Abstract
Serotonin is a neuromodulator implicated in depression that is often measured in real-time by fast-scan cyclic voltammetry (FSCV). A specialized “Jackson” waveform (JW, 0.2, 1.0 V, −0.1 V, 0.2 V, 1000 V/s) was developed to reduce serotonin fouling, but the 1.0 V switching potential limits sensitivity and electrodes still foul. The goal of this study was to test the effects of extending the FSCV switching potential to increase serotonin sensitivity and decrease fouling. We compared the Jackson waveform, the dopamine waveform (DA, −0.4 V, 1.3 V, 400 V/s), and two new waveforms: the extended serotonin waveform (ESW, 0.2, 1.3, −0.1, 0.2, 1000 V/s) and extended hold serotonin waveform (EHSW, 0.2, 1.3 (hold 1 ms), −0.1, 0.2, 400 V/s). The EHSW was the most sensitive (LOD = 0.6 nM), and the JW the least sensitive (LOD = 2.4 nM). With the Jackson waveform, electrode fouling was significant with repeated injections of serotonin or exposure to its metabolite, 5-hydroxyindoleacetic acid (5-HIAA). Using the extended waveforms, electrodes fouled 50% less than with the Jackson waveform for both analytes. No electrode fouling was observed with the dopamine waveform because of the negative holding potential. The Jackson waveform was the most selective for serotonin over dopamine (800x), and the ESW was also highly selective. All waveforms were useful for measuring serotonin with optogenetic stimulation in Drosophila larvae. These results provide new FSCV waveforms to measure dynamic serotonin changes with different experimental requirements, like high sensitivity (EHSW), high selectivity (ESW, JW), or eliminating electrode fouling (DA).
Keywords: fast-scan cyclic voltammetry, serotonin, 5-hydroxyindoleacetic acid, dopamine, Drosophila melanogaster, optogenetics, CsChrimson
Graphical Abstract

Introduction
Serotonin is a major neuromodulator in the brain that is important for mental health by regulating sleep, mood, and appetite.1,2 The serotonergic system is one of the main targets of antidepressants that treat depression and anxiety disorders, but their efficacies vary in individuals.3 Serotonin concentrations in the extracellular space are tightly regulated by serotonin receptors and serotonin transporters (SERT).3–5 Therefore, fast analytical techniques are needed to monitor real-time serotonin changes in the brain. Electrochemical techniques are commonly used to study neurotransmitters in vivo in order to understand their effects on specific behaviors and dysfunction in neurological diseases.1,2,6–8 In particular, fast-scan cyclic voltammetry (FSCV) at carbon-fiber microelectrodes (CFMEs) applies linear ramp potentials at fast scan rates for high sensitivity and rapid temporal resolution detection of neurotransmitter concentration changes.1,6,9–12 FSCV has revealed the dynamic co-release of serotonin and histamine in mammals13 and the mechanism of selective serotonin reuptake inhibitors (SSRIs) to increase serotonin concentrations.14–16 In addition, FSCV has been used to measure rapid release and uptake of serotonin in Drosophila larval ventral nerve cords.17–19 However, serotonin remains difficult to study with FSCV because it’s oxidative byproducts foul CFMEs during long-term experiments.20–23
Serotonin and its major downstream metabolite, 5-hydroxyindoleacetic acid (5-HIAA), produce highly reactive radicals during oxidation that polymerize to form films on the surface of the CFME.22,23 These films hinder electron transfer and cause electrode fouling, which decreases sensitivity and limits accurate measurements in vivo.21 The standard FSCV waveform for serotonin, termed the “Jackson” waveform, was proposed to ameliorate these issues and sweeps from 0.2 V to 1.0 V to −0.1 to 0.2 V at 1000 V/s.20 The Jackson waveform was originally applied to beveled disk electrodes, however, the Hashemi group showed it can also be applied to cylindrical CFMEs.21 The Jackson waveform is highly selective for serotonin compared to dopamine, but electrodes still foul with repeated measurements and long exposure to 5-HIAA.21,24 Surface coatings such as Nafion are commonly used to mitigate this fouling,21 however they slow electrode responses.25 A new FSCV waveform that prevents electrode fouling while maintaining high sensitivity and selectivity would be beneficial for studying real-time serotonin release.
Modified FSCV waveforms have been investigated for several neurotransmitters to understand how waveform parameters affect CFME sensitivity and fouling.8,24,26–28 The Wightman group extended the switching potential of the dopamine waveform from 1.0 V to 1.3 V to increase sensitivity,27 and later demonstrated higher switching potentials (≥1.3 V) broke carbon-carbon bonds on the surface of the fiber,29 also increasing surface oxide groups.8,11,30 The higher switching potential also renews the surface to remove impurities.29 Likewise, Keithley et al. designed “sawhorse” waveforms with an extended hold at 1.3 V and observed greater CFME sensitivity using higher scan rates (≥1000 V/s).26 Modified sawhorse and extended switching potential waveforms also improved adenosine and histamine detection.24,31 Although extending the switching potential enhanced detection of these neurotransmitters, the Jackson waveform has not been revisited in 25 years to improve serotonin detection.20
The goal of this study was to develop practical new waveforms for serotonin detection to reduce electrode fouling and increase sensitivity. We hypothesized that extending the switching potential would decrease fouling by renewing the CFME surface, and that holding at a higher switching potential would enhance these effects.29 We designed new serotonin waveforms to extend the Jackson waveform to 1.3 V with varied scan rates, and tested sawhorse waveforms to hold at 1.3 V. The traditional dopamine waveform was also tested. Electrodes fouled the most using the Jackson waveform with repeated serotonin measurements and long exposure to 5-HIAA, while electrodes using the dopamine waveform did not foul. The extended waveforms with 1.3 V switching potentials had decreased electrode fouling compared to the Jackson waveform and had the highest electrode sensitivity. Waveforms were characterized in vitro with optogenetic stimulation in fruit fly larvae and all were useful for stable serotonin detection. Overall, our study develops extended waveforms for serotonin detection that provide high sensitivity and low electrode fouling for measurements in vivo.
Experimental
Chemicals
Serotonin hydrochloride was purchased from Sigma Aldrich (St. Louis, MO). Dopamine hydrochloride and 5-hydroxyindoleacetic acid were purchased from Acros Organics (Morris Plains, NH). A 1 mM stock solution of each chemical was prepared in 0.1 M HClO4. Final working solutions were prepared by diluting a stock in phosphate buffer saline (PBS) (131.25 mM NaCl, 3.00 mM KCl, 10 mM NaH2PO4, 1.2 mM MgCl2, 2.0 mM Na2SO4, and 1.2 mM CaCl2 with the final pH adjusted to 7.4 with 1 M NaOH).
Microelectrode Preparation
CFMEs were prepared as previously described.24 A T-650 carbon fiber (Cytec Engineering Materials, West Patterson, NJ) with 7 μm diameter was aspirated into a standard 1.28 mm inner diameter × 0.68 mm outer diameter glass capillary tube (A-M Systems, Sequim, WA) with a vacuum pump. A capillary was then pulled by a vertical puller (Narishige, Tokyo, Japan) to make two electrodes. The exposed fiber was cut to 25–75 μm. The CFME was epoxied by dipping the tip of the fiber into a solution of 14% m-phenylenediamine hardener (Acros Organics, Morris Plains, NH) in Epon Resin 828 (Miller-Stephenson, Danbury, CT) at 80–85 °C for 30–40 seconds. The CFMEs were cured at 100 °C overnight and 150 °C for at least 8 hours.
Electrochemical Instrumentation
Initial flow cell fouling experiments were performed using a two-electrode system with a CFME working electrode backfilled with 1 M KCl.17,18,24,29 All potential measurements are reported versus a chloridized Ag/AgCl wire reference electrode, and experiments were performed in a grounded Faraday cage. Before experiments, electrode tips were soaked in isopropyl alcohol for a minimum of 10 minutes to clean the surface. Electrodes were connected to a ChemClamp potentiostat and headstage (Dagan, Minneapolis, MN). Data were collected with HDCV Analysis software (Department of Chemistry, University of North Carolina at Chapel Hill). Supplemental Figure 1 shows background charging currents for each waveform tested and describes background subtraction procedures. The flow-injection system consists of a six-port loop injector with an air actuator (Valco Instruments, Houston, TX). PBS buffer and test solutions were flowed at 2 mL/min using a syringe pump (Harvard Apparatus, Holliston, MA) through a flow cell with the CFME tip inserted in solution. Analyte was flowed by the electrode for 5 seconds. D. melanogaster in vitro experiments were performed using the same two-electrode system, except electrodes were connected to a WaveNeuro system (Pine Research, Durham, NC). CFMEs were precalibrated and post calibrated in vitro using a flow injection analysis to flow 1 μM serotonin solution by the electrode to determine the current response (Figure S2). The concentration of serotonin was determined in vitro using this calibration factor, since the measured oxidation peak current is linear with the serotonin concentration.32
Waveform Parameters
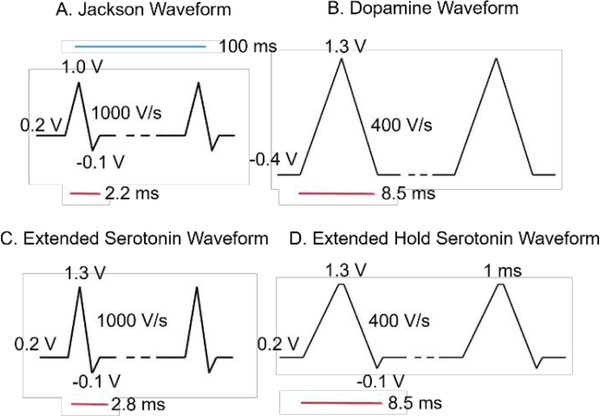
The traditional serotonin “Jackson” waveform (JW) proposed in Jackson et al., scans from 0.2 V to a switching potential of 1.0 V to −0.1 V back to the holding potential of 0.2 V at 1000 V/s (Fig. 1).20 The traditional dopamine waveform (DA) was tested that scans from −0.4 V to 1.3 V at 400 V/s. The extended serotonin waveform (ESW) extends the Jackson waveform to a switching potential at 1.3 V (0.2 V, 1.3 V, −0.1 V, 0.2 V, 1000 V/s). A sawhorse waveform, known as the extended hold serotonin waveform (EHSW) was similar to the ESW but the switching potential was held for 1 ms at 1.3 V (0.2 V, 1.3 V (1 ms), −0.1 V, 0.2 V, 400 V/s). A frequency of 10 Hz was used for all waveforms. A 2 KHz low-pass filter was applied for 400 V/s scan rates, and 10 kHz filter for 1000 V/s scan rates.
Figure 1.
Waveforms tested. A. Traditional serotonin “Jackson” waveform with a 1.0 V switching potential and 1000 V/s scan rate. B. Traditional dopamine waveform with a −0.4 V holding potential, extended 1.3 V switching potential, and 400 V/s scan rate. C. Extended serotonin waveform (ESW) with 1.3 V switching potential and 1000 V/s scan rate. D. Extended hold serotonin waveform (EHSW) with a 1 ms hold at 1.3 V and 400 V/s scan rate. All waveforms were repeated at 10 Hz.
Drosophila melanogaster Experiments
Methods were previously described in Privman et al. 2015.32 Virgin females with UAS-CsChrimson (Stockline #55136, Bloomington Drosophila Stock Center, Bloomington, IN) were crossed with tph-Gal4 (Serotonin driver line), a gift from Dr. Jay Hirsh (University of Virginia, Biology Department) and resulting heterozygous larvae were shielded from light and raised on standard food mixed 250:1 with 100 mM all-trans retinal (Sigma-Aldrich). The ventral nerve cords (VNCs) of third instar “wandering” larvae were dissected out in PBS buffer kept on ice. A VNC was placed on an uncoated Petri dish dorsal side down, and a small slice of the lateral optic lobe was removed using the tip of a 22-gauge hypodermic needle. The electrode was implanted from the lateral edge of the tissue into the dorsal medial protocerebrum. Dissection and electrode insertion were conducted under low light conditions. The electrode was allowed to equilibrate in the tissue for 10 minutes in the dark prior to data collection.
Optogenetic release of serotonin was stimulated by activating CsChrimson ion channels with red light from a 617 nm fiber-coupled high-power LED with a 200 μm core optical cable (ThorLabs, Newton, NJ, USA).32 The fiber was centered above the VNC using a micromanipulator and the light was modulated with transistor–transistor logic (TTL) inputs to a T-cube LED controller (ThorLabs), which was connected to the FSCV breakout box. TTL input was driven by electrical pulses controlled by the WaveNeuro system and HDCV software, which were used to control frequency, pulse width, and number of pulses. For in vitro experiments, 120 biphasic pulses were delivered at 60 Hz and pulse width of 4 ms. Stimulations were repeated every 5 minutes to allow the releasable pool of serotonin to replenish itself.17
Serotonin Imaging in Larvae Ventral Nerve Cords
Drosophila were bred to yield a tph-Gal4/CyO; UAS-mCD8-GFP cross, a gift from Dr. Jeffery Copeland (Eastern Mennonite University, Biology Department). Third instar larvae were collected and dissected as stated above but raised in normal light conditions.32 For imaging preparation, several VNCs were collected and placed in a Petri dish with cold PBS on ice. VNCs were preserved by removing PBS and pipetting 2–3 mL of 4% paraformaldehyde in PBS solution (Alfa Aesar, Ward Hill, MA). The petri dishes were covered in Parafilm (Bemis, Neenah, WI) and gently rocked for 20 minutes on a Nutating Mixer (VWR International, Radnor, PA). Paraformaldehyde was removed and 2 mL PBS was applied for 20 minutes as an initial wash, followed by two 5-minute wash steps. A glass slide was prepared by placing preserved specimens dorsal side up in a 60 μL aliquot of Vectashield (Vector Laboratories, Burlingame, CA). A Zeiss AxioZoom macroscope (Carl Zeiss Microscopy, Germany) was used to image GFP expression with Image J software (National Institutes of Health).
Statistics
Data are the mean ± the standard error of the mean (SEM) for n number of electrodes. Statistics were performed in GraphPad Prism 8.0 (GraphPad Software, La Jolla, CA). For One-Way ANOVA, Two-Way ANOVA, and Tukey’s post-hoc test, significance was determined at 95% confidence level.
For sensitivity and selectivity determination, the limit of detection (LOD) for serotonin and dopamine were calculated from the lowest concentrations tested: 100 nM serotonin (all waveforms), 100 nM dopamine (DA and EHSW), 1 μM dopamine (ESW), and 10 μM dopamine (JW). LOD is calculated by a ratio method, as the ratio of the measured S/N to the tested concentration is equal to the LOD divided by 3. Noise was determined by calculating the standard deviation (SD) of the baseline current from 0–3 s in the i vs t trace (n = 30).
Results and Discussion
Waveform Characteristics
We designed and tested new serotonin waveforms by varying the switching potential, holding potential, and scan rate to determine their sensitivity, selectivity, and electrode fouling behaviors. Figure 1 shows the main waveforms tested and Table 1 summarizes waveform parameters. The Jackson waveform was compared to the traditional dopamine waveform that uses a negative holding potential and extended switching potential. The extended serotonin waveform (ESW) extends the Jackson waveform to 1.3 V, but uses the same 1000 V/s scan rate. The extended hold serotonin waveform (EHSW) is a sawhorse waveform that extends the applied switching potential at 1.3 V for 1 ms with a slower scan rate at 400 V/s. A 1 ms hold was chosen because holds ≥1 ms do not produce higher or different current responses, but a 1 ms hold oxidizes the surface more than a 0.5 ms hold.31 Our hypothesis is that extending the switching potential will decrease fouling and increase sensitivity for serotonin by continuously regenerating the carbon fiber surface.29 In addition to the main waveforms, scan rate was also varied for the ESW and EHSW (Figure S3).
Table 1.
Summary of waveform parameters.
| Waveform | Switching Potential (V) | Holding Potential (V) | Scan Rate (V/s) |
|---|---|---|---|
| Jackson | 1.0 | 0.2 | 1000 |
| Dopamine | 1.3 | −0.4 | 400 |
| ESW | 1.3 | 0.2 | 1000 |
| EHSW | 1.3 (1 ms) | 0.2 | 400 |
Figure 2 shows example false color plots and cyclic voltammograms (CVs) for each waveform. The Jackson waveform (Fig. 2A) uses a high scan rate that shifts the oxidation peak to approximately 0.9 V. The characteristic reduction peak at 0.0 V is difficult to see on the CV because of the fast scan rate and potential that sweeps to only −0.1 V. However, the reduction peak is observed on the false color plot. The serotonin CV for the dopamine waveform shows full oxidation and reduction peaks at 0.6 V and 0.0 V, respectively (Fig. 2B). The serotonin CV for the ESW is similar to the Jackson waveform; however, the oxidation peak is fully observed because of the extension to 1.3 V (Fig. 2C). The cyclic voltammogram for the EHSW (Fig. 2D) is similar to the dopamine waveform and shows similar oxidation and reduction peaks. The reduction peak is easier to identify than the Jackson waveform and ESW because of the slower scan rate (400 V/s).
Figure 2.
Example false color plots (above) and cyclic voltammograms (CV, below) for all waveforms for 1 μM serotonin injection in a flow cell. Color plots show oxidation (green) and reduction (blue) for serotonin. A. The Jackson waveform displays a shifted oxidation peak in the CV, and the reduction peak is harder to see because of the −0.1 V negative potential. Both oxidation and reduction peaks are observed in the color plot. B. The dopamine waveform shows a complete CV with fully resolved oxidation and reduction peaks at approximately 0.6 V and 0.0 V, respectively. C. The extended serotonin waveform (ESW) extends the switching potential to 1.3 V with 1000 V/s scan rate and thus, the CV is similar to the Jackson waveform, except its oxidation peak is fully resolved. D. The extended hold serotonin waveform (EHSW) applies a 1 ms hold at 1.3 V (400 V/s) and the CV shows fully resolved oxidation and reduction peaks similar to the dopamine waveform.
Repeated serotonin measurement fouling
Serotonin undergoes irreversible oxidation and produces a series of radicals that dimerize and extend to form a polymer (Figure S4).20–23 This serotonin polymer electropolymerizes to the carbon fiber and forms films that hinder electron transfer. To test electrode fouling, 25 repeated serotonin injections were made for 5 seconds every 30 seconds using flow injection analysis. A CFME fouls if the current decreases from the initial current of the first injection. Figure 3A-D shows cyclic voltammograms for the initial (black) and 25th injections (colored) for each waveform. Electrodes using the Jackson waveform (Fig. 3A) fouled the most, with a 39 ± 3% average current decrease after 25 injections (n = 6). No fouling was observed with electrodes using the dopamine waveform (Fig. 3B) and current decreased only 5 ± 2%. Electrode fouling was similar for the ESW (Fig. 3C) and EHSW (Fig. 3D) with 19 ± 2% and 18 ± 4% current decrease, respectively. Fig. 3E compares electrode fouling for the repeated injections among waveforms. There were signficant overall effects of waveform applied (Two-Way ANOVA, F(3,20) = 26.75, p = 0.0001, n = 6) and injection number (F(24,480) = 66.34, p = 0.0001) with significant interaction between the groups (F(72,480) = 11.51, p = 0.0001). Tukey’s post-hoc test revealed significant differences in electrode fouling with the Jackson waveform compared to the dopamine waveform, ESW, and EHSW (all p = 0.0001). However, no differences in electrode fouling were observed between the other waveforms (p > 0.05). To test the effect of scan rate on electrode fouling, the ESW and EHSW were tested at 400 and 1000 V/s (Fig. S3). No differences were observed with scan rate for the EHSW. However, electrodes using the ESW fouled more at 400 V/s (39 ± 2%) compared to 1000 V/s (19 ± 2%), so 1000 V/s was chosen as the optimal scan rate.
Figure 3.
Repeated serotonin measurement electrode fouling was determined by injecting 1 μM serotonin for 5 seconds every 30 seconds repeated 25 times in a flow cell. Cyclic voltammograms show initial (1st, black) and final (25th, color) injections for each waveform. A. The current for the 25th CV is significantly reduced with the Jackson waveform. B. Electrode current responses using the dopamine waveform were stable. Electrodes using the C.ESW and D. EHSW had slight decreases in current for the 25th injection. E. Comparison of all waveforms for fouling with repeated injections of 1 μM serotonin. Plot shows normalized current (to the first injection) with standard error of the mean (SEM) error bars. There were significant main effects of waveform (Two-Way ANOVA, F(3,20) = 26.75, p = 0.0001, n = 6) and injection number (F(24,480) = 66.34, p = 0.0001) on current detected. Electrode fouling with the Jackson waveform was significantly different compared to electrodes using the dopamine waveform, ESW, and EHSW (Tukey’s post-hoc, p = 0.0001). No differences in fouling were observed between the other waveforms (p > 0.05).
Fouling after long exposure to 5-hydroxyindoleacetic acid
In mammals, the majority of serotonin in cerebral spinal fluid and blood quickly metabolizes to 5-hydroxyindoleacetaldehyde by monoamine oxidase-A and further oxidizes to 5-hydroxyindoleacetic acid (5-HIAA), which is present in mammalian tissue at concentrations 1000-fold greater than serotonin.21 5-HIAA fouls CFMEs through a similar oxidation scheme to serotonin and produces a radical intermediate that dimerizes and electropolymerizes to the CFME (Fig. S4). To determine electrode fouling effects with long 5-HIAA exposure, the current response to a 1 μM 5-HIAA injection was recorded, the CFME was soaked in 5-HIAA for 1 hour with a waveform applied, and then the current response to 5-HIAA was analyzed again. Control experiments were similar, but the electrode was soaked in PBS for an hour with a waveform applied. CFMEs were additionally soaked in 1 μM serotonin for 1 hour to compare fouling behaviors to highly concentrated serotonin for an extended period of time (Fig. S5).
Figure 4A-D shows cyclic voltammograms for the initial and final injections of 5-HIAA for each waveform. In Fig. 4A, electrodes using the Jackson waveform show dramatically reduced currents after the waveform is applied for 1 hour in 5-HIAA. In comparison, using the dopamine waveform, currents are higher after 1 hour of soaking in 5-HIAA (Fig. 4B). For electrodes using the ESW (Fig. 4C) and EHSW (Fig. 4D), currents decreased around a third. Fig. 4E shows a comparison of currents 1 hour after soaking in PBS (control) or 5-HIAA with the different waveforms applied. There were significant effects of waveform (Two-Way ANOVA, F(3,28) = 38.16, p = 0.0001, n = 6 for 5-HIAA, n = 3 for PBS) and soaking in either 5-HIAA or PBS (F(1,28) = 26.47, p = 0.0001) on current response with significant interaction between the groups (F(3,28) = 14.79, p = 0.0001). Electrodes using the Jackson waveform fouled the most in 5-HIAA with a 65 ± 4% decrease, and current responses were significantly different compared to the control (Fig. 4E, Tukey’s post-hoc, p = 0.0001). The ESW and EHSW had similar electrode fouling, with 34 ± 3% and 28 ± 4% current decrease, respectively. However, due to differences in variance, only EHSW current responses are significantly different in 5-HIAA and PBS (p = 0.01). No electrode fouling was observed with the dopamine waveform, and the final currents were higher than the initial currents (145 ± 11%, p > 0.05), which indicates that the CFME is activated by the waveform. Long exposure to 1 μM serotonin showed similar trends to 5-HIAA even though electrodes fouled severely (Fig. S5). CFMEs using the Jackson waveform fouled the most (85 ± 1%) compared with the ESW (65± 4%) and EHSW (63± 3%), while electrodes showed remarkably less fouling with the dopamine waveform (22 ± 1%). This serotonin concentration is larger than estimated basal levels (60 nM)33 so fouling in vivo may not be as severe.
Figure 4.
Electrode fouling after long exposure to 5-HIAA. A 1 μM 5-HIAA injection was recorded and the CFME was soaked in 1 μM 5-HIAA for 1 hour with the waveform continuously applied. A final 5-HIAA injection was performed to determine electrode fouling. The control was soaking in PBS for 1 hour between 5-HIAA injections. A. Example data using the Jackson waveform shows substantial fouling. B. No fouling was observed with the dopamine waveform in 5-HIAA, and final current values were higher than initial currents. Electrodes using the C. ESW waveform and D. EHSW fouled moderately. E. Bar graph compares responses for electrodes soaked in 5-HIAA and PBS. There were significant overall effects of waveform (Two-Way ANOVA, F(3,28) = 38.16, p = 0.0001) and soaking in 5-HIAA (n = 6) or PBS (n = 3) (F(1,28) = 26.47, p = 0.0001). Electrode fouling was significantly different for the Jackson waveform (F(7,28) = 36.82, Tukey’s post-hoc, p = 0.0001) and EHSW (p = 0.01), but not the dopamine waveform (p = 0.1) or ESW (p = 0.055).
For both serotonin and 5-HIAA fouling experiments, extending the switching potential to 1.3 V decreased electrode fouling by removing electropolymerized films.29 In Jackson et al., they proposed using a high scan rate of 1000 V/s to “outrun” serotonin fouling film formation.20 We observed less electrode fouling using the ESW at 1000 V/s than 400 V/s, but the sawhorse waveform shows no differences with scan rate (Fig. S3). The extended hold at 1.3 V allows more time for the carbon surface to regenerate, so fouling was similar regardless of scan rate.29 Although electrode fouling was still observed with the new serotonin waveforms, fouling was half that observed using the traditional Jackson waveform for both analytes.
Using the dopamine waveform, electrodes did not foul with repeated serotonin injections or long exposure to 5-HIAA. The holding potential at −0.4 V is applied for over 90% of a waveform cycle and helps attract the positively charged amine group to the electrode.11,27 However, during serotonin oxidation, the highly reactive radical is delocalized over the indole ring structure.22,23 This delocalization of the radical gives it a partial negative charge, which could reduce adsorption with a negative holding potential. Likely, serotonin dimers are still produced, but if they polymerize onto the carbon fiber, the higher switching potential regenerates the surface by continuously breaking carbon-carbon bonds.29 Similarly, 5-HIAA and its radical possess a negatively charged carboxyl group at physiological pH, which is also repelled by the negative holding potential. In Figure 4E, the dopamine waveform produced higher current responses after 1 hour because the extended switching potential increases surface oxide groups, increasing the current.29 Conversely, the waveforms that use a positive holding potential at 0.2 V attract serotonin radicals and 5-HIAA onto the fiber and produce worse electrode fouling (Fig. 3E and Fig. 4E). Although extending the switching potential reduces electrode fouling, a negative holding potential is critical to eliminate it.
Waveform sensitivity and selectivity determination
After understanding electrode fouling behaviors, we investigated CFME responses with serotonin and dopamine for each waveform to determine sensitivity and selectivity. Figure 5 shows example cyclic voltammograms for 100 nM serotonin and dopamine with each waveform and Table 2 gives the average results. Each electrode was used to investigate current responses for both analytes, so responses could be compared (n = 6). Electrodes using all waveforms detected 100 nM serotonin; however, 100 nM dopamine was only detected with electrodes using the dopamine waveform (Fig. 5B) and EHSW (5D) and not with the Jackson waveform (5A) or ESW (5C). Current responses for serotonin were highest using the EHSW, followed by the ESW, dopamine waveform, and Jackson waveform.
Figure 5.
Example current responses for 100 nM serotonin and dopamine (Table 2 shows averaged results from 6 electrodes). A. When using the Jackson waveform, the electrode detected 100 nM serotonin, but not 100 nM dopamine. B. Both dopamine and serotonin were detected at 100 nM with the dopamine waveform, and the cyclic voltammograms has higher currents for serotonin. C. Using the ESW, the CFME detected 100 nM serotonin, but not 100 nM dopamine. D. With the EHSW, the electrode detected both 100 nM serotonin and dopamine, and the currents for both analytes were the highest compared to the other waveforms.
Table 2.
Serotonin and Dopamine limit of detection and selectivity ratio
| Waveform | Average LOD 5-HT (nM) | Average LOD DA (nM) | Ratio [DA] [5-HT] |
|---|---|---|---|
| Jackson | 2.4 ± 1.0 | 2000 ± 600 | 833 |
| Dopamine | 1.5 ± 0.3 | 9.4 ± 0.2 | 6.3 |
| ESW | 0.8 ± 0.2 | 189 ± 5 | 236 |
| EHSW | 0.6 ± 0.2 | 1.4 ± 0.2 | 2.3 |
n=6 electrodes each
Table 2 shows the limit of detection (LOD) for serotonin (5-HT) and dopamine (DA). The limit of detection (LOD) for both analytes was calculated from the lowest concentrations detected, and the LOD for serotonin was lower than dopamine for all waveforms. CFMEs using the Jackson waveform produced the highest LOD for serotonin at 2.4 ± 1.0 nM (n = 6), while the LOD was lowest for the EHSW (0.6 ± 0.2 nM). Electrodes using the ESW and EHSW were the most sensitive for serotonin, with LODs in the hundreds of picomolar range. Interestingly, with the traditional “dopamine” waveform, CFMEs were 6-times more sensitive for serotonin than dopamine (Fig. 5B). With the Jackson waveform, electrodes showed the greatest selectivity for serotonin, with an 800-fold higher LOD, while electrodes using ESW also had a 200-fold higher LOD for serotonin. With the dopamine waveform and EHSW, electrodes were not highly selective and produced much lower ratios (closer to 1) implying more equal sensitivity to both analytes. The LOD for dopamine with the Jackson waveform is higher than physiological concentrations typically measured in vivo, so electrodes should not detect it during experiments.11
Extending the switching potential increases CFME sensitivity for both serotonin and dopamine by increasing adsorption through increased oxide groups.27,29 However, this decreases chemical selectivity. Electrodes using the Jackson waveform showed the greatest selectivity, followed by the ESW. In Jackson et al., the −0.1 V potential was designed to allow part of the reduction peak to be observed in the cyclic voltammogram.20 This limited potential sweep also favors serotonin detection because it reduces around 0.0 V, while dopamine reduces at −0.2 V. When the potential is swept only to −0.1 V, the oxidized dopamine-o-quinone product is not recycled back to dopamine in order to be detected again on the next scan.
Interestingly, with the EHSW, electrodes did not show enhanced selectivity because of their prolonged exposure to the extended switching potential. Electrodes using this sawhorse waveform were the most sensitive to both serotonin and dopamine, however applying 1.3 V for 1 ms or longer compromises selectivity for sensitivity by increasing adsorption.27,29 Keithley et al. examined sawhorse waveforms for dopamine detection and held at the switching potential for 0.55 ms.26 Their LOD of 0.9 nM for dopamine is similar to our EHSW LOD for dopamine at 1.4 ± 0.2 nM and LOD for serotonin at 0.6 ± 0.2 nM.
Characterization of optogenetically-stimulated serotonin release using various waveforms in Drosophila ventral nerve cords
Previous work in the Venton lab has shown real-time serotonin and dopamine FSCV detection in larvae and adult Drosophila melanogaster (fruit flies).17–19,32 Here, each waveform was investigated for biological applications by detecting serotonin release in isolated fruit fly larvae ventral nerve cords (VNCs). Stimulations were performed with optogenetics by inserting a geneticallyencoded, light-sensitive cation channel (CsChrimson)34 in cells expressing tryptophan hydroxylase (tph).32 Tryptophan hydroxylase is the rate-determining enzymatic step that converts tryptophan to serotonin. CsChrimson is a form of Channelrhodopsin that responds to red light, causing exocytosis when activated. A short flash of red light onto the larval VNC causes release of only serotonin without interference from dopamine.35
Figure 6A shows a confocal image of serotonin neurons in a 5-day old, third-instar larva. Neurons are visualized with GFP expression with a tph-Gal4/CyO; UAS-mCD8-GFP cross. Serotonin cell bodies are located on either side of the midline, although projections fill the neuropil. The CFME was inserted to the side of the midline for optimal serotonin detection and the waveform applied continuously during the experiment (Fig. 6B). The fiber optic cable was positioned above the VNC to deliver red light (617 nm) stimulations, which were 2 seconds long and delivered every 5 minutes to allow the releasable pool of serotonin to replenish itself.17
Figure 6.
Serotonin neuron imaging and CFME placement in Drosophila larvae ventral nerve cords (VNC). A. A tph-Gal4/CyO; UAS-mCD8-GFP cross shows GFP expression of serotonin neuron clusters in the larva VNC. B. Image shows optimal CFME placement in the neuropil of the VNC to detect the highest concentrations of serotonin.
Figure 7A-D shows example false color plots, i vs t plots, and serotonin cyclic voltammograms (inset) for the initial (colored) and final (6th, black) optogenetic stimulations for each waveform. The false color plots and cyclic voltammograms for each waveform are similar to their corresponding examples in Figure 2A-D. The i vs t plots show stable electrode current responses when stimulations are repeated every 5 minutes, regardless of the waveform used. There was no significant effect of waveform on current stability for repeated injections in Drosophila (Fig. S6, One-Way ANOVA, F(3,20) = 1.747, p = 0.1897, n = 4). Ambient levels of serotonin did not foul electrodes and stable serotonin detection was achieved because the 5 minute wait period between stimulations allows the releasable pool to replenish itself.17 Monoamine oxidase-A, which catalyzes the breakdown of serotonin to 5-HIAA, has not been identified in Drosophila.36 Instead, serotonin undergoes sugar-conjugated acetylation reactions and is recycled into the larva’s body and chitin in the adult’s exoskeleton. Thus, fouling during in vitro detection may not be as much of a prevalent problem in this model organism compared to mammals.21 Future experiments should compare the waveforms in more complex in vivo mammalian models to determine stability where 5-HIAA fouling is more prevalent.21 Although detection was stable, a comparison of pre and post calibrated electrodes show CFMEs foul 20–50% due to protein adsorption onto the CFME from inserting it into the VNC tissue (Fig. S2).37
Figure 7.
Optogenetic stimulation of serotonin in Drosophila with different waveforms. Repeated stimulations were performed by shining a red light on the ventral nerve cord for 2 seconds every 5 minute. False color plots show serotonin release on the first stimulation. Current responses were compared for the first and final (6th) stimulations. Electrodes using the (A) Jackson waveform, (B) dopamine waveform, (C) ESW, and (D) EHSW all produced stable measurements (n = 4).
Comparison of serotonin waveforms and future applications
Our results show electrodes foul severely when the Jackson waveform is used to detect repeated serotonin measurements or when 5-HIAA is present. However, the Jackson waveform is highly selective for serotonin and does not detect physiological concentrations of dopamine.11 The Jackson waveform is best suited for complex in vivo experiments where serotonin is detected with the possible interference of dopamine; for example, in regions like the striatum where interference needs to be avoided.1 With the Jackson waveform, Nafion-coated electrodes are required to mitigate serotonin and 5-HIAA fouling.21 Nafion is a cation-exchange polymer that shields serotonin and 5-HIAA from electropolymerizing to the surface of the fiber. Although it reduces electrode fouling, thick Nafion layers decrease response times in vivo, so caution is necessary when determining kinetic information during these experiments.25
Our work also shows that changing the applied FSCV waveform reduces electrode fouling without applied polymer coatings. With the extended serotonin waveform (ESW), electrodes had comparable selectivity to the Jackson waveform with reduced fouling and higher sensitivity. The ESW could be applied in vivo without Nafion if selectivity and faster responses are required. Nafion coatings may not be stable with a 1.3 V switching potential because of surface regeneration.29 With the extended hold serotonin waveform (EHSW), electrode responses were not as selective, but were the most sensitive for both serotonin and dopamine. The EHSW would be beneficial for optogenetic experiments where either serotonin or dopamine was specifically stimulated, especially if high sensitivity was necessary.
A major finding in this work is that there is no electrode fouling for serotonin with the dopamine waveform. Further, with the dopamine waveform, electrodes were more sensitive for serotonin than dopamine, but they were not highly selective. The dopamine waveform’s anti-fouling nature is due to its extended switching potential, which renews the carbon electrode surface, and negative holding potential, which reduces adsorption of serotonin and its oxidation products. In Moran et al., a unique waveform (−0.6 V to 1.4 V and back, 400 V/s) was used to detect both serotonin and dopamine in a single cyclic voltammogram, with machine learning to distinguish the compounds.1,38 Serotonin and dopamine are usually identified by their different reduction peaks (0.0 V and −0.2 V, respectively), but these can shift in vivo from tissue fouling and protein adsorption.37,39 While the sensitivity of CFMEs with the dopamine waveform has been investigated previously,27,29 it should be recognized that the CFME is more sensitive to serotonin with this waveform and that small concentrations of serotonin will easily interfere with dopamine measurements. Ultimately, using the dopamine waveform is beneficial because it produces high sensitivity and causes less electrode fouling, and it is useful in experiments where selectivity is not a problem. For example, the dopamine waveform is useful for detecting serotonin in optogenetic experiments, where the channel is genetically targeted to one cell type, so selectivity is not an issue.17,32
Overall, this work shows many waveforms can be applied to CFMEs to detect serotonin. Electrodes using the Jackson waveform are the most selective for serotonin, but electrodes using the ESW show higher sensitivity than the Jackson waveform while maintaining high selectivity. With the EHSW and dopamine waveform, electrodes also have excellent sensitivity. All extended waveforms show less (ESW, EHSW) or no (DA) electrode fouling. Each waveform can be applied to Drosophila for stable serotonin detection; however, future in vivo applications of each waveform should be based on the properties desired for an experiment.
Conclusions
Overall, this work evaluated different FSCV waveforms for serotonin detection and detailed different advantages and disadvantages for each waveform. Detection using the Jackson waveform is the most selective for serotonin but fouling is the most problematic. The ESW shows higher electrode sensitivity while maintaining high selectivity. With the EHSW and dopamine waveform, electrodes have excellent sensitivity. All extended waveforms show reduced electrode fouling compared to the Jackson waveform, and the dopamine waveform shows no electrode fouling with serotonin or 5-HIAA. Each waveform can be applied to CFMEs for stable serotonin detection in Drosophila; however, future in vivo applications should be based on experimental designs. For example, the dopamine waveform can be used in experiments where fouling is an issue if the analyte being detected is known to be serotonin, so selectivity is not a concern. The ESW is a better choice for experiments requiring selectivity between dopamine and serotonin, and will limit fouling. All waveforms should be further investigated in mammalian models, but this work developed a toolkit of serotonin waveforms that can be tuned to the requirements of an individual experiment, and will facilitate a better understanding of the role of serotonin in illnesses such as depression.
Supplementary Material
Acknowledgments
We thank Dr. Mimi Shin, Dr. Eduard Dumitrescu, and Dr. Pumidech (Max) Puthongkham (University of Virginia, Chemistry Department) for their assistance with instrumentation, techniques, and Drosophila maintenance. We also thank Dr. Jeffery Copeland (Eastern Mennonite University, Biology Department) for his guidance on Drosophila genetic manipulations. This work was funded by NIH R01MH085159.
Footnotes
Conflicts of Interest
There are no conflicts to declare.
Supporting Information
Supplementary information is included. Supplementary Figure 1: Fast-scan cyclic voltammetry (FSCV) background (BG) charging current subtraction, Supplementary Figure 2: Precalibration and post calibration responses of electrode after Drosophila tissue implantation, Supplementary Figure 3: Extended serotonin waveform scan rate comparison, Supplementary Figure 4: Serotonin and 5-hydroxyindoleacetic acid oxidation scheme, Supplementary Figure 5: Fouling after long exposure to serotonin, Supplementary Figure 6: Optogenetic stimulation stability with different waveforms.
References
- (1).Moran RJ; Kishida KT; Lohrenz T; Saez I; Laxton AW; Witcher MR; Tatter SB; Ellis TL; Phillips PE; Dayan P; Montague PR The Protective Action Encoding of Serotonin Transients in the Human Brain. Neuropsychopharmacology 2018, 43 (6), 1425–1435. 10.1038/npp.2017.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2).Shin M; Wang Y; Borgus JR; Venton BJ Electrochemistry at the Synapse. Annu. Rev. Anal. Chem. 2019, 12 (1), 297–321. 10.1146/annurev-anchem-061318-115434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (3).Fakhoury M Revisiting the Serotonin Hypothesis: Implications for Major Depressive Disorders. Mol. Neurobiol 2016, 53 (5), 2778–2786. 10.1007/s12035-015-9152-z. [DOI] [PubMed] [Google Scholar]
- (4).Wood KM; Zeqja A; Nijhout HF; Reed MC; Best J; Hashemi P Voltammetric and Mathematical Evidence for Dual Transport Mediation of Serotonin Clearance in Vivo. J. Neurochem 2014, 130 (3), 351–359. 10.1111/jnc.12733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Saylor RA; Hersey M; West A; Buchanan AM; Berger SN; Nijhout HF; Reed MC; Best J; Hashemi P In Vivo Hippocampal Serotonin Dynamics in Male and Female Mice: Determining Effects of Acute Escitalopram Using Fast Scan Cyclic Voltammetry. Front. Neurosci 2019, 13, 362 10.3389/fnins.2019.00362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Venton BJ; Wightman RM Psychoanalytical Electrochemistry: Dopamine and Behavior. Anal. Chem 2003, 75 (19), 414 A–421 A. [Google Scholar]
- (7).Ganesana M; Lee ST; Wang Y; Venton BJ Analytical Techniques in Neuroscience: Recent Advances in Imaging, Separation, and Electrochemical Methods. Anal. Chem 2017, 89 (1), 314–341. 10.1021/acs.analchem.6b04278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (8).Puthongkham P; Venton BJ Recent Advances in Fast-Scan Cyclic Voltammetry. Analyst 2020, 145, 1087–1102. 10.1039/C9AN01925A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Lama RD; Charlson K; Anantharam A; Hashemi P Ultrafast Detection and Quantification of Brain Signaling Molecules with Carbon Fiber Microelectrodes. Anal. Chem 2012, 84 (19), 8096–8101. 10.1021/ac301670h. [DOI] [PubMed] [Google Scholar]
- (10).Huffman ML; Venton BJ Carbon-Fiber Microelectrodes for in Vivo Applications. Analyst 2009, 134 (1), 18–24. 10.1039/b807563h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Venton BJ; Cao Q Fundamentals of Fast-Scan Cyclic Voltammetry for Dopamine Detection. Analyst 2020, 145, 1158–1168. 10.1039/C9AN01586H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (12).Andrews AM Why Monitor Molecules in Neuroscience? ACS Chem. Neurosci 2017, 8 (2), 211–212. 10.1021/acschemneuro.7b00052. [DOI] [PubMed] [Google Scholar]
- (13).Hashemi P; Dankoski EC; Wood KM; Ambrose RE; Wightman RM In Vivo Electrochemical Evidence for Simultaneous 5-HT and Histamine Release in the Rat Substantia Nigra Pars Reticulata Following Medial Forebrain Bundle Stimulation. J. Neurochem 2011, 118 (5), 749–759. 10.1111/j.1471-4159.2011.07352.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).Dankoski EC; Carroll S; Wightman RM Acute Selective Serotonin Reuptake Inhibitors Regulate the Dorsal Raphe Nucleus Causing Amplification of Terminal Serotonin Release. J. Neurochem 2016, 136 (6), 1131–1141. 10.1111/jnc.13528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (15).Dankoski EC; Agster KL; Fox ME; Moy SS; Wightman RM Facilitation of Serotonin Signaling by SSRIs Is Attenuated by Social Isolation. Neuropsychopharmacology 2014, 39 (13), 2928–2937. 10.1038/npp.2014.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (16).Wood KM; Hashemi P Fast-Scan Cyclic Voltammetry Analysis of Dynamic Serotonin Reponses to Acute Escitalopram. ACS Chem. Neurosci 2013, 4 (5), 715–720. 10.1021/cn4000378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (17).Borue X; Condron B; Venton BJ Both Synthesis and Reuptake Are Critical for Replenishing the Releasable Serotonin Pool in Drosophila. J. Neurochem 2010, 113 (1), 188–199. 10.1111/j.1471-4159.2010.06588.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (18).Borue X; Cooper S; Hirsh J; Condron B; Venton BJ Quantitative Evaluation of Serotonin Release and Clearance in Drosophila. J. Neurosci. Methods 2009, 179 (2), 300–308. 10.1016/j.jneumeth.2009.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Xiao N; Privman E; Venton BJ Optogenetic Control of Serotonin and Dopamine Release in Drosophila Larvae. ACS Chem. Neurosci 2014, 5 (8), 666–673. 10.1021/cn500044b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (20).Jackson BP; Dietz SM; Wightman RM Fast-Scan Cyclic Voltammetry of 5-Hydroxytryptamine. Anal. Chem 1995, 67 (6), 1115–1120. 10.1021/ac00102a015. [DOI] [PubMed] [Google Scholar]
- (21).Hashemi P; Dankoski EC; Petrovic J; Keithley RB; Wightman RM Voltammetric Detection of 5-Hydroxytryptamine Release in the Rat Brain. Anal. Chem 2009, 81 (22), 9462–9471. 10.1021/ac9018846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (22).Wrona MZ; Dryhurst G Electrochemical Oxidation of 5-Hydroxytryptamine in Aqueous Solution at Physiological PH. Bioorg. Chem 1990, 18 (3), 291–317. 10.1016/0045-2068(90)90005-P. [DOI] [Google Scholar]
- (23).Anne A; Lemordant D; Dryhurst G; Wrona MZ Mechanism and Products of Electrochemical Oxidation of 5, 7-Dihydroxytryptamine. J. Am. Chem. Soc 1989, 111 (2), 719–726. 10.1021/ja00184a050. [DOI] [Google Scholar]
- (24).Puthongkham P; Lee ST; Venton BJ Mechanism of Histamine Oxidation and Electropolymerization at Carbon Electrodes. Anal. Chem 2019, 91 (13), 8366–8373. 10.1021/acs.analchem.9b01178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (25).Ross AE; Venton BJ Nafion-CNT Coated Carbon-Fiber Microelectrodes for Enhanced Detection of Adenosine. Analyst 2012, 137 (13), 3045–3051. 10.1039/c2an35297d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (26).Keithley RB; Takmakov P; Bucher ES; Belle AM; Owesson-White CA; Park J; Wightman RM Higher Sensitivity Dopamine Measurements with Faster-Scan Cyclic Voltammetry. Anal. Chem 2011, 83 (9), 3563–3571. 10.1021/ac200143v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Heien MLAV; Phillips PEM; Stuber GD; Seipel AT; Wightman RM Overoxidation of Carbon-Fiber Microelectrodes Enhances Dopamine Adsorption and Increases Sensitivity. Analyst 2003, 128, 1413–1419. 10.1039/b307024g. [DOI] [PubMed] [Google Scholar]
- (28).Robinson DL; Venton BJ; Heien MLA V; Wightman, R. M. Detecting Subsecond Dopamine Release with Fast-Scan Cyclic Voltammetry in Vivo. Clin. Chem 2003, 49 (10), 1763–1773. https://doi.org/10.137¾9.10.1763. [DOI] [PubMed] [Google Scholar]
- (29).Takmakov P; Zachek MK; Keithley RB; Walsh PL; Donley C; McCarty GS; Wightman RM Carbon Microelectrodes with a Renewable Surface. Anal. Chem 2010, 82 (5), 2020–2028. 10.1021/ac902753x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (30).McCreery RL Advanced Carbon Electrode Materials for Molecular Electrochemistry. Chem. Rev 2008, 108 (7), 2646–2687. 10.1021/cr068076m. [DOI] [PubMed] [Google Scholar]
- (31).Ross AE; Venton BJ Sawhorse Waveform Voltammetry for Selective Detection of Adenosine, ATP, and Hydrogen Peroxide. Anal. Chem 2014, 86 (15), 7486–7493. 10.1021/ac501229c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (32).Privman E; Venton BJ Comparison of Dopamine Kinetics in the Larval Drosophila Ventral Nerve Cord and Protocerebrum with Improved Optogenetic Stimulation. J. Neurochem 2015, 135 (4), 695–704. 10.1111/jnc.13286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (33).Abdalla A; Atcherley CW; Pathirathna P; Samaranayake S; Qiang B; Peña EA; Morgan SL; Heien ML; Hashemi P In Vivo Ambient Serotonin Measurements at Carbon-Fiber Microelectrodes. Anal. Chem 2017, 89 (18), 9703–9711. 10.1021/acs.analchem.7b01257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (34).Klapoetke NC; Murata Y; Kim SS; Pulver SR; Birdsey-Benson A; Cho YK; Morimoto TK; Chuong AS; Carpenter EJ; Tian Z; Wang J; Xie Y; Yan Z; Zhang Y; Chow BY; Surek B; Melkonian M; Jayaraman V; Constantine-Paton M; Wong GKS; Boyden ES Independent Optical Excitation of Distinct Neural Populations. Nat. Methods 2014, 11 (3), 338–346. 10.1038/nmeth.2836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (35).Shin M; Copeland JM; Venton BJ Drosophila as a Model System for Neurotransmitter Measurements. ACS Chem. Neurosci 2018, 9 (8), 1872–1883. 10.1021/acschemneuro.7b00456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Dewhurst SA; Croker SG; Ikeda K; Mccaman RE Metabolism of Biogenic Amines in Nervous Tissue in Drosophila. Comp. Biochem. Physiol 1972, 43 (1966), 975–981. [DOI] [PubMed] [Google Scholar]
- (37).Norde W Driving Forces for Protein Adsorption at Solid Surfaces. Macromol. Symp 1996, 103, 5–18. 10.1002/masy.19961030104. [DOI] [Google Scholar]
- (38).Kishida KT; Saez I; Lohrenz T; Witcher MR; Laxton AW; Tatter SB; White JP; Ellis TL; Phillips PEM; Montague PR Subsecond Dopamine Fluctuations in Human Striatum Encode Superposed Error Signals about Actual and Counterfactual Reward. Proc. Natl. Acad. Sci 2016, 113 (1), 200–205. 10.1073/pnas.1513619112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (39).Seaton BT; Hill DF; Cowen SL; Heien ML Mitigating the Effects of Electrode Biofouling-Induced Impedance for Improved Long-Term Electrochemical Measurements In Vivo. Anal. Chem 2020, No. 2 10.1021/acs.analchem.9b05194. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.