Abstract
Background: Renal cell carcinoma (RCC) is the most common cancer of the kidney. The most common histotype is clear-cell (cc) RCC. Hydrogen sulfide (H2S) is an angiogenic and anti-apoptotic gasotransmitter that is elevated under pseudohypoxic conditions. H2S is endogenously produced by three enzymes: Cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST). Seeing as increased expression of these enzymes has been observed in other human cancer types, this study aimed to quantify H2S-producing enzyme expression in human RCC samples and evaluate whether it correlated with clinical outcomes. Patients and Methods: Eighty-eight human kidney tissue specimens, with healthy and cancerous tissue components, were immunohistochemically stained for CSE, CBS, and MPST. The mean pixel intensity of positively stained areas was quantified. A retrospective analysis was conducted to obtain patient demographics, rates of metastasis/recurrence, and prognostic characteristics. Statistical correlations between enzyme expressions and subsequent patient outcomes were evaluated. Results: There was significantly greater expression of CSE, CBS, and MPST in cc-RCC compared to paired healthy tissue (p<0.0001). The difference in expression of CSE in cancerous versus normal tissue was significantly greater than that for CBS and MPST (p<0.0001 and p<0.01, respectively). Enzyme expression patterns in cancerous versus normal tissue did not correlate with nuclear grade, stage, histological type or cancer recurrence/metastasis. Conclusion: To our knowledge, this is the first report of the differential increase in expression of CSE, CBS, and MPST in human RCC. Although these patterns do not appear to correlate with cancer recurrence, metastasis, size or nuclear grade, their differential increase suggests a potential therapeutic target.
Keywords: Renal cell carcinoma, hydrogen sulfide, cystathionine γ-lyase, CSE, cystathionine β-synthase, CBS, 3-mercaptopyruvate sulfurtransferase, MPST
Renal cell carcinoma (RCC) is the most common cancer of the kidney. It accounts for 80-90% of all kidney cancer cases and has a cancer-specific mortality rate of 30-40% (1,2). Incidence rates of RCC have increased over the past few decades, and this is largely attributed to increased detection by diagnostic tools (1,3). RCC is subdivided into various histotypes based on tumour morphology, genetic signature, clinical manifestation, and area of the nephron where the tumour is present. The most common type of RCC is clear-cell (cc) RCC, accounting for 80% of all cases. Other types included papillary, chromophobe and others, which can be classified as non-cc-RCC (4).
In cc-RCC, the lack of functional Von-Hippel Lindau (VHL) tumour-suppressor gene prevents the degradation of hypoxia-inducible factor-alpha (HIFa) under normal oxygen conditions. This leads to the inappropriate expression of hypoxia response genes during normoxia, resulting in a condition called pseudohypoxia. The hypoxia response pathways are constitutively active in pseudohypoxia, which causes the resulting cells to be proliferative, invasive, and angiogenic (5). A shift in metabolism, known as the Warburg effect, also occurs, wherein cells have a strong preference for glycolysis over aerobic respiration, even when oxygen is readily available (6). The inactivation of VHL and the Warburg effect provide cancerous cells with an environment that further facilitates rapid proliferation.
Hydrogen sulfide (H2S) is a small, membrane-permeable gasotransmitter that is produced in nearly all mammalian cells by three independent enzymes: Cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST) (7). H2S modifies cellular protein activity by reacting with cysteine thiol groups in a process called S-sulfhydration (8). Endogenous H2S production is critical for the normal functioning of various systems of the human body, including the central nervous system, cardiovascular system, digestive system, endocrine system, and respiratory system (9).
Under hypoxic conditions, H2S pushes cells towards glycolysis by increasing glyceraldehyde-3 phosphate dehydrogenase activity. It also promotes anti-apoptotic signalling by S-sulfhydration of nuclear factor kappa-light-chain-enhancer of activated B-cells (NFĸB) and modulates angiogenesis by interacting with vascular endothelial growth factor (10). However, when the H2S level becomes too high under normoxic conditions, it acts as a mitochondrial poison that inhibits complex-IV of the electron transport chain, which prevents aerobic respiration (11). The level of endogenous H2S production is monitored precisely by the body. Unlike hormones and neurotransmitters, gasotransmitters such as H2S are not suitable for long-term storage. As soon as H2S is produced, it is immediately able to react with its surroundings, therefore production must be precisely regulated at the level of enzyme expression and activity (12).
Given its involvement in physiological functions, H2S production has been linked to various types of cancer including colon, ovarian, breast, liver (13-15). Szabo et al. have shown that CBS is up-regulated in colon cancer. They observed increased expression of CBS in both human tumour samples and malignant cell lines when compared to non-malignant samples. Increased endogenous H2S production due to increased CBS expression and activity contributes to cell migration and bioenergetics (16). Additionally, overexpression of CSE was observed in ovarian cancer cell lines that expressed little to no CBS, unlike ovarian epithelial cells that expressed CBS, suggesting an inverse correlation between expression of these two H2S-producing enzymes. Furthermore, the same study found that silencing CBS significantly inhibited ovarian cancer cell proliferation as it reduced H2S production, activated tumour suppressors (including p53), and impaired cytoprotection by inhibiting NFĸB (17).
Data on enzymes responsible for endogenous H2S production are controversial, showing them to be increased (6) or decreased (6,18) in RCC cell lines. H2S levels were observed to be higher in VHL-deficient cc-RCC cell lines and inhibition of CBS and CSE significantly reduced the growth, survival, and metabolic output of cc-RCC cell lines (6). However, another study showed H2S-producing enzymes to be reduced up to 17-fold in cc-RCC cell lines (18).
The aim of this study was therefore to determine whether the expression patterns of H2S-producing enzymes increased or not in human RCC tissues and whether these correlated with subsequent clinical outcomes. If any correlations between CSE, CBS or MPST expression and patient outcomes emerge, they may support the use of this approach as a potential prognostic tool for patients with RCC.
Patients and Methods
Immunohistochemistry. This study was approved by the University of Western Ontario’s Research Ethics Board (REB 106749). Eighty-eight human kidney tissue specimens were collected from storage at the University Hospital – London Health Sciences Centre Pathology Department. These specimens were obtained from patients dating back from 2012 who presented with malignant kidney mass and underwent partial or radical nephrectomy. Each specimen had tumour tissue adjacent to normal, healthy tissue. From each tissue sample, three slices were obtained from the tumour section and three slices from the normal tissue section. Samples were then stained with antibodies for CBS (mouse anti-human CBS; Abnova, Walnut, CA, USA), CSE (rabbit anti-human CSE; Sigma-Aldrich, St. Louis, MO, USA), and MPST (rabbit anti-human MPST; Sigma-Aldrich). The primary antibody for CBS was used at a 250:1 dilution, whereas antibodies for CSE and MPST were used at a 1500:1 dilution, based on preliminary optimization to ensure strong enzyme staining of our tissue samples.
Imaging and analysis. Immunohistochemically stained samples were imaged with a Nikon camera mounted on a bright-field microscope (Nikon Eclipse 90i; Nikon, Melville NY, USA). Images were taken at 10× magnification. Since tumours are heterogeneous, images were taken from three random areas on each slide to account for this.
All images were analyzed using ImageJ software (Wayne Rasband, National Institutes of Health, Bethesda, MA, USA). The colour threshold tool on ImageJ was used to quantify areas positive for enzyme staining to compare expression patterns in cancerous versus healthy tissue. The mean pixel intensity (MPI) of the stained areas, which was taken to be representative of the relative enzyme expression in that area of tissue, was used for subsequent data analysis. The MPI for the three tumour and three normal tissue sections were averaged for each specimen. To ensure consistency of analysis, the optimal threshold parameters for each enzyme stain were saved as a template and applied to all images analyzed for that particular enzyme. The area quantified was confirmed with a clinical pathologist to ensure accuracy. Subsequently, in order to determine the fold-increase of the expression of each enzyme in the patient-matched biopsies, the ratio of the MPI of each enzyme stain of RCC relative to that of normal tissue samples was calculated.
GraphPad Prism Version 8.0 (GraphPad Software, Inc., La Jolla, CA, USA) was used for data analysis. A paired sample t-test was conducted to compare relative enzyme expression between cancerous and healthy tissue for each enzyme. A one-way repeated measures ANOVA analysis and Tukey’s post hoc test were conducted to determine statistical differences between ratios of enzyme expression.
Retrospective analysis. Retrospective analysis was performed using PowerChart software (Cerner, Kansas City, MO, USA) to obtain data of patient demographics, nuclear grade, tumour size, metastasis, and recurrence. These data were cross-referenced with immunohistochemistry results to detect any relationships between expression of H2S-producing enzymes and patient outcomes. Two-way ANOVA analyses and Tukey’s post hoc tests were conducted to determine statistical significance for each correlation.
Results
Expression of H2S-producing enzymes increases in RCC versus paired healthy renal tissue. Eighty-eight human kidney samples and demographics were analyzed. Demographics of the patients in this study are shown in Table I.
Table I. Patient demographics.
PN: Partial nephrectomy; RN: radical nephrectomy; L: left; R: right; SD: standard deviation.
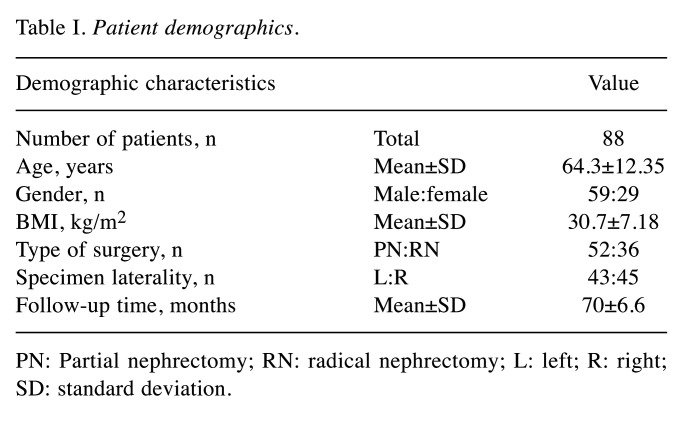
In order to evaluate the expression of H2S-producing enzymes in RCC compared with normal renal tissue, immunohistochemistry was used to stain three healthy and three tumour sections from each patient for CBS, CSE and MPST expression. Side-by-side sample images of paired normal and cc-RCC sections stained for CSE, CBS and MPST expression are shown in Figure 1.
Figure 1. Immunohistochemistry staining of formalin-fixed, paraffin-embedded human kidney tissue. Each specimen consisted of normal tissue adjacent to renal cell carcinoma (RCC) tissue. Each section was immunohistochemically stained with antibodies for cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST). All images were taken at 10× magnification using bright-field microscopy.
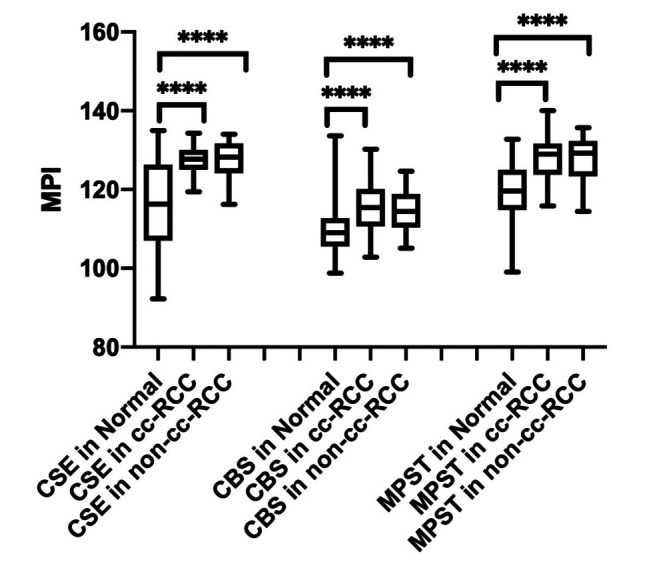
We found that the MPI of CSE, CBS, and MPST were significantly greater both in cc-RCC and non-cc-RCC tissue versus normal tissue (p<0.0001). There was no significant difference in MPI between cc-RCC and non-cc-RCC for CSE (p=0.914), CBS (p=0.662), and MPST (p=0.681) (Figure 2).
Figure 2. Expression patterns of cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST) in normal (N), clear-cell renal cell carcinoma (cc-RCC) and non-cc-RCC tissue. Box and whisker graph of expression in patient-matched biopsies from healthy (normal, n=88) and cancerous (cc-RCC, n=68; non-cc-RCC, n=20) kidney tissues which were fixed, paraffin-embedded, sectioned, and immunohistochemically stained for CSE, CBS, and MPST. Data are presented as the mean±SD of the mean pixel intensity. ****Significantly different at p<0.0001 by two-way ANOVA and Tukey’s post hoc tests.
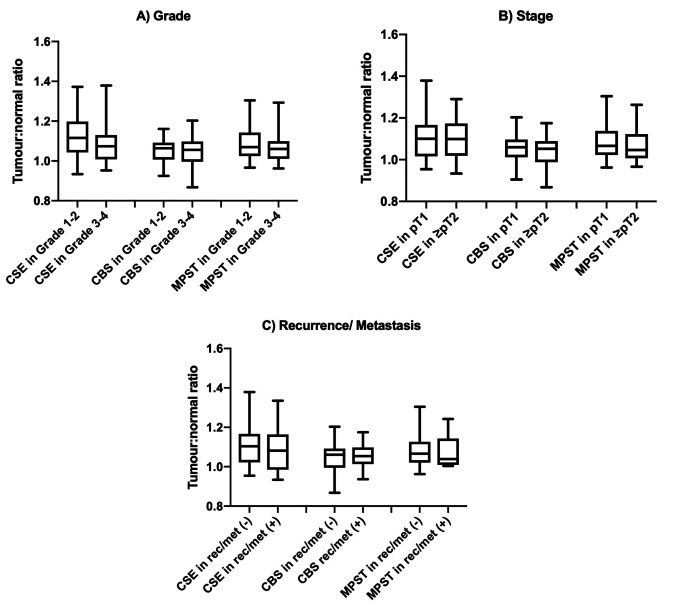
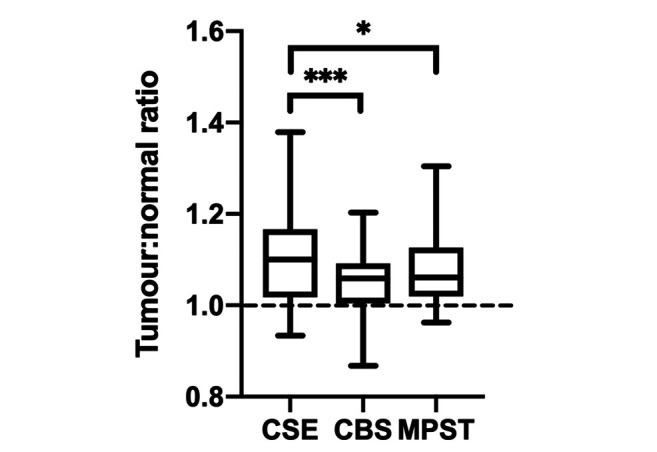
H2S-producing enzymes exhibit a differential increase in expression. To determine the fold-increase of each enzyme, the ratio of the difference in MPI of CSE, CBS, and MPST between RCC and normal samples, was calculated. The three H2S-producing enzymes showed differential changes in expression when comparing the ratio of expression in tumour versus normal kidney tissue (Figure 3). There was a 1.10-fold increase in expression of CSE in RCC tissue versus normal, healthy tissue. The ratio for CSE was found to be significantly greater than those for CBS (1.051±0.06, p<0.001) and MPST (1.083±0.08, p=0.01). There was no significant difference in the MPI ratio between CBS and MPST (p=0.65).
Figure 3. The ratio of tumour:normal tissue expression of cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST) in human kidney tissue. Box and whisker graph showing the mean pixel intensity ratio of tumour:normal tissue with min-max values. The dotted line represents a 1:1 ratio of mean pixel intensity, where there is no difference in enzyme expression between tumour and normal tissue. Significantly different at *p<0.01 and ***p<0.001 by one-way repeated measures ANOVA and Tukey’s post hoc tests.
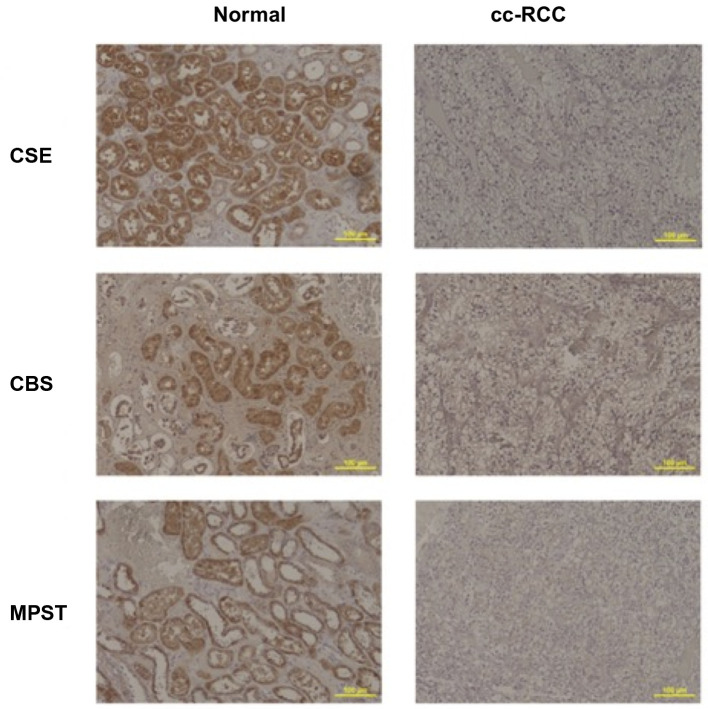
H2S-producing enzyme expression patterns in RCC specimens do not correlate to grade, pathological stage, recurrence or metastasis. In order to determine whether patterns of endogenous H2S-producing enzyme expression correlated with subsequent pathological and clinical outcomes, the ratios of enzyme expression of patient-matched biopsies from healthy and RCC kidney tissue were compared between patients whose RCC scored low (1 or 2) or high (3 or 4) on the Fuhrman grading system, had different pathological stages in the pathology report, and whose cancer did or did not recur/metastasize after surgical treatment. There were no significant differences in the MPI ratio for CSE, CBS and MPST based on correlation with grade (p=0.061, p=0.95 and p=0.212), stage (p=0.714, p=0.266 and p=0.336), and metastasis/recurrence (p=0.352, p=0.731 and p=0.709) (Figure 4).
Figure 4. Mean pixel intensity (MPI) ratio of tumour:normal tissue expression of cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (MPST). Box and violin graph represented the mean pixel intensity ratio between patients whose renal cell carcinoma scored low (1 or 2, n=40) or high grade (3 or 4, n=46) on the Fuhrman grading system (A), with lower (pT1, n=62) or higher stage (≥pT2, n=26) in the pathology report (B), and whose cancer either did (positive, n=16) or did not (negative, n=72) recur/metastasize in the followup period (C). Data are presented as a ratio of mean tumour:normal pixel intensity±SD. There were no significant differences between groups by two-way repeated-measures ANOVA and Tukey’s post hoc tests.
Discussion
The incidence of RCC has been rising by 2% annually over the past two decades due to the increased use of diagnostic tools for other reasons (3). Due to the lack of specific early symptoms and the absence of general screening tests, more than 30% of patients with RCC have disease at the metastatic stage at the time of diagnosis (19). Despite diagnostic advances, RCC is still the most lethal common urological cancer (1). The recent studies have reported increased H2S levels and H2S-producing enzyme expression in several types of urological cancer, including RCC (6,20-22). Thus, this study aimed to evaluate whether the expression patterns of H2S-producing enzymes in human RCC samples correlate with subsequent clinical outcomes. The results showed a differential increase in the expression of CSE, CBS, and MPST in both cc and non-cc-RCC tissue samples; however, the expression patterns did not correlate with clinical outcomes.
H2S has been a focus in cancer research because of its involvement in cytoprotection (23), angiogenesis (13), and anaerobic metabolism (24). In hypoxia, H2S accumulates and helps maintain cell function by up-regulating glycolysis to provide ATP via anaerobic metabolism, up-regulating cytoprotective pathways by promoting anti-apoptotic signaling of NFĸB, and promoting new blood vessel formation to deoxygenated tissue by interacting with vascular endothelial growth factor (13,25,26). Since it is a membrane-permeable gasotransmitter and direct measurement of H2S in the cell is not easy, we measured H2S-producing enzyme expression and not the H2S level directly, the increase in H2S-producing enzyme expression observed in this study suggests that higher H2S activity may also be present in human cc and non-cc-RCC samples. Thus, the findings of this study contribute to the literature surrounding the role of H2S and H2S-producing enzymes in RCC, using human RCC samples.
Previous studies have examined H2S-producing enzyme expression profiles in other types of human cancer. Szabo et al. showed that CBS was up-regulated but CSE unchanged in human colon cancer, resulting in an increased rate of H2S production. They further discovered that colon cancer-derived epithelial cell lines also up-regulated CBS and increased H2S production (16). This indicates that in the colon cancer, CBS is the main regulator in supporting growth, proliferation, and angiogenesis of the cancerous cells. Another study by Bhattacharyya et al. demonstrated an inverse correlation between the expression of CBS and CSE in human ovarian cancer. They observed overexpression of CSE in ovarian cell epithelium; however, CBS was overexpressed in ovarian cancer cell lines (17). Unlike these studies, we found a differential increase in the expression of all three H2S-producing enzymes in the cc and non-cc RCC samples. Additionally, CSE exhibited significantly higher tumour: normal tissue expression than CBS and MPST, suggesting its importance in the pathology of RCC specifically.
The observed increase in the expression of H2S-producing enzymes suggests that inhibiting these enzymes may represent a novel therapeutic strategy for limiting RCC tumour growth. Previous studies have evaluated the effects of inhibiting key H2S-producing enzymes in various tumour models. Szabo et al. showed that silencing of CBS in colon cancer cells reduced xenograft growth of patient-derived colon cancer xenograft in mice and suppressed neovessel density (16). Bhattacharyya et al. showed the silencing of CBS inhibited ovarian cancer cell proliferation by reducing H2S production, activating tumour suppressors such as p53, and impairing cytoprotection by inhibiting NFĸB (17). Based on the anti-tumour effects of inhibiting H2S-producing enzymes in these studies, it is likely that this approach would also limit RCC tumour growth. Sonke et al. have shown that inhibition of CBS and CSE significantly reduced the growth, survival, and metabolic output of murine RCC cells. Furthermore, they also showed that inhibition of endogenous H2S production in chick embryos also restricts the neovascularization and growth of cc-RCC xenografts (6). Another study by Breza et al. showed that silencing of especially of CSE might contribute to resistance of induction of apoptosis (18). Further research is needed to establish whether inhibiting H2S-producing enzymes, particularly CSE, inhibits the growth of murine RCC tumours in vivo and human RCC cell lines.
While we are the first to show that CSE, CBS, and MPST are differentially increased in both cc and non-cc human RCC tissue samples, quantifying enzyme expression is not sufficient to conclude that H2S production is increased in human RCC. To determine the activity of the H2S-producing enzymes, H2S levels need to be measured or functional assays need to be performed using human RCC samples. Additionally, a larger cohort of patient samples is needed to confirm whether the differential increase in CSE, CBS and MPST expression correlates with clinical outcomes. While no correlations were observed in this study, Shackelford et al. showed that CBS protein level increased in RCC tumours with higher Fuhrman grades (20). If it is established that H2S-producing enzyme activity increases in RCC and is correlated with clinical outcomes, healthcare professionals can consider using a nomogram that correlates expression of H2S-producing enzyme in a renal biopsy with patient demographics to give a calculated score that predicts the likelihood of a mass becoming aggressive, metastasizing, or recurring. This would allow healthcare teams to tailor an individualized treatment strategy and make an informed decision on the direction of oncological treatment.
In conclusion, this is the first study to report the differential increase in the expression of CSE, CBS, and MPST in human RCC. Since these patterns did not correlate with cancer stage, recurrence, metastasis, or nuclear grade in this study, further research is needed to establish whether H2S-producing enzyme expression might serve as a novel diagnostic/prognostic marker. Additionally, targeted therapies aimed at inhibiting these H2S-producing enzymes may represent a novel therapeutic approach towards limiting RCC tumour growth and disease progression.
Conflicts of Interest
The Authors declared that they have no conflicts of interest in regard to this study.
Authors’ Contributions
S. Emrullah: Data collection or management, data analysis, article writing; P. Katharine: data collection or management, data analysis, article writing; J. Smriti: data analysis, article writing; A. Masoud: article editing; G. Manal: project development; S. Alp: project development, article writing/editing.
References
- 1.Motzer RJ, Agarwal N, Beard C, Bhayani S, Bolger GB, Carducci MA, Chang SS, Choueiri TK, Hancock SL, Hudes GR, Jonasch E, Josephson D, Kuzel TM, Levine EG, Lin DW, Margolin KA, Michaelson MD, Olencki T, Pili R, Ratliff TW, Redman BG, Robertson CN, Ryan CJ, Sheinfeld J, Spiess PE, Wang J, Wilder RB, Nationall Comprehensive Cancer Network Kidney cancer. J Natl Compr Canc Netw. 2011;9:960–977. doi: 10.6004/jnccn.2011.0082. [DOI] [PubMed] [Google Scholar]
- 2.Rossi SH, Hsu R, Blick C, Goh V, Nathan P, Nicol D, Fleming S, Sweeting M, Wilson ECF, Stewart GD. Meta-analysis of the prevalence of renal cancer detected by abdominal ultrasonography. Br J Surg. 2017;104:648–659. doi: 10.1002/bjs.10523.. [DOI] [PubMed] [Google Scholar]
- 3.Bhatt JR, Finelli A. Landmarks in the diagnosis and treatment of renal cell carcinoma. Nat Rev Urol. 2014;11:517–25. doi: 10.1038/nrurol.2014.194.. [DOI] [PubMed] [Google Scholar]
- 4.Muglia VF, Prando A. Renal cell carcinoma: histological classification and correlation with imaging findings. Radiol Bras. 2015;48:166–174. doi: 10.1590/0100-3984.2013.1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haase VH. Renal cancer: Oxygen meets metabolism. Exp Cell Res. 2012;318:1057–1067. doi: 10.1016/j.yexcr.2012.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sonke E, Verrydt M, Postenka CO, Pardhan S, Willie CJ, Mazzola CR, Hammers MD, Pluth MD, Lobb I, Power NE, Chambers AF, Leong HS, Sener A. Inhibition of endogenous hydrogen sulfide production in clear-cell renal cell carcinoma cell lines and xenografts restricts their growth, survival and angiogenic potential. Nitric oxide Biol Chem. 2015;49:26–39. doi: 10.1016/j.niox.2015.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kimura H. Hydrogen sulfide: its production, release and functions. Amino Acids. 2011;41:113–121. doi: 10.1007/s00726-010-0510-x. [DOI] [PubMed] [Google Scholar]
- 8.Hancock JT, Whiteman M. Hydrogen sulfide and cell signaling: Team player or referee. Plant Physiol Biochem. 2014;78:37–42. doi: 10.1016/j.plaphy.2014.02.012. [DOI] [PubMed] [Google Scholar]
- 9.Wang R. Physiological implications of hydrogen sulfide: A whiff exploration that blossomed. Physiol Rev. 2012;92:791–896. doi: 10.1152/physrev.00017.2011. [DOI] [PubMed] [Google Scholar]
- 10.Sen N, Paul BD, Gadalla MM, Mustafa AK, Sen T, Xu R, Kim S, Snyder SH. Hydrogen sulfide-linked sulfhydration of NF-ĸB mediates its antiapoptotic actions. Mol Cell. 2012;45:13–24. doi: 10.1016/j.molcel.2011.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Petersen LC. The effect of inhibitors on the oxygen kinetics of cytochrome c oxidase. BBA - Bioenerg. 1977;460:299–307. doi: 10.1016/0005-2728(77)90216-X. [DOI] [PubMed] [Google Scholar]
- 12.Paul BD, Snyder SH. H2S signalling through protein sulfhydration and beyond. Nat Rev Mol Cell Biol. 2012;13:499–507. doi: 10.1038/nrm3391. 507. [DOI] [PubMed] [Google Scholar]
- 13.Coletta C, Papapetropoulos A, Erdelyi K, Olah G, Módis K, Panopoulos P, Asimakopoulou A, Gerö D, Sharina I, Martin E, Szabo C. Hydrogen sulfide and nitric oxide are mutually dependent in the regulation of angiogenesis and endothelium-dependent vasorelaxation. Proc Natl Acad Sci USA. 2012;109:9161–9166. doi: 10.1073/pnas.1202916109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Szabó C, Papapetropoulos A. Hydrogen sulphide and angiogenesis: mechanisms and applications. Br J Pharmacol. 2011;164:853–65. doi: 10.1111/j.1476-5381.2010.01191.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hellmich MR, Szabo C. Hydrogen sulfide and cancer. Handb Exp Pharmacol. 2015;230:233–41. doi: 10.1007/978-3-319-18144-8_12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Szabo C, Coletta C, Chao C, Modis K, Szczesny B, Papapetropoulos A, Hellmich MR. Tumor-derived hydrogen sulfide, produced by cystathionine-β-synthase, stimulates bioenergetics, cell proliferation, and angiogenesis in colon cancer. Proc Natl Acad Sci USA. 2013;110:12474–12479. doi: 10.1073/pnas.1306241110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bhattacharyya S, Saha S, Giri K, Lanza IR, Nair KS, Jennings NB, Rodriguez-Aguayo C, Lopez-Berestein G, Basal E, Weaver AL, Visscher DW, Cliby W, Sood AK, Bhattacharya R, Mukherjee P. Cystathionine beta-synthase (CBS) contributes to advanced ovarian cancer progression and drug resistance. PLoS One. 2013;8:e79167. doi: 10.1371/journal.pone.0079167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Breza J, Soltysova A, Hudecova S, Penesova A, Szadvari I, Babula P, Chovancova B, Lencesova L, Pos O, Breza J, Ondrias K, Krizanova O. Endogenous H2S producing enzymes are involved in apoptosis induction in clear cell renal cell carcinoma. BMC Cancer. 2018;18:591. doi: 10.1186/s12885-018-4508-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Motzer RJ, Russo P. Systemic therapy for renal cell carcinoma. J Urol. 2000;163:408–417. doi: 10.1016/s0022-5347(05)67889-5. [DOI] [PubMed] [Google Scholar]
- 20.Shackelford RE, Abdulsattar J, Wei EX, Cotelingam J, Coppola D, Herrera GA. Increased nicotinamide phosphoribosyltransferase and cystathionine-β-synthase in renal oncocytomas, renal urothelial carcinoma, and renal clear cell carcinoma. Anticancer Res. 2017;37:3423–3427. doi: 10.21873/anticanres.11709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gai JW, Qin W, Liu M, Wang HF, Zhang M, Li M, Zhou WH, Ma QT, Liu GM, Song WH, Jin J, Ma HS. Expression profile of hydrogen sulfide and its synthases correlates with tumor stage and grade in urothelial cell carcinoma of bladder. Urol Oncol Semin Orig Investig. 2016;34:166.e15–166.e20. doi: 10.1016/j.urolonc.2015.06.020. [DOI] [PubMed] [Google Scholar]
- 22.Zhao K, Li S, Wu L, Lai C, Yang G. Hydrogen sulfide represses androgen receptor transactivation by targeting at the second zinc finger module. J Biol Chem. 2014;289:20824–20835. doi: 10.1074/jbc.M114.559518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ahn H, Lee E, Kim K, Lee C. Effect of glutathione and its related enzymes on chemosensitivity of renal cell carcinoma and bladder carcinoma cell lines. J Urol. 1994;151:263–267. doi: 10.1016/S0022-5347(17)34929-7. [DOI] [PubMed] [Google Scholar]
- 24.Xue R, Hao D-D, Sun J-P, Li W-W, Zhao M-M, Li X-H, Chen Y, Zhu J-H, Ding Y-J, Liu J, Zhu Y-C. Hydrogen sulfide treatment promotes glucose uptake by increasing insulin receptor sensitivity and ameliorates kidney lesions in type 2 diabetes. Antioxid Redox Signal. 2013;19:5–23. doi: 10.1089/ars.2012.5024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cai W, Wang M, Ju L, Wang C, Zhu Y. Hydrogen sulfide induces human colon cancer cell proliferation: role of AKT, ERK and p21. Cell Biol Int. 2010;34:565–572. doi: 10.1042/cbi20090368. [DOI] [PubMed] [Google Scholar]
- 26.Yang G, Zhao K, Ju Y, Mani S, Cao Q, Puukila S, Khaper N, Wu L, Wang R. Hydrogen sulfide protects against cellular senescence via S-sulfhydration of KEAP1 and activation of NRF2. Antioxid Redox Signal. 2013;18:1906–1919. doi: 10.1089/ars.2012.4645. [DOI] [PubMed] [Google Scholar]