Abstract
Three-dimensional (3-D) neural cultures represent a promising platform for studying disease and drug screening. Tools and methodologies for measuring the electrophysiological function in these cultures are needed. Therefore, the purpose of this work was primarily to develop a methodology to interface engineered 3-D dissociated neural cultures with commercially available 3-D multi-electrode arrays (MEAs) reliably over 3 weeks to enable the recording of their electrophysiological activity. We further compared the functional output of these cultures to their structural and synaptic network development over time. We reliably interfaced a primary rodent neuron-astrocyte (2:1) 3-D co-culture (2500 cells/mm3 plating cell density) in Matrigel™ (7.5 mg/mL) that was up to 750 µm thick (30–40 cell-layers) with spiked 3-D MEAs while maintaining high viability. Using these MEAs we successfully recorded the spontaneous development of neural network-level electrophysiological activity and measured the development of putative synapses and neuronal maturation in these co-cultures using immunocytochemistry over 3 weeks in vitro. Planar (2-D) MEAs interfaced with these cultures served as recording controls. Neurons within this interfaced 3-D culture-MEA system exhibited considerable neurite outgrowth, networking, neuronal maturation, synaptogenesis, and culture-wide spontaneous firing of synchronized spikes and bursts of action potentials. Network-wide spikes and synchronized bursts increased rapidly (first detected at 2 days) during the first week in culture, plateaued during the second week, and reduced slightly in the third week, while maintaining high viability throughout the 3-week culturing period. Early electrophysiology activity occurred prior to neuronal process maturation and significant synaptic density increases in the second week. We successfully interfaced 3-D neural co-cultures with 3-D MEAs and recorded the electrophysiological activity of these cultures over 3 weeks. The initial period of rapid increase in electrophysiological activity, followed by a period of neuronal maturation and high-level of synapse formation in these cultures suggests a developmental homeostatic process. This methodology can enable future applications both in fundamental investigations of neural network behavior and in translational studies involving drug testing and neural interfacing.
Keywords: Three-dimensional, Electrophysiology, Multi-electrode array, MEA, In vitro, Neuron, Astrocyte, Synapse, Matrigel, Culture
Introduction
Novel three-dimensional (3-D) neural culture models represent a promising platform for studying disease and drug screening. Traditionally, neural networks have been studied either in two-dimensional (2-D) dissociated or brain slice culture models in vitro (e.g., [1–4]). These culture models come with their own advantages and disadvantages. For example, 2-D cultures are the simplest to cultivate. However, they may not represent the cell–cell configurations, 3-D architecture, and network-level functions that are typical in vivo. Brain slice culturing models; on the other hand, preserve some of the native neural network connectivity, 3-D architecture, and function. However, these are prone to poor viability, tissue break-down, and settling because of mass transport limitations [5]. 3-D neural culture models provide an alternative to these existing culture model; for example, assembling dissociated cells in 3-D may permit the creation of tissue-mimetics with more control over the cell-level parameters, such as cell type(s), ratio, and density than brain slice preparations, and more in vivo-like tissue organization than 2-D cultures [6–10]. To this end, we previously established a 3-D co-culture model by mixing dissociated rat embryonic cortical neurons and neonatal cortical astrocytes homogeneously within the bioactive matrix Matrigel™ and cultured under defined serum-free media conditions [11].
There has been increasing interest in the last decade to develop in vitro 3-D neural cell culture models [11–13] and correspondingly increasing reports of measurement of electrophysiological activity of dissociated 3-D neural cultures are being published using different culture configurations and recording techniques [14–21]. Previously, performing patch clamp recordings, our group showed that dissociated cortical neurons cultured in the 3-D neural culture model that we developed within Matrigel™ are electrophysiologically active [14]. Similar measurements revealed that electrophysiological activity characteristics of hippocampal neurons cultured in 3-D collagen hydrogels were comparable to neurons cultured on top of 2-D collagen coated surfaces [22]. Individual neuron-level electrophysiological recording; however, are not able to assess network-level activity and culture development over time. Differences between 2-D and 3-D neural network-level activity has been demonstrated using planar MEAs [16, 19, 23], but the functional development of 3-D cultures over time has not been investigated using dissociated cortical neuronal-astrocyte co-cultures.
In this work, we integrated 3-D neuronal-astrocytic co-cultures with either spiked (i.e. 3-D) or planar (i.e. 2-D) multi-electrode arrays (MEA) to characterize network activity. The purpose of this investigation was not to compare 3-D cultures with 2-D cultures, per se, but to specifically develop a methodology to interface a novel engineered 3-D dissociated neural culture model with commercially available 3-D multi-electrode arrays (MEAs) reliably over 3 weeks to enable the recording of their electrophysiological function. Therefore, 2-D cultures were not investigated in this study. The traditional planar (2-D) MEAs were used as a recording control for the relatively newer spiked (3-D) MEAs in this study, which focused specifically on recording the electrophysiological function of the 3-D co-cultures. We compared the rates of MEA-acquired extracellular field-potential measurements of action potential spikes and bursts to markers of synaptic distribution and neuronal maturation in the neuronal-astrocytic 3-D co-cultures with respect to days in vitro. Additionally, to test the bioactive responsiveness of these cultures, electrophysiological activity was also recorded before, during, and after exposing these 3-D co-cultures to the following chemical perturbations: bicuculline methiodide (BMI, a GABAA-receptor antagonist) to block inhibitory synaptic transmission [24], and gramicidin (a plasmalemma transient-pore forming antibiotic) to disrupt electrophysiological function [25] in the presence of cell-impermeant calcein (to verify pore formation).
Materials and methods
Multi-electrode arrays (MEAs) and cell culture chambers
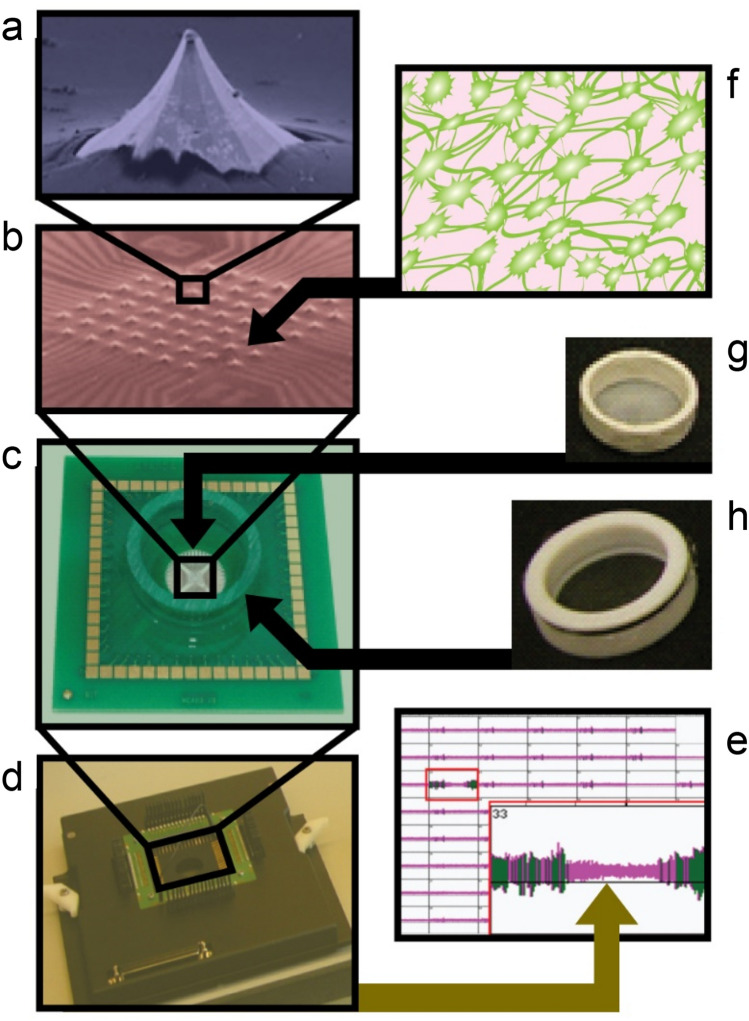
Both spiked (3-D) MEAs (Ayanda Biosystems, Lausanne, Switzerland) and optical planar (2-D) MEAs (Multi Channel Systems, Reutlingen, Germany) were used. The planar MEAs were used as controls for the spiked (3-D) MEAs. Furthermore, their “optical” capability, enabled by a thin 180 μm glass bottom and optically transparent indium tin oxide conductive leads, enabled the continual imaging of up to several hundred microns into the 3-D co-culture while being attached to the planar (2-D) MEAs, without disturbing culturing conditions. In contrast, the spiked (3-D) MEAs, with a 700 μm glass bottom thickness, presented platinum-tipped, micro-electrode spikes aiding in extracellular field potential recording up to 50–70 µm into the 3-D co-culture thickness (Fig. 1a, b). Imaging of 3-D co-cultures apposed to the spiked MEAs was done by inverting the MEAs over the objective of an inverted confocal microscope, with a semi-permeable Teflon membrane acting as a barrier. This configuration allowed closer access of microscope objective to the 3-D cell culture, enabling imaging of several hundred microns into the thickness of the cultures, while maintaining sterility and conditions necessary for long-term culturing. Both planar and spiked MEAs presented a 8 × 8 matrix of electrodes (excluding corner electrodes) spaced 200 μm from each other, with a characteristic recording base dimension of 30 µm (planar MEA electrode) and 40 µm (spiked MEA electrode), respectively (Fig. 1b, c). Figure 2c, d illustrate the experimental set-up of the 3-D co-cultures interfaced with the spiked (3-D) MEAs and planar (2-D) MEAs, respectively.
Fig. 1.
The spiked (3-D) MEA cell culture interface for electrophysiological recording. Components of the in vitro recording system are shown: a An enlarged view of a single spike electrode (50–70 µm in height, 40 × 40 µm based dimension), b an array of these electrodes (59 electrodes with 200 µm inter-electrode spacing), c the MEA culture dish, d a Multi Channel Systems preamplifier, e a multichannel recording output snapshot showings spikes and bursts in dark vertical lines that cross a horizontal threshold line, f a schematic of the 3-D neural culture, g nylon-mesh insert for culture immobilization, h and FEP-membrane cap for reducing evaporative losses and the possibility of infection. (a–c courtesy: Ayanda Biosystems, Lausanne, Switzerland; Heuschkel et al. [31])
Fig. 2.
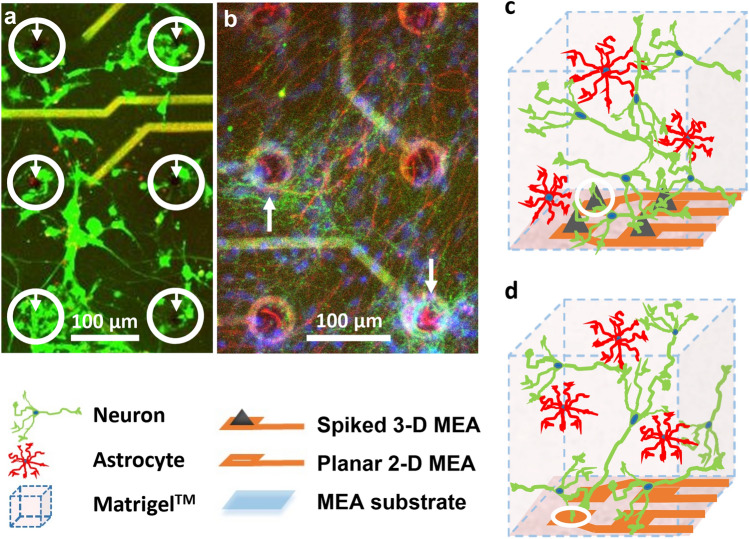
Cells and electrodes are closely apposed at the 3-D culture electrode array interface. a Live cells (fluorescing green by AM-cleavage) and dead cells with compromised membranes (nuclei fluorescing red by EthD-1 binding to DNA) are seen in a confocal z-stack image projection of a 105 μm thick region of a 3-D co-culture on a spiked MEA at 24 days in vitro. The co-cultures maintained significant viability for more than 3 weeks in culture, with cells showing in vivo-like somatic shapes and networking via process outgrowth as well as cluster formation. Cells within the white circles are seen apposed around the 3-D electrode spikes demarked by white arrows. b Neurons, astrocytes, and cellular nuclei are seen immuno-stained green (Tau 5), red (GFAP), and blue (Hoechst), respectively, showing random distribution of the two cell types and the apposition of approximately 5–10 cells per electrode in a confocal z-stack image projection of a 80 μm thick culture region on a MEA at 22 days in vitro as indicated by white arrows. This indicates that each electrode likely records activity from multiple cells. c Schematic of the 3-D neuron-astrocyte co-culture interfaced to the spiked (3-D) MEA. d Schematic of the 3-D co-culture interfaced with planar (2-D) MEA. The planar MEAs served as a control for the spiked MEA. In c and d, neurons are depicted in green and astrocytes in red and scale is exploded for illustration. (Color figure online)
Non-MEA custom-made cell culture chambers, which were used for 3-D cell culture viability and immunocytochemistry measurements, were prepared by pressing silicone rings (internal diameter 1.6 cm; Sylgard 184 and 186, 50:50, Dow Corning, Midland, MI) on air glow discharge treated glass cover slips (1 × 10−1 mbar pressure, 25 mA discharge current, and negative polarity for 1 min; EMS-100, Electron Microscopy Sciences, Hatfield, PA). The glow discharge was also used on corresponding MEA culturing surfaces to render them hydrophilic. The silicone rings stuck to the glass coverslips created 3-D cell culture chambers that were similar to the MEA cell culture wells, but with a thin glass bottom that enabled continual upright confocal imaging of the cultures while maintaining sterility and conditions necessary for long-term culturing.
Subsequent to sterilization, all cell culture surfaces were coated with poly-d-lysine (100 µg/ml, MW > 300,000 Da; Sigma, St. Louis, MO), rinsed with water, and then coated with laminin (20 µg/ml, Invitrogen, Carlsbad, CA). Immediately prior to plating, the laminin solution was removed and the chambers were rinsed with calcium- and magnesium-free Hanks balanced salt solution (CMF-HBSS, Invitrogen).
Cell harvest
All procedures involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) of the Georgia Institute of Technology. Sasco Sprague–Dawley rats (Charles River, Wilmington, MA) were anesthetized and euthanized at embryonic day 18 (E18). Rat fetuses were removed by Caesarean section, and the cerebral cortices were isolated in ice cold CMF-HBSS and dissociated in pre-warmed trypsin (0.25% + 1 mM EDTA, Invitrogen) at 37 °C for 10 min followed by agitation in deoxyribonuclease I (DNase I, 0.15 mg/mL, Sigma) at room temperature for 5–15 s. Dissociated cortical cells were centrifuged at 1000 rpm for 3 min, gently triturated and resuspended in a defined plating medium (Neurobasal medium + 2% B-27 + 500 µM l-glutamine; Invitrogen).
Astrocytes were obtained from postnatal day 1 Sasco Sprague–Dawley rat cortices and used between passages 4 and 10. Briefly, isolated cortices were minced, digested in trypsin at 37 °C for 3–5 min, followed by DNase I treatment and gentle mechanical trituration. Cells were plated in DMEM/F12 with 10% fetal bovine serum (Invitrogen). Mechanical agitation detached less adherent cells (primarily neurons) from flask bottoms at 24 and 72 h, followed by passaging (at 90% confluence) to give a population of > 95% type I astrocytes.
3-D cortical neural cell co-cultures
3-D co-cultures were generated for electrophysiological recording (n = 9 on spiked MEAs; n = 3 on planar MEAs), viability assessment (n = 3 each at 5, 8, 11, 16, and 24 days in vitro on non-MEA chambers) and immunocytochemical phenotypic marking (n = 4 each at 5, 8, 11, and 16 days in vitro for the markers probed on non-MEA chambers). A limited number of viability and immunocytochemical measurements were also done at terminal time-points on the MEA cultures to verify cell culture apposition to the electrode arrays. The 3-D co-cultures were plated with neurons and astrocytes at 2:1 ratio and 2500 cells/mm3 cell density uniformly distributed throughout a 7.5 mg/mL growth factor reduced Matrigel™ matrix (BD Biosciences, San Jose, CA) to produce cultures of up to 750 μm in thickness (Fig. 1b, c, f). Matrigel™ is well known to promote ECM-mediated neurite outgrowth [26, 27] and growth factor mediated processes [28]. These plating parameters are based on previously published cell culture optimization work from our laboratory [29]. After matrix gelation in a cell culture incubator for 45 min, 1 mL of plating medium (37 °C) was gently added to each culture well, followed by complete replacement by co-culture feeding medium (Neurobasal medium + 2% B-27 + 500 µM l-glutamine + 1% G-5; Invitrogen) at 24 h. Nylon mesh inserts (70–200 µm pores) were placed on top of the cultures to prevent their detachment from MEAs (Fig. 1g). Finally, all cultures were sealed with O2- and CO2-permeable fluorinated ethylene propylene (FEP) membrane caps that prevented evaporative losses and; thereby, osmolality shocks to the cultures, while also reducing the risk of infection [30] (Fig. 1h).
Cell viability
Cell viability was quantified using fluorescent probes to distinguish between live and dead cells in cultures (Viability/Cytotoxicity Kit; Molecular Probes, Eugene, OR). Cultures were incubated with 2 μM calcein AM and 4 μM ethidium homodimer-1 at 37 °C for 30 min and rinsed with 0.1 M Dulbecco’s phosphate buffered saline (D-PBS, Invitrogen) followed by confocal microscopic imaging. The percentage of viable cells per 3-D reconstructed image was determined from the number of live cells (green fluorescing via AM-cleavage) and the number of cells with compromised membranes (nuclei fluorescing red via EthD-1/DNA intercalation). In addition to viability assessment, this staining also allowed qualitative observation of neural process sprouting, extension, and networking over the 3-week culture period.
Electrophysiological recording
Electrophysiological activity was recorded daily (for 24 days) at 37 °C by interfacing the MEA cultures (in random order) with a Multi Channel Systems MEA60 preamplifier system (Fig. 1d, e). Silicone/gold-based “zebra connectors” ensured proper electrical contact of the preamplifier pins with MEA pads. Noisy channels—baseline noise greater than ± 17.5 µV for spiked MEAs, or greater than ± 20–30 µV for planar MEAs, based on manufacturer specifications—were excluded from analyses. All data recorded with an amplifier gain of 1200 and a sampling frequency of 25 kHz were thresholded through a 200 Hz high pass filter.
Spike and burst detection and analysis
A 5 min voltage fluctuation stream was analyzed from each viable MEA channel recording to quantify spikes of action potentials (singular voltage fluctuations beyond a threshold) and bursts (clusters of spikes that fulfilled a minimum criterion). MC Rack software (Multi Channel Systems) detected spikes by tagging a voltage trace whenever it exceeded a baseline-threshold of − 17.5 µV for the spiked MEA data [31], or 3 times the standard deviation of the data stream for the planar MEA data (Fig. 1e). Three times standard deviation was chosen as the spike detection threshold for the planar MEA data, because we observed that it was significantly off-set from base-line noise reducing the possibility of detecting false positive spikes for our 3-D cultures (which usually exhibit lower signal-to-noise ratios compared to their traditional 2-D monolayer counter-parts). Likewise, − 17.5 µV, which was significantly greater than the noise range for the spiked MEA, both as characterized by the company and by our own independent observations, was selected as threshold for the spiked MEA signal. This process was conservative at not filtering out actual signal. Furthermore, external electromagnetic interference (noise) was blocked out by conducting all recordings in a Faraday cage set-up in the dark.
Next, Neuroexplorer Software (Nex Technologies, MA) was used to rasterize the spikes and quantify the spikes and bursts. Bursts were identified by a previously developed burst detector algorithm [32]. According to this algorithm, the minimum criteria for a burst were a cluster of 4 spikes over a period of 20 ms with an inter-burst interval of at least 10 ms. This method was sensitive for processing our 3-D cell culture electropysiological activity. The acquired spike and burst values are a function of the detection criteria. Therefore, it is important to note that their relative trends are more important than absolute values. Average spike (spikes/second) and burst (bursts/minute) rates were calculated for each replicate MEA culture by adding the number of spikes and bursts from all the viable channels from a recording session and dividing by the number of viable channels and the recording period to give average parameter rates per electrode, respectively. Therefore, both the average spike and burst rates are multi-channel/network-level parameters, as opposed to single-channel/cell. Since, bursts of action potentials were mostly synchronized across all the recording electrodes, and most spikes were seen within these synchronized bursts, this methodology to give a measure of multi-channel/network-level bursting activity was considered appropriate. Each channel recording within an MEA was considered an N of 1 while performing statistical analysis. Statistical analysis was performed using one- and two-way ANOVA after verifying normal distribution of data and equal variances between treatment groups, followed by Tukey’s pair-wise comparison (p < 0.05 considered significant).
Additionally, to test the bioactive responsiveness of the cultures, electrophysiological activity was also recorded before, during, and after exposing a subset of 3-D co-cultures (21 days in vitro, n = 3) to the following chemical perturbations: 10–200 μM bicuculline methiodide (BMI), a GABAA-receptor antagonist to block inhibitory synaptic transmission and have an indirect excitatory effect on network activity [24]; and 0.01–1 μg/ml gramicidin, a plasmalemma transient-pore forming antibiotic that depolarizes the neurons [25], in the presence of cell-impermeant calcein (to verify pore formation). Multiple complete media changes were done as rinses after each chemical exposure.
Immunocytochemistry
Cultures were fixed in 3.7% formaldehyde in phosphate buffered saline (PBS) at room temperature for 30 min and permeabilized using 0.3% Triton X100 + 4% goat serum (Invitrogen) in PBS for 20 min. Next, the cultures were incubated with different combinations of following primary antibodies (in PBS + 4% goat serum) at 18–24 °C: (1) MAP-2ab, a microtubule-associated protein expressed predominantly in mature neuronal cell bodies and dendrites (MAB3418, 1:200; Chemicon, Temecula, CA) [33–35]; (2) Tau-5, a microtubule-associated protein localized primarily in neuronal cell bodies and axons (MS247P, 1:200; NeoMarkers, Fremont, CA) [36, 37]; (3) GFAP, a structural intermediate filament protein found in astrocytes (MAB360, 1:400; Chemicon) [38, 39]; and 4) Synapsin I, a phospho-protein localized to the cytoplasmic surface of pre-synaptic vesicles (A6442, 1:200; Invitrogen) [40, 41]. After overnight primary antibody incubation, the appropriate secondary fluorophore-conjugated antibody (Alexa Fluor 488 goat anti-rabbit, 1:500; or Alexa Flour 546 goat anti-mouse, 1:500; Molecular probes, Eugene, OR) in PBS + 4% serum was added at 18–24 °C for 2 h. Negative staining controls followed the same procedure, but without the primary antibodies. Counterstaining for nuclei was done using Hoechst 33,258 (1:1000, Molecular Probes).
Image analysis
For viability and immunocytochemical assessments, at least 3 different randomly selected regions from each culture were imaged as z-stacks using a Zeiss 510 Confocal Laser Scanning Microscope (Carl Zeiss AG, Göttingen, Germany). LSM Image Browser (Carl Zeiss AG) converted the z-stacks into 3-D projections, from which the viability and immunocytochemistry-based parameters were manually quantified. For the immunocytochemistry, the number of MAP2-positive cell somata, Synapsin I positive puncta, and Hoechst-positive nuclei were quantified for each 3-D projected confocal z-stack to calculate mature neuronal and putative synaptic densities (#/mm3) with respect to time in culture. To prevent user bias, each image was independently quantified by two operators, and only those corresponding counts that were within ± 10% of each other were considered and averaged. Statistical analysis was performed using one-way ANOVA after verifying normal distribution of data and equal variances between treatment groups followed by a post hoc test (p < 0.05 considered significant).
Results
3-D culture morphology and viability
Initially, cells that were uniformly dispersed in 3-D within the Matrigel™ at the time of plating extended processes in all directions rapidly (up to 100 µm long within 24 h post-plating) and continued to do so over the culture period. By week two, cellular processes, which had increased in number and extended up to several hundreds of microns, formed dense neural networks in 3-D. These cultures maintained high viability (97 ± 3%) (Fig. 2a), with neuronal and astrocytic cell bodies and processes randomly distributed throughout the matrix as well as apposed to the electrode arrays throughout the 3 week culturing period without detachment (Fig. 2b).
Electrophysiological activity
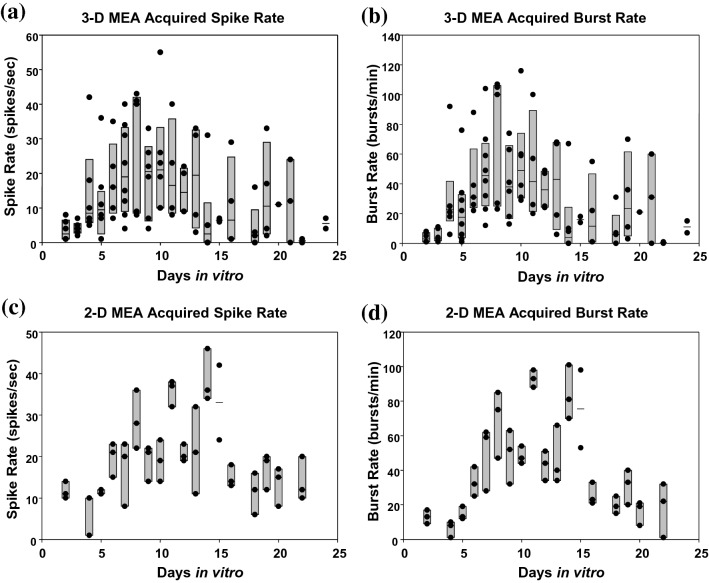
Spontaneous electrophysiological activity was successfully recorded from these 3-D co-cultures for up to 24 days in vitro using both the spiked (3-D) and planar (2-D) MEAs. These extracellular electrophysiological recordings showed both individual spikes (amplitudes in the 25 to 250 μV range) and clusters of spikes (bursts) of action potential. Random spiking, as well as the beginning of synchronized bursting activity, were both observed as early as 2 days in vitro, indicating the concurrent development of functional synapses along with network formation. Overall, during the culturing period tested, both spike and burst rates increased rapidly during the first week in culture, reached a plateau during the second week, and gradually dropped off in the third week (Fig. 3). As days in culture progressed, the spiking activity rarely occurred outside of culture-wide bursts. This observation, shared by both the planar (2-D) and spiked (3-D) MEA recorded data, indicates the synchronization across the culture of most spikes within bursts, explaining the similar spike and burst rate development profiles with respect to days in vitro. Furthermore, similar spike and burst rates were detected by both the planar (2-D) MEAs and spiked (3-D) MEAs from these 3-D neural co-cultures using our data acquisition and processing method, indicating that our methods with the two different types of MEAs were comparable.
Fig. 3.
The development of electrophysiological activity in 3-D neural co-cultures in terms of spike and burst rates with respect to days in vitro recorded via spiked (3-D) MEAs (a and b) and planar (2-D) MEAs (c and d), respectively. The spike and burst rate values and development trends were similar in both spiked and planar MEA recordings for these 3-D neural co-cultures. The gray boxes show the 95% confidence interval at each time point (day 15 only had 2 cultures sampled), and each dark circle indicates activity of a unique culture. As a common trend, the spike and the burst rates increased rapidly in the first week in culture, maximized during the second week (suggesting a homeostatic process or saturation) and gradually dropped in the third week. The burst rate distribution is similar in trend to the spike rate distribution mainly because a majority of spikes lie within bursts
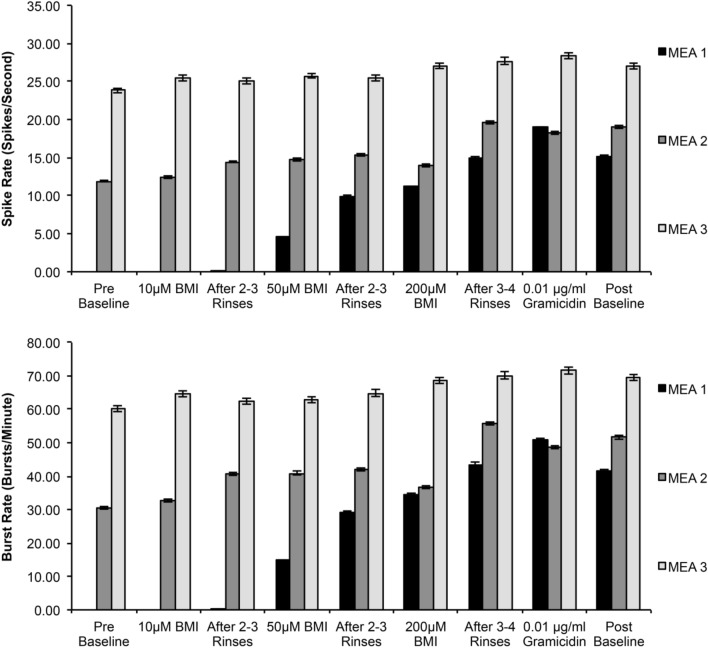
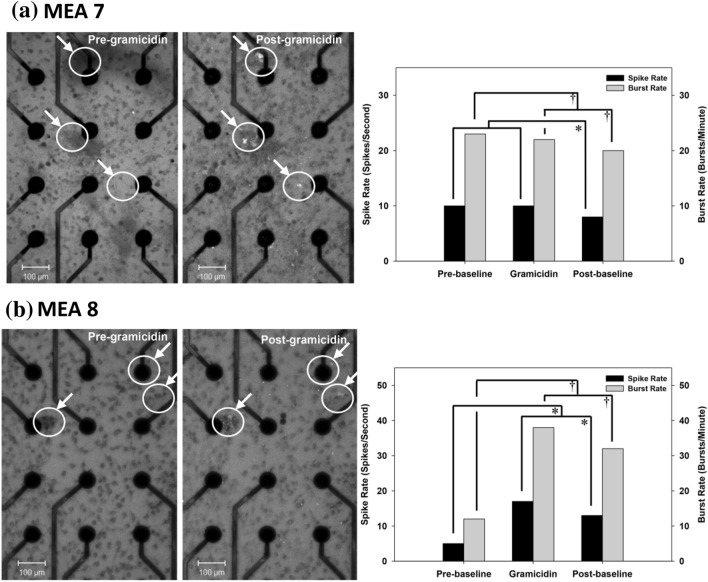
The response to BMI was varied. In 2 out of 3 cultures at 21 days in vitro, adding 10–200 μM BMI caused significant spike and burst rate increases, with only marginal increases in the third culture (Fig. 4). It should be noted that the culture with only marginal increases had higher than average baseline spike and burst rates prior to the chemical perturbation. Likewise, the addition of gramicidin (1 µg/ml) evoked varied electrophysiological activity changes in two separate cultures, although subsequent media rinses reduced activity significantly (Fig. 5). Pore formation upon addition of gramicidin was confirmed by measuring an increase in cellular permeability as noted by fluorescent calcein intake (Fig. 5).
Fig. 4.
The electrophysiological response of 3-D neural co-cultures at 21 days in vitro to chemical perturbations via BMI and gramicidin in terms of changes in spike (a) and burst rates (b). BMI is expected to have an excitatory effect by blocking the inhibitory synaptic transmission, whereas gramicidin depolarizes neurons by creating ion-permissive pores in the plasmalemma. Note that the increase in electrophysiological activity of the cultures is in inverse proportion to base-line activity levels prior to chemical perturbation. Random network connectivity, differing inhibitory synapse numbers, and differing saturation limits for chemical excitation may explain the variability in response between the cultures. Rinses did not completely reverse the pharmacological effect indicating that the thickness and density of the cultures may limit chemical diffusion
Fig. 5.
Exposure of spiked MEA-interfaced 3-D co-cultures to gramicidin concomitantly with calcein verified poration (left) and alterations in the electrophysiological activity of the cultures (right). 2-D projections of 20 µm thick image z-stacks of 25 days in vitro 3-D co-cultures on MEAs 7 and 8 pre- and post-addition of 1 µg/ml gramicidin are shown in (a) and (b), respectively. Encircled regions show cellular calcein uptake post addition of gramicidin indicating increased cell membrane permeability. The corresponding electrophysiological activity response of these two cultures is shown in the graphs on the right for MEAs 7 and 8, respectively. Significant differences for spike rates (*) and burst rates (†) are seen at p < 0.05. Following rinsing electrophysiological activity reduced significantly for both the cultures as seen in the post-baseline activity. Note that the culture for which gramicidin exposure increased electrophysiological activity displayed the lower pre-baseline activity
3-D culture maturation
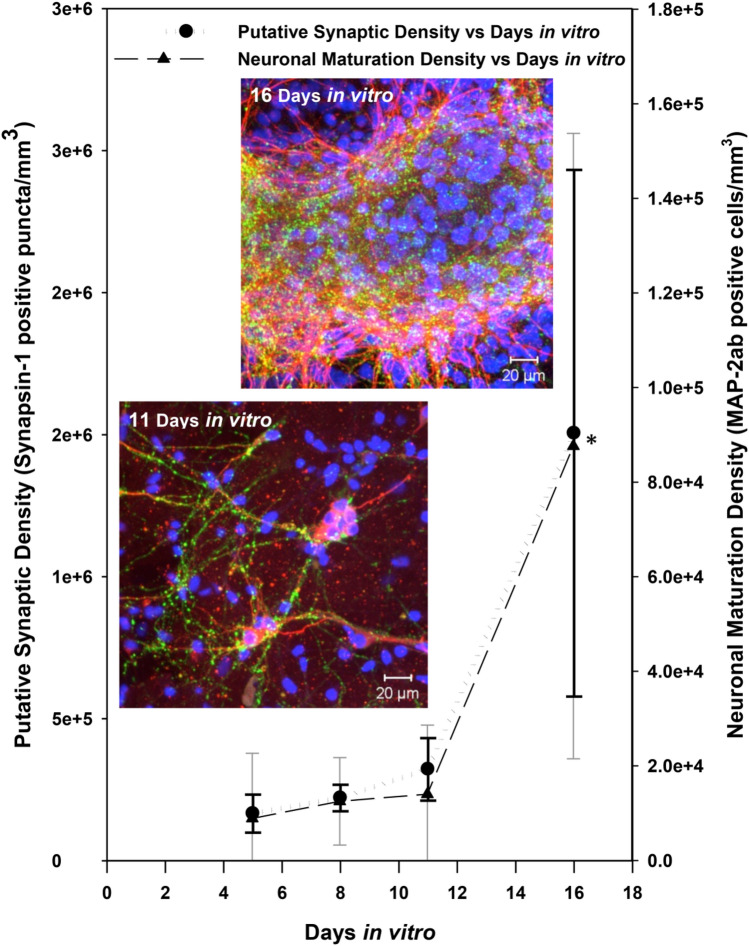
The putative synaptic density (number of synapsin-1 positive puncta per unit volume) and maturing neuronal density (number of MAP-2ab positive cell bodies per unit volume) increased dramatically midway through the second week in culture (Fig. 6). Some clustering of cells was also seen at the later time points owing to culture remodeling associated with 3-D network formation and astrocyte proliferation.
Fig. 6.
Neuronal network maturation and synaptic development increase with respect to days in vitro. The mature neuronal and the putative synaptic marker densities increased in tandem slowly in the first 1.5 weeks in culture and rapidly in the second half of the second week. * indicates significant differences for both markers at 16 days in vitro compared to previous time points, p < 0.05. Insets show 3-D z-stack image projections of 11 days (~ 51 µm thick z-section) and 16 days (~ 55 µm thick z-section) old cultures, respectively. Dramatic changes are seen in the mature neuronal density (red MAP-2ab + ve), putative synaptic density (green Synapsin 1 + ve), and nuclear density (Blue Hoechst + ve) indicating significant culture development and remodeling. (Color figure online)
Discussion
This study demonstrated the ability to interface engineered 3-D neural co-cultures with 3-D spiked MEAs and to measure electrophysiological activity and the corresponding network development as a function of days in vitro. The 3-D co-cultures were generated with primary cortical neurons and astrocytes using a high seeding density to mimic brain tissue and a laminin-rich neuro-permissive extracellular matrix micro-environment to promote network formation [11, 42]. Twice the number of neurons compared to astrocytes (2:1) were plated, to permit astrocytic support for the neurons while also seeking to control astrocytic proliferation to minimize potential interference in neuron-electrode coupling. These conditions and supplemented culturing media supported rapid neural network formation (neurite outgrowth on the order of 100 µm per day) and superior culture viability (97%) over the 3 week culture period compared to other 3-D neural culturing systems reported in literature [22, 43]. High cell density and three-dimensionality is known to promote neural network formation via enhanced neuronal connections through increased near-neighbor interactions in all three dimensions [44]. Likewise, neurite outgrowth has been shown to depend on ECM conditions in other 2-D and 3-D culturing systems as well (e.g., [45–47]).
The 3-D neuronal-astrocytic co-cultures were interfaced with spiked (3-D) and planar (2-D) MEAs following an adhesion-promoting treatment protocol, which comprised of a glow discharge treatment of the MEA culturing surfaces to render them hydrophilic [48], followed by coating with the net positively-charged polypeptide poly-d-lysine to promote long-range electrostatic interactions [49], followed by coating with the extracellular matrix protein laminin to promote both cell adhesion and cohesive interactions with Matrigel™—the hydrogel scaffold of these 3-D co-cultures. This treatment protocol along with the placement of the breathable Nylon mesh inserts on top of the cultures achieved long-term attachment of the 3-D co-cultures to the MEA surfaces enabling sustained interfacing with an electrophysiological recording and stimulation system. Finally, sealing this culturing system from the non-recording interface with O2- and CO2-permeable fluorinated ethylene propylene (FEP) membrane caps prevented evaporative losses, and thereby, osmolality shocks to the cultures, while also reducing the risk of infection. Together, this MEA-interfaced 3-D culture system enabled successful and stable recording of the electrophysiological activity of these 3-D co-cultures over a 3-week culturing period.
The 3-D co-cultures, investigated in this study, exhibited not only action potential individual spikes, but also the beginning of network-wide synchronized bursts of clustered spikes as early as 2 days in vitro (Fig. 3). This early bursting indicates initiation of networking and synaptogenesis as also verified by live-dead staining and immunocytochemical staining (See Figs. 2, 6). Dissociated cerebellar neurons plated on top of 3-D collagen hydrogels have been previously shown to express neuronal morphological and electrophysiological properties as early as 24 to 30 h in culture using the patch clamp method at the individual cell-level [50]. Overall, the spike and burst rates increased rapidly during the first week in culture, maintained relatively high activity levels during the second week, and then declined somewhat during the third week in culture (Fig. 3), suggesting physiological maturation concurrent with network development as also corroborated by an increase in neuronal maturation and putative synaptic marker density (Fig. 6). Furthermore, the electrophysiological activity development in these 3-D co-cultures with respect to time as recorded using the spiked (3-D) MEAs mirrored the recording from these cultures using the planar (2-D) MEAs. Similarly, the degree of synchronization in 3-D networks was seen to decrease after 2 weeks using calcium imaging in dissociated cerebral hippocampal neurons cultured in 3-D graphene foam scaffolds by others [19].
The cortical neurons in these cultures are expected to be a mixture of both excitatory (glutamatergic pyramidal and stellate) neurons and inhibitory (glycinergic or GABAergic) interneurons [51]. The initial rise in neural network activity in the first week of 3-D culture can be explained by the rapid neurite outgrowth and synaptic networking. It is postulated that the rapid rise in early electrophysiological activity is because of major excitatory transmission during the early period of cell culture as observed by others using traditional 2-D dissociated cultures and computational modeling [52, 53]. It has previously been noted that the inhibitory neurotransmitter GABA acts as an excitatory neurotransmitter initially during development and transitions to an inhibitory function later by the mid-second week in 2-D cultures [54, 55]. Likewise, GABAA receptors are known to initially serve excitatory function, and have been shown to develop prior to excitatory glutamate receptors in dissociated 2-D cortical cultures [56]; the later gradually increase over the first week in culture to a maximum by the mid-second week [57]. Furthermore, it is known that functional inhibitory synapses develop later than excitatory synapses upon the transition of GABA and glycine to inhibitory function [58, 59]. Therefore, given that our 3-D co-cultures maintained a high viability (97%) throughout the culturing period, the relatively levelled-off network electrophysiological activity during the latter part of the culturing period (Fig. 3), which is characterized also by increased synaptic density (Fig. 6), is postulated to be due to a relative increase in inhibitory neuronal synapses [60]. This hypothesis needs further investigation in our 3-D co-cultures system.
In addition to the postulated dynamics between inhibitory and excitatory neuronal behavior, cell maturation and synapse formation can also shed light on the structure–function development in these co-cultures. Our 3-D cultures matured after about 2 weeks as evidenced by peak MAP-2ab levels [34], which is consistent with previous reports [51, 61]. We also observed a concomitant stark increase in synaptic staining during this later period (Fig. 6), which is similar to other reports [62, 63]. The initial period of rapid increase of electrophysiological activity, followed by a period of neuronal maturation and high-level of synapse formation suggests a developmental homeostatic process between excitatory and inhibitory behavior [64, 65], albeit additional immunocytochemical staining and functional assessments are needed to confirm this hypothesis. Therefore, our MEA-interfaced 3-D co-culture system can serve as an enabling platform for the determination of structure–function relationships that govern the formation and maturation of 3-D neural networks.
Furthermore, in order to probe the bioactive-responsiveness of these cultures to the disinhibition of excitatory synaptic transmission, we added the drug bicuculline methiodide (BMI) at 21 days in vitro. We observed a general increase in electrophysiological activity in these cultures upon administration of BMI (Fig. 4), with the magnitude of increase inversely related to baseline activity levels prior to BMI administration, similar to patterns seen by others in 2-D cultures [24]. This observation lends preliminary empirical support to our postulation that significant intrinsic inhibition suppresses the innate excitatory electrophysiological activity of these cultures by 3 weeks in vitro, albeit further studies are certainly required to fully characterize the excitatory and inhibitory behavior in these 3-D co-cultures. Likewise, addition of the depolarizing agent gramicidin to a 3-D co-culture that already exhibited higher electrophysiological activity did not increase activity any further, whereas it did in a co-culture with relatively lower activity (Fig. 5). This behavioral response to gramicidin may be influenced by the same principle that limits the response to BMI, in that each neural network/culture may have a saturation level of activity. We previously observed similar heterogeneous behavior to gramicidin in 2-D neuronal cultures, including an initial increase in activity followed by a reduction [66], indicating a disruption of network behavior as expected from pore formation in the neuronal plasma membranes. The variability and delayed-responsiveness of the electrophysiological activity-changes in these co-cultures (Figs. 4, 5) also suggests that convective diffusion during chemical stimulation and washout is impeded by the density and thickness of these 3-D cultures, lending support for more controlled microfluidic platforms for chemical stimulation or drug screening studies (e.g., [67]). Moreover, culture-to-culture variability in MEA recordings has also been reported for the traditional 2-D monolayer cultures in the literature [4].
Similarities are found in the temporal trend of electrophysiological activity development in our 3-D co-cultures with that in brain slice cultures [68] as well as dissociated 2-D neural cultures [4, 69]. Despite the random network organization of our 3-D co-cultures, they characteristically displayed bursts in electrophysiological activity similar to the neuronal avalanches observed in unstimulated brain slices [3]. These cultures displayed neural network development via early neurite outgrowth, networking, synaptogenesis, and spontaneous electrophysiological activity, followed by subsequent maturation. The high plating cell density and three-dimensionality may promote this network development and maturation process by increasing the likelihood of nearest neighbor cell-to-cell interactions and thereby synchronized electrophysiological activity development throughout the culture. The electrophysiological activity may be influenced by the changing balance between inhibitory and excitatory processes over time, which warrants further investigation.
In summary, this is the first report of successful interfacing of a 3-D co-culture of dissociated primary cortical neurons and astrocytes in a 3-D Matrigel™ construct with both the spiked (3-D) and planar (2-D) MEA configurations, and the recording of the development of network-level electrophysiological activity in these co-cultures with respect to time in culture. This work shows that physiologically-relevant 3-D microenvironments can be created with control over cell density, cell types, 3-D matrix, and media conditions, serving as platforms to probe neural structure–function relationships during network development. Such ability to detect electrophysiological activity across a 3-D network using 3-D multielectrode technology would especially be attractive in more sophisticated layered and oriented neural 3-D culture systems [15, 17, 70–72]. Well-characterized 3-D neural cultures may act as robust test-beds for pharmacological screening; investigating trauma, disease, regenerative strategies, and hybrid-neural interfaces; and for understanding structure–function relationships in neural information processing.
Acknowledgements
The authors thank Dr. Brock Wester, Megan Springman, Winston Pewin, Tulika Raj, Nishil Patel, Angela Liu, Melody Keith, Willa Ni, and Rebekah Hamrick for technical and editorial assistance, and NIBIB/NINDS (BRP EB000786) for funding.
Compliance with ethical standards
Conflict of interest
The authors have no conflicts of interest to disclose.
Ethical approval
All procedures involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) of the Georgia Institute of Technology.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Novak JL, Wheeler BC. Multisite hippocampal slice recording and stimulation using a 32 element microelectrode array. J Neurosci Methods. 1988;23(2):149–159. doi: 10.1016/0165-0270(88)90187-2. [DOI] [PubMed] [Google Scholar]
- 2.De Simoni A, Griesinger CB, Edwards FA. Development of rat CA1 neurones in acute versus organotypic slices: role of experience in synaptic morphology and activity. J Physiol. 2003;550(1):135–147. doi: 10.1113/jphysiol.2003.039099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beggs JM, Plenz D. Neuronal avalanches are diverse and precise activity patterns that are stable for many hours in cortical slice cultures. J Neurosci. 2004;24(22):5216–5229. doi: 10.1523/JNEUROSCI.0540-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wagenaar D, Pine J, Potter S. An extremely rich repertoire of bursting patterns during the development of cortical cultures. BMC Neurosci. 2006;7(1):1–18. doi: 10.1186/1471-2202-7-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gähwiler B, Capogna M, Debanne D, McKinney R, Thompson S. Organotypic slice cultures: a technique has come of age. Trends Neurosci. 1997;20(10):471–477. doi: 10.1016/s0166-2236(97)01122-3. [DOI] [PubMed] [Google Scholar]
- 6.Hoffman RM. To do tissue culture in two or three dimensions? That is the question. Stem Cells. 1993;11(2):105–111. doi: 10.1002/stem.5530110205. [DOI] [PubMed] [Google Scholar]
- 7.Fawcett JW, Barker RA, Dunnett SB. Dopaminergic neuronal survival and the effects of bFGF in explant, three dimensional and monolayer cultures of embryonic rat ventral mesencephalon. Exp Brain Res. 1995;106(2):275–282. doi: 10.1007/BF00241123. [DOI] [PubMed] [Google Scholar]
- 8.Schindler M, Nur-E-Kamal A, Ahmed I, Kamal J, Liu H, Amor N, Ponery A, Crockett D, Grafe T, Chung H. Living in three dimensions: 3D nanostructured environments for cell culture and regenerative medicine. Cell Biochem Biophys. 2006;45(2):215–227. doi: 10.1385/CBB:45:2:215. [DOI] [PubMed] [Google Scholar]
- 9.Puschmann TB, Zandén C, De Pablo Y, Kirchhoff F, Pekna M, Liu J, Pekny M. Bioactive 3D cell culture system minimizes cellular stress and maintains the in vivo-like morphological complexity of astroglial cells. Glia. 2013;61(3):432–440. doi: 10.1002/glia.22446. [DOI] [PubMed] [Google Scholar]
- 10.Duval K, Grover H, Han L-H, Mou Y, Pegoraro AF, Fredberg J, Chen Z. Modeling physiological events in 2D vs. 3D cell culture. Physiology. 2017;32(4):266–277. doi: 10.1152/physiol.00036.2016. [DOI] [PMC free article] [PubMed] [Google Scholar] [Research Misconduct Found]
- 11.LaPlaca MC, Vernekar VN, Shoemaker JT, Cullen DK (eds) (2010) Three-dimensional neuronal cultures. In: Berthiaume JMF (eds), Methods in bioengineering: 3D tissue engineering, Artech House Publishers, London.
- 12.Shuler ML, Hickman JJ. Toward in vitro models of brain structure and function. Proc Natl Acad Sci. 2014;111(38):13682–13683. doi: 10.1073/pnas.1414484111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hopkins AM, DeSimone E, Chwalek K, Kaplan DL. 3D in vitro modeling of the central nervous system. Prog Neurobiol. 2015;125:1–25. doi: 10.1016/j.pneurobio.2014.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Irons H, Cullen D, Shapiro N, Lambert N, Lee R, LaPlaca M. Three-dimensional neural constructs: a novel platform for neurophysiological investigation. J Neural Eng. 2008;5(3):333–341. doi: 10.1088/1741-2560/5/3/006. [DOI] [PubMed] [Google Scholar]
- 15.Pautot S, Wyart C, Isacoff EY. Colloid-guided assembly of oriented 3D neuronal networks. Nat Methods. 2008;5(8):735–740. doi: 10.1038/NMETH.1236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Frega M, Tedesco M, Massobrio P, Pesce M, Martinoia S. Network dynamics of 3D engineered neuronal cultures: a new experimental model for in vitro electrophysiology. Sci Rep. 2014;4:5489. doi: 10.1038/srep05489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tang-Schomer MD, White JD, Tien LW, Schmitt LI, Valentin TM, Graziano DJ, Hopkins AM, Omenetto FG, Haydon PG, Kaplan DL. Bioengineered functional brain-like cortical tissue. Proc Natl Acad Sci. 2014;2014:201324214. doi: 10.1073/pnas.1324214111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee W, Shim S, Park JH, Kim SJ. A three-dimensional neural cell construct for implantable neural interface. Biomed Eng Lett. 2016;6(3):172–180. [Google Scholar]
- 19.Severino FPU, Ban J, Song Q, Tang M, Bianconi G, Cheng G, Torre V. The role of dimensionality in neuronal network dynamics. Sci Rep. 2016;6:29640. doi: 10.1038/srep29640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Valderhaug VD, Glomm WR, Sandru EM, Yasuda M, Sandvig A, Sandvig I. Formation of neural networks with structural and functional features consistent with small-world network topology on surface-grafted polymer particles. R Soc Open Sci. 2019;6(10):191086. doi: 10.1098/rsos.191086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Soscia DA, Lam D, Tooker AC, Enright HA, Triplett M, Karande P, Peters SK, Sales AP, Wheeler EK, Fischer NO. A flexible 3-dimensional microelectrode array for in vitro brain models. Lab Chip. 2020;20(5):901–911. doi: 10.1039/c9lc01148j. [DOI] [PubMed] [Google Scholar]
- 22.Xu T, Molnar P, Gregory C, Das M, Boland T, Hickman JJ. Electrophysiological characterization of embryonic hippocampal neurons cultured in a 3D collagen hydrogel. Biomaterials. 2009;30(26):4377–4383. doi: 10.1016/j.biomaterials.2009.04.047. [DOI] [PubMed] [Google Scholar]
- 23.Bourke JL, Quigley AF, Duchi S, O’Connell CD, Crook JM, Wallace GG, Cook MJ, Kapsa RM. Three-dimensional neural cultures produce networks that mimic native brain activity. J Tissue Eng Regen Med. 2018;12(2):490–493. doi: 10.1002/term.2508. [DOI] [PubMed] [Google Scholar]
- 24.Keefer EW, Gramowski A, Stenger DA, Pancrazio JJ, Gross GW. Characterization of acute neurotoxic effects of trimethylolpropane phosphate via neuronal network biosensors. Biosens Bioelectron. 2001;16(7–8):513–525. doi: 10.1016/s0956-5663(01)00165-8. [DOI] [PubMed] [Google Scholar]
- 25.Doebler JA. Effects of neutral ionophores on membrane electrical characteristics of NG108-15 cells. Toxicol Lett. 2000;114(1–3):27–38. doi: 10.1016/s0378-4274(99)00193-9. [DOI] [PubMed] [Google Scholar]
- 26.Madison R, da Silva CF, Dikkes P, Chiu T-H, Sidman RL. Increased rate of peripheral nerve regeneration using bioresorbable nerve guides and a laminin-containing gel. Exp Neurol. 1985;88(3):767–772. doi: 10.1016/0014-4886(85)90087-1. [DOI] [PubMed] [Google Scholar]
- 27.Kleinman H, McGarvey M, Hassell J, Star V, Cannon F, Laurie G, Martin G. Basement membrane complexes with biological activity. Biochemistry. 1986;25(2):312–318. doi: 10.1021/bi00350a005. [DOI] [PubMed] [Google Scholar]
- 28.Vukicevic S, Kleinman H, Luyten F, Roberts A, Roche N, Reddi A. Identification of multiple active growth factors in basement membrane Matrigel suggests caution in interpretation of cellular activity related to extracellular matrix components. Exp Cell Res. 1992;202(1):1–8. doi: 10.1016/0014-4827(92)90397-q. [DOI] [PubMed] [Google Scholar]
- 29.Cullen DK, Vukasinovic J, Glezer A, LaPlaca MC. Microfluidic engineered high cell density three-dimensional neural cultures. J Neural Eng. 2007;4(2):159. doi: 10.1088/1741-2560/4/2/015. [DOI] [PubMed] [Google Scholar]
- 30.Potter SM, DeMarse TB. A new approach to neural cell culture for long-term studies. J Neurosci Methods. 2001;110(1–2):17–24. doi: 10.1016/s0165-0270(01)00412-5. [DOI] [PubMed] [Google Scholar]
- 31.Heuschkel M, Wirth C, Steidl E-M, Buisson B. Development of 3-D multi-electrode arrays for use with acture tissue slices. In: Taketani M, Baudry M, editors. Advances in network electrophysiology using multi-electrode arrays. New York: Springer; 2006. pp. 69–111. [Google Scholar]
- 32.Legendy C, Salcman M. Bursts and recurrences of bursts in the spike trains of spontaneously active striate cortex neurons. J Neurophysiol. 1985;53(4):926–939. doi: 10.1152/jn.1985.53.4.926. [DOI] [PubMed] [Google Scholar]
- 33.Caceres A, Banker G, Binder L. Immunocytochemical localization of tubulin and microtubule-associated protein 2 during the development of hippocampal neurons in culture. J Neurosci. 1986;6(3):714–722. doi: 10.1523/JNEUROSCI.06-03-00714.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nunez J. Immature and mature variants of MAP2 and tau proteins and neuronal plasticity. Trends Neurosci. 1988;11(11):477–479. doi: 10.1016/0166-2236(88)90004-5. [DOI] [PubMed] [Google Scholar]
- 35.Goedert M, Crowther R, Garner C. Molecular characterization of microtubule-associated proteins tau and MAP2. Trends Neurosci. 1991;14(5):193–199. doi: 10.1016/0166-2236(91)90105-4. [DOI] [PubMed] [Google Scholar]
- 36.Binder LI, Frankfurter A, Rebhun LI. The distribution of tau in the mammalian central nervous system. J Cell Biol. 1985;101(4):1371–1378. doi: 10.1083/jcb.101.4.1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Migheli A, Butler M, Brown K, Shelanski M. Light and electron microscope localization of the microtubule-associated tau protein in rat brain. J Neurosci. 1988;8(6):1846–1851. doi: 10.1523/JNEUROSCI.08-06-01846.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bignami A, Dahl D, Rueger DC. Glial fibrillary acidic protein (GFA) in normal neural cells and in pathological conditions. In: Fedoroff S, Hertz, L, editors. Advances in cellular neurobiology. Elsevier; 1980. pp. 285–310.
- 39.Cullen D, Simon C, LaPlaca M. Strain rate-dependent induction of reactive astrogliosis and cell death in three-dimensional neuronal–astrocytic co-cultures. Brain Res. 2007;1158:103–115. doi: 10.1016/j.brainres.2007.04.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fletcher TL, Cameron P, De Camilli P, Banker G. The distribution of synapsin I and synaptophysin in hippocampal neurons developing in culture. J Neurosci. 1991;11(6):1617–1626. doi: 10.1523/JNEUROSCI.11-06-01617.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cullen DK, Gilroy ME, Irons HR, LaPlaca MC. Synapse-to-neuron ratio is inversely related to neuronal density in mature neuronal cultures. Brain Res. 2010;1359:44–55. doi: 10.1016/j.brainres.2010.08.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cullen DK, Vernekar VN, LaPlaca MC. Trauma-induced plasmalemma disruptions in three-dimensional neural cultures are dependent on strain modality and rate. J Neurotrauma. 2011;28(11):2219–2233. doi: 10.1089/neu.2011.1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.O’Connor S, Stenger D, Shaffer K, Ma W. Survival and neurite outgrowth of rat cortical neurons in three-dimensional agarose and collagen gel matrices. Neurosci Lett. 2001;304(3):189–193. doi: 10.1016/s0304-3940(01)01769-4. [DOI] [PubMed] [Google Scholar]
- 44.Peretz H, Talpalar AE, Vago R, Baranes D. Superior survival and durability of neurons and astrocytes on 3-dimensional aragonite biomatrices. Tissue Eng. 2007;13(3):461–472. doi: 10.1089/ten.2005.0522. [DOI] [PubMed] [Google Scholar]
- 45.Yu X, Dillon GP, Bellamkonda RV. A laminin and nerve growth factor-laden three-dimensional scaffold for enhanced neurite extension. Tissue Eng. 1999;5(4):291–304. doi: 10.1089/ten.1999.5.291. [DOI] [PubMed] [Google Scholar]
- 46.Lin P-W, Wu C-C, Chen C-H, Ho H-O, Chen Y-C, Sheu M-T. Characterization of cortical neuron outgrowth in two- and three-dimensional culture systems. J Biomed Mater Res Part B Appl Biomater. 2005;75B(1):146–157. doi: 10.1002/jbm.b.30276. [DOI] [PubMed] [Google Scholar]
- 47.Lam D, Enright HA, Cadena J, Peters SK, Sales AP, Osburn JJ, Soscia DA, Kulp KS, Wheeler EK, Fischer NO. Tissue-specific extracellular matrix accelerates the formation of neural networks and communities in a neuron-glia co-culture on a multi-electrode array. Sci Rep. 2019;9(1):1–15. doi: 10.1038/s41598-019-40128-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vernekar VN, Cullen DK, Fogleman N, Choi Y, Garcia AJ, Allen MG, Brewer GJ, LaPlaca MC. SU-8 2000 rendered cytocompatible for neuronal bioMEMS applications. J Biomed Mater Res A. 2009;89(1):138–151. doi: 10.1002/jbm.a.31839. [DOI] [PubMed] [Google Scholar]
- 49.Vernekar VN, Latour RA., Jr Adsorption thermodynamics of a mid-chain peptide residue on functionalized SAM surfaces using SPR. Mater Res Innov. 2005;9(2):53–55. [Google Scholar]
- 50.Coates P, Fermini B, Strahlendorf J, Strahlendorf H. Utilization of three-dimensional culture for early morphometric and electrophysiological analyses of solitary cerebellar neurons. Dev Neurosci. 1992;14:35–43. doi: 10.1159/000111645. [DOI] [PubMed] [Google Scholar]
- 51.De Lima AD, Voigt T. Identification of two distinct populations of aminobutyric acidergic neurons in cultures of the rat cerebral cortex. J Compar Neurol. 1997;388(4):526–540. [PubMed] [Google Scholar]
- 52.Ramakers G, Van Galen H, Feenstra M, Corner M, Boer G. Activity-dependent plasticity of inhibitory and excitatory amino acid transmitter systems in cultured rat cerebral cortex. Int J Dev Neurosci Off J Int Soc Dev Neurosci. 1994;12(7):611–621. doi: 10.1016/0736-5748(94)90013-2. [DOI] [PubMed] [Google Scholar]
- 53.Van Ooyen A, Van Pelt J, Corner M. Implications of activity dependent neurite outgrowth for neuronal morphology and network development. J Theor Biol. 1995;172(1):63–82. doi: 10.1006/jtbi.1995.0005. [DOI] [PubMed] [Google Scholar]
- 54.Leinekugel X, Khalilov I, McLean H, Caillard O, Gaiarsa JL, Ben-Ari Y, Khazipov R. GABA is the principal fast-acting excitatory transmitter in the neonatal brain. Adv Neurol. 1999;79:189–201. [PubMed] [Google Scholar]
- 55.Ben-Ari Y. Developing networks play a similar melody. Trends Neurosci. 2001;24(6):353–360. doi: 10.1016/s0166-2236(00)01813-0. [DOI] [PubMed] [Google Scholar]
- 56.Koller H, Siebler M, Schmalenbach C, Muller H. GABA and glutamate receptor development of cultured neurons from rat hippocampus, septal region, and neocortex. Synapse. 1990;5(1):59–64. doi: 10.1002/syn.890050105. [DOI] [PubMed] [Google Scholar]
- 57.Lesuisse C, Martin L. Long-term culture of mouse cortical neurons as a model for neuronal development, aging, and death. J Neurobiol. 2002;51(1):9–23. doi: 10.1002/neu.10037. [DOI] [PubMed] [Google Scholar]
- 58.Luhmann H, Prince D. Postnatal maturation of the GABAergic system in rat neocortex. J Neurophysiol. 1991;65(2):247–263. doi: 10.1152/jn.1991.65.2.247. [DOI] [PubMed] [Google Scholar]
- 59.Burgard EC, Hablitz JJ. Developmental changes in NMDA and non-NMDA receptor-mediated synaptic potentials in rat neocortex. J Neurophysiol. 1993;69(1):230–240. doi: 10.1152/jn.1993.69.1.230. [DOI] [PubMed] [Google Scholar]
- 60.Fellous JM, Sejnowski TJ. Regulation of persistent activity by background inhibition in an in vitro model of a cortical microcircuit. Cereb Cortex. 2003;13(11):1232–1241. doi: 10.1093/cercor/bhg098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.O’Shaughnessy TJ, Lin HJ, Ma W. Functional synapse formation among rat cortical neurons grown on three-dimensional collagen gels. Neurosci Lett. 2003;340(3):169–172. doi: 10.1016/s0304-3940(03)00083-1. [DOI] [PubMed] [Google Scholar]
- 62.Van Huizen F, Romijn H, Habets A. Synaptogenesis in rat cerebral cortex cultures is affected during chronic blockade of spontaneous bioelectric activity by tetradotoxin. Brain Res. 1985;19(1):67–80. doi: 10.1016/0165-3806(85)90232-9. [DOI] [PubMed] [Google Scholar]
- 63.De Lima A, Merten M, Voigt T. Neuritic differentiation and synaptogenesis in serum-free neuronal cultures of the rat cerebral cortex. J Compar Neurol. 1997;382(2):230–246. doi: 10.1002/(sici)1096-9861(19970602)382:2<230::aid-cne7>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 64.Turrigiano GG. Homeostatic plasticity in neuronal networks: the more things change, the more they stay the same. Trends Neurosci. 1999;22(5):221–227. doi: 10.1016/s0166-2236(98)01341-1. [DOI] [PubMed] [Google Scholar]
- 65.Corner M, Van Pelt J, Wolters P, Baker R, Nuytinck R. Physiological effects of sustained blockade of excitatory synaptic transmission on spontaneously active developing neuronal networks—an inquiry into the reciprocal linkage between intrinsic biorhythms and neuroplasticity in early ontogeny. Neurosci Biobehav Rev. 2002;26(2):127–185. doi: 10.1016/s0149-7634(01)00062-8. [DOI] [PubMed] [Google Scholar]
- 66.Prado GR, Ross JD, DeWeerth SP, LaPlaca MC. Mechanical trauma induces immediate changes in neuronal network activity. J Neural Eng. 2005;2(4):148. doi: 10.1088/1741-2560/2/4/011. [DOI] [PubMed] [Google Scholar]
- 67.Li X, Valadez AV, Zuo P, Nie Z. Microfluidic 3D cell culture: potential application for tissue-based bioassays. Bioanalysis. 2012;4(12):1509–1525. doi: 10.4155/bio.12.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Uesaka N, Hirai S, Maruyama T, Ruthazer ES, Yamamoto N. Activity dependence of cortical axon branch formation: a morphological and electrophysiological study using organotypic slice cultures. J Neurosci. 2005;25(1):1–9. doi: 10.1523/JNEUROSCI.3855-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Siebler M, Köller H, Stichel CC, Müller HW, Freund HJ. Spontaneous activity and recurrent inhibition in cultured hippocampal networks. Synapse. 1993;14(3):206–213. doi: 10.1002/syn.890140304. [DOI] [PubMed] [Google Scholar]
- 70.Kunze A, Giugliano M, Valero A, Renaud P. Micropatterning neural cell cultures in 3D with a multi-layered scaffold. Biomaterials. 2011;32(8):2088–2098. doi: 10.1016/j.biomaterials.2010.11.047. [DOI] [PubMed] [Google Scholar]
- 71.Ren T, Grosshäuser B, Sridhar K, Nieland TJ, Tocchio A, Schepers U, Demirci U. 3-D geometry and irregular connectivity dictate neuronal firing in frequency domain and synchronization. Biomaterials. 2019;197:171–181. doi: 10.1016/j.biomaterials.2019.01.017. [DOI] [PubMed] [Google Scholar]
- 72.Musick K, Khatami D, Wheeler BC. Three-dimensional micro-electrode array for recording dissociated neuronal cultures. Lab Chip. 2009;9(14):2036–2042. doi: 10.1039/b820596e. [DOI] [PMC free article] [PubMed] [Google Scholar]