Abstract
Polymeric nanomaterials have become a prominent area of research in the field of drug delivery. Their application in nanomedicine can improve bioavailability, pharmacokinetics, and, therefore, the effectiveness of various therapeutics or contrast agents. There are many studies for developing new polymeric nanocarriers; however, their clinical application is somewhat limited. In this review, we present new complex and multifunctional polymeric nanocarriers as promising and innovative diagnostic or therapeutic systems. Their multifunctionality, resulting from the unique chemical and biological properties of the polymers used, ensures better delivery, and a controlled, sequential release of many different therapeutics to the diseased tissue. We present a brief introduction of the classical formulation techniques and describe examples of multifunctional nanocarriers, whose biological assessment has been carried out at least in vitro. Most of them, however, also underwent evaluation in vivo on animal models. Selected polymeric nanocarriers were grouped depending on their medical application: anti-cancer drug nanocarriers, nanomaterials delivering compounds for cancer immunotherapy or regenerative medicine, components of vaccines nanomaterials used for topical application, and lifestyle diseases, ie, diabetes.
Keywords: polymeric nanocarriers, biomedical application, anti-cancer, cancer immunotherapy, modern vaccines, regenerative medicine, lifestyle diseases – diabetes
Introduction
Medical application of nanomaterials is becoming increasingly crucial in diagnostics (delivery of contrast agents for imaging) as well as in prophylaxis (vaccines) and therapy of various diseases (drug delivery systems). Nanocarriers employed in medicine improve the bioavailability, pharmacokinetics, and effectiveness of various therapeutics or contrast agents, ensuring: increased hydrophilicity, reduced interaction with plasma and cellular proteins, and better accumulation in target tissues. The most promising purpose of exploiting nanoparticles in medicine is their potential to localize (or be targeted) in a specific manner to the site of disease and reduce or eliminate the possible side effects.1 Targeting can be broadly classified into three regimes; passive, active, andphysical.2 Passive is based on accumulating nanoparticulate systems containing drugs through leaky vasculature of a diseased area. It was found in tumors, infarcts, and inflammation regions. In such regions with increased vascular permeability, nanoparticulate systems can selectively accumulate and release a drug that exerts a therapeuticeffect.3 This phenomenon allows the delivery of drugs to the tumors, for which “enhanced permeability and retention” (EPR) effect is observed.2,4,5 The EPR effect is predominantly described for solid tumors where fenestrations between epithelial cells, disorders in the pericyte coverage, and the lack of a smooth muscle layer of new blood vessels are sufficient for nanomedicines accumulation. Additionally, impaired lymphatic drainage in this tissue is responsible for nanocarriers retention.6
Another approach, active targeting, is based on the specific recognition of targeted diseased region/cells’ ligands. Finally, physical targeting includes pH-sensitive, temperature-sensitive, redox potential sensitive, ultrasound sensitive, and magnetic sensitive systems. As a result of nanomaterials’ research, over 50 nanoformulations for oral, intravenous, subcutaneous, intrabronchial, topical, or intramuscular administration have already been approved by the American Food and Drug Administration (FDA) or the European Medicines Agency (EMA) for human use.7,8 The most clinically approved nanomedicines belong to liposomes and polymeric nanoparticles; however, the number of nanomaterials accepted by the FDA for medical applications is still low. An example of a polymeric nanomedicine is Apealea/Paclical containing micellar paclitaxel (paclitaxel formulated with the surfactant XR17) used to treat epithelial ovarian cancer, primary peritoneal cancer, and Fallopian tube cancer.9 The new polymer nanomaterials’ safety and therapeutic efficacy recently verified several clinical studies that mostly concerned nanocarriers of anti-cancer drugs. Phase three of clinical trials have already been completed for yet another paclitaxel formulation - NK105, a “core-shell-type” polymeric micellar nanoparticle formulation used in patients with metastatic or recurrent breast cancer.10 Still, other studies concerned nanomedicines such as cyclodextrin-based polymer-camptothecin CRLX101 or conjugated docetaxel CRLX-301. Soon, the first phase of clinical trials will begin on polymeric nanoparticles loaded with monoclonal antibody Cetuximab and decorated with somatostatin analog for the targeting of colon cancer (data from clinicaltrials.gov). In connection with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, at least several different nano-based-tools have been developed that could be useful in the treatment of patients with the coronavirus disease-19 (COVID-19).11,12 Clinical trials have recently started for a full-length recombinant SARS-CoV-2 glycoprotein nanoparticle vaccine adjuvanted with the saponin-based Matrix M (NVX-CoV2373, developed by Novavax, NCT04368988) or inhaled nanoparticle formulation of Remdesivir (developed by NeuroActiva, NCT04480333, data from clinicaltrials.gov).13
Nanocarriers were initially developed to improve the pharmacokinetics of drugs, but they unexpectedly received much attention from researchers and have become the center of this promising trend of multifunctional nanomedicine.14 Unlike molecular drugs or imaging agents, nanoparticles can easily integrate more than one kind of contrast or therapeutic agent, making them potential multifunctional nanoplatforms. The concept of multifunctional nanocarriers also refers to the precise delivery of more than one drug simultaneously, which increases the effectiveness of therapy or overcome resistance to a given compound. Delivery of more than one agent also allows the use of various therapeutic methods (for example, chemotherapy and immunotherapy, chemotherapy and photodynamic therapy, induction of the immune response to the selected antigen, and enhancement of this response).
There are constant enormous demands on new, advanced, multifunctional nanomaterials to be used in the future of medicine. First of all, they should be biodegradable and biocompatible. They are also supposed to be very stable in the body, have optimal pharmacokinetics and biodistribution, ensure the protection of healthy tissues, and accumulate drugs in diseased tissue at the right time of the disease process. Additionally, they should deliver chemically diverse compounds for including small drugs (antibiotics, cytostatics), nucleic acids, peptides, proteins, and magnetic nanoparticles. Appropriate synthesis methods should allow the biological activity of transported drugs to be preserved. It is especially important when providing active therapeutic proteins (enzymes, cytokines) or antigens that are the essential components of vaccines.
The purpose of this work is to present the latest scientific reports on polymeric nanocarriers for medical applications. We selected papers that were published in 2016–2020. We describe nanomaterials whose biological assessment has been carried out at least in vitro. Most of them have also been tested in vivo.
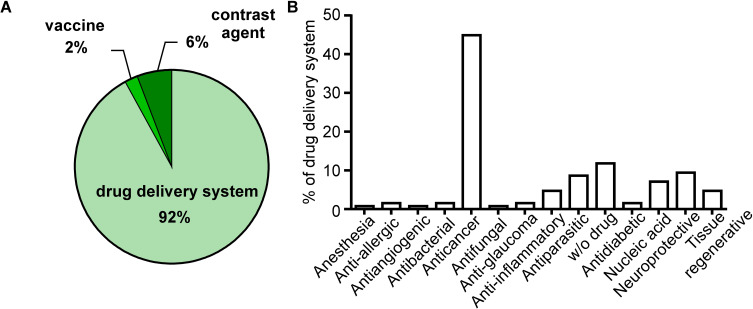
A literature search (PubMED) revealed that polymeric nanocarriers are most often used as drug delivery systems. Since effective cancer treatment still seems to be the most significant medical challenge, most of the new nanomaterials that have recently been obtained and characterized are primarily anti-cancer drug carriers (Figure 1). In this work, we also present less numerous polymer nanomaterials to treat other diseases such as diabetes, atherosclerosis, or diseases based on chronic inflammation.
Figure 1.
The main biomedical application of polymeric nanocapsules/nanocarriers.
Notes: PubMed resources searched in all fields using the term “polymeric nanocapsules” filtered on “in the last five years” (2016–2020). Seven hundred seventy results were obtained (on March 25, 2020), and the authors selected 320 publications in which nanocapsules were used in the biomedical field (excluding diagnostic application and review papers). (A) Polymer nanocapsules were divided into three categories depending on their application in medicine. (B) Since the vast majority were nanocapsules used as a drug delivery system (294 from 320 publications), the authors present the types of transported drugs, w/o drugs stand for nanocapsules which were obtained empty and have potential as drug transporter.
We focused primarily on these polymeric nanocarriers, which in our opinion, can be described as multifunctional, with high capacity as innovative diagnostic and therapeutic systems for the clinical field in the treatment of various diseases, and with the considerable potential for improving human health.
Polymeric Nanocarriers: Materials and Formulation Techniques
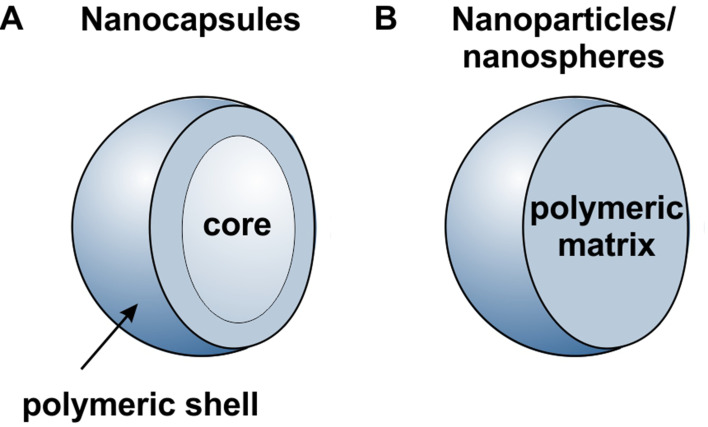
Nanocarriers are generally considered as particulate structures with a diameter smaller than 1000 nm. Depends on the morphology, nanoparticles, nanospheres, and nanocapsules can be defined as presented in Figure 2. Nanoparticles/nanospheres are generally homogenous matrix systems in which the drug is dispersed in the material, forming them or can be adsorbed in their pores or at their surface, or the drug can be conjugated to them. On the other hand, nanocapsules’ structure can be divided into two parts, the core and the shell.15 The core material can be solid, liquid, or gas, depending on the type of application, while the shell is usually formed from the polymeric material. Generally, the drug is located in the core part, while the shell protects them from the external environment. However, incorporation in shell material and adsorption at their surface is also possible, allowing simultaneous delivery of one or several drugs in different nanocapsule compartments. The shell may be made permeable, semi-permeable, or impermeable, depending on the application. Permeable or semi-permeable shells are used for controlled release applications. The release rate from capsules is governed mainly by the shell’s thickness, its pore’s size, and the core’s material.
Figure 2.
Schematic illustration of nanocarriers (A) nanocapsules (B) nanoparticle/nanospheres.
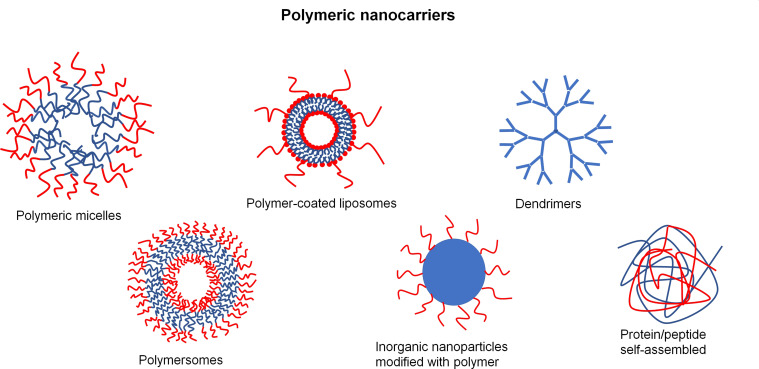
There are three main classical polymeric nanocarriers preparations: layer-by-layer method, nanoprecipitation method, and emulsion templated method. Emulsion templated methods mainly include emulsification-diffusion, emulsification-evaporation, emulsification-coacervation, multiple emulsification, and emulsion/polymer coating.15–19 Nevertheless, other methods have been used, such as methodologies for preparing polymer-coated liposomes, polymersomes, dendrimers, or self-assembled polymeric micelles inorganic nanoparticles modified with polymeric materials as well as protein/peptide self-assembled nanocarriers.20–29 Sketches of selected polymeric nanocarriers are presented in Figure 3.
Figure 3.
Schematic representation of selected polymeric nanocarriers.
On the other hand, the mentioned methods of preparation of polymeric nanocarriers could be divided mainly by two approaches: (i) dispersion of the preformed polymers; and (ii) polymerization of monomers. The commonly used polymers are: Arabian gum, gelatin, ethylcellulose, hydroxypropylmethylcellulose phthalate, poly-e-caprolactone (PCL), poly(lactide) (PLA), poly(lactide-co-glycolide) (PLGA), poly(alkyl cyanoacrylate) (PACA), while the polyelectrolytes are: polyethyleneimine (PEI), polyacrylic acid (PAA,), poly(diallyldimethylammonium, chloride) (PDADMAC), poly(sodium 4-styrene sulfonate) (PSS), and poly(allylamine hydrochloride) (PAH), dextran sulfate (DS), poly(glutamic acid) (PGA), alginates and alginic acid (ALG), hyaluronic acid (HYL), chitosan (CHIT), poly(lysine) (PLL). Synthetic polymers have higher purity and better reproducibility than natural ones, while natural ones possess better biocompatibility.17,19,30 Another class of polymeric nanoparticles is based on proteins or peptides. They are biological macromolecules composed of amino acids, with proved biocompatibility and bioavailability. Protein/peptide nanoparticles are widely used to enhance the bioavailability of drugs.27 Surface modification of nanocarriers for the preparation of multifunctional drug delivery systems eg for passive, active, and physical targeting, are performed.21,31,32
Polymeric nanocarriers have been increasing in interest and applied in the pharmaceutical field, especially as drug delivery systems (Figure 1). Since several specialized reviews have already discussed nanotechnology and polymeric materials for the formulation of nanocarriers,15–17,19,26 in this review, we present a brief introduction of the classical formulation techniques.
The Layer-by-Layer Method
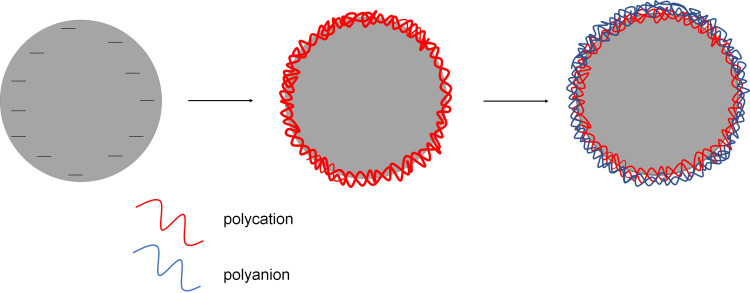
The sequential adsorption of oppositely charged nanoobjects (Layer-by-Layer (LbL) method) is a powerful approach for fabricating multilayer thin films.33,34 The origin of multilayered adsorption can be traced back to the mid-1960s when Iler proposed deposition of microparticles onto solid substrates in the LbL manner.35 In the 1990s, Decher and Hong expended the LbL by using a combination of linear polycations and polyanions.33 The layer-by-layer adsorption of charged nanoobjects like polyelectrolytes, nanoparticles, proteins, organic molecules, etc., is considered as a convenient method to obtain multilayer “nanocarriers” shells on colloidal cores (Figure 4), and those types of capsules have been the subject of intensive research since 1998 when Sukhoukovr proposed the method of forming polyelectrolyte multilayers shell on solid microparticles.36 Furthermore, the layer-by-layer method was applied for liquid and gas cores.37,38
Figure 4.
Schematic illustration of the layer by layer method.
The layer-by-layer (LbL) method’s main advantage are the ease of manipulation and the multifunctionality that comes from the possibility of modifying the polyelectrolyte shell by organic molecules, polymers, inorganic nanoparticles, carbon nanotubes, antibodies, lipids, or nanoparticles.39–41 That multifunctionality is of fundamental importance for the preparation of multifunctional drug delivery systems. For passive targeting, the polyelectrolyte shell is modified by the adsorption of so-called “stealth” polymers like poly(ethylene glycol) (PEG), poly(acrylamide), poly(vinylpyrrolidone), polysaccharides, or dextrans.42–44 That modification prolongs circulation time and allows nanocarriers’ accumulation in the targeted area through an EPR effect.4,5,45 For active targeting, polyelectrolyte shells can be functionalized by adsorption/incorporation of targeting ligands like antibodies or folic acids present on the surface of the targeted cells; for physical targeting, by the incorporation of stimuli-responsive building blocks in multilayered shell eg pH-responsive polymers, magnetic nanoparticles, etc. For example, the incorporation of magnetic nanoparticles that allows controlling magnetically responsive nanocarriers using an external magnetic field could be indicated.46–48 That incorporation allows the application of such nanocarriers as a tool for cell-biology research to separate and purify cell populations, tissue repair, magnetic drug delivery, magnetic resonance imaging (MRI), magnetic hyperthermia, etc.49–54 Excellent books and comprehensive reviews of LbL nanocarriers have been written.55–58
The Nanoprecipitation Method
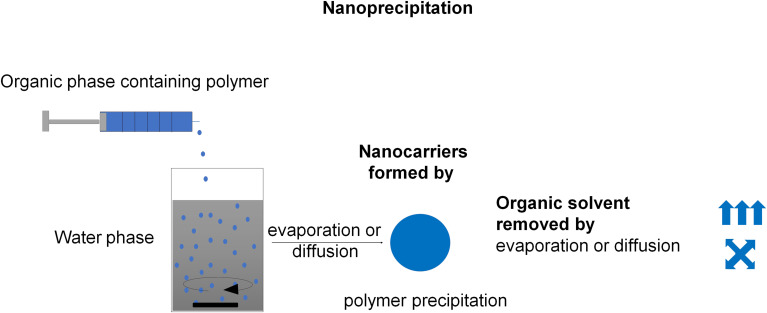
The nanoprecipitation method is also known as solvent-displacement, or interfacial deposition method, or solvent shifting method. According to Fessi et al, the nanocarrier synthesis needs both solvent and non-solvent phases (usually, the solvent and non-solvent phases are called organic and aqueous phases, respectively).59 A polymer and active substance solution in a miscible organic solvent or a mixture of solvents (ie ethanol, acetone, hexane, methylene chloride, or dioxane) is added to a nonsolvent or mixture of nonsolvents supplemented with one or more surfactants. It can be performed either by dialysis or by the dropping technique (Figure 5). After adding the organic phase to the water phase, the polymer diffuses with the organic solvent and precipitates at the interface between oil and water, forming nanocarriers.60 The principle of nanocarriers formation by nanoprecipitation is a fluctuation of polymer concentration at the interface between solvent and precipitant during the solvent to nonsolvent exchange.61 The initial supersaturation at the interface layer occurs, followed by the interfacial deposition of the polymer.17 The nanocarriers’ characteristics can be mainly influenced by polymer concentration, injection method of the organic phase (dialysis or dropping), the volume ratios components, and the nature of materials.61 The interfacial deposition method has been used intensively in the last decade, due to the simple operation without applied external high energy force and extensive applicability for various payloads. The studies of polymeric nanocarriers formulation by the nanoprecipitation method are excellently summarized in the following review.61–64
Figure 5.
Schematic illustration of the nanoprecipitation method. Nanocarriers are formed by polymer precipitation caused by organic solvent removal by evaporation or diffusion. Data from Weiss et al11 and Keech et al13.
The Nanoemulsion Templated Methods
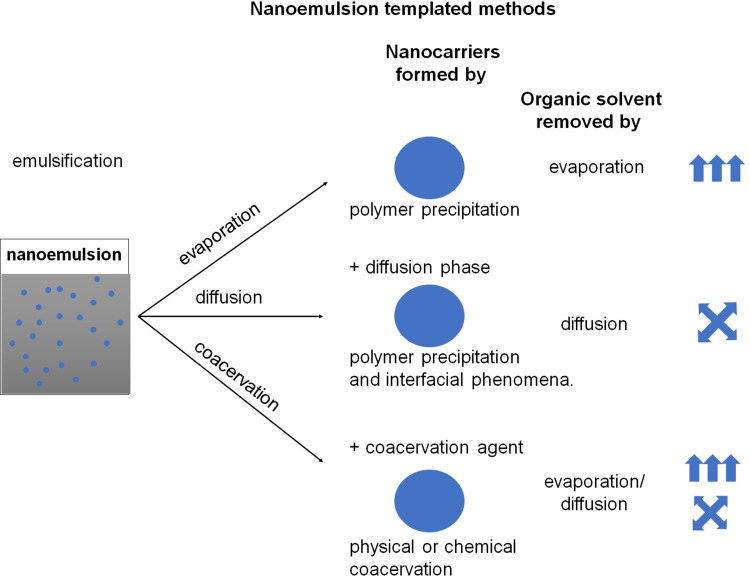
Nanoemulsions are thermodynamically unstable but kinetically stabilized systems.65,66 For nanoemulsion preparation, the organic or aqueous phase is emulsified in the aqueous or organic phase in the presence of a surfactant (stabilizing agents). During the nanoemulsion formation, surfactants are self-assembled at the interface between two immiscible phases to reduce the interfacial tension, thereby achieving a stable state. The various methods of formation of nanoemulsion including a high energy ones (eg, ultrasonication, high mechanical shear forces) and low energy ones (eg, phase inversion composition, phase inversion temperature, membrane emulsification, self-emulsification) have been developed.65,66 The size of the droplets of nanoemulsion is in the range of about 20–500 nm with a low polydispersity index. Nanodroplets found application as a template for polymeric nanocarrier synthesis both from preformed polymers and monomers in a polymerization process. The second approach is generally restricted to synthetic polymer materials, in which case one of the phases usually contains monomers, which in the polymerization process form polymeric nanocarriers. Here four main methods (Figure 6): emulsification-diffusion, emulsification-evaporation, emulsification-coacervation, multiple emulsification will be described; nevertheless, other nanoemulsions templating methods have been used such as nanoemulsion/polymer coating or nanoemulsion polymerization.19,67–69
Figure 6.
Schematic illustration of the nanoemulsion templated methods. Nanocarriers are formed by polymer precipitation, interfacial phenomena, or coacervation caused by organic solvent removal by evaporation or diffusion. 11 13Data from Weiss et al11 and Keech et al13.
Emulsification-Evaporation
In this method, the polymer and active substance containing the oil phase are emulsified into an aqueous solution in the presence of surfactants to make oil in water nanoemulsion. After the formation of nanoemulsion, the organic solvent is evaporated either by increasing the temperature/under pressure or by continuous stirring results in polymeric nanocarriers’ formation. Nanocarriers are formed by polymer precipitation during an evaporation procedure. The influence of process parameters on the properties of prepared polymeric nanocarriers has been discussed.16,17,19,26,32
Emulsification-Diffusion
In this method, the oil phase contains polymers and active substances dissolved in the organic solvent (eg dichloromethane or chloroform) and another solvent that is miscible with water (eg acetone, ethanol, or ethyl acetate), thereby removing them by diffusion into water.16,17,19,26,32 Such an oil phase is emulsified into the aqueous solution in the presence of surfactants to make oil in water nanoemulsion; subsequently, due to the spontaneous diffusion of water-miscible solvent, nanocarriers are formed by a combination of polymer precipitation and interfacial phenomena.
Emulsification-Coacervation
In the emulsification-coacervation method, polymeric nanocarriers are formed and stabilized by physical coacervation or chemical crosslinking at the interface. This method is principally used for materials (monomers or polymers) possessing crosslinking function groups. Physical coacervation (eg based on electrostatic interaction), as well as chemical ones (eg by chemical crosslinking or interfacial polymerization), have been proposed.16,17,19,26,32
Multiple Emulsification
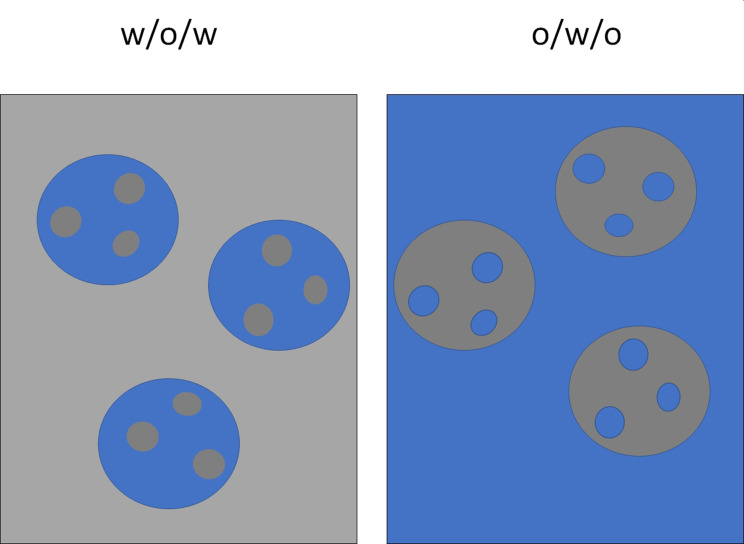
Multiple emulsions are heterodisperse systems called “emulsions of emulsions” formed when oil droplets in a continuous water phase contain dispersed water droplets (w/o/w).19 A hydrophilic active substance is dissolved in water, that is dispersed into the oil phase containing a dissolved polymer in the presence of a stabilizer. This primary/internal emulsion containing hydrophilic active substance is emulsified in an outer water phase again in the presence of a stabilizer. The procedure for obtaining the polymeric nanocarriers is similar to the single nanoemulsion technique for solvent removal, ie, diffusion, evaporation, coacervation, or a combination of these approaches. A reverse system w/o/w may also be formed (Figure 7).19,70
Figure 7.
Schematic representation of water in oil in water (w/o/w) and oil in water in oil o/w/o emulsions.
Polymeric Nanocarriers for Anti-Cancer Drugs
Chemotherapy remains one of the most important treatments for all cancer types, but its efficacy is lower than expected. The right approach to improve it seems to be developing better formulations, including nanoformulations of already approved drugs. However, the clinic’s nanoformulations are less toxic than conventional therapy; their therapeutic efficiency remains unchanged. Therefore, even better nanoformulations of anti-cancer drugs are continually being sought. Various polymeric nanocarriers have recently been successfully obtained and characterized in vivo and seem promising as the oral or intravenous transporters of the following chemotherapeutic agents: gemcitabine, diphenyl diselenide, etoposide, thiazolidinediones, imiquimod, methotrexate, or tamoxifen (Table 1).
Table 1.
Polymeric Nanocapsules Obtained and Characterized in vivo for Delivery of Various Anticancer Compounds
| Nanocarriers | Drug | Route of Administration | Animal Model | Targeting |
|---|---|---|---|---|
| PCL nanocarriers127 | 2-(2-Methoxyphenyl)-3-((Piperidin-1-yl)ethyl)thiazolidin-4-one | Orally | Healthy Wistar rats | Passive |
| PCL nanocarriers128 | Diphenyl diselenide | i.g. | Wistar rats, healthy or tumor-bearing rats after injection of C6 (rat glioma) cells in the right striatum | Passive |
| Polyurethane nanocarriers129 | Doxorubicin | - | Zebrafish | Active – mitochondria |
| PEGylated polypyrrole nanocarriers79 | Doxorubicin | i.v. | Tumor-bearing athymic nude mice after subcutaneous injection of Skov3 (human ovarian carcinoma) cells | Active – folate receptor |
| Spherical SiO2 structured PDADMAC, PAZO NaGdF4, Yb, Tm@NaYF4@NaGdF4, Nd@NaYF130 | Doxorubicin | i.v. | Tumor-bearing BALB/c nude mice after subcutaneous injection of U87-MG (human glioblastoma) cells | Light-triggered drug delivery |
| Poly-L-arginine nanocarriers131 | Elisidepsin | Orally | Healthy mice | Passive |
| PEGylated b-poly(L-aspartic acid) nanocarriers132 | Erlotinib | i.v. | Tumor-bearing BALB/c nude mice after subcutaneous injection of HCC-827 (human lung adenocarcinoma) cells | Passive |
| PEGylated lipid based nanosuspention133 | Etoposide | i.v. | Healthy Kunming mice and tumor-bearing BALB/c mice after subcutaneous injection of A20 (murine lymphoma) cells | Passive |
| PCLlipid core nanocarriers134 | Eugenol or acetyl eugenol | Orally or i.p. | Tumor-bearing C57Bl6 mice after subcutaneous injection of B16F10 (murine melanoma) cells | Passive |
| PEGylated lipid squalene nanocarriers135 | Gemcitabine | Convection-enhanced delivery (the infusion of therapeutic molecules directly into the brain) | Healthy Sprague- Dawley or tumor-bearing Fischer 344 rats after intracranial implantation of RG2 (rat glioma) cells | Passive |
| PEGylated PLGA nanocarriers136 | Glabrescione b | i.v. | Tumor-bearing SCID/Beige mice after subcutaneous injection of PANC-1 (human pancreas carcinoma) cells | Passive |
| PCLnanocarriers137 | Methotrexate | Orally or i.v. | Healthy or tumor-bearing C57Bl/6 mice after intracranial implantation of GL261 (murine glioma) cells | Passive |
| Chitosan and Eudragit S100 nanocarriers138 | Psoralidin | Orally | Healthy Sprague-Dawley rats | Passive |
| PCL nanocarriers139 | Resveratrol | i.p. | Tumor-bearing C57BL/6J mice after subcutaneous injection of B16F10 (murine melanoma) cells | Passive |
| PLA nanocarriers140 | Tamoxifen | i.v. | Tumor-bearing Swiss albino mice after subcutaneous injection of MCF-7 (human breast cancer estrogen receptor-positive) cells | Passive |
| Polydopamine nanocarriers141 | Zeolitic nanocrystals | i.p. | Healthy Sprague- Dawley rats | Passive |
Abbreviations: i.g., intragastric; i.p., intraperitoneal; i.v., intravenous; PCL, poly-e-caprolactone; PEG, poly(ethylene glycol); PLA, poly(lactide).
Very complex nanocarriers have been developed to deliver some drugs, eg, doxorubicin, to simultaneously increase drug delivery and precisely control drug release in the tumor. The free doxorubicin may cause cardiotoxicity in some patients leading to the discontinuation of therapy.71 Wang et al proposed the use of polymer nanocarriers made of reduction-responsive arginine-tethered amphiphilic copolymers and microwave absorbing ammonium bicarbonate for doxorubicin encapsulation. These nanocarriers can easily penetrate the tumor and are efficiently taken up by cancer cells. After irradiation by the pulsed microwave in the cell, in nanocarriers, ammonium bicarbonate converts the adsorbed energy to kinetic energy, which may generate thermocavitation and production of gas bubbles in cells. Consequently, intracellular structures are destroyed, and the doxorubicin releases from nanocarriers immediately target tumor cells.72 Still, other polymeric nanocarriers have been designed to release doxorubicin under the influence of reactive oxygen species (ROS). Because the oxidative environment is characteristic of tumors or tissues affected by chronic inflammation, the release of drugs can be most active there. It has been demonstrated for nanocarriers made of a biocompatible copolymer, namely phenylboronic acid pinacol ester-functionalized poly (ethylene glycol)-block-poly (phthalic anhydride-alter-glycidyl propargyl ether). Due to the phenylboronic acid ester’s presence, this nanocarrier was sensitive to oxidative stress and demonstrated an improved anti-cancer effect in a breast cancer mouse model.73 Another exciting approach is multidrug therapy using optimally selected agents, simultaneously or sequentially administered, to achieve a synergistic therapeutic effect. Attempts are being made to develop various nanomaterials, including polymeric nanocarriers, that would allow more than one drug to be delivered efficiently.74 Zhou et al proposed an attractive approach for sequential co-delivery of gefitinib (a small-molecule inhibitor of the epidermal growth factor receptor - EGFR) and doxorubicin. They obtained nanocarriers based on drug-polymer conjugation and ion pairing. Gefitinib was complexed with dioleoyl phosphatidic acid (DOPA) and encapsulated by coprecipitation with doxorubicin-conjugated poly(L-lactide)-block-polyethylene glycol (DOX-PLA-b-PEG). The biological activity of these nanocarriers was compared in vitro with Doxil (PEGylated liposome with doxorubicin - FDA approved doxorubicin nanoformulation), demonstrating a better anti-cancer effect against A549 (human non-small cell lung cancer) and MDA-MB-468 (human mammary adenocarcinoma, triple-negative breast cancer cells).
Moreover, the analysis of pharmacokinetic and biodistribution in the mouse model confirmed the high nanocapsule accumulation inside the breast tumors. However, the authors have not yet conducted studies on the developed nanocarriers’ anti-tumor activity in vivo.75 There are many examples of polymeric nanocarriers equipped with more than one chemotherapeutic. Nanoparticles with popular drugs, eg, doxorubicin, paclitaxel, curcumin, 5-fluorouracil, were proposed. In all cases, more than one anti-cancer agent’s encapsulation allows a better therapeutic effect than one drug (summarized in Table 2).
Table 2.
Polymeric Nanocarriers Obtained and Characterized in vivo for Delivery of More Than One Chemotherapeutic
| Nanocarriers | Drug | Route of Administration | Animal Model | Targeting |
|---|---|---|---|---|
| PLGA nanocarriers142 | Doxorubicin; berberine | i.v. | Healthy Sprague- Dawley rats | – |
| Poly(methyl vinyl ether-co-maleic anhydride) nanocarriers143 | Doxorubicin; selol | i.v. | Tumor-bearing BALB/c mice after subcutaneous injection of 4T1 (mouse mammary carcinoma) cells | – |
| PLGA nanocarriers144 | Oxaliplatin, irinotecan, and 5-fluorouracil | i.v. | Tumor-bearing BALB/c (nu/nu) nude mice after subcutaneous injection of SW1990 or Panc-1 (human pancreatic cancer) cells | – |
| PEGylated lipid bilayer coated mesoporous silica nanocarriers145 | Paclitaxel; curcumin | i.v. | Tumor-bearing BALB/c nude mice after subcutaneous injection of CIPp (canine breast cancer) cells | – |
| PLGA nanocarriers146,147 | Paclitaxel; perfluorooctyl bromide | i.v. | Tumor-bearing Fox1nu (nu/nu) mice after subcutaneous injection of CT-26 (murine colon cancer) cells | – |
Abbreviations: i.v., intravenous; PEG, poly(ethylene glycol); PLGA, poly(lactide-co-glycolide).
A more complex multifunctional nanocarrier system, namely RGD-PS-PEG calcium phosphate nanoparticles, was obtained and characterized previously by the team of Dong et al. They used the polymer RGD-PS-PEG (RGD corresponds to peptide H- (D-Val) -Arg-Gly-Asp-Glu-OH; PS-PEG is organic phosphatidylserine-polyethylene glycol) self-assembling into micelles to encapsulate the anti-cancer drug Novantrone (mitoxantrone hydrochloride). Moreover, calcium phosphate that adsorbs Verapamil was used to form an inorganic shell on the organic core (RGD-PS-PEG with Novantrol) to stabilize these nanoparticles.76 Because Verapamil is a P-gp inhibitor, these nanocarriers co-deliver a chemotherapeutic and anti-multidrug resistance agent. It is worth emphasizing that, in this case, the carrier was additionally actively targeted by the covalent coupling of PS-PEG with the RGD peptide. RGD is a cell adhesion motif displayed on extracellular matrix and is a ligand, for example, for αvβ3 or α5β3 integrins. The expression of these integrins is high on the tumor endothelium cells. Therefore RGD-functionalized nanocarriers may increase drug delivery to the tumor.77 In vivo studies performed after intravenous administration of RGD-PS-PEG calcium phosphate nanoparticles to animal bearing MCF7/MDR tumors (multidrug-resistant human breast cancer cells MC7) indeed revealed the increased concentration of encapsulated drugs in tumors, improved therapeutic effect, and reduced toxicity against healthy organs. Other examples of active delivery to the tumor are nanocarriers targeting folate receptors, namely hybrid lipid-core nanoparticles for co-delivery of doxorubicin and tanespimycin (inhibitor of a heat shock protein 90, chaperon of critical signaling proteins controlling, for example, the proliferation of cancer cells) or polypyrrole nanocarriers also with doxorubicin. Both these delivery systems have surface modified with PEG-folate. These nanocarriers’ in vivo results showed the beneficial effect of functionalization on drug accumulation, resulting in better therapeutic efficacy and lower toxicity to healthy tissues.78,79
There are also attempts at dual encapsulation of chemotherapy and inorganic nanoparticles. Jeon et al obtained paclitaxel loaded nanocarriers coated with crosslinked gold nanoparticles and poly(amidoamine) dendrimers. The therapeutic efficacy of this combination was confirmed in a mouse tumor model.80 Besides, superparamagnetic iron oxide nanoparticles (SPIONs) can be used for magnetic targeting. PLGA-PEG nanocarriers with SPION and docetaxel showed faster and stronger accumulation, followed by better tumor growth inhibition after local exposure to the magnetic field.81 Because encapsulated inorganic compounds are predominantly applied as contrast agents, PLGA-PEG nanocarriers were also used as a magnetically-targeted triple-modal imaging system. This time SPIONs and indocyanine green (fluorescence near-infrared dye) were encapsulated in the hydrophobic core, and diethylene triamine penta acetic acid was conjugated to PLGA-PEG in order to Indium-111 (radioisotope) chelation. This approach was tested on a mice model after intravenous injection and allowed for successful tumor visualization via fluorescence, MRI, and nuclear imaging.
Furthermore, the accumulation of nanocarriers, hence the signal intensity, was more significant after adding an external magnetic field around the tumor.82 Other examples of MRI contrast agents are semicrystalline polyurea nanocarriers with Gadobutrol (commercial contrast agent of gadolinium(III) complex), which poses enhanced tissue contrast compared to the free agent.83 Also, PEGylated nanocarriers with cubic cobalt ferrite nanocubes were detectable via MRI in the tumor tissue after its intratumoral injection.84 An exciting example is actively targeted perfluoropropane filled covered with gold nanoshelled PLGA nanocarriers functionalized with anti-VEGFR2 (vascular endothelial growth factor receptor type 2 overexpressed on tumor surrounding vascular endothelial cells) and anti-p53 antibodies for precise ultrasound molecular imaging of breast cancer. The authors have shown that dual-targeted nanocarriers accumulate in the tumor (orthotopic mice breast cancer) about two times more than non-targeted, ensuring accurate breast cancer detection. Interestingly, regardless of double targeting, the amount of the injected dosage detected in the tumor was 5%, which is still lower than expected.85
The easily modifiable thickness of the polymeric shell allows, for example, to obtain thin-shelled nanocarriers to deliver perfluorooctyl bromide, which finds application in ultrasonography as a contrasting agent. The liquid core of nanocarriers and the appropriately thin shell made of PLGA-PEG ensure very good echogenicity and acoustic response. Besides, nanocarriers’ surface functionalization with a monoclonal therapeutic antibody specific for EGFR (Cetuximab) improves the accumulation of capsules within tumors that overexpress this receptor.86
Anti-cancer theranostics uses advanced nanomaterials to simultaneously and accurately detect disease processes in the patient’s body and deliver drugs directly to areas affected by the disease. An interesting example is amphiphilic nanocarriers synthesized using block copolymer, succinic anhydride functionalized poly (2-diisopropylaminoethyl methacrylate) -block poly (2-aminoethyl methacrylate hydrochloride) with hydrophobic ruthenium entrapped in the core. The acidic tumor environment allows this compound’s active release, a known inhibitor of DNA replication with fluorescent properties.87 Exciting examples of anti-cancer theranostics are multifunctional polymeric PLGA nanocarriers designed for the High-Intensity Focused Ultrasound (HIFU) combined with transarterial chemoembolization treatment of hepatocellular carcinoma model in rabbits. The nanocapsule shell contained magnetic Fe3O4 as a contrast compound for MRI, and hydrophobic perfluorohexane was encapsulated into the core. HIFU is a method for local cancer treatment based on high temperatures leading to tissue destruction. Because perfluorohexane may increase energy deposition in cancer tissue, it is an enhancing agent for HIFU. Simultaneously, the embolization of blood vessels in the tumor allows retaining more nanocarriers within a tumor and improving therapy’s efficacy.88
Multifunctional, intelligent, and dual-stimulus responsive nanocarriers, functionalized with folate for active delivery of doxorubicin and indocyanine green (ICG), have also been obtained using PLGA – polyethylene glycol – poly (N-isopropyl acrylamide). ICG is known as a near-infrared fluorescence (NIR) imaging agent, as well as the photosensitizer in photodynamic therapy (PDT), or photothermal agent in photothermal therapy (PTT). At the same time, Fe/FeO nanocrystals were tethered with PLGA polymer, finally forming the shell of multifunctional nanocarriers. Fe/FeO nanocrystals are MRI contrasting agents, and they may provide additional, precise diagnostic information about the exact tumor location and size. After irradiation with NIR laser, nanocarriers shrink and degrade, releasing doxorubicin and ICG while Fe/FeO simultaneously degrades in the acidic tumor environment, leading to the overproduction of ROS as a result of the Fenton reaction. Results obtained in mouse tumor models suggest that it may help overcome hypoxia-related tumor resistance to PDT.89 Wang et al showed that the problem of insufficient oxygen in the tumor significantly limited the effectiveness of PDT, which can be solved by the simultaneous delivery of a photosensitizer, in this case, meso-tetra (p-hydroxyphenyl) porphine and catalase, an enzyme that decomposes endogenous tumor H2O2 into water and oxygen. This approach was possible due to the use of unique polymeric nanocarriers obtained due to the covalent coupling of catalase, meso-tetra (p-hydroxyphenyl) porphine, and PEG, and additionally labeled with 99mTc4+ for in vivo single-photon emission computed tomography imaging.90
ICG converts NIR light energy into local heat leading to cancer tissue ablation. However, cellular stress-induced during this therapy may activate autophagy in cancer cells, which is a complex process that eliminates damaged cellular structures. It can promote tumor cell survival and further tumor progression. That is why Wu et al have proposed yet another multifunctional system for the delivery of ICG and improve PTT’s effectiveness. It uses nanocarriers made of malPEG-b-poly (l-phenylalanine-co-l-aspartic acid), loaded with ICG and primaquine - autophagy inhibitor. The nanocarriers’ surface has been modified by covalently attaching to the polymer the so-called cell-penetrating peptide (CPP, Cys-KRPTMRFRYTWNPMK), allowing shielding of the entire capsule as a result of hyaluronic acid (HA) binding. HA actively directs nanocarriers to the tumor due to interaction with CD44 (HA receptor). Then, HA corona is decomposed in the tumor’s acidic environment, exposing CPP that facilitates nanocarriers uptake. The anti-cancer effects of these nanocarriers have already been confirmed by in vivo studies.91
The intelligent, PEGylated bismuth selenide nanocarriers were designed to deliver doxorubicin and yet another agent used in PDT/PTT, namely chlorin (Ce6). The entrapment of small drugs in bismuth selenide is simple because of their mesoporous and hollow structure (sponge-like structure). It is worth emphasizing that because previous studies confirmed the usefulness of bismuth selenide as a computed tomography contrasting agent, these nanocarriers enabled bimodal imaging of tumors, fluorescence, and CT. Fluorescence imaging confirmed efficient tumor accumulation of nanocarriers and remarkable tumor eradication. It resulted from the synergistic effect of doxorubicin and PTT/PDT-induced tumor ablation. At the same time, toxicity analysis revealed, the nanocarriers were not harmful to healthy tissues. The high therapy specificity resulted from the selective drug release primarily in the tumor. Protonation of the amino group of DOX under acidic tumor conditions altered, leading to increased drug hydrophilicity and its release from nanocarriers.
Additionally, upon NIR laser irradiation, there was also a release of drugs from the nanocarriers in the tumor.92 PEGylated, biodegradable hollow mesoporous organosilica nanocarriers were obtained to deliver up to 3 different anti-cancer compounds. In addition to ICG, they contained gemcitabine and 17 AAG, the heat-shock protein 90 inhibitors. The use of 17AAG allows to overcome tumor thermoresistance, the phenomenon regulated by heat shock proteins that are responsible for the proper functioning of selected cancer defense mechanisms, that affect anti-cancer effects of PTT.93
A unique example of theranostic nanocarriers can be peptide nanoparticles that possess fluorescent properties. The team of Fan et al obtained self-assembling nanoparticles based on octa-peptide cyclo - [(D-Ala-L-Glu-D-Ala-L-Trp) 2-] and Zn2+ ions and used them to deliver a chemotherapeutic compound, epirubicin. Studies performed on the mouse esophagus cancer model demonstrated that Zn2 + - coordinated cyclic peptides forming these nanoparticles emit NIR fluorescence due to quantum confinement within the nanostructures allowing tumors to be imaged without the use of any other imaging compounds. The presence of RGD on their surface ensured sufficient accumulation of nanocapsules in cancer.29
It is worth asking why so few polymeric nanocarriers intended for the transport of cancer drugs are functionalized with specific ligands (ie anti-CD19 or anti-Her2/ErbB2 monoclonal antibodies for active targeting of nanocarriers to cancer cells overexpressing these antigens).94
It is believed that actively targeted nanoparticles more effectively affect cancer. However, retention of even modified nanomaterials in tumors depends on EPR.95 Since most studies on the EPR effect have been conducted on rodent models, it is doubtful whether this effect occurs in humans and whether its functioning depends on the type of cancer.96,97 Also, in the case of human-emerging tumors, significant tumor volumes are elements of the so-called tumor microenvironment such as tumor-associated fibroblasts, immune cells, and extracellular matrix, which impede the nanoformulation of penetration into the tumor.98 Additionally, intratumor heterogeneity makes it difficult to select the appropriate molecular target and hampers actively targeted nanoparticles.99 It should also be mentioned that the vast majority of ligands for actively targeted nanodrugs only affect cancer cells without considering other tumor microenvironment forming cells. The problem is also the lack of efficient and cheap methods ensuring the attachment of ligands, especially those with a complex structure than RGD or folate (eg antibodies) while maintaining their biological activity. This makes the production of targeted nanodrugs expensive and cost-inefficient.100 These factors probably contribute to the phenomenon observed in the clinic, indicating the lack of improvement in the effectiveness of many drugs given in nanoformulation compared to their free forms.
Polymeric Nanocarriers as Helpful Tools in Mobilizing an Immune Response Directed Against Cancer or Pathogens
Tumor Targeted Immunotherapy
The application of nanomaterials in anti-cancer therapies is not limited to the transport of chemotherapeutics. Numerous studies indicate that nanomaterials, including polymeric nanocarriers, may also be used in cancer immunotherapy. Briefly, immunotherapy’s essential goal is to stimulate immune cells, primarily cytotoxic T lymphocytes (CTL), capable of eliminating cancer cells. Since strict CTL activation depends on their cooperation with antigen-presenting cells such as dendritic cells (DC) and macrophages, it is crucial to properly stimulate these cells’ activity, eg, by specific adjuvants. An interesting approach was using intradermally administered nanocarriers made of PLGA for providing low-molecular agonists (imidazoquinoline-based esters) of endosomal pathogen recognizing Toll-like receptor 7/9 (TLR7/9). Due to encapsulation, these compounds underwent effective delivery and uptake by DC, enhancing on their surface the expression of CTL co-stimulatory molecules (CD80, CD40, CD86) and inducing the expression of selected cytokines. This activation of dendritic cells led to CTL stimulation and a better anti-tumor immune response.101 In turn, the use of poly (β-amino ester)s copolymers assembling into nanocarriers with encapsulated IL-12 were efficiently delivered to the tumor after intravenous administration. This approach provides reversion of the tumor infiltrated macrophages from M2 to M1 phenotype, which resulted in a more effective anti-tumor response. It is worth emphasizing that the administration of IL-12 in nanocarriers allowed for a significant reduction of side effects resulting from this proinflammatory cytokine’s systemic activity.102 With the help of polymeric nanocarriers, cancer cells’ surface can also undergo modification by exposing them to specialized immune cells such as natural killer cells (NK cells). This approach effectively stimulates immune cells, but limits their activity only to the tumor, bypassing other tissues. Zheng et al received very complex polymeric nanocarriers, in which the first layer was made of bovine serum albumin, with a covalently attached antibody and phenylboronic acid. The outer layer was glucose-modified poly (2-methacryloyloxyethyl phosphorylcholine) -b-poly (N- (3-aminopropyl) -methacrylamine) associated with the albumin layer due to the interaction between phenylboronic acid and glucose. Because this interaction is fragile in the acid environment of the tumor, it is possible to disassemble the outer layer easily. It results in simultaneous exposure of phenylboronic acid capable of binding sialic acids present on the surface of the cancer cells. Then, nanocarriers bind to the surface of cancer cells, and due to the presence of an antibody to lure NK cells. These cells may employ a direct killing mechanism involving antibody-dependent cell-mediated cytotoxicity.103 As demonstrated recently, an interesting and even more comprehensive approach to overcoming tumor tolerance may be possible using nanocarriers whose shell is made of antigen (e.g, protein, overexpressed on tumor cells). Simultaneously, inside the carriers, there are two different adjuvants (resiquimod and muramyl dipeptide) stimulating DC. Such complex nanocarriers are, therefore, a therapeutic anti-cancer nano-vaccine. However, the immunostimulatory activity of these nanocarriers still requires verification in vivo.102
Components of Modern Vaccines
Nanoparticles indeed offer many advantages as modern therapeutic and preventive vaccines for tumor, viral, and bacterial antigens (e.g, recombinant proteins and nucleic acid) delivering. They might help effectively protect the integrity of the antigen, increase its immunogenicity, and, as a consequence, improve vaccine efficacy. For example, incorporating the antigen into the nanoparticles allows for more effective delivery primarily from the injection site (subcutaneous or intramuscular injection) via the lymphatic system to secondary lymphoid organs where adaptive immune response develops. However, not without significance is the fact that nanomaterials with antigen, due to their size, shape, density, surface charge, and chemical complexity, can mimic the pathogen. Thus nano-vaccines more effectively stimulate immune cells (B cells and T cells), leading to efficient antibody synthesis and long-term immunity. This approach has already been successfully implemented to prepare clinically approved the influenza vaccine Inflexal/Crucell, where virus haemagglutinin was incorporated into liposomes.104 Currently, there are various polymeric nanocarriers in development for the delivery of vaccine components. In that case, an antigen is displayed on the nanocarrier surface. It associates with shell-forming polymers already after the synthesis. The effectiveness of this process is monitored by analyzing changes in the zeta potential values. The research carried out on the mouse model revealed that the polymers that build nanocarrier shells are essential for the antigen’s immunogenicity embedded in/on the surface. Peleteiro et al confirmed the higher efficacy of protamine nanocarriers over polyarginine ones in the induction of an immune response against the recombinant hepatitis B antigen. Vaccination of animals with protamine nanocarriers resulted in the higher production of antigen-specific antibodies and selected cytokines, including IL-17A and IL-1β. These cytokines, due to their proinflammatory activity, play a fundamental role in creating the right environment for the developing immune response.105
An effective, but the slightly different solution is the use of two-layer nanocarriers, in which the antigen is trapped between two polymeric layers with opposite charges. Thus, an antigen is not exposed on the surface of the nanocarriers. Crecente-Canto analyzed in vivo on the mice model two different nanocapsule types: nanocarriers made of chitosan associated with bacterial antigen and nanocarriers in which the antigen bound to the chitosan layer was covered with a second layer of dextran sulfate. He concluded that coating the antigen with an additional layer of polymer may induce a better immune response. It is probably due to the increased stability of the delivered antigen, which is longer available for antigen-presentingcells.106 It is worth emphasizing that the effectiveness of many vaccines based on the polymeric nanocarriers, although satisfactory, still seems lower than vaccines based on the antigen administered together with the standard adjuvant Alum.105
Many researchers claim the effectiveness of nano-vaccines can be significantly increased by additional encapsulation molecules that modulate the immune response, such as imiquimod, CpG or polyI: C.107,108 Interestingly, Fichter et al proposed the use of nanocarriers built exclusively from hepatitis C virus nonstructural protein 5A and functionalized with adjuvant monophosphoryl lipid A (MPLA) as a vaccine against the hepatitis C virus. Studies conducted on a mouse model showed that after intravenous administration, these nanocarriers efficiently and preferentially accumulated primarily in the liver, and thanks to MPLA, which is a ligand for TLR4, were detectable in liver-resident antigen-presenting cells such as Kupffer cells (specialized macrophages located in the liver) or DCs leading to induction of intrahepatic immune response specific to the viral antigen. The authors also emphasized that an essential advantage of using nanocarriers made solely of antigen is the lack of induction of an immune response directed against the additional material present in nanocarriers providing viral antigen.109 Finally, polymeric nanocarriers can be useful in immunotherapy, aiming to induce immune tolerance to allergens (antigens). Briefly, this therapy involves supplying the body with a small amount of antigen to inhibit the synthesis of the underlying allergy molecules, namely IgE class antibodies and cytokines such as IL-4, IL-5, and IL-13. In vivo tests carried out by Smarr et al have shown that an effective and safe approach for the abrogation of allergen-specific response and inhibition of allergic inflammation can be the use of antigen-conjugated biodegradable PLGA, and antigen-encapsulated PLGA. It is worth emphasizing that, in this case, the antigen is encapsulated or covalently bound to the nanocapsule-building polymer.110
Regenerative Medicine and Topical Application
Nanomaterials have also found a broad application in regenerative medicine, which aims to develop and apply therapies that support the healing of tissue injuries resulting from diseases or mechanical damage. Encapsulation of phenytoin (stimulate fibroblast for cytokine and growth factor production) into PCL nanocarriers significantly reduced drug skin permeabilization compared to a free drug when supplied with chitosan hydrogel.111 Another example of polymeric nanocarriers embedded in a hydrogel was utilized as local anesthesia of the intraoral mucosa. The standard application of EMLA (a mixture of lidocaine and prilocaine) is limited because it inconveniences the patient (eg bitter taste and burning). Muniz et al proposed PCL nanocarriers for double encapsulation of lidocaine and prilocaine trapped in CARBOPOL (acrylic acid copolymer matrix) hydrogel. Studies in a rat model have shown that nanoformulation provides a better anesthesia duration compared to commercially available EMLA and free drugs embedded in the hydrogel.112
At the same time, tissue regeneration is a multistep process, and the use of more than one active agent and long-term sequential release of the drug at the appropriate stage of the healing process is required. An interesting example of a nanosystem used for wound healing is dual encapsulation of bromelain and salvianolic acid B into an ultrafine core-shell nanofiber built from PCL, poly (vinyl alcohol), and gelatin. The nanofibers were designed to release bromelain sequentially at an early stage, and salvianolic acid B at a later stage of tissue healing. It is essential because bromelain poses anti-inflammatory, antimicrobial, and anticoagulant properties necessary at the initial inflammation phase. Salvianolic acid B promotes wound healing by stimulating angiogenesis, re-epithelialization, and cell proliferation at proliferative, maturation, and remodeling phases of skin regeneration. During the in vitro evaluation, the authors proved biocompatibility, antibacterial, and proangiogenic properties of tested nanofibers. Besides, they confirmed that the subsequent sequential release of two compounds provides accelerated wound healing in rats compared to saline control and unloaded nanofibers.113
Because in the late phase of bone healing, the surrounding environment becomes more alkaline Tian et al proposed pH-dependent nanocarriers with growth factor - bone morphogenic protein-2 (nBMP-2). The authors used free-radical polymerization of N-(3-aminopropyl) methacrylamide, acrylamide, and glycerol dimethacrylate (degradable cross-linker) to synthesize alkaline-degradable polymeric shells. This solution provides a sustained release of BMP-2 at the therapeutic window. During research on the posterolateral spinal fusion in rats, nBMP-2 poses superior characteristics upon free BMP-2 and provides a better quality of newly formed bone and reduced undesirable inflammatory edema.114 Additionally, proteins like growth factors are sensitive to tissue proteases, which provide their short half-life in the body. Thus, the nanocarriers application also reduces the risk of transported cargo proteolysis.
Due to their pleiotropic properties, polyphenols were proposed for osteoarthritis treatment. To dual transport resveratrol and curcumin, self-assembling lipid-core nanocarriers with grape seed oil cores and PCL shells were proposed. Because of differences in the polyphenols solubility in oil, these drugs pose subsequent sequential releases from nanocarriers. The results indicate that this combination of polyphenols administered in nanocarriers protects chondrocytes against oxidative stress and shows better therapeutic efficacy against rat arthritis without undesired toxicity.115,116
There are also examples of the use of gene therapy in the treatment of osteoporosis. Sezlev Bilecen et al successfully proposed PLGA nanocarriers with siRNA inhibiting RANK expression (Receptor activator of nuclear factor κ B) protein, essential in the activation of osteoclasts.117 Furthermore, nanocarriers built of amphiphilic gelatin with encapsulated TGF-β1 (transforming growth factor-β1) and magnetic nanoparticles were proposed for cartilage regeneration. This approach combines magnetic guidance with chondrocyte stimulation via growth factor.118 The mentioned nanoformulation still requires evaluation of toxicity as well as therapeutic efficiency in vivo.
An interesting example is drug delivery as eye drops, which is limited due to the rapid dilution and clearance of the drug that follows tear production and blink reflex. The effectiveness of nanoparticles in this area is due to their improved corneal interaction. Reimondez-Troitiño et al presented oil nanodroplets surrounded by cell-penetrating cationic polypeptides - protamine or polyarginine for corneal wound healing. Even empty nanocarriers administered in eye drops were able to accelerate wound healing in vivo process similarly to commercially available Cacicol®. The use of protamine nanocarriers resulted from a decrease degree of corneal haze following tissue healing compared to polyarginine. This effect was probably due to the antifibrotic activity of protamine. However, additional encapsulation of cyclosporin A or vitamin A did not improve the therapy’s effectiveness compared to empty nanocarriers. All nanoformulations were non-toxic in a mouse ocular tolerance test.119
Lifestyle Diseases – Diabetes
Recently published papers indicate that polymeric nanocarriers could be used in the future for the oral delivery of drugs, in particular biological drugs such as peptides and proteins. Oral administration requires developing a formulation that will protect them from degradation and loss of biological activity in the gastrointestinal tract and allow their effective absorption.
Lifestyle diseases are a combination of diet, physical inactivity, and consumption of stimulants. They emerge more often each year, and polymeric nanocarriers have already been proposed as a drug delivery system for venous thromboembolism, hypertension, peripheral arterial disease, and inflammatory bowel disease (summaries in Table 3). Among these diseases, diabetes requires a new drug formulation improvement of patient safety. Many studies involve the development of an oral insulin formulation. This peptide hormone is widely used in treating type I diabetes and advanced type II diabetes when patients have inefficient insulin synthesis resulting from the destruction of the pancreatic beta-cells caused by prolonged hyperglycemia or dyslipidemia. Because insulin is a drug taken only by subcutaneous injection, developing an oral formulation would significantly improve patients’ comfort of life and reduce therapy costs. Lee et al received insulin loaded pluronic based nanocarriers. Interestingly, to ensure the nanocarriers’ best absorption in the small intestine, they armed them with two different ligands: chitosan and hexamer peptide AT-1002. Due to its interaction with the intestinal mucosa, chitosan allows nanocarriers to adhere to epithelial cells and improves interstitial permeability. Its activity is synergistic with the activity of AT-1002. AT-1002 is a peptide derived from zonula occludens toxin (ZOT), a virulence factor of Vibrio cholera, a pathogen responsible for the diarrhoeal disease cholera.120 Because AT-1002, like ZOT reversibly and transiently enhances the intestinal permeability by rearranging the intestinal cell cytoskeleton strategically located to modulate tight intercellular junctions, ensuring effective transport of nanocarriers across the epithelium to the blood.121 In vivo studies conducted in diabetic rats confirmed that the nanocarriers prepared in this way were effectively absorbed and that the encapsulated insulin retained biological activity. The slow insulin release profile from nanocarriers allowed normal blood glucose levels to be maintained up to 12 hours after oral administration.120 Later, Sun et al proposed a unique synthesis method termed flash nanocomplexation to obtain polyelectrolyte nanocomplexes to deliver insulin. First, insulin-loaded N- (2-hydroxy) -propyl-3-trimethylammonium-7-chloride modified chitosan/sodium tripolyphosphate nanocomplexes were produced as a nanocore. In the next step, they encapsulated nanocores into enteric Eudragit L100-55 polymer (polymer with mucoadhesive properties). Its task is to protect nanocomplexes with the drug against degradation in the digestive system and achieve a specific release of nanocomplexes in the ileum. Importantly, the authors claimed that nanocarrier synthesis’s optimized process is a scalable manner allowing easy clinical translation. The effectiveness of the obtained nanocarriers revealed studies on diabetic rats.122
Table 3.
Polymeric Nanocarriers Obtained and Characterized in vivo for Delivery of Different Drugs for Lifestyle Disease
| Nanocarriers | Transporting Agent | Route of Administration | Animal Model | Application | Targeting |
|---|---|---|---|---|---|
| PEGylated nanocarriers148 | Rivaroxaban | Orally or i.v. | Healthy or blood vessel inflammation-induced albino rats | Venous thromboembolism treatment | – |
|
Chitosan and gum arabic polymeric nanocarriers and polysorbate-80 nanocarriers149 |
Glycyrrhizin and thymoquinone | Orally | Streptozotocin and nicotinamide–induced diabetic Wistar albino rats | Diabetes mellitus treatment | – |
| Eudragit RSPO-Lutrol F 127 nanocarriers150 | Felodipine | Orally | Healthy Sprague- Dawley rats | Hypertension treatment | – |
|
PLGA nanocarriers151 |
Curcumin capped gold nanoparticles | s.c. | Enalapril-induced cardiac hypertrophy Wister rats | Cardiac hypertrophy reduction | – |
|
PCL-PEG nanocarriers152 |
Cilostazol | Orally | Healthy Wistar rats | Peripheral arterial disease treatment | – |
| Eudragit S 100 nanocarriers153 | Budesonide | Orally | Colitis-induced Wister albino rats | Inflammatory bowel disease treatment | pH-sensitive |
Abbreviations: i.v., intravenous; s.c., subcutaneous; PCL, poly-e-caprolactone; PEG, poly(ethylene glycol); PLGA, poly(lactide-co-glycolide).
Other Diseases
Nasal polymeric oil-core nanocarriers with shells covered with Eudragit RS100 (poly (ethyl acrylate-co-methyl methacrylate-co-trimethylamine methacrylate chloride) and with encapsulated steroid triamcinolone acetonide revealed excellent efficacy for the treatment of allergic rhinitis. Unique mucoadhesive properties of Eudragit polymers enabled excellent retention of nanocarriers in the nasal cavity, high local absorption of the drug, which limited its systemic distribution, and the resulting side effects.123 As other numerous studies have shown, polymeric nanocarriers could be used to formulate and deliver a wide variety of anti-inflammatory drugs (Table 4).
Table 4.
Polymeric Nanocarriers Obtained and Characterized in vivo for Delivery of Anti-Inflammatory Drugs
| Nanocarriers | Drug | Route of Administration | Animal Model | Targeting |
|---|---|---|---|---|
| Hyaluronic acid niosomes154 | Curcumin | Orally | Rats with carrageenan-induced paw edema | – |
| Starch-based nanocarriers155 | Neutrophil elastase inhibitor (ER143) | Topical | The croton oil-induced ear inflammation in BALB/c mice | – |
| PCL nanocarriers156 | Nerolidol | i.p. | Zymosan-induced arthritis in Swiss mice | – |
| PCL nanocarriers157 | Resveratrol | Orally | Challenged with LPS A/J mice | – |
| PEGylated nanocarriers158 | Meloxicam | Orally | Healthy C57BL/6 mice | – |
Abbreviations: i.p., intraperitoneal; PCL, poly-e-caprolactone; PEG, poly(ethylene glycol).
Gene therapy is a sophisticated approach to the treatment of different diseases based on nucleic acid delivery. A new method of CRISPR gene editing was developed. Chen et al utilized polymeric nanocarriers to transport the preassembled complex of Cas9 nuclease and a single-guide RNA (Cas RNP), which provided safe and effective CRISPR-mediated therapeutic gene editing. Because Cas RNP was surrounded by oppositely charged polymers, imidazole-containing monomer, covalently crosslinked glutathione, and acrylate methoxy poly(ethylene glycol), nanocarriers underwent endosomal escape followed by cytoplasmatic degradations. Subsequently, Cas RNP was able to enter the nucleus. Moreover, for in vivo experiments, nanocarriers were decorated with all-trans retinoic acid, which binds to the interphotoreceptor retinoid-binding protein. Results obtained for either subretinal or intramuscular injection have shown more effective genome editing compared to unencapsulated Cas RNP.124
One of the biggest challenges of modern medicine is undoubtedly treating central nervous system diseases such as neurodegenerative disease and cancer. The fundamental problem is a weak blood-brain barrier (BBB) permeability for small molecule drugs and therapeutic proteins. Hence, many studies on developing systems that allow the effective transport of therapeutics through BBB-forming endothelial cells. One of the more interesting approaches is the use of nanocarriers obtained from the degradable crosslinker, poly (lactide) -b-poly (ethylene glycol) -b-poly (lactide) -diacrylate triblock copolymer and 2-methacryloyloxyethyl phosphorylcholine. The latter compound is made of choline and acetylcholine analogs. Receptors for these neurotransmitters are commonly found on cells located within the central nervous system and on endothelial barrier cells of the brain; hence they can facilitate transcytosis of functionalized nanocarriers carrying therapeutic proteins (monoclonal antibodies or growth factors supporting the regeneration of damaged brain areas).125,126 Other examples of polymeric nanocarriers transporting drugs for neurological disorders are summarized in Table 5.
Table 5.
Polymeric Nanocarriers Obtained and Characterized in vivo for Delivery of Drugs for Neurological Disorders
| Nanocarriers | Transporting Agent | Route of Administration | Animal Model | Application | Targeting |
|---|---|---|---|---|---|
| PCL nanocarriers159 | p,p’-methoxyl-diphenyl diselenide | i.g. | Swiss mice with the chronic pain-like behavior induced by the partial sciatic nerve ligation surgery | Neuropathic pain treatment | – |
| Polysorbate 80; chitosan PEGylated nanocarriers160 | Clozapine | i.p. or i.v. | Healthy or amphetamine-induced pseudo-psychosis Wistar rats | Schizophrenia treatment | – |
| NFL-TBS.40–63 polypeptide nanocarriers161 | – | Injection in the brain right lateral ventricle or the spinal cord | Healthy Sprague- Dawley rats | Neuronal disorders treatment | Neural stem cells |
| PLGA nanocarriers containing triphenylphosphonium162 | Quercetin | Oral gavage | Cerebral ischemia-induced in Wistar rats | Neuro-protective role in cerebral ischemia-reperfusion | Mitochondria |
| Lecithin/chitosan nanocarriers163 | Simvastatin | Intranasally | Healthy Wistar rats | Neuro-degenerative diseases treatment | – |
Abbreviations: i.g., intragastric; i.p., intraperitoneal; i.v., intravenous; PCL, poly-e-caprolactone; PEG, poly(ethylene glycol); PLGA, poly(lactide-co-glycolide).
Furthermore, polymer nanocarriers have been used to treat various infectious diseases as transporters of antibacterial, antifungal, and anti-parasitic compounds (summarized in Table 6).
Table 6.
Polymeric Nanocarriers Obtained and Characterized in vivo for Delivery of Drugs Against Infectious Diseases
| Nanocarriers | Transporting Agent | Route of Administration | Animal Model | Application | Targeting |
|---|---|---|---|---|---|
| Eudragit L-100 nanocarriers164 | Curcumin | Orally | Healthy or Listeria monocytogenes-infected gerbils | Bacterial infection treatment | pH-sensitive |
| Cellulose acetate phthalate nanocarriers165 | Eugenol; Chlorhexidine | Orally | Healthy volunteers with periodontal disease | Bacterial infection treatment (periodontal disease) | pH-sensitive |
| PCL nanocarriers166 | Itraconazole | Topical application in the vagina | BALB/c mice inoculated intravaginally with Candida albicans | Fungal infection treatment (vulvovaginal candidiasis) | – |
| PLGA-PEG nanocarriers167–170 | Lychnopholide | Orally or i.v. | Healthy or Trypanosoma cruzi- infected Swiss mice; healthy C57BL/6 mice | Parasite infection treatment (Chagas disease) | – |
| PCL nanocarriers171 | Quercetin; penta-acetylated derivative of quercetin | Intragastric gavage | Leishmania amazonensis-infected BALB/c mice | Parasite infection treatment (Leishmaniasis) | – |
| Eudragit® RS100 or polysorbate 80 nanocarriers172 | Quinine | i.v. | Plasmodium berghei-infected Wistar rats | Parasite infection treatment (malaria) | – |
| Heparin nanocarriers173 | Artesunate | i.v. | Healthy BALB/c mice | Parasite infection treatment (malaria) | – |
| Polysorbate 80 nanocarriers or Eudragit® RS100 nanocarriers174,175 | Quinine; curcumin | i.p. | Wild-type N2 Bristol strain of Caenorhabditis elegans; Plasmodium berghei-infected Swiss mice | Parasite infection treatment (malaria) | – |
| PCL nanocarriers176 | Artemether | Orally | Healthy and Plasmodium berghei-infected C57BL/6 mice | Parasite infection treatment (malaria) | – |
Abbreviations: i.g., intragastric; i.p., intraperitoneal; i.v., intravenous; PCL, poly-e-caprolactone; PEG, poly(ethylene glycol); PLGA, poly(lactide-co-glycolide).
Final Remarks
The requirements that medicine places in modern therapies, especially those useful in treating such complex diseases as cancer, chronic inflammatory diseases, or autoimmune diseases, lead scientists to seek more and more advanced therapeutics. Modern therapies are simultaneously based on many drugs, often biochemically different, but their administration is expected to be safe and as low as possible for the patient. Hence the necessity to design biocompatible drug nanocarriers efficiently delivering drugs to the diseased tissue and ensuring their specific release. Recently performed studies indicate that polymeric nanomaterials could help in developing such nanocarriers and therapies of the future. The biocompatible polymers used to synthesize polymeric nanomaterials ensure low toxicity and allow obtaining complex and various nanocarriers. Polymeric nanocarriers are flexible in size, efficiently transport, and rapidly release cargo in the target tissue dependent on pH or temperature stimuli. However, these nanocarriers are often so complicated that it is difficult to predict the cost of mass production and how stable in vivo they would ultimately be. The usefulness of many polymeric nanomaterials has not even been studied in biological systems. Therefore, our knowledge about numerous advanced and complex nanosystems is still low. It is necessary to develop optimal schemes allowing for a detailed characterization of toxicity and verifying these nanomaterials’ effectiveness in vivo.
Acknowledgments
KSz acknowledges financial assistance from the statutory research fund of ICSC PAS.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Santos HA, Bimbo LM, Herranz B, Shahbazi MA, Hirvonen J, Salonen J. Nanostructured porous silicon in preclinical imaging: moving from bench to bedside. J Mater Res. 2013;28(2):152–164. doi: 10.1557/jmr.2012.271 [DOI] [Google Scholar]
- 2.Prasad D, Chauhan H. Key targeting approaches for pharmaceutical drug delivery. Am Pharm Rev. 2013;16(6). [Google Scholar]
- 3.Maeda H. Vascular permeability in cancer and infection as related to macromolecular drug delivery, with emphasis on the EPR effect for tumor-selective drug targeting. Proc Jpn Acad Ser B Phys Biol Sci. 2012;88(3):53–71. doi: 10.2183/pjab.88.53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yuan F, Dellian M, Fukumura D, et al. Vascular permeability in a human tumor xenograft: molecular size dependence and cutoff size. Cancer Res. 1995;55(17):3752–3756. [PubMed] [Google Scholar]
- 5.Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46(12Part 1):6387–6392. [PubMed] [Google Scholar]
- 6.Fang J, Sawa T, Maeda H. Factors and mechanism of “EPR” effect and the enhanced antitumor effects of macromolecular drugs including SMANCS. In: Maeda H., Kabanov A., Kataoka K., Okano T. editors, Advances in Experimental Medicine and Biology. Vol. 519. 2004:29–49. doi: 10.1007/0-306-47932-x_2 [DOI] [PubMed] [Google Scholar]
- 7.Anselmo AC, Mitragotri S. Nanoparticles in the clinic. Bioeng Transl Med. 2016;1(1):10–29. doi: 10.1002/btm2.10003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anselmo AC, Mitragotri S. Nanoparticles in the clinic: an update. Bioeng Transl Med. 2019;4(3):e10143. doi: 10.1002/btm2.10143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Borgå O, Lilienberg E, Bjermo H, Hansson F, Heldring N, Dediu R. Pharmacokinetics of total and unbound paclitaxel after administration of paclitaxel micellar or nab-paclitaxel: an open, randomized, cross-over, explorative study in breast cancer patients. Adv Ther. 2019;36(10):2825–2837. doi: 10.1007/s12325-019-01058-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fujiwara Y, Mukai H, Saeki T, et al. A multi-national, randomised, open-label, parallel, Phase III non-inferiority study comparing NK105 and paclitaxel in metastatic or recurrent breast cancer patients. Br J Cancer. 2019;120(5):475–480. doi: 10.1038/s41416-019-0391-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Weiss C, Carriere M, Fusco L, et al. Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano. 2020;14(6):6383–6406. doi: 10.1021/acsnano.0c03697 [DOI] [PubMed] [Google Scholar]
- 12.Florindo HF, Kleiner R, Vaskovich-Koubi D, et al. Immune-mediated approaches against COVID-19. Nat Nanotechnol. 2020;15(8):630–645. doi: 10.1038/s41565-020-0732-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Keech C, Albert G, Cho I, et al. Phase 1–2 Trial of a SARS-CoV-2 recombinant spike protein nanoparticle vaccine. N Engl J Med. 2020:NEJMoa2026920. doi: 10.1056/NEJMoa2026920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee DE, Koo H, Sun IC, Ryu JH, Kim K, Kwon IC. Multifunctional nanoparticles for multimodal imaging and theragnosis. Chem Soc Rev. 2012;41(7):2656–2672. doi: 10.1039/c2cs15261d [DOI] [PubMed] [Google Scholar]
- 15.Kothamasu P, Kanumur H, Ravur N, Maddu C, Parasuramrajam R, Thangavel S. Nanocapsules: the weapons for novel drug delivery systems. BioImpacts. 2012;2(2):71–81. doi: 10.5681/bi.2012.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Couvreur P, Barratt G, Fattal E, Legrand P, Vauthier C. Nanocapsule technology: a review. Crit Rev Ther Drug Carrier Syst. 2002;19(2):99–134. doi: 10.1615/CritRevTherDrugCarrierSyst.v19.i2.10 [DOI] [PubMed] [Google Scholar]
- 17.Deng S, Gigliobianco MR, Censi R, Di Martino P. Polymeric nanocapsules as nanotechnological alternative for drug delivery system: current status, challenges and opportunities. Nanomaterials. 2020;10(5):847. doi: 10.3390/nano10050847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Benita S. Microparticulate drug delivery systems: release kinetic models. Microspheres Microcapsules Liposomes. 1998;2:155–181. [Google Scholar]
- 19.Mora-Huertas CE, Fessi H, Elaissari A. Polymer-based nanocapsules for drug delivery. Int J Pharm. 2010;385(1–2):113–142. doi: 10.1016/j.ijpharm.2009.10.018 [DOI] [PubMed] [Google Scholar]
- 20.Ottenbrite RM, Kim SW. Polymeric Drugs and Drug Delivery Systems. CRC Press; 2019. doi: 10.1201/9780429136405 [DOI] [Google Scholar]
- 21.Rostamizadeh K, Torchilin VP. Polymeric nanomicelles as versatile tool for multidrug delivery in chemotherapy. In: Ranjita Shegokar editor, Nanopharmaceuticals. Zimmern: Elsevier; 2020:45–72. doi: 10.1016/b978-0-12-817778-5.00003-8 [DOI] [Google Scholar]
- 22.Pattni BS, Chupin VV, Torchilin VP. New developments in liposomal drug delivery. Chem Rev. 2015;115(19):10938–10966. doi: 10.1021/acs.chemrev.5b00046 [DOI] [PubMed] [Google Scholar]
- 23.Rahnfeld L, Luciani P. Injectable lipid-based depot formulations: where do we stand? Pharmaceutics. 2020;12(6):567. doi: 10.3390/PHARMACEUTICS12060567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pinto Reis C, Neufeld RJ, Ribeiro AJ, Veiga F. Nanoencapsulation I. Methods for preparation of drug-loaded polymeric nanoparticles. Nanomedicine. 2006;2(1):8–21. doi: 10.1016/j.nano.2005.12.003 [DOI] [PubMed] [Google Scholar]
- 25.Kumari A, Singla R, Guliani A, Yadav SK. Nanoencapsulation for drug delivery. EXCLI J. 2014;13:265–286. doi: 10.17877/DE290R-15592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Soppimath KS, Aminabhavi TM, Kulkarni AR, Rudzinski WE. Biodegradable polymeric nanoparticles as drug delivery devices. J Control Release. 2001;70(1–2):1–20. doi: 10.1016/S0168-3659(00)00339-4 [DOI] [PubMed] [Google Scholar]
- 27.Sun M, Hu H, Sun L, Fan Z. The application of biomacromolecules to improve oral absorption by enhanced intestinal permeability: A mini-review. Chin Chem Lett. 2020;31(7):1729–1736. doi: 10.1016/j.cclet.2020.02.035 [DOI] [Google Scholar]
- 28.Fu D, Liu D, Zhang L, Sun L. Self-assembled fluorescent tripeptide nanoparticles for bioimaging and drug delivery applications. Chin Chem Lett. 2020. doi: 10.1016/j.cclet.2020.07.011 [DOI] [Google Scholar]
- 29.Fan Z, Chang Y, Cui C, et al. Near infrared fluorescent peptide nanoparticles for enhancing esophageal cancer therapeutic efficacy. Nat Commun. 2018;9(1):1–11. doi: 10.1038/s41467-018-04763-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Khoee S, Yaghoobian M. An investigation into the role of surfactants in controlling particle size of polymeric nanocapsules containing penicillin-G in double emulsion. Eur J Med Chem. 2009;44(6):2392–2399. doi: 10.1016/j.ejmech.2008.09.045 [DOI] [PubMed] [Google Scholar]
- 31.Torchilin VP. Multifunctional nanocarriers. Adv Drug Deliv Rev. 2006;58(14):1532–1555. doi: 10.1016/j.addr.2006.09.009 [DOI] [PubMed] [Google Scholar]
- 32.Singh R, Lillard JW. Nanoparticle-based targeted drug delivery. Exp Mol Pathol. 2009;86(3):215–223. doi: 10.1016/j.yexmp.2008.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Decher G, Hong JD, Schmitt J. Buildup of ultrathin multilayer films by a self-assembly process: III. Consecutively alternating adsorption of anionic and cationic polyelectrolytes on charged surfaces. Thin Solid Films. 1992;210–211(PART 2):831–835. doi: 10.1016/0040-6090(92)90417-A [DOI] [Google Scholar]
- 34.Decher G. Fuzzy nanoassemblies: toward layered polymeric multicomposites. Science. 1997;277(5330):1232–1237. doi: 10.1126/science.277.5330.1232 [DOI] [Google Scholar]
- 35.Iler RK. Multilayers of colloidal particles. J Colloid Interface Sci. 1966;21(6):569–594. doi: 10.1016/0095-8522(66)90018-3 [DOI] [Google Scholar]
- 36.Sukhorukov GB, Donath E, Lichtenfeld H, et al. Layer-by-layer self assembly of polyelectrolytes on colloidal particles. Colloids Surf a Physicochem Eng Asp. 1998;137(1–3):253–266. doi: 10.1016/S0927-7757(98)00213-1 [DOI] [Google Scholar]
- 37.Szczepanowicz K, Bazylińska U, Pietkiewicz J, Szyk-Warszyńska L, Wilk KA, Warszyński P. Biocompatible long-sustained release oil-core polyelectrolyte nanocarriers: from controlling physical state and stability to biological impact. Adv Colloid Interface Sci. 2015;222:678–691. doi: 10.1016/j.cis.2014.10.005 [DOI] [PubMed] [Google Scholar]
- 38.Szczepanowicz K, Dronka-Góra D, Para G, Warszyński P. Encapsulation of liquid cores by layer-by-layer adsorption of polyelectrolytes. J Microencapsul. 2010;27(3):198–204. doi: 10.3109/02652040903052069 [DOI] [PubMed] [Google Scholar]
- 39.Shchukin DG, Sukhorukov GB. Nanoparticle synthesis in engineered organic nanoscale reactors. Adv Mater. 2004;16(8):671–682. doi: 10.1002/adma.200306466 [DOI] [Google Scholar]
- 40.Katagiri K, Caruso F. Monodisperse polyelectrolyte-supported asymmetric lipid-bilayer vesicles. Adv Mater. 2005;17(6):738–743. doi: 10.1002/adma.200401441 [DOI] [Google Scholar]
- 41.Angelatos AS, Katagiri K, Caruso F. Bioinspired colloidal systems via layer-by-layer assembly. Soft Matter. 2006;2(1):18–23. doi: 10.1039/b511930h [DOI] [PubMed] [Google Scholar]
- 42.Torchilin VP, Trubetskoy VS. Which polymers can make nanoparticulate drug carriers long-circulating? Adv Drug Deliv Rev. 1995;16(2–3):141–155. doi: 10.1016/0169-409X(95)00022-Y [DOI] [Google Scholar]
- 43.Moghimi SM, Hunter AC, Murray JC. Long-circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev. 2011;53(2):283–318. [PubMed] [Google Scholar]
- 44.Kingshott P, Griesser HJ. Surfaces that resist bioadhesion. Curr Opin Solid State Mater Sci. 1999;4(4):403–412. doi: 10.1016/S1359-0286(99)00018-2 [DOI] [Google Scholar]
- 45.Maeda H, Greish K, Fang J. The EPR effect and polymeric drugs: A paradigm shift for cancer chemotherapy in the 21st century. Adv Polym Sci. 2006;193(1):103–121. doi: 10.1007/12_026 [DOI] [Google Scholar]
- 46.Torchilin VP. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat Rev Drug Discov. 2014;13(11):813–827. doi: 10.1038/nrd4333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Polyak B, Friedman G. Magnetic targeting for site-specific drug delivery: applications and clinical potential. Expert Opin Drug Deliv. 2009;6(1):53–70. doi: 10.1517/17425240802662795 [DOI] [PubMed] [Google Scholar]
- 48.Torchilin VP. Drug targeting. Eur J Pharm Sci. 2000;11(SUPPL. 2):S81–S91. doi: 10.1016/S0928-0987(00)00166-4 [DOI] [PubMed] [Google Scholar]
- 49.Häfeli U, Schütt W, Teller J, Zborowski M. Scientific and Clinical Applications of Magnetic Carriers. Springer Science & Business Media; 1997. [Google Scholar]
- 50.Pankhurst QA, Connolly J, Jones SK, Dobson J. Applications of magnetic nanoparticles in biomedicine. J Phys D Appl Phys. 2003;36(13):R167. doi: 10.1088/0022-3727/49/50/501002 [DOI] [Google Scholar]
- 51.Reimer P, Weissleder R. Development and experimental use of receptor-specific MR contrast media. Radiologe. 1996;36(2):153–163. doi: 10.1007/s001170050053 [DOI] [PubMed] [Google Scholar]
- 52.Arbab AS, Bashaw LA, Miller BR, et al. Characterization of biophysical and metabolic properties of cells labeled with superparamagnetic iron oxide nanoparticles and transfection agent for cellular MR imaging. Radiology. 2003;229(3):838–846. doi: 10.1148/radiol.2293021215 [DOI] [PubMed] [Google Scholar]
- 53.Gupta AK, Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials. 2005;26(18):3995–4021. doi: 10.1016/j.biomaterials.2004.10.012 [DOI] [PubMed] [Google Scholar]
- 54.Chen S, Weissleder R. Polymer-Coated Iron Oxide Nanoparticles for Medical Imaging. Massachusetts Institute of Technology; 2010. [Google Scholar]
- 55.Vilela C, Figueiredo ARP, Silvestre AJD, Freire CSR. Multilayered materials based on biopolymers as drug delivery systems. Expert Opin Drug Deliv. 2017;14(2):189–200. doi: 10.1080/17425247.2016.1214568 [DOI] [PubMed] [Google Scholar]
- 56.Correa S, Dreaden EC, Gu L, Hammond PT. Engineering nanolayered particles for modular drug delivery. J Control Release. 2016;240:364–386. doi: 10.1016/j.jconrel.2016.01.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Picart C, Caruso F, Voegel J. Layer-By-Layer Films for Biomedical Applications. John Wiley & Sons; 2015. [Google Scholar]
- 58.Johnston APR, Cortez C, Angelatos AS, Caruso F. Layer-by-layer engineered capsules and their applications. Curr Opin Colloid Interface Sci. 2006;11(4):203–209. doi: 10.1016/j.cocis.2006.05.001 [DOI] [Google Scholar]
- 59.Fessi H, Puisieux F, Devissaguet J Process for preparing a colloidal and disperse system in the shape of nanocapsules; 1988.
- 60.Schubert S, Delaney, Jr JT, Schubert US. Nanoprecipitation and nanoformulation of polymers: from history to powerful possibilities beyond poly(lactic acid). Soft Matter. 2011;7(5):1581–1588. doi: 10.1039/c0sm00862a [DOI] [Google Scholar]
- 61.Gericke M, Schulze P, Heinze T. Nanoparticles based on hydrophobic polysaccharide derivatives—formation principles, characterization techniques, and biomedical applications. Macromol Biosci. 2020;20(4):1900415. doi: 10.1002/mabi.201900415 [DOI] [PubMed] [Google Scholar]
- 62.Galindo-Rodriguez S, Allémann E, Fessi H, Doelker E. Physicochemical parameters associated with nanoparticle formation in the salting-out, emulsification-diffusion, and nanoprecipitation methods. Pharm Res. 2004;21(8):1428–1439. doi: 10.1023/B:PHAM.0000036917.75634.be [DOI] [PubMed] [Google Scholar]
- 63.Hornig S, Heinze T, Becer CR, Schubert US. Synthetic polymeric nanoparticles by nanoprecipitation. J Mater Chem. 2009;19(23):3838–3840. doi: 10.1039/b906556n [DOI] [Google Scholar]
- 64.Fessi H, Puisieux F, Devissaguet JP, Ammoury N, Benita S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int J Pharm. 1989;55(1):R1–R4. doi: 10.1016/0378-5173(89)90281-0 [DOI] [Google Scholar]
- 65.Che Marzuki NH, Wahab RA, Abdul Hamid M. An overview of nanoemulsion: concepts of development and cosmeceutical applications. Biotechnol Biotechnol Equip. 2019;33(1):779–797. doi: 10.1080/13102818.2019.1620124 [DOI] [Google Scholar]
- 66.Gupta A, Eral HB, Hatton TA, Doyle PS. Nanoemulsions: formation, properties and applications. Soft Matter. 2016;12(11):2826–2841. doi: 10.1039/c5sm02958a [DOI] [PubMed] [Google Scholar]
- 67.Tiarks F, Landfester K, Antonietti M. Preparation of polymeric nanocapsules by miniemulsion polymerization. Langmuir. 2001;17(3):908–918. doi: 10.1021/la001276n [DOI] [Google Scholar]
- 68.Landfester K. Encapsulation through (Mini)Emulsion polymerization. In: Swapan Kumar Ghosh editor, Functional Coatings, Weinheim:John Wiley and Sons; 2006;29–66. doi: 10.1002/3527608478.ch2 [DOI] [Google Scholar]
- 69.Khan I, Saeed K, Khan I. Nanoparticles: properties, applications and toxicities. Arab J Chem. 2019;12(7):908–931. doi: 10.1016/j.arabjc.2017.05.011 [DOI] [Google Scholar]
- 70.Grigoriev DO, Miller R. Mono- and multilayer covered drops as carriers. Curr Opin Colloid Interface Sci. 2009;14(1):48–59. doi: 10.1016/j.cocis.2008.03.003 [DOI] [Google Scholar]
- 71.Dos Santos DS, Dos Santos Goldenberg RC. Doxorubicin-induced cardiotoxicity: from mechanisms to development of efficient therapy. In: Wenyong Tan editor. Cardiotoxicity. London: InTech; 2018;3–24. doi: 10.5772/intechopen.79588 [DOI] [Google Scholar]
- 72.Wang Z, Zhang Y, Cao B, et al. Explosible nanocapsules excited by pulsed microwaves for efficient thermoacoustic-chemo combination therapy. Nanoscale. 2019;11(4):1710–1719. doi: 10.1039/c8nr08498j [DOI] [PubMed] [Google Scholar]
- 73.Zhang Y, He P, Liu X, et al. A PEGylated alternating copolymer with oxidation-sensitive phenylboronic ester pendants for anticancer drug delivery. Biomater Sci. 2019;7(9):3898–3905. doi: 10.1039/c9bm00884e [DOI] [PubMed] [Google Scholar]
- 74.Harter P, Sehouli J, Kimmig R, et al. Addition of vandetanib to pegylated liposomal doxorubicin (PLD) in patients with recurrent ovarian cancer. A randomized phase I/II study of the AGO study group (AGO-OVAR 2.13). Invest New Drugs. 2013;31(6):1499–1504. doi: 10.1007/s10637-013-0011-3 [DOI] [PubMed] [Google Scholar]
- 75.Zhou Z, Jafari M, Sriram V, et al. Delayed sequential co-delivery of gefitinib and doxorubicin for targeted combination chemotherapy. Mol Pharm. 2017;14(12):4551–4559. doi: 10.1021/acs.molpharmaceut.7b00669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dong Y, Liao H, Yu J, et al. Incorporation of drug efflux inhibitor and chemotherapeutic agent into an inorganic/organic platform for the effective treatment of multidrug resistant breast cancer. J Nanobiotechnology. 2019;17(1):125. doi: 10.1186/s12951-019-0559-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang F, Li Y, Shen Y, Wang A, Wang S, Xie T. The functions and applications of RGD in tumor therapy and tissue engineering. Int J Mol Sci. 2013;14(7):13447–13462. doi: 10.3390/ijms140713447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gupta B, Pathak S, Poudel BK, et al. Folate receptor-targeted hybrid lipid-core nanocapsules for sequential delivery of doxorubicin and tanespimycin. Colloids Surf B Biointerfaces. 2017;155:83–92. doi: 10.1016/j.colsurfb.2017.04.010 [DOI] [PubMed] [Google Scholar]
- 79.Chen J, Li X, Sun Y, et al. Synthesis of size-tunable hollow polypyrrole nanostructures and their assembly into folate-targeting and pH-responsive anticancer drug-delivery agents. Chem Eur J. 2017;23(68):17279–17289. doi: 10.1002/chem.201702945 [DOI] [PubMed] [Google Scholar]
- 80.Jeong Y, Kim ST, Jiang Y, et al. Nanoparticle-dendrimer hybrid nanocapsules for therapeutic delivery. Nanomedicine. 2016;11(12):1571–1578. doi: 10.2217/nnm-2016-0034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Al-Jamal KT, Bai J, Wang JTW, et al. Magnetic Drug targeting: preclinical in vivo studies, mathematical modeling, and extrapolation to humans. Nano Lett. 2016;16(9):5652–5660. doi: 10.1021/acs.nanolett.6b02261 [DOI] [PubMed] [Google Scholar]
- 82.Bai J, Wang JTW, Rubio N, et al. Triple-modal imaging of magnetically-targeted nanocapsules in solid tumours in vivo. Theranostics. 2016;6(3):342–356. doi: 10.7150/thno.11918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schlegel I, Renz P, Simon J, et al. Highly Loaded semipermeable nanocapsules for magnetic resonance imaging. Macromol Biosci. 2018;18(4):e1700387. doi: 10.1002/mabi.201700387 [DOI] [PubMed] [Google Scholar]
- 84.Calcagno V, Vecchione R, Quagliariello V, et al. Oil core-PEG shell nanocarriers for in vivo MRI imaging. Adv Healthcare Mater. 2019;8(3):1801313. doi: 10.1002/adhm.201801313 [DOI] [PubMed] [Google Scholar]
- 85.Xu L, Du J, Wan CF, et al. Ultrasound molecular imaging of breast cancer in MCF-7 orthotopic mice using gold nanoshelled poly(lactic-co-glycolic acid) nanocapsules: A novel dual-targeted ultrasound contrast agent. Int J Nanomedicine. 2018;13:1791–1807. doi: 10.2147/IJN.S153993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Achmad A, Yamaguchi A, Hanaoka H, Tsushima Y. Thin-shelled PEGylated perfluorooctyl bromide nanocapsules for tumor-targeted ultrasound contrast agent. Contrast Media Mol Imaging. 2018;2018:1725323. doi: 10.1155/2018/1725323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chen L, Fu C, Deng Y, Wu W, Fu A. A pH-sensitive nanocarrier for tumor targeting: delivery of ruthenium complex for tumor theranostic by pH-sensitive nanocapsule. Pharm Res. 2016;33(12):2989–2998. doi: 10.1007/s11095-016-2021-2 [DOI] [PubMed] [Google Scholar]
- 88.You Y, Wang Z, Ran H, et al. Nanoparticle-enhanced synergistic HIFU ablation and transarterial chemoembolization for efficient cancer therapy. Nanoscale. 2016;8(7):4324–4339. doi: 10.1039/c5nr08292g [DOI] [PubMed] [Google Scholar]
- 89.Wang Z, Ju Y, Ali Z, et al. Near-infrared light and tumor microenvironment dual responsive size-switchable nanocapsules for multimodal tumor theranostics. Nat Commun. 2019;10(1):1–12. doi: 10.1038/s41467-019-12142-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wang H, Chao Y, Liu J, et al. Photosensitizer-crosslinked in-situ polymerization on catalase for tumor hypoxia modulation & enhanced photodynamic therapy. Biomaterials. 2018;181:310–317. doi: 10.1016/j.biomaterials.2018.08.011 [DOI] [PubMed] [Google Scholar]
- 91.Wu X, Wu Y, Wang Z, et al. A cascade-targeting nanocapsule for enhanced photothermal tumor therapy with aid of autophagy inhibition. Adv Healthcare Mater. 2018;7(11):1800121. doi: 10.1002/adhm.201800121 [DOI] [PubMed] [Google Scholar]
- 92.Sun L, Li Q, Zhang L, et al. Stimuli responsive PEGylated bismuth selenide hollow nanocapsules for fluorescence/CT imaging and light-driven multimodal tumor therapy. Biomater Sci. 2019;7(7):3025–3040. doi: 10.1039/c9bm00351g [DOI] [PubMed] [Google Scholar]
- 93.Wu J, Bremner DH, Niu S, et al. Chemodrug-gated biodegradable hollow mesoporous organosilica nanotheranostics for multimodal imaging-guided low-temperature photothermal therapy/chemotherapy of cancer. ACS Appl Mater Interfaces. 2018;10(49):42115–42126. doi: 10.1021/acsami.8b16448 [DOI] [PubMed] [Google Scholar]
- 94.Alqaraghuli HGJ, Kashanian S, Rafipour RA. Review on targeting nanoparticles for breast cancer. Curr Pharm Biotechnol. 2019;20(13):1087–1107. doi: 10.2174/1389201020666190731130001 [DOI] [PubMed] [Google Scholar]
- 95.Lee H, Hoang B, Fonge H, Reilly RM, Allen C. In vivo distribution of polymeric nanoparticles at the whole-body, tumor, and cellular levels. Pharm Res. 2010;27(11):2343–2355. doi: 10.1007/s11095-010-0068-z [DOI] [PubMed] [Google Scholar]
- 96.Prabhakar U, Maeda H, Jain RK, et al. Challenges and key considerations of the enhanced permeability and retention effect for nanomedicine drug delivery in oncology. Cancer Res. 2013;73(8):2412–2417. doi: 10.1158/0008-5472.CAN-12-4561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nakamura Y, Mochida A, Choyke PL, Kobayashi H. Nanodrug delivery: is the enhanced permeability and retention effect sufficient for curing cancer? Bioconjug Chem. 2016;27(10):2225–2238. doi: 10.1021/acs.bioconjchem.6b00437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Neesse A, Michl P, Frese KK, et al. Stromal biology and therapy in pancreatic cancer. Gut. 2011;60(6):861–868. doi: 10.1136/gut.2010.226092 [DOI] [PubMed] [Google Scholar]
- 99.Gerlinger M, Rowan AJ, Horswell S, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366(10):883–892. doi: 10.1056/NEJMoa1113205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cutler DM. Early returns from the era of precision medicine. JAMA. 2020;323(2):109–110. doi: 10.1001/jama.2019.20659 [DOI] [PubMed] [Google Scholar]
- 101.Kim H, Niu L, Larson P, et al. Polymeric nanoparticles encapsulating novel TLR7/8 agonists as immunostimulatory adjuvants for enhanced cancer immunotherapy. Biomaterials. 2018;164:38–53. doi: 10.1016/j.biomaterials.2018.02.034 [DOI] [PubMed] [Google Scholar]
- 102.Wang Y, Lin YX, Qiao SL, et al. Polymeric nanoparticles enable reversing macrophage in tumor microenvironment for immunotherapy. Biomaterials. 2017;112:153–163. doi: 10.1016/j.biomaterials.2016.09.034 [DOI] [PubMed] [Google Scholar]
- 103.Zheng C, Wang Q, Wang Y, et al. In situ modification of the tumor cell surface with immunomodulating nanoparticles for effective suppression of tumor growth in mice. Adv Mater. 2019;31(32):1902542. doi: 10.1002/adma.201902542 [DOI] [PubMed] [Google Scholar]
- 104.Bachmann MF, Jennings GT. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat Rev Immunol. 2010;10(11):787–796. doi: 10.1038/nri2868 [DOI] [PubMed] [Google Scholar]
- 105.Peleteiro M, Presas E, González-Aramundiz JV, et al. Polymeric nanocapsules for vaccine delivery: influence of the polymeric shell on the interaction with the immune system. Front Immunol. 2018;9:791. doi: 10.3389/fimmu.2018.00791 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Crecente-Campo J, Lorenzo-Abalde S, Mora A, et al. Bilayer polymeric nanocapsules: a formulation approach for a thermostable and adjuvanted E. coli antigen vaccine. J Control Release. 2018;286:20–32. doi: 10.1016/j.jconrel.2018.07.018 [DOI] [PubMed] [Google Scholar]
- 107.Correia-Pinto JF, Peleteiro M, Csaba N, González-Fernández Á, Alonso MJ. Multi-enveloping of particulated antigens with biopolymers and immunostimulant polynucleotides. J Drug Deliv Sci Technol. 2015;30:424–434. doi: 10.1016/j.jddst.2015.08.010 [DOI] [Google Scholar]
- 108.Vicente S, Peleteiro M, Díaz-Freitas B, Sanchez A, González-Fernández Á, Alonso MJ. Co-delivery of viral proteins and a TLR7 agonist from polysaccharide nanocapsules: A needle-free vaccination strategy. J Control Release. 2013;172(3):773–781. doi: 10.1016/j.jconrel.2013.09.012 [DOI] [PubMed] [Google Scholar]
- 109.Fichter M, Piradashvili K, Pietrzak-Nguyen A, et al. Polymeric hepatitis C virus non-structural protein 5A nanocapsules induce intrahepatic antigen-specific immune responses. Biomaterials. 2016;108:1–12. doi: 10.1016/j.biomaterials.2016.08.046 [DOI] [PubMed] [Google Scholar]
- 110.Smarr CB, Yap WT, Neef TP, et al. Biodegradable antigen-associated PLG nanoparticles tolerize Th2-mediated allergic airway inflammation pre- and postsensitization. Proc Natl Acad Sci U S A. 2016;113(18):5059–5064. doi: 10.1073/pnas.1505782113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Cardoso AM, de Oliveira EG, Coradini K, et al. Chitosan hydrogels containing nanoencapsulated phenytoin for cutaneous use: skin permeation/penetration and efficacy in wound healing. Mater Sci Eng C. 2019;96:205–217. doi: 10.1016/j.msec.2018.11.013 [DOI] [PubMed] [Google Scholar]
- 112.Muniz BV, Baratelli D, Di Carla S, et al. Hybrid hydrogel composed of polymeric nanocapsules co-loading lidocaine and prilocaine for topical intraoral anesthesia. Sci Rep. 2018;8(1):1–12. doi: 10.1038/s41598-018-36382-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Shoba E, Lakra R, Kiran MS, Korrapati PS. Fabrication of core-shell nanofibers for controlled delivery of bromelain and salvianolic acid B for skin regeneration in wound therapeutics. Biomed Mater. 2017;12(3). doi: 10.1088/1748-605X/aa6684 [DOI] [PubMed] [Google Scholar]
- 114.Tian H, Du J, Wen J, et al. Growth-factor nanocapsules that enable tunable controlled release for bone regeneration. ACS Nano. 2016;10(8):7362–7369. doi: 10.1021/acsnano.5b07950 [DOI] [PubMed] [Google Scholar]
- 115.Kann B, Spengler C, Coradini K, et al. Intracellular delivery of poorly soluble polyphenols: elucidating the interplay of self-assembling nanocarriers and human chondrocytes. Anal Chem. 2016;88(14):7014–7022. doi: 10.1021/acs.analchem.6b00199 [DOI] [PubMed] [Google Scholar]
- 116.Coradini K, Friedrich RB, Fonseca FN, et al. A novel approach to arthritis treatment based on resveratrol and curcumin co-encapsulated in lipid-core nanocapsules: in vivo studies. Eur J Pharm Sci. 2015;78:163–170. doi: 10.1016/j.ejps.2015.07.012 [DOI] [PubMed] [Google Scholar]
- 117.Sezlev Bilecen D, Uludag H, Hasirci V. Development of PEI-RANK siRNA complex loaded PLGA nanocapsules for the treatment of osteoporosis. Tissue Eng Part A. 2019;25(1–2):34–43. doi: 10.1089/ten.TEA.2017.0476 [DOI] [PubMed] [Google Scholar]
- 118.Chiang CS, Chen JY, Chiang MY, et al. Using the interplay of magnetic guidance and controlled TGF-β release from protein-based nanocapsules to stimulate chondrogenesis. Int J Nanomedicine. 2018;13:3177–3188. doi: 10.2147/IJN.S156284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Reimondez-Troitiño S, Alcalde I, Csaba N, et al. Polymeric nanocapsules: a potential new therapy for corneal wound healing. Drug Deliv Transl Res. 2016;6(6):708–721. doi: 10.1007/s13346-016-0312-0 [DOI] [PubMed] [Google Scholar]
- 120.Lee JH, Sahu A, Choi W Il, Lee JY, Tae G. ZOT-derived peptide and chitosan functionalized nanocarrier for oral delivery of protein drug. Biomaterials. 2016;103:160–169. doi: 10.1016/j.biomaterials.2016.06.059 [DOI] [PubMed] [Google Scholar]
- 121.Wang WL, Lu RL, DiPierro MR, Fasano A. Zonula occludin toxin, a microtubule binding protein. World J Gastroenterol. 2000;6(3):330–334. doi: 10.3748/wjg.v6.i3.330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sun L, Liu Z, Tian H, et al. Scalable manufacturing of enteric encapsulation systems for site-specific oral insulin delivery. Biomacromolecules. 2019;20(1):528–538. doi: 10.1021/acs.biomac.8b01530 [DOI] [PubMed] [Google Scholar]
- 123.Sallam MA, Helal HM, Mortada SM. Rationally designed nanocarriers for intranasal therapy of allergic rhinitis: influence of carrier type on in vivo nasal deposition. Int J Nanomedicine. 2016;11:2345–2357. doi: 10.2147/IJN.S98547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chen G, Abdeen AA, Wang Y, et al. A biodegradable nanocapsule delivers a Cas9 ribonucleoprotein complex for in vivo genome editing. Nat Nanotechnol. 2019;14(10):974–980. doi: 10.1038/s41565-019-0539-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wu D, Qin M, Xu D, et al. A bioinspired platform for effective delivery of protein therapeutics to the central nervous system. Adv Mater. 2019;31(18):1807557. doi: 10.1002/adma.201807557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Xu D, Wu D, Qin M, et al. Efficient delivery of nerve growth factors to the central nervous system for neural regeneration. Adv Mater. 2019;31(33):1900727. doi: 10.1002/adma.201900727 [DOI] [PubMed] [Google Scholar]
- 127.da Silveira EF, Ferreira LM, Gehrcke M, et al. 2-(2-Methoxyphenyl)-3-((Piperidin-1-yl)ethyl)thiazolidin-4-one-loaded polymeric nanocapsules: in vitro antiglioma activity and in vivo toxicity evaluation. Cell Mol Neurobiol. 2019;39(6):783–797. doi: 10.1007/s10571-019-00678-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ferreira LM, Azambuja JH, da Silveira EF, et al. Antitumor action of diphenyl diselenide nanocapsules: in vitro assessments and preclinical evidence in an animal model of glioblastoma multiforme. J Trace Elem Med Biol. 2019;55:180–189. doi: 10.1016/j.jtemb.2019.06.010 [DOI] [PubMed] [Google Scholar]
- 129.Pramanik SK, Sreedharan S, Singh H, et al. Mitochondria targeting non-isocyanate-based polyurethane nanocapsules for enzyme-triggered drug release. Bioconjug Chem. 2018;29(11):3532–3543. doi: 10.1021/acs.bioconjchem.8b00460 [DOI] [PubMed] [Google Scholar]
- 130.Zhao T, Wang P, Li Q, et al. Near-infrared triggered decomposition of nanocapsules with high tumor accumulation and stimuli responsive fast elimination. Angew Chem Int Ed. 2018;57(10):2611–2615. doi: 10.1002/anie.201711354 [DOI] [PubMed] [Google Scholar]
- 131.Lollo G, Gonzalez-Paredes A, Garcia-Fuentes M, Calvo P, Torres D, Alonso MJ. Polyarginine nanocapsules as a potential oral peptide delivery carrier. J Pharm Sci. 2017;106(2):611–618. doi: 10.1016/j.xphs.2016.09.029 [DOI] [PubMed] [Google Scholar]
- 132.Kim J, Ramasamy T, Choi JY, et al. PEGylated polypeptide lipid nanocapsules to enhance the anticancer efficacy of erlotinib in non-small cell lung cancer. Colloids Surf B Biointerfaces. 2017;150:393–401. doi: 10.1016/j.colsurfb.2016.11.002 [DOI] [PubMed] [Google Scholar]
- 133.Yin X, Han L, Mu S, et al. Preparation and evaluation of etoposide-loaded lipid-based nanosuspensions for high-dose treatment of lymphoma. Nanomedicine. 2019;14(11):1403–1427. doi: 10.2217/nnm-2018-0502 [DOI] [PubMed] [Google Scholar]
- 134.Drewes CC, Fiel LA, Bexiga CG, et al. Novel therapeutic mechanisms determine the effectiveness of lipid-core nanocapsules on melanoma models. Int J Nanomedicine. 2016;11:1261–1279. doi: 10.2147/IJN.S101543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gaudin A, Song E, King AR, et al. PEGylated squalenoyl-gemcitabine nanoparticles for the treatment of glioblastoma. Biomaterials. 2016;105:136–144. doi: 10.1016/j.biomaterials.2016.07.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Ingallina C, Costa PM, Ghirga F, et al. Polymeric glabrescione B nanocapsules for passive targeting of Hedgehog-dependent tumor therapy. Nanomedicine. 2017;12(7):711–728. doi: 10.2217/nnm-2016-0388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Pereira NRC, Loiola RA, Rodrigues SF, et al. Mechanisms of the effectiveness of poly(ε-caprolactone) lipid-core nanocapsules loaded with methotrexate on glioblastoma multiforme treatment. Int J Nanomedicine. 2018;13:4563–4573. doi: 10.2147/IJN.S168400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Yin J, Xiang C, Song X. Nanoencapsulation of psoralidin via chitosan and Eudragit S100 for enhancement of oral bioavailability. Int J Pharm. 2016;510(1):203–209. doi: 10.1016/j.ijpharm.2016.05.007 [DOI] [PubMed] [Google Scholar]
- 139.Carletto B, Berton J, Ferreira TN, et al. Resveratrol-loaded nanocapsules inhibit murine melanoma tumor growth. Colloids Surf B Biointerfaces. 2016;144:65–72. doi: 10.1016/j.colsurfb.2016.04.001 [DOI] [PubMed] [Google Scholar]
- 140.Ravikumara NR, Bharadwaj M, Madhusudhan B. Tamoxifen citrate-loaded poly(d,l) lactic acid nanoparticles: evaluation for their anticancer activity in vitro and in vivo. J Biomater Appl. 2016;31(5):755–772. doi: 10.1177/0885328216670561 [DOI] [PubMed] [Google Scholar]
- 141.Ran J, Wang C, Zhang J, et al. New insight into Polydopamine@ZIF-8 Nanohybrids: a zinc-releasing container for potential anticancer activity. Polymers. 2018;10(5):476. doi: 10.3390/polym10050476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Khan I, Joshi G, Nakhate KT, Kumar R, Gupta U. Nano-co-delivery of berberine and anticancer drug using PLGA nanoparticles: exploration of better anticancer activity and in vivo kinetics. Pharm Res. 2019;36(10):149. doi: 10.1007/s11095-019-2677-5 [DOI] [PubMed] [Google Scholar]
- 143.Ganassin R, Horst FH, Camargo NS, et al. Selol nanocapsules with a poly(methyl vinyl ether-co-maleic anhydride) shell conjugated to doxorubicin for combinatorial chemotherapy against murine breast adenocarcinoma in vivo. Artif Cells Nanomed Biotechnol. 2018;46(sup2):1046–1052. doi: 10.1080/21691401.2018.1478423 [DOI] [PubMed] [Google Scholar]
- 144.Xu Y, Hu B, Xu J, Wu J, Ye B. Preparation of biodegradable polymeric nanocapsules for treatment of malignant tumor using coaxial capillary microfluidic device. Cancer Biother Radiopharm. 2020. doi: 10.1089/cbr.2019.3412 [DOI] [PubMed] [Google Scholar]
- 145.Gao J, Fan K, Jin Y, et al. PEGylated lipid bilayer coated mesoporous silica nanoparticles co-delivery of paclitaxel and curcumin leads to increased tumor site drug accumulation and reduced tumor burden. Eur J Pharm Sci. 2019;140:105070. doi: 10.1016/j.ejps.2019.105070 [DOI] [PubMed] [Google Scholar]
- 146.Boissenot T, Fattal E, Bordat A, et al. Paclitaxel-loaded PEGylated nanocapsules of perfluorooctyl bromide as theranostic agents. Eur J Pharm Biopharm. 2016;108:136–144. doi: 10.1016/j.ejpb.2016.08.017 [DOI] [PubMed] [Google Scholar]
- 147.Boissenot T, Bordat A, Larrat B, et al. Ultrasound-induced mild hyperthermia improves the anticancer efficacy of both Taxol® and paclitaxel-loaded nanocapsules. J Control Release. 2017;264:219–227. doi: 10.1016/j.jconrel.2017.08.041 [DOI] [PubMed] [Google Scholar]
- 148.Abouhussein DMN, Bahaa El Din Mahmoud D, Mohammad FE. Design of a liquid nano-sized drug delivery system with enhanced solubility of rivaroxaban for venous thromboembolism management in paediatric patients and emergency cases. J Liposome Res. 2019;29(4):399–412. doi: 10.1080/08982104.2019.1576732 [DOI] [PubMed] [Google Scholar]
- 149.Rani R, Dahiya S, Dhingra D, et al. Antidiabetic activity enhancement in streptozotocin + nicotinamide-induced diabetic rats through combinational polymeric nanoformulation. Int J Nanomedicine. 2019;14:4383–4395. doi: 10.2147/IJN.S205319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Geroge JK, Verma PRP, Venkatesan J, et al. Studies on core-shell nanocapsules of felodipine: in vitro-in vivo evaluations. AAPS PharmSciTech. 2017;18(8):2871–2888. doi: 10.1208/s12249-017-0770-9 [DOI] [PubMed] [Google Scholar]
- 151.Liu Y, Liu Q, Liu Y, Ju F, Ma Q, He Q. In vivo evaluation of enhanced drug carrier efficiency and cardiac anti-hypertrophy therapeutic potential of nano-curcumin encapsulated photo-plasmonic nanoparticles combined polymerized nano-vesicles: A novel strategy. J Photochem Photobiol B. 2019;199:111619. doi: 10.1016/j.jphotobiol.2019.111619 [DOI] [PubMed] [Google Scholar]
- 152.Gomes MLS, da Silva Nascimento N, Borsato DM, et al. Long-lasting anti-platelet activity of cilostazol from poly(ε-caprolactone)-poly(ethylene glycol) blend nanocapsules. Mater Sci Eng C. 2019;94:694–702. doi: 10.1016/j.msec.2018.10.029 [DOI] [PubMed] [Google Scholar]
- 153.Qelliny MR, Aly UF, Elgarhy OH, Khaled KA. Budesonide-loaded Eudragit S 100 nanocapsules for the treatment of acetic acid-induced colitis in animal model. AAPS PharmSciTech. 2019;20(6):237. doi: 10.1208/s12249-019-1453-5 [DOI] [PubMed] [Google Scholar]
- 154.Sadeghi Ghadi Z, Ebrahimnejad P. Curcumin entrapped hyaluronan containing niosomes: preparation, characterisation and in vitro/in vivo evaluation. J Microencapsul. 2019;36(2):169–179. doi: 10.1080/02652048.2019.1617360 [DOI] [PubMed] [Google Scholar]
- 155.Marto J, Ruivo E, Lucas SD, et al. Starch nanocapsules containing a novel neutrophil elastase inhibitor with improved pharmaceutical performance. Eur J Pharm Biopharm. 2018;127:1–11. doi: 10.1016/j.ejpb.2018.01.011 [DOI] [PubMed] [Google Scholar]
- 156.Barros Silva Soares de Souza EP, Trindade GDGG, Lins Dantas Gomes MV, et al. Anti-inflammatory effect of nano-encapsulated nerolidol on zymosan-induced arthritis in mice. Food Chem Toxicol. 2020;135:110958. doi: 10.1016/j.fct.2019.110958 [DOI] [PubMed] [Google Scholar]
- 157.de Oliveira MTP, de Sa Coutinho D, de Souza ÉT, et al. Orally delivered resveratrol-loaded lipid-core nanocapsules ameliorate LPS-induced acute lung injury via the ERK and PI3K/Akt pathways. Int J Nanomedicine. 2019;14:5215–5228. doi: 10.2147/IJN.S200666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nishihira VSK, Fontana BD, Ianiski FR, et al. PEGylated meloxicam-loaded nanocapsules reverse in vitro damage on caspase activity and do not induce toxicity in cultured human lymphocytes and mice. Biomed Pharmacother. 2018;107:1259–1267. doi: 10.1016/j.biopha.2018.08.120 [DOI] [PubMed] [Google Scholar]
- 159.Marcondes Sari MH, Zborowski VA, Ferreira LM, et al. Enhanced pharmacological actions of p,p’-methoxyl-diphenyl diselenide-loaded polymeric nanocapsules in a mouse model of neuropathic pain: behavioral and molecular insights. J Trace Elem Med Biol. 2018;46:17–25. doi: 10.1016/j.jtemb.2017.11.002 [DOI] [PubMed] [Google Scholar]
- 160.Vieira SM, Michels LR, Roversi K, et al. A surface modification of clozapine-loaded nanocapsules improves their efficacy: a study of formulation development and biological assessment. Colloids Surf B Biointerfaces. 2016;145:748–756. doi: 10.1016/j.colsurfb.2016.05.065 [DOI] [PubMed] [Google Scholar]
- 161.Carradori D, Saulnier P, Préat V, Des Rieux A, Eyer J. NFL-lipid nanocapsules for brain neural stem cell targeting in vitro and in vivo. J Control Release. 2016;238:253–262. doi: 10.1016/j.jconrel.2016.08.006 [DOI] [PubMed] [Google Scholar]
- 162.Ghosh S, Sarkar S, Choudhury ST, Ghosh T, Das N. Triphenyl phosphonium coated nano-quercetin for oral delivery: neuroprotective effects in attenuating age related global moderate cerebral ischemia reperfusion injury in rats. Nanomedicine. 2017;13(8):2439–2450. doi: 10.1016/j.nano.2017.08.002 [DOI] [PubMed] [Google Scholar]
- 163.Clementino A, Batger M, Garrastazu G, et al. The nasal delivery of nanoencapsulated statins – an approach for brain delivery. Int J Nanomedicine. 2016;11:6575–6590. doi: 10.2147/IJN.S119033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Jaguezeski AM, Souza CF, Perin G, et al. Effect of free and nano-encapsulated curcumin on treatment and energetic metabolism of gerbils infected by Listeria monocytogenes. Microb Pathog. 2019;134:103564. doi: 10.1016/j.micpath.2019.103564 [DOI] [PubMed] [Google Scholar]
- 165.Vidal-Romero G, Zambrano-Zaragoza ML, Martínez-Acevedo L, Leyva-Gómez G, Mendoza-Elvira SE, Quintanar-Guerrero D. Design and evaluation of pH-dependent nanosystems based on cellulose acetate phthalate, nanoparticles loaded with chlorhexidine for periodontal treatment. Pharmaceutics. 2019;11(11):604. doi: 10.3390/pharmaceutics11110604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Lucena PA, Nascimento TL, Gaeti MPN, et al. In vivo vaginal fungal load reduction after treatment with itraconazole-loaded polycaprolactone-nanoparticles. J Biomed Nanotechnol. 2018;14(7):1347–1358. doi: 10.1166/jbn.2018.2574 [DOI] [PubMed] [Google Scholar]
- 167.Branquinho RT, De Mello CGC, Oliveira MT, et al. Lychnopholide in poly(D,L-lactide)-block-polyethylene glycol nanocapsules cures infection with a drug-resistant Trypanosoma cruzi strain at acute and chronic phases. Antimicrob Agents Chemother. 2020;64(4). doi: 10.1128/AAC.01937-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Branquinho RT, Pound-Lana G, Marques Milagre M, et al. Increased body exposure to new anti-trypanosomal through nanoencapsulation. Sci Rep. 2017;7(1). doi: 10.1038/s41598-017-08469-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Branquinho RT, Roy J, Farah C, et al. Biodegradable polymeric nanocapsules prevent cardiotoxicity of anti-trypanosomal lychnopholide. Sci Rep. 2017;7. doi: 10.1038/srep44998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.De Mello CGC, Branquinho RT, Oliveira MT, et al. Efficacy of lychnopholide polymeric nanocapsules after oral and intravenous administration in murine experimental Chagas disease. Antimicrob Agents Chemother. 2016;60(9):5215–5222. doi: 10.1128/AAC.00178-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Sousa-Batista AJ, Poletto FS, Philipon CIMS, Guterres SS, Pohlmann AR, Rossi-Bergmann B. Lipid-core nanocapsules increase the oral efficacy of quercetin in cutaneous leishmaniasis. Parasitology. 2017;144(13):1769–1774. doi: 10.1017/S003118201700097X [DOI] [PubMed] [Google Scholar]
- 172.Michels LR, Maciel TR, Nakama KA, et al. Effects of surface characteristics of polymeric nanocapsules on the pharmacokinetics and efficacy of antimalarial quinine. Int J Nanomedicine. 2019;14:10165–10178. doi: 10.2147/IJN.S227914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Ismail M, Du Y, Ling L, Li X. Artesunate-heparin conjugate based nanocapsules with improved pharmacokinetics to combat malaria. Int J Pharm. 2019;562:162–171. doi: 10.1016/j.ijpharm.2019.03.031 [DOI] [PubMed] [Google Scholar]
- 174.Velasques K, Maciel TR, de Castro Dal Forno AH, et al. Co-nanoencapsulation of antimalarial drugs increases their in vitro efficacy against Plasmodium falciparum and decreases their toxicity to Caenorhabditis elegans. Eur J Pharm Sci. 2018;118:1–12. doi: 10.1016/j.ejps.2018.03.014 [DOI] [PubMed] [Google Scholar]
- 175.Gomes GS, Maciel TR, Piegas EM, et al. Optimization of curcuma oil/quinine-loaded nanocapsules for malaria treatment. AAPS PharmSciTech. 2018;19(2):551–564. doi: 10.1208/s12249-017-0854-6 [DOI] [PubMed] [Google Scholar]
- 176.Souza ACM, Mosqueira VCF, Silveira APA, et al. Reduced cardiotoxicity and increased oral efficacy of artemether polymeric nanocapsules in Plasmodium berghei-infected mice. Parasitology. 2018;145(8):1075–1083. doi: 10.1017/S0031182017002207 [DOI] [PubMed] [Google Scholar]