Abstract
Background
Maternal anemia, low birth weight, intrauterine growth retardation and premature births are complications associated with malaria in pregnancy (MIP). In order to prevent malaria, Wold Health Organization (WHO) recommends at least three doses of Sulphadoxine Pyrimethamine (SP) for Intermittent Preventive Treatment of Malaria in Pregnancy (IPTp-SP3+) during the antenatal period. This study aimed to determine the prevalence and the health system determinants of uptake of three or more doses (IPTp 3+) in Busia County, Kenya.
Methods
A facility-based cross-sectional study conducted at Busia County Referral Hospital between June and July 2023. Mothers’ in the Maternity Unit with term deliveries were consecutively sampled. Data on health system factors was collected using interviewer administered semi-structured questionnaires and interview guides. The statistical significance of health system factors affecting IPTp-SP3+ was dertemined using the odds ratio (OR) and adjusted OR.
Results
The prevalence of IPTp-SP 3+ uptake was 43.0%. Health system factors significantly associated with IPTp-SP 3+ uptake were facility being too far (aOR = 0.575, 95% CI: 0.351- 0.941, p = 0.028) and waiting time being too long (aOR = 0.606, 95% CI: 0.385-0.955, p = 0.031).
Conclusion
Prevalence of uptake of IPTp 3+ is still low, with less than half of the pregnant mothers complying. The County and National Ministries of Health should make a concerted effort to ensure availability and affordability of SP drugs and also ensure access to and customer satisfaction at the healthcare facilities.
Introduction
Despite considerable global efforts to eradicate malaria, it still poses a health risk in tropical and subtropical regions, with Sub-Saharan Africa bearing most of the disease’s fatalities. About half of all people on earth are susceptible to malaria. According to the World Malaria Report1, there were 241 million cases of malaria around the world in 2020, coming from 85 malaria-endemic nations. This is a considerable rise from the 227 million cases recorded in 2019, with most occurring in Africa1. During that time, estimates show that 627,000 people died from malaria-related reasons. It has been projected that Africa accounts for an increase in malaria infections from 213 million to 228 million and deaths from 534,000 to 602,000 between 2019 and 2020, accounting for 95% and 96% of all cases and deaths worldwide, respectively1. At the local level, it is thought that 70% of Kenyans are at risk for malaria, despite the disease not equally spread. The regions closest to Lake Victoria and the coastal regions are most vulnerable to malaria infection. The Lake Endemic region has the most significant incidence (28%) of the 8% nationwide prevalence rate2. Kenya’s Busia County has the highest frequency of 39% and is in the Lake Endemic Zone.
Pregnant women are particularly susceptible to malaria infection and death. Malaria in pregnancy (MiP) is associated with various health risks and issues, such as maternal anemia, newborns with low birth weight (LBW), intrauterine growth retardation (IGR), premature births, and neonatal death3. LBW results from inadequate nutrition of the fetus caused by malaria parasites and is a contributing factor to low infant development and survival in Africa. In places where malaria is endemic, LBW is predicted to cause 6% of infant fatalities4. In 2020, malaria was expected to have affected 11.6 million pregnant women in 33 African nations, leading to 819,000 LBW infants1. Preventing MiP reduces the risk of severe maternal anemia by 38%, low birthweight by 43%, and perinatal mortality by 27%. According to data from the Ministry of Health (MOH) in Kenya, hospitalized incidences of malaria among pregnant women in 2019 varied from 1% to 60%5. Additionally, according to MOH estimates, 6.3% of women who saw an ANC doctor for the first time ended up in the hospital because of malaria5.
Intermittent preventative therapy during pregnancy (IPTp) using sulfadoxine-pyrimethamine (SP) is a cost-effective and safe strategy to reduce the impact of MiP and unfavorable pregnancy and delivery outcomes6. According to the World Malaria Report1, 32% of all pregnant women in 33 countries had at least three IPTp doses. In 2012, the World Health Organization (WHO) updated recommendations for giving IPTp, indicating that pregnant women should receive three or more doses. The World Malaria Report has indicated that if at least 90% of all pregnant women took three or more doses of SP as part of IPTp, the number of LBW babies would significantly decline by as much as 206,000 births1. Even though the Kenyan government implemented these regulations in 2013 and saw quick advancements, the acceptance of +3 doses of IPTp-SP has been incredibly sluggish. The Kenya Malaria Indicator Survey in 2020 indicated only 49% and 46% of Kenyan women aged 15-49 years in the Coast and Lake endemic zones got IPTp-SP+3, respectively7.
There is plausity of information in Kenya, especially in the endemic lake region, on the uptake of three or more doses of IPTp-SP3+. This study aimed to determine the prevalence of health system-related factors contributing to IPTp-SP uptake at Busia County Referral Hospital in Kenya.
Methods
Study Design
A facility-based cross-sectional research study investigated the prevalence and health system factors influencing the uptake of three or more doses of SP during IPTp-SP for MiP.
Study Site
Busia County Referral Hospital, in Busia County, Kenya, served as the research site given that 157,650 women in the County are at risk of contracting malaria8.
Inclusion Criteria
The research participants included in the study were mothers with term births (>37 weeks), in possession of their ANC booklet attending the Busia County referral hospital for delivery or who had recently given birth (within 24 hours) and presented themselves to the facility.
Exclusion Criteria
Mothers with HIV taking daily prophylactic cotrimoxazole, mothers allergic to Sulfur, mothers treated for malaria during their pregnancy, distressed mothers unable to complete the study, and preterm mothers (<37 weeks) were excluded from the study.
Sample size determination and sampling method
Mothers who met the inclusion criteria were included in the study sample until the expected sample size was attained. When mothers checked into the facility’s maternity ward, were scheduled for delivery, and had a term delivery, they were approached and informed about the research. Informed consent was provided in writing by those willing to participate in the study. Cochran’s method9 was used to determine a sample size of 384 mothers.
Data Collection
The study participants completed semi-structured questionnaires covering aspects of the health system such as user charges, working hours, waiting time, stockouts, mothers’ perceptions of staff knowledge on IPTp-SP, and facility initiatives to educate patients about IPTp-SP. Data collection period was June 7, 2023 − July 2, 2023. Two research assistants were trained to help with data collection. Data was collected through interviewing mothers and entered into Microsoft Excel. The primary investigator cleaned, verified and evaluated the data to ensure its precision and comprehensiveness.
Data Analysis
Measures of central tendency, percentages, and frequencies were reported for descriptive statistics and prevalence data. Factors influencing uptake of three or more doses of IPTp-SP were assessed using Crude Odds ratio (OR) and adjusted OR to determine statistical significance. A p-value of 0.05 with a 95% confidence interval (CI) was considered for inferential statistics. SPSS version 25 was used to analyse.
Ethics Approval And Consent To Particpate
The Jomo Kenyatta University of Agriculture and Technology (JKUAT) Ethical Review Committee approved the study through the ethical clearance- JKU/ISERC/02316/0849.
Permit to conduct the study was granted by the National Commission for Science, Technology and Innovation (NACOSTI)-NACOSTI/P/23/25738. Consent was sought from Busia County Department of Health, Busia County Referral Hospital and all the study participants signed an informed consent form before participating in the study.
Results
Sociodemographic factors
The majority (33.6%) of the participants were between 20–25 years, with the mean age being 26.51±6.1 years. As shown in Table 1, most participants were married (79.9%), had only completed secondary school (43.8%), were jobless (52.9%), lived in rural regions (92.2%), and made less than KES 10,000 a month (87.5%).
Table 1. Baseline Sociodemographic Characteristics.
| Variable | Frequency (N/384) | Percentage |
|---|---|---|
| Age | ||
| ≤ 19 Yrs. | 51 | 13.3% |
| 20 − 25 Yrs. | 129 | 33.6% |
| 26 − 30 Yrs. | 106 | 27.6% |
| 31 − 35 Yrs. | 60 | 15.6% |
| > 36 Yrs. | 38 | 9.9% |
| Mean age = 26.51 Standard Deviation = 6.1 | ||
| Education Level | ||
| No education | 9 | 2.3% |
| Primary | 148 | 38.5% |
| Secondary | 163 | 42.4% |
| Tertiary | 64 | 16.7% |
| Marital Status | ||
| Single | 76 | 19.8% |
| Married | 307 | 79.9% |
| Separated/Widowed | 1 | 0.3% |
| Occupation | ||
| Unemployed | 203 | 52.9% |
| Student | 28 | 7.3% |
| Casual Laborer | 38 | 9.9% |
| Self-employed | 87 | 22.7% |
| Formal employment | 28 | 7.3% |
| Residency | ||
| Rural | 354 | 92.2% |
| Urban | 30 | 7.8% |
| Monthly Income (Ksh.) | ||
| < 10000 | 336 | 87.5% |
| 10000 − 20000 | 46 | 12.0 |
| 20001 − 50000 | 2 | 0.5% |
| 50001+ | 0 | 0% |
Prevalence of Uptake of Three or More Doses of IPTp-SP
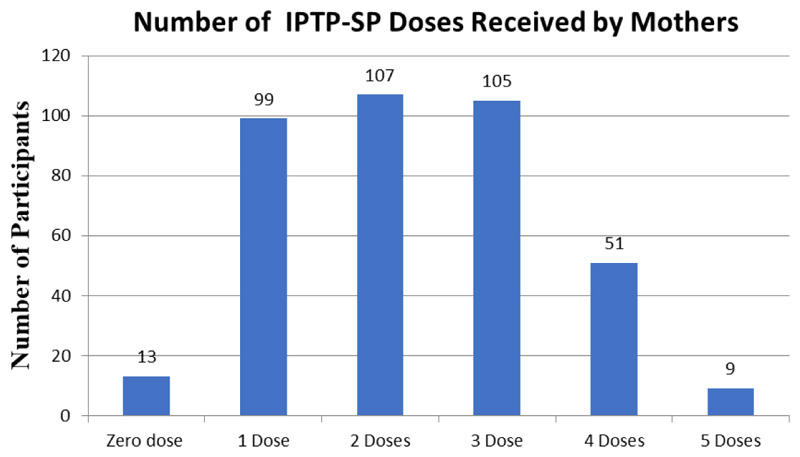
27.9% of the participants took two IPTp-SP doses during their pregnancy period, 27.3% took three doses, 13.3% took four doses, 25.8% took one dose, 2.3% took five doses, and 3.4% did not use a single dose of IPTP-SP (Figure 1). 43.0% of participants took three or more doses of IPTP-SP.
Fig. 1. The number of IPTp-SP doses participants received.
Health System Factors Influencing the Uptake of 3 ≥ Doses of IPTp-SP
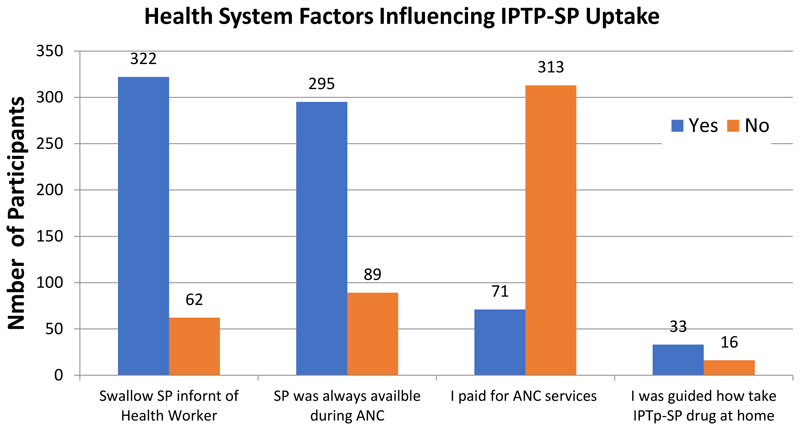
Most participants (86.8%) who took the IPTp-SP medication acknowledged that they had taken it in front of the healthcare worker, and 67.3% received instructions from the healthcare worker on how to do so independently. The SP medication was not always available in the institution, as evidenced by the 76.8% of respondents who disagreed that it was always available. An evaluation of the cost of obtaining ANC services revealed that, of those who reported using the services, 81.5% said they did not pay, and 18.5% said they did pay (Fig. 2).
Fig. 2. Health system factors influencing uptake of IPTP-SP.
Chi-square test for association was performed to determine the association between health system factors and IPTp-SP medications uptake. As seen in Table 2 below, three factors showed statistically significant association with IPTp-SP medication uptake, including SP available always (χ2 (1) = 5.683, p = 0.017), facility being too far (χ2 (1) = 6.900, p = 0.009), and waiting too long to receive a dose (χ2 (1) = 6.396, p = 0.011).
Table 2. Chi-Square Test for Association between Health System Factors and IPTp-SP Uptake.
| Variable | ≥ 3 IPTp-SP Drugs Uptake (N = 165) N (%) |
≤3 IPTp-SP Drug Uptake (N = 219) N (%) |
P Value |
|---|---|---|---|
| SP available Always No Yes |
48 (29.1) 117(70.9) |
41 (18.7) 178(81.3) |
0.017 |
| Swallowed SP at Facility No Yes |
20 (12.1) 145 (87.9) |
42 (19.2) 177 (80.8) |
0.063 |
| Facility too Far No Yes |
31 (18.8) 134 (81.2) |
67 (30.6) 152 (69.4) |
0.009 |
| Staff Professional No Yes |
4 (2.4) 161 (97.6) |
3 (1.4) 216 (98.6) |
0.445 |
| Paid for ANC Services No Yes |
133 (80.6) 32 (19.4) |
180 (82.2) 39 (17.8) |
0.692 |
| SP Charged Highly No Yes |
6 (3.6) 159 (94.6) |
9 (4.1) 210 (95.9) |
0.813 |
| Waiting Time long No Yes |
41 (24.8) 124 (75.2) |
81 (37.0) 138 (63.0) |
0.011 |
| Staff Shortage No Yes |
129 (78.2) 36 (21.8) |
166 (75.8) 53 (24.2) |
0.584 |
| Facility Closed at Times No Yes |
152 (92.1) 13 (7.9) |
201 (91.8) 18 (8.2) |
0.904 |
Bivariate logistic regression was done to determine the strength of association between the significant health-system factors and IPTp-SP medication uptake. As seen in Table 3, the odds of receiving three or more doses of IPTp-SP (“yes” category) was 1.781 times greater when a participant indicated that SP was always available (OR = 1.781, 95% CI: 1.105 - 2.872, p = 0.018). The odds of receiving three or more doses of IPTp-SP (“yes” category) was 47.5% less likely for participants who indicated that the facility was too far as opposed to those who indicated that the facility was not too far (OR = 0.525, 95% CI: 0.323 − 0.852, p = 0.009). Additionally, the odds of receiving three or more doses of IPTp-SP (“yes” category) was 43.7% less likely for participants who indicated that the waiting time was too long as opposed to those who indicated that the waiting time was not too long (OR = 0.563, 95% CI: 0.360 - 0.881, p = 0.012).
Table 3. Bivariate and Multivariable Association for Significant Health System Factors and IPTp-SP Uptake.
| Variable | Crude Odds Ratio (95% CI) |
P − value | Adjusted Odds Ratio (95% CI) |
P − value |
|---|---|---|---|---|
| SP Always Available No Yes |
ref 1.781 (1.105-2.872) |
0.018 | ref 1.598 (0.982-2.602) |
0.059 |
| Facility too Far No Yes |
ref 0.525 (0.323 − 0.852) |
0.009 | ref 0.575 (0.351-0.941) |
0.028 |
| Waiting time too long No Yes |
ref 0.563 (0.360-0.881) |
0.012 | ref 0.606 (0.385-0.955) |
0.031 |
Multivariate logistic regression of the three health-system factors was done to determine whether the variables added significantly to the model. The shown in Table 3, facility being too far (aOR = 0.575, 95% CI: 0.351-0.941, p = 0.028) and waiting time being too long (aOR = 0.606, 95% CI: 0.385 - 0.955, p = 0.031) added significantly to the model, but SP always available (aOR = 1.598, 95% CI: 0.982-2.602, p = 0.059) did not add significantly to the model.
The odds of receiving three or more doses of IPTp-SP was 42.5% less likely among participants who indicated that the facility was too far as opposed to those who indicated that the facility was not too far and 39.4% less likely among participants who indicated that the waiting time was too long as opposed to those that indicated that the waiting time was not too long.
Discussion
The Prevalence of Uptake of Three or More Doses of IPTp-SP
According to the results of the present study, the prevalence of uptake of three or more doses of IPTp-SP was 43%. The findings are consistent with Karoki et al.10, who found a prevalence of 49% incidence in Kenya. The disparities between the overall endemic regions in the Kenyan prevalence of 49% compared to our rate of 43% may be explained by the findings in Karoki et al.10 that the prevalence was lower in the lake region than the coastal zone. These findings are inconsistent with Pons-Duran et al.11, who found a prevalence of 63% in Mozambique and less than 25% in the DRC, Madagascar, and Nigeria. They agree with Martin et al.12, who observed a 22.3% frequency in Uganda. Regardless, this study’s findings provide a more accurate assessment of the SP drug compliance rate. They show a lack of adherence to WHO guidelines recommending that all pregnant women in malaria-endemic areas take three or more IPTp-SP medications during their pregnancy. Health system factors may help explain the below-average prevalence uptake of three or more doses of IPTp-SP.
Health System Factors Influencing the Uptake of 3 ≥ Doses of IPTp-SP
This study demonstrated that the SP availability at the study site was a significant factor in uptake of three or more doses of IPTp-SP. The mothers’ distance to the institution constituted another issue affecting the uptake of 3 ≥ doses of IPTp-SP. Another health system factor was the lengthy wait times. The study’s findings bring new information to the body of prior research while comparable to the findings of other investigations. Stockout had a detrimental effect on SP medication uptake, according to Bajaria et al. [13]. They suggested improved ways to guarantee that the pharmaceuticals are refilled before depletion13. It has been claimed that strengthening Uganda’s public health institutions’ supply chain system has reduced the issue of SP medicine shortages in hospitals, which has increased the number of pregnant women utilizing the medication14. According to Ogba et al.15, the government should create public-private partnerships to enhance the sustainable supply of IPTp-SP medications in facilities so that all pregnant women may access them whenever needed or recommended.
Similar to the current study, Darteh et al.16 found that distance from the facility hindered SP medication uptake. Traveling long distances to ANC facilities may affect the number of visits, particularly in rural areas with inadequate communication and transportation infrastructure. Mothers who travel great distances to receive ANC therapies may not be patient, particularly in crowded settings. This affects their attitudes toward the necessity of prenatal clinic services even in the absence of illness. Lengthy waiting time makes consumers impatient and discourages them from coming back. According to Amankwah and Anto17, mothers may also bear some of the expense of treatment since they choose to choose institutions that provide reasonably priced services and don’t get the minimum number of ANC visits as recommended by the WHO.
Staff shortages and low motivation were among the problems cited by Ogba et al.15 that contributed to the prolonged waiting period and compromised the number of SP medicines the patient could get. With their dispersed location, rural facilities in Kenya and Busia County have a staffing problem18. Utilizing community health volunteers, other community-based tactics, and decongesting facilities may help bridge the gap by bringing services closer to moms19. While it’s true that lengthy wait times could not be resolved in the short term, they should nonetheless be gradually addressed.
Limitations
The antecedent-consequent bias is a prevalent feature of cross-sectional study design and therefore not the best for establishing cause- and-effect relationships. The recruited mothers had given birth after 28 weeks of pregnancy, there is a chance that bias would have been introduced in the inclusion of participants, especially in the determination of the results on the health system factors affecting uptake, however this study population is the best to determine the prevalence because these are mothers who have gone through the pregnancy to term and it is expected they have gotten the optimal doses of IPTp-SP.
Conclusion
Less than half of the pregnant women in Busia County are taking the three or more doses of IPTp-SP. Even though it is often understood that preventing pregnancy issues requires preventing malaria, there are essential elements that impede the medication’s appropriate uptake. The medication availability, distance to the ANC facility, and waiting period were health system-related factors substantially linked to the uptake of three or more doses of IPTp-SP. These results show that the availability of services and access at ANC significantly determines the number of doses of IPTp-SP medications taken to prevent malaria.
This study has demonstrated the significance of increasing the number of mothers receiving ANC services, educating the public about IPTp-SP drug benefits, dosages, and access points, managing stock out or SP costs, and minimizing doses missed due to circumstances outside the clients’ control.
Contributor Information
A.N. Miatu, School of Public Health, Department of Environmental Health and Disease Control, Jomo Kenyatta University of Agriculture and Technology, P.O. Box 62 000 − 00200 Nairobi, Kenya
B.R. Cheriro, School of Public Health, Department of Environmental Health and Disease Control, Jomo Kenyatta University of Agriculture and Technology, P.O. Box 62 000 − 00200 Nairobi, Kenya
S.P. Kamija, School of Global and Public Health, Kamuzu University of Health Sciences, Private Bag 360 Chichiri, Blantyre 3, Malawi
References
- 1.World Health Organization. World Malaria Report. 2021. [accessed: 22/6/2022]. https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2021 .
- 2.Kenya Malaria Indicator Survey. Kenya Malaria Indicator Survey. 2015. https://dhsprogram.com/pubs/pdf/MIS22/MIS22.pdf 22/6/2022.
- 3.Wanyonyi WA, Mulambalah CS, Mulama DH, Omukunda E. Malaria prevalence and risk analysis among pregnant women in Bungoma county, Kenya. Med Sci. 2019;8(2):301–6. [Internet] Available from: www.medicinescience.org. [Google Scholar]
- 4.Guyatt HL, Snow RW. Malaria in pregnancy as an indirect cause of infant mortality in sub-Saharan Africa. Trans R Soc Trop Med Hyg. 2001;95(6):569–76. doi: 10.1016/s0035-9203(01)90082-3. [Internet] [DOI] [PubMed] [Google Scholar]
- 5.Nyamu GW, Kihara JH, Oyugi EO, Omballa V, El-Busaidy H, Jeza VT. Prevalence and risk factors associated with asymptomatic Plasmodium falciparum infection and anemia among pregnant women at the first antenatal care visit: A hospital based cross-sectional study in Kwale County, Kenya. PLoS One. 2020;15(10):e0239578. doi: 10.1371/journal.pone.0239578. [Internet] Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0239578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.WHO. WHO | Intermittent preventative treatment to reduce the risk of malaria during pregnancy. WHO; 2019. [cited 2023 Nov 29]. [Internet] Available from: https://www.who.int/tools/elena/interventions/iptp-pregnancy. [Google Scholar]
- 7.Kenya Malaria Indicator Survey. Kenya Malaria Indicator Survey. 2020. https://dhsprogram.com/pubs/pdf/MIS36/MIS36.pdf: 22/6/2022.
- 8.Busia County. County Government of Busia County Integrated Development Plan 2018-2022 II Busia County Vision. 2022. [Internet] Available from: https://www.devolution.go.ke/wp-content/uploads/2020/02/Busia-CIDP-2018-2022.pdf.
- 9.Israel GD. Determining Sample Size 1. 1992. pp. 1–5. [Internet] Available from: https://www.psycholosphere.com/ Determining sample size by Glen Israel.pdf.
- 10.Karoki SM, Kariuki L, Owiti PO, Takarinda KC, Kizito W, Edwards JK, et al. Intermittent preventive treatment and Bed Nets uptake among pregnant women in Kenya. East Afr Med J. 2016;93(10):S16–21. [Google Scholar]
- 11.Pons-Duran C, Llach M, Sacoor C, Sanz S, Macete E, Arikpo I, et al. Coverage of intermittent preventive treatment of malaria in pregnancy in four sub-Saharan countries: findings from household surveys. Int J Epidemiol. 2021;50(2):550–9. doi: 10.1093/ije/dyaa233. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Martin MK, Venantius KB, Patricia N, Bernard K, Keith B, Allen K, et al. Correlates of uptake of optimal doses of sulfadoxine-pyrimethamine for prevention of malaria during pregnancy in East-Central Uganda. Malar J. 2020;19(1) doi: 10.1186/s12936-020-03230-8. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bajaria S, Festo C, Mrema S, Shabani J, Hertzmark E, Abdul R. Assessment of the impact of availability and readiness of malaria services on uptake of intermittent preventive treatment in pregnancy (IPTp) provided during ANC visits in Tanzania. Malar J. 2019;18(1) doi: 10.1186/s12936-019-2862-3. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rassi C, Graham K, Mufubenga P, King R, Meier J, Gudoi SS. Assessing supply-side barriers to uptake of intermittent preventive treatment for malaria in pregnancy: a qualitative study and document and record review in two regions of Uganda. Malar J. 2016;15(1) doi: 10.1186/s12936-016-1405-4. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ogba P, Baumann A, Chidwick H, Banfield L, DiLiberto DD. Barriers and facilitators to access and uptake of intermittent preventive treatment with sulfadoxine-pyrimethamine among pregnant women in Nigeria: a scoping review. MalariaWorld J. 2022;13:4. [Internet] [PMC free article] [PubMed] [Google Scholar]
- 16.Darteh EKM, Dickson KS, Ahinkorah BO, Owusu BA, Okyere J, Salihu T, et al. Factors influencing the uptake of intermittent preventive treatment among pregnant women in sub-Saharan Africa: a multilevel analysis. Arch Public Heal. 2021;79(1):1–9. doi: 10.1186/s13690-021-00707-z. [Internet] Available from: https://archpublichealth.biomedcentral.com/articles/10.1186/s13690-021-00707-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amankwah S, Anto F. Factors Associated with Uptake of Intermittent Preventive Treatment of Malaria in Pregnancy: A Cross-Sectional Study in Private Health Facilities in Tema Metropolis, Ghana. J Trop Med. 2019;2019 doi: 10.1155/2019/9278432. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mutanyi JA, Onguru DO, Ogolla SO, Adipo LB. Determinants of the uptake of intermittent preventive treatment of malaria in pregnancy with sulphadoxine pyrimethamine in Sabatia Sub County, Western Kenya. Infect Dis poverty. 2021;10(1) doi: 10.1186/s40249-021-00887-4. [Internet] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Odjidja EN, Duric P. Evaluation of demand and supply predictors of uptake of intermittent preventive treatment for malaria in pregnancy in Malawi. MalariaWorld J. 2017;8(20):20. [Internet] [PMC free article] [PubMed] [Google Scholar]