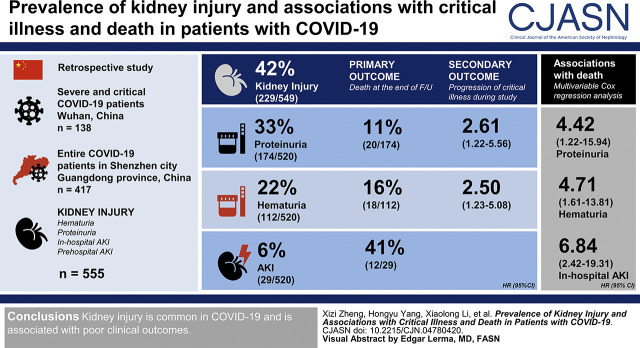
Visual Abstract
Keywords: COVID-19, acute kidney injury, proteinuria, hematuria, critical illness
Abstract
Background and objectives
Coronavirus disease 2019 is spreading rapidly across the world. This study aimed to assess the characteristics of kidney injury and its association with disease progression and death of patients with coronavirus disease 2019.
Design, setting, participants, & measurements
This is a retrospective study. Two representative cohorts were included. Cohort 1 involved severe and critical patients with coronavirus disease 2019 from Wuhan, China. Cohort 2 was all patients with coronavirus disease 2019 in Shenzhen city (Guangdong province, China). Any kidney injury was defined as the presence of any of the following: hematuria, proteinuria, in-hospital AKI, or prehospital AKI. AKI was defined according to the Kidney Disease Improving Global Outcomes (KDIGO) creatinine criteria. The primary outcome was death at the end of follow-up. The secondary outcome was progression to critical illness during the study period.
Results
A total of 555 patients were enrolled; 42% of the cases (229 of 549) were detected with any kidney injury, 33% of the cases (174 of 520) were detected with proteinuria, 22% of the cases (112 of 520) were detected with hematuria, and 6% of the cases (29 of 520) were detected with AKI. Of the 29 patients with AKI, 21 cases were recognized as in-hospital AKI, and eight were recognized as prehospital AKI. Altogether, 27 (5%) patients died at the end of follow-up. The death rate was 11% (20 of 174) in patients with proteinuria, 16% (18 of 112) in patients with hematuria, and 41% (12 of 29) in the AKI settings. Multivariable Cox regression analysis showed that proteinuria (hazard ratio, 4.42; 95% confidence interval, 1.22 to 15.94), hematuria (hazard ratio, 4.71; 95% confidence interval, 1.61 to 13.81), and in-hospital AKI (hazard ratio, 6.84; 95% confidence interval, 2.42 to 19.31) were associated with death. Among the 520 patients with noncritical illness at admission, proteinuria (hazard ratio, 2.61; 95% confidence interval, 1.22 to 5.56) and hematuria (hazard ratio, 2.50; 95% confidence interval, 1.23 to 5.08) were found to be associated with progression to critical illness during the study period.
Conclusions
Kidney injury is common in coronavirus disease 2019, and it is associated with poor clinical outcomes.
Podcast
This article contains a podcast at https://www.asn-online.org/media/podcast/CJASN/2020_09_18_CJN04780420.mp3
Introduction
Since December 2019, an ongoing outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection emerged in Wuhan, China (1,2). The disease (coronavirus disease 2019 [COVID-19]) spread rapidly to other parts of the world and has resulted in >1.2 million cases and 67,000 deaths as of April 7, 2020 (3).
Although SARS-CoV-2 infection mainly presents as acute respiratory symptoms, with some patients rapidly developing acute respiratory distress syndrome (4–7), extrapulmonary involvement including the gastrointestinal tract, liver, cardiovascular system, and kidney has been reported (8–11). The incidence of AKI reported in early studies of SARS-CoV-2 infection varied substantially from 0.5% to 9% (9,12–14). A recent study showed that AKI could affect 29% of critically ill patients (4). It is thought that the occurrence of AKI is part of the multiorgan dysfunction that developed in critically ill patients infected with SARS-CoV-2, and the variety of AKI incidence reflects the wide clinical spectrum of COVID-19 in terms of disease severity and outcome. Until now, there is limited information regarding the whole picture of kidney involvement in this disease, and it is unknown whether kidney injury at early stage would have any association with disease progression.
In this study, we carried out a retrospective cohort study on the kidney involvement of COVID-19 in two large populations: one is all patients with confirmed COVID-19 in Shenzhen, a large city with a total population of 13 million in South China, and the other is patients with severe COVID-19 from Wuhan city needing medical care in the hospital. We describe clinical characteristics of kidney injury and investigate its association with the disease progression and outcomes in the studied population.
Materials and Methods
Study Design and Participants
This was a retrospective cohort study that included two populations from two cities. Cohort 1 involved all consecutive patients with severe and critical COVID-19 from Peking University Medical Team (deployed in Wuhan, China) from February 8 to 28, 2020. Cohort 2 was all patients with COVID-19 who were diagnosed in Shenzhen city (Guangdong province, China) and transferred to Shenzhen Third People’s Hospital from January 8 to February 25, 2020 (Shenzhen cohort). All patients in this study were diagnosed with COVID-19 according to the World Health Organization interim guidance (15). The study was approved by the ethics committees of the Peking University First Hospital and the Shenzhen Third People’s Hospital.
Procedures
The epidemiologic, clinical, laboratory, management, and outcome data were extracted from electronic medical records and reviewed by a trained team of physicians. The clinical data and outcomes were monitored up to March 1, 2020.
Cases were confirmed by a positive result of the nucleic acid of 2019-nCoV by RT-PCR of nasopharyngeal swabs. Baseline disease severity was classified, mainly on the basis of respiratory indicators, into four categories: mild, moderate, severe, and critical within the first 24 hours of admission according to Guidance for Corona Virus Disease 2019: Prevention, Control, Diagnosis and Management issued by the National Health Committee of the People’s Republic of China (16) (Supplemental Table 1). A severe case was defined as having one of the following: (1) respiratory rate >30/min, (2) oxygen saturation ≤93%, or (3) PaO2-FiO2 ratio ≤300 mm Hg. A critical case was defined when meeting any of the following: shock (defined as with systolic BP below 90 mm Hg for >1 hour and/or requiring the administration of vasopressor); respiratory failure requiring mechanical ventilation; or extrapulmonary organ failure requiring intensive care (defined as with hemodynamic instability, including the presence of systolic BP <90 mm Hg, chest pain, dyspnea, syncope, or altered mental status).
Any kidney injury was defined as the presence of any of the following: hematuria, proteinuria, in-hospital AKI, or prehospital AKI. Urine samples were tested by an automatic urine biochemical analyzer at hospital admission, and none of the enrolled patients were on urethral catheterization at the time. Proteinuria or hematuria was defined when the semiquantitative dipstick test read “1+” or more. We identified AKI on the basis of change in serum creatine (sCr) levels during hospitalization. The criteria included the 2012 Kidney Disease Improving Global Outcomes (KDIGO) AKI sCr definition: an increase in sCr level by 0.3 mg/dl within 48 hours or by 50% within 7 days (17). For those who had recovering AKI at admission, we expanded the criteria as follows: a decrease in sCr level of 50% during the hospital stay using the lowest sCr value during hospitalization as the comparator. We then identified prehospital AKI when the patient met any of the following criteria: (1) an increased sCr level at admission and a trend of decreasing sCr levels during the hospital stay or (2) an increased sCr level at admission and an sCr level that continued to increase or remained at a high level during the hospital stay, with preadmission sCr values establishing the existence of AKI.
Baseline sCr was used to determine the peak stage of AKI in patients who were identified as having in-hospital AKI by the AKI definition criteria. We defined baseline sCr as the lowest sCr value that was available within 3 months prior to admission and throughout the hospital stay (from admission to the end of follow-up). Among the 21 patients with in-hospital AKI, five patients had sCr value available within 3 months prior to admission, and the other 16 patients had their baseline sCr defined as the lowest sCr during the hospitalization. The date of in-hospital AKI onset was defined as the earliest day of an sCr change meeting the KDIGO criteria, and the maximum AKI stage was defined using the KDIGO creatinine criteria.
The primary outcome was death at the end of follow-up. The secondary outcome was progression to critical illness during the study period. Patients with critical illness at admission were included in the outcome analysis for “death” but not included in the outcome analysis for “progression to critical illness.” Critical illness was defined using the same classification as critical case.
Statistical Analyses
Categorical variables were described as frequency and percentages, and continuous variables were described as means with SD or median with 25th and 75th percentiles as appropriate. Continuous variables were compared by one-way ANOVA test if data were with normal distribution; otherwise, they were compared by the Mann–Whitney test. Categorical variables were compared by the chi-squared test or the Fisher test as appropriate.
Cumulative rates of outcome events were described by the Kaplan–Meier method. Follow-up time for each patient was started from the date of admission and ended at the date of occurrence of the outcome or the prespecified administrative end date (March 1, 2020), as appropriate. The associations between kidney disease indicators and study outcomes were examined using Cox regression analysis. The covariates were limited to age; sex; disease severity; the values of lactate dehydrogenase, C-reactive protein, troponin, d-dimer, and lymphocyte count at hospital admission; and comorbidities, such as hypertension, diabetes, coronary heart disease, chronic obstructive pulmonary disease or asthma, CKD, and malignant tumor history, on the basis of the published studies of the known factors associated with poor outcome of patients with COVID-19 (12,18–21). All of the covariates were evaluated/measured at the time point of admission. When analyzing the association between AKI and disease progression, in-hospital AKI was set as a time-dependent covariate. Cox proportional hazards assumptions were tested with the Schoenfeld residuals. No violations of the Cox proportional hazards assumptions were detected. In multivariable models, we assessed interactions of kidney disease indicators with other covariates. None were eligible for retention (all P values for interaction were 0.05). Patients with missing values in proteinuria, hematuria, and AKI were excluded when assessing the association of those indicators with study outcomes. Other missing values were filled with mean values for the continuous variables or categorized as a separate group for the categorical variables before entering into the multivariable regression model to avoid losing study samples. In addition, the continuous variables under skewed distribution were logarithm transformed in the regression analyses. We also did sensitivity analysis by Poisson regression. All statistical analyses were performed using SPSS 16 software (SPSS Inc.). P=0.05 was considered statistically significant.
Results
Altogether, 555 patients with confirmed COVID-19 were included in this study, with a median follow-up time of 19 (14, 24) days; 417 were all patients with COVID-19 who were diagnosed in Shenzhen city and cared for in one designated hospital (Shenzhen cohort), and the other 138 patients were from Wuhan city with severe disease conditions needing medical care in the hospital (Wuhan cohort). As shown in Table 1, patients in the Wuhan cohort were much older than those in the Shenzhen cohort (median 66 versus 47 years), with more patients having concomitant diseases (78% versus 22%, respectively). At admission, the majority of the Shenzhen cohort was staged as mild and moderate (78%; 326 of 417), with 17% (72 of 417) of the patients staged as severe and 5% (19 of 417) of the patients staged as critical. All of the patients in the Wuhan cohort were staged as either severe (88%; 122 of 138) or critical (12%; 16 of 138). The laboratory characteristics and treatment of the two cohorts are listed in Supplemental Table 2.
Table 1.
Baseline characteristics and outcomes of patients diagnosed with coronavirus disease 2019 in one of two Chinese cohorts: consecutive patients with severe coronavirus disease 2019 and patients with critical coronavirus disease 2019 from Peking University Medical Team admitted from February 8 to 28, 2020 (Wuhan cohort) and all patients with coronavirus disease 2019 diagnosed in Shenzhen city and transferred to Shenzhen Third People’s Hospital from January 8 to February 25, 2020 (Shenzhen cohort)
| Clincal Characteristics | Total, n=555 | Wuhan, n=138 | Shenzhen, n=417 |
|---|---|---|---|
| Men, no. (%) | 269 (48) | 71 (51) | 198 (47) |
| Age, yr | 52 (36, 64) | 66 (54, 71) | 47 (34, 60) |
| Duration from onset of symptoms to admission, d | 5 (3, 11) | 14 (11, 18) | 4 (2, 7) |
| Disease severity at admission, no. (%) | |||
| Mild | 12 (2) | 0 (0) | 12 (3) |
| Moderate | 314 (57) | 0 (0) | 314 (75) |
| Severe | 194 (35) | 122 (88) | 72 (17) |
| Critical | 35 (6) | 16 (12) | 19 (5) |
| Concomitant diseases, no. (%) | |||
| Any | 198 (36) | 108 (78) | 90 (22) |
| Hypertension | 155 (28) | 92 (67) | 63 (15) |
| Diabetes | 53 (10) | 28 (20) | 25 (6) |
| Coronary heart disease | 41 (7) | 26 (19) | 15 (4) |
| COPD or asthma | 34 (6) | 21 (15) | 13 (3) |
| CKDa | 10 (2) | 9 (7) | 1 (0.2) |
| Malignant tumor | 4 (1) | 3 (2.2) | 1 (0.2) |
| Outcome, no. (%) | |||
| Death | 27 (5) | 24 (17) | 3 (0.7) |
| Progression to critical illnessb | 40 (8) | 18 (15) | 22 (6) |
COPD, chronic obstructive pulmonary disease.
CKD was defined as abnormalities of kidney structure or function, present for >3 months, according to the 2012 Kidney Disease Improving Global Outcomes CKD definition on the basis of the past medical history reported by the patients along with their medical records when available.
Patients who were critical at admission were excluded for the outcome of disease progression to critical illness (n=35).
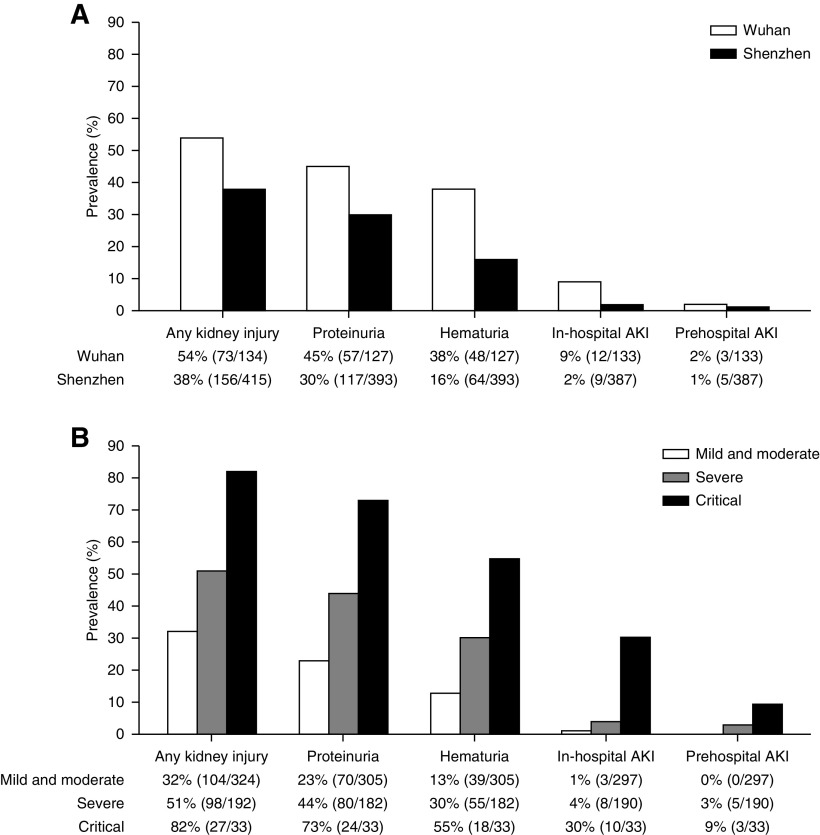
Six patients having neither urinalysis at admission nor repeated sCr tests during hospital stay were excluded from further evaluation of kidney injury indicators, of which two patients were classified as having critical illness at admission and died during follow-up. Any kidney injury was observed in 42% of the remaining 549 patients (229 of 549), with proteinuria, hematuria, and AKI detected in 33% (174 of 520), 22% (112 of 520), and 6% (29 of 520), respectively. Of the 29 patients with AKI, 21 patients were recognized as having in-hospital AKI (72%), and eight patients were recognized as having prehospital AKI (28%). As shown in Figure 1A, proteinuria was the most common kidney injury indicator (30%; 117 of 393) in the Shenzhen cohort, followed by hematuria (16%; 64 of 393), in-hospital AKI (2%; nine of 387), and prehospital AKI (1%; five of 387). In the Wuhan cohort, however, the prevalence of each indicator was higher than that in the Shenzhen cohort, with proteinuria detected in 45% (57 of 127), hematuria detected in 38% (48 of 127), in-hospital AKI detected in 9% (12 of 133), and prehospital AKI detected in 2% (three of 133). The prevalence of kidney injury indicators was significantly higher with greater disease severity in the whole population and in each cohort as well, with no differences between the two cohorts when patients had the same disease severity (Figure 1B, Supplemental Figure 1).
Figure 1.
The prevalence of kidney injury indicators increased in parallel with disease severity of COVID-19. The prevalence of kidney injury according to (A) cohorts and (B) disease severity at admission. Disease severity was classified as mild and moderate, severe, or critical; 35 individuals for proteinuria, 35 individuals for hematuria, 35 individuals for in-hospital AKI, 35 individuals for prehospital AKI, and six individuals for any kidney injury were excluded due to missing data.
Of the 21 patients with in-hospital AKI, the median time to AKI onset with respect to hospital admission was 9 (4, 13) days, and the median time to onset of COVID-19 symptoms was 18 (14, 22) days. The peak stages of in-hospital AKI were stage 1 in 38% (eight of 21), stage 2 in 19% (four of 21), and stage 3 in 43% (nine of 21). Kidney replacement therapy was required in 24% (seven of 29) of all patients with AKI, 29% (six of 21) of all patients with in-hospital AKI, and 12% (one of eight) of all patients with prehospital AKI. In the eight patients with prehospital AKI, three patients (38%) had critical illness at hospital admission, and another two patients (25%) progressed to critical illness during hospitalization. Of the 21 patients with in-hospital AKI, 15 patients (71%) met the criteria for critical illness either at admission (n=10) or during hospital stay (n=5). Among these 15 patients with critical illness, 14 (93%) developed AKI after the identification of critical illness.
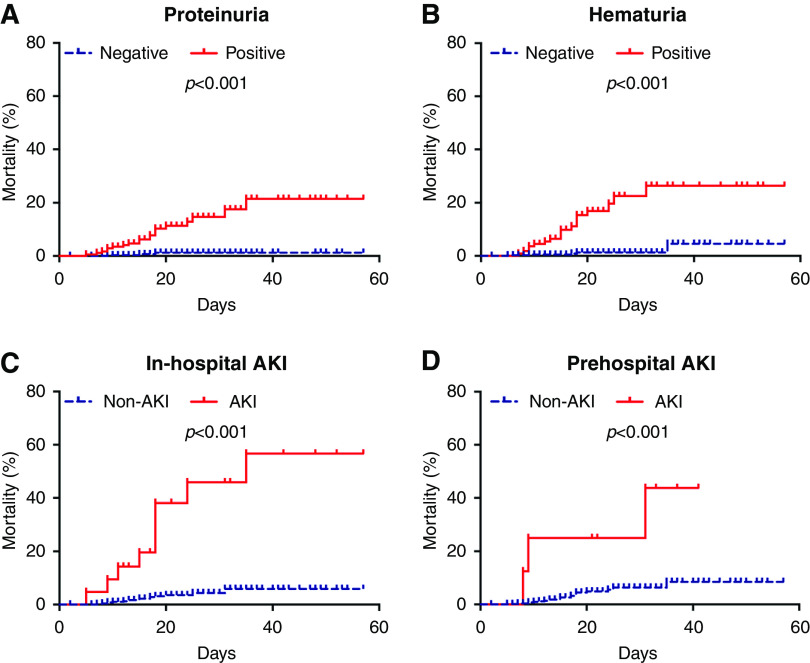
By March 1, 2020, altogether 27 patients (5%; 27 of 555) died, including 24 patients (17%; 24 of 138) in the Wuhan cohort and three patients (0.7%; three of 417) in the Shenzhen cohort. The death rate was 11% (20 of 174) in patients with proteinuria, 16% (18 of 112) in patients with hematuria, 41% (12 of 29) in all AKI settings, 43% (nine of 21) in patients with in-hospital AKI, and 38% (three of eight) in patients with prehospital AKI. Kaplan–Meier analysis showed a significantly higher death rate for patients with any kidney injury, including proteinuria, hematuria, in-hospital AKI, and prehospital AKI (all P<0.001) (Figure 2). After adjusting age, sex, disease severity at admission, lactate dehydrogenase, C-reactive protein, troponin, d-dimer, lymphocyte count, and comorbidities, multivariable Cox regression analysis showed that proteinuria (hazard ratio [HR], 4.42; 95% confidence interval [95% CI], 1.22 to 15.94), hematuria (HR, 4.71; 95% CI, 1.61 to 13.81), and in-hospital AKI (HR, 6.84; 95% CI, 2.42 to 19.31) were associated with death (Table 2).
Figure 2.
Kaplan–Meier analysis showed a significantly higher death rate for patients with proteinuria, hematuria, in-hospital AKI, and prehospital AKI. Cumulative incidence for death of patients subgrouped by (A) proteinuria, (B) hematuria, (C) in-hospital AKI, and (D) prehospital AKI.
Table 2.
Association of kidney injury indicators with death and disease progression in patients with coronavirus disease 2019
| Outcomes | Exposed (No. of Events/No. of Patients) | Unexposed (No. of Events/No. of Patients) | Hazard Ratio (95% Confidence Interval) | P Value |
|---|---|---|---|---|
| In-hospital death, n=555a | ||||
| Proteinuria | 20/174 | 3/346 | 4.42 (1.22 to 15.94) | 0.02 |
| Hematuria | 18/112 | 5/408 | 4.71 (1.61 to 13.81) | 0.02 |
| In-hospital AKI | 9/21 | 16/499 | 6.84 (2.42 to 19.31) | <0.001 |
| Prehospital AKI | 3/8 | 22/512 | 1.06 (0.29 to 3.91) | 0.92 |
| Any kidney injury | 23/229 | 2/320 | 5.01 (1.13 to 22.15) | 0.03 |
| Progression to critical illness, n=520b | ||||
| Proteinuria | 24/150 | 12/337 | 2.61 (1.22 to 5.56) | 0.01 |
| Hematuria | 18/94 | 18/393 | 2.50 (1.23 to 5.08) | 0.01 |
| In-hospital AKI | 1/7 | 39/480 | 1.58 (0.21 to 11.68) | 0.66 |
| Prehospital AKI | 2/5 | 38/482 | 1.03 (0.22 to 4.75) | 0.97 |
| Any kidney injury | 27/200 | 13/316 | 1.89 (0.95 to 3.78) | 0.07 |
Hazard ratios of each variable were obtained using separate proportional hazard Cox models after adjustment for age, sex, disease severity at admission, lactate dehydrogenase, C-reactive protein, troponin, d-dimer, lymphocyte count, and comorbidities. Comorbidities include CKD, hypertension, diabetes, coronary heart disease, chronic pulmonary disease, or malignant tumor history. In-hospital AKI was set as a time-dependent covariate.
For the outcome of in-hospital death, 35 individuals for proteinuria, 35 individuals for hematuria, 35 individuals for in-hospital AKI, 35 individuals for prehospital AKI, and six individuals for any kidney injury were excluded due to missing data.
For the outcome of progression to critical illness, 33 individuals for proteinuria, 33 individuals for hematuria, 33 individuals for in-hospital AKI, 33 individuals for prehospital AKI, and four individuals for any kidney injury were excluded due to missing data.
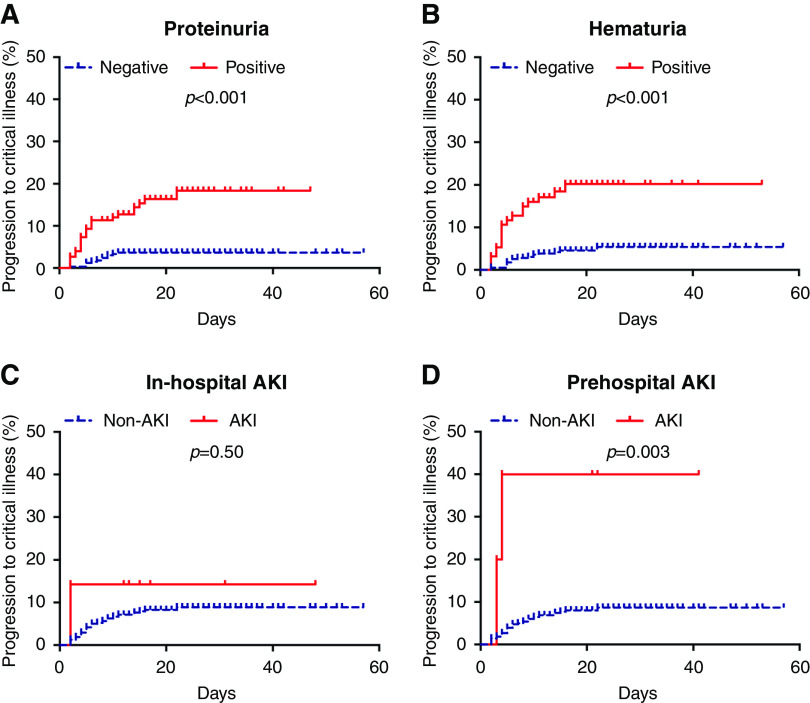
Among the 520 noncritical patients at admission, 40 patients progressed to critical illness during the observational period. The proportions of patients who achieved the end point were 16% (24 of 150) and 4% (12 of 337) in those with and without proteinuria, respectively; 19% (18 of 94) and 5% (18 of 393) in those with and without hematuria, respectively; 40% (two of five) and 8% (38 of 482) in those with and without prehospital AKI, respectively; and 14% (one of seven) and 8% (39 of 480) in those with and without in-hospital AKI, respectively (Table 2). Kaplan–Meier analysis showed a significantly higher progression rate for patients with proteinuria, hematuria, and prehospital AKI at admission (all P<0.01) (Figure 3). In multivariable analysis, only proteinuria (HR, 2.61; 95% CI, 1.22 to 5.56) and hematuria (HR, 2.50; 95% CI, 1.23 to 5.08) were associated with disease progression (Table 2).
Figure 3.
Kaplan–Meier analysis showed a significantly higher progression rate for patients with proteinuria, hematuria, and prehospital AKI at admission. Cumulative incidence for disease progression to critical illness of patients subgrouped by (A) proteinuria, (B) hematuria, (C) in-hospital AKI, and (D) prehospital AKI.
Results of sensitivity analysis by Poisson regression were consistent with the Cox regression (Supplemental Table 3). We observed no significant association between corticosteroid use with outcomes. Rate ratio was 1.53 (95% CI, 0.86 to 2.70) for the association with death and 1.00 (95% CI, 0.42 to 2.37) for disease progression by Poisson regression.
Discussion
In this study involving two large cohorts, we observed a high prevalence of kidney injury in the overall population of patients with COVID-19 (38%) represented by the Shenzhen cohort and in severe hospitalized patients (54%) represented by the Wuhan cohort. Kidney injury mainly presented as proteinuria and hematuria, both of which were independently associated with death or progression to critical illness. Although the incidence of AKI was relatively low in the whole population of patients with COVID-19, the rate was insignificantly higher when patients reached a critical stage. The finding that both proteinuria and hematuria in COVID-19 were associated with disease progression or death highlights the importance of urine analysis or even a dipstick test in patients with SARS-CoV-2 infection.
Kidney involvement in SARS-CoV-2 has not drawn much attention, with the understanding that AKI mainly developed in patients with severe cases (14,22,23) as a part of multiorgan dysfunction due to virus infection and cytokine storm. In a recent study on 710 patients with COVID-19 from Tongji Hospital, Wuhan, the prevalence rates of proteinuria, hematuria, and AKI were reported as 44%, 27%, and 5%, respectively, and kidney involvement was found to be associated with death (14). However, the status of kidney involvement in the whole SARS-CoV-2–infected population, especially in those with mild to moderate disease conditions, is still unknown. In this study, we included two representative cohorts, the Shenzhen cohort with all patients with COVID-19 in the whole city and the Wuhan cohort with patients with severe disease conditions needing hospitalization (this cohort is similar to but not the same as the previously published cohort [14]). It is important to note that not only in patients with severe COVID-19, but also in the general infected population, kidney injury was common, with higher frequency, particularly of AKI, in patients with severe COVID-19. Furthermore, in noncritical patients, proteinuria and hematuria were strongly associated with subsequent disease progression. Our data indicate the importance of evaluation of kidney injury in all of the patients with SARS-CoV-2 infection.
COVID-19 has already become a global pandemic, and the health system across the world is facing big challenges and great pressures. Although the majority of SARS-CoV-2 infections are mild and moderate, about 5% of the patients will progress and be classified as having severe or critical conditions, with significantly higher mortality (24,25). Therefore, the ability to identify patients at high risk for disease progression at an early stage is of critical importance. Several studies have defined oxygen saturation, respiratory rate, blood leukocyte/lymphocyte count, and chest x-ray/computed tomography manifestations as risk factors for poor clinical outcomes. Our study showed that kidney injury could be a new risk factor for disease progression or death. Especially in noncritical patients, kidney involvement presenting as proteinuria or hematuria was a stable risk factor for poor outcomes. These findings support adding kidney injury indicators into the patient’s severity score system. A simple urine dipstick could potentially help with risk evaluation and patient stratification.
The mechanism of kidney involvement of SARS-CoV-2 remains unclear. SARS-CoV-2 uses the SARS-CoV-2 receptor ACE2 for host cell entry. Analysis using a kidney single-cell sequencing database showed that ACE2 was highly expressed in the tubular epithelium, especially in proximal tubular cells, suggesting that tubular epithelium maybe a target for infection (26). Recently, viral RNA was identified in urine samples of patients infected with SARS-CoV-2 (27), with angiotensin-converting enzymes expressed on kidney tubular cells being identified as binding partners (28). Therefore, direct tubular cell injury through virus-binding angiotensin-converting enzyme may be one of the mechanisms (29). Other postulated mechanisms, such as cytokine storm, sepsis, hypoperfusion, factors related to treatment, etc., could also contribute to kidney injury. In this study, over two thirds of the in-hospital AKI episodes were developed after patients reached critical illness, whereas in prehospital AKI settings, 62% of the patients had AKI under less severe disease conditions, which indicates the comprehensive pathogenic factors in the development of COVID-19–related kidney injuries.
In this study, we used two representative COVID-19 populations to demonstrate that kidney injury was a stable risk factor for disease progression. However, this study had several limitations. First, because of the retrospective study design, CKD was defined on the basis of the past medical history reported by the patients along with their medical records when available. It is possible that some patients might not know of their underlying CKD, and therefore, this would lead to an overestimation of the incidence of COVID-19–related kidney injury, especially when urinalysis abnormalities occurred without AKI. Second, because the Shenzhen cohort included the infected patients from the whole city, most of the patients had nonsevere illness, and therefore, SOFA or APACHE scores were evaluated. Third, the number of deaths that occurred in the study is small, which has limited the power to evaluate the actual effect value of kidney injury on death. Fourth, kidney biopsy was not performed, and therefore, the pathology lesions of patients with COVID-19 and kidney injury could not be investigated. The limited postmortem biopsies data suggest that tubular interstitial injury was the main pathologic change, with protein exudation detected in the glomerular Bowman’s capsule (30).
In conclusion, our study shows that kidney injury is common in patients with COVID-19, even in those at mild and moderate disease stages. Kidney injury is associated with poor clinical outcomes; proteinuria and hematuria in noncritical patients are independently associated with the progression to respiratory failure or death, whereas AKI mainly occurs in critical patients. We recommend that all patients with SARS-CoV-2 infection receive kidney evaluation to aid in early identification of high-risk patients.
Disclosures
All authors have nothing to disclose.
Funding
This work was supported by China National Funds for Distinguished Young Scientists grants 81625004 and 81925006 and Beijing Outstanding Young Scientist Program grant BJJWZYJH01201910001006.
Supplementary Material
Acknowledgments
We acknowledge all health care workers of the Peking University medical team in Wuhan and Shenzhen Third People’s Hospital involved in the diagnosis and treatment of patients; we thank all patients involved in the study.
The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Dr. Jicheng Lv and Dr. Li Yang conceived and designed the study; Dr. Yaping Dong, Dr. Wanyin Hou, Dr. Haichao Li, Dr. Xiaolong Li, Dr. Yang Li, Dr. Jinwei Wang, Dr. Lingyi Xu, Dr. Hongyu Yang, Dr. Qi Yu, Dr. Xin Zhang, Dr. Xizi Zheng, and Dr. Youlu Zhao collected the clinical data and processed statistical data; Dr. Hongyu Yang and Dr. Xizi Zheng drafted the manuscript; Dr. Hong Gao, Dr. Feng Hu, Dr. Jicheng Lv, and Dr. Li Yang revised the final manuscript; and all authors approved the final manuscript for submission.
Data Sharing Statement
The datasets used and analyzed during this study are available from the corresponding author on reasonable request.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
Supplemental Material
This article contains the following supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.04780420/-/DCSupplemental.
Supplemental Figure 1. The prevalence of kidney injury in (A) severe and (B) critical patients.
Supplemental Table 1. Clinical classifications of disease severity of patients with COVID-19.
Supplemental Table 2. Baseline laboratory data and treatment of patients with COVID-19.
Supplemental Table 3. Association of kidney injury indicators with death and disease progression in patients with COVID-19.
References
- 1.Lu H, Stratton CW, Tang YW: Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle. J Med Virol 92: 401–402, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hui DS, I Azhar E, Madani TA, Ntoumi F, Kock R, Dar O, Ippolito G, Mchugh TD, Memish ZA, Drosten C, Zumla A, Petersen E: The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health. The latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis 91: 264–266, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization : Coronavirus Disease 2019 (COVID-19) Situation Report 77, 2020. Available at: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200406-sitrep-77-covid-19.pdf?sfvrsn=21d1e632_2. Accessed April 7, 2020
- 4.Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, Wu Y, Zhang L, Yu Z, Fang M, Yu T, Wang Y, Pan S, Zou X, Yuan S, Shang Y: Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study [published correction appears in Lancet Respir Med 8: e26, 2020 10.1016/S2213-2600(20)30103-X]. Lancet Respir Med 8: 475–481, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS: Pathological findings of COVID-19 associated with acute respiratory distress syndrome [published correction appears in Lancet Respir Med 8: e26, 2020 10.1016/S2213-2600(20)30085-0]. Lancet Respir Med 8: 420–422, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tian S, Hu W, Niu L, Liu H, Xu H, Xiao SY: Pulmonary pathology of early-phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J Thorac Oncol 15: 700–704, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kui L, Fang YY, Deng Y, Liu W, Wang MF, Ma JP, Xiao W, Wang YN, Zhong MH, Li CH, Li GC, Liu HG: Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin Med J (Engl) 133: 1025–1031, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chang D, Lin M, Wei L, Xie L, Zhu G, Dela Cruz CS, Sharma L: Epidemiologic and clinical characteristics of novel coronavirus infections involving 13 patients outside Wuhan, China. JAMA 323: 1092–1093, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z: Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323: 1061–1069, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tang N, Li D, Wang X, Sun Z: Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 18: 844–847, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang C, Shi L, Wang F-S: Liver injury in COVID-19: Management and challenges. Lancet Gastroenterol Hepatol 5: 428–430, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B: Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497–506, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L: Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 395: 507–513, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, Li J, Yao Y, Ge S, Xu G: Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int 97: 829–838, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.World Health Organization: Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected: Interim guidance, 2020. Available at: https://extranet.who.int/iris/restricted/bitstream/handle/10665/330854/WHO-nCoV-Clinical-2020.2-eng.pdf?sequence=1&isAllowed=y. Accessed March 1, 2020
- 16.National Health Committee of the People’s Republic of China : Guidance for Corona Virus Disease 2019: Prevention, Control, Diagnosis and Management, 1st Ed., Beijing, China, People’s Medical Publication House, 2020 [Google Scholar]
- 17.Kidney Disease Improving Global Outcomes: Acute Kidney Injury Work Group : KDIGO clinical practice guideline for acute kidney injury Section 2: AKI Definition. Available at https://kdigo.org/guidelines/acute-kidney-injury/. Accessed March 25, 2020.
- 18.Liang W, Liang H, Ou L, Chen B, Chen A, Li C, Li Y, Guan W, Sang L, Lu J, Xu Y, Chen G, Guo H, Guo J, Chen Z, Zhao Y, Li S, Zhang N, Zhong N, He J; China Medical Treatment Expert Group for COVID-19 : Development and validation of a clinical risk score to predict the occurrence of critical illness in hospitalized patients with COVID-19. JAMA Intern Med 180: 1–9, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19 : Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 382: 1708–1720, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B: Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395: 1054–1062, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu Z, McGoogan JM: Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese center for disease control and prevention [published online ahead of print February 24, 2020]. JAMA 10.1001/jama.2020.2648 [DOI] [PubMed] [Google Scholar]
- 22.Yang Xiaobo, Yu Yuan, Xu Jiqian, Shu Huaqing, Xia Jia ’an, Liu Hong, Wu Yongran, Zhang Lu, Yu Zhui, Fang Minghao, Yu Ting, Wang Yaxin, Pan Shangwen, Zou Xiaojing, Yuan Shiying, Shang You: Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir Med 8[5]: 475–481, 2020. 10.1016/S2213-2600(20)30079-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang Yang, Lu Xiaofan, Li Yongsheng, Chen Hui, Chen Taige, Su Nan, Huang Fang, Zhou Jing, Zhang Bing, Yan Fangrong, Wang Jun: Clinical course and outcomes of 344 intensive care patients with COVID-19. Am. J. Respir. Crit. Care Med 201[11]: 1430–1434, 2020. 10.1164/rccm.202003-0736LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wu J, Lu AD, Zhang LP, Zuo YX, Jia YP: [Study of clinical outcome and prognosis in pediatric core binding factor-acute myeloid leukemia]. Zhonghua Xue Ye Xue Za Zhi 40: 52–57, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guan W, Ni Z-y, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19 : Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 382: 1708–1720, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zou X, Chen K, Zou J, Han P, Hao J, Han Z: Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med 14: 185–192, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sun Jing, Zhu Airu, Li Heying, Zheng Kui, Zhuang Zhen, Chen Zhao, Shi Yongxia, Zhang Zhaoyong, Chen Si-Bei, Liu Xuesong, Dai Jun, Li Xiaobo, Huang Shuxiang, Huang Xiaofang, Luo Ling, Wen Liyan, Zhuo Jianfen, Li Yuming, Wang Yanqun, Zhang Lu, Zhang Yanjun, Li Fang, Feng Liqiang, Chen Xinwen, Zhong Nanshan, Yang Zifeng, Huang Jicheng, Zhao Jincun, Li Yi-Min: Isolation of infectious SARS-CoV-2 from urine of a COVID-19 patient. Emerg Microbes Infect 9[1]: 991–993, 2020. 10.1080/22221751.2020.1760144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang H, Penninger JM, Li Y, Zhong N, Slutsky AS: Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med 46: 586–590, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hoffmann Markus, Kleine-Weber Hannah, Schroeder Simon, Krüger Nadine, Herrler Tanja, Erichsen Sandra, Schiergens Tobias S, Herrler Georg, Wu Nai-Huei, Nitsche Andreas, Müller Marcel A, Drosten Christian, Pöhlmann Stefan: SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181[2]: 271–280.e8, 2020. 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Su H, Yang M, Wan C, Yi LX, Tang F, Zhu HY, Yi F, Yang HC, Fogo AB, Nie X, Zhang C: Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int 98: 219–227, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.