 µg/mL group; dp < 0.01 versus the 10 µg/mL. (C) Alizarin Red S staining of calcium deposits formed in rat BM-MSCs after incubation with various doses of naringin (1, 10 and 50 µg/mL) for 21 days. (D) RT-PCR gene expression analysis of osteogenic markers after a 7-day treatment of human BM-MSCs with different naringin doses. Data represented in mean ± s.d. (n=6). *p < 0.05 and **p < 0.01 compared with the control group (n=6). (E) ALP staining of human BM-MSCs 7 days after drug administration. Adapted from references [25,29] with permission from Elsevier. Reference [26] adapted under a Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0/).
In the context of stem cell-based bone therapies the osteogenic effect of naringin was also assessed in human BM-MSCs. Peng-Zhang and his team observed a dose-dependent proliferative and osteogenic activity of naringin in human BM-MSCs up to 100 µg/mL [29]. The effect of the highest doses (10 and 100 µg/mL) on ALP activity was significant after only a 24 h incubation period and increased over 7 days. In addition, all naringin-treated groups exhibited a remarkable improvement in the expression of osteogenic markers ALP, OC, Col I and osteopontin (OPN) (Figure 2D). Moreover, Von Kossa staining of calcium nodes and ALP staining following naringin treatment further supported the dose-dependent effect of this flavonoid (Figure 2E).
Numerous studies have investigated the role of different signaling pathways in determining the osteogenic activity of naringin in other cell types, such studies are important since different cell types can respond differently to the flavonoid [26,29,30]. Liu and co-workers found that naringin-induced osteogenic activity in human amniotic fluid-derived stem cells (hAFSCs) was related to stimulation of the bone morphogenetic protein (BMP) and Wnt/β-catenin signaling pathways [31]. These pathways play a crucial role in osteogenesis regulation, adipogenesis repression and prevention of osteoblastic apoptosis [32]. Similar to previous studies, the authors demonstrated a dose-dependent increase in proliferative and osteogenic activities of this flavonoid (1 to 100 µg/mL), but not at the highest dose (200 µg/mL). Accordingly, calcium content after 28 days of naringin treatment was markedly increased at 100 µg/mL. Reverse transcription polymerase chain reaction (RT-PCR) gene expression analysis of naringin-treated cells revealed significant upregulation of osteogenic marker genes (OPN and Col I) and a remarkable increase in osteoprotegerin (OPG) expression., once again suggesting a dual-effect of naringin in both promoting osteogenic proliferation/differentiation while inhibiting osteoclastogenesis. Interestingly, the expression levels of BMP-related regulators (RUNX2 and BMP-4), as well as Wnt-related genes (β-catenin and cyclin D1) were upregulated in naringin-treated cells, and the ALP activities were significantly reduced in the presence of inhibitors for these pathways (accordingly, noggin for BMP and DKK-1 for Wnt-signaling).
Also, Pang and colleagues confirmed the significant osteogenic role of naringin in UMR 106 osteoblast-like cells via estrogen receptor (ER)-dependent pathways [33]. Importantly, this study demonstrated that naringin exerts tissue-selective oestrogenic effects on bone and possibly in adipose tissue, but not in the uterus. It was proposed that this selective behavior is determined by differential phosphorylation of ERα and ERE-dependent transcriptional activity. In addition, the authors observed that naringin (10 nM) markedly enhanced OPG mRNA expression, and that this effect was reversed in the presence of an ER-antagonist (ICI 182780). OPG secretion is linked to inhibition of osteoclastogenesis, which suggests a potential anti-resorptive capacity of naringin.
In fact, besides its known pro-osteogenic effect, naringin has demonstrated to be capable of inhibiting bone resorption in vitro via osteoclasts [30,34]. In this case, the anti-inflammatory effect of naringin appears to play a role in its anti-resorptive activities. Such is supported by OPG and NF-κB pathway involvement in osteoclastogenesis [5].
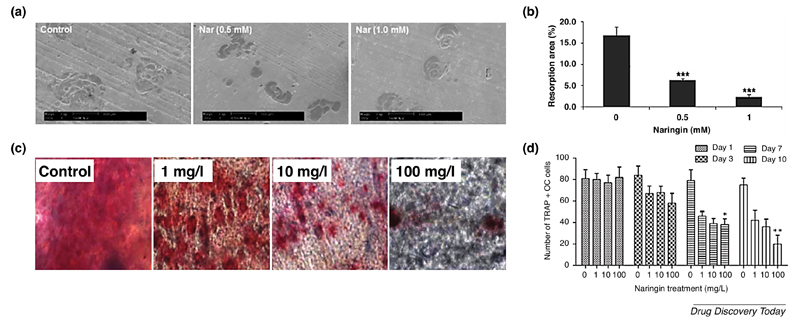
In this context, Ang and co-workers observed that naringin inhibits osteoclastogenesis and bone resorption by suppressing RANKL-induced NF-κB activation and phosphorylation of extracellular receptor kinase (ERK) [30]. Notably, the flavonoid was able to reduce the bone resorption area whilst maintaining the cell number of osteoclast-like cells (Figure 3A, B). It is also worth noting that murine macrophage RAW 264.7 cells, which were used to generate osteoclast-like cells, showed a remarkable tolerance against naringin dosages, with only 10% apoptosis after a 24 h treatment with 1 mM of naringin (approximately 580 µg/mL). Nevertheless, another study achieved significant bone resorption inhibition at much lower dosages of the flavonoid [34]. In this study, Xu and colleagues investigated the naringin effect in repressing osteoclastogenesis of a rat calvarial bone culture, after incubation with different naringin doses (1, 10 and 100 mg/L). The tartrate-resistant alkaline phosphatase (TRAP) staining of osteoclasts after treatment with different naringin doses for 10 days revealed a dose-dependent inhibition of osteoclastogenesis (Figure 3C). Moreover, naringin markedly suppressed osteoclastogenesis in a time-dependent manner, from 1 to 10 days of in vitro 2D culture (Figure 3D). After incubation with different naringin concentrations, the authors observed a 74 % (100 mg/L), 52 % (10 mg/L) and 41 % (1 mg/L) reduction in the number of TRAP-stained osteoclasts.
Figure 3.
Naringin effectively inhibits bone resorption in vitro. (A) Representative scanning electron microscopy (SEM) micrographs of bone resorption pits induced by osteoclast-like cells in bovine bone slices after incubation with different naringin doses (0, 0.5 and 1 mM) for 24 h. (B) Total resorption pit areas of each treatment group as measured under SEM. (C) Microscopic view of TRAP-stained osteoclasts in calvarial bone cultures treated with different doses of naringin for 10 days. (D) Number of TRAP-stained osteoclast cells treated with different doses of naringin for 1, 3,7 and 10 days. *p < 0.05, **p < 0.01, ***p < 0.001. All data above is represented in mean ± s.d. (n=3). Adapted from reference [30] with permission Wiley. Adapted from reference [34] under a Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0/).
The above findings demonstrate that naringin is capable of significantly repressing osteoclastogenesis and reduce bone resorption areas. This is an important finding for applications in bone degenerative disorders, such as post-menopausal osteoporosis, characterized with osteolytic degradation due to markedly increased bone turnover. Taken together, the pre-clinical in vitro studies highlight a tremendous potential for naringin applications as a bone therapeutic and for committing stem cells into the osteoblastic lineage.
Pre-clinical in vivo studies
The former examples provide an important body of knowledge regarding the potential of naringin for treating different bone disorders. Adding to this, various in vivo studies that will be discussed in the following paragraphs provide further evidence of naringin realistic potential to be used as therapeutic alternative in a foreseeable future.
Naringin has improved overall bone health in both healthy and gonadectomized animal models (such as rat and mouse) in vivo. In healthy mice, naringin daily oral administration significantly enhanced femoral bone mass by increasing both trabecular and cortical bone [35]. In another study in healthy mice, Yin and co-workers achieved the first validation of successful stem cell-based therapy involving naringin for improving bone formation in vivo. Initially, human periodontal dental ligament stem cells (hPDLSCs) were seeded in a nanohydroxyapatite scaffold and cultured in naringin-containing (1 µM) medium for 1 week, before implantation into healthy mice. The transplant was harvested 8 weeks later and naringin-treated group exhibited improved trabecular bone maturity surrounding the scaffold, as well as locally increased OPN and OC expression by 50 % [36].
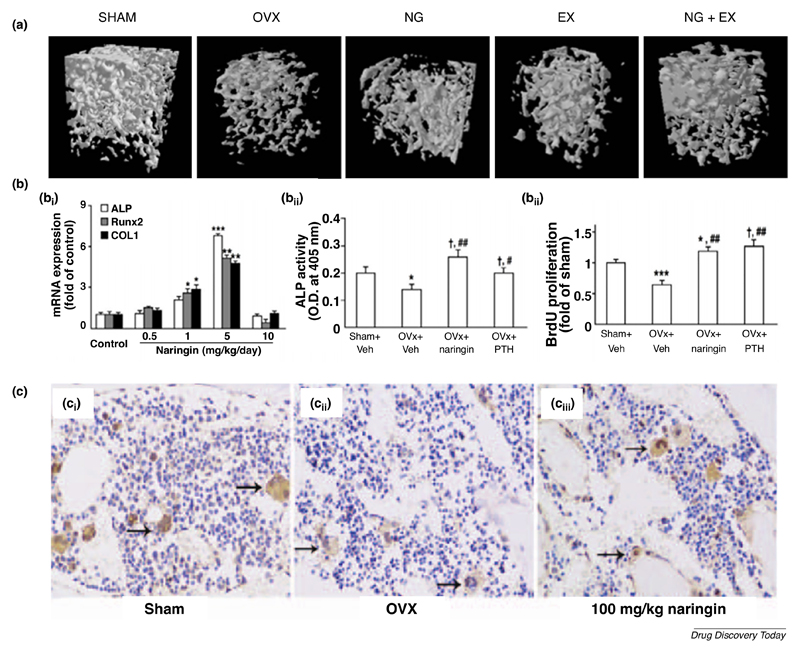
Currently, most in vivo studies involving naringin have been performed in ovariectomized mice. Pang and colleagues demonstrated that treatment of ovariectomized mice with naringin (200 and 400 mg/kg per day) for 6 weeks significantly improved bone quality at the distal femur, proximal tibia and lumbar spine [33]. In addition, naringin suppressed the ovariectomized-induced increase in urinary calcium excretion as well as losses in bone mass and strength. However, in this study, naringin treatment failed to significantly decrease urinary deoxypyridoline level in ovariectomized mice, a collagen degradation product that reflects bone resorption rate. In another study, ovariectomized mice were daily treated with various naringin doses (60, 300 and 1500 mg/kg) via oral gavage, leading to effective recovery of ovariectomized-induced bone loss [25]. The authors found that naringin at 300 mg/kg provided an optimal increase in bone mass density (BMD), bone volume as well as trabecular thickness, while decreasing trabecular space. Furthermore, naringin treatment did not change the uterus weight significantly, suggesting that naringin did not elicit off-target estrogenic effects. Interestingly, a study by Sun and colleagues investigated the effect of a combination regimen of oral naringin (300 mg/kg) with treadmill exercise in ovariectomized rats for 60 days [37]. Authors found that naringin + exercise regime led to stronger effects on osteoporosis than either monotherapy on bone mass preservation and improved bone strength (Figure 4A). In a different report, Wang and colleagues observed an improved bone strength in ovariectomized mice even at lower doses (5 mg/kg) of naringin (Figure 4B1) [38]. This dose markedly improved ALP, RUNX2 and Col I expression in vivo (Figure 4B2). In particular, the authors observed that co-administration of AMPK and Akt inhibitors partly reversed naringin effects in vivo, suggesting that the osteogenic activity of this flavonoid is in part via its stimulation of the Wnt/β-catenin signaling upon interaction with AMPK and Akt. Moreover, ovariectomized mice achieved equally increased cell proliferation when treated with naringin or conventional parathyroid hormone, but naringin-treated mice were characterized with the highest enhancement in ALP activity (Figure 4B3). Adding to this evident potential, a recent study found that naringin (100 and 200 mg/kg) significantly inhibited the ovariectomized-induced reduction in bone marrow microvessels, regulating the function of endothelial cells while promoting angiogenesis in bone (Figure 4C) [39]. In parallel, oral administration of naringin (300 mg/kg) has recently been shown to augment the vascularization of the callus in osteoporotic fractures in ovariectomized rats, by significantly improving the expression of VEGF and VEGF receptor-2 [40]. This lead to an increase in vessel numbers and thickness, as well as larger neovascularization areas. Overall, this strategy resulted in accelerated bone healing at 2, 4 and 8 weeks post-fracture, in a dose dependent manner (40, 100 and 300 mg/kg). The development of bone vasculature is particularly relevant for both the treatment of osteoporosis but also in the context of tissue engineering and regenerative medicine [41]. In particular, angiogenesis is fundamental for engineering a clinically relevant-sized tissue, which requires a vascular network for properly supplying cells beyond the diffusional limit for oxygen and nutrients [42].
Figure 4.
Pro-osteogenic and pro-angiogenic protective activities of naringin in vivo. (A) 3D reconstruction of trabecular microarchitecture within the distal femoral metaphyseal region in Sham, ovariectomized (OVX), naringin (NG) or exercise (EX) monotherapy or combination regimen groups. (B1) Optimal naringin-induced osteogenic gene (ALP, RUNX2, Col I) expression at 5 mg/kg (n=5). *p < 0.05; **p < 0.01; ***p <0.001 compared to control group. (B2, B3) Naringin-induced (5 mg/kg) increase in ALP activity and osteoblastic cell proliferation. Data represented in mean ± SEM (n=5 and n=3, respectively). #p < 0.05; ##p < 0.01 compared with the OVX group. (C) Naringin markedly inhibited (100 mg/kg) the OVX-induced reduction in bone marrow microvessels (black arrows). Adapted from reference [37] under a Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0/). Adapted from reference [38] under a CC-BY-NC license. The final, published version of this article is available at http://www.karger.com/?doi= 10.1159/000430319. Adapted from reference [39] under a CC BY-NC-ND 4.0 International license.
These findings regarding the pro-angiogenic activity of naringin are supported by the previous in vitro study demonstrating naringin-induced osteogenic differentiation of BM-MSCs via activation of the Notch signaling pathway [26]. Endothelial Notch activity plays a key role in simultaneously promoting bone angiogenesis and osteogenesis and therefore, could be one of the main mechanisms behind the osteoprotective effect of naringin [43].
Alternatively, other studied animal models support the above findings regarding the clinical potential of naringin in improving bone health status. Wei and co-workers demonstrated the anti-osteoporotic activity of naringin in a retinoic acid-induced osteoporosis rat model [44]. Naringin treatment with different doses (20, 40, 100 mg/kg) led to improvements in bone weight index, length and diameter of the femur bone, as well as bone ash content and levels of calcium and phosphorus. In orchidectomized rats, treatment with naringin at 200 ppm for 2 months significantly increased serum IGF-I, femoral bone density and calcium content, as well as suppressed plasma TRAP activity, associated with bone resorption [45]. Alternatively, subcutaneously administered naringin (10 mg/kg) is also described to promote bone formation in a titanium particle-induced diabetic murine calvarial osteolytic model [46]. Moreover, naringin embedded in a collagen bone graft also promoted bone formation in a rabbit bone defect model [47].
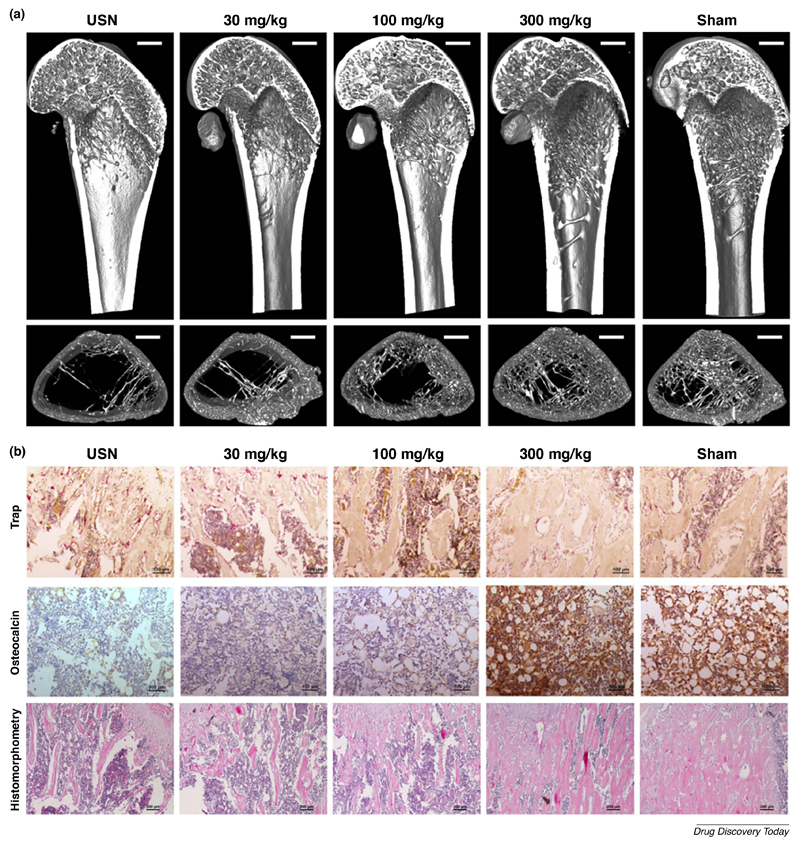
Naringin exhibited the most therapeutic potential in disuse-induced osteoporosis animal models, caused by mechanical unloading and characteristic in bedridden or low-mobility patients, as well as astronauts [48]. A study by Ma and co-workers investigated naringin-treatment in denervated bone induced by sciatic neurectomy [49]. Disuse-induced bone loss in rats was induced by sciatic neurectomy, resulting in reduced BMD and trabecular microarchitecture in the distal femur, as well as increased urinary deoxypyridoline levels. The authors confirmed the dose-dependent (30, 100, 300 mg/kg) recovery induced by naringin treatment in restoring trabecular microarchitecture as well as bone formation rates to Sham-levels (Figure 5A). Moreover, the highest naringin dose had a profound effect in improving osteogenesis and inhibiting osteoclastogenesis in vivo, as indicated by OC and TRAP immunohistochemistry analysis in the distal femoral metaphysis (Figure 5B), as well as reducing urinary deoxypyridoline. Naringin-treatment also successfully prevented biomechanical deterioration of ipsilateral femur in immobilized rats (Figure 5B). This protective effect is thought to be modulated via naringin-induced increase in Semaphorin 3A expression in vivo, a local factor of the bone microenvironment that simultaneously promotes bone formation while reducing bone resorption [49]. However, Semaphorin 3A deficiency is directly implicated in the disruption of bone remodeling and subsequent bone loss in diabetic rats [50]. This might explain why naringin successfully prevented bone loss in a rat model of streptozotocin-induced diabetes [51]. Considering this, these studies suggest that naringin could be a promising therapeutic alternative in treating disuse osteoporosis and for improving bone health in patients with diabetes mellitus type 1.
Figure 5.
Naringin therapeutic activity in disuse osteoporosis induced by unilateral sciatic neurectomy (USN). (A) Representative 3D images showing the trabecular microarchitecture in the distal femoral metaphysis of each group. (B) Immunohistochemical staining of OC and osteoclasts (TRAP+) in the distal femoral metaphysis, as well as histomorphometric analysis of naringin-prevented deterioration of the ipsilateral femurs due to immobilization. Adapted from reference [49] under a Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0/).
Naringin has also shown particular promise in treating osteoarticular degenerative diseases, such as osteoarthritis, ankylosis spondylitis and autoimmune arthritis. In such disorders, the characteristic destruction of inflamed cartilage tissue significantly affects the surrounding bone tissues and heavily impairs patient quality of life.
Naringin was used in a recent study that involved a mice model of ankylosis spondylitis. Analysis of harvested tissues showed that flavonoid intraperitoneal administration attenuated the expression of pro-inflammatory cytokines (such as TNF-α, IL-1β and IL-6) and inhibited the production of STAT3 and JAK2 (implicated in the sensitivity to this disease), in a dose-dependent manner [52]. In parallel, naringin also improved oxidative stress markers in treated mice by restoring the activity of superoxide dismutase, catalase and glutathione peroxidase in vivo. Recently, orally-administered naringin has also shown remarkable potential in preventing cartilage destruction in both surgically and monosodium iodoacetate-induced osteoarthritis models in vivo [53,54]. In these studies, naringin has been reported to improve the weight-bearing ability of osteoarthritic rats, as well as inhibit key enzymatic mediators of osteoarthritis progression (such as MMP-13 and ADAMTS-5) in affected mice.
In addition, some studies link the anti-arthritic activity of naringin to the regulation of inflammatory proteins and cell mediators responsible for facilitating joint cellular infiltration [55,56]. Indeed, in a mice model of autoimmune arthritis, naringin was able to modulate T regulatory cells and significantly upregulated IL-4 expression, which is described to ameliorate cartilage proteoglycan depletion induced by pro-inflammatory cytokines [55]. Moreover, in a collagen-induced arthritis mice model, naringin oral treatment significantly mitigated intercondylar knee joint damage, pannus formation and synovium infiltration [56].
These studies highlight the potential of naringin for therapeutic applications in osteoarticular degenerative diseases, yet there are still few studies in the literature exploring this promising phytotherapeutic alternative. In fact, beyond the classic anti-inflammatory and anti-oxidant activities implicated in the therapeutic potential of this flavanone in osteoarthritis, there are other interesting targets such as estrogen receptors that are currently linked to osteo- and rheumatoid arthritis therapeutic alternatives [57,58]. As explained earlier, the interaction between naringin and estrogen receptors is well explored in the context of osteoporotic scenarios, but this is still undervalued in the context of osteoarticular therapies.
Overall, the above pre-clinical studies provide significant evidence for naringin multifactorial osteostimulative and chondroprotective effects, either in bone fracture healing or for bone erosive and osteoarticular degenerative disorders.
However, so far, naringin has been mostly explored for treating bone degenerative disorders. It is worth noting that bone remodeling is a product of concomitant interaction between bone formation and resorption, meaning that disruption of this balance leads to bone dysfunction [49]. In this context, clinically available therapeutics such as antiresorptive bisphosphonates and anabolic parathyroid hormone can reduce bone formation and promote bone resorption, respectively, which might explain why combination therapy approaches between clinical therapeutics or with mechanical-loading have been investigated with the aim of improving bone quality [59,60]. Furthermore, long-term disuse osteoporosis has been described as less sensitive to bisphosphonate treatment than other types of osteoporosis [61]. Alternatively, bone formation is significantly reduced with ageing, mostly attributed to a shift from osteogenesis to predominant adipogenesis in the marrow stroma, leading to senile osteoporosis [62]. Naringin treatment could aid in revascularizing the affected skeletal sites and ultimately rejuvenate such microenvironment to a healthier phenotype.
Taken together, the simultaneous antiresorptive, osteostimulative and antiadipogenic role of naringin ultimately play a role in improving bone health and establish this flavonoid as a valuable candidate for pursuing alternative clinical treatment options for some of the most common bone disorders or injuries due to fractures.
Current obstacles in naringin clinical translation
Despite its promise for therapeutic action in several pathologies, in particular for preventing degenerative diseases and improving bone health status, naringin is yet to be approved for clinical administration, either as a single therapy or in combination with other bioactives. This fact is mainly correlated with flavonoids extensive in vivo metabolism, a critical factor that limits their therapeutic efficacy [63]. In addition, naringin exhibits low bioavailability (8.8 %) following oral administration due to its poor water solubility and dissolution rates [64,65]. In fact, the poor water solubility of naringin is considered the rate-limiting step for its absorption in the body, thus leading to inferior therapeutic efficacy [65]. Moreover, this drug is extensively degraded in acidic pH and enzymatically cleaved by β-glucosidases in the gut, inherent to intestinal microflora [66].
The effects of naringin have been mainly explored via oral absorption, however its absorption in the gastrointestinal tract is slow and irregular [67]. Moreover, the intestinal microbiota plays a crucial role in defining the bioavailability of flavonoids such as naringin. It is important to emphasize that this microbiome is characterized by substantial inter-individual heterogeneity, hence, ultimately the microbiome defines the clinical efficacy of dietary flavonoids [63]. On account of these limitations, recently there have been some in vitro attempts for improving flavonoids bioavailability and absorption, by increasing its solubility and dissolution rates, as well as protection from intestinal degradation upon encapsulation in nanoparticles, microparticles or water-soluble fibers [65,67–69]. It has been suggested that minimal absorption rates of such drugs can severely restrict their clinical applications [14].
Adding to this, naringin can also be degraded during blood circulation if administered via intravenous route. Indeed, flavonoids such as naringin are unstable during circulation and easily undergo oxidation both in serum, and in the liver, where they are generally degraded by hepatic β-glucosidases [66]. It is also reported that naringin spontaneously interacts with bovine serum albumin under physiological conditions, an aspect that can play a major role in defining its pharmacokinetic profile, facilitating excretion and hence influencing its bioavailability [70]. Such parameter is also influenced by the decreased solubility of flavanones under physiological conditions (pH = 7.4), due to degradation into chalcone structures [65]. Yet so far, no attempts have been made towards formulating oral or intravenously administrable nanocarriers for the controlled delivery of naringin to skeletal sites. As recognized from previously highlighted studies with other natural compounds (e.g. quercetin), controlled delivery via nanocarriers can significantly improve their in vivo therapeutic effect [14]. There have been some recent developments concerning the formulation of naringin within suitable carriers for oral absorption, but research is still at an early stage and studies are yet to validate this approach in vivo. Concerning the parenteral route of administration, literature is still scarce and there is a great untapped potential to be exploited on this approach.
Nanotechnology-based drug delivery systems allow for the improvement of solubility, bioavailability and pharmacokinetics of entrapped pharmaceutics, protecting them from degradation and unspecific interactions while prolonging their circulation times [71,72]. Furthermore, these nanocarriers can provide a sustained release profile and can be modified with specific targeting moieties for improving accumulation at the desired locations [73]. Hence, the bioavailability of naringin could be vastly improved by inclusion in a nanocarrier, ultimately leading to locally increased concentrations in the bone microenvironment. Recognizing the potential of nanotechnology for flavonoid delivery, the following chapter will explore current approaches in the literature concerning the incorporation of naringin within biomaterial-based nano/microplatforms with potential skeletal delivery.
Biomaterial-based platforms for naringin incorporation
As aforementioned, naringin has low bioavailability and undergoes extensive metabolism in vivo. Such has motivated researchers to explore biomaterial-based platforms for immobilizing or protecting naringin from degradation, and for achieving a sustained spatiotemporally controlled release with the aim of improving its therapeutic effect.
Regarding the production of naringin-biomaterial hybrids, Ji and co-workers incorporated the flavanone within an electrospun nanoscaffold comprised of polycaprolactone (PCL) and poly(ethylene glycol)-b-polycaprolactone (PEG-b-PCL) nanofibers (~ 242 nm) to serve as a bone regenerating implant [74]. PCL/PEG-b-PCL/naringin nanoscaffolds elicited an increased MC3T3-E1 proliferation, as well as enhanced osteogenic differentiation (evaluated via ALP activity) after 14 days of culture in medium with no osteogenic supplements. Moreover, for cells cultured in these nanoscaffolds, Alizarin Red S staining showed improved calcium mineralization nodules after only 10 days. In addition, the authors studied the effect of the naringin nanoscaffold in suppressing osteoclastogenesis in a critical size defect model of mouse calvarial bone. After 14 days of implantation, the defects treated with PCL/PEG-b-PCL/naringin nanoscaffolds showed a significant decrease in TRAP staining when compared to treatment with blank PCL nanoscaffolds. These results corroborate naringin-induced osteoclastogenesis suppression and suggest potential applications of this drug-loaded nanofiber scaffold in bone tissue engineering. Regarding this application, Chen and co-workers developed a porous biodegradable composite comprised by genipin-crosslinked gelatin, and β-Ca3(PO4)2ceramic microparticles (GGT) mixed with naringin (10 mg/mL). These composites were formulated with the aim to enhance bone repair in vivo in a rabbit calvarial defect model [75]. The obtained radiographic analysis and histological H&E staining revealed that, 8 weeks post-implantation, naringin-loaded GGT composites promoted a significant deposition of new bone formation when compared to GGT controls. Moreover, complete osseointegration of the biodegradable implant could be readily observed, with newly formed bone replacing a significant amount of the naringin-loaded GGT composites.
Recently, Guo and colleagues developed a porous poly(l-lactide) (PLLA) scaffold incorporating anti-inflammatory drug parthenolide and spray-dried naringin-loaded chitosan microspheres embedded in the matrix [76]. The regenerative performance of this dual drug delivery scaffold was studied in a rat model of periodontal fenestration defects. Analysis of µCT data revealed that 8 weeks post-implantation, the dual delivery scaffold significantly enhanced bone volume and decreased inflammatory response in the defect, when compared to the PLLA group. Histological H&E analysis corroborated the superior performance of the dual delivery scaffold in improving periodontal tissue regeneration. Also, IL-6 immunostaining showed that the dual delivery scaffold achieved the least positive staining areas for the cytokine, highlighting its anti-inflammatory activity. The authors therefore suggest a possible application as an adjuvant for the treatment of periodontitis. However, it should be noted that MC3T3-E1 cell proliferation assays showed that both the dual delivery scaffold and the parthenolide-loaded group significantly decreased cells proliferation in comparison with the naringin-loaded group. These results suggest that this flavanone can partially rescue the observed cytotoxic effect of anti-inflammatory drug parthenolide.
In parallel, Chang and colleagues have recently developed a naringin-loaded (0.85 %) pH-responsive hydrogel for periodontitis treatment, taking advantage of the characteristic pH reduction hallmark in inflammation sites [77]. In this study, the authors formulated a hydrogel comprised by carboxymethyl-hexanoyl chitosan, β-glycerophosphate and glycerol via a thermally induced sol-to-gel transition. Notably, the hydrogel underwent instant sol-to-gel transition at 37 ºC. Interestingly, a 2-fold higher amount naringin was released in acidic conditions (pH = 5.5) when compared to physiological conditions (pH = 7.4), accordingly 51 % versus 24 % of total naringin released in the first 4 h. Here, a silk-induced acute periodontitis mice model was used to evaluate the hydrogel biological performance. The naringin-loaded hydrogel markedly attenuated local inflammation and periodontal breakdown after 7 days. Gene expression analysis also showed a remarkable downregulation of Toll-like receptor 2, TNF-α and RAGE expression, but not myd88, which is why the authors suggested that this therapeutic activity might be achieved via a LPS-mediated myd88-independent mechanism.
Yu and colleagues developed a mineralized collagen coating with embedded naringin-loaded metal-organic frameworks (MOFs) [78]. By taking advantage of the inherent antimicrobial and pro-osteogenic activities of this flavanone, the authors aimed to design a multifunctional osseointegrating orthopedic coating. Such collagen/MOFs/naringin coatings significantly improved MSCs cell adhesion over other control groups. Moreover, RT-PCR analysis of several osteogenic gene markers at 14 days of culture on different coating substrates showed improved expression of Col I, OC and RUNX2 for the collagen/MOFs/naringin group. In addition, Alizarin Red S staining indicated that this coating lead to the highest MSCs mineralization after 21 days of culture. Furthermore, the promising osteogenic activity of the collagen/MOFs/naringin coating is complemented with enhanced antibacterial activity against Staphylococcus Aureus at 3, 5 and 7 days of culture in comparison to other coating groups.
Alternatively, Feng and co-workers developed naringin-loaded water-soluble ternary nanoparticles comprised by amylose, α-linoleic acid and β-lactoglobulin as a delivery platform [79]. As determined by high-performance liquid chromatography, these carriers exhibited high naringin encapsulation efficiency (79.7 ± 4.2%) and may inspire further studies aiming to improve naringin bioavailability using food grade formulations. Moreover, naringin has also been entrapped within different spray-dried microparticles (e.g. gastro-resistant cellulose acetate phthalate and maltodextrin) with the aim of improving its dissolution rates and bioavailability upon oral delivery [67,68]. More recently, other studies focused on the incorporation of naringin in different nanocarriers, either as a stabilizing agent with gum tragacanth in gold nanoparticles or by studying its inclusion complex with food grade β-cyclodextrin [69,80].
Overall, these studies suggest promising future applications for naringin delivered through biomaterials. Yet, so far, delivery of naringin to skeletal sites has been mostly explored in the context of impregnation in implantable porous composites, polymeric scaffolds or in surface coatings [74–76,78]. Apart from such examples, no study has described the in vivo performance of naringin-loaded delivery systems. It is undoubtedly crucial to investigate whether incorporating naringin within such nano/microcarriers can augment this flavonoid bioactivity in different diseases. In addition, different bone disorders are characterized by specific pathophysiological conditions that represent unique exploitable opportunities for nanocarriers endowed with stimuli-responsiveness [81]. Harnessing such biological triggers to our advantage may further improve the current potential of bone therapeutics, either synthetic or naturally-available.
Conclusions and Future Perspectives
The search for safer and more suitable osteoinductive agents has lead researchers to explore the potential of natural-based compounds such as naringin. As highlighted by the numerous pre-clinical studies investigating this flavanone’s therapeutic activities, there is unquestionable potential in naringin application for bone diseases or for instructing stem cells osteogenic differentiation. Indeed, naringin has shown promise for applications in skeletal disorders for which current pharmaceutic strategies are lacking, while also providing a relatively safe option for osteoinduction, particularly when compared to other commonly used osteoinductive drugs. The studies involving the loading and controlled release of this flavanone are emerging and their outcome could pave the way for future biomedical applications. Such could potentially lead to the establishment of nature-derived products as valuable sources for new pharmaceutics that find application in cell-based therapies for tissue engineering and regenerative medicine.
Supplementary Material
Acknowledgements
The authors would like to acknowledge the support of the European Research Council grant agreement ERC-2014-ADG-669858 for project ATLAS. The authors also acknowledge the financial support by the Portuguese Foundation for Science and Technology (FCT) through a Post-doctoral grant (SFRH/BPD/119983/2016, Vítor Gaspar). The authors also acknowledge the project CICECO – Aveiro Institute of Materials, POCI-01-0145-FEDER-007679 (FCT Ref. UID/CTM/50011/2013), financed by national funds through the FCT/MEC and when appropriate co-financed by FEDER under the PT2020 Partnership Agreement.
Footnotes
Conflict of interest
The authors disclose that they have no conflict of interest.
References
- [1].Rodan GA. Therapeutic Approaches to Bone Diseases. Science (80-. ) 2000;289:1508–1514. doi: 10.1126/science.289.5484.1508. [DOI] [PubMed] [Google Scholar]
- [2].Roberts TT, Rosenbaum AJ. Bone grafts, bone substitutes and orthobiologics. Organogenesis. 2012;8:114–124. doi: 10.4161/org.23306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Dimitriou R, Jones E, McGonagle D, Giannoudis PV. Bone regeneration: current concepts and future directions. BMC Med. 2011;9:66. doi: 10.1186/1741-7015-9-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Che C-T, Wong M, Lam C. Natural Products from Chinese Medicines with Potential Benefits to Bone Health. Molecules. 2016;21:239. doi: 10.3390/molecules21030239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].An J, Hao D, Zhang Q, Chen B, Zhang R, Wang Y, Yang H. Natural products for treatment of bone erosive diseases: The effects and mechanisms on inhibiting osteoclastogenesis and bone resorption. Int Immunopharmacol. 2016;36:118–131. doi: 10.1016/j.intimp.2016.04.024. [DOI] [PubMed] [Google Scholar]
- [6].An J, Yang H, Zhang Q, Liu C, Zhao J, Zhang L, Chen B. Natural products for treatment of osteoporosis: The effects and mechanisms on promoting osteoblast-mediated bone formation. Life Sci. 2016;147:46–58. doi: 10.1016/j.lfs.2016.01.024. [DOI] [PubMed] [Google Scholar]
- [7].Brandi ML, Gennari L, Cerinic MM, Becherini L, Falchetti A, Masi L, Gennari C, Reginster J. Genetic markers of osteoarticular disorders : facts and hopes. Arthritis Res Ther. 2001;3:270–280. doi: 10.1186/ar316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Santo VE, Gomes ME, Mano JF, Reis RL. Controlled Release Strategies for Bone, Cartilage, and Osteochondral Engineering—Part II: Challenges on the Evolution from Single to Multiple Bioactive Factor Delivery. Tissue Eng Part B Rev. 2013;19:327–352. doi: 10.1089/ten.teb.2012.0727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Lee JS, Lee JS, Lee MS, An S, Yang K, Lee K, Yang HS, Lee H, Cho S-W. Plant Flavonoid-Mediated Multifunctional Surface Modification Chemistry: Catechin Coating for Enhanced Osteogenesis of Human Stem Cells. Chem Mater. 2017;29:4375–4384. doi: 10.1021/acs.chemmater.7b00802. [DOI] [Google Scholar]
- [10].Zhao J, Ohba S, Komiyama Y, Shinkai M, Chung U, Nagamune T. Icariin: A Potential Osteoinductive Compound for Bone Tissue Engineering. Tissue Eng Part A. 2010;16:233–243. doi: 10.1089/ten.TEA.2009.0165. [DOI] [PubMed] [Google Scholar]
- [11].Wang P, Zhang F, He Q, Wang J, Shiu HT, Shu Y, Tsang WP, Liang S, Zhao K, Wan C. Flavonoid Compound Icariin Activates Hypoxia Inducible Factor-1α in Chondrocytes and Promotes Articular Cartilage Repair. PLoS One. 2016;11:e0148372. doi: 10.1371/journal.pone.0148372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Liu H, Xiong Y, Zhu X, Gao H, Yin S, Wang J, Chen G, Wang C, Xiang L, Wang P, Fang J, et al. Icariin improves osteoporosis, inhibits the expression of PPARγ, C/EBPα, FABP4 mRNA, N1ICD and jagged1 proteins, and increases Notch2 mRNA in ovariectomized rats. Exp Ther Med. 2017;13:1360–1368. doi: 10.3892/etm.2017.4128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Weaver CM, Alekel DL, Ward WE, Ronis MJ. Flavonoid Intake and Bone Health. J Nutr Gerontol Geriatr. 2012;31:239–253. doi: 10.1080/21551197.2012.698220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Ahmad N, Banala VT, Kushwaha P, Karvande A, Sharma S, Tripathi AK, Verma A, Trivedi R, Mishra PR. Quercetin-loaded solid lipid nanoparticles improve osteoprotective activity in an ovariectomized rat model: a preventive strategy for post-menopausal osteoporosis. RSC Adv. 2016;6:97613–97628. doi: 10.1039/C6RA17141A. [DOI] [Google Scholar]
- [15].Gattuso G, Barreca D, Gargiulli C, Leuzzi U, Caristi C. Flavonoid composition of citrus juices. Molecules. 2007;12:1641–1673. doi: 10.3390/12081641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Chen R, Qi Q-L, Wang M-T, Li Q-Y. Therapeutic potential of naringin: an overview. Pharm Biol. 2016;54:3203–3210. doi: 10.1080/13880209.2016.1216131. [DOI] [PubMed] [Google Scholar]
- [17].Dahan A, Altman H. Food–drug interaction: grapefruit juice augments drug bioavailability—mechanism, extent and relevance. Eur J Clin Nutr. 2004;58:1–9. doi: 10.1038/sj.ejcn.1601736. [DOI] [PubMed] [Google Scholar]
- [18].Ferlazzo N, Cirmi S, Russo M, Trapasso E, Ursino MR, Lombardo GE, Gangemi S, Calapai G, Navarra M. NF-κB mediates the antiproliferative and proapoptotic effects of bergamot juice in HepG2 cells. Life Sci. 2016;146:81–91. doi: 10.1016/j.lfs.2015.12.040. [DOI] [PubMed] [Google Scholar]
- [19].Marino A, Paterniti I, Cordaro M, Morabito R, Campolo M, Navarra M, Esposito E, Cuzzocrea S. Role of natural antioxidants and potential use of bergamot in treating rheumatoid arthritis. PharmaNutrition. 2015;3:53–59. doi: 10.1016/j.phanu.2015.03.002. [DOI] [Google Scholar]
- [20].Janda E, Lascala A, Martino C, Ragusa S, Nucera S, Walker R, Gratteri S, Mollace V. Molecular mechanisms of lipid- and glucose-lowering activities of bergamot flavonoids. PharmaNutrition. 2016;4:S8–S18. doi: 10.1016/j.phanu.2016.05.001. [DOI] [Google Scholar]
- [21].Bharti S, Rani N, Krishnamurthy B, Arya DS. Preclinical evidence for the pharmacological actions of naringin: A review. Planta Med. 2014;80:437–451. doi: 10.1055/s-0034-1368351. [DOI] [PubMed] [Google Scholar]
- [22].Li L, Zeng Z, Cai G. Comparison of neoeriocitrin and naringin on proliferation and osteogenic differentiation in MC3T3-E1. Phytomedicine. 2011;18:985–989. doi: 10.1016/j.phymed.2011.03.002. [DOI] [PubMed] [Google Scholar]
- [23].Wu J Bin, Fong YC, Tsai HY, Chen YF, Tsuzuki M, Tang CH. Naringin-induced bone morphogenetic protein-2 expression via PI3K, Akt, c-Fos/c-Jun and AP-1 pathway in osteoblasts. Eur J Pharmacol. 2008;588:333–341. doi: 10.1016/j.ejphar.2008.04.030. [DOI] [PubMed] [Google Scholar]
- [24].Fan J, Li J, Fan Q. Naringin promotes differentiation of bone marrow stem cells into osteoblasts by upregulating the expression levels of microRNA-20a and downregulating the expression levels of PPARγ. Mol Med Rep. 2015;12:4759–4765. doi: 10.3892/mmr.2015.3996. [DOI] [PubMed] [Google Scholar]
- [25].Xu Z, Li N, Wooley PH, Yang S-Y, Jiang Y. Naringin promotes osteoblast differentiation and effectively reverses ovariectomy-associated osteoporosis. J Orthop Sci. 2013;18:478–485. doi: 10.1007/s00776-013-0362-9. [DOI] [PubMed] [Google Scholar]
- [26].Yu G, Zheng G, Chang B, Hu Q, Lin F, Liu D, Wu C, Du S, Li X. Naringin Stimulates Osteogenic Differentiation of Rat Bone Marrow Stromal Cells via Activation of the Notch Signaling Pathway. Stem Cells Int. 2016;2016:1–8. doi: 10.1155/2016/7130653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Rickard DJ, Sullivan TA, Shenker BJ, Leboy PS, Kazhdan I. Induction of Rapid Osteoblast Differentiation in Rat Bone Marrow Stromal Cell Cultures by Dexamethasone and BMP-2. Dev Biol. 1994;161:218–228. doi: 10.1006/dbio.1994.1022. [DOI] [PubMed] [Google Scholar]
- [28].Ghali O, Broux O, Falgayrac G, Haren N, van Leeuwen JPTM, Penel G, Hardouin P, Chauveau C. Dexamethasone in osteogenic medium strongly induces adipocyte differentiation of mouse bone marrow stromal cells and increases osteoblast differentiation. BMC Cell Biol. 2015;16:9. doi: 10.1186/s12860-015-0056-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Zhang Peng, Dai K, Yan S, Yan W, Zhang Chao, Chen D, Xu Bo, Xu Z. Effects of naringin on the proliferation and osteogenic differentiation of human bone mesenchymal stem cell. Eur J Pharmacol. 2009;607:1–5. doi: 10.1016/j.ejphar.2009.01.035. [DOI] [PubMed] [Google Scholar]
- [30].Ang ESM, Yang X, Chen H, Liu Q, Zheng MH, Xu J. Naringin abrogates osteoclastogenesis and bone resorption via the inhibition of RANKL-induced NF-kB and ERK activation. FEBS Lett. 2011;585:2755–2762. doi: 10.1016/j.febslet.2011.07.046. [DOI] [PubMed] [Google Scholar]
- [31].Liu M, Li Y, Yang S-T. Effects of naringin on the proliferation and osteogenic differentiation of human amniotic fluid-derived stem cells. J Tissue Eng Regen Med. 2014;11:276–284. doi: 10.1002/term.1911. [DOI] [PubMed] [Google Scholar]
- [32].Krishnan V, Byrant HU, MacDougald OA. Regulation of bone mass by Wnt signaling. J Clin Invest. 2006;116:1202–1209. doi: 10.1172/JCI28551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Pang W-Y, Wang X-L, Mok S-K, Lai W-P, Chow H-K, Leung P-C, Yao X-S, Wong M-S. Naringin improves bone properties in ovariectomized mice and exerts oestrogen-like activities in rat osteoblast-like (UMR-106) cells. Br J Pharmacol. 2010;159:1693–1703. doi: 10.1111/j.1476-5381.2010.00664.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Xu T, Wang L, Tao Y, Ji Y, Deng F, Wu X-H. The Function of Naringin in Inducing Secretion of Osteoprotegerin and Inhibiting Formation of Osteoclasts, Evidence-Based Complement. Altern Med. 2016;2016:1–7. doi: 10.1155/2016/8981650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Hirata M, Matsumoto C, Takita M, Miyaura C, Inada M. Naringin Suppresses Osteoclast Formation and Enhances Bone Mass in Mice. J Heal Sci. 2009;55:463–467. doi: 10.1248/jhs.55.463. [DOI] [Google Scholar]
- [36].Yin L, Cheng W, Qin Z, Yu H, Yu Z, Zhong M, Sun K, Zhang W. Effects of Naringin on Proliferation and Osteogenic Differentiation of Human Periodontal Ligament Stem Cells In Vitro and In Vivo. Stem Cells Int. 2015;2015:1–9. doi: 10.1155/2015/758706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Sun X, Fengbo L, Xinlong M, Jianxiong M, Zhao B, Zhang Y, Yanjun L, Jianwei L, Meng X. The Effects of Combined Treatment with Naringin and Treadmill Exercise on Osteoporosis in Ovariectomized Rats. Sci Rep. 2015;5 doi: 10.1038/srep13009. 13009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Wang D, Ma W, Wang F, Dong J, Wang D, Sun B, Wang B. Stimulation of Wnt/β-Catenin Signaling to Improve Bone Development by Naringin via Interacting with AMPK and Akt. Cell Physiol Biochem. 2015;36:1563–1576. doi: 10.1159/000430319. [DOI] [PubMed] [Google Scholar]
- [39].Shangguan W, Zhang Y, Li Z, Tang L, Shao J, Li H. Naringin inhibits vascular endothelial cell apoptosis via endoplasmic reticulum stress- and mitochondrial-mediated pathways and promotes intraosseous angiogenesis in ovariectomized rats. Int J Mol Med. 2017;40:1741–1749. doi: 10.3892/ijmm.2017.3160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Song N, Zhao Z, Ma X, Sun X, Ma J, Li F, Sun L, Lv J. Naringin promotes fracture healing through stimulation of angiogenesis by regulating the VEGF/VEGFR-2 signaling pathway in osteoporotic rats. Chem Biol Interact. 2017;261:11–17. doi: 10.1016/j.cbi.2016.10.020. [DOI] [PubMed] [Google Scholar]
- [41].Santo VE, Gomes ME, Mano JF, Reis RL. Controlled Release Strategies for Bone, Cartilage, and Osteochondral Engineering—Part I: Recapitulation of Native Tissue Healing and Variables for the Design of Delivery Systems. Tissue Eng Part B Rev. 2013;19:308–326. doi: 10.1089/ten.teb.2012.0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Rouwkema J, Khademhosseini A. Vascularization and Angiogenesis in Tissue Engineering: Beyond Creating Static Networks. Trends Biotechnol. 2016;34:733–745. doi: 10.1016/j.tibtech.2016.03.002. [DOI] [PubMed] [Google Scholar]
- [43].Ramasamy SK, Kusumbe AP, Wang L, Adams RH. Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature. 2014;507:376–380. doi: 10.1038/nature13146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Wei M, Yang Z, Li P, Zhang Y, Sse WC. Anti-osteoporosis activity of naringin in the retinoic acid-induced osteoporosis model. Am J Chin Med. 2007;35:663–667. doi: 10.1142/S0192415X07005156. [DOI] [PubMed] [Google Scholar]
- [45].Mandadi K, Ramirez M, Jayaprakasha GK, Faraji B, Lihono M, Deyhim F, Patil BS. Citrus bioactive compounds improve bone quality and plasma antioxidant activity in orchidectomized rats. Phytomedicine. 2009;16:513–520. doi: 10.1016/j.phymed.2008.09.001. [DOI] [PubMed] [Google Scholar]
- [46].Zhou X, Zhang P, Zhang C, Zhu Z. Promotion of bone formation by naringin in a titanium particle-induced diabetic murine calvarial osteolysis model. J Orthop Res. 2009;28:451–456. doi: 10.1002/jor.21002. [DOI] [PubMed] [Google Scholar]
- [47].Wong RWK, Rabie ABM. Effect of naringin collagen graft on bone formation. Biomaterials. 2006;27:1824–1831. doi: 10.1016/j.biomaterials.2005.11.009. [DOI] [PubMed] [Google Scholar]
- [48].Lin C, Jiang X, Dai Z, Guo X, Weng T, Wang J, Li Y, Feng G, Gao X, He L. Sclerostin Mediates Bone Response to Mechanical Unloading Through Antagonizing Wnt/β-Catenin Signaling. J Bone Miner Res. 2009;24:1651–1661. doi: 10.1359/jbmr.090411. [DOI] [PubMed] [Google Scholar]
- [49].Ma X, Lv J, Sun X, Ma J, Xing G, Wang Y, Sun L, Wang J, Li F, Li Y, Zhao Z. Naringin ameliorates bone loss induced by sciatic neurectomy and increases Semaphorin 3A expression in denervated bone. Sci Rep. 2016;6 doi: 10.1038/srep24562. 24562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Ma R, Wang L, Zhao B, Liu C, Liu H, Zhu R, Chen B, Li L, Zhao D, Mo F, Li Y, et al. Diabetes Perturbs Bone Microarchitecture and Bone Strength through Regulation of Sema3A/IGF-1/β-Catenin in Rats. Cell Physiol Biochem. 2017;41:55–66. doi: 10.1159/000455936. [DOI] [PubMed] [Google Scholar]
- [51].Rivoira M, Rodríguez V, Picotto G, Battaglino R, Tolosa de Talamoni N. Naringin prevents bone loss in a rat model of type 1 Diabetes mellitus. Arch Biochem Biophys. 2018;637:56–63. doi: 10.1016/j.abb.2017.12.001. [DOI] [PubMed] [Google Scholar]
- [52].Liu K, Wu L, Shi X, Wu F. Protective effect of naringin against ankylosing spondylitis via ossification, inflammation and oxidative stress in mice. Exp Ther Med. 2016;12:1153–1158. doi: 10.3892/etm.2016.3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Zhao Y, Li Z, Wang W, Zhang H, Chen J, Su P, Liu L, Li W. Naringin Protects Against Cartilage Destruction in Osteoarthritis Through Repression of NF-κB Signaling Pathway. Inflammation. 2016;39:385–392. doi: 10.1007/s10753-015-0260-8. [DOI] [PubMed] [Google Scholar]
- [54].Xu Q, Zhang Z, Sun W. Effect of Naringin on Monosodium Iodoacetate-Induced Osteoarthritis Pain in Rats. Med Sci Monit. 2017;23:3746–3751. doi: 10.12659/MSM.902396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Ahmad SF, Zoheir KMA, Abdel-Hamied HE, Ashour AE, Bakheet SA, Attia SM, Abd-Allah ARA. Amelioration of autoimmune arthritis by naringin through modulation of T regulatory cells and Th1/Th2 cytokines. Cell Immunol. 2014;287:112–120. doi: 10.1016/j.cellimm.2014.01.001. [DOI] [PubMed] [Google Scholar]
- [56].Kawaguchi K, Maruyama H, Hasunuma R, Kumazawa Y. Suppression of inflammatory responses after onset of collagen-induced arthritis in mice by oral administration of the Citrus flavanone naringin. Immunopharmacol Immunotoxicol. 2011;33:723–729. doi: 10.3109/08923973.2011.564186. [DOI] [PubMed] [Google Scholar]
- [57].Lugo L, Villalvilla A, Largo R, Herrero-Beaumont G, Roman-Blas JA. Selective estrogen receptor modulators (SERMs): New alternatives for osteoarthritis? Maturitas. 2014;77:380–384. doi: 10.1016/j.maturitas.2014.01.016. [DOI] [PubMed] [Google Scholar]
- [58].Sapir-Koren R, Livshits G. Postmenopausal osteoporosis in rheumatoid arthritis: The estrogen deficiency-immune mechanisms link. Bone. 2017;103:102–115. doi: 10.1016/j.bone.2017.06.020. [DOI] [PubMed] [Google Scholar]
- [59].Cosman F. Combination therapy for osteoporosis: a reappraisal. Bonekey Rep. 2014;3:1–8. doi: 10.1038/bonekey.2014.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Camargos GV, Bhattacharya P, van Lenthe GH, Del Bel Cury AA, Naert I, Duyck J, Vandamme K. Mechanical competence of ovariectomy-induced compromised bone after single or combined treatment with high-frequency loading and bisphosphonates. Sci Rep. 2015;5 doi: 10.1038/srep10795. 10795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Li CY, Price C, Delisser K, Nasser P, Laudier D, Clement M, Jepsen KJ, Schaffler MB. Long-Term Disuse Osteoporosis Seems Less Sensitive to Bisphosphonate Treatment Than Other Osteoporosis. J Bone Miner Res. 2004;20:117–124. doi: 10.1359/JBMR.041010. [DOI] [PubMed] [Google Scholar]
- [62].Duque G, Troen BR. Understanding the Mechanisms of Senile Osteoporosis: New Facts for a Major Geriatric Syndrome. J Am Geriatr Soc. 2008;56:935–941. doi: 10.1111/j.1532-5415.2008.01764.x. [DOI] [PubMed] [Google Scholar]
- [63].Cassidy A, Minihane A-M. The role of metabolism (and the microbiome) in defining the clinical efficacy of dietary flavonoids. Am J Clin Nutr. 2017;105:10–22. doi: 10.3945/ajcn.116.136051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Manach C, Williamson G, Morand C, Scalbert A, Rémésy C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr. 2005;81:230S–242S. doi: 10.1093/ajcn/81.1.230S. doi:81/1/230S [pii] [DOI] [PubMed] [Google Scholar]
- [65].Kanaze FI, Kokkalou E, Niopas I, Georgarakis M, Stergiou A, Bikiaris D. Dissolution enhancement of flavonoids by solid dispersion in PVP and PEG matrixes: A comparative study. J Appl Polym Sci. 2006;102:460–471. doi: 10.1002/app.24200. [DOI] [Google Scholar]
- [66].Walle T. Absorption and metabolism of flavonoids. Free Radic Biol Med. 2004;36:829–837. doi: 10.1016/j.freeradbiomed.2004.01.002. [DOI] [PubMed] [Google Scholar]
- [67].Lauro MR, De Simone F, Sansone F, Iannelli P, Aquino RP. Preparations and release characteristics of naringin and naringenin gastro-resistant microparticles by spray-drying. J Drug Deliv Sci Technol. 2007;17:119–124. doi: 10.1016/S1773-2247(07)50018-3. [DOI] [Google Scholar]
- [68].Pai DA, Vangala VR, Ng JW, Ng WK, Tan RBH. Resistant maltodextrin as a shell material for encapsulation of naringin: Production and physicochemical characterization. J Food Eng. 2015;161:68–74. doi: 10.1016/j.jfoodeng.2015.03.037. [DOI] [Google Scholar]
- [69].Rao K, Imran M, Jabri T, Ali I, Perveen S, Shafiullah, Ahmed S, Shah MR. Gum tragacanth stabilized green gold nanoparticles as cargos for Naringin loading: A morphological investigation through AFM. Carbohydr Polym. 2017;174:243–252. doi: 10.1016/j.carbpol.2017.06.071. [DOI] [PubMed] [Google Scholar]
- [70].Roy AS. A spectroscopic study of the interaction of the antioxidant naringin with bovine serum albumin. J Biophys Chem. 2010;1:141–152. doi: 10.4236/jbpc.2010.13017. [DOI] [Google Scholar]
- [71].Singh R, Lillard JW. Nanoparticle-based targeted drug delivery. Exp Mol Pathol. 2009;86:215–223. doi: 10.1016/j.yexmp.2008.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Santo VE, Gomes ME, Mano JF, Reis RL. From nano- to macro-scale: nanotechnology approaches for spatially controlled delivery of bioactive factors for bone and cartilage engineering. Nanomedicine. 2012;7:1045–1066. doi: 10.2217/nnm.12.78. [DOI] [PubMed] [Google Scholar]
- [73].Low SA, Kopeček J. Targeting polymer therapeutics to bone. Adv Drug Deliv Rev. 2012;64:1189–1204. doi: 10.1016/j.addr.2012.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Ji Y, Wang L, Watts DC, Qiu H, You T, Deng F, Wu X. Controlled-release naringin nanoscaffold for osteoporotic bone healing. Dent Mater. 2014;30:1263–1273. doi: 10.1016/j.dental.2014.08.381. [DOI] [PubMed] [Google Scholar]
- [75].Chen K-Y, Lin K, Chen Y, Yao C. A Novel Porous Gelatin Composite Containing Naringin for Bone Repair. Evidence-Based Complement Altern Med. 2013;2013:1–10. doi: 10.1155/2013/283941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Guo Z, Bo D, He P, Li H, Wu G, Li Z, Zhou C, Li Q. Sequential controlled-released dual-drug loaded scaffold for guided bone regeneration in a rat fenestration defect model. J Mater Chem B. 2017;5:7701–7710. doi: 10.1039/c7tb00909g. [DOI] [PubMed] [Google Scholar]
- [77].Chang P-C, Chao Y-C, Hsiao M-H, Chou H-S, Jheng Y-H, Yu X-H, Lee N, Yang C, Liu D-M. Inhibition of Periodontitis Induction Using a Stimuli-Responsive Hydrogel Carrying Naringin. J Periodontol. 2017;88:190–196. doi: 10.1902/jop.2016.160189. [DOI] [PubMed] [Google Scholar]
- [78].Yu M, You D, Zhuang J, Lin S, Dong L, Weng S, Zhang B, Cheng K, Weng W, Wang H. Controlled release of naringin in metal-organic framework-loaded mineralized collagen coating to simultaneously enhance osseointegration and antibacterial activity. ACS Appl Mater Interfaces. 2017;9:19698–19705. doi: 10.1021/acsami.7b05296. [DOI] [PubMed] [Google Scholar]
- [79].Feng T, Wang K, Liu F, Ye R, Zhu X, Zhuang H, Xu Z. Structural characterization and bioavailability of ternary nanoparticles consisting of amylose, α-linoleic acid and β-lactoglobulin complexed with naringin. Int J Biol Macromol. 2017;99:365–374. doi: 10.1016/j.ijbiomac.2017.03.005. [DOI] [PubMed] [Google Scholar]
- [80].Yan H-H, Zhang J-Q, Ren S-H, Xie X-G, Huang R, Jin Y, Lin J. Experimental and computational studies of naringin/cyclodextrin inclusion complexation. J Incl Phenom Macrocycl Chem. 2017;88:15–26. doi: 10.1007/s10847-017-0704-x. [DOI] [Google Scholar]
- [81].Lavrador P, Gaspar VM, Mano JF. Stimuli-responsive nanocarriers for delivery of bone therapeutics – Barriers and progresses. J Control Release. 2018;273:51–67. doi: 10.1016/j.jconrel.2018.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.