Abstract
The ongoing coronavirus disease 19 caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has become fatal for the world with affected population crossing over 25 million in more than 217 countries, consequently declared a global pandemic by the World Health Organization. Unfortunately, neither specific prophylactic or therapeutic drugs nor vaccines are available. To address the unmet medical needs, we explored a strategy identifying new compounds targeting the main protease (Mpro) of SARS-CoV-2. Targeting the SARS-CoV-2 Mpro crystal structure (PDB ID: 6LU7) a combination of in silico screening, molecular docking, and dynamic approaches, a set of 5000 compounds of the ZINC database were screened. As a result, we identified and ranked the top 20 compounds based on the scores of ligand-interaction, their drug-likeness properties, and their predicted antiviral efficacies. The prominent drug-like and potent inhibitory compounds are 2-[2-(2-aminoacetyl) aminoacetyl] amino-3-(4-hydroxyphenyl)-propanamide (ZINC000004762511), 6′-fluoroaristeromycin (ZINC000001483267) and cyclo (L-histidyl-L-histidyl) (ZINC000005116916) scaffolds. Further in vitro and in vivo validations are required to demonstrate anti-SARS-CoV-2 activities.
Electronic supplementary material
The online version of this article (10.1007/s13337-020-00627-6) contains supplementary material, which is available to authorized users.
Keywords: COVID-19, SARS-CoV-2, Therapy, Protease, Zinc, Inhibitors
In the late December 2019, hospitals in Wuhan, Hubei, China reported cases of unexplained pneumonia, later named the coronavirus disease 2019 (COVID-19). The responsible causative agent for COVID-19 was identified as the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1–3]. As of now, according to the World Health Organization (WHO) around 25 million laboratory-confirmed cases have been reported worldwide and more than 800,000 of deaths resulting in a fatality rate of 3.2% [2].
European countries and the United States of America have become the epicenters of the SARS-CoV-2 outbreak [3]. In the majority of cases, COVID-19 is either asymptomatic or causing only minor clinical symptoms; however, in patients with medical preconditions, the fatality rates are alarming [4, 5]. As of now, no success is seen in the front of having a specific therapeutic drug or prophylactic vaccine. The current approach to treat COVID-19 relies on the the use of hydroxychloroquine, chloroquine, azithromycin, remdesivir, lopinavir-ritonavir, favipiravir, ribavirin, interferon, and convalescent plasma [6–11]. To quickly address the unmet medical needs, in silico approaches can accelerate drug discovery and development, and can be a complementary method for the classical screening and identification of specific drugs against COVID-19 [12, 13]. Taking the advantage of the main protease (Mpro) structure that became available recently [14], we carried out a virtual in silico screening of nearly 5000 ZINC compound database to identify new inhibitors targeting the SARS-CoV-2.
The Mpro (PDB ID: 6LU7) structure of SARS-CoV-2 was obtained from protein data bank (PDB) (https://www.rcsb.org/) [14]. The active site prediction was performed using Computed Atlas for Surface Topography of Proteins (CASTp) (https://sts.bioe.uic.edu/castp/index.html?201l).
The ZINC database [15] was used, and a set of 5000 public-available compounds were downloaded in.mol2 format. The predicted active sites from CASTp were then used for molecular simulations, which were performed by using MtiOpenScreen [16], a virtual screening online server-based on AutoDock Vina. The grid box was created based on predicted active site residues using MtiOpenScreen [16] option that allows selecting the docking grid based on specific residues.
To explore more the interaction between our target and top-ranked ligands, we used Ligplot+ [17]. This tool allows analyzing the ligand-receptor interactions by plotting hydrogen bonding and hydrophobic interactions.
Absorption, Distribution, Metabolism, and Excretion (also known as ADME) evaluation has been applied to investigate the pharmacological activity of the compound. The characterization of ADME and Toxicity was performed based on the SWISS ADME server (https://www.swissadme.ch). The rules of Lipinski, Ghose, and Veber applied to predict drug-likeness following molecular weight, LogP, HBD, and number of HPA parameters.
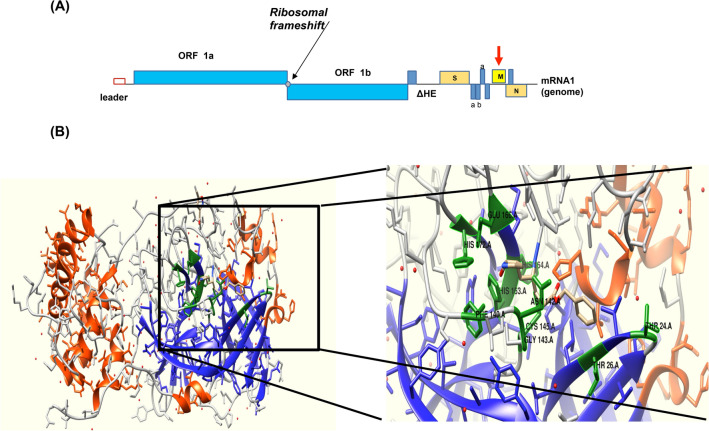
The Mpro plays a pivotal role in the replication and transcription of SARS-CoV-2 and thus becomes a apposite drug target [14]. It forms a homodimer of two chains A and B, which are complexed to a native ligand (Fig. 1a). The active site pockets resulted from CASTp has shown that potential site to be localized between THR24, 26, PHE140, ASN142, GLY143, CYS145, HIS163, 164, 172, and GLU166 residues positions in the chain A of the SARS-CoV-2 (Fig. 1b).
Fig. 1.
Structure of SARS-CoV-2 main protease Mpro. a Schematic presentation of the SARS-CoV-2 genome organization. b The 3D of Mpro structure. The strand structure is represented in blue and the helix is red. The predicted binding site is represented in green
The 5000 compounds from the ZINC database were docked using Autodock Vina. Table 1 and supplementary table 1 show the top 20 compounds with binding energies ranging from − 7 to − 6 kcal/mol of chain A active site of Mpro (Supplementary figure 1).
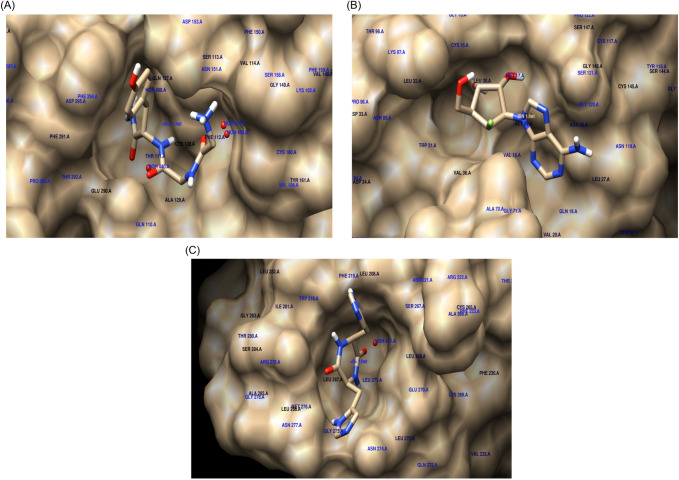
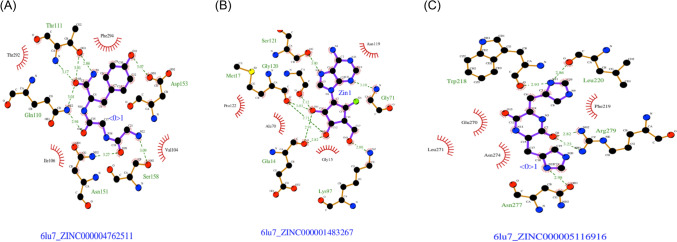
The docking simulations showed, by a ranking of binding energies score, that compound 2-[2-(2-aminoacetyl)aminoacetyl]amino-3-(4-hydroxyphenyl)–propanamide (ZINC000004762511) has the best predicted binding energy of −7 kcal/mol. The interaction between the top-ranked hits, according to its docking score and SARS-CoV-2 Mpro, has been explored using ligplot+ [16]. The data showed that ZINC000004762511 has Asn151, Thr111, Asp153, Gln110, and Ser158 residues interact with ligand through hydrogen bonding, and Ile106, Thr292, Phe294, and Val104 have a hydrophobic binding (Figs. 2a, 3a). The ZINC000004762511 compound is known as glycylglycyl–L–tyrosinamide, and until to date has not yet been described to have antiviral efficacies.
Fig. 2.
Docking simulations. The SARS-CoV-2 Mpro structure is represented in a surface format with a label of each residue interacting in the zone with the ligand. a A complex of the 2-[2-(2-aminoacetyl) aminoacetyl]amino-3-(4-hydroxyphenyl)-propanamide compound with the structure of main protease Mpro of the SARS-CoV-2. b ZINC000001483267 compound. c ZINC000005116916 compound. Amino acids labels are shown in blue and ligand atoms are shown in red, green and blue
Fig. 3.
Main protease Mpro of the SARS-CoV-2-ligand interactions with ranked compounds. a ZINC000004762511 compound. b ZINC000001483267 compound. c ZINC000005116916 compound. Residues colored in purple showing hydrogen bonding with residues. Green dash lines represent the hydrogen bond interactions. Green, red, blue, and black are highlighted atoms of ligan
Moreover, we found that the 6′-fluoroaristeromycin (ZINC000001483267) has a score of − 7 kcal/mol and ligand-interaction show that this compound has Asp289, Glu288, Lys5, Lys137, and Arg131 as hydrogen bonding residues and Thr198, Asp197, Thr199, and Glu290 residues forming hydrophobic binding) (Figs. 2b, 3a). Important to note, it was reported that this compound is an inhibitor of the S-adenosylhomocysteine hydrolase [18].
Furthermore, our data demonstrated that the ZINC000005116916 compound has a lower binding energy of − 6.1 kcal/mol (Figs. 2c, 3c). It interacts through hydrophobic binding with Trp218, Leu220, Asn277, and Arg279 residues. In addition, hydrophobic binding interactions were formed by Leu271, Glu270, Asn274, and Phe219 residues. Furthermore, this compound is known as cyclo(L-histidyl-L-histidyl) and is a part of cyclic peptides that are reported to have a worthy biological function pertinent for the treatment of cardiovascular-related disease, cancer, and infectious diseases [19, 20]. In contrast, regarding other ranked compounds, no available data was published so far.
Based on our computational strategy, the pharmacokinetic properties and drug-likeness of the top 20 ranked scoring molecules that show the potential of Mpro inhibitors of the SARS-CoV-2 are shown in supplementary table 1. As per the pharmacokinetic properties, most of the compounds showed low gastrointestinal absorption except six compounds that exhibit higher absorption. These compounds didn’t show blood–brain barrier (BBB) penetrability and no inhibition to (CYP1A2, CYP2C19, CYP2C9, CYP2D6, CYP3A4) except one compounds (ZINC000005116916). The drug-likeness forecast following the selected Lipinski, Ghose, and Veber rules and bioavailability scores (Supplementary Table 2), the results of our data recommend that majority of the compounds have no PAIN (pan-assay interference compounds) alerts; in other terms, none of the analyzed hits has reported having false-positive results in high-throughput screenings, and only three compounds have Brenk alerts, a filter determined based on a list of fragments to be putatively toxic [21]. In addition, most of the predicted compounds have shown no Lipinski's violations except five compounds (Supplementary Table 2). The prediction of pharmacokinetics and drug-likeness properties remain a powerful step in in silico drug design due to its capacity to orient and to help scientist to make a decision about which compound to test in vitro according to predicted models.
In the absence of FDA-approved therapies, COVID-19 will continue to raise fatality rates. Several studies were initiated to discover new treatments or to repurpose know drugs quickly [9, 11, 22]. However, these treatments are now under clinical trial investigations [7, 23, 24–26]. We applied a computer-aided drug design to identify new SARS-CoV-2 Mpro inhibitors, and identified 20 new prospective inhibitors. Further in vitro and in vivo validations are required to show their efficacy as SARS-CoV-2 main protease inhibitors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This study was supported by Institut Pasteur du Maroc.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Coronaviridae study group of the international committee on taxonomy of V. The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020. [DOI] [PMC free article] [PubMed]
- 2.WHO. Coronavirus disease (COVID-19) outbreak. Weekly Epidemiological Update. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200831-weekly-epi-update-3.pdf?sfvrsn=d7032a2a_4. Accessed 4 Sept 2020.
- 3.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W, China Novel Coronavirus I, Research T A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ahn JY, Sohn Y, Lee SH, Cho Y, Hyun JH, Baek YJ, Jeong SJ, Kim JH, Ku NS, Yeom JS, Roh J, Ahn MY, Chin BS, Kim YS, Lee H, Yong D, Kim HO, Kim S, Choi JY. Use of convalescent plasma therapy in two COVID-19 patients with acute respiratory distress syndrome in Korea. J Korean Med Sci. 2020;35:e149. doi: 10.3346/jkms.2020.35.e149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Duan K, Liu B, Li C, Zhang H, Yu T, Qu J, Zhou M, Chen L, Meng S, Hu Y, Peng C, Yuan M, Huang J, Wang Z, Yu J, Gao X, Wang D, Yu X, Li L, Zhang J, Wu X, Li B, Xu Y, Chen W, Peng Y, Hu Y, Lin L, Liu X, Huang S, Zhou Z, Zhang L, Wang Y, Zhang Z, Deng K, Xia Z, Gong Q, Zhang W, Zheng X, Liu Y, Yang H, Zhou D, Yu D, Hou J, Shi Z, Chen S, Chen Z, Zhang X, Yang X. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Nat Acad Sci. 2020;117(17):9490–9496. doi: 10.1073/pnas.2004168117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J, Giordanengo V, Vieira VE, Dupont HT, Honore S, Colson P, Chabriere E, La Scola B, Rolain JM, Brouqui P, Raoult D. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrobi Agents. 2020;56:105949. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 9.Kupferschmidt K, Cohen J. Race to find COVID-19 treatments accelerates. Science. 2020;367:1412–1413. doi: 10.1126/science.367.6485.1412. [DOI] [PubMed] [Google Scholar]
- 10.Kruse RL. Therapeutic strategies in an outbreak scenario to treat the novel coronavirus originating in Wuhan, China. F1000Research. 2020;9:72. doi: 10.12688/f1000research.22211.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, Ruan L, Song B, Cai Y, Wei M, Li X, Xia J, Chen N, Xiang J, Yu T, Bai T, Xie X, Zhang L, Li C, Yuan Y, Chen H, Li H, Huang H, Tu S, Gong F, Liu Y, Wei Y, Dong C, Zhou F, Gu X, Xu J, Liu Z, Zhang Y, Li H, Shang L, Wang K, Li K, Zhou X, Dong X, Qu Z, Lu S, Hu X, Ruan S, Luo S, Wu J, Peng L, Cheng F, Pan L, Zou J, Jia C, Wang J, Liu X, Wang S, Wu X, Ge Q, He J, Zhan H, Qiu F, Guo L, Huang C, Jaki T, Hayden FG, Horby PW, Zhang D, Wang C. A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. New England J Med. 2020;382:1787–1799. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Elmessaoudi-Idrissi M, Blondel A, Kettani A, Windisch MP, Benjelloun S, Ezzikouri S. Virtual screening in hepatitis B virus drug discovery: current stateof- the-art and future perspectives. Curr Med Chem. 2018;25:2709–2721. doi: 10.2174/0929867325666180221141451. [DOI] [PubMed] [Google Scholar]
- 13.Shah B, Modi P, Sagar SR. In silico studies on therapeutic agents for COVID-19: Drug repurposing approach. Life Sci. 2020;252:117652. doi: 10.1016/j.lfs.2020.117652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jin Z, Du X, Xu Y, Deng Y, Liu M, Zhao Y, Zhang B, Li X, Zhang L, Peng C, Duan Y, Yu J, Wang L, Yang K, Liu F, Jiang R, Yang X, You T, Liu X, Yang X, Bai F, Liu H, Liu X, Guddat LW, Xu W, Xiao G, Qin C, Shi Z, Jiang H, Rao Z, Yang H. Structure of M pro from SARS-CoV-2 and discovery of its inhibitors. Nature. 2020;9:1–5. doi: 10.1038/s41586-020-2223-y. [DOI] [PubMed] [Google Scholar]
- 15.Sterling T, Irwin JJ. ZINC 15–Ligand discovery for everyone. J Chem Inf Model. 2015;55:2324–2337. doi: 10.1021/acs.jcim.5b00559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Labbe CM, Rey J, Lagorce D, Vavrusa M, Becot J, Sperandio O, Villoutreix BO, Tuffery P, Miteva MA. MTiOpenScreen: a web server for structure-based virtual screening. Nucleic Acids Res. 2015;43:W448–W454. doi: 10.1093/nar/gkv306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Laskowski RA, Swindells MB. LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J Chem Inf Model. 2011;51:2778–2786. doi: 10.1021/ci200227u. [DOI] [PubMed] [Google Scholar]
- 18.Cools M, Balzarini J, De Clercq E. Mechanism of antiviral and cytotoxic action of (+/-)-6' beta-fluoroaristeromycin, a potent inhibitor of S-adenosylhomocysteine hydrolase. Mol Pharmacol. 1991;39:718–724. [PubMed] [Google Scholar]
- 19.McCleland K, Milne PJ, Lucieto FR, Frost C, Brauns SC, Van De Venter M, Du Plessis J, Dyason K. An investigation into the biological activity of the selected histidine-containing diketopiperazines cyclo(His-Phe) and cyclo(His-Tyr) J Pharm Pharmacol. 2004;56:1143–1153. doi: 10.1211/0022357044139. [DOI] [PubMed] [Google Scholar]
- 20.Fernandez-Lopez S, Kim HS, Choi EC, Delgado M, Granja JR, Khasanov A, Kraehenbuehl K, Long G, Weinberger DA, Wilcoxen KM, Ghadiri MR. Antibacterial agents based on the cyclic D L-alpha-peptide architecture. Nature. 2001;412:452–455. doi: 10.1038/35086601. [DOI] [PubMed] [Google Scholar]
- 21.Brenk R, Schipani A, James D, Krasowski A, Gilbert IH, Frearson J, Wyatt PG. Lessons learnt from assembling screening libraries for drug discovery for neglected diseases. ChemMedChem. 2008;3:435–444. doi: 10.1002/cmdc.200700139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martinez MA. Compounds with therapeutic potential against novel respiratory 2019 coronavirus. Antimicrob Agents Chemother. 2020;64:e00399–20. doi: 10.1128/AAC.00399-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Borba MGS, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, Brito M, Mourão MPG, Brito-Sousa JD, Baía-da-Silva D, Guerra MVF, Hajjar LA, Pinto RC, Balieiro AAS, Pacheco AGF, Santos JDO, Jr, Naveca FG, Xavier MS, Siqueira AM, Schwarzbold A, Croda J, Nogueira ML, Romero GAS, Bassat Q, Fontes CJ, Albuquerque BC, Daniel-Ribeiro CT, Monteiro WM, Lacerda MVG. CloroCovid-19 Team. Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA Netw Open. 2020;3:e208857. doi: 10.1001/jamanetworkopen.2020.8857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chiotos K, Hayes M, Kimberlin DW, Jones SB, James SH, Pinninti SG, Yarbrough A, Abzug MJ, MacBrayne CE, Soma VL, Dulek DE, Vora SB, Waghmare A, Wolf J, Olivero R, Grapentine S, Wattier RL, Bio L, Cross SJ, Dillman NO, Downes KJ, Timberlake K, Young J, Orscheln RC, Tamma PD, Schwenk HT, Zachariah P, Aldrich M, Goldman DL, Groves HE, Lamb GS, Tribble AC, Hersh AL, Thorell EA, Denison MR, Ratner AJ, Newland JG, Nakamura MM. Multicenter initial guidance on use of antivirals for children with COVID-19/SARS-CoV-2. J Pediatric Infect Dis Soc. 2020;piaa045.
- 25.Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, Feldt T, Green G, Green ML, Lescure FX, Nicastri E, Oda R, Yo K, Quiros-Roldan E, Studemeister A, Redinski J, Ahmed S, Bernett J, Chelliah D, Chen D, Chihara S, Cohen SH, Cunningham J, D'Arminio Monforte A, Ismail S, Kato H, Lapadula G, L'Her E, Maeno T, Majumder S, Massari M, Mora-Rillo M, Mutoh Y, Nguyen D, Verweij E, Zoufaly A, Osinusi AO, DeZure A, Zhao Y, Zhong L, Chokkalingam A, Elboudwarej E, Telep L, Timbs L, Henne I, Sellers S, Cao H, Tan SK, Winterbourne L, Desai P, Mera R, Gaggar A, Myers RP, Brainard DM, Childs R, Flanigan T. Compassionate use of remdesivir for patients with severe Covid-19. N Engl J Med. 2020;382:2327–2336. doi: 10.1056/NEJMoa2007016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ezzikouri S, Nourlil J, Benjelloun S, Kohara M, Tsukiyama-Kohara K. Coronavirus disease 2019-Historical context, virology, pathogenesis, immunotherapy, and vaccine development. Hum Vaccin Immunother. 2020;5:1–9. doi: 10.1080/21645515.2020.1787068. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.