Abstract
Background
Fabry disease (FD) is an X-linked lysosomal storage disorder caused by α-galactosidase A (α-Gal A) deficiency, resulting in multi-organ accumulation of sphingolipid, namely globotriaosylceramide (Gb3). This triggers ventricular myocardial hypertrophy, fibrosis, and inflammation, driving arrhythmia and sudden death. Atrial fibrillation (AF) is common, yet the cellular mechanisms accounting for this are unknown.
Methods
To address this, we conducted electrocardiography (ECG) analysis from a large cohort of 115 adults with FD at varying cardiomyopathy stages. ECG P-wave characteristics were compared with non-FD controls. Cellular contractile and electrophysiological function were examined in a novel atrial cellular FD model developed and imputed into in-silico atrial models to provide insight into mechanisms of arrhythmia. Induced pluripotent stem cells were genome-edited using CRISPR-Cas9 to introduce the GLA p. N215S variant and differentiated into atrial cardiomyocytes (iPSC-CMs). Contraction, calcium handling and electrophysiology experiments were conducted. Bi-atrial in-silico models were developed with cellular changes as in GLA p. N215S iPSC-CMs.
Results
ECG analysis demonstrated P-wave duration and PQ interval shortening in FD adults prior to onset of cardiomyopathy. FD patients exhibited a higher incidence of premature atrial contractions and increased risk of AF compared to healthy controls. GLA p. N215S iPSC-CMs were deficient in α-Gal A and exhibited Gb3 accumulation. Atrial GLA p. N215S iPSC-CMs demonstrated a more positive diastolic membrane potential, faster action potential upstroke velocity, greater incidence of delayed afterdepolarizations, greater contraction force and alterations in calcium handling compared to wildtype iPSC-CMs. Simulations with these changes in the in-silico models resulted in similar P-wave morphology changes to those seen in early FD cardiomyopathy and increased AF vulnerability.
Conclusions
These findings provide novel insights into underpinning mechanisms for atrial arrhythmia and a rationale for early P-wave changes in FD. These may be targeted to develop therapeutic strategies to reduce the arrhythmic burden in FD.
Keywords: Fabry, atrial cardiomyocytes, arrhythmia, globotriaosylceramide (Gb3), electrocardiography, Induced pluripotent stem cells (iPSCs)
Nonstandard Abbreviations and Acronyms
- α-GAL A
Alpha-Galactosidase A
- AF
Atrial Fibrillation
- ANOVA
Analysis of Variance
- AP
Action Potential
- APD
Action Potential Duration
- AP
Action Potential
- AV
Atrioventricular
- BP
Blood Pressure
- CMs
Cardiomyocytes
- CMR
Cardiovascular Magnetic Resonance
- CRISPR
Clustered Regularly Interspaced Short Palindromic Repeats
- CTD
Calcium Transient Duration
- DAD
Delayed Afterdepolarization
- DNA
Deoxyribonucleic Acid
- ECG
Electrocardiography
- ERT
Enzyme Replacement Therapy
- EPIC
Echocardiography Imaging System
- FACS
Fluorescence Activated Cell Sorting
- FD
Fabry Disease
- Gb3
Globotriaosylceramide
- GLA
Galactosidase-α
- HS
High-Sensitivity
- iPSCs
Induced Pluripotent Stem Cells
- IQR
Interquartile Range
- IRAS
Integrated Research Application System
- LA
Left Atrial
- LVEDP
Left Ventricular End-Diastolic Pressure
- LVH
Left Ventricular Hypertrophy
- MRI
Magnetic Resonance Imaging
- NTpro-BNP
N-Terminal pro B-Type Natriuretic Peptide
- OCT
Oral Chaperone Therapy
- PAC
Premature Atrial Complex
Portable Document Format
- qPCR
Quantitative Polymerase Chain Reaction
- S1-S2
Electrophysiological Simulation Protocol
- SD
Standard Deviation
- TTE
Transthoracic Echocardiography
- UK
United Kingdom
- WT
Wildtype
Introduction
Fabry disease (FD) is a lysosomal storage disorder due to pathogenic variants in the galactosidase-α (GLA) gene resulting in the deficiency of α-galactosidase A (α-GAL A)1. This results in the accumulation of glycosphingolipids, in particular globotriaosylceramide (Gb3), in lysosomes of multiple organs2, with cardiovascular involvement the most common cause of morbidity and mortality3. Glycosphingolipid accumulation takes place in all cardiac cell types, triggering left ventricular hypertrophy (LVH), myocardial fibrosis and inflammation, and arrhythmia. Global prevalence is estimated to range from 1:40,000 to 1:170,000 live births4. Symptoms such as palpitations are common and occur in up to 50% of women and 75% of men. The incidence of atrial fibrillation (AF) is high at over 12%5. Multiple structural changes are thought to contribute to the causation of AF including passive left atrial (LA) dilatation due to elevated left ventricular end-diastolic pressure (LVEDP), as well as direct atrial Gb3 accumulation resulting in atrial myopathy6. In all populations, AF is associated with greater morbidity, including stroke, heart failure, and cardiovascular mortality7. The risks from AF are high in those with FD, with stroke prevalence reported up to 6.9% in males and 4.3% in females8. Furthermore, heart failure symptoms are reported in up to 25% of FD patients in AF in large cohort studies9.
Electrical changes can be detected early in FD on 12-lead electrocardiography (ECG)10. Preliminary evidence from small cohorts of FD patients without LVH, suggest shortening of the P-wave duration and QRS-width, indicative of accelerated depolarization. Increased repolarization times have also been demonstrated in the same cohort manifesting as QTc prolongation10–12. However, detailed ECG analysis measured at varying disease stages in larger cohorts of FD patients is currently lacking.
The cellular mechanisms underpinning pro-arrhythmic atrial electrical remodelling in FD are currently unknown. Enhancing our mechanistic understanding of atrial involvement in FD could identify early therapeutic targets, allow for robust risk-stratification for stroke, and may trigger initiation of disease-specific therapy to reduce the burden of cardiac complications.
We conducted signal averaged P-wave analysis from ECGs acquired in adults with FD and correlated findings with an atrial model of FD using genome edited induced pluripotent stem cell-derived atrial cardiomyocytes (iPSC-CMs). To provide further insights, we developed in-silico atrial models with clinical and cellular data imputed to understand how they may provoke arrhythmia.
Methods
The data that support the findings of this study are available from the corresponding author upon reasonable request.
12-lead electrocardiography (ECG)
Routinely collected 12-lead ECGs were acquired from 115 adults with FD attending the centre for rare disease at the Queen Elizabeth Hospital, Birmingham, UK, between July 2014 and November 2023. The most recent ECGs were obtained from patients in sinus rhythm, to include those who subsequently developed AF. Patients were scored for cardiac disease stage using a combination of biochemical, imaging, and electrocardiographic parameters as previously described13. Two Fabry experts (AR and RPS) reviewed the staging of patients, with discrepancies resolved by consensus. ECGs were also acquired from 40 age/sex-matched healthy non-FD controls for comparison.
ECGs were independently analysed by two experienced readers (CO and HC). Firstly, the presence of premature atrial complexes (PACs) was quantified. Secondly, 10 seconds of the lead II signal from the ECG portable document formats (PDFs) were digitised using an in-house developed algorithm and analyzed as previously described14. Briefly, all R-waves were identified and averaged to produce the averaged ECG complex. The two observers then individually identified P-wave start and end, QRS start and end, and T-wave start and end. The isoelectric line was defined from start to end of the P-wave, and duration and amplitude parameters automatically measured. Inter-observer variability is illustrated in Supplemental Figure 1.
Transthoracic Echocardiography (TTE)
TTE data were routinely collected by an accredited sonographer (AMA) using ie33 and EPIC ultrasound systems (Phillips) according to the British Society for Echocardiography minimum dataset15. Biplane left atrial (LA) volume was collected from the TTE report where available.
Haematology and Biochemistry
Data for haematology and biochemistry tests performed at the nearest date to ECG acquirement were additionally extracted and included high-sensitivity (HS) troponin I, and N-terminal pro B-type natriuretic peptide (NTpro-BNP). NTpro-BNP was measured by sandwich immunoassay with magnetic particle separation and chemiluminescent detection on an E170 analyser (Roche Diagnostics, Burgess Hill, United Kingdom)
Generation of genome-edited iPSCs
iPSCs were engineered with the pathogenic variant via CRISPR-Cas9 mediated genome-engineering to introduce the GLA p. N215S variant into male KOLF2 iPSC line as detailed in (Supplemental Methods 1.0) GLA p. N215S gives rise to a predominantly cardiac phenotype in FD, without overt effects in other organs16. It is the most prevalent mutation in the United Kingdom17. As wildtype (WT) controls, cells which were not carrying the GLA variant but had undergone the same genome editing procedure were used. Three independent clones were established, checked for pluripotency and correct karyotype (Supplemental Figures 2-4).
Generation of atrial iPSC-derived cardiomyocytes
GLA p. N215S iPSCs were differentiated into atrial iPSC-CMs using an established protocol18 (Supplemental Figure 5). Under the same conditions, WT iPSCs were differentiated into atrial iPSC-CMs. For all investigations, a minimum of three independent batches of WT and GLA p. N215S iPSC-CMs were utilized. Glucose starvation and culture to day 30 were performed to ensure maturity.
Confirmation of model
The model was confirmed and validated by Western blotting for α-GAL A deficiency in GLA p. N215S iPSC-CMs, immunofluorescence for Gb3 over-accumulation in GLA p. N215S iPSC-CMs and quantitative polymerase chain reaction (qPCR) for expression of atrial and ventricular markers. Detailed methods are described in (Supplemental Methods 2.0, 3.0 and 4.0).
Assessment of electrophysiological properties: Patch clamping
Stimulated and spontaneous APs were recorded from iPSC-CMs using a manual current clamp configuration. iPSC-CMs were seeded onto Geltrex (Thermo Fisher Scientific A1413201) coated coverslips 30 days after the initiation of differentiation. APs were recorded at 37 °C using an internal solution containing (in mM): KCl 135, NaCl 10, MgATP 5, HEPES 10, EGTA 0.1, pH 7.2 (adjusted using KOH). Micropipette resistances were between 3-4 MΩ. The extracellular solution contained (in mM): NaCl 145, KCl 5.4, MgSO4 0.83, NaH2PO4 0.33, HEPES 5, Glucose 11, CaCl2 1.8, pH 7.4 (NaOH). A 60 second period after breakthrough allowed cell stabilization. Spontaneous APs were recorded for 60 seconds prior to stimulation. A continuous hyperpolarizing current was then applied to the cells to hold the diastolic membrane potential at − 75 mV prior to recording of stimulated Aps19,20. Only those cells that required a hyperpolarizing current of less than 150 pA were used19. APs were stimulated using a 1 nA, 2 ms current injection at a frequency of 1 Hz20.10 successive spontaneous and stimulated APs were analyzed from each cell using custom algorithms developed in MATLAB.
Delayed afterdepolarizations (DADs) may develop after the repolarization phase of an action potential, are triggered by dysregulated Ca2+ homeostasis, and are thought to be pro-arhythmic in nature. These were visualized and quantified during analysis. DADs were defined as a sustained spontaneous depolarisation of greater than or equal to 10mV taking place following terminal repolarization, during the diastolic interval. Quantification of atrial ion channel expression was assessed using qPCR as described in Supplemental Methods 4.0.
Assessment of contractile properties
Contractile properties were assessed using a GoPro Inc. camera at 20x magnification. Video recordings were obtained in multiple areas of the same well and for multiple wells of beating iPSC-CM monolayers. These were 20 second videos recorded at room temperature in a quiet setting with no other activity in the room of recording to ensure minimal interference. Videos were converted to TIFF format using DaVinci Resolve (Blackmagic Design). A TIFF-stack and single sub-stack file was then created using a script run in ImageJ. The sub-stack file was then run using a MUSCLEMOTION21 script on ImageJ (add-on Macro); a validated tool for quantitative analysis of cardiac contraction by determining changes in pixel intensity between image frames. Outputs expressed were measures of movement during contraction and relaxation. Three 20-second videos were taken in each well with six wells for each batch The MUSCLEMOTION script generates a graphical representation of contraction and relaxation; expressed outputs are outlined below:
Contraction amplitude – Peak contraction from beginning of wave.
Peak amplitude – Peak contraction force from 0.
Relaxation time – Interval between peak contraction and beginning of following contraction.
Time to peak - Interval between beginning of contraction and peak contraction.
Peak to-peak - Interval between each contraction.
Assessment of calcium transients: Optical mapping
At day 17 of the differentiation protocol, atrial iPSC-CMs were split into dishes to form a confluent beating monolayer of atrial iPSC-CMs. Between days 23 and 27, calcium optical mapping was performed using intracellular calcium dye Fura-2-AM (Invitrogen). Cells were incubated with 5 µM Fura-2-AM in 1 mL RPMI B27 + Insulin (Thermo Fisher Scientific 17504044) at 37°C for 20 minutes, followed by a further 20 minutes in Tyrode’s solution (Concentrations in mM: NaCl 129, KCl 5.4, HEPES 10, MgCl2 48, CaCl2 1.8, and D-Glucose 9.99, pH 7.44-7.48).
Dye loaded cells were excited at 380 nm, and emission imaged at 10x magnification through 510/40 nm bandpass filter. Images were collected at 1.7 ms exposure time (588 Hz) using an Evolve delta 512x512 EMCCD camera. Binning was set to 10, giving a final resolution of 51x51 pixels, pixel size = 8 µm. Images were collected for 10 to 30 seconds using WinFluor (University of Strathclyde, UK), and converted to .MAT files for analysis with ElectroMap22. Images were pre-conditioned with a 3x3 gaussian spatial filter. Time to peak was measured from 10 to 90% calcium intrusion before peak. Calcium transient duration (CTD) was measured to 30, 50 and 80% extrusion from both maximum upstroke time (the maximum positive differential) and peak amplitude. Calcium conduction velocity was measured using published methods with a 5x5 local window size23.
Computer modelling and simulation of human atrial electrophysiology
The methodology employed for modelling and simulation is detailed in Supplemental Methods 5.0 and briefly summarized here. In-silico populations of atrial cardiomyocyte models were developed to replicate the mechanisms underlying electrophysiological differences between GLA p. N215S and WT atrial iPSC-CMs. Two populations were built, one considering atrial cardiomyocyte models with action potential and calcium transient characteristics as reported in GLA p. N215S atrial iPSC-CMs, and one as in WT atrial iPSC-CMs. From these populations, the 10 atrial cardiomyocyte models that better captured the electrophysiological changes induced by GLA p. N215S iPSC-CMs and 10 representatives of WT were integrated in a human bi-atrial model to produce 20 virtual patients.
The atrial anatomy was derived from a human bi-atrial statistical shape model, based on 47 (40% female) clinical computed tomography and cardiac magnetic resonance scans24. This bi-atrial anatomy presented accurate fibre orientation, intra-atrial bridges, electrophysiological differences in different atrial regions, and the characteristic LA (and equivalent right atrial) volume of patients showing PACs on the ECG. PACs were also replicated in-silico, by assessing changes in the P-wave morphology after applying ectopic stimuli in different locations of the human atrial model (i.e., crista terminalis and pulmonary veins). The comparison of the P-wave morphology between in-silico and clinical PACs was used to estimate the initiation side of abnormal electrical impulses. Subsequently, the arrhythmogenicity of the electrophysiological substrate induced by FD was assessed by applying burst stimulation on the ectopic sides identified as generators PACs.
First, 10 sinus rhythm beats were applied at a cycle length of 700 ms (i.e., 85 bpm) in the 20 virtual patients (i.e., 10 representatives of GLA p. N215S and 10 of WT iPSC-CMs). Subsequently, 12 burst stimulation protocols were independently assessed in each virtual patient using a constant coupling interval of 400 ms between the last sinus rhythm and the first ectopic beat (i.e., shortest interval enabling capture). In all 12 protocols, the burst considered five periodic stimuli but was applied at different cycle lengths, ranging from 150 to 260 ms with 10 ms increment25. Arrhythmia vulnerability was computed in each virtual patient as the number of bursts inducing AF over the total number of burst protocols (i.e., 12).
The three-dimensional monodomain equation of the transmembrane voltage and all ECG calculations were solved using the high-performance open-source MonoAlg3D26.
Statistical Analysis
Where two variables were being compared, normality was assessed using the Shapiro-Wilk test. If the data was normally distributed and groups had similar standard deviation (SD), unpaired t-tests were performed. If the data was normally distributed with unequal SDs, unpaired t-tests with Welch’s correction were performed. Where data was not normally distributed, the Mann-Whitney test was used. Where more than two variables were being compared, an Ordinary one-way ANOVA was performed with Turkey’s multiple comparisons test for normally distributed data. For non-normally distributed data where more than two variables were compared, the Kruskal-Wallis test was performed. For categorical and contingency data, Fishers exact test was performed. Continuous variables are reported as mean ± SD where approximately normally distributed, with non-normal variables reported as median (interquartile range; IQR) unless stated otherwise. Data was analysed and presented in GraphPad Prism version 10.0.0 with p<0.05 deemed to be indicative of statistical significance throughout. A full list of abbreviations is detailed in Supplemental Methods 6.
The use of 12-lead ECGs in the FD cohort was approved by West Midlands – South Birmingham Research Ethics Committee (23/WM/0180 IRAS 325613). The studies were conducted in accordance with the local legislation and institutional requirements. The Ethics Committee / institutional review board waived the requirement of written informed consent for participation from the participants or participant’s legal guardians / next of kin because data were acquired from a research database using routinely collected clinical data for the purpose of research. The use of 12-lead ECGs in healthy controls was approved by the West Midlands Solihull Research Ethics Committee (17/WM/0048) and approved by the Health Research Council. All healthy controls gave informed consent to take part in accordance with the principles set out in the Declaration of Helsinki.
iPSCs were engineered with the pathogenic GLA p. N215S variant with the support of the Genome Engineering Core Facility at the Wellcome Centre for Human Genetics, University of Oxford. The variant was introduced into male iPSCs (KOLF2 line). KOLF2 are Human Induced Pluripotent Stem Cell Initiative lines from a consortium at the Sanger Institute. They do not fall under the Human Tissue Act (2004) as the volunteer has given prior consent under an open access agreement.
Results
Early atrial changes on ECGs of FD patients
The cohort demographics and clinical characteristics are described in (Table 1). The patients were predominantly female, of middle age and white Caucasian which reflects our clinical patient population. 43 (37%) had a classical FD mutation and 72 (63%) had the GLA p. N215S non-classical cardiac variant. At the time of ECG acquisition, 35 (30%) patients were on enzyme replacement therapy (ERT) and 26 (23%) on oral chaperone therapy (OCT). AF was present in 16 (14%) and therefore the ECG analyzed was that recorded when they were last in sinus rhythm. Age, male predominance, body-mass index, systolic BP and LA volume increased with cardiac disease stage. There was also a trend to worsening of cardiac, renal and AF-associated biomarkers with elevations in NTpro-BNP, HS troponin I and serum creatinine with cardiac disease stage.
Table 1. Cohort Characteristics of FD and Healthy control cohorts, total and according to cardiac disease stage.
| Control (N=40) |
All (N=115) | Stage 1 (N=39) | Stage 2 (N=30) | Stage 3 (N=24) | Stage 4 (N=22) | P-Value | |
|---|---|---|---|---|---|---|---|
| Demographics | |||||||
| Age (Years) | 51.0 ± 11.7 | 45.4 ± 17.4 | 30.4 ± 11.9 | 42.6 ± 13.3 | 57.6 ± 10.0 | 62.4 ± 12.3 | <0.0001 |
| Female Sex N (%) | 27 (68) | 63 (55) | 31 (80) | 13 (43) | 11 (46) | 8 (36) | |
| Ethnicity N (%) | |||||||
| White Caucasian | 34 (85) | 93 (81) | 28 (72) | 23 (77) | 21 (88) | 21 (96) | |
| Black and Minority Ethnic | 6 (15) | 22 (19) | 11 (28) | 7 (23) | 3 (12) | 1 (4) | |
| Height (m) | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | 0.2190 |
| Weight (kg) | 76 ± 12 | 76 ± 21 | 77 ± 18 | 72 ± 18 | 82 ± 27 | 76 ± 23 | 0.4457 |
| BMI (kg/m2) | 27 ± 4 | 27 ± 7 | 28 ± 5 | 25 ± 6 | 29 ± 8 | 27 ± 7 | 0.0258 |
| Systolic BP (mmHg) | 122 ± 16 | 133 ± 17 | 127 ± 14 | 132 ± 20 | 141 ± 15 | 136 ± 19 | 0.0125 |
| Diastolic BP (mmHg) | 79 ± 9 | 79 ± 11 | 79 ± 8 | 75 ± 12 | 82 ± 11 | 80 ± 12 | 0.1541 |
| Bloods | |||||||
| Creatinine (umol/L) | 74 [60-96] | 61 [65-70] | 74 [67-90] | 89 [61-110] | 94 [81-117] | <0.0001 | |
| NT-proBNP (ng/L) | 128 [51-490] | 51 [32-115] | 68 [41-104] | 243 [147-641] | 880 [356-2343] | <0.0001 | |
| Troponin I (ng/L) | 8 [4-37] | 5 [4-5] | 7 [4-12] | 29 [14-78] | 122 [60-230] | <0.0001 | |
| Haemoglobin (g/L) | 135 ± 14 | 131 ± 10 | 138 ± 14 | 138 ± 17 | 133 ± 16 | 0.7930 | |
| Comorbidities | |||||||
| Stroke N (%) | 0 (0) | 9 (8) | 1 (3) | 3 (10) | 1 (4) | 4 (18) | |
| Hypercholesterolaemia N (%) | 7 (18) | 35 (30) | 3 (8) | 6 (20) | 16 (67) | 10 (46) | |
| Ischaemic Heart Disease N (%) | 0 (0) | 7 (6) | 0 (0) | 2 (7) | 1 (4) | 4 (18) | |
| Statin Therapy N (%) | 5 (13) | 34 (30) | 1 (3) | 7 (23) | 14 (58) | 12 (55) | |
| Current Smoker N (%) | 1 (2.5) | 8 (7.0) | 1 (2.6) | 3 (10.0) | 2 (8.3) | 2 (91) | |
| Fabry Therapy | |||||||
| Enzyme Replacement Therapy N (%) | 35 (30) | 7 (18) | 10 (33) | 8 (33) | 10 (45) | ||
| Oral Chaperone Therapy N (%) | 26 (22.6) | 2 (5.1) | 8 (26.7) | 11 (45.8) | 5 (22.7) | ||
| ECG and TTE | |||||||
| Rate (bpm) | 63 ± 15 | 64 ± 13 | 68 ± 12 | 61 ± 11 | 64 ± 15 | 60 ± 12 | 0.0888 |
| P wave duration (ms) | 110 ± 17 | 106 ± 18 | 96 ± 16 | 108 ± 14 | 110 ± 15 | 116 ± 20 | <0.0001 |
| P wave area (mV/ms) | 6.2 ± 3.3 | 7.9 ± 3.9 | 5.9 ± 3.7 | 8.8 ± 3.8 | 8.2 ± 3.7 | 8.5 ± 3.9 | 0.0015 |
| P wave amplitude (mV) | 0.085 ± 0.038 | 0.12 ± 0.041 | 0.10 ± 0.048 | 0.13 ± 0.043 | 0.12 ± 0.041 | 0.12 ± 0.035 | <0.0001 |
| PQ interval (ms) | 171 ± 30 | 159 ± 34 | 145 ± 26 | 162 ± 33 | 159 ± 26 | 177 ± 46 | 0.0013 |
| QRS duration (ms) | 89 ± 18 | 104 ± 22 | 94 ± 17 | 98 ± 16 | 107 ± 18 | 124 ± 27 | <0.0001 |
| QTc interval (ms) | 406 ± 53 | 402 ± 48 | 403 ± 25 | 390 ± 52 | 428 ± 48 | 428 ± 49 | 0.0086 |
| LA volume (ml) | 63 ± 17 | 68 ± 34 | 56 ± 15 | 54 ± 25 | 86 ± 38 | 84 ± 43 | <0.0001 |
BMI: body mass index, BP: Blood pressure, LA: left atrium, ECG: electrocardiography, TTE: transthoracic echocardiography, NT-proBNP: N-terminal pro brain natriuretic peptide. Data presented as N (%) or mean ± SD. P-values derived from ordinary one-way ANOVA performed for normally distributed data. Kruskal-Wallis test performed for non-normally distributed data.
P-wave morphology changes in non-FD controls, stage 1 FD and stage 4 FD are illustrated in (Figure 1A). There was a significant shortening of P-wave duration in adults with stage 1 cardiac phenotype FD compared to non-FD controls (96 ± 16 ms versus 110 ± 17 ms; p=0.0026) (Figure 1B). PQ interval shortening in FD stage 1 was also present when compared to non-FD controls (145 ± 27 ms versus 171 ± 30 ms; p=0.0043) (Figure 1C). Prolongation of P-wave duration and PQ interval were seen to be associated with worsening severity of FD cardiac phenotype (stages 1-4) (Figures 1A-C and Table 1). Prolongation of P-wave duration and PQ interval in the later stages of cardiac disease was associated with greater LA volume on TTE (Figure 1D). P-wave duration and PQ interval were also shorter in patients with stage 1 FD in the GLA p. N215S cohort compared to healthy controls (96 ± 18 ms versus 110 ± 17 ms; p=0.0026)
Figure 1.
12-lead ECG analysis: (A) P-wave morphology changes on ECG (control vs stage 1 vs stage 4) (B-C) P-wave duration and PQ interval changes in controls and FD cardiomyopathy stages (D) LA volume changes in controls and FD cardiomyopathy stages on TTE. (E) Proportion of patients with AF Fabry vs control (F) P-wave duration in AF vs no AF (G) PAC on ECG (lead II) of adult with FD (H) Proportion of PACs Fabry Vs control (I) ECG (lead II) of AF in a patient with FD stage 1. Ordinary one-way ANOVA with Turkey’s multiple comparisons statistical test used comparing stages. Data presented as median ± 25th-75th percentile with whiskers indicating minimum and maximum values. Mann Whitney Test used to compare P-wave duration in AF vs no AF. Data presented as median ± 25th-75th percentile with whiskers indicating the minimum and maximum values. Fisher’s exact test used for proportion of patients with PACs and AF. * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001
More FD patients had AF compared to non-FD controls (Figure 1E). In the sub-analysis of FD patients with AF (N=16) using their most recent ECGs in sinus rhythm, a more prolonged P-wave duration was documented compared to FD patients remaining in sinus rhythm (116 ± 13 ms versus 104 ± 18 ms; p=0.0094) (Figure 1F). The presence of PACs on ECG (Figure 1G) was observed in 14/115 adults with FD vs 0/40 for controls (Figure 1H). Prevalence of PACs in cardiac disease stages was not significantly different; Stage 1 (N=4/39), stage 2 (N=1/30), stage 3 (N=5/24) and stage 4 (N=4/22). The presence of AF on the ECG of a patient with FD stage 1 is illustrated in Figure 1I.
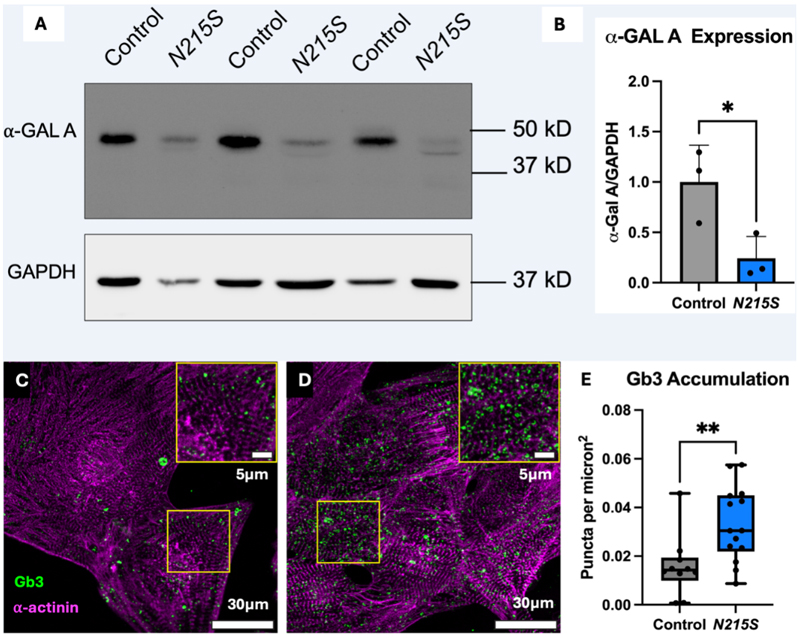
Successful generation of atrial FD model
Sanger sequencing confirmed the presence of GLA p. N215S at the CRISPR target site, fluorescence activated cell sorting (FACS) analysis confirmed high expression of pluripotent stem cell markers and normal chromosomal count was confirmed on karyotype analysis (Supplemental Figures 2-4). α-GAL A is a homodimer, consisting of two 49 kDA subunits. Denaturing western blotting confirmed reduced expression of α-GAL A in GLA p.N215S iPSC-CMs compared to WT (Figure 2A and Supplemental Figure 6). Significantly lower expression of α-GAL A in GLA p.N215S iPSC-CMs was also confirmed when normalized to WT (0.24 ± 0.22-fold change from WT at 1.0 ± 0.37; p=0.0368) (Figure 2B). When stained with a Gb3 antibody and visualised using confocal fluorescence microscopy, mature atrial GLA p.N215S iPSC-CMs displayed greater accumulation of Gb3 compared to WT (Figure 2C-D). On Gb3 quantification, GLA p.N215S iPSC-CMs exhibited a greater number of puncta per micron2 compared to WT (0.031: IQR 0.22-0.45 puncta per micron2 versus 0.014: IQR 0.099-0.019 puncta per micron2; p=0.0073) (Figure 2E). Markers of atrial and ventricular expression on qPCR are illustrated in (Supplemental Figure 7A-D). When normalized to GAPDH, atrial iPSC-CMs had greater expression of atrial marker MYL7 (1.9: IQR 1.2-3.7-fold change in atrial iPSC-CMs versus 0.87: IQR 0.82-1.4-fold change in ventricular iPSC-CMs: p=0.0184) and a trend to increased expression of MYH6, with reduced expression of ventricular markers MYL2 (0.14: IQR 0.067-0.023-fold change in atrial iPSC-CMs versus 0.62: IQR 0.39-4.1-fold change in ventricular iPSC-CMs; p=0.0021) and MYH7 (0.31: IQR 0.11-0.91-fold change in atrial iPSC-CMs versus 0.93: IQR 0.86-1.2-fold change in ventricular iPSC-CMs; p=0.0142) compared with ventricular iPSC-CM expression. All iPSC-CMs expressed the cardiac marker troponin (TNNT2) on qPCR (Supplemental Figure 7E).
Figure 2. Confirmation of FD model.
(A) Western blot for α-GAL A protein levels (control vs N215S) in iPSC-CMs. (B) Quantification of α-GAL A expression (control vs N215S) in iPSC-CMs. Data presented as mean ± SD with whiskers indicating the minimum and maximum values (C-D) Immunofluorescence Gb3 (green) stain using confocal microscopy (A: control B: N215S) in atrial iPSC-CMs (α-actinin, purple) (E) Gb3 accumulation (control vs N215S) in atrial iPSC-CMs. Data presented as median ± 25th-75th percentile with whiskers indicating the minimum and maximum values. Mann Whitney U statistical test used,* p=<0.05, ** p=<0.01.
Quicker upstroke in atrial APs of GLA p. N215S IPSC-CMs identified
Findings from single cell patch clamping assessment of atrial APs are summarised in (Figure 3). In the first step, we assessed spontaneous action potentials. GLA p. N215S atrial iPSC-CMs demonstrated a more positive resting diastolic membrane potential (Figure 3A) (-49 ± 7.1 mV versus -52 ± 7.1 mV; p=0.0153). These values suggest generation of mature of atrial iPSC-CMs in keeping with published literature27–29. There were no significant changes in firing frequency or APD30, 50, 70 or 90.
Figure 3.
(A-C) Diastolic membrane potential from the APs of spontaneously beating iPSC-CMs, upstroke and amplitude from stimulated APs from iPSC-CMs at 1Hz (WT vs N215S). (D) Single cell atrial stimulated APs (WT vs N215S) illustrating quicker upstroke in N215S. (E) Stimulated atrial APs (WT vs N215S) illustrating DAD activity in N215S (circled in red) compared to no DAD activity in WT. (F) DAD quantification from the stimulated APs of iPSC-CMs documenting a greater number of N215S iPSC-CMs with DAD activity compared to WT. Welch’s unpaired t-test statistical test used for Diastolic membrane potential. Data presented as mean ± SD with whiskers indicating minimum and maximum values. Mann Whitney U test used for Stimulated Upstroke and Amplitude. Data presented as median ± 25th-75th percentile with whiskers indicating the minimum and maximum values. Fisher’s exact test used for comparison of number of DADs. * p ≤ 0.05, *** p ≤ 0.001, **** p ≤ 0.0001.
In the second step, we assessed stimulated APs. When stimulating cells at a held resting potential of -75 mV with 1 nA, 2 ms current injection, GLA p. N215S atrial iPSC-CMs had a significantly faster action potential upstroke velocity (87: IQR 75-113 mV/ms versus 78: IQR 67-88 mV/ms; p=0.004) and a greater action potential amplitude (118 ± 20 mV versus 110 ± 16 mV; p=0.0095) (Figure 3B-D). There were no significant changes in APD30, APD50, APD70 or APD90 (Supplemental Figure 8A-D). APD90 values for GLA p. N215S atrial iPSC-CMs and WT were in a similar range to several previous studies investigating atrial iPSC-CMs28–31, and shorter than several studies on ventricular iPSC-CMs29,30,32,33. Similarly, no changes were observed in triangulation when measured as APD90:APD50 ratio (Supplemental Figure 8E) (1.38 ± 0.02 in WT vs 1.39 ± 0.02 in GLA p. N215S atrial iPSC-CMs; p=0.8). Previous studies have used a triangulation value of 1.3 as a cut-off for defining cells as atrial or ventricular30. By this measure, 63% of WT and 57% GLA p. N215S atrial iPSC-CMs exhibit “atrial-like” AP morphology.
The presence of DADs in a GLA p. N215S atrial iPSC-CM stimulated AP are illustrated in (Figure 3E). Quantitative assessment of stimulated APs showed an increased numbers of cells with APs displaying DADs in GLA p. N215S atrial iPSC-CMs (N=46) compared with WT (N=19) (Figure 3F) (p=0.0002). There were no significant changes identified in atrial ion channel expression for relevant sodium and potassium channels (Supplemental Figure 9A-H)
Greater contraction in atrial GLA p. N215S iPSC-CMs identified
Findings from MUSCLEMOTION analysis of contraction in the 2D monolayers are summarised in (Figure 4). Graphical representations of contraction and relaxation are illustrated for GLA p. N215S iPSC-CMs and WT iPSC-CMs (Figure 4A). Data from MUSCLEMOTION findings are expressed as normalized percentage changes of the mean of the WT. GLA p. N215S iPSC-CMs had a significantly longer contraction duration (119: IQR 103-145 % versus 98: IQR 86-115 %; p=0.0055) (Figure 4B) and time to peak (150: IQR 84-212 % versus 98: IQR 82-118 %; p=0.0296) (Figure 4C), as well as a longer peak-to-peak distance (145 ± 59 % versus 100 ± 16.9 %; p=0.0032) (Figure 4G) indicating a slower beat rate compared to WT. GLA p. N215S iPSC-CMs also generated a significantly higher contraction (141: IQR 103-194 % versus 101: IQR 65-129 %; p=0.0074) and peak (119: IQR 100-133 % versus 95: IQR 75-116 %; p=0.0047) amplitude (Figure 4D and E) compared to WT. There were no differences in relaxation time observed between GLA p. N215S iPSC-CMs and WT (Figure 4F).
Figure 4.
(A) Graphical outputs from MUSCLEMOTION in an N215S atrial iPSC-CM and (B) WT-iPSC-CM. (C-H) Intervals and contractility parameters from atrial iPSC-CMs (N215S vs WT) Values expressed as a percentage difference of the mean value for the WT of each batch. Mann Whitney U statistical test used for Contraction Duration, Time to Peak, Contraction Amplitude, Peak Amplitude and Relaxation time. Data presented as median ± 25th-75th percentile with whiskers indicating the minimum and maximum values. Welch’s unpaired t-test used for Peak to Peak time. Data presented as mean ± SD with whiskers indicating minimum and maximum values. ns not significant, * p ≤ 0.05, ** p ≤ 0.01.
Prolonged CTDs in atrial GLA p. N215S iPSC-CMs identified
To assess calcium handling, optical mapping was performed in the cellular monolayers. CTD was prolonged to all extrusion levels in GLA p. N215S atrial iPSC-CMs across the monolayers (Figure 5A-C): CTD30 (265 ± 35 ms versus 190 ± 37 ms; p=<0.0001), CTD50 (357 ± 37 ms versus 252 ± 54 ms; p=<0.0001) and CTD80 (587: IQR 535-647 ms versus 358: IQR 337-453 ms; p=<0.0001) (Figures 5D-F). GLA p. N215S atrial iPSC-CMs also displayed a significantly slower time-to-peak (Figure 5G) (96 ± 26 ms versus 77 ± 18 ms; p=0.0034), and larger peak amplitude (Figure 5H) (16325 ± 2595 au versus 13998 ± 2894 au; p=0.0004) suggesting a greater total cycling of calcium, and a slower beating frequency (Figure 5I) (35: IQR 27-38 bpm versus 56: IQR 40-70 bpm; p=<0.0001) consistent with contraction analysis data. Calcium conduction velocity was increased in the GLA p. N215S atrial iPSC-CMs compared to controls (0.89 ± 0.21 cm/s versus 0.63 ± 0.17 cm/s, p=<0.0001) (Figures 5J-L).
Figure 5.
(A) WT atrial iPSC-CM CTD map indicating shorter CTDs (blue), (B) N215S atrial iPSC-CM CTD map indicating longer CTDs (red), (C) Atrial calcium transients (N215S vs WT) illustrating changes in morphology and CTDs, (D-F) Atrial iPSC-CM CTD30, 50, 80 (N215S vs WT), (G-I) Interval and amplitude parameters (N215S vs WT), (J-L) Calcium conduction velocity assessment (N215S vs WT). Mann Whitney U statistical test used for CTD80, CTD50, Time to peak, Peak Amplitude, Frequency and calcium conduction velocity. Data presented as median ± 25th-75th percentile with whiskers indicating the minimum and maximum values. Welch’s Unpaired t-test used for CTD30. Data presented as mean ± SD with whiskers indicating minimum and maximum values * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001.
Increased AF vulnerability in FD patient models
An in-silico population of 222 human atrial cardiomyocyte models reproduced the faster upstroke and higher action potential amplitude observed in GLA p. N215S atrial iPSC-CMs (Figure 6A-B), as well as the slower time-to-peak, larger peak amplitude and longer duration of the calcium transient (Supplemental Figures 10A-E, 11A-D). Compared to 251 atrial cell models without these alterations, the calcium changes observed in GLA p. N215S atrial iPSC-CMs favoured an increase in the APD restitution slope and the appearance of APD alternans (Figure 6A).
Figure 6. In silico investigation of the proarrhythmic profile of Fabry Disease (FD).
A) Action potential duration (APD) restitution slope, action potential amplitude and maximum upstroke velocity of 251 and 222 human atrial cardiomyocyte models representative of WT and GLA p. N215S atrial iPSC-CMs, respectively. Time course of the transmembrane voltage of a representative cardiomyocyte model in each subgroup, showing action potential alternans in the model representative of GLA p. N215S atrial iPSC-CMs (in blue). B) Schematic representation of the human atrial model inside a torso with the electrode location. It is illustrated the location of the sinus node (stimulation during sinus rhythm, SR) and the direction of propagation of an ectopic beat originating from the left pulmonary veins. Abbreviations: RA-LA: right and left arm; RL-LL: right and left leg. C) Comparison of clinical and simulated P-waves, considering the representative atrial cardiomyocyte models shown in A). Simulated P-wave duration in the 10 virtual patients with FD and 10 patients in control conditions. Representative atrial activation map of one virtual patient in each subgroup. D) AF vulnerability showing that AF is induced only when considering the electrophysiological changes caused by FD. E) Consecutive snapshots of the atrial transmembrane voltage (Vm) and corresponding ECG in control conditions and FD. The blue arrows on top of the atrial anatomy indicate the presence of rotors. Since the QRS is not simulated, the atrial repolarization (Ta-wave) can be seen on the ECG. Abbreviations: RA-LA: right and left atrium; rPV-lPV: right and left pulmonary veins.
From these in-silico populations, 10 atrial cardiomyocyte models representative of FD, with faster upstroke, higher AP amplitude and APD alternans, and 10 without these alterations, representative of control, were used to describe the electrophysiology of a human bi-atrial model (right and left atrial volumes of 122 mL and 85 mL, respectively). During sinus rhythm, significantly shorter P-waves (i.e., faster atrial depolarization) were obtained in virtual patients with the FD phenotype, as observed clinically in patients with FD (Figure 6C). Faster depolarization was also evidenced by the shorter activation times and the faster calcium conduction velocity demonstrated in GLA p. N215S atrial iPSC-CMs compared to WT (Figure 5J-L)
This 3D atrial anatomy was used to estimate the ectopic origin of PACs in patients with FD. For this, ectopic stimuli were applied in different atrial sites of the human bi-atrial model (Supplemental Figure 12A) and the resulting changes in the P-wave morphology were compared against PACs on ECG. The comparison between simulated and clinical ECGs evidenced the left pulmonary veins as a potential ectopic trigger of PACs in patients with FD (Supplemental Figure 12B-C), in agreement with the greater number of DADs observed in GLA p. N215S atrial iPSC-CMs (Figure 3).
Thus, 12 AF-induction stimulation protocols were applied in the left pulmonary veins of the 10 virtual patients with FD and of the 10 control patients. A significant increase in AF vulnerability was observed in virtual patients with the FD phenotype (Figure 6D), since the associated electrophysiological changes favoured AF induction even in the absence of structural remodelling (Figure 6E, Supplemental video 1).
Discussion
In a deeply phenotyped cohort of patients with FD, staged according to degree of cardiomyopathy, we demonstrate P-wave duration and PQ interval shortening in adults with FD, even in those without an overt cardiac disease phenotype. This has not previously been demonstrated in “cardiac phenotype-negative” patients and for the first time suggests early atrial electrical remodelling secondary to Gb3 accumulation in patients with classical disease and with the GLA p. N215S variant, previously thought to be “late-onset”. P-wave duration and PQ interval shortening have been demonstrated previously in a smaller study of (N=30) FD patients without LVH, however these patients did not undergo multiparametric CMR and biomarker assessment to exclude early-onset cardiomyopathy due to myocardial Gb3 accumulation10. Crista terminalis and atrioventricular (AV) nodal Gb3 accumulation may account for P-wave duration shortening via accelerated conduction34–36. As cardiac disease stage progresses, P-wave duration and PQ interval appears to prolong, and a positive correlation is observed with LA volume on TTE. These findings suggest multiple mechanisms underlying the pathophysiology of atrial myopathy in FD. Firstly, in early disease, intrinsic electrophysiological cellular changes, presumably due to Gb3 accumulation, cause abnormalities in sodium and calcium handling that help to account for the early PQ interval and P-wave duration shortening on 12-lead ECG seen in stage-1 FD patients. These may be pro-arrhythmic in nature12. As Gb3 accumulation progresses and LA volume increases due to the passive effects of elevated LVEDP due to LVH, this structural alteration causes ‘pseudo-normalization’ of the P-wave duration and PQ interval, which eventually becomes prolonged, further increasing the susceptibility to the onset of AF37. These mechanisms according to disease stage are summarised in (Figure 7). These are important findings as they provide insight into underpinning mechanisms and may serve as biomarkers in the assessment of treatment response.
Figure 7.
Stages of cardiomyopathy in FD. Abbreviations: GLS: global longitudinal strain, LV: left ventricular, LGE: late gadolinium enhancement, LVH: left ventricular hypertrophy, NT-proBNP: N-terminal-pro brain natriuretic peptide.
Our cellular model suggests that enzyme deficiency and accumulation of unmetabolized Gb3 in atrial iPSC-CMs are associated with key alterations in cellular contractility, intracellular calcium handling, AP morphology and DAD occurrence. Similar findings have been demonstrated in ventricular iPSC-CMs from patient-derived iPSCs with FD including higher spontaneous AP frequency, shorter AP duration, increased sodium current density, and increased upstroke velocity; all suggesting increased excitability38. The same study demonstrated disruption to intracellular calcium handling, with transients exhibiting a greater amplitude and reduction in peak-width duration. This study, however, did not investigate atrial iPSCs and was not guided by in-vivo clinical data. We have demonstrated, for the first time, alterations in the atrial APs in GLA p. N215S iPSC-CMs. The shifting of diastolic membrane potential to a more positive value which is closer to the threshold potential for depolarization is pro-arrhythmic as it causes an increases myocyte excitability and may contribute to an increased risk of ectopic activity39. We have also demonstrated an increased AP upstroke velocity in atrial GLA p. N215S iPSC-CMs compared to WT. The AP upstroke is largely controlled by voltage-gated sodium channels driving the inward sodium current40. An increase in calcium conduction velocity, while generally considered anti-arrhythmic, has been proposed as a substrate for arrhythmia in atrial and ventricular CMs12. It has been suggested that rapid intra-atrial conduction velocity counteracts the fast inter-atrial conduction via Bachmann’s bundle, allowing for co-ordinated atrial depolarization. Changes in AP upstroke have also been demonstrated in patient-derived stem cell-derived ventricular CMs in FD38. Our findings in atrial CMs provide a mechanism for the observation of P-wave duration and PQ shortening as the earliest electrical manifestation of FD cardiomyopathy on 12-lead ECG10. The ECG data from adults with FD together with our data from atrial iPSC-CM, suggests intrinsic early electrical changes which may be secondary to Gb3 accumulation.
Contraction analysis results demonstrate significantly higher contraction and peak amplitude with prolonged peak-to-peak times in GLA p. N215S iPSC-CMs compared with WT21. Our study, for the first time, demonstrates intrinsic changes to the properties of atrial myocytes at a cellular level manifesting as increases in contraction amplitude, which may reflect greater contraction force, that are not due to passive effects of the elevated LVEDP and LVH. Published clinical data demonstrates that impairments in LA strain (contractile function) were weakly associated with parameters of LV diastolic and systolic function, implying that long standing atrial structural changes accounting for impaired LA strain in FD may overcome and negate the cellular effects which tend to increase contractile force.6
The changes observed in this study of a slower beat rate in GLA p. N215S iPSC-CMs compared to WT are in keeping with published literature which records that patients with FD are often bradycardic. Our data suggest the bradycardia may be a result of atrial cellular changes caused by Gb3 accumulation.
Another novel finding of this study is the identification of alterations in intracellular calcium handling in atrial GLA p. N215S iPSC-CMs including a greater transient amplitude and prolonged duration. Although it should be noted that amplitude measurements from optical mapping of non-ratiometric indicators are influenced by several non-physiological factors, including inhomogeneous loading and excitation, these results suggest an overall increase in calcium that is cycled into and out of the cytosol with each excitation41. This may explain the increase in contraction amplitude observed in these cells, and also aligns with published data in patient derived ventricular iPSC-CMs in FD which demonstrate a similar finding of greater amplitude38. Furthermore, the finding of a greater number of atrial GLA p. N215S iPSC-CMs exhibiting DAD activity compared to WT is consistent with a greater quantity of intracellular calcium which predispose to DADs42,43. DADs can trigger focal activity that can lead to arrhythmogenesis, a potential mechanism explaining the increased arrhythmia-burden in these patients, including for AF44. The higher level of DADs in GLA p. N215S iPSC-CMs may relate to a shift of the diastolic membrane potential in atrial APs to a more positive value, closer to the depolarization threshold, increasing myocyte excitability and focal ectopic activity risk. Further investigations are required to directly assess subcellular calcium dynamics in these cells.
When reproducing the observed calcium handling and action potential findings from GLA p. N215S iPSC-CMs in an in-silico atrial population, we found simulated P-waves with a shorter P-wave duration and PQ interval than P-waves without the cellular findings imputed. The P wave morphology was like that seen in the P-waves of patients with FD stage 1. This striking observation provides further evidence that intrinsic changes in atrial calcium handling and electrophysiology may, account for the abnormalities in P-wave duration and PQ interval seen in early FD cardiomyopathy. We also demonstrate an increase in the APD restitution slope, the appearance of APD alternans and (in our bi-atrial tissue model) AF inducibility by ectopic activity in the pulmonary veins. APD restitution is the rate-dependant adaptation of the AP45. Changes in APD restitution may provoke re-entrant arrythmia particularly in the context of a prolonged repolarization phase and in conjunction with premature stimuli46,47. Further investigations are warranted to assess restitution effects in FD cardiac tissue.
Our ECG analysis demonstrated increased PAC occurrence in FD patients likely arising from ectopic activity in the pulmonary veins. Subsequently, in our FD bi-atrial model informed by our observed clinical and cellular findings, AF was induced by two ectopic beats originating from the left superior pulmonary vein. These findings confirm the pro-arrhythmic nature of observed cellular changes induced by GLA p. N215S FD. The in-vivo, in-vitro and modelling experiments used in this model for FD may be applicable to other atrial cardiomyopathies with the potential of developing atrial arrhythmia-specific therapy
Limitations
This study involves one of the largest datasets of patients with FD. However, when comparing to studies on related cardiac conditions, the cohort size is comparatively small since FD is a rare disease. Whilst data is available on LA volumes in FD patients, atrial contraction analysis is currently not assessed as part of the dataset. Future work will incorporate LA strain assessment to assess atrial contraction to compare with the iPSC-CM contractility data.
iPSC-CMs are a valuable, widely used, human cellular model for cardiac diseases, but we acknowledge their inherent immaturity, which is recognised within the scientific community48–50. While the diastolic membrane potential of the cells in our model is more positive than of human adult atrial cardiomyocytes, it is in keeping with other studies.27–29 and the cells reflect well key molecular aspects of FD, namely α-Gal A deficiency and Gb3 accumulation. Future studies, e.g. co-culturing iPSC-CM in 3D, together with other cardiac cells, have the potential to mimic cardiac electrical phenotypes of FD even better18,51.
Moreover, with the cellular data, there can be several variables in the differentiation process when culturing atrial cardiomyocytes which affects the quality and subsequent molecular and functional properties of the cells. To mitigate this, we conducted experiments on multiple batches of high-quality cells in a coordinated beating monolayer, suitable for analysis. With the APDs, whilst changes were not observed, this has less relevance when comparing with the 12-lead ECGs of FD patients as any change reflects atrial repolarisation which is difficult to quantify on ECG. Regarding the calcium handling data, calcium conduction velocity measurements derived from calcium transient data are limited in assessing action potential propagation speed due to uncertainties in the temporal relationship between electrical depolarisation and the rise in cytosolic calcium. For these reasons, future experiments should be performed in this model using transmembrane voltage dyes. Furthermore, we were not able to pace our cell monolayers, and therefore future work will address arrhythmia inducibility in our cell model using programmed simulation such as S1-S2 protocols, as spontaneous arrhythmias were not observed
Regarding the human modelling and simulation component, similar results and the same conclusions were obtained at single-cell, using different ionic models, supporting the primary outcome of this work. Therefore, only one cellular model was used for whole-organ simulations. However, future work should attempt to include different and more sophisticated Ca2+ handling systems in the 3D atrial models. Similarly, the population of action potential models was calibrated with human data from patients in sinus rhythm and AF. While the biomarkers of both populations greatly overlapped, it would be interesting to further analyse the results obtained if only sinus rhythm biomarkers are used for calibration.
Conclusion
In summary, in this study of adults with FD and atrial iPSC-CMs with the GLA p. N215S variant for FD, we confirm early P-wave changes in “phenotype negative” patients. In an atrial myocyte model of FD, we found novel cellular changes including alterations in contraction, intracellular calcium handling, and atrial electrophysiology which may account for the early electrical changes observed on 12-lead ECGs of FD patients. In an in-silico atrial model, the cellular changes produced a similar P-wave morphology to that seen in clinical early-stage FD cardiomyopathy and predicted an increase in AF vulnerability. This study provides new insights into the underlying mechanisms contributing to the arrhythmic substrate in FD and validates the use of iPSC models to understand arrhythmia mechanisms. These identified changes may act as targets for cardioprotective therapy to reduce the burden of arrhythmia and stroke in FD.
Supplementary Material
What does this study add?
Signal-averaged P-wave analysis in adults with FD of varying stages of cardiomyopathy demonstrate P-wave duration and PQ shortening in adults with FD, but no overt cardiovascular phenotype. P-wave duration and PQ interval prolong as cardiomyopathy progresses, likely due to atrial structural remodelling including dilatation and impaired atrial strain.
We have identified several mechanisms for atrial arrhythmogenesis in FD. We demonstrate alterations in contraction, calcium handling and excitability in gene-edited GLA p. N215S atrial IPSC-cardiomyocytes which under-express □-GAL A and accumulate Gb3, compared to WT. We confirm arrhythmia inducibility when imputing our cellular findings in an in-silico atrial model.
The atrial iPSC-CM data supports the early clinical ECG findings in FD, suggesting electrical instability and remodelling in atrial cardiomyocytes begins early and progresses insidiously, forming a substrate for atrial arrythmia. The combined use of in-vivo and in-vitro data with an in-silico model may be applicable to other atrial cardiomyopathies.
What is known?
Arrhythmia and stroke are responsible for large burden of cardiovascular mortality in FD. Whilst predisposing factors and pathophysiological mechanisms for ventricular arrhythmia are established, there is less evidence for mechanisms of atrial myopathy and arrhythmia. Yet, AF prevalence is high and likely a significant contributor to stroke burden.
The earliest detectable cardiac manifestations on ECG are shortening or prolongation of the PQ interval. The mechanisms of PQ shortening are poorly understood.
GLA p. N215S is a non-classical cardiac variant of FD, previously thought to be “late onset”, with manifestations predominantly presenting late in adulthood.
Acknowledgments
The authors would like to acknowledge the support and work of the Centre for Rare Diseases and Department of Echocardiography at the Queen Elizabeth Hospital, Birmingham, United Kingdom.
Sources of Funding
This study has been supported by an Accelerator Award from the British Heart Foundation (AA/18/2/34218) to AR, the Metchley Park Medical Society Research Consumables Grant (C111 10005 60492) to AR, a grant from the Society of Mucopolysaccharide Diseases (MPS) (DD-ROY2024) to AR, the Takeda RaILRoAD Grant (IIR-GBR-001662) to AR/TG/RPS, the Sanofi RaILRoAD Grant (GZ-2017-11698) to AR/TG/RPS, the Medical Research Council (MR/V009540/1) to KG, National Centre for the 3Rs/British Heart Foundation (BHF) studentship (NC/T001747/1) to MC/KG, Wellcome Trust (221650/Z/20/Z) to COS, BHF (FS/PhD/20/29093) to APH, (PG/17/55/33087, RG/17/15/33106, FS/19/12/342040, FS/PhD/22/29309) to DP and (IA/F/23/275037) to KG, a Wellcome Trust Fellowship in Basic Biomedical Sciences (214290/Z/18/Z) to BR, the EPSRC project CompBioMed X (EP/X019446/1) to BR, the European Union’s Horizon 2020 research and by the EPSRC Impact Acceleration Account Award (UKRI Grant Reference—EP/X525777/1) to AD.
Footnotes
Disclosures: None
References
- 1.Desnick RJ, Blieden LC, Sharp HL, Hofschire PJ, Moller JH. Cardiac valvular anomalies in Fabry disease. Clinical, morphologic, and biochemical studies. Circulation. 1976 Nov;54(5):818–25. doi: 10.1161/01.cir.54.5.818. [DOI] [PubMed] [Google Scholar]
- 2.Desnick RJ, Wasserstein MP. Fabry disease: clinical features and recent advances in enzyme replacement therapy. Adv Nephrol Necker Hosp. 2001;31:317–39. [PubMed] [Google Scholar]
- 3.Mehta A, Beck M, Eyskens F, et al. Fabry disease: a review of current management strategies. QJM. 2010 Sep;103(9):641–59. doi: 10.1093/qjmed/hcq117. [DOI] [PubMed] [Google Scholar]
- 4.Biegstraaten M, Arngrímsson R, Barbey F, et al. Recommendations for initiation and cessation of enzyme replacement therapy in patients with Fabry disease: the European Fabry Working Group consensus document. Orphanet J Rare Dis. 2015 Mar 27;10:36. doi: 10.1186/s13023-015-0253-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vijapurapu R, Roy A, Demetriades P, et al. Systematic review of the incidence and clinical risk predictors of atrial fibrillation and permanent pacemaker implantation for bradycardia in Fabry disease. Open Heart. 2023 Jul;10(2) doi: 10.1136/openhrt-2023-002316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pichette M, Serri K, Pagé M, Bichet DG, Poulin F. Impaired Left Atrial Function in Fabry Disease: A Longitudinal Speckle-Tracking Echocardiography Study. J Am Soc Echocardiogr. 2017;30(2):170–179.:e2. doi: 10.1016/j.echo.2016.10.014. [DOI] [PubMed] [Google Scholar]
- 7.Kotecha D, Piccini JP. Atrial fibrillation in heart failure: what should we do? Eur Heart J. 2015 Dec 07;36(46):3250–7. doi: 10.1093/eurheartj/ehv513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sims K, Politei J, Banikazemi M, Lee P. Stroke in Fabry disease frequently occurs before diagnosis and in the absence of other clinical events: natural history data from the Fabry Registry. Stroke. 2009 Mar;40(3):788–94. doi: 10.1161/STROKEAHA.108.526293. [DOI] [PubMed] [Google Scholar]
- 9.Linhart A, Germain DP, Olivotto I, et al. An expert consensus document on the management of cardiovascular manifestations of Fabry disease. Eur J Heart Fail. 2020;22(7):1076–1096. doi: 10.1002/ejhf.1960. [DOI] [PubMed] [Google Scholar]
- 10.Namdar M, Steffel J, Vidovic M, et al. Electrocardiographic changes in early recognition of Fabry disease. Heart. 2011 Mar;97(6):485–90. doi: 10.1136/hrt.2010.211789. [DOI] [PubMed] [Google Scholar]
- 11.Namdar M, Kampmann C, Steffel J, et al. PQ interval in patients with Fabry disease. Am J Cardiol. 2010 Mar 01;105(5):753–6. doi: 10.1016/j.amjcard.2009.10.056. [DOI] [PubMed] [Google Scholar]
- 12.Namdar M. Electrocardiographic Changes and Arrhythmia in Fabry Disease. Front Cardiovasc Med. 2016;3:7. doi: 10.3389/fcvm.2016.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pieroni M, Moon JC, Arbustini E, et al. Cardiac Involvement in Fabry Disease: JACC Review Topic of the Week. J Am Coll Cardiol. 2021 Feb 23;77(7):922–936. doi: 10.1016/j.jacc.2020.12.024. [DOI] [PubMed] [Google Scholar]
- 14.Sommerfeld LC, Holmes AP, Yu TY, et al. Reduced plakoglobin increases the risk of sodium current defects and atrial conduction abnormalities in response to androgenic anabolic steroid abuse. J Physiol. 2024 Feb 12; doi: 10.1113/JP284597. [DOI] [PubMed] [Google Scholar]
- 15.Robinson S, Rana B, Oxborough D, et al. A practical guideline for performing a comprehensive transthoracic echocardiogram in adults: the British Society of Echocardiography minimum dataset. Echo Res Pract. 2020 Dec;7(4):G59–G93. doi: 10.1530/ERP-20-0026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Germain DP, Brand E, Burlina A, et al. Phenotypic characteristics of the p.Asn215Ser (p.N215S) GLA mutation in male and female patients with Fabry disease: A multicenter Fabry Registry study. Mol Genet Genomic Med. 2018 Apr; doi: 10.1002/mgg3.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gilchrist M, Casanova F, Tyrrell JS, et al. Prevalence of Fabry disease-causing variants in the UK Biobank. J Med Genet. 2023 Apr;60(4):391–396. doi: 10.1136/jmg-2022-108523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cumberland MJ, Euchner J, Azad AJ, et al. Generation of a human iPSC-derived cardiomyocyte/fibroblast engineered heart tissue model. F1000Res. 2023;12:1224. doi: 10.12688/f1000research.139482.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Workman AJ, Kane KA, Rankin AC. The contribution of ionic currents to changes in refractoriness of human atrial myocytes associated with chronic atrial fibrillation. Cardiovasc Res. 2001 Nov;52(2):226–35. doi: 10.1016/s0008-6363(01)00380-7. [DOI] [PubMed] [Google Scholar]
- 20.Holmes AP, Saxena P, Kabir SN, et al. Atrial resting membrane potential confers sodium current sensitivity to propafenone, flecainide and dronedarone. Heart Rhythm. 2021 Jul;18(7):1212–1220. doi: 10.1016/j.hrthm.2021.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sala L, van Meer BJ, Tertoolen LGJ, et al. MUSCLEMOTION: A Versatile Open Software Tool to Quantify Cardiomyocyte and Cardiac Muscle Contraction In Vitro and In Vivo. Circ Res. 2018 Feb 02;122(3):e5–e16. doi: 10.1161/CIRCRESAHA.117.312067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.O’Shea C, Holmes AP, Yu TY, et al. ElectroMap: High-throughput open-source software for analysis and mapping of cardiac electrophysiology. Sci Rep. 2019;9(1):1389. doi: 10.1038/s41598-018-38263-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bayly PV, KenKnight BH, Rogers JM, Hillsley RE, Ideker RE, Smith WM. Estimation of conduction velocity vector fields from epicardial mapping data. IEEE Trans Biomed Eng. 1998 May;45(5):563–71. doi: 10.1109/10.668746. [DOI] [PubMed] [Google Scholar]
- 24.Dasí A, Nagel C, Pope MTB, et al. In Silico TRials guide optimal stratification of ATrIal FIbrillation patients to Catheter Ablation and pharmacological medicaTION: the i-STRATIFICATION study. Europace. 2024 Jun 03;26(6) doi: 10.1093/europace/euae150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dasí A, Roy A, Sachetto R, Camps J, Bueno-Orovio A, Rodriguez B. drug trials for precision medicine in atrial fibrillation: From ionic mechanisms to electrocardiogram-based predictions in structurally-healthy human atria. Front Physiol. 2022;13:966046. doi: 10.3389/fphys.2022.966046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sachetto Oliveira R, Martins Rocha B, Burgarelli D, Meira W, Constantinides C, Weber Dos Santos R. Performance evaluation of GPU parallelization, space-time adaptive algorithms, and their combination for simulating cardiac electrophysiology. Int J Numer Method Biomed Eng. 2018 Feb;34(2) doi: 10.1002/cnm.2913. [DOI] [PubMed] [Google Scholar]
- 27.Vaidyanathan R, Markandeya YS, Kamp TJ, Makielski JC, January CT, Eckhardt LL. IK1-enhanced human-induced pluripotent stem cell-derived cardiomyocytes: an improved cardiomyocyte model to investigate inherited arrhythmia syndromes. Am J Physiol Heart Circ Physiol. 2016 Jun 01;310(11):H1611–21. doi: 10.1152/ajpheart.00481.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Reyat JS, Sommerfeld LC, O’Reilly M, et al. PITX2 deficiency leads to atrial mitochondrial dysfunction. Cardiovasc Res. 2024 Aug 12; doi: 10.1093/cvr/cvae169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Thorpe J, Perry MD, Contreras O, et al. Development of a robust induced pluripotent stem cell atrial cardiomyocyte differentiation protocol to model atrial arrhythmia. Stem Cell Res Ther. 2023 Jul 27;14(1):183. doi: 10.1186/s13287-023-03405-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Moretti A, Bellin M, Welling A, et al. Patient-specific induced pluripotent stem-cell models for long-QT syndrome. N Engl J Med. 2010 Oct 07;363(15):1397–409. doi: 10.1056/NEJMoa0908679. [DOI] [PubMed] [Google Scholar]
- 31.Benzoni P, Campostrini G, Landi S, et al. Human iPSC modelling of a familial form of atrial fibrillation reveals a gain of function of If and ICaL in patient-derived cardiomyocytes. Cardiovasc Res. 2020 May 01;116(6):1147–1160. doi: 10.1093/cvr/cvz217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Priori SG, Napolitano C, Di Pasquale E, Condorelli G. Induced pluripotent stem cell-derived cardiomyocytes in studies of inherited arrhythmias. J Clin Invest. 2013 Jan;123(1):84–91. doi: 10.1172/JCI62838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simons E, Loeys B, Alaerts M. iPSC-Derived Cardiomyocytes in Inherited Cardiac Arrhythmias: Pathomechanistic Discovery and Drug Development. Biomedicines. 2023 Jan 25;11(2) doi: 10.3390/biomedicines11020334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jastrzebski M, Bacior B, Dimitrow PP, Kawecka-Jaszcz K. Electrophysiological study in a patient with Fabry disease and a short PQ interval. Europace. 2006 Dec;8(12):1045–7. doi: 10.1093/europace/eul121. [DOI] [PubMed] [Google Scholar]
- 35.Aryana A, Fifer MA, Ruskin JN, Mela T. Short PR interval in the absence of preexcitation: a characteristic finding in a patient with Fabry disease. Pacing Clin Electrophysiol. 2008 Jun;31(6):782–3. doi: 10.1111/j.1540-8159.2008.01088.x. [DOI] [PubMed] [Google Scholar]
- 36.Frustaci A, Morgante E, Russo MA, et al. Pathology and function of conduction tissue in Fabry disease cardiomyopathy. Circ Arrhythm Electrophysiol. 2015 Aug;8(4):799–805. doi: 10.1161/CIRCEP.114.002569. [DOI] [PubMed] [Google Scholar]
- 37.Ali ZS, Bhuiyan A, Vyas P, et al. PR prolongation as a predictor of atrial fibrillation onset: A state-of-the-art review. Curr Probl Cardiol. 2024 Apr;49(4):102469. doi: 10.1016/j.cpcardiol.2024.102469. [DOI] [PubMed] [Google Scholar]
- 38.Birket MJ, Raibaud S, Lettieri M, et al. A Human Stem Cell Model of Fabry Disease Implicates LIMP-2 Accumulation in Cardiomyocyte Pathology. Stem Cell Reports. 2019;13(2):380–393. doi: 10.1016/j.stemcr.2019.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Parham WA, Mehdirad AA, Biermann KM, Fredman CS. Hyperkalemia revisited. Tex Heart Inst J. 2006;33(1):40–7. [PMC free article] [PubMed] [Google Scholar]
- 40.Berecki G, Wilders R, de Jonge B, van Ginneken AC, Verkerk AO. Re-evaluation of the action potential upstroke velocity as a measure of the Na+ current in cardiac myocytes at physiological conditions. PLoS One. 2010 Dec 31;5(12):e15772. doi: 10.1371/journal.pone.0015772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Baines O, Sha R, Kalla M, et al. Optical mapping and optogenetics in cardiac electrophysiology research and therapy: a state-of-the-art review. Europace. 2024 Feb 01;26(2) doi: 10.1093/europace/euae017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mechmann S, Pott L. Identification of Na-Ca exchange current in single cardiac myocytes. Nature. 1986;319(6054):597–9. doi: 10.1038/319597a0. 1986 Feb 13-19. [DOI] [PubMed] [Google Scholar]
- 43.Asakura K, Cha CY, Yamaoka H, et al. EAD and DAD mechanisms analyzed by developing a new human ventricular cell model. Prog Biophys Mol Biol. 2014 Sep;116(1):11–24. doi: 10.1016/j.pbiomolbio.2014.08.008. [DOI] [PubMed] [Google Scholar]
- 44.Kistamás K, Veress R, Horváth B, Bányász T, Nánási PP, Eisner DA. Calcium Handling Defects and Cardiac Arrhythmia Syndromes. Front Pharmacol. 2020;11:72. doi: 10.3389/fphar.2020.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shattock MJ, Park KC, Yang HY, et al. Restitution slope is principally determined by steady-state action potential duration. Cardiovasc Res. 2017 Jun 01;113(7):817–828. doi: 10.1093/cvr/cvx063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Coronel R, Wilms-Schopman FJ, Opthof T, Janse MJ. Dispersion of repolarization and arrhythmogenesis. Heart Rhythm. 2009 Apr;6(4):537–43. doi: 10.1016/j.hrthm.2009.01.013. [DOI] [PubMed] [Google Scholar]
- 47.Nicolson WB, McCann GP, Smith MI, et al. Prospective evaluation of two novel ECG-based restitution biomarkers for prediction of sudden cardiac death risk in ischaemic cardiomyopathy. Heart. 2014 Dec;100(23):1878–85. doi: 10.1136/heartjnl-2014-305672. [DOI] [PubMed] [Google Scholar]
- 48.Vučković S, Dinani R, Nollet EE, et al. Characterization of cardiac metabolism in iPSC-derived cardiomyocytes: lessons from maturation and disease modeling. Stem Cell Res Ther. 2022 Jul 23;13(1):332. doi: 10.1186/s13287-022-03021-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ormrod B, Ehler E. Induced pluripotent stem cell-derived cardiomyocytes-more show than substance? Biophys Rev. 2023 Dec;15(6):1941–1950. doi: 10.1007/s12551-023-01099-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Joshi J, Albers C, Smole N, Guo S, Smith SA. Human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs) for modeling cardiac arrhythmias: strengths, challenges and potential solutions. Front Physiol. 2024;15:1475152. doi: 10.3389/fphys.2024.1475152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ibarra DE, Jewett ME, Jarrell DK, Pinales A, VeDepo MC, Jacot JG. Bioreactor Design for Culturing Vascularized Engineered Tissue in Flow Conditions. Tissue Eng Part A. 2024 Jun;30(11-12):304–313. doi: 10.1089/ten.tea.2023.0201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gehmlich K, Dodd MS, Allwood JW, et al. Changes in the cardiac metabolome caused by perhexiline treatment in a mouse model of hypertrophic cardiomyopathy. Mol Biosyst. 2015 Feb;11(2):564–73. doi: 10.1039/c4mb00594e. [DOI] [PubMed] [Google Scholar]
- 53.Courtemanche M, Ramirez RJ, Nattel S. Ionic mechanisms underlying human atrial action potential properties: insights from a mathematical model. Am J Physiol. 1998 Jul;275(1):H301–21. doi: 10.1152/ajpheart.1998.275.1.H301. [DOI] [PubMed] [Google Scholar]
- 54.Herrera NT, Zhang X, Ni H, et al. Dual effects of the small-conductance Ca. Am J Physiol Heart Circ Physiol. 2023 Oct 01;325(4):H896–H908. doi: 10.1152/ajpheart.00362.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Muszkiewicz A, Britton OJ, Gemmell P, et al. Variability in cardiac electrophysiology: Using experimentally-calibrated populations of models to move beyond the single virtual physiological human paradigm. Prog Biophys Mol Biol. 2016 Jan;120(1-3):115–27. doi: 10.1016/j.pbiomolbio.2015.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sánchez C, Bueno-Orovio A, Wettwer E, et al. Inter-subject variability in human atrial action potential in sinus rhythm versus chronic atrial fibrillation. PLoS One. 2014;9(8):e105897. doi: 10.1371/journal.pone.0105897. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.