Abstract
SARS-CoV-2 causes a severe pneumonia (COVID-19) that affects essentially elderly people. In COVID-19, macrophage infiltration into the lung causes a rapid and intense cytokine storm leading finally to a multi-organ failure and death. Comorbidities such as metabolic syndrome, obesity, type 2 diabetes, lung and cardiovascular diseases, all of them age-associated diseases, increase the severity and lethality of COVID-19. Mitochondrial dysfunction is one of the hallmarks of aging and COVID-19 risk factors. Dysfunctional mitochondria is associated with defective immunological response to viral infections and chronic inflammation. This review discuss how mitochondrial dysfunction is associated with defective immune response in aging and different age-related diseases, and with many of the comorbidities associated with poor prognosis in the progression of COVID-19. We suggest here that chronic inflammation caused by mitochondrial dysfunction is responsible of the explosive release of inflammatory cytokines causing severe pneumonia, multi-organ failure and finally death in COVID-19 patients. Preventive treatments based on therapies improving mitochondrial turnover, dynamics and activity would be essential to protect against COVID-19 severity.
Keywords: COVID-19, SARS-CoV-2, Mitochondria, Inflammaging, Inflammation, Cytokine storm, Mitochondrial health, Mitochondrial nutrients, Mitochondrial turnover
Highlights
-
•
Mitochondrial dysfunction is associated with aging and age-related diseases and metabolic syndrome.
-
•
Unbalanced activity of mitochondria is involved in inflammation and dysfunctional response to virus infection.
-
•
COVID-19 severity is higher in aged and metabolic syndrome related diseases.
-
•
Improvement of mitochondrial activity during aging can improve response to virus infections including respiratory virus.
-
•
Reduction of dysfunctional mitochondria can reduce the inflammatory response to SARS-CoV-2 infection.
1. Introduction
Coronavirus Disease 2019 (COVID-19) is a complex respiratory and thrombogenic disease caused by a new strain of coronavirus known as Severe Acute Respiratory Syndrome-Coronavirus 2 (SARS-CoV-2). Many of the patients infected with SARS-CoV-2 are asymptomatic or show low intensity symptoms. However, around 20% of the patients, mainly elderly people, manifest severe symptoms and high mortality ratio (Chen et al., 2020). COVID-19 predominant symptoms are high fever, intense cough, pneumonia and myalgia, mainly leading to acute respiratory distress syndrome (ARDS) (Rothan and Byrareddy, 2020). Diarrhea and nausea can precede fever or respiratory symptoms (Huang et al., 2020). These symptoms can be aggravated in senior patients aged more than 70 years (Annweiler et al., 2020).
ARDS associated with COVID-19 is reminiscent of the cytokine release syndrome (CRS)-induced ARDS caused by the first SARS-CoV and of the secondary hemophagocytic lymphohistiocytosis (sHLH) associated with the disease caused by Middle East Respiratory Syndrome Coronavirus (MERS-CoV) (Moore and June, 2020). As in the case of SARS and MERS, the severity of COVID-19 also correlates with high levels of interleukin-6 (IL-6) and other proinflammatory cytokines (Chen et al., 2020; Ruan et al., 2020). This exaggerated innate immunity response is also associated with the severity of influenza infection in aged individuals (Chen et al., 2020) indicating its importance in the lethality by respiratory viruses. In fact, as in the case of COVID-19, adults over 65 show high vulnerability to influenza virus (McGuire, 2019; Etard et al., 2020) indicating that age is the main factor in the severity of viral respiratory infections.
Clinical severity of COVID-19 correlates with lymphopenia, low blood lymphocyte count and essentially with CRS, indicating a disturbance of the immunological function and an unbalanced immune response (Yang et al., 2020). Anti-cytokine therapies have been proposed as therapeutic options to reverse hyper-inflammation and decrease COVID-19 severity (Roshanravan et al., 2020). Further, T cell exhaustion associated with dendritic cell dysfunction has been also associated with the immunopathology of COVID-19 (Zheng et al., 2020a; Zheng et al., 2020b) and an active role of local SARS-CoV-2 infected neutrophils and macrophages in inflammatory reactions has been recently proposed (Jafarzadeh et al., 2020).
The severity of COVID-19 is associated with the dysfunction of the immune system found in aged individuals and recently, this aged immunity has been considered to exacerbate COVID-19 (Akbar and Gilroy, 2020). Many of the age-associated diseases such as metabolic syndrome (MS), type 2 diabetes (T2D), obesity, hypertension and cardiovascular diseases (CVD) also aggravate the severity of COVID-19 (Pirola and Sookoian, 2020; Zheng et al., 2020c). The deterioration of the immune system associated with aging is called immunosenescence (Currie, 1992), which is defined as the gradual deterioration of the immune system associated with the natural age progression (De Martinis et al., 2007; Pangrazzi and Weinberger, 2020). Immunosenescence is key in the deteriorated immune response against viral infections. This restricted immune response prevents successful inhibition of viral spread at early stages of infection exacerbating morbidity and mortality in this population (Jing et al., 2009; Montecino-Rodriguez et al., 2013). Importantly, aging is also associated with a proinflammatory profile known as “inflammaging” (Franceschi et al., 2017). In inflammaging, activated cells such as microglia and macrophages or dendritic cells increase the activity of nuclear factor kappa light chain enhancer of activated B-cells (NF-κB), cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), leading to the release of proinflammatory cytokines such as interleukin 6 (IL6), IL1β, and tumor necrosis factor alpha (TFNα) among others (Dantzer et al., 2008; Agostinho et al., 2010). This chronic inflammation has been associated with the inhibition of antigen-specific immunity and the low response of aged people to vaccines (Akbar and Gilroy, 2020; Parmigiani et al., 2013).
Deterioration of mitochondrial function is key in the progression of aging and age-related diseases (Lopez-Lluch et al., 2015). Mitochondrial dysfunction is also involved in the deterioration of the immune system being a key factor in inflammaging, higher susceptibility to viral infections and impaired T cell immunity found in aged people and in age-associated diseases (McGuire, 2019). In the present review, we show evidence indicating that mitochondrial dysfunction is a key factor for the severity of COVID-19 disease and for triggering the cytokine storm associated with the devastating symptoms that collapse lungs and lead to death to COVID-19 patients. Improvement of mitochondrial activity in the elderly would serve as preventive therapy to improve the immune response and reduce mortality caused by this disease and other viral respiratory diseases such as influenza.
2. Mitochondrial dysfunction and aging
The highest severity of COVID-19 is found in aged male patients and suffering overweight or obesity (Mauvais-Jarvis, 2020). Mitochondrial dysfunction is deeply associated with two of these factors, aging and obesity (Lopez-Lluch, 2017b). Mitochondria are essential for many key activities in cells involved not only in energy production but also in the synthesis of nucleotides, modification of membrane phospholipids and regulation of calcium homeostasis. Further, dysfunctional mitochondria are the main sources of reactive oxygen species (mtROS) that increase oxidative damage during aging and metabolic diseases (Lopez-Lluch et al., 2018). The accumulation of damaged mitochondria by disruption or malfunction of different nutrient sensor mechanisms in the cells is a hallmark aging and metabolic syndrome including obesity (Lopez-Lluch et al., 2018).
Age-related mitochondrial dysfunction is also a consequence of the unbalance of mitochondrial dynamics, mito/autophagy and biogenesis, producing the accumulation of damaged mitochondria (Lopez-Lluch, 2017a, Lopez-Lluch, 2017b; Lopez-Lluch et al., 2015). Defective autophagy is involved in aging progression and in the pathophysiology of many metabolic and age-related diseases affecting different organs and tissues (Evans et al., 2017; Y.H. Lee et al., 2019; Lin et al., 2019; Yamaguchi, 2019). The accumulation of deficient mitochondria through a decrease in mitochondrial turnover has been associated with many age-related diseases including not only metabolic diseases (Natarajan et al., 2020), but also cardiovascular (Shemiakova et al., 2020) and neurodegenerative diseases (Wu et al., 2019) sarcopenia (Shally and McDonagh, 2020) or cancer (Moro, 2019) among others. Furthermore, many prolongevity effectors induce autophagy as a common denominator of their respective anti-aging effect (Stead et al., 2019).
In the case of metabolic syndrome, a key factor in COVID-19 severity (Stefan et al., 2020), mitochondrial dysfunction is key in the increase of insulin resistance associated with type 2 diabetes connected with the increase of mutations in mtDNA, changes in ATP levels, generation of ROS and unbalanced mitochondrial turnover (Skuratovskaia et al., 2020). The metabolic inflexibility caused by the accumulation of damaged mitochondria in both, metabolic syndrome and aging, produces an inadequate metabolic substrate disposal affecting glucose and free fatty acid metabolism that is associated with a cluster of metabolic abnormalities that affects many organs and tissues including immune cells (Chakravarthy and Neuschwander-Tetri, 2020).
Immunosenescence has been described as the aged-dependent deterioration of the immune system (Currie, 1992), and as the adaptation of the immune system to deteriorative changes over the time (De Martinis et al., 2007). Whatever it is, immunosenescence, as many other physiological dysfunctions in aging, has been partially associated with dysfunctional mitochondria (McGuire, 2019). Mitochondrial dysfunction associated with a defective mitochondrial turnover can specially affect T-helper CD4+ lymphocytes that control the whole immune response (Bektas et al., 2019). On the other hand, dysfunction of mitochondria produces deterioration of T-cell activity that, in a vicious cycle, increases senescence in many other tissues by activating cytokine storm (Desdín-Micó et al., 2020).
Importantly, several inflammatory disorders associated with aging and mitochondrial dysfunction such as neurodegenerative diseases, diabetes and atherosclerosis among others, with a clear proinflammatory profile has been associated with the chronic aberrant NLRP3 inflammasome activation (Kelley et al., 2019). Interestingly, the relationship of mitochondrial dysfunction with inflammation increases just after the fifth decade of life, just when COVID-19 shows a rise in its severity and mortality (McGuire, 2019). Thus, it seems clear that mitochondrial dysfunction is an important factor in the proinflammatory profile caused by the release of inflammatory cytokines produced by activation of NLRP3 inflammasome and other mechanisms over-activated in aging and in metabolic diseases.
3. Mitochondrial dysfunction in inflammatory response
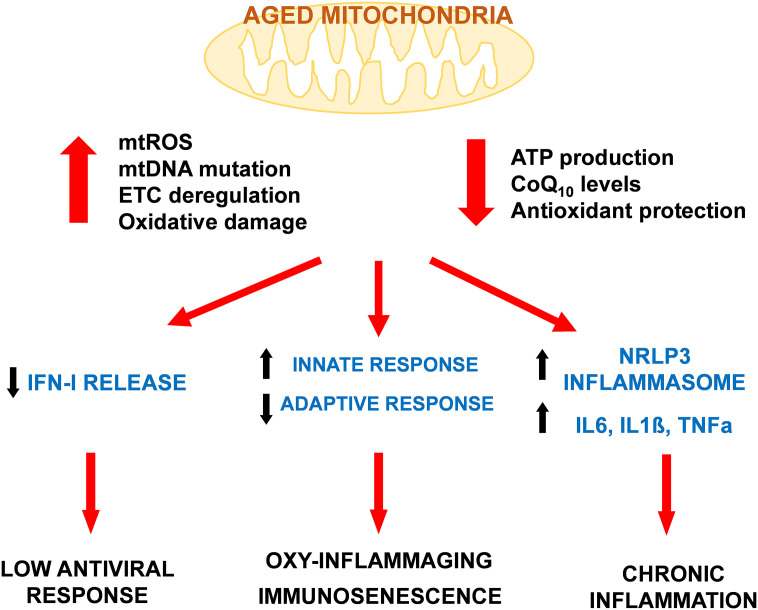
The response of the immune system is very complex and depends on the type of pathogen or agent to be blocked (Justiz Vaillant and Jan, 2020). Many factors influence the immune response and one of them is mitochondria, that has been strongly associated with the activity of the innate response (Sandhir et al., 2017). Inflammatory response depends on the innate immunity that is strongly affected by dysfunctional mitochondrial activity (Missiroli et al., 2020) (Fig. 1 ).
Fig. 1.
Mitochondrial dysfunction associated with aged mitochondria reduces the efficiency of immune system. The accumulation of mitochondrial damage together with the reduction of the efficiency in the production of energy affects immune system capacity by reducing the capacity to respond to viral infections through a lower release of IFN-I. Aged mitochondria is also involved in the unbalance of immune system by increase of innate response and decrease of the adaptive response found in immunosenescence. Mitochondrial dysfunction releases many damage signals to cytosol that end in the activation of inflammasome and the release of inflammatory cytokines that cause the chronic inflammation associated with aging and age-related diseases.
Many different pathogens are recognized through their repetitive molecular signatures by pattern recognition receptors (PRRs). These receptors are present on the cell surface and in intracellular compartments of innate cells. Their main receptors are Toll-like receptors (TLRs), retinoic acid-inducible gene I-like receptors (RLRs), nucleotide oligomerization domain receptors (NLRs) and cytosolic DNA sensors (cyclic GMP-AMP synthase (cGAS) and Stimulator of interferon genes (STING)) (Thompson et al., 2011). These receptors are activated by pathogen-associated molecular patterns (PAMPs) and also by damage-associated molecular patterns (DAMPs) (Tang et al., 2012). PAMPs are found in pathogens, whereas DAMPs are produced into the cells during stress, ROS release, apoptosis or necrosis and mainly induce inflammatory response. In this response, NLRP3 (NLR family, pyrin domain containing 3) inflammasome triggers the activation of Caspase 1 (CASP1) and activates the secretion of proinflammatory cytokines such as IL1β and IL18 (Zhao and Zhao, 2020).
Mitochondrial dysfunction plays an essential role in the activation of NLRP3 since DAMPs can be released especially by damaged mitochondria (Janeway and Medzhitov, 2002). To date, many mitochondrial DAMPs have been identified: cardiolipin, n-formyl-peptides, mitochondrial transcription factor A (TFAM), mitochondrial ROS (mtROS) and mitochondrial DNA (mtDNA) because they mimic bacterial structures (McGuire, 2019). These DAMPs can also act independently of NLRP3. TFAM activates immune cells through interaction with receptors of advanced glycation end products (RAGE) and TLR9 (Julian et al., 2013). The release of mtDNA from dysfunctional mitochondria has been also associated with the rise of NLRP3-independent proinflammatory cytokines such as TNFα, IL6, regulatory upon activation, normal T-cell expressed and secreted (RANTES, CCL5) or interleukin 1 receptor antagonist (IL1RA) (Pinti et al., 2014).
The release of mtROS is key in the activation of the inflammatory immune response and in aging progression (De la Fuente and Miquel, 2009). The increase of the release of mtROS produced by mitochondrial dysfunction during aging can be responsible of the maintenance of a chronic state of inflammation that would reduce longevity and affect many of age-related diseases in a theory of aging known as oxi-inflammaging (De la Fuente and Miquel, 2009). This increase in the oxidative damage produced in immune cells have been associated with immunosenescence, the deterioration of immune system during aging (Garrido et al., 2019).
Oxidative stress produced by mitochondrial dysfunction is an essential factor in the appearance of “inflammaging”, the chronic inflammation profile associated with aging (Franceschi and Campisi, 2014). This chronic inflammation profile is caused, at least in part, by the accumulation of damaged macromolecules during aging by both, the increase of oxidative stress and/or by inadequate elimination due to autophagy decline (Franceschi and Campisi, 2014). The hallmarks of inflammaging are chronic and high levels of inflammatory biomarkers such as C-reactive protein or IL6 (Franceschi and Campisi, 2014). Activated cells such as glia, macrophages or dendritic cells suffer the overactivation of NF-κB, COX-2 and iNOS leading to the release of proinflammatory cytokines such as IL6, IL1β, TNFα and also the release of mtROS (Agostinho et al., 2010). As strategy, many prolongevity effectors that reduce the release of mtROS would be used to reduce the inflammatory profile through improvement of mitochondrial activity (De la Fuente et al., 2011).
Apart of the activation of NLRP3, high levels of mtROS are also involved in other immunological mechanisms involved in inflammation such as hypoxia inducible factor 1 alpha (HIF1α) and NF-κB (Brunelle et al., 2005). Activation of HIF1α by mtROS can induce the expression of one of its gene targets, the cytokine IL1β resulting in a proinflammatory phenotype (McGettrick and O'Neill, 2020; Tannahill et al., 2013). On the other hand, mtROS induce NF-κB-dependent inflammation (Herb et al., 2019), although in a regulatory feedback, induction of NF-κB by mtROS can also act as a signal to induce the removal of dysfunctional mitochondria that would reduce inflammation (Zhong et al., 2016). Furthermore, the response of NK cells to IL2 is defective in old individuals in an effect associated with the increase of oxidative stress caused by defective mitochondria (Miranda et al., 2018).
On the other hand, many studies have shown the relationship of defective mito/autophagy processes with the inflammatory profile associated with aging. Disruption of mitochondrial turnover contributes to the accumulation of damaged mitochondria and higher mtROS production that aggravates proinflammatory processes contributing to the impairment of the progression of different age-related diseases (Caruso et al., 2009) accordingly with the oxy-inflammaging theory. In the Baltimore Longitudinal Study on Aging, the accumulation of under-degraded mitochondria in autophagosomes of CD4+ T cells indicates the accumulation of dysfunctional mitochondria by deficiency of autophagy, which was associated with chronic inflammation in the elderly (Bektas et al., 2019). Chronification of local inflammation in atherosclerosis has been also associated with defective mitophagy in aged individuals (Orekhov et al., 2020).
On the contrary, several studies have demonstrated that improving mitochondrial turnover contributes to the decrease of inflammation and restoration of the activity of the immune system. Restoration of mitochondrial turnover through induction of mito/autophagy, suppress the inflammatory response by inhibiting NLRP3 activation in different models of sepsis (Kim et al., 2016). Removal of damaged mitochondria through activation of autophagy in murine macrophages protects against inflammation induced by alcohol in liver (Liang et al., 2019). In influenza infection, decrease of mtROS reduced inflammation, neutrophils infiltration and virus load without affecting B and T cell activities (To et al., 2020). In a recent review, Xu et al. (2020) summarize the accumulated evidence that implicates the elimination of dysfunctional mitochondria with the maintenance of the immune system during aging.
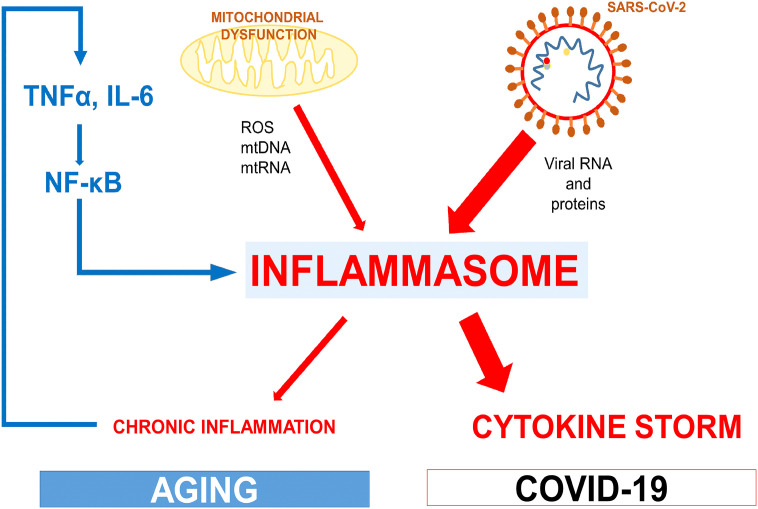
Therefore, we can speculate that the accumulation of dysfunctional mitochondria could be partially behind the high mortality rate by COVID-19 in the elderly mainly due to mitochondrial insufficiency in senescent macrophages and lymphocytes that produce an exacerbated inflammatory response (Fig. 2 ).
Fig. 2.
Age-related mitochondrial dysfunction increases the inflammatory response to SARS-CoV-2. The activation of the inflammasome caused by mitochondrial dysfunction during aging and age-related diseases contributes to the chronic inflammation and the release of inflammatory cytokines in the elderly. This system produces a vicious cycle that feed the activity of the inflammasome. When SARS-CoV-2 invades the organism, macrophages showing a high activity of inflammasome can enhance the release of inflammatory cytokines generating the characteristic cytokine storm associated with COVID-19.
4. Role of mitochondria in antiviral response
Mitochondrial dysfunction affects the activity of the immune system against virus infection. The release of interferon alpha (IFNα) and β (IFN type I (IFN-I)) is essential for antiviral response since these cytokines limit viral replication, improve antigen presentation and promote antigen-specific T and B response. Mitochondrial antiviral signaling (MAVS) participates in the expression of IFN-I (Akiyama et al., 2014). MAVS is a 56 kDa protein located on the outer membrane of mitochondria, peroxisomes and mitochondria associated membrane (MAMs) in endoplasmic reticulum. MAVS aggregates following retinoic acid-inducible gene (RIG-I) and melanoma differentiation associated factor 5 (MDA-5) binding (McGuire, 2019) and serves as scaffold to recruit TNF receptor associated factors (TRAFs). This process results in the phosphorylation of interferon-regulatory factors (IRFs) and its translocation to the nucleus to induce the synthesis of IFN (Lazear et al., 2013).
The activity of MAVS depends upon intact mitochondrial membrane potential and OXPHOS activity (Koshiba et al., 2011) suggesting that damaged mitochondria impair MAVS activity. However, if this relationship is associated with the deficient response to viral infections during aging, especially to respiratory infections (Haq and McElhaney, 2014), remains to be clarified, although some studies suggest a direct relationship. Silencing MAVS expression through RNA interference abolishes the activation of NF-κB and IRF3 in response to viral infection, permitting viral replication (Seth et al., 2005). Further, aged monocytes showing impaired mitochondrial respiratory capacity (Pence and Yarbro, 2018), suffer a decrease in the activation of IRF3 and 7 suggesting a link between mitochondrial dysfunction, MAVS malfunction and decrease of IFN-I release (Molony et al., 2017). Recently, MAVS-dependent signaling has been revised in deep in the immune response against emerging RNA viruses such as Ebola, Zika, Influenza A virus or SARS-CoV converting mitochondria as central hub of the response against these and other viruses (Dutta et al., 2020).
Another important process to be taken into consideration is that infection by virus also promotes mtROS increase. As has been demonstrated in the previous section, mtROS drive innate immune inflammation that exacerbates viral pathogenesis (To et al., 2020). This can be accompanied by the fact that many viruses produce modifications in mitochondrial activity that reduce the capacity of cells to induce IFN-I-dependent antiviral responses (Anand and Tikoo, 2013). Further, viruses show increased replication efficiency in senescent cells with low mitochondrial capacity, suggesting that accumulation of senescent cells, containing high levels of dysfunctional mitochondria, during aging and age-related diseases may promote the progression of virus infection (Malavolta et al., 2020). In the case of COVID-19, antivirus response of the immune system is compromised by a low release of type I and III IFN accompanied by high expression of proinflammatory cytokines (Blanco-Melo et al., 2020). This unbalanced response could be associated with the accumulation of damaged mitochondria in aging and obesity that strongly affect antiviral immunological response (Tavernarakis, 2020).
T lymphocytes are key in the protection against virus infections. The activation of lymphocytes also depends on the mitochondrial activity. After T-cell receptor stimulation, substantial changes required for T-cell activation occurs in metabolism including mitochondrial OXPHOS activation (Tarasenko et al., 2017). T cell proliferation and activation require ATP produced by mitochondrial activity to promote the increase of glycolysis (van der Windt et al., 2013) and reprogrammation of the whole metabolism (Chang et al., 2013). This reprogrammation of the metabolism is essential to support TCR signaling and nucleic acid synthesis needed for the transition of quiescent T-cells to the proliferative state (Ron-Harel et al., 2016; Weinberg et al., 2015).
OXPHOS activity is also essential in the reactivation of memory CD8+ T-cells. These cells have high respiratory capacity and high mitochondrial mass that allow them to rapidly reactivate after recognition of cognate antigens (van der Windt et al., 2013). The decline of OXPHOS in these cells is manifested as impaired immune memory (Goronzy and Weyand, 2017) and loss of capacity to quickly respond to secondary infections.
It seems clear that mitochondrial activity is essential for the appropriate response of lymphocytes against virus infections and the accumulation of damaged mitochondria in these cells will impair defense against these infections.
5. Age-related coenzyme q depletion and immune system
Coenzyme Q10 (CoQ10) is an essential component of the mitochondrial OXPHOS and antioxidant in cell membranes and its depletion is associated with severe mitochondrial-diseases (Lopez-Lluch et al., 2010). It is known that levels of CoQ decrease in many tissues and organs during aging (Kalen et al., 1989). Taken into consideration the important role of mitochondria in immunosenescence and inflammaging, a reduction of CoQ10 levels in immune cells could impair the progression of immune system unbalance and increase the low grade inflammatory profile found in the elderly.
The decrease of CoQ levels during aging also affects thymus (Bliznakov et al., 1978), and mitochondrial activity of CoQ10 has been considered essential for the optimal function of the immune system (Folkers and Wolaniuk, 1985). Thus, we can postulate that this age-related CoQ10 decrease can be also partially responsible of the unbalance of the immune system found in immunosenescence and inflammaging. However, no studies have been performed to directly associate CoQ10 depletion and inflammaging or immunosenescence although indirect studies seems to indicate a direct relationship (Novoselova et al., 2009). CoQ10 supplementation improves the activity of the immune system in infections. CoQ10 as other natural occurring antioxidants such as α-tocopherol reduce the LPS-induced inflammatory response in mice in a model of acute inflammation by modulating the activation of the NF-κB signaling pathway (Novoselova et al., 2009). On the other hand, supplementation with CoQ10 reduces the levels of the proinflammatory cytokine IL17 and the levels of anti-inflammatory cytokines in a mice model of colitis (Lee et al., 2017). The effect was associated with the activation of the AMPK and FOXP3 regulatory pathways involved in mitochondrial turnover. The same anti-inflammatory response was found in graft vs. host disease (Lee et al., 2016). Further, in a model of carrageenan-induced inflammation in the air pouch, CoQ10, in combination with the known immunomodulator β-glucan, reduced leukocyte infiltration in lung, nitrite and proinflammatory cytokines IL1α and β levels (Vetvicka and Vetvickova, 2018). In humans, CoQ10 supplementation significantly lower inflammation markers in individuals with metabolic syndrome (Dludla et al., 2020) or with diabetic nephropathy (Heidari et al., 2018). CoQ10 supplementation also reduces the proinflammatory marker, β-defensin 2 (hBD2) indicating a reduction in inflammatory status associate with diabetes (Brauner et al., 2014). On the other hand, a recent meta-analysis has demonstrated that CoQ10 supplementation increases antioxidant defences although did not affect inflammatory cytokines in patients with coronary artery disease (Jorat et al., 2019). On the other hand, another systematic review and meta-analysis about the effect of CoQ10 in markers of inflammation involving nine trials and 428 subjects, demonstrated that CoQ10 supplementation significantly decreased TNFα although, in this case, did not affected IL6 levels (Zhai et al., 2017). Probably these discrepancies are mainly due to the type of disease and the supplementation procedures and further studies and clinical trials are needed to clarify the role of CoQ10 supplementation in chronic inflammation.
We have also found that CoQ10 primary deficiency produces a change in the transcriptomic profile in cells, that acquire a defensive phenotype (Fernandez-Ayala et al., 2013). Among the gene ontology pathways repressed in primary CoQ10 deficiency, many immune genes and immunity-related gene ontology pathways were found downregulated, like the cellular signaling mechanisms that respond to IFNα and, in general, to IFN-I. On the other hand, the mechanisms repressed were those associated with negative regulation of viral genome replication and viral reproduction, cytokine mediated signaling pathways and other pathways in immune system response (Fernandez-Ayala et al., 2013). All these pathways indicate a key role of CoQ10 in antiviral defense and have been associated with immunosenescence and with the severity of COVID-19.
All these studies indicate the importance of CoQ10 in the activity of mitochondria and their effect on the immune system and on inflammation. For this reason, reduction of the levels of CoQ10 in leukocytes during aging could be considered a key factor in age-related chronic inflammation and an important factor for the severity of COVID-19. The study of the relationship of CoQ10 levels in leukocytes and COVID-19 severity and even other respiratory diseases such as flu could clarify this hypothesis and provide clues to use CoQ10 as supplement to improve immune system activity in the elderly.
6. COVID-19, cytokine storm and interferon signal
The lethality of COVID-19 depends of a systemic hyper-inflammation known as cytokine storm or macrophage activation syndrome (MAS) (Bektas et al., 2020). The worst prognosis in the disease caused by SARS-CoV-2 virus occurs in patients with adult respiratory distress syndrome (ARDS) (Grasselli et al., 2020). Although with some differences with the ARDS produced by SARS-CoV, an extremely powerful inflammation during the latter phase of the disease is the main cause of ARDS-mediated death (Grasselli et al., 2020) in COVID-19 as it was in the case of SARS (de Wit et al., 2016). In cytokine storm, immune and non-immune cells release large amounts of proinflammatory cytokines that generates a great distortion in the immune response. To date, the proinflammatory activity of lung macrophages seems to be the main responsible of the trigger of cytokine storm in patients of COVID-19 (Pagliaro, 2020). Furthermore, the enormous release of proinflammatory cytokines is responsible of the exaggerated internal inflammatory processes suffered by survivors of the acute phase of the disease (Bektas et al., 2020).
A persistent inflammation caused by over-activation of the innate immune system leads to cytokine storm and ARDS (Teijaro et al., 2014). In the case of COVID-19, blood plasma shows significant high levels of proinflammatory cytokines such as IL6, IL1β, IL1RA, IL7, IL8, TNFα, several chemokines such as CXCL9 and CXCL16, chemoattractant for T and NK cells, CCL8 and CCL4, which recruit monocytes/macrophages, and CXCL8, a classic neutrophil chemoattractant (Proudfoot, 2002). IL2, IL7, IL10, TFNα, Granulocyte colony-stimulating factor (GCSF), and the chemokines Interferon gamma-induced protein 10 (IP10, CXCL10), Monocyte chemoattractant protein 1 (MCP1, CCL2), Macrophage Inflammatory Proteins (MIP1α, CCL3) have been associated with the patients that need intensive care, indicating the relationship of inflammatory cytokines and chemoattractants of monocytes and neutrophils with the severity of the disease (Rothan and Byrareddy, 2020).
Another feature of COVID-19 severity is a defective IFN-I response. Analysis performed in postmortem samples from COVID-19 positive patients, show enrichment in innate and humoral responses, but no of antiviral type I and III interferons (IFN-I or IFN-III) (Blanco-Melo et al., 2020). In vitro studies have demonstrated that replication of the virus is sensible to IFN-I since treatment of cells with IFNβ resulted in a dramatic reduction of virus title (Blanco-Melo et al., 2020) and a recent clinical study indicates that treatment with IFNβ1 can reduce the mortality in severe COVID-19 patients (Rahmani et al., 2020). Lower activity of IFN-I also correlates with the increase in the recruitment of inflammatory monocyte-macrophage populations, one of the hallmarks of the infection with SARS-CoV (Channappanavar et al., 2016) and probably associated with the cytokine storm involved in COVID-19. In fact, a study carried out in different COVID-19 patients from different countries found a common denominator in a high proinflammatory signal from myeloid cells together with a low production of IFNα from dendritic cells (Arunachalam et al., 2020). In general, we can then consider that SARS-CoV-2 infection produces a unique and inappropriate inflammatory response characterized by the release of low levels of the anti-viral type I and III interferons (IFN-I and III) and high levels of CXCLs and IL6 (Blanco-Melo et al., 2020).
For coronaviruses, the key pattern recognition in macrophages depends on TLR7, which is activated by viral RNA in endosomes (Cervantes-Barragan et al., 2007). Activation of TLR7 is needed for the production of INFα (IFN-I), IL12 and IL6. This response is required for the production and activation of CD8+ T-cells, CD4+ and B lymphocytes (Zhou et al., 2017). Another pathways involved in this response the RLRs, MDA-5 and laboratory of genetics and physiology 2 (LGP2), which recognize cytosolic viral RNA (Zust et al., 2011) and the cGAS-STING pathway, which recognizes cytosolic DNA (Sun et al., 2013).
Activation of all these pathways induce a signaling cascade leading to the expression of IFN-I and other inflammatory cytokines. However, in the case of infection with coronaviruses, some papain-like proteases block TLR7 (Li et al., 2016) and STING-dependent signaling (Sun et al., 2012) probably downregulating the activation of IRFs and blocking the early transport of IRFs to the nucleus (Roth-Cross et al., 2007; Spiegel et al., 2005). The importance of the TLR7 signal pathway has been very recently highlighted by the fact that several young patients suffering severed COVID-19 and death showed loss-of-function variants of the TLR7 gene affecting downstream IFN-I signaling (van der Made et al., 2020). The same relationship has been found in patients with TLR3 and IRF7 loss-of function mutations partially explaining the great differences in the response to the virus (Zhang et al., 2020). To this scenario, we have also to add the production of autoantibodies against different forms of IFN-I in some of the more severe COVID-19 patients (Bastard et al., 2020) indicating the importance of a correct IFN response in survival to this disease.
Importantly, cytotoxic CD8+ T-cells requires mitochondrial-mediated IFN-I signaling for optimal protection. Activated naïve CD8+ T cells require the increase of mitochondrial mass and activation of mitochondria to produce IL2 and TNFα, IFNγ (IFN-II) and other mediators (Fischer et al., 2018). In a model of viral choriomeningitis infection in mice, the mitochondrial cyclophilin D was essential to produce IFN-I and maintain survival of CD8+ T-cells (Condotta et al., 2020). On the other hand, high IL6 levels inhibit CD8+ cytotoxic T-cells by blocking the secretion of IFNγ (Ahmadpoor and Rostaing, 2020), paralyzing yet more the cell-mediated antiviral response during cytokine storm (Velazquez-Salinas et al., 2019).
7. COVID-19-severity risk factors are associated with mitochondrial dysfunction
Accumulation of abnormal mitochondria contributes to aging and age-related and metabolic diseases, including metabolic syndrome, cancer, neurodegenerative disease, type II diabetes, etc. (Lopez-Lluch, 2017b). COVID-19 severity is linked to several previous pathological situations mainly, hypertension, diabetes and coronary heart disease, lung diseases, metabolic syndrome and obesity, and, importantly, older age (Engin et al., 2020; Zaki et al., 2020; F. Zhou et al., 2020). In all these diseases, mitochondrial dysfunction plays a very important role (Hernandez-Camacho et al., 2018; Lopez-Lluch, 2017b; Lopez-Lluch et al., 2018). Moreover, most of them are associated with a pro-inflammatory profile in which mitochondrial dysfunction plays an essential role (Guerrero-Ros et al., 2020; Maddaloni and Buzzetti, 2020; McMaster et al., 2015; Prasun, 2020). The direct relationship between mitochondrial dysfunction and comorbidities associated with COVID-19 severity helps to understand the importance of maintaining mitochondrial health in the progression and severity of COVID-19. Following, we briefly summarize the relationship of these comorbidities with mitochondria and COVID-19 progression.
7.1. Diabetes
Although the relationship between diabetes mellitus and severe complications in COVID-19 are not completely clear, levels of proinflammatory cytokines in plasma in this disease have been associated with the severity of COVID-19 infection (Roy et al., 2020). The phagocytic activity of professional phagocytic cells including macrophages and the innate cell-mediated immunity is impaired in diabetic patients (Ma and Holt, 2020). Diabetic patients also show elevated proinflammatory cytokines levels such as IL1β, IL6 and TNFα, the same cytokines induced by mitochondrial dysfunction (Geerlings and Hoepelman, 1999; Maddaloni and Buzzetti, 2020; Pal and Bhansali, 2020). In these patients, mtROS induces lysosomal dysfunction that impairs autophagic flux and contributes to M1 macrophage polarization to the inflammatory phenotype (Yuan et al., 2019). It seems clear that, mitochondrial dysfunction in diabetic patients contributes importantly to the low-grade inflammatory profile associated with this disease that is aggravated during aging and has been associated with higher severity in COVID-19 infection.
7.2. Obesity
One of the main risk factors in COVID-19 disease is obesity (Kwok et al., 2020). This relationship has been associated with a proinflammatory profile and metabolic dysregulation causing severe symptoms and complications such as hypercoagulopathy (Pasquarelli-do-Nascimento et al., 2020). Regarding mitochondrial activity, lipid challenge in obesity causes dysregulation of the immune response. High levels of fatty acids inhibit the activity of CD4+ T-cells in an effect linked to the malfunction of autophagosome formation and mitochondrial degradation (Guerrero-Ros et al., 2020). This disruption blocks mitochondrial turnover accompanied by the increase in mitochondrial ROS and decrease in ATP levels as hallmarks of mitochondrial dysfunction (Guerrero-Ros et al., 2020). Other studies have demonstrated that alterations in lipids and high-fat loading interfere in autophagic process and contribute to the accumulation of damaged mitochondria (Koga et al., 2010; Rodriguez-Navarro et al., 2012).
7.3. Hypertension
Hypertension is another main risk factors in COVID-19 infection. In this relationship, inflammation also contributes importantly to the pathogenesis since inflammation impairs hypertension control (Mischel et al., 2015) and hypertension induces the release of inflammatory cytokines in vascular tissues (McMaster et al., 2015). Proinflammatory cytokines such as IL17, IL6, TNFα and IFNγ, are found in high levels in hypertensive patients (McMaster et al., 2015). In this process, mitochondrial dysfunction plays also a very active role. Mitochondrial dysfunction in macrophage population strongly contributes to the M1/M2 unbalance favoring the activity of pro-inflammatory M1 macrophages. Hypertension associated with obesity and other metabolic factors induces the unbalance of macrophage population increasing the amount and activity of M1 and decreasing the function of the anti-inflammatory and respiratory M2 macrophages (Mouton et al., 2020).
On the other hand, improvement of mitochondrial activity reduces inflammation associated with hypertension. Dihydrofolate reductase KO mice suffers hypertension and abdominal aortic aneurism after infusion with angiotensin II. In these mice, scavenging of mtROS completely abrogates the development of hypertension and aneurism indicating the importance of mitochondrial dysfunction in these processes (Li et al., 2019). Moreover, mitochondrial Sirt3 depletion induces vascular inflammation and oxidative stress in hypertension whereas its overexpression reduces vascular dysfunction (Dikalova et al., 2020). Further, pharmacological restoration of mitophagy reduces hypertension-related stroke occurrence (Forte et al., 2019). All these studies indicate the importance of mitochondrial activity in the control of hypertension and probably in the response to COVID-19.
7.4. Metabolic syndrome
Severity of COVID-19 increases also in patients suffering metabolic syndrome (MS) (Bansal et al., 2020). MS is considered as the co-occurrence of obesity, dyslipidemia and hypertension, main factors for COVID-19 severity (Zaki et al., 2020). Obesity and MS patients show a very compromised immune system (Andersen et al., 2016), even affecting lymphocytes populations (Rodríguez et al., 2018), that decrease the response of the organism to vaccines such as influenza and other viral infections (Frasca and Blomberg, 2020).
In MS, mitochondrial dysfunction contributes very importantly to oxidative stress and systemic inflammation (Prasun, 2020). Mitochondrial abnormalities are also associated with cardiac contractile dysfunction occurring in obesity, type 2 diabetes and insulin resistance associated with MS (Bugger and Abel, 2008). In this relationship, mitochondrial stress and unfolded protein response (mtUPR) play a key role (Hu and Liu, 2011). In fact, the use of therapies focussed to improve the capacity of mitochondria to reduce stress damage and decrease mtUPR are considered an effective approach to avoid mitochondrial dysfunction associated with many of the physiological disorders associated with MS (Hu and Liu, 2011). Further, as strategy for the treatment of MS, enhancing mitochondrial biogenesis through pharmacological and non-pharmacological approaches and increase of antioxidant capacity has been recently proposed (Prasun, 2020).
7.5. Cardiovascular disease
Another of the main groups in risk for suffering severe COVID-19 is composed of individuals suffering cardiovascular disease (CVD) (Nishiga et al., 2020). CVD is strongly aggravated by MS, hypertension, obesity and diabetes; therefore, all the indications above indicated for these diseases can be applied also to CVD. Hypertrophy, fibrosis and inflammation are common denominators of aged cardiovascular system, and all of them are accompanied by the accumulation of dysfunctional mitochondria due to mito/autophagy decline among other factors (Morciano et al., 2020). Accumulation of dysfunctional mitochondria in CVD by a shift or mitochondrial dynamics to fission indicates the importance of a right mitochondrial equilibrium in the progression of this disease (Cooper et al., 2020). On the other hand, induction of Peroxisome Proliferator-Activated Receptor Gamma Coactivator-1a (PGC1a), a widely known transcription factor that induces mitochondrial biogenesis and turnover, protects heart against oxidative stress, microcirculation and mitochondrial dysfunction (Arad et al., 2020).
Again, the relationship between inflammation and mitochondrial dysfunction seems to play an essential role in the progression and severity of CVD in elderly people (Ferrucci and Fabbri, 2018). Therapeutic agents and interventions targeting mitochondria decreasing mtROS production have been extensively studied improving the progression of CVD (Veloso et al., 2019). Cardioprotective signaling pathways seems to converge on mitochondria especially to prevent inflammatory injury associated with CVD (Koenitzer and Freeman, 2010). Further, mitochondrion-targeted antioxidants show cardioprotective effects linked to reduction of NF-κB signaling and reduction of the expression of inflammatory cytokines (Liu et al., 2019). Supplementation with Coenzyme Q10 plus selenium reduces the mortality by approximately 50% in patients with CVD in an effect associated to its bioenergetics, antioxidant and anti-inflammatory function (Mantle and Hargreaves, 2019).
7.6. Lung diseases
Among the diseases associated with COVID19 severity, lung diseases are the most important since massive bilateral pneumonia is one of the main symptoms of this disease (P. Zhou et al., 2020). Mitochondrial dysfunction has been also associated with many of the lung diseases such as ARDS, pneumonia, chronic lung diseases, or bronchial asthma among others (Ten and Ratner, 2020). Lung physiology is also affected during aging. Age-associated chronic obstructive pulmonary disease (COPD) shows many of the hallmarks of aging including impaired autophagy/mitophagy, accumulation of damaged mitochondria and low grade chronic inflammation (Barnes, 2019). In lung dysfunction, peripheral blood mononuclear cells (PBMCs) and platelets actively participate in inflammation contributing to the severity and lethality of COVID-19 (Riou et al., 2020).
Removal of dysfunctional mitochondria has been considered a key therapeutic process for the treatment of respiratory diseases (Cloonan and Choi, 2016). Activation of autophagy improves the initiation and the progression of lung diseases during aging reducing mitochondrial dysfunction and associated inflammation (Nakahira et al., 2014). However, other studies have indicated that autophagic clearance of mitochondria may aggravate the pathogenesis of COPD by increasing cell death (Ryter and Choi, 2015). Interestingly, resveratrol (RSV), a polyphenol used in aging studies that promotes mitochondrial biogenesis and turnover (Baur et al., 2006), and also shows anti-inflammatory properties (Tung et al., 2015), has been recently considered as putative candidate to counteract lung and muscle impairments associated with COPD (Beijers et al., 2018). Furthermore, CR, the strongest non-genetic intervention in longevity, reverses several age-related changes in lung by reducing inflammation and improving mitochondrial turnover (Hegab et al., 2019). Clearly, removal of damaged mitochondria plays, again, a key protective role in the progression of respiratory diseases associated with aging and risk factor for COVID-19.
8. Age-related immune system distortion can be prevented by prolongevity factors
Prolongevity interventions can modulate the evolution of immunosenescence and inflammation in organisms (Mau et al., 2020). The most powerful interventions to delay aging are CR (Lopez-Lluch and Navas, 2016), physical exercise (Ciolac et al., 2020) and some dietary bioactive compounds or therapies based on the induction of mito/autophagy (Lopez-Lluch et al., 2018) and on maintenance of NAD+ (Braidy and Liu, 2020). Caloric restriction (CR), delays T-cell senescence in non-human primates indicating its capacity to affect immunosenescence (Messaoudi et al., 2006). Exercise improves immunological and anti-inflammatory response decreasing morbidity and mortality (Turner, 2016). Prolongevity nutritional compounds such as polyphenols improve the immune function and reduce proinflammatory profiles in many model animals (Olcum et al., 2020; Tung et al., 2015).
The research about the influence of CR in humans is very complex and has not produced clear results to date, however, studies performed in aged long-lived nonhuman primates have demonstrated that CR delays T-cell senescence (Messaoudi et al., 2006). Other studies in mice have demonstrated that CR improves CD4+ and CD8+ T-cells activity in the spleen, mesenteric lymph nodes, peripheral blood, thymus and salivary glands (Jolly, 2004). In mice, CR prevents thymic deterioration and improves the response of T-cells to IL-2 (Yang et al., 2009). Taken into consideration the importance of bioenergetics in the activity of T-cells (Choi et al., 2017) and the effect of CR on mitochondrial activity and turnover (Lopez-Lluch et al., 2006), the maintenance of mitochondrial activity in the immune system can be considered one of the main effect by which CR delays immunosenescence.
Exercise, a well-known anti-aging factor, also improves immunological and anti-inflammatory response (Turner, 2016). Physical activity in old mice (J.Y. Lee et al., 2019) and in humans (Sellami et al., 2018) improves immunological response. Even in old patients showing cognitive dysfunction, aerobic exercise reduces the levels of the most significant age-related inflammatory cytokines, IL6 and TNFα (Stigger et al., 2019). Part of the positive effect of exercise can be due to a decrease of the amount of dysfunctional mitochondria by inducing mitochondrial biogenesis (Lumini et al., 2008). Further, the immunomodulatory effect of exercise can be also associated with the stimulation of the synthesis of CoQ10 improving both, mitochondrial and antioxidant activities (Rodriguez-Bies et al., 2015; Rodriguez-Bies et al., 2010).
Bioactive compounds have shown positive effects of the immune function in many different models of immunological dysfunction such as ovariectomized mice (Baeza et al., 2010), normal aging (Yuan et al., 2012) or in senescence accelerated mice (Zhang et al., 2012). In humans, the studies with natural extracts rich in bioactive compounds have shown conflicting results. In some experiments, extracts rich in bioactive compounds have demonstrated capacity to activate immune cells (Tumova et al., 2017), although with other extracts, no improvement was found (Hunter et al., 2012). On the other hand, high flavonoid cocoa supplements have recently demonstrated their capacity to reduce oxidative stress in plasma and inflammation in older subjects (Munguia et al., 2019). Mediterranean diet, rich in plant foods, is associated with reduced risk of developing age-related chronic diseases by inducing protection against oxidative stress and improving mitochondrial activity that could be the cause of a reduced inflammation level (Tosti et al., 2018).
Among the most studied bioactive compounds, polyphenols have demonstrated their capacity to modulate mitochondrial physiology and reduce mitochondrial dysfunction (Baur et al., 2006; Tung et al., 2014). Polyphenols are nowadays considered potential antiaging agents because of their ability to modulate oxidative damage, inflammation, cell senescence and autophagy (Russo et al., 2020). RSV is not only useful reducing the levels of TNFα in aged animals (Tung et al., 2015), it also reduces the effect of TFNα in an in vitro model of uveitis in which reduces the response to TNFα and the release of proinflammatory cytokines (Paladino et al., 2020). A recent revision about the effect of different compounds on chronic low grade inflammation in humans and model animals indicated that RSV and metformin, a widely used pharmacological compound for T2DM treatment, show anti-inflammatory capacity and can be considered promising therapies for inflammation and frailty in the elderly (Chen et al., 2019). Interestingly, metformin also inhibits the expression of NF-kB gene reducing the inflammatory response (Kanigur Sultuybek et al., 2019) through normalizing mitochondrial function through enhancing mitochondrial clearance by mito/autophagy (Bharath et al., 2020). In fact, metformin, has been recently proposed as pharmacological compound against SARS-CoV-2 infection through its anti-inflammatory property by modulating mitochondrial ROS/Ca+ release (Menendez, 2020) and by regulating mTOR/AMPK signaling involved in mitochondrial turnover (Ramaiah, 2020). Another compound used as supplement that modulates several aspects of metabolism and mitochondrial mechanisms is CoQ10. Low levels of plasma CoQ10 have been associated with inflammation and mortality in CVD patients (Shimizu et al., 2017). Furthermore, supplementation with CoQ10 can prevent the processes related to chronic oxidative stress and inflammation during aging (Lopez-Lluch, 2020; Lopez-Moreno et al., 2018). Many in vitro and clinical studies indicate that CoQ10 is a promising anti-inflammatory molecule in metabolic and age-related diseases (Schmelzer et al., 2010; Shimizu et al., 2017; Suárez-Rivero et al., 2019; Yubero-Serrano et al., 2012) although the variability in the bioavailability of this compound in humans can affect the conclusions in different clinical studies (Lopez-Lluch et al., 2019).
Maintenance of NAD+ levels is important in aging (Braidy and Liu, 2020). Dysregulation of mitochondrial activity can affect NAD+ levels (Chinopoulos, 2020) and introduce a new important factor in aging and the activity of the immune system (Audrito et al., 2020). Decline in NAD+ has been reported in degenerative diseases associated with oxidative stress and inflammation (Braidy and Liu, 2020). Therefore, supplementation with NAD+ has been suggested as therapy for age-associated disorders reactivating defective DNA repair and mitophagy, thus, avoiding accumulation of dysfunctional mitochondria (Babbar et al., 2020). Decrease of the levels of the NAD+ precursor, nicotinamide mononucleotide (NMN) has been also associated with a proinflammatory profile in neurodegenerative diseases (Yao et al., 2017). This proinflammatory profile can be reversed by chronic supplementation with nicotinamide (NAM), and other precursors of NAD+, that improve bioenergetics and reduce inflammation in a dietary stress model in mice (Braidy and Liu, 2020; Mitchell et al., 2019). On the other hand, treatment with NMN improves cardiac muscle contractility and protects against inflammation in mice suffering cardiomyopathies (Zhang et al., 2017) by improving mitochondrial function and reducing IL6 and TNFα levels in many tissues (Sims et al., 2018).
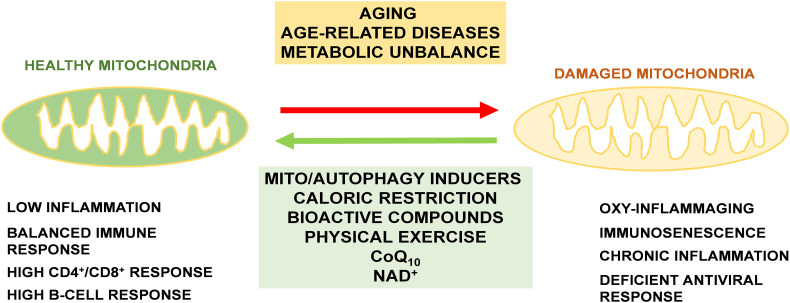
In general, it seems clear that many prolongevity effectors improve immune system in the elderly by regulating mitochondrial function and inflammation level. Taken into consideration the importance of bioenergetics, mitochondrial activity and antioxidant protection in the immune response, we can speculate that maintenance of mitochondrial homeostasis by bioactive compounds or treatments that activate mitochondrial physiology, dynamics, removal and biosynthesis can be effective in the decrease of the inflammatory profile and in the immunosenescence associated with aging and age-associated diseases (Fig. 3 ). These treatments are shown as promising therapies to improve mitochondrial function and reduce inflammation in the elderly, and would protect them against viral respiratory infections that are more severe when patients show previous inflammatory conditions.
Fig. 3.
Essential role of mitochondrial health maintenance in the immune response. Aging and age-related diseases, including many metabolic diseases, aggravate mitochondrial dysfunction and the accumulation of damaged mitochondria in cells including immune cells. The use of prolongevity procedures such as CR, bioactive compounds or physical exercise can reverse this accumulation by inducing mitochondrial turnover and biogenesis. Among these compounds, mito/autophagy inducers, CoQ10 and NAD+ supplementation can help to maintain high levels or healthy mitochondria during aging and avoid immunological unbalance associated with COVID-19 severity.
9. Conclusions
SARS-CoV-2 causes a complex and rapid response that collapse lungs through the induction of a cytokine storm. The disease produced by SARS-CoV-2, COVID-19, is highly severe in old patients and in persons showing previous risk factors that are related with age-dependent chronic diseases such as CVD, obesity, T2DM, MS or lung diseases. Elderly people and patients suffering these chronic diseases show a low level of chronic inflammation that is associated with the dysfunction of mitochondria (Parker et al., 2019). Induction of defective mitochondria removal through activation of the mito/autophagy process reduces the inflammatory profile (Raz et al., 2017) and would improve the response to new pathogens such as SARS-CoV-2 even in old people.
We postulate here that the maintenance of mitochondrial health through healthy life habits, pharmacological compounds o dietary supplements able to improve mitochondrial activity, dynamics and turnover would reduce the levels of inflammatory cytokines and the severity of COVID-19 and other respiratory diseases such as seasonal flu. These and other “geroprotective” treatments must be used to treat or prevent COVID-19 (Promislow, 2020). Interventions and compounds able to improve mitochondrial health in the elderly would reduce or even eliminate the cytokine storm induced by SARS-CoV-2 infection and, therefore, reduce the grade of ARDS associated with COVID-19. No existing effective treatment against COVID-19 once cytokine storm and ARDS has started, prevention of the stronger effect induced by SARS-CoV-2 appears the most promising strategy. We think this strategy must be based in the improvement of the mitochondrial activity that would reduce the proinflammatory profile associated with aging and age-related diseases.
Declarations
Funding
No funding was required for this article.
Conflicts of interest/competing interests
Authors declare no conflicts of interest.
Ethics approval
No applicable.
Consent to participate
No applicable.
Consent for publication
No applicable.
Availability of data and material
No applicable.
Code availability
No applicable.
Authors' contributions
All the authors have actively participated in the search of literature search, writing and revision of this manuscript and approval of the final version.
Section Editor: Werner Zwerschke
References
- Agostinho P., Cunha R.A., Oliveira C. Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer's disease. Curr. Pharm. Des. 2010;16:2766–2778. doi: 10.2174/138161210793176572. [DOI] [PubMed] [Google Scholar]
- Ahmadpoor P., Rostaing L. Why the immune system fails to mount an adaptive immune response to a COVID-19 infection. Transplant Int. Off. J. Eur. Soc. Organ Transplant. 2020 doi: 10.1111/tri.13611. [DOI] [PubMed] [Google Scholar]
- Akbar A.N., Gilroy D.W. Aging immunity may exacerbate COVID-19. Science. 2020;369:256–257. doi: 10.1126/science.abb0762. [DOI] [PubMed] [Google Scholar]
- Akiyama T. Mitochondria-nucleus shuttling FK506-binding protein 51 interacts with TRAF proteins and facilitates the RIG-I-like receptor-mediated expression of type I IFN. PLoS One. 2014;9 doi: 10.1371/journal.pone.0095992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anand S.K., Tikoo S.K. Viruses as modulators of mitochondrial functions. Adv. Virol. 2013;2013 doi: 10.1155/2013/738794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen C.J., Murphy K.E., Fernandez M.L. Impact of obesity and metabolic syndrome on immunity. Adv. Nutr. 2016;7:66–75. doi: 10.3945/an.115.010207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Annweiler C. National French survey of COVID-19 symptoms in people aged 70 and over. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arad M. Therapeutic approaches to diabetic cardiomyopathy: targeting the antioxidant pathway. Prostaglandins Other Lipid Mediators. 2020 doi: 10.1016/j.prostaglandins.2020.106454. [DOI] [PubMed] [Google Scholar]
- Arunachalam P.S. Systems biological assessment of immunity to mild versus severe COVID-19 infection in humans. Science. 2020 doi: 10.1126/science.abc6261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Audrito V., Messana V.G., Deaglio S. NAMPT and NAPRT: two metabolic enzymes with key roles in inflammation. Front. Oncol. 2020;10:358. doi: 10.3389/fonc.2020.00358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babbar M. Mitophagy and DNA damage signaling in human aging. Mech. Ageing Dev. 2020;186 doi: 10.1016/j.mad.2020.111207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baeza I. Soybean and green tea polyphenols improve immune function and redox status in very old ovariectomized mice. Rejuvenation Res. 2010;13:665–674. doi: 10.1089/rej.2010.1049. [DOI] [PubMed] [Google Scholar]
- Bansal R., Gubbi S., Muniyappa R. Metabolic syndrome and COVID 19: endocrine-immune-vascular interactions shapes clinical course. Endocrinology. 2020 doi: 10.1210/endocr/bqaa112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes P.J. Pulmonary diseases and ageing. Subcell. Biochem. 2019;91:45–74. doi: 10.1007/978-981-13-3681-2_3. [DOI] [PubMed] [Google Scholar]
- Bastard P. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020;370 doi: 10.1126/science.abd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baur J.A. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444:337–342. doi: 10.1038/nature05354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beijers R., Gosker H.R., Schols A. Resveratrol for patients with chronic obstructive pulmonary disease: hype or hope? Curr. Opin. Clin. Nutr. Metab. Care. 2018;21:138–144. doi: 10.1097/mco.0000000000000444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bektas A. Age-associated changes in human CD4(+) T cells point to mitochondrial dysfunction consequent to impaired autophagy. Aging (Albany NY) 2019;11:9234–9263. doi: 10.18632/aging.102438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bektas A. A public health perspective of aging: do hyper-inflammatory syndromes such as COVID-19, SARS, ARDS, cytokine storm syndrome, and post-ICU syndrome accelerate short- and long-term inflammaging? Immun. Ageing. 2020;17:23. doi: 10.1186/s12979-020-00196-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bharath L.P. Metformin enhances autophagy and normalizes mitochondrial function to alleviate aging-associated inflammation. Cell Metab. 2020;32:44–55. doi: 10.1016/j.cmet.2020.04.015. e46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanco-Melo D. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020 doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bliznakov E.G. Coenzyme Q deficiency in aged mice. J. Med. 1978;9:337–346. [PubMed] [Google Scholar]
- Braidy N., Liu Y. NAD+ therapy in age-related degenerative disorders: a benefit/risk analysis. Exp. Gerontol. 2020;132 doi: 10.1016/j.exger.2020.110831. [DOI] [PubMed] [Google Scholar]
- Brauner H. Markers of innate immune activity in patients with type 1 and type 2 diabetes mellitus and the effect of the anti-oxidant coenzyme Q10 on inflammatory activity. Clin. Exp. Immunol. 2014;177:478–482. doi: 10.1111/cei.12316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunelle J.K. Oxygen sensing requires mitochondrial ROS but not oxidative phosphorylation. Cell Metab. 2005;1:409–414. doi: 10.1016/j.cmet.2005.05.002. [DOI] [PubMed] [Google Scholar]
- Bugger H., Abel E.D. Molecular mechanisms for myocardial mitochondrial dysfunction in the metabolic syndrome. Clin. Sci. (Lond.) 2008;114:195–210. doi: 10.1042/CS20070166. [DOI] [PubMed] [Google Scholar]
- Caruso C. Mechanisms of immunosenescence. Immun. Ageing. 2009;6:10. doi: 10.1186/1742-4933-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cervantes-Barragan L. Control of coronavirus infection through plasmacytoid dendritic-cell-derived type I interferon. Blood. 2007;109:1131–1137. doi: 10.1182/blood-2006-05-023770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakravarthy M.V., Neuschwander-Tetri B.A. The metabolic basis of nonalcoholic steatohepatitis. Endocrinol. Diabetes Metab. 2020;3 doi: 10.1002/edm2.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang C.H. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell. 2013;153:1239–1251. doi: 10.1016/j.cell.2013.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Channappanavar R. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice. Cell Host Microbe. 2016;19:181–193. doi: 10.1016/j.chom.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., Liu S., Leng S.X. Chronic low-grade inflammatory phenotype (CLIP) and senescent immune dysregulation. Clin. Ther. 2019;41:400–409. doi: 10.1016/j.clinthera.2019.02.001. [DOI] [PubMed] [Google Scholar]
- Chen J., Kelley W.J., Goldstein D.R. Role of aging and the immune response to respiratory viral infections: potential implications for COVID-19. J. Immunol. 2020;205:313–320. doi: 10.4049/jimmunol.2000380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinopoulos C. Acute sources of mitochondrial NAD(+) during respiratory chain dysfunction. Exp. Neurol. 2020;327 doi: 10.1016/j.expneurol.2020.113218. [DOI] [PubMed] [Google Scholar]
- Choi I.Y., Lee C., Longo V.D. Nutrition and fasting mimicking diets in the prevention and treatment of autoimmune diseases and immunosenescence. Mol. Cell. Endocrinol. 2017;455:4–12. doi: 10.1016/j.mce.2017.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciolac E.G., Rodrigues da Silva J.M., Vieira R.P. Physical exercise as an Immunomodulator of chronic diseases in aging. J. Phys. Act. Health. 2020:1–11. doi: 10.1123/jpah.2019-0237. [DOI] [PubMed] [Google Scholar]
- Cloonan S.M., Choi A.M. Mitochondria in lung disease. J. Clin. Invest. 2016;126:809–820. doi: 10.1172/jci81113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Condotta S.A. Cyclophilin D regulates antiviral CD8(+) T cell survival in a cell-extrinsic manner. ImmunoHorizons. 2020;4:217–230. doi: 10.4049/immunohorizons.2000016. [DOI] [PubMed] [Google Scholar]
- Cooper H.A. Targeting mitochondrial fission as a potential therapeutic for abdominal aortic aneurysm. Cardiovasc. Res. 2020 doi: 10.1093/cvr/cvaa133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Currie M.S. Immunosenescence. Compr. Ther. 1992;18:26–34. [PubMed] [Google Scholar]
- Dantzer R. From inflammation to sickness and depression: when the immune system subjugates the brain. Nature Reviews. Neuroscience. 2008;9:46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De la Fuente M., Miquel J. An update of the oxidation-inflammation theory of aging: the involvement of the immune system in oxi-inflamm-aging. Curr. Pharm. Des. 2009;15:3003–3026. doi: 10.2174/138161209789058110. [DOI] [PubMed] [Google Scholar]
- De la Fuente M. Strategies to improve the functions and redox state of the immune system in aged subjects. Curr. Pharm. Des. 2011;17:3966–3993. doi: 10.2174/138161211798764861. [DOI] [PubMed] [Google Scholar]
- De Martinis M. Apoptosis remodeling in immunosenescence: implications for strategies to delay ageing. Curr. Med. Chem. 2007;14:1389–1397. doi: 10.2174/092986707780831122. [DOI] [PubMed] [Google Scholar]
- Desdín-Micó G. T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science. 2020 doi: 10.1126/science.aax0860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dikalova A.E. Mitochondrial deacetylase Sirt3 reduces vascular dysfunction and hypertension while Sirt3 depletion in essential hypertension is linked to vascular inflammation and oxidative stress. Circ. Res. 2020;126:439–452. doi: 10.1161/circresaha.119.315767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dludla P.V. Coenzyme Q(10) supplementation improves adipokine levels and alleviates inflammation and lipid peroxidation in conditions of metabolic syndrome: a meta-analysis of randomized controlled trials. Int. J. Mol. Sci. 2020;21 doi: 10.3390/ijms21093247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dutta S., Das N., Mukherjee P. Picking up a fight: fine tuning mitochondrial innate immune defenses against RNA viruses. Front. Microbiol. 2020;11:1990. doi: 10.3389/fmicb.2020.01990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engin A.B., Engin E.D., Engin A. Two important controversial risk factors in SARS-CoV-2 infection: obesity and smoking. Environ. Toxicol. Pharmacol. 2020 doi: 10.1016/j.etap.2020.103411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etard J.F. Potential lethal outbreak of coronavirus disease (COVID-19) among the elderly in retirement homes and long-term facilities, France, March 2020. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2020;25 doi: 10.2807/1560-7917.ES.2020.25.15.2000448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans T.D. Target acquired: selective autophagy in cardiometabolic disease. Sci. Signal. 2017;10 doi: 10.1126/scisignal.aag2298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Ayala D.J. Survival transcriptome in the coenzyme Q10 deficiency syndrome is acquired by epigenetic modifications: a modelling study for human coenzyme Q10 deficiencies. BMJ Open. 2013;3 doi: 10.1136/bmjopen-2012-002524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrucci L., Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018;15:505–522. doi: 10.1038/s41569-018-0064-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer M. Early effector maturation of naïve human CD8(+) T cells requires mitochondrial biogenesis. Eur. J. Immunol. 2018;48:1632–1643. doi: 10.1002/eji.201747443. [DOI] [PubMed] [Google Scholar]
- Folkers K., Wolaniuk A. Research on coenzyme Q10 in clinical medicine and in immunomodulation. Drugs Exp. Clin. Res. 1985;11:539–545. [PubMed] [Google Scholar]
- Forte M. Pharmacological restoration of autophagy reduces hypertension-related stroke occurrence. Autophagy. 2019:1–14. doi: 10.1080/15548627.2019.1687215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franceschi C., Campisi J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014;69(Suppl. 1):S4–S9. doi: 10.1093/gerona/glu057. [DOI] [PubMed] [Google Scholar]
- Franceschi C. Inflammaging and 'Garb-aging'. Trends in Endocrinology and Metabolism: TEM. 2017;28:199–212. doi: 10.1016/j.tem.2016.09.005. [DOI] [PubMed] [Google Scholar]
- Frasca D., Blomberg B.B. The impact of obesity and metabolic syndrome on vaccination success. Interdiscip. Top Gerontol. Geriatr. 2020;43:86–97. doi: 10.1159/000504440. [DOI] [PubMed] [Google Scholar]
- Garrido A. Oxidative-inflammatory stress in immune cells from adult mice with premature aging. Int. J. Mol. Sci. 2019;20 doi: 10.3390/ijms20030769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geerlings S.E., Hoepelman A.I. Immune dysfunction in patients with diabetes mellitus (DM) FEMS Immunol. Med. Microbiol. 1999;26:259–265. doi: 10.1111/j.1574-695X.1999.tb01397.x. [DOI] [PubMed] [Google Scholar]
- Goronzy J.J., Weyand C.M. Successful and maladaptive T cell aging. Immunity. 2017;46:364–378. doi: 10.1016/j.immuni.2017.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grasselli G. Pathophysiology of COVID-19-associated acute respiratory distress syndrome: a multicentre prospective observational study. Lancet Respir. Med. 2020 doi: 10.1016/s2213-2600(20)30370-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerrero-Ros I. The negative effect of lipid challenge on autophagy inhibits T cell responses. Autophagy. 2020;16:223–238. doi: 10.1080/15548627.2019.1606635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haq K., McElhaney J.E. Ageing and respiratory infections: the airway of ageing. Immunol. Lett. 2014;162:323–328. doi: 10.1016/j.imlet.2014.06.009. [DOI] [PubMed] [Google Scholar]
- Hegab A.E. Calorie restriction enhances adult mouse lung stem cells function and reverses several ageing-induced changes. J. Tissue Eng. Regen. Med. 2019;13:295–308. doi: 10.1002/term.2792. [DOI] [PubMed] [Google Scholar]
- Heidari A. Effects of coenzyme Q10 supplementation on gene expressions related to insulin, lipid, and inflammation pathways in patients with diabetic nephropathy. Iran J Kidney Dis. 2018;12:14–21. [PubMed] [Google Scholar]
- Herb M. Mitochondrial reactive oxygen species enable proinflammatory signaling through disulfide linkage of NEMO. Sci. Signal. 2019;12 doi: 10.1126/scisignal.aar5926. [DOI] [PubMed] [Google Scholar]
- Hernandez-Camacho J.D. Coenzyme Q10 supplementation in aging and disease. Front. Physiol. 2018;9:44. doi: 10.3389/fphys.2018.00044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu F., Liu F. Mitochondrial stress: a bridge between mitochondrial dysfunction and metabolic diseases? Cell. Signal. 2011;23:1528–1533. doi: 10.1016/j.cellsig.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395:10223. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter D.C. Consumption of gold kiwifruit reduces severity and duration of selected upper respiratory tract infection symptoms and increases plasma vitamin C concentration in healthy older adults. Br. J. Nutr. 2012;108:1235–1245. doi: 10.1017/S0007114511006659. [DOI] [PubMed] [Google Scholar]
- Jafarzadeh A. Contribution of monocytes and macrophages to the local tissue inflammation and cytokine storm in COVID-19: lessons from SARS and MERS, and potential therapeutic interventions. Life Sci. 2020 doi: 10.1016/j.lfs.2020.118102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janeway C.A., Jr., Medzhitov R. Innate immune recognition. Annu. Rev. Immunol. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- Jing Y. Aging is associated with a numerical and functional decline in plasmacytoid dendritic cells, whereas myeloid dendritic cells are relatively unaltered in human peripheral blood. Human Immunology. 2009;70:777–784. doi: 10.1016/j.humimm.2009.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jolly C.A. Dietary restriction and immune function. J. Nutr. 2004;134:1853–1856. doi: 10.1093/jn/134.8.1853. [DOI] [PubMed] [Google Scholar]
- Jorat M.V. The effects of coenzyme Q10 supplementation on biomarkers of inflammation and oxidative stress in among coronary artery disease: a systematic review and meta-analysis of randomized controlled trials. Inflammopharmacology. 2019;27:233–248. doi: 10.1007/s10787-019-00572-x. [DOI] [PubMed] [Google Scholar]
- Julian M.W. Mitochondrial transcription factor A, an endogenous danger signal, promotes TNFα release via RAGE- and TLR9-responsive plasmacytoid dendritic cells. PLoS One. 2013;8 doi: 10.1371/journal.pone.0072354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Justiz Vaillant A.A., Jan A. StatPearls Publishing, StatPearls Publishing LLC; Treasure Island (FL): 2020. Physiology, Immune Response, StatPearls. [PubMed] [Google Scholar]
- Kalen A., Appelkvist E.L., Dallner G. Age-related changes in the lipid compositions of rat and human tissues. Lipids. 1989;24:579–584. doi: 10.1007/BF02535072. [DOI] [PubMed] [Google Scholar]
- Kanigur Sultuybek G., Soydas T., Yenmis G. NF-κB as the mediator of metformin's effect on ageing and ageing-related diseases. Clin. Exp. Pharmacol. Physiol. 2019;46:413–422. doi: 10.1111/1440-1681.13073. [DOI] [PubMed] [Google Scholar]
- Kelley N. The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int. J. Mol. Sci. 2019;20 doi: 10.3390/ijms20133328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim M.J. SESN2/sestrin2 suppresses sepsis by inducing mitophagy and inhibiting NLRP3 activation in macrophages. Autophagy. 2016;12:1272–1291. doi: 10.1080/15548627.2016.1183081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenitzer J.R., Freeman B.A. Redox signaling in inflammation: interactions of endogenous electrophiles and mitochondria in cardiovascular disease. Ann. N. Y. Acad. Sci. 2010;1203:45–52. doi: 10.1111/j.1749-6632.2010.05559.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koga H., Kaushik S., Cuervo A.M. Altered lipid content inhibits autophagic vesicular fusion. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2010;24:3052–3065. doi: 10.1096/fj.09-144519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshiba T. Mitochondrial membrane potential is required for MAVS-mediated antiviral signaling. Sci. Signal. 2011;4:ra7. doi: 10.1126/scisignal.2001147. [DOI] [PubMed] [Google Scholar]
- Kwok S. Obesity: a critical risk factor in the COVID-19 pandemic. Clin. Obes. 2020 doi: 10.1111/cob.12403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazear H.M. IRF-3, IRF-5, and IRF-7 coordinately regulate the type I IFN response in myeloid dendritic cells downstream of MAVS signaling. PLoS Pathog. 2013;9 doi: 10.1371/journal.ppat.1003118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S.H. Coenzyme Q10 exerts anti-inflammatory activity and induces Treg in graft versus host disease. J. Med. Food. 2016;19:238–244. doi: 10.1089/jmf.2015.3535. [DOI] [PubMed] [Google Scholar]
- Lee S.Y. Coenzyme Q10 inhibits Th17 and STAT3 signaling pathways to ameliorate colitis in mice. J. Med. Food. 2017;20:821–829. doi: 10.1089/jmf.2016.3859. [DOI] [PubMed] [Google Scholar]
- Lee J.Y., Paik I.Y., Kim J.Y. Voluntary exercise reverses immune aging induced by oxidative stress in aging mice. Exp. Gerontol. 2019;115:148–154. doi: 10.1016/j.exger.2018.08.009. [DOI] [PubMed] [Google Scholar]
- Lee Y.H. β-Cell autophagy: mechanism and role in β-cell dysfunction. Mol. Metab. 2019;27s:S92–s103. doi: 10.1016/j.molmet.2019.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S.W. SARS coronavirus papain-like protease inhibits the TLR7 signaling pathway through removing Lys63-linked Polyubiquitination of TRAF3 and TRAF6. Int. J. Mol. Sci. 2016;17 doi: 10.3390/ijms17050678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q. Knockout of dihydrofolate reductase in mice induces hypertension and abdominal aortic aneurysm via mitochondrial dysfunction. Redox Biol. 2019;24 doi: 10.1016/j.redox.2019.101185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang S. Murine macrophage autophagy protects against alcohol-induced liver injury by degrading interferon regulatory factor 1 (IRF1) and removing damaged mitochondria. J. Biol. Chem. 2019;294:12359–12369. doi: 10.1074/jbc.RA119.007409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin T.A., Wu V.C., Wang C.Y. Autophagy in chronic kidney diseases. Cells. 2019;8 doi: 10.3390/cells8010061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. SS31 ameliorates sepsis-induced heart injury by inhibiting oxidative stress and inflammation. Inflammation. 2019;42:2170–2180. doi: 10.1007/s10753-019-01081-3. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Essential role of mitochondrial dynamics in muscle physiology. Acta Physiol. 2017;219:20–21. doi: 10.1111/apha.12750. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Mitochondrial activity and dynamics changes regarding metabolism in ageing and obesity. Mech. Ageing Dev. 2017;162:108–121. doi: 10.1016/j.mad.2016.12.005. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Springer International Publishing; Switzerland: 2020. Coenzyme Q in Aging. [DOI] [Google Scholar]
- Lopez-Lluch G., Navas P. Calorie restriction as an intervention in ageing. J. Physiol. 2016;594:2043–2060. doi: 10.1113/JP270543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Lluch G. Calorie restriction induces mitochondrial biogenesis and bioenergetic efficiency. Proc. Natl. Acad. Sci. U. S. A. 2006;103:1768–1773. doi: 10.1073/pnas.0510452103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Lluch G. Is coenzyme Q a key factor in aging? Mech. Ageing Dev. 2010;131:225–235. doi: 10.1016/j.mad.2010.02.003. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Mitochondrial responsibility in ageing process: innocent, suspect or guilty. Biogerontology. 2015;16:599–620. doi: 10.1007/s10522-015-9585-9. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Mitochondrial dysfunction in metabolism and ageing: shared mechanisms and outcomes? Biogerontology. 2018;19:461–480. doi: 10.1007/s10522-018-9768-2. [DOI] [PubMed] [Google Scholar]
- Lopez-Lluch G. Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutrition. 2019;57:133–140. doi: 10.1016/j.nut.2018.05.020. [DOI] [PubMed] [Google Scholar]
- Lopez-Moreno J. Mediterranean diet supplemented with coenzyme Q10 modulates the postprandial metabolism of advanced glycation end products in elderly men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2018;73:340–346. doi: 10.1093/gerona/glw214. [DOI] [PubMed] [Google Scholar]
- Lumini J.A. Beneficial effects of exercise on muscle mitochondrial function in diabetes mellitus. Sports Med. 2008;38:735–750. doi: 10.2165/00007256-200838090-00003. [DOI] [PubMed] [Google Scholar]
- Ma R.C.W., Holt R.I.G. COVID-19 and diabetes. Diabetic Med. J. Brit. Diabetic Assoc. 2020;37:723–725. doi: 10.1111/dme.14300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maddaloni E., Buzzetti R. Covid-19 and diabetes mellitus: unveiling the interaction of two pandemics. Diabetes Metab. Res. Rev. 2020 doi: 10.1002/dmrr.3321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Made C.I. Presence of genetic variants among young men with severe COVID-19. Jama. 2020;324:1–11. doi: 10.1001/jama.2020.13719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malavolta M. Exploring the relevance of senotherapeutics for the current SARS-CoV-2 emergency and similar future global health threats. Cells. 2020;9 doi: 10.3390/cells9040909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantle D., Hargreaves I. Coenzyme Q10 and degenerative disorders affecting longevity: an overview. Antioxidants. 2019;8 doi: 10.3390/antiox8020044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mau T. Life-span extension drug interventions affect adipose tissue inflammation in aging. J. Gerontol. A Biol. Sci. Med. Sci. 2020;75:89–98. doi: 10.1093/gerona/glz177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauvais-Jarvis F. Aging, male sex, obesity, and metabolic inflammation create the perfect storm for COVID-19. Diabetes. 2020;69:1857–1863. doi: 10.2337/dbi19-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGettrick A.F., O'Neill L.A.J. The role of HIF in immunity and inflammation. Cell Metab. 2020 doi: 10.1016/j.cmet.2020.08.002. [DOI] [PubMed] [Google Scholar]
- McGuire P.J. Mitochondrial dysfunction and the aging immune system. Biology (Basel) 2019;8 doi: 10.3390/biology8020026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMaster W.G. Inflammation, immunity, and hypertensive end-organ damage. Circ. Res. 2015;116:1022–1033. doi: 10.1161/circresaha.116.303697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menendez J.A. Metformin and SARS-CoV-2: mechanistic lessons on air pollution to weather the cytokine/thrombotic storm in COVID-19. Aging (Albany NY) 2020;12:8760–8765. doi: 10.18632/aging.103347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messaoudi I. Delay of T cell senescence by caloric restriction in aged long-lived nonhuman primates. Proc. Natl. Acad. Sci. U. S. A. 2006;103:19448–19453. doi: 10.1073/pnas.0606661103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miranda D. Deficient mitochondrial biogenesis in IL-2 activated NK cells correlates with impaired PGC1-alpha upregulation in elderly humans. Exp. Gerontol. 2018;110:73–78. doi: 10.1016/j.exger.2018.05.014. [DOI] [PubMed] [Google Scholar]
- Mischel N.A. (In)activity-related neuroplasticity in brainstem control of sympathetic outflow: unraveling underlying molecular, cellular, and anatomical mechanisms. Am. J. Physiol. Heart Circ. Physiol. 2015;309:H235–H243. doi: 10.1152/ajpheart.00929.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Missiroli S. The role of mitochondria in inflammation: from cancer to neurodegenerative disorders. J. Clin. Med. 2020;9 doi: 10.3390/jcm9030740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell S.J. Daily fasting improves health and survival in male mice independent of diet composition and calories. Cell Metab. 2019;29:221–228. doi: 10.1016/j.cmet.2018.08.011. e223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molony R.D. Aging impairs both primary and secondary RIG-I signaling for interferon induction in human monocytes. Sci. Signal. 2017;10 doi: 10.1126/scisignal.aan2392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montecino-Rodriguez E., Berent-Maoz B., Dorshkind K. Causes, consequences, and reversal of immune system aging. The Journal of Clinical Investigation. 2013;123:958–965. doi: 10.1172/JCI64096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore J.B., June C.H. Cytokine release syndrome in severe COVID-19. Science. 2020;368:473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- Morciano G. Mitophagy in cardiovascular diseases. J. Clin. Med. 2020;9 doi: 10.3390/jcm9030892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moro L. Mitochondrial dysfunction in aging and cancer. J. Clin. Med. 2019;8 doi: 10.3390/jcm8111983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouton A.J. Obesity, hypertension, and cardiac dysfunction: novel roles of immunometabolism in macrophage activation and inflammation. Circ. Res. 2020;126:789–806. doi: 10.1161/circresaha.119.312321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munguia L. High flavonoid cocoa supplement ameliorates plasma oxidative stress and inflammation levels while improving mobility and quality of life in older subjects: a double-blind randomized clinical trial. J. Gerontol. A Biol. Sci. Med. Sci. 2019;74:1620–1627. doi: 10.1093/gerona/glz107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakahira K. Autophagy: a crucial moderator of redox balance, inflammation, and apoptosis in lung disease. Antioxid. Redox Signal. 2014;20:474–494. doi: 10.1089/ars.2013.5373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natarajan V. Mitochondrial dysfunction in age-related metabolic disorders. Proteomics. 2020;20 doi: 10.1002/pmic.201800404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishiga M. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020;17:543–558. doi: 10.1038/s41569-020-0413-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novoselova E.G. Naturally occurring antioxidant nutrients reduce inflammatory response in mice. Eur. J. Pharmacol. 2009;615:234–240. doi: 10.1016/j.ejphar.2009.05.004. [DOI] [PubMed] [Google Scholar]
- Olcum M. Phytochemicals and NLRP3 inflammasome inhibitory effects of phytochemicals on NLRP3 inflammasome activation: a review. Phytomedicine. 2020 doi: 10.1016/j.phymed.2020.153238. [DOI] [PubMed] [Google Scholar]
- Orekhov A.N. Possible role of mitochondrial DNA mutations in chronification of inflammation: focus on atherosclerosis. J. Clin. Med. 2020;9 doi: 10.3390/jcm9040978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagliaro P. Is macrophages heterogeneity important in determining COVID-19 lethality? Med. Hypotheses. 2020;143 doi: 10.1016/j.mehy.2020.110073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal R., Bhansali A. COVID-19, diabetes mellitus and ACE2: the conundrum. Diabetes Res. Clin. Pract. 2020;162 doi: 10.1016/j.diabres.2020.108132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paladino R.A. Resveratrol reverses the effect of TNF-α on inflammatory markers in a model of autoimmune uveitis. Eur. J. Integr. Med. 2020;36 doi: 10.1016/j.eujim.2020.101137. [DOI] [Google Scholar]
- Pangrazzi L., Weinberger B. T cells, aging and senescence. Exp. Gerontol. 2020;134 doi: 10.1016/j.exger.2020.110887. [DOI] [PubMed] [Google Scholar]
- Parker D. Age-related adverse inflammatory and metabolic changes begin early in adulthood. J. Gerontol. A Biol. Sci. Med. Sci. 2019;74:283–289. doi: 10.1093/gerona/gly121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parmigiani A. Impaired antibody response to influenza vaccine in HIV-infected and uninfected aging women is associated with immune activation and inflammation. PLoS One. 2013;8 doi: 10.1371/journal.pone.0079816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquarelli-do-Nascimento G. Hypercoagulopathy and adipose tissue exacerbated inflammation may explain higher mortality in COVID-19 patients with obesity. Front. Endocrinol. (Lausanne) 2020;11:530. doi: 10.3389/fendo.2020.00530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pence B.D., Yarbro J.R. Aging impairs mitochondrial respiratory capacity in classical monocytes. Exp. Gerontol. 2018;108:112–117. doi: 10.1016/j.exger.2018.04.008. [DOI] [PubMed] [Google Scholar]
- Pinti M. Circulating mitochondrial DNA increases with age and is a familiar trait: implications for “inflamm-aging”. Eur. J. Immunol. 2014;44:1552–1562. doi: 10.1002/eji.201343921. [DOI] [PubMed] [Google Scholar]
- Pirola C.J., Sookoian S. Age but not sex may explain the negative effect of arterial hypertension and diabetes on COVID-19 prognosis. The Journal of Infection. 2020 doi: 10.1016/j.jinf.2020.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasun P. Mitochondrial dysfunction in metabolic syndrome. Biochim. Biophys. Acta Mol. Dis. 2020 doi: 10.1016/j.bbadis.2020.165838. [DOI] [PubMed] [Google Scholar]
- Promislow D.E.L. A geroscience perspective on COVID-19 mortality. J. Gerontol. A Biol. Sci. Med. Sci. 2020 doi: 10.1093/gerona/glaa094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proudfoot A.E. Chemokine receptors: multifaceted therapeutic targets. Nat. Rev. Immunol. 2002;2:106–115. doi: 10.1038/nri722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahmani H. Interferon β-1b in treatment of severe COVID-19: a randomized clinical trial. Int. Immunopharmacol. 2020;88 doi: 10.1016/j.intimp.2020.106903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramaiah M.J. mTOR inhibition and p53 activation, microRNAs: the possible therapy against pandemic COVID-19. Gene Rep. 2020;20 doi: 10.1016/j.genrep.2020.100765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz Y. Activation-induced autophagy is preserved in CD4+ T-cells in familial longevity. J. Gerontol. A Biol. Sci. Med. Sci. 2017;72:1201–1206. doi: 10.1093/gerona/glx020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riou M. New insights into the implication of mitochondrial dysfunction in tissue, peripheral blood mononuclear cells, and platelets during lung diseases. J. Clin. Med. 2020;9 doi: 10.3390/jcm9051253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodríguez C.P. Peripheral lymphocytes, obesity, and metabolic syndrome in young adults: an immunometabolism study. Metab. Syndr. Relat. Disord. 2018;16:342–349. doi: 10.1089/met.2018.0005. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Bies E. Muscle physiology changes induced by every other day feeding and endurance exercise in mice: effects on physical performance. PLoS One. 2010;5 doi: 10.1371/journal.pone.0013900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Bies E., Navas P., Lopez-Lluch G. Age-dependent effect of every-other-day feeding and aerobic exercise in ubiquinone levels and related antioxidant activities in mice muscle. J. Gerontol. A Biol. Sci. Med. Sci. 2015;70:33–43. doi: 10.1093/gerona/glu002. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Navarro J.A. Inhibitory effect of dietary lipids on chaperone-mediated autophagy. Proc. Natl. Acad. Sci. U. S. A. 2012;109:E705–E714. doi: 10.1073/pnas.1113036109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ron-Harel N. Mitochondrial biogenesis and proteome remodeling promote one-carbon metabolism for T cell activation. Cell Metab. 2016;24:104–117. doi: 10.1016/j.cmet.2016.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roshanravan N. Targeting cytokine storm to manage patients with COVID-19: a mini-review. Arch. Med. Res. 2020 doi: 10.1016/j.arcmed.2020.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothan H.A., Byrareddy S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020;109 doi: 10.1016/j.jaut.2020.102433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth-Cross J.K. Inhibition of the alpha/beta interferon response by mouse hepatitis virus at multiple levels. J. Virol. 2007;81:7189–7199. doi: 10.1128/jvi.00013-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy S., Mazumder T., Banik S. The Association of Cardiovascular Diseases and Diabetes Mellitus with COVID-19 (SARS-CoV-2) and their possible mechanisms. SN Compr. Clin. Med. 2020:1–6. doi: 10.1007/s42399-020-00376-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Q. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Medicine. 2020;46:846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo G.L. Mechanisms of aging and potential role of selected polyphenols in extending healthspan. Biochem. Pharmacol. 2020;173 doi: 10.1016/j.bcp.2019.113719. [DOI] [PubMed] [Google Scholar]
- Ryter S.W., Choi A.M. Autophagy in lung disease pathogenesis and therapeutics. Redox Biol. 2015;4:215–225. doi: 10.1016/j.redox.2014.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandhir R., Halder A., Sunkaria A. Mitochondria as a centrally positioned hub in the innate immune response. Biochim. Biophys. Acta. Mol. Dis. 2017;1863:1090–1097. doi: 10.1016/j.bbadis.2016.10.020. [DOI] [PubMed] [Google Scholar]
- Schmelzer C. Supplementation with the reduced form of coenzyme Q10 decelerates phenotypic characteristics of senescence and induces a peroxisome proliferator-activated receptor-alpha gene expression signature in SAMP1 mice. Mol. Nutr. Food Res. 2010;54:805–815. doi: 10.1002/mnfr.200900155. [DOI] [PubMed] [Google Scholar]
- Sellami M. Effects of acute and chronic exercise on immunological parameters in the elderly aged: can physical activity counteract the effects of aging? Front. Immunol. 2018;9:2187. doi: 10.3389/fimmu.2018.02187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seth R.B. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-kappaB and IRF 3. Cell. 2005;122:669–682. doi: 10.1016/j.cell.2005.08.012. [DOI] [PubMed] [Google Scholar]
- Shally A., McDonagh B. The redox environment and mitochondrial dysfunction in age-related skeletal muscle atrophy. Biogerontology. 2020;21:461–473. doi: 10.1007/s10522-020-09879-7. [DOI] [PubMed] [Google Scholar]
- Shemiakova T. Mitochondrial dysfunction and DNA damage in the context of pathogenesis of atherosclerosis. Biomedicines. 2020;8 doi: 10.3390/biomedicines8060166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu M. Low circulating coenzyme Q10 during acute phase is associated with inflammation, malnutrition, and in-hospital mortality in patients admitted to the coronary care unit. Heart Vessel. 2017;32:668–673. doi: 10.1007/s00380-016-0923-x. [DOI] [PubMed] [Google Scholar]
- Sims C.A. Nicotinamide mononucleotide preserves mitochondrial function and increases survival in hemorrhagic shock. JCI Insight. 2018;3 doi: 10.1172/jci.insight.120182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skuratovskaia D. Mitochondrial destiny in type 2 diabetes: the effects of oxidative stress on the dynamics and biogenesis of mitochondria. PeerJ. 2020;8 doi: 10.7717/peerj.9741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegel M. Inhibition of Beta interferon induction by severe acute respiratory syndrome coronavirus suggests a two-step model for activation of interferon regulatory factor 3. J. Virol. 2005;79:2079–2086. doi: 10.1128/jvi.79.4.2079-2086.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stead E.R. Agephagy - adapting autophagy for health during aging. Front. Cell Develop. Biol. 2019;7:308. doi: 10.3389/fcell.2019.00308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefan N. Obesity and impaired metabolic health in patients with COVID-19. Nat. Rev. Endocrinol. 2020;16:341–342. doi: 10.1038/s41574-020-0364-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stigger F.S. Effects of exercise on inflammatory, oxidative, and neurotrophic biomarkers on cognitively impaired individuals diagnosed with dementia or mild cognitive impairment: a systematic review and meta-analysis. J. Gerontol. A Biol. Sci. Med. Sci. 2019;74:616–624. doi: 10.1093/gerona/gly173. [DOI] [PubMed] [Google Scholar]
- Suárez-Rivero J.M. Atherosclerosis and coenzyme Q(10) Int. J. Mol. Sci. 2019:20. doi: 10.3390/ijms20205195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun L. Coronavirus papain-like proteases negatively regulate antiviral innate immune response through disruption of STING-mediated signaling. PLoS One. 2012;7 doi: 10.1371/journal.pone.0030802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun L. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science. 2013;339:786–791. doi: 10.1126/science.1232458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang D. PAMPs and DAMPs: signal 0s that spur autophagy and immunity. Immunol. Rev. 2012;249:158–175. doi: 10.1111/j.1600-065X.2012.01146.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tannahill G.M. Succinate is an inflammatory signal that induces IL-1beta through HIF-1alpha. Nature. 2013;496:238–242. doi: 10.1038/nature11986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarasenko T.N. Cytochrome c oxidase activity is a metabolic checkpoint that regulates cell fate decisions during T cell activation and differentiation. Cell Metab. 2017;25:1254–1268. doi: 10.1016/j.cmet.2017.05.007. e1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavernarakis N. Inflammation brakes mitochondrial metabolism in obesity. Nat. Immunol. 2020;21:1143–1145. doi: 10.1038/s41590-020-0780-8. [DOI] [PubMed] [Google Scholar]
- Teijaro J.R. Mapping the innate signaling cascade essential for cytokine storm during influenza virus infection. Proc. Natl. Acad. Sci. U. S. A. 2014;111:3799–3804. doi: 10.1073/pnas.1400593111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ten V.S., Ratner V. Mitochondrial bioenergetics and pulmonary dysfunction: current progress and future directions. Paediatr. Respir. Rev. 2020;34:37–45. doi: 10.1016/j.prrv.2019.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson M.R. Pattern recognition receptors and the innate immune response to viral infection. Viruses. 2011;3:920–940. doi: 10.3390/v3060920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- To E.E. Mitochondrial reactive oxygen species contribute to pathological inflammation during influenza A virus infection in mice. Antioxid. Redox Signal. 2020;32:929–942. doi: 10.1089/ars.2019.7727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tosti V., Bertozzi B., Fontana L. Health benefits of the Mediterranean diet: metabolic and molecular mechanisms. J. Gerontol. A Biol. Sci. Med. Sci. 2018;73:318–326. doi: 10.1093/gerona/glx227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tumova L. Azorella compacta infusion activates human immune cells and scavenges free radicals in vitro. Pharmacogn. Mag. 2017;13:260–264. doi: 10.4103/0973-1296.204558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tung B.T. Modulation of endogenous antioxidant activity by resveratrol and exercise in mouse liver is age dependent. J. Gerontol. A Biol. Sci. Med. Sci. 2014;69:398–409. doi: 10.1093/gerona/glt102. [DOI] [PubMed] [Google Scholar]
- Tung B.T. Anti-inflammatory effect of resveratrol in old mice liver. Exp. Gerontol. 2015;64:1–7. doi: 10.1016/j.exger.2015.02.004. [DOI] [PubMed] [Google Scholar]
- Turner J.E. Is immunosenescence influenced by our lifetime “dose” of exercise? Biogerontology. 2016;17:581–602. doi: 10.1007/s10522-016-9642-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velazquez-Salinas L. The role of interleukin 6 during viral infections. Front. Microbiol. 2019;10:1057. doi: 10.3389/fmicb.2019.01057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veloso C.D. A mitochondrial approach to cardiovascular risk and disease. Curr. Pharm. Des. 2019;25:3175–3194. doi: 10.2174/1389203720666190830163735. [DOI] [PubMed] [Google Scholar]
- Vetvicka V., Vetvickova J. Combination therapy with glucan and coenzyme Q10 in murine experimental autoimmune disease and cancer. Anticancer Res. 2018;38:3291–3297. doi: 10.21873/anticanres.12594. [DOI] [PubMed] [Google Scholar]
- Weinberg S.E., Sena L.A., Chandel N.S. Mitochondria in the regulation of innate and adaptive immunity. Immunity. 2015;42:406–417. doi: 10.1016/j.immuni.2015.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Windt G.J. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc. Natl. Acad. Sci. U. S. A. 2013;110:14336–14341. doi: 10.1073/pnas.1221740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit E. SARS and MERS: recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016;14:523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Chen M., Jiang J. Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion. 2019;49:35–45. doi: 10.1016/j.mito.2019.07.003. [DOI] [PubMed] [Google Scholar]
- Xu Y., Shen J., Ran Z. Emerging views of mitophagy in immunity and autoimmune diseases. Autophagy. 2020;16:3–17. doi: 10.1080/15548627.2019.1603547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi O. Autophagy in the heart. Circ. J. 2019;83:697–704. doi: 10.1253/circj.CJ-18-1065. [DOI] [PubMed] [Google Scholar]
- Yang H., Youm Y.H., Dixit V.D. Inhibition of thymic adipogenesis by caloric restriction is coupled with reduction in age-related thymic involution. J. Immunol. 2009;183:3040–3052. doi: 10.4049/jimmunol.0900562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. The Lancet. Respiratory medicine. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao Z. Nicotinamide mononucleotide inhibits JNK activation to reverse Alzheimer disease. Neurosci. Lett. 2017;647:133–140. doi: 10.1016/j.neulet.2017.03.027. [DOI] [PubMed] [Google Scholar]
- Yuan J. Dietary intake of resveratrol enhances the adaptive immunity of aged rats. Rejuvenation Res. 2012;15:507–515. doi: 10.1089/rej.2012.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan Y. Mitochondrial ROS-induced lysosomal dysfunction impairs autophagic flux and contributes to M1 macrophage polarization in a diabetic condition. Clin. Sci. 2019;133:1759–1777. doi: 10.1042/CS20190672. [DOI] [PubMed] [Google Scholar]
- Yubero-Serrano E.M. Mediterranean diet supplemented with coenzyme Q10 modifies the expression of proinflammatory and endoplasmic reticulum stress-related genes in elderly men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2012;67:3–10. doi: 10.1093/gerona/glr167. [DOI] [PubMed] [Google Scholar]
- Zaki N., Alashwal H., Ibrahim S. Association of hypertension, diabetes, stroke, cancer, kidney disease, and high-cholesterol with COVID-19 disease severity and fatality: a systematic review. Diabetes Metab. Syndrome. 2020;14:1133–1142. doi: 10.1016/j.dsx.2020.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai J. Effects of coenzyme Q10 on markers of inflammation: a systematic review and meta-analysis. PLoS One. 2017;12 doi: 10.1371/journal.pone.0170172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L. Decreasing pro-inflammatory cytokine and reversing the immunosenescence with extracts of Pu-erh tea in senescence accelerated mouse (SAM) Food Chem. 2012;135:2222–2228. doi: 10.1016/j.foodchem.2012.07.033. [DOI] [PubMed] [Google Scholar]
- Zhang R. Short-term administration of nicotinamide mononucleotide preserves cardiac mitochondrial homeostasis and prevents heart failure. J. Mol. Cell. Cardiol. 2017;112:64–73. doi: 10.1016/j.yjmcc.2017.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science. 2020;370 doi: 10.1126/science.abd4570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng H.Y. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cellular & Molecular Immunology. 2020 doi: 10.1038/s41423-020-0401-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng M. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cellular & molecular immunology. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Z. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. The Journal of Infection. 2020 doi: 10.1016/j.jinf.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao C., Zhao W. NLRP3 inflammasome-a key player in antiviral responses. Front. Immunol. 2020;11:211. doi: 10.3389/fimmu.2020.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong Z. NF-κB restricts inflammasome activation via elimination of damaged mitochondria. Cell. 2016;164:896–910. doi: 10.1016/j.cell.2015.12.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y. Post-translational regulation of antiviral innate signaling. Eur. J. Immunol. 2017;47:1414–1426. doi: 10.1002/eji.201746959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou F. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zust R. Ribose 2'-O-methylation provides a molecular signature for the distinction of self and non-self mRNA dependent on the RNA sensor Mda5. Nat. Immunol. 2011;12:137–143. doi: 10.1038/ni.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No applicable.