Dear Sir,
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) continues to spread rapidly all over the world; however, the mechanism of the disease is not yet fully understood. Why do many people who come into contact with the virus not get sick and remain asymptomatic? Why do some people recover with tiny symptoms, while other people have a severe infection or die? SARS-CoV-2 infection can lead to severe or fatal outcomes in patients with comorbid diseases such as hypertension, diabetes, obesity, and heart failure. SARS-CoV-2 binds to angiotensin-converting enzyme (ACE)-2, like SARS-CoV, enters the cells and causes infection. The renin-angiotensin system (RAS) plays a crucial role in the virus entry to cells and the progression of the virus-induced disease. Whether ACE2 upregulation will increase viral load remains unclear.1 ACE2 up-regulation increases angiotensin 1–7 formation and may have a protective effect against SARS-COV-2 caused acute respiratory distress syndrome (ARDS) and heart damage.1 Angiotensin II level is high in patients with the novel coronavirus disease 2019 (COVID-19). According to an extensive view, SARS-CoV-2 binds to ACE2, causing ACE2 to become dysfunctional.2 Therefore, increased angiotensin II level leads to ARDS and heart damage.2 Interestingly, most infected people have no symptoms, and they do not have heart or lung damage. The effects of SARS-CoV-2 on peripheral RAS have been highlighted so far. We believe that the central RAS involvement of the virus has vital implications for COVID-19 progression.
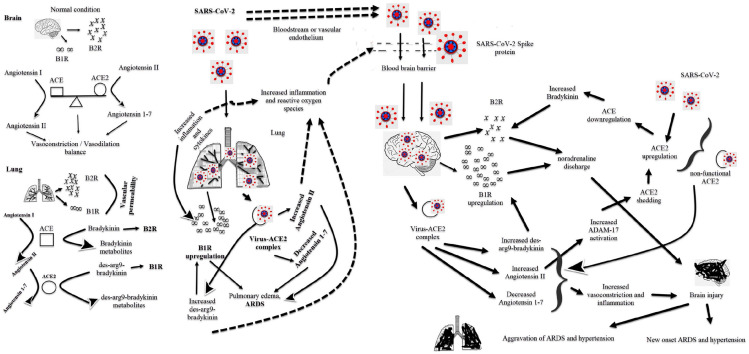
Regulation of pulmonary vascular tone is vital for the maintenance of pulmonary functions. RAS regulates vasoconstriction and vasodilation of the pulmonary vascular system. Bradykinin and kinin are responsible for the permeability and vasodilation of the pulmonary vascular system (Figure 1). Inflammation leads to an increase in bradykinin-1 receptor (B1R) in the lungs. They increase vascular permeability by binding bradykinin to the bradykinin-2 receptor (B2R) and des-arg9-bradykinin binding to the B1R. Increased pulmonary vascular permeability causes pulmonary edema.3,4 SARS-CoV-2 can cause ARDS via the bradykinin pathway in the lung (Figure 1).3,4 The brain is one of the tissues containing ACE2, such as the lung, heart, pancreas, kidney, and vascular endothelium, and SARS-CoV-2 can easily infect the brain.5,6 The virus infects the brain after can cross the blood-brain barrier either by direct transport through the bloodstream or indirectly by binding to the vascular endothelium (Figure 1).7 The virus may cause cerebrovascular disorders, epileptic seizures, and neurodegenerative diseases with central involvement.5,8 Neuroinflammation of SARS-CoV-2 can lead to an increased risk of hospital stay and mortality. The virus can cause brain damage through the bradykinin and des-arg9-bradykinin pathway, as in the lung. Bradykinin has two receptors, and the brain and spinal cord have a few numbers of B1R, while many tissues have B2R.9,10 There is also abundant B2R in the brain. Bradykinin is only a substrate for ACE, and it is not a substrate for ACE2.9,10 The mechanism by which bradykinin causes central hypertension through B2R is well known. Especially, bradykinin injection into the lateral ventricle causes severe systemic hypertension through B2R.10–12 It was observed that bradykinin into the brain caused a high increase in central blood pressure in hypertensive rats than in normotensive rats.13 The central or intrathecal bradykinin injection leads to noradrenaline release and sympathoneuronal pathway activation.11 The development of central hypertension through B1R is less well known than B2R. Not bradykinin, des-arg9-bradykinin binds B1R and is a substrate of ACE2. B1R upregulation occurs in patients with hypertension, heart failure, diabetes, and obesity.14 COVID-19 is aggressive and severe in patients with comorbid diseases such as hypertension, diabetes, obesity, and chronic obstructive pulmonary disease. Some stimulants such as tissue injuries, pro-inflammatory cytokines, and bacterial lipopolysaccharides cause B1R upregulation.12,14 The virus can render it dysfunctional by forming a complex with ACE2 in brain tissue. Thus, the virus can increase angiotensin II levels (Figure 1). It may also cause an abundant release of pro-inflammatory cytokines. Stimulation of B1R causes nitric oxide release. Increased nitric oxide turns peroxynitrite and superoxide radicals.14 The virus disrupts the structure of the blood-brain barrier either directly through its spike protein or due to increased oxidative stress via B1R (Figure 1).14,15 Disrupted blood-brain barrier facilitates the passage of harmful substances such as reactive oxygen species and cytokines from the peripheral circulation to the brain.
Figure 1.
Lung and brain damage of the virus.
The virus-ACE2 complex causes ACE2 to lose its function, and ACE2 does not degrade des-arg9-bradykinin and angiotensin II. The increased levels of des-arg9-bradykinin, angiotensin II and cytokines cause B1R upregulation. B1R upregulation in the brain increases noradrenaline discharge, leading to severe central and peripheral hypertension (Figure 1).16 High angiotensin II levels lead to ADAM17 activation. The activation induces ACE2 shedding, and as a result, ACE2 upregulation occurs.14 ACE2 upregulation causes the downregulation of ACE and increases bradykinin levels. High bradykinin levels trigger noradrenaline discharge in the brain (Figure 1).11,12 ACE2 protects tissues against the toxic effects of angiotensin II by converting angiotensin II to angiotensin 1–7.17 Angiotensin 1–7 is a vasodilator peptide and protects tissues against ischemia. Angiotensin 1–7 acts as a vasodilator and antioxidant in the brain as in the peripheral system.18 It lowers blood pressure by reducing norepinephrine release and shows a neuroprotective effect.18,19 The invasion of ACE2 by the virus reduces the formation of angiotensin 1–7. Low angiotensin 1–7 may not have neuroprotective and blood pressure lowering effects. Since the vasodilator-vasoconstrictor balance in the brain is disturbed, blood pressure may increase with the stimulation of the central pathway (Figure 1).
In conclusion, the virus increases angiotensin II, bradykinin, and des-arg9-bradykinin levels in the pulmonary vascular bed and peripheral vascular system. Increasing these peptides can lead to ARDS and hypertension (Figure 1). The virus can reach the brain and cause neuroinflammation and increase the level of these peptides. Neuroinflammation of the virus can have two consequences. First, the virus can directly trigger the development of ARDS and hypertension via the central pathway. Second, the simultaneous involvement of both peripheral and central vascular systems may worsen the clinical course of ARDS and hypertension. Brain involvement of the virus may increase the duration of the hospital stay and mortality rate.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Erkan Cure  https://orcid.org/0000-0001-7807-135X
https://orcid.org/0000-0001-7807-135X
References
- 1. Cure E, Cumhur Cure M. Comment on “Organ-protective effect of angiotensin-converting enzyme 2 and its effect on the prognosis of COVID-19”. J Med Virol. Epub ahead of print April 2020. DOI: 10.1002/jmv.25848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Vaduganathan M, Vardeny O, Michel T, et al. Renin-Angiotensin-Aldosterone system inhibitors in patients with Covid-19. N Engl J Med 2020; 382(17): 1653–1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. de Maat S, de Mast Q, Danser AHJ, et al. Impaired breakdown of bradykinin and its metabolites as a possible cause for pulmonary edema in COVID-19 infection. Semin Thromb Hemost 2020; 46(7): 835–837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. van de Veerdonk FL, Netea MG, van Deuren M, et al. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife 2020; 9: e57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Karadaş Ö, Öztürk B, Sonkaya AR. A prospective clinical study of detailed neurological manifestations in patients with COVID-19. Neurol Sci 2020; 41(8): 1991–1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Virhammar J, Kumlien E, Fällmar D, et al. Acute necrotizing encephalopathy with SARS-CoV-2 RNA confirmed in cerebrospinal fluid. Neurology 2020; 95(10): 445–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Chigr F, Merzouki M, Najimi M. Comment on “The neuroinvasive potential of SARS-CoV-2 may play a role in the respiratory failure of COVID-19 patients”. J Med Virol 2020; 92(7): 703–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cure E, Cumhur Cure M, Kucuk A. Comment on “Smooth or risky revisit of an old malaria drug for COVID-19?” J Neuroimmune Pharmacol 2020; 15(3): 345–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Sodhi CP, Wohlford-Lenane C, Yamaguchi Y, et al. Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg(9) bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration. Am J Physiol Lung Cell Mol Physiol 2018; 314(1): L17–L31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lindsey CJ, Nakaie CR, Martins DT. Central nervous system kinin receptors and the hypertensive response mediated by bradykinin. Br J Pharmacol 1989; 97(3): 763–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Qadri F, Bäurle L, Häuser W, et al. Centrally bradykinin B2-receptor-induced hypertensive and positive chronotropic effects are mediated via activation of the sympathetic nervous system. J Hypertens 1999; 17(9): 1265–1271. [DOI] [PubMed] [Google Scholar]
- 12. Cloutier F, de Sousa Buck H, Ongali B, et al. Pharmacologic and autoradiographic evidence for an up-regulation of kinin B(2) receptors in the spinal cord of spontaneously hypertensive rats. Br J Pharmacol 2002; 135(7): 1641–1654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Martins DT, Fior DR, Nakaie CR, et al. Kinin receptors of the central nervous system of spontaneously hypertensive rats related to the pressor response to bradykinin. Br J Pharmacol 1991; 103(4): 1851–1856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Xu J, Sriramula S, Xia H, et al. Clinical relevance and role of neuronal AT(1) receptors in ADAM17-Mediated ACE2 shedding in neurogenic hypertension. Circ Res 2017; 121(1): 43–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Buzhdygan TP, DeOre BJ, Baldwin-Leclair A, et al. The SARS-CoV-2 spike protein alters barrier function in 2D static and 3D microfluidic in vitro models of the human blood-brain barrier. bioRxiv [Preprint] 2020. DOI: 10.1101/2020.06.15.150912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sriramula S. Kinin B1 receptor: a target for neuroinflammation in hypertension. Pharmacol Res 2020; 155: 104715. [DOI] [PubMed] [Google Scholar]
- 17. Cure E, Cumhur Cure M. Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers may be harmful in patients with diabetes during COVID-19 pandemic. Diabetes Metab Syndr 2020; 14(4): 349–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Du D, Chen J, Liu M, et al. The effects of angiotensin II and angiotensin-(1-7) in the rostral ventrolateral medulla of rats on stress-induced hypertension. PLoS One 2013; 8(8): e70976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jiang T, Gao L, Lu J, et al. ACE2-Ang-(1-7)-Mas axis in brain: a potential target for prevention and treatment of ischemic stroke. Curr Neuropharmacol 2013; 11(2): 209–217. [DOI] [PMC free article] [PubMed] [Google Scholar]