Abstract
The approach to transplantation in human immunodeficiency virus (HIV)-positive patients has been conservative due to fear of exacerbating an immunocompromised condition. As a result, HIV-positive patients with diabetes were initially excluded from beta cell replacement therapy. Early reports of pancreas transplant in patients with HIV described high rates of early graft loss with limited follow up. We report long-term follow up of islet or pancreas transplantation in HIV-positive Type I diabetic patients who received a kidney transplant concurrently, or had previously undergone kidney transplantation. Although four patients developed polyoma viremia, highly active antiretroviral therapy and adequate infectious prophylaxis was successful in providing protection until CD4+ counts recovered. Coordination with HIV providers is critical to reduce the risk of rejection by minimizing drug-drug interactions. Also, protocols for prophylaxis of opportunistic infections, and strategies for monitoring and treating BK viremia are important given the degree of immunosuppression required. This series demonstrates that type 1 diabetic patients with well-controlled HIV and renal failure can be appropriate candidates for beta cell replacement, with a low rate of infectious complications, early graft loss and rejection, so excellent long-term graft survival is possible. Additionally, patients with HIV and cardiovascular contraindications can undergo islet infusion.
Keywords: HIV, pancreas transplant, islet transplant, pancreas-kidney transplant, immune suppression, Polyoma virus
1. Introduction
cART has facilitated reconstitution of the immune system in people infected with HIV, making solid organ transplantation a realistic option. Although several trials have shown liver and kidney transplantation to be a safe and effective therapy in the HIV-positive recipient, early trials reported an unexpectedly high incidence of rejection, reflecting the necessity for aggressive immunosuppression in this group of patients that are theoretically immunocompromised. Successful pancreas and pancreatic islet transplantation in the HIV-negative recipient has generally required aggressive induction therapy with a lymphocyte depleting agent to overcome both the allo- and auto-immune response in the recipient with Type 1 diabetes. For these reasons, pancreas and islet transplantation in HIV-positive recipients has lagged behind other solid organ transplants due to the anticipated aggressive immunosuppression regimens and concerns that these aggressive regimens would be poorly tolerated given their impact on CD4 T-cell biology and increased potential for infection. In this report we discuss the long-term follow up of our first eight cases of solid organ pancreas transplantation and two cases of islet transplantation in HIV-positive, uremic Type I diabetic patients also requiring kidney transplantation. Additionally, we offer an in depth description of our protocols and the outcomes we have experienced, including the challenges we face in this patient population.
2. Materials and Methods
2.1. Patient selection
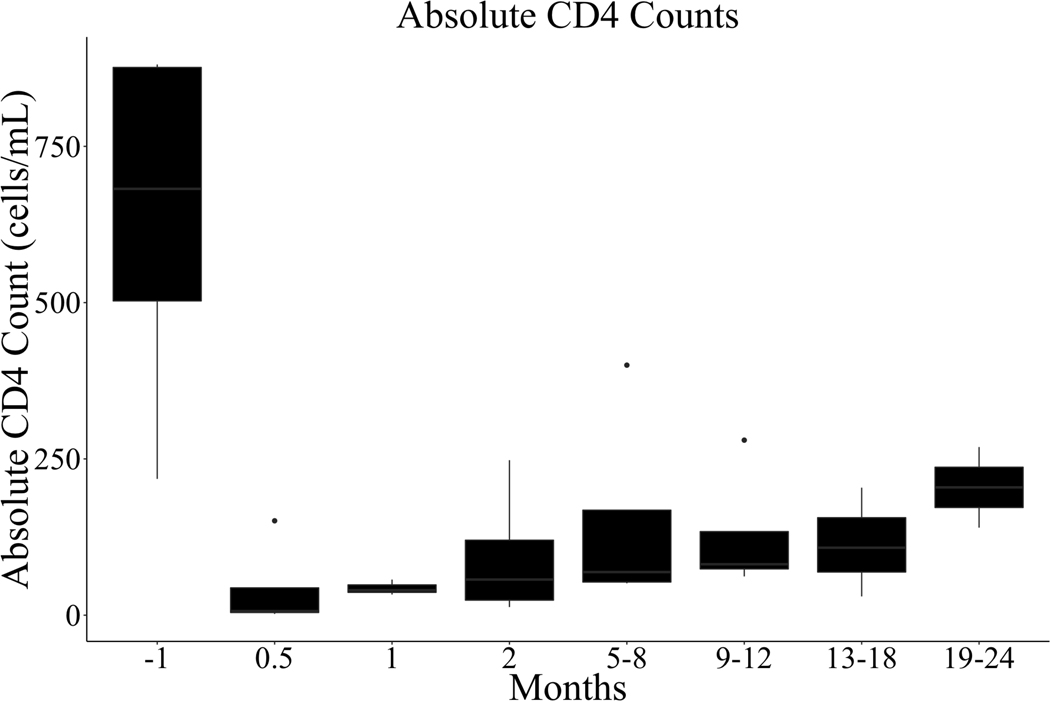
Type I diabetes patients were considered candidates for pancreas transplant if HIV was well-controlled on a stable cART regimen with concurrent renal failure or had recently undergone kidney transplantation. Islet preparation and infusion was performed under an FDA approved IND (#11088). Specific inclusion criteria included: 1) ≥ 5 years of insulin-dependent Type 1 diabetes mellitus; 2) stimulated C-peptide levels < 0.5ng/mL (islet after kidney transplant (IAK)); 4) body mass index < 32 kg/m2; 5) insulin requirement < 55 units/day; 6) creatinine clearance > 60 mL/min/m2 for beta cell replacement following kidney transplantation (IAK or pancreas after kidney transplant (PAK)); 7) no history of malignancy within 10 years (except treated basal or squamous cell carcinoma of the skin); 8) stable CD4 counts > 200 cells/mL; 9) undetectable viral load on a stable anti-retroviral regimen at the time of transplant; and 10) no history of any opportunistic infections which cannot be controlled with medical management (coccidioidomycosis or JC virus). Two patients underwent islet infusions rather than solid organ pancreas transplantation because their cardiovascular risk was too high to tolerate pancreas transplantation. Box plots showing absolute CD4 counts (cells/mL) were created using the ggplot2 package in RStudio (Version 1.1.453). The Committee on Human Research at University of California, San Francisco approved this study (IRB # 13–11589).
2.2. Islet preparation and transplantation
Islet preparation and infusion was performed under an FDA approved IND (#11088). Islets were procured from deceased donor pancreata with Collagenase NB1 blend (Serva Electrophoresis) and LIBERASE HI (Roche). Islets were maintained in culture for 36 to 48 hours and assessed for sterility and viability before suspension in transplant medium supplemented with heparin (70U/kg recipient body weight). Inclusion criteria for release of islets for clinical use included: ≥ 4,000 islet equivalents (IE)/kg recipient body weight, viability ≥ 70%, purity ≥ 20,000 IEQ/mL settled tissue volume, settled tissue volume ≤ 15mL, glucose stimulated insulin release index ≥ 1.0, endotoxin level ≤ 5.0 EU/kg recipient body weight, and negative gram stain of islet culture fluid.
Percutaneous portal vein catheterization was used for intraportal islet infusion. Recipients received heparin for 48 hours after islet infusion followed by 5 days of subcutaneous enoxaparin. Recipients who were not insulin independent 2–3 months after transplantation, but who had detectable C-peptide were listed for a second infusion.
2.3. Pancreas transplantation
Standard heterotopic pancreas transplantation was performed with enteric exocrine drainage and systemic venous drainage.
2.4. Immunosuppression and prophylaxis
Thymoglobulin (6 mg/kg) was used as the induction agent in all solid organ pancreas recipients. Immunosuppression regimen at the time of pancreas transplantation, the most recent maintenance immunosuppression, and cART at the time of transplant and time of rejection (if applicable) are listed in Table 1. Postoperative prophylaxis against Pneumocystis jiroveci pneumonia, cytomegalovirus, and mycobacterium avium complex infections included sulfamethoxazole/trimethoprim double strength three times weekly, valganciclovir 900mg daily or Acyclovir 800mg four times a day adjusted for renal function, and weekly azithromycin 1200mg, respectively. Fungal prophylaxis was fluconazole 100 mg weekly for 3 months. Given the higher risk of rejection in this patient population steroid withdrawal was not employed and patients remained on Prednisone 5 mg daily in the long term, unless a significant contraindication developed. Tacrolimus trough goal was 8–10 ng/ml for the first 1 year, then 5–8 ng/ml for the long term. Screening of the serum for polyoma virus (BK) was performed monthly for the first year, and was considered positive if any virus was detected. Monitoring was continued monthly until the virus was no longer detectable, if needed. Once BK was detected, MMF was reduced by 50%. MMF was changed to Leflunomide in the case of persistent BK viremia. We did not monitor for BK virus in the urine.
Table 1.
Pancreas Transplant Recipients
| Patient | P#1 | P#2 | P#3 | P#4 | P#5 | P#6 | P#7 | P#8 |
|---|---|---|---|---|---|---|---|---|
| Age at transplant (years) | 50 | 33 | 49 | 40 | 32 | 38 | 49 | 50 |
| Gender | Male | Male | Male | Male | Male | Male | Female | Male |
| Height (cm) | 177.8 | 177.8 | 177.8 | 169.9 | 172.7 | 163 | 167.6 | 185.4 |
| Weight (kg) | 72 | 70.3 | 72.57 | 68 | 76.7 | 52.6 | 73.5 | 86.6 |
| BMI (kg/m2) | 22.8 | 22.2 | 23 | 23.6 | 25.7 | 19.9 | 26.2 | 25.2 |
| Previous transplant | No | No | No | No | Yes (kidney) | No | No | No |
| cPRA | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 4% |
| Length of pre-transplant DM1 (years) | 26 | 21 | 25 | 27 | 16 | 22 | 28 | 26 |
| Induction for pancreas transplant | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) | Thymo (6mg/kg) |
| Immunosuppression regimen at the time of pancreas transplant | MMF, cyclosporine, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone |
| LOS | 7 | 9 | 15 | 8 | 7 | 13 | 7 | 20 |
| Melena requiring in PRBC transfusion (units) | Yes (3) | No | Yes (4) | Yes (7) | No | Yes (1) | No | No |
| Required reoperation | Yes, for bleeding on POD 2 | No | No | No | No | No | No | No |
| Readmission within 30 days (reason) | Yes (CSA toxicity) | No | No | No | Yes (fluid collection requiring antibiotics) | Yes (dehydration) | No | No |
| Splenic vein thrombosis on US | No | No | No | No | No | No | No | Yes |
| Most recent immunosuppression | MMF, sirolimus* | MMF, cyclosporine | MMF, tacrolimus, prednisone | MMF, tacrolimus, prednisone* | MMF, sirolimus, tacrolimus, prednisone | MMF, tacrolimus, prednisone | Everolimus, tacrolimus | MMF, tacrolimus, prednisone |
| cART regimen at time of pancreas graft transplant | Stavudinea, ritonavirb, lamivudinea, saquinavirb | Abacavira, lamivudinea, efavirenzc | Abacavira, fosamprenavirb, lamivudinea | Stavudinea, efavirenzc, lamivudinea | Abacavira/ lamivudinea, efavirenzc | Abacavira, lamivudinea, raltegravird | Abacavira, emtrictabinea, raltegravird | Dolutegravird/ rilpivirinec |
| cART regimen at time of rejection (if applicable) | NA | Abacavira, lamivudinea, efavirenzc | NA | NA | Abacavira/ lamivudinea, efavirenzc | NA | NA | NA |
| Most recent cART regimen | Stavudinea, ritonavirb, lamivudinea, saquinavirb | Abacavira/ dolutegravird/ lamivudinea | Abacavira, dolutegravird, emtricitabinea/ rilpivirinec/ tenofovira, fosamprenavirb | Abacavira, efavirenzc, lamivudinea | Rilpivirinec, raltegravird, abacavira/ lamivudinea | Abacavira/ dolutegravird /lamivudinea | Abacavira/ dolutegravird/ lamivudinea | Emtricitabinea-tenofovira alafenamidea, dolutegravird, doravirinec |
| Evidence of pancreas rejection | No | Mild to moderate | No | No | Severe | No | No | No |
| Treatment of rejection | N/A | Thymo, prednisone | N/A | N/A | Thymo, prednisone | N/A | N/A | N/A |
| Currently insulin independent | Yes* | No* | Yes | Yes* | Yes | Yes | Yes | Yes |
| Follow-up (months) | 48* | 126* | 99 | 65* | 92 | 44 | 27 | 7 |
| Most recent HbA1c | 4.4* | 6.5* | 5.9 | 5.9* | 5.4 | 4.6 | 3.9 | 4.5 |
| O.N. or O.I. | None* | BK viremia | None | None* | BK viremia, UTI | BK viremia | BK viremia | None |
At the time of death
nucleotide reverse transcriptase inhibitor,
protease inhibitor,
non-nucleotide reverse transcriptase inhibitor,
integrase inhibitor
O.N. = Opportunistic neoplasm; O.I. = Opportunistic infection
Our early HIV-positive kidney alone transplant recipients were placed on cyclosporine (CSA), as there were early reports CSA may be a good drug to suppress HIV. In fact, there were early trials to test CSA as a strategy to control HIV. As a result the IAK patients were on CSA instead of tacrolimus.
3. Results
3.1. Pancreas solid organ transplant recipients
The mean age at the time of pancreas transplantation was 43 years, ranging from 33 to 52 years; seven of eight recipients were male. The average time from initiation of exogenous insulin to transplantation was 24 years. Seven of eight patients underwent simultaneous kidney and pancreas transplant (SPK), and one received a PAK (Table 1), and the donor characteristics are similar to what one would expect for deceased pancreas donors (Table 2). All donors were HIV-negative. A single donor was HCV antibody positive, but HCV nucleic acid amplification testing was negative. The average follow-up for the eight patients was 5.3 years, ranging from 0.6 to 10.6 years. Induction therapy with thymoglobulin resulted in profound lymphocyte depletion, but CD4 counts recovered by one year after transplant (Figure 1), although they did not return to pre-induction levels. One patient (P#7) had evidence of a “blip” in HIV. This patient had a detectable HIV viral load 9 months after transplant, but on subsequent testing the HIV viral load was negative in the absence of any changes in cART or immunosuppression. Two patients, P#1 and P#7, were transitioned to an mTOR inhibitor due to intolerance of calcineurin inhibitor, and intolerance of MMF, respectively.
Table 2.
Donor Characteristics
| Donor | P#1 | P#2 | P#3 | P#4 | P#5 | P#6 | P#7 | P#8 | I#1 | I#2 | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organ Type | SPK | SPK | SPK | SPK | PAK | Kidney | SPK | SPK | SPK | IAK #1 | IAK #2 | IAK #1 | IAK #2 |
| Age | 27 | 18 | 37 | 21 | 15 | 34 | 19 | 21 | 20 | 22 | 30 | 50 | 52 |
| Gender | Male | Male | Male | Male | Male | Male | Male | Male | Male | Male | Male | Male | Male |
| Height (cm) | 188 | 187 | 174 | 180 | 173 | 181 | 175 | 183 | 171 | 180 | 183 | 188 | 178 |
| Weight (kg) | 86.4 | 77.8 | 73.6 | 65.8 | 70.6 | 71.8 | 81.6 | 80.5 | 62.0 | 97.5 | 140.5 | 95.5 | 90.0 |
| BMI (kg/m2) | 24.5 | 22.5 | 24.3 | 20.3 | 23.6 | 21.9 | 26.6 | 24.0 | 21.2 | 30.1 | 42.0 | 27.0 | 28.4 |
| HTN (yes/no) | no | no | no | no | no | no | no | no | no | no | no | no | Yes |
| DM (yes/no) | no | no | no | no | no | no | no | no | no | no | no | no | no |
| Creatinine (mg/dl) | 1.6 | 1 | 0.9 | 1 | 0.9 | 0.76 | 0.74 | 0.9 | 0.7 | 1.1 | 1.5 | 0.9 | 1.1 |
| Hep C status | False positive* | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative |
| HIV status | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative | negative |
| pDRI | 0.86 | NA** | 1.51 | NA** | 0.73 | 0.80 | 0.80 | 0.79 | |||||
| KDPI | 13% | 1% | 31% | 6% | NA | 2% | 2% | 6% | |||||
| Donor type | Deceased | Deceased | Deceased | Deceased | Deceased | Living related | Deceased | Deceased | Deceased | Deceased | Deceased | Deceased | Deceased |
| DCD (yes/no) | no | no | no | no | no | NA | no | no | no | no | no | no | no |
| Cause of death | Head trauma | Head trauma | CVA | Head trauma | CVA | NA | Head trauma | Head trauma | Anoxia | Head trauma | Head trauma | Head trauma | Head trauma |
| CIT Kidney (min) | 766 | 665 | 678 | 731 | 26 | 583 | 885 | 645 | |||||
| WIT Kidney (min) | 29 | NA** | 40 | 29 | 22 | 29 | 97 | 21 | |||||
| CIT Pancreas (min) | 609 | NA** | 600 | NA** | 427 | 476 | 800 | 549 | 435 | 360 | 360 | 300 | |
| WIT Pancreas (min) | 21 | NA** | 30 | 20 | 21 | 25 | 80 | 26 | |||||
pDRI= Pancreas donor risk index
Donor tested positive for Hepatitis C on serologic testing but was negative on nucleic acid amplification testing
Data are not available due to the remote timeframe
Figure 1.
Box plot of absolute CD4 counts (cells/mL). Multiple time points both pre- and post-transplant are shown. The middle bar indicates the median value, and lower and upper hinges represent the 25th to 75th interquartile range, respectively.
The average length of stay after transplant was 10.8 days, and the perioperative complications are described in Table 1. Four patients had malacotic stools and were transfused packed red blood cells during the index hospitalization. Patient P#1 was the only patient that required reoperation, and this was for bleeding two days after transplant. No patients required abdominal reoperation or percutaneous drainage for peri-pancreatic fluid collections. Three patients were re-admitted in the first month, P#1 for CSA toxicity, P#6 with dehydration, and P#7 for a peri-pancreatic fluid collection that was treated with antibiotics. Patient P#8 developed partial thrombosis in the splenic vein of the allograft the day after transplant. Ultrasound demonstrated persevered diastolic flow in the pancreatic parenchyma, and the graft was functioning well, so this patient was treated with anticoagulation and the thrombus resolved within a month. This patient also suffered worsening of peripheral vascular disease and right toe ischemia 2 months after transplant and eventually required right transmetatarsal amputation. Patient P#5 was readmitted 3 months after transplant with an uncomplicated urinary tract infection (UTI).
There were two episodes of biopsy-proven pancreas rejection in two patients with clinical suspicion of rejection (escalation in amylase/lipase, and/or swelling of allograft on ultrasound). Patient P#2 had a moderate acute cell-mediated rejection (grade II) episode 11 months after transplant treated with of thymoglobulin (6 mg/kg), prednisone bolus with taper, and a continued maintenance therapy of tacrolimus and sirolimus. This patient had excellent pancreas function for many years but returned to insulin therapy 7.5 years after transplant. This patient also experienced an episode of biopsy-proven acute mixed rejection (grade I) of the kidney allograft nine years after transplant, and eventually returned to hemodialysis before passing away. Patient P#5, who received a pancreas transplant 15 months after his kidney transplant, experienced an episode of biopsy-proven severe acute cell-mediated rejection (grade III) of the pancreas allograft one month after transplant that was treated with thymoglobulin and a steroid taper. This patient had no additional rejection episodes and has ongoing excellent pancreas graft function 7.6 years after transplant. None of the patients developed de novo donor specific antibody, and no patients were diagnosed with antibody mediated rejection.
The most recent hemoglobin A1c (HbA1c) levels in the ten patients range from 3.9% −7.6% (mean 5.1%). The average length of insulin independence to date is 4.9 years. Only patient P#2, described above, had pancreas graft failure during the study period at 7.5 years. Table 3 shows overall patient and pancreas graft survival. One-year and three-year patient and pancreas graft survival was 100% for recipients that reached these end points. Five year patient and pancreas graft survival was 80% (4/5) and 100% (4/4), respectively, for patients that reached the end point.
Table 3.
Pancreas solid organ recipient and death censored graft survival
| 1 year survival* | 3 year survival* | 5 year survival* | |
|---|---|---|---|
| Patient survival | 7/7 (100%) | 6/6 (100%) | 4/5 (80%) |
| Pancreas graft survival | 7/7 (100%) | 6/6 (100%) | 4/4 (100%) |
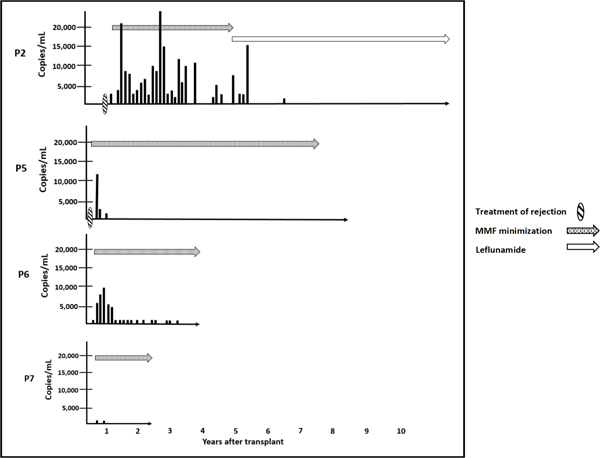
A total of 4 opportunistic infections (OI) were identified, all of which were BK viremia (Figure 2). All patients were treated with lowering of immunosuppression doses (mycophenolate mofetil (MMF) minimization); in addition, patient P#2 was also switched from MMF to Leflunomide 20mg maintenance daily. Patient P#2 experienced BK viremia following treatment for pancreas rejection 11 months after transplant, with BK virus levels peaking 2.6 years after transplant at 25,780 copies/mL. This patient had excellent graft function for years after the BK viremia resolved, but eventually returned to hemodialysis 8 years later and died from an intracerebral hemorrhage more than 10 years after transplant. Patient P#5 also developed asymptomatic BK viremia following treatment for pancreas rejection 1 month after transplant with a serum BK level of 12,614 copies/mL 3 months after transplantation. Patients P#6 and P#7 developed asymptomatic BK viremia (9,220 copies/mL 4 months, and <1,000 copies/mL at 4 months after transplant, respectively) without a preceding rejection episode. There was no biochemical evidence of BK nephropathy in any patient. The absolute CD4 count at the time of detectable BK viremia for patients in this study group was mean (range) 38.3 (13–57). In addition to BK viremia, patient P#5 experienced a recurrence of squamous cell carcinoma of the scalp. No other neoplasms were identified in any of the patients. There was no progression of HIV based on stable CD4 counts and continued undetectable HIV virus in all patients.
Figure 2.
Serum BK virus levels in the four patients with this opportunistic infection.
Three patients (P#1, P#2, P#4) passed away during the study period, as a result of cardiovascular events. Two of these three patients (P#1, P#4) had functioning renal allografts and were insulin-dependent. None of the deaths were considered to be a postoperative complication for the purposes of this analysis.
3.2. Islet recipients
The mean age at the time of the first islet infusion was 50 years, (range: 48 to 52 years), and both recipients were male. The average time from initiation of exogenous insulin to transplantation was 31 years. Both patients underwent kidney transplant prior to islet infusion. IAK was performed 6 years after living donor kidney transplant in patient I#1, and 7 years after deceased donor kidney transplantation in patient I#2. In both cases, the indication was severe hypoglycemic unawareness combined with cardiovascular risk factors that precluded solid organ pancreas transplantation. The average follow-up was 4.8 years, ranging from 4.6 to 4.9 years (Table 4).
Table 4.
IAK recipients
| I#1 | I#2 | |
|---|---|---|
| Age at first infusion (years) | 51 | 47 |
| Gender | Male | Male |
| Height (cm) | 183.0 | 160.0 |
| Weight (kg) | 77.1 | 65.8 |
| BMI (kg/m2) | 23.1 | 25.7 |
| Previous transplant | Yes (kidney) | Yes (kidney) |
| cPRA | NA | NA |
| Length of pre-transplant DM1 (years) | 27 | 35 |
| Type of kidney transplant | Living unrelated | Cadaveric |
| Years between kidney transplant and first IAK infusion | 6 | 7 |
| First beta cell infusion | ||
| Total IEQ (Islets/kg body weight) | 459,290 (5,957) | 682,760 (10,376) |
| Induction | Thymo (3mg/kg) | Thymo (3mg/kg) |
| Immunosuppression regimen at the time of infusion | Cyclosporine, prednisone | Sirolimus, prednisone |
| Evidence of beta cell rejection | No | Yes |
| Second beta cell infusion | ||
| Total IEQ (Islets/kg body weight) | 711,070 (9,223) | 375,860 (5,712) |
| Reason for second infusion | Ongoing insulin requirement | Ongoing insulin requirement |
| Induction | Daclizumab | Basiliximab |
| Immunosuppression regimen at the time of second infusion | Cyclosporine, prednisone | Cyclosporine, sirolimus, prednisone |
| Most recent immunosuppression regimen | Cyclosporine, prednisone* | Sirolimus, prednisone* |
| Evidence of beta cell rejection | No | Yes |
| Treatment of rejection | N/A | None |
| Currently insulin independent | Yes* | No* |
| Follow-up (months) | 52.0* | 59.3* |
| Most recent HbA1c | 6.0* | 7.6* |
| O.N. or O.I. | None* | Squamous cell carcinoma of the ear |
At the time of the patient’s death
O.N. = Opportunistic neoplasm; O.I. = Opportunistic infection
IAK recipients received thymoglobulin for a total of 3 mg/kg prior to their initial islet infusion. Both IAK patients received second islet infusions with basiliximab or daclizumab induction. Immunosuppression regimens at the time of the first infusion and most recent maintenance immunosuppression regimens are listed in Table 4. Postoperatively, cART remained unchanged for each patient and prophylaxis against opportunistic infections was the same as the pancreas recipient group.
Patient I#1 was on 15 units of long acting insulin and 4–10 units of short acting insulin with meals prior to the first infusion of 459,290 purified IEQ. His insulin requirement fell after the first transplant to zero long acting insulin and 5–7 units of short acting insulin per day. Seven weeks later he underwent a second infusion of 682,760 IEQ that resulted in insulin independence. A mixed meal tolerance test 1 year after the second infusion showed a fasting c-peptide of 3.3 ng/mL, and stimulated peak of 8.3 ng/mL. He was insulin independent at the time of his death 52 months after the initial islet infusion. Patient I#2 was on 30 units of insulin per day prior to the first infusion of 711,070 IEQ. His insulin requirement fell after the first transplant to 8 units per day. Nine weeks later he underwent a second infusion of 375,860 IEQ. After the second infusion he remained on spot dosing of insulin up to 4 units per day for 8 weeks, and his c-peptide ranged from 1.5 to 2.4 ng/mL over that time. He did not have a mixed meal tolerance test. Then, his insulin requirement gradually increased up to 20 units per day, and his c-peptide fell to <0.01 ng/mL 10 months after the initial islet infusion, and he remained on insulin with undetectable c-peptide until he died 59 months after the initial islet infusion, so this patient was therefore considered to have beta cell graft failure. The most recent HbA1c levels in the IAK patients ranged from 6.0% −7.6% (mean 6.8%). The average length of insulin independence to date was 2.4 years, ranging from 2.4 – 55.1 months.
There were no postoperative complications in the IAK patients. One opportunistic infection was identified. Patient I#2 developed squamous cell carcinoma during the study period. Both IAK patients, I#1 and I#2, died of cardiovascular events; I#1 had a functioning beta graft at the time of his death. Both patients had functioning renal grafts at the time of their deaths.
4. Discussion
Solid organ transplantation in the HIV-positive recipient is increasingly accepted as both safe and effective. Early outcomes following kidney transplantation one and three year patient/graft survival) were similar to HIV-negative recipients, although two to three fold increases in rejection rates were observed.1 Five year outcomes following liver transplantation in HIV/HBV-coinfected patients were also equivalent, suggesting that if the coinfection is controlled, liver transplantation can have comparable results to HIV-negative recipients.2 With the advent of the new direct acting HCV agents, liver transplantation is now feasible in the challenging cohort of HIV/HCV coinfected liver recipients. Interestingly, high rates of rejection have also been observed in the HIV-infected liver recipients.3 Due to the intense burden of immune suppression required for pancreas transplantation necessary to overcome both the allo- and auto-immune responses, there has been limited enthusiasm for pancreas transplantation in patients with HIV, Type 1 diabetes and renal failure.
The first pancreas transplant in a patient know to be HIV-positive was described in 20034 but such transplants were avoided by many centers due to concerns over severe lymphocyte depletion from thymoglobulin that is typically used as induction therapy for pancreas transplantation.1 The literature contains scattered reports of HIV-positive patients undergoing pancreas transplantation,5–9 with the largest being a case series of 4 HIV-positive SPK recipients with short term follow up. In that small series,6 all four patients experienced postoperative infections (median 30 days, range 9–128), with two of the four patients requiring re-laparotomy for surgical and infectious complications. Of the four patients, one experienced treatable acute rejection of the kidney, and another developed pancreatic graft failure 64 months after transplant.
In the eight pancreas transplant recipients presented here, surgical complications, specifically bleeding and splenic vein thrombosis were seen at approximately the expected rate. Thrombosis in the splenic vein resolved with anticoagulation, as expected.10 No patients required reoperation or percutaneous drain placement for intra-abdominal infection or leak. With an average follow-up of 5.3 years, there were two episodes of biopsy-proven acute rejection of the pancreas allograft and one of the kidney allograft. The higher incidence of rejection reported in kidney and liver recipients was, fortunately, not observed in this series, and we feel preventing rejection is a critical in HIV-infected patients because they are at high risk of infectious complications following the treatment of rejection. The CD4 counts in the patients presented here recovered at a similar pace as HIV-positive patients given thymoglobulin for kidney transplant.11 This is markedly prolonged compared to the HIV-negative recipients.12 It was reassuring to see again that HIV viral breakthrough was not a significant issue in this patient population. A single patient had a detectable HIV viral load 9 months after transplant, and he subsequently cleared the virus without any changes in cART or immunosuppression, similar to what is seen in the majority of HIV-positive solid organ recipients – excellent viral suppression and only rare cases where immunosuppression has to be changed.
In HIV-infected patients consideration of drug-drug interactions is critical to reduce the risk of rejection. Protease inhibitors are largely cleared by the same cytochrome P450 enzymes as calcineurin inhibitors, even when used in low doses, making dosing of calcineurin inhibitors very challenging. Several centers have found that antiretroviral regimens based on integrase inhibitors were associated with lower rejection rates, presumably due to much greater ease of achieving effective and stable maintenance immunosuppressive coverage in patients not taking protease inhibitors. Our anti-retroviral regimen has evolved in recipients of all HIV-positive solid organ transplants to avoid protease inhibitors based on these problematic drug-drug interactions. In this series of pancreas transplant recipients, we shifted to a non-protease inhibitor based regimen after the 3rd patient, and only two patients were on protease inhibitor therapy at the time of transplant. This strategy, in part, may be responsible for the lower rates of rejection noted in this series, and we would recommend a switch to integrase-based regimen prior to transplantation. Of course, this switch requires oversight by an experienced HIV provider to assure that control of the HIV virus can be maintained. Of the eight pancreas transplant recipients in our series, only four patients experienced infections (one UTI requiring hospitalization and four cases of BK viremia). All four patients were managed with reduction of MMF. BK viremia occurs in approximately 35% of non HIV-infected pancreas transplant recipients,13 but published data on BK viremia in HIV-infected transplant recipients is sparse. An international review suggested the risk of OI in HIV-infected liver or kidney transplant recipients to be 5–10%, but it is not clear whether or not BK viremia was considered an OI in those studies.14 Our findings suggest that BK viremia is not significantly more common in HIV-infected pancreas recipients. Moreover, BK nephropathy in these patients is rare, which is an important point because the risk of renal allograft loss from BK nephropathy is significant (43–89%).15 Two of our four patients with BK viremia developed it after being treated for rejection, and it is also interesting to note the CD4 counts at the time of BK viremia in our four patients were low.
Two patients had cardiovascular risk factors that precluded solid organ pancreas transplantation. For IAK, we have elected to use an insulin requirement cut off of 55 units/day, slightly stricter than the commonly used 1 unit/kg/day, as we have had challenges in rendering recipients insulin independent with islet transplantation when insulin requirements were above that level. Both of these patients required two infusions of islets to achieve insulin independence. Only one patient remained insulin independent for the long term, and this probably is due in part to the inefficient engraftment that is commonly observed following islet infusion. Nonetheless, it is interesting that both recipients were free of hypoglycemic events, even though insulin was reinitiated in one of the two recipients. In addition to the beta cell toxicity of immunosuppression and cART regimens, insulin independence by islet transplantation alone is limited by islet engraftment when infused into the portal system. The limitations of the surgical procedure as well as the toxicity of the immunosuppression and cART regimens could be responsible for the inability of patient I#2 to achieve insulin independence. It is challenging to achieve long term insulin independence in large men with islet transplantation using current state of the art for islet transplantation. Both of these recipients died from a cardiovascular event, reinforcing our decision not to proceed with solid organ pancreas transplantation based on cardiovascular risk factors. It also highlights the cardiovascular comorbidities seen in patients with HIV on cART compounded by history of diabetes and immunosuppressive medications.
As medical therapies for Type 1 diabetes improve, and Type 2 diabetes continues to become more common, we must consider the possibility of beta cell replacement in patients with HIV and Type 2 diabetes. To date, we have not encountered such a patient, but we see no absolute contraindications to solid organ pancreas transplantation. Currently, we are performing SPKs in people with Type 2 diabetes if their insulin requirements are < 1unit/kg/day and BMI <30. At this point, we would not entertain islet cell infusions, as it is unlikely that enough beta cell mass could be provided to overcome the insulin resistance. This is no different than our strategy for HIV-negative people with Type 2 diabetes. The ratio of male to female patients presented here is likely a result of the demographics of the population of patients with HIV in our area.
In conclusion, this report demonstrates that HIV-positive people with Type 1 diabetes can safely undergo pancreas and kidney transplantation with good long term graft survival. Cardiovascular morbidity in the population remains high. If cardiovascular risks preclude solid organ transplantation, islet transplantation can be considered to prevent hypoglycemic unawareness. Strategies to avoid protease inhibitors to decrease rejection rates are preferred, since treating rejection increases the risk of complications in this population. Lymphocyte depletion after induction immunotherapy is profound and recovers slowly, but few opportunistic infections or neoplasms developed with over 5 years of follow up in these patients. Although BK viremia was observed in one third of the patients, this frequently followed the treatment of rejection. The rate of BK viremia does not differ significantly from the expected rate in non HIV-infected pancreas transplant recipients, and BK nephropathy was not seen. Early detection of BK viremia followed by modulation of immunosuppression provided resolution of the viremia without deterioration in kidney function.
Acknowledgments / Funding
There was no external funding for this work. The authors would like to acknowledge all of the patients with HIV who allowed us to care for them over the years, and the nurses and coordinators who cared for these patients; Laurie Carlson and Bruncelia (Cely) Hynson. Arya Zarinsefat is supported by the Filling a Void of Research Training for Transplant Surgeons (FAVOR) T32 (National Institute of Allergy and Infectious Diseases) grant (T32AI125222).
Abbreviations:
- (BK)
polyoma virus
- (cART)
combined antiretroviral therapy
- (CSA)
cyclosporine
- (HbA1c)
Hemoglobin A1c
- (HIV)
human immunodeficiency virus
- (IAK)
islets after kidney transplant
- (OI)
opportunistic infections
- (MMF)
mycophenolate mofetil
- (PAK)
pancreas after kidney transplant
- (SPK)
simultaneous kidney and pancreas transplant
- (UTI)
urinary tract infection
Footnotes
Data Availability Statement:
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Disclosure
The authors of this manuscript have no conflicts of interest to disclose as described by the American Journal of Transplantation.
References
- 1.Stock PG, Barin B, Murphy B, et al. Outcomes of Kidney Transplantation in HIV-Infected Recipients. N Engl J Med. 2010;363(21):2004–2014. doi: 10.1056/NEJMoa1001197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Coffin CS, Stock PG, Dove LM, et al. Virologic and Clinical Outcomes of Hepatitis B Virus Infection in HIV-HBV Coinfected Transplant Recipients. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2010;10(5):1268–1275. doi: 10.1111/j.1600-6143.2010.03070.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Terrault NA, Roland ME, Schiano T, et al. Outcomes of Liver Transplantation in HCV-HIV Coinfected Recipients. Liver Transpl. 2012;18(6):716–726. doi: 10.1002/lt.23411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Toso C, Berney T, Oberholzer J, et al. Kidney-Pancreas Transplantation in a Long-Term Non-Progressor HIV-Infected Recipient. Am J Transplant. 2003;3(5):631–633. doi: 10.1034/j.1600-6143.2003.00119.x [DOI] [PubMed] [Google Scholar]
- 5.Akhtar MZ, Patel N, Devaney A, et al. Simultaneous pancreas kidney transplantation in the HIV-positive patient. Transplant Proc. 2011;43(10):3903–3904. doi: 10.1016/j.transproceed.2011.08.093 [DOI] [PubMed] [Google Scholar]
- 6.Grossi PA, Righi E, Gasperina DD, et al. Report of four simultaneous pancreas-kidney transplants in HIV-positive recipients with favorable outcomes. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2012;12(4):1039–1045. doi: 10.1111/j.1600-6143.2011.03906.x [DOI] [PubMed] [Google Scholar]
- 7.Genzini T, Noujaim HM, Mota LT, et al. Simultaneous pancreas-kidney transplantation in a human immunodeficiency virus-positive recipient: a case report. Transplant Proc. 2010;42(2):591–593. doi: 10.1016/j.transproceed.2010.01.007 [DOI] [PubMed] [Google Scholar]
- 8.Miro JM, Ricart MJ, Trullas JC, et al. Simultaneous pancreas-kidney transplantation in HIV-infected patients: a case report and literature review. Transplant Proc. 2010;42(9):3887–3891. doi: 10.1016/j.transproceed.2010.09.003 [DOI] [PubMed] [Google Scholar]
- 9.Mittal S, Choudhary P, Callaghan C, Hilton R, Drage M. First case of pancreas transplant alone in a patient with diabetes and HIV infection. Int J STD AIDS. 2016;27(14):1350–1353. doi: 10.1177/0956462416645996 [DOI] [PubMed] [Google Scholar]
- 10.Harbell JW, Morgan T, Feldstein VA, et al. Splenic Vein Thrombosis Following Pancreas Transplantation: Identification of Factors That Support Conservative Management. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2017;17(11):2955–2962. doi: 10.1111/ajt.14428 [DOI] [PubMed] [Google Scholar]
- 11.Carter JT, Melcher ML, Carlson LL, Roland ME, Stock PG. Thymoglobulin-associated Cd4+ T-cell depletion and infection risk in HIV-infected renal transplant recipients. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2006;6(4):753–760. doi: 10.1111/j.1600-6143.2006.01238.x [DOI] [PubMed] [Google Scholar]
- 12.Gurkan S, Luan Y, Dhillon N, et al. Immune reconstitution following rabbit antithymocyte globulin. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2010;10(9):2132–2141. doi: 10.1111/j.1600-6143.2010.03210.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Westphal SG, Lyden ER, Langewisch ED, Miles CD. BK viremia surveillance and outcomes in simultaneous pancreas-kidney transplant recipients. Clin Transplant. 2017;31(8):e13010. doi: 10.1111/ctr.13010 [DOI] [PubMed] [Google Scholar]
- 14.Miro JM, Agüero F, Duclos-Vallée J-C, et al. Infections in solid organ transplant HIV-infected patients. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis. 2014;20 Suppl 7:119–130. doi: 10.1111/1469-0691.12754 [DOI] [PubMed] [Google Scholar]
- 15.Mujtaba M, Fridell J, Sharfuddin A, et al. BK virus nephropathy in simultaneous pancreas kidney transplant: a potentially preventable cause of kidney allograft loss. Clin Transplant. 2012;26(2):E87–93. doi: 10.1111/j.1399-0012.2012.01599.x [DOI] [PubMed] [Google Scholar]