Emerging evidence suggests that people with chronic kidney disease have a higher risk of severe infection with coronavirus disease 2019 (COVID-19) [1]. Furthermore, acute kidney injury is common amongst patients hospitalised with COVID-19 compared to historical controls (56.9% vs. 25.1%) [2]. An estimated 726,000 people in the United States (US) are affected by end-stage renal disease (ESRD), and one-year mortality for people with ESRD who receive dialysis is approximately 20–25% [3]. Reports from China, England, Italy and France have suggested between 9% and 27% of patients with ESRD who tested positive for COVID-19 died, compared to 4% globally as of July 2020 [4]. The extent to which COVID-19 may worsen outcomes for people with ESRD is unclear. Therefore, the objective of this study was to compare 30-day mortality for people with ESRD and COVID-19 (cases) to matched people with ESRD before the COVID-19 pandemic (historical controls).
The study data were provided by TriNetX, a global federated health research network with access to electronic medical records (EMRs) from participating healthcare organisations, including academic medical centres, speciality physician practices and community hospitals, predominately in the United States (US). The TriNetX network was searched on November 3, 2020. ESRD was defined using the International Classification of Disease (ICD)−10-Clinical Modification (CM) code N18.6. Cases were aged ≥18 years with COVID-19 and ESRD (ICD-10-CM code N18.6) recorded in EMRs between January 20, 2020 (date COVID-19 first confirmed in the US) [5] and October 3, 2020 (to allow for at least 30 days follow-up for all included patients). Historical controls were aged ≥18 years with ESRD (ICD-10-CM code N18.6) recorded in EMRs between January 20, 2019 and October 3, 2019. For both the case and control groups, only patients with first-recording of ESRD in EMRs during the specified time periods were included, therefore, patients were excluded if they had the ICD-10-CM code N18.6 included in EMRs prior to the specified dates. At the time of the search, 44 participating healthcare organisations had data available for patients meeting the study inclusion criteria. COVID-19 was identified using criteria provided by TriNetX based on Centres for Disease Control and Prevention (CDC) coding guidelines using ICD-10-CM codes, or specific laboratory Logical Observation Identifiers Names and Codes [6].
All statistical analyses were completed on the TriNetX platform. Baseline characteristics were compared with chi-squared tests or independent-sample t-tests. Cases and historical controls were 1:1 propensity score matched using logistic regression for age, sex, race, respiratory diseases, diseases of the nervous system, hypertension, heart failure, atrial fibrillation and flutter, other cardiac arrhythmias, ischaemic heart disease, cerebrovascular diseases, diabetes mellitus, neoplasms and mental, behavioural and neurodevelopmental disorders. Kaplan-Meier survival curves with log-rank tests and Risk Ratios with 95% Confidence Intervals (CIs) for 30-day mortality comparing cases and historical controls were produced. Statistical significance was set at p<0.05.
To gain access to the data in the TriNetX research network, a request can be made to TriNetX (https://live.trinetx.com), but costs may be incurred, a data sharing agreement would be necessary, and no patient identifiable information can be obtained.
Of the participating sites within the TriNetX network, 27,961 patients met the inclusion criteria for historical controls with ESRD and 865 patients met the inclusion criteria for cases with ESRD and COVID-19. Of the patients with ESRD and COVID-19, 73.2% (n = 633) were recorded as having a hospital inpatient visit two weeks before or after their COVID-19 diagnosis. Compared to cases, historical controls were younger, had a higher proportion of females, had a higher proportion of people identified as white and had a lower proportion of people with a history of the health conditions included in subsequent propensity score matching (Table 1 ). Table 1 also shows the characteristics of the cases and historical controls after 1:1 propensity score matching. After 1:1 propensity score matching, there were 863 patients in each cohort and the cohorts were well balanced (all differences on age, race, sex and included health conditions p>0.05).
Table 1.
Baseline characteristics of the patients with end stage renal disease with and without COVID-19, before and after propensity score matching.
| Before propensity score matching |
After propensity score matching |
|||||
|---|---|---|---|---|---|---|
| ESRD and COVID-19 (n = 865) | ESRD, no COVID-19 (n = 27,961) | P-value | ESRD and COVID-19 (n = 863) | ESRD, no COVID-19 (n = 863) | P-value | |
| Age (years), mean (SD) | 61.2 (14.3) | 60.0 (15.0) | 0.02 | 61.2 (14.3) | 61.3 (14.8) | 0.90 |
| Female | 37.9 (328) | 42.6 (11,907) | 0.006 | 38.0 (328) | 38.5 (332) | 0.84 |
| Race | ||||||
| White | 36.1 (312) | 52.1 (14,554) | <0.001 | 36.2 (312) | 36.8 (318) | 0.76 |
| Black or African American | 37.9 (328) | 26.6 (7426) | <0.001 | 37.8 (326) | 36.3 (313) | 0.52 |
| Asian | 3.6 (31) | 3.6 (1011) | 0.96 | 3.6 (31) | 4.5 (39) | 0.33 |
| Native Hawaiian or other Pacific Islander | 1.2 (10) | 0.3 (75) | <0.001 | 1.2 (10) | 1.2 (10) | 1.00 |
| American Indian or Alaska Native | 1.2 (10) | 0.8 (219) | 0.22 | 1.2 (10) | 1.2 (10) | 1.00 |
| Unknown | 21.4 (185) | 16.7 (4676) | <0.001 | 21.4 (185) | 21.7 (187) | 0.91 |
| Hypertensive diseases | 51.2 (443) | 41.1 (11,492) | <0.001 | 51.3 (443) | 54.5 (470) | 0.19 |
| Ischaemic heart diseases | 22.2 (192) | 17.2 (4815) | 0.001 | 22.2 (192) | 22.9 (198) | 0.73 |
| Heart Failure | 22.3 (193) | 16.3 (4570) | <0.001 | 22.4 (193) | 21.7 (187) | 0.73 |
| Cerebrovascular diseases | 14.3 (124) | 8.6 (2401) | <0.001 | 14.1 (122) | 14.6 (126) | 0.78 |
| Atrial fibrillation and flutter | 11.3 (98) | 8.2 (2300) | 0.001 | 11.4 (98) | 10.4 (90) | 0.54 |
| Other cardiac arrhythmias | 12.5 (108) | 8.4 (2358) | <0.001 | 12.5 (108) | 12.3 (106) | 0.88 |
| Diabetes Mellitus | 39.5 (342) | 29.6 (8263) | <0.001 | 39.4 (340) | 42.6 (368) | 0.17 |
| Neoplasms | 16.9 (146) | 14.8 (4147) | 0.10 | 16.9 (146) | 16.2 (140) | 0.70 |
| Diseases of the respiratory system | 46.1 (399) | 26.0 (7272) | <0.001 | 46.0 (397) | 46.2 (399) | 0.92 |
| Diseases of the nervous system | 36.2 (313) | 25.0 (6992) | <0.001 | 36.0 (311) | 39.7 (308) | 0.88 |
| Mental, behavioural and neurodevelopmental disorders | 27.3 (236) | 20.8 (5816) | <0.001 | 27.3 (236) | 25.8 (223) | 0.48 |
ESRD: end stage renal disease; SD: standard deviation.
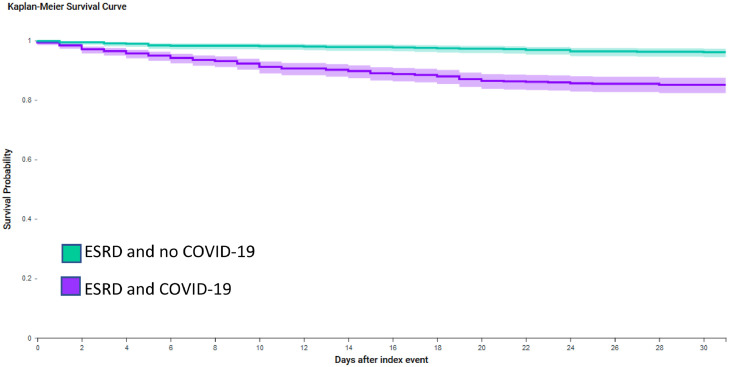
Before propensity score matching, 30-day mortality was higher in cases than historical controls (Log Rank test p<0.0001). For cases, 30-day mortality was 12.7% (n = 110 of 865 patients), compared to 3.0% of historical controls (n = 847 of 27,961 patients). The Risk Ratio for 30-day mortality was 4.20 (95% CI: 3.48, 5.06) for cases compared to historical controls. After propensity score matching, 30-day mortality remained higher in cases than historical controls (12.7% of cases (n = 110 of 863 patients) compared to 3.4% of historical controls (n = 29 of 863 patients); Log Rank test p<0.0001, Fig. 1 ). The Risk Ratio for 30-day mortality was 3.80 (95% CI: 2.55, 5.65) for cases compared to historical controls after propensity score matching.
Fig. 1.
Kaplan-Meier survival curve of 30-day mortality for cases (adults with end stage renal disease and COVID-19) compared to historical controls (adults with end stage renal disease without COVID-19).
ESRD: end stage renal disease. Propensity score matched for age, sex, race, respiratory diseases, diseases of the nervous system, hypertension, heart failure, atrial fibrillation and flutter, other cardiac arrhythmias, ischaemic heart disease, cerebrovascular diseases, diabetes mellitus, neoplasms, mental, behavioural and neurodevelopmental disorders. Purple line is cases (adults with end stage renal disease and COVID-19) and green line is historical controls (adults with end stage renal disease without COVID-19).
Sensitivity analyses were conducted excluding people with kidney transplant identified from ICD-10-CM or procedure codes, up to 30-days after ESRD was coded in EMRs, and showed no important difference in the results (data not shown).
The current study of over 1700 people with ESRD showed that COVID-19 was associated with significantly higher 30-day mortality when compared to propensity score matched patients without COVID-19.
In this study, 13% of people with ESRD died with COVID-19, compared to 3% of historical controls. Evidence from studies in China suggest people with chronic kidney disease are at a higher risk of severe infection with COVID-19 [1]. Recent studies from the United Kingdom (UK) and the US suggested chronic kidney disease associates with higher mortality for patients with COVID-19 [7, 8]. Furthermore, a study of patients hospitalised with COVID-19 in the US suggested higher in-hospital deaths in patients with ESRD [9]. However, patients with ESRD have substantially higher mortality than the general population so this may be expected. By comparing people with ESRD and COVID-19 to historical matched controls with ESRD without COVID-19, the results of the current study suggest that COVID-19 further increases mortality nearly 4-fold in people with ESRD.
The study utilised a global federated research network with a large number of patients with ESRD and propensity score matched those with ESRD and COVID-19 to historical controls with ESRD without COVID-19, captured in the same time frame 12-months prior. The main limitation is that characterisation of ESRD and other health conditions is based on ICD codes from EMRs, and previous studies have suggested reporting of conditions with ICD codes may vary by patient characteristics and between different healthcare organisations [10]. Although patients were matched for many co-morbidities and demographic factors, residual confounding may still be possible.
In conclusion, this study suggests that mortality for people with ESRD and COVID-19 is significantly higher than for people with ESRD without COVID-19. Measures to prevent infection are particularly important for people with ESRD, as well as the targeting of early interventions such as increased monitoring post-infection and should be considered for people with ESRD.
Declaration of Competing Interest
Stephanie L Harrison and Benjamin JR Buckley: None declared. Elnara Fazio-Eynullayeva and Paula Underhill are employees of TriNetX Inc. Deirdre A Lane has received investigator-initiated educational grants from Bristol-Myers Squibb (BMS), has been a speaker for Boehringer Ingeheim, and BMS/Pfizer and has consulted for BMS, Boehringer Ingelheim, and Daiichi-Sankyo. Gregory YH Lip: consultant for Bayer/Janssen, BMS/Pfizer, Medtronic, Boehringer Ingelheim, Novartis, Verseon and Daiichi-Sankyo and speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim, and Daiichi-Sankyo. No fees are directly received to Gregory Lip personally.
Acknowledgments
Acknowledgements
The authors acknowledge the participating healthcare organisations who allowed TriNetX Inc. access to their electronic medical records, the identities of the healthcare organisations must remain anonymous due to data privacy restrictions.
Funding
No specific funding was received for this study. TriNetX Inc. funded the acquisition of the data used.
References
- 1.Henry B.M., Lippi G. Chronic kidney disease is associated with severe coronavirus disease 2019 (COVID-19) infection. Int Urol Nephrol. 2020 doi: 10.1007/s11255-020-02451-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fisher M., Neugarten J., Bellin E., Yunes M., Stahl L., Johns T.S. AKI in Hospitalized Patients with and without COVID-19: a Comparison Study. Journal of the American Society of Nephrology. 2020;31:2145. doi: 10.1681/ASN.2020040509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention . Vol. 2019. US Department of Health and Human Services, Centers for Disease Control and Prevention; Atlanta, GA: 2019. (Chronic kidney disease in the united states). [Google Scholar]
- 4.Valeri A. COVID-19 and ESRD: entering a New Era of Uncertainty. Kidney Int Rep. 2020;5:1381–1382. doi: 10.1016/j.ekir.2020.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holshue M.L., DeBolt C., Lindquist S., Lofy K.H., Wiesman J., Bruce H. First Case of 2019 Novel Coronavirus in the United States. The New England journal of medicine. 2020;382:929–936. doi: 10.1056/NEJMoa2001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Centers for Disease Control and Prevention. ICD-10-CM Official Coding Guidelines - Supplement Coding encounters related to COVID-19 Coronavirus Outbreak. 2020.
- 7.Docherty A.B., Harrison E.M., Green C.A., Hardwick H.E., Pius R., Norman L. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi: 10.1136/bmj.m1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Harrison S.L., Fazio-Eynullayeva E., Lane D.A., Underhill P., Lip G.Y.H. Comorbidities associated with mortality in 31,461 adults with COVID-19 in the United States: a federated electronic medical record analysis. PLoS Med. 2020;17 doi: 10.1371/journal.pmed.1003321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ng J.H., Hirsch J.S., Wanchoo R., Sachdeva M., Sakhiya V., Hong S. Outcomes of patients with end-stage kidney disease hospitalized with COVID-19. Kidney Int. 2020 doi: 10.1016/j.kint.2020.07.030. S0085-2538(20)30945-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chong W.F., Ding Y.Y., Heng B.H. A comparison of comorbidities obtained from hospital administrative data and medical charts in older patients with pneumonia. BMC Health Serv Res. 2011;11:105. doi: 10.1186/1472-6963-11-105. [DOI] [PMC free article] [PubMed] [Google Scholar]