Abstract
Esophageal squamous cell carcinoma (ESCC) is a common subtype of esophageal cancer that is particularly prevalent in East Asian countries. Our previous expression profile analysis showed that the gene encoding protein tyrosine kinase 7 (PTK7) is upregulated in ESCC tissues. Here, we aimed to validate PTK7 as a prognostic factor and a candidate target for molecular treatment of ESCC. Both RT‐PCR and Western blot analysis of tissues from ESCC patients revealed that PTK7 was significantly upregulated in tumor tissue samples of ESCC. Immunohistochemical staining of PTK7 showed that increased expression of PTK7 was inversely correlated with overall survival (P = 0.021). In vitro knockdown of PTK7 inhibited proliferation, survival, wound healing, and invasion of ESCC cells. In addition, PTK7 knockdown decreased phosphorylation of Akt, Erk, and focal adhesion kinase (FAK), important determinants of cell proliferation, survival, and migration. Therefore, our findings suggest that PTK7 has potential as a prognostic marker for ESCC and might also be a candidate for targeted therapy in the treatment of ESCC.
Esophageal cancer is one of the most devastating malignancies. Despite recent developments in its management, advanced esophageal cancer is associated with high rates of local recurrence and distant metastasis.1, 2 Although there has been improvement in clinical outcome through multidisciplinary treatment strategies,3, 4, 5 such as total mediastinal lymph node dissection,6 and new diagnostic modalities such as positron emission tomography,7 these developments have not proven satisfactory in preventing the high recurrence rates. Currently, the prognosis is only promising if the tumor is detected early and resected completely.
Esophageal squamous cell carcinoma (ESCC) is the most common histologic subtype of esophageal cancer in East Asian countries.8 It has a unique profile of clinical and anatomical characteristics that is distinct from those of esophageal adenocarcinoma, which is more prevalent in North America, UK, and Australia.8 To develop prognostic diagnosis and effective therapeutics for ESCC, the identification of biomarkers is of vital importance. In a gene expression profiling analysis to pursue biomarkers, we identified protein tyrosine kinase 7 (PTK7; also known as colon carcinoma kinase‐4 or CCK‐4), as a candidate biomarker for ESCC.9
Protein tyrosine kinase 7 is a receptor tyrosine kinase‐like molecule containing an extracellular domain with seven immunoglobulin‐like loops, a transmembrane domain, and a defective tyrosine kinase domain that resembles a catalytic domain but lacks catalytic activity.10, 11, 12 Mice expressing a truncated form of PTK7 protein die perinatally, revealing a defect in neural tube closure and stereociliary bundle orientation. These findings implicate PTK7 as a regulator of planar cell polarity (PCP).13 It has been shown that PTK7 recruits RACK1, which affects Dsh recruitment by interaction with PKCδ1.14 Interaction between PTK7 and Dsh at the plasma membrane activates non‐canonical Wnt signaling, which then directs PCP.15 Also, PTK7 interacts with β‐catenin, enhancing β‐catenin‐dependent transcriptional events.16 These studies suggest that PTK7 plays a role in the canonical Wnt pathway, the non‐canonical Wnt pathway, and the PCP signaling pathway.
Increased PTK7 expression has been reported in several types of cancer, including colon cancer,11, 17 gastric cancer,18 lung cancer,19 acute myeloid leukemia,20, 21 and liposarcoma.22 Expression of PTK7 in leukemia cells enhances cell migration and survival.20 In addition, PTK7 knockdown has been shown to inhibit proliferation and invasion of liposarcoma cells,22 as well as proliferation and antiapoptotic activity of colon cancer cells.23 Moreover, treatment with the entire extracellular domain of PTK7 (soluble PTK7) acting as a decoy receptor or knockdown of PTK7 prevented vascular endothelial growth factor‐induced migration, invasion, and tube‐formation of HUVECs, and angiogenesis in vivo.24 However, PTK7 expression has been shown to be decreased in metastatic melanoma25 and clear cell renal cell carcinoma.26 In addition, chromosome 6p,27 on which the PTK7 gene is located, was found to be deleted in breast carcinoma28 and malignant melanoma.29
Although the role of PTK7 in different cancers has not been studied comprehensively, PTK7 is likely to have a tumor‐promoting role in general. To evaluate the prognostic value of PTK7 in ESCC, we first analyzed expression of PTK7 by RT‐PCR and Western blotting using tumor and adjacent normal tissue samples from ESCC patients. Second, we examined the expression pattern of PTK7 in surgical specimens of 157 ESCC patients by immunohistochemical (IHC) staining and investigated the correlation between expression of PTK7 and parameters of clinical outcome. Finally, we analyzed the oncogenic properties of PTK7 and PTK7‐mediated signaling pathways by PTK7 knockdown in ESCC cell lines.
Materials and Methods
Patients and preparation of tissue samples
This study was approved by the institutional review board of Korea Cancer Center Hospital. For RT‐PCR and western blot analysis, frozen samples of tumor and adjacent healthy esophageal tissue were obtained from patients who underwent surgery for ESCC in Korea Cancer Center Hospital (Seoul, Korea). For IHC staining, paraffin blocks were obtained from 157 patients who underwent curative surgical resection for thoracic ESCC from 1999 to 2006 at Korea Cancer Center Hospital and were followed up until January 2012. Surgical resection was carried out with total mediastinal, lesser curvature, and common hepatic lymph node dissection, followed by evaluation of the resection margins. Pathologic staging was based on the 7th edition of the American Joint Committee on Cancer's Cancer Staging Manual.
RNA isolation and RT‐PCR
Total RNA was isolated from frozen esophageal samples (ESCC tissue and adjacent normal epithelium) using an RNeasy Mini Kit (Qiagen, Cambridge, MA, USA). The RT‐PCR was carried out as described previously.24
Western blot analysis
Protein extraction from frozen tissues was carried out using tissue extraction reagent II (Invitrogen, Carlsbad, CA, USA) containing protease inhibitors (Roche, Basel, Switzerland). Cultured cells were lysed with lysis buffer (50 mM Tris‐HCl, pH 7.4, 150 mM NaCl, 1% Triton X‐100, 5 mM NaF, 1 mM Na3VO4) supplemented with protease inhibitor cocktail I (Calbiochem, La Jolla, CA, USA) for 10 min at 4°C. Lysates containing 50 μg protein were subjected to SDS‐PAGE and transferred onto a nitrocellulose or PVDF membrane. Western blotting was carried out using: anti‐phospho‐Erk, anti‐Erk, anti‐focal adhesion kinase (FAK), and anti‐β‐actin (Santa Cruz Biotechnology, Santa Cruz, CA, USA); anti‐phospho‐Akt (Ser473) and anti‐Akt (Cell Signaling Technology, Beverly, MA, USA); anti‐phospho‐FAK (Tyr397) (Millipore, Billerica, MA, USA); and HRP‐conjugated secondary antibodies (KOMA Biotech, Seoul, Korea and Santa Cruz Biotechnology). Rabbit anti‐PTK7 anti‐serum, which recognizes the extracellular domain of both human and murine PTK7, was used as described previously.24 The immune reactions were visualized using Immobilon Western Chemiluminescent HRP Substrate (Millipore) and a LAS‐3000 imaging system (Fujifilm, Tokyo, Japan).
Immunohistochemical staining
The IHC staining was carried out using a tissue array technique. Paraffin blocks with a diameter of 3 mm were punched out and arranged into 6 × 4 block tissue arrays, which were sectioned at 4‐μm thickness. After paraffin removal and dehydration, the sections were quenched with 3% H2O2 in methanol for 20 min and transferred to warm 10 mM citrate buffer solution (pH 6) for 10 min. After washing with PBS (137 mM NaCl, 10 mM Na2HPO4, 2.7 mM KCl, and 2 mM KH2PO4; pH 7.4), the sections were incubated with primary antibody in a humid chamber at room temperature for 1 h. The sections were then washed with PBS, incubated with HRP polymer conjugate for 15 min at room temperature, and DAB Chromogen (ScyTek Laboratory, West Logan, UT, USA) was added for 5 min at room temperature. After staining, micrographs were taken and evaluated for intensity of staining and percentage of stained cells by a pathologist who was blind to the clinical information. The intensity of staining was scored in a 0–10 scale, with the following intensity score ranges: 0–3, weak; 4–7, moderate; and 8–10, strong.
Cell culture
Human ESCC cell lines TE‐5, TE‐9, TE‐10, TE‐11, and TE‐14 were obtained from the RIKEN BioResource Center (Tsukuba, Japan). Human colon cancer SW480 cell line was obtained from the Korean Cell Line Bank (Seoul, Korea). The HEK293 cell line expressing SV40 T antigen (HEK293T) was kindly provided by Professor Jang‐Hee Hahn (Kangwon National University, Chuncheon, Korea). These cells were grown in DMEM (HyClone, South Logan, UT, USA) supplemented with 5% FBS (HyClone), 100 U/mL penicillin, and 100 μg/mL streptomycin. All cells were grown at 37°C in 5% CO2 and 95% air.
Generation of PTK7‐knockdown lentivirus and infection of ESCC cells
Lentiviruses were produced in HEK293T cells cotransfected with: 10 μg pLKO.1‐Control, pLKO.1‐shRNA‐PTK7‐6433, or pLKO.1‐shRNA‐PTK7‐6434 plasmid (Sigma‐Aldrich, St. Louis, MO, USA); 7.4 μg packaging vector psPAX2 (Addgene, Cambridge, MA, USA); and 2.6 μg envelope vector pMD2.G (Addgene). The ESCC cells were infected with lentiviruses according to the manufacturer's recommendations (Addgene). The infected cells were cultured with 2.5 μg/mL puromycin for 14 days, to produce puromycin‐resistant colonies as a mixed culture.
Cell proliferation, survival, wound healing, and invasion assays
For cell the proliferation assay,30 cells (0.1 mL, 4 × 104 cells/mL) in a 96‐well plate were grown in DMEM supplemented with 5% FBS over a 7‐day period. For the cell survival assay,31 cells (0.1 mL, 5 × 104 cells/mL) were plated in 96‐well plates, allowed to attach for 4 h, and treated with 0–80 μM 5‐fluorouracil (5‐FU) for 48 h. Viable cells were measured with the colorimetric MTT assay.
For the wound healing assay, a wound was introduced by scraping the monolayer with a micropipette tip. The cells were incubated for 24 h in DMEM supplemented with 5% FBS, fixed with 3.7% formamide, and evaluated by light microscopy.
The invasion assay was carried out using a Transwell system (Corning‐Costar, Lowell, MA, USA) as described previously.24 Briefly, detached cells (0.1 mL, 5 × 105 cells/mL) were loaded into the upper chamber which was coated with 10 μL of 0.1% gelatin and 80 μL of 0.3 μg/mL growth‐factor reduced Matrigel (BD Biosciences, Bedford, MA, USA) on the lower and upper surfaces, respectively. DMEM (0.6 mL) supplemented with 5% FBS as a chemoattractant was added to the lower chamber. After 24 h, invaded cells on the lower surface of the filter were stained with H&E and analyzed under a light microscope.
Statistical analysis
The IHC grade and various survival parameters (sex, age, pathologic stage, differentiation, and adjuvant and neoadjuvant therapy) were analyzed with respect to recurrence‐free survival and overall survival using a multivariate Cox proportional hazard model. The software spss version 14.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis.
All data were obtained from at least three independent experiments and expressed as the mean ± standard deviation. Statistical significance was analyzed by Student's t‐test. A P‐value less than 0.05 was considered significant.
Results
Expression of PTK7 increased in ESCC samples
Expression levels of PTK7 mRNA and protein in tumor and normal tissue pairs from 20 and 10 ESCC patients, respectively, were determined by RT‐PCR and Western blot analysis. The ESCC tissue showed significantly increased expression of PTK7 at both the mRNA and protein levels compared with normal esophageal epithelial tissue (Fig. 1).
Figure 1.

Expression of protein tyrosine kinase 7 (PTK7) in tumor samples from 10 patients with esophageal squamous cell carcinoma. (a) Levels of PTK7 mRNA determined by RT‐PCR. The GAPDH mRNA levels are shown for normalization. Numbers 1–20 are patient identifiers. (b) Western blot showing PTK7 protein levels. β‐Actin levels are shown for normalization. Numbers 1–10 are patient identifiers. N, healthy esophageal epithelium, adjacent to the tumor; T, tumor tissue sample.
Characteristics of ESCC patients analyzed for IHC staining
Among the 157 ESCC patients analyzed for IHC staining, the mean age was 62.4 years and 150 patients were male. All patients underwent a trans‐thoracic esophagectomy, after which the esophagus was reconstructed by interposition of the stomach or colon. There were 19 patients with pathologic stage I disease, 36 with stage II, 40 with stage IIIA, 26 with stage IIIB, and 36 with stage IIIC (Table 1). The mean duration of follow‐up was 44.5 months.
Table 1.
Clinical parameters and recurrence‐free and overall survival after surgery in patients with esophageal squamous cell carcinoma (n = 157)
| Prognostic factor | No. (%) | Recurrence‐free survival | Overall survival | ||||
|---|---|---|---|---|---|---|---|
| HR | 95%CI | P‐value | HR | 95%CI | P‐value | ||
| Gender | 0.710 | 0.800 | |||||
| Female | 7 (4.5) | 1.00 | 1.00 | ||||
| Male | 150 (95.5) | 1.22 | 0.44–3.35 | 0.710 | 1.14 | 0.41–3.15 | 0.800 |
| Age, years (>65) | 62.4 ± 7.6 | 0.610 | 0.150 | ||||
| <65 | 96 (61.1) | 1.00 | 1.00 | ||||
| ≥65 | 61 (38.9) | 1.37 | 0.89–2.22 | 0.610 | 1.37 | 0.89–2.22 | 0.150 |
| Pathologic stage | 0.001* | 0.005* | |||||
| Stage I | 19 (12.1) | 1.00 | 1.00 | ||||
| Stage II | 36 (22.9) | 1.02 | 0.73–1.49 | 0.760 | 1.17 | 0.98–1.35 | 0.570 |
| Stage IIIA | 40 (25.5) | 1.75 | 1.10–2.95 | 0.029* | 1.68 | 1.11–3.31 | 0.025* |
| Stage IIIB | 26 (16.6) | 2.07 | 1.12–3.21 | 0.015* | 1.92 | 1.13–3.98 | 0.210 |
| Stage IIIC | 36 (22.9) | 2.68 | 1.39–5.20 | 0.003* | 2.98 | 1.51–5.85 | 0.002* |
| Neoadjuvant chemotherapy | 0.216 | 0.089 | |||||
| No | 150 (95.5) | 1.00 | 1.00 | ||||
| Yes | 7 (4.5) | 1.22 | 0.72–4.10 | 0.216 | 2.03 | 0.89–4.60 | 0.089 |
| Adjuvant chemotherapy | 0.520 | 0.560 | |||||
| No | 129 (82.2) | 1.00 | 1.00 | ||||
| Yes | 28 (17.8) | 0.85 | 0.51–1.41 | 0.520 | 0.86 | 0.52–1.43 | 0.560 |
| Adjuvant radiotherapy | 0.890 | 0.880 | |||||
| No | 134 (85.4) | 1.00 | 1.00 | ||||
| Yes | 23 (14.6) | 0.89 | 0.60–1.81 | 0.890 | 1.04 | 0.60–1.81 | 0.880 |
| Degree of differentiation | 0.910 | 0.890 | |||||
| Well | 36 (22.9) | 1.00 | 1.00 | ||||
| Moderate | 103 (65.6) | 1.14 | 0.74–1.74 | 0.840 | 1.10 | 0.71–1.69 | 0.820 |
| Poor | 18 (11.5) | 1.39 | 0.51–2.84 | 0.900 | 1.19 | 0.69–1.99 | 0.880 |
| IHC staining grade | 0.131 | 0.163 | |||||
| Grade 1 | 67 (42.7) | 1.00 | |||||
| Grade 2 | 61 (38.9) | 1.23 | 0.80–1.79 | 0.360 | 1.16 | 0.75–1.79 | 0.510 |
| Grade 3 | 29 (18.5) | 1.61 | 0.97–2.65 | 0.064 | 1.81 | 1.09–2.99 | 0.021* |
*P < 0.05. Immunohistochemical (IHC) staining: grade 1, weak staining or fewer than 40% of tumor cells stained; grade 2, moderate to strong staining and 40–70% of tumor cells stained; grade 3, moderate to strong staining and more than 70% of tumor cells stained. CI, confidence interval; HR, hazard ratio.
Most ESCC tissues stained for PTK7
Each specimen was subjected to IHC staining of PTK7. Most normal epithelia were not stained, with a few exceptions showing stained benign epithelia or stroma. In contrast, most tumor cell nests stained positive for PTK7 expression (Fig. 2). Both the intensity of staining and the percentage of stained tumor cells were semiquantitatively scored. Examples for the lowest (Score 0) and highest (Score 10) intensity of staining are shown in Figure 2(a,b), respectively. The specimens were classified according to intensity and number of stained tumor cells on a 3‐point scale (Fig. 2c). The number of specimens with each staining grade was 67 for grade 1, 61 for grade 2, and 29 for grade 3 (Fig. 2d–f). Approximately 60% of specimens showed moderate to strong staining for PTK7 (grade 2 or 3). Among the 67 cases classified as grade 1, three specimens were completely negative for PTK7 whereas another 17 specimens showed staining in fewer than 10% of tumor cells. Apart from these exceptions, over 80% of the total cases displayed PTK7 expression. In most specimens classified as grade 2 or 3, PTK7 staining mainly localized to the cell membranes of tumor cells. Notably, in well‐stained tumor regions, unstained portions of tumor cell nests were sporadically observed (Fig. 2g). In addition, in fewer than 10% of the specimens, several cells within the stromal tissue surrounding a tumor cell nest stained positive for PTK7 (Fig. 2h). Intraepithelial neoplastic lesions around tumor cells which were found in some specimens were usually stained for PTK7. These findings suggest that PTK7 expression is upregulated even at the early stages of tumor development.
Figure 2.
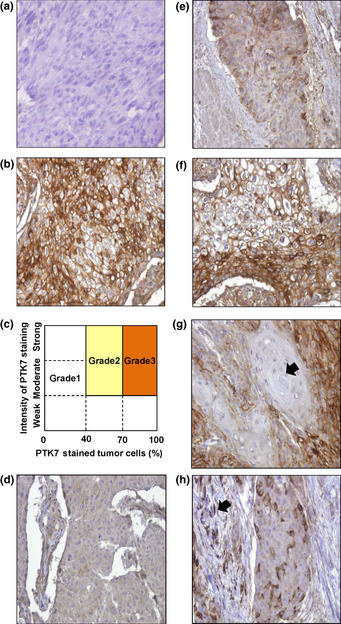
Immunohistochemical (IHC) analysis of esophageal squamous cell carcinoma tissue stained for protein tyrosine kinase 7 (PTK7). Representative images of poorly stained tumor cells (Score 0) (a) and heavily stained tumor cells (Score 10) (b). (c) Classification of IHC staining grade. Grade 1, weak staining or fewer than 40% of tumor cells stained; grade 2, moderate to strong staining and 40–70% of tumor cells stained; grade 3, moderate to strong staining and more than 70% of tumor cells stained. (d–f) Representative images of PTK7 IHC grades 1–3, respectively. (g) PTK7‐negative tumor cell nest. Some differentiated tumor cell nests (arrow) were found to be negative for PTK7 expression. (h) PTK7‐positive stromal cells. Some stromal cells (arrow) around a tumor cell nest were positive for PTK7. Magnification, ×200.
High expression of PTK7 correlates with poor prognosis in patients with ESCC
Using the follow‐up data of the 157 patients in conjunction with the results from the PTK7 IHC staining experiments, we analyzed which tumor characteristics affected recurrence‐free survival and overall survival. The mortality rate during the follow‐up period was 71% (n = 113). Analysis of recurrence‐free survival showed that pathologic stage was the only significant parameter (P = 0.001) when analyzed with a multivariate Cox proportional hazard model (Table 1). The PTK7 staining grade determined by IHC did not correlate significantly with recurrence‐free survival, although patients with IHC staining classified as grade 3 tended to have poorer recurrence‐free survival than patients with grade 1 or 2 staining (Fig. 3a). However, analysis of overall survival showed that the IHC staining grade clearly correlates with prognosis of ESCC (Fig. 3b). Multivariate analysis of overall survival also showed that grade 3 staining was associated with worse prognosis than grade 1 (P = 0.021) and indicated a correlation between prognosis and the pathologic stage of the tumor (Table 1). However, there was no significant association between the IHC staining grade and clinical parameters (Table S1). In conclusion, our analysis reveals that PTK7 IHC staining grade and pathologic stage were significant factors influencing overall survival, whereas age, gender, and neoadjuvant and adjuvant therapy did not affect prognosis of ESCC patients significantly.
Figure 3.

Analysis of survival rates of patients with esophageal squamous cell carcinoma according to protein tyrosine kinase 7 (PTK7) immunohistochemical staining grade. (a) Kaplan–Meier curves for recurrence‐free survival of each grade. (b) Kaplan–Meier curves for overall survival of each grade. Grade 1, weak staining or fewer than 40% of tumor cells stained; grade 2, moderate to strong staining and 40–70% of tumor cells stained; grade 3, moderate to strong staining and more than 70% of tumor cells stained.
Knockdown of PTK7 decreases the oncogenic potential of ESCC cells in vitro
To investigate the oncogenic role of PTK7 in ESCC, a knockdown model for the PTK7 gene was applied in ESCC cell lines. Among ESCC cell lines TE‐5, TE‐9, TE‐10, TE‐11, and TE‐14, the expression level of PTK7 was highest in TE‐10 and TE‐11 cells (Fig. S1a). Therefore, PTK7 knockdown was carried out in TE‐10 and TE‐11 cells by infection with one of two different PTK7‐silencing lentiviruses, shRNA‐PTK7‐6433 and shRNA‐PTK7‐6434 (Fig. S1b).
Using the PTK7 knockdown ESCC cells, we analyzed the effect of PTK7 on cell proliferation and survival using an MTT assay. In cell proliferation assay, the number of TE‐10 and TE‐11 cells infected with control lentiviral vector increased to 313% and 306% of the original number, respectively, after 7 days of culture in the presence of serum (Fig. 4a). However, in the same cell lines infected with PTK7‐knockdown lentiviruses, the number of cells was approximately 65–75% that of the control cells at 5 and 7 days of culture. In the survival assay of 5‐FU‐treated TE‐10 cells, treatment with 20 μM 5‐FU, at which the most significant statistical difference was observed, showed 80% survival of control cells but 55% survival of PTK7‐knockdown cells (Fig. 4b).
Figure 4.
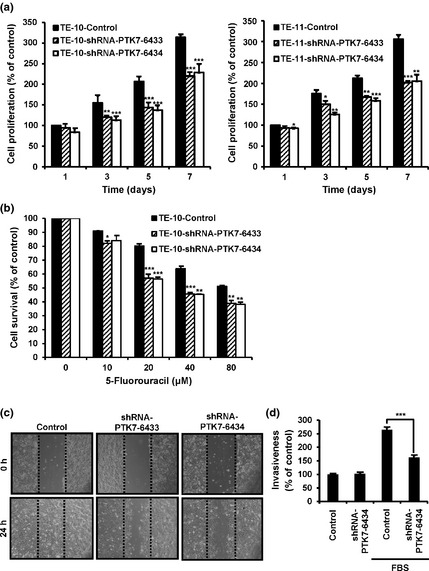
Effects of protein tyrosine kinase 7 (PTK7) knockdown on oncogenic properties of esophageal squamous cell carcinoma (ESCC) cell lines. The ESCC cells were infected with lentivirus harboring a puromycin resistance gene (control vs shRNA‐PTK7‐6433 vs shRNA‐PTK7‐6434) and mixed cultures of puromycin‐resistant colonies were used. (a) Effect of PTK7 knockdown on cell proliferation in TE‐10 (left panel) and TE‐11 (right panel) cells. (b) Effect of PTK7 knockdown on cell survival of 5‐fluorouracil (5‐FU)‐treated TE‐10 ESCC cells. (c) Effect of PTK7 knockdown on wound healing in TE‐10 ESCC cells. Light micrographs (×100) showing wound healing in monolayers of TE‐10 cells with or without PTK7 knockdown. (d) Effect of PTK7 knockdown on Matrigel invasion of TE‐10 ESCC cells. *P < 0.05; **P < 0.01; ***P < 0.001 vs control.
We next investigated the role of PTK7 on wound healing and invasion of TE‐10 cells. In the wound healing assay, control cells repaired the wound in 24 h, whereas PTK7‐knockdown cells showed significantly delayed wound repair (Fig. 4c). Regarding invasiveness, in the absence of serum the invasion of control cells was similar to that of PTK7‐knockdown cells. When serum was added as an attractant, invasion of control cells increased 2.6‐fold, whereas invasion of PTK7‐knockdown cells was approximately 60% of that shown by the control cells (Fig. 4d).
Knockdown of PTK7 inhibits activation of Akt, MAPKs, and FAK in ESCC cells
Activation of PI3‐Kinase/Akt and MAPK pathways is an important process during tumorigenesis and is closely linked to cell proliferation and survival.32 In addition, FAK activation is involved in cell migration and invasion.33 Therefore, we analyzed the role of PTK7 in the activation of Akt, MAPKs, and FAK. In both TE‐10 and TE‐11 cells, PTK7 knockdown significantly decreased the phosphorylation levels of Akt, Erk, JNK, p38 MAPK, and FAK (Fig. 5). This result suggests that PTK7 modulates signaling pathways involving Akt, Erk, JNK, p38 MAPK, and FAK to enhance the proliferation, survival, migration, and invasion of ESCC cells.
Figure 5.

Effect of protein tyrosine kinase 7 (PTK7) knockdown on activation of signal molecules in TE‐10 and TE‐11 esophageal squamous cell carcinoma cells. Expression of signaling proteins Akt, Erk, JNK, p38 MAPK, and focal adhesion kinase (FAK) was examined by Western blot analysis in TE‐10 (left panel) and TE‐11 (right panel) cells with or without PTK7 knockdown. The GAPDH levels are shown for normalization.
Discussion
Esophageal squamous cell carcinoma is the most prevalent subtype of esophageal cancer in East Asian countries, accounting for over 90% of total cases, whereas esophageal adenocarcinoma is more prevalent in North America, UK, and Australia. The carcinogenic mechanisms and biological and clinical characteristics of ESCC differ from those seen in adenocarcinoma. In ESCC, smoking, alcohol abuse, lower socioeconomic status, and consumption of extremely hot beverages have been reported as significant predisposing factors. For adenocarcinoma, known risk factors are gastroesophageal reflux disease and obesity.34
In our preliminary study, we detected upregulation of the PTK7 mRNA level by gene expression profiling in ESCC, but not in normal esophageal tissues or in other thoracic squamous cell carcinoma specimens (data not shown). In the present study, we confirm that PTK7 is significantly upregulated compared with adjacent normal tissue at both the mRNA and protein levels in tumor samples of ESCC patients. Moreover, we found moderate to strong staining for PTK7 through IHC in over 60% of ESCC specimens. Multivariate analysis correlating clinical outcome parameters of ESCC patients with tumor characteristics yielded pathologic stage as the only statistically significant determinant of recurrence‐free survival. However, when correlating tumor characteristics with overall survival, PTK7 IHC staining grade and pathologic stage were identified as independent prognostic factors. Thus, we propose that PTK7 IHC staining grade could be used as a prospective prognostic determinant of ESCC.
Although previous studies have reported a decrease in PTK7 expression levels in metastatic melanoma25 and clear cell renal cell carcinoma,26 PTK7 has been shown to be upregulated in many types of human malignancy, including colon cancer, lung carcinoma, liposarcoma, gastric cancer, and acute myeloid leukemia.11, 17, 18, 19, 20, 22 Prebet et al.20 reported that increased expression of PTK7 is correlated with poor clinical outcome in acute myeloid leukemia, a finding that was linked to increased cell migration and resistance to apoptosis. Meng et al.23 showed that knockdown of PTK7 in colon cancer cells inhibits proliferation and induces caspase‐10‐dependent apoptosis through the mitochondrial pathway.
Consistent with those studies, we showed here that PTK7 knockdown decreases proliferation of TE‐10 and TE‐11 ESCC cells and survival of TE‐10 cells, and inhibits wound healing and invasion of TE‐10 cells. A previous study showed that soluble PTK7 treatment, which has an effect comparable to that of PTK7 knockdown, inhibited vascular endothelial growth factor‐induced activation of FAK and paxillin in HUVECs.24 Focal adhesion kinase is involved in the adhesion and migration of cells35 and, more specifically, in tumor invasion and lymph node metastasis in ESCC.36 Activation of Akt and MAPKs promotes cell survival, proliferation, and migration.37, 38 We found that knockdown of PTK7 decreased phosphorylation of Akt, Erk, JNK, p38 MAPK, and FAK in ESCC cells. Therefore, given our in vitro findings regarding PTK7‐dependent proliferation, survival, wound healing, and invasion, we propose that PTK7 may be involved in tumorigenesis and metastasis of ESCC cells, specifically through the activation of factors that promote proliferation, cell survival, migration, and invasion.
In summary, we showed upregulation of PTK7 in clinical samples of ESCC and highlighted the oncogenic role of PTK7 in ESCC cells in vitro. Increased expression of PTK7 was inversely correlated with overall survival of ESCC patients, and grading of IHC samples based on PTK7 expression was able to predict ESCC prognosis in a multivariate analysis. Our findings consistently suggest that PTK7 is a potentially valuable prognostic marker for ESCC and might be applied as a therapeutic target molecule in the management of ESCC.
Disclosure Statement
The authors have no conflict of interest.
Supporting information
Table S1. χ2‐test for the cross‐tabulations of immunohistochemical staining grade and other categorical clinical variables (n = 157).
Fig. S1. Protein tyrosine kinase 7 (PTK7) expression and knockdown in esophageal squamous cell carcinoma cell lines.
Acknowledgments
This work was supported by grants from the Basic Research Program (2012R1A1A2007638 to S.‐T.L.) of the National Research Foundation, Ministry of Education, Science, and Technology, Korea and from the National R&D Program for Cancer Control (1120110 to S.‐T.L. and 1120260 to J.H.P), Ministry of Health and Welfare, Korea. W.‐S.S. and H.‐W.N. were pre‐doctoral trainees of the Brain Korea 21 program from the Ministry of Education, Science, and Technology, Korea.
(Cancer Sci 2013; 104: 1120–1126)
References
- 1. Wobst A, Audisio RA, Colleoni M, Geraghty JG. Oesophageal cancer treatment: studies, strategies and facts. Ann Oncol 1998; 9: 951–62. [DOI] [PubMed] [Google Scholar]
- 2. Miyata H, Yamasaki M, Kurokawa Y et al Survival factors in patients with recurrence after curative resection of esophageal squamous cell carcinomas. Ann Surg Oncol 2011; 18: 3353–61. [DOI] [PubMed] [Google Scholar]
- 3. Kranzfelder M, Schuster T, Geinitz H, Friess H, Buchler P. Meta‐analysis of neoadjuvant treatment modalities and definitive non‐surgical therapy for oesophageal squamous cell cancer. Br J Surg 2011; 98: 768–83. [DOI] [PubMed] [Google Scholar]
- 4. Chiu PW, Chan AC, Leung SF et al Multicenter prospective randomized trial comparing standard esophagectomy with chemoradiotherapy for treatment of squamous esophageal cancer: early results from the Chinese University Research Group for Esophageal Cancer (CURE). J Gastrointest Surg 2005; 9: 794–802. [DOI] [PubMed] [Google Scholar]
- 5. Kelsen DP, Ginsberg R, Pajak TF et al Chemotherapy followed by surgery compared with surgery alone for localized esophageal cancer. N Engl J Med 1998; 339: 1979–84. [DOI] [PubMed] [Google Scholar]
- 6. Urschel JD, Vasan H. A meta‐analysis of randomized controlled trials that compared neoadjuvant chemoradiation and surgery to surgery alone for resectable esophageal cancer. Am J Surg 2003; 185: 538–43. [DOI] [PubMed] [Google Scholar]
- 7. Gebski V, Burmeister B, Smithers BM, Foo K, Zalcberg J, Simes J. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta‐analysis. Lancet Oncol 2007; 8: 226–34. [DOI] [PubMed] [Google Scholar]
- 8. Hongo M, Nagasaki Y, Shoji T. Epidemiology of esophageal cancer: Orient to Occident. Effects of chronology, geography and ethnicity. J Gastroenterol Hepatol 2009; 24: 729–35. [DOI] [PubMed] [Google Scholar]
- 9. Kwon YJ, Lee SJ, Koh JS, Kim SH, Kim YJ, Park JH. Expression patterns of aurora kinase B, heat shock protein 47, and periostin in esophageal squamous cell carcinoma. Oncol Res 2009; 18: 141–51. [DOI] [PubMed] [Google Scholar]
- 10. Lee ST, Strunk KM, Spritz RA. A survey of protein tyrosine kinase mRNAs expressed in normal human melanocytes. Oncogene 1993; 8: 3403–10. [PubMed] [Google Scholar]
- 11. Mossie K, Jallal B, Alves F, Sures I, Plowman GD, Ullrich A. Colon carcinoma kinase‐4 defines a new subclass of the receptor tyrosine kinase family. Oncogene 1995; 11: 2179–84. [PubMed] [Google Scholar]
- 12. Park SK, Lee HS, Lee ST. Characterization of the human full‐length PTK7 cDNA encoding a receptor protein tyrosine kinase‐like molecule closely related to chick KLG. J Biochem 1996; 119: 235–9. [DOI] [PubMed] [Google Scholar]
- 13. Lu X, Borchers AG, Jolicoeur C, Rayburn H, Baker JC, Tessier‐Lavigne M. PTK7/CCK‐4 is a novel regulator of planar cell polarity in vertebrates. Nature 2004; 430: 93–8. [DOI] [PubMed] [Google Scholar]
- 14. Wehner P, Shnitsar I, Urlaub H, Borchers A. RACK1 is a novel interaction partner of PTK7 that is required for neural tube closure. Development 2011; 138: 1321–7. [DOI] [PubMed] [Google Scholar]
- 15. Shnitsar I, Borchers A. PTK7 recruits dsh to regulate neural crest migration. Development 2008; 135: 4015–24. [DOI] [PubMed] [Google Scholar]
- 16. Puppo F, Thome V, Lhoumeau AC et al Protein tyrosine kinase 7 has a conserved role in Wnt/beta‐catenin canonical signalling. EMBO Rep 2011; 12: 43–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Saha S, Bardelli A, Buckhaults P et al A phosphatase associated with metastasis of colorectal cancer. Science 2001; 294: 1343–6. [DOI] [PubMed] [Google Scholar]
- 18. Gorringe KL, Boussioutas A, Bowtell DD. Novel regions of chromosomal amplification at 6p21, 5p13, and 12q14 in gastric cancer identified by array comparative genomic hybridization. Genes Chromosom Cancer 2005; 42: 247–59. [DOI] [PubMed] [Google Scholar]
- 19. Endoh H, Tomida S, Yatabe Y et al Prognostic model of pulmonary adenocarcinoma by expression profiling of eight genes as determined by quantitative real‐time reverse transcriptase polymerase chain reaction. J Clin Oncol 2004; 22: 811–9. [DOI] [PubMed] [Google Scholar]
- 20. Prebet T, Lhoumeau AC, Arnoulet C et al The cell polarity PTK7 receptor acts as a modulator of the chemotherapeutic response in acute myeloid leukemia and impairs clinical outcome. Blood 2010; 116: 2315–23. [DOI] [PubMed] [Google Scholar]
- 21. Muller‐Tidow C, Schwable J, Steffen B et al High‐throughput analysis of genome‐wide receptor tyrosine kinase expression in human cancers identifies potential novel drug targets. Clin Cancer Res 2004; 10: 1241–9. [DOI] [PubMed] [Google Scholar]
- 22. Gobble RM, Qin LX, Brill ER et al Expression profiling of liposarcoma yields a multigene predictor of patient outcome and identifies genes that contribute to liposarcomagenesis. Cancer Res 2011; 71: 2697–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Meng L, Sefah K, O'Donoghue MB et al Silencing of PTK7 in colon cancer cells: caspase‐10‐dependent apoptosis via mitochondrial pathway. PLoS One 2010; 5: e14018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Shin WS, Maeng YS, Jung JW, Min JK, Kwon YG, Lee ST. Soluble PTK7 inhibits tube formation, migration, and invasion of endothelial cells and angiogenesis. Biochem Biophys Res Commun 2008; 371: 793–8. [DOI] [PubMed] [Google Scholar]
- 25. Easty DJ, Mitchell PJ, Patel K, Florenes VA, Spritz RA, Bennett DC. Loss of expression of receptor tyrosine kinase family genes PTK7 and SEK in metastatic melanoma. Int J Cancer 1997; 71: 1061–5. [DOI] [PubMed] [Google Scholar]
- 26. Behbahani TE, Thierse C, Baumann C et al Tyrosine kinase expression profile in clear cell renal cell carcinoma. World J Urol 2012; 30: 559–65. [DOI] [PubMed] [Google Scholar]
- 27. Banga SS, Ozer HL, Park SK, Lee ST. Assignment of PTK7 encoding a receptor protein tyrosine kinase‐like molecule to human chromosome 6p21.1–>p12.2 by fluorescence in situ hybridization. Cytogenet Cell Genet 1997; 76: 43–4. [DOI] [PubMed] [Google Scholar]
- 28. Piao Z, Lee KS, Kim H, Perucho M, Malkhosyan S. Identification of novel deletion regions on chromosome arms 2q and 6p in breast carcinomas by amplotype analysis. Genes Chromosom Cancer 2001; 30: 113–22. [PubMed] [Google Scholar]
- 29. Baudrier‐Regnier A, Bodenant C, Proust F, Delangre T, Hemet J, Laquerriere A. An isochromosome 6p in a primary meningeal malignant melanoma. Cancer Genet Cytogenet 2000; 119: 80–2. [DOI] [PubMed] [Google Scholar]
- 30. Na HW, Shin WS, Ludwig A, Lee ST. The cytosolic domain of protein‐tyrosine kinase 7 (PTK7), generated from sequential cleavage by a disintegrin and metalloprotease 17 (ADAM17) and gamma‐secretase, enhances cell proliferation and migration in colon cancer cells. J Biol Chem 2012; 287: 25001–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Iizuka N, Hirose K, Noma T et al The nm23‐H1 gene as a predictor of sensitivity to chemotherapeutic agents in oesophageal squamous cell carcinoma. Br J Cancer 1999; 81: 469–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Steelman LS, Abrams SL, Whelan J et al Contributions of the Raf/MEK/ERK, PI3K/PTEN/Akt/mTOR and Jak/STAT pathways to leukemia. Leukemia 2008; 22: 686–707. [DOI] [PubMed] [Google Scholar]
- 33. Zhao J, Guan JL. Signal transduction by focal adhesion kinase in cancer. Cancer Metastasis Rev 2009; 28: 35–49. [DOI] [PubMed] [Google Scholar]
- 34. Enzinger PC, Mayer RJ. Esophageal cancer. N Engl J Med 2003; 349: 2241–52. [DOI] [PubMed] [Google Scholar]
- 35. Webb DJ, Donais K, Whitmore LA et al FAK‐Src signalling through paxillin, ERK and MLCK regulates adhesion disassembly. Nat Cell Biol 2004; 6: 154–61. [DOI] [PubMed] [Google Scholar]
- 36. Miyazaki T, Kato H, Nakajima M et al FAK overexpression is correlated with tumour invasiveness and lymph node metastasis in oesophageal squamous cell carcinoma. Br J Cancer 2003; 89: 140–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell 2007; 129: 1261–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Johnson GL, Lapadat R. Mitogen‐activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002; 298: 1911–2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. χ2‐test for the cross‐tabulations of immunohistochemical staining grade and other categorical clinical variables (n = 157).
Fig. S1. Protein tyrosine kinase 7 (PTK7) expression and knockdown in esophageal squamous cell carcinoma cell lines.


