ABSTRACT
While COVID-19 has primarily been characterized by the respiratory impact of viral pneumonia, it affects every organ system and carries a high consequent risk of death in critically ill patients. Higher sequential organ failure assessment (SOFA) scores have been associated with increased mortality in patients critically ill patients with COVID-19. It is important that clinicians managing critically ill COVID-19 patients be aware of the multisystem impact of the disease so that care can be focused on the prevention of end-organ injuries to potentially improve clinical outcomes. We review the multisystem complications of COVID-19 and associated treatment strategies to improve the care of critically ill COVID-19 patients.
KEYWORDS: COVID-19, coronavirus, COVID-19 multi-system effects, coronavirus multi-system effects, COVID-19 complications, COVID-19 mechanism of injury, COVID-19 management
Introduction
Coronavirus disease 2019 (COVID-19), the disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first described in Wuhan, China in December 2019 and rapidly evolved into a worldwide pandemic [1]. Of patients affected with the virus, approximately 5% to 14% will become critically ill [2–4]. While COVID-19 generally begins as a respiratory tract infection, it can have damaging effects on every organ system. When the virus does spread systemically, the result is often multisystem critical illness associated with a high risk of death. Higher sequential organ failure assessment (SOFA) scores have been associated with increased mortality in critically ill patients with COVID-19 [5]. It is important that clinicians managing these critically ill patients be aware of the multisystem impact of the disease so that care can be focused on the prevention of end-organ injuries to potentially improve clinical outcomes. Here, we review the multisystem complications of COVID-19 and treatment strategies to improve the care of critically ill COVID-19 patients.
General pathophysiology
The multisystem manifestations of COVID-19 (Figures 1–3) result from a combination of the direct effects of the viral infection and the indirect effects of the body’s significant inflammatory response to the virus. Post mortem findings in COVID-19 patients show viral elements within endothelial cells, an accumulation of inflammatory cells, and cellular apoptosis in multiple organs [6]. These widespread effects largely reflect the virus’ ability to use the angiotensin-converting enzyme 2 (ACE2) receptors to gain entry into endothelial cells [7]. ACE2 breaks down angiotensin II, a pro-inflammatory factor in the lung, and viral inhibition of this enzyme may also be a contributing factor to the lung injury and multiorgan dysfunction that result from SARS-CoV-2 infection [8]. The indirect effects of the virus result from the host’s response to the viral infection, and are associated with a cytokine storm characterized by very high circulating levels of pro-inflammatory cytokines, including tumor necrosis factor (TNF)-α, interleukins, granulocyte-colony stimulating factor, and chemokines [9]. This hyper-inflammatory response combined with hypercoagulability [10] can lead to venous thromboembolism, further increasing the risk of multisystem failure and mortality.
Figure 1.
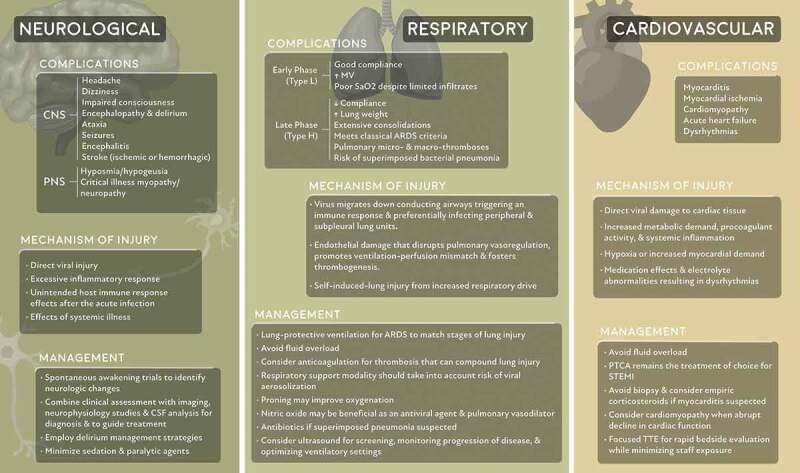
Neurological, respiratory, and cardiovascular system complications and their mechanism of injury and management
Figure 2.
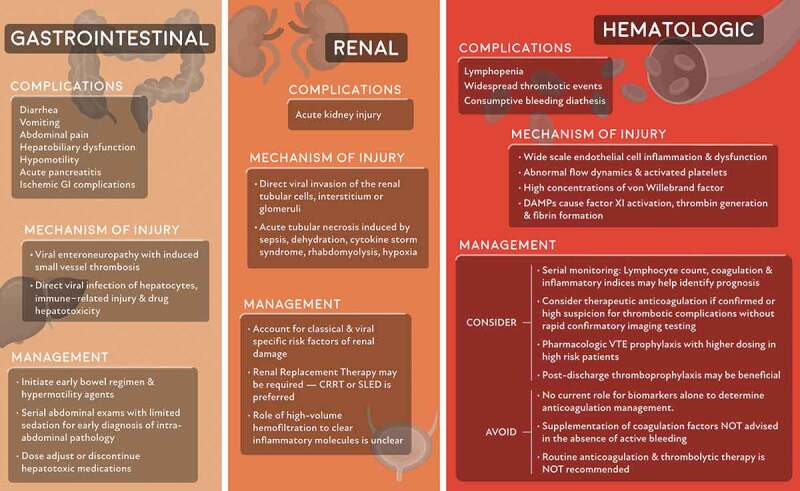
Gastrointestinal, renal, and hematologic system complications and their mechanism of injury and management
Figure 3.

Endocrine, dermatologic, and musculoskeletal system complications and their mechanism of injury and management
Respiratory
The severity of COVID-19-related respiratory disease varies significantly, from mild symptoms requiring minimal oxygen support with nasal cannula to acute hypoxemic respiratory failure requiring intubation and mechanical ventilation. For about 80% of the patients, the disease will be mild and mostly restricted to the upper airways. The remaining 20% of the patients will develop pulmonary infiltrates as the virus reaches the alveoli, where it preferentially infects the peripheral and subpleural units [4].
In an earlier phase of the disease, patients often show preserved lung mechanics with normal compliance, despite having severely impaired oxygenation and increased minute ventilation [11]. There have been a number of phenotypes that have been proposed to characterize COVID-19 pneumonia (i.e. ‘L’ vs ‘H’ phenotype), however, these phenotypes are controversial [12]. On CT imaging, ground-glass opacities are seen in up to 98% of the patients, which commonly suggests interstitial, rather than alveolar, edema. These characteristics have been categorized as ‘type L’ and include a low lung elastance, low recruitability, and a poor response to positive end-expiratory pressure (PEEP). In some patients, the disease progresses into a clinical scenario that resembles typical acute respiratory distress syndrome (ARDS), which may be due to the development of ventilator-induced lung injury, worsening of COVID-19 disease, or bacterial superinfection [13]. These patients are found to have high lung elastance, extensive consolidations on CT imaging, and significant response to higher PEEP. These lung findings are categorized as ‘type H.’ Standard lung-protective ventilation should be used for all stages of the disease, despite the wide spectrum of clinical characteristics.
There has been significant debate regarding the optimal form of respiratory support for non-intubated patients with increasing oxygen requirements and dyspnea. Early intubation has been encouraged to reduce the risk of virus aerosolization and self-induced lung injury. However, early intubation may result in unnecessary intubation of patients who would have otherwise improved with less invasive support [14]. Other strategies that may improve ventilation in these patients include prone positioning and nitric oxide, which is being tested both as an antiviral agent and for its benefits as a pulmonary vasodilator. While prone positioning in awake spontaneously breathing patients is generally well tolerated and has been shown to improve oxygenation and reduce respiratory rate, the effects on clinical outcomes is unclear [15]. The use of noninvasive ventilation and high flow nasal cannula in COVID-19 patients is controversial given the potential risk for aerosolization and exposure to health care workers, and can be minimized when delivered in a negative pressure environment. In-hospital airway management is challenging and we have previously described a practical, stepwise protocol for safe in-hospital airway management in COVID-19 patients [16].
Hematologic system
COVID-19 has profound effects on the hematologic system. Lymphopenia is a characteristic laboratory finding in COVID-19 patients and has prognostic potential in determining severe cases [17]. Throughout the disease course, serial monitoring of lymphocyte count dynamics and inflammatory indices such as lactate dehydrogenase, C-reactive protein, and interleukin-6, may allow identification of patients with poor prognosis and trigger timely intervention. Other biomarkers, including high serum procalcitonin and ferritin, have emerged as poor prognostic factors [5].
Patients with COVID-19 infection present a complicated clinical paradigm as current research has suggested that they are at risk of both impaired hemostasis as well as thrombotic events. Elevated fibrinogen and D-dimer levels are the most common coagulopathy seen in hospitalized COVID-19 patients. While patients with severe COVID-19 infection commonly meet the clinical criteria for disseminated intravascular coagulopathy (DIC), their thrombocytopenia is generally mild, and microangiopathy is often not present. However, given that some patients can develop fulminant DIC with consumption of coagulation factors, platelet count, PT/aPTT, D-dimer, and fibrinogen should be monitored closely. While abnormal coagulation parameters are common in patients with COVID-19 infection, COVID-19 infection seems to be rarely associated with bleeding. Correction of the coagulopathy should only be pursued in patients with active bleeding or those requiring an invasive procedure.
Patients with COVID-19 are also at an increased risk of venous thromboembolic (VTE) disease. VTE disease is likely due to the combined effects of systemic inflammation, abnormal coagulation, multiorgan dysfunction, and critical illness [18]. Multiple studies have shown the correlation between elevated d-dimer and disease severity [5,19,20]. Clinically, d-dimer levels have been followed in patients with COVID-19 infection to determine not only disease severity and clinical progression but also risk of thromboembolic events. While elevated serum d-dimer levels have been associated with increased mortality in COVID-19 patients, there is no current evidence supporting the use of d-dimer levels to guide anticoagulation. Pharmacologic VTE prophylaxis is generally recommended in all hospitalized COVID-19 patients with no specific contraindication. A prospective cohort study that examined the rate of thromboembolic events in 184 ICU patients all receiving standard dosing of VTE prophylaxis and found a 31% incidence of thromboembolic events. Given this finding, the authors suggest increased dosing of prophylaxis medications [21]. The current evidence is limited on the optimal dosing of VTE prophylaxis medication in critically ill patients with COVID-19 and studies to evaluate therapeutic anticoagulation in critically ill patients with COVID-19 are currently ongoing (NCT04359277). Furthermore, post-hospital discharge VTE prophylaxis should be considered in these patients on a case-by-case basis. Assessment for VTE is challenging in these patients, and imaging should only be pursued when clinical examination and condition support the laboratory studies. The decision for treatment with anticoagulation should be made based on imaging findings when possible. A review by Flaczyk et al. compares published guidelines for management of COVID-19 associated coagulopathy in critically ill patients and provides a framework for patient management [22].
Neurological system
Neurologic manifestations have been reported in more than one-third of patients hospitalized with COVID-19 [23]. Potential mechanisms of neurologic injury from COVID-19 include direct viral damage of nervous tissue, injury resulting from the excessive inflammatory response, unintended host immune response effects after the acute infection (e.g., Guillain-Barré syndrome as reported in a case series of four patients [24]), and injury resulting from the effects of systemic illness. Most COVID-related neurologic complications in critically ill patients fall into this latter category, and manifest as encephalopathy, delirium, and critical illness myopathy.
A variety of neurological manifestations have been reported, including headache, encephalitis, stroke, seizures, hyposmia, and hypogeusia [25]. The prevalence of hyposmia and hypogeusia suggests direct viral infection of the olfactory nerve [23] and SARS-CoV 2 has been detected in human neuronal cells on postmortem analysis [26,27]. Several studies have also reported meningoencephalitis manifested by headache, fever, altered mental status, and signs of meningeal irritation as a presenting symptom of COVID-19 that may be secondary to the indirect inflammatory effects of the virus [28–32]. A case of COVID-associated acute necrotizing hemorrhagic encephalitis within the temporal lobes and thalami has also been reported [30]. SARS-CoV-2 has been detected in the CSF of a patient with encephalitis [29]. There have been several cases of young patients presenting with large-vessel strokes [33] and 5.7% of a hospitalized cohort in Wuhan had an acute stroke (80% ischemic, 20% hemorrhagic) [23]. Given the prevalence of hypercoagulability, neurologic thromboembolic events should be strongly considered in a patient with a fluctuating neurologic examination [34]. Careful clinical assessment in conjunction with imaging and cerebrospinal fluid examination may be essential for diagnosing COVID-related neurological disorders.
A significant proportion of patients receive high amounts of sedation to ensure comfort and facilitate ventilator synchrony [35]. Neuromuscular blockade has also been utilized when the effects of sedation are inadequate. Spontaneous awakening trials are vital to alert clinicians to neurologic changes and enable them to take appropriate action. This neurologic assessment is especially important before initiating therapeutic anticoagulation, given the significant morbidity in patients with unidentified intracranial hemorrhage. A growing body of literature is reporting high rates of acute encephalopathy and agitated delirium in critically ill COVID-19 patients [36–39]. Delirium management strategies should be routinely employed, given the prevalence of the disorder in COVID-19 patients.
Cardiovascular system
Between 5% and 25% of the hospitalized COVID-19 patients will have evidence of myocardial involvement [40,41], and preexisting cardiovascular disease has been linked to more severe infections [42]. Cardiovascular complications include infarction, myocarditis, heart failure, and dysrhythmias. There are multiple proposed etiologies for adverse cardiovascular outcomes, including an increased metabolic demand, a hyperinflammatory state, and increased procoagulant activity [42]. There is also evidence that the virus may cause direct damage to the heart via ACE2 receptors located within the cardiac tissue.
Angiotensin II is converted to angiotensin (1–7) by ACE2, and the downregulation of ACE2 among COVID-19 patients has been correlated with increased viral load [43]. Since angiotensin (1–7) has anti-inflammatory and anti-fibrotic effects that counter the pro-inflammatory effects of angiotensin II, there has been concern that patients taking angiotensin-converting enzyme inhibitors or angiotensin receptor blockers might be more susceptible to viral infection and propagation [44,45]. However, the reduction in angiotensin II activity and upregulation of the anti-inflammatory ACE2 effects from these drugs may be beneficial in COVID-19 infection [46]. Many use the lack of evidence of harm in COVID-19 to support the continuation of such medications [45], given the risk of heart failure exacerbation with their withdrawal [47].
Myocardial injury and myocarditis
Elevated troponins have been found in many patients with COVID-19, which may be secondary to increased cardiac physiologic demand, hypoxia, and/or direct myocardial injury. Myocarditis has also been identified on autopsy of some patients with COVID-19 [48,49] and presents with variable clinical severity in COVID-19 patients. Fulminant viral myocarditis [48,50] or diffuse lymphocytic infiltrates characteristic of viral myocarditis [43,51] are uncommon findings in COVID-19 patients. The patients who have developed COVID-19 myocarditis are typically younger and healthier, often without underlying cardiovascular disease. Myocarditis may result from a cytokine storm in patients who mount a vigorous immune response to the virus.
Differentiating myocarditis from acute coronary syndrome (ACS) can be challenging [52]. Serum troponin values will be elevated in both conditions, and electrocardiogram (ECG) in patients with myocarditis can demonstrate a range of findings, in some cases mimicking ACS. Echocardiographic evaluation is more likely to show global dysfunction with myocarditis, while a focal wall motion abnormality is more suggestive of ACS. ECG and echocardiographic abnormalities are markers of severity in COVID-19 patients, and are correlated with worse outcomes; moreover, troponin elevations in severe COVID-19 patients have been directly associated with an increased risk of mortality. Diagnostic confirmation of COVID-19 myocarditis via biopsy is unlikely to change management and therefore is not generally recommended. Instead, it may be reasonable to empirically treat COVID-19 patients with suspected myocarditis with a course of corticosteroids.
Acute myocardial infarction
Given the hyperinflammatory and hypercoagulable state, COVID-19 patients may be at increased risk for acute myocardial infarction. Patients should be directed toward appropriate standard of care therapy [53] while also avoiding unnecessary and costly procedures that can both lead to morbidity and increase the risk of infectious exposure to staff.
Acute heart failure and cardiomyopathy
Approximately a quarter of patients presenting with COVID-19 will develop acute heart failure, with the majority lacking a prior diagnosis of hypertension or cardiovascular disease. It is unclear if heart failure in these patients is a new cardiomyopathy or an exacerbation of an undiagnosed condition. Given the high prevalence of cardiac dysfunction and concern for causing pulmonary edema, fluids should be administered judiciously in COVID-19 patients.
A diagnosis of stress cardiomyopathy should be considered in any critically ill COVID-19 patient with an abrupt decline in cardiac function. Focused echocardiography can be performed at the bedside to allow for rapid evaluation while minimizing exposure to additional staff. Typical echocardiographic findings include impaired systolic function with global hypokinesis or regional wall motion abnormalities that are not limited to a coronary distribution [54]. In stress cardiomyopathy, systolic dysfunction is typically transient, with most patients recovering within days to weeks. However, due to ongoing infection and respiratory compromise, the prognosis for COVID-19 patients who develop stress cardiomyopathy is often poor. Management is mostly supportive, and the use of mechanical circulatory support should be considered for patients in whom myocardial recovery is likely.
Pediatric multi-system inflammatory syndrome
A ‘pediatric multi-system inflammatory syndrome temporally associated with COVID-19’ (PIM-TS) syndrome has been described in children, which shares certain features of toxic shock syndrome and atypical Kawasaki disease and has been linked to a number of deaths and rapid decompensation in children [55]. Common features include rash, fever, gastrointestinal symptoms, and severe cardiac dysfunction from myocarditis and cardiogenic shock. Repeat echocardiography, cardiac catheterization, supportive treatment, steroids, and IVIG may be required.
Gastrointestinal system
Gastrointestinal (GI) manifestations of COVID-19 are common and include diarrhea, vomiting, and abdominal pain. The SARS‐CoV-2 RNA can be readily detected in stool specimens, even where respiratory tests are negative [56]. SARS‐CoV-2 has a tropism for the GI tract, with the viral ACE2 receptor found on gastrointestinal epithelial cells. Critically ill patients with COVID-19 are at high risk of hepatobiliary, hypomotility, and ischemic GI complications [57], with small vessel thrombosis and viral enteroneuropathy as likely etiologic factors. Consequently, serial abdominal exams with limited sedation are required when there is clinical suspicion of intra-abdominal pathology.
Liver injury occurs in 15% to 78% of COVID-19 patients, with the most common finding being abnormal transaminase levels [58,59]. Most liver injuries are mild and transient, but more severe liver damage can occur and is more likely in patients with severe COVID‐19 infection. Mechanisms of liver injury may include direct viral infection of hepatocytes, immune‐related injury, and drug hepatotoxicity. In patients with transaminitis, medications with potential hepatotoxicity, including acetaminophen, statins, and hydroxychloroquine, should be adjusted or discontinued.
Renal system
Acute kidney injury (AKI) is a common complication of COVID-19 infection, occurring in 0.5–15% of hospitalized COVID-19 patients [5], and up to 23% of critically ill patients [60,61]. The median onset of AKI from hospitalization ranges from 7 [62] to 15 days [5], and it has been identified as an independent risk factor for morbidity and mortality [62]. AKI can occur through several proposed mechanisms, including acute tubular necrosis induced by sepsis, fluid restriction, rhabdomyolysis, or hypoxia. Furthermore, intrinsic tissue injury by direct viral invasion of the renal tubular cells, interstitum, or glomeruli has also been proposed. Varying degrees of acute tubular necrosis, lymphocytic infiltration, and viral RNA have been found on postmortem examination of COVID-19 patients, suggesting the direct invasion of kidney tubules [63].
Management of AKI in COVID-19 patients must account for classical and viral-specific risk factors of renal damage. In a recent study, approximately 30–40% of critically ill patients with AKI required renal replacement therapy (RRT) [34,61]. The preferred modality is continuous RRT or sustained low-efficiency dialysis, although the American Society of Nephrology notes that whatever is available in these resource-scarce times will suffice. If the AKI is secondary to cytokine storm, some have questioned whether using high-volume hemofiltration would be preferred to clear inflammatory molecules, but current evidence shows no difference in outcome compared to standard volume filtration [18].
Endocrine system
While the effects of COVID-19 on the endocrine system remain largely unknown, given the expression of ACE2 on the majority of endocrine glands, dysfunction of these systems should be considered in critically ill patients.
Pancreas
The ACE2 receptor is expressed throughout the pancreas and dysfunction of both the exocrine and endocrine systems is seen in patients with COVID-19 infection. While pancreatitis is uncommon, elevated lipase or amylase have been seen in up to 17% of the patients with severe disease [64]. Similar to other infections, patients with diabetes mellitus (DM) are more at risk for COVID-19 than the general population and are more likely to have a severe course due to compromised innate immunity and downregulated ACE2 levels [18]. COVID-19 patients with DM have higher serum levels of inflammatory biomarkers and are more susceptible to cytokine storm [18]. Furthermore, infection with SARS-CoV-1 was shown to cause new-onset diabetes in approximately 8% of the patients during hospitalization that may not be classic type 1 or type 2 diabetes but a new type of diabetes [65,66]. In some patients, hyperglycemia persisted for 3 years after recovery from the virus. While a similar effect has not yet been reported in COVID-19, close monitoring of blood glucose during the acute and convalescent phase of the illness is indicated.
Obesity
Obesity is associated with severe COVID-19, which may be explained by ACE2 expression in adipose tissue. Furthermore, visceral and subcutaneous adipose tissue produces pro-inflammatory cytokines that are found in greater abundance in obese patients with COVID-19 as compared to non-obese patients [67]. This may predispose obese patients with COVID 19 to an exaggerated cytokine response in the presence of SARS-CoV-2, manifesting as more severe disease and ARDS [68].
Hypothalamus–pituitary-adrenal gland
Hypothalamic and pituitary tissues express ACE2 and given the evidence of viral injury of nervous tissue, it is reasonable to assume that SARS-CoV-2 may affect the hypothalamus–pituitary as well, either directly or via immune-mediated hypophysitis [69]. Accordingly, central hypocortisolism should be suspected in critically ill COVID-19 patients with unexplained hypotension or shock. Diabetes insipidus is common in patients with pituitary–hypothalamic disorders and, in conjunction with insensible water loss from fever and the conservative fluid management strategy employed in critically ill COVID patients, could result in hypovolemia and hypernatremia.
Impairment of the host’s cortisol stress response was a primary immunologic strategy utilized by SARS-CoV-1 for facilitating viral spread [70]. The similarities between the two viral strains suggest that SARS-CoV-2 may employ the same strategy. Therefore, patients with severe COVID-19 may be more prone to develop critical illness-related corticosteroid insufficiency. The absence of lymphopenia in patients with COVID-19 could be used as a marker of hypocortisolism (absolute or relative) and clinicians may want to adopt a low threshold for initiating glucocorticoid therapy in the presence of shock.
Thyroid
Data regarding the impact of COVID-19 on thyroid function are limited. A case report suggests that COVID-19 may lead to subacute thyroiditis in some patients, which is suspected to have viral or postviral origin [71]. Recent guidelines advise patients with underlying hypothyroidism or hyperthyroidism to continue prescribed medications since uncontrolled thyroid disease may increase the risk for viral infection and complications [72].
Dermatologic
A variety of cutaneous manifestations such as erythematous rash, generalized urticaria, and chickenpox-like lesions have been associated with COVID-19, and can present even before the onset of symptoms and with an incidence of up to 36% of the patients depending on the cutaneous manifestation [73,74]. Interestingly, there was no association between cutaneous findings and disease severity [74]. Urticarial eruptions typically preceded additional symptoms of COVID-19 infection, were noted to occur even in the absence of fevers, and tended to be consistent on histology with a viral exanthem [53,75,76]. Acrocyanosis and limb ischemia have been described in a cohort of critically ill patients with elevated D-dimer, fibrinogen, and fibrinogen degradation products [77]. In this small cohort, 57% of the patients developed DIC. Livedo reticularis, a cutaneous manifestation commonly associated with DIC, was seen in two patients with only mild-moderate disease [78]. The authors hypothesize that these findings may represent microthrombosis of cutaneous vasculature, as similar pathophysiology has been noted in other organ systems in COVID-19 patients. Chilblains-like lesions, which are erythematous areas on the feet, described colloquially as ‘COVID toes,’ may represent endothelitis secondary to systemic COVID-19. In a small cohort of patients with severe COVID-19 and purpuric skin rash, biopsy demonstrated a thrombogenic vasculopathy of both affected and normal-appearing skin as well as localization of SARS-CoV-2 spike glycoprotein causing complement activation [79]. These findings are consistent with a catastrophic microvascular injury mediated by systemic viral spread and associated with a procoagulant state.
Current evidence does not suggest that dermatologic findings are associated with disease severity, but may reveal microthrombosis, hypercoagulability, and DIC. In the correct clinical setting, these findings may encourage providers to consider therapeutic anticoagulation.
Musculoskeletal
Myalgias are a common presenting symptom of COVID-19 occurring in more than one-third of patients [62,80–82]. Elevated creatinine kinase levels are prevalent in hospitalized patients [80] and are more common in patients with severe disease [3,80]. While myositis and rhabdomyolysis was documented as a manifestation of SARS-CoV-1 infection, RNA from SARS-CoV-2 has not been found within myocytes to date [26]. Critically ill patients in general are at risk of developing myopathy and neuropathy due to prolonged immobility, systemic inflammation, corticosteroids, and the use of neuromuscular blocking agents [83,84]. Early mobilization and initiation of physical therapy is important in these patients to ensure the best functional outcome.
Conclusion
While COVID-19 generally starts as a local upper respiratory tract infection, it can spread to affect multiple organ systems with significant morbidity and mortality. Clinicians should actively seek evidence of multiorgan system involvement in these patients to guide early management to potentially improve outcomes. The long-term implications of COVID-19 are only beginning to be appreciated, but will likely include cognitive, physical, and psychological impairment.
Authors’ contributions
GWD, LEG, LZ, CWD, ZS, DAD, SAL, MGC, and EAB wrote and reviewed the article.
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Competing interests
The authors declare to have no competing interests.
Declaration of interest
GWD reports no conflict of interest. LEG reports no conflict of interest. LZ reports no conflict of interest. CWD reports no conflict of interest. ZS reports no conflict of interest. DAD reports no conflict of interest. SAL reports no conflict of interest. MGC reports no conflict of interest. EAB reports no conflict of interest.
References
- 1.Coronavirus Resource Center . Available from: https://coronavirus.jhu.edu/
- 2.Grasselli G, Zangrillo A, Zanella A, et al. COVID-19 Lombardy ICU Network. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020 Apr 28;323(16):1574–1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Richardson S, Hirsch JS, Narasimhan M, et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA. 2020 May 26;323(20):2052–2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417–1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Monteil V, Kwon H, Prado P, et al. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181(4):905–913. e907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gheblawi M, Wang K, Viveiros A, et al. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ Res. 2020;126(10):1456–1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ye Q, Wang B, Mao J.. The pathogenesis and treatment of the `Cytokine Storm’ in COVID-19. J Infect. 2020;80(6):607–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iba T, Levy JH, Levi M, et al. Coagulopathy of Coronavirus Disease 2019. Crit Care Med. 2020 Sep;48(9):1358–1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gattinoni L, Coppola S, Cressoni M, et al. COVID-19 Does Not Lead to a “Typical„ Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2020 May 15;201(10):1299–1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bos LDJ, Sinha P, Dickson RP. The perils of premature phenotyping in COVID: a call for caution. Eur Respir J. 2020. DOI: 10.1183/13993003.01768-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marini JJ, Gattinoni L. Management of COVID-19 respiratory distress. JAMA. 2020;323(22):2329. [DOI] [PubMed] [Google Scholar]
- 14.Arulkumaran N, Brealey D, Howell D, et al. Use of non-invasive ventilation for patients with COVID-19: a cause for concern? Lancet Respir Med. 2020;8(6):e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Telias I, Katira BH, Brochard L. Is the prone position helpful during spontaneous breathing in patients with COVID-19? JAMA. 2020;323(22):2265. [DOI] [PubMed] [Google Scholar]
- 16.Sullivan EH, Gibson LE, Berra L, et al. In-hospital airway management of COVID-19 patients. Crit Care. 2020;24(1):292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tan L, Wang Q, Zhang D, et al. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduct Target Ther. 2020;5(1):33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Clark E, Molnar AO, Joannes-Boyau O, et al. High-volume hemofiltration for septic acute kidney injury: a systematic review and meta-analysis. Crit Care. 2014;18(1):R7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tang N, Bai H, Chen X, et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18(5):1094–1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tang N, Li D, Wang X, et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(4):844–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020 Jul;191:145–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Flaczyk A, Rosovsky RP, Reed CT, Bankhead-Kendall BK, Bittner EA, Chang MG. Comparison of published guidelines for management of coagulopathy and thrombosis in critically ill patients with COVID 19: implications for clinical practice and future investigations. Crit Care. 2020 Sep 16;24(1):559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;77(6):683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Toscano G, Palmerini F, Ravaglia S, et al. Guillain-Barré Syndrome Associated with SARS-CoV-2. N Engl J Med. 2020 Jun 25;382(26):2574–2576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Asadi-Pooya AA, Simani L. Central nervous system manifestations of COVID-19: a systematic review. J Neurol Sci. 2020;413:116832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ding Y, He L, Zhang Q, et al. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J Pathol. 2004;203(2):622–630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Paniz-Mondolfi A, Bryce C, Grimes Z, et al. Central nervous system involvement by severe acute respiratory syndrome Coronavirus −2 (SARS-CoV-2). J Med Virol. 2020;92:699–702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duong L, Xu P, Liu A. Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav Immun. 2020;87:33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Moriguchi T, Harii N, Goto J, et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis. 2020;94:55–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Poyiadji N, Shahin G, Noujaim D, et al. COVID-19-associated Acute Hemorrhagic Necrotizing Encephalopathy: Imaging Features. Radiology. 2020 Aug;296(2):E119–E120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ye M, Ren Y, Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav Immun. 2020 Aug;88:945–946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yin R, Feng W, Wang T, Chen G, Wu T, Chen D, Lv T, Xiang D. Concomitant neurological symptoms observed in a patient diagnosed with coronavirus disease 2019. J Med Virol. 2020 Apr 15:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Oxley TJ, Mocco J, Majidi S, et al. Large-vessel stroke as a presenting feature of Covid-19 in the young. N Engl J Med. 2020;382(20):e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hanidziar D, Bittner EA. In response: rethinking sedation during prolonged mechanical ventilation for COVID-19 respiratory failure. Anesth Analg. 2020. DOI: 10.1213/ANE.0000000000004961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cipriani G, Danti S, Nuti A, et al. A complication of coronavirus disease 2019: delirium. Acta Neurol Belg. 2020;120(4):927–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kotfis K, Williams Roberson S, Wilson JE, et al. COVID-19: ICU delirium management during SARS-CoV-2 pandemic. Crit Care. 2020;24(1):176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rogers JP, Chesney E, Oliver D, et al. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7(7):611–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Varatharaj A, Thomas N, Ellul MA, et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry. 2020. DOI: 10.1016/S2215-0366(20)30287-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lippi G, Lavie CJ, Sanchis-Gomar F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): evidence from a meta-analysis. Prog Cardiovasc Dis. 2020;63(3):390–391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shi S, Qin M, Shen B, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020;5(7):802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Keyaerts E, Vijgen L, Chen L, et al. Inhibition of SARS-coronavirus infection in vitro by S-nitroso-N-acetylpenicillamine, a nitric oxide donor compound. Int J Infect Dis. 2004;8(4):223–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8(4):e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Patel AB, Verma A. COVID-19 and angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: what is the evidence? JAMA. 2020. DOI: 10.1001/jama.2020.4812 [DOI] [PubMed] [Google Scholar]
- 46.Sommerstein R, Kochen MM, Messerli FH, et al. Coronavirus disease 2019 (COVID-19): do angiotensin-converting enzyme inhibitors/angiotensin receptor blockers have a biphasic effect? J Am Heart Assoc. 2020;9(7):e016509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vaduganathan M, Vardeny O, Michel T, et al. Renin-angiotensin-aldosterone system inhibitors in patients with Covid-19. N Engl J Med. 2020;382(17):1653–1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Inciardi RM, Lupi L, Zaccone G, Italia L, Raffo M, Tomasoni D, Cani DS, Cerini M, Farina D, Gavazzi E, Maroldi R, Adamo M, Ammirati E, Sinagra G, Lombardi CM, Metra M. Cardiac Involvement in a Patient With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020 Jul 1;5(7):819–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sala S, Peretto G, Gramegna M, et al. Acute myocarditis presenting as a reverse Tako-Tsubo syndrome in a patient with SARS-CoV-2 respiratory infection. Eur Heart J. 2020;41(19):1861–1862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hua A, O'Gallagher K, Sado D, et al. Life-threatening cardiac tamponade complicating myo-pericarditis in COVID-19. Eur Heart J. 2020 Jun 7;41(22):2130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fox S, Akmatbekov A, Harbert J, et al., Pulmonary and cardiac pathology in covid-19: the first autopsy series from New Orleans. 2020. [DOI] [PMC free article] [PubMed]
- 52.Long B, Brady WJ, Koyfman A, et al. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020;38(7):1504–1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.van Damme C, Berlingin E, Saussez S, et al. Acute urticaria with pyrexia as the first manifestations of a COVID-19 infection. J Eur Acad Dermatol Venereol. 2020 Jul;34(7):e300–e301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bybee KA, Kara T, Prasad A, et al. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med. 2004;141(11):858–865. [DOI] [PubMed] [Google Scholar]
- 55.Whittaker E, Bamford A, Kenny J, et al. Clinical characteristics of 58 children with a pediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2. JAMA. 2020;324(3):259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cheung KS, Hung IFN, Chan PPY, et al. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology. 2020 Jul;159(1):81–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kaafarani HMA, Moheb ME, Hwabejire JO, et al. Gastrointestinal complications in critically ill patients with COVID-19. Ann Surg. 2020;272:e61-e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cai Q, Huang D, Yu H, et al. COVID-19: Abnormal liver function tests. J Hepatol. 2020 Sep;73(3):566–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Xu L, Liu J, Lu M, et al. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40(5):998–1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Arentz M, Yim E, Klaff L, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA. 2020;323(16):1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cheng Y, Luo R, Wang K, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97(5):829–838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Diao B, Wang C, Wang R, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection In; 2020. [DOI] [PMC free article] [PubMed]
- 64.Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rubino F, Amiel SA, Zimmet P, et al. New-Onset Diabetes in Covid-19. N Engl J Med. 2020 Aug 20;383(8):789–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yang JK, Lin SS, Ji XJ, et al. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010;47(3):193–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Richard C, Wadowski M, Goruk S, et al. Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res Care. 2017;5(1):e000379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sattar N, McInnes IB, McMurray JJV. Obesity Is a Risk Factor for Severe COVID-19 Infection: Multiple Potential Mechanisms. Circulation. 2020 Jul 7;142(1):4–6. [DOI] [PubMed] [Google Scholar]
- 69.Pal R. COVID-19, hypothalamo-pituitary-adrenal axis and clinical implications. Endocrine. 2020 May;68(2):251–252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wheatland R. Molecular mimicry of ACTH in SARS - implications for corticosteroid treatment and prophylaxis. Med Hypotheses. 2004;63(5):855–862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Brancatella A, Ricci D, Viola N, et al. Subacute Thyroiditis After Sars-COV-2 Infection. J Clin Endocrinol Metab. 2020 Jul 1;105(7):dgaa276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.BTA/SFE Statement regarding issues specific to thyroid dysfunction during the COVID -19 pandemic. [cited 2020 May 20]; Available from: https://www.british-thyroid-association.org/sandbox/bta2016/management-of-thyroid-dysfunction-during-covid-19_final.pdf. [Google Scholar]
- 73.Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98(2):75–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Eur Acad Dermatol Venereol. 2020 May;34(5):e212–e213. [DOI] [PubMed] [Google Scholar]
- 75.Amatore F, Macagno N, Mailhe M, et al. SARS-CoV-2 infection presenting as a febrile rash. J Eur Acad Dermatol Venereol. 2020. DOI: 10.1111/jdv.16528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Henry D, Ackerman M, Sancelme E, et al. Urticarial eruption in COVID-19 infection. J Eur Acad Dermatol Venereol. 2020. DOI: 10.1111/jdv.16472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zhang Y, Cao W, Xiao M, et al. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E006. [DOI] [PubMed] [Google Scholar]
- 78.Manalo IF, Smith MK, Cheeley J, et al. A dermatologic manifestation of COVID-19: transient Livedo Reticularis. J Am Acad Dermatol. 2020. DOI: 10.1016/j.jaad.2020.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sun P, Qie S, Liu Z, et al. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: A single arm meta-analysis. J Med Virol. 2020 Jun;92(6):612–617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zhu J, Ji P, Pang J, et al. Clinical characteristics of 3062 COVID-19 patients: A meta-analysis. J Med Virol. 2020 Apr 15:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Behbehani NA, Al-Mane F, D’Yachkova Y, et al. Myopathy following mechanical ventilation for acute severe asthma: the role of muscle relaxants and corticosteroids. Chest. 1999;115(6):1627–1631. [DOI] [PubMed] [Google Scholar]
- 84.De Jonghe B, Sharshar T, Lefaucheur JP, et al. Paresis acquired in the intensive care unit: a prospective multicenter study. JAMA. 2002;288(22):2859–2867. [DOI] [PubMed] [Google Scholar]


