To the editor
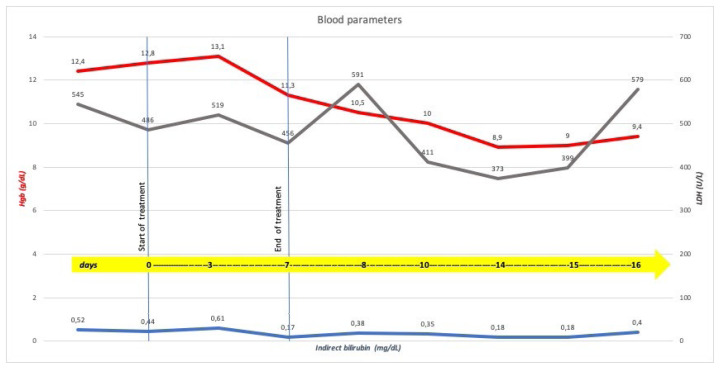
In March 2020, “Coronavirus Disease 2019” (COVID-19) infection outbreak has been declared a pandemic by the World Health Organization, and until now, there are no proven drugs for the treatment of “Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)” infection; therefore affected symptomatic patients are treated with drugs already used for other infections1 such as hydroxychloroquine (HCQ) or chloroquine, administered alone or in combination with other medications. Hence the growing attention about G6PD (Glucose-6-Phosphate Dehydrogenase)-deficiency since that chloroquine or HCQ could trigger severe hemolysis in subjects with this inherited abnormality.2 From a literature review, four case reports have been recently published about the use of HCQ in patients with COVID-19 infection and G6PD-deficiency, developing acute hemolytic anemia (AHA);3–6 we are reporting the case of a patient with SARS-CoV-2 infection and G6PD-deficiency treated with HCQ without laboratory evidence of hemolysis. The patient, a 61-year-old Caucasian man, was admitted to the Intensive Care Unit of “A.O.U. Consorziale, Policlinico di Bari-Italy” for severe acute respiratory failure, requiring invasive mechanical ventilation, and fever. Chest X-ray showed multiple bilateral lung opacities. The suspected COVID-19 infection was confirmed by a real-time-PCR assay on a nasopharyngeal swab. According to the institutional protocol, treatment with HCQ (200 mg, thrice a day), darunavir (800 mg, once a day), and azithromycin (500 mg, once a day) was carried on for seven days. The patient’s medical history included hypertension controlled with nebivolol and aspirin, along with some relatives having a suspected condition of “favism,” but there was no personal history of drug-induced hemolysis. Since recent indications7 for G6PD testing include a family history of G6PD-deficiency, enzymatic activity was analyzed; a diagnosis of G6PD-deficiency was made with a dosage of 5.6 UI/g Hb (normal range: 6.9–9.0; reticulocytes: 1.63%; residual enzyme activity: 79%) and in consideration of ongoing treatment, close monitoring of complete blood count and hemolysis parameters was carried on, without stopping treatment. A decreased value of Hgb was reported, without laboratory evidence of hemolysis during and after treatment (Figure 1); no transfusion support with red blood cells was needed. Molecular analysis was not performed, but considering that the incidence of class III (WHO classification) G6PD deficiency in our region is high, according to our previous paper,8 the patient may be a carrier of the Seattle variant, usually associated with a low risk of AHA. G6PD deficiency is a global health problem representing a paradigmatic example of a highly specific interaction between an inherited abnormality and exogenous agents that trigger hemolysis;7 it frequently occurs in Africa, Asia, and the Mediterranean region9 and remains the most common human enzymatic disorder of red blood cells worldwide. G6PD, whose gene maps to the long arm of the X chromosome (band Xq28), is expressed in all tissue cells where it catalyzes the first step in the pentose phosphate pathway. In the red blood cell, this is the unique pathway for NADPH production, which is required to maintain glutathione in the reduced state. Failure of this process impairs the red cell’s ability to deal with oxidative stress, which may lead to hemolytic episodes and anemia that can be severe and, in some cases, fatal. Often G6PD deficiency is an asymptomatic condition that remains undetected until subjects are exposed to an exogenous hemolytic trigger such as fava beans ingestion, taking drugs with intracellular oxidizing action, exposure to substances with intracellular oxidizing action, bacterial and viral infections. We identify some interesting issues concerning the management of the case report described. First, “G6PD deficiency” does not necessarily mean “hemolysis” depending on the severity of the deficiency and residual enzymatic activity. Second, the enzymatic activity deficiency is not a predictive parameter of the severity of the clinical condition, as we reported in 2001.8 Although it is not yet clearly understood whether genetic or extragenetic, other mechanisms must exist that offer protection from the oxidative stresses that play a role in the clinical expression of G6PD deficiency. Third, since G6PD-deficiency could be a susceptibility factor to SARS-CoV2 infection10 and that this inherited abnormality is not so rare, it should be sought in case of unexplained hemolysis or before administering drugs, a potential trigger of hemolysis. According to literature data, we recommend caution with using HCQ in all subjects with G6PD-deficiency, including those with COVID-19 infection; at the same time, we believe that the use of this drug in patients with G6PD deficiency should be investigated with further studies, elucidating the role of residual enzyme activity and genotype, factors that may influence AHA risk. Moreover, although G6PD deficiency remains a contraindication to HCQ use, our report suggests that several variables may influence AHA risk, and these aspects should be investigated in further studies to discriminate patients who may have some benefits from this specific drug.
Figure 1.
Timeline of blood parameters and treatment
Acknowledgments
The authors thank Mark Mirizio for the language revision of the manuscript.
Footnotes
Competing interests: The authors declare no conflict of Interest.
References
- 1.Costanzo M, De Giglio MAR, Roviello GN. SARS-CoV-2: Recent Reports on Antiviral Therapies Based on Lopinavir/Ritonavir, Darunavir/Umifenovir, Hydroxychloroquine, Remdesivir, Favipiravir and other Drugs for the Treatment of the New Coronavirus. Curr Med Chem. 2020;27(27):4536–4541. doi: 10.2174/0929867327666200416131117. [DOI] [PubMed] [Google Scholar]
- 2.ISS COVID-19 Rare Diseases Working Group. Interim guidance for the appropriate support of people with enzymopenia G6PD (favism) in the current SARS-CoV-2 emergency scenario. Version April 14, 2020. Roma: Istituto Superiore di Sanità; 2020. (Rapporto ISS COVID-19, 14/2020- English version) [Google Scholar]
- 3.Beauverd Y, Adam Y, Assouline B, Samii K. COVID-19 infection and treatment with hydroxychloroquine cause severe haemolysis crisis in a patient with glucose-6-phosphate dehydrogenase deficiency [published online ahead of print, 2020 Apr 23] Eur J Haematol. 2020 doi: 10.1111/ejh.13432. doi: 10.1111/ejh.13432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Maillart E, Leemans S, Van Noten H, et al. A case report of serious haemolysis in a glucose-6-phosphate dehydrogenase-deficient COVID-19 patient receiving hydroxychloroquine. Infect Dis (Lond) 2020;52(9):659–661. doi: 10.1080/23744235.2020.1774644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sasi S, Yassin MA, Nair AP, Al Maslamani MS. A Case of COVID-19 in a Patient with Asymptomatic Hemoglobin D Thalassemia and Glucose-6-Phosphate Dehydrogenase Deficiency. Am J Case Rep. 2020;21:e925788. doi: 10.12659/AJCR.925788. Published 2020 Jul 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De Franceschi L, Costa E, Dima F, Morandi M, Olivieri O. Acute hemolysis by hydroxycloroquine was observed in G6PD-deficient patient with severe COVD-19 related lung injury. Eur J Intern Med. 2020;77:136–137. doi: 10.1016/j.ejim.2020.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Roper D, Layton M, Rees D, et al. Laboratory diagnosis of G6PD deficiency. A British Society for Haematology Guideline. Br J Haematol. 2020;189(1):24–38. doi: 10.1111/bjh.16366. [DOI] [PubMed] [Google Scholar]
- 8.Pietrapertosa A, Palma A, Campanale D, Delios G, Vitucci A, Tannoia N. Genotype and phenotype correlation in glucose-6-phosphate dehydrogenase deficiency. Haematologica. 2001;86(1):30–35. [PubMed] [Google Scholar]
- 9.Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet. 2008;371(9606):64–74. doi: 10.1016/S0140-6736(08)60073-2. [DOI] [PubMed] [Google Scholar]
- 10.Kassi EN, Papavassiliou KA, Papavassiliou AG. G6PD and chloroquine: Selecting the treatment against SARS-CoV-2? J Cell Mol Med. 2020;24(9):4913–4914. doi: 10.1111/jcmm.15312. [DOI] [PMC free article] [PubMed] [Google Scholar]